Abstract
Lipids, which along with carbohydrates and proteins are among the most important nutrients for the living organism, have a variety of biological functions that can be applied widely in biomedicine. A fatty acid, the most fundamental biological lipid, may be classified by length of its aliphatic chain, and the short-, medium-, and long-chain fatty acids and each have distinct biological activities with therapeutic relevance. For example, short-chain fatty acids have immune regulatory activities and could be useful against autoimmune disease; medium-chain fatty acids generate ketogenic metabolites and may be used to control seizure; and some metabolites oxidized from long-chain fatty acids could be used to treat metabolic disorders. Glycerolipids play important roles in pathological environments, such as those of cancers or metabolic disorders, and thus are regarded as a potential therapeutic target. Phospholipids represent the main building unit of the plasma membrane of cells, and play key roles in cellular signaling. Due to their physical properties, glycerophospholipids are frequently used as pharmaceutical ingredients, in addition to being potential novel drug targets for treating disease. Sphingolipids, which comprise another component of the plasma membrane, have their own distinct biological functions and have been investigated in nanotechnological applications such as drug delivery systems. Saccharolipids, which are derived from bacteria, have endotoxin effects that stimulate the immune system. Chemically modified saccharolipids might be useful for cancer immunotherapy or as vaccine adjuvants. This review will address the important biological function of several key lipids and offer critical insights into their potential therapeutic applications.
Keywords: Lipids, Fatty acids, Glycerolipids, Phospholipids, Biomedical applications
INTRODUCTION
Lipids are chemically defined as hydrophobic molecules that are insoluble in water but soluble in apolar solvents. In the biological context, lipids are essential molecules that function as the major structural components of living cells and play important roles in various cellular functions (Fahy et al., 2011). For many years, lipids were known only as energy-storing molecules and the building blocks of cellular membranes. However, with the progress of advanced analytical techniques and bioassays, additional classes and functions of lipids are increasingly coming to light. Today, lipids are known to be involved in numerous cellular signaling pathways, including those that regulate cell proliferation, survival, apoptosis, and cell metabolism (Alabdulkarim et al., 2012). Due to their excellent biocompatibility, lipids are also used as efficient and safe carriers for drug delivery in various formulations (Yang and Merlin, 2020).
Lipids are classified not only as hydrophobic compounds but also as amphiphilic molecules with both hydrophilic and hydrophobic properties. This property of lipid molecules means that they arrange themselves in the biophysical environment as bilayer structures (also called “micelles”) wherein the hydrophobic part is sequestered to the interior and the hydrophilic part is exposed to the aqueous environment. This self-organization determines the morphology and functions of lipids in living organisms. For example, bilayers of phospholipids comprise the cellular and subcellular membranes of living cells. Their amphiphilic properties govern their interactions with transmembrane proteins and modulate their activity (Watson, 2015). Lipids may also be located in droplets stored within cells (Welte and Gould, 2017) or circulate in blood as lipoprotein complexes (Fernandez et al., 2013). Glycolipids play important roles in the immune system (Zhang et al., 2019a).
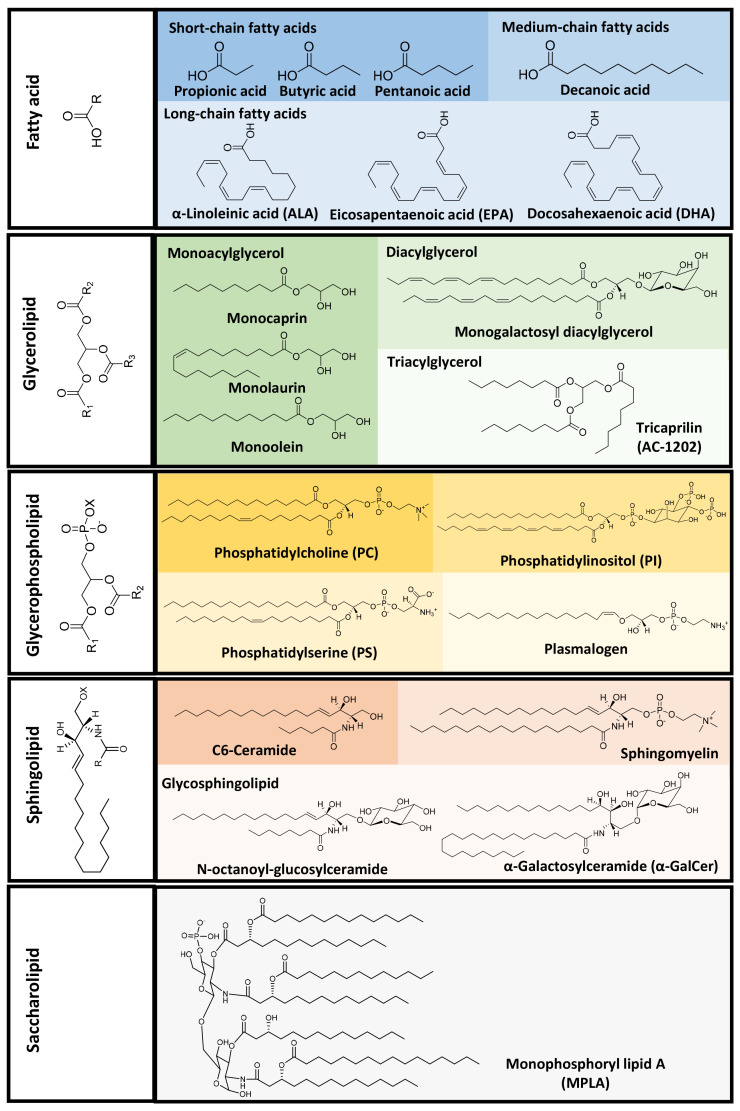
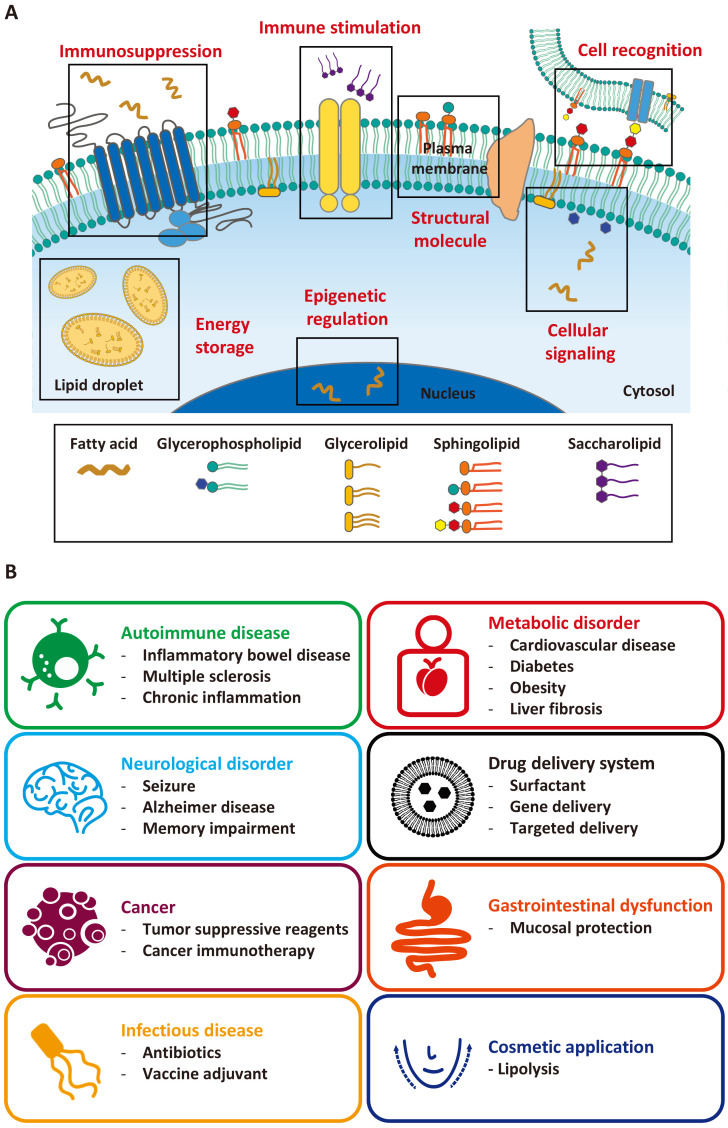
Dysregulated lipid levels have been reported in various diseases, such as cancer, obesity, diabetes, and cardiovascular, autoimmune, neurodegenerative diseases (Leuti et al., 2020). This, combined with their important roles in various biological conditions, suggests that lipids might be potential therapeutic targets in disease treatment. In this review, we categorize lipids based on their structures and relevant biological activities (Fig. 1), update and summarize the recent applications of bioactive lipids in disease treatment and biomedicine (Fig. 2), and discuss nano/micro formulations that have improved the therapeutic effects of bioactive lipids.
Fig. 1.
Structure-based classification of lipids and representative lipid molecules for biomedical application.
Fig. 2.
Biomedical activities and applications of lipids. (A) Lipids play a wide range of biological roles in regulating cell homeostasis through various processes, including cell structure formation, energy storage, and cellular signaling. (B) Biomedical applications of lipids in human medicine.
FATTY ACIDS
Fatty acids are hydrocarbon derivatives that have a carboxylic acid with a long aliphatic chain (Kimura et al., 2020). Generally, naturally occurring aliphatic chains of fatty acids have 4-28 carbon atoms connected by single or double bonds. They can be classified as saturated fatty acids, which have only single carbon-carbon bonds in the aliphatic chain, or unsaturated fatty acids, which have one or more double bonds in the aliphatic chain (Currie et al., 2013). Examples of biomedical applications of fatty acids are listed in Table 1.
Table 1.
Biomedical activity and application of fatty acids
| Class | Name | Activity | Application | Ref. |
|---|---|---|---|---|
| Short-chain fatty acids | Propionic acid | Immunomodulatory effect on mature dendritic cells | Anti-inflammatory activity | Nastasi et al., 2015 |
| Enhance regulatory T cell metabolism and function | Multiple sclerosis | Duscha et al., 2020 | ||
| Alteration of bone marrow hematopoiesis | Allergic airway disease | Trompette et al., 2014 | ||
| Butyric acid | Microencapsulated butyric acid for regulation of microbiota (ButyRose) | Inflammatory bowel disease | Facchin et al., 2020 | |
| Enhanced epithelial barrier function through IL-10 receptor | Inflammatory bowel disease | Zheng et al., 2017 | ||
| Alleviates mouse colitis by regulating gut microbiota dysbiosis | Inflammatory bowel disease | Dou et al., 2020 | ||
| Regulation of intestinal homeostasis through actin-associated protein synaptopodin | Inflammatory bowel disease | Wang et al., 2020 | ||
| Intestinal macrophage regulation via histone deacetylase inhibition | Inflammatory bowel disease | Chang et al., 2014 | ||
| Pentanoic acid | Inducing IL-10 production in lymphocytes by reprogramming their metabolic activity | Experimental autoimmune encephalomyelitis | Luu et al., 2019 | |
| Medium-chain fatty acids | Decanoic acid | Synergistic action against AMPA receptors and seizures with perampanel | Seizure control | Augustin et al., 2018a |
| Inhibition of glutamate-induced currents derived from various types of AMPA receptor | Seizure control | Chang et al., 2016 | ||
| Long-chain fatty acids | α-linolenic acid | Promoting bioconversion and subsequent oxylipin formation | High fat diet induced Insulin resistance | Fan et al., 2020 |
| Enhanced production of oxylipins in M1 macrophage | Anti-inflammatory activity | Pauls et al., 2018 | ||
| Activation of alternatively activated macrophages via oxylipin profile change | Anti-inflammatory activity | Pauls et al., 2020 | ||
| Modulation of phagocytosis and endosomal pathways of extracellular Tau in microglia | Alzheimer’s disease | Desale and Chinnathambi, 2021 | ||
| Alleviates dextran sulfate sodium-induced ulcerative colitis in mice | Inflammatory bowel disease | Kim et al., 2020 | ||
| Eicosapentaenoic acid | Improvement of hepatic metabolism and reduces inflammation in mice and HepG2 cells | Nonalcoholic fatty liver disease | Albracht-Schulte et al., 2019 | |
| Inhibition NLRP3 inflammasome activation | Acute cerebral infarction (ACI) | Mo et al., 2020 | ||
| Docosahexaenoic acid | Enhanced M2 macrophage polarization via the p38 signaling pathway and autophagy | Chronic inflammation | Kawano et al., 2019 | |
| Modulate monocyte inflammatory response | Chronic inflammation | So et al., 2021 |
Short-chain fatty acids
Short-chain fatty acids have aliphatic chains with fewer than six carbon atoms and play important roles in immune cell activity and modulation of G protein-coupled receptor signaling (Kim, 2021; Moniri and Farah, 2021). Short-chain fatty acids are produced by the anaerobic fermentation of dietary fibers in the intestine (Koh et al., 2016). They exert their biological activity via inhibition of histone deacetylase (HDAC); this activity is not only confined to the intestinal area but is also detected in the blood, leading to systemic effects (Parada Venegas et al., 2019). Gut microbiota-derived short-chain fatty acids, such as acetic acid (C2:0), propionic acid (C3:0), and butyric acid (C4:0), are key immune system-regulating metabolites and their dysfunction is related to inflammatory disease (Lavelle and Sokol, 2020).
Propionic acid which is a major short-chain fatty acid produced from gut microbiota fermentation, protects against allergic inflammation in the lung (Trompette et al., 2014). Propionic acid acts as a ligand of G protein-coupled receptor 41 (GPR41) and G protein-coupled receptor 43 (GPR43) (Husted et al., 2017). An immunomodulatory effect of propionic acid was also observed in human cells. Based on the bioactivity of propionic acid, it has been studied for the treatment of autoimmune diseases, such as multiple sclerosis (Duscha et al., 2020). After supplementation of propionic acid for 14 days, the gut microbiome of multiple sclerosis patients was altered and the imbalance between regulatory T cells (Tregs) and helper T cell type 17 (Th17) in the central nervous system was improved. As a result, the subcortical gray matter of the patients was increased. These findings support the potential clinical application of propionic acid.
Butyric acid is another widely studied short-chain fatty acid. Similar to propionic acid, butyric acid shows anti-inflammatory activity. It plays important roles in maintaining the homeostasis of commensal bacteria through the regulation of intestinal macrophages (Chang et al., 2014). Butyric acid was reported to induce anti-inflammatory characteristics in bone marrow-derived macrophages and colonic lamina propria macrophages. Mechanistically, butyric acid reduced the lipopolysaccharide (LPS) stimulation-induced secretions of nitric oxide, tumor necrosis factor-α (TNF-α), and IL-6 through histone deacetylase inhibition, but not via the G-protein coupled receptor. Despite these anti-inflammatory effects, however, butyric acid treatment did not ameliorate colitis in dextran sulfate sodium (DSS)-treated model mice due to a lack of sufficient production of protective cytokines, such as IL-6.
