Dear Editor,
The Qinghai-Tibet Plateau (QTP) is the highest and largest plateau in the world; its altitude exceeds 4500 m on average, posing great physiological challenges such as severe hypoxia for endemic wild mammals on the plateau.1 To cope with such a harsh environment, the phylogenetically distant QTP endemic mammals have independently evolved similar morphological and physiological traits. For example, compared with the lowland mammals, the QTP mammals have a larger ratio of heart mass to body mass (HM/BM)2,3 and prominently lower pulmonary artery pressure (PAP),1,4 leading to a high efficiency of oxygen transport under chronically hypoxic environment. Although species-specific genetic adaptations have been described in several QTP endemic mammals,5,6 little is known about the extent to which common molecular bases contribute to the evolution of the convergent traits across phylogenetically distant mammalian lineages on the QTP.
To address this question, we de novo sequenced and assembled two draft genomes of the plateau pika (Ochotona curzoniae; Lagomorpha) and the plateau zokor (Myospalax baileyi; Rodentia) (Supplementary information, Tables S1–S6). Together with yak5 and Tibetan antelope,6 the four species span the major phylogenetic lineages of QTP endemic mammals that independently evolved similar phenotypic traits (i.e., higher HM/BM and lower PAP) as a response to highland environment over millions of years. To examine the genome-wide convergent/parallel evolution among these QTP mammals, we chose their respective known closest lowland, living-in-normoxia relatives with genomic sequences available, including cow (Bos taurus), sheep (Ovis aries), mouse (Mus musculus), and rabbit (Oryctolagus cuniculus), as well as five outgroup mammal species as controls (Fig. 1a), which could ensure the reliability of inferring ancestral sequences and detecting the molecular convergences or parallels. Across these 13 mammal species, we finally obtained a total of 6643 high-confidence one-to-one orthologous protein-coding genes. Following the evolutionary analyses of our previous study,7 we discovered 16 and 11 genes under parallel evolution among at least three of four QTP species (Supplementary information, Table S7) and among three of four lowland relatives (Supplementary information, Table S8), respectively, but found no convergent genes in both groups. The genes under parallel evolution are expected to contribute to the molecular basis of the convergent phenotypes among the highland mammals. To test this prediction, we performed the functional enrichment analyses and found that these genes from the QTP mammals are enriched in the functional categories related to heart development compared with those from their lowland counterparts (P = 6.2e−5, two-tailed Student’s t-test; Supplementary information, Tables S9, S10). This result is to some extent coincident with the observation that the QTP mammals have a higher HM/BM value and a more powerful cardiovascular system.2,3
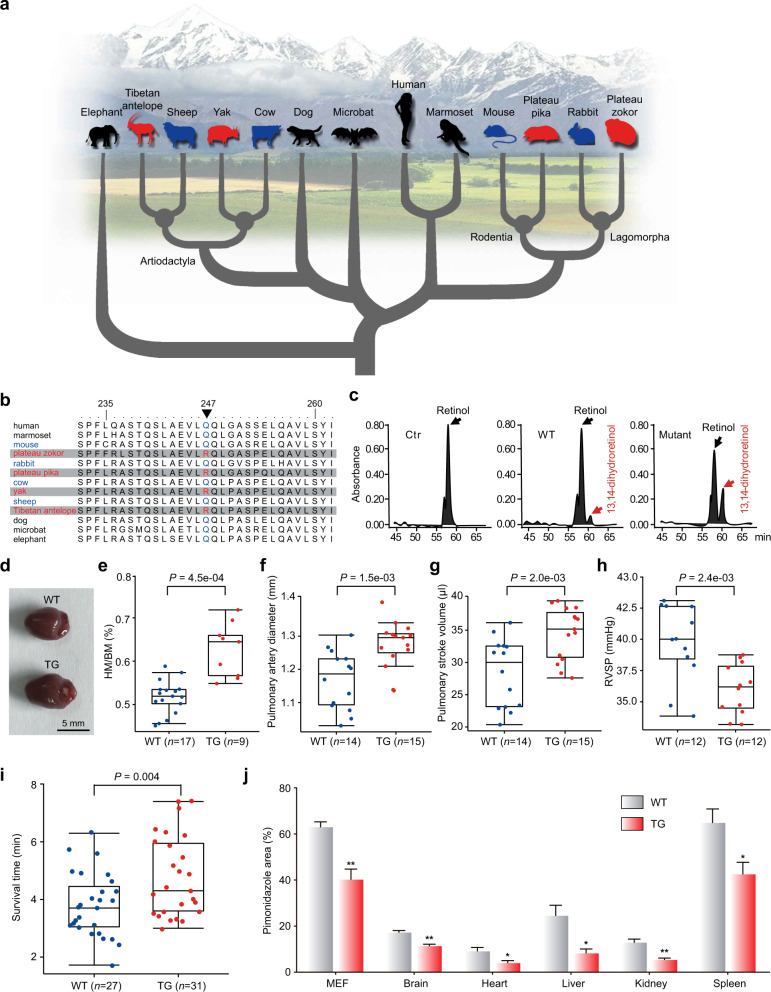
Fig. 1. Substitution of a parallel amino acid of RETSAT among QTP mammals endows mice with convergent phenotypes and enhanced hypoxia tolerance.
