Abstract
Background
Obesity is associated with restrictive ventilatory defects and a faster rate of decline in FVC. This association is not exclusively mediated by mechanical factors and may reflect direct pulmonary injury by adipose-derived mediators.
Research Question
Is adipose tissue involved in the pathogenesis of interstitial lung disease (ILD)?
Study Design and Methods
We evaluated the association of CT measures of pericardial, abdominal visceral, and abdominal subcutaneous adipose tissue with high-attenuation areas (HAAs) and interstitial lung abnormalities (ILAs) in a large multicenter cohort study of community-dwelling adults, using multivariable-adjusted models. We secondarily evaluated the association of adipose depot size with FVC and biomarkers of obesity and inflammation.
Results
In fully adjusted models, every doubling in pericardial adipose tissue volume was associated with a 63.4-unit increase in HAA (95% CI, 55.5-71.3), 20% increased odds of ILA (95% CI, –2% to 50%), and a 5.5% decrease in percent predicted FVC (95% CI, –6.8% to –4.3%). IL-6 levels accounted for 8% of the association between pericardial adipose tissue and HAA. Every doubling in visceral adipose tissue area was associated with a 41.5-unit increase in HAA (95% CI, 28.3-54.7), 30% increased odds of ILA (95% CI, –10% to 80%), and a 5.4% decrease in percent predicted FVC (95% CI, –6.6% to –4.3%). IL-6 and leptin accounted for 17% and 18%, respectively, of the association between visceral adipose tissue and HAA.
Interpretation
Greater amounts of pericardial and abdominal visceral adipose tissue were associated with CT measures of early lung injury and lower FVC in a cohort of community-dwelling adults. Adipose tissue may represent a modifiable risk factor for ILD.
Key Words: high-attenuation areas, IL-6, interstitial lung abnormalities, interstitial lung disease, leptin, obesity
Abbreviations: CRP, C-reactive protein; HAA, high-attenuation area; HU, Hounsfield unit; ILA, interstitial lung abnormalities; ILD, interstitial lung disease; IPF, idiopathic pulmonary fibrosis; IQR, interquartile range; MCP-1, monocyte chemoattractant protein-1; MESA, Multi-Ethnic Study of Atherosclerosis; PAT, pericardial adipose tissue volume; SAT, subcutaneous adipose tissue area; TNF-α, tumor necrosis factor α; VAT, visceral adipose tissue area
FOR EDITORIAL COMMENT, SEE PAGE 400
The prevalence of obesity is rapidly increasing in the United States and is expected to affect 40% of the population by 2030.1 Obesity, particularly visceral obesity, is associated with restrictive ventilatory defects in older adults2,3 and a greater rate of decline in lung function in the general population.4 This does not appear to be exclusively mediated by mechanical factors5 and may reflect direct injury by circulating inflammatory cytokines or adipokines.6, 7, 8, 9 A better understanding of the role of obesity in lung injury and early fibrosis may identify a novel target for the prevention and treatment of interstitial lung disease (ILD).
In states of obesity, adipocytes undergo hypertrophy to store excess fatty acids. Adipocyte hypertrophy results in recruitment of proinflammatory macrophages, and increased production of IL-6, leptin, and resistin.10, 11, 12 All of these mediators have been shown to contribute to lung injury in animal models of pulmonary fibrosis.7,13, 14, 15 However, a direct link between adiposity and lung injury, remodeling, and early fibrosis in humans has not been previously established.
Prior work evaluating the association between BMI and early parenchymal lung injury is inconsistent. Greater BMI may be associated with increased high-attenuation areas,16 but there is no consistent association between BMI and interstitial lung abnormalities.17 BMI is a poor measure of adiposity.18 It varies significantly by sex and race/ethnicity19 and provides no insight into mechanistic links between obesity and early parenchymal lung injury. In contrast, CT scanning can measure adipose tissue depot size and has been used to establish mechanistic links between adiposity and other forms of lung injury.20
We sought to determine whether adipose tissue depot size was associated with two CT measures of early lung injury and fibrosis, and FVC, in a large cohort of community-dwelling adults. We and others have previously used and validated both an automated, quantitative measure termed high-attenuation areas (HAAs) and a qualitative measure termed interstitial lung abnormalities (ILAs).16,21,22 Both measures have been used to identify novel risk factors for early ILD.14,16,17,21,22 Here, we hypothesized that greater amounts of pericardial, abdominal visceral, and abdominal subcutaneous adipose tissue would be associated with increased HAAs, greater odds of ILAs, and reduced FVC. We secondarily hypothesized that the association between adipose depots and both HAAs and ILAs would be partially mediated through circulating markers of obesity (leptin, adiponectin, resistin) and inflammation (IL-6, C-reactive protein [CRP], tumor necrosis factor [TNF]-α).
Methods
Study Participants
The National Heart, Lung, and Blood Institute-funded Multi-Ethnic Study of Atherosclerosis (MESA) is an ongoing multicenter prospective cohort study that enrolled 6,814 adults between the ages of 45 and 84 years without clinically evident cardiovascular disease. Enrollment occurred between 2000 and 2002, with six subsequent follow-up examinations (e-Table 1). MESA and its ancillary studies were approved by individual site institutional review boards (e-Appendix 1). All participants provided informed consent.
Pericardial Adipose Tissue Volume
Our primary exposure was pericardial adipose tissue (PAT) volume measured by cardiac CT imaging performed at MESA examination 1 (e-Table 1) on electron beam tomography scanners at three sites (Imatron C-150; Imatron, Inc.) and multidetector CT imaging at three sites (Volume Zoom [Siemens] or GE LightSpeed [GE Healthcare]), using previously published techniques.16,23 PAT was defined as all tissue with the density of adipose (–190 to –30 Hounsfield units [HU]), located between 15 mm above and 30 mm below the superior extent of the left coronary artery, bordered anteriorly by the chest wall, and posteriorly by the aorta and bronchus using volume analysis software (GE Healthcare). This limited region of PAT highly correlates with total volume of PAT (r = 0.93) with high intra- and interobserver reliability (κ = 0.999 and 0.997, respectively).23
Abdominal Adipose Tissue Area
Our secondary exposures were visceral adipose tissue (VAT) and subcutaneous adipose tissue (SAT) cross-sectional areas. At examinations 2 and 3 (2002-2005; e-Table 1), a random subset of 1,947 participants underwent abdominal imaging for measures of aortic calcium, using the scanners noted above. Using previously described techniques to interrogate these scans,24 abdominal adipose tissue cross-sectional area was measured at five locations between L2/L3 and L4/L5. Primary analyses were performed using areas at L4/L5, with sensitivity analyses performed at L3/L4.20,25 All tissue within the peritoneal cavity with the density of adipose tissue was identified as VAT. All tissue with the density of adipose and located outside of the peritoneal cavity but not within the muscle was identified as SAT. Using previously described methods, imputation was used to estimate missing SAT area on scans in which the subcutaneous tissue was incompletely imaged.26 These imputed SAT measures were used in primary analyses. Sensitivity analyses were performed, including only subjects with completely imaged SAT.