Other studies have shown that butyric acid may have therapeutic potential for inflammatory bowel disease due to its ability to modulate epithelial function or gut microbiota (Zheng et al., 2017; Dou et al., 2020; Facchin et al., 2020; Wang et al., 2020). Butyric acid, but not other short-chain fatty acids, up-regulated the actin-binding protein, synaptopodin, at the intestinal epithelial tight junction, which is important for epithelial barrier function and wound healing. Moreover, butyric acid supplementation alleviated the phenotypes in synaptopodin-deleted mice experiencing DSS-induced colitis (Wang et al., 2020). In terms of modulating the gut microbiota, orally administered sodium butyric acid showed therapeutic efficacy in the DSS-induced colitis mouse model and increased the populations of Lachnoclostridium and Lachnospiraceae (Dou et al., 2020), and administration of butyric acid was reported to change the gut-microbiota environment in inflammatory bowel disease patients (Facchin et al., 2020). In the latter work, oral administration of microencapsulated sodium butyric acid improved the gut microbiota composition of ulcerative colitis and Crohn’s disease patients, as evidenced by alterations in short-chain fatty acid-producing microorganisms (Lachnospiraceae spp. and Butyricicoccus, respectively). Sodium butyric acid also reportedly improved the quality of life among ulcerative colitis patients.
Pentanoic acid (C5:0, also called valeric acid) has also been reported as an immunoregulatory short-chain fatty acid that might be a potential therapeutic for autoimmune disease (Luu et al., 2019). Pentanoate elevated the glucose oxidation of lymphocytes via mTOR activation to suppress IL-17A expression and enhance IL-10 expression. Consequently, in experimental colitis and multiple sclerosis mouse models, pentanoate ameliorated the disease via regulatory B cell-mediated protection.
Medium-chain fatty acids
Medium-chain fatty acids have six to 12 carbon atoms in their aliphatic chains; they represent a unique form of dietary fat and show variable biological functions (Kimura et al., 2020). Physicochemical features of fatty acids have been reported to be affected by the hydrocarbon tail lengths (Ramirez et al., 2001; Kimura et al., 2020). The decrease of the hydrocarbon tail lengths is known to increase the water solubility of fatty acids. Thus, compared to long-chain fatty acids, medium-chain fatty acids show increased water solubility, resulting in higher oral absorption (Chen et al., 2021). Medium-chain fatty acids are transported directly to the liver without requiring the carnitine carrier system, and are therefore rapidly metabolized and converted to fuel. After medium-chain fatty acids are degraded via β-oxidation, they produce large amounts of acetyl-CoA and ketone bodies, such as acetone, acetoacetate and β-hydroxybutyric acid (McGarry and Foster, 1980).
Ketone bodies are utilized as an energy source for the heart, brain, and skeletal muscles. They also have other biological functions: For example, ketone bodies reportedly control the electrical activity of neurons by regulating transporters (e.g., vesicular glutamate transporters) and ion channels (e.g., ATP-sensitive K+ channels and voltage-dependent Ca2+ channels) (Juge et al., 2010; Tanner et al., 2011; Kadowaki et al., 2017). In the therapeutic context, ketone bodies and ketone body-generating medium-chain fatty acids may affect neurological diseases, and a medium-chain triglyceride-based ketogenic diet has been applied for seizure control since 1920s (Warren et al., 2018).
Although seizure control requires ketone bodies originating from medium-chain fatty acids, several studies found that medium-chain fatty acids themselves might directly control neuronal activity (Sada and Inoue, 2018). Decanoic acid (C10:0), which is a monocarboxylic saturated fatty acid with 10 carbon atoms, inhibited the activation of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor subunits stimulated by glutamate-induced currents, showing a greater impact on the GluA2/3 subunit versus the GluA1 or GluA1/2 subunits (Chang et al., 2016). Decanoic acid also showed a synergistic interaction with the current epilepsy drug, perampanel, in antagonizing AMPA receptors and treating seizures (Augustin et al., 2018a).
Long-chain fatty acids
Long-chain fatty acids have 12 or more carbon atoms in their aliphatic chains; they are generally synthesized in animal or plant cells and generate energy via their β-oxidation-mediated degradation to acetyl-CoA. Even-number saturated fatty acids, such as palmitic acid (C16:0) and stearic acid (C18:0), are the predominant fatty acids used for energy storage in animal tissues (Kimura et al., 2020). However, some unsaturated long-chain fatty acids have other biological functions, as well as pharmacological activities that may be useful for therapeutic applications (Valenzuela et al., 2021).
Polyunsaturated long-chain fatty acids are particularly important signaling molecules for physiological homeostasis. Polyunsaturated long-chain fatty acids can be classified based on the position of their double bond (Simopoulos, 2016). Omega-6 fatty acids, which have a double bond located at six carbon atoms away from the terminal methyl end of their backbone, are synthesized from linoleic acid (LA, C18:2n-6), which is an essential fatty acid that must be consumed from food as human body cannot synthesize (Simopoulos, 2016). Omega-3 fatty acids have a double bond located at three carbon atoms away from the terminal methyl end of their aliphatic chain, and are synthesized from the essential fatty acid, α-linolenic acid (ALA, C18:n-3). Omega-6 fatty acids generally show pro-inflammatory activity, while omega-3 fatty acids show anti-inflammatory activity (Serhan et al., 2008; Serhan and Chiang, 2013). As a result, omega-3 fatty acids can show beneficial effects in chronic inflammatory disease or metabolic disorders (Valenzuela et al., 2021). GRP40 and GRP120 have been identified as receptors of omega-3 and omega-6 fatty acids (Oh et al., 2010).
ALA is considered to have therapeutic potential. It is a precursor of oxylipins, such as eicosapentaenoic acid (C20:5n-3) and docosahexaenoic acid (C22:6n-22), which are oxygenated metabolites that have anti-inflammatory activities (Serhan et al., 2008; Serhan and Chiang, 2013). At the cellular level, ALA has been reported to regulate macrophages via oxylipin production, as evidenced by reduced LPS-triggered secretion of IL-1β, IL-6, and TNF-α by macrophages and increased phagocytic activity and altered production of the chemokine, monocyte chemoattractant protein-1, by M2-like cells (Pauls et al., 2018, 2020). ALA-induced enhancement of migratory ability increases phagocytosis and endosomal processing of extracellular Tau in microglia (Desale and Chinnathambi, 2021). ALA has shown anti-inflammatory activities in animal models of metabolic or inflammatory diseases. For example, ingestion of ALA-enriched butter elevated the blood level of eicosapentaenoic acid and suppressed the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), and these changes were associated with improvement of insulin resistance and tissue inflammation in high fat diet-fed mice (Fan et al., 2020). ALA treatment also halted colon contraction and improved clinical symptoms in the DSS-induced colitis mouse model (Kim et al., 2020).
Eicosapentaenoic acid and docosahexaenoic acid-derived mediators (such as resolvins, protectins, and maresins) can contribute to the resolution of inflammatory conditions and are thus termed “pro-resolving” mediators (Serhan et al., 2008; Bäck et al., 2019). Eicosapentaenoic acid is oxidized to E-series resolvins (RvE1, RvE2, and RvE3) by cyclooxygenase and lipoxygenase. RvE1 and RvE2 are endogenous antagonists of the leukotriene B4 (LTB4) receptor, BLT-1, and can thereby block the chemotaxis of polymorphous-nuclear neutrophils and activation of NF-κB. These resolvins also activate ChemR23/ERV, which regulates pro-inflammatory cytokine production (Spite et al., 2014). Eicosapentaenoic acid was further reported to exert anti-inflammatory activity by inhibiting nucleotide binding oligomerization domain-like receptor pyrin domain-containing 3 (NLRP3) and to show therapeutic efficacy in acute cerebral infarction or nonalcoholic fatty liver disease (Albracht-Schulte et al., 2019; Mo et al., 2020). Docosahexaenoic acid may be metabolized to D-series resolvins (RvD1-6), protectins (PD1/NPD1), maresins, and RvD1, which shares working mechanisms similar to those of RvE1 and RvE2 (Serhan and Chiang, 2013). Similar to eicosapentaenoic acid, docosahexaenoic acid has been demonstrated to act as an anti-inflammatory molecule that regulates macrophages and monocytes (Kawano et al., 2019; So et al., 2021).
GLYCEROLIPIDS
A glycerolipid is classified as a monoacylglycerol (MAG), diacylglycerol, or triacylglycerol (TAG) based on how many ester bonds are formed between the fatty acid with the glycerol backbone. Glycerolipids can be found in the membranes of prokaryotes and eukaryotes and in animal fat tissues, and are usually considered to act as an energy reservoir. The hydrolysis of the ester linkage between glycerol and fatty acid is the initiating step of fat metabolism. Many types of glycerolipids are found in human tissues, with some having been shown to have therapeutic effects (Table 2).
Table 2.
Biomedical activity and application of glycerolipids
| Subclass | Name | Activity | Medical application | Ref. |
|---|---|---|---|---|
| Monoacylglycerol | Monocaprylate | Formation of transient or permanent pores in the membrane | Antimicrobial | Hyldgaard et al., 2012 |
| MonolaurinMoncaprin | Bactericidal activity against H. pylori | Infectious disease | Bergsson et al., 2002 | |
| Monoolein | Antimicrobial activity against S. aureus and E. coli | Antimicrobial | Jumina et al., 2019 | |
| MAG-DHA | Downregulation of NF-kB and upregulation of PTEN | Non-small cell lung cancer | Morin and Fortin, 2017 | |
| Diacylglycerol | Monogalactosyl diacylglycerol | Inhibition of CD31 positive tumor blood vessel growth. | Colon cancer | Maeda et al., 2013 |
| 1,3-Diacylglycerol | Increasing oxidation of fatty acid and suppressing chylomicron formation | Obesity and insulin resistance | Saito et al., 2007 | |
| Triacylglycerol | Medium-chain triglyceride | Suppression of IL-6, iNOS, cyclooxygenase-2 and IL-10 and activation of NF-kB and p38 MAPK pathways | Insulin resistance | Geng et al., 2016 |
| Medium-chain triglyceride | Metabolized to ketone bodies to the brain | Neurological and metabolic disorders | Augustin et al., 2018b | |
| Tricaprilin (AC-1202) | Reducing oxidative damage and improvement of respiration rate in brain | Alzheimer’s disease | Henderson et al., 2009 |
Monoacylglycerols
Monoacylglycerols, which can exhibit different forms depending on the site at which the fatty acids form ester bonds with a hydroxyl group of glycerol, are generated by enzymatic hydrolysis of triacylglycerols or diacylglycerols. Monoacylglycerols and diacylglycerols are often used as food emulsifiers. Integration of a monoacylglycerol into the membrane of microbial cells is a common means to disrupt the membrane or induce cell lysis (Hyldgaard et al., 2012). The electron transport chain and oxidative phosphorylation play important roles in energy production for bacteria. Monoacylglycerols disrupt the electron transport chain by binding to electron carriers and block oxidative phosphorylation by reducing the membrane potential and proton gradient. Among the monoacylglycerols, monocaprin and monolaurin showed the strongest bactericidal effects on Helicobacter pylori (H. pylori) (Bergsson et al., 2002).
Diacylglycerols
There are two forms of diacylglycerol: 1,2-diacylglycerols and 1,3-diacylglycerols. Diacylglycerols are used commercially as surfactants and fat emulsifiers and, within the cell, act as intermediates and cellular messengers. Phosphatidylinositol-4,5-bisphospate (PI(4,5)P2) bound to the cellular membrane can be hydrolyzed into diacylglycerol, and this plasma membrane-localized diacylglycerol can activate protein kinase C. In T cells, this activation stimulates transcription factors, such as NF-κB and AP-1, to activate T cell receptor. Diacylglycerols also play important roles in activating immune cells, and several studies have tried to utilize diacylglycerol for immunotherapy. For example, researchers inhibited diacylglycerol kinase (DGK), which is the negative regulator of diacylglycerol-mediated signaling, to enhance the anti-cancer effect of natural killer (NK) cells. The same authors also observed that knocking out DGKζ in NK cells enhanced downstream receptor activation (Yang et al., 2016). Another study found that inhibition of DGK amplified the diacylglycerol signaling cascade of T cells (Arranz-Nicolás et al., 2018).
Chimeric antigen receptor-T cells showed higher expression of DGKα/ζ compared to spleen-derived wild type cells, which has been proposed as a strategy to advance chimeric antigen receptor-T cell immunotherapy (Moon et al., 2014). Oral consumption of spinach-derived monogalactosyl diacylglycerol showed a tumor-suppressive effect in a murine model: Colon tumor (Colon 26)-inoculated groups fed with 4 mg/kg or 20 mg/kg exhibited decreases in the tumor load compared with the vehicle control (Maeda et al., 2013). Diacylglycerols have also shown promising therapeutic effects against metabolic disorders. High fat diet-induced obese and brown adipose tissue-deficient mice were fed with diacylglycerol oil or triacylglycerol oil for 15 weeks. Mice fed diacylglycerol oil showed a slight decrease in body weight but an increase in glucose intolerance, compared to the triacylglycerol oil-fed group (Saito et al., 2007). Finally, a study showed that accumulated hepatic diacylglycerol activated PKCε, which phosphorylated insulin receptor kinase at T1160 to inhibit IRK activation and cause insulin resistance (Lyu et al., 2018). However, the importance of diacylglycerol in lipid-induced hepatic insulin resistance remains controversial.
Triacylglycerols
Triacylglycerols are tri-esters composed of glycerol bound to three fatty acid molecules, and can be subcategorized into saturated and unsaturated groups based on the presence of double bonds in the fatty acid chain. They are commonly used as an index for metabolic syndrome. Orally applied medium-chain triglycerides have been examined as potential therapeutics against metabolic disorders and inflammation. Inflammatory signals are known to activate JNK and IKK and target insulin receptor substrate to inhibit the insulin signaling cascade. Oral application of medium-chain triglycerides was found to alleviate insulin resistance in high fat diet-induced obese mice by reducing the inflammatory response in obesity. Mice fed with medium-chain triglycerides also gained less body weight, and eventually lost up to 15% of their body weight (Geng et al., 2016).
The triglyceride-glucose index is used as a marker for symptomatic coronary artery disease and atherosclerosis (Da Silva et al., 2019); high triglyceride-indexed patients tend to have artery stiffness and coronary artery calcification (Won et al., 2018). A medium-chain triglyceride-based ketogenic diet is becoming a prevalent trend for treating metabolic disorders. Moreover, recent studies have focused on applying medium-chain triglycerides to treat Alzheimer’s disease. The brain uses two main types of fuel in its metabolic pathways: ketone and glucose. At an early stage of Alzheimer’s disease, cerebral glucose hypometabolism is observed, and cells metabolize ketones rather than glucose.
