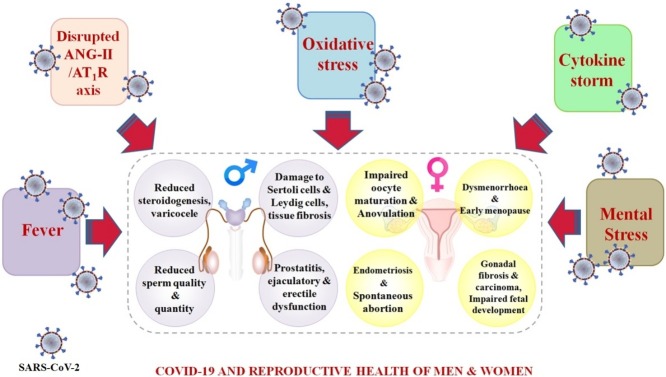
Graphical abstract
Keywords: COVID-19, Reproductive health, Renin Angiotensin System, Cytokine storm, Oxidative stress
Abstract
COVID-19 is the ongoing health emergency affecting individuals of all ages around the globe. Initially, the infection was reported to affect pulmonary structures. However, recent studies have delineated the impacts of COVID-19 on the reproductive system of both men and women. Hence, the present review aims to shed light on the distribution of SARS-CoV-2 entry factors in various reproductive organs. In addition, impacts of COVID-19 mediators like disrupted renin angiotensin system, oxidative stress, cytokine storm, fever, and the mental stress on reproductive physiology have also been discussed. For the present study, various keywords were used to search literature on PubMed, ScienceDirect, and Google Scholar databases. Articles were screened for relevancy and were studied in detail for qualitative synthesis of the review. Through our literature review, we found a multitude of effects of COVID-19 mediators on reproductive systems. Studies reported expression of receptors like ACE-2, TMPRSS2, and CD147 in the testes, epididymis, prostrate, seminal vesicles, and ovarian follicles. These proteins are known to serve as major SARS-CoV-2 entry factors. The expression of lysosomal cathepsins (CTSB/CTSL) and/ neuropilin-1 (NRP-1) are also evident in the testes, epididymis, seminal vesicles, fallopian tube, cervix, and endometrium. The binding of viral spike protein with ACE-2 was found to alter the renin-angiotensin cascade, which could invite additional infertility problems. Furthermore, COVID-19 mediated cytokine storm, oxidative stress, and elevated body temperature could be detrimental to gametogenesis, steroidogenesis, and reproductive cycles in patients. Finally, social isolation, confinement, and job insecurities have fueled mental stress and frustration that might promote glucocorticoid-mediated subnormal sperm quality in men and higher risk of miscarriage in women. Hence, the influence of COVID-19 on the alteration of reproductive health and fertility is quite apparent.
1. Introduction
Reproduction is a fundamental process that permits organisms to pass on their genetic imprint to the individuals of the next generation. Inability to bear a child is a major setback on the social, mental, and physical wellbeing of both men and women. Unfortunately, infertility has become a common health problem affecting around 8–12 % of the global population. Infertility rates differ greatly in various regions of the world, while the males could be responsible for up to 50 % of overall infertility cases [1]. Notably, numerous biotic and abiotic factors are known to have an adverse influence on the fertility of humans. Exposure to toxicants, malnutrition, psychological stress, and viral infections are just some of the known factors which impact reproductive health through various mechanisms [2,3].
At present, the world is suffering from a global health emergency in the form of Coronavirus disease-19 (COVID-19), caused by the SARS-CoV-2 virus. In December 2019, the epicenter of the disease was identified as the Hubei Province of Wuhan City, China, after which, as of August 26, 2021, it has spread to 213 nations and territories, resulting in 4,459,381 deaths worldwide (https://covid19.who.int/). These infections trigger strong inflammatory responses to confer acute pneumonitis, bronchitis, dyspnea, and respiratory failure in patients [4]. Severe lung injury characterized by “Ground Glass Opacities” is prevalent in infected patients.
Beside respiratory troubles, other ailments are also evident in the majority of COVID-19 cases [5]. The SARS-CoV-2 virus targets cardiac, gastro-intestinal, hepatic, cutaneous, and renal organs to elicit organopathy and uses angiotensin converting enzyme 2 (ACE-2), transmembrane serine protease 2 (TMPRSS2), and other entry factors to colonize host cells. Hence, organs expressing these entry factors are at greater risk of COVID-19 mediated pathology and organ dysfunction. Studies have claimed the expression of these entry factors in gonads and other accessory reproductive organs [6,7]. Therefore, it is possible that this virus might be able to invade reproductive organs and disrupt the reproductive health of infected individuals.
Epidemiological reports demonstrating the impacts of SARS-CoV-2 infection on the reproductive health of patients have been depicted in Table 1 . A variety of effects have been reported. A recent study has reported the occurrence of bilateral orchitis in a 37-year-old man who had suffered from COVID-19 [8]; inflammation associated with orchiepididymitis has been reported in a pediatric patient of confirmed COVID-19 infection [9]; and impaired sperm quality has been reported from patients with moderate infection [10]. It is further suggested that enhanced immune response in testicular cells might adversely impact the process of spermatogenesis [11]. Furthermore, females of child bearing age, when infected with COVID-19, exhibited decrease in menstrual volume and a prolongation of their menstrual cycle.
Table 1.
Reported cases demonstrating impacts of COVID-19 on reproductive health of patients.
| No. of cases studied | Gender | Impacts on reproductive health | Reference |
|---|---|---|---|
| 01 | Male | Bilateral orchitis (scrotal erythema, testicular pain) | [8] |
| 10 | Male | Decreased sperm quality | [10] |
| 01 | Male | Orchiepididymitis with lymphocytopenia and elevated C-reactive protein | [9] |
| 01 | Male | Ischemia-related priapism | [152] |
| 01 | Male | Testicular pain | [153] |
| 06 | Male | Interstitial edema, congestion, red blood cell exudation in testes, and epididymides; thinning of seminiferous tubules; oligozoospermia | [11] |
| 05 | Male | Numerous degenerated germ cells; altered transcriptome | [154] |
| 11 | Male | Sertoli cells with swelling, vacuolation and cytoplasmic rarefaction; reduced number of Leydig cells | [155] |
| 50 | Female | Decreased menstrual volume; cycle prolongation. | [12] |
| 04 | Female | Hypoxemia‐related fetal distress during pregnancy. | [156] |
| 05 | Female | Maternal morbidity and preterm birth | [157] |
| 27 | Female | Placental injury characterized by maternal vascular malperfusion (higher retroplacental hematomas; accelerated villous maturation; distal villous hyperplasia; atherosis, fibrinoid necrosis, mural hypertrophy of membrane arterioles; vessel ectasia and persistence of intramural endovascular trophoblast) | [158] |
The aftermath of all these events might be deleterious for fertility in future [12]. Incidences of spontaneous miscarriage and preterm delivery have been reported in the past among coronavirus infected pregnant women [13]. Systematic review and meta-analysis studies have indicated the enhanced risk of maternal death during pregnancy in COVID-19 patients [14], and vertical transmission of the virus particle from infected mother to their new born has also been documented [15].
It is also worth noting that the COVID-19 pandemic has delayed medical procedures of assisted reproductive technology (ART) throughout the world. ART is a set of medical procedures that are widely used to treat infertility. A Research highlighted the link of pandemic to anxiety and depression in 100 Turkish women whose urgent ART treatment was postponed [16]. Both mental health and depression are also considered as risk factors for infertility. Higher depression and anxiety levels are associated with lower fertility rates in communities. For example, in one study, COVID 19 infected pregnant women had greater levels of IL6 and TNFγ than the normal individuals [17]. Excessive production of IL6 and TNFγ have also been associated with adverse pregnancy outcomes such as preterm delivery, preterm/premature rupture of the membrane, and pregnancy loss [18]. ACE gene polymorphism is also associated with infertility problems in women. Lumbers et al. [19] have discussed the role of dysregulated renin-angiotensin pathway in the development of preeclampsia, which is a pregnancy associated complication marked by high blood pressure as well as indications of liver and kidney damage.
Thus, the present review provides insight on the distribution of various entry factors/receptors responsible for SARS-CoV-2 invasion in male and female reproductive organs, and further discusses the effects of COVID-19 mediators such as disturbed renin-angiotensin system, cytokine storm, oxidative stress, fever, and mental stress on reproductive health.
2. Search criteria
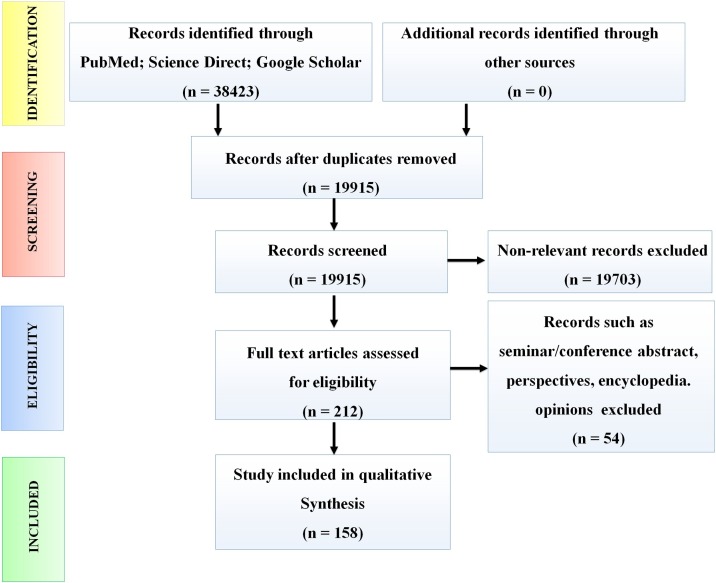
A systematic review of the literature has been conducted in accordance with the guideline of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [20]. Search terms specified in supplemental Table 1 were used to retrieve publications from PubMed, ScienceDirect, and Google Scholar scientific databases. Literatures published up to the 30th of April 2021, were manually screened by co-authors for the removal of duplicate entries. Relevant articles were screened independently by co-authors PR and AG based on title, abstract, and keywords. Discrepancies were resolved after joint article review and discussion. Seminar/conference abstracts, opinions, perspectives, and encyclopedias were excluded. After preliminary screening, the full texts of relevant articles were assessed for qualitative synthesis of the review. A schematic presentation of the screening process has been depicted in Fig. 1 .
Fig. 1.
Schematic representation of workflow demonstrating the literature search strategy.
3. Reproductive organs expressing entry factors for SARS-CoV-2
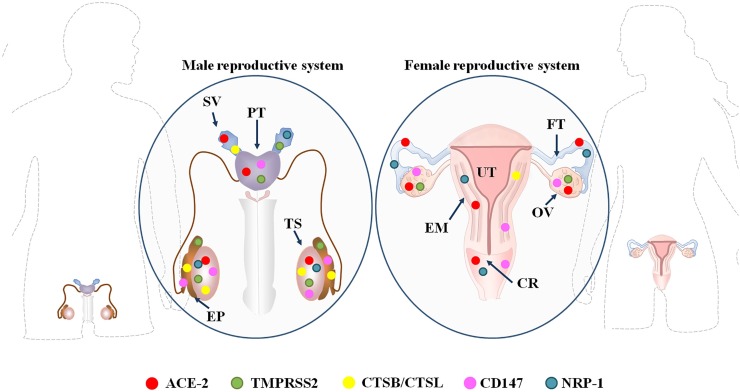
To facilitate the entry of SARS-CoV-2 into the host cell, the viral spike glycoprotein attaches to the ACE-2 receptor on the cell membrane. The conformational change of the spike protein is then ensured by the host cell’s proteolytic enzymes mainly TMPRSS2. Other receptors such as neuropilin-1 (NRP-1), cluster of differentiation 147 (CD147), furin, lysosomal cathepsin L (CTSL), and cathepsin B (CTSB) also facilitate viral invasion (Fig. 2 ).
Fig. 2.
