Abstract
We report a case of a 62‐year‐old man who developed cerebral venous sinus thrombosis with subarachnoid hemorrhage and concomitant thrombocytopenia, which occurred 13 days after ChAdOx1 nCov‐19 injection. The patient died in the intensive care unit after heparin infusion and platelet transfusion. The key clinical purpose of this case report is to better understand how to confirm vaccine‐induced immune thrombotic thrombocytopenia (VITT). VITT diagnosis was made using 14C‐serotonin release and flow cytometry evaluating activation and platelet microvesicles on washed platelets. Four control patients were examined: a patient with heparin‐induced thrombocytopenia (HIT), two patients with thrombotic events without thrombocytopenia after ChAdOx1 nCov‐19 or BNT162b2, and a patient with suspected HIT and an excluded diagnosis. We evidenced in the VITT case a high level of IgG anti–platelet factor 4–heparin antibodies associated with a high level of platelet activation in the absence of heparin. Conversely, the functional assays were negative in the patients with thrombosis without thrombocytopenia.
Keywords: COVID‐19, platelet factor 4, thrombocytopenia, thrombosis, vaccines
Essentials.
Vaccine‐induced immune thrombotic thrombocytopenia (VITT) can appear after vaccination.
VITT is characterized by positive antiplatelet factor 4 antibodies.
Heparin infusion and platelet transfusion induce worsening of the disease.
We confirm that VITT has a specific profile using specific platelet activation assays.
1. INTRODUCTION
Respiratory tract infection by severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) is responsible for a systemic inflammatory disease (coronavirus disease 2019 [COVID‐19]) associated with coagulopathy and endothelial damage that lead to microthrombosis.1, 2, 3, 4, 5, 6, 7 The COVID‐19 pandemic is still poorly controlled, and mass vaccination programs are ongoing to lower both the SARS‐CoV‐2 circulation and the pressure on health systems.8, 9, 10 We previously analyzed the frequency of anti–COVID‐19 vaccination thrombotic events reported in the World Health Organization Global Database for Individual Case Safety Reports (VigiBase). It was found that thrombotic events, including cerebral venous thrombosis, might occur in association with all three vaccines available in Europe, without evidence of a link between the vaccines and the thrombotic events.11 However, three studies recently described thrombotic thrombocytopenia after ChAdOx1 nCov‐19 (vaxzevria, the Oxford–AstraZeneca vaccine) respectively in 11, 5, and 23 patients.12, 13, 14 Moreover, Muir et al15 described one patient with the same syndrome 3 days after Ad26.COV2.S vaccination (Johnson & Johnson/Janssen vaccine). This rare syndrome has been called vaccine‐induced immune thrombotic thrombocytopenia (VITT) or thrombosis with thrombocytopenia syndrome as defined by the World Health Organization.
2. CASE REPORT
We report here a 62‐year‐old French man admitted into an emergency department with epigastric discomfort and severe right‐sided abdominal pain occurring 11 days after his first intramuscular injection with the ChAdOx1 nCov‐19 vaccine. The patient’s body mass index was 32.7 kg/m2, and he had no history of thrombosis, nor did his family. He had a medical history of diabetes mellitus, hypereosinophilic asthma treated with benralizumab, and obstructive sleep apnea. This patient was immediately transferred to the gastroenterology unit for endoscopic retrograde cholangiopancreatography for suspected bile‐duct stone migration. He showed no fever, and physical examination was mundane, with the exception of an intense tenderness on abdominal palpation. Upon admission, laboratory examination revealed a moderate increase in C‐reactive protein to 56 mg/L (normal value, <5 mg/L), moderate cytolysis without cholestasis, leukocyte count of 21.3 × 109/L, hemoglobin level of 174 g/L, and platelet count of 100 × 109/L. A SARS‐CoV‐2 reverse‐transcriptase polymerase chain reaction assay of a nasopharyngeal swab was negative. Abdominal computed tomography (CT) imaging was suggestive of bile‐duct stone migration due to macrolithiasis in the gallbladder. The following day (day 12), the platelet count had fallen to 10 × 109/L.
