To the Editor
Psoriasis is a highly visible, chronic, immune-mediated inflammatory skin disorder that affects 2–3% of the US population (Lowes et al., 2014). Tumor necrosis factor-α (TNF-α) and IL-17A synergistically up-regulate the production of other cytokines, chemokines, and antimicrobial peptides from keratinocytes and regional immune cells, initiating and perpetuating the immune activation of psoriasis (Chiricozzi et al., 2011, Di Cesare et al., 2009, Ettehadi et al., 1994, Harden et al., 2015, Lowes et al., 2005). Humanized antibodies and inhibitory fusion proteins inhibiting TNF-α are commercially available for treating moderate to severe psoriasis (Lowes et al., 2007). These systemically administered biologic agents are targeted to disease pathogenesis and have better efficacy and safety than broad immunosuppressants such as cyclosporine and methotrexate. Their high cost and potential adverse effects limit systemic administration for milder disease, but their high molecular weight precludes topical formulation.
We have generated a TNF-α–suppressing antisense spherical nucleic acid (SNA), a promising construct to emerge from the field of nanotechnology (Banga et al., 2014, Cutler et al., 2012, Giljohann et al., 2009, Zheng et al., 2012). These 3-dimensional (3D) arrangements of densely packed and radially oriented oligonucleotides (see Supplementary Figure S1 online) impart properties distinct from linear nucleic acids, especially skin penetration capability without physical or chemical skin disruptors and increased cellular uptake. SNAs use scavenger receptors to enter cells, whereas other oligonucleotide delivery systems (e.g., cationic lipids or polymers) often disrupt anionic cell membranes for delivery (Choi et al., 2013). Liposomal-cored SNAs (L-SNAs) are physiologically compatible but share characteristics of the early-generation trackable gold-cored SNAs (Banga et al., 2014, Randeria et al., 2015, Zheng et al., 2012). Using both a human 3D cytokine-induced raft model and the mouse imiquimod (IMQ)-generated psoriasis-like model, we found that TNF-targeting L-SNAs prevent the phenotype clinically, histologically, and transcriptionally, suggesting a topical treatment paradigm for psoriasis.
For L-SNA generation, oligonucleotides and 50-nm–diameter liposomes (100:1) were self-assembled to form L-SNAs (see Supplementary Figure S1 and Supplementary Materials online). mRNA expression was assessed by quantitative PCR (see Supplementary Table S1 online). In all studies, data were analyzed by analysis of variance (group) or paired t testing (individual comparisons), with P less than 0.05 considered significant.
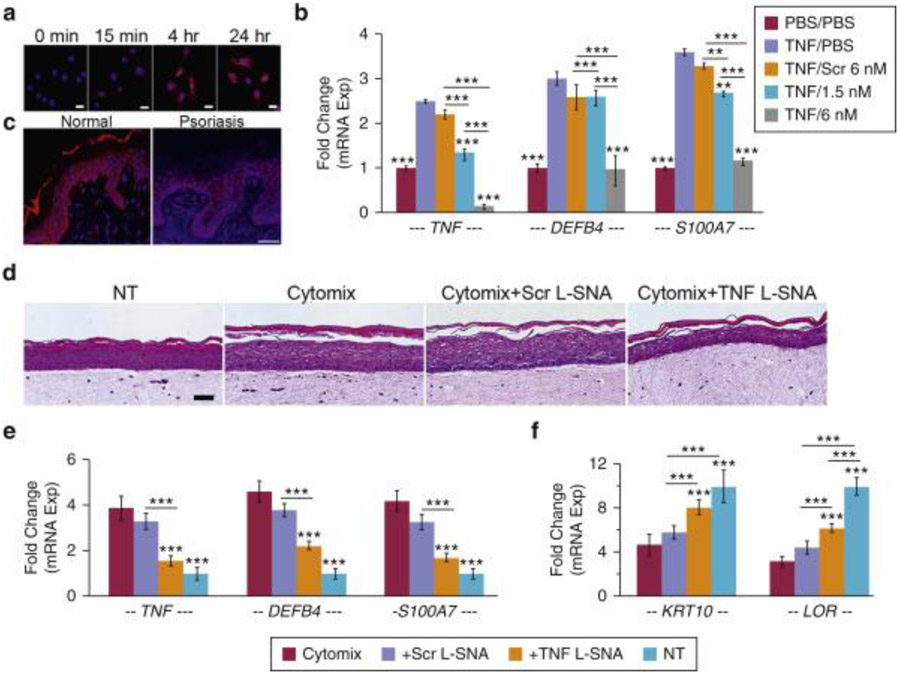
Cyanine 5–labeled TNF L-SNAs were taken into normal human epidermal keratinocyte (NHEK) cytoplasm within 15 minutes (Figure 1a) of exposure (keratinocytes for NHEK studies were prepared from otherwise discarded unidentified foreskins, obtained through expedited institutional approval that required no written informed parental consent). TNF L-SNAs knocked down mRNA expression in TNF-α–induced NHEKs by 48 hours (TNF by 93%, DEFB4 (encoding β-defensin 2A) by 62%, S100A7 by 64%; all P < 0.001 vs. scrambled L-SNA control samples [Scr]) (Figure 1b). L-SNAs penetrated human abdominoplasty and psoriatic skin within 24 hours (Figure 1c), suggesting possible translation to human psoriasis. 