An orally dosed triacylglyceride is hydrolyzed into medium-chain fatty acids by lipases in the gastrointestinal tract; these fatty acids are absorbed and metabolized through β-oxidation, leading to the production of ketone bodies (β-hydroxybutyric acid, acetoacetate, and acetone) (Augustin et al., 2018b). Different kinds of triacylglycerols are undergoing clinical trials in Alzheimer’s disease patients. One example is the orally administrable ketogenic compound, AC-1202, which is a medium-chain triglyceride composed of glycerin and caprylic acid. AC-1202 was shown to increase the serum level of the ketone body, β-hydroxybutyric acid, and significantly increase the cognitive subscale of the Alzheimer’s Disease Assessment Scale compared with placebo (Henderson et al., 2009).
GLYCEROPHOSPHOLIPIDS
Glycerophospholipids and phospholipids are the main lipid constituents of the eukaryotic cellular membrane. In general, phospholipids are amphiphilic molecules with structures containing a polar portion and a non-polar portion (van Meer et al., 2008). Glycerophospholipids share the basic structure of a glycerol backbone conjugated with hydrophobic acyl chains, plus a phosphate group and a hydroxyl group. The chemical structures of phospholipids vary based on the head polar groups, the properties (number, length, and saturation) of the hydrophobic acyl chains, and the type of bond formed between the hydrophobic chain and the glycerol (Li et al., 2015).
Phospholipids can be grouped into five classes based on the polar head group: phosphatidylcholine, phosphatidylethanolamine, phosphatidylglycerol, phosphatidylinositol, and phosphatidylserine (Harayama and Riezman, 2018). The molecular species in each class vary relative to the acyl chains at the sn1 and sn2 positions. In general, a saturated or monosaturated fatty acid is located at the sn1 position and a polyunsaturated fatty acid with a longer acyl chain is located at the sn2 position (Zarringhalam et al., 2012).
Phospholipids are among the most basic but important compounds that contribute to life activities. They are found ubiquitously in humans, animals, plants, and bacteria, and are considered to be the bare bones of cellular and sub-cellular membranes, in which they assemble as a bilayer (van Meer et al., 2008). Phospholipids contribute to the formation of circulating lipoproteins, which act as carriers to transport hydrophobic triglycerides and cholesterol through the bloodstream (Boffa and Koschinsky, 2019). Phospholipids also act as a natural emulsifier in the body, such as by emulsifying fat-soluble substances in the presence of cholesterol and bile acid in the gallbladder, to facilitate the absorption of these substances (Ko et al., 2020). They function as pulmonary surfactants to reduce the surface tension of the air/liquid interface in tissues, such as the pleura and alveoli of the lung (Agassandian and Mallampalli, 2013). Finally, phospholipids may act as signaling mediators for many important cellular functions (van Meer et al., 2008). Examples of bioactive glycerophospholipids are listed in Table 3.
Table 3.
Biomedical activity and application of glycerophospholipids
| Subclass | Name | Activity | Medical application | Ref. |
|---|---|---|---|---|
| PC | PC | Lipolysis in human fat tissue | Cosmetic procedure | Thomas et al., 2016 |
| Lipolysis in malar, jawline, submental areas and upper arm | Cosmetic procedure | Thomas et al., 2018 | ||
| Promote adipocyte-specific lipolysis and apoptosis through TNF-α or IL-1β signaling | Adipocyte lipolysis and apoptosis | Jung et al., 2019 | ||
| Reduce gastric bleeding, prevent GI permeability disturbance due to LPS | Gastrointestinal tract inflammation | Dial et al., 2008 | ||
| Improves intestinal barrier function. | Drug-induced Liver injury | Chen et al., 2019 | ||
| Polyenylphosphatidylcholine | Protective effects against lipid peroxidation, oxidative stress and hepatic fibrosis | Fatty liver, metabolic comorbidities | Maev et al., 2020 | |
| PI | Phosphatidylinositol (3, 4, 5) – trisphosphate (PI(3,4,5)P3) |
Signaling molecule processed by phosphatidylinositol 3-kinase | Insulin resistance | Kachko et al., 2015 |
| PS | PS | Suppress the TNF-α production of peritoneal macrophage | Inflammatory disease | Klein et al., 2020 |
| Interfere with the pro-inflammatory cytokine formation of phagocyte | Inflammatory disease | Szondy et al., 2017 | ||
| Lecithin-transphosphatidylated PS | Restored age-associated decreases of choline transporter | Memory impairment | Lee et al., 2015 | |
| Plasmalogen | Ethanolamine plasmalogen | Correlation of declined circulating plasmalogen and cognitive function | Alzheimer’s disease | Wood et al., 2010 |
| Enhanced inhibition activity of phosphatidylethanolamine on γ-secretase | Alzheimer’s disease | Onodera et al., 2015 | ||
| Improvement of cognitive functions | Alzheimer’s disease | Fujino et al., 2017 | ||
| Improved memory impairment in amyloid β-infused rats | Alzheimer’s disease | Yamashita et al., 2017 |
Phosphatidylcholine (PC)
PC is a main component of lecithin. Classified as glycerophospholipid, PC is composed of choline as its polar head, fatty acids as its hydrophobic domains, and glycerol as their connector. The fatty acids of PC generally include one mono-unsaturated fatty acid and one saturated fatty acid. PC dominates among the phospholipids that comprise the cell membrane, particularly in the exoplasmic leaflet of the plasma membrane (Harayama and Riezman, 2018). PC has excellent biocompatibility and safety, as it originated from nature, and has long been approved as a major ingredient in various commercial liposome formulations (Barenholz, 2012). Beyond its use as a component of lipid-based carriers, PC can also perform potential therapeutic activities, such as inducing lipolysis, reducing inflammatory responses, and protecting the liver from drug-induced injury (Jung et al., 2019).
PC has been used to induce lipolysis in adipose tissues, such as via injection in cosmetic surgery (Jung et al., 2018). The stored triglycerides released from lysed adipocytes are subsequently solubilized and intracellular lipases are activated. A mixture of PC and sodium deoxycholate has been used worldwide for many years as an off-label strategy to reduce the cosmetic volume of subcutaneous fat tissues in the clinic. Currently, only deoxycholate is FDA-approved for this purpose; it is a component of Kybella (Allergan, Irvine, CA, USA), the subcutaneous injection of which is used to improve the appearance of moderate to severe convexity or fullness associated with submental fat in adults.
Although not yet approved by FDA, PC has been proven to be effective in lipolysis in animals and humans when combined with deoxycholate. It has been shown to promote lipolysis effects with fewer side effects than deoxycholate alone (Salti et al., 2008), and local application of a PC/deoxycholate cocktail has been repeatedly shown to reduce unwanted fat with improved effects at the face (malar, jawline, and submental areas) and upper arm and average effects at the thighs and knees (Thomas et al., 2016, 2018).
PC treatment was also reported to improve intestinal barrier functions. PC is an important surface-active compound that generates the hydrophobic biophysical surface property of the intestine, thereby protecting against microorganisms and toxic byproducts. Orally administered PC (100 mg/kg) was found to protect the intestinal mucosa from LPS-induced damage and potentially prevent LPS-induced gastric bleeding (Dial et al., 2008). The study revealed that PC acted directly in the mucosa to prevent LPS from disturbing the permeability of the gastrointestinal tract.
Patients with liver cirrhosis or hepatocarcinoma show considerable increases of lysophosphatidylcholine in the fecal, which indicates the reduction of PC in the intestinal lumina (Huang et al., 2013). A recent study reported that oral PC improved liver function following CCl4-induced injury (Chen et al., 2019). Oral PC could be directly absorbed into the mucus layer, thereby restores the intestinal barrier function, triggers reduction of the portal pressure and improves the liver function. In clinical trials, polyenylphosphatidylcholine, an unsaturated fatty acid PC, is used as an adjunctive therapy in improving the liver function of patients with nonalcoholic fatty liver disease and metabolic comorbidities (Maev et al., 2020). The protective effect of polyenylphosphatidylcholine against nonalcoholic fatty liver disease suggests its function in suppressing lipid peroxidation, oxidative stress, and hepatic fibrosis.
Phosphatidylinositol (PI)
PI is a low-abundance phospholipid that constitutes 5-8% of the total phospholipid component of the cell membrane. It shares some structural similarity with the major phospholipids, but is distinguished by the presence of an inositol sugar moiety at the polar head. The mammalian PI derivatives tend to have stearic acid (C18:0) or arachidonic acid (C20:4) as the fatty acid chains linked to their glycerol backbone. Among them, phosphatidylinositol-4,5-diphosphate (PI(4,5)P2) is the most abundant phosphoinositide, accounting for 1-2% of total lipids. It regulates various aspects of cellular function, including cytoskeletal organization, membrane trafficking, and ion channels (Schink et al., 2016). PI(3,4,5)P3, which is mainly generated by phosphorylation of PI(4,5)P2 via phosphatidylinositol 3-kinase, plays important roles as a signal mediator for various cellular functions, such as cell motility, cell metabolism, cell survival, and gene regulation (Czech, 2003; Marat and Haucke, 2016).
PI products have also been shown to trigger downstream signals and induce translocation of the insulin-sensitive glucose transporter, GLUT4, from internal membrane pools to the plasma membrane in muscle and fat cells (Draznin, 2006; Bridges and Saltiel, 2015). Therefore, PI(3,4,5)P3 could be helpful in relieving insulin resistance. Indeed, intracellular delivery of PI(3,4,5)P3 using polymeric nanoparticles was shown to overcome insulin resistance. The researchers condensed polyethyleneimine (PEI) 25 kDa and PI(3,4,5)P3 into PEI-25/PIP3 nanoparticles whose cationic charge (derived from PEI25) caused PI(3,4,5)P3 to be retained at the cell perimeter. This strategy was thought to mimic the extent and cellular organization of PI(3,4,5)P3, which is preferable for insulin stimulation (Kachko et al., 2015).
Phosphatidylserine (PS)
PS is an anionic phospholipid found abundantly in the inner leaflet of eukaryotic membranes. Structurally, PS consists of a glycerol backbone, two fatty acid chains of variable length and saturation, and a phosphate group conjugated with a polar serine moiety. When cells undergo apoptosis, the asymmetric distribution of PS across the lipid bilayer is disrupted by the scramblase enzyme, which is activated during apoptosis. PS is exposed in the outer leaflet, where it is recognized by phagocytes as an “eat me” signal.
Phagocytosis of apoptotic cells is triggered by the interaction of PS with PS-specific receptors, such as stabilin-2, brain-specific angiogenesis inhibitor 1 (BAI1), and members of the T-cell immunoglobulin and mucin (TIM) family. The phagocytosis of PS-exposing cells is also mediated by bridging molecules, such as milk fat globulin-EGF-factor 8 and growth arrest-specific gene 6, which bind to PS-exposing cells and bridge the interaction of phagocytes and apoptotic cells. Both interaction pathways generate an “eat me” signal to phagocytes, resulting in the clearance and removal of apoptotic cells. This particular bioactivity of PS has been recognized as a major feature of immunotherapy and has been applied in recent studies.
In order to promote drug delivery into activated microglia, researchers prepared PS-microbubbles by mixing PS with 2-distearoyl-sn-glycero-3-phosphocholine (DSPC), cholesterol, 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethyleneglycol)-2000] (DSPE-PEG2000), and 1,2-dipalmitoyl-sn-glycero-3-phospho-L-serine (Zhao et al., 2018). The PS-decorated microbubbles were specifically taken up by microglia in inflammatory areas of a model of cerebral ischemia. The uptake of PS-microbubbles by macrophages was double that seen for control-microbubbles. Following intravenous administration of PS-microbubbles, ultrasound was used to decompose the particles from micro- to nano-sized, to facilitate penetration of the blood-brain barrier. In a similar study, acetylated dextran nanoparticles were coated with 1, 2-dipalmitoyl-sn-glycero-3-phospho-L-serine to promote the uptake of curcumin by macrophages (Shah et al., 2019). PS-coated dextran nanoparticles were taken up at a 4-fold higher rate by RAW 264.7 macrophages compared to control A549 lung cancer cells.
PS delivered in a micelle formulation was recently shown to stimulate the polarization of macrophages from the pro-inflammatory to anti-inflammatory phenotype (Klein et al., 2020). This was consistent with previous reports associating PS treatment with down-regulation of pro-inflammatory cytokines (e.g., TNF-α and IL-6) and increased secretion of anti-inflammatory mediators (e.g., IL-10 and TGF-β) (Szondy et al., 2017). Indeed, micelles enriched with 1,2-dioleoyl-sn-glycero-3-phospho-L-serine (DOPS) significantly reduced the release of TNF-α by LPS-activated macrophages, indicating an anti-inflammatory effect (Klein et al., 2020).
Oral administration of PS was found to improve memory impairment in aged rats (Lee et al., 2015). A dose of 20 mg/kg PS was associated with recovery of age-related decreases in the mRNA levels of choline transporter and muscarinic acetylcholine receptor type 1, and a dose of 50 mg/kg yielded significant memory improvements. This suggests that PS may improve age-related memory deficits and thus could hold promise for use in other diseases associated with neurodegeneration.
Plasmalogen
Plasmalogen is a major component of the membrane phospholipids found in the brain, kidneys, lungs, and skeletal muscles; it accounts for 18-20% of all phospholipids (Braverman and Moser, 2012). Structurally, plasmalogen consists of a glycerol backbone, a vinyl ether linkage at sn1, a fatty acid ester linkage at sn2, and a phosphate group at sn3 that is tethered with either ethanol amine (to yield ethanolamine plasmalogen) or choline (to yield choline plasmalogen). Ethanolamine plasmalogen is widely distributed in the brain, while choline plasmalogen is found more in heart muscle. Plasmalogen has attracted recent interest because it appears to play important roles in various diseases, such as Alzheimer’s disease, cardiovascular disease, cancer, and respiratory disease (Braverman and Moser, 2012; Sutter et al., 2016; Messias et al., 2018).
Increasing evidence suggests that ethanolamine plasmalogen is deficient in Alzheimer’s disease: in Alzheimer’s disease patients, the ethanolamine plasmalogen level was found to be reduced in post-mortem brain samples as well as in cerebrospinal fluid, plasma, serum, and red blood cells. Compared to heathy brain tissues, ethanolamine plasmalogen was reduced by 70% in the brains of Alzheimer’s patients (Wood et al., 2010; Onodera et al., 2015). It is unclear whether ethanolamine plasmalogen deficiency is a cause or a result of Alzheimer’s disease, but studies have shown that increasing plasma levels of ethanolamine plasmalogen can improve disease symptoms (Fujino et al., 2017; Yamashita et al., 2017). In a study on amyloid β-infused rats, oral administration of ethanolamine plasmalogen at 209 μmol/kg body weight/day for 4 weeks improved the working memory-related learning ability in tested animals (Yamashita et al., 2017). In a recent human trial, oral administration of ethanolamine plasmalogen at a dose of 1 mg/day for 24 weeks significantly improved memory among female patients with Alzheimer’s disease and those under the age of 77 (Fujino et al., 2017).