Further laboratory evaluation was negative for hemolysis, with normal haptoglobin and lactate dehydrogenase levels and absence of schizocytes. A disseminated intravascular coagulopathy (DIC) panel demonstrated a D‐dimer level above 4000 ng/mL, measured using an immuno‐turbidimetric assay (STA‐Liatest D‐Di PLUS) on a routine laboratory coagulometer STA‐R Max Diagnostica Stago (Asnieres‐sur‐seine, France; normal range, 0‐500 ng/mL), and a normal level of fibrinogen 4.1 g/L, a soluble fibrin monomer complex level of >150 µg/mL (normal value, <7 µg/mL), and a high‐sensitivity cardiac troponin I level at 4224 ng/mL (normal value, <11.6 ng/mL) were found (without ST‐segment elevation but the presence of pathological Q waves in lead III on the initial electrocardiogram [ECG]). The international normalized ratio was 1.11 and prothrombin time 14.6 seconds (vs 13.2 seconds for control). In the aftermath, the patient had a 39℃ fever, weakness in the right arm, and mental confusion, requiring his transfer to the intensive care unit, and was administered antibiotics and one unit of platelet concentrate. Following his transfer to the intensive care unit, several ECGs were performed, which indicated acute ischemia with ST‐segment elevation in contiguous leads V6‐V9 and ST depressions in leads V1‐V3, with an increase in cardiac troponin I level at 7217 ng/mL. A head CT scan revealed superior sagittal sinus thrombosis. Intravenous unfractionated heparin (UFH) at a therapeutic regimen was started, despite high risk of hemorrhage transformation. The patient’s condition worsened dramatically 3 hours later, with heart failure, myocardial infarction, neurological aggravation, and respiratory distress that required intubation. Livedo reticularis appeared on the lower limbs, abdomen, and trunk. Repeated head CT imaging revealed a large left parietal hematoma over a rupture of the thrombosed cortical vein. At this stage, these syndromes led us to envisage two competing diagnoses: (i) heparin‐induced thrombocytopenia (HIT) was initially suspected due to a suggestive clinical presentation, but the patient had not received heparin therapy before intensive care unit admission or during the previous 3 months; and (ii) catastrophic antiphospholipid syndrome was suspected and treated accordingly with intravenous methylprednisolone (1 g).
The patient died a few hours later, at day 13 following ChAdOx1 nCov‐19 injection. The results of the autopsy confirmed death by superior sagittal sinus thrombosis with subarachnoid hemorrhage and a rupture of the thrombosed cortical vein. The autopsy also confirmed multivisceral thrombosis in small, medium, and large vessels in many organs, such as the heart (Figure 1A), the liver (Figure 1B), the kidney, and the spleen, accompanied by typical lesions of ischemic myocardial and hepatic necrosis. Macroscopically, the heart and liver appeared heterogeneous. Microscopically, in the liver, we observed the presence of steatosis (30%), central lobular necrosis, and vascular and endothelial thrombi in medium‐sized vessels. In the heart, there was gross morphologic evidence of early myocardial infarction, with an organizing thrombus in a branch of the left circumflex coronary. A standard immunohistochemical method detected a heterogeneous desmin depletion. The blood vessels had clumped red blood cells in the lumen, forming fibrino‐cruoric microthrombi.
FIGURE 1.

Autopsy findings. Autopsy findings with multivisceral thrombosis in the heart (A) and liver (B) at original magnification, ×100
3. METHODS
Informed consent was received from the patient’s family. All specific biological explorations were conducted following the death of this patient, as VITT was not a clearly defined syndrome during the hospitalization of this patient in March 2021.