3D human psoriatic rafts were generated by adding TNF-α, IL-17A, and IL-22 (each at a concentration of 10 ng/ml) to the medium beginning 6 days after NHEK lifting. Histologic (hematoxylin and eosin), immunologic (ELISA and Western blot), and transcriptional alterations in psoriasis markers were present within 3 days of cytokine initiation and were further increased by 6 days (see Supplementary Materials and Supplementary Figure S2a-d online). TNF L-SNAs at 50 nmol/L (Scr and phosphate buffered saline control samples) were applied to the raft center every other day using a ring to prevent leakage, beginning 3 days after cytokine initiation. The rafts were harvested at 7 days after cytokine initiation and 24 hours after the last L-SNA application. TNF L-SNAs improved differentiation (Figure 1d), reduced acanthosis (P < 0.05) (see Supplementary Figure S3a and b online), and normalized psoriatic marker mRNA expression (Figure 1e and f) of the psoriasis raft model to resemble rafts not exposed to cytokines.
Figure 1. TNF L-SNAs prevent the psoriatic phenotype in 3D raft models.
(a) Time-dependent uptake of 6 nmol/L cyanine 5–L-SNA in NHEKs. DAPI-stained nuclei. Scale bars = 20 μm. (b) Quantitative PCR of NHEKs pretreated with 10 ng/ml human TNF-α or PBS for 24 hours and incubated with L-SNAs or PBS (0 nmol/L L-SNA) for 48 hours. (c) Cyanine 5–L-SNA (30 μmol/L) in Aquaphor/PBS (1:1) was applied to normal or psoriatic human skin explants in lifted cultures and imaged after 24 hours. Scale bars = 50 μm. (d) Hematoxylin and eosin staining of untreated (NT) and cytokine-treated (cytomix) psoriasis-like rafts after two topical treatments with 50 nmol/L TNF L-SNAs or controls. Scale bars = 50 μm. (e, f) Quantitative PCR analysis of NT or cytokine-treated rafts. Mean ± standard error of the mean, n = 3/group, three runs. Asterisks over columns are in comparison with TNF/PBS (lilac) in b and versus cytomix/burgundy columns in e and f; other comparisons are defined by the ends of the horizontal bar with asterisks (**P < 0.01, ***P < 0.001). 3D, three dimensional; Exp, expression; hr, hour; L-SNA, liposomal-cored spherical nucleic acid; M, mol/L; min, minute; NHEK, normal human epidermal keratinocyte; NT, not treated; PBS, phosphate buffered saline; Scr, scrambled liposomal-cored spherical nucleic acid control sample; SNA, spherical nucleic acid; TNF-α, tumor necrosis factor-α.
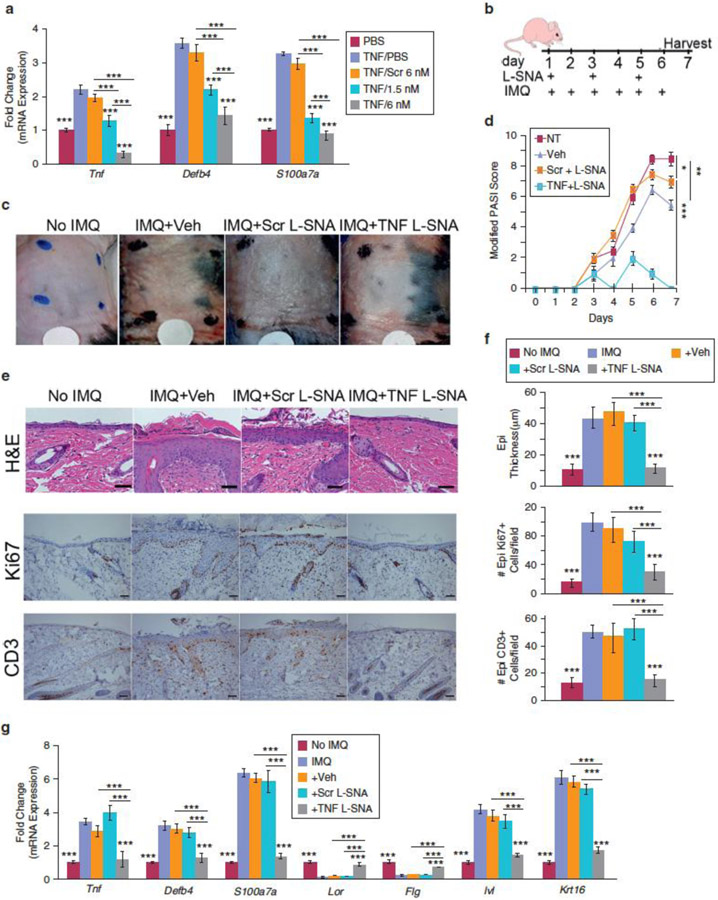
The human TNF-targeted oligonucleotide sequence is 89% homologous with mouse Tnf and is able to knock down TNF-α–induced mouse fibroblast Tnf, Defb4, and S100a7a versus Scr by 84%, 56%, and 70%, respectively (all P < 0.001) (Figure 2a). The psoriasis-like model was established in 6-week-old C57BL/6 male mice by daily application of 62.5 mg IMQ cream (5%) for 6 days (van der Fits et al., 2009). Although no ideal psoriasis mouse model exists and the IMQ model has limitations (Hawkes et al., 2016), its reproducible inflammatory skin response