SPHINGOLIPIDS
Sphingolipids, which are a main membrane lipid, have one polar head and two aliphatic chains, and play important roles in cell recognition and signaling (Hannun and Obeid, 2008). Sphingolipids are composed of a sphingoid base having an amino alcohol group; other components are connected to a sphingoid base through amide, glycoside, or phosphodiester bonds (Hannun and Obeid, 2018).
The addition of head groups to ceramide produces different types of sphingolipids, such as sphingomyelins, cerebrosides, and gangliosides (Hannun and Obeid, 2018). Sphingomyelins consist of a ceramide backbone and phosphocholine or phosphoethanolamine, and thus resemble phospholipids. They are also an important component of the cell membrane, especially that of the myelin sheath that covers neuronal axons (Slotte, 2013). The 1-hydroxyl group of ceramides can be conjugated with carbohydrate residues by β-glycoside linkage to generate glycosphingolipids, such as cerebrosides, globosides, and gangliosides (Zhang et al., 2019a).
Cerebrosides have a ceramide backbone with a single sugar, such as glucose or galactose, and are commonly called glucocerebrosides (glucosylceramides) or galactocerebrosides (galactosylceramides), respectively. Generally, galactocerebrosides are abundant in neural tissue, while glucocerebrosides are found in other tissues. Gangliosides are glycosphingolipids with a ceramide backbone with an oligosaccharide that must contain at least one sialic acid residue; this provides a negative charge at physiological pH that distinguishes gangliosides from cerebrosides and globosides, which have neutral charges (Cutillo et al., 2020). Gangliosides are present in the outer membrane of cells and are primarily responsible for recognizing signaling molecules (Cutillo et al., 2020).
Sphingolipids have been reported to have multiple biological functions. In cellular level, they are involved in providing the structural integrity of cell membrane, cell recognition, cell adhesion, endoplasmic reticulum stress, and controlling the function of cilia and microvilli (Park and Park, 2020). In central nervous system, sphingolipids are known to be players of brain development (Dasgupta and Ray, 2017). In gastrointestinal tract, sphingolipids play a role in regulating oral absorption of some nutrients (Kurek et al., 2013). In immune systems, sphingolipids have been reported to function in the differentiation, maturation, and immune responses of lymphocytes including B cells, and T cells (Zhang et al., 2020).
Due to the various biological functions, sphingolipids and their derivatives have been studied for therapeutic applications of diverse diseases such as cancer, and autoimmune diseases (Table 4). In cancer immunotherapy, targeting specific types of glycosphingolipids has been shown to modulate the immune microenvironments of tumors (Yu et al., 2020). Safingol and fingolimod are examples of sphinolipid analogues for clinical applications (Choi and Song, 2020). Safingol can exert anticancer effect by suppressing the activity of sphingosine kinases. Fingolimod is clinically used for treatment of multiple sclerosis. Fingolimod is an analogue of the sphingolipids that modulates the sphingosine-1-phosphate receptor 1. Phosphorylated fingolimod is reported to internalize the receptors and block the migration of lymphocytes to the central nervous system (Brinkmann et al., 2010).
Table 4.
Biomedical activity and application of sphingolipids
| Subclass | Name | Activity | Medical application | Ref. |
|---|---|---|---|---|
| Ceramide | C6 ceramide | Increasing vincristine sensitivity via AMP activated protein kinase–p53 signaling | Colon cancer, pancreatic cancer | Chen et al., 2015 |
| Oxidized graphene nanoparticles as a delivery system for the pro-apoptotic C6 ceramide | Cervical cancer | Suhrland et al., 2019 | ||
| Graphene oxide nanoparticle delivery system enhance anti-cancer activity of C6 ceramide | Hepatocellular carcinoma | Wang et al., 2019 | ||
| Liposomal short-chain C6 ceramide induces potent anti-osteosarcoma activity | Osteosarcoma | Zhai et al., 2015 | ||
| Sphingo-myelin | Sphingomyelin | Sphingomyelin-based nanosystems (SNs) for the anticancer miRNA therapeutics | Colon cancer | Nagachinta et al., 2020 |
| Site-activated drug releasing lipid-iron nanoparticle | Cancer | Medina et al., 2020 | ||
| Engineered liposomes sequester bacterial exotoxins and protect from invasive infections | Antibiotics | Henry et al., 2015 | ||
| Specific drug release lipid nanoparticles at infection sites | Antibiotics | Zhang et al., 2021 | ||
| Glyco-sphingolipid | N-octanoyl-glucosylceramide | Improvement of doxorubicin delivery and efficacy in solid tumors | Squamous cell carcinoma, Melanoma | van Lummel et al., 2011 |
| Improvement of efficacy of liposomal doxorubicin | Lobular breast carcinoma | van Hell et al., 2013 | ||
| Formation of transient micro-channels within the cell membrane | Uterine sarcoma | Zalba et al., 2020 | ||
| α-Galactosyl-ceramide (α-GalCer) | Antigen-presenting cell immunotherapy by the activation of endogenous iNKT cells | Non-small cell lung cancer | Toyoda et al., 2020 | |
| mRNA nanovaccine with α-GalCer induces antitumor immunity by NKT cells | Melanoma | Verbeke et al., 2019 |
Ceramides
A ceramide is composed of a sphingoid base and a fatty acid (Cha et al., 2016). Ceramides can be synthesized from L-serine and palmitoyl-CoA via the de novo pathway, from sphingomyelins via the sphingomyelinase pathway, and from complex sphingolipids via the salvage pathway (Simon et al., 2021). Generally, ceramides are associated with cell death, differentiation, proliferation, and inflammation (Turpin-Nolan and Brüning, 2020; Zhang et al., 2020).
Lipid-based nano-carrier systems, such as liposomes, are one of the promising candidates that have been studied for their potential to improve the physicochemical limitations of ceramides. For example, C6 ceramide encapsulated liposomes are known to induce caspase-mediated apoptosis more potently in osteosarcoma cells than free C6 ceramide, thereby providing better safety against non-cancerous bone cells. Moreover, administration of liposomal C6 ceramide with methotrexate had synergistic effects compared to monotherapy (Zhai et al., 2015). However, although liposome formulations can provide improved physical properties and efficacy for C6 ceramide, several drawbacks, such as poor loading efficiency due to other lipids and cofactors, limit their use (Khazanov et al., 2008).
Graphene nanosheets can also be used to deliver lipids attached via hydrophobic interactions (Shim et al., 2016). Graphene nanosheets and oxidized graphene nanoribbons have been studied for the delivery of C6 ceramide. Mass spectrometry showed that the loading efficiencies of C6 ceramide onto the graphene nanosheets and oxidized graphene nanoribbons were 51.5% and 57%, respectively, and these formulations were found to reduce the viability of HeLa cells through caspase-3 dependent apoptosis (Suhrland et al., 2019). Additionally, modified C6 ceramide-loaded graphene nanosheets showed promising anti-cancer effects in a hepatocellular carcinoma model. The graphene oxide nanosheets were conjugated with polyethyleneglycol and PEI to enhance cellular uptake via a clathrin-mediated mechanism. The surface-modified nanosheets inhibited tumor growth and improved the survival duration in a hepatocellular carcinoma xenografts mouse model, and exhibited synergistic effects with sorafenib (Wang et al., 2019).
Sphingomyelin
Sphingomyelin is a major structural lipid that contributes to forming the plasma membranes of mammalian cells (van Meer et al., 2008). Sphingomyelin has phosphocholine or phosphoethanolamine and thus shares physicochemical properties with phospholipids such as PC (Slotte, 2013). Similar to PC and other phospholipids, sphingomyelin provides high bioavailability for liposomal drug delivery systems. A sphingomyelin-based nanosystem was recently investigated for miRNA delivery (Nagachinta et al., 2020). The lipid formulations comprised vitamin E, stearylamine or N-[1-(2,3-dioleoyloxy)propyl]-N,N,N-trimethylammonium methylsulfate (DOTAP), PEG12-C18, and sphingomyelin.
Two strategies for loading miRNA145 were investigated: attaching the miRNA to the surface of a lipid nanoparticle composed of stearylamine and sphingomyelin; and encapsulating a lipid complex composed of miRNA and DOTAP inside the sphingomyelin lipid layer (SNs-Lpx). The generated sphingomyelin-based miRNA-encapsulated lipid nanoparticles showed higher cellular uptake, transfection efficiency, and anticancer effects compared to miRNA-tethered liposomes composed of stearylamine and sphingomyelin. This likely reflects that the encapsulation of miRNA inside the sphingomyelin lipid layer avoided its premature release (Nagachinta et al., 2020).
Sphingomyelin has also been included in environmentally reactive drug delivery systems developed using enzymes that are induced by specific environmental cues. The members of the acid sphingomyelinase family of sphingomyelinases are activated by oxidative stress or ionizing radiation (Paris et al., 2001) and function to convert sphingomyelins to ceramide (Ogretmen, 2017). Iron nanoparticle-containing sphingomyelin lipid nanoparticles have been developed for site-specific drug release (Medina et al., 2020). In this study, the lipid nanoparticles were composed of DSPC, DOTAP, cholesterol, and sphingomyelin. The particles were designed to be broken down by acid sphingomyelinase-secretory cells, such as those represented by the cisplatin-exposed SCC-9 cell line (Lacour et al., 2004), and could be remotely localized to the tumor site by altering magnetic field (AMF), due to the presence of iron nanoparticles in the lipid layer. In the orthotropic SCC-9 tumor bearing mouse model, lipid-iron nanoparticles were specifically delivered to tumor tissue, where they released indocyanine green and cisplatin for diagnostic and therapeutic purposes, respectively (Medina et al., 2020).
Pore-forming toxins originating from bacteria have the ability to form membrane pores in the host cell membrane (Parker and Feil, 2005). Cholesterol-dependent cytolysis involving the actions of the pore-forming toxins such as pneumolysin, streptolysin O, tetanolysin, and α-hemolysin is important for the progression of infectious diseases. Thus, cholesterol and sphingomyelin-based liposomes are being investigated as a means to attenuate these toxins by mimicking the mammalian cell membrane (Henry et al., 2015). Cholesterol-rich sphingomyelin liposomes were generated and found to competitively prevent the binding of pneumolysin, α-hemolysin, and phospholipase C. In other work, liposomes were used to protect mice from sepsis caused by Staphylococcus aureus (S. aureus) and Streptococcus pneumoniae (S. pneumoniae) and prevent pneumococcal pneumonia (Henry et al., 2015).
Pore-forming toxins also recognize certain lipids on the surface of the host membrane; these include sphingomyelin, which is well represented on animal cell membranes (Parker and Feil, 2005). As a result, sphingomyelin liposomes that respond to pore-forming toxins and release encapsulated antibiotics were developed as a means to combat infectious diseases. These liposomes comprised unsaturated fatty acid-bound phospholipids that provide fluidity to the membrane, and encapsulated cholesterol, sphingomyelin, and vancomycin inside the hydrophilic core. The liposomal vancomycin was released in the α-hemolysin rich environment, and its antibacterial efficacy was demonstrated in both in vitro and in vivo models (Zhang et al., 2021).
Glycosphingolipids
Cerebroside is a ceramide that binds to a single sugar molecule, usually glucose or galactose (Zhang et al., 2019a). Glucosylceramides (or glucosylcerebrosides), especially those conjugated with a short-chain fatty acid, have been studied to control cell plasma membrane fluidity. N-octanoyl-glucosylceramide was found to cause incomplete lipid packing and enhance the cellular uptake of amphiphilic drugs, such as doxorubicin (van Lummel et al., 2011). This short-chain glucosylceramide generated transient membrane channels to mediate the translocation of doxorubicin, and thus improving its efficacy (van Hell et al., 2013). This strategy may be a promising solution for multi-drug resistant (MDR) cancer therapy. Doxorubicin-resistant cancer cells have relatively rigid lipid membranes compared to those of doxorubicin-sensitive cancer cells, and this rigidity can interfere with drug absorption. As a result, the use of N-octanoyl-glucosylceramide modified liposomes loaded with doxorubicin yielded improved doxorubicin accumulation in tumor tissues compared to a conventional liposome formulation (Zalba et al., 2020).
Glycosphingolipids, including cerebroside, play important roles in the immune system. Immune cells have their own distinct glycosphingolipid expression patterns in the cell membrane, and these patterns are associated with cell proliferation and/or differentiation. For example, glycosphingolipids are essential components of glycosphingolipid-enriched microdomains (GEMs) and control cell activity (Zhang et al., 2019a). α-Galactosylceramide (α-GalCer), which is composed of α-linked galactose and ceramide, is a lipid antigen for invariant natural killer T cells (iNKT cells). α-GalCer is recognized by the invariant T cell receptor (invTCR) of iNKT cells via the MHC class I-like molecule, CD1d; this triggers the iNKT cells to release Th1 and Th2 cytokines, which activate leukocytes, such as dendritic cells and T cells (Zhang et al., 2019b). Therefore, α-GalCer has been investigated for cancer immunotherapy through iNKT cell activation (Toyoda et al., 2020). In a study on an mRNA vaccine nanoparticle formulation, α-GalCer was used to target NKT cells and DOTAP provided cationic properties for mRNA loading. The α-GalCer-based nanoparticles were found to activate both innate and adaptive immunity and exhibit improved anticancer efficacy (Verbeke et al., 2019).
The lack of satisfactory results in clinical studies using α-GalCer alone underlined the need to develop stronger immune-activating reagents, prompting researchers to develop α-GalCer analogs for cancer immunotherapy and test vaccine adjuvants, such as 7DW8-5 (Li et al., 2010). Indeed, 7DW8-5-encapsulating liposome therapy showed synergistic effects with CAR-T cell therapy in solid tumors. As a glycolipid adjuvant, 7DW8-5 was entrapped inside the liposome with a PI3K inhibitor intended to recover the immunosuppressive tumor microenvironment, and the particle was modified with the tumor-targeting peptide, iRGD (Zhang et al., 2018). 7DW8-5 has also been applied to manufacture influenza vaccines and strengthen the production of influenza virus-specific antibodies in mouse models (Feng et al., 2019).
SACCHAROLIPIDS
Saccharolipids have a structure in which fatty acids are linked to the sugar backbone. The different types of saccharolipids are classified by the different structures formed by the sugar moiety, and there are two main subclasses: acylaminosugars and acyltrehaloses. Examples of biomedical applications of saccharolipids are listed in Table 5.
Table 5.
