Abstract
Background
Trabeculectomy is performed as a treatment for glaucoma to lower the intraocular pressure (IOP). Mitomycin C (MMC) is an antimetabolite used during the initial stages of a trabeculectomy to prevent excessive postoperative scarring and thus reduce the risk of failure.
Objectives
To assess the effects of intraoperative MMC compared to placebo in trabeculectomy.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) (which contains the Cochrane Eyes and Vision Group Trials Register) (The Cochrane Library Issue 4, 2009), MEDLINE (January 1966 to January 2010), EMBASE (January 1980 to January 2010), LILACS (Latin American and Caribbean Health Sciences Literature Database) (January 1982 to January 2010), OpenSIGLE (January 2010) and the UK Clinical Trials Gateway (UKCTG) (January 2010). We also wrote to investigators of trials included in the review to ask if they were aware of any other studies. There were no language or date restrictions in the search for trials. The electronic databases were last searched on 19 January 2010.
Selection criteria
We included randomised controlled trials (RCTs) of intraoperative MMC compared to placebo or no adjunct in trabeculectomy surgery.
Data collection and analysis
Two authors independently assessed trial quality and extracted data. We contacted trial investigators for missing information.
Main results
Eleven trials, involving a total of 698 participants, were included. The trials enrolled three types of participants (high risk of failure, trabeculectomy combined with cataract surgery, no previous surgical intervention). Mitomycin C appears to reduce the relative risk of failure of trabeculectomy both in eyes at high risk of failure (relative risk 0.32, 95% confidence interval: 0.20 to 0.53) and those undergoing surgery for the first time (relative risk 0.29, 95% confidence interval 0.16 to 0.53). No significant effect on failure was noted in the group undergoing trabeculectomy combined with cataract extraction. Mean IOP was significantly reduced at 12 months in all three participant groups receiving MMC compared to placebo. No significant increase in permanent sight‐threatening complications was detected. However, none of the trials were large enough or of sufficient duration to address the long‐term risk of bleb infection and endophthalmitis which has been reported in observational studies. Some evidence exists that MMC increases the risk of cataract.
Authors' conclusions
Intraoperative MMC reduces the risk of surgical failure in eyes that have undergone no previous surgery and in eyes at high risk of failure. Compared to placebo it reduces mean IOP at 12 months in all groups of participants in this review. Apart from an increase in cataract formation following MMC, there was insufficient power to detect any increase in other serious side effects such as endophthalmitis.
Plain language summary
Mitomycin C for glaucoma surgery
Surgical treatment of glaucoma is usually reserved for serious cases which cannot be controlled by other means such as topical medication or laser. Surgery can be performed in most types of glaucoma in order to reduce the pressure inside the eye which if left uncontrolled can irreversibly damage the optic nerve leading to loss of sight. There are a number of variants of drainage surgery but the most commonly performed procedure is trabeculectomy in which a guarded channel is fashioned through the wall of the eye under the upper lid and the fluid is allowed to collect under the conjunctiva (the membrane lining the eyeball) to form a drainage bleb. Scarring during the healing process can cause this channel to close and the operation to fail with a rise in pressure. Mitomycin C is a powerful agent which prevents scarring by inhibiting the multiplication of cells which produce scar tissue. This review asks whether there is evidence that its use during the initial stages of surgery to prevent the excessive conjunctival scarring reduces the risk of failure of the operation. Three types of patient were included: those at high risk of failure because of previous failed surgery or other complications, those having combined cataract and glaucoma surgery and those having primary trabeculectomy ‐ an operation for the first time for their glaucoma. The review found evidence that Mitomycin C reduces the risk of surgical failure in both high risk and primary surgery but no evidence on combined cataract and glaucoma surgery. But the risk of adverse effects including an increased risk of cataracts (not in the combined group) was also noted. There were only a few studies on each category of patients and most were of only poor or moderate quality.
Summary of findings
for the main comparison.
| Intraoperative Mitomycin C compared with no antimetabolite or placebo for trabeculectomy surgery for glaucoma | ||||||
|
Patient or population: People undergoing trabeculectomy surgery with glaucoma Settings: Eye clinics and hospitals Intervention: Intraoperative Mitomycin C applied in any dose for any duration Comparison: Placebo application or nothing | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| control | mitomycin C | |||||
| Trabeculectomy failure at 12 months |
Low risk population | RR 0.37 (0.26 to 0.51) | 698 (11) | +++O moderate | medium risk population poorly designed studies may underestimate effect |
|
| 280 per 1000 | 77 per 1000 ([value] to [value]) | |||||
| Medium risk population | ||||||
| 127 per 1000 | 135 per 1000 ([value] to [value]) | |||||
| High risk population | ||||||
| 505 per 1000 | 156 per 1000 ([value] to [value]) | |||||
| mean intraocular pressure mmHg at 12 months |
The mean IOP ranged across control groups from 15.9 to 23.9 mmHg | The mean IOP in the intervention groups was 11.1 to 14.6 mmHg | the WMD was ‐4.1 mmHg [‐4.68 to ‐3.34] mmHg |
380 [8] | +++O moderate | |
| complications wound leak by 12 months |
Low risk population | RR 1.84 (0.72 to 4.66) | 333 (7) | ++OO low | no events reported in trials of high risk patients | |
| 45 per 1000 | 114 per 1000 ([value] to [value]) | |||||
| Medium risk population | ||||||
| 84 per 1000 | 112 per 1000 ([value] to [value]) | |||||
| High risk population | ||||||
| inestimable | inestimable | |||||
| complications hypotony occurring up to 12 months |
Low risk population | RR 1.8 (0.79 to 4.12) | 488 (10) | ++OO low | inconsistently defined and reported | |
| 58 per 1000 | 61 per 1000 ([value] to [value]) | |||||
| Medium risk population | ||||||
| 14 per 1000 | 37 per 1000 ([value] to [value]) | |||||
| High risk population | ||||||
| 31 per 1000 | 83 per 1000 ([value] to [value]) | |||||
| complications shallow anterior chamber occurring within 12 months |
Low risk population | RR 1.14 (0.42 to 3.07) | 441 (10) | ++OO low | inconsistently defined and reported | |
| 169 per 1000 | 151 per 1000 ([value] to [value]) | |||||
| Medium risk population | ||||||
| 0 per 1000 | 9 per 1000 ([value] to [value]) | |||||
| High risk population | ||||||
| 145 per 1000 | 200 per 1000 ([value] to [value]) | |||||
| cataract formation by 12 months | Low risk population | RR 1.8 (1.00 to 3.22) | 482 (7) | +++O moderate | outcome not relevant to medium risk population because these are combined cataract extraction and glaucoma procedures | |
| 93 per 1000 | 190 per 1000 ([value] to [value]) | |||||
| High risk population | ||||||
| 57 per 1000 | 80 per 1000 ([value] to [value]) | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk Ratio; [other abbreviations, e.g.. OR, etc] | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
Background
Introduction
Lowering intraocular pressure (IOP) was established as a treatment for glaucoma more than 100 years ago. For some time, there was a lack of good evidence that lowering IOP is effective in preventing continuing loss of visual function in glaucoma (Rossetti 1993), but more recently, new trials have been completed which indicate lowering IOP reduces the risk of converting to POAG from Ocular Hypertension (OHTS 2002) and the risk of progression in early manifest disease (EMGT 2002). More recently still, a systematic review of the effectiveness of lowering IOP in glaucoma concludes that there is now better evidence to support this intervention (Maier 2005).
In terms of controlling IOP and preventing loss of visual field, a recent Cochrane review (Burr 2004) comparing the effectiveness of surgery versus medicine for glaucoma found there was evidence of slightly better IOP control with surgery though no evidence of any difference in visual field progression. There was evidence of loss of some detailed vision in the short‐term for those having surgery and some disbenefits in terms of ocular discomfort and vision related quality of life.
Glaucoma surgery of this kind is conducted under local or general anaesthesia often as a 'day‐case' or outpatient procedure in western countries but practice varies widely according to local resources and access to follow‐up care.
The trabeculectomy operation involves separating the conjunctiva from the sclera by making an incision at the junction of the cornea and the sclera (on the part of the eye normally hidden under the upper eyelid), to form a conjunctival flap that is folded back to expose the underlying sclera. A half‐thickness incision is made in to the sclera (usually 4 x 4 mm) at the corneo‐scleral junction. The half‐thickness scleral flap is raised towards the limbus and a small section of the sclera under the flap is removed (sclerostomy) allowing aqueous to leave the anterior chamber of the eye. The scleral flap is repositioned and loosely sutured. The flap guards the sclerostomy, preventing excessive egress of aqueous that leads to hypotony (a very soft eye). Finally the conjunctiva is replaced. Aqueous passes through the sclera and collects under the conjunctiva as a bleb. Fluid in the bleb is absorbed by capillaries and lymphatics within the conjunctiva or evaporates across the conjunctiva. Final IOP is determined by many factors including the size of the bleb, the thickness of the conjunctiva and how adherent the conjunctiva around the bleb is to the sclera. If the conjunctiva overlying the operation site scars down onto the scleral flap then less aqueous can leave the eye, resulting in the return of raised IOP.
