We report the genome sequences and the identification of genetic variations in eight clinical samples of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Samples were collected from nasopharyngeal swabs of symptomatic and asymptomatic individuals from five care homes for elderly and infirm persons in Israel. The sequences obtained are valuable, as they carry a newly reported nonsynonymous substitution located within the nucleoprotein open reading frame.
ABSTRACT
We report the genome sequences and the identification of genetic variations in eight clinical samples of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Samples were collected from nasopharyngeal swabs of symptomatic and asymptomatic individuals from five care homes for elderly and infirm persons in Israel. The sequences obtained are valuable, as they carry a newly reported nonsynonymous substitution located within the nucleoprotein open reading frame.
ANNOUNCEMENT
Shortly after a severe acute respiratory syndrome emerged in Wuhan, China, in December 2019 (1, 2), a new Betacoronavirus strain of the Coronaviridae family named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was identified as the etiological agent of a disease that was later termed coronavirus disease 19 (COVID-19) (2, 3). In this report, we describe the sequencing of eight SARS-CoV-2 samples obtained from specimens from five care homes for elderly and infirm persons in Israel. This study is in line with the ethical statement of the associate director general of the Israeli Ministry of Health. The individuals were initially identified as positive for COVID-19 by reverse transcriptase quantitative PCR (RT-PCR) and exhibited low cycle threshold (CT) values ranging from 12.8 to 16.8, implying a high viral load. Partial clinical information indicated that at least 2 of the 8 samples (i.e., EPI_ISL_594157 and EPI_ISL_594158) originated from asymptomatic individuals.
Samples were collected directly from swabs, and RNA was extracted with a QIAamp viral RNA minikit (Qiagen) according to the manufacturer’s protocol, using 60 µl of AVE buffer for elution. A SMARTer stranded total RNA-Seq pico input mammalian v2 kit (TaKaRa) was used for library construction prior to sequencing on a MiSeq instrument (Illumina). Whole-genome, paired-end sequencing was conducted in a duplex or triplex format with a read length of 150 nucleotides.
FastQC (https://www.bioinformatics.babraham.ac.uk/projects/fastqc) with default settings was used for quality control of the data. Trimming and removal of low-quality reads were performed using Trim Galore! v0.6.3 (http://www.bioinformatics.babraham.ac.uk/projects/trim_galore/) with default settings. Bowtie 2 (4) with default parameters was used for filtering of the results and for mapping the filtered reads against the reference Wuhan strain (GenBank accession number NC_045512). Reads mapped to SARS-CoV-2 were used as input data for the SPAdes assembler v3.13.0 (5) or the DNAStar software (SeqMan NGen v17.0; DNAStar, Madison, WI), resulting in a single contig for each sample. The genomic features of the samples are summarized in Table 1. Variant calling was performed using the SAMtools software package (6) with default parameters; a variant quality score cutoff of 100 was applied for all samples. A phylogenetic analysis generated using Nextstrain (7), rooted relative to the early samples from Wuhan, revealed that two of the eight samples (i.e., EPI_ISL_594155 and EPI_ISL_594156) belong to clade 20B, while the rest belong to clade 20C.
TABLE 1.