Biomedical activity and application of saccharolipids
| Subclass | Name | Activity | Medical application | Ref. |
|---|---|---|---|---|
| Lipid A | LPS | Upregulation of pro-inflammatory cytokines | Wound healing | Kostarnoy et al., 2013 |
| Generate tolerogenic dendritic cells to exhaust effector CD4 T cells | Experimental autoimmune encephalomyelitis | Zhou et al., 2014 | ||
| Increase endogenous TNF-α expression | Cancer | Goto et al., 1996 | ||
| Upregulation of MHC molecule on GSCs | Cancer | Han et al., 2017 | ||
| Monophosphoryl lipid A | TLR4 agonist | Vaccine adjuvant | Saito et al., 2010 | |
| Enhancing CD4 T cell activation and initial clonal expansion | Vaccine adjuvant | Thompson et al., 2005 |
Lipopolysaccharide (LPS)
LPS consists of a lipid and O-antigen-containing polysaccharide. It is also known as endotoxin, as it is a toxin located within bacterial cells. Lipid A, which is an acylaminosugar present in gram-negative bacteria, is a component of LPS, along with O-antigen and oligosaccharides. The structure of LPS allows bacteria to maintain the integrity of the cell wall, to which it is tethered by lipid A. When the cell wall is decomposed, lipid A is released from the bacterial wall to exert a toxic effect on the human body. The complement cascade is initiated by C3a and C5a in macrophages, which causes the release of histamine and triggers an inflammatory response via the release of TNF, cytokines, interleukins, and prostaglandins. Topical administration of bacterial LPS was used to treat skin wounds in mice. It promoted inflammation resolution and macrophage infiltration, thereby enhancing collagen synthesis. Administration at the wound site increased the levels of the CC chemokines (CCL2/monocyte chemoattractant protein-1, CCL7/MCP-3, CCL3/MIP-1α, and CCL5/RANTES), which are associated with macrophage infiltration, and increased the collagen synthesis-related cytokines, TGF-β1 and fibroblast growth factor 2 (FGF2) (Kostarnoy et al., 2013).
LPS treatment generated apoptotic dendritic cells, which triggered the generation of tolerant dendritic cells to deplete effector CD4+ T cells in an autoimmune encephalomyelitis murine model, supporting the potential of LPS-induced dendritic cell therapy for multiple sclerosis (Zhou et al., 2014). There is controversy over the use of LPS in antitumor cancer therapy. LPS treatment was reported to confer antitumor effects by upregulating endogenous TNF, and LPS combined with cyclophosphamide synergistically increased the antitumor effect of TNF (Goto et al., 1996). LPS-pretreated glioma stem-like cells (GSCs) showed upregulation of MHC molecules, and LPS-pretreated GSC-inoculated rats had a longer mean survival time compared to those treated with control GSC (67.0 ± 31.8 days vs. 16.0 ± 2.5 days, respectively) (Han et al., 2017). A high dose of LPS in colorectal cancer was found to interfere with the effect of anti-programmed death-ligand 1 monoclonal antibody (anti-PD-L1 mAb) treatment. However, LPS-targeting fusion protein-loaded nanoparticles were found to improve colorectal cancer immunotherapy outcomes (Song et al., 2018).
LPS, which is derived from Gram-negative bacteria, is often heterogeneous. Differences in the lengths and compositions of the terminal glycan chains may lead to different experimental consequences, such as in the context of Toll-like receptor 4 (TLR4). To address this issue, researchers developed di[3-deoxy-D-manno-octulosonyl]-lipid A (Kdo2-Lipid A), which is a homologous subclass of LPS that exhibits endotoxin activity similar to LPS and is highly selective for TLR4. Kdo2-Lipid A outperforms LPS in yielding highly reproducible results with 10-fold higher endotoxin activity (Saito et al., 2010).
Several limitations have been proposed for LPS-based immunotherapy, including the development of LPS resistance, the existence of sensitivity differences between in vivo mouse models and humans, and the risk of adverse effects such as systemic inflammatory response syndrome, generalized Schwartzman reaction, hepatotoxicity, and Arthus reaction (Shetab Boushehri and Lamprecht, 2019). Various efforts have been made to overcome these issues, such as developing lipid A moieties to reduce the risk of side effects or resistance. Among them, tolerin and SDZ MRL953 could prevent sepsis, ONO-4007 showed tumor regression, and monophosphoryl lipid A was used as cancer vaccine adjuvant (Rockwell et al., 2010). Other efforts include the development of controlled drug-delivery strategies, such as by engineering sustained release of LPS from nanoparticles, the use of microneedles for passive targeting of antigen-presenting cells, and targeting of intracellular LPS to downregulate unwanted pro-inflammatory pathways (Shetab Boushehri et al., 2019).
Monophosphoryl lipid A
LPS has been excluded from clinical trials due to its high toxicity, but monophosphoryl lipids exhibit far less toxicity. Monophosphoryl lipid A is a low-toxicity derivative of LPS that can be used as a TLR4 agonist and to promote CD4+ T cell activation. In an early study, monophosphoryl lipid A was emulsified in water with 1,2-dipalmitoyl-sn-glycero-3-phosphocholine and administered intravenously, and this formulation triggered CD4+ T cell activation and initial clonal expansion comparable to that obtained with LPS, but with lower toxicity (Thompson et al., 2005). Since then, monophosphoryl lipid A has been approved as a vaccine adjuvant in liposome formulations (Bashiri et al., 2020).
CURRENT CHALLENGES AND PERSPECTIVES
The successful use of lipids in preventing and treating chronic diseases has been increasingly demonstrated in recent studies. Their therapeutic effect is seen both as monotherapy and as an adjuvant in combination with other drugs or substances (MacLeod et al., 2011). Due to the special physicochemical properties of lipids, including their low water solubility, high oil-water partition coefficient, high melting point, and crystal state at room temperature, therapeutic lipids face issues with their formulation, dosing, absorption and stability (McClements, 2010). Moreover, lipids are susceptible to oxidation over time, which can reduce their therapeutic activity and/or produce toxic by-products (Khanum and Thevanayagam, 2017). To overcome these problems, several strategies have been studied to extend the application and efficacy of bioactive lipids, as described in the following.
Fatty acids are mostly administered by the oral route (N’Goma et al., 2012). Because fatty acids are not freely dispersed in the aqueous phase, they tend to form large fat globules in the gastrointestinal tract. The enzymatic de-esterification of these lipids occurs at the water-oil interface. Therefore, the smaller the lipid droplets, the faster they hydrolyze and higher the bioavailability (Maki and Dicklin, 2019). Of note, the absorption of these lipids is highly dependent on dietary fat, which activates the release of bile acids to emulsify the consumed lipids. Therefore, high intra-individual variation in bioavailability has been a challenge in clinical trials (Bisschop et al., 2004). Efforts have been made to reduce the size of lipid droplets in the gastrointestinal tract using surfactants or emulsifiers. For example, omega-3 ethyl ester exhibited improved bioavailability when combined with a non-ionic surfactant mixture consisting of polysorbate 80 and polyoxyethylene-polyoxypropylene block copolymer (Lopez-Toledano et al., 2019).
Another problem with lipid formulations is the risk of lipid peroxidation, which occurs by via pathways in bulk lipid materials and emulsification systems. Lipid peroxidation, which is generally caused by free radicals or enzymes (Khanum and Thevanayagam, 2017), generates secondary oxidation products, such as aldehydes, ketones, alcohols, hydrocarbons, organic acids, and epoxy compounds (Vieira et al., 2017). These substances potentially cause oxidative stress and damage to the human body (Ramana et al., 2013). To prevent peroxidation, many strategies have been proposed for the design and preparation of lipid products. During the preparation phase, lipid peroxidation may be reduced by carefully controlling the materials and manufacturing conditions, such as by using high-purity ingredients, monitoring the pH levels during formulation and storage, and reducing or removing trace metals. During the design phase, lipid peroxidation can be reduced by including antioxidants, such as ascorbic acid, α-tocopherol, propyl gallate, or ascorbyl palmitate, in the aqueous phase (Khanum and Thevanayagam, 2017).
Many lipids have been approved for human use in both clinical trials and commercial products. They come in a variety of pharmaceutical forms or supplement products. In the context of drug delivery systems, lipids are used in a variety of nano- and micro-formulations, such as liposomes, micelles, and solid lipid nanoparticles. In addition to natural lipids, synthetic lipids are also used in specific applications, such as gene transfer. mRNA vaccines are being increasingly investigated in lipid-based formulations, due to the many reports indicating the safety and efficacy of lipid-based delivery systems. Several lipid nanoparticle-based SARS-CoV-2 mRNA vaccines have been approved for human use. For example, lipid nanoparticles made of the proprietary ionic lipid SM-102, polyethyleneglycol 2000, dimyristoyl glycerol, cholesterol, and DSPC were developed by Moderna (Cambridge, MA, USA) and achieved a vaccine effect of 94.5% against SARS-CoV-2. BioNTech SE (Mainz, Germany) developed another effective lipid nanoparticle candidate. Formulated with the lipids, ((4-hydroxybutyl)azanediyl) bis(hexane-6,1-diyl)bis(2-hexyldecanoate), 2 [(polyethylene glycol)-2000]-N,N-ditetradecylacetamide, 1,2-distearoyl-sn-glycero-3-phosphocholine, and cholesterol, it showed promising protective effects (Milane and Amiji, 2021). Additional lipid nanoparticle formulations are currently being developed for mRNA vaccine delivery in other diseases, such as cancer and infectious diseases. (Miao et al., 2019; Zhang et al., 2019c).
CONCLUSION
New lipid species and their biological functions in pathology are increasingly being elucidated. Lipids show potential as new drug candidates and components of lipid-based nanomedicines. With the increased interest in lipid-based nanomaterials for delivering genes and vaccines, the study of functional lipids will continue to grow. The potential therapeutic applications of lipids have been supported in various diseases, emphasizing the importance of lipids in biomedicine. Although issues with the physical stability and formulation needs of lipid-based drugs still remain as challenges, continued advances in basic science are expected to facilitate the discovery of new species and roles of lipid in the future, further extending the application of lipids in efforts to treat and/or prevent various diseases.
ACKNOWLEDGMENTS
This research was supported by grants from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT, Republic of Korea (NRF-2021R1A2B5B03002123; NRF-2018R1A5A2024425).
REFERENCES
- Agassandian M., Mallampalli R. K. Surfactant phospholipid metabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids. 2013;1831:612–625. doi: 10.1016/j.bbalip.2012.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alabdulkarim B., Bakeet Z. A. N., Arzoo S. Role of some functional lipids in preventing diseases and promoting health. J. King Saud Univ. Sci. 2012;24:319–329. doi: 10.1016/j.jksus.2012.03.001. [DOI] [Google Scholar]
- Albracht-Schulte K., Gonzalez S., Jackson A., Wilson S., Ramalingam L., Kalupahana N. S., Moustaid-Moussa N. Eicosapentaenoic acid improves hepatic metabolism and reduces inflammation independent of obesity in high-fat-fed mice and in HepG2 cells. Nutrients. 2019;11:599. doi: 10.3390/nu11030599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arranz-Nicolás J., Ogando J., Soutar D., Arcos-Pérez R., Meraviglia-Crivelli D., Mañes S., Mérida I., Ávila-Flores A. Diacylglycerol kinase α inactivation is an integral component of the costimulatory pathway that amplifies TCR signals. Cancer Immunol. Immunother. 2018;67:965–980. doi: 10.1007/s00262-018-2154-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augustin K., Khabbush A., Williams S., Eaton S., Orford M., Cross J. H., Heales S. J. R., Walker M. C., Williams R. S. B. Mechanisms of action for the medium-chain triglyceride ketogenic diet in neurological and metabolic disorders. Lancet Neurol. 2018a;17:84–93. doi: 10.1016/S1474-4422(17)30408-8. [DOI] [PubMed] [Google Scholar]
- Augustin K., Williams S., Cunningham M., Devlin A. M., Friedrich M., Jayasekera A., Hussain M. A., Holliman D., Mitchell P., Jenkins A., Chen P. E., Walker M. C., Williams R. S. B. Perampanel and decanoic acid show synergistic action against AMPA receptors and seizures. Epilepsia. 2018b;59:e172–e178. doi: 10.1111/epi.14578. [DOI] [PubMed] [Google Scholar]
- Bäck M., Yurdagul A., Tabas I., Öörni K., Kovanen P. K. Inflammation and its resolution in atherosclerosis: mediators and therapeutic opportunities. Nat. Rev. Cardiol. 2019;16:389–406. doi: 10.1038/s41569-019-0169-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barenholz Y. Doxil® - The first FDA-approved nano-drug: lessons learned. J. Control. Release. 2012;160:117–134. doi: 10.1016/j.jconrel.2012.03.020. [DOI] [PubMed] [Google Scholar]
- Bashiri S., Koirala P., Toth I., Skwarczynski M. Carbohydrate immune adjuvants in subunit vaccines. Pharmaceutics. 2020;12:965. doi: 10.3390/pharmaceutics12100965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergsson G., Steingrímsson Ó., Thormar H. Bactericidal effects of fatty acids and monoglycerides on Helicobacter pylori. Int. J. Antimicrob. Agents. 2002;20:258–262. doi: 10.1016/S0924-8579(02)00205-4. [DOI] [PubMed] [Google Scholar]