Various risk factors are thought to produce exaggerated postoperative wound healing, producing a hypercellular response in the subconjunctival tissues thereby restricting the flow of aqueous across the conjunctiva. Risk factors for postoperative failure include previous surgical interventions such as cataract surgery or previous glaucoma surgery and prolonged exposure to eye drops (Lavin 1990) especially sympathomimetics such as adrenaline. Age is inversely related to risk and being of black African ethnic origin is thought by some to be a risk factor (Broadway 1994).
Antimetabolites, including Mitomycin C (MMC) and 5‐Fluorouracil, were first used to modify the wound healing response in the early 1980s. Mitomycin C's alkylating properties inhibit DNA replication, which led to its use first as an anti‐cancer drug. Much of the laboratory work on MMC's effectiveness followed a clinical report by Chen (Chen 1990). At the concentrations used clinically MMC inhibits or kills the fibroblast cells involved in the scarring response (Khaw 1992b). It increases the success rate of filtration surgery in animal models of filtration surgery (Khaw 1992a). The main side effect from inhibiting wound healing is that the conjunctiva overlying the sclerostomy may become very thin. In the early postoperative period, flow of aqueous through the sclerostomy may be greater leading to hypotony. With longer follow‐up holes can form in the conjunctiva that permit bacteria to enter the eye, triggering endophthalmitis. Finally it has been argued that MMC penetrates the sclera and has a direct toxic effect on the ciliary body and epithelium, possibly reducing the production of aqueous.
Rationale for a systematic review
We have already reviewed the effect of postoperative subconjunctival 5‐fluorouracil injections compared to control on trabeculectomy (Wormald 2001). There have been no systematic reviews undertaken to summarise the totality of evidence for MMC augmentation of trabeculectomy. A review of wound healing in glaucoma filtering surgery was written before Chen first used MMC (Skuta 1987). A subsequent editorial in 1992 called for more randomised controlled trials on the use of antimetabolites in filtering surgery to answer important questions on who should and should not receive these agents (Parrish 1992). In reviewing combined glaucoma and cataract surgery in another editorial, Shields suggested that antimetabolites might be useful in improving the success of this procedure but again called for more evidence of effectiveness (Shields 1993). A more recent editorial (Higginbotham 1996) raised valid concerns that the widespread use of antimetabolites in glaucoma surgery has the potential to do as much harm as good and urged caution. Chen's survey in the US and Japan reveals wide variation in practice reflecting the underlying uncertainty over the indications for antimetabolite usage (Chen 1997).
Objectives
The objective of this review was to assess the effects of intraoperative Mitomycin C compared to placebo on the success rate of trabeculectomy and to examine the balance of risk and benefit at one year follow‐up.
Methods
Criteria for considering studies for this review
Types of studies
We included only RCTs. The minimum length of follow‐up was 12 months.
Types of participants
We considered three separate subgroup populations: 1. High risk of failure: people who have had previous glaucoma drainage surgery or previous surgery involving anything more than trivial conjunctival incision, including cataract surgery; people with one or more of the following forms of glaucoma: glaucoma secondary to intraocular inflammation, congenital glaucoma and neovascular glaucoma. 2. Combined surgery: people undergoing trabeculectomy with extra‐capsular cataract extraction and intraocular lens implant. 3. Primary trabeculectomy: people who have received no previous surgical intervention as defined above. This group may include people who have had previous medical therapy, laser procedures or both.
Types of interventions
We included trials in which intraoperative Mitomycin C was administered at any concentration and dose compared to placebo or control.
Types of outcome measures
Primary outcomes
The primary outcomes were the proportion of failed trabeculectomies at 12 months after surgery and the mean IOP at 12 months after surgery. Failure was defined in this review as repeat surgery or uncontrolled IOP (usually more than 22 mmHg) despite additional topical or systemic medications.
Secondary outcomes
The secondary outcomes were adverse event rates in either group with reference to:
wound leaks: the presence of a positive Seidel test (visible aqueous flow with the tear film stained with Fluorescein);
hypotony: IOP is below 5 mmHg or is associated with complications such as macular oedema and sight loss or choroidal detachments;
late endophthalmitis: an infection of the globe contents that, even with prompt aggressive treatment, often results in substantial loss of visual function. 'Late' here implies infection arising from organisms gaining access to the globe through thin‐walled drainage blebs or frank breaks in the conjunctival epithelium after the immediate postoperative period when infectious agents may have entered the eye during the surgical procedure;
expulsive haemorrhage: choroidal haemorrhage usually at the time of surgery or during the early postoperative period while the eye is still soft leading to marked rise in IOP;
shallow anterior chamber: prolonged shallowing of the anterior chamber giving rise to concern over possible contact of the lens with the cornea, occurring as a result of excessive drainage or choroidal effusions or both, leading to anterior displacement of the ciliary body, iris and lens;
cataract: reduction in the optical clarity of the eyes' natural lens producing sufficient visual disturbance to require surgery.
Tertiary outcomes were quality of life assessments or patient perspectives.
Other definitions for general eye‐related terms can be found in the glossary contained within the Eyes and Vision Group Module in The Cochrane Library.
Search methods for identification of studies
Electronic searches
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) (which contains the Cochrane Eyes and Vision Group Trials Register) (The Cochrane Library Issue 4, 2009), MEDLINE (January 1966 to January 2010), EMBASE (January 1980 to January 2010), LILACS (Latin American and Caribbean Health Sciences Literature Database) (January 1982 to January 2010), OpenSIGLE (January 2010) and the UK Clinical Trials Gateway (UKCTG) (January 2010).There were no language or date restrictions in the search for trials. The electronic databases were last searched on 19 January 2010.
See: Appendices for details of search strategies for CENTRAL (Appendix 1), MEDLINE (Appendix 2), EMBASE (Appendix 3), LILACS (Appendix 4), OpenSIGLE (Appendix 5) and UKCTG (Appendix 6).
Searching other resources
We searched the reference lists of identified trials to find additional trials. We used the Science Citation Index to find studies which had cited the identified trials. We contacted the investigators of the identified trials and we asked a leading practitioner in the field of wound healing to contact his colleagues to identify additional published and unpublished studies.
Data collection and analysis
Selection of trials
Two authors independently screened the titles and abstracts resulting from the searches. We obtained full copies of any report referring to possibly or definitely relevant trials. There was no language restriction in the selection of trials and resources were available for translation where necessary. We graded all selected studies according to their concealment of treatment allocation. We put studies into one of three categories as follows.
Category A: adequate concealment
Centralised randomisation.
Sequential administration of pre‐numbered or coded containers.
On‐site computer system.
Serially numbered sealed opaque envelopes.
Other approaches appearing to offer adequate concealment.
Category B: unclear
Randomisation used but method not stated.
List or table used.
Envelopes but no qualifying statement.
An apparently adequate concealment but other information in trial indicates concealment may not have been adequate.
Category C: inadequate
Alternation.
Case record numbers.
Dates of birth.
Days of the week.
Any allocation that is entirely transparent before allocation.
We included in this review only trials coded as category A or B.
Assessment of methodological quality
We assessed study quality using guidelines produced by the Cochrane Eyes and Vision Group. Studies were assessed by using the replies 'Yes', 'No', and 'Unclear' to the following questions about each trial.
Performance bias (1) Were the recipients of care unaware of their assigned treatment? (2) Were persons providing care unaware of the assigned treatment?
Detection bias (3) Were persons responsible for outcome assessments unaware of the assigned treatment?
Attrition bias (4) Were rates of follow‐up similar in the comparison groups? (5) Was the analysis 'intention‐to‐treat'?
Any trial that received a 'No' answer to any question was deemed to be at high risk of bias.
Data collection
Two authors independently extracted data using a form developed by the Cochrane Eyes and Vision Group. We compared the results and resolved discrepancies by discussion.
Data synthesis
Data synthesis was performed according to the Cochrane Eyes and Vision Group statistical guidelines. We summarised data for the probability of failure at 12 months using relative risk. Statistical heterogeneity was detected only in the overall analysis of mean IOP (Chi2 test). Thus for the analysis of mean IOP we used both fixed and random‐effects models. Otherwise we only applied fixed‐effects models. We used odds ratios to summarise data for rarer events such as most complications. We performed a sensitivity analysis to determine the effect of excluding trials deemed to be at high risk of bias.
Results
Description of studies
Results of the search
The Initial electronic searches revealed more than 70 reports. We identified 11 RCTs that met the review inclusion criteria. One trial (Shin 1996) was excluded because it was unclear whether it included data from a similar trial the year before. Attempts made to contact the lead author were unsuccessful. Another trial (Chen 2001) was excluded because (a) although the mean follow up was 28 months, no results were expressed for one year and (b) because it was not possible to determine whether the high proportion of participants in the study with angle‐closure glaucoma had any prior inflammatory or surgical events, thus preventing assessment of the participants' risk of failure. Despite contact with the authors of identified trials and with researchers active in the field we have not identified any further relevant studies.