a Phylogeny of the mammals used to examine genome-wide convergent/parallel evolution in this study. Endemic species on the QTP and their phylogenetically close lowland relatives are highlighted in red and blue, respectively. b The black triangle indicates the parallel amino acid of RETSAT. c Enzymatic activity of RETSAT is compared among the negative control, WT RETSAT (247Q), and mutant RETSAT (247R). The black arrow points to the peak of retinol and the red arrow points to the peak of 13, 14-dihydroretinol. d Representative images of hearts of the TG and WT mice. e The HM/BM value of the TG mice is larger than that of the WT mice. f–h Echocardiography analyses between the TG and WT mice. RVSP, right ventricular systolic pressure determined by the correlation with the ratio of pulmonary acceleration time (PAT) to pulmonary ejection time (PET). i The TG mice (n = 31) live longer in the acute hypoxic condition (4% O2) compared with the WT mice (n = 27). j Relative hypoxic areas stained using pimonidazole dye in MEFs and vital organs (brain, heart, liver, kidney, and spleen) are smaller in the TG mice than in the WT mice. Data are shown as means ± SEM; *P < 0.05; **P < 0.01. Boxes in e–h indicate the first, the median, and the third quartiles. Whiskers denote upper and lower bounds. All P values are from two-sided Student’s t-tests. The representative images are shown in Supplementary information, Fig. S6.
Among the 16 parallel genes in the QTP mammals, only the retinol saturase gene (RETSAT) contains a single parallel amino acid change from glutamine (Q) to arginine (R) at position 247 in all four QTP mammals (Fig. 1b). Previous studies showed that RETSAT saturates the 13–14 double bond of all-trans-retinol to produce all-trans-13,14-dihydroretinol,8 and is involved in vitamin A metabolism,9 lipid metabolism, and production of reactive oxygen species.10 However, this gene has never been implicated in hypoxia adaptation in QTP mammals. We thus performed in vitro and in vivo experiments to test whether this QTP-specific parallel substitution of RETSAT is functionally significant in the evolution of convergent phenotypes of the QTP mammals. We incubated the retinoid substrates with the HEK293T cells expressing RETSAT. The HPLC analyses of cell lysates showed that RETSAT with the highland substitution (247R) has a higher enzymatic activity than that with the wild-type (WT) 247Q (Fig. 1c; Supplementary information, Fig. S1), indicating that this substitution (Q247R) causes a biochemical functional change of RETSAT. Next, we created a line of genetically engineered mice by mutating glutamine to arginine at position 247 of RETSAT (Supplementary information, Fig. S2). The mice carrying the homozygous Q247R mutation (RETSATR/R; transgenic (TG)) have significantly higher HM than the WT mice (P = 1.1e−4, two-tailed Student’s t-test) (Fig. 1d; Supplementary information, Fig. S3a, b) without significant difference in BM (Supplementary information, Fig. S3c). Consequently, the HM/BM values of the TG mice are significantly larger than those of the WT mice (P = 4.5e−4, two-tailed Student’s t-test; Fig. 1e). Further, our echocardiographic analyses showed that compared with the WT mice, the TG mice exhibit a larger pulmonary artery diameter (Fig. 1f), higher pulmonary stroke volume (Fig. 1g), larger pulmonary cardiac output (Supplementary information, Fig. S4a), and lower right ventricular systolic pressure (RVSP) (Fig. 1h; Supplementary information, Fig. S4b). In addition, the TG mice transport oxygen to the liver more efficiently than the WT mice (P = 1.1e−5, two-tailed Student’s t-test; Supplementary information, Fig. S5). Taken together, the TG mice display multiple similar phenotypes required for adapting to the highland environment for the QTP endemic mammalian lineages,1–4 suggesting that a single parallel amino acid substitution can lead to convergent phenotypes.
