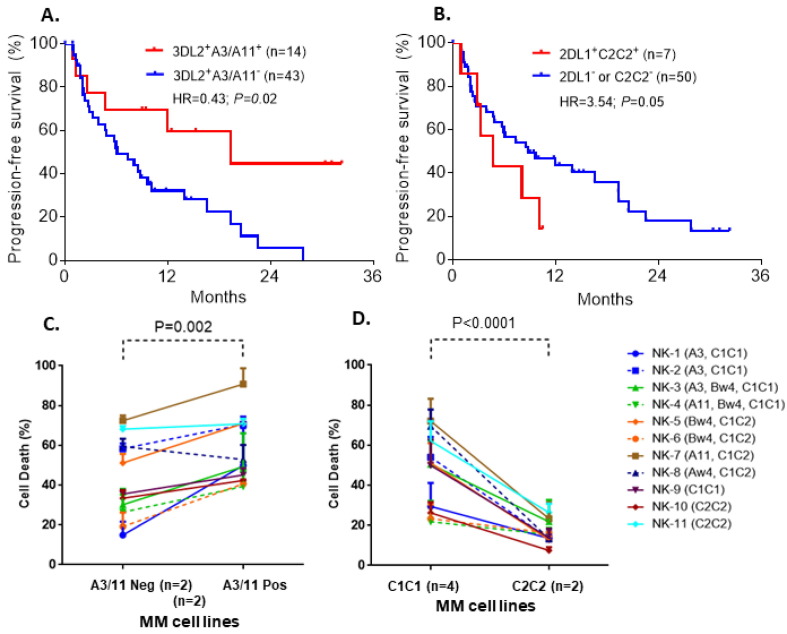
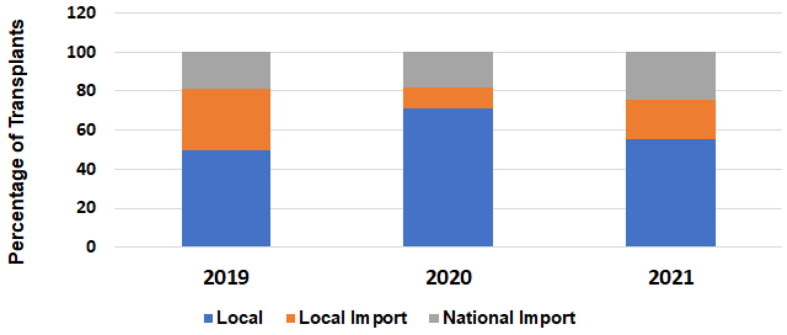
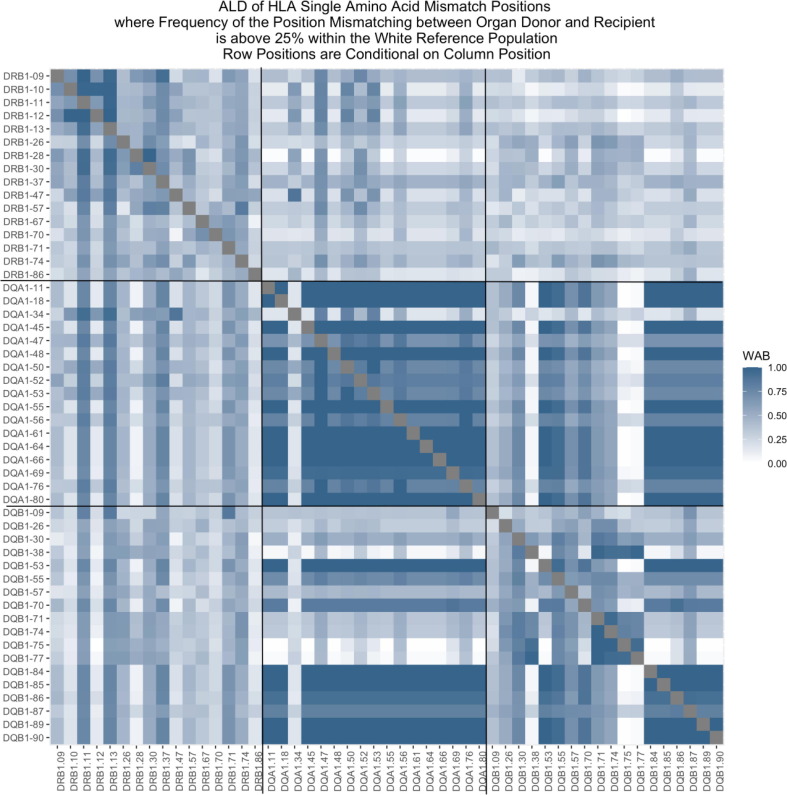
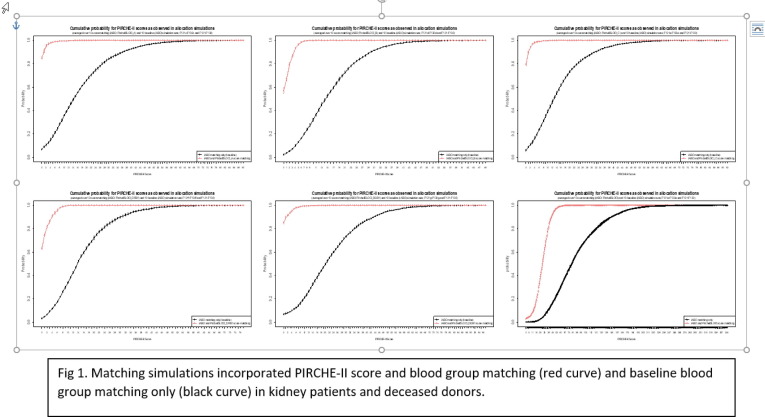
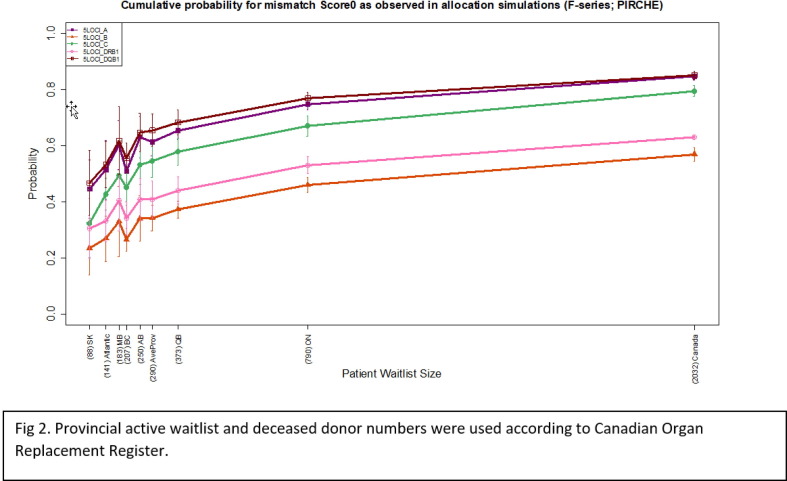
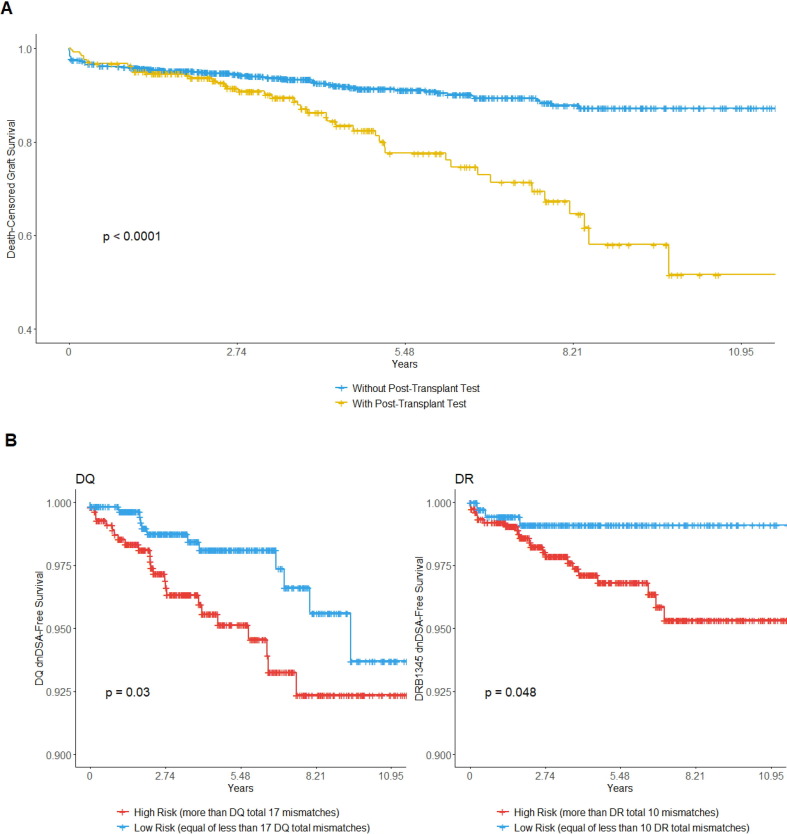
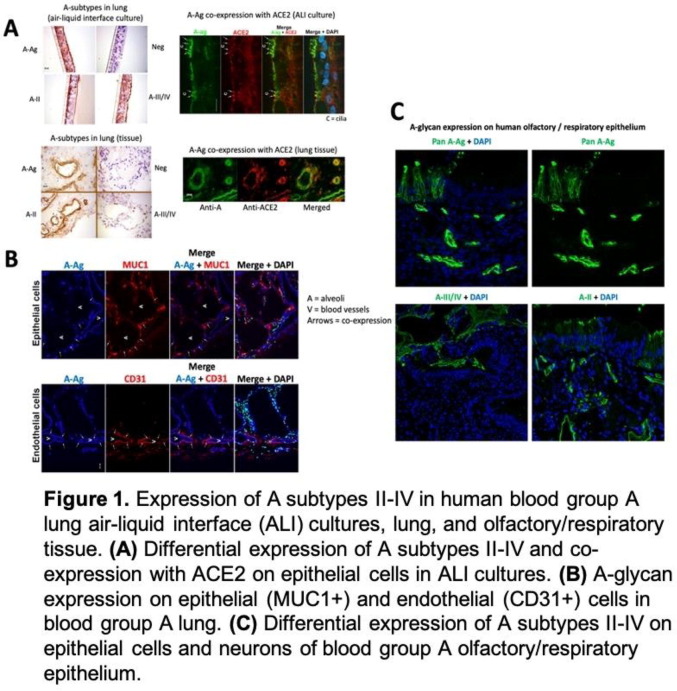
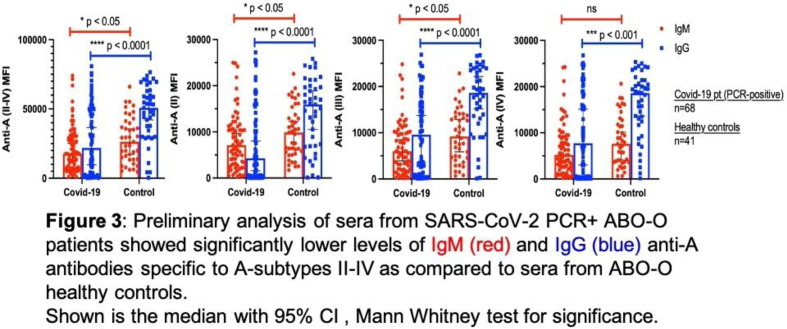
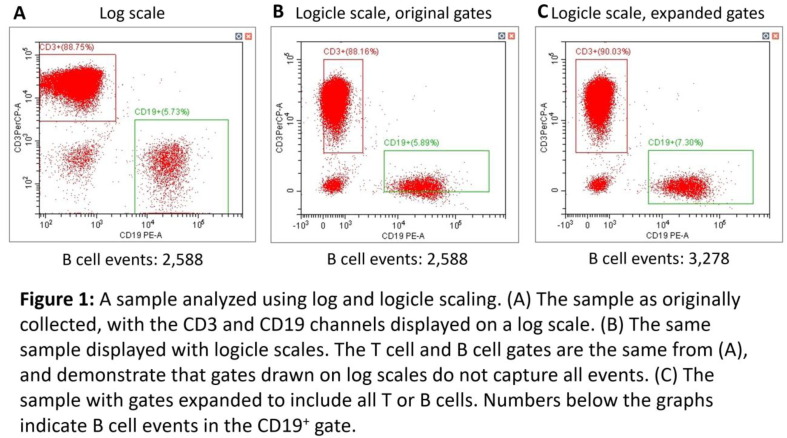
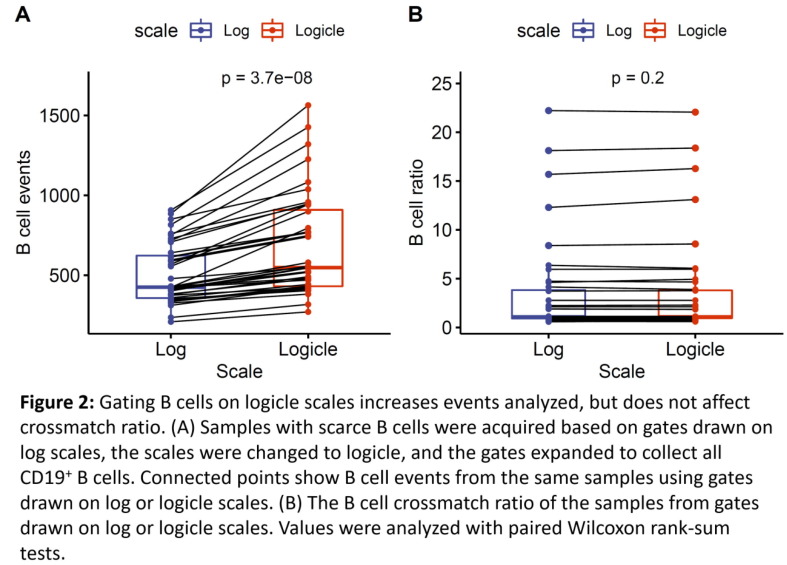
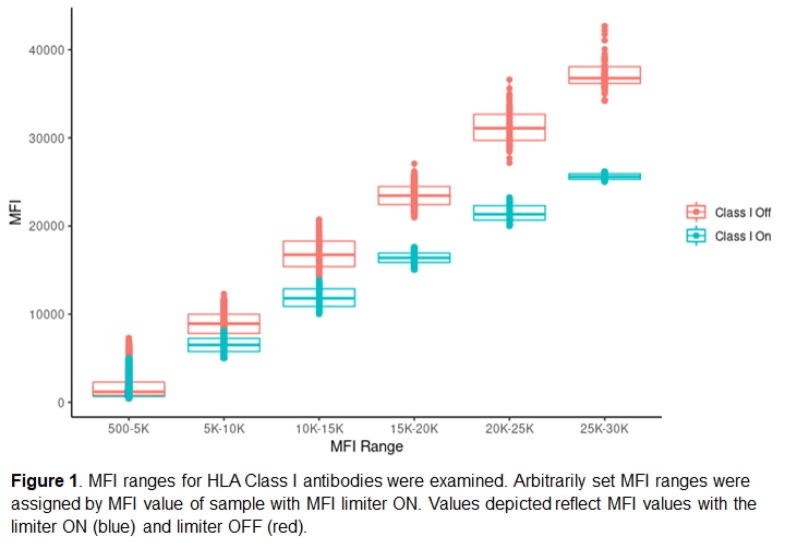
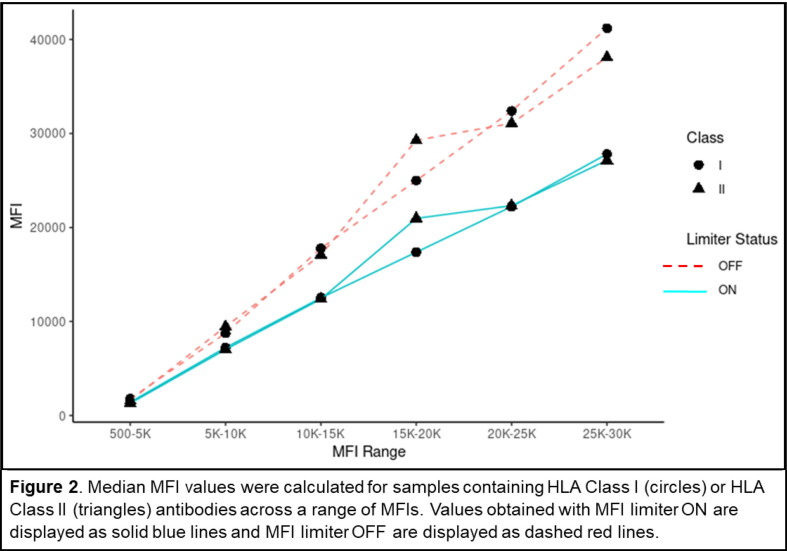
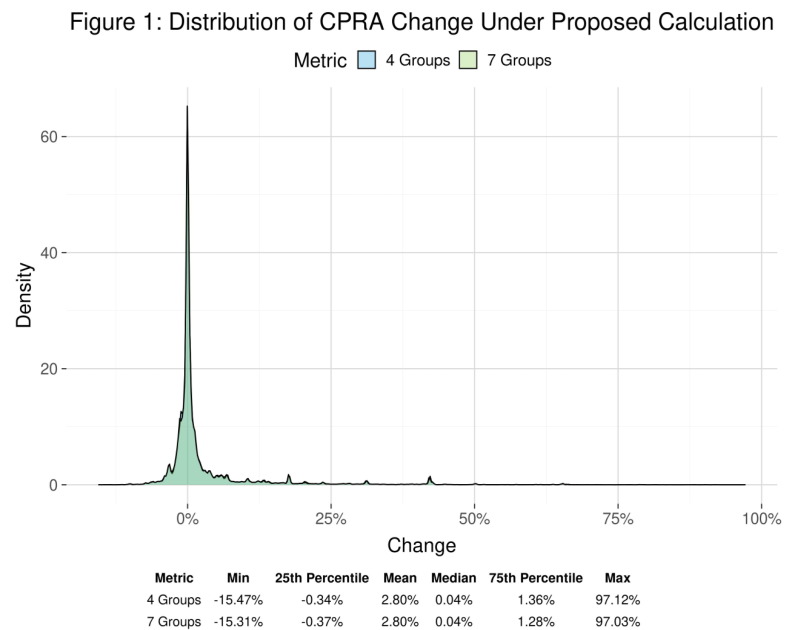
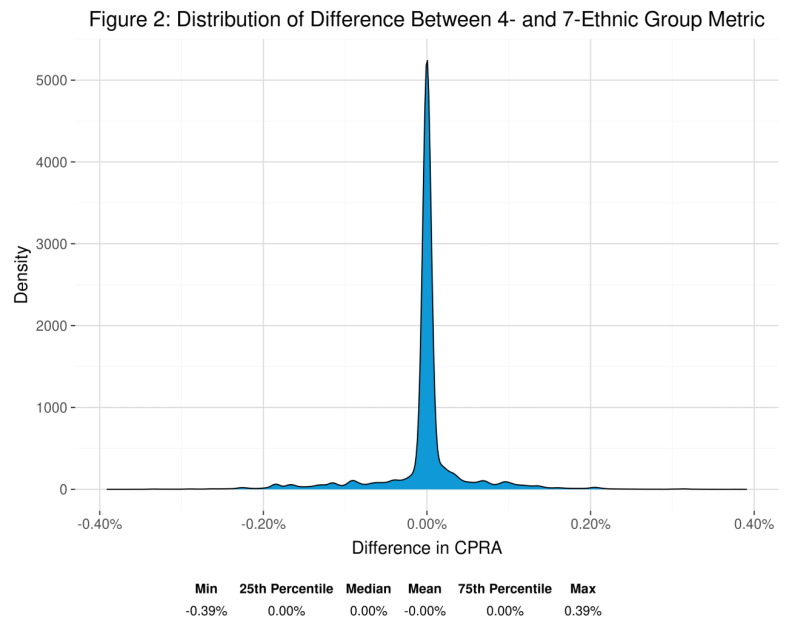
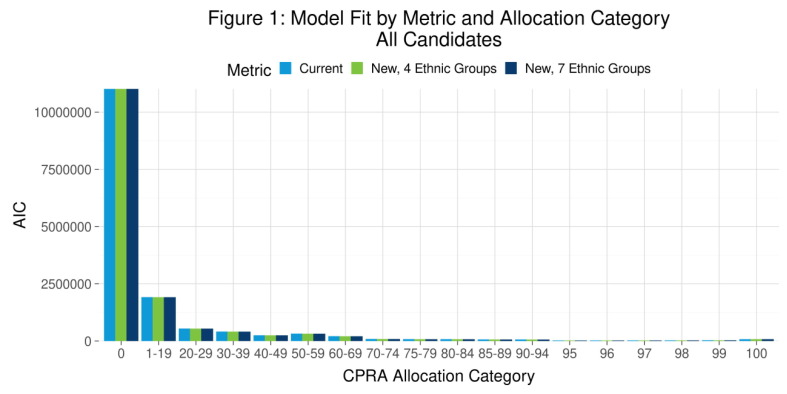
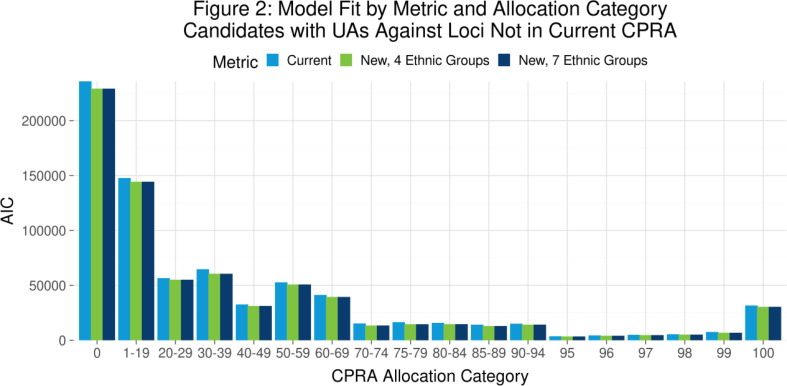
P01
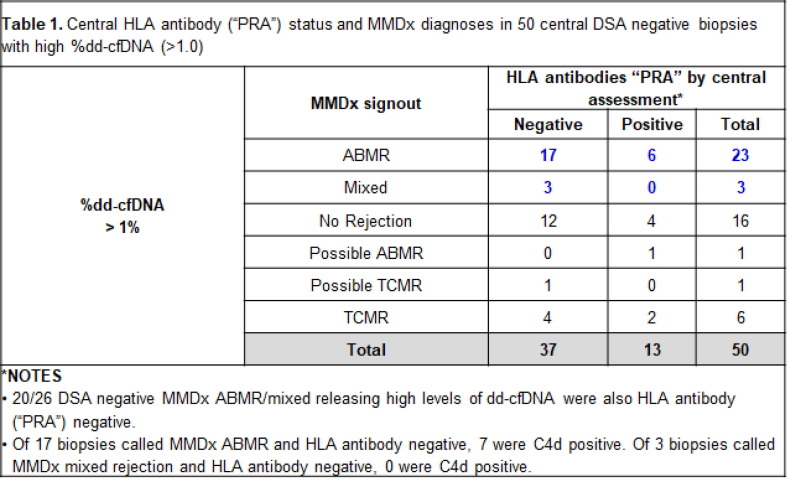
Case studies
Title: POSITIVE FLOW CYTOMETRY CROSSMATCH WITH DISCREPANT ANTIBODY TESTING RESULTS FOLLOWING COVID-19 VACCINATION
Authors: <u>Q. Xu</u>, P. Sood, D. Helmick, J. Lomago, A. Tevar, A. Zeevi, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, UNITED STATES|
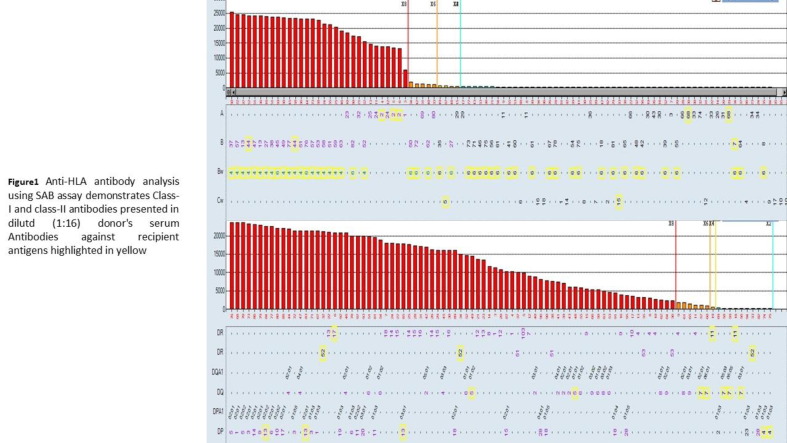
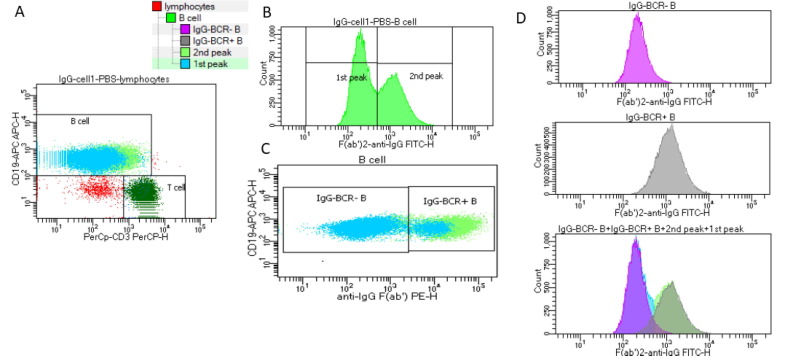
Aim: The impact of COVID-19 vaccination on the alloimmunity of transplant candidates is unknown. We report a case of unexpected positive B-cell flow cytometry crossmatch (FCXM) following COVID-19 vaccination in a patient waiting for second kidney transplantation. Methods: The standard 3-color FCXM was performed with pronase-treated lymphocytes. Solid-phase assays, including single-antigen and multi-antigen beads from two vendors (One Lambda and Immucor) were used to determine the specificity of anti-HLA antibodies. Results: The preliminary B-cell FCXM, using serum samples collected before receiving a two-dose COVID-19 vaccine (Pfizer-BioNTech), were negative. When a serum collected 37-days post-vaccination was used in the final FCXM, there was a surprisingly strong positive FCXM on B cells (Table 1). The patient had no recent significant medical/surgical events, nor did he receive any blood transfusion or therapeutical antibodies such as Rituxan. T cell FCXM were negative with both sera, so did the anti-class I antibodies. With the sensitive single-antigen beads assays, no HLA class II donor-specific antibody (DSA) was detected in both sera (Fig. 1). Possible prozone effects were excluded using treatment of EDTA, heat, or dilutions. No DSA was found in the C1q assay (One Lambda), or a single-antigen beads assay from an alternative vendor (Immucor), or a new Reflex beads (One Lambda). However, DSA to HLA-DR7 were clearly identified with multi-antigen PRA beads in the serum post-vaccination (Fig. 2) but not pre-vaccination (Fig. 3). The B cell FCXM were strongly positive with HLA-DR7-positive surrogate donor # 4 & #5, while negative with HLA-DR7-negative surrogate donor # 1–3 (Table 1), which confirms the HLA-DR7 alloreactivity. The antigen configurations on B lymphocytes are similar to that on the multi-antigen beads while distinct to the single-antigen beads. HLA-DR7 was the repeating mismatched antigen with the failing first kidney allograft. The newly emerged antibody to HLA-DR7 probably is the consequence of bystander activation of memory response by the COVID-19 vaccination. Conclusion: This case highlights the importance of utilizing multiple assays, including multi-antigen beads and cell-based crossmatch. COVID-19 immunization may deserve special attention when assessing the immunological risk before and after organ transplantation.