Distribution of SARS-CoV-2 entry factors in male and female reproductive organs.
Abbreviations: SV: Seminal vesicle; PT: Prostate gland; EP: Epididymis; TS: Testis; OV: Ovary; EM: Endometrium; FT: Fallopian tube; UT: Uterus; CR: Cervix; ACE-2: Angiotensin Converting Enzyme 2; TMPRSS2: Type II Membrane Serine Proteases; CSTB/CSTL: Lysosomal Cathepsin B / Cathepsin L; CD147: Cluster of Differentiation 147; NRP-1: Neuropilin-1.
The entry factors are found throughout reproductive tissues. One such molecule angiotensin converting enzyme 2 (ACE-2), is a membrane-bound Zinc metalloproteinase with a catalytic subunit that is directed towards the extracellular space in various body tissues. It converts angiotensin II to angiotensin 1–7. SARS-CoV-2 spike protein can interact with ACE-2 and trigger the loss of ACE-2 function by endocytosis. The down-regulation of ACE-2 may then further aggravate the pathological impacts of COVID-19 in various organs. ACE-2 is also expressed in male reproductive organs and differentiating spermatogenic cells. Sertoli cells, Leydig cells, and testicular germ cells have all been observed to express ACE-2 on their cell surface [21,22]. Single cell RNA-seq analysis has detected the presence of ACE-2 in Sertoli cells and differentiating spermatogonial cells, mainly in leptotene spermatocyte type-1, leptotene spermatocyte type-3, zygotene spermatocyte, diplotene spermatocyte, and spermatid stage-1 [6].
Other entry factors have also been found to be expressed throughout male reproductive tissues. CD147 receptors are expressed in all testicular cells, with maximum expression in differentiating gametocytes [6]. Endosomal cysteine proteases such as CTSB and CTSL are abundant in Leydig cells, Sertoli cells, seminal vesicles, the epididymis, and germ cells and participate in the priming of S protein [23,24]. Expression of TMPRSS2, an important serine protease needed for viral entry into the host, is also evident in primordial germ cells and the acrosomal region of developing spermatogonia [6,7]. Finally as per the human protein atlas, neuropilin-1 (NRP-1) is expressed in Leydig cells and seminal vesicles.
Female reproductive organs such as uterus, vagina, ovary, placenta etc. also express ACE-2 as well as other entry factor [25]. Syncytiotrophoblast, cytotrophoblast, endothelium, and the vascular smooth muscles of primary and secondary placental villi have been observed to contain ACE-2. The endometrium also expresses ACE-2, as well as TMPRSS2/4, CTSL, and CTSB. Presence of ACE-2 is evident in arterial and venous endothelium, as well as the smooth muscles of the umbilical cord. The expression of these entry factors reaches its maximum during the secretory phase of the menstrual cycle [26]. Expression of ACE-2 is also at its peak during the early gestational period. During late gestation, ACE-2 transcripts are synthesized in the labyrinth placenta, amniotic, and yolk sac epithelium.
As per the human protein atlas, low to medium-level expression of NRP-1 is evident in the vagina, the fallopian tube, endometrium, and cervix. Human ovaries also express ACE-2 in their primordial, primary, secondary, and antral follicles, the stroma, and the corpora lutea [27]. Notably, ACE-2 expression is upregulated during the course of follicle development and further increases after gonadotropin stimulation [28]. Also of note, expression of CD147 has been detected in granulosa cells (follicles of all stages), the ovarian surface epithelium, and granulose cells, as well as the theca lutein of corpora lutea [29]. Finally, one of the key proteases in granulosa cells of ovarian follicles is the cathepsin L that mediates follicular rupture during ovulation [30].
4. COVID-19 and mediators of infertility
4.1. COVID-19 and the disruption of the renin angiotensin system
Renin Angiotensin System (RAS) is precisely tuned machinery that controls fluid homeostasis within the body. Renin, ACE-1, ACE-2, and ACE-3 are the fundamental components of the RAS. In recent years, the influence of the RAS on different reproductive phenomena has been investigated. In males, it has been found that the RAS regulates tubular contractibility, spermatogenesis, sperm maturation, capacitation, acrosomal exocytosis, and fertilization capacity while, in females, it has been found that the RAS maintains ovarian blood pressure and several autocrine/paracrine signaling pathways that control fertility [31].
Renin is an aspartyl proteinase that catalyzes the conversion of angiotensinogen to angiotensin I (ANG-I). ANG-I is then further cleaved into angiotensin II (ANG-II) by ACE-1, after which ANG-II is hydrolyzed to angiotensin 1–7 (Ang 1–7) by a mono-carboxypeptidase, (i.e. ACE-2). ANG-II is also produced by catalytic activity of non-ACE-2 related enzymes like serine-protease Chymase [32]. ANG-II is a biologically active octapeptide that works through the ANG-II type-I receptor (AT1R) to promote vasoconstriction, inflammation, and oxidative stress [33]. Higher ANG-II contents in seminal plasma are evident in sub-fertile subjects. In addition, ANG-II negatively affects sperm count and sperm morphology [34] by promoting vascular hypertension. ANG-II mediated hyperproduction of proinflammatory cytokine IL6 in testicular peritubular cells is implicated in male infertility. Intriguingly, ANG-II also stimulates metalloproteinase-2 (MMP2)/MMP14 through PI3K/AKT and MAPK signaling cascades that facilitate cellular proliferation and angiogenesis. Furthermore, excessive ANG-II inhibits adenylate cyclase in Leydig cells to subvert testosterone production [35].
In females, the ANG-II/AT1R-axis is responsible for endometrial, breast, and cervical carcinoma [36,37]. ANG-II promotes ovarian cancer metastasis by modulating the activity of stearoyl-CoA desaturase-1 and endoplasmic reticulum stress in patients [38]. Moreover, women with upregulated ANG-II/AT1R signaling are vulnerable to pre-eclampsia during pregnancy [39]. ANG-II also evokes endometrial fibrosis by boosting cell proliferation and expression of α-smooth muscle actin (α-SMA), transforming growth factor beta 1 (TGF-β1), Insulin-like growth factor 1 (IGF-1), type I collagen, and fibronectin [40].
The ANG-II/AT1R-axis is also related to and regulated by the ACE-2/Ang 1–7 dependent molecular nexus [41]. Ang 1–7 works through the Mas receptor to regulate reproductive physiology and fertility. ACE-2/Ang 1–7/Mas-mediated signaling pathway controls steroidogenesis, epididymal contractibility, and sperm cell motility in males. For example, in one study, sub-fertile men with low ACE-2, Ang 1–7, and Mas levels had impaired spermatogenesis. Meanwhile, in females, ovarian steroidogenesis, follicular development, oocyte maturation, ovulation, and atresia are regulated by ACE-2 mediated signaling cascades [42]. Furthermore, the ACE-2/Ang 1–7/Mas axis also promotes the meiotic resumption of oocytes, which is precisely controlled by luteinizing hormone [43].
Unfortunately, coronavirus infection can down-regulate ACE-2 receptors, leading to reduced conversion of ANG-II to Ang 1–7 and excessive accumulation of ANG-II (Box 1 ). The Coronavirus modulates ACE-2 expression via the endocytosis of the receptor-ligand complex or the activation of the TNF-α converting enzyme (TACE)-mediated shedding of the receptor ectodomain [44]. This paradoxically benefits virions with respect to their replication and packaging inside the host cell [45]. Hence, coronavirus mediated disruption of the ACE-2/Ang 1–7/Mas-axis could impair the normal reproductive physiology and fertility of both men and women.
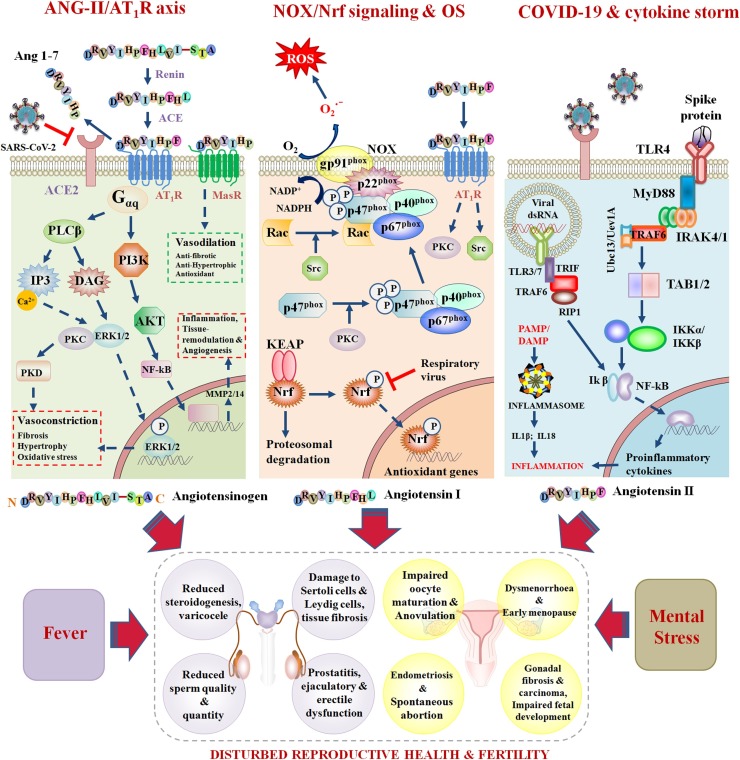
Box 1. ANG-II/AT1R axis.
ANG-II performs most of its physiological functions through angiotensin type-I receptors (AT1Rs) coupled with intracellular-Gαq/11-Gβγ complexes. The binding of ANG-II with AT1R activates downstream signaling molecules namely phospholipase-C (PLC), phospholipase-A2 (PLA2), and phospholipase-D (PLD). PLC generates inositol-1,4,5-triphosphate (IP3) and diacylglycerol (DAG) within the subcellular moiety. IP3 triggers Ca2+-efflux from endoplasmic reticulum to promote vasoconstriction while DAG activates protein kinase C (PKC) involved in ERK pathway. PLD hydrolyzes phosphatidylcholine to phosphatidic acid (PA). PA is converted into DAG for sustained availability of PKC. PLA2 generates arachidonic acid (AA) that catalyzes NAD(P)H oxidation. ANG-II also promotes the synthesis of leukotrienes responsible for prolonged vasoconstriction and inflammatory diseases. The pleiotrophic effect of ANG-II is mediated by NAD(P)H oxidase (NOX) induced ROS production and up-regulation of NF-kB. NF-kB blocks the expression of E-cadherin and fuels cancer cell metastasis. ANG-II/AT1Rs interaction activates c-Src and Calcium-dependent Pyk2 involved in EGFR phosphorylation and Shc/Grb2 complex formation. This scaffold promotes Ras-MEK1/2- ERK1/2 cascade for the transcriptional activation of several genes involved in tissue remodeling, hypertrophy, inflammation, and organ damage. ACE-2 down-regulates the aforementioned signaling pathways and reverses the pathophysiology imposed by ANG-II/AT1R-axis. Moreover, ACE-2/Ang(1–7)/Mas-axis inhibits Stim1/Orai1-mediated SOCE (store-operated calcium entry) to repress PAK1/NF-kB/Snail signaling implicated in breast cancer metastasis.
Alt-text: Box 1
4.2. Cytokine storms and reproductive dysfunction
The severity of COVID-19 in patients is associated with exaggerated immune responses like “cytokine storm” and intense inflammation. Following COVID-19 infection, the defense machinery of the body becomes active and immune cells infiltrate the lungs, leading to the hyperactivation of macrophages and monocytes. This is then followed by the excessive production of proinflammatory cytokines, namely interleukin-6 (IL6), interleukin-1β (IL1β), tumor necrosis factor α (TNFα), and chemokines like monocyte chemoattractant protein-1 (MCP-1/CCL2). Although these cytokine storms are an effort of the immune system to protect the host from the infection, the excessive release of proinflammatory cytokines could injure multiple organs throughout the body. Typically, cytokines are essential for maintenance of structural and functional integrity of gonads, nevertheless, upon profound immune stimulation, proinflammatory cytokines can modulate the cellular microenvironment in a way that impairs reproductive physiology (Box 2 ).