Updated searches conducted in March 2005, which included searching three additional databases (SIGLE, National Research Register and LILACS), yielded no new trials.
The update search in January 2010 retrieved 209 reports and one potentially relevant trial was found (Reibaldi 2008). This is a report of long‐term outcomes of the use of lower dose topical MMC in primary trabeculectomy compared to a smaller control group who received balanced salt solution. We have not yet been able to retrieve outcomes at one year but hope to receive these from the author (with whom we have been in touch) in due course. The Kaplan Meier survival curves printed in this report appear to show no differences in survival for at least the first 15 months of follow‐up for any of the reported outcome measures; IOP less than 18 mmHg, IOP less than 14 mmHg and visual field progression.
Included studies
Types of participants
Trials reported participants from three subgroups: those at high risk of failure (Andreanos 1997; Turacli 1996; Wu 1996), those undergoing combined cataract and trabeculectomy (Carlson 1997; Cohen 1996; Shin 1995) and those undergoing trabeculectomy for the first time after failure to control IOP with medical intervention (Costa 1996; Martini 1997; Robin 1997; Szymanski 1997). Although one study concerned the use of MMC in combined surgery, it was included in the high risk group because the patients had already undergone filtering surgery (Shin 1998). This study also included a primary glaucoma triple procedure group matched to the secondary group (having previously undergone trabeculectomy) for numerous characteristics and allocated to MMC or control on the basis of the matching. This group was therefore indirectly randomised and could theoretically be included in the combined procedure comparisons. However, we had no way of being sure whether these patients had been participants in the previously reported trial (Shin 1995). No trial included more than one type of participant.
Types of intervention
Mitomycin C was administered intraoperatively in concentrations of 0.1 to 0.5 mg/ml saline for durations varying from one to five minutes.
Types of outcome measure
All the trials reported failure rates at 12 to 25 months follow‐up. Exact definitions of failure varied across trials but an inclusive definition is IOP at or above 21 mmHg with or without medications (Table 2). Three trials (Robin 1997; Shin 1998; Szymanski 1997) failed to report mean IOP and standard deviation at 12 months. One trial (Wu 1996) reported IOP using kilopascal (kPa) units that required conversion to mmHg by multiplying by a factor of 7.5. Complications reported in the trials included wound leaks, choroidal effusions, supra choroidal haemorrhage, hypotony, hyphaema and cataract. None of the trial reports provided any information relating to the participants' perspective. See 'Characteristics of included studies' table.
1. Mean baseline IOP and failure criteria.
| Trial | Mean baseline IOP | Failure criterion |
| Shin 1995 | 20.8 mmHg | < 20 mmHg plus > 20% fall in baseline IOP, without topical medication |
| Cohen 1996 | 21.3 mmHg | Eye requires additional glaucoma surgery |
| Costa 1996 | 25.6 mmHg | < 15 mmHg, without topical medication |
| Turacli 1996 | 31.9 mmHg | < 20 mmHg or > 25% fall in baseline IOP, without topical medication |
| Wu 1996 | 40.3 mmHg | < 21 mmHg , without topical medication |
| Andreanos 1997 | 32.4 mmHg | < 21 mmHg, without topical medication |
| Carlson 1997 | 18.8 mmHg | Not specified but results show all eyes were < 21 mmHg, topical medication was used |
| Martini 1997 | 28.2 mmHg | < 18 mmHg on or off topical medication |
| Robin 1997 | 29.7 mmHg | < 19 mmHg on or off topical medication |
| Szymanski 1997 | Not specified | < 15 mmHg on topical medication |
| Shin 1998 | 17.6 mmHg | Eye requires glaucoma surgery, needling or topical medication |
Risk of bias in included studies
The general quality of the reporting of studies was low. Only three studies were initially graded at low risk of bias (Carlson 1997; Cohen 1996; Robin 1997). A response from one author allowed us to upgrade the quality assessment of that trial (Cohen 1996). The remaining studies were all graded at high risk of bias.
We have subsequently added risk of bias analyses Figure 1 and Figure 2 and summary of findings tables Table 1. This has lead to a re‐evaluation of some studies including Cohen 1996 where we identified high risk of bias on two counts. Details of these concerns are provided under the risk of bias table for each study.
1.
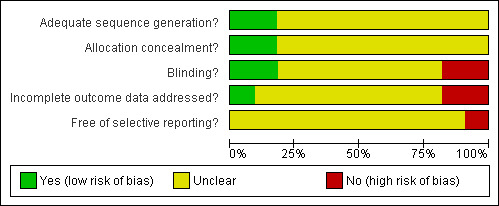
Methodological quality graph: review authors' judgements about each methodological quality item presented as percentages across all included studies.
2.

Methodological quality summary: review authors' judgements about each methodological quality item for each included study.
Effects of interventions
See: Table 1
Primary outcomes
Failure at 12 months (SeeAnalysis 1.1) 1. High risk of failure group A total of 193 participants who were classified as being at high risk of failure were randomised in four trials (Andreanos 1997; Shin 1998; Turacli 1996; Wu 1996). The results show a statistically significant benefit of intraoperative MMC, that is, protection against failure of surgery at 12 months (relative risk (RR) 0.32, 95% confidence interval (CI): 0.20 to 0.53).
1.1. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 1 Failure at 12 months.
2. Combined surgery group A total of 167 participants who were undergoing combined surgery were randomised in three trials (Carlson 1997; Cohen 1996; Shin 1995). No benefit of intraoperative MMC was found (RR 0.81 95% CI: 0.37 to 1.80).
3. Primary trabeculectomy group A total of 338 participants who were having surgery for the first time were randomised in four trials (Costa 1996; Martini 1997; Robin 1997; Szymanski 1997). The results show a statistically significant benefit of MMC (RR 0.29, 95% CI: 0.16 to 0.53), a 71% reduction in risk of failure.
Mean IOP at 12 months ‐ fixed‐effect reported (SeeAnalysis 1.2) 1. High risk of failure group Three trials reported that, compared to placebo, MMC produced a statistically significant difference in the decrease of IOP from baseline and 12 months (Andreanos 1997; Turacli 1996; Wu 1996). The weighted mean difference in all trials combined indicates that MMC lowers IOP by 5.31 mmHg more than placebo (95% CI: 3.85 to 6.76 mmHg). The fourth study (Shin 1998) did not report mean IOP of the MMC and control treatment arms.
1.2. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 2 Mean intraocular pressure at 12 months.
2. Combined surgery Of the three relevant trials, two indicate that MMC is statistically significantly better than control in effecting a decrease in IOP at 12 months compared to baseline (Carlson 1997; Cohen 1996) whereas one does not (Shin 1995). When all three studies are combined, the overall effect favours MMC over placebo (mean difference in decrease from baseline 3.34 mmHg, 95% CI: 2.51 to 4.16 mmHg).
3. Primary trabeculectomy Only two trials provide IOP figures at 12 months (Costa 1996; Martini 1997) and the mean IOP reduction in the trials is similar. The pooled estimate of effect is a lowering of IOP that favours MMC over placebo (mean difference in decrease from baseline 5.41 mmHg, 95% CI: 3.48 to 7.34 mmHg).
Secondary outcomes
Wound leak (SeeAnalysis 1.3) Wound leaks were reported as outcomes in two trials in the high risk of failure groups (Turacli 1996; Wu 1996). There were no events in either MMC or placebo groups in these trials. In the combined surgery group, all three trials reported wound leaks although the incidence of leaks was very variable across studies. The pooled effect shows no significant differences in wound leaks between groups using MMC and those using placebo. Wound leak was reported as an outcome in two primary trabeculectomy trials (Costa 1996; Szymanski 1997). Although there were more events in the MMC group this difference was not significant. Wound leak was mentioned in one further trial in the primary trabeculectomy group (Robin 1997) but not in sufficient detail to be included in the analysis.
1.3. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 3 Complications ‐ wound leak.
Hypotony (SeeAnalysis 1.4) Although the point estimate in all three risk groups show an increase in the risk of hypotony with MMC, the confidence intervals are either side of unity meaning that the estimates are not significant. This is repeated when all the trials are analysed together.
1.4. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 4 Complications ‐ hypotony.
Endophthalmitis (SeeAnalysis 1.5) Only three trials reported endophthalmitis as an outcome. Two of the trials included participants in the combined surgery group (Carlson 1997; Shin 1995). Two cases were reported and both occurred in the MMC group. The confidence intervals of the point estimate for the pooled effect are however wide and the results are consistent with there being no effect. Robin 1997, from the primary trabeculectomy group, reported that no cases of endophthalmitis occurred (0/229 in the MMC group compared to 0/71 in the control group).