Biomarkers
All biomarkers were measured in serum samples collected between 2004 and 2005 (examination 3, e-Table 1) and stored in a –80°C freezer. Inflammatory biomarkers (IL-6, TNF-α, CRP) were measured in fasting samples, using an ultrasensitive enzyme-linked immunosorbent assay for IL-6 (Quantikine HS human IL-6 immunoassay; R&D Systems), Bio-Rad Luminex flow cytometry for TNF-α (Millipore), and a particle-enhanced immune photometric assay (BNII nephelometer for CRP; Dade-Behring, Inc). Adipokines (adiponectin, leptin, resistin) were measured by Bio-Rad Luminex flow cytometry (Millipore) at the University of Vermont (Burlington, VT).
Measures of Parenchymal Lung Injury
We examined two validated CT phenotypes of early parenchymal lung injury and fibrosis: a quantitative phenotype (HAAs), and a qualitative phenotype (ILAs).16,22,27 HAA was measured as all voxels of lung tissue with attenuation values between –600 and –250 HU on examination 1 (cardiac CT imaging). This range captures ground-glass and reticular changes while excluding denser areas of consolidation, nodules, and vasculature.16 ILA was visually assessed on examination 5 (full lung CT imaging) by one of five trained radiologists and defined according to Fleischner Society guidelines as the presence of ground glass, reticular changes, emphysematous cysts, honeycombing, or traction bronchiectasis affecting > 5% of any lung zone in a nondependent fashion.16,27,28 Radiologists were blinded to measures of adiposity.
Spirometry
At examinations 3 and 4 (2004-2007; e-Table 1), spirometry was performed in accordance with the American Thoracic Society/European Respiratory Society guidelines.29
Statistical Analysis
The distributions of adipose depot measures and serum biomarkers were right-skewed, and therefore they were log2 transformed. Changes in the outcome are reported per doubling of the predictor. The distribution of HAA was right-skewed; transformation to –1/HAA2 for regression modeling purposes has been validated and is reported here as HAA.30
We performed Spearman correlations between adipose tissue depot size and height, weight, and BMI to confirm validity. Given that nonlinear associations are often identified in studies of obesity,31,32 we tested for nonlinear relationships between predictors and outcomes using generalized additive models with the “gam” function in R with a loess smoothing function. Associations appeared to be linear, and therefore we used linear regression to examine the cross-sectional associations of adipose tissue depots with HAA and FVC. We used logistic regression to examine the associations of adipose depots with ILA. HAA models were adjusted for factors known to influence HAA measurement, including the following: study site, imaged lung volume, and radiation dose (milliamperes). We adjusted for potential confounders including age, sex, race/ethnicity, height, smoking status, cigarette pack-years, and percent emphysema (percent voxels with density less than 950 HU) in fully adjusted models.16 A priori subgroup analyses were performed by sex, age (< 65 or ≥ 65 years), BMI, race, and smoking status using Wald and likelihood ratio tests.
To account for mechanical effects of adiposity (ie, atelectasis), we performed sensitivity analyses that (1) included BMI in models of PAT, and (2) included both VAT and SAT in the same model for assessments of abdominal adipose depots. Given high correlations between these measures, their inclusion in the same model may result in bias, and these estimates should be interpreted with caution.
We assessed the association between biomarkers of obesity (leptin, resistin, adiponectin) and inflammation (IL-6, CRP, TNF-α) with PAT volume and VAT area using Spearman correlations. For biomarkers that were significantly associated with adipose measures, we estimated the proportion of the total association explained by each biomarker, using multilevel mediation analyses with the ml_mediation package in STATA (StataCorp) with covariates noted above clustered by study site.
All analyses were performed with STATA/IC version 15.1 (StataCorp), and R version 3.3.1 (R Foundation for Statistical Computing).
Results
Study Participants
A total of 6,814 subjects were enrolled in MESA and underwent cardiac CT imaging performed at examination 1. For 6,784 subjects, measures of both HAA and PAT were available (e-Fig 1). Forty-seven percent of the subjects were male, the median (interquartile range [IQR]) age was 62 (53-70) years, 39% were white, 22% were Hispanic, the median BMI was 27.6 kg/m2 (IQR, 24.5-31.2), 50% were never smokers, 36% were former smokers, and median pack-years was 15 (IQR, 5-32) (e-Table 2). A total of 1,923 subjects underwent abdominal CT imaging at examination 2 or 3, with available measures of HAA (e-Fig 1). VAT measurement was available on all abdominal scans, and SAT was completely imaged on 1,388 (86%) scans and imputed on 234 scans (14%). Subjects in the lowest tertile of PAT were less likely to be male or white, had lower weight and BMI, and lower rates of smoking (Table 1). Median PAT, VAT, and SAT values for the PAT cohort were 71 cm3 (IQR, 49-100), 137 cm2 (IQR, 97-193), and 236 cm2 (IQR, 170-310), respectively, and were similar among subjects with available ILA and FVC (e-Table 2). Subjects with available measures of abdominal VAT, ILA, and FVC appeared similar to the full cohort on baseline characteristics (e-Table 2).
Table 1.
Baseline Characteristics by Tertile of Pericardial Adipose Tissue Volumea
| Characteristic | Lowest Tertile of PAT (n = 2,262) | Middle Tertile of PAT (n = 2,261) | Highest Tertile of PAT (n = 2,261) |
|---|---|---|---|
| Age, y | 57 (51-67) | 63 (54-70) | 65 (57-72) |
| Sex (male) | 762 (34) | 986 (44) | 1,444 (64) |
| Race | |||
| White | 794 (35) | 803 (36) | 1,018 (45) |
| Asian | 268 (12) | 320 (14) | 209 (9) |
| Black | 833 (37) | 628 (28) | 423 (19) |
| Hispanic | 367 (16) | 510 (23) | 611 (27) |
| Weight, kg | 60 (69-79) | 76 (67-88) | 87 (77-99) |
| Height, cm | 164 (158-171) | 165 (158-173) | 169 (161-176) |
| BMI, kg/m2 | 24.8 (22.5-28.0) | 27.5 (25.5-30.8) | 30.2 (27.4-34.0) |
| BMI category | |||
| < 18.5 | 53 (2) | 5 (0) | 0 (0) |
| 18.5-25.0 | 1,105 (49) | 601 (27) | 180 (8) |
| 25.0-30.0 | 778 (34) | 966 (43) | 910 (40) |
| 30-35 | 233 (10) | 468 (21) | 723 (32) |
| > 35 | 93 (4) | 221 (10) | 448 (20) |
| Smoking status | |||
| Never smoker | 1,239 (55) | 1,181 (52) | 981 (43) |
| Former smoker | 700 (31) | 780 (35) | 995 (44) |
| Current smoker | 316 (14) | 292 (13) | 278 (12) |
| Pack-years | 12 (3-25) | 15 (5-31) | 20 (6-38) |
| PAT volume, cm3 | 42 (34-49) | 71 (64-79) | 115 (100-142) |
| VAT area, cm2 | 91 (65-117) | 133 (105-172) | 198 (154-245) |
| SAT area, cm2 | 217 (145-283) | 239 (172-320) | 253 (200-336) |
Continuous variables are reported as median (interquartile range); categorical variables are reported as No. (%). PAT = pericardial adipose tissue; SAT = abdominal subcutaneous adipose tissue; VAT = abdominal visceral adipose tissue.