Serum samples were obtained from this patient at day 13 for exploration by platelet‐activation assays. We used a standard enzyme‐linked immunosorbent assay to detect IgG platelet factor 4 (PF4)–heparin antibodies (anti‐PF4/H). The presence of IgG anti‐PF4/H in the patient’s plasma was evaluated by ELISA (Zymutest HIA, Hyphen Biomed, Neuville‐sur‐Oise, France), according to the manufacturer’s protocol. An optical density (OD) of 0.5 was defined as the positive cutoff value. We evaluated platelet‐activating antibodies with the 14C‐serotonin release assay (SRA) as previously described.16, 17, 18 We also measured platelet‐activating antibodies by flow cytometric assay based on a platelet microvesicle assay (PMA), which detected the activation of washed platelets induced by activating heparin‐dependent antibodies.19 The PMA was performed as previously described by Maeda et al19 by the evaluation of platelet‐activating profiles, which detects the activation of washed platelets induced by activating antibodies in plasma or serum. In the SRA and PMA, platelet activation was challenged by an Fc‐receptor–blocking monoclonal antibody (anti‐CD32/FcγRIIA, clone IV.3; STEMCELL Technologies, Vancouver, BC, Canada) to confirm FcγRIIA engagement in platelets.12 Blood from healthy donors selected as platelet donors known to react well in the SRA was used, as previously suggested by Warkentin et al.18 These normal healthy donors had been free from aspirin and nonsteroidal anti‐inflammatory drugs for at least 10 days. The healthy donors were known to react well to SRA, and their washed platelets were used at a platelet concentration of ≈300 × 109/L. Twenty microliters of serum were incubated with the washed platelet solution (75 μL), with 5 μL of buffer alone, or with UFH at three different final concentrations of 0.1, 0.5, and 100 U/mL. The mixtures were placed in flat‐bottomed polystyrene microtiter wells and gently agitated for 1.5 hours at room temperature. After agitation, these samples were subjected to flow cytometric analysis with Navios (Beckman‐Coulter, Krefeld, Germany) to quantify the extent of washed‐platelet activation. A phycoerythrine‐labeled anti‐CD42b murine monoclonal antibody (SZ2, Beckman‐Coulter, Indianapolis, IN, USA) was used to label platelets and platelet‐derived microvesicles. We observed the distribution of CD42b‐positive events, which contained platelet‐derived microvesicles created by activating antibodies. The left upper quadrant corresponded to activated platelet and platelet‐derived microvesicles. The platelet activation index (iPA) was calculated as previously described.19 This iPA was obtained after sample analysis with flow cytometry to quantify the extent of washed‐platelet activation. iPA is defined as the ratio (percentage) of the number of fluorescent events in the left upper quadrant to the total number of fluorescent events in the left and right upper quadrants combined. In general, samples with an iPA of ≥1% at UFH 0.1 and/or 0.5 U/mL and <10% at UFH 100 U/mL, and <10% at UFH 0.1 U/mL with monoclonal antibody anti‐FcγRIIA were considered positive. A negative result was defined as an iPA of <10% under all reaction conditions, with an iPA of ≥10% induced by the weak positive control.
4. RESULTS AND DISCUSSION
At day 13 after ChAdOx1 nCov‐19 injection, our 62‐year‐old patient had high levels of IgG anti‐PF4/H, as indicated by strikingly high OD values (OD, 1.57) in serum from day 13. The functional activity of serum from this patient (patient A) was compared with that of serum from four other patients: (i) patient B: a 63‐year‐old woman with positive IgG anti‐PF4/H and negative SRA (HIT suspicion 5 days following mitral valve replacement complicated by acute kidney injury requiring renal replacement therapy and danaparoid substitutive therapy), used as negative control; (ii) patient C: a 71‐year‐old man with well‐known HIT, used as a positive control (positive IgG anti‐PF4/H and positive SRA); (iii) patient D: a 63‐year‐old man with bilateral subsegmental pulmonary embolism (PE) associated with a deep vein thrombosis treated by rivaroxaban without thrombocytopenia at day 11 following ChAdOx1 nCov‐19 injection and negative anti‐PF4/H; and (iv) patient E: a 76‐year‐old man with severe bilateral proximal PE requiring thrombolysis without thrombocytopenia at day 4 after BNT162b2 (comirnaty, Pfizer–BioNTech vaccine) injection and negative anti‐PF4/H.