simulates psoriasis clinically, histologically, and transcriptionally. Every other day, mice were treated with a template-defined area with topical formulations of 50 μmol/L TNF L-SNA, 50 μmol/L Scr, Aquaphor (Beiersdorf, Wilton, CT)/phosphate buffered saline (1:1) vehicle, or nothing (untreated). On days with both therapy and IMQ application, the IMQ was applied 10 minutes after the L-SNA or vehicle formulations (Figure 2b). Preliminary studies confirmed that serial application of L-SNAs and IMQ did not alter the effect of either, regardless of the order applied, and showed 50 μmol/L to be the optimal TNF L-SNA dose (see Supplementary Figure S4a and b online). Erythema, scaling, and thickness were serially scored by blinded reviewers as 0 (none) to 4 (very severe) and added together for a modified Psoriasis Area Severity Index score. The clinical (Figure 2c and d), histological/immunohistological (Figure 2e and f), and transcriptional (Figure 2g) profiles of IMQ-treated mouse skin treated with TNF L-SNA were indistinguishable from normal mouse skin without IMQ-induction of the psoriasiform phenotype. Vehicle/Scr-treated mice had improved erythema/scaling, although only on harvest day and not nearly to the extent of TNF L-SNA–treated mice (Figure 2c and d,and see Supplementary Figure S5 online); no histological or transcriptional improvement was noted in these control-treated, IMQ-induced mice versus IMQ-only–treated mice (Figure 2c-g). Histological sections of control-treated, IMQ-induced samples showed parakeratosis, hypogranulosis, acanthosis, increased Ki67 expression, and prominent T-cell infiltrates (Figure 2e and f). Control, but not TNF L-SNA–treated, mice had increased expression of Krt16 (proliferation), Tnf, S100a7a and Defb4 (T helper type 17 pathway), and Ivl (encoding involucrin) and decreased Lor (loricrin) and Flg (filaggrin) (differentiation markers) (Figure 2g).
Figure 2. Topically applied TNF L-SNAs prevent development of the imiquimod-induced psoriasis-like phenotype in mice.
(a) Mouse J2 fibroblasts were pretreated with or without 10 ng/ml mouse TNF for 24 hours and then with 1.5 nmol/L or 6 nmol/L TNF L-SNAs, 6 nmol/L Scr L-SNAs, or PBS for 48 hours. (b) Application schedule. (c, d) Skin was assessed clinically by modified Psoriasis Area Severity Index score; all data shown are with IMQ treatment, with modified Psoriasis Area Severity Index scores for untreated mice all equal to 0. (e) Hematoxylin and eosin and immunohistochemical staining was performed (scale bars = 50 µm), and (f) the extent of epidermal hyperplasia, hyperproliferation, and T-cell infiltration was quantified. (g) Real-time quantitative polymerase chain reaction analysis of mouse skin. Mean ± standard error of the mean, n = 12 mice/group. Asterisks over columns are (a) versus TNF/0 nmol/L-treated NHEK or (f, g) IMQ-only–treated mice controls; others as indicated (***P < 0.001). Epi, epidermal; H&E, hematoxylin and eosin; IMQ, imiquimod; L-SNA, liposomal-cored spherical nucleic acid; M, mol/L; NT, not treated; PBS, phosphate buffered saline; Scr, scrambled liposomal-cored spherical nucleic acid control sample; TNF, tumor necrosis factor-α; Veh, vehicle.