Missing values: Smoking status on 22 subjects, and cigarette pack-years on 94 subjects.
PAT was strongly correlated with abdominal VAT (ρ = 0.67), moderately correlated with BMI (ρ = 0.51) and weight (ρ = 0.49), and weakly correlated with abdominal SAT (ρ = 0.20) and height (ρ = 0.17) (e-Table 3). Abdominal VAT was moderately correlated with BMI (ρ = 0.61) and weight (ρ = 0.54), and weakly correlated with abdominal SAT (ρ = 0.33). Abdominal SAT was strongly correlated with BMI (ρ = 0.73) and moderately correlated with height (ρ = 0.46).
High-Attenuation Areas
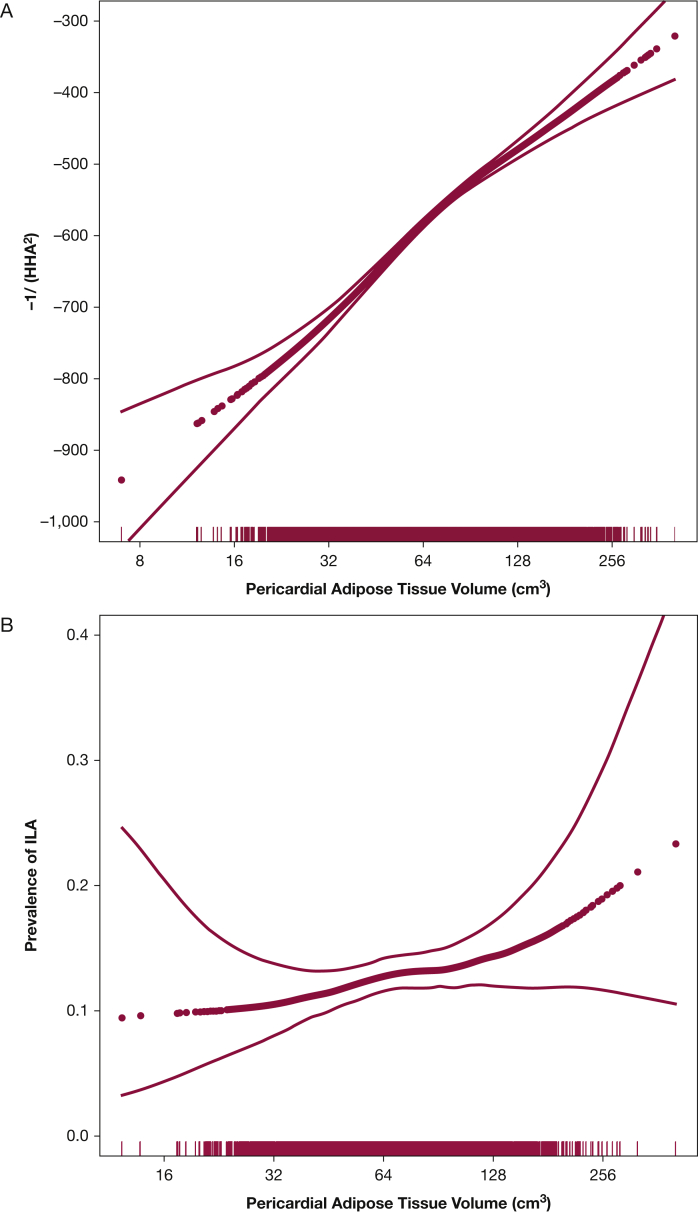
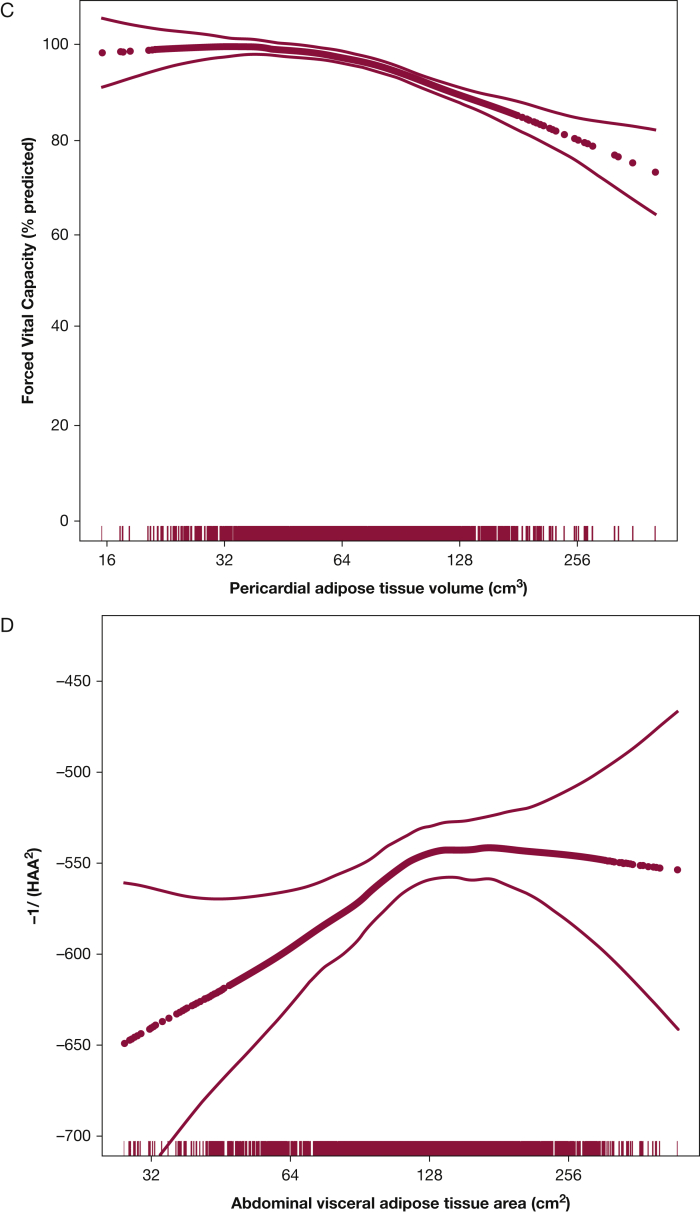
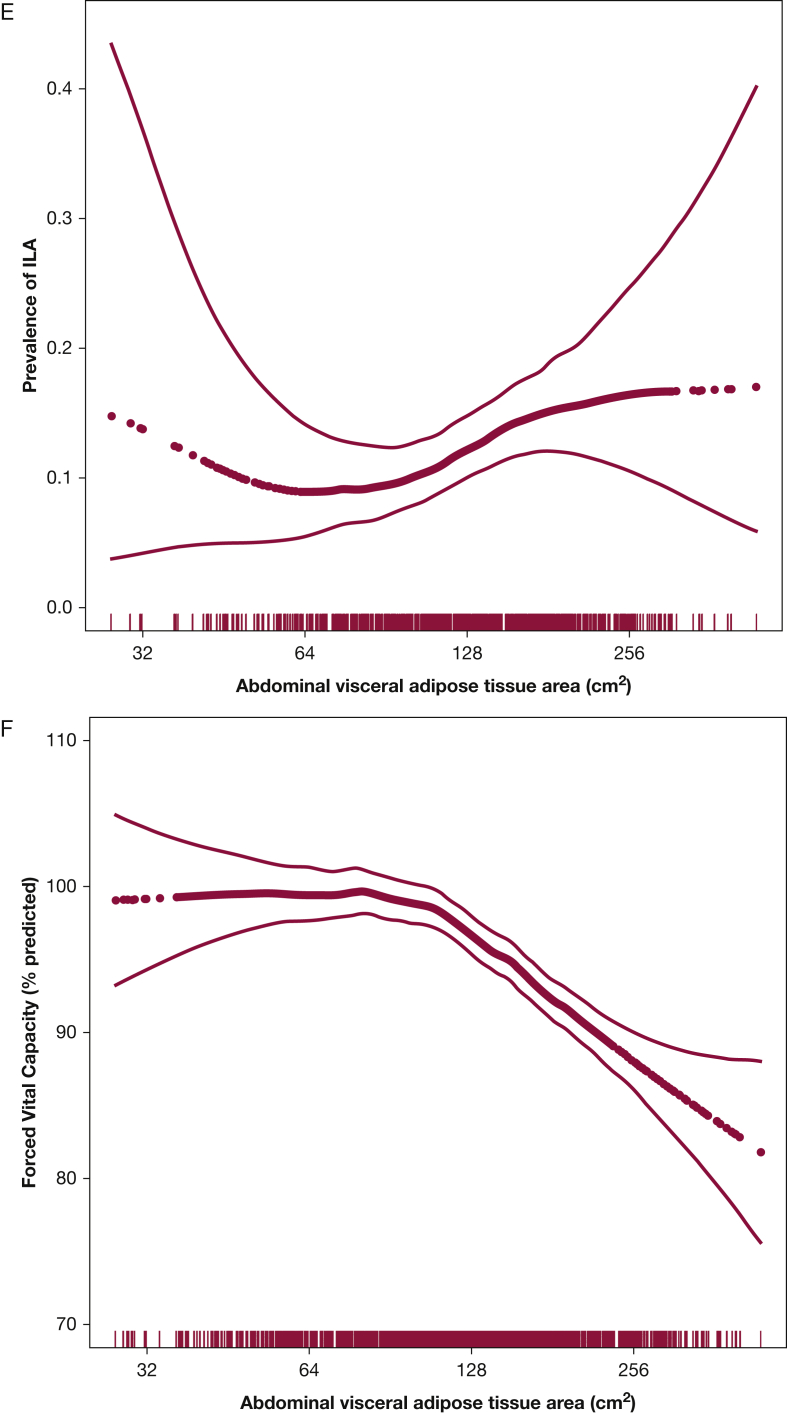
Every doubling in PAT volume was associated with 63.4-unit higher HAA (fully adjusted 95% CI, 55.5-71.3; P < .001) (Table 2, Fig 1A), and every doubling in abdominal VAT area was associated with 41.5-unit higher HAA (fully adjusted 95% CI, 28.3-54.7; P < .001) (Fig 1D). Abdominal SAT area was not associated with HAA in base models (9.3 units; 95% CI, –9.4 to 28.1; P = .33) (e-Fig 2A) but was associated with HAA in fully adjusted models (56.1 units; 95% CI, 38.8-73.5; P < .001).
Table 2.
Associations of Adipose Tissue Depots With High-Attenuation Areas, Interstitial Lung Abnormalities, and FVC
| Model | No. | Change in HAA per Doubling PAT Volume | 95% CI | P Value | No. | Odds of ILA per Doubling PAT Volume | 95% CI | P Value | No. | Change in FVC per Doubling PAT Volume | 95% CI | P Value |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PAT | ||||||||||||
| Model 1a | 6,784 | 82.0 | 73.4 to 90.5 | < .001 | 2,423 | 1.4 | 1.2 to 1.6 | < .001 | 1,415 | –4.1 | –5.2 to –2.9 | < .001 |