Figure 2 shows the SRA profile of the suspected VITT patient (Figure 2A for patient A) and two controls of negative and positive HIT diagnosis with positive anti‐PF4/H (Figure 2B and 2C for patients B and C, respectively). Patient A showed increased reactivity with buffer control and UFH 0.1 and 0.5 U/mL. Platelet reactivity was blocked by UFH 100 U/mL and also when the blocking monoclonal antibody against FcγRIIa was added to UFH 0.1 U/mL, as for patient C with HIT (Figure 2C).
FIGURE 2.
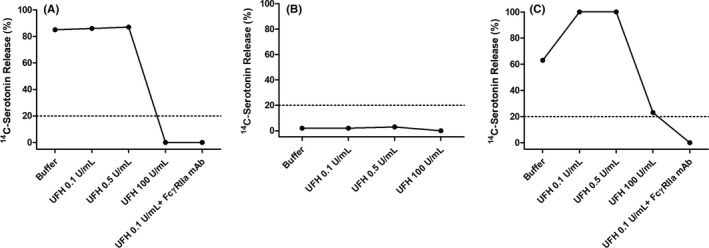
Functional testing by 14C‐serotonin release assay. (A) Male patient with cerebral venous sinus thrombosis and subarachnoid hemorrhage and thrombocytopenia following ChAdOx1 nCov‐19 injection (patient A). (B) Patient with suspected heparin‐induced thrombocytopenia with positive IgG anti–platelet factor 4–heparin antibodies (PF4/H) and negative serotonin release assay (SRA); used as negative control (patient D). (C) HIT‐diagnosed patient (positive IgG anti‐PF4/H and positive SRA), used as a positive control (patient E). UFH, unfractionated heparin
We confirmed with the PMA (Figure 3) that serum from patient A strongly activated platelets independently of classical doses of UFH (iPA = 83%, 89%, and 52%, respectively, at basal level, with 0.1 U/mL, and with 0.5 U/mL), in contrast to the patient with HIT (patient C; iPA = 5%, 32%, and 85%, respectively, at basal level, with 0.1 U/mL, and with 0.5 U/mL). This effect was completely inhibited by a high concentration of UFH or the addition of blocking monoclonal antibody against FcγRIIa to UFH 0.1 U/mL, as found with the serum from patient C. In contrast, thrombotic events without thrombocytopenia after ChAdOx1 nCov‐19 (patient D) or BNT162b2 (patient E) and in the HIT‐suspected patient with excluded diagnosis (patient B) all had an iPA <10%.
FIGURE 3.