Based on the observation that TG mice have similar phenotypes to the QTP mammals, we hypothesized that these mice could survive better under hypoxic conditions than the WT mice. In survival experiments, we indeed found that the survival time of the TG mice is significantly longer than that of the WT littermates under an acute hypoxia condition (4% O2) (P = 0.00397, two-tailed Student’s t-test; Fig. 1i). The result is highly consistent even when considering the female and male animals, respectively (Supplementary information, Fig. S6). Further, the TG mice also display hypoxia resistance at cellular and tissue levels. After culturing mouse embryonic fibroblast (MEF) cells under hypoxic conditions (1% O2), the pimonidazole staining analyses showed that the relative hypoxic area is significantly smaller in the TG mice than in the WT mice (Fig. 1j; Supplementary information, Fig. S7a). Similarly, significantly smaller hypoxia areas are observed in the vital organs including brain, heart, liver, kidney, and spleen of the TG mice after hypoxic treatment (Fig. 1j; Supplementary information, Fig. S7b–f). These experiments further demonstrate that the TG mice can be more resistant to hypoxia stress than the WT littermates.
Our study not only identifies parallel genetic changes among multiple phylogenetically independent lineages of the QTP mammals at the genomic scale, but also demonstrates that a single amino acid substitution in RETSAT can qualitatively create some of the convergent phenotypes of the QTP mammals. Notably, this parallel substitution was genomically screened out and tested for adaptive selection among only 13 mammalian species, which may bias our evolutionary and statistical analyses due to the small sample size. To overcome this limitation, we collected the RETSAT protein sequences from a total of 137 mammalian species (Supplementary information, Table S11) to test the selection of this substitution11 and confirmed that natural selection, rather than chance, underlies the occurrence of the substitution (P < 10–4; Supplementary information, Table S12). In addition, we observed that this parallel substitution occurs in some non-hypoxia-tolerant mammals. Although this observation is not surprising and does not overturn our conclusion because (i) the pure patterns of molecular convergences or parallels rarely exist when taxonomic sampling depth increases12 and (ii) the occurrence of the parallel substitution in some background species does not necessarily preclude a functional effect on the protein13 as our experiments proved for the Q247R of RETSAT, we proposed the following possibilities why the parallel substitution occurs in non-hypoxia-tolerant mammals. One possibility is that some non-hypoxia-tolerant species with the parallel substitution own a large HM/BM value as we observed in the QTP mammals such as degu14 and white-tailed deer.15 Another possibility is that this parallel substitution may have pleiotropic effects on phenotypes that are not related to the increase of HM/BM value, which warrants further exploration. Our study represents a typical empirical example for revealing a close relationship between molecular convergences and convergent phenotypes in mammals. The identification of a novel major effect gene sheds new light on the genetic mechanisms underlying hypoxia adaptation and provides new targets for the prevention and treatment of common mountain diseases, such as pulmonary arterial hypertension and right ventricle hypertrophy.
Supplementary information
Acknowledgements
We thank Zhengting Zou for modifying the computational program of convCalScript and Zhong-Ru Gu for his advice in genome assembly. This work was supported jointly and equally by the Second Tibetan Plateau Scientific Expedition and Research Program (2019QZKK0501) and the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB13020400). This work was also supported by the National Natural Science Foundation of China (U1902216, 81772996, 81672764, 31621062, 31871270, 31922010, 31871277, 3190013), Science and Technology Department of Qinghai Province (Sanjiangyuan National Park Animal Genome Program), and Yunnan Applied Basic Research Projects (2016FA009, 2019FJ009). C.Y., S.P. and Z.L. were also supported by the Youth Innovation Promotion Association, CAS, and the Youth Talent Support Program of Yunnan Province (2019FI008).