- Bisschop P. H., Bandsma R. H. J., Stellaard F., Ter Harmsel A., Meijer A. J., Sauerwein H. P., Kuipers F., Romijn J. A. Low-fat, high-carbohydrate and high-fat, low-carbohydrate diets decrease primary bile acid synthesis in humans. Am. J. Clin. Nutr. 2004;79:570–576. doi: 10.1093/ajcn/79.4.570. [DOI] [PubMed] [Google Scholar]
- Boffa M. B., Koschinsky M. L. Oxidized phospholipids as a unifying theory for lipoprotein(a) and cardiovascular disease. Nat. Rev. Cardiol. 2019;16:305–318. doi: 10.1038/s41569-018-0153-2. [DOI] [PubMed] [Google Scholar]
- Braverman N. E., Moser A. B. Functions of plasmalogen lipids in health and disease. Biochim. Biophys. Acta Mol. Basis Dis. 2012;1822:1442–1452. doi: 10.1016/j.bbadis.2012.05.008. [DOI] [PubMed] [Google Scholar]
- Bridges D., Saltiel A. R. Phosphoinositides: key modulators of energy metabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids. 2015;1851:857–866. doi: 10.1016/j.bbalip.2014.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinkmann V., Billich A., Baumruker T., Heining P., Schmouder R., Francis G., Aradhye S., Burtin P. Fingolimod (FTY720): discovery anddevelopment of an oral drug to treatmultiple sclerosis. Nat. Rev. Drug Discov. 2010;9:883–897. doi: 10.1038/nrd3248. [DOI] [PubMed] [Google Scholar]
- Cha H. J., He C., Zhao H., Dong Y., An I. S., An S. Intercellular and intracellular functions of ceramides and their metabolites in skin (review) Int. J. Mol. Med. 2016;38:16–22. doi: 10.3892/ijmm.2016.2600. [DOI] [PubMed] [Google Scholar]
- Chang P. V., Hao L., Offermanns S., Medzhitov R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc. Natl. Acad. Sci. U.S.A. 2014;111:2247–2252. doi: 10.1073/pnas.1322269111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang P., Augustin K., Boddum K., Williams S., Sun M., Terschak J. A., Hardege J. D., Chen P. E., Walker M. C., Williams R. S. B. Seizure control by decanoic acid through direct AMPA receptor inhibition. Brain. 2016;139:431–443. doi: 10.1093/brain/awv325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., She Y., Lei J., Wang D., Wu S., Men K. Medium chain fatty acids: extraction, isolation, purification, bioactive properties and application. IOP Conf. Ser. Earth Environ. Sci. 2021;705:012013. doi: 10.1088/1755-1315/705/1/012013. [DOI] [Google Scholar]
- Chen M., Huang H., Zhou P., Zhang J., Dai Y., Yang D., Fan X., Pan H. Oral phosphatidylcholine improves intestinal barrier function in drug-induced liver injury in rats. Gastroenterol. Res. Pract. 2019;2019:8723460. doi: 10.1155/2019/8723460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M. B., Jiang Q., Liu Y. Y., Zhang Y., He B. S., Wei M. X., Lu J. W., Ji Y., Lu P. H. C6 ceramide dramatically increases vincristine sensitivity both in vivo and in vitro, involving AMP-activated protein kinase-p53 signaling. Carcinogenesis. 2015;36:1061–1070. doi: 10.1093/carcin/bgv094. [DOI] [PubMed] [Google Scholar]
- Choi M. K., Song I. S. Recent advances in the formulation of sphingolipid anticancer therapeutics. J. Pharm. Investig. 2020;50:295–307. doi: 10.1007/s40005-020-00475-y. [DOI] [Google Scholar]
- Currie E., Schulze A., Zechner R., Walther T. C., Farese R. V. Cellular fatty acid metabolism and cancer. Cell Metab. 2013;18:153–161. doi: 10.1016/j.cmet.2013.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutillo G., Saariaho A. H., Meri S. Physiology of gangliosides and the role of antiganglioside antibodies in human diseases. Cell. Mol. Immunol. 2020;17:313–322. doi: 10.1038/s41423-020-0388-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czech M. P. Dynamics of phosphoinositides in membrane retrieval and insertion. Annu. Rev. Physiol. 2003;65:791–815. doi: 10.1146/annurev.physiol.65.092101.142522. [DOI] [PubMed] [Google Scholar]
- Da Silva A., Caldas A. P. S., Hermsdorff H. H. M., Bersch-Ferreira Â. C., Torreglosa C. R., Weber B., Bressan J. Triglyceride-glucose index is associated with symptomatic coronary artery disease in patients in secondary care. Cardiovasc. Diabetol. 2019;18:89. doi: 10.1186/s12933-019-0893-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasgupta S., Ray S. W. Diverse biological functions of sphingolipids in the cns: ceramide and sphingosine regulate myelination in developing brain but stimulate demyelination during pathogenesis of multiple sclerosis. J. Neurol. Psychol. 2017;5:10.13188/2332-3469.1000035. doi: 10.13188/2332-3469.1000035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desale S. E., Chinnathambi S. α-Linolenic acid modulates phagocytosis and endosomal pathways of extracellular tau in microglia. Cell Adh. Migr. 2021;15:84–100. doi: 10.1080/19336918.2021.1898727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dial E. J., Zayat M., Lopez-Storey M., Tran D., Lichtenberger L. Oral phosphatidylcholine preserves the gastrointestinal mucosal barrier during LPS-induced inflammation. Shock. 2008;30:729–733. doi: 10.1097/SHK.0b013e318173e8d4. [DOI] [PubMed] [Google Scholar]
- Dou X., Gao N., Yan D., Shan A. Sodium butyrate alleviates mouse colitis by regulating gut microbiota dysbiosis. Animals. 2020;10:1154. doi: 10.3390/ani10071154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Draznin B. Molecular mechanisms of insulin resistance: serine phosphorylation of insulin receptor substrate-1 and increased expression of p85 α: the two sides of a coin. Diabetes. 2006;55:2392–2397. doi: 10.2337/db06-0391. [DOI] [PubMed] [Google Scholar]
- Duscha A., Gisevius B., Hirschberg S., Yissachar N., Stangl G. I., Eilers E., Bader V., Haase S., Kaisler J., David C., Schneider R., Troisi R., Zent D., Hegelmaier T., Dokalis N., Gerstein S., Del Mare-Roumani S., Amidror S., Staszewski O., Poschmann G., Stühler K., Hirche F., Balogh A., Kempa S., Träger P., Zaiss M. M., Holm J. B., Massa M. G., Nielsen H. B., Faissner A., Lukas C., Gatermann S. G., Scholz M., Przuntek H., Prinz M., Forslund S. K., Winklhofer K. F., Müller D. N., Linker R. A., Gold R., Haghikia A. Propionic acid shapes the multiple sclerosis disease course by an immunomodulatory mechanism. Cell. 2020;180:1067–1080.e16. doi: 10.1016/j.cell.2020.02.035. [DOI] [PubMed] [Google Scholar]
- Facchin S., Vitulo N., Calgaro M., Buda A., Romualdi C., Pohl D., Perini B., Lorenzon G., Marinelli C., D'Incà R., Sturniolo G. C., Savarino E. V. Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel disease. Neurogastroenterol. Motil. 2020;32:13–25. doi: 10.1111/nmo.13914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fahy E., Cotter D., Sud M., Subramaniam S. Lipid classification, structures and tools. Biochim. Biophys. Acta. 2011;1811:637–647. doi: 10.1016/j.bbalip.2011.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan R., Kim J., You M., Giraud D., Toney A. M., Shin S. H., Kim S. Y., Borkowski K., Newman J. W., Chung S. α-Linolenic acid-enriched butter attenuated high fat diet-induced insulin resistance and inflammation by promoting bioconversion of n-3 PUFA and subsequent oxylipin formation. J. Nutr. Biochem. 2020;76:108285. doi: 10.1016/j.jnutbio.2019.108285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng H., Nakajima N., Wu L., Yamashita M., Lopes T. J. S., Tsuji M., Hasegawa H., Watanabe T., Kawaoka Y. A glycolipid adjuvant, 7DW8-5, enhances the protective immune response to the current split influenza vaccine in mice. Front. Microbiol. 2019;10:2157. doi: 10.3389/fmicb.2019.02157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez C., Sandin M., Sampaio J. L., Almgren P., Narkiewicz K., Hoffmann M., Hedner T., Wahlstrand B., Simons K., Shevchenko A., James P., Melander O. Plasma lipid composition and risk of developing cardiovascular disease. PLoS ONE. 2013;8:e71846. doi: 10.1371/journal.pone.0071846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujino T., Yamada T., Asada T., Tsuboi Y., Wakana C., Mawatari S., Kono S. Efficacy and blood plasmalogen changes by oral administration of plasmalogen in patients with mild Alzheimer's disease and mild cognitive impairment: a multicenter, randomized, double-blind, placebo-controlled trial. EBioMedicine. 2017;17:199–205. doi: 10.1016/j.ebiom.2017.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geng S., Zhu W., Xie C., Li X., Wu J., Liang Z., Xie W., Zhu J., Huang C., Zhu M., Wu R., Zhong C. Medium-chain triglyceride ameliorates insulin resistance and inflammation in high fat diet-induced obese mice. Eur. J. Nutr. 2016;55:931–940. doi: 10.1007/s00394-015-0907-0. [DOI] [PubMed] [Google Scholar]
- Goto S., Sakai S., Kera J., Suma Y., Soma G. I., Takeuchi S. Intradermal administration of lipopolysaccharide in treatment of human cancer. Cancer Immunol. Immunother. 1996;42:255–261. doi: 10.1007/s002620050279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han S., Wang C., Qin X., Xia J., Wu A. LPS alters the immuno-phenotype of glioma and glioma stem-like cells and induces in vivo antitumor immunity via TLR4. J. Exp. Clin. Cancer Res. 2017;36:83. doi: 10.1186/s13046-017-0552-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannun Y. A., Obeid L. M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018;19:175–191. doi: 10.1038/nrm.2017.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannun Y. A., Obeid L. M. Principles of bioactive lipid signalling: lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 2008;9:139–150. doi: 10.1038/nrm2329. [DOI] [PubMed] [Google Scholar]
- Harayama T., Riezman H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 2018;19:281–296. doi: 10.1038/nrm.2017.138. [DOI] [PubMed] [Google Scholar]
- Henderson S. T., Vogel J. L., Barr L. J., Garvin F., Jones J. J., Costantini L. C. Study of the ketogenic agent AC-1202 in mild to moderate Alzheimer's disease: a randomized, double-blind, placebo-controlled, multicenter trial. Nutr. Metab. 2009;6:31. doi: 10.1186/1743-7075-6-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry B. D., Neill D. R., Becker K. A., Gore S., Bricio-Moreno L., Ziobro R., Edwards M. J., Mühlemann K., Steinmann J., Kleuser B., Japtok L., Luginbühl M., Wolfmeier H., Scherag A., Gulbins E., Kadioglu A., Draeger A., Babiychuk E. B. Engineered liposomes sequester bacterial exotoxins and protect from severe invasive infections in mice. Nat. Biotechnol. 2015;33:81–88. doi: 10.1038/nbt.3037. [DOI] [PubMed] [Google Scholar]
- Huang H. J., Zhang A. Y., Cao H. C., Lu H. F., Wang B. H., Xie Q., Xu W., Li L. J. Metabolomic analyses of faeces reveals malabsorption in cirrhotic patients. Dig. Liver Dis. 2013;45:677–682. doi: 10.1016/j.dld.2013.01.001. [DOI] [PubMed] [Google Scholar]
- Husted A. S., Trauelsen M., Rudenko O., Hjorth S. A., Schwartz T. W. GPCR-mediated signaling of metabolites. Cell Metab. 2017;25:777–796. doi: 10.1016/j.cmet.2017.03.008. [DOI] [PubMed] [Google Scholar]
- Hyldgaard M., Sutherland D. S., Sundh M., Mygind T., Meyer R. L. Antimicrobial mechanism of monocaprylate. Appl. Environ. Microbiol. 2012;78:2957–2965. doi: 10.1128/AEM.07224-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juge N., Gray J. A., Omote H., Miyaji T., Inoue T., Hara C., Uneyama H., Edwards R. H., Nicoll R. A., Moriyama Y. Metabolic control of vesicular glutamate transport and release. Neuron. 2010;68:99–112. doi: 10.1016/j.neuron.2010.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jumina J., Lavendi W., Singgih T., Triono S., Kurniawan Y. S., Koketsu M. Preparation of monoacylglycerol derivatives from indonesian edible oil and their antimicrobial assay against Staphylococcus aureus and Escherichia coli. Sci. Rep. 2019;9:10941. doi: 10.1038/s41598-019-47373-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung T. W., Kim S. T., Lee J. H., Chae S. I., Hwang K. W., Chung Y. H., Kim H. C., Abd El-Aty A. M., Lee T. J., Park E. S., Jeong J. H. Phosphatidylcholine causes lipolysis and apoptosis in adipocytes through the tumor necrosis factor alpha-dependent pathway. Pharmacology. 2018;101:111–119. doi: 10.1159/000481571. [DOI] [PubMed] [Google Scholar]
- Jung T. W., Park T., Park J., Kim U., Je H. D., Kim H. D., Cho S. W., Abd El-Aty A. M., Song J. H., Kim H. C., Shin Y. K., Jeong J. H. Phosphatidylcholine causes adipocyte-specific lipolysis and apoptosis in adipose and muscle tissues. PLoS ONE. 2019;14:e0214760. doi: 10.1371/journal.pone.0214760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kachko I., Traitel T., Goldbart R., Silbert L., Katz M., Bashan N., Jelinek R., Rudich A., Kost J. Polymeric carrier-mediated intracellular delivery of phosphatidylinositol-3,4,5-trisphosphate to overcome insulin resistance. J. Drug Target. 2015;23:698–709. doi: 10.3109/1061186X.2015.1052076. [DOI] [PubMed] [Google Scholar]
- Kadowaki A., Sada N., Juge N., Wakasa A., Moriyama Y., Inoue T. Neuronal inhibition and seizure suppression by acetoacetate and its analog, 2-phenylbutyrate. Epilepsia. 2017;58:845–857. doi: 10.1111/epi.13718. [DOI] [PubMed] [Google Scholar]
- Kawano A., Ariyoshi W., Yoshioka Y., Hikiji H., Nishihara T., Okinaga T. Docosahexaenoic acid enhances M2 macrophage polarization via the p38 signaling pathway and autophagy. J. Cell. Biochem. 2019;120:12604–12617. doi: 10.1002/jcb.28527. [DOI] [PubMed] [Google Scholar]
- Khanum R., Thevanayagam H. Lipid peroxidation: its effects on the formulation and use of pharmaceutical emulsions. Asian J. Pharm. Sci. 2017;12:401–411. doi: 10.1016/j.ajps.2017.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khazanov E., Priev A., Shillemans J. P., Barenholz Y. Physicochemical and biological characterization of ceramide-containing liposomes: paving the way to ceramide therapeutic application. Langmuir. 2008;24:6965–6980. doi: 10.1021/la800207z. [DOI] [PubMed] [Google Scholar]
- Kim C. H. Control of lymphocyte functions by gut microbiota-derived short-chain fatty acids. Cell. Mol. Immunol. 2021;18:1161–1171. doi: 10.1038/s41423-020-00625-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Ahn M., Choi Y., Kang T., Kim J., Lee N. H., Kim G. O., Shin T. Alpha-linolenic acid alleviates dextran sulfate sodium-induced ulcerative colitis in mice. Inflammation. 2020;43:1876–1883. doi: 10.1007/s10753-020-01260-7. [DOI] [PubMed] [Google Scholar]