Box 2. SARS-CoV-2 mediated Cytokine storm.
Infection with SARS-CoV-2 causes a cytokine storm that can lead to organ damage. The S1 subunit of the viral spike protein interacts with TLR7/8 to recruit downstream adaptors (MyD88, IRAK 4/1, and TRAF6). TRAF6 with dimeric ubiquitin conjugating enzyme complex (Ubc13-Uev1A) activates TAK1/TAB1/2 and IKKα/IKKβ. IKKα/IKKβ phosphorylates Ikβ to promote the nuclear translocation of NF-kβ involved in the transcription of various proinflammatory cytokines responsible for the cytokine storm. Endosomal dsRNAs through TLR3/4 are also implicated in the direct activation of NF-kβ through the recruitment of adaptor proteins namely TRIF, TRAF6, and RIP1. PAMPs and damage associated molecular patterns (DAMPs) also interact with NLRP3 to assemble procaspases-1 and ASC (apoptosis-associated speck-like protein containing CARD) into a multimeric component i.e. inflammasome. Activated caspase-1 cleaves proIL1β and proIL18 to generate the active version of proinflammatory cytokines. In addition, caspase-1 cleaves Gasdermin-D at Asp276 to separate N- and C-terminus domains. Active N-termini create pores in lipid bilayer to elicit pyroptosis and subsequent organ damage. Intense inflammation invites epididymal and testicular immunopathologies in men whereas impairs ovulation and endometrial receptivity in females.
Alt-text: Box 2
Pathogen associated molecular patterns (PAMPs) activate pathogen recognition receptors on macrophages, mast cells, Sertoli cells, and developing spermatozoa to generate a plethora of cytokines that might be associated with reduced sperm count and impaired sperm motility [46,47]. Dyspermia such as oligozoospermia, asthenozoospermia, and teratozoospermia have been diagnosed in cases concerning higher levels of proinflammatory cytokines namely IL1β, IL2, IL6, IL8, IL10, IL18, and TNFα in infertile men compared to fertile subjects [48,49]. There are many ways by which proinflammatory cytokines impact infertility. IL6, through transcription factor Zfp637, interferes with sperm cell differentiation to impede spermatogenesis [50]. Reproductive complications driven by testicular inflammation can develop, such as epididymitis, which affects almost 400 per 10,000 males per year [51]. Inflammatory signals can thereafter recruit mast cells and lead to mast cell proliferation, and cause extravasation into the testicular environment. Higher numbers of mast cells, Th17 cells, and their products like IL17a further contribute to infertility [52,53].
Further effects of cytokines on male reproductive systems have also been documented. In one survey including 400 males, prostatitis-like symptoms were positively associated with serum IL8 concentration [54]. In other studies, TNFα was shown to trigger the activation of NF-kB and DAX-1 to inhibit transactivation of orphan nuclear receptors involved in steroidogenesis [55,56]. Toll-like receptors (TLRs) are activated by SARS-CoV-2 proteins. Activated TLR2 can lower testosterone synthesis in Leydig cells. IL17/IL18 have also displayed a negative correlation with spermatozoa concentration and motility in infertile subjects [57,58]. Moreover, cytokine induced recruitment of mast cells and macrophages may result in testicular fibrosis, reduced spermatogonial cells, and concomitant thickening of lamina propria [59]. Another concern is the blood-testis barrier, a dynamic structure where proinflammatory cytokines can deregulate Cldn11 in Sertoli cells to damage gap junctions [60]. Finally, elevated contents of IL6 and TNFR2 have also been associated with seminomas [61].
The female reproductive system is also sensitive to proinflammatory cytokines within the body. Normal titers of TNFα, IL1β, and IL6 are central to the ovarian cycle and follicular development [62]. Elevated levels of TH1 cytokines, viz. TNFα, IFNγ, and IL10 are associated with infertility and recurrent spontaneous abortion [63,64]. Dysregulation of cytokine titers fuels thrombotic/inflammatory reactions at uteroplacental blood vessels, leading to fetal resorption [65]. TGFβ, a proinflammatory mediator up-regulating MMP-2 and MMP-9 [66], has been implicated in endometrial extracellular matrix turnover during implantation. Any alteration in this turnover can promote idiopathic recurrent spontaneous miscarriage (IRSM) [67]. In one study, higher concentrations of proinflammatory cytokines such as IL1β, TNFα, and IFNγ were linked to IRSM [68].
Further examples of cytokines’ effects on female reproductive health also exist. Increased levels of IL2 have been detected in women with recurrent spontaneous abortion within 24 h, compared to women who have no abortion history [69]. Intrauterine growth restriction has been detected with elevated levels of IL10 in amniotic fluid of women at mid-trimester [70]. Aberrant expression of IL10 in decidual T-lymphocytes is evident in women with recurrent miscarriage [71]. Dysregulation of inflammatory mediators are known to promote endometriosis-associated reproductive failure [72]. Cyclooxygenase 2 (COX2) production is enhanced by cytokines, and inhibitors of COX2 are implicated in treatment of dysmenorrhoea and heavy menstrual blood loss [73]. This indicates the possible contribution of COX2 to female infertility. Moreover, COX2 has links with endometrial carcinomas [74]. Finally, synthesis of Prostaglandin E2 (PGE2) is triggered by proinflammatory cytokines [75]. Dysregulated PGE2 is responsible for menorrhagic endometrium and excessive menstrual bleeding in women [76,77].
Therefore, the above suggested that cytokine storms caused by COVID-19 could be detrimental to the reproductive organs and reproductive physiology of both men and women.
4.3. Oxidative stress and disturbed reproductive health
Oxidative stress (OS) is a physiological condition in which the redox equilibrium is disrupted as a result of excessive reactive oxygen species (ROS) production at the subcellular level. SARS-CoV-2 mediated disruption of the RAS can lead to accumulation of ANG-II in blood plasma, and then promote ROS production via the NADPH oxidase (NOX)-protein kinase C (PKC) dependent pathway (Box 3 ) [78]. Hypoxia is a common symptom of COVID-19 and it acts as a stimulant for ROS generation [79]. Meta-analysis of 1210 COVID-19 cases has revealed reduced hemoglobin levels (5.9 g/L-7.1 g/L) [80] which might be related to anemic hypoxia and cellular ROS production in patients.
Box 3. NOX and Nrf cascades linked to Oxidative Stress.
The interaction between ANG-II and AT1R triggers PKC and Src kinase mediated signaling pathways. PKC activates NOX, which is involved in ROS production. NOX is a multimeric protein composed of two membrane subunits (gp91-phox, p22-phox), three cytosolic subunits (p47-phox, p67-phox, p40-phox), and one G-protein Rac. Glycoprotein (gp)91-phox is NOX-2 specific and its homologs (DUOX1 and DUOX2) are detected in NOX-1, 3, 4, and 5. PKC phosphorylates p47-phox to facilitate its binding with the p67-phox/p40-phox complex. A trimeric complex is thus formed that translocates to the cell membrane and interacts with p22-phox. Src kinase activates Rac which independently move to the membrane and forms the active NOX complex. NOX then oxidizes molecular Oxygen into ROS. Nrf2 is a transcription factor, playing crucial role in redox-homeostasis. The activity of Nrf2 is regulated by Keap1 (Kelch-like ECH-associated protein-1) via cullin-3 dependent proteosomal degradation. Several kinases (PKC, PI3K/Akt, GSK-3β, JNK) phosphorylate Nrf2 and block its degradation. Phosphorylated Nrf2 then translocates into the nucleus and forms a heterodimer with Maf (masculoaponeurotic-fibrosarcoma) to bind antioxidant response elements (AREs) on nuclear DNA. AREs are implicated in the synthesis of endogenous antioxidants that alleviate OS. Respiratory viral infections can inhibit Nrf2 signaling and therefore promote ROS production and subsequent OS in host.
Alt-text: Box 3
The nuclear factor erythroid 2–related factor 2 (Nrf2) is a transcription factor that maintains redox balance in a living system. Respiratory viruses can abrogate the Nrf pathway to impose OS [81]. Moreover, proinflammatory cytokines can increase ROS production [82], putting redox-equilibrium in jeopardy. Excessive production of ROS and disturbed antioxidant defense machinery have been found to be evident during coronavirus infection [83]. Some researchers have argued that coronavirus induced lung injury is triggered by OS and NF-kB signaling in patients [84].
Excessive ROS and subsequent OS in male gonads can impair genesis, motility, and fertilization capacity of mature sperms. Surprisingly, a Canadian research group has discovered substantial ROS generation in 40 % of the semen collected from infertile men [85]. Related to this, the cell membranes of spermatozoa are rich in polyunsaturated fatty acids (PUFAs) that are extremely vulnerable to ROS-induced lipid peroxidation (LPO). LPO promotes the disruption of membrane fluidity and the rapid loss of ATP from spermatozoa, leading to axonal damage, midpiece defects, and reduced sperm motility and viability [86]. Excessive ROS is also positively correlated with varicocele in men [87], which is a leading cause of male infertility.
Other effects of ROS and OS on male gonads have also been observed. For example genetic materials of spermatozoa are highly vulnerable to ROS-induced base modification and degradation [88]. Single or double stranded DNA breaks in gametes can lower the reproductive potential of fertile men. Moreover, infertile men with greater ROS levels tend to have more apoptotic spermatozoa than control individuals [89]. Massive ROS also impairs sperm motility, morphology, and ability to penetrate oocytes [90], and Sertoli cells injured by ROS lead to decreased sperm count and motility [91]. Additionally, Steroidogenesis is sensitive to redox dyshomeostasis and increases in the activities of COX2 and MAPK [92], and chemicals inducing ROS production impede testosterone synthesis in Leydig cells [93,94]. Finally, OS promotes long-term changes in epididymis and maturing spermatozoa resulting in declined sperm quality [95].
Optimum levels of ROS are implicated in the proper maintenance of female reproductive health. However, recent studies have proposed that excessive ROS production might be detrimental to reproductive events in females. Erroneous redox balance can promote endometriosis, coupled with an inflammatory response in the peritoneal cavity [96]. OS also appears to play a crucial role in the malignant transformation and progression of ovarian cancer [97]. Moreover, OS is involved in the aetiology of errant embryo implantation and development [98]. Burgeoning evidences also advocate the role of OS in pathogenesis of polycystic ovary syndrome (PCOS), indicating the potential contribution of ROS to infertility [99]. Furthermore, in women with PCOS, an OS-induced proinflammatory state might result in ovulatory failure and hyperandrogenism [100].
Taking all of the above into consideration, disturbed redox states in men and women could be detrimental to their reproductive physiology and fertility.
4.4. Elevated body temperature and altered reproductive physiology
The majority of COVID-19 cases report moderate to severe fever, characterized by an oral temperature of 100.4 ° F and above [101]. Higher body temperatures during COVID-19 infection might be a significant harbinger of poor outcomes [102]. Body temperature in a healthy person ranges from 97.7 to 99.5 ° F [103] and is crucial to maintain the normal physiology of the body. Disruption of thermal homeostasis impairs the proper activity of organs, including those involved in reproductive biology.
The epithelia of the scrotal testes are highly sensitive to changes in temperature of even a few degrees [104]. Slight changes in testicular temperature can cause incidences of morphological abnormality in developing spermatozoa and spermatids. Additional body temperature increases also curtail the sperm-storage capacity of the cauda epididymidis [104]. Furthermore, high fevers (>39 °C) for 3 or more days have been associated with declined sperm concentration and semen quality in patients [105]. Fever of up to 41.2 °C for 10 days was sufficient enough to impair spermatogenesis in typhoid patients, leading to an azoospermic state [106]. Similar impacts on sperm count, coupled with an increase in small-head sperm, were reported as a response of an acute fever of 38°-39 °C during a throat infection [107]. Sub-normal sperm count, sperm motility, and sperm DNA integrity have been documented in semen samples collected from fertile volunteers presenting a 2-day fever of 39–40 °C [108]. Excess temperature could also invite germ cell apoptosis [109], and Leydig cells are vulnerable to morphological deterioration following exposure to high temperature [110]. Finally, hypothermic states promote the death of Sertoli cells, which might hamper sperm quality and fertility [111].