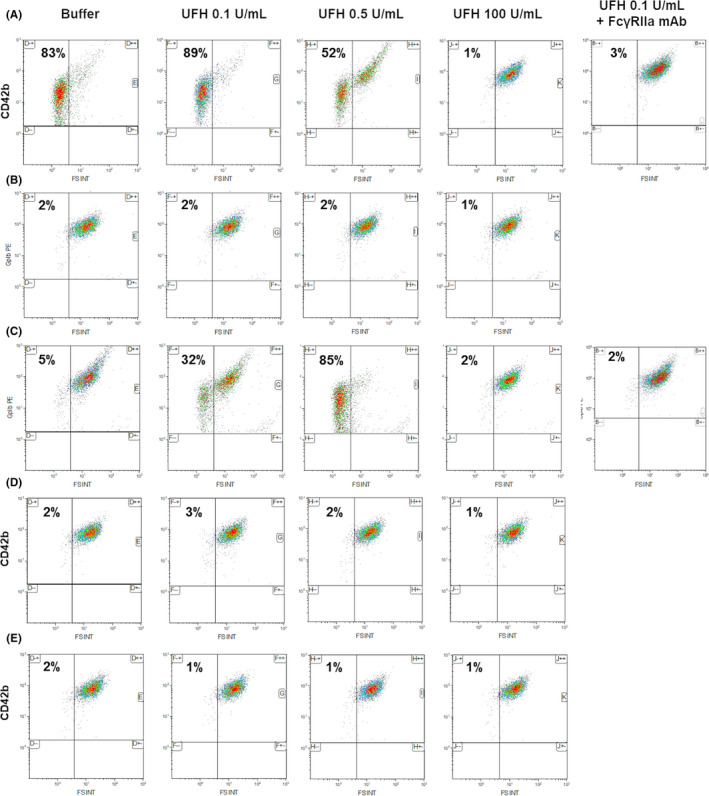
Functional testing by flow cytometry of platelet microvesicle assay (PMA) detecting. Washed platelets from one selected healthy donor were activated by serum containing or not activating antibodies and heparin at a therapeutic concentration. We observed the distribution of CD42b‐positive events, which contained platelet‐derived microvesicles created by activating antibodies. The platelet activation index result for each patient and condition is indicated in all upper left quadrant in this figure. (A) Male patient with cerebral venous sinus thrombosis and subarachnoid hemorrhage and thrombocytopenia following ChAdOx1 nCov‐19 injection. (B) Patient with suspected heparin‐induced thrombocytopenia with positive IgG anti‐PF4/H and negative serotonin release assay (SRA); used as negative control. (C) HIT‐diagnosed patient used as a positive control (positive IgG anti–platelet factor 4–heparin antibodies (PF4/H) and positive SRA), used as a positive control. (D) Patient with pulmonary embolism (PE) associated with a deep venous thrombosis without thrombocytopenia following ChAdOx1 nCov‐19 injection and negative IgG anti‐PF4/H. (E) Patient with PE without thrombocytopenia following BNT162b2 injection and negative IgG anti‐PF4/H. mAb, monoclonal antibody; UFH, unfractionated heparin
We present here one case of cerebral venous sinus thrombosis with subarachnoid hemorrhage and DIC with concomitant thrombocytopenia, which occurred 13days after ChAdOx1 nCov‐19 injection. At the time of the patient’s management, some cases of vaccine‐induced cerebral thrombosis had been discussed, but the relevance of anti‐PF4/H had yet been proposed. ISTH guidance on VITT was published on April 20, 2021,20 and cases studies were available in preprint from Andreas Greinacher on March 30, 2021, and on the New England Journal of Medicine website on April 9, 2021.12 The diagnosis of HIT was raised but was not retained in the absence of exposure to heparin during our patient’s hospitalization in March 2021. In light of the current data about VITT and recommendations, we confirm here with this case that platelet transfusions or heparin should not be recommended and can induce clinical deterioration.
High levels of IgG anti‐PF4/H were found to be associated with positivity of functional testing by flow cytometry evaluating activated‐platelet and platelet microvesicles on washed platelets, in contrast to a previous report using P‐selectin evaluation of platelets.14 In contrast to patients with classical HIT, the functional assays (PMA and SRA) of our patient were not dependent on low doses of UFH but were sensitive to inhibition by high‐dose UFH or by the Fc‐receptor–blocking monoclonal antibody. This patient received UFH therapy before tested blood samples were obtained. Even if we cannot rule out the presence of previous circulating complexes, containing antibodies bound to PF4 and heparin, the severe thrombocytopenia before UFH introduction allows us to be confident with our results. The results of the PMA suggest that the serum of this 62‐year‐old patient contained immune complexes with a specificity close to those classically observed in HIT. Our findings confirm that a syndrome similar to HIT may occur in a subgroup of patient’s following ChAdOx1 nCov‐19 injection. Strikingly, the case described here is a man >60 years, and, when associated with other cases described by Scully et al,14 we can probably affirm that VITT can be suspected in all ages, as described in reports from the Medicines and Healthcare Products Regulatory Agency in United Kingdom (report from May 13, 2021). VITT incidence appears to be probably very low, and the beneficial effect of anti–COVID‐19 vaccination fully outweighs this rare adverse effect.