| Model 2b | 6,690 | 63.4 | 55.5 to 71.3 | < .001 | 2,394 | 1.2 | 0.98 to 1.5 | .07 | 1,400 | –5.5 | –6.8 to –4.3 | < .001 |
| Model 3c | 6,690 | 19.6 | 10.6 to 28.7 | < .001 | 2,394 | 1.3 | 1.01 to 1.6 | .04 | 1,400 | –4.3 | –5.8 to –2.7 | < .001 |
| No. | Change in HAA per Doubling VAT Area | 95% CI | P Value | No. | Odds of ILA per Doubling VAT Area | 95% CI | P Value | No. | Change in FVC per Doubling VAT Area | 95% CI | P Value | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VAT | ||||||||||||
| Model 1a | 1,923 | 57.4 | 42.6 to 77.1 | < .001 | 861 | 1.4 | 1.03 to 1.9 | .03 | 1,419 | –4.1 | –5.2 to –3.0 | < .001 |
| Model 2b | 1,838 | 41.5 | 28.3 to 54.7 | < .001 | 844 | 1.3 | 0.9 to 1.8 | .21 | 1,404 | –5.4 | –6.6 to – 4.3 | < .001 |
| Model 3c | 1,562 | 22.1 | 5.2 to 38.9 | .01 | 726 | 1.4 | 0.9 to 2.1 | .16 | 1,229 | –4.0 | -5.5 to –2.6 | < .001 |
| No. | Change in HAA per Doubling SAT Area | 95% CI | P Value | No. | Odds of ILA per Doubling SAT Area | 95% CI | P Value | No. | Change in FVC per Doubling SAT Area | 95% CI | P Value | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SAT | ||||||||||||
| Model 1a | 1,622 | 9.3 | –9.4 to 28.1 | .33 | 740 | 1.1 | 0.8 to 1.5 | .73 | 1,241 | –5.0 | –6.3 to –3.8 | < .001 |
| Model 2b | 1,605 | 56.1 | 38.8 to 73.5 | < .001 | 726 | 1.0 | 0.7 to 1.5 | .96 | 1,229 | –5.3 | –6.7 to –3.9 | < .001 |
| Model 3c | 1,562 | 41.2 | 21.5 to 61.0 | < .001 | 726 | 0.8 | 0.5 to 1.4 | .49 | 1,229 | –2.6 | –4.3 to –0.9 | .002 |
HAA = high-attenuation areas operationalized as (–1/percent HAA2); ILA = interstitial lung abnormalities; PAT = pericardial adipose tissue; SAT = subcutaneous adipose tissue; VAT = visceral adipose tissue.