Author contributions
P.S., Y.C., and X.-J.Z. designed and supervised the project. P.S. coordinated the project. X.-J.Z. led the genome assembly group. Y.C. led the functional experiment group. P.S. led the evolutionary genome analysis group. T.-Z.Z. collected samples. D.X., S.P., and X.-J.Z. performed genome assembly and annotation. D.X., Z.L., H.Y., T.Z., Y.G., P.C., and P.S. analyzed genomic data. C.Y., Q.S., X.Z., M.L., and Y.C. performed functional experiments. D.X., Z.L., Y.C., C.Y., X.-J.Z., and P.S. wrote the manuscript.
Data availability
The raw sequence data reported in this study have been deposited in the Genome Sequence Archive in BIG Data Center, Beijing Institute of Genomics, Chinese Academy of Sciences (accession numbers CRA000080, CRA000079). They are publicly accessible at https://bigd.big.ac.cn/gsa. The assemblies for the plateau zokor and plateau pika have been deposited in the BIG Genome Warehouse (GWH) under project numbers GWHABJY00000000 and GWHABJX00000000. The RETSAT protein sequence alignment across 137 mammals has been uploaded to the Dryad Digital Repository (doi:10.5061/dryad.ttdz08kxg).
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Dongming Xu, Cuiping Yang, Qiushuo Shen, Shengkai Pan, Zhen Liu, Tongzuo Zhang
Contributor Information
Xiangjiang Zhan, Email: zhanxj@ioz.ac.cn.
Yongbin Chen, Email: ybchen@mail.kiz.ac.cn.
Peng Shi, Email: ship@mail.kiz.ac.cn.
Supplementary information
The online version contains supplementary material available at 10.1038/s41422-021-00517-6.
References
- 1.Storz JF, et al. J. Exp. Biol. 2010;213:4125–4136. doi: 10.1242/jeb.048181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Qi X, et al. Acta Physiol. Sin. 2008;60:348–354. [PubMed] [Google Scholar]
- 3.Rong C, et al. Am. J. Vet. Res. 2012;73:809–813. doi: 10.2460/ajvr.73.6.809. [DOI] [PubMed] [Google Scholar]
- 4.Anand I, et al. Thorax. 1986;41:696–700. doi: 10.1136/thx.41.9.696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Qiu Q, et al. Nat. Genet. 2012;44:946–949. doi: 10.1038/ng.2343. [DOI] [PubMed] [Google Scholar]
- 6.Ge RL, et al. Nat. Commun. 2013;4:1858. doi: 10.1038/ncomms2860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Z, et al. Sci. Adv. 2018;4:eaat8821. doi: 10.1126/sciadv.aat8821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moise AR, et al. J. Biol. Chem. 2004;279:50230–50242. doi: 10.1074/jbc.M409130200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Moise AR, et al. Mol. Pharmacol. 2009;76:1228–1237. doi: 10.1124/mol.109.060038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pang XY, et al. Arch. Biochem. Biophys. 2017;633:93–102. doi: 10.1016/j.abb.2017.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zou Z, et al. Mol. Biol. Evol. 2015;32:2085–2096. doi: 10.1093/molbev/msv091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Thomas GW, et al. Genome Biol. Evol. 2017;9:213–221. doi: 10.1093/gbe/evw306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee JH, et al. Sci. Adv. 2018;4:eaat9660. doi: 10.1126/sciadv.aat9660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Meijler FL, et al. Heart Rhythm. 2005;2:188–196. doi: 10.1016/j.hrthm.2004.11.018. [DOI] [PubMed] [Google Scholar]
- 15.Crile GC, et al. Ohio J. Sci. 1939;40:219–259. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The raw sequence data reported in this study have been deposited in the Genome Sequence Archive in BIG Data Center, Beijing Institute of Genomics, Chinese Academy of Sciences (accession numbers CRA000080, CRA000079). They are publicly accessible at https://bigd.big.ac.cn/gsa. The assemblies for the plateau zokor and plateau pika have been deposited in the BIG Genome Warehouse (GWH) under project numbers GWHABJY00000000 and GWHABJX00000000. The RETSAT protein sequence alignment across 137 mammals has been uploaded to the Dryad Digital Repository (doi:10.5061/dryad.ttdz08kxg).