We confirm here also the specificity of this anti‐PF4–related thrombosis by exploring IgG anti‐PF4/H in two other patients with venous thrombosis after COVID‐19 vaccination (one receiving the BNT162b2 and the other receiving the ChAdOx1 nCov‐19 injection). Of note, these two venous thrombotic events were not associated with thrombocytopenia. This association between thrombosis and immune thrombocytopenia was observed only with the ChAdOx1 nCov‐19 and Ad26.COV2.S COVID‐19 vaccines, whereas thrombotic events have been observed with all available vaccines in Europe, probably linked more to the population that the vaccination itself.11 In the SRA and PMA, platelet activation was inhibited by the Fc‐receptor–blocking monoclonal antibody (anti‐CD32), confirming the engagement of FcγRIIA in platelets and allowing us to confirm antibody‐dependent platelet activation and to exclude a nonspecific mechanism, such as inflammation.21
This case reports VITT in a man >60 years old and a flow cytometry assay using washed platelets derived from an HIT diagnostic functional assay. VITT is a very rare life‐threatening adverse event. Further studies are needed to investigate the prevalence of this coagulopathy associated with multiple thromboses after ChAdOx1 nCov‐19 or Ad26.COV2.S COVID‐19 vaccines and for other vaccines to decipher the particular populations that need to be vaccinated with mRNA or adenovirus vaccines.
RELATIONSHIP DISCLOSURE
None of these conflicts are related to this paper: RC received consultant fees from Aspen. DS received consultant fees, lecture fees, or travel awards from Aspen, Bayer, Carmat, Alliance BMS‐Pfizer, Léo Pharma, and Boehringer‐Ingelheim. OS received grants, personal fees, or nonfinancial support from Bayer, Alliance BMS‐Pfizer, Sanofi Aventis, Daiichi Sankyo, MSD, Boston Scientifics, and Chiesi. NG discloses consulting fees by Boehringer‐Ingelheim, Bayer, Bristol‐Myers Squibb/Pfizer, and Léo‐Pharma. The remaining authors report nothing to disclose.
AUTHOR CONTRIBUTIONS
AB and DMS wrote the paper and supervised the study. AB, DB, CJ, MM, BT, RC, and OS included and treated the patients. FB‐J, NG, DH, and DMS supervised the biological explorations. SB and Florence Desvard performed biological exploration.
ACKNOWLEDGEMENTS
We would like to acknowledge all nurses, technicians, and physicians involved in the clinical departments of the Annecy hospital and the George Pompidou European Hospital for their help in taking care of patients included here. We thank the unit of clinical research URC HEGP CIC‐EC1418 (Pauline Jouany and Dr Juliette Djadi‐Prat) for their involvement in SARCODO/COVID projects.
Bérezné A, Bougon D, Blanc‐Jouvan F, et al. Deterioration of vaccine‐induced immune thrombotic thrombocytopenia treated by heparin and platelet transfusion: Insight from functional cytometry and serotonin release assay. Res Pract Thromb Haemost. 2021;5:e12572. 10.1002/rth2.12572
Handling Editor: Pantep Angchaisuksiri.
Funding information
D.M. Smadja’s work was funded with grants from the French national agency for research ANR SARCODO (Fondation de France; NCT04624997).
Contributor Information
Nicolas Gendron, @gendronnico.
Benjamin Terrier, @terrierben.
David M. Smadja, Email: david.smadja@aphp.fr, @davidmsmadja.