Slight changes in core body temperature (0.3 °C to 0.7 °C) are obvious during the ovulatory menstrual cycle. Body temperature is increased in the post-ovulatory luteal phase when progesterone is high, compared to the preovulatory follicular phase but further hike in core temperature can modulate reproductive events. For example, mouse oocytes incubated at 38.5–39 °C temperature showed decrease in rate of blastocyst formation and had low potential for implantation [112]. In vitro studies on swine ovarian follicles have revealed that high temperatures can reduce ovarian follicle responsiveness to hormonal stimulators and ovarian hormone production [113]. Another in vitro study found that when the follicular wall is exposed to high temperature, follicular steroidogenesis, androstenedione, and estradiol levels are reduced [114]. In mammals, high temperature triggers irregularities in cycle length, ovulation rate, morphological abnormalities in ova, and reduction in the size of the fetus [115]. Temperatures above optimum levels also impede secretion of reproductive hormones such as luteinizing hormone (LH), follicle stimulating hormone (FSH), and progesterone in bovine species [116]. Heat stress also has the potential to reduce levels of gonadotropin receptors, aromatase activity of granulosa cells, and follicular fluid concentrations of estradiol in rat follicles [117]. Moreover, high temperatures can threaten development of embryos in mice [118].
Hence, the above discussions suggests that elevated body temperatures during COVID-19 infection might play a role in the reproductive pathology of patients.
4.5. Mental stress and reproductive impairment
People in the modern world where they are able to travel rapidly and communicate with each other have been forced to live in confinement, with social isolation, and restrictions. Job insecurity and financial problems, or bereavement due to loss of near ones and global lock-down, has caused severe emotional upheaval in large number of people across the planet. The unprecedented situation caused by the COVID-19 outbreak has fueled depression, anxiety, hysteria, mood swings, irritability, insomnia, anger, and emotional exhaustion [119,120]. These psychological symptoms are symptoms of severe mental stress that could be detrimental to the reproductive health of humans.
Mental stress activates several components of hypothalamus-pituitary-adrenal (HPA) axis to stimulate the production of stress hormones like corticotropin-releasing hormone (CRH), adrenocorticotropic hormone (ACTH), and glucocorticoids (GCs) [121]. Glucocorticoids have a direct inhibitory influence on gonadotropin releasing hormone (GnRH) secreted from the arcuate nucleus of the hypothalamus. GnRH stimulates gonadotrophs of the anterior pituitary gland to release FSH and LH, which are involved in the maintenance of testicular function. Thus, inhibition of GnRH function can impede optimum testicular physiology. GCs induced by mental stress further interact with GC receptors in Leydig cells to suppress enzymes implicated in testosterone biosynthesis [122].
Psychological stress also seems to have further effects on male reproductive health outcomes. For example, a stressed environment fuels cortisol-mediated apoptosis in germ cells and Leydig cells. Low testosterone levels are also responsible for defective blood-testis barriers and subsequent germ cell loss [123,124]. Furthermore, in one study, men experiencing stress at work and a recent bereavement have displayed reduced semen quantity, sperm count, and sperm motility [125]. In another study, fertile men reporting more than two recent stressful events had low sperm concentration and motility [126]. Semen samples collected from healthy medical students just before final examination (well regarded as a stressful period) have shown reduced sperm concentration and motility compared to stress-free subjects [127]. Emotional stress in patients due to enrollment in an in vitro fertilization (IVF) program has also been found to negatively affect semen quality parameters. Finally, chronic or acute psychological stress has implications on erectile and ejaculatory dysfunction [128].
In females, psychological stress induces a clinical condition called hypothalamic amenorrhea, characterized by disrupted menstruation [129]. Intriguingly, excess stress perception can be a risk factor for intense premenstrual pain, ovarian troubles, preterm delivery as well as early onset of perimenopause [130,131]. Stress induced GCs also impede the secretion of major reproductive hormones such as LH, ovarian estrogen, and progesterone harming uterus and fetus. Elevated levels of the stress hormone cortisol, during pregnancy, has also been associated with spontaneous abortion [132]. In women with Cushing syndrome (hypercortisolism), chances for miscarriage or preterm delivery are also high [133]. Finally, an inadequate level of progesterone during pregnancy is positively associated with fetal loss [134], and mental stress might be an etiological factor for unsuccessful IVF and live births [135].
5. The role of COVID-19 mediators on IVF and other ARTs
ARTs including IVF and intracytoplasmic sperm injection (ICSI) are currently being used to treat infertility. However, in the case of semen samples with high DNA damage, ARTs may not be so effective. Sometimes, compromised spermatozoa lead to high rates of miscarriage after ICSI [136]. Infertile men also fail to initiate pregnancy after ARTs, which might be due to defective spermatozoa. Defects in spermatozoa might also be caused by seminal OS [137]. Similarly, OS is responsible for the poor oocyte quality that reduces the rate of success in IVF [138]. Additionally, cytokine storm and inflammation can fuel several gynecological problems, particularly in women of reproductive age. Inflammation can affect ovulation and hormone production, and might also be associated with endometriosis, as inflammation alters receptivity of endometrium. Furthermore, it also plays a major role in tissue repair and remodeling.
A variety of relationships between external factors such as cytokines and ARTs have been observed. In one study, Harrity et al. [139] assessed intracellular cytokine ratios in patients with a history of implantation failure and/or pregnancy loss. They opined that higher intracellular cytokine levels might be responsible for ART implantation failure and miscarriage in the studied patients. In another study, elevated TNF-α/IL10 cytokine ratios were correlated with an increased risk of IVF failure [140]. Inagaki et al. also [141] analyzed intrauterine cytokine concentrations in women having recurrent failed embryo transfer. They observed that, in women with recurrent spontaneous abortion (n = 13), IL1β concentration was significantly higher in comparison to the control group, whereas concentrations of IFNγ and IL10 were significantly lower. Hence, cytokine concentrations might play a critical role in IVF patients with recurrent implantation failure.
Exposure to partner's seminal plasma with high levels of IL18 during the IVF/ICSI cycle also increases the risk of pregnancy failure in women [142]. Interestingly, stress-related over-expression of immune (IL18; IL1β) and hormonal factors in seminal plasma link male stress with embryo implantation failure [143]. Studies also indicate that stress-induced norepinephrine production could directly impact embryo development in the oviduct via adrenergic receptors, and further suggests that maternal stress can influence the embryo even in very early pregnancy [144]. Furthermore, it has been shown that pregnancies where elevated maternal cortisol (a commonly used stress marker) levels are recorded during the first three weeks following conception display a higher probability of spontaneous abortion. This could negatively affect IVF and other ARTs [145].
According to An et al. [146], anxiety is also negatively associated with live birth rates, and positively associated with serum norepinephrine and cortisol values in IVF patients. Zhou et al. [147] examined the amount of the enzyme alpha-amylase secreted in saliva (SAA) and correlated it with pregnancy outcomes in infertile couples undergoing in vitro fertilization and embryo transplantation (IVF-ET). High SAA levels in female or male partners individually, or in both, resulted in an increased risk of pregnancy failure compared to individuals with low SAA levels. The SAA levels were also found to be directly correlated with the level of follicle-stimulating hormone, and inversely proportional to the anti-Müllerian hormone level and endometrial thickness. Additionally, parameters portraying semen quality of male partners, such as density, survival rate, sperm rapid progressive motility and progressive motility were significantly lower in individuals with high SAA when compared with low SAA group. Thus high SAA level can be considered as an objective indicator of high stress that increases the risk of pregnancy failure in infertile couples undergoing IVF-ET.
Fever has also been reported to have negative effects on follicular development and ovarian estradiol production in some women undergoing controlled ovarian stimulation. Awwad et al. [148] demonstrated that fever-exposed IVF/ICSI cycles were associated with a significantly lower number of pre-ovulatory follicles, higher number of medium-size follicles, and reduced serum estradiol per follicle. These women, as a result need significantly longer durations of ovarian stimulation. Additionally, acute febrile fever can also harm sperm count, sperm motility, and sperm DNA integrity in men undergoing ART [149].
Finally, SARS-CoV-2 infection blocks ACE-2 receptors and increases the level of ANG-II. High levels of follicular ANG-II are associated with the development of severe ovarian hyperstimulation syndrome in patients undergoing IVF procedure [150]. In males, excessive ANG-II inhibits adenylate cyclase in Leydig cells to reduce testosterone production [35]. Notably, the RAS is also responsible for the maintenance of seminal plasma electrolytes, regulation of steroidogenesis, spermatogenesis and sperm functions [151]; however, disruption of RAS can alter these processes and affect IVF/ICSC cycles.
Thus, mediators of COVID-19 have significant potential to alter the outcome of IVF and other ARTs.
6. Concluding remarks
The COVID-19 pandemic is an ongoing biological disaster affecting the human population through multiple avenues. Reproductive organs are sensitive to pathogenic insult, and hence might be targeted by SARS-CoV-2 infection. SARS-CoV-2-induced ACE-2 shedding, cytokine storm, oxidative stress, and elevated body temperature are all potential threats to reproductive health and physiology (Fig. 3 ). Social isolation, confinement, and job insecurities due to the COVID-19 outbreak have also invited mild to severe mental stress to swamp the general population, which could peril the fertility and reproductive success of infected individuals. Due to the presence of entry factors for SARS-CoV-2 in various reproductive tissues, virions can invade reproductive organs and might disrupt the fertility efficiency of couples planning to conceive in the near future. Orchitis, low sperm quality, oligozoospermia, and priapism are even evident in some COVID-19 cases and miscarriage, preterm delivery, and prolonged cycle have been reported in some infected women. However, these outcomes might also be triggered by certain drugs and antibiotics used during the treatment of patients, or some of these conditions could be preexisting.
Fig. 3.
COVID-19 fuels several mediators of infertility.
ANG-II/AT1R axis. The sequential proteolytic cleavage of Angiotensinogen produces Angiotensin-II (ANG-II), which acts as ligand for Angiotensin type-1 receptor (AT1R). ACE-2 regulates the amount of ANG-II in the body by converting it into Ang 1–7. Ang 1–7 then counter-regulates ANG-II signaling using Mas-receptor. The binding of the SARS-CoV-2 spike protein with the ACE-2 receptor causes ectodomain shedding and ACE-2 down-regulation. This results in massive accumulation of ANG-II and profound activation of the ANG-II/AT1R axis. The ANG-II/AT1R axis promotes ERK1/2 and NF-kB signaling involved in vasoconstriction, fibrosis, hypertrophy, and oxidative stress (OS).
NOX/Nrf signaling and OS. Ang 1–7 triggers the activation of PKC and Src, which help in the assembly of NADPH Oxidase (NOX) involved in ROS production and OS. Nrf catalyzes the transcriptional activation of endogenous antioxidants to combat OS. SARS-CoV-2 reduces Ang 1–7 levels and blocks Nrf activation to promote redox dyshomeostasis, which has been linked to reproductive problems.
COVID-19 and cytokine storm. Spike protein/dsRNA of the virus interacts with TLRs to recruit downstream adaptors for the massive production of proinflammatory cytokines. Moreover, pathogen associated molecular patterns (PAMP) and damage associated molecular patterns (DAMP) can directly elicit the assembly of inflammasomes that are involved in the activation of proinflammatory cytokines.
Fever and mental stress are obvious outcomes of COVID-19. All of these mediators have the potential to damage the reproductive health of men and women.