Model 1: HAA analyses adjusted for study site, imaged lung volume, and radiation dose; ILA and FVC analyses are unadjusted.
Model 2: Model 1 + age at examination 1, sex, race, height, smoking status, percent emphysema, cigarette pack-years, and study site.
Model 3: Model 2 + BMI (in PAT models), SAT (in VAT models), or VAT (in SAT models).
Figure 1.
Associations between pericardial adipose tissue volume and (A) high-attenuation areas (P for association, <.001; P for nonlinearity, .02), (B) interstitial lung abnormalities (P for association, <.001; P for nonlinearity, .43), and (C) FVC (P for association, < .001; P for nonlinearity, .02). Association between abdominal visceral adipose tissue area and (D) high-attenuation areas (P for association, .002; P for nonlinearity, .13), (E) interstitial lung abnormalities (P for association, .04; P for nonlinearity, .15), and (F) FVC (P for association, .001; P for nonlinearity, .001). Models are adjusted for age, sex, and smoking status. Thick dotted black line represents the effect estimates. Surrounding thin lines represent 95% CIs. Vertical lines along the x axis each represent a single study subject.
Results were similar in sensitivity analyses adjusting for BMI and total adiposity (Table 2), among subjects with completely imaged abdominal SAT (e-Table 4), and using measures from the L3/L4 vertebral level (e-Table 5).
Interstitial Lung Abnormalities
Interstitial lung abnormalities were present in 294 participants (12%) after excluding those with scans indeterminate for ILA. Those with and without ILA had a mean (SD) BMI of 28.1 (4.8) kg/m2 and 28.1 (5.1) kg/m2, respectively. In fully adjusted models, every doubling in PAT volume was associated with 20% increased odds of ILA (OR, 1.2; 95% CI, 0.98-1.5; P = .07) (Table 2, Fig 1B). Similarly, every doubling in abdominal VAT area was associated with increased odds of ILA, although this was not statistically significant in fully adjusted models (OR, 1.3; 95% CI, 0.9-1.8; P = .21) (Fig 1E). There was no association between abdominal SAT and ILA (Table 2, e-Fig 2B). Similar associations were present in sensitivity analyses (Table 2, e-Tables 4 and 5).
Spirometry
Every doubling in PAT volume was associated with a 5.5% lower percent predicted FVC in fully adjusted models (95% CI, –6.8 to –4.3; P < .001) (Fig 1C). Similarly, greater abdominal VAT area and greater abdominal SAT area were associated with significantly lower FVC in unadjusted and fully adjusted models (Table 2, Fig 1F, e-Fig 2C). Associations were similar in sensitivity analyses (Table 2, e-Tables 4 and 5).
Subgroup Analyses
Sex modified the associations between PAT (P for interaction, < .001), abdominal VAT (P for interaction, .15), and HAA (Table 3). Associations were stronger in women, in whom every doubling in PAT volume was associated with 76.7-unit higher HAA (95% CI, 65.5-87.9) and every doubling in VAT area was associated with 52.5-unit higher HAA (95% CI, 33.9-71.2). Among men, every doubling in PAT volume was associated with 47.6-unit higher HAA (95% CI, 36.8-58.5), and every doubling in VAT was associated with 25.9-unit higher HAA (95% CI, 7.1-44.8). The association between PAT and ILA also appeared to be modified by sex (P for interaction, .01). In contrast to HAA, PAT seemed to be associated with a greater risk of ILA among men (OR, 1.5; 95% CI, 1.1-2.0) than among women (OR, 1.0; 95% CI, 0.8-1.3).
Table 3.
Associations of Pericardial Adipose Tissue Volume and Abdominal Visceral Adipose Tissue Area With High-Attenuation Areas, Interstitial Lung Abnormalities, and FVC Within Prespecified Subgroups by Sex
| Subgroup | No. | Change in HAA per Doubling in PAT Volume | 95% CI | P for Interaction | No. | Change in ILA per Doubling in PAT Volume | 95% CI | P for Interaction | No. | Change in Percent Predicted FVC per Doubling in PAT Volume | 95% CI | P for Interaction |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | < .001 | .01 | .18 | |||||||||
| Male | 3,144 | 47.6 | 36.8 to 58.5 | 1,125 | 1.5 | 1.1 to 2.0 | 722 | –6.7 | –8.3 to –5.0 | |||
| Female | 3,546 | 76.7 | 65.5 to 87.9 | 1,269 | 1.0 | 0.8 to 1.3 | 678 | –4.8 | –6.7 to –2.9 |
| Subgroup | No. | Change in HAA per Doubling in VAT Area | 95% CI | P for Interaction | No. | Change in ILA per Doubling in VAT Area | 95% CI | P for Interaction | No. | Change in Percent Predicted FVC per Doubling in VAT Area | 95% CI | P for Interaction |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | .15 | .11 | .10 | |||||||||
| Male | 921 | 25.9 | 7.1 to 44.8 | 424 | 1.6 | 0.8 to 2.7 | 725 | –6.6 | –8.3 to –4.9 | |||
| Female | 917 | 52.5 | 33.9 to 71.2 | 420 | 1.1 | 0.7 to 1.8 | 679 | –4.9 | –6.6 to –3.3 |
Models adjusted for study site, age, height, race, smoking status, cigarette pack-years, study site, percent emphysema (FVC and HAA models), imaged lung volume (HAA models), and radiation dose (HAA model). HAA = high-attenuation area; ILA = interstitial lung abnormalities; PAT = pericardial adipose tissue; VAT = visceral adipose tissue.
There did not appear to be effect modification by age (e-Tables 6-8). Smoking status may modify the association between PAT (P for interaction, .03), abdominal VAT (P for interaction, < .001), abdominal SAT (P for interaction, .005), and FVC with a stronger association between adiposity and FVC among ever smokers compared with never smokers (e-Tables 6-8). BMI may modify the association between abdominal SAT and HAA (P for interaction, < .001) with a strong effect among a small group of underweight subjects (e-Table 6).