REFERENCES
- 1.Debuc B, Smadja DM. Is COVID‐19 a new hematologic disease? Stem Cell Rev Rep. 2021;17(1):4‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Smadja DM, Guerin CL, Chocron R, et al. Angiopoietin‐2 as a marker of endothelial activation is a good predictor factor for intensive care unit admission of COVID‐19 patients. Angiogenesis. 2020;23(4):611‐620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Philippe A, Chocron R, Gendron N, et al. Circulating von Willebrand factor and high molecular weight multimers as markers of endothelial injury predict COVID‐19 in‐hospital mortality. Angiogenesis. 2021;24(3):505‐517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Khider L, Gendron N, Goudot G, et al. Curative anticoagulation prevents endothelial lesion in COVID‐19 patients. J Thromb Haemost. 2020;18(9):2391‐2399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chocron R, Galand V, Cellier J, et al. Anticoagulation prior to hospitalization is a potential protective factor for COVID‐19: insight from a French multicenter cohort study. J Am Heart Assoc. 2021:e018288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Libby P, Luscher T. COVID‐19 is, in the end, an endothelial disease. Eur Heart J. 2020;41(32):3038‐3044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Smadja DM, Philippe A, Bory O, et al. Placental growth factor level in plasma predicts COVID‐19 severity and in‐hospital mortality. J Thromb Haemost. 2021;19(7):1823‐1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid‐19 vaccine. N Engl J Med. 2020;383(27):2603‐2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA‐1273 SARS‐CoV‐2 vaccine. N Engl J Med. 2021;384(5):403‐416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV‐19 vaccine (AZD1222) against SARS‐CoV‐2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99‐111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Smadja DM, Yue Q‐Y, Chocron R, Sanchez O, Lillo‐Le Louet A. Vaccination against COVID‐19: insight from arterial and venous thrombosis occurrence using data from VigiBase. Eur Respir J. 2021;58(1):2100956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic thrombocytopenia after ChAdOx1 nCov‐19 Vaccination. N Engl J Med. 2021;384(22):2092‐2101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schultz NH, Sorvoll IH, Michelsen AE, et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV‐19 vaccination. N Engl J Med. 2021;384(22):2124‐2130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Scully M, Singh D, Lown R, et al. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV‐19 vaccination. N Engl J Med. 2021;384(23):2202‐2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Muir KL, Kallam A, Koepsell SA, Gundabolu K. Thrombotic thrombocytopenia after Ad26.COV2.S vaccination. N Engl J Med. 2021;384(20):1964‐1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gruel Y, Vayne C, Rollin J, et al. Comparative analysis of a French prospective series of 144 patients with heparin‐induced thrombocytopenia (FRIGTIH) and the literature. Thromb Haemost. 2020;120(7):1096‐1107. [DOI] [PubMed] [Google Scholar]
- 17.Lillo‐Le Louet A, Boutouyrie P, Alhenc‐Gelas M, et al. Diagnostic score for heparin‐induced thrombocytopenia after cardiopulmonary bypass. J Thromb Haemost. 2004;2(11):1882‐1888. [DOI] [PubMed] [Google Scholar]
- 18.Warkentin TE, Arnold DM, Nazi I, Kelton JG. The platelet serotonin‐release assay. Am J Hematol. 2015;90(6):564‐572. [DOI] [PubMed] [Google Scholar]
- 19.Maeda T, Nakagawa K, Murata K, et al. Identifying patients at high risk of heparin‐induced thrombocytopenia‐associated thrombosis with a platelet activation assay using flow cytometry. Thromb Haemost. 2017;117(1):127‐138. [DOI] [PubMed] [Google Scholar]
- 20.Nazy I, Sachs UJ, Arnold DM, et al. Recommendations for the clinical and laboratory diagnosis of VITT against COVID‐19: Communication from the ISTH SSC Subcommittee on Platelet Immunology. J Thromb Haemost. 2021;19(6):1585‐1588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stokes KY, Granger DN. Platelets: a critical link between inflammation and microvascular dysfunction. J Physiol. 2012;590(5):1023‐1034. [DOI] [PMC free article] [PubMed] [Google Scholar]