Finally, it is important to note that the majority of investigations conducted so far in this field are case studies and therefore warrant further studies in more representative samples for the proper validation of results. The avoidance of infection is the best choice that can be taken to reduce COVID-19-mediated reproductive pathology. People should follow the proper guidelines issued by WHO and local governments to minimize the risk of COVID-19 infection and the subsequent impact of infection on the reproductive health of both men and women.
CRediT authorship contribution statement
Prem Rajak: Conceptualization, Writing - original draft, Writing - review & editing, Supervision. Sumedha Roy: Conceptualization, Writing - review & editing, Supervision. Moumita Dutta: Writing - review & editing. Sayanti Podder: Writing - review & editing. Saurabh Sarkar: Writing - review & editing. Abhratanu Ganguly: Writing - original draft. Moutushi Mandi: Writing - review & editing. Salma Khatun: Writing - review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The authors would like to thank Mr. Francisco Javier Miralles, an undergraduate student at the University of Washington, for his valuable contribution in the edits and modification of the manuscript. Mr. Miralles aspires to be a physician-scientist, in order to create the research and policy that will change how the American healthcare system operates and afford to all patients the right to heal from autoimmune and chronic conditions.
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.repbio.2021.100559.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- 1.Vander Borght M., Wyns C. Fertility and infertility: definition and epidemiology. Clin Biochem. 2018;62:2–10. doi: 10.1016/j.clinbiochem.2018.03.012. [DOI] [PubMed] [Google Scholar]
- 2.Ali R.I., Ibrahim M.A. Malathion induced testicular toxicity and oxidative damage in male mice: the protective effect of curcumin. Egypt J Forensic Sci. 2018;8:70. [Google Scholar]
- 3.Garolla A., Pizzol D., Bertoldo A., Menegazzo M., Barzon L., Foresta C. Sperm viral infection and male infertility: focus on HBV, HCV, HIV, HPV, HSV, HCMV, and AAV. J Reprod Immunol. 2013;100:20–29. doi: 10.1016/j.jri.2013.03.004. [DOI] [PubMed] [Google Scholar]
- 4.Greenland J.R., Michelow M.D., Wang L., London M.J. COVID-19 infection: implications for perioperative and critical care physicians. Anesthesiology. 2020;132:1346–1361. doi: 10.1097/ALN.0000000000003303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mokhtari T., Hassani F., Ghaffari N., Ebrahimi B., Yarahmadi A., Hassanzadeh G. COVID-19 and multiorgan failure: a narrative review on potential mechanisms. J Mol Histol. 2020;51:613–628. doi: 10.1007/s10735-020-09915-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liu X., Chen Y., Tang W., Zhang L., Chen W., Yan Z., et al. Single-cell transcriptome analysis of the novel coronavirus (SARS-CoV-2) associated gene ACE2 expression in normal and non-obstructive azoospermia (NOA) human male testes. Sci China Life Sci. 2020;63:1006–1015. doi: 10.1007/s11427-020-1705-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Larasati T., Noda T., Fujihara Y., Shimada K., Tobita T., Yu Z., et al. Tmprss12 is required for sperm motility and uterotubal junction migration in mice†. Biol Reprod. 2020;103:254–263. doi: 10.1093/biolre/ioaa060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bridwell Re, Merrill Dr, Griffith Sa, Wray J., Oliver Jj. A coronavirus disease 2019 (COVID-19) patient with bilateral orchitis. Am J Emerg Med. 2021;42(260) doi: 10.1016/j.ajem.2020.08.068. e3-260.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gagliardi L., Bertacca C., Centenari C., Merusi I., Parolo E., Ragazzo V., et al. Orchiepididymitis in a boy with COVID-19. Pediatr Infect Dis J. 2020;39:e200–2. doi: 10.1097/INF.0000000000002769. [DOI] [PubMed] [Google Scholar]
- 10.Holtmann N., Edimiris P., Andree M., Doehmen C., Baston-Buest D., Adams O., et al. Assessment of SARS-CoV-2 in human semen-a cohort study. Fertil Steril. 2020;114:233–238. doi: 10.1016/j.fertnstert.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li H., Xiao X., Zhang J., Zafar M.I., Wu C., Long Y., et al. Impaired spermatogenesis in COVID-19 patients. EClinicalMedicine. 2020;28 doi: 10.1016/j.eclinm.2020.100604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li K., Chen G., Hou H., Liao Q., Chen J., Bai H., et al. Analysis of sex hormones and menstruation in COVID-19 women of child-bearing age. Reprod Biomed Online. 2021;42:260–267. doi: 10.1016/j.rbmo.2020.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong S.F., Chow K.M., Leung T.N., Ng W.F., Ng T.K., Shek C.C., et al. Pregnancy and perinatal outcomes of women with severe acute respiratory syndrome. Am J Obstet Gynecol. 2004;191:292–297. doi: 10.1016/j.ajog.2003.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Matar R., Alrahmani L., Monzer N., Debiane L.G., Berbari E., Fares J., et al. Clinical presentation and outcomes of pregnant women with Coronavirus Disease 2019: a Systematic Review and Meta-analysis. Clin Infect Dis. 2021;72:521–533. doi: 10.1093/cid/ciaa828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dong L., Tian J., He S., Zhu C., Wang J., Liu C., et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. JAMA. 2020;323:1846–1848. doi: 10.1001/jama.2020.4621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tokgoz V.Y., Kaya Y., Tekin A.B. The level of anxiety in infertile women whose ART cycles are postponed due to the COVID-19 outbreak. J Psychosom Obstet Gynaecol. 2020:1–8. doi: 10.1080/0167482X.2020.1806819. [DOI] [PubMed] [Google Scholar]
- 17.Tanacan A., Yazihan N., Erol S.A., Anuk A.T., Yucel Yetiskin F.D., Biriken D., et al. The impact of COVID-19 infection on the cytokine profile of pregnant women: a prospective case-control study. Cytokine. 2021;140 doi: 10.1016/j.cyto.2021.155431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qiu X., Zhang L., Tong Y., Qu Y., Wang H., Mu D. Interleukin-6 for early diagnosis of neonatal sepsis with premature rupture of the membranes: a meta-analysis. Medicine (Baltimore) 2018;97 doi: 10.1097/MD.0000000000013146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lumbers E.R., Delforce S.J., Arthurs A.L., Pringle K.G. Causes and consequences of the dysregulated maternal renin-angiotensin system in preeclampsia. Front Endocrinol (Lausanne) 2019;10:563. doi: 10.3389/fendo.2019.00563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moher D., Liberati A., Tetzlaff J., PRISMA Group Altman D. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62:1006–1012. doi: 10.1016/j.jclinepi.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 21.Shen Q., Xiao X., Aierken A., Yue W., Wu X., Liao M., et al. The ACE2 expression in Sertoli cells and germ cells may cause male reproductive disorder after SARS-CoV-2 infection. J Cell Mol Med. 2020;24:9472–9477. doi: 10.1111/jcmm.15541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang Z., Xu X. scRNA-seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS-CoV-2 infection in spermatogonia, Leydig and Sertoli cells. Cells. 2020;9:e920. doi: 10.3390/cells9040920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gye M.C., Kim S.T. Expression of cathepsin L in human testis under diverse infertility conditions. Arch Androl. 2004;50:187–191. doi: 10.1080/01485010490425223. [DOI] [PubMed] [Google Scholar]
- 24.Chung S.S., Zhu L.J., Mo M.Y., Silvestrini B., Lee W.M., Cheng C.Y. Evidence for cross-talk between Sertoli and germ cells using selected cathepsins as markers. J Androl. 1998;19:686–703. [PubMed] [Google Scholar]
- 25.Jing Y., Run-Qian L., Hao-Ran W., Hao-Ran C., Ya-Bin L., Yang G., et al. Potential influence of COVID-19/ACE2 on the female reproductive system. Mol Hum Reprod. 2020;26:367–373. doi: 10.1093/molehr/gaaa030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Henarejos-Castillo I., Sebastian-Leon P., Devesa-Peiro A., Pellicer A., Diaz-Gimeno P. SARS-CoV-2 infection risk assessment in the endometrium: viral infection-related gene expression across the menstrual cycle. Fertil Steril. 2020;114:223–232. doi: 10.1016/j.fertnstert.2020.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Reis F.M., Bouissou D.R., Pereira V.M., Camargos A.F., dos Reis A.M., Santos R.A. Angiotensin-(1-7), its receptor Mas, and the angiotensin-converting enzyme type 2 are expressed in the human ovary. Fertil Steril. 2011;95:176–181. doi: 10.1016/j.fertnstert.2010.06.060. [DOI] [PubMed] [Google Scholar]
- 28.Pereira V.M., Reis F.M., Santos R.A., Cassali G.D., Santos S.H., Honorato-Sampaio K., et al. Gonadotropin stimulation increases the expression of angiotensin-(1--7) and MAS receptor in the rat ovary. Reprod Sci. 2009;16:1165–1174. doi: 10.1177/1933719109343309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Smedts A.M., Lele S.M., Modesitt S.C., Curry T.E. Expression of an extracellular matrix metalloproteinase inducer (basigin) in the human ovary and ovarian endometriosis. Fertil Steril. 2006;86:535–542. doi: 10.1016/j.fertnstert.2006.01.042. [DOI] [PubMed] [Google Scholar]
- 30.Robker R.L., Russell D.L., Espey L.L., Lydon J.P., O’Malley B.W., Richards J.S. Progesterone-regulated genes in the ovulation process: ADAMTS-1 and cathepsin L proteases. Proc Natl Acad Sci USA. 2000;97:4689–4694. doi: 10.1073/pnas.080073497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brannstrom M., Zackrisson U., Hagstrom H.G., Josefsson B., Hellberg P., Granberg S., et al. Preovulatory changes of blood flow in different regions of the human follicle. Fertil Steril. 1998;69:435–442. doi: 10.1016/s0015-0282(97)00544-x. [DOI] [PubMed] [Google Scholar]
- 32.Unger T., Li J. The role of the renin–angiotensin-aldosterone system in heart failure. J Renin Angiotensin Aldosterone Syst. 2004;5(Suppl. 1):S7–10. doi: 10.3317/jraas.2004.024. [DOI] [PubMed] [Google Scholar]
- 33.Bader M., Ganten D. Update on tissue renin-angiotensin systems. J Mol Med. 2008;86:615–621. doi: 10.1007/s00109-008-0336-0. [DOI] [PubMed] [Google Scholar]
- 34.Colli L.G., Belardin L.B., Echem C., Akamine E.H., Antoniassi M.P., Andretta R.R., et al. Systemic arterial hypertension leads to decreased semen quality and alterations in the testicular microcirculation in rats. Sci Rep. 2019;9:11047. doi: 10.1038/s41598-019-47157-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Khanum A., Dufau M.L. Angiotensin II receptors and inhibitory actions in Leydig cells. J Biol Chem. 1988;263:5070–5074. [PubMed] [Google Scholar]