1.5. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 5 Complications ‐ late endophthalmitis.
Expulsive haemorrhage (SeeAnalysis 1.6) No trial reported cases of expulsive haemorrhage.
1.6. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 6 Complications ‐ expulsive haemorrhage.
Shallow anterior chamber (SeeAnalysis 1.7) Shallow anterior chamber occurred at markedly varying rates from 0/57 to 8/30 across MMC and control groups. This probably reflects variations in definition and also surgical technique. However, for each risk group and overall, there was no significant difference with respect to shallow anterior chamber.
1.7. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 7 Complications ‐ shallow anterior chamber.
Cataract (SeeAnalysis 1.8) In one trial, which included participants from the primary trabeculectomy group, there was a statistically significant increase in the risk of cataract associated with the use of MMC (Robin 1997). Using a fixed‐effect model, the pooled estimates of effect showed that the risk of cataract was possibly increased with MMC use in trials of participants in the primary trabeculectomy group (RR 1.93, 95% CI: 0.98 to 3.80), as well as for all participant groups analysed together (RR 1.80, 95% CI: 1.00 to 3.22).
1.8. Analysis.
Comparison 1 Intraoperative Mitomycin C versus control, Outcome 8 Complications ‐ cataract.
Numbers needed to harm Cataract was the only side effect to be significantly increased by using MMC. The numbers needed to harm with MMC to cause the production of one additional cataract was 15.
Sensitivity analyses
Repeat analysis using only the three studies that we rated as being at low risk of bias did not produce any different conclusions. No sensitivity analysis could be performed for cataract since two of the three trials were performed on eyes undergoing trabeculectomy combined with cataract extraction.
Table 1 gives an overview of the evidence of safety and effectiveness which this review provides and it can be seen that, at best, the trials provide evidence of only moderate quality ‐ this being due to poor trial design and risk of bias in almost all studies.
Discussion
The main finding is that while intraoperative Mitomycin C (MMC) is better than placebo in terms of lower one year failure rates in people at high risk of failure or undergoing primary surgery, there is no significant effect on those undergoing combined cataract extraction and trabeculectomy. This is despite the fact that MMC has a significantly greater IOP lowering effect than placebo across all three participant groups. In the combined trabeculectomy and cataract extraction studies the mean preoperative IOP ranges from 17.6 to 21.3 mmHg. In the primary trabeculectomy group the figure is 25.6 to 29.7 mmHg, and in the high risk group it is 20.8 to 40.3 mmHg with three of the four trials having a baseline IOP > 30 mmHg (Table 2). Using an IOP threshold of 21 mmHg as an outcome measure, it is much less likely that failure will occur if participants enter the study with an IOP that labels them as a success. Much larger samples would be required to show an effect, if it is present.
The risk of cataract is the only side effect significantly increased by MMC, furthermore this effect is only seen in one trial. The failure to confirm many ophthalmologists' anecdotal evidence of increased complications such as hypotony and endophthalmitis is most likely due to:
low event rates;
Low event rates coupled with variations in diagnosis mean that very large numbers may be required to show a significant effect. Other descriptive work has suggested that MMC results in an incidence of endophthalmitis of one per cent per year (Greenfield 1996); our figures reveal a rate lower than this: only two cases of endophthalmitis were reported, both were in combined surgery participants receiving intraoperative MMC. With no cases occurring in the placebo group, even if this represents a real difference between treatment and control, the combined size of the trials is not enough to eliminate the possibility that the observed difference is due to chance. Risk of cataract formation was the only side effect shown to be significantly increased by MMC, it was also the side effect with highest event rate. Thus the combined trials were powerful enough to detect the effect of MMC.
increasing success in preventing complications;
As surgeons have recognised MMC's ability to produce wound leak and hypotony they have developed techniques to reduce them. Retrospective studies have quoted figures from 1.3% to 13% (Costa 1993; Suner 1997; Zacharia 1993). Meticulous care with conjunctival handling plus careful suturing of the conjunctiva and sclera has reduced the incidence of such complications (Suner 1997).
variable definitions.
Side effects such as wound leak can be hard to assess. Anterior chamber depth was not assessed using a pachymeter in any of these studies.
It remains essential to be vigilant about such complications even with low event rates. The outcome of endophthalmitis is poor, as is the long‐term outcome from mitomycin hypotony. Only one of Costa's five patients with hypotony regained preoperative visual acuity after five months of follow‐up (Costa 1993). A recent review of the experience of one centre using MMC in trabeculectomy for normal tension glaucoma found a dramatically increased incidence of late bleb leaks more than three months after the surgery (Membrey 2000).
Given that trabeculectomy is performed with the intention of preserving vision, any reduction in visual function due to cataract is detrimental. All intraocular surgery is recognised as potentially being cataractogenic. Trabeculectomy is no exception, involving intraocular inflammation, alteration of aqueous flow, changes in the blood eye barrier and prolonged exposure to topical steroids. Two glaucoma trials involving trabeculectomy have reported high rates of cataract. In the AGIS study 37.1% of white participants undergoing trabeculectomy developed cataract (AGIS 1998a), while in the Collaborative Normal‐tension Glaucoma Study Group trial 38% of patients treated with filtration surgery developed lens opacities (CNTG 1998a; CNTG 1998b). In the latter study it was possible to detect a difference in visual field outcomes between treated and untreated patients only by censoring the data of those patients developing cataract (CNTG 1998b). Unfortunately the use of antimetabolites was not reported in these studies. However if, as suggested in this review, the use of intraoperative MMC produces an additional risk of cataract formation, then any field preserving effect achieved by MMC's additional IOP lowering may be offset by an increase in cataract formation.
A major problem when discussing the results is the high risk of bias in most studies, as determined using simple criteria. The failure of many trialists to respond to requests for more information means that all interpretation should continue to be performed with caution.
This systematic review has only been able to deal with the effect of MMC on failure, IOP and complications over one year. A patient with glaucoma may be followed for decades; both patient and ophthalmologist would benefit from information on the preservation of visual function. In none of the studies in this review was visual field preservation used as an outcome measure. Furthermore our analysis of failure at one year is hindered by the variable criteria used for this outcome (Table 2). The criteria employed include different threshold IOPs for failure and vary as to whether being on drops constitutes failure. We have reviewed data from the trials using variable criteria in a bid to be inclusive. However, permitting patients on drops to be considered successes will underestimate the size of MMC's effect. We have not presented a sub‐analysis segregating trials with different failure criteria because of the heterogeneous nature of the data.
A notable omission from all the trials was any report of the participants' perception of their treatment, where they are exposed to potentially vision threatening risks of hypotony, cataract and endophthalmitis. It is also worth noting that the relative risk of failing following intraoperative MMC are similar to those obtained in the Cochrane systematic review of postoperative 5‐Fluorouracil injections (Wormald 2001). However, the poor quality of the presented evidence means that any interpretation should be made with caution.
We have not analysed the variations in dose of MMC applied. The dose of MMC applied in the reviewed studies was varied by altering application time (one to five minutes) and concentration (0.1 to 0.5 mg/ml saline). It is known from work using radio labelled 5‐Fluorouracil on animal cadaver eyes that peak conjunctival and scleral concentrations are reached within three minutes of application (Wilkins 2000). What is not known is the effect of varying Mitomycin time and application on tissue levels and on efficacy. Two studies dealt with variable application times and concentrations of MMC (Robin 1997; Shin 1998) and neither was able to demonstrate a significant difference in risk of failure between the different treatment groups. Robin 1997 did find a significantly increased risk of cataract in eyes treated with 0.2 mg/ml for four minutes compared to the other groups: control, 0.2 mg/ml for two minutes and 0.4 mg/ml for two minutes.
Publication bias is always a concern in a systematic review of this nature. Negative studies are less likely to be published and our results should be interpreted with this in mind. We have attempted to identify all trials that have been conducted but there is still a possibility that further trials have been conducted but not reported. We hope that readers of this review who are aware of relevant unpublished data will contact us through the available comments and criticism mechanism in The Cochrane Library so that the review can be modified accordingly.
Authors' conclusions
Implications for practice.
Intraoperative Mitomycin C appears to reduce the risk of surgical failure in eyes that have undergone no previous surgery and in eyes at high risk of failure. Compared to placebo it reduces mean IOP at 12 months in all groups of participants in this review. Apart from an increase in cataract formation following Mitomycin C use, no demonstrable significant increase in other side effects was detected. However, it is possible that low event rates and varying definitions would prevent the detection of a true increase in complications such as infection and hypotony. The quality of evidence supporting these conclusions is at best moderate and often low.
Implications for research.
Given the significant treatment effect shown in individual trials and in this systematic review, future trials involving patients at high risk and patients having a first trabeculectomy are unlikely to compare MMC with placebo. Any definitive report on the risk of side effects of using MMC would require a large number of patients to be randomised to either MMC or placebo. Thus the issue of risk and benefit from MMC is unlikely to be fully answered. Future trials are likely to compare MMC application to either intraoperative 5‐Fluorouracil, postoperative 5‐Fluorouracil or both, since they have come to represent the current minimum practice. However it is worth pointing out the evidence supporting this is not high quality.