Fig. 1. The post-vaccination serum was tested with the single-antigen beads. The profile was similar when the pre-vaccination serum was tested with the single-antigen beads
Fig. 2. The post-vaccination serum tested with the multi-antigen PRA beads; mismatched donor HLA-DR7 was highlighted in the blue box.
Fig. 3. The pre-vaccination serum tested with the multi-antigen PRA beads; mismatched donor HLA-DR7 was highlighted in the blue box
P02
Case studies
Title: A MULTI-PLATFORM APPROACH TO DETERMINE WHICH HLA ANTIGENS SHOULD BE CONSIDERED UNACCEPTABLE
Authors: <u>D.H. Fallon</u>, M. Kincade, N. Higgins, W. Goggins, Transplant Immunology, IU Health, Indianapolis, Indiana, UNITED STATES|
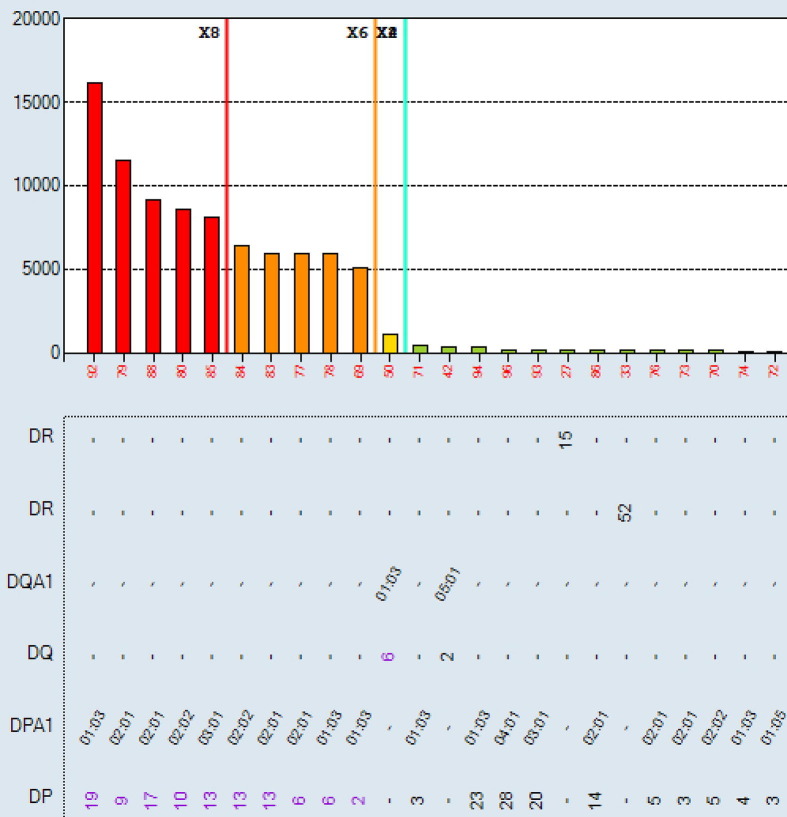
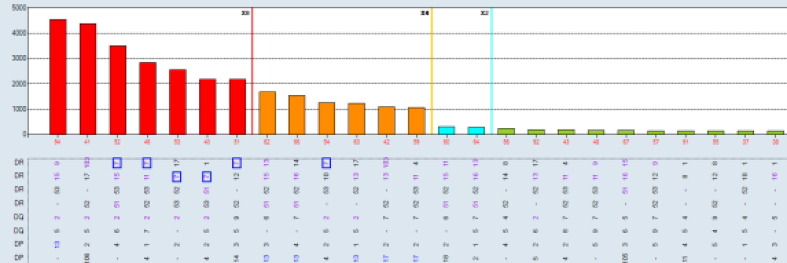
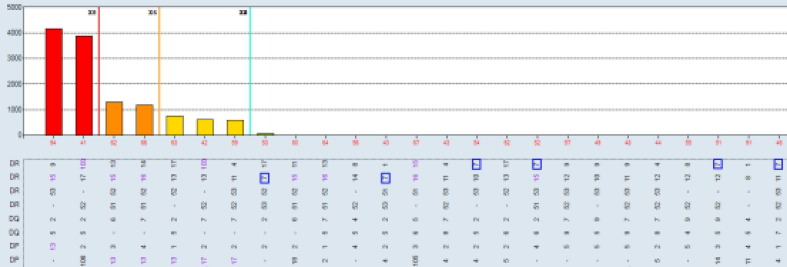
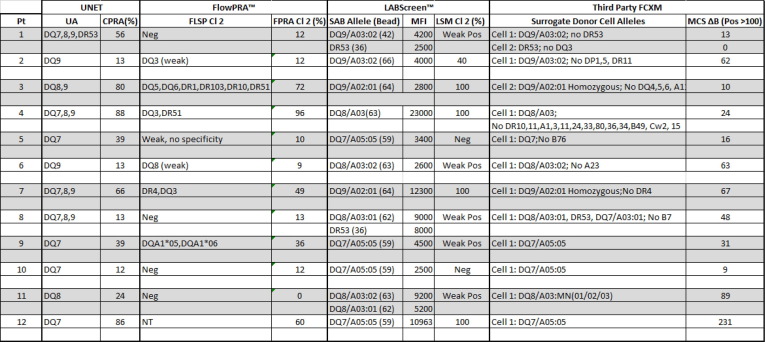
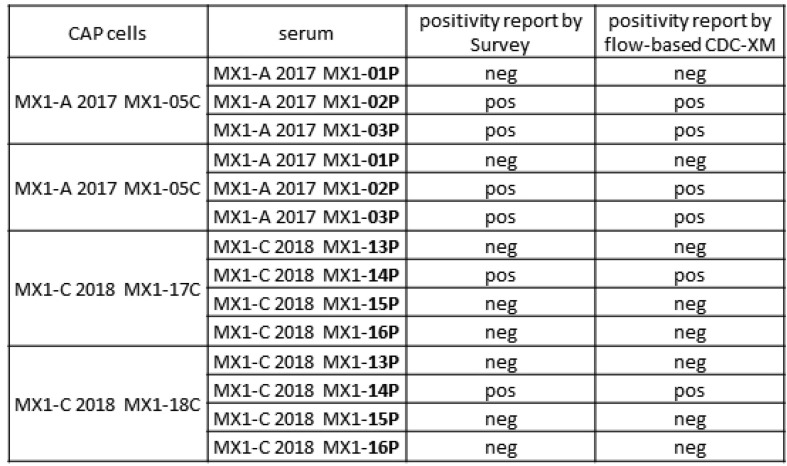
Aim: Single antigen bead (SAB) assays are used to detect and measure the strength of HLA Ab. However, it has become well established that detection of certain specificities are falsely positive.This study compares SAB data to other Ab testing platforms (One Lambda FlowPRA™ screening beads (FPRA) and Specificity Beads (FLSP), LABScreen™ SAB and Mixed beads (LSPRA) ) as a multipronged approach to list only those unacceptable antigens (UA) in UNET that are truly positive. Methods: The dataset includes all listed patients at our center with DQ3 and/or DR53 (total n = 39 patients; DQ3 only n = 27; DR53 only n = 5; Both DQ3 and DR53 n = 7). Patient were selected along the following parameters: <90% CPRA; no previous transplant with donor alleles DQ3 or DR53 (known sensitization to specific Ab) and SAB testing with threshold values >2500 MFI. Ab screening was performed on patient serum using four distinct platforms: FPRA, FLSP, SAB, and LSM. FCXM was acquired on a BD Lyric using surrogate donor cells selected for specific alleles (MCS Delta B cell pos >100). Results: Out of 12 pts with SAB MFI >2500: 11pts have neg FCXM results with donor surrogate cells (91.7%); 6 (of 11) pts are neg on FLSP (54.5%); 7 pts are neg or weakly pos on LSM (58.3%); and 1pt has a neg FPRA (8.3%). Using these results, UAs in UNET will be re-evaluated for 11pts that were previously listed by SAB results only. Conclusion: The SAB testing platform relies primarily on testing of microparticles coated with recombinat HLA antigens, while FPRA, LSM and FLSP all use target beads coated with native HLA antigens. While SAB testing is a revolutinary approach to HLA antibody identification, some specificities that are detected on the recombinant targets are falsely positive, perhaps due to denatured antigens occuring as part of the manufacturing process, or because peptides presented by certain HLA antigens are being detected as opposed to the HLA antigens themselves. Thus, while an extremely sensitive assay, the specificty of the SAB approach is subopitmal. As such, SAB testing should not be a stand alone test but combined with results from other solid phase and cell based assays where the target antigens are in their native form. Concordnace among the tests would provide more confidence that the antibodies detected are “true” positives. A multi-platform testing approach will allow for resolution on many of these cases.
P03
Case studies
Title: CHRONIC ANTIBODY-MEDICATED REJECTION IN A LIVER ALLOGRAFT RECIPIENT
Authors: <u>K. Gvozdjan</u>, J. Patel, Z. Du, Pathology and Laboratory Medicine, Loyola University Medical Center, Maywood, Illinois, UNITED STATES|
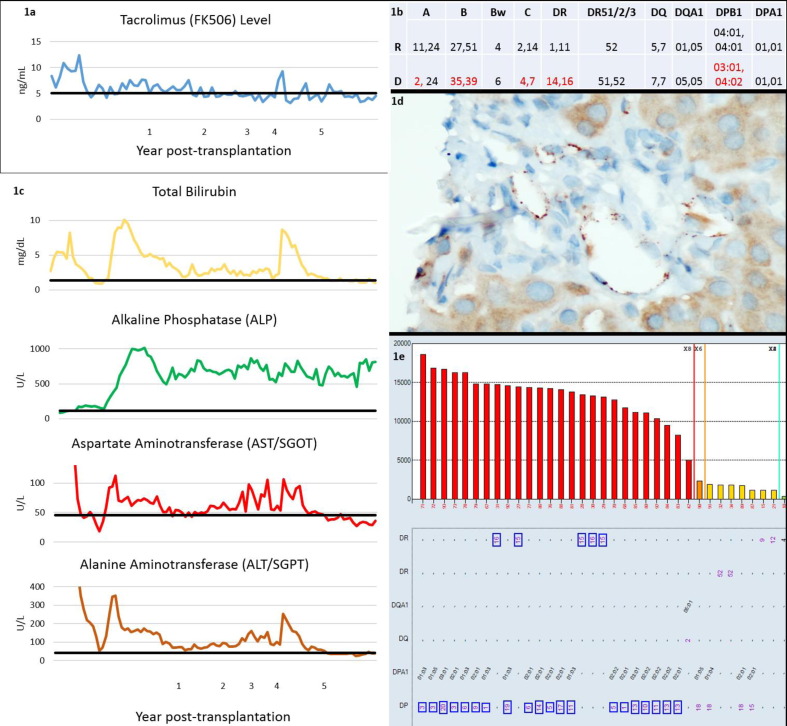
Aim: Antibody mediated rejection (AMR) can cause liver allograft loss; however, most transplantation programs do not routinely monitor donor-specific HLA antibodies (DSA) due to the known tolerance to DSA in the liver allograft. We, herein, present a case of a 70-year-old Caucasian male with history of NASH cirrhosis and hepatocellular carcinoma, 5.5 years status-post liver transplantation, on tacrolimus (Figure 1a) and mycophenolic acid, with clinically suspected chronic AMR with de novo DSA. The recipient (R) had 0% PRA pretransplantation and received an ABO compatible, deceased-donor (D) liver allograft with HLA mismatches as shown in Figure 1b. Follow-up DSA testing was not performed until his most recent visit, when the patient presented with a new-onset ascites requiring large volume paracentesis. Liver function tests suggested long-term injury to the biliary system and hepatocytes (Figure 1c). Liver allograft biopsy showed periportal fibrosis with focal bridging, mild portal inflammation with lymphoplasmacytic infiltration. Focal bile duct injury was present. C4d immunostain showed C4d expression in portal vascular endothelial cells (score 2 of 3, Figure 1d). Due to the suspicion for AMR, DSA testing was performed using Labscreen single antigen beads kits (ThermoFisher One Lambda, West Hills, CA). Anti-DR16 (MFI: 14,032) and -DP03:01 (MFI: 17,262) DSA were identified. Altogether, the patient's anti-HLA antibody profile showed strong anti-DR15, 16 and anti-DPB1*01,03,05,06,09,10,11,13,14,17,19,20 alloantibody reactivities (Figure 1e) that target 142 M3 and 84DEAV epitopes, respectively. DSA results supported the clinical and biopsy findings of chronic AMR. The patient remained on tacrolimus and mycophenolic acid immunosuppressive regimen, and repeat DSA test showed only a slight decrease in MFI of anti-DR16 (MFI: 11,949) and anti-DPB1*03:01 (MFI: 14,511) after one month. We, therefore, present the biochemical, immunologic, and pathologic changes in a case of chronic AMR of liver allograft that demonstrates the pathogenic potential of de novo DSA in this classically antibody tolerant organ, and suggest that routine DSA monitoring may be beneficial in liver allograft recipients. Methods: NA Results: NA Conclusion: NA
P04
Case studies
Title: AN HLA-B39-DONOR SPECIFIC ANTIBODY CAUSES KIDNEY TRANSPLANT FAILURE IN AN HLA-B39 PATIENT
Authors: <u>Z. Du</u>, J. Patel, R. Raz, B. Norton, I. Hamad, K. Gvozdjan, Pathology, Loyola University Medical Center, Maywood, Illinois, UNITED STATES|
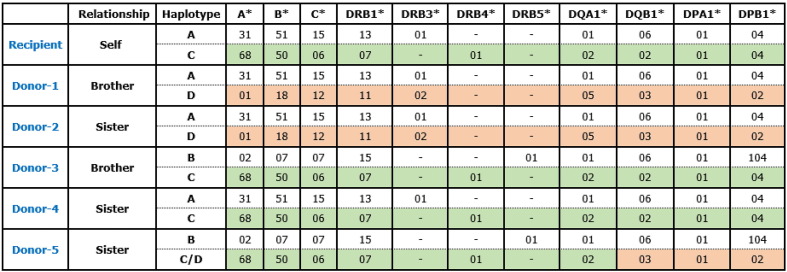
Aim: <b> Human leukocyte antigen (HLA) donor specific antibody (DSA) is the main cause of long term graft failure in kidney transplantation. HLA matching is currently conducted at the HLA antigen level in solid organ transplantation. Alleleic specific antibodies are often detected and its clinical significance is not well defined. Here we reported a strong HLA-B39 specific antibody in an HLA-B39 antigen positive kidney transplant patient. </b> Methods: <b> A 46 years old Hispanic female received a deceased donor kidney transplant 19 years ago under re-transplantation evaluation. She matched HLA-B antigens with her donor (B39 and B48 in both the recipient and the donor) and lost her graft recently. HLA IgG antibody identification by Luminex single antigen bead array assays and HLA typing by LabType SSOP (One Lambda, ThermoFisher Scientific) were performed. </b> Results: <b> HLA antibody identification by single antigen bead assays revealed strong anti-B39 (MFI > 20,000) in the patient. To answer if it is an allele specific antibody came from the donor, we further retyped the patient's HLA and revealed her HLA-B*39:08 which is a different allele from the single antigen B3901 coated on the bead in the single antigen bead test panel.</b><b > Compared with both alleles of amino acid sequences, four amino acid differences at positions of 63 (N > E), 67 (C > S), 74 (D > Y) and 156 (L > R) between B*39:01 (the donor's) and B*39:08 (the patient) indicate the strong donor immunogenicity. Unfornately, we could not re-type the donor due to the material was not available. This allele specific anti-B3901 is predicted as DSA and might be the cause of allograft loss in the patient. </b> Conclusion: <b> Although both the recipient and the donor carried the same antigen HLA-B39 in the patient, different alleles of HLA antigen triggered the allele specific DSA and further could be the cause of allograft loss. In addition, this study suggested high resolution HLA typing played an important role in solid organ transplantation in order to provide a better match donor and interpret DSA in post-transplant monitoring.</b>
P05
Case studies
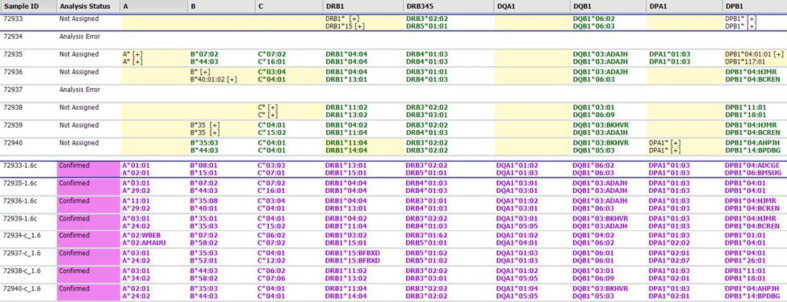
Title: AN HLA-IDENTICAL CHILD AS A DONOR FOR HEMATOPOIETIC CELL TRANSPLANTATION: IS THIS A HAPLOIDENTICAL OR MATCHED- “SIBLING” TRANSPLANT?