- 36.Uemura H. Angiotensin II receptor blocker shows antiproliferative activity in prostate cancer cells: a possibility of tyrosine kinase inhibitor of growth factor. Mol Cancer Ther. 2003;2:1139–1147. [PubMed] [Google Scholar]
- 37.Piastowska-Ciesielska A.W., Płuciennik E., Wójcik-Krowiranda K., Bieńkiewicz A., Bednarek A., Ochędalski T. Analysis of the expression of angiotensin II type 1 receptor and VEGF in endometrial adenocarcinoma with different clinicopathological characteristics. Tumour Biol. 2012;33:767–774. doi: 10.1007/s13277-011-0292-0. [DOI] [PubMed] [Google Scholar]
- 38.Zhang Q., Yu S., Lam M.M.T., Poon T.C.W., Sun L., Jiao Y., et al. Angiotensin II promotes ovarian cancer spheroid formation and metastasis by upregulation of lipid desaturation and suppression of endoplasmic reticulum stress. J Exp Clin Cancer Res. 2019;38:116. doi: 10.1186/s13046-019-1127-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Leung P.S., Tsai S.J., Wallukat G., Leung T.N., Lau T.K. The upregulation of angiotensin II receptor AT(1) in human preeclamptic placenta. Mol Cell Endocrinol. 2001;2001(184):95–102. doi: 10.1016/s0303-7207(01)00637-2. [DOI] [PubMed] [Google Scholar]
- 40.Shan T., Shang W., Zhang L., Zhao C., Chen W., Zhang Y., et al. Effect of angiotensin-(1-7) and angiotensin II on the proliferation and activation of human endometrial stromal cells in vitro. Int J Clin Exp Pathol. 2015;8:8948–8957. [PMC free article] [PubMed] [Google Scholar]
- 41.Alenina N., Xu P., Rentzsch B., Patkin E.L., Bader M. Genetically altered animal models for Mas and angiotensin-(1-7) Exp Physiol. 2008;93:528–537. doi: 10.1113/expphysiol.2007.040345. [DOI] [PubMed] [Google Scholar]
- 42.Costa A.P., Fagundes-Moura C.R., Pereira V.M., Silva L.F., Vieira M.A., Santos R.A. Angiotensin-(1-7): a novel peptide in the ovary. Endocrinology. 2003;144:1942–1948. doi: 10.1210/en.2002-220787. [DOI] [PubMed] [Google Scholar]
- 43.Honorato-Sampaio K., Pereira V.M., Santos R.A., Reis A.M. Evidence that angiotensin-(1–7) is an intermediate of gonadotrophin-induced oocyte maturation in the rat preovulatory follicle. Exp Physiol. 2012;97:642–650. doi: 10.1113/expphysiol.2011.061960. [DOI] [PubMed] [Google Scholar]
- 44.Jia H.P., Look D.C., Tan P., Shi L., Hickey M., Gakhar L., et al. Ectodomain shedding of angiotensin converting enzyme 2 in human airway epithelia. Am J Physiol Lung Cell Mol Physiol. 2009;297:L84–96. doi: 10.1152/ajplung.00071.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Stoddart C.A., Geleziunas R., Ferrell S., Linquist-Stepps V., Moreno M.E., Bare C., et al. Human immunodeficiency virus type 1 Nef-mediated downregulation of CD4 correlates with Nef enhancement of viral pathogenesis. J Virol. 2003;77:2124–2133. doi: 10.1128/JVI.77.3.2124-2133.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fujita Y., Mihara T., Okazaki T., Shitanaka M., Kushino R., Ikeda C., et al. Toll-like receptors (TLR) 2 and 4 on human sperm recognize bacterial endotoxins and mediate apoptosis. Hum Reprod. 2011;26:2799–2806. doi: 10.1093/humrep/der234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Klein B., Haggeney T., Fietz D., Indumathy S., Loveland K.L., Hedger M., et al. Specific immune cell and cytokine characteristics of human testicular germ cell neoplasia. Hum Reprod. 2016;31:2192–2202. doi: 10.1093/humrep/dew211. [DOI] [PubMed] [Google Scholar]
- 48.Alsaimary I.E.A. Evaluation of serum levels of pro-inflammatory cytokines (interleukins 2, 6, 8) in fertile and infertile men. Donnish J Microbiol Biotechnol Res. 2014;1:23–34. [Google Scholar]
- 49.Havrylyuk A., Chopyak V., Boyko Y., Kril I., Kurpisz M. Cytokines in the blood and semen of infertile patients. Cent Eur J Immunol. 2015;3:337–344. doi: 10.5114/ceji.2015.54596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang H., Yin Y., Wang G., Liu Z., Liu L., Sun F. Interleukin-6 disrupts blood-testis barrier through inhibiting protein degradation or activating phosphorylated ERK in Sertoli cells. Sci Rep. 2014;4:4260. doi: 10.1038/srep04260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nicholson A., Rait G., Murray-Thomas T., Hughes G., Mercer C.H., Cassell J. Management of epididymo-orchitis in primary care: results from a large UK primary care database. Br J Gen Pract. 2010;60:e407–22. doi: 10.3399/bjgp10X532413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Meineke V., Frungieri M.B., Jessberger B., Vogt H., Mayerhofer A. Human testicular mast cells contain tryptase: increased mast cell number and altered distribution in the testes of infertile men. Fertil Steril. 2000;74:239–244. doi: 10.1016/s0015-0282(00)00626-9. [DOI] [PubMed] [Google Scholar]
- 53.Duan Y.G., Yu CF Novak N., Bieber T., Zhu C.H., Schuppe H.C., et al. Immunodeviation towards a Th17 immune response associated with testicular damage in azoospermic men. Int J Androl. 2011;34:e536–45. doi: 10.1111/j.1365-2605.2010.01137.x. [DOI] [PubMed] [Google Scholar]
- 54.Lotti F., Corona G., Mondaini N., Maseroli E., Rossi M., Filimberti E., et al. Seminal, clinical and colour-Doppler ultrasound correlations of prostatitis-like symptoms in males of infertile couples. Andrology. 2014;2:30–41. doi: 10.1111/j.2047-2927.2013.00156.x. [DOI] [PubMed] [Google Scholar]
- 55.Hong C.Y., Park J.H., Ahn R.S., Im S.Y., Choi H.S., Soh J., et al. Molecular mechanism of suppression of testicular steroidogenesis by proinflammatory cytokine tumor necrosis factor alpha. Mol Cell Biol. 2004;24:2593–2604. doi: 10.1128/MCB.24.7.2593-2604.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sadasivam M., Ramatchandirin B., Balakrishnan S., Prahalathan C. TNF-mediated suppression of Leydig cell steroidogenesis involves DAX-1. Inflamm Res. 2015;64:549–556. doi: 10.1007/s00011-015-0835-8. [DOI] [PubMed] [Google Scholar]
- 57.Qian L., Shi Q., Gu Y., Song J., Zhou M., Hua M. The relationship between IL-17 and male infertility: semen analysis. African J Microbiol Res. 2012;6:5672–5677. [Google Scholar]
- 58.Matalliotakis I.M., Cakmak H., Fragouli Y., Kourtis A., Arici A., Huszar G. Increased IL-18 levels in seminal plasma of infertile men with genital tract infections. Am J Reprod Immunol. 2006;55:428–433. doi: 10.1111/j.1600-0897.2006.00380.x. [DOI] [PubMed] [Google Scholar]
- 59.Schuppe H.C., Bergmann M. Springer; London: 2013. Inflammatory conditions of the testis; pp. 113–122. [Google Scholar]
- 60.Oh Y.S., Jo N.H., Park J.K., Gye M.C. Changes in inflammatory cytokines accompany deregulation of Claudin-11, resulting in inter-Sertoli tight junctions in varicocele rat testes. J Urol. 2016;196:1303–1312. doi: 10.1016/j.juro.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 61.Białas M., Fiszer D., Rozwadowska N., Kosicki W., Jedrzejczak P., Kurpisz M. The role of IL-6, IL-10, TNF-alpha and its receptors TNFR1 and TNFR2 in the local regulatory system of normal and impaired human spermatogenesis. Am J Reprod Immunol. 2009;62:51–59. doi: 10.1111/j.1600-0897.2009.00711.x. [DOI] [PubMed] [Google Scholar]
- 62.Vital Reyes V.S., Téllez Velasco S., Hinojosa Cruz J.C. Ortiz Romero Mde J, Chavarría Olarte ME, Reyes Fuentes a. Serum levels of IL-1beta, IL-6 and TNF-alpha in infertile patients with ovarian dysfunction. Ginecol Obstet Mex. 2005;73:604–610. [PubMed] [Google Scholar]
- 63.Reid J.G. The carriage of pro-inflammatory cytokine gene polymorphisms in recurrent pregnancy loss. Am J Reprod. Immunol. 2001;45:35–40. doi: 10.1111/j.8755-8920.2001.450106.x. [DOI] [PubMed] [Google Scholar]
- 64.Mahdi B.M. Role of some cytokines on reproduction. Middle East Fertil Soc J. 2011;16:220–223. [Google Scholar]
- 65.Clark D.A., Chaouat G., Arck P.C., Mittruecker H.W., Levy G.A. Cytokine-dependent abortion in CBA DBA/2 mice is mediated by the procoagulant fgl2 prothrombinase. J Immunol. 1998;15:545–559. [PubMed] [Google Scholar]
- 66.Kim E.S., Kim M.S., Moon A. TGF-beta-induced upregulation of MMP-2 and MMP-9 depends on p38 MAPK, but not ERK signaling in MCF10A human breast epithelial cells. Int J Oncol. 2004;25:1375–1382. [PubMed] [Google Scholar]
- 67.Jokimaa V., Oksjoki S., Kujari H., Vuorio E., Anttila L. Altered expression of genes involved in the production and degradation of endometrial extracellular matrix in patients with unexplained infertility and recurrent miscarriages. Mol Hum Reprod. 2002;8:1111–1116. doi: 10.1093/molehr/8.12.1111. [DOI] [PubMed] [Google Scholar]
- 68.Banerjee P., Jana S.K., Pasricha P., Ghosh S., Chakravarty B., Chaudhury K. Proinflammatory cytokines induced altered expression of cyclooxygenase-2 gene results in unreceptive endometrium in women with idiopathic recurrent spontaneous miscarriage. Fertil Steril. 2013;99 doi: 10.1016/j.fertnstert.2012.08.034. 179-87.e2. [DOI] [PubMed] [Google Scholar]
- 69.Hadinedoushan H., Mirahmadian M., Aflatounian A. Increased natural killer cell cytotoxicity and IL-2 production in recurrent spontaneous abortion. Am J Reprod Immunol. 2007;58:409–414. doi: 10.1111/j.1600-0897.2007.00524.x. [DOI] [PubMed] [Google Scholar]
- 70.Heyborne K.D., McGregor J.A., Henry G., Witkin S.S., Abrams J.S. Interleukin-10 in amniotic fluid at midtrimester: immune activation and suppression in relation to fetal growth. Am J Obstet Gynecol. 1994;171:55–59. doi: 10.1016/s0002-9378(94)70077-x. [DOI] [PubMed] [Google Scholar]
- 71.Piccinni M.P., Beloni L., Livi C., Maggi E., Scarselli G., Romagnani S. Defective production of both leukemia inhibitory factor and type 2 T-helper cytokines by decidual T cells in unexplained recurrent abortions. Nat Med. 1998;4:1020–1024. doi: 10.1038/2006. [DOI] [PubMed] [Google Scholar]
- 72.Gupta S., Goldberg J.M., Aziz N., Goldberg E., Krajcir N., Agarwal A. Pathogenic mechanisms in endometriosis-associated infertility. Fertil Steril. 2008;90:247–257. doi: 10.1016/j.fertnstert.2008.02.093. [DOI] [PubMed] [Google Scholar]
- 73.Daniels S.E., Talwalker S., Torri S., Snabes M.C., Recker D.P., Verburg K.M. Valdecoxib, a cyclooxygenase-2-specific inhibitor, is effective in treating primary dysmenorrhea. Obstet Gynecol. 2002;100:350–358. doi: 10.1016/s0029-7844(02)02085-9. [DOI] [PubMed] [Google Scholar]
- 74.Jabbour H.N., Milne S.A., Williams A.R.W., Anderson R.A., Boddy S.C. Expression of COX-2 and PGE synthase and synthesis of PGE2 in endometrial adenocarcinoma a possible autocrine/paracrine regulation of neoplastic cell function via EP2/EP4 receptors. Br J Cancer. 2001;85:1023–1031. doi: 10.1054/bjoc.2001.2033. [DOI] [PubMed] [Google Scholar]
- 75.Molina-Holgado E., Ortiz S., Molina-Holgado F., Guaza C. Induction of COX-2 and PGE(2) biosynthesis by IL-1beta is mediated by PKC and mitogen-activated protein kinases in murine astrocytes. Br J Pharmacol. 2000;131:152–159. doi: 10.1038/sj.bjp.0703557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Smith S.K., Abel M.H., Kelly R.W., Baird D.T. A role for prostacyclin (PGi2) in excessive menstrual bleeding. Lancet. 1981;1:522–524. doi: 10.1016/s0140-6736(81)92862-2. [DOI] [PubMed] [Google Scholar]