The ability of TNF L-SNA to penetrate human psoriatic skin and completely reverse the development of 3D human and IMQ-treated mouse model phenotypes suggests the therapeutic potential for topically applied SNA-mediated antisense therapy. Although TNF-α was chosen for this proof-of-concept study, the ease of altering the molecular target by changing the oligonucleotide sequence emphasizes the broad potential applicability of SNA topical therapeutics.
Supplementary Material
Acknowledgments
We thank Chad Mirkin for developing the SNA technology. We thank Denise Akuamoah for assistance in dosing IMQ and L-SNAs. This research was supported by NIAMS R01 AR060810 (ASP), NIAMS R41 AR066438 (DAG, ASP), the International Institute for Nanotechnology seed grant, and the Max Moser psoriasis research fund. Core resources were provided by Northwestern University’s Skin Disease Research Center (NIAMS P30AR057216) and the Northwestern University Center for Advanced Microscopy (NCI CCSG P30CA060553).
Abbreviations
- 3D
three dimensional
- IMQ
imiquimod
- L-SNA
liposomal-cored spherical nucleic acid
- NHEK
normal human epidermal keratinocyte
- Scr
scrambled liposomal-cored spherical nucleic acid control sample
- SNA
spherical nucleic acid
- TNF-α
tumor necrosis factor-α
Footnotes
Conflict of Interest
WD, RK, and DAG are employees of Exicure, Inc. ASP is a member of the Exicure Scientific Advisory Board and has received Exicure, Inc. stock options.
References
- Banga RJ, Chernyak N, Narayan SP, Nguyen ST, Mirkin CA. Liposomal spherical nucleic acids. J Am Chem Soc 2014;136:9866e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiricozzi A, Guttman-Yassky E, Suarez- Farinas M, Nograles KE, Tian S, Cardinale I, et al. Integrative responses to IL-17 and TNF- alpha in human keratinocytes account for key inflammatory pathogenic circuits in psoriasis. J Invest Dermatol 2011;131:677e87. [DOI] [PubMed] [Google Scholar]
- Choi CH, Hao L, Narayan SP, Auyeung E, Mirkin CA. Mechanism for the endocytosis of spherical nucleic acid nanoparticle conjugates. Proc Natl Acad Sci USA 2013;110(19): 7625e30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutler JI, Auyeung E, Mirkin CA. Spherical nucleic acids. J Am Chem Soc 2012;134:13791. [DOI] [PubMed] [Google Scholar]
- Di Cesare A, Di Meglio P, Nestle FO. The IL-23/ Th17 axis in the immunopathogenesis of psoriasis. J Invest Dermatol 2009;129:1339–1350 [DOI] [PubMed] [Google Scholar]
- Ettehadi P, Greaves MW, Wallach D, Aderka D, Camp RD. Elevated tumour necrosis factor- alpha (TNF-alpha) biological activity in psoriatic skin lesions. Clin Exp Immunol 1994;96:146e51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giljohann DA, Seferos DS, Prigodich AE, Patel PC, Mirkin CA. Gene regulation with polyvalent siRNA-nanoparticle conjugates. J Am Chem Soc 2009;131:2072e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harden JL, Krueger JG, Bowcock AM. The immunogenetics of psoriasis: A comprehensive review. J Autoimmun 2015;64:66e73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawkes JE, Gudjonsson JE, Ward NL. The snowballing literature on imiquimod-induced skin inflammation in mice: a critical appraisal. J Invest Dermatol 2016;137:546e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowes MA, Bowcock AM, Krueger JG. Pathogen- esis and therapy of psoriasis. Nature 2007;445(7130):866e73. [DOI] [PubMed] [Google Scholar]
- Lowes MA, Chamian F, Abello MV, Fuentes-Duculan J, Lin SL, Nussbaum R, et al. Increase in TNF-alpha and inducible nitric oxide synthase-expressing dendritic cells in psoriasis and reduction with efalizumab (anti-CD11a). Proc Natl Acad Sci USA 2005;102(52): 19057e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowes MA, Suarez-Farinas M, Krueger JG. Immunology of psoriasis. Annu Rev Immunol 2014;32:227e55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randeria PS, Seeger MA, Wang XQ, Wilson H, Shipp D, Mirkin CA, et al. SiRNA-based spherical nucleic acids reverse impaired wound healing in diabetic mice by ganglioside GM3 synthase knockdown. Proc Natl Acad Sci USA 2015;112(18): 5573–5578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Fits L, Mourits S, Voerman JS, Kant M, Boon L, Laman JD, et al. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol 2009;182:5836e45. [DOI] [PubMed] [Google Scholar]
- Zheng D, Giljohann DA, Chen DL, Massich MD, Wang XQ, Iordanov H, et al. Topical delivery of siRNA-based spherical nucleic acid nanoparticle conjugates for gene regulation. Proc Natl Acad Sci USA 2012;109(30):11975e80. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.