Authors: <u>N. Brown</u>, M. Kamoun, F. Johnson, Pathology & Laboratory Medicine, Penn Medicine, Philadelphia, Pennsylvania, UNITED STATES|D. Porter, A. Loren, Medicine, Penn Medicine, Philadelphia, Pennsylvania, UNITED STATES|
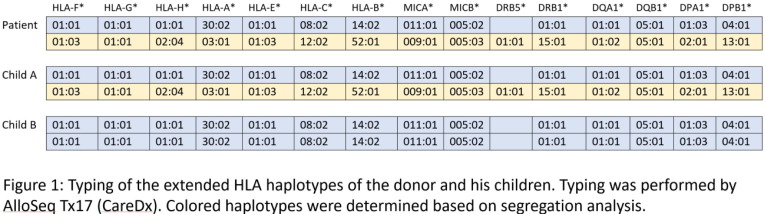
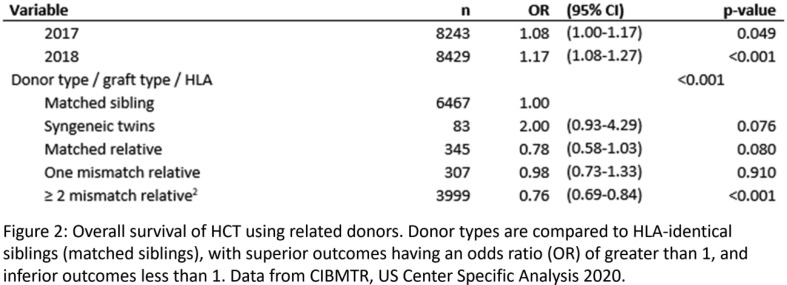
Aim: HLA-identical siblings are the preferred donors for hematopoietic cell transplant (HCT), but available in only approximately 30% of cases. Haploidentical transplantation dramatically expands the donor pool to parents, children or haplo-matched siblings, but requires a different conditioning regimen and more intensive graft-vs-host disease (GVHD) prophylaxis. Here we present a 60-year-old man of Hispanic Latino ancestry diagnosed with acute myeloid leukemia who was considered for HCT. Methods: High-resolution HLA typing by NGS (AlloSeq Tx17, CareDx) was performed on peripheral blood specimens for the patient and his two adult children as presumptive haploidentical donors. Verification typing was performed on a buccal swab for the patient and blood samples for the children. Results: Surprisingly, Child A was found to be HLA identical to the patient (Fig. 1). While the HLA type of the patient’s spouse was not available, fully-homozygous Child B indicated that the patient and his spouse apparently carry the same A*30:02 ∼ C*08:02 ∼ B*14:02 ∼ DRB1*01:01 ∼ DQB1*05:01 haplotype, fairly common in the Hispanic population with a frequency of 3.9x10 < sup > -4</sup>, based on National Marrow Donor Program data. Using segregation analysis, the patient and Child A also carry the A*03:01 ∼ C*12:02 ∼ B*52:01 ∼ DRB1*15:01 ∼ DQB1*05:01 haplotype, which is considerably rarer at 1.5x10 < sup > -6</sup > . The most parsimonious explanation is that Child A inherited the more common haplotype from the mother, and the less common haplotype from the father, and so Child A being HLA identical to the patient is entirely plausible. As the extended HLA haplotype of the HLA-F, G, H, E, MICA and MICB loci, included in the typing panel, was also identical between the patient and Child A (Table 1), this donor was selected for transplant. Conclusion: No published data on outcomes using HLA-identical child donors exists. The Center for International Blood & Marrow Transplant Registry 2020 Center Specific Analysis demonstrated that transplants with HLA-identical related (non-sibling) donors (denoted as “matched relative” in Fig. 2) have similar outcomes to matched sibling donors. We therefore decided to use conditioning and immunosuppressive regimens of HLA-identical sibling donors. We conclude that all first-degree, HLA-identical relatives should be treated similarly when choosing a conditioning regimen and GVHD prophylactic strategy.
P06
Case studies
Title: LOSS OF HETEROZYGOSITY LEADING TO INCORRECT HLA TYPING FOR PLATELET REFRACTORY PATIENT
Authors: M.C. Horwath, J.C. Wade, E.A. Dent, J. Hauenstein, T. Tvrdik, D. Saxe, K.K. Deeb, H.M. Gebel, R.A. Bray, <u> H.C. Sullivan</u>, Pathology and Laboratory Medicine, Emory Univesity, Atlanta, Georgia, UNITED STATES|
Aim: Repeat platelet (PLT) transfusions can lead to allosensitization to Class I HLA antigens and human platelet antigens (HPA). These antibodies can lead to platelet refractoriness (PR). Management of PR due to anti-HLA antibodies includes provision of HLA matched (HLAm) PLTs. Obtaining HLAm PLTs is predicated on correct recipient HLA typing. Here we present an interesting case involving loss of heterozygosity (LOH) at the HLA-A locus. Methods: HLA typing was performed by real time PCR (Linkage Biosciences), sequence specific oligonucleotide (SSO) LABType Typing Test (One Lambda), and Next Generation Sequencing (NGS) performed at a reference laboratory (Histogenetics, Inc.). Class I HLA antibody specificity testing was done by LABScreen Single Antigen assay (One Lambda). Chromosomal Microarray (SNP Copy Number (SNP-CN); Thermo Fisher Scientific CytoScan HD platform) was utilized to assess LOH. Results: A 30-year-old female, with leukocytosis and thrombocytopenia, failed to respond to PLT transfusion. Evaluation for PR revealed numerous HLA antibodies, and peripheral blood (PB) was sent for HLA typing. Though real-time PCR yielded inconclusive results, NGS demonstrated homozygosity at the HLA-A locus. These results were used to obtain HLAm PLTs. The patient was subsequently diagnosed with acute myelogenous leukemia (AML) and chemotherapy was initiated. Post treatment, a new HLA typing was ordered for stem cell transplantation evaluation. This time NGS demonstrated heterozygosity at the HLA-A locus, which was confirmed by SSO. Chromosomal microarray analysis demonstrated LOH of the HLA-A locus within the histocompatibility complex on chromosome 6p in the pre-treatment sample and heterozygosity in the post-treatment sample. Retrospectively, the pathology report revealed that the initial typing was performed on PB containing 93% myeloid blast cells while subsequent typing was performed on PB containing no abnormal cells. Conclusion: LOH is well established in the setting of AML. This case represents acquired LOH affecting only the HLA-A locus. Testing of PB with predominantly aberrant cells resulted in inaccurate HLA typing in the PR workup. Given that patients with hematologic malignancies are often evaluated for PR, obtaining clinical status at the time of sample collection is important. For patients with high leukemic burden, obtaining a non-PB sample (e.g., buccal swab) for HLA typing warrants consideration.
P07
Case studies
Title: CD3 + T CELL LINEAGE SPECIFIC MATERNAL ENGRAFTMENT IN A PEDIATRIC PATIENT WITH SEVERE COMBINED IMMUNODEFICIENCY DISORDER
Authors: <u>G. Balgansuren</u>, N. Shelton, L. Regen, D. McLendon, S. Russell, P. Peterson, A. Ng, D. Smith, C. McFarland, CIL, SCCA, Seattle, Washington, UNITED STATES|
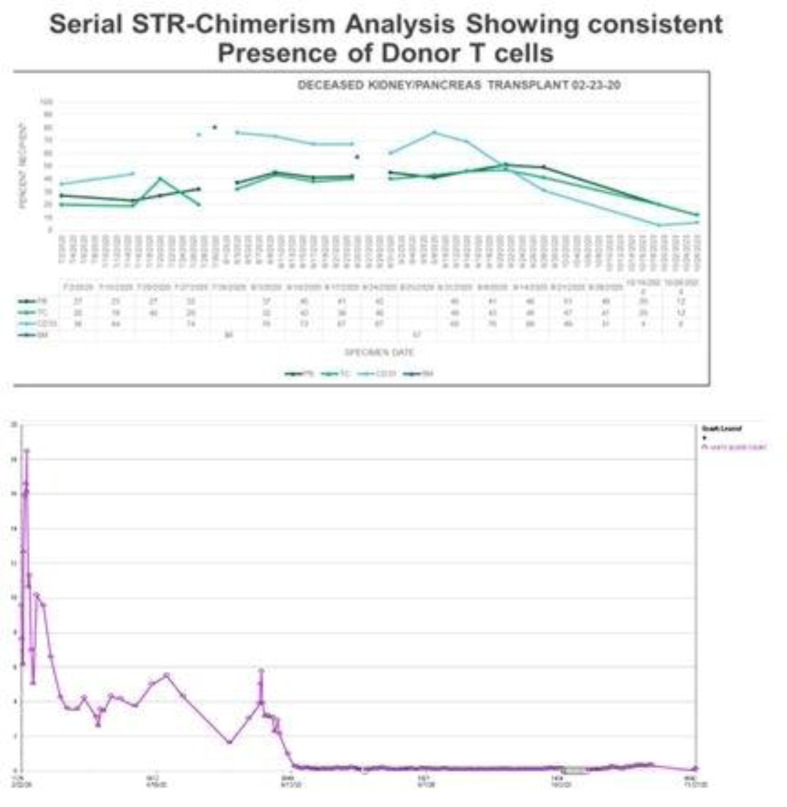
Aim: The human placenta allows for bidirectional passage of nucleated cells between mother and fetus, in which the mother’s cells are usually eradicated by the immune system in healthy infants. Severe Combined Immunodeficiency Disorder (SCID) infants may lack the functional immunity to reject circulating maternal T cells resulting in persistent transplacental maternal engraftment (TME), which can be associated with GVHD. A premature infant newborn diagnosed with SCID and complete maternal engraftment will be discussed in this study. Methods: Chimerism tests were performed using the PCR based Promega PowerPlex 16 STR System and capillary electrophoresis to detect genetic polymorphisms in short tandem repeat (STR) loci. Patient and donor baseline PBMC (patient's pre-HCT) and patient's post-HCT sorted cells were used to extract DNA for the chimerism tests. The lineage specific cell populations for CD3+, CD4+, CD8+, CD19+, CD56+, and CD34 + antigens were sorted by fluorescence activated cell sorting. High resolution HLA typing for patient, parents and sibling were performed by NGS technology (Illumina, MiSeq platform) using TruSight HLA v2 Sequencing Panel with TruSight Assign analysis software. Results: The patient was born prematurely at 33 weeks and 4 days of gestation due to placental abruption and uterine rupture. SCID was diagnosed from newborn’s screening with likely T neg, B neg and NK pos phenotype. The patient had graft-versus-host-disease (GVHD) with skin, liver, gut and bone marrow involvement, secondary to maternal engraftment, which had been confirmed by chimerism testing 10 days after birth. Following HCT at 3 months of age with an HLA-matched sibling, partial donor engraftment was first detected at day + 42 in myeloid cells followed by gradually increasing donor engraftment of NK, B and T cell lineages through day + 410 post-HCT. Sorted CD4 + and CD8 + T cells tested only once at day + 42 revealed that the engrafted maternal T cells were exclusively of CD3 + CD4 + phenotype, comprising 15% of the CD4 + T cells in circulation. Evidence of maternal engraftment was absent after day + 83 post-HCT. Conclusion: Our findings suggest that CD3 + CD4 + lineage specific T cells might be the main contributor for pre-HCT GVHD secondary to maternal engraftment.
P08
Case studies
Title: CASE STUDY: SUBSTANTIAL CHIMERISM DETECTED IN NON-HEMATOPOIETIC TISSUE OF PRIOR CORD BLOOD RECIPIENT A DECADE AFTER TRANSPLANT
Authors: <u>H.R. McMurray</u>, A. Busacco, M.L. Coppage, Pathology and Laboratory Medicine, University of Rochester Medical Center, Rochester, New York, UNITED STATES|
Aim: The patient in this case was referred for updated HLA genotyping to enable an unrelated donor (URD) search, following relapse of precursor B cell acute lymphoblastic leukemia (pre-B-ALL) that was first diagnosed in 2010. At the time of initial diagnosis, the patient was treated with epratuzumab, clofarabine and cytarabine and entered remission. Consolidation therapy of hematopoietic stem cell transplant was performed with dual cord blood units (CBUs) for the allograft. <br /> <br /> After a 10 year remission, this patient was diagnosed with relapsed disease, confirmed by bone marrow biopsy and PET scan. At relapse, the patient’s blood showed 18% blasts. Chimerism study of blood revealed representation of the patient’s cells at 4–8%, with the remainder of hematopoietic tissue being derived from one of the donor CBUs. This “one cord wins” phenomenon of a single CBU dominating hematopoietic repopulation is well-documented in the literature. Methods: Consistent with chimerism results, HLA typing by next-generation sequencing (NGS) of a current blood sample showed typing matching one of the original CBUs with sequence matching the recipient nearly undetectable. Despite the sensitivity of NGS, DNA matching the genotype of the second unit that was transplanted was not detected in any testing. Results: To enable URD selection, we performed genotyping on DNA isolated from a current buccal swab of the patient. Surprisingly, NGS of the buccal DNA showed substantial representation of donor sequences in the genotyping data, up to 30% of reads for some loci. While rare, chimerism in non-hematopoietic tissue following allograft has been reported in the forensics literature, where buccal swabs and even hair follicles can sometimes show donor-derived sequences. Our results are consistent with such findings. Conclusion: Because of this unique situation, we utilized the patient’s original DNA sample from 2010 for genotyping by NGS and were able to identify the patient’s true alleles, including a rare B*14:37 allele. Thus, if allogeneic stem cell transplant is chosen as the treatment plan (vs. possible CAR-T cell therapy), the patient will likely be transplanted with additional cord blood units.
P09
Case studies
Title: HOMOZYGOUS OR HETEROZYGOUS, THAT IS THE QUESTION
Authors: <u>K. Hacke</u>, A. Hodgkins, A. Morris, C. Sholar, S. Fossey, DCI Transplant Immunology Laboratory, Nashville, Tennessee, UNITED STATES|J. Edwards, CareDx Pty Ltd., Fremantle, Western Australia, AUSTRALIA|E. Gomes, C. Wirtz, CareDx, Inc., South San Franc
Aim: Our laboratory routinely performs HLA typing using next generation sequencing (NGS) for all transplant patients. Methods: We identified a renal recipient with a homozygous HLA-A locus (A*23:01, 23:01); homozygosity supported by sequence specific primer PCR (SSP) and reverse single stranded oligonucleotide probe PCR (revSSOP). We received additional family members for evaluation (son and grandchild). Results: Both relatives were a one haplotype match with the recipient at all loci, with the exception of the HLA-A locus. Each family member was homozygous for different HLA-A alleles. The son was typed as HLA-A*11:01, 11:01; the grandchild was typed as HLA-A*03:01, 03:01. Additionally, sequences consistent with HLA-G*01:05 N and HLA-H*02:05 were present for all family members. These sequences are associated with A*30:01:01. Together, these results suggested a shared familial haplotype with a possible HLA-A genetic deletion. Analysis of the copy numbers present at the HLA-A locus revealed a copy number of 1.2 when normalized with the full sequencing run, consistent with allele drop out or loss of heterozygosity (LOH). The sequence data from each individual was analyzed using motif finding software specifically targeting the HLA-A locus. This analysis revealed motifs present within exon 1 and 2 of HLA-A*30:01:01 and identified an 817 bp gene deletion spanning the end of intron 2, extending into exon 4 in DNA from each of the family members. In summary, our detailed NGS sequencing data analysis confirmed a shared familial haplotype carrying an HLA-A*30 allele with a partial gene deletion. This deletion was not detected by multiple different methodologies. Conclusion: This case strongly points out the necessity to carefully evaluate homozygous HLA typing results even in the era of HLA typing by NGS.
P10
Case studies
Title: POSITIVE FCXM WITH NO IDENTIFIED DONOR-SPECIFIC ANTIBODIES: DISCORDANT RESULTS IN 3 KIDNEY RECIPIENT CASES
Authors: M. Michel, S. Leckie, <u>A.E. Sidahmed</u>, Pathology and lab medicine, LHSC, London, Ontario, CANADA|S. DeChickera, Medicine, Nephrology, LHSC, London, Ontario, CANADA|
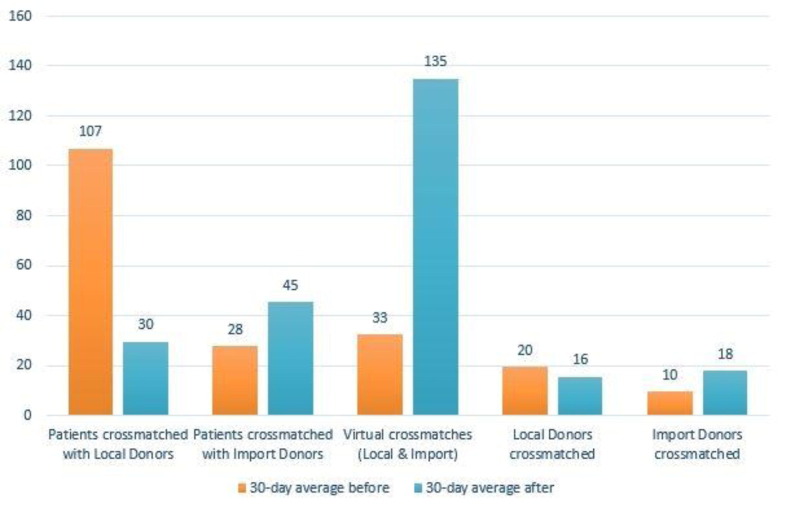
Aim: The FCXM assay is the most sensitive cell-based method to detect DSAs and its prognostic value in a pre-transplant screening is well established. SPA are more sensitive for lower titer antibodies and are better at distinguishing between specific HLA antigens. Complimenting FCXM with SPA offers the potential to better discriminate immunologically relevant positive FCXMs from false-positive results. This is key to maximize the number of safe, compatible transplants to occur. There are still challenges, however, in their interpretation. In this report, we present 3 cases of discordant testing results in potential kidney transplant recipients: positive B cell FCXM and lack of detectable DSAs using routine SPA testing. Methods: A total of 3 recipient samples and their respective donors (4) were used in analysis. Samples were collected from peripheral blood draws. CDCXM, FCXM, HLA typing, and HLA antibody screening was performed on samples accordingly. Results: Auto-crossmatches for all 3 potential kidney recipients were negative. Serial dilution of serum samples also had no effect on HLA antibody testing. Other trouble shooting, namely DTT, absorb out, increased EDTA volume, and heating serum 56oC, all still resulted in negative antibody screening. The Luminex assay was also run using our institution’s usual protocol (Halifax assay) and using manufacturer’s protocol, and all were negative. Finally, we analyzed recipients’ serum for potential non-HLA antigens using One Lambda’s available kits in triplicate, which were also negative. After discussion with One Lambda technical support, they performed SAB testing and expanded single antigen bead assays (ExPlex). Using their ExPlex kits, they identified previously unidentified DSAs. Surrogate crossmatches using these DSAs for each recipient yielded a clinically significant DSA for one recipient and insignificant for another, who proceeded to successful crossmatch with his unrelated living donor. Conclusion: Discussion: A positive FCXM in the context of a negative HLA antibody screen can be interpreted as ‘false positive’, but these cases demonstrate that sometimes routine testing or one test protocol is not enough for all cases. False negative DSA results could adversely affect clinical decisions before and after transplantation, while overcalling these scenarios can result in patients remaining on dialysis and increased risk of mortality.