Biomarkers
Biomarker measures were available for 1,468 subjects. As previously reported, there was a moderate correlation between PAT volume and IL-6 (e-Fig 3A).33,34 There was no correlation between PAT volume and CRP, adiponectin, or leptin (e-Figs 3B-3D). Abdominal VAT was moderately correlated with IL-6 (ρ = 0.32, P < .001) (e-Fig 4A) and leptin (ρ = 0.28, P < .001) (e-Fig 4B), weakly correlated with CRP (ρ = 0.25, P < .001) (e-Fig 4D), and moderately inversely correlated with adiponectin (ρ = –0.31, P < .001) (e-Fig 4C). Neither PAT volume nor abdominal VAT area was significantly associated with resistin or TNF-α (e-Figs 2D and 2F, e-Figs 3E and 3F).
In mediation analyses, IL-6 accounted for 8% of the total association between PAT and HAA and 17% of the total association between VAT and HAA (e-Table 9). Leptin accounted for 18% of the total association between VAT area and HAA, and CRP and adiponectin accounted for 11% and 4% of the total association, respectively.
Discussion
Greater amounts of pericardial and abdominal visceral adipose tissue were associated with greater high-attenuation areas on chest CT imaging, higher prevalence of interstitial lung abnormalities, and lower FVC among community-dwelling adults. These associations may vary by sex, and may be partially mediated by plasma IL-6 and leptin. The strength of our study lies in the consistency of our associations across three separate measures of early parenchymal lung injury and fibrosis in a large, multiethnic cohort of community-dwelling adults.
Our findings are consistent with epidemiologic studies demonstrating an association between diabetes, high-fat diet, and idiopathic pulmonary fibrosis (IPF), as well as known associations between greater pericardial adipose tissue volume or greater abdominal obesity with decreased FVC in the general population.2, 3, 4,35, 36, 37 Prior work has been limited to measures of pulmonary function, which are strongly influenced by the mechanical effects of obesity. We demonstrate that these restrictive ventilatory defects may not exclusively reflect the mechanical effects of obesity, but may also reflect parenchymal changes caused by systemic inflammation and circulating adipokines. BMI is a poor measure of adiposity and varies significantly by sex and race/ethnicity.18,19 The use of adipose depot size, rather than BMI,17 allowed us to identify a novel association between pericardial and visceral adiposity and subclinical ILD. Although mechanical factors may partially explain our findings, they are unlikely to represent the only mechanism linking adiposity and ILD. First, the strength of the association between adiposity and early parenchymal injury was greatest for PAT, which does not exert mechanical effects on the lung. Second, ILAs were qualitatively assessed and less prone to mismeasurement by body habitus. Third, significant associations between adiposity and early lung injury remain after controlling for measures of total adiposity and BMI. Fourth, we demonstrated a significant association with circulating measures of inflammation, suggesting alternate mechanisms.
Multiple nonmechanical mechanisms may link adiposity and ILD. Obese adipose tissue is characterized by adipocyte hypertrophy, resulting in recruitment of proinflammatory adipose tissue macrophages,10 and production of IL-6 and monocyte chemoattractant protein-1 (MCP-1).8 Direct drainage of pericardial adipose into the pulmonary circulation may increase the potential for lung injury by adipose-derived inflammatory cytokines and adipokines.
IL-6 has been implicated in the pathogenesis of IPF. IL-6 and MCP-1 are increased in serum and BAL during acute exacerbations of IPF.6,9 IL-6 blockade attenuates fibrosis in mouse models of bleomycin-induced lung injury.13 MCP-1 is an important stimulus for recruitment of profibrotic macrophages and fibroblasts to the lung and has been targeted in the treatment of IPF.9,38 We found that IL-6 partially mediated the association between adipose depots and HAA, suggesting that inflammation may be an important link between adiposity and lung injury.
The adipokines leptin and resistin are produced by adipocytes in high-energy states11,12 and have been linked to lung injury. Leptin, which partially mediates the association between abdominal VAT and HAA in our analyses, is increased in the BAL fluid of patients with acute lung injury and may increase macrophage-mediated inflammatory cytokine production.39,40 In bleomycin-induced models of acute lung injury in mice, leptin resistance decreases fibrosis and inflammation.15,41 Notably, we failed to identify a significant association between adipose depots and serum resistin despite having previously demonstrated a significant association between resistin and HAA.14 This suggests that resistin may play an independent role in lung injury that does not simply reflect greater adiposity.42
This study has significant strengths. We used a large population-based multicenter multiethnic prospective cohort, making our findings generalizable to the greater population. We demonstrated consistent associations across two separate adipose depots and two separate measures of early parenchymal lung injury, as well as reduced FVC, suggesting reproducibility. Extensive sensitivity analyses demonstrated consistent effects.
There are also limitations to this study. First, ILA was measured at examination 5 whereas adipose tissue and lung function measures were from earlier examinations. Reassuringly, weight was highly correlated across study visits, suggesting that there were no significant changes in body composition over time. Second, although we identified a significant association between SAT and HAA in adjusted models, the absence of an association in unadjusted models suggests effect modification. This appears to be driven by a small number of subjects with BMI less than 18.5 kg/m2. Weight loss is associated with disease progression in IPF.43 Although our analyses suggest that greater SAT may be associated with greater HAA among underweight subjects, greater SAT may actually be associated with a higher FVC in this subgroup. Given the very small sample size, these results should be interpreted with caution. Further work should evaluate changes in adipose depot size in patients with IPF with weight loss. Third, we identified a significant association between abdominal SAT and FVC despite no clear association between SAT and subclinical ILD, suggesting that factors other than parenchymal changes may explain the association between adiposity and FVC. Fourth, although we replicated these findings across three measures of lung injury, we have not confirmed these findings in other cohorts. Fifth, ILAs were assessed by only one of five radiologists, introducing potential misclassification.
Interpretation
Greater amounts of pericardial and abdominal visceral adipose tissue were associated with interstitial lung abnormalities, increased high-attenuation areas, and decreased FVC in a large, diverse multicenter study. This association may be partially mediated by leptin and IL-6. Quantification of adipose tissue on CT imaging identified adiposity as a novel target for prevention of ILD and highlights the importance of the nonmechanical effects of adipose tissue on the lung. Further work should investigate potential roles for IL-6, MCP-1, and leptin in lung injury and fibrosis.
Take-home Points.
Study Question: Is adipose tissue involved in the pathogenesis of interstitial lung disease?
Results: Greater amounts of pericardial and abdominal visceral adipose tissue were associated with increased high-attenuation areas and interstitial lung abnormalities, and decreased forced vital capacity in a large multi-center cohort of community-dwelling adults.
Interpretation: Adipose tissue may represent a modifiable risk factor for interstitial lung disease.