The link between IOP and visual field preservation is assumed in the surgical studies reviewed here. Future studies will need to quantify MMC's effect in terms of visual fields. They will also need longer follow‐up to ascertain whether the effect is sustained.
Future MMC trials involving combined procedures will need to modify the design of the trials included in this review; the low preoperative IOP in the existing combined studies militates against finding a significant outcome effect.
What's new
| Date | Event | Description |
|---|---|---|
| 18 January 2010 | New search has been performed | Issue 2 2010: updated searches yielded one potential trial which has been placed in the 'studies awaiting classification' section until one year outcomes are available. Following the completion of risk of bias tables for all included trials, some studies have been re‐assessed. A summary of findings table has also been completed. |
History
Review first published: Issue 1, 2001
| Date | Event | Description |
|---|---|---|
| 2 November 2008 | Amended | Converted to new review format. |
| 16 August 2005 | New citation required and conclusions have changed | Substantive amendment |
Acknowledgements
We are grateful to David Broadway for peer review comments on this review and to the Australasian Cochrane Centre for preparing the synopsis for this review. The Editorial base created and ran the search strategies for the electronic databases.
Appendices
Appendix 1. CENTRAL search strategy
#1 MeSH descriptor Glaucoma #2 MeSH descriptor Filtering Surgery #3 MeSH descriptor Trabeculectomy #4 MeSH descriptor Sclerostomy #5 ((surg* near glaucoma) or filter* or filtrat*) #6 surg* near intra ocular pressure #7 trabeculectom* #8 sclerostom* #9 (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8) #10 MeSH descriptor Mitomycin #11 mitomycin* #12 mytomycin* #13 mitomicin* #14 mytomicin* #15 (#10 OR #11 OR #12 OR #13 OR #14) #16 (#9 AND #15)
Appendix 2. MEDLINE search strategy
1. randomized controlled trial.pt. 2. (randomized or randomised).ab,ti. 3. placebo.ab,ti. 4. dt.fs. 5. randomly.ab,ti. 6. trial.ab,ti. 7. groups.ab,ti. 8. or/1‐7 9. exp animals/ 10. exp humans/ 11. 9 not (9 and 10) 12. 8 not 11 13. exp glaucoma/ 14. exp filtering surgery/ 15. exp trabeculectomy/ 16. exp sclerostomy/ 17. ((surg$ or filter$ or filtrat$) adj5 glaucoma$).tw. 18. ((intra ocular pressure$ or IOP) adj5 surg$).tw. 19. trabeculectom$.tw. 20. sclerostom$.tw. 21. or/13‐20 22. exp mitomycin/ 23. mitomycin$.tw. 24. mytomycin$.tw. 25. mitomicin$.tw. 26. mytomicin$.tw. 27. or/22‐26 28. 21 and 27 29. 12 and 28
The search filter for trials at the beginning of the MEDLINE strategy is from the published paper by Glanville et al (Glanville 2006).
Appendix 3. EMBASE search strategy
1. exp randomized controlled trial/ 2. exp randomization/ 3. exp double blind procedure/ 4. exp single blind procedure/ 5. random$.tw. 6. or/1‐5 7. (animal or animal experiment).sh. 8. human.sh. 9. 7 and 8 10. 7 not 9 11. 6 not 10 12. exp clinical trial/ 13. (clin$ adj3 trial$).tw. 14. ((singl$ or doubl$ or trebl$ or tripl$) adj3 (blind$ or mask$)).tw. 15. exp placebo/ 16. placebo$.tw. 17. random$.tw. 18. exp experimental design/ 19. exp crossover procedure/ 20. exp control group/ 21. exp latin square design/ 22. or/12‐21 23. 22 not 10 24. 23 not 11 25. exp comparative study/ 26. exp evaluation/ 27. exp prospective study/ 28. (control$ or prospectiv$ or volunteer$).tw. 29. or/25‐28 30. 29 not 10 31. 30 not (11 or 23) 32. 11 or 24 or 31 33. exp glaucoma/ 34. exp filtering surgery/ 35. exp trabeculectomy/ 36. exp sclerostomy/ 37. ((surg$ or filter$ or filtrat$) adj5 glaucoma$).tw. 38. ((intra ocular pressure$ or IOP) adj5 surg$).tw. 39. trabeculectom$.tw. 40. sclerostom$.tw. 41. or/33‐40 42. exp mitomycin/ 43. mitomycin$.tw. 44. mytomycin$.tw. 45. mitomicin$.tw. 46. mytomicin$.tw. 47. or/42‐46 48. 41 and 47 49. 32 and 48
Appendix 4. LILACS search strategy
glaucom$ or intra ocular pressure or intraocular pressure or trabeculectom$ or slcerostom$ and mitomycin or mytomycin or mitomicin or mytomicin
Appendix 5. OpenSIGLE search strategy
glaucoma* or trabeculect* or sclerostom* or intraocular pressure or intra ocular pressure AND mitomycin or mytomycin or mitomicin or mytomicin
Appendix 6. UKCTG search strategy
(mitomycin OR mytomycin OR mitomicin OR mytomicin) AND glaucoma%
Data and analyses
Comparison 1. Intraoperative Mitomycin C versus control.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Failure at 12 months | 11 | 698 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.37 [0.26, 0.51] |
| 1.1 High risk | 4 | 193 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.20, 0.53] |
| 1.2 Cataract extraction combined with trabeculectomy | 3 | 167 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.37, 1.80] |
| 1.3 Primary trabeculectomy | 4 | 338 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.29 [0.16, 0.53] |
| 2 Mean intraocular pressure at 12 months | 8 | 380 | Mean Difference (IV, Fixed, 95% CI) | ‐4.01 [‐4.68, ‐3.34] |
| 2.1 High risk | 3 | 144 | Mean Difference (IV, Fixed, 95% CI) | ‐5.31 [‐6.76, ‐3.85] |
| 2.2 Cataract extraction combined with trabeculectomy | 3 | 148 | Mean Difference (IV, Fixed, 95% CI) | ‐3.34 [‐4.16, ‐2.51] |
| 2.3 Primary trabeculectomy | 2 | 88 | Mean Difference (IV, Fixed, 95% CI) | ‐5.41 [‐7.34, ‐3.49] |
| 3 Complications ‐ wound leak | 7 | 333 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.84 [0.72, 4.66] |
| 3.1 High risk | 2 | 98 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Cataract extraction combined with trabeculectomy | 3 | 178 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.88 [0.68, 5.16] |
| 3.3 Primary trabeculectomy | 2 | 57 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.65 [0.16, 17.47] |
| 4 Complications ‐ hypotony | 10 | 488 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.80 [0.79, 4.12] |
| 4.1 High risk | 4 | 193 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.83 [0.76, 10.48] |
| 4.2 Cataract extraction combined with trabeculectomy | 3 | 178 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.65 [0.34, 7.94] |
| 4.3 Primary trabeculectomy | 3 | 117 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.23, 4.68] |
| 5 Complications ‐ late endophthalmitis | 2 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 Cataract extraction combined with trabeculectomy | 2 | 107 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.02 [0.20, 20.17] |
| 6 Complications ‐ expulsive haemorrhage | 10 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 6.1 High risk | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 Cataract extraction combined with trabeculectomy | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.3 Primary trabeculectomy | 4 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Complications ‐ shallow anterior chamber | 10 | 441 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.38 [0.73, 2.63] |
| 7.1 High risk | 3 | 144 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.49 [0.62, 3.60] |
| 7.2 Cataract extraction combined with trabeculectomy | 3 | 178 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.44 [0.13, 91.79] |
| 7.3 Primary trabeculectomy | 4 | 119 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.14 [0.42, 3.07] |
| 8 Complications ‐ cataract | 7 | 482 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.80 [1.00, 3.22] |
| 8.1 High risk | 3 | 144 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.38 [0.45, 4.24] |
| 8.2 Primary trabeculectomy | 4 | 338 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.93 [0.98, 3.80] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Andreanos 1997.
| Methods | Prospective, randomised study with 18 month follow‐up. | |
| Participants | 46 patients with uncontrolled glaucoma who had all had previous filtration surgery. | |
| Interventions | Standard trabeculectomy with or without MMC 0.4 mg/ml for 2 to 3 minutes. | |
| Outcomes | 83.3% IOP < 20 in MMC group. 63.6% IOP < 20 in control group. | |
| Notes | Prolonged hypotony observed in higher percentage in MMC group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Method of sequence generation not stated. |
| Allocation concealment? | Unclear risk | No information on allocation concealment. |
| Blinding? All outcomes | Low risk | Probably single masked ‐ "Postoperative follow up was done by someone unaware of the patient's group". Patient's awareness of treatment group not stated. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Follow‐up ranged from 11 to 34 months with mean of 18 months after the second operation" (meaning the operation at which MMC was applied). It is not clear when the proportion of participants with controlled pressure were counted but the mean postoperative pressures are quoted at 18 months. |
| Free of selective reporting? | Unclear risk | No power statement and no indication of an a priori analysis plan. |
Carlson 1997.