P11
Case studies
Title: IRREVERSIBLE ANTIBODY MEDIATED REJECTION ASSOCIATED WITH MEMORY IMMUNE RESPONSE INDUCTION OF STRONG DONOR SPECIFIC ANTIBODIES POST LUNG TRANSPLANTATION
Authors: <u>A. Zhang</u>, D. Thomas, Allogen Laboratories, Cleveland Clinic, Cleveland, Ohio, UNITED STATES|M. Budev, Cleveland Clinic, Cleveland, Ohio, UNITED STATES|
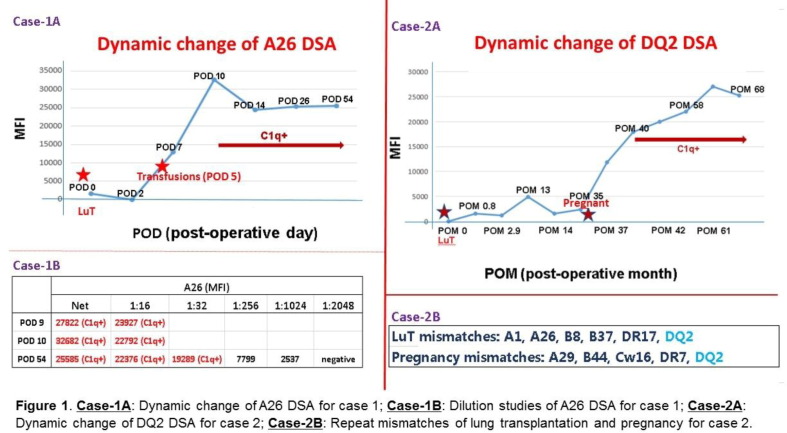
Aim: Antibody-mediated rejection (AMR) due to HLA donor specific antibody (DSA) is a well-recognized cause of allograft dysfunction in lung transplantation (LuT). Allo-reactive memory cells can elicit a potent increase in DSA resulting in poor LuT outcome. Here, we present two failed LuT cases caused by irreversible AMR. Methods: none Results: <b><u>Case 1</u></b>: A 53 yo female with cystic fibrosis (CF) underwent LuT in 2020. Virtual cross-match at pre-LuT was negative with borderline A26 DSA (1600 MFI). Pulmonary edema was noted during intra-operation indicating the existence of immune activation. Patient received plasmapheresis (PLEX)/IvIg, rATG and high dose steroids on post-operative day (POD) 1. A26 DSA reduced to negative (<1000 MFI) on POD 2. During POD 5–8, patient was multiply transfused. Worsening edema, fever, and rapid expansion of white cell counts were observed. Concurrently, A26 DSA rebounded to 13,000 MFI. A26 DSA continually increased to 32,682 MFI (C1q + ) on POD 10 and then remained C1q + until POD 54 with no response to enhanced treatment of PLEX/IvIg/Bortezomib (Figure 1. Case-1A, 1B). Patient expired due to profound irreversible AMR related graft failure at 2 months post LuT. < b><u>Case 2</u></b>: A 33 yo female with CF underwent LuT in 2014. Flow cross-match at pre-LuT was negative without DSA. As demonstrated in Figure 1. Case-2A, weak DQ2 DSA (1600 MFI) was detected post-operative month (POM) 0.8. Subsequently, DQ2 DSA continued to rise and fall between 1200 and 4900 MFI. In 2017 (POM 35), patient became pregnant with DQ2 as one of the pregnancy mismatches (Case-2B). At POM 37, DQ2 DSA rapidly escalated and developed into C1q + at POM 40. Consequently, AMR was treated with rATG/PLEX/IvIg/Carfilzomib during POM 40–42. Unfortunately, DQ2 DSA did not respond to desensitization. Severe graft dysfunction was declared at POM 68. Patient was listed for urgent re-LuT. Conclusion: Prompt surveillance of rebounding DSA caused by potential memory response (Case 1 and 2) and immune activation status (Case 1) is critical for timely adjustment of immune therapies for prevention of graft loss due to irreversible AMR post LuT.
P12
Case studies
Title: CASE OF THE MISSING DONOR B CELLS: OCRELIZUMAB SUSPECT APPREHENDED
Authors: <u>M.J. Thiessen</u>, Histocompatibility, Alberta Precision Laboratories, University of Alberta Hospitals, Edmonton, Alberta, CANADA|P. Campbell, A.M. Halpin, Laboratory Medicine and Pathology, Histocompatibility, Alberta Precision Laboratories, Universit
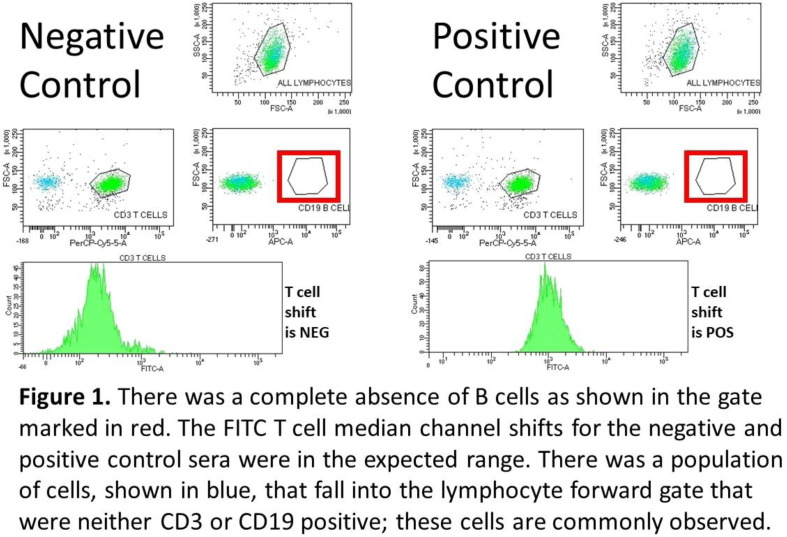
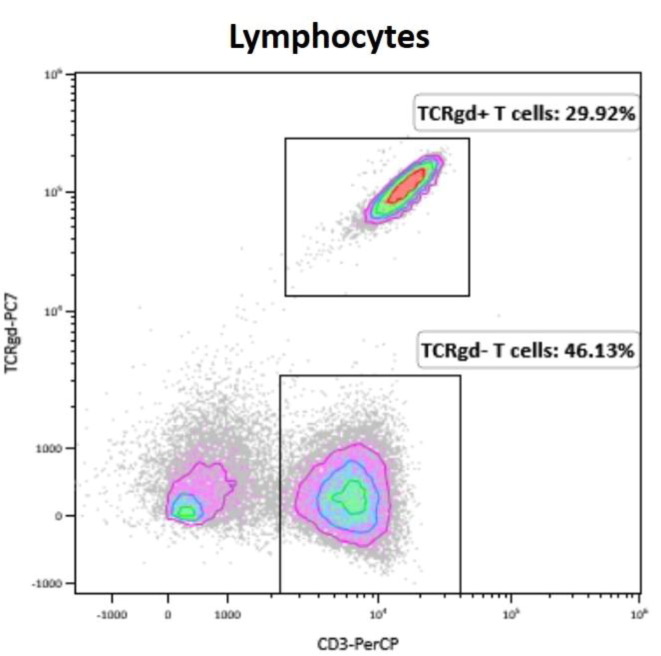
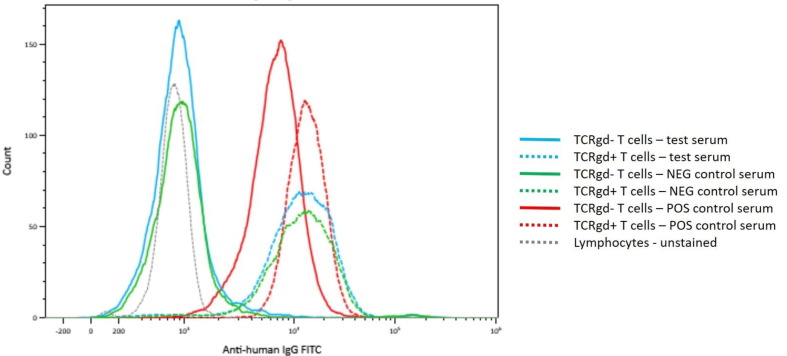
Aim: T and B cell flow crossmatch (FCXM) are often performed pre-transplant, in parallel with solid phase HLA antibody testing, to assess compatibility. Here we present a case in which B cells were absent in the XM and the subsequent investigation of this issue. Methods: The HLA Laboratory received a section of spleen from a deceased donor. Standard Ficoll cell isolation was performed; cells were further purified by EasySep™ HLA. The ‘Halifaster’ FCXM was performed, including pronase treatment; CD3 and CD19 were used to label T and B cells, respectively. FITC-labelled anti-human IgG was used as secondary antibody. Recipient samples included a heart/lung recipient (5% cPRA) and liver recipient with no previously detected HLA antibodies. Results: No B cells were detected in the FCXM (Figure 1). Repeat FCXM was performed to rule out technical error but the same result was observed. FCXM was reported to the director who followed up with the organ procurement team. Additional medical history was provided as well as a drug history of ocrelizumab, an anti-CD20 therapy. As there were no donor-specific HLA antibodies, no recent sensitizing events, and patient samples were recently screened for HLA antibodies, a virtual B cell XM was reported along with the physical T cell FCXM. Conclusion: Deceased donor histocompatibility testing can yield unexpected results that create additional work on call. The prior treatment of the donor with ocrelizumab resulted in the absence of donor B cells in the donor’s spleen, a sample normally rich with B cells. Our laboratory was able to report a FCXM for the T cells and a virtual crossmatch for the B cells. Technologist time and on-call stress could have been reduced had the donor’s medical history been provided. Communication with the donor procurement team will assist the laboratory in trouble-shooting donor issues. These recipients had low or negative cPRA so did not pose as much of a challenge as if the patients were highly sensitized. The total lack of CD19 + B cells is interesting as we know that therapies such as ocrelizumab eliminate B cell populations in peripheral blood but it is rare to see this glimpse into human secondary lymphoid organs. This drug may decrease B cell niches in transplanted organs which could offer theoretical advantage in recipients although this would be challenging to study as this is likely a rare event.
P13
Case studies
Title: EPITOPE ANALYSIS MAY BE BETTER THAN CREG ASSOCIATIONS FOR DETERMINATION OF RELEVANT DSA
Authors: T. Harville, Pathology, Laboratory Medicine, and Internal Medicine, University of Arkansas for Medical Sciences, Little Rock, Arkansas, UNITED STATES|<u>J. Liu</u>, S. Pandey, Pathology, Laboratory Medicine, University of Arkansas for Medical Sciences, Li
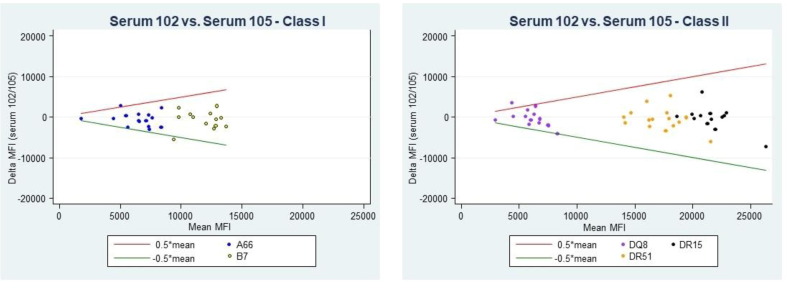
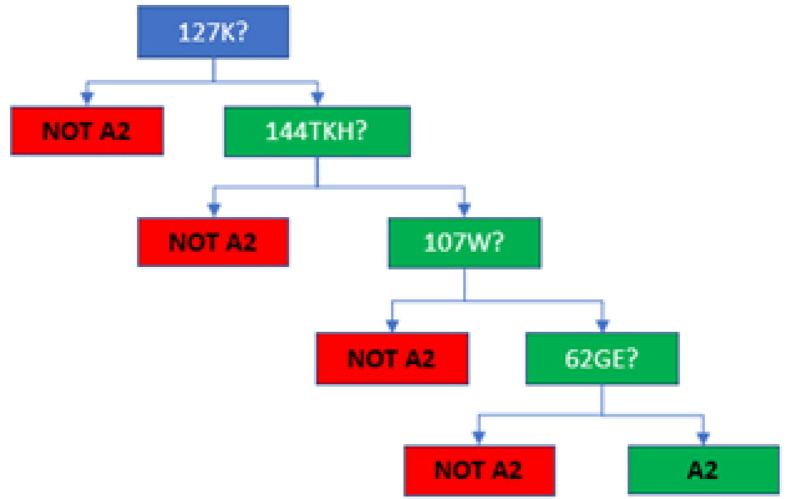
Aim: A patient received cardiac transplantation with concurrently performed Flow Cytometric Crossmatch (FCXM); T- and B-cells were positive with both current and one-month-old sera. The patient had not had HLA typing performed prior to transplantation. At the time of transplantation, anti-HLA-A2 (all alleles) donor-specific antibody (DSA) had been detected with MFI of ∼ 1800–1950 on the one-month-old serum, along with lower levels of anti-HLA-B57 and -B58, while antibodies against other A2 CREG members, HLA-A23, -A24, -A68, and -A69, were near 0 MFI (Tables 1 and 2). The transplant center uses 2000 MFI for avoidances, thus, HLA-A2 was not being avoided. It is typically expected that pathogenic anti-HLA-A2 antibodies would be shared across other A2 CREG members. Without knowing the patient's HLA type though, antibody spread across A2 CREG could not be fully assessed. Thus, a virtual assessment of the donor type with potential DSA prior to transplantation was incomplete, and the lack of A2 CREG antibody spread gave false reassurance that the potential DSA anti-HLA-A2 antibody may not be highly significant, since the MFI was below the avoidance cut-off of the transplantation center. The positive FCXM dashed this reassurance. The patient was typed post-transplantation revealing the expression of HLA-A*68:01 (Table 3), explaining the absence of antibody reactivity to HLA-A68 and -A69. Methods: Epitope analyses were performed with the MatchMaker™ function in One Lambda Fusion™. Results: Epitope analyses were performed with the MatchMaker™ function in One Lambda Fusion™, which indicated that HLA-A2 (all alleles), -B57, and -B58 share epitope 62GE, while HLA-A23, -A24, -A68, and -A69 do not (Table 4 and data not shown). Any other epitope for A2 CREG, for example, 44RM (found on HLA-A2, -A23, -A24, -A68, -A69, and -B57) is also present on some 29 other HLA types (for example, HLA-B76), for which antibodies have not been detected. The absence of such antibodies indicates that other A2 CREG epitopes are not relevant in this case. Conclusion: This interesting case illustrates the need for patient and donor typing results, as well as the anti-HLA antibody results to assess fully for potential meaningful DSA. While CREG associations have been useful, epitope analyses can be very useful to determine the most likely relevant DSA, based on the antibody patterns, and shared epitopes of the HLA types.
P14
Case studies
Title: RELATIONSHIP BETWEEN MFI, FlowPRA AND FCXM EMPLOYING A MONOTYPIC DRB4(DR53) ALLOANTIBODY.
Authors: J. Abraham, D. Flynn, C. Tate, H.C. Sullivan, H.M. Gebel, <u>R.A. Bray</u>, Emory, Atlanta, Georgia, UNITED STATES|
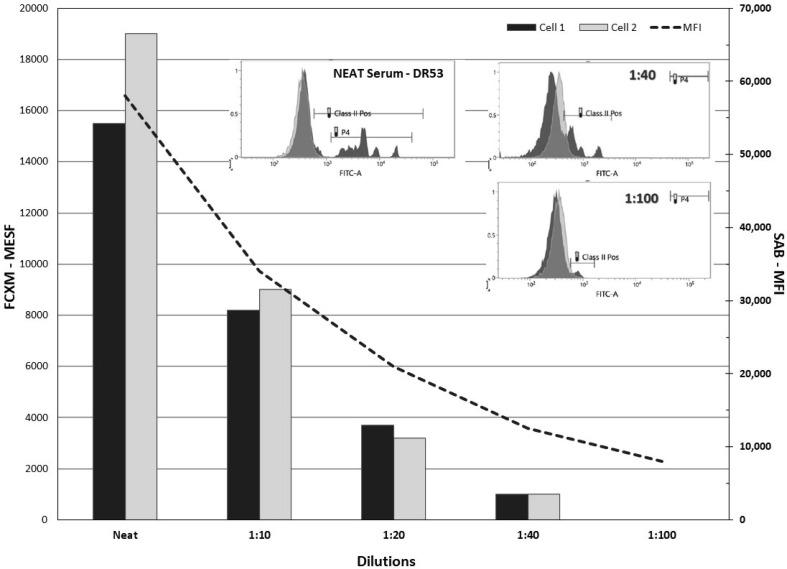
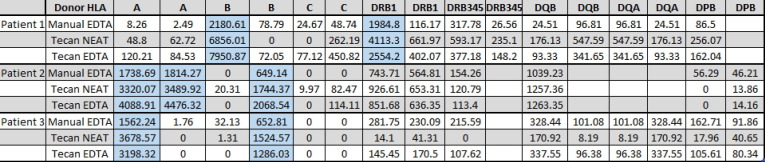
Aim: Evaluating the relationship between Mean Fluorescence Intensity (MFI) from single antigen bead (SAB) testing, reactivity against HLA phenotype-bearing beads and flow-cytometric crossmatches (FCXM) with appropriate target cells has been attempted by many groups. However, most studies utilized human allosera that tended to be poly-reactive possessing multiple class I and/or II specificities which presented distinct challenges when selecting appropriate target cells for a FCXM. In this report, we present a patient who developed a monotypic, high MFI, DRB4 (DR53) alloantibody subsequent to renal transplantation. Studies were performed to assess the relationship between MFI, FCXM and FlowPRA reactivity for this unique alloantibody. Methods: Briefly, we performed serial dilutions of the monotypic DR53 alloantibody (Neat to 1:800) assessing MFI (Luminex FM-3D), MESF and FlowPRA reactivity at each dilution. MFI from SAB testing (One Lambda, Inc.) was compared to FCXM using a hemizygous DR53 target cell (Cell 1; DR4, DR7; DR53, DR53N) and a homozygous DR53 target (Cell 2; DR4, DR7; DR53, DR53). The FCXM was performed on the BD FACSLyric and reported as a change in Molecules of Equivalent Soluble Fluorochrome (MESF). Results: Results are shown in < b>Figure 1</b>. DR53 alloreactivity, initially ∼ 58,000 MFI, was easily detected out to a 1:400 dilution (∼1,700 MFI) and still reactive at 1:800 (∼500 MFI) by SABs. FlowPRA reactivity was lost by ∼ 1:200 (∼4,000 MFI) and the FCXM became negative with both target cells at >1:20 dilution. Conclusion: Studies employing monotypic HLA allosera to assess reactivity across multiple antibody detection platforms are rare. Moreover, few if any studies have utilized a monotypic DR53 serum. Although only a single patient study, our work clearly points out the sensitivity differences between platforms. Specifically, for this DR53 antibody, SABs could detect antibody reactivity at significantly lower levels than FlowPRA beads or target cells (SABs > FlowPRA > FCXM). This difference is likely due to the level of the DR53 antigen expression on the various targets. Interestingly, there was no significant difference in reactivity between a hemizygous vs homozygous DR53 bearing cell. In summary, studies utilizing monotypic allosera can provide a better understanding of the relationship between the various antibody assays and ultimately help decipher their clinical relevance.
P15
Case studies
Title: SUCCESSFUL DETECTION AND MANAGEMENT OF MEMORY RESPONSE IN A HEART TRANSPLANT PATIENT
Authors: <u>C. Bornstein</u>, J. Cone Sullivan, A. Rabson, J.J. Xin, Pathology, Tufts Medical Center, Boston, Massachusetts, UNITED STATES|A. Vest, Cardiology, Tufts Medical Center, Boston, Massachusetts, UNITED STATES|
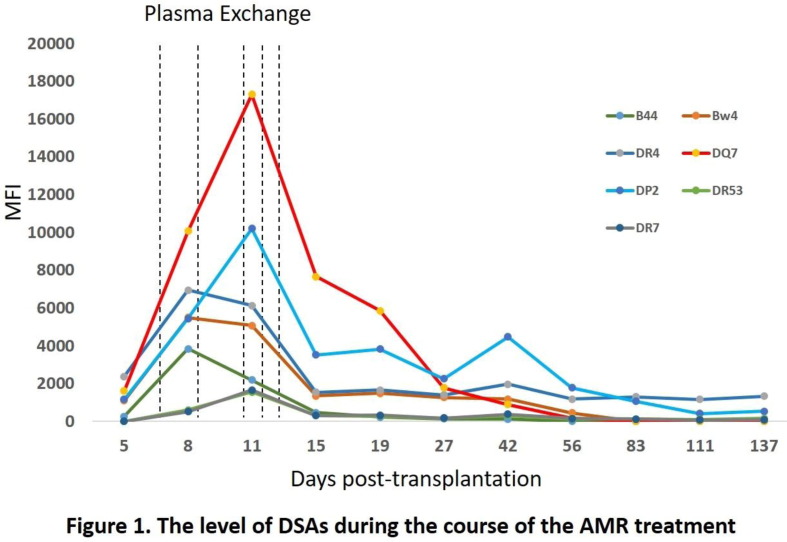
Aim: Memory immune response can cause accelerated organ rejection after transplant. We present an interesting case and share our experience. Methods: The patient’s clinical course and the testing results were retrospectively reviewed and analyzed. Results: A 52 year old female with a pre-transplant calculated panel reactive antibodies (cPRA) around 75% underwent heart transplantation with thymoglobulin induction due to sensitization (only 2 doses due to leucopenia) after a negative prospective T cell CDC crossmatch. HLA antibody testing and biopsy was performed 5 days post-transplantation (DPT). Four de novo DSAs (DR4, DQ7, DR53 and DP2) and two pre-existing DSAs (B44 and Bw4) were detected. There was no histopathologic evidence of antibody–mediated rejection (AMR), but retrospective T and B cell flow crossmatches were positive. One session of plasma exchange was followed by an unexpected increase in DSA levels (Figure 1, mean fluoresce intensity, MFI, doubled from DPT 5 to 8). A memory response was suspected and the patient received additional plasma exchanges. On 11 DPT, while three DSAs (B44, Bw4, and DR53) appeared to trend down, two DSAs (DQ7 and DP2) continued increasing (MFI almost doubled by 8 DPT). On 15 DPT, all DSA levels dropped to the similar levels as detected on 8 DPT. Two doses (375 mg/m < sup > 2</sup > ) of rituximab were administered. C1q testing evaluated complement-fixing ability; only DQ7 DSA was C1q positive on 11 DPT (peak level) but negative on 15 (DPT). The DSAs have trended down to a stable low level for the past 3 months. The third weekly biopsy showed faint C4d staining (<10% capillaries). Subsequent biopsies and echocardiograms have been unremarkable and the patient is currently stable 6 months post-transplant. This cases demonstrates the value of DSA testing for sensitized patients within 1 week post-transplantation. Conclusion: A rapid increase in pre-existing DSAs or emerging “de novo” DSAs are important indicators of a memory response. Early recognition and reduction in DSA levels is critical to manage the complement-fixing ability of DSAs and avoid graft damage.