Acknowledgments
Author contributions: M. R. A. takes responsibility for the data analysis and the content of this manuscript. Study design: M. R. A., J. S. K., A. P.; data acquisition, analysis, interpretation: M. R. A., J. S. K., M. A., J. T. G., E. A. H., J. D., R. G. B., A. P.; first draft of the manuscript: M. R. A., A. P.; final manuscript approval: all of the authors.
Financial/nonfinancial disclosures: The authors have reported to CHEST the following: M. R. A. receives grant support from the National Institutes of Health, the Parker B. Francis Foundation, the Stony Wold-Herbert Foundation, and the American Society of Transplantation. J. S. K. receives grant support from the Pulmonary Fibrosis Foundation. J. T. G. reports grants from Pfizer, personal fees from Gilead, personal fees from Eli Lilly, personal fees from AbbVie, personal fees from Bristol Myers Squibb, and personal fees from UCB outside of the submitted work; E. A. H. is a founder and shareholder of VIDA Diagnostics, a company commercializing lung image analysis software developed, in part, at the University of Iowa. R. G. B. receives grant support from the National Institutes of Health and the COPD Foundation. None declared: M. A., J. D., A. P.
Role of sponsors: The sponsors had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript.
Additional information: The e-Appendix, e-Figures, and e-Tables can be found in the Supplemental Materials section of the online article.
Footnotes
FUNDING/SUPPORT: The Multi-Ethnic Study of Atherosclerosis (MESA) is supported by the National Heart, Lung, and Blood Institute (NHLBI) in collaboration with MESA investigators. Support for MESA is provided by the NHLBI [contracts 75N92020D00001, HHSN268201500003sI, N01-HC-95159, 75N92020D00005, N01-HC-95160, 75N92020D00002, N01-HC-95161, 75N92020D00003, N01-HC-95162, 75N92020D00006, N01-HC-95163, 75N92020D00004, N01-HC-95164, 75N92020D00007, N01-HC-95165, N01-HC-95166, N01-HC-95167, N01-HC-95168, and N01-HC-95169], and by the National Center for Advancing Translational Sciences (NCATS) [grants UL1-TR-000040, UL1-TR-001079, and UL1-TR-001420]. This work was additionally supported by the NIH [K23 HL140199, K23 HL 150280], the Stony Wold-Herbert Foundation, the Parker B. Francis Foundation, and the Pulmonary Fibrosis Foundation.
Supplementary Data
References
- 1.Finkelstein E.A., Khavjou O.A., Thompson H. Obesity and severe obesity forecasts through 2030. Am J Prev Med. 2012;42(6):563–570. doi: 10.1016/j.amepre.2011.10.026. [DOI] [PubMed] [Google Scholar]
- 2.Leone N., Courbon D., Thomas F. Lung function impairment and metabolic syndrome: the critical role of abdominal obesity. Am J Respir Crit Care Med. 2009;179(6):509–516. doi: 10.1164/rccm.200807-1195OC. [DOI] [PubMed] [Google Scholar]
- 3.Kwack W.G., Kang Y.S., Jeong Y.J. Association between thoracic fat measured using computed tomography and lung function in a population without respiratory diseases. J Thorac Dis. 2019;11(12):5300–5309. doi: 10.21037/jtd.2019.11.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Peralta G.P., Marcon A., Carsin A.E. Body mass index and weight change are associated with adult lung function trajectories: the prospective ECRHS study. Thorax. 2020;75(4):313–320. doi: 10.1136/thoraxjnl-2019-213880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fimognari F.L., Pasqualetti P., Moro L. The association between metabolic syndrome and restrictive ventilatory dysfunction in older persons. J Gerontol A Biol Sci Med Sci. 2007;62(7):760–765. doi: 10.1093/gerona/62.7.760. [DOI] [PubMed] [Google Scholar]
- 6.Papiris S.A., Tomos I.P., Karakatsani A. High levels of IL-6 and IL-8 characterize early-on idiopathic pulmonary fibrosis acute exacerbations. Cytokine. 2018;102:168–172. doi: 10.1016/j.cyto.2017.08.019. [DOI] [PubMed] [Google Scholar]
- 7.Chu S.G., Villalba J.A., Liang X. Palmitic acid-rich high-fat diet exacerbates experimental pulmonary fibrosis by modulating endoplasmic reticulum stress. Am J Respir Cell Mol Biol. 2019;61(6):737–746. doi: 10.1165/rcmb.2018-0324OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bruun J.M., Lihn A.S., Pedersen S.B., Richelsen B. Monocyte chemoattractant protein-1 release is higher in visceral than subcutaneous human adipose tissue (AT): implication of macrophages resident in the AT. J Clin Endocrinol Metab. 2005;90(4):2282–2289. doi: 10.1210/jc.2004-1696. [DOI] [PubMed] [Google Scholar]
- 9.Suga M., Iyonaga K., Ichiyasu H., Saita N., Yamasaki H., Ando M. Clinical significance of MCP-1 levels in BALF and serum in patients with interstitial lung diseases. Eur Respir J. 1999;14(2):376–382. doi: 10.1034/j.1399-3003.1999.14b23.x. [DOI] [PubMed] [Google Scholar]
- 10.Weisberg S.P., McCann D., Desai M., Rosenbaum M., Leibel R.L., Ferrante A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796–1808. doi: 10.1172/JCI19246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maffei M., Halaas J., Ravussin E. Leptin levels in human and rodent: measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat Med. 1995;1(11):1155–1161. doi: 10.1038/nm1195-1155. [DOI] [PubMed] [Google Scholar]
- 12.Steppan C.M., Bailey S.T., Bhat S. The hormone resistin links obesity to diabetes. Nature. 2001;409(6818):307–312. doi: 10.1038/35053000. [DOI] [PubMed] [Google Scholar]
- 13.Le T.T., Karmouty-Quintana H., Melicoff E. Blockade of IL-6 trans signaling attenuates pulmonary fibrosis. J Immunol. 2014;193(7):3755–3768. doi: 10.4049/jimmunol.1302470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim J.S., Anderson M.R., Podolanczuk A.J. Associations of serum adipokines with subclinical interstitial lung disease among community-dwelling adults: the Multi-Ethnic Study of Atherosclerosis (MESA) Chest. 2020;157(3):580–589. doi: 10.1016/j.chest.2019.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bellmeyer A., Martino J.M., Chandel N.S., Scott Budinger G.R., Dean D.A., Mutlu G.M. Leptin resistance protects mice from hyperoxia-induced acute lung injury. Am J Respir Crit Care Med. 2007;175(6):587–594. doi: 10.1164/rccm.200603-312OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Podolanczuk A.J., Oelsner E.C., Barr R.G. High attenuation areas on chest computed tomography in community-dwelling adults: the MESA study. Eur Respir J. 2016;48(5):1442–1452. doi: 10.1183/13993003.00129-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Putman R.K., Hatabu H., Araki T. Association between interstitial lung abnormalities and all-cause mortality. JAMA. 2016;315(7):672–681. doi: 10.1001/jama.2016.0518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shah N.R., Braverman E.R. Measuring adiposity in patients: the utility of body mass index (BMI), percent body fat, and leptin. PLoS One. 2012;7(4) doi: 10.1371/journal.pone.0033308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Heymsfield S.B., Peterson C.M., Thomas D.M., Heo M., Schuna J.M., Jr. Why are there race/ethnic differences in adult body mass index-adiposity relationships? A quantitative critical review. Obes Rev. 2016;17(3):262–275. doi: 10.1111/obr.12358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Anderson M.R., Udupa J.K., Edwin E. Adipose tissue quantification and primary graft dysfunction after lung transplantation: the Lung Transplant Body Composition study. J Heart Lung Transplant. 