- 77.Makarainen L., Ylikorkala O. Primary and myoma-associated menorrhagia role of prostaglandins and effects of ibuprofen. Br J Obstet Gynaecol. 1986;93:974–978. doi: 10.1111/j.1471-0528.1986.tb08019.x. [DOI] [PubMed] [Google Scholar]
- 78.Dikalov S.I., Nazarewicz R.R. Angiotensin II-induced production of mitochondrial reactive oxygen species: potential mechanisms and relevance for cardiovascular disease. Antioxid Redox Signal. 2013;19:1085–1094. doi: 10.1089/ars.2012.4604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Azimi I., Petersen R.M., Thompson E.W., Roberts-Thomson S.J., Monteith G.R. Hypoxia-induced reactive oxygen species mediate N-cadherin and SERPINE1 expression, EGFR signalling and motility in MDA-MB-468 breast cancer cells. Sci Rep. 2017;7:15140. doi: 10.1038/s41598-017-15474-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lippi G., Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hematol Transfus Cell Ther. 2020;42:116–117. doi: 10.1016/j.htct.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hosakote Y.M., Jantzi P.D., Esham D.L., Spratt H., Kurosky A., Casola A., et al. Viral-mediated inhibition of antioxidant enzymes contributes to the pathogenesis of severe respiratory syncytial virus bronchiolitis. Am J Respir Crit Care Med. 2011;183:1550–1560. doi: 10.1164/rccm.201010-1755OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yang D., Elner S.G., Bian Z.M., Till G.O., Petty H.R., Elner V.M. Pro-inflammatory cytokines increase reactive oxygen species through mitochondria and NADPH oxidase in cultured RPE cells. Exp Eye Res. 2007;85:462–472. doi: 10.1016/j.exer.2007.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.van den Brand J.M., Haagmans B.L., van Riel D., Osterhaus A.D., Kuiken T. The pathology and pathogenesis of experimental severe acute respiratory syndrome and influenza in animal models. J Comp Pathol. 2014;151:83–112. doi: 10.1016/j.jcpa.2014.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Smith J.T., Willey N.J., Hancock J.T. Low dose ionizing radiation produces too few reactive oxygen species to directly affect antioxidant concentrations in cells. Biol Lett. 2012;8:594–597. doi: 10.1098/rsbl.2012.0150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Iwasaki A., Gagnon C. Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil Steril. 1992;57:409–416. doi: 10.1016/s0015-0282(16)54855-9. [DOI] [PubMed] [Google Scholar]
- 86.Chen S.J., Allam J.P., Duan Y.G., Haidl G. Influence of reactive oxygen species on human sperm functions and fertilizing capacity including therapeutical approaches. Arch Gynecol Obstet. 2013;288:191–199. doi: 10.1007/s00404-013-2801-4. [DOI] [PubMed] [Google Scholar]
- 87.Will M.A., Swain J., Fode M., Sonksen J., Christman G.M., Ohl D. The great debate: varicocele treatment and impact on fertility. Fertil Steril. 2011;95:841–852. doi: 10.1016/j.fertnstert.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zribi N., Chakroun N.F., Elleuch H., Abdallah F.B., Ben Hamida A.S., Gargouri J., et al. Sperm DNA fragmentation and oxidation are independent of malondialdheyde. Reprod Biol Endocrinol. 2011;9:47. doi: 10.1186/1477-7827-9-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Agarwal A., Said T.M. Role of sperm chromatin abnormalities and DNA damage in male infertility. Hum Reprod Update. 2003;9:331–345. doi: 10.1093/humupd/dmg027. [DOI] [PubMed] [Google Scholar]
- 90.Koppers A.J., Mitchell L.A., Wang P., Lin M., Aitken R.J. Phosphoinositide 3-kinase signalling pathway involvement in a truncated apoptotic cascade associated with motility loss and oxidative DNA damage in human spermatozoa. Biochem J. 2011;436:687–698. doi: 10.1042/BJ20110114. [DOI] [PubMed] [Google Scholar]
- 91.Krishnamoorthy G., Selvakumar K., Venkataraman P., Elumalai P., Arunakaran J. Lycopene supplementation prevents reactive oxygen species mediated apoptosis in Sertoli cells of adult albino rats exposed to polychlorinated biphenyls. Interdiscip Toxicol. 2013;6:83–92. doi: 10.2478/intox-2013-0015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang Y., Chen F., Ye L., Zirkin B., Chen H. Steroidogenesis in Leydig cells: effects of aging and environmental factors. Reproduction. 2017;154:R111–22. doi: 10.1530/REP-17-0064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Shi Z., Feng Y., Wang J., Zhang H., Ding L., Dai J. Perfluorododecanoic acid-induced steroidogenic inhibition is associated with steroidogenic acute regulatory protein and reactive oxygen species in cAMP-stimulated Leydig cells. Toxicol Sci. 2010;114:285–294. doi: 10.1093/toxsci/kfq014. [DOI] [PubMed] [Google Scholar]
- 94.Sai L., Li X., Liu Y., Guo Q., Xie L., Yu G., et al. Effects of chlorpyrifos on reproductive toxicology of male rats. Environ Toxicol. 2014;29:1083–1088. doi: 10.1002/tox.21838. [DOI] [PubMed] [Google Scholar]
- 95.Wu P.Y., Scarlata E., O’Flaherty C. Long-term adverse effects of oxidative stress on rat epididymis and spermatozoa. Antioxidants (Basel) 2020;9:170. doi: 10.3390/antiox9020170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Augoulea A., Mastorakos G., Lambrinoudaki I., Christodoulakos G., Creatsas G. The role of the oxidative-stress in the endometriosis-related infertility. Gynecol Endocrinol. 2009;25:75–81. doi: 10.1080/09513590802485012. [DOI] [PubMed] [Google Scholar]
- 97.White M., Cohen J., Hummel C., Burky R., Cruz A., Farias-Eisner R. The role of oxidative stress in ovarian cancer: implications for the treatment of patients. Cancer. 2014:41–50. [Google Scholar]
- 98.Guérin P., El Mouatassim S., Ménézo Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum Reprod Update. 2001;7:175–189. doi: 10.1093/humupd/7.2.175. [DOI] [PubMed] [Google Scholar]
- 99.Sulaiman M.A.H., Al-Farsi Y.M., Al-Khaduri M.M., Saleh J., Waly M.I. Polycystic ovarian syndrome is linked to increased oxidative stress in Omani women. Int J Womens Health. 2018;10:763–771. doi: 10.2147/IJWH.S166461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.González F., Rote N.S., Minium J., Kirwan J.P. Reactive oxygen species-induced oxidative stress in the development of insulin resistance and hyperandrogenism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91:336–340. doi: 10.1210/jc.2005-1696. [DOI] [PubMed] [Google Scholar]
- 101.Disabled World. Rev . 2020. Human body temperature: COVID-19 : fever : normal : low.https://www.disabled-world.com/calculators-charts/degrees.php [Google Scholar]
- 102.Tharakan S., Nomoto K., Miyashita S., Ishikawa K. Body temperature correlates with mortality in COVID-19 patients. Crit Care. 2020;24:298. doi: 10.1186/s13054-020-03045-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hutchison J.S., Ward R.E., Lacroix J., Hébert P.C., Barnes M.A., Bohn D.J., et al. Hypothermia pediatric head injury trial investigators and the canadian critical care trials group. Hypothermia therapy after traumatic brain injury in children. N Engl J Med. 2008;358:2447–2456. doi: 10.1056/NEJMoa0706930. [DOI] [PubMed] [Google Scholar]
- 104.Bedford J.M. Effects of elevated temperature on the epididymis and testis: experimental studies. Adv Exp Med Biol. 1991;286:19–32. doi: 10.1007/978-1-4684-5913-5_3. [DOI] [PubMed] [Google Scholar]
- 105.Jung A., Schuppe H.C., Schill W.B. Fieber als Ursache einer temporären Fertilitätseinschränkung des Mannes [Fever as etiology of temporary infertility in the man] Hautarzt. 2001;52:1090–1093. doi: 10.1007/s001050170018. [DOI] [PubMed] [Google Scholar]
- 106.Koentjoro-Soehadi L. Azoospermia caused by typhoid fever. A case report. Andrologia. 1982;14 doi: 10.1111/j.1439-0272.1982.tb03092.x. 31-2,34. [DOI] [PubMed] [Google Scholar]
- 107.Andrade-Rocha F.T. Temporary impairment of semen quality following recent acute fever. Ann Clin Lab Sci. 2013;43:94–97. [PubMed] [Google Scholar]
- 108.Sergerie M., Mieusset R., Croute F., Daudin M., Bujan L. High risk of temporary alteration of semen parameters after recent acute febrile illness. Fertil Steril. 2007;88 doi: 10.1016/j.fertnstert.2006.12.045. 970.e1-7. [DOI] [PubMed] [Google Scholar]
- 109.Yin Y., Hawkins K.L., DeWolf W.C., Morgentaler A. Heat stress causes testicular germ cell apoptosis in adult mice. J Androl. 1997;18:159–165. [PubMed] [Google Scholar]
- 110.Aktas C., Kanter M. A morphological study on Leydig cells of scrotal hyperthermia applied rats in short-term. J Mol Histol. 2009;40:31–39. doi: 10.1007/s10735-009-9210-9. [DOI] [PubMed] [Google Scholar]
- 111.Kulibin A.Y., Malolina E.A. Sertoli cells cultured under high-temperature and hypoxic conditions. Cell Tiss Biol. 2014;8:97–106. [PubMed] [Google Scholar]
- 112.Wang J.Z., Sui H.S., Miao D.Q., Liu N., Zhou P., Ge L., et al. Effects of heat stress during in vitro maturation on cytoplasmic versus nuclear components of mouse oocytes. Reproduction. 2009;137:181–189. doi: 10.1530/REP-08-0339. [DOI] [PubMed] [Google Scholar]
- 113.Sirotkin A.V., Kacaniova M. The effect of high temperature on swine ovarian function in vitro. Vet Med. 2010;55:377–382. [Google Scholar]
- 114.Bridges P.J., Brusie M.A., Fortune J.E. Elevated temperature (heat stress) in vitro reduces androstenedione and estradiol and increases progesterone secretion by follicular cells from bovine dominant follicles. Domest Anim Endocrinol. 2005;29:508–522. doi: 10.1016/j.domaniend.2005.02.017. [DOI] [PubMed] [Google Scholar]
- 115.Hafez E.S.E. Effects of high temperature on reproduction. Int J Biometeorol. 1964;7:223–230. [Google Scholar]
- 116.Wise M.E., Armstrong D.V., Huber J.T., Hunter R., Wiersma F. Hormonal alterations in the lactating dairy cow in response to thermal stress. J Dairy Sci. 1988;71:2480–2485. doi: 10.3168/jds.S0022-0302(88)79834-3. [DOI] [PubMed] [Google Scholar]
- 117.Shimizu T., Ohshima I., Ozawa M., Takahashi S., Tajima A., Shiota M., et al. Heat stress diminishes gonadotropin receptor expression and enhances susceptibility to apoptosis of rat granulosa cells. Reproduction. 2005;129:463–472. doi: 10.1530/rep.1.00502. [DOI] [PubMed] [Google Scholar]
- 118.Roth Z., Aroyo A., Yavin S., Arav A. The antioxidant epigallocatechin gallate (EGCG) moderates the deleterious effects of maternal hyperthermia on follicle-enclosed oocytes in mice. Theriogenology. 2008;70:887–897. doi: 10.1016/j.theriogenology.2008.05.053. [DOI] [PubMed] [Google Scholar]