- Kimura I., Ichimura A., Ohue-Kitano R., Igarashi M. Free fatty acid receptors in health and disease. Physiol. Rev. 2020;100:171–210. doi: 10.1152/physrev.00041.2018. [DOI] [PubMed] [Google Scholar]
- Klein M. E., Rieckmann M., Lucas H., Meister A., Loppnow H., Mäder K. Phosphatidylserine (PS) and phosphatidylglycerol (PG) enriched mixed micelles (mm): a new nano-drug delivery system with anti-inflammatory potential? Eur. J. Pharm. Sci. 2020;152:105451. doi: 10.1016/j.ejps.2020.105451. [DOI] [PubMed] [Google Scholar]
- Ko C. W., Qu J., Black D. D., Tso P. Regulation of intestinal lipid metabolism: current concepts and relevance to disease. Nat. Rev. Gastroenterol. Hepatol. 2020;17:169–183. doi: 10.1038/s41575-019-0250-7. [DOI] [PubMed] [Google Scholar]
- Koh A., De Vadder F., Kovatcheva-Datchary P., Bäckhed F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell. 2016;165:1332–1345. doi: 10.1016/j.cell.2016.05.041. [DOI] [PubMed] [Google Scholar]
- Kostarnoy A. V., Gancheva P. G., Logunov D. Y., Verkhovskaya L. V., Bobrov M. A., Scheblyakov D. V., Tukhvatulin A. I., Filippova N. E., Naroditsky B. S., Gintsburg A. L. Topical bacterial lipopolysaccharide application affects inflammatory response and promotes wound healing. J. Interferon Cytokine Res. 2013;33:514–522. doi: 10.1089/jir.2012.0108. [DOI] [PubMed] [Google Scholar]
- Kurek K., Łukaszuk B., Piotrowska D. M., WiesioBek P., Chabowska A. M., Zendzian-Piotrowska M. Metabolism, physiological role, and clinical implications of sphingolipids in gastrointestinal tract. BioMed Res. Int. 2013;2013:908907. doi: 10.1155/2013/908907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacour S., Hammann A., Grazide S., Lagadic-Gossmann D., Athias A., Sergent O., Laurent G., Gambert P., Solary E., Dimanche-Boitrel M. T. Cisplatin-induced CD95 redistribution into membrane lipid rafts of HT29 human colon cancer cells. Cancer Res. 2004;64:3593–3598. doi: 10.1158/0008-5472.CAN-03-2787. [DOI] [PubMed] [Google Scholar]
- Lavelle A., Sokol H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 2020;17:223–237. doi: 10.1038/s41575-019-0258-z. [DOI] [PubMed] [Google Scholar]
- Lee B., Sur B. J., Han J. J., Shim I., Her S., Lee Y. S., Lee H. J., Hahm D. H. Oral administration of squid lecithin-transphosphatidylated phosphatidylserine improves memory impairment in aged rats. Prog. Neuro-Psychopharmacol. Biol. Psychiatry. 2015;56:1–10. doi: 10.1016/j.pnpbp.2014.07.004. [DOI] [PubMed] [Google Scholar]
- Leuti A., Fazio D., Fava M., Piccoli A., Oddi S., Maccarrone M. Bioactive lipids, inflammation and chronic diseases. Adv. Drug Deliv. Rev. 2020;159:133–169. doi: 10.1016/j.addr.2020.06.028. [DOI] [PubMed] [Google Scholar]
- Li J., Wang X., Zhang T., Wang C., Huang Z., Luo X., Deng Y. A review on phospholipids and their main applications in drug delivery systems. Asian J. Pharm. Sci. 2015;10:81–98. doi: 10.1016/j.ajps.2014.09.004. [DOI] [Google Scholar]
- Li X., Fujio M., Imamura M., Wu D., Vasan S., Wong C. H., Ho D. D., Tsuji M. Design of a potent CD1d-binding NKT cell ligand as a vaccine adjuvant. Proc. Natl. Acad. Sci. U.S.A. 2010;107:13010–13015. doi: 10.1073/pnas.1006662107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Toledano M. A., Saxena V., Legassie J. D., Liu H., Ghanta A., Riseman S., Cocilova C., Daak A., Thorsteinsson T., Rabinowicz A. L., Sancilio F. D. Advanced lipid technologies® (ALT®): a proven formulation platform to enhance the bioavailability of lipophilic compounds. J. Drug Deliv. 2019;2019:1957360. doi: 10.1155/2019/1957360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luu M., Pautz S., Kohl V., Singh R., Romero R., Lucas S., Hofmann J., Raifer H., Vachharajani N., Carrascosa L. C., Lamp B., Nist A., Stiewe T., Shaul Y., Adhikary T., Zaiss M. M., Lauth M., Steinhoff U., Visekruna A. The short-chain fatty acid pentanoate suppresses autoimmunity by modulating the metabolic-epigenetic crosstalk in lymphocytes. Nat. Commun. 2019;10:760. doi: 10.1038/s41467-019-08711-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyu K., Zhang D., Nozaki Y., Zhang Y., Bhanot S., Cline G., Samual V., Shulman G. I. Membrane sn-1,2 diacylglycerol mediates lipid-induced hepatic insulin resistance in vivo. Diabetes. 2018;67:243–LB. doi: 10.2337/db18-243-LB. [DOI] [Google Scholar]
- MacLeod M. K. L., McKee A. S., David A., Wang J., Mason R., Kappler J. W., Marrack P. Vaccine adjuvants aluminum and monophosphoryl lipid a provide distinct signals to generate protective cytotoxic memory CD8 T cells. Proc. Natl. Acad. Sci. U.S.A. 2011;108:7914–7919. doi: 10.1073/pnas.1104588108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeda N., Kokai Y., Hada T., Yoshida H., Mizushina Y. Oral administration of monogalactosyl diacylglycerol from spinach inhibits colon tumor growth in mice. Exp. Ther. Med. 2013;5:17–22. doi: 10.3892/etm.2012.792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maev I. V., Samsonov A. A., Palgova L. K., Pavlov C. S., Shirokova E. N., Vovk E. I., Starostin K. M. Effectiveness of phosphatidylcholine as adjunctive therapy in improving liver function tests in patients with non-alcoholic fatty liver disease and metabolic comorbidities: real-life observational study from Russia. BMJ Open Gastroenterol. 2020;7:e000368. doi: 10.1136/bmjgast-2019-000368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maki K. C., Dicklin M. R. Strategies to improve bioavailability of omega-3 fatty acids from ethyl ester concentrates. Curr. Opin. Clin. Nutr. Metab. Care. 2019;22:116–123. doi: 10.1097/MCO.0000000000000537. [DOI] [PubMed] [Google Scholar]
- Marat A. L., Haucke V. Phosphatidylinositol 3-phosphates-at the interface between cell signalling and membrane traffic. EMBO J. 2016;35:561–579. doi: 10.15252/embj.201593564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClements D. J. Design of nano-laminated coatings to control bioavailability of lipophilic food components. J. Food Sci. 2010;75:30–42. doi: 10.1111/j.1750-3841.2009.01452.x. [DOI] [PubMed] [Google Scholar]
- McGarry J. D., Foster D. W. Regulation of hepatic fatty acid oxidation and ketone body production. Annu. Rev. Biochem. 1980;49:395–420. doi: 10.1146/annurev.bi.49.070180.002143. [DOI] [PubMed] [Google Scholar]
- Medina T. P., Gerle M., Humbert J., Chu H., Köpnick A. L., Barkmann R., Garamus V. M., Sanz B., Purcz N., Will O., Appold L., Damm T., Suojanen J., Arnold P., Lucius R., Willumeit-Römer R., Açil Y., Wiltfang J., Goya G. F., Glüer C. C., Medina O. P. Lipid-iron nanoparticle with a cell stress release mechanism combined with a local alternating magnetic field enables site-activated drug release. Cancers (Basel) 2020;12:3767. doi: 10.3390/cancers12123767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messias M. C. F., Mecatti G. C., Priolli D. G., De Oliveira Carvalho P. Plasmalogen lipids: functional mechanism and their involvement in gastrointestinal cancer. Lipids Health Dis. 2018;17:41. doi: 10.1186/s12944-018-0685-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao L., Li L., Huang Y., Delcassian D., Chahal J., Han J., Shi Y., Sadtler K., Gao W., Lin J., Doloff J. C., Langer R., Anderson D. G. Delivery of mRNA vaccines with heterocyclic lipids increases anti-tumor efficacy by sting-mediated immune cell activation. Nat. Biotechnol. 2019;37:1174–1185. doi: 10.1038/s41587-019-0247-3. [DOI] [PubMed] [Google Scholar]
- Milane L., Amiji M. Clinical approval of nanotechnology-based SARS-CoV-2 mRNA vaccines: impact on translational nanomedicine. Drug Deliv. Transl. Res. 2021;11:1309–1315. doi: 10.1007/s13346-021-00911-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mo Z., Tang C., Li H., Lei J., Zhu L., Kou L., Li H., Luo S., Li C., Chen W., Zhang L. Eicosapentaenoic acid prevents inflammation induced by acute cerebral infarction through inhibition of NLRP3 inflammasome activation. Life Sci. 2020;242:117133. doi: 10.1016/j.lfs.2019.117133. [DOI] [PubMed] [Google Scholar]
- Moon E. K., Wang L. C., Dolfi D. V., Wilson C. B., Ranganathan R., Sun J., Kapoor V., Scholler J., Puré E., Milone M. C., June C. H., Riley J. L., Wherry E. J., Albelda S. M. Multifactorial T-cell hypofunction that is reversible can limit the efficacy of chimeric antigen receptor-transduced human T cells in solid tumors. Clin. Cancer Res. 2014;20:4262–4273. doi: 10.1158/1078-0432.CCR-13-2627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moniri N. H., Farah Q. Short-chain free-fatty acid G protein-coupled receptors in colon cancer. Biochem. Pharmacol. 2021;186:114483. doi: 10.1016/j.bcp.2021.114483. [DOI] [PubMed] [Google Scholar]
- Morin C., Fortin S. Docosahexaenoic acid monoglyceride increases carboplatin activity in lung cancer models by targeting EGFR. Anticancer Res. 2017;37:6015–6023. doi: 10.21873/anticanres.12048. [DOI] [PubMed] [Google Scholar]
- Nagachinta S., Bouzo B. L., Vazquez-Rios A. J., Lopez R., de la Fuente M. Sphingomyelin-based nanosystems (SNS) for the development of anticancer miRNA therapeutics. Pharmaceutics. 2020;12:189. doi: 10.3390/pharmaceutics12020189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nastasi C., Candela M., Bonefeld C. M., Geisler C., Hansen M., Krejsgaard T., Biagi E., Andersen M. H., Brigidi P., Ødum N., Litman T., Woetmann A. The effect of short-chain fatty acids on human monocyte-derived dendritic cells. Sci. Rep. 2015;5:16148. doi: 10.1038/srep16148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- N'Goma J. C. B., Amara S., Dridi K., Jannin V., Carrière F. Understanding the lipid-digestion processes in the GI tract before designing lipid-based drug-delivery systems. Ther. Deliv. 2012;3:105–124. doi: 10.4155/tde.11.138. [DOI] [PubMed] [Google Scholar]
- Ogretmen B. Sphingolipid metabolism in cancer signalling and therapy. Nat. Rev. Cancer. 2017;18:33–50. doi: 10.1038/nrc.2017.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh D. Y., Talukdar S., Bae E. J., Imamura T., Morinaga H., Fan W., Li P., Lu W. J., Watkins S., M., Olefsky J. M. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell. 2010;142:687–698. doi: 10.1016/j.cell.2010.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onodera T., Futai E., Kan E., Abe N., Uchida T., Kamio Y., Kaneko J. Phosphatidylethanolamine plasmalogen enhances the inhibiting effect of phosphatidylethanolamine on γ-secretase activity. J. Biochem. 2015;157:301–309. doi: 10.1093/jb/mvu074. [DOI] [PubMed] [Google Scholar]
- Parada Venegas D., De La Fuente M. K., Landskron G., González M. J., Quera R., Dijkstra G., Harmsen H. J. M., Faber K. N., Hermoso M. A. Corrigendum: short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 2019;10:1486. doi: 10.3389/fimmu.2019.01486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paris F., Fuks Z., Kang A., Capodieci P., Juan G., Ehleiter D., Haimovitz-Friedman A., Cordon-Cardo C., Kolesnick R. Endothelial apoptosis as the primary lesion initiating intestinal radiation damage in mice. Science. 2001;293:293–297. doi: 10.1126/science.1060191. [DOI] [PubMed] [Google Scholar]
- Park W. J., Park J. W. The role of sphingolipids in endoplasmic reticulum stress. FEBS Lett. 2020;594:3632–3651. doi: 10.1002/1873-3468.13863. [DOI] [PubMed] [Google Scholar]
- Parker M. W., Feil S. C. Pore-forming protein toxins: from structure to function. Prog. Biophys. Mol. Biol. 2005;88:91–142. doi: 10.1016/j.pbiomolbio.2004.01.009. [DOI] [PubMed] [Google Scholar]
- Pauls S. D., Rodway L. A., Winter T., Taylor C. G., Zahradka P., Aukema H. M. Alpha-linolenic acid enhances the phagocytic and secretory functions of alternatively activated macrophages in part via changes to the oxylipin profile. Int. J. Biochem. Cell Biol. 2020;119:105662. doi: 10.1016/j.biocel.2019.105662. [DOI] [PubMed] [Google Scholar]
- Pauls S. D., Rodway L. A., Winter T., Taylor C. G., Zahradka P., Aukema H. M. Anti-inflammatory effects of α-linolenic acid in M1-like macrophages are associated with enhanced production of oxylipins from α-linolenic and linoleic acid. J. Nutr. Biochem. 2018;57:121–129. doi: 10.1016/j.jnutbio.2018.03.020. [DOI] [PubMed] [Google Scholar]
- Ramana K. V., Srivastava S., Singhal S. S. Lipid peroxidation products in human health and disease. Oxid. Med. Cell. Longev. 2013;2013:583438. doi: 10.1155/2013/583438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez M., Amate L., Gil A. Absorption and distribution of dietary fatty acids from different sources. Early Hum. Dev. 2001;65 Suppl:S95–S101. doi: 10.1016/S0378-3782(01)00211-0. [DOI] [PubMed] [Google Scholar]
- Rockwell C. E., Morrison D. C., Qureshi N. Lipid a-mediated tolerance and cancer therapy. Adv. Exp. Med. Biol. 2010;667:81–99. doi: 10.1007/978-1-4419-1603-7_8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sada N., Inoue T. Electrical control in neurons by the ketogenic diet. Front. Cell. Neurosci. 2018;12:208. doi: 10.3389/fncel.2018.00208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito O., Svensson C. I., Buczynski M. W., Wegner K., Hua X. Y., Codeluppi S., Schaloske R. H., Deems R. A., Dennis E. A., Yaksh T. L. Spinal glial TLR4-mediated nociception and production of prostaglandin E2 and TNF. Br. J. Pharmacol. 2010;160:1754–1764. doi: 10.1111/j.1476-5381.2010.00811.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito S., Hernandez-Ono A., Ginsberg H. N. Dietary 1,3-diacylglycerol protects against diet-induced obesity and insulin resistance. Metabolism. 2007;56:1566–1575. doi: 10.1016/j.metabol.2007.06.024. [DOI] [PubMed] [Google Scholar]
- Salti G., Ghersetich I., Tantussi F., Bovani B., Lotti T. Phosphatidylcholine and sodium deoxycholate in the treatment of localized fat: a double-blind, randomized study. Dermatol. Surg. 2008;34:60–66. doi: 10.1097/00042728-200801000-00010. [DOI] [PubMed] [Google Scholar]
- Schink K. O., Tan K. W., Stenmark H. Phosphoinositides in control of membrane dynamics. Annu. Rev. Cell Dev. Biol. 2016;32:143–171. doi: 10.1146/annurev-cellbio-111315-125349. [DOI] [PubMed] [Google Scholar]