2019;38(12):1246–1256. doi: 10.1016/j.healun.2019.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hunninghake G.M., Hatabu H., Okajima Y. MUC5B promoter polymorphism and interstitial lung abnormalities. N Engl J Med. 2013;368(23):2192–2200. doi: 10.1056/NEJMoa1216076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Podolanczuk A.J., Oelsner E.C., Barr R.G. High-attenuation areas on chest computed tomography and clinical respiratory outcomes in community-dwelling adults. Am J Respir Crit Care Med. 2017;196(11):1434–1442. doi: 10.1164/rccm.201703-0555OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ding J., Kritchevsky S.B., Harris T.B. The association of pericardial fat with calcified coronary plaque. Obesity (Silver Spring) 2008;16(8):1914–1919. doi: 10.1038/oby.2008.278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rao V.N., Zhao D., Allison M.A. Adiposity and incident heart failure and its subtypes: MESA (Multi-Ethnic Study of Atherosclerosis) JACC Heart Fail. 2018;6(12):999–1007. doi: 10.1016/j.jchf.2018.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McClellan T., Allen B.C., Kappus M. Repeatability of computerized tomography-based anthropomorphic measurements of frailty in patients with pulmonary fibrosis undergoing lung transplantation. Curr Probl Diagn Radiol. 2017;46(4):300–304. doi: 10.1067/j.cpradiol.2016.12.009. [DOI] [PubMed] [Google Scholar]
- 26.Mongraw-Chaffin M., Allison M.A., Burke G.L. CT-derived body fat distribution and incident cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. J Clin Endocrinol Metab. 2017;102(11):4173–4183. doi: 10.1210/jc.2017-01113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Washko G.R., Hunninghake G.M., Fernandez I.E. Lung volumes and emphysema in smokers with interstitial lung abnormalities. N Engl J Med. 2011;364(10):897–906. doi: 10.1056/NEJMoa1007285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hatabu H., Hunninghake G.M., Richeldi L. Interstitial lung abnormalities detected incidentally on CT: a position paper from the Fleischner Society. Lancet Respir Med. 2020;8(7):726–737. doi: 10.1016/S2213-2600(20)30168-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miller M.R., Hankinson J., Brusasco V. American Thoracic Society/European Respiratory Society (ATS/ERS) Task Force. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. doi: 10.1183/09031936.05.00034805. [DOI] [PubMed] [Google Scholar]
- 30.Easthausen I., Podolanczuk A., Hoffman E. Reference values for high attenuation areas on chest CT in a healthy, never-smoker, multi-ethnic sample: the MESA study. Respirology. 2020;25(8):855–862. doi: 10.1111/resp.13783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Anderson M.R., Geleris J., Anderson D.R. Body mass index and risk for intubation or death in SARS-CoV-2 infection: a retrospective cohort study. Ann Intern Med. 2020;173(10):782–790. doi: 10.7326/M20-3214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Anderson M.R., Kolaitis N.A., Gao Y. A nonlinear relationship between visceral adipose tissue and frailty in adult lung transplant candidates. Am J Transplant. 2019;19(11):3155–3161. doi: 10.1111/ajt.15525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McClain J., Hsu F., Brown E. Pericardial adipose tissue and coronary artery calcification in the Multi-Ethnic Study of Atherosclerosis (MESA) Obesity (Silver Spring) 2013;21(5):1056–1063. doi: 10.1002/oby.20090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ong K.L., Ding J., McClelland R.L. Relationship of pericardial fat with biomarkers of inflammation and hemostasis, and cardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis. 2015;239(2):386–392. doi: 10.1016/j.atherosclerosis.2015.01.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hickson D.A., Liu J., Bidulescu A., Burchfiel C.M., Taylor H.A., Petrini M.F. Pericardial fat is associated with impaired lung function and a restrictive lung pattern in adults: the Jackson Heart Study. Chest. 2011;140(6):1567–1573. doi: 10.1378/chest.11-0258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Miyake Y., Sasaki S., Yokoyama T. Dietary fat and meat intake and idiopathic pulmonary fibrosis: a case-control study in Japan. Int J Tuberc Lung Dis. 2006;10(3):333–339. [PubMed] [Google Scholar]
- 37.Kim Y.J., Park J.W., Kyung S.Y. Clinical characteristics of idiopathic pulmonary fibrosis patients with diabetes mellitus: the national survey in Korea from 2003 to 2007. J Korean Med Sci. 2012;27(7):756–760. doi: 10.3346/jkms.2012.27.7.756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Moore B.B., Kolodsick J.E., Thannickal V.J. CCR2-mediated recruitment of fibrocytes to the alveolar space after fibrotic injury. Am J Pathol. 2005;166(3):675–684. doi: 10.1016/S0002-9440(10)62289-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gainsford T., Willson T.A., Metcalf D. Leptin can induce proliferation, differentiation, and functional activation of hemopoietic cells. Proc Natl Acad Sci U S A. 1996;93(25):14564–14568. doi: 10.1073/pnas.93.25.14564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ubags N.D., Vernooy J.H., Burg E. The role of leptin in the development of pulmonary neutrophilia in infection and acute lung injury. Crit Care Med. 2014;42(2):e143–e151. doi: 10.1097/CCM.0000000000000048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jain M., Budinger G.R., Lo A. Leptin promotes fibroproliferative acute respiratory distress syndrome by inhibiting peroxisome proliferator-activated receptor-γ. Am J Respir Crit Care Med. 2011;183(11):1490–1498. doi: 10.1164/rccm.201009-1409OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jiang S., Park D.W., Tadie J.M. Human resistin promotes neutrophil proinflammatory activation and neutrophil extracellular trap formation and increases severity of acute lung injury. J Immunol. 2014;192(10):4795–4803. doi: 10.4049/jimmunol.1302764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nakatsuka Y., Handa T., Kokosi M. The clinical significance of body weight loss in idiopathic pulmonary fibrosis patients. Respiration. 2018;96(4):338–347. doi: 10.1159/000490355. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.