P16
Case studies
Title: SEASONAL INFLUENZA VACCINE IMPACTED HLA ANTIBODY FORMATION IN A KIDNEY TRANSPLANT RECIPIENT WITH HISTORY OF BLOOD TRANSFUSION: A CASE STUDY.
Authors: <u>A. Mostafa</u>, D. Webster, T. Pearce, K. Gorkoff, C. Shanofer, Saskatchewan Health Authority, Saskatoon, Saskatchewan, CANADA|
Aim: It is critical for each laboratory to validate and optimize thier assays for thier centers. Indeed, the performance of multiple assays should be considered, as no single test is without its own limitation. Methods: 59-year old male Asian Canadian was diagnosed with end-stage renal disease secondary to both obstructive nephropathy as well as hypertension. He was referred to the kidney transplant clinic for an upcoming living donor renal transplant from his sister. The patient had a history of blood transfusion in 2016. The initial flow cell crossmatch (FCXM) was performed in May 2018. FCXM was negative for both T and B cells. In order to confirm the FCXM results and to properly assess the patient, SAB (LABScreen single antigens beads, One Lambda) was done on patient sera and the results were negative for both HLA class I and II antibodies with 0% PRA. < br /> The transplantation of the patient was delayed due to some medical concerns that required the patient to be hospitalized and his transplantation was temporary suspended. One year later, the patient was fit to be added to the transplantation list. The patient received the influenza seasonal vaccination and the initial FCXM was repeated. Results: Surprisingly the patient reacted strongly positive for both T and B cells FCXM. SAB for HLA class II did not show any change from previous and the PRA was 0%. For HLA class I antibodies we noticed the presence of A1, 23, 24, 80; B44, 45, 76 with a PRA of 51%. A1 and B44 were DSA’s. We performed epitope analysis using the matchmaker in the Fusion 4.4 software to see if there is any shared epitope(s) that explained the pattern of antibodies. We found that two epitopes, 166DG and 166ES explain the entire specificity. The increase in the breadth of HLA antibodies was mainly within the same cross-reactive antigen group (CREG), indicating an expansion of existing specificities without development of new specificities. A1 is a DSA with an MFI of 1200, but it shares the same epitope of A80 with an MFI of 15000. This could explain the strong positivity of the T cell and B cell FCXM Conclusion: In conclusion, HLA antibody formation after the seasonal vaccination are due to activation of pre-existing memory B cells, which result in increase of strength of HLA antibodies.
P17
Case studies
Title: SEROLOGICAL DEFINITION OF B*15:15 - B62 OR B75?
Authors: <u>D. Youngs</u>, I. Gimferrer, E. Chou-Wu, E. Doran, Bloodworks Northwest, Seattle, Washington, UNITED STATES|
Aim: The WHO Nomenclature Report lists HLA-B*15:15 with a serological specificity of B62, but sequence alignment with other B62 and B75 alleles suggests B*15:15 is more likely to behave serologically as B75. Defining B*15:15 as B62 or B75 is important when allocating organs from B*15:15 donors to candidates who have antibodies that are B62+/B75- or B75+/B62-. As single antigen antibody testing panels do not include B*15:15, we evaluated the serological specificity of B*15:15 by crossmatching B*15:15-positive cells against patient sera whose antibody reactivity differentiated B62 from B75. Methods: Two donors were typed as B*15:15 by One Lambda's SABR typing kit, the software of which follows the WHO report and assigns B*15:15 as B62. Testing with One Lambda's LABScreen Single Antigen antibody kit identified 12 patients who were positive with B62 (B*15:01 on LSA panel) and negative with B75 (B*15:02 and B*15:11 on LSA panel), and 17 patients who were positive with B75 and negative with B62. Flow crossmatches were performed between cells of the two B*15:15 donors and sera from three patients with B62+/B75- antibodies, and three patients with B75+/B62- antibodies (because of additional reactivity with other donor HLA, the other 23 patients were not informative and were not crossmatched). The patients' sera were also tested against B62 (B*15:01) and B75 (B*15:02) cells as controls, which excluded three additional sera as uninformative for this analysis. Results: The two patients whose antibodies were B62+/B75- were crossmatch-negative with both B*15:15 donors. The one patient whose antibodies were B75+/B62- was crossmatch-positive with both B*15:15 donors. Conclusion: Crossmatches with patient sera that have antibodies that discriminate B62 from B75 demonstrate that B*15:15 behaves as a B75, not as a B62, and is consistent with sequence comparisons that suggest amino acid position 63 is key in differentiating B75 from B62 (63 N contributes to defining B75, while 63E contributes to defining B62). For candidates with antibodies that are B62+/B75- or B75+/B62- it is important that potential donors are assigned the appropriate B15 serological split based on how the antigen behaves serologically. In order to appropriately exclude potential recipients who have antibodies against B75, and to avoid inappropriate exclusion of potential recipients who have antibodies against B62 but not against B75, organ donors who type as B*15:15 should be reported as B75.
P18
Case studies
Title: DE NOVO HLA ANTIBODY PRODUCTION FOLLOWING CLINICAL TRIAL CMV VACCINE
Authors: <u>M. Nevski</u>, M. McAuliffe, G. Wedermyer, C.L. Roark, B.M. Freed, ClinImmune Labs, Aurora, Colorado, UNITED STATES|S. Davis, University of Colorado, Aurora, Colorado, UNITED STATES|
Aim: A 38-year old male (CMV negative) with chronic kidney disease was evaluated for living kidney transplant. His sister (CMV positive) was identified as a suitable living donor. Preliminary testing showed a negative T and B flow crossmatch and no DSA (cPRA = 0%). CMV disease can be a complication of immunosuppression following kidney transplantation, requiring prophylactic antiviral treatment. The recipient enrolled in a double-blind clinical trial for a bivalent recombinant CMV vaccine. He received the final of the three dose trial vaccine series eight days before his serum sample was drawn for PRA and final crossmatch. Unexpectedly, his cPRA increased from 0% to 80% and his final crossmatch was T and B cell positive with DSA detected to A2 (MFI range = 4974 – 5552) and B7 (MFI = 14391). Low resolution HLA-A, B, DR testing was performed to confirm sample identity. A redraw was also requested which confirmed the positive T and B cell crossmatch and increased cPRA (81%). Subsequent C1q testing identified C1q-binding antibodies, including C1q-binding DSA (B7, MFI = 4019). Through coordination with the clinical team, the only sensitizing event identified was the CMV trial vaccination. There were no recent or historic transfusions or transplants. The transplant was cancelled and the donor and recipient entered NKR. Retrospective record review identified another clinical trial participant (26 yo male) whose serum samples showed a strong, <i>de novo</i> A3 antibody (MFI = 18613) one month following his vaccine series. C1q testing was positive for A3 (MFI = 20995) and his flow crossmatch using an A3-positive surrogate donor was also positive. This patient also had no history of sensitization. Since A3 was not a target of DSA, this antibody did not impact transplantation. In both cases, the antibodies that appeared after vaccination decreased in strength to baseline levels over 8 months (38 yo patient) and 12 months (26 yo patient). Since this clinical trial remains blinded, it is difficult to determine the cause of the HLA sensitizations detected in the participants, although strong, <i>de novo</i> antibody production without a source of sensitization is difficult to explain. The HLA sensitization suggests that anti-CMV antibodies may cross react with HLA antigens. Further research to determine the incidence of HLA sensitization following CMV infection may help to explain the sensitizations seen in this trial. Methods: x Results: x Conclusion: x
P19
Case studies
Title: A NEW LOOK AT OLD ANTIBODIES UTILIZING CURRENTLY AVAILABLE TECHNOLOGIES
Authors: <u>M. Rewinski</u>, P. Doyle, J. Devivo, S. Vicki, D. Durkin, R. Caruk, L. Bow, Transplant Immunology, Hartford Healthcare, Hartford, Connecticut, UNITED STATES|
Aim: BO is a 30 year old Hispanic male with FSGS disease who received a zero antigen matched deceased donor kidney transplant (Tx) in 2015. Four months post-Tx, BO showed a slight elevation in class II antibodies (Abs) as detected by luminex single antigen (SA) beads, specifically donor specific antibodies (DSA) to DQA1*05:01 (MFI = 2,800). Methods: Reactivity to this particular bead was frequently observed in isolation and in unsensitized patients, so was often considered nonspecific or false positive. DSA to DQ2, represented on four other SA beads separate from the bead associated with DQA1*05:01, was considered negative (MFI < 1,000). One year later, DSA MFI levels to DQA1*05:01 increased to 9,600 and remained > 7,000 over the next few years, despite plasmapheresis and IVIG treatment, but overall Ab reactivity to DQ2 was still negative by our lab’s standards (MFI 〈9 0 0). Results: About five years post-Tx, BO then experienced robust Ab mediated rejection (AMR) with high level DSA to class II DQ2 (MFI > 20,00), with all five DQ2 beads expressing strong positivity, including the one associated with DQA1*05:01. Upon further analysis of this current luminex SA data, there appeared to be a second, strongly positive Ab reactivity to the DQA1*04,05,06 epitope (MFI > 20,000). With the recent addition of another luminex SA assay to our laboratory, we decided to confirm the newly developed Ab pattern. The results of the second luminex SA assay clarified the class II Ab profile, showing more definitively two distinct DSA to DQ2 and the DQA1*04,05,06 epitope. This second luminex SA assay provides four DQA1*05:01 beads, two of which are associated with DQB chains other than DQ2, and three beads each to cover DQA1*04:01 and DQA1*06:01, also associated with different DQB chains compared to the first luminex assay. Conclusion: The enhanced DQA and DQB bead associations of the second luminex assay helped confirm the results of the first luminex assay. If the initial post-Tx testing on BO included the second luminex assay, then perhaps the Ab reactivity to DQA1*05:01 could have been confirmed as true DSA, not false positive, and treatment plan adjusted accordingly at that time. This case study emphasizes the importance of using more than one Ab testing algorithm, incorporating beads from different vendors (along with surrogate crossmatch testing) to help elucidate difficult Ab patterns and/or suspected false positive, nonspecific reactivity.
P20
Case studies
Title: DONOR SPECIFIC ANTIBODY AFTER HEART TRANSPLANT: IS C1Q ANTIBODY GENERATED FROM MEMORY B-CELLS?
Authors: <u>L. Helander</u>, C.L. Roark, B.M. Freed, University of Colorado/ClinImmune, Aurora, Colorado, UNITED STATES|M.D. Everitt, Cardiology, Children's Hospital Colorado, Aurora, California, UNITED STATES|
Aim: In 2013, a 27 year old woman with Ebstein’s anomaly was tested using the OneLambda single antigen bead (SAB) assay to determine her HLA sensitization following pregnancy. Her antibody profile showed defined and tentative Class I and II antibodies (cPRA = 39%). Her antibody level to A2 was tentative (MFI range = 853–1268), but by early 2014 the reactivity to A2 was negative (MFIs < 1000). In late 2014, the patient received an ABO matched, 6/6 HLA-mismatched heart transplant (A2,A30,B42,B44,DR8,DR15) for cardiac failure. The retrospective T and B cell flow crossmatch was weak positive; attributed to DR15 donor specific antibody (DSA) with a plan to give thymoglobulin as treatment. Within 7 days, her cPRA = 100% with the return of A2 antibody (MFI range = 18608–19701). Antibody mediated rejection (AMR) was confirmed on cardiac biopsy. The C1q binding assay showed complement binding DSA (A2,B44,DR8,DR15). Plasmapheresis (TPE), IVIG, Rituximab and steroids reduced her cPRA to 7% with a negative C1q assay by 9 weeks post-transplant. < br /> <br /> Similarly in 2009, a male with multiple congenital cardiac anomalies presented with defined A2 antibody (MFI range = 1755–2914). By 2013, A2 antibody was no longer detectable. In 2021, the patient’s received an ABO compatible, 1 antigen match/5 mismatched (A2,B8,B27,DR8,DR17) heart transplant. The retrospective T and B cell flow crossmatch was negative and no DSA was present. 13 days post-transplant, there was evidence of graft dysfunction with the cPRA = 100% and DSA to A2 saturating the limit of the SAB assay at 1:4 dilutions (MFI estimated > 50,000). DSA to Cw7 (MFI = 8016) and DQ4 (MFI = 2453) were also present. The C1q binding assay showed complement binding by the A2 antibody only (MFI range = 18907–20531). Cardiac biopsy confirmed AMR. Desensitization using TPE, Eculizumab, Bortezomib and steroids reduced the C1q–binding A2 antibody into the negative range within 5 days. Methods: NA Results: NA Conclusion: We hypothesize the complement activating antibodies are the result of memory B-cells and/or long-lived plasma cells. Memory cell activation leading to AMR is a rare occurrence, but these cases highlight the limitation of current testing to identify the risk of previous sensitization. Memory B-cell assays should be considered for screening high risk patients or consideration given to listing historical but absent antibodies as unacceptable antigen matches for organ transplant.
P21
Case studies
Title: NOVEL HLA-A*01 ALLELE IDENTIFIED IN A COMMON CAUCASIAN HAPLOTYPE
Authors: <u>M. Lee</u>, N. Smith, I. Salvador, A. Comeaux, R. Vorhaben, A. Usmani, C. Lacelle, University of Texas Southwestern Medical Center, Dallas, Texas, UNITED STATES|
Aim: Herein we describe a novel HLA-A*01 allele identified in a patient with myelodysplastic syndrome. Methods: High-resolution HLA genotyping was performed using an in house developped sequence based typing method and analyzed using SBTengine software (GenDX, Netherlands). The typing was also confirmed using next generation sequencing (FastPlex NGS kit from One Lambda). Antibody testing was performed using One Lambda single antigen beads. Results: We identified a novel HLA-A*01 allele in a patient with myelodysplastic syndrome. The new allele was identified in two samples collected 4 years apart and differs from the common HLA-A*01:01 allele by an A to G substitution in the A*01:01 exon 3 sequence, at nucleotide position 508, which causes an amino acid change from lysine to glutamic acid at codon 146. The new HLA-A*01 allele was found in the common HLA-A*01, B*08:01, C*07:01, DRB1*03:01 (DR17) and DQB1*02:01 Caucasian haplotype. The haplotype was confirmed using the typing from the 3 patient’s children except for HLA-C. The patient is HLA-C*07:01, 07:18 and the only child typed for HLA-C has both the HLA-C*07:01 and C*07:18 alleles. However, the most likely HLA-C present in the haplotype containing the novel HLA-A*01 allele is the C*07:01 since the C*07:18 allele is commonly found in the other patient’s haplotype of HLA-A*29:02, B58:01, C*07:18, DRB1*13:03 and DQB1*03:01. Interestingly, two of the patient’s three children inherited an A*01:01, B08:01, C*07:01, DRB1*01:01 and DQB1*05:01 haplotype from their father. While the patient mounted antibodies due to pregnancy to the other paternal haplotype mismatches, no antibodies against the HLA-A*01:01 allele were identified by single antigen assay despite the patient being sensitized to HLA-A*01:01 by pregnancy. The patient also received multiple blood transfusions and likely has encountered HLA-A*01:01 in donor blood as well. This indicates that HLA-A*01:01 may not be immunogenic in individuals having the HLA-A*01:NEW allele. Conclusion: This case study identified a novel HLA-A*01 allele present in a common Caucassian haplotype. Individuals carrying this new allele do not appear to mount antibodies to HLA-A*01:01.
P22
Case studies
Title: A PATIENT WITH REPEATEDLY POSITIVE ALLOGENEIC CROSSMATCHES DUE TO NON-HLA ANTIBODIES
Authors: <u>E. Chou-Wu</u>, D. Youngs, I. Gimferrer, Immunogenetics/HLA, Bloodworks NW, Seattle, Washington, UNITED STATES|A. Weiss, Nephrology and Renal Transplantation, Virginia Mason Medical Center, Seattle, Washington, UNITED STATES|
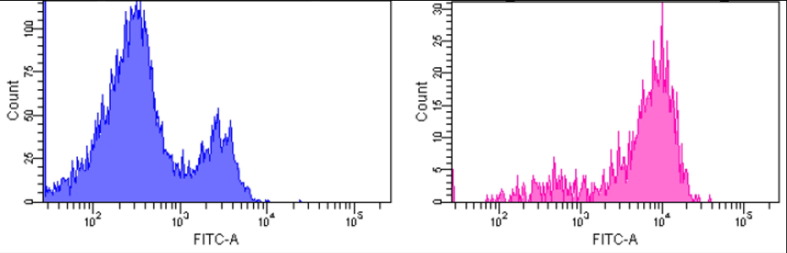
Aim: A 64 year-old African American female with a CPRA of 26% was listed for kidney transplantation in 2018 due to hypertensive nephrosclerosis. Methods: During 2019–2021, a total of 19 allogeneic lymphocyte flow cytometric crossmatches (FXM) were performed and, despite the absence of donor-specific HLA antibodies (DSA) and negative T and B cell autologous FXMs in all the cases, positive reactivities were consistently observed with B cells and a subset of T cells (Figure 1, average B cell channel shift= +64, established positive cutoff= +23; T cell channel shift= +8, established positive cutoff= +14 on a 256-channel scale). Furthermore, crossmatches with aortic endothelial cells from two deceased donors without HLA DSAs were also strongly positive with an average median channel shift of 210 (cutoff = 50). Results: The patient’s medical record showed that she is G3, P3 with no history of previous transplant, transfusion, autoimmune diseases, or treatment of therapeutic biological products. Additional antibody testing, including anti-AT1R, MICA, HNA (human neutrophil antigen), HPA (human platelet antigen), and a commercially available bead-based non-HLA antibody panel, were all negative. These results indicated that the patient has antibodies directed against frequent epitopes on endothelial cells, B lymphocytes, and a subset of T cells that are absent from the autologous cells. The targets of these antibodies, whether unique or shared among different cell types, remain to be identified. Conclusion: This complex case underscores the need for available assays to allow characterization of non-HLA antibodies and further examine their roles in solid organ transplantation. The patient remains untransplanted to date due to the increased risk of allograft rejection presented by anti-endothelial cell antibodies and the lack of specified antibody targets for post-transplant monitoring.
Fig. 1. Representative histograms of allogeneic T cell (a) and B cell (b) flow cytometric crossmatches.
P23
Case studies
Title: HIDDEN NON-HLA ANTIBODIES IN A HIGHLY SENSITIZED PATIENT IN NEED OF SECOND KIDNEY TRANSPLANT
Authors: S. DIonne, L. Santiago, <u>S. Shetty</u>, VRL Eurofins, Centennial, Colorado, UNITED STATES|
Aim: <b> A 68-year-old Caucasian male previously received a related LD kidney (2016) which subsequently failed 3 years later in 2019 due non-HLA antibody related early acute rejection with allograft dysfunction. He has end-stage renal disease due to obstructive uropathy. The patient was ultimately listed in UNOS for a 2nd transplant. The patient had a CPRA of 99.56% with both Class I and II antibodies.</b> Methods: <b> A well-matched DD kidney offer was received. Negative results were precdecited by virtual crossmatch. Both the T/B cell results were Negative upon Flow crossmatch. Table 1 outlines the HLA typing, antigen mismatches and donor-specific unacceptable antigens. At the same time this STAT crossmatch was performed, results for esoteric reference lab testing were received. </b><br /> <b> The patient had AT1R antibodies (15 U/mL; reference range > 10 U/mL risk for endothelial cell dysfunction). XM-One crossmatch results showed both the IgG/IgM results were positive. Channel shifts were 209 and 129 (cutoff at 40 and 80, respectively). The Transplant Center declined this offer, despite the lack of DSAs. </b > Results: <b > This case elucidates the need to develop a plan for transplant using both positive/negative outcomes of anticipated test results. Due to the high CPRA and a low probability that another donor of this kind would present in the coming year, understanding the meaning of esoteric results need to be taken into consideration. Had these test results been available two days after re-transplantation, what would the clinical course look like for this patient? Would the center be prepared to deal with rejection of this kind? In instances in which non-routine testing is ordered for patients on the waitlist, a clear line of communication is needed between the Transplant Center and the lab. Perhaps the patient should have been made Inactive on the Waitlist until the full panel of testing was completed and an appropriate plan could be generated for the best care/transplant outcome.</b > Conclusion: Whether the presence of pre-transplant AT1R-Ab positivity justifies prophylactic desensitization is still debated but due to anti-endothelial antibodies, the complexity of this case heightens. < b > A desensitization regimen would likely need to be developed for this patient, and surveillance allograft biopsies post-transplant would be warranted. Conversely, the patient could be referred to another center that has experience with complex cases. </b>
P24
Case studies
Title: A RARE CASE OF RECOMBINATION BETWEEN DR and DQ IN A SAUDI DONOR
Authors: A. MOHAMMED, M. Awaji, M. ALZAHRANI, D. AlAbduladheem, <u > R. Al attas</u>, histocompatibility and immunogenetics, king fahad specialist hospital Dammam, Dammam, SAUDI ARABIA|<u > R. Al attas</u>, Saudi society of bone marrow transplantation, Riyadh, SAUDI A
Aim: Crossover at HLA region is a rare event, resulting in a new haplotype that could decrease the chance for (HSCT) patient to find a matched family donor. The estimated incidence of crossover between chromosomes in paternal or maternal chromosomes occurring during meiotic cell division of the germ cells is 1%. The proportion of any particular recombination depends on the distance between the relevant HLA loci. Generally, the more distant the loci are, the greater the chance of recombination. Therefore, Crossover events were observed between nearly all the neighboring HLA loci (A-C, C-B, B-DR, DQ-DP) except for HLA-DR and DQ where there is strong associations between genes within the 85-kb region from DQB1 to DRB1. Here, we report an unusual recombination between HLA HLA-DR and DQ. Methods: A potential Hematopoietic stem transplantation (HSCT) patient and five siblings were tested for HLA class I genes (<i > A</i>, <i > B</i > and < i > C</i > ) and HLA class II genes (<i > DRB1</i>, <i > DRB3/4/5</i>, <i > DQA1</i>, <i > DQB1</i>, <i > DPA1</i > and < i > DPB1</i > ) typing using reverse specific sequence oligonucleotides probe (rSSO) after DNA extraction and amplification by PCR. Results: HLA haplotype analysis revealed the presence of a cross over in the HLA class II region in one of the potential donors. A recombination occurred between DRB1/ DQA1 and DQB1 genes [Figure 1] resulting in a novel haplotype containing DRB1-DRB4-DQA1 and DQB1-DPA1-DPB1 hybrid. Conclusion: Although hotspots of recombination in the HLA region do not usually include the region encompasses DR and DQ loci. Yet, crossover, within this region can rarely occur.