| Methods | Prospective, double‐masked study with 20 month mean follow‐up. | |
| Participants | 29 patients with visually significant cataract and glaucoma. | |
| Interventions | Phacotrabeculectomy with either MMC 0.5 mg/ml for 3.5 minutes or placebo. | |
| Outcomes | 85% of MMC group had IOP 5‐15. 42% of control group had IOP 5‐15. | |
| Notes | Limbus‐based flap with MMC under partial‐thickness flap. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No mention of randomisation method or sequence generation. |
| Allocation concealment? | Unclear risk | Possible ‐ said to be double masked. "A methylcellulose sponge ... was used to apply either the MMC (0.5 mg/ml) or a placebo developed by the authors using dilute gentian violet to match the appearance of the mitomycin.". However no statement is made as to how these solutions were selected or allocated. |
| Blinding? All outcomes | Unclear risk | As above but no detail is provided as to how patient's intervention group was unmasked at the analysis stage. Presumably both patients and investigators were unaware of the intervention status during follow‐up and the authors state at the beginning of the discussion ‐ "In this randomised double masked study..". |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "More than 90% of patients were reviewed at the 8‐ and 12‐ month visit. Follow‐up of one MMC patient ended at 6 months because of a patient's death from pneumonia. All other patients were examined within 4 months of the end of study." |
| Free of selective reporting? | Unclear risk | "Comparisons of IOP and visual acuity were performed using repeated measures analysis of variance, which permits comparison of groups over the entire period studied." There is no power statement and no clear a priori statement of the primary outcome measures of the study. |
Cohen 1996.
| Methods | Prospective, randomised, masked study with 12 month follow‐up. | |
| Participants | 72 patients with cataract and glaucoma for combined surgery. | |
| Interventions | Standard trabeculectomy with peri‐operative sponge soaked with MMC 0.5 mg/ml for 2.5 minutes or placebo applied to scleral incision. | |
| Outcomes | Mean IOP at 12 months: 14.5 (MMC). 17.15 (placebo). | |
| Notes | 17 participants not included in 12 month review. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | "In a masked and random fashion, eyes were assigned to receive either MMC or placebo balanced salt solution." No information is provided on sequence generation. |
| Allocation concealment? | Unclear risk | As above. No mention of allocation concealment beyond "masked and random fashion". |
| Blinding? All outcomes | Unclear risk | Apart from the statement above, follow up examinations are described as follows. "Filtering blebs were evaluated by a masked observer..". "The masked observer obtained study data at 3, 6 and 12 months after surgery". These were separate visits from those to the operating surgeon. This does imply the operating surgeon was in fact aware of the allocation status of the patient which somewhat contradicts the statement above. |
| Incomplete outcome data addressed? All outcomes | High risk | Post randomisation exclusions occurred including protocol violations and reasons relating to the outcome measures ‐ such as uncontrolled intraocular pressure. |
| Free of selective reporting? | High risk | A worrying sentence appears in the statistical analysis paragraph as follows: The study initially planned for 100 patients. When 72 eyes were enrolled, it was found that the variability was less than assumed before surgery, and there was sufficient power to evaluate adequately the ocular hypotensive efficacy of MMC. |
Costa 1996.
| Methods | Prospective, randomised study with 24 month follow‐up. | |
| Participants | 28 patients with uncontrolled POAG or CACG. | |
| Interventions | Standard trabeculectomy with MMC 0.2 mg/ml for 3 minutes or placebo. | |
| Outcomes | 75.7% MMC group were successes. 28.5% of control group were successes. | |
| Notes | 57.1% of thin ischaemic blebs observed in MMC eyes in Palmer's series. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No statement of sequence generation method for random allocation. |
| Allocation concealment? | Low risk | "The surgeon, who was not aware of the solution used intraoperatively, concluded the surgery". Allocation was probably concealed therefore. |
| Blinding? All outcomes | Unclear risk | No mention is made of masking any participant beyond the statement above about the operating surgeon. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Mean follow‐up for each group provided only, with no information about specific losses to follow‐up in either. |
| Free of selective reporting? | Unclear risk | There is no power calculation with no clear statement of primary or secondary outcomes or adverse events. |
Martini 1997.
| Methods | Prospective, randomised one year study. | |
| Participants | 66 eyes of 48 patients with uncontrolled glaucoma. | |
| Interventions | Standard trabeculectomy or with MMC 0.1mg/ml. | |
| Outcomes | Success rate: 96.6% in MMC group. 73.3% in control group. | |
| Notes | 8 participants needed anti‐glaucoma medication to keep IOP < 18 mmHg. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | "Patients were randomised in two groups using a computer‐generated randomisation chart (Stat View 4 program)". |
| Allocation concealment? | Unclear risk | "a) thirty eyes received trabeculectomy with interoperative low‐dose MMC; b) thirty eyes underwent standard trabeculectomy. Twelve patients needed bilateral surgery; the first eye was assigned randomly to MMC or no treatment and the second eye was automatically assigned to the other group.". From this statement it is clear that 12 eyes were not randomised but it not clear how the authors then managed to have 30 eyes in either group. |
| Blinding? All outcomes | Unclear risk | "The control visits were done by personnel unaware of which group the patient or eye belonged to and recorded on special cards which were later analysed for statistical purposes." |
| Incomplete outcome data addressed? All outcomes | Low risk | There appears to be no loss of follow‐up at one year for either group. |
| Free of selective reporting? | Unclear risk | There is no power statement or evidence of a priori analysis plan. |
Robin 1997.
| Methods | Prospective, double‐masked, placebo‐controlled one year study. | |
| Participants | 300 patients with POAG, PACG, pigmentary and exfoliation glaucoma. | |
| Interventions | Trabeculectomy with placebo, MMC 0.2 mg/ml for 2 or 4 minutes, or MMC 0.4 mg/ml for 2 minutes. | |
| Outcomes | 88% < 18 mmHg. 88% IOP < 18 mmHg. 89% IOP fall > 25% 93% off all medication. 80% met all 3. | |
| Notes | 79 participants lost to 12 month follow‐up. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | No information provided on sequence generation but verbal communication with the statistician involved confirmed allocation sequence was generated by computer generated block randomisation and delivered to the operating theatre in sequential sealed opaque envelopes. |
| Allocation concealment? | Low risk | "We randomised patients into 4 treatment groups in a prospective double masked fashion. The study medication was given to the surgeon by the operating room technician after the conjunctival dissection. Each eye received two consecutive sponges ...". |
| Blinding? All outcomes | Low risk | As well as the statement above, the following is stated: "only one physician (R.K.), masked to the treatment group, performed all of the measurements including the evaluation of cataracts. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | There is very little information on losses to follow‐up save "The mean length of follow‐up was similar in each group". But 221 out of 302 randomised patients provided outcome data at 12 months ‐ 26% loss without any information about characteristics of the those lost to follow‐up. |
| Free of selective reporting? | Unclear risk | On discussing this trial with one of the authors, it transpires that this trial was terminated prematurely because of the rising incidence of cataract in the treatment arms. The original sample size was 600 though this can not be now accurately determined. There is no power statement but it is likely that the study was powered to detect differences in IOP between treatment groups. |
Shin 1995.
| Methods | Prospective, randomised study with 27 months follow‐up. | |
| Participants | 174 POAG patients undergoing glaucoma triple surgery. | |
| Interventions | Triple procedure with or without MMC 0.5 mg/ml for 1, 3 or 5 minutes. | |
| Outcomes | 100% IOP < 14.1 in control group. 100% IOP < 13.9 in MMC groups. | |
| Notes | Prognostic factors for filtration failure seen in control group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No information on sequence generation provided. |
| Allocation concealment? | Unclear risk | No information on allocation concealment provided. |
| Blinding? All outcomes | High risk | The surgeon was clearly aware of the treatment status of patients since the only variable in the surgeries was the time of application of the MMC sponge or none. No mention is made of masked outcome assessment to treatment status. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Losses to follow‐up occurred but were not commented upon. In the MMC for 1 minute group, only 13 out of 21 patients completed the 12 month follow‐up. In the MMC for 5 minute group, 12 out out of 15 completed 12 months. Follow‐up was complete for the control group and the 3 minute group at 12 months. |
| Free of selective reporting? | Unclear risk | There is no power statement and definitions of success and qualified success are described as arbitrary. |
Shin 1998.