- Serhan C. N., Chiang N. Resolution phase lipid mediators of inflammation: agonists of resolution. Curr. Opin. Pharmacol. 2013;13:632–640. doi: 10.1016/j.coph.2013.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serhan C. N., Chiang N., Van Dyke T. E. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008;8:349–361. doi: 10.1038/nri2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah N. K., Gupta S. K., Wang Z., Meenach S. A. Enhancement of macrophage uptake via phosphatidylserine-coated acetalated dextran nanoparticles. J. Drug Deliv. Sci. Technol. 2019;50:57–65. doi: 10.1016/j.jddst.2019.01.013. [DOI] [Google Scholar]
- Shetab Boushehri M. A., Lamprecht A. Challenges of using lipopolysaccharides for cancer immunotherapy and potential delivery-based solutions thereto. Ther. Deliv. 2019;10:165–187. doi: 10.4155/tde-2018-0076. [DOI] [PubMed] [Google Scholar]
- Shetab Boushehri M. A., Yazeji T., Stein V., Lamprecht A. Modulation of nanostructure-based lipopolysaccharide active immunotherapy in cancer: size and composition determine short- and long-term tolerability. Mol. Pharm. 2019;16:4507–4518. doi: 10.1021/acs.molpharmaceut.9b00631. [DOI] [PubMed] [Google Scholar]
- Shim G., Kim M. G., Park J. Y., Oh Y. K. Graphene-based nanosheets for delivery of chemotherapeutics and biological drugs. Adv. Drug Deliv. Rev. 2016;105:205–227. doi: 10.1016/j.addr.2016.04.004. [DOI] [PubMed] [Google Scholar]
- Simon M. V., Basu S. K., Qaladize B., Grambergs R., Rotstein N. P., Mandal N. Sphingolipids as critical players in retinal physiology and pathology. J. Lipid Res. 2021;62:100037. doi: 10.1194/jlr.TR120000972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simopoulos A. P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients. 2016;8:128. doi: 10.3390/nu8030128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slotte J. P. Biological functions of sphingomyelins. Prog. Lipid Res. 2013;52:424–437. doi: 10.1016/j.plipres.2013.05.001. [DOI] [PubMed] [Google Scholar]
- So J., Wu D., Lichtenstein A. H., Tai A. K., Matthan N. R., Maddipati K. R., Lamon-Fava S. EPA and DHA differentially modulate monocyte inflammatory response in subjects with chronic inflammation in part via plasma specialized pro-resolving lipid mediators: a randomized, double-blind, crossover study. Atherosclerosis. 2021;316:90–98. doi: 10.1016/j.atherosclerosis.2020.11.018. [DOI] [PubMed] [Google Scholar]
- Song W., Tiruthani K., Wang Y., Shen L., Hu M., Dorosheva O., Qiu K., Kinghorn K. A., Liu R., Huang L. Trapping of lipopolysaccharide to promote immunotherapy against colorectal cancer and attenuate liver metastasis. Adv. Mater. 2018;30:e1805007. doi: 10.1002/adma.201805007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spite M., Clària J., Serhan C. N. Resolvins, Specialized proresolving lipid mediators, and their potential roles in metabolic diseases. Cell Metab. 2014;19:21–36. doi: 10.1016/j.cmet.2013.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suhrland C., Truman J. P., Obeid L. M., Sitharaman B. Oxidized graphene nanoparticles as a delivery system for the pro-apoptotic sphingolipid C6 ceramide. J. Biomed. Mater. Res. A. 2019;107:25–37. doi: 10.1002/jbm.a.36474. [DOI] [PubMed] [Google Scholar]
- Sutter I., Klingenberg R., Othman A., Rohrer L., Landmesser U., Heg D., Rodondi N., Mach F., Windecker S., Matter C. M., Lüscher T. F., von Eckardstein A., Hornemann T. Decreased phosphatidylcholine plasmalogens - a putative novel lipid signature in patients with stable coronary artery disease and acute myocardial infarction. Atherosclerosis. 2016;246:130–140. doi: 10.1016/j.atherosclerosis.2016.01.003. [DOI] [PubMed] [Google Scholar]
- Szondy Z., Sarang Z., Kiss B., Garabuczi É., Köröskényi K. Anti-inflammatory mechanisms triggered by apoptotic cells during their clearance. Front. Immunol. 2017;8:909. doi: 10.3389/fimmu.2017.00909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanner G. R., Lutas A., Martínez-François J. R., Yellen G. Single KATP channel opening in response to action potential firing in mouse dentate granule neurons. J. Neurosci. 2011;31:8689–8696. doi: 10.1523/JNEUROSCI.5951-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas M. K., D'Silva J. A., Borole A. J. Injection lipolysis with a cocktail of phosphatidylcholine and deoxycholate: an indian experience. Plast. Reconstr. Surg. Glob. Open. 2016;4:e861. doi: 10.1097/GOX.0000000000000492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas M. K., D'Silva J. A., Borole A. J. Injection lipolysis: a systematic review of literature and our experience with a combination of phosphatidylcholine and deoxycholate over a period of 14 years in 1269 patients of Indian and South East Asian origin. J. Cutan. Aesthet. Surg. 2018;11:222–228. doi: 10.4103/JCAS.JCAS_117_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson B. S., Chilton P. M., Ward J. R., Evans J. T., Mitchell T. C. The low-toxicity versions of LPS, MPL adjuvant and RC529, are efficient adjuvants for CD4+ T cells. J. Leukoc. Biol. 2005;78:1273–1280. doi: 10.1189/jlb.0305172. [DOI] [PubMed] [Google Scholar]
- Toyoda T., Kamata T., Tanaka K., Ihara F., Takami M., Suzuki H., Nakajima T., Ikeuchi T., Kawasaki Y., Hanaoka H., Nakayama T., Yoshino I., Motohashi S. Phase II study of α-galactosylceramide-pulsed antigen-presenting cells in patients with advanced or recurrent non-small cell lung cancer. J. Immunother. Cancer. 2020;8:e000316. doi: 10.1136/jitc-2019-000316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trompette A., Gollwitzer E. S., Yadava K., Sichelstiel A. K., Sprenger N., Ngom-Bru C., Blanchard C., Junt T., Nicod L. P., Harris N. L., Marsland B. J. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 2014;20:159–166. doi: 10.1038/nm.3444. [DOI] [PubMed] [Google Scholar]
- Turpin-Nolan S. M., Brüning J. C. The role of ceramides in metabolic disorders: when size and localization matters. Nat. Rev. Endocrinol. 2020;16:224–233. doi: 10.1038/s41574-020-0320-5. [DOI] [PubMed] [Google Scholar]
- Valenzuela P. L., Carrera-Bastos P., Gálvez B. G., Ruiz-Hurtado G., Ordovas J. M., Ruilope L. M., Lucia A. Lifestyle interventions for the prevention and treatment of hypertension. Nat. Rev. Cardiol. 2021;18:251–275. doi: 10.1038/s41569-020-00437-9. [DOI] [PubMed] [Google Scholar]
- van Hell A. J., Melo M. N., van Blitterswijk W. J., Gueth D. M., Braumuller T. M., Pedrosa L. R. C., Song J. Y., Marrink S. J., Koning G. A., Jonkers J., Verheij M. Defined lipid analogues induce transient channels to facilitate drug-membrane traversal and circumvent cancer therapy resistance. Sci. Rep. 2013;3:1949. doi: 10.1038/srep01949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lummel M., van Blitterswijk W. J., Vink S. R., Veldman R. J., van der Valk M. A., Schipper D., Dicheva B. M., Eggermont A. M. M., ten Hagen T. L. M., Verheij M., Koning G. A. Enriching lipid nanovesicles with short-chain glucosylceramide improves doxorubicin delivery and efficacy in solid tumors. FASEB J. 2011;25:280–289. doi: 10.1096/fj.10-163709. [DOI] [PubMed] [Google Scholar]
- van Meer G., Voelker D. R., Feigenson G. W. Membrane lipids: where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008;9:112–124. doi: 10.1038/nrm2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verbeke R., Lentacker I., Breckpot K., Janssens J., Van Calenbergh S., De Smedt S. C., Dewitte H. Broadening the message: a nanovaccine co-loaded with messenger RNA and α-Galcer induces antitumor immunity through conventional and natural killer T cells. ACS Nano. 2019;13:1655–1669. doi: 10.1021/acsnano.8b07660. [DOI] [PubMed] [Google Scholar]
- Vieira S. A., Zhang G., Decker E. A. Biological implications of lipid oxidation products. J. Am. Oil Chem. Soc. 2017;94:339–351. doi: 10.1007/s11746-017-2958-2. [DOI] [Google Scholar]
- Wang R. X., Lee J. S., Campbell E. L., Colgan S. P. Microbiota-derived butyrate dynamically regulates intestinal homeostasis through regulation of actin-associated protein synaptopodin. Proc. Natl. Acad. Sci. U.S.A. 2020;117:11648–11657. doi: 10.1073/pnas.1917597117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S. B., Ma Y. Y., Chen X. Y., Zhao Y. Y., Mou X. Z. Ceramide-graphene oxide nanoparticles enhance cytotoxicity and decrease HCC xenograft development: a novel approach for targeted cancer therapy. Front. Pharmacol. 2019;10:69. doi: 10.3389/fphar.2019.00069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren E. C., Walker M. C., Williams R. S. B. All you need is fats-for seizure control: using amoeba to advance epilepsy research. Front. Cell. Neurosci. 2018;12:199. doi: 10.3389/fncel.2018.00199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson H. Biological membranes. Essays Biochem. 2015;59:43–70. doi: 10.1042/bse0590043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welte M. A., Gould A. P. Lipid droplet functions beyond energy storage. Biochim. Biophys. Acta Mol. Cell Biol. Lipids. 2017;1862:1260–1272. doi: 10.1016/j.bbalip.2017.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Won K. B., Park G. M., Lee S. E., Cho I. J., Kim H. C., Lee B. K., Chang H. J. Relationship of insulin resistance estimated by triglyceride glucose index to arterial stiffness. Lipids Health Dis. 2018;17:268. doi: 10.1186/s12944-018-0914-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood P. L., Mankidy R., Ritchie S., Heath D., Wood J. A., Flax J., Goodenowe D. B. Circulating plasmalogen levels and Alzheimer disease assessment scale-cognitive scores in Alzheimer patients. J. Psychiatry Neurosci. 2010;35:59–62. doi: 10.1503/jpn.090059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita S., Hashimoto M., Haque A. M., Nakagawa K., Kinoshita M., Shido O., Miyazawa T. Oral administration of ethanolamine glycerophospholipid containing a high level of plasmalogen improves memory impairment in amyloid β-infused rats. Lipids. 2017;52:575–585. doi: 10.1007/s11745-017-4260-3. [DOI] [PubMed] [Google Scholar]
- Yang C., Merlin D. Lipid-based drug delivery nanoplatforms for colorectal cancer therapy. Nanomaterials. 2020;10:1424. doi: 10.3390/nano10071424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang E., Singh B. K., Paustian A. M. S., Kambayashi T. Diacylglycerol kinase ζ is a target to enhance NK cell function. J. Immunol. 2016;197:934–941. doi: 10.4049/jimmunol.1600581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J., Hung J. T., Wang S. H., Cheng J. Y., Yu A. L. Targeting glycosphingolipids for cancer immunotherapy. FEBS Lett. 2020;594:3602–3618. doi: 10.1002/1873-3468.13917. [DOI] [PubMed] [Google Scholar]
- Zalba S., Seynhaeve A. L. B., Brouwers J. F., Süss R., Verheij M., Ten Hagen T. L. M. Sensitization of drug resistant sarcoma tumors by membrane modulation: via short chain sphingolipid-containing nanoparticles. Nanoscale. 2020;12:16967–16979. doi: 10.1039/D0NR02257H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarringhalam K., Zhang L., Kiebish M. A., Yang K., Han X., Gross R. W., Chuang J. Statistical analysis of the processes controlling choline and ethanolamine glycerophospholipid molecular species composition. PLoS ONE. 2012;7:e37293. doi: 10.1371/journal.pone.0037293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai L., Sun N., Han Z., Jin H. C., Zhang B. Liposomal short-chain C6 ceramide induces potent anti-osteosarcoma activity in vitro and in vivo. Biochem. Biophys. Res. Commun. 2015;468:274–280. doi: 10.1016/j.bbrc.2015.10.113. [DOI] [PubMed] [Google Scholar]
- Zhang C., Maruggi G., Shan H., Li J. Advances in mRNA vaccines for infectious diseases. Front. Immunol. 2019c;10:594. doi: 10.3389/fimmu.2019.00594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang F., Stephan S. B., Ene C. I., Smith T. T., Holland E. C., Stephan M. T. Nanoparticles that reshape the tumor milieu create a therapeutic window for effective T-cell therapy in solid malignancies. Cancer Res. 2018;78:3718–3730. doi: 10.1158/0008-5472.CAN-18-0306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S., Lu X., Wang B., Zhang G., Liu M., Geng S., Sun L., An J., Zhang Z., Zhang H. A soft anti-virulence liposome realizing the explosive release of antibiotics at an infectious site to improve antimicrobial therapy. J. Mater. Chem. B. 2021;9:147–158. doi: 10.1039/D0TB02255A. [DOI] [PubMed] [Google Scholar]
- Zhang T., De Waard A. A., Wuhrer M., Spaapen R. M. The role of glycosphingolipids in immune cell functions. Front. Immunol. 2019a;10:90. doi: 10.3389/fimmu.2019.00090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X., Matsuda M., Yaegashi N., Nabe T., Kitatani K. Regulation of necroptosis by phospholipids and sphingolipids. Cells. 2020;9:627. doi: 10.3390/cells9030627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Springfield R., Chen S., Li X., Feng X., Moshirian R., Yang R., Yuan W. α-GalCer and iNKT cell-based cancer immunotherapy: realizing the therapeutic potentials. Front. Immunol. 2019b;10:1126. doi: 10.3389/fimmu.2019.01126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao R., Jiang J., Li H., Chen M., Liu R., Sun S., Ma D., Liang X., Wang S. Phosphatidylserine-microbubble targeting-activated microglia/macrophage in inflammation combined with ultrasound for breaking through the blood-brain barrier. J. Neuroinflammation. 2018;15:334. doi: 10.1186/s12974-018-1368-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng L., Kelly C. J., Battista K. D., Schaefer R., Lanis J. M., Alexeev E. E., Wang R. X., Onyiah J. C., Kominsky D. J., Colgan S. P. Microbial-derived butyrate promotes epithelial barrier function through IL-10 receptor-dependent repression of claudin-2. J. Immunol. 2017;199:2976–2984. doi: 10.4049/jimmunol.1700105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou F., Ciric B., Zhang G. X., Rostami A. Immunotherapy using lipopolysaccharide-stimulated bone marrow-derived dendritic cells to treat experimental autoimmune encephalomyelitis. Clin. Exp. Immunol. 2014;178:447–458. doi: 10.1111/cei.12440. [DOI] [PMC free article] [PubMed] [Google Scholar]