Family HLA typing
P25
Case studies
Title: GRAFT-VERSUS-HOST DISEASE (GVHD) POST LIVER TRANSPLANTATION (LT); A CASE REPORT
Authors: S. Ghandorah, Z. AlQahtani, H. A. Alharbi, A. Aqool, M. Qahtani, S. Kebasi, <u > R. Al attas</u>, histocompatibility and immunogenetics, king fahad specialist hospital Dammam, Dammam, SAUDI ARABIA|<u > R. Al attas</u>, Saudi society of bone marrow transplanta
Aim: The development of GVHD after liver transplantation is uncommon complication post LT with an incidence rate of 0.5% to 2% & a mortality rate of 85%. It arises when activated donor lymphocytes transplanted along with liver graft mounting a destructive immune response against tissues of the immunocompromised recipient. Here, we describe a case of an eleven months old child who had LT from his HLA- haploidentical 35- old mother. Two months later he presented with generalized skin rash involving the whole body & oral mucosa. Histology of the skin biopsy showed hyperkeratosis and vacuolization of the basal layer surrounded by lymphocytes, suggestive of GVHD.Chimerism study using short tandem repeat (STR) on peripheral blood (PB) sample revealed 39% of maternal DNA mixed with the recipient DNA. Revising HLA typing of both donor & recipient showed homozygous maternal HLA type in all loci. Patient typing is HLA-A 02,68; HLA-B 07,44; HLA-C 05,15; HLA-DRB1 17, 11; HLA-DR 52; DQA 01, 05; DQ 7, 05; DPA 01, 02; DPB1 04, 13. The Donor typing is HLA-A 68; HLA-B 07; HLA-C 15; HLA-DR 11; HLA-DR 52; DQA 05; DQB1 7; DPA 01; DPB1 04. Mother serum showed broad HLA- sensitization (figure 1). Methods: <b > STR</b>: PB DNA was extracted using Qiagen extraction kit & analysed using AmpFlSTR® Identifiler® Plus PCR Amplification Kit, includes16-STR panel on 3500xL Genetic Analyzer. < b > HLA </b > typing was determined using Sequence-Specific Oligonucleotide typing method, antibody performed by LSM and SAB using One Lambda® protocol. Results: This case represent classical risk factors reported in literatures to develop GVHD including young, multiparous mother, HLA- homozygous donor, & strongly sensitized mother against paternal antigens represented in tissues of her immunocompromised child. Conclusion: Although GVHD is a rare complication after liver transplantation, high index of suspicion & early diagnosis is critical to exclude competing etiologies and prevent the high mortality . Unlike hematopoietic stem cell transplantation, routine HLA testing is not often required before liver transplantation. Recognition of GVHD - risk factors in our case by proper HLA-work-up pre transplantation could have discovered these risk factors and might resulted in melioration or even prevention of this potentially fatal condition through proper donor selection. < br /> Chimerism analysis is an excellent diagnostic tool to confirm clinical and histological suspicion of GVHD.
P26
Case studies
Title: THE FIRST DOCUMENTED EVIDENCE OF DARATUMUMAB INTERFERING WITH FLOW CYTOMETRIC CROSSMATCH
Authors: <u > S. Ho</u>, Gift of Hope Organ & Tissue Donor Network, Itasca, Illinois, UNITED STATES|<u > S. Ho</u>, K. Putnam, C. Peiter, W. Herczyk, J. Gerlach, Gift of Life Michigan, Ann Arbor, Michigan, UNITED STATES|<u > S. Ho</u>, J. Gerlach, Michigan State Univers
Aim: Multiple myeloma (MM) is characterized by cancerous plasma cells which often leads to renal failure and transplant (Tx). Daratumumab (Dara) is an IgG1κ human mAb for treating MM by depleting CD38 + B cells and plasma cells. Herein, we report the first case of Dara interference of flow cytometric crossmatch (XM) repeatedly causing aberrant false positives which inadvertently delayed Tx for a renal waitlist patient. We show that a simple cell treatment can effectively mitigate Dara interference preserving the utility of pre-Tx XM in Dara-treated patients. Methods: XM were performed on pronased donor lymphocytes using a BD FACSLyric flow cytometer with staining of CD45/leukocyte, CD3/T, CD19/B, G46-2.6/HLA-I and Tu39/HLA-II. Cell surface CD38 expression was characterized using HB7 anti-human CD38 mAb and analyzed in median channel (MC) values. Results: Patient is a 67-yo white female with ESRD secondary to MM who continuously receives Dara infusions. Her sensitization included an autologous HCT, transfusions and pregnancies. While solid phase testing confirmed the absence of HLA antibody upon listing, 26/27 deceased donor XM were unexpectedly positive (T + B+, n = 21; T-B+, n = 5), presumably caused by Dara binding to donor cell surface CD38. The strength of the positive XM MC shift was wide-ranging (T cell, +45–158; B cell, +74–239; cutoff T + 39B + 70), which could be attributed to the highly variable CD38 expression level on donor lymphocytes (T cell, 238–428; B cell, 237–433; n = 78). Overall, T cells were found to have a lower CD38 expression profile than B cells (296 ± 40 vs 317 ± 38; p < 0.001), and no significant difference was observed between deceased and living donors. Finally, lymphocyte treatment with mild 0.05 M DTT (10 mins 37 °C) prior to pronase was able to cleave off most of the cell surface CD38 and effectively abrogated the Dara-induced false positive XM without affecting XM sensitivity, specificity or HLA expression. Conclusion: The patient was eventually transplanted across a strong positive T and B cell XM with an out-of-state deceased donor. She is currently doing well > 6 mos post-Tx with no HLA antibodies detected to date. To our knowledge, this is the first case documenting Daratumumab interference in routine XM. We also demonstrated that CD38 expression varies greatly on lymphocytes, and that cell treatment with DTT can effectively mitigate the interference from Dara and preserve the utility of XM.
P27
Case studies
Title: THE IMPORTANCE OF HIGH-RESOLUTION HLA TYPING FOR 0 ANTIGEN MISMATCH KIDNEYS: A PEDIATRIC CASE REPORT
Authors: <u > K. Kaneta</u>, Pediatrics, Children's Hospital Los Angeles Department of Pediatrics, Los Angeles, California, UNITED STATES|R. Lestz, Nephrology, Children's Hospital Los Angeles Department of Pediatrics, Los Angeles, California, UNITED STATES|N. Fotiad
Aim: Human leukocyte antigen (HLA) mismatches inform decisions on organ allocation and influence long-term graft outcomes. Low-resolution HLA typing is the standard for OPTN/UNOS allocation, but typing using next generation sequencing (NGS) has increased precision which can identify potential targets for B-cell mediated rejection that would have not been recognized using low-resolution typing. This case illustrates the importance and potential clinical significance of NGS for donors considered to be 0 - ABDR mismatched. Methods: Low resolution HLA typing was obtained from UNOS. HLA A, B, C, DRB1 and DQB1 types were determined using next generation sequencing (NGS) for both donor and recipient. HLA epitopes were determined using HLA Matchmaker eplet matching (ABC v4.0 and DRDQDP v3.1). Results: A 5-year-old boy with a history of end stage renal disease secondary to nephrotic syndrome underwent a deceased-donor kidney transplantation in March 2020. Using UNOS criteria, the donor was 0-ABDR mismatched. Retrospective NGS typing revealed several HLA mismatches (table 1). Using HLA Matchmaker there were 7 eplet mismatches. Conclusion: This case highlights the importance of high-resolution HLA typing to identify potential clinically significant HLA mismatches. It should not be assumed that a 0-ABDR mismatch identified by low resolution typing has no risk for development of donor specific HLA antibodies, or that humoral rejection in a 0-ABDR mismatched donor must be caused by non-HLA antibodies. This is significant for organ allocation and also relevant to post transplant management, particularly in the pediatric population. With the move towards individualized protocols for optimal immunosuppression, it is increasingly important to accurately balance the risk of graft rejection while also minimizing the risk of infection. This case illustrates that a 0-ABDR mismatch may appear to be an opportunity for the clinician to limit immunosuppression, however, management may differ when mismatches are identified through NGS. Furthermore, preventing development of donor specific antibodies is an important consideration in the pediatric patient who will likely require multiple kidney transplants in their lifetime.
P28
Case studies
Title: CROSSMATCH FOR ABO INCOMPATIBILITY LIVING DONOR KIDNEY TRANSPLANTATION WORKUP
Authors: <u > J.C. Ho</u>, S.K. Cheung, S.L. Ng, K.W. Tam, J. Kwok, Transplantation and Immunogenetics, Queen Mary Hospital, Pokfulam, Hong Kong, HONG KONG|
Aim: Kidney transplantation remains the best treatment for patients suffering from end stage kidney diseases; however, there is a severe shortage of deceased donors in Hong Kong. Living donor kidney transplantation provides an alternative source of donor graft. Often at time, it was hindered by blood group incompatibility of the related donor. ABO-incompatibility (ABOi) living kidney donor transplantation has become a possible alternative to expand the donor pool. Methods: Between August 2018 and June 2021, 11 pairs of live donor/recipient pairs with ABOi have undergone route immunological risk assessment including HLA antibody screening by Luminex, T- cell and B-cell CDC and flow cytometric crossmatch. Results: 8 out of 11 pairs were negative for both T-cell and B-cell CDC and flow cytometric crossmatch. Of the 3 pairs with positive crossmatch, one pair was found to be positive for both T- and B-cell. The positive B-cell crossmatch was probably due to the presence of a moderate level of anti-DQ9 donor-specific HLA IgG antibody. Of the other two pairs with positive T-cell flow cytometric crossmatch, HLA antibodies were absence in the one pair while low level of HLA antibodies against Class I and Class II antigen in the other pair. However, the donor of the later pair was actually a HLA matched sibling with zero mismatched at A, B, DRB1 and DQB1 loci. Therefore, the positive crossmatches for these two pairs are probably due to the presence of non-HLA antibodies. A significantly higher titers of donor-specific isohemagglutins IgG antibody (1:512) were found from these two pairs compared with those pairs with a negative crossmatch. Conclusion: Presence of high titer of donor-specific isohemagglutins antibodies may cause a positive T-cell crossmatch in the absence of the donor-specific anti-HLA antibodies. Special caution has to be made in interpreting the crossmatch results for ABOi kidney transplant workup.
P29
Case studies
Title: ANTIBODY-MEDIATED REJECTION IN DSA NEGATIVE FULLY HLA MATCHED KIDNEY TRANSPLANT PATIENT
Authors: <u > E. Lazar-Molnar</u>, Pathology, University of Utah, Salt Lake City, Utah, UNITED STATES|D. Raghavan, Division of Nephrology, University of Utah, Salt Lake City, Utah, UNITED STATES|P.F. Halloran, Medicine, Division of Nephrology, University of Alberta
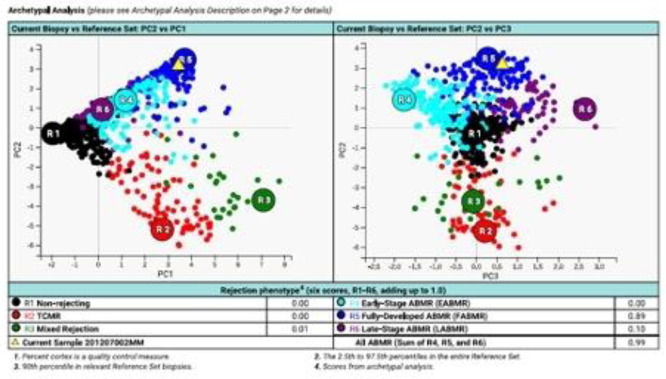
Aim: Case: A 43 y old F patient with a history of end stage renal disease due to lupus nephritis, with a cPRA of 100% received a deceased donor kidney offer with zero antigen mismatch, and was transplanted with a 4 < sup > th</sup > kidney with a compatible T and B cell flow cytometry crossmatch. Methods: Following transplant she had good urine output but serum creatinine did not decrease as expected and she underwent kidney biopsy on post-operative day 7. The biopsy indicated no acute T cell mediated rejection (TCMR), but was positive for C4d staining in peritubular capillaries. There was no microcirculatory inflammation. She was treated with plasmapheresis, IVIG and high dose steroids, which improved her creatinine level. 3 months post-transplant a repeat biopsy showed focal endothelialitis, which was read as acute TCMR, Banff IIA, with mild peritubular capillaritis, C4d staining was negative. The patient was started on iv steroids. Molecular microscope (MMDx) testing did not detect TCMR, but rather severe antibody mediated rejection (ABMR). Treatment was changed to plasmapheresis and IVIG followed by rituximab, which improved her serum creatinine. Results: Confirmatory high-resolution HLA typing on the patient and her donor confirmed that they were matched not just for classical HLA genes but also for non-classical HLA-E, F, G, H and minor antigens MICA and MICB at the 3-field level. < br /> Non-HLA antibody testing detected AT1R antibodies above the upper limit of the assay, and the patient was started on losartan, with no significant change. 6 months post-transplant a repeat biopsy indicated mild peritubular capillaritis and glomerulitis but C4d was negative and no acute TCMR was detected. MMDx showed severe fully developed ABMR but no TCMR. Compared to the biopsy 3 months earlier, there was some increase in inflammatory features. The patient was again treated with plasmapheresis and IVIG, and additionally received bortezomib, which led to a decrease in serum creatinine. Conclusion: Challenge: The problem of early-onset DSA negative ABMR in a highly sensitized patient raises many issues, and suggests mechanisms that have not been characterized. The AT1R autoantibody has a high positivity rate in the post-transplant population and has little predictive value. Other possibilities include alloantibody directed against non-HLA alloantigens expressed on endothelial cells or alternative NK cell recognition systems.
Figure 1. MMDx result indicating fully developed ABMR 3 months post-transplant in a fully HLA matched 100% cPRA patient.
P30
Case studies
Title: AMBIGUOUS HLA TYPING RESOLVED BY PATTERN ANALYSIS
Authors: A. Parkinson, K. Chandler, C. D’Haene, S. Mendelsohn, S. Shelton, E. Schwedler, <u > M. Cusick</u>, Pathology, University of Michigan Medicine, Ann Arbor, Michigan, UNITED STATES|E.H. Rozemuller, GenDx, Utrecht, NETHERLANDS|
Aim: In DQB1 and DPB1, extra patterns were identified in hematopoietic cell transplant (HCT) patient buccal swab DNA HLA typed by next generation sequencing (NGS). Therefore, we wanted to determine which alleles were represented in these NGS patterns using a “multipattern analyzer”. Methods: High resolution typing by NGS (GenDx) and sequence-based typing (SBT) (One Lambda) performed on Illumia MiSeq and 3500xL, respectively. Reverse Sequence Specific Oligonucleotide Probe hybridization (rSSOP) (One Lambda) was used for confirmation. NGS analysis was performed using NGSgo software and multipattern analysis was done by GenDx, which was used strictly as an experimental approach and not clinical use. Multipattern analysis for DQB1 was of exon 2 and 3 and exon 2 for DPB1. Results: Patient is a 53-year-old Caucasian male who received a HCT in 2008 and was being evaluated for a second HCT due to Acute Myeloid Leukemia relapse in 2021. The recipient had 9/10 match (1 mismatch at DQB1) with previous donor. Neither the patient nor the donor was typed for HLA-DPB1 for the first pre-transplant work up. HLA-DPB1 typing was performed for donor but not the recipient due to no patient DNA being available prior to 1 < sup > st</sup > HCT. Multiple buccal swabs were obtained, and extreme care was taken to avoid cheek bleeding but recipient DPB1 typing was repeatedly ambiguous for HLA-DPB1. Donor typing is DQB1*03:02, 06:03 and patient DQB1*03:04, 06:03 (pre-1 < sup > st</sup > HCT). Donor and patient share DQB1*06:03, confirmed by high fraction % (Fig). Interestingly, donor DQB1*03:02 accounted for a much higher fraction (28%) compared to recipient DQB1*03:04 (10.6%) (fig). For DPB1, 4 different alleles for DPB1 (2 of which matched the SBT donor typing) and the other 2 were attributed to the patient (fig). Conclusion: We were unable to report the patient HLA-DPB1 due to contamination. This left the patient at risk for GVHD due to potential unpermissive DPB1 mismatch. Fortunately, the patient did receive a HCT with a permissive DPB1 donor based on these experimental results and is doing well post-HCT. These results suggest the ambiguous typing results may be due to GVHD. Therefore, caution should be used when interpreting typing results in a repeat HCT patient. Lastly, further investigation is needed to validate the multipattern analyzer.
P31
Case studies
Title: LUNG TRANSPLANTATION IN A POST COVID-19 PNEUMONIA PATIENT: A LIFE-SAVING OPPORTUNITY
Authors: <u > G.L. Crowther</u>, M.E. Jeresano, D.P. Lucas, M. Bettinotti, Pathology, Johns Hopkins University, Baltimore, Maryland, UNITED STATES|
Aim: While the long-term effects of COVID-19 infections are still relatively unknown, patients with COVID-19 associated irreversible lung injury are in need of immediate therapies. Lung transplantation has emerged as a potentially lifesaving treatment for patients affected by COVID-19 associated respiratory failure. We describe the course of a bilateral lung transplant for a post COVID-19 infection patient. Methods: DSA was evaluated using Luminex immunoassays (Lifecodes classes I and II single antigen; Immucor-Lifecodes, Stamford, CT and Single Antigen Beads; One Lambda, Canoga Park, CA). Sera were pretreated using Melon Columns™. Results: A 61 year old male with no significant medical history was admitted with COVID-19 associated pneumonia two months prior to listing. His recovery from COVID-19 was complicated by significant fibrosis and a non-healing bronchopleural fistula caused by prolonged ventilator use. The fistula was determined to be non-repairable via surgery. As a life-saving treatment, the patient was urgently evaluated and listed for lung transplantation. The patient was put on veno-venous extracorporeal membrane oxygenation (ECMO) and remained on ECMO until a suitable donor was found one month post listing. The transplant surgery had several complications due to the severity of the bronchopleural fistula, including the removal of an abscess in the apex and severe upper lobe parenchymal disruption. These complications resulted in an extended surgery requiring massive volumes of blood products. At time of transplant, the patient was positive for HLA class I antibody, but negative for HLA donor-specific antibody (HLA-DSA). The patient’s post-transplant recovery was affected by numerous bacterial infections as well as the presence of de novo HLA-DSA one month post transplant. Additionally, the patient developed AKI (acute kidney injury) due to prolonged ECMO prior to transplant and ATI (acute tubular injury), likely from his COVID-19 pneumonia. The patient remains hospitalized and under close medical care. Conclusion: Lung transplantation as a life-saving treatment for patients with irreversible end-stage lung injury due to COVID-19 related pulmonary fibrosis may follow a challenging post-transplant course and requires more in-depth clinical studies.
P32
Case studies
Title: HLA LOSS IN PEDIATRIC HYPODIPLOID ACUTE LYMPHOBLASTIC LEUKEMIA (ALL): A CASE FOR A PRIORI HAPLOIDENTICAL DONOR SELECTION FOR HEMATOPOIETIC CELL TRANSPLANT (HCT)
Authors: <u > P. Arnold</u>, Pathology, St. Jude Children's Research Hospital, Memphis, Tennessee, UNITED STATES|A.C. Talleur, B.M. Triplett, Bone Marrow Transplant and Cellular Therapy, St. Jude Children's Research Hospital, Memphis, Tennessee, UNITED STATES|
Aim: Loss or downregulation of HLA is rare in hematologic malignancies, except in the setting of acute myeloid leukemia (AML) relapse after mismatched HCT. Approximately one-third of AML relapses following haploidentical (haplo)HCT involve genomic loss of mismatched HLA in leukemic cells. HLA loss often occurs via uniparental disomy, whereby the HLA region on chromosome 6p is lost, and the remaining chromosome is copied by the cell’s replication machinery. Loss of HLA allows leukemic cell evasion of donor immune surveillance, rendering donor lymphocyte infusion inefficacious. Compared to patients with AML, HLA loss in ALL is rare, both at diagnosis and post-transplant relapse. Hypodiploid ALL, characterized by extensive chromosomal loss, is associated with poor response to therapy. The patient described here is a 6-year-old male who presented to St. Jude with hyperleukocytosis of CD19 + B-ALL. Diagnostic cytogenetics and genomics studies revealed 26 chromosomes, consistent with near-haploid ALL. Methods: The patient received chemotherapy, autologous CD19-directed chimeric antigen receptor T cell therapy (CAR-T), and a 5/10 HLA matched T-cell depleted haploHCT from his mother. Post-transplant the patient engrafted and achieved 100% donor chimerism, which he maintained until a year later, when he relapsed with 60% donor chimerism in the bone marrow. Relapse was characterized by leukemic cell loss of the inherited paternal HLA haplotype as measured by qPCR chimerism. At this time, the original pre-transplant diagnostic sample was analyzed by qPCR for HLA loss and HLA typing was performed. Results: At diagnosis, the leukemia exhibited loss of the paternally derived chromosome 6 and was homozygous for the maternally inherited HLA haplotype. The patient received additional chemotherapy and CD19-directed CAR-T cells, then proceeded to 2 < sup > nd</sup > haploHCT using his father as donor, who was single HLA haplotype matched to the patient but completely HLA mismatched to the leukemia cells. Approximately 8 months after 2 < sup > nd</sup > HCT, the patient continues to have 100% donor 2 chimerism and remains in remission. Conclusion: This case highlights the potential of assessing HLA loss in leukemia patients at diagnosis, particularly in patients with known chromosomal abnormalities. This information may facilitate optimal donor selection if haploHCT is an option.