| Methods | Prospective, randomised study with 2 years follow‐up. Published as part of larger non‐randomised case control study. | |
| Participants | 49 eyes of 49 patients who had previous filtration surgery undergoing cataract extraction and repeat trabeculectomy. 21 of these were randomised to MMC and 28 to no antimetabolite. These were labelled the Secondary Glaucoma Triple Procedure group (SGTP). Another group of patients "exactly" matched for age, race, gender, cup:disc ratio and systemic diseases (Diabetes Mellitus and Systemic Hypertension) was recruited for comparison in a case‐control design to undergo primary trabeculectomy combined with cataract extraction and were allocated to MMC also on the basis of matching and allocation of the SGTP group. These were termed the Primary Glaucoma Triple Procedure group (PGTP). It is not clear whether these patients might have been participants in other reported trials such as Shin 1995 or Shin 1996. | |
| Interventions | Triple procedure with and without MMC 0.5 mg/ml for 1 minute in the SGTP and PGTP group to determine the relative effectiveness of MMC in each group. | |
| Outcomes | Survival for triple procedure with MMC significantly higher than triple procedure alone on Kaplan‐Meier plot over 30/12. | |
| Notes | Failure rates extracted from Kaplan‐Meier survival plots. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No mention is made of the randomisation sequence generation. |
| Allocation concealment? | Unclear risk | No mention is made of the allocation method or attempt at concealment. |
| Blinding? All outcomes | Unclear risk | No mention is made of masking of any patient group of observers of outcome. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Some loss to follow‐up occurred ‐ the lowest end of the range of follow‐up is 11 months. One might assume that most participants completed 12 month follow‐up. |
| Free of selective reporting? | Unclear risk | No mention of sample size calculation made and numerous outcomes are reported. There is no separate prior publication of the trial protocol. |
Szymanski 1997.
| Methods | Prospective, randomised study with 18 month follow‐up. The primary aim of the trial was to study the effect of MMC on corneal endothelial permeability. | |
| Participants | 29 patients with POAG under 50 years of age. | |
| Interventions | Trabeculectomy with placebo or MMC 0.2 mg/ml or 0.5 mg/ml. | |
| Outcomes | MMC eyes had IOP < 15. Non‐MMC eyes had IOP < 15 on medication. | |
| Notes | Exact information on outcomes at 12 months was not provided but follow‐up exceeded 12 months in all participants. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | "Twenty‐nine patients were divided into three groups. The choice of operative procedure and assignment to groups was made randomly." |
| Allocation concealment? | Unclear risk | No information on allocation method provided. |
| Blinding? All outcomes | Unclear risk | No statement on masking of any participants or observers noted. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Complete follow‐up at 12 months occurred but detailed reporting of control at this time point not given. |
| Free of selective reporting? | Unclear risk | No sample size calculation or prior publication of the protocol. |
Turacli 1996.
| Methods | Prospective, randomised study with 30 month follow‐up. | |
| Participants | 58 patients with POAG, closed‐angle and secondary glaucoma. | |
| Interventions | Standard trabeculectomy with or without MMC. | |
| Outcomes | 90% of MMC eyes had > 25% IOP fall. 72.4% of Control group had > 25% IOP fall. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No statement on sequence generation. |
| Allocation concealment? | Unclear risk | No statement on allocation concealment. |
| Blinding? All outcomes | Unclear risk | No mention of masking of either observers of outcome or participants. |
| Incomplete outcome data addressed? All outcomes | Unclear risk | The mean follow‐up for three groups in the trial only were reported ‐ the means for the three arms were similar but the ranges differed. It is not stated at which time point the outcome measures for success were obtained. |
| Free of selective reporting? | Unclear risk | There is no power statement or any a priori statement of primary outcomes or adverse events. |
Wu 1996.
| Methods | Prospective, randomised study with 10 months follow‐up. | |
| Participants | 40 eyes of 30 patients with uveitic, aphakic, neovascular, traumatic and infantile glaucoma. | |
| Interventions | Standard trabeculectomy with or without MMC 0.4 mg/ml for 5 minutes. | |
| Outcomes | MMC group had success rate of 90.5%. Control group had success rate of 26.3%. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | No information on sequence generation. |
| Allocation concealment? | Unclear risk | No information on allocation concealment. |
| Blinding? All outcomes | High risk | It would appear no attempt was made to mask patients or observers of outcome. |
| Incomplete outcome data addressed? All outcomes | High risk | "The MMC group follow‐up varied from 6 to 18 months (mean 8.8 months); the control group follow‐up ranged from 6 to 28 month (mean 11.3 months)." From this statement it would appear that there maybe unequal follow up in control and intervention groups. Success rates are reported but the time point at which success was judged is not stated. One must assume that failure was not estimated at a fixed time point which may lead to bias if the because the control group was followed for a longer period. |
| Free of selective reporting? | Unclear risk | Few outcomes are presented but no sample size or power calculation nor indeed a prior statement on timing of measurement of primary endpoint is made. |
IOP: Intraocular pressure MMC: Mitomycin C mmHg: Millimetres of mercury CACG: Congenital angle‐closure glaucoma POAG: Primary open‐angle glaucoma PACG: Primary angle‐closure glaucoma
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Chen 2001 | High proportion of participants had angle closure glaucoma. Not clear from study whether patients had had previous inflammatory or surgical events e.g. episode of acute angle closure glaucoma or surgical iridectomy. Unclear what time point results are reported for. Mean follow‐up 28 months. No results reported for 1 year. |
| Shin 1996 | Unable to determine whether it included data from similar report the previous year (Shin 1995). Contact with lead author yielded no reply. |
Differences between protocol and review
A previous version of this review included searching the National Research Register. This resource has now been archived and the UK Clinical Trials Gateway is now being searched. For the updated review in Issue 2, 2010, risk of bias tables have been completed for all included studies as well as the completion of a summary of findings table.
Contributions of authors
Mark Wilkins screened the search results, graded selected trials, extracted data, wrote the review and updated the review until Issue 2, 2010. Andrea Indar screened the search results, graded selected trials and extracted data. Richard Wormald conceived the idea of the review, advised on data extraction, statistical analysis, writing of the review. Richard Wormald also updated the review for Issue 2, 2010.
Sources of support
Internal sources
Moorfields Eye Hospital NHS Trust, UK.
External sources
Medical Research Council, UK.
Declarations of interest
None known.
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Andreanos 1997 {published data only}
- Andreanos D, Georgopoulos GT, Vergados J, Papaconstantinou D, Liokis N, Theodossiadis P. Clinical evaluation of the effect of mitomycin‐C in re‐operation for primary open angle glaucoma. European Journal of Ophthalmology 1997;7(1):49‐54. [DOI] [PubMed] [Google Scholar]
Carlson 1997 {published data only}
- Carlson DW, Alward WL, Barad JP, Zimmerman MB, Carney BL. A randomized study of mitomycin augmentation in combined phacoemulsification and trabeculectomy. Ophthalmology 1997;104(4):719‐24. [DOI] [PubMed] [Google Scholar]
Cohen 1996 {published data only}
- Cohen JS, Greff LJ, Novack GD, Wind BE. A placebo‐controlled, double‐masked evaluation of mitomycin C in combined glaucoma and cataract procedures. Ophthalmology 1996;103(11):1934‐42. [DOI] [PubMed] [Google Scholar]
Costa 1996 {published data only}
- Costa VP, Comegno PE, Vasconcelos JP, Malta RF, Jose NK. Low‐dose mitomycin C trabeculectomy in patients with advanced glaucoma. Journal of Glaucoma 1996;5(3):193‐9. [PubMed] [Google Scholar]
Martini 1997 {published data only}