Genome features of eight SARS-CoV-2 clinical samples
| Sample | Genbank accession no. | Total no. of reads | No. of mapped reads | Avg coverage (×) | Assembly length (bp) | Overall G+C content (%) |
|---|---|---|---|---|---|---|
| NH-MA | MW227568 | 1,151,461 | 6,406 | 37 | 29,894 | 37.94 |
| NH-GD3 | MW201578 | 2,299,402 | 20,459 | 128 | 29,895 | 37.94 |
| NH-NM | MW193889 | 1,759,712 | 51,125 | 214 | 29,899 | 37.94 |
| NH-GD2 | MW201577 | 1,245,093 | 28,057 | 143 | 29,870 | 37.94 |
| NH-GD1 | MW237708 | 1,099,546 | 14,081 | 62 | 29,895 | 37.94 |
| NH-AS | MW201576 | 3,106,246 | 36,205 | 245 | 29,927 | 37.92 |
| NH-M2 | MW194121 | 6,485,364 | 59,944 | 297 | 29,930 | 37.91 |
| NH-M1 | MW228070 | 3,982,658 | 16,558 | 113 | 29,942 | 37.99 |
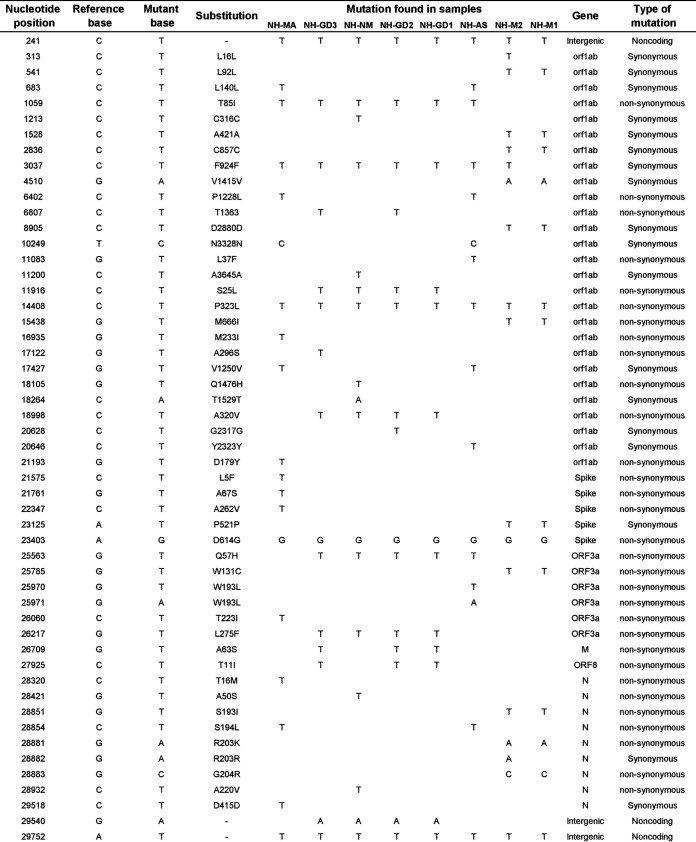
The variant calling process revealed a total of 52 unique single-nucleotide polymorphism (SNP) replacements. A total of 31 substitutions were nonsynonymous, 4 of which mapped to the Spike coding region; 18 substitutions were of the synonymous type, and the remaining 3 substitutions occurred in noncoding regions (Fig. 1). The eight samples share one common mutation in an intergenic region (position 241, C to T) and two common mutations in coding regions (positions 23403, A to G, and 14408, C to T), resulting in the well-documented D614G substitution and the P323L replacement, respectively (Fig. 1). Apart from the abundant D614G replacement, six other nonsynonymous abundant replacements found in this study (i.e., T85I, L37F, S25L, P323L, A320V, and Q57H; Fig. 1) were previously reported as a result of hot spot mutations (8–10).
FIG 1.
Mutations detected in the SARS-CoV-2 sequences generated in this study. The reference base was retrieved from the reference Wuhan strain (GenBank accession number NC_045512). Samples are named according to the isolate feature in the corresponding GenBank record (see Table 1).
While most of the nonsynonymous replacements were previously reported (11), the A50S substitution (located in the nucleocapsid protein) identified in the EPI_ISL_594161 sample, was not documented before (GISAID [12, 13], as of November 2020).
Although several papers documented a list of viral factors that are correlated with COVID-19 severity (9, 14–16), there is still more to it than meets the eye. Thus, mapping and identification of new mutations may contribute to a better understanding of the viral factors related to clinical outcomes of the disease.
Data availability.
The genome sequences have been deposited at the GISAID EpiCoV coronavirus SARS-CoV-2 platform database under the identifiers EPI_ISL_594155, EPI_ISL_594156, EPI_ISL_594157, EPI_ISL_594158, EPI_ISL_594159, EPI_ISL_594160, EPI_ISL_594161, and EPI_ISL_594162 and in the NCBI GenBank database under the accession numbers MW228070, MW194121, MW201576, MW227568, MW237708, MW201577, MW193889, and MW201578. The raw reads have been submitted to the NCBI Sequence Read Archive under the study reference number PRJNA672811.
ACKNOWLEDGMENT
We thank Emanuelle Mamroud for fruitful discussions and support throughout the project.