P33
Case studies
Title: THE UNIQUE CHALLENGES OF EVALUATING A BONE MARROW TRANSPLANT RECIPIENT FOR LUNG TRANSPLANTATION
Authors: <u > M.E. Jeresano</u>, G.L. Crowther, S. Jeresano, D.P. Lucas, M. Bettinotti, Pathology, Johns Hopkins School of Medicine, Baltimore, Maryland, UNITED STATES|
Aim: Evaluation for lung transplantation of a patient with a previous bone marrow transplant (BMT) presents unique challenges. Swift determination of the appropriate HLA typing and ABO at the time of active lung listing is crucial. We describe a candidate whose complex treatment regimen for recurrent desmoplastic small round cell tumors as well as BMT complications led to pulmonary fibrosis and the urgent need for lung transplantation. Methods: The patient is a 24 year old HLA antibody negative male who had undergone haplo-identical BMT from his father. At the time of evaluation for lung transplantation, chimerism testing showed complete engraftment of the BMT donor. Meaning, the patient’s body (except the hematological tissue) was expected to express the constitutional, original HLA antigens and his immunological competent cells were expected to express the BMT donor’s HLA antigens. Total engraftment was further supported by the observance of only the BMT donor’s HLA antigens in typing results from the patient’s peripheral blood sample post BMT. The patient also converted to the BMT donor’s ABO type O, introducing the possibility of blood group anti-A or B antibodies. Therefore, the interdisciplinary team decided the best strategy to list the patient in UNET for lung transplantation was as follows:<br /> 1. List patient’s constitutional HLA typing < br /> 2. Match the blood group of lung allograft to the BMT donor Results: The rationale of listing the patient’s constitutional HLA typing was that engraftment may be transient and the patient may revert to his original immune system. Furthermore, the patient did not appear to make anti-self HLA antibodies, so there was minimal risk of acute rejection of an organ carrying any of the HLA-antigens of the mismatched haplotype. Lastly, a blood group O lung donor would not express A or B antigens. Fortunately, within weeks of being waitlisted, a compatible deceased donor was found and a successful bilateral lung transplantation was performed. Donor lungs are stable 6 months post transplantation with no evidence of antibody mediated rejection (AMR). Conclusion: Collaboration and clear communication among the lung transplantation, HLA, molecular pathology and transfusion medicine teams was crucial for the timely listing and successful outcome of this patient.
P34
Case studies
Title: COMPLEX IMMUNOLOGICAL INTERACTIONS BETWEEN SYSTEMIC LUPUS ERYTHEMATOSUS, HUMAN PAPILLOMAVIRUS, AND HLA ALLOSENSITIZATION IN A PATIENT WITH END-STAGE RENAL DISEASE
Authors: <u > V. Bravo-Egana</u>, H. Sanders, Histocompatibility/Immunology Laboratory, Medical College of Georgia/Augusta University, Augusta, Georgia, UNITED STATES|
Aim: Recent publications support the idea that exposure to infectious antigens may induce immune responses against homologous proteins, including development of autoantibodies and anti-HLA antibodies. The link between lupus and HPV is especially well documented. Multiple immunological mechanisms contribute to activation of innate and adaptive immunity in response to infections and autoimmune activity. When these elements converge in a kidney transplant candidate, many questions arise, most of which lack concrete answers. Methods: A 56-year-old White female with a history of prior kidney transplant has a primary diagnosis of SLE, as well as a history of HPV infection. She suffered an episode of acute rejection consistent with immunological hyper-responsiveness, resulting in a significant allosensitization profile. Her case is also notable for extreme autoreactivity, with autologous and allogeneic crossmatches (XM) exhibiting strong reactivity even in the absence of DSA. Multiple rounds of auto-adsorption are required to reduce the levels of autoantibody in her serum so that allogeneic XM can be meaningfully interpreted. However, the levels of these autoantibodies increased dramatically over time, and our auto-adsorption protocol was no longer sufficient to reduce XM reactivity into the negative range. A paired donation offer was rejected because it was not known whether autoreactivity levels were masking alloantibodies. Results: Specific questions persist about these phenomena, including how they may be interacting, and continue to pose a contraindication for transplantation? How can immunological memory be assessed when high autoreactivity may have masked historical antibodies? Is there a practical serum treatment for managing increasing autoreactivity? Is the HPV infection contributing to this escalation, perhaps even in a positive feedback loop? Can control of the HPV infection be balanced against the demands of post-transplant immunosuppression? Will post-transplant sensitizing events trigger acute rejection episodes? Conclusion: Until we are able to answer these questions, the immunological risk versus benefit of transplantation for this patient will be difficult to assess.
P35
Case studies
Title: HLA ANTIBODIES IN SERUM OF OLDER INFANTS CAN BE MATERNALLY DERIVED
Authors: K. Foreman, J. Menteer, R. Lestz, L. Baxter-Lowe, Keck School of Medicine, University of Southern California, Los Angeles, California, UNITED STATES|<u > N. Fotiadis</u>, L. Baxter-Lowe, Pathology and Laboratory Medicine, Children's Hospital Los Angeles, Lo
Aim: HLA antibodies (cPRA 77%) were detected in a 7-month-old candidate for heart transplantation who had no history of sensitizing events. The child has dilated cardiomyopathy, required a mechanical ventricular assist device (VAD) and had no evidence of autoimmune disease. To avoid unnecessarily excluding donors from the extremely limited pediatric donor pool, the possibility of maternally derived antibodies was investigated. Methods: HLA antibodies in serum from the patient and mother were characterized using LABScreen Single Antigen reagents (One Lambda). Serial dilutions were utilized to titer antibodies in maternal serum. Patient HLA types were determined by Next Generation Sequencing (Omixon). During VAD implantation, we initiated cardiopulmonary bypass with a 3- vol exchange transfusion to eliminate HLA antibodies. Results: Antibodies against the B7 CREG and a partial A2 CREG were detected in patient serum (1025–6290 MFI). No self-reactive antibodies were detected in the patient’s serum. Maternal serum showed all of the specificities observed in the patient’s serum and additional specificities including antibodies against several of the patient’s antigens (B35, DR4, DR53, DQ8). The strongest antibodies in the maternal serum (1:256 titer) were the strongest antibodies in the patient’s serum. In maternal serum, the strongest patient-specific antibody was B*35:01 with a titer of 1:128, but there was no evidence of this specificity in the patient’s serum. < br /> In patient serum tested one day after exchange transfusion and VAD placement, the B7 CREG and partial A2 CREG HLA antibodies were reduced to 100–400 MFI. These antibodies remained above the baseline but below reportable levels without significant change over the 6 month period following VAD placement prior to heart transplant. Conclusion: Maternal HLA IgG antibodies can persist at substantial levels in infant serum for at least 7 months after birth. When neonates and young children who are candidates for transplantation present with HLA antibodies, the possibility of maternal origin should be considered. Maternal antibodies can be removed and will not recur, potentially broadening the donor pool and/or reducing the likelihood of peri-transplant immune reactions. In this case, anti-infant maternal HLA antibodies were selectively absent in the patient serum but the mechanisms are unknown.
P36
Case studies
Title: COVID-19 VACCINATION INDUCED DONOR–SPECIFIC HLA ANTIBODY IN A RENAL TRANSPLAN PATIENT – A CASE REPORT
Authors: <u > A. ABU-KHADER</u>, I. Galaszkiewicz, F. Khan, N. Berka, Histocompatibility and Immunogenetics Laboratory, Alberta Precision Laboratories, University of Calgary, Calgary, Alberta, CANADA|<u > A. ABU-KHADER</u>, F. Khan, N. Berka, Department of Pathology a
Aim: Donor-specific HLA antibody (DSA) is associated with the risk of allograft loss due to antibody-mediated rejection (AMR). The ongoing pandemic is stipulated to impact transplant recipients both in terms of their chance to receive an allograft as well as the prognosis of transplant. Many reports emphasized the ability of the Covid-19 infection to induce the humoral immunity and the ability to form memory immune responses in patients who received solid organ transplants. Herein we report the formation of de novo DSA in a renal patient on waiting list after receiving mRNA Covid-19 vaccine, with no previous graft and no blood transfusion. The patient also received dTap-IPV (Diphtheria, Toxoid, Pertussis, Polio, and Tetanus Toxoid) and Hepatitis B vaccines in the last six months before Covid-19 vaccine. Methods: We present the case of a 42-year-old man, who has consistently tested negative for Covid-19 virus and is been worked up for a renal transplant. The patient has received two doses of a Covid-19 mRNA-based vaccination. We conducted both allo and auto preliminary and final flow cytometry crossmatches (FCXM) with a potential living donor. Patient’s sera were analyzed for HLA antibodies by LABScreen® Single Antigen bead (SAB), Luminex before and after the first dose of Covid-19 vaccination. Results: Before Covid-19 mRNA vaccination, HLA antibody screening, SAB testing, and preliminary T-cell and B-cell FCXM were all negative. Patient has no sensitization events before and after vaccination, however after twenty days of Covid-19 vaccination, final FCXM results for both T-cell (MCS 3SD shift = 185, our lab cutoff = 66) and B-cell (MCS 3SD shift = 149, our lab cutoff = 97) was strongly positive. We further performed class I Ab screening and SAB testing and our analysis confirmed the formation of new de novo DSA against B57 (MFI = 12,211), a de novo non-DSA against B58 (MFI = 10,920). Final auto-FCXM results were also negative for both T-cell (MCS 3SD shift = -18) and B-cell (MCS 3SD shift = -40) ruling out potential autoantibodies. Conclusion: These findings suggest the ability of this patients’ immune system to induce the formation of anti-HLA antibodies (both DSA and non-DSA), after mRNA Covid-19 vaccination. Caution must be considered in renal patients waiting for solid organ transplant to screen for development of de novo DSA after single or double doses of Covid-19 vaccines.
P37
Case studies
Title: DETECTION OF A COMPLEX NOVEL ALLELE BY GENE TARGET ENRICHMENT WITH HYBRID CAPTURE
Authors: <u > L.T. Madden</u>, J. Edwards, H. Hogan, Research and Development, CareDx, Applecross, Western Australia, AUSTRALIA|V. Van Sandt, J. Kerkhofs, S. De Pelsmaeker, L. Daniëls, Rode Kruis-Vlaanderen, Mechelen, BELGIUM|
Aim: A limitation of gene target enrichment by PCR in sequencing-based HLA typing assays is allele dropout due to amplification primer site polymorphisms or amplification primer site loss due to sequence deletion. Gene target enrichment by hybrid capture is not prone to the same limitations. We report a novel allele that contains a ∼ 1500 bp deletion in < i > HLA-C</i > starting in intron 7 and continuing into the 3′UTR. This deletion was detected by hybrid capture but not by PCR based NGS. Methods: HLA-C results from two samples resulted in detection of 2 × alleles by SSO and a single allele by PCR based NGS. The samples were then tested using AlloSeq™ Tx 17, a 17 locus MHC typing assay that uses hybrid capture for sequence target enrichment, revealing the presence of 2 × alleles for which a confirmed genotype was not possible in the AlloSeq™ Assign® sequence analysis software. We used an in-house software application to extract motifs specific for the novel allele at the 3′ end of the gene and analyzed these against a whole genome reference using Geneious Prime® (2021.1.1 Auckland N.Z., “Geneious”) in order to characterize the deletion. Results: The two samples, confirmed as being non-related, were typed as HLA-C*05/C*07 and HLA-C*03/C*05 by SSO, respectively. PCR based NGS failed to detect the C*05 allele in both samples. Typing by AlloSeq™ Tx 17 detected a potential C*05 allele but accurate genotyping was not possible due to several sequence mismatches at the 3’ of HLA-C that were consistent with loss of heterozygosity. Furthermore, the C*05 specific reads terminated 7 bases into intron 7. Alignment in Geneious confirmed the presence of a deletion of approximately 1500 bp starting in intron 7 for both samples. PCR based NGS assays use amplification primers in the 5’ and 3’ UTR regions of HLA class I to obtain full gene sequence. It is likely that the HLA-C*05 allele deletion included the amplification primer site and resulted in complete dropout of this allele by PCR enrichment. Conclusion: This case study highlights the limitations of gene target enrichment by PCR for HLA typing. Gene target enrichment using probes for hybrid capture that span the length of genes do not have the same limitations and, in this case, enable accurate characterization of the novel allele. The impact of the deletion on HLA-C expression has not been elucidated.
P38
Case studies
Title: GRAFT VS HOST DISEASE WITH ACELLULAR BONE MARROW FOLLOWING KIDNEY/PANCREAS TRANSPLANT
Authors: <u > S. Kanangat</u>, A. Vardouniotis, I. Skaljic, S. Piggot, M.H. Prod, Pathology, Rush University Medical Center, Chicago, Illinois, UNITED STATES|O. Olaitan, University Transplant Program, Rush University Medical Center, Chicago, Illinois, UNITED STATES|
Aim: To describe a fatal case of a graft vs host disease (GVHD) following Kidney Pancreas transplant resulting in acellular bone marrow Methods: The HLA typing of the donor and recipient was done by NGS (CareDx Allo Seq), post-transplant chimerism analysis by PowerPlex 16, and Immunophenotyping of peripheral blood (PB) and bone marrow (BM) by 10 color flow cytometry. Biopsies were performed to confirm GVHD and to determine if there was an immunological rejection of the allograft. Results: A 47-year-old male who had a SPK transplant maintained on CellCept and Prograf, was admitted for infectious vs toxic-metabolic etiology, acute liver injury with elevated LFTs, diarrhea and fatigue. Patient was started empirically on cefepime, vanc, acyclovir, ampicillin and doxycycline due to high suspicion of meningitis/encephalitis. Blood count deteriorated rapidly -[Ref attached Fig] and BM biopsy showed aplastic, hypocellular, fatty marrow suggesting acute toxic effect. The skin biopsy confirmed acute GVHD. Genomic DNA from the patient’s PB had both recipient and donor HLA present. Chimerism analysis showed persistent presence of allograft donor derived T cells by analysis of informative loci- alleles Ref attached Fig]. There were no HLA DSAs, and no indications of graft dysfunctions or immunological rejections. On 120 of hospitalization, the patient was moved to hospice and died shortly. Conclusion: GVHD following solid organ transplantation (SOT) is very rare and it is exceedingly rare after renal transplant, with fatality rate of ∼ 70% <sup > 1–2</sup > . Early detection may help to effectively treat the GVHD; however, many cases may go unnoticed as drug associated reactions. Migration of donor immune cells from the allograft to the recipient’s lymphoid organs have has been documented in human and animal studies. The outcome is determined by the balance between the donor’s cells and the recipient’s immune system, which is apparently compromised in transplant setting. Early recognition of GVHD following SOT is key to successful treatment < sup > 2.</sup > . Reliable methods for early differentiation of drug related toxicities from acute GVHD are warrented. < br /> References < br /> 1. H. Evin Gulbahce HE et al . Graft-vs-Host Disease After Solid Organ Transplant Am J Clin Pathol 2003;119:568 N. < br /> 2. Zacharias, N et al. Graft-versus-Host Disease after Living-Unrelated Kidney Transplantation 2014, Article ID 971,426
P39
Case studies
Title: THE IMPORTANCE OF EXAMINING THE FUNCTIONAL IMPACT OF NOVEL ALLELES: A CASE STUDY OF A NOVEL NULL DRB1 ALLELE
Authors: <u > J.L. Duke</u>, D. Ferriola, M. Ponisciak, S. Heron, S. Morris, A. Dinh, D.S. Monos, Immunogenetics Laboratory, Department of Pathology and Laboratory Medicine, The Children's Hospital of Philadelphia, Gap, Pennsylvania, UNITED STATES|A. Dinh, D.S. Mono
Aim: Detecting novel alleles through next generation sequencing for HLA typing is not uncommon, however the functional impact of novelties is not always immediately apparent. Here we present work undertaken to elucidate the functional impact of a novel DRB1 allele. Methods: Blood from a 5yo female with sickle cell disease being evaluated for an unrelated donor stem cell transplant was HLA typed using the Omixon Holotype V2 kit and sequenced on the Illumina MiSeq. Results: The initial evaluation of the patient indicated DRB1*07:01:01 and a novel DRB1 allele, identical to the DRB1*14:54:01 allele apart from a single base deletion of ‘G’ in exon 4, codon 216c. The deletion occurs in the fourth to last codon of the transmembrane region, causes a frameshift, and is suspected to also impact translation through the end of the protein. The full extent of the deletion on the protein was unknown as the Omixon V2 kit does not amplify beyond exon 4 of DRB1, and thus it was resequenced with in-house primers that characterize from exon 2 to the 3′ untranslated region. No further differences identified. The novelty was confirmed in a buccal sample from the patient and upon evaluation of her parents, found to be inherited on her paternal haplotype. The functional impact of the novel allele is not initially evident as most of the change to the DRB1 protein chain impacts the cytoplasmic tail. We questioned whether the deletion would modify the expression of the DR molecule on the cell surface or the ability of the protein to transmit signals within the cell. In order to fully advise our clinical program on the proper search strategy for this patient, we performed serological HLA typing, whereby the patient is serologically negative for DR14, indicating the novel allele is not present on the cell surface and the novelty encodes a null DRB1 allele. Conclusion: Due to the novel, but null, DRB1*14 allele, and the presence of DRB3*02:02:01, it will be difficult to find a well-matched unrelated donor for this patient and it has been recommended to pursue a cord blood transplant. This case illustrates the importance of fully investigating novel alleles, from the nucleotide sequence, translation of codons to the protein, through to the functional consequences of the novelty, as the result can change the way the clinical program will evaluate the patient and proceed with the appropriate search for a donor.
P40
Title: ANTIBODY-INDEPENDENT ASSOCIATIONS BETWEEN HLA MISMATCHES AND HISTOLOGY SUGGESTIVE OF ANTIBODY-MEDIATED INJURY UNCOVER THE NON-SPECIFICITY OF MICROVASCULAR LESIONS.
Authors: <u > A. Senev</u>, M. Naesens, Department of Microbiology, Immunology and Transplantation, KU Leuven, Leuven, BELGIUM|
Aim: The histology of antibody-mediated rejection (ABMR < sub > h</sub > ) after kidney transplantation is observed frequently in absence of detectable donor-specific anti-HLA antibodies (HLA-DSA). While there is an active interest in the role of non-HLA antibodies in this phenotype, it remains unknown whether HLA mismatches play an antibody-independent role in this phenotype of microcirculation inflammation. Methods: All consecutive adult kidney recipients transplanted at a single center between 2004 and 2013 were eligible for this study. The transplant pairs were retrospectively genotyped at high resolution for 11 HLA loci. < i > HLAMatchmaker</i>, <i > EMS-3D</i>, <i > HLA-EMMA</i > and < i > PIRCHE-II</i > tools were used to determine the degree of HLA molecular mismatches. The recipients with available biopsy follow-up were retrospectively genotyped for the presence of 14 KIR genes. Results: Of 893 kidney recipients, 121 (13.5%) developed ABMR < sub > h</sub > in the absence of HLA-DSA. In multivariable Cox analysis, all different calculations of the degree of HLA mismatch associated with the risk for developing HLA-DSA negative ABMR < sub > h </sub>(<i><b > Figure 1</b></i > ). This association was neither dependent on the correlation between HLA mismatch and T-cell mediated rejection nor on the formation of < i > de novo</i > HLA-DSA. Additional analyses also showed that HLA mismatch scores were associated with an increased incidence of ABMR < sub > h</sub > in the absence of HLA-DSA and anti-HLA antibodies, independently of the presence of high Missing self. Finally, glomerulitis and C4d deposition in peritubular capillaries also associated with the degree of HLA mismatch in the absence of circulating anti-HLA antibodies. Conclusion: The histology of antibody-mediated rejection and its defining lesions are also observed in patients without circulating anti-HLA antibodies and relate to the degree of HLA mismatch. This indicates that the donor-recipient HLA-mismatch partially explains the development of the histological of ABMR in an HLA antibody-independent process. These data highlight the need to clearly differentiate the Banff histological picture from the underlying pathophysiology of the kidney transplant biopsies.
P41
Clinical research, Solid organ
Title: INVESTIGATING THE ROLE OF A LARGE PANEL OF NON-HLA ANTIBODIES IN THE OCCURRENCE OF HISTOLOGY OF ANTIBODY-MEDIATED REJECTION AFTER KIDNEY TRANSPLANTATION.