- Martini E, Laffi GL, Sprovieri C, Scorolli L. Low‐dosage mitomycin C as an adjunct to trabeculectomy. A prospective controlled study. European Journal of Ophthalmology 1997;7(1):40‐8. [DOI] [PubMed] [Google Scholar]
Robin 1997 {published data only}
- Robin AL, Ramakrishnan R, Krishnadas R, Smith SD, Katz JD, Selvaraj S, et al. A long‐term dose‐response study of mitomycin in glaucoma filtration surgery. Archives of Ophthalmology 1997;115(8):969‐74. [DOI] [PubMed] [Google Scholar]
Shin 1995 {published data only}
- Shin DH, Simone PA, Song MS, Reed SY, Juzych MS, Kim C, et al. Adjunctive subconjunctival mitomycin C in glaucoma triple procedure. Ophthalmology 1995;102(10):1550‐8. [DOI] [PubMed] [Google Scholar]
Shin 1998 {published data only}
- Shin DH, Kim YY, Sheth N, Ren J, Shah M, Kim C, et al. The role of adjunctive mitomycin C in secondary glaucoma triple procedure as compared to primary glaucoma triple procedure. Ophthalmology 1998;105(4):740‐5. [DOI] [PubMed] [Google Scholar]
Szymanski 1997 {published data only}
- Szymanski A, Gierek Lapinska A, Koziak M, Gierek Ciaciura S. A fluorophotometric study of corneal endothelium after trabeculectomy using different concentrations of Mitomycin‐C. International Ophthalmology 1996;20(1‐3):95‐9. [DOI] [PubMed] [Google Scholar]
Turacli 1996 {published data only}
- Turacli E, Gunduz K, Aktan G, Tamer C. A comparative clinical trial of mitomycin C and cyclosporin A in trabeculectomy. European Journal of Ophthalmology 1996;6(4):398‐401. [DOI] [PubMed] [Google Scholar]
Wu 1996 {published data only}
- Wu L, Yin J. The effect of mitomycin C on filtration surgery of glaucoma with poor prognosis. Chinese Journal of Ophthalmology 1996;32(1):32‐4. [PubMed] [Google Scholar]
References to studies excluded from this review
Chen 2001 {published data only}
- Chen X, Liu X, Wu D. Application of mitomycin c and 5‐fluorouracil in the glaucoma filtering surgery. Chinese Ophthalmic Research 2001;19(4):347‐9. [Google Scholar]
Shin 1996 {published data only}
- Shin DH, Hughes BA, Song MS, Kim C, Yang KJ, Shah MI, et al. Primary glaucoma triple procedure with or without adjunctive mitomycin. Prognostic factors for filtration failure. Ophthalmology 1996;103(11):1925‐33. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Reibaldi 2008 {published data only}
- Reibaldi A, Uva MG, Longo A. Nine‐year follow‐up of trabeculectomy with or without low‐dosage mitomycin‐c in primary open‐angle glaucoma. British Journal of Ophthalmology 2008;92(12):1666‐70. [DOI] [PubMed] [Google Scholar]
Additional references
AGIS 1998a
- AGIS. The Advanced Glaucoma Intervention Study (AGIS): 4. Comparison of treatment outcomes within race. Seven‐year results. Ophthalmology 1998;105(7):1146‐64. [DOI] [PubMed] [Google Scholar]
Broadway 1994
- Broadway D, Grierson I, Hitchings R. Racial differences in the results of glaucoma filtration surgery: are racial differences in the conjunctival cell profile important?. British Journal of Ophthalmology 1994;78(6):466‐75. [DOI] [PMC free article] [PubMed] [Google Scholar]
Burr 2004
- Burr J, Azuara‐Blanco A, Avenell A. Medical versus surgical interventions for open angle glaucoma. Cochrane Database of Systematic Reviews 2004, Issue 2. [DOI: 10.1002/14651858.CD004399.pub2] [DOI] [PubMed] [Google Scholar]
Chen 1990
- Chen CW, Huang HT, Bair JS, Lee CC. Trabeculectomy with simultaneous topical application of mitomycin‐C in refractory glaucoma. Journal of Ocular Pharmacology 1990;6(3):175‐82. [DOI] [PubMed] [Google Scholar]
Chen 1997
- Chen P, Yamamoto T, Sawada A, Parrish R, Kitazawa Y. Use of antifibrosis agents and glaucoma drainage devices in the American and Japanese Glaucoma Societies. Journal of Glaucoma 1997;6(3):192‐6. [PubMed] [Google Scholar]
CNTG 1998a
- Collaborative Normal‐Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal‐tension glaucoma. American Journal of Ophthalmology 1998;126(4):498‐505. [DOI] [PubMed] [Google Scholar]
CNTG 1998b
- Collaborative Normal‐Tension Glaucoma Study Group. Comparison of glaucomatous progression between untreated patients with normal‐tension glaucoma and patients with therapeutically reduced pressures. American Journal of Ophthalmology 1998;126(4):487‐97. [DOI] [PubMed] [Google Scholar]
Costa 1993
- Costa VP, Wilson RP, Moster MR, Schmidt CM, Gandham S. Hypotony maculopathy following the use of topical mitomycin C in glaucoma filtration surgery. Ophthalmic Surgery 1993;24(6):389‐94. [PubMed] [Google Scholar]
EMGT 2002
- Heijl A, Leske MC, Bengtsson B, Hyman L, Bengtsson B, Hussein M, et al. Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Archives of Ophthalmology 2002;120(10):1268‐79. [DOI] [PubMed] [Google Scholar]
Glanville 2006
- Glanville JM, Lefebvre C, Miles JN, Camosso‐Stefinovic J. How to identify randomized controlled trials in MEDLINE: ten years on. Journal of the Medical Library Association 2006;94(2):130‐6. [PMC free article] [PubMed] [Google Scholar]
Greenfield 1996
- Greenfield DS, Suner IJ, Miller MP, Kangas TA, Palmberg PF, Flynn HW Jr. Endophthalmitis after filtering surgery with mitomycin. Archives of Ophthalmology 1996;114(8):943‐9. [DOI] [PubMed] [Google Scholar]
Higginbotham 1996
- Higginbotham EJ, Stevens RK, Musch DC, Karp KO, Lichter PR, Bergstrom TJ, et al. Bleb‐related endophthalmitis after trabeculectomy with mitomycin C. Ophthalmology 1996;103(4):650‐6. [DOI] [PubMed] [Google Scholar]
Khaw 1992a
- Khaw PT, Sherwood MB, Doyle JW, Smith MF, Grierson I, McGorray S, et al. Intraoperative and post operative treatment with 5‐fluorouracil and mitomycin‐c: long term effects in vivo on subconjunctival and scleral fibroblasts. International Ophthalmology 1992;16(4‐5):381‐5. [DOI] [PubMed] [Google Scholar]
Khaw 1992b
- Khaw PT, Sherwood MB, MacKay SL, Rossi MJ, Schultz G. Five‐minute treatments with fluorouracil, floxuridine, and mitomycin have long‐term effects on human Tenon's capsule fibroblasts. Archives of Ophthalmology 1992;110(8):1150‐4. [DOI] [PubMed] [Google Scholar]
Lavin 1990
- Lavin MJ, Wormald RPL, Migdal CS, Hitchings RA. The influence of prior therapy on the success of trabeculectomy. Archives of Ophthalmology 1990;108(11):1543‐8. [DOI] [PubMed] [Google Scholar]
Maier 2005
- Maier PC, Funk J, Schwarzer G, Antes G, Falck‐Ytter YT. Treatment of ocular hypertension and open angle glaucoma: meta‐analysis of randomised controlled trials. BMJ 2005;331(7509):134. [DOI] [PMC free article] [PubMed] [Google Scholar]
Membrey 2000
- Membrey WL, Poinoosawmy DP, Bunce C, Hitchings RA. Glaucoma surgery with or without adjunctive antiproliferatives in normal tension glaucoma: 1 intraocular pressure control and complications. British Journal of Ophthalmology 2000;84(6):586‐90. [DOI] [PMC free article] [PubMed] [Google Scholar]
OHTS 2002
- Kass MA, Heuer DK, Higginbotham EJ, Johnson CA, Keltner JL, Miller JP, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open‐angle glaucoma. Archives of Ophthalmology 2002;120(6):701‐13. [DOI] [PubMed] [Google Scholar]
Parrish 1992
- Parrish RK 2d. Who should receive antimetabolites after filtering surgery. Archives of Ophthalmology 1992;110(8):1069‐71. [DOI] [PubMed] [Google Scholar]
Rossetti 1993
- Rossetti L, Marchetti I, Orzalesi N, Scorpiglione N, Torri V, Liberati A. Randomized clinical trials on medical treatment of glaucoma. Are they appropriate to guide clinical practice?. Archives of Ophthalmology 1993;111(1):96‐103. [DOI] [PubMed] [Google Scholar]
Shields 1993
- Shields MB. Another reevaluation of combined cataract and glaucoma surgery. American Journal of Ophthalmology 1993;115(6):806‐11. [DOI] [PubMed] [Google Scholar]
Skuta 1987
- Skuta GL, Parrish RK 2nd. Wound healing in glaucoma filtering surgery. Survey of Ophthalmology 1987;32(3):149‐70. [DOI] [PubMed] [Google Scholar]
Suner 1997
- Suner IJ, Greenfield DS, Miller MP, Nicolela MT, Palmberg PF. Hypotony maculopathy after filtering surgery with mitomycin C. Incidence and treatment. Ophthalmology 1997;104(2):207‐14. [DOI] [PubMed] [Google Scholar]
Wilkins 2000
- Wilkins MR, Occleston NL, Kotecha A, Waters L, Khaw PT. Sponge delivery variables and tissue levels of 5‐fluorouracil. British Journal of Ophthalmology 2000;84(1):92‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wormald 2001
- Wormald R, Wilkins MR, Bunce C. Postoperative 5‐Fluorouracil for glaucoma surgery. Cochrane Database of Systematic Reviews 2001, Issue 3. [DOI: 10.1002/14651858.CD001132] [DOI] [PubMed] [Google Scholar]
Zacharia 1993
- Zacharia PT, Deppermann SR, Schuman JS. Ocular hypotony after trabeculectomy with mitomycin C. American Journal of Ophthalmology 1993;116(3):314‐26. [DOI] [PubMed] [Google Scholar]