- 119.Xiang Y.T., Yang Y., Li W., Zhang L., Zhang Q., Cheung T., et al. Timely mental health care for the 2019 novel coronavirus outbreak is urgently needed. Lancet Psychiatry. 2020;7:228–229. doi: 10.1016/S2215-0366(20)30046-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Serafini G., Parmigiani B., Amerio A., Aguglia A., Sher L., Amore M. The psychological impact of COVID-19 on the mental health in the general population. QJM. 2020;113:531–537. doi: 10.1093/qjmed/hcaa201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Cacioppo J.T., Berntson G.G., Malarkey W.B., Kiecolt-Glaser J.K., Sheridan J.F., Poehlmann K.M., et al. Autonomic, neuroendocrine, and immune responses to psychological stress: the reactivity hypothesis. Ann N Y Acad Sci. 1998;840:664–673. doi: 10.1111/j.1749-6632.1998.tb09605.x. [DOI] [PubMed] [Google Scholar]
- 122.Xiao Y.C., Huang Y.D., Hardy D.O., Li X.K., Ge R.S. Glucocorticoid suppresses steroidogenesis in rat progenitor Leydig cells. J Androl. 2010;31:365–371. doi: 10.2164/jandrol.109.009019. [DOI] [PubMed] [Google Scholar]
- 123.Kotitschke A., Sadie-Van Gijsen H., Avenant C., Fernandes S., Hapgood J.P. Genomic and nongenomic cross talk between the gonadotropin-releasing hormone receptor and glucocorticoid receptor signaling pathways. Mol Endocrinol. 2009;23:1726–1745. doi: 10.1210/me.2008-0462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Smith L.B., Walker W.H. The regulation of spermatogenesis by androgens. Semin Cell Dev Biol. 2014;30:2–13. doi: 10.1016/j.semcdb.2014.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jurewicz J., Hanke W., Sobala W., Merecz D., Radwan M. The effect of stress on the semen quality [Polish] Med Pr. 2010;61:607–613. [PubMed] [Google Scholar]
- 126.Gollenberg A.L., Liu F., Brazil C., Drobnis E.Z., Guzick D., Overstreet J.W., et al. Semen quality in fertile men in relation to psychosocial stress. Fertil Steril. 2010;93:1104–1111. doi: 10.1016/j.fertnstert.2008.12.018. [DOI] [PubMed] [Google Scholar]
- 127.Eskiocak S., Gozen A.S., Taskiran A., Kilic A.S., Eskiocak M., Gulen S. Effect of psychological stress on the L-arginine-nitric oxide pathway and semen quality. Braz J Med Biol Res. 2006;39:581–588. doi: 10.1590/s0100-879x2006000500003. [DOI] [PubMed] [Google Scholar]
- 128.Althof S.E., Needle R.B. Psychological factors associated with male sexual dysfunction: screening and treatment for the urologist. Urol Clin North Am. 2011;38:141–146. doi: 10.1016/j.ucl.2011.02.003. [DOI] [PubMed] [Google Scholar]
- 129.Genazzani A.D., Ricchieri F., Lanzoni C., Strucchi C., Jasonni V.M. Diagnostic and therapeutic approach to hypothalamic amenorrhea. Ann N Y Acad Sci. 2006;1092:103–113. doi: 10.1196/annals.1365.009. [DOI] [PubMed] [Google Scholar]
- 130.Barsom S.H., Mansfield P.K., Koch P.B., Gierach G., West S.G. Association between psychological stress and menstrual cycle characteristics in perimenopausal women. Womens Health Issues. 2004;14:235–241. doi: 10.1016/j.whi.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 131.Allsworth J.E., Zierler S., Lapane K.L., Krieger N., Hogan J.W., Harlow B.L. Longitudinal study of the inception of perimenopause in relation to lifetime history of sexual or physical violence. J Epidemiol Community Health. 2004;58:938–943. doi: 10.1136/jech.2003.017160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Nepomnaschy P.A., Sheiner E., Mastorakos G., Arck P.C. Stress, immune function, and women’s reproduction. Ann N Y Acad Sci. 2007;1113:350–364. doi: 10.1196/annals.1391.028. [DOI] [PubMed] [Google Scholar]
- 133.Pourali L., Vatanchi A.M., Hamidi A. A case of Cushing’s syndrome in pregnancy. Iran J Med Sci. 2017;42:607–610. [PMC free article] [PubMed] [Google Scholar]
- 134.Nepomnaschy P.A., Welch K.B., McConnell D.S., Low B.S., Strassmann B.I., England B.G. Cortisol levels and very early pregnancy loss in humans. Proc Natl Acad Sci U S A. 2006;103:3938–3942. doi: 10.1073/pnas.0511183103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Klonoff-Cohen H., Chu E., Natarajan L., Sieber W. A prospective study of stress among women undergoing in vitro fertilization or gamete intrafallopian transfer. Fertil Steril. 2001;76:675–687. doi: 10.1016/s0015-0282(01)02008-8. [DOI] [PubMed] [Google Scholar]
- 136.Carrell D.T., Wilcox A.L., Lowy L., Peterson C.M., Jones K.P., Erickson L., et al. Elevated sperm chromosome aneuploidy and apoptosis in patients with unexplained recurrent pregnancy loss. Obstet Gynecol. 2003;101:1229–1235. doi: 10.1016/s0029-7844(03)00339-9. [DOI] [PubMed] [Google Scholar]
- 137.Saleh R.A., Agarwal A. Oxidative stress and male infertility: from research bench to clinical practice. J Androl. 2002;23:737–752. [PubMed] [Google Scholar]
- 138.Tamura H., Takasaki A., Miwa I., Taniguchi K., Maekawa R., Asada H., et al. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J Pineal Res. 2008;44:280–287. doi: 10.1111/j.1600-079X.2007.00524.x. [DOI] [PubMed] [Google Scholar]
- 139.Harrity C., Shkrobot L., Walsh D., Marron K. ART implantation failure and miscarriage in patients with elevated intracellular cytokine ratios: response to immune support therapy. Fertil Res Pract. 2018;4:7. doi: 10.1186/s40738-018-0052-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Winger E.E., Reed J.L., Ashoush S., El-Toukhy T., Ahuja S., Taranissi M. Degree of TNF-α/IL-10 cytokine elevation correlates with IVF success rates in women undergoing treatment with Adalimumab (Humira) and IVIG. Am J Reprod Immunol. 2011;65:610–618. doi: 10.1111/j.1600-0897.2010.00946.x. [DOI] [PubMed] [Google Scholar]
- 141.Inagaki N., Stern C., McBain J., Lopata A., Kornman L., Wilkinson D. Analysis of intra-uterine cytokine concentration and matrix-metalloproteinase activity in women with recurrent failed embryo transfer. Hum Reprod. 2003;18:608–615. doi: 10.1093/humrep/deg139. [DOI] [PubMed] [Google Scholar]
- 142.Nikolaeva M.A., Babayan A.A., Stepanova E.O., Smolnikova V.Y., Kalinina E.A., Fernández N., et al. The relationship of seminal transforming growth factor-β1 and interleukin-18 with reproductive success in women exposed to seminal plasma during IVF/ICSI treatment. J Reprod Immunol. 2016;117:45–51. doi: 10.1016/j.jri.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 143.Nikolaeva M., Arefieva A., Babayan A., Chagovets V., Kitsilovskaya N., Starodubtseva N., et al. Immunoendocrine markers of stress in seminal plasma at IVF/ICSI failure: a preliminary study. Reprod Sci. 2021;28:144–158. doi: 10.1007/s43032-020-00253-z. [DOI] [PubMed] [Google Scholar]
- 144.Cikos S., Rehák P., Czikková S., Veselá J., Koppel J. Expression of adrenergic receptors in mouse preimplantation embryos and ovulated oocytes. Reproduction. 2007;133:1139–1147. doi: 10.1530/REP-07-0006. [DOI] [PubMed] [Google Scholar]
- 145.Nepomnaschy P.A., Welch K.B., McConnell D.S., Low B.S., Strassmann B.I., England B.G. Cortisol levels and very early pregnancy loss in humans. Proc Natl Acad Sci U S A. 2006;103:3938–3942. doi: 10.1073/pnas.0511183103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.An Y., Sun Z., Li L., Zhang Y., Ji H. Relationship between psychological stress and reproductive outcome in women undergoing in vitro fertilization treatment: psychological and neurohormonal assessment. J Assist Reprod Genet. 2013;30:35–41. doi: 10.1007/s10815-012-9904-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Zhou F.J., Cai Y.N., Dong Y.Z. Stress increases the risk of pregnancy failure in couples undergoing IVF. Stress. 2019;22:414–420. doi: 10.1080/10253890.2019.1584181. [DOI] [PubMed] [Google Scholar]
- 148.Awwad J., Ghazeeri G., Toth T., Hannoun A., Abdallah M.A., Farra C. Fever in women may interfere with follicular development during controlled ovarian stimulation. Int J Hyperthermia. 2012;28:742–746. doi: 10.3109/02656736.2012.724516. [DOI] [PubMed] [Google Scholar]
- 149.Sergerie M., Mieusset R., Croute F., Daudin M., Bujan L. High risk of temporary alteration of semen parameters after recent acute febrile illness. Fertil Steril. 2007;88 doi: 10.1016/j.fertnstert.2006.12.045. 970.e1-7. [DOI] [PubMed] [Google Scholar]
- 150.Surcel M., Surcel M., Zlatescu-Marton C., Micu R., Nemeti G.I., Axente D.D., et al. The role of high follicular levels of angiotensin ii and vascular endothelial growth factor in anticipating the development of severe ovarian hyperstimulation syndrome in patients with prophylactic cabergoline therapy undergoing an in vitro fertilization procedure. Acta endocrinologica (Bucharest, Romania. 2005;2020(16):30–36. doi: 10.4183/aeb.2020.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Gianzo M., Subirán N. Regulation of male fertility by the renin-angiotensin system. Int J Mol Sci. 2020;21:7943. doi: 10.3390/ijms21217943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lamamri M., Chebbi A., Mamane J., Abbad S., Munuzzolini M., Sarfati F., et al. Priapism in a patient with coronavirus disease 2019 (COVID-19) Am J Emerg Med. 2021;39 doi: 10.1016/j.ajem.2020.06.027. 251.e5-251.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Kim J., Thomsen T., Sell N., Goldsmith A.J. Abdominal and testicular pain: an atypical presentation of COVID-19. Am J Emerg Med. 2020;38(1542) doi: 10.1016/j.ajem.2020.03.052. e1-1542.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Ma X., Guan C., Chen R., Wang Y., Feng S., Wang R., et al. Pathological and molecular examinations of postmortem testis biopsies reveal SARS-CoV-2 infection in the testis and spermatogenesis damage in COVID-19 patients. Cell Mol Immunol. 2021;18(2):487–489. doi: 10.1038/s41423-020-00604-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Yang M., Chen S., Huang B., Zhong J.M., Su H., Chen Y.J., et al. Pathological findings in the testes of COVID-19 patients: clinical implications. Eur Urol Focus. 2020;6(5):1124–1129. doi: 10.1016/j.euf.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Alay I., Yildiz S., Kaya C., Yasar K.K., Aydin O.A., Karaosmanoglu H.K., et al. The clinical findings and outcomes of symptomatic pregnant women diagnosed with or suspected of having coronavirus disease 2019 in a tertiary pandemic hospital in Istanbul, Turkey. J Obstet Gynaecol Res. 2020 doi: 10.1111/jog.14493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Sentilhes L., De Marcillac F., Jouffrieau C., Kuhn P., Thuet V., Hansmann Y., et al. Coronavirus disease 2019 in pregnancy was associated with maternal morbidity and preterm birth. Am J Obstet Gynecol. 2020;223 doi: 10.1016/j.ajog.2020.06.022. 914.e1-914.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Jaiswal N., Puri M., Agarwal K., Singh S., Yadav R., Tiwary N., et al. COVID-19 as an independent risk factor for subclinical placental dysfunction. Eur J Obstet Gynecol Reprod Biol. 2021;259:7–11. doi: 10.1016/j.ejogrb.2021.01.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.