Authors: <u > A. Senev</u>, M. Naesens, Department of Microbiology, Immunology and Transplantation, KU Leuven, Leuven, BELGIUM|B. Ray, C. Heylen, J. Hariharan, Immucor, Norcross, Georgia, UNITED STATES|
Aim: Many kidney allografts fail due to the occurrence of antibody-mediated rejection (ABMR), related to donor-specific anti-HLA antibodies (HLA-DSA). However, the histology of ABMR can be observed also in patients without HLA-DSA. While some non-HLA antibodies have been related to the histology of ABMR, it is still not well known to what extent they contribute to kidney allograft injury. Here we aimed to investigate the role of 82 different non-HLA antibodies in the occurrence of histology of ABMR after kidney transplantation. Methods: We included all patients who underwent kidney transplantation between 2004 and 2013 in a single center and who had biobanked pretransplant serum. The sera were retrospectively tested for the presence of 82 different non-HLA antibodies using a prototype bead assay on Luminex (Immucor, Inc). For each target, a ratio was calculated between the measured MFI value and the cut-off MFI defined by the vendor. Antibodies were assigned as positive if the calculated ratio was ≥ 1. Histology of ABMR was defined according to the first 2 Banff 2019 criteria for ABMR. Results: 874 patients had available pretransplant serum and were included in this analysis. Of them, 133 (15.2%) received a repeat kidney allograft, and 100 (11.4%) had pretransplant HLA-DSA. In total, 204 (23.3%) patients developed histology of ABMR after kidney transplantation. In 78 patients (38.2%) the histology of ABMR was explained by the presence of pretransplant or < i > de novo</i > HLA-DSA. We performed univariable Cox analysis to investigate the associations between the histology of ABMR and 80 different pretransplant antibodies. We identified 11 different pretransplant antibodies that associated with an increased rate of histology of ABMR (p < 0.05) (<b > Figure 1A</b > ). In a sensitivity Cox analysis restricted to HLA-DSA negative patients (N = 774), we identified antibodies against 16 non-HLA antigenic targets associated with the histology of ABMR developed in the absence of HLA-DSA (p < 0.05) (<b > Figure 1B</b > ). < b> </b > Conclusion: We found pretransplant antibodies against multiple non-HLA antigenic targets to be associated with increased rates of histology of ABMR developed in the absence of HLA-DSA in kidney transplant recipients. Whether these associations represent causality warrants further studies.
P42
Clinical research, Solid organ
Title: THE PREVALENCE AND TARGETS OF ALLELE-SPECIFIC ANTIBODIES IN PATIENTS WAITLISTED FOR SOLID ORGAN TRANSPLANTATION
Authors: <u > A.J. Gareau</u>, J. Jones, J. Metz, D.P. Lucas, S. Krummey, M. Bettinotti, Johns Hopkins University, Baltimore, Maryland, UNITED STATES|
Aim: Allele-specific antibody can present a significant barrier to solid organ transplantation, particularly in highly sensitized patients waiting for a re-transplant. These offers initially appear to be matched at the serological level, but many patients have high-level antibody at the allele level that cannot always be resolved in time to accept a deceased donor offer with current typing methods. The aim of this study was to identify the primary targets of these allele-specific antibodies to inform future testing methods. Methods: HLA antibody specificities listed as unacceptable were compiled from our laboratory database at the allele level from 4,358 waitlisted patients. HLA-DPB1 specificities were excluded. HLA-A, -B, and -DRB1 specificities with an MFI > 6,000 on single antigen bead testing were considered consistent with a positive T or B cell crossmatch on flow cytometry, and an MFI > 12,000 was used as a cutoff for HLA-C, DRB3/4/5, DQA1 or DQB1 specificities. Results: A total of 2904 unique allele-specific antibody assignments from 1103 patients were identified. The majority of these specificities (63%, n = 1829) were against class II alleles, with the remaining 37% (n = 1075) against class I alleles. Class I allele-specific antibody primarily targeted HLA-A (70%, n = 750), with 29% (308/1075) against HLA-B and a small number against HLA-C (17/1075). Of the class II specificities, 40% were directed against HLA-DRB1 (739/1829) with the majority against DR4 (234/739, 32%) and DR14 (297/739, 40%). Specificities targeting DQB1 alleles were overwhelmingly directed against DQ6 (480/510, 94%) with DQB1*06:01 (120/480, 25%) and DQB1*06:03 (118/480, 25%) representing the majority of assignments. There were 532 unique assignments against DRB3/4/5, with the majority (432/532, 81%) against DRB3. Of the class II targets, 42% of the allele-specific antibodies were at flow level or above for DRB1, 31% were at or above flow level for DQB1, and 34% were at or above flow level for DRB3/4/5. Conclusion: Specific alleles can sometimes be ruled out for a potential donor, but this is not universal. Molecular typing methods are needed that allow for definitive identification at the allele level in a deceased donor on-call scenario. This would further increase access to transplantation for highly sensitized patients.
P43
Clinical research, HCT
Title: DONOR-SPECIFIC ANTI-HLA ANTIBODIES ARE ASSOCIATED WITH POOR OUTCOMES FOLLOWING HAPLOIDENTICAL TRANSPLANTATION WITH POSTTRANSPLANT CYCLOPHOSPHAMIDE IN PEDIATRIC PATIENTS WITH NONMALIGNANT DISORDERS
Authors: <u > A.C. Lima</u>, J. Getz, G. Borsato do Amaral, N. Farah Pereira, Immunogenetics Laboratory, Federal University of Parana, Curitiba, Paraná, BRAZIL|C. Bonfim, G. Loth, S. Kanaan Nabhan, R. Pasquini, Bone Marrow Transplantation Unit, Federal University of
Aim: Donor-specific HLA antibodies (DSAs) have been recognized as a major risk factor for graft failure (GF) in adult patients with malignancies undergoing haploidentical transplantation with posttransplant cyclophosphamide (haplo-PTCy). However, the impact of DSAs after pediatric haplo-PTCy for nonmalignant disorders (NMDs) has been poorly reported. Thus, we retrospectively evaluated the role of DSAs in a cohort of 59 pediatric patients with NMDs who underwent haplo-PTCy at our institution. Methods: DSA testing was performed with Single Antigen Beads kits (LABScreen ExPlex and Immucor LIFECODES) using EDTA-treated sera. Mean fluorescence intensity (MFI) > 1000 was considered positive, and MFI < 1000 and > 500 was considered potentially positive, based on epitope reactivity patterns. Suspected false-positive reactions were assessed with acid-treated beads or adsorption/elution. Neutrophil and platelet engraftment and GF were the primary endpoints, whereas event-free survival (GF or death) and overall survival were the secondary endpoints. Multivariable analyses were carried out with Fine-Gray competing risk regression or Cox proportional-hazards regression models. Statistical analyses were performed using EZR version 1.53. Results: Overall, 15 patients (25.4%) had DSAs > 500 MFI. Of them, 4 had false-positive DSAs (median MFI = 1762). Of the 11 patients with true DSA reactivity, 5 had 1 DSA, 5 had 2 DSAs, and 1 had 3 DSAs, with a median MFI of 4162 (range, 527–24200) at haplo-PTCy. Four patients underwent desensitization with rituximab and plasmapheresis, and seven were untreated. All patients with treated DSAs had sustained engraftment. In the multivariable analyses adjusted for confounders, untreated DSAs were associated with lower neutrophil recovery, increased GF, inferior event-free survival, and poor overall survival (Table 1). Moreover, both treated and untreated DSAs adversely affected platelet recovery (Table 1). Conclusion: Our results indicate that the presence of DSAs is an independent predictor of poor outcomes following pediatric haplo-PTCy for NMDs.
P44
Clinical research, Solid organ
Title: DSA-NEGATIVE HLA ANTIBODY-NEGATIVE MOLECULAR ABMR RELEASES DONOR-DERIVED CELL-FREE DNA IN THE TRIFECTA STUDY
Authors: <u > P.F. Halloran</u>, J. Reeve, K. Madill-Thomsen, University of Alberta, Edmonton, Alberta, CANADA|L.G. Hidalgo, University of Wisconsin, Madison, Wisconsin, UNITED STATES|Z. Demko, A. Prewett, P. Billings, Natera Inc., San Carlos, California, UNITED STA
Aim: Trifecta (ClinicalTrials.gov #NCT04239703) is a prospective, investigator-initiated, IRB-approved, and consented study of the relationships between three central molecular tests in kidney transplant indication biopsies: molecular biopsy diagnoses (Molecular Microscope < sup>®</sup > Diagnostic System; MMDx), %dd-cfDNA, and HLA antibody testing. Methods: All central measurements were available for 219 biopsies. MMDx was assessed at ATAGC. Natera Inc. performed central %dd-cfDNA testing (Prospera). One Lambda Inc. performed HLA antibody testing, which LGH interpreted as DSA using local donor-recipient genotyping. We recorded local standard-of-care histology and DSA data. Results: Central and local DSA generally agreed: 97/103 (94%) of DSA-negative (DSA-) biopsies by local testing were also negative by central testing. < br /> <br /> By MMDx, 56 biopsies were called ABMR/mixed, of which 21 (38%) were DSA + by central testing (Figure 1) and 43 (77%) had %dd-cfDNA > 1.0. Of 35 DSA- ABMR/mixed, 26 (74%) had %dd-cfDNA > 1.0. Of 26 DSA- ABMR/mixed with %dd-cfDNA > 1.0, 20 were also negative for all HLA antibody (“PRA-”) by central testing (Table 1). Similar trends were seen for histologic ABMR: many were DSA-, some were PRA- and yet gave %dd-cfDNA > 1.0. < br /> <br /> Moreover, 7/20 PRA-/DSA- ABMR with %dd-cfDNA > 1.0 were C4d-positive, indicating complement activation. < br /> <br /> ABMR molecular classifiers and %dd-cfDNA both predicted central DSA better than local DSA (AUC 0.79 vs. 0.70, and AUC 0.73 vs. 0.65, respectively). As predictors of MMDx ABMR/mixed biopsy status, the balanced accuracy (mean of specificity + sensitivity) of %dd-cfDNA > 1.0 was higher than that of DSA (%dd-cfDNA 0.78; central DSA 0.64; local DSA 0.67). Similarly, %dd-cfDNA predicted histologic ABMR/mixed better than DSA (AUCs %dd-cfDNA 0.71; central DSA 0.66; local DSA 0.64). Conclusion: In < b> </b > Trifecta, most molecular ABMR/mixed biopsies have %dd-cfDNA > 1.0 but many are DSA negative, and some have no HLA antibody. ABMR without DSA may be explained by undetected alloantibodies (HLA, non-HLA) or autoantibodies, but also raises the possibility of another biologic mechanism targeting donor microcirculation e.g. NK activation by missing self. We conclude that in the Trifecta indication biopsy population, %dd-cfDNA is a robust feature of active ABMR regardless of DSA or of HLA antibody, and should be integrated into the ABMR diagnostic criteria.
P45
Clinical research, Solid organ
Title: ANTITHYMOCYTE GLOBULIN IS ASSOCIATE WITH A LOWER INCIDENCE OF DE NOVO DONOR-SPECIFIC ANTIBODY DETECTION IN LUNG TRANSPLANT RECIPIENTS A SINGLE-CENTER EXPERIENCE
Authors: T. Narula, S. Khouzam, <u > M. Elrefaei</u>, Mayo Clinic, Jacksonville, Florida, UNITED STATES|
Aim: De novo donor-specific HLA antibodies (DSA) are associated with a significant negative impact on lung transplant outcomes. Induction immunosuppression has improved the long-term outcomes after lung transplant.This is the first report exploring the association of induction immunosuppression on the development of de novo DSA in lung transplant recipients. Methods: Sixty-seven consecutive primary lung transplant recipients (LTR) were followed for t3 years post-transplant. 41 of 67 (61%) LTR received induction immunosuppression using a single dose of rabbit Antithymocyte Globulin (rATG; 1.5 mg/kg) within 24 h of transplant. All recipients had negative cytotoxicity, and flow cytometry crossmatches on the day of transplant. Serum samples at 1, 3, 6, and 12 months post-lung transplant were assessed for the presence of de novo HLA antibodies. Results: De novo HLA DSA were detected in 22/67 (32.8%) LTR within 1-year post-transplant. Of these, 9/41 (21.9 %) occurred in the induction therapy group and 13/26 (50%) in the non-induction group. Class II DSA were detected in 3/41 (7.3%) LTR who received induction as compared to 9/26 (34.6%) LTR without Induction immunosuppression (p = 0.005). However, the differences in overall survival or freedom from CLAD rates between the 2 groups were not statistically significant (p > 0.05). Conclusion: <br /> Induction immunosuppression utilizing a modified regimen of single-dose rATG is associated with a significant reduction in de novo DSA production in LTR
P46
Clinical research, Solid organ
Title: IMPACTS OF THE 2021 CHANGES TO DECEASED DONOR KIDNEY ALLOCATION- ORGAN PROCUREMENT ORGANIZATION AND TRANSPLANT PROGRAM PERSPECTIVE
Authors: <u > P. Rao</u>, G.P. Morris, Pathology, University of California San Diego, La Jolla, California, UNITED STATES|C. Wainaina, J. Trageser, Lifesharing, San Diego, California, UNITED STATES|
Aim: OPTN removed local donation service area (LDSA) for deceased donor kidney allocation and transitioned to a 250 nautical mile (NM) radius from the donor hospital on March 15, 2021. This change was made with a goal of reducing access disparities based on location, though a potential for negative impacts on efficiency of allocation was questioned. Our laboratory provides histocompatibility testing for both individual transplant centers as well as the local Organ Procurement Organization, providing an opportunity to examine how the NM allocation policy affected histocompatibility evaluation and organ placement for deceased donor kidneys. Methods: We reviewed laboratory testing and placement of locally recovered kidneys as well as for deceased donor kidneys transplanted at our center since implementation of the NM allocation (3/15/2021 – 4/30/2021), with comparison to COVID-era (9/1/2020 – 2/28/2021) and pre-COVID-era LDSA allocation (9/1/2019 – 2/28/2020). Results: Examining placement of locally-recovered kidneys, NM allocation reduced allocation to LDSA transplant centers, from 64.0% in both pre-COVID and COVID-era LDSA-based allocation to 18.5% (P < 0.0001). Placement of kidneys to centers within the 250 NM radius increased from 9.9% in pre-COVID and 3.6% in COVID-era LDSA-based allocation to 48.1% (P < 0.0001). Allocation of kidneys to regional centers not in the 250 NM circle was not increased. Fewer potential recipients from local transplant programs were tested per local donor (4.4 NM-era vs. 7.4 LDSA-era). From the transplant program perspective, the number of deceased donor kidney transplant offers from outside the LDSA evaluated by cellular crossmatch testing increased dramatically, from 9.3/month in pre-COVID and 7.7/month COVID-era LDSA allocation to 25.0/month (P < 0.0001). Conclusion: Implementation of the 250 NM allocation has resulted in a significant change to the offers for allocation of deceased donor kidneys. Our data indicate that the new allocation system has increased the portion of offers extended to patients outside of the LDSA, and subsequently increased allocation of these organs outside of the LDSA and within the 250 NM radius. It is currently too early to determine the net impact of decreased local organ offers and increased offers from outside the LSA on individual transplant programs.
P47
Clinical research, Solid organ
Title: NOVEL BEAD-BASED ASSAY ASSESSES THE IMPACT OF IMLIFIDASE ON ABO IGG ANTIBODIES
Authors: <u > A.M. Halpin</u>, B. Motyka, T. Ellis, J. Pearcey, L.J. West, Pediatrics, University of Alberta, Edmonton, Alberta, CANADA|<u > A.M. Halpin</u>, Histocompatibility, Alberta Precision Laboratories, Edmonton, Alberta, CANADA|<u > A.M. Halpin</u>, L.J. West, L
Aim: The accurate assessment and measurement of ABO antibodies (ABO-Abs) is critical in ABO-incompatible (ABOi) transplantation and IgG and IgM isotypes need to be considered carefully. Standard hemagglutination methods may not accurately detect the effectiveness of ABO-Ab removal strategies. Imlifidase cleaves IgG, which prevents Fc-mediated effector functions. Our aim was to assess the impact of imlifidase using a novel bead-based assay to measure IgG and IgM ABO-Abs and compare the results with titres determined using a traditional hemagglutination assay. Methods: Luminex beads were coupled to specific A glycan I-VI and B glycan I-VI subtypes. These beads were used to measure IgG and IgM glycan subtype-specific ABO-Abs, expressed in mean fluorescence intensity (MFI), in sera of healthy ABO-O adults (n = 105). A subset of samples with high IgG ABO-Abs (n = 13) was treated < i > in vitro</i > with imlifidase and assessed with the Luminex assay. A further subset of imlifidase-treated samples (n = 5) was additionally tested by standard hemagglutination at room temperature; 50uL of serially diluted serum was incubated with 25uL of 1% commercial A1 and B reagent red cells at room temperature. The titre was reported as the last dilution that caused agglutination. Results: IgG and IgM ABO-Abs levels vary widely in healthy individuals and include high MFI ABO-Abs to IgM and IgG. < i > In vitro</i > treatment with imlifidase effectively reduced MFI signal for IgG ABO-Ab but IgM ABO-Ab MFI signal was unaffected (Fig 1A). Similar hemagglutination titres were found despite widely different ABO antibody profiles (Fig 1B). Hemagglutination results were unchanged in the imlifidase-treated samples despite the change in the antibody profiles as detected by the bead-based assay (Fig 1B). Conclusion: This Luminex ABO antibody assay enables measurement of both IgM and IgG ABO-Abs and their subtype-specificities and allows immunologic risk assessment of each isotype in ABOi transplantation. Hemagglutination cannot adequately identify ABO antibody profiles for management of ABOi organ transplantation and is not useful in monitoring the effectiveness of imlifidase treatment. Use of single antigen ABO beads measures the precise effect of imlifidase and provides a tool to investigate the potential role of imlifidase in ABOi transplantation in individuals with high levels of IgG ABO-Abs.
P48
Clinical research, Solid organ
Title: INCIDENCE OF HLA VERSUS NON-HLA ANTIBODY-MEDIATED REJECTION: A SINGLE-CENTER EXPERIENCE WITH HEART TRANSPLANTATION
Authors: <u > J.J. Xin</u>, A. Rabson, J. Cone Sullivan, Pathology, Tufts Medical Center, Boston, Massachusetts, UNITED STATES|A. Vest, Tufts Medical Center, Boston, Massachusetts, UNITED STATES|
Aim: Antibody Mediated Rejection (AMR) may occur in the absence of HLA Donor Specific Antibodies (DSA) and may be due to non-HLA antibodies. We sought to understand the frequency with which DSA versus non-HLA antibodies were implicated in AMR. Methods: We retrospectively reviewed cardiac allograft recipients followed at our single-center, academic center between January 2019 - May 2021. DSA were routinely monitored following cardiac allograft transplant. Patients with negative DSA (Mean Fluorescence Intensity, MFI < 1000) or low-level DSA (MFI 1000–5000) were tested for non-HLA antibodies including Anti-Angiotensin II Type 1 Receptor (AT1R), Anti-Major Histocompatibility Complex Class I Chain-Related A (MICA) and Anti-Endothelial Cell antibodies (AECA). AECA were detected by endothelial cell crossmatch. Results: Of 219 patients tested for DSA, 45 (21%) patients had clinically suspected AMR per symptoms, hemodynamics and/or echocardiogram. Of these, 27 (60%) patients had high-level DSA (MFI > 5000), 9 (20%) had low-level, and 9 (20%) were DSA negative (Fig. 1A). All patients with low-level DSA (n = 9/9, 100%) were positive for ≥ 1 non-HLA antibody (AT1R n = 4, MICA n = 1, AECA n = 1; both MICA and AECA n = 2). Among patients with high-level DSAs, DQ, DQA and DR53 were the most common DSA specificities (Fig 1B). Among patients with low-level DSAs, DSA specificities were relatively evenly distributed between HLA Class I and II, and of concurrent non-HLA antibodies, AT1R was more frequent than MICA or AECA (Fig 1C). All DSA negative patients with AMR had high-level (>20 U/ml) AT1R antibody (n = 7 > 40 U/ml, n = 1 31 U/ml, n = 1 21 U/ml). 2 were AECA positive (Fig 1D). Conclusion: DSA and non-HLA antibodies may independently and/or synergistically mediate cardiac allograft dysfunction. AT1R was commonly detected (n = 13/45, 29%) in our cardiac allograft dysfunction population. If AMR is clinically suspected and no DSAs or only low-level DSAs are detected, it is worth looking further at the non-HLA antibodies.
P49
Clinical research, Immunotherapy
Title: INDIVIDUALIZED GENETIC MAKEUP THAT CONTROLS NATURAL KILLER CELL FUNCTION INFLUENCES THE EFFICACY OF ISATUXIMAB IMMUNOTHERAPY IN PATIENTS WITH MULTIPLE MYELOMA.
Authors: <u > R. Rajalingam</u>, H. Sun, D. Kong, Immunogenetics and Transplantation Laboratory, University of California San Francisco, San Francisco, California, UNITED STATES|S. Macé, M. Chiron, Translational and Experimental Medicine, Sanofi Research & Developme
Aim: Phase-Ib clinical trial with Isatuximab-Lenalidomide(Len)-Dexamethasone(Dex) showed an improved progression-free survival (PFS) in patients with relapsed or refractory multiple myeloma (RRMM), but the efficacy varied by patient. Antibody-dependent cell-mediated cytotoxicity (ADCC) by natural killer (NK) cells plays a crucial role in arbitrating antitumor activities of therapeutic antibodies. We tested if patient-specific genetic makeup known to set NK cell functional threshold influence response to Isatuximab-Len-Dex therapy. Methods: We characterized 57 RRMM patients receiving Isatuximab-Len-Dex for polymorphisms of killer-cell