REFERENCES
- 1.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W, China Novel Coronavirus Investigation, Research Team . 2020. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. 2020. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Langmead B, Salzberg SL. 2012. Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM, Nikolenko SI, Pham S, Prjibelski AD, Pyshkin AV, Sirotkin AV, Vyahhi N, Tesler G, Alekseyev MA, Pevzner PA. 2012. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R, Genome Project Data Processing Subgroup . 2009. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25:2078–2079. doi: 10.1093/bioinformatics/btp352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hadfield J, Megill C, Bell SM, Huddleston J, Potter B, Callender C, Sagulenko P, Bedford T, Neher RA. 2018. Nextstrain: real-time tracking of pathogen evolution. Bioinformatics 34:4121–4123. doi: 10.1093/bioinformatics/bty407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Alouane T, Laamarti M, Essabbar A, Hakmi M, Bouricha EM, Chemao-Elfihri MW, Kartti S, Boumajdi N, Bendani H, Laamrti R, Ghrifi F, Allam L, Aanniz T, Ouadghiri M, El Hafidi N, El Jaoudi R, Benrahma H, Elattar J, Mentag R, Sbabou L, Nejjari C, Amzazi S, Belyamani L, Ibrahimi A. 2020. Genomic diversity and hotspot mutations in 30,983 SARS-CoV-2 genomes: moving toward a universal vaccine for the “confined virus”? bioRxiv https://www.biorxiv.org/content/10.1101/2020.06.20.163188v1. [DOI] [PMC free article] [PubMed]
- 9.Laha S, Chakraborty J, Das S, Manna SK, Biswas S, Chatterjee R. 2020. Characterizations of SARS-CoV-2 mutational profile, spike protein stability and viral transmission. Infect Genet Evol 85:104445. doi: 10.1016/j.meegid.2020.104445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Patro LPP, Sathyaseelan C, Uttamrao PP, Rathinavelan T. 2020. Global variation in the SARS-CoV-2 proteome reveals the mutational hotspots in the drug and vaccine candidates. bioRxiv https://www.biorxiv.org/content/10.1101/2020.07.31.230987v3.
- 11.Singer J, Gifford R, Cotten M, Robertson D. 2020. CoV-GLUE: a Web application for tracking SARS-CoV-2 genomic variation. Preprints doi: 10.20944/preprints202006.0225.v1. [DOI] [Google Scholar]
- 12.Elbe S, Buckland-Merrett G. 2017. Data, disease and diplomacy: GISAID's innovative contribution to global health. Glob Chall 1:33–46. doi: 10.1002/gch2.1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shu Y, McCauley J. 2017. GISAID: global initiative on sharing all influenza data - from vision to reality. Euro Surveill 22:30494. https://www.eurosurveillance.org/content/10.2807/1560-7917.ES.2017.22.13.30494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aiewsakun P, Wongtrakoongate P, Thawornwattana Y, Hongeng S, Thitithanyanont A. 2020. SARS-CoV-2 genetic variations associated with COVID-19 severity. medRxiv https://www.medrxiv.org/content/10.1101/2020.05.27.20114546v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Toyoshima Y, Nemoto K, Matsumoto S, Nakamura Y, Kiyotani K. 2020. SARS-CoV-2 genomic variations associated with mortality rate of COVID-19. J Hum Genet 65:1075–1082. doi: 10.1038/s10038-020-0808-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang X, Tan Y, Ling Y, Lu G, Liu F, Yi Z, Jia X, Wu M, Shi B, Xu S, Chen J, Wang W, Chen B, Jiang L, Yu S, Lu J, Wang J, Xu M, Yuan Z, Zhang Q, Zhang X, Zhao G, Wang S, Chen S, Lu H. 2020. Viral and host factors related to the clinical outcome of COVID-19. Nature 583:437–440. doi: 10.1038/s41586-020-2355-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The genome sequences have been deposited at the GISAID EpiCoV coronavirus SARS-CoV-2 platform database under the identifiers EPI_ISL_594155, EPI_ISL_594156, EPI_ISL_594157, EPI_ISL_594158, EPI_ISL_594159, EPI_ISL_594160, EPI_ISL_594161, and EPI_ISL_594162 and in the NCBI GenBank database under the accession numbers MW228070, MW194121, MW201576, MW227568, MW237708, MW201577, MW193889, and MW201578. The raw reads have been submitted to the NCBI Sequence Read Archive under the study reference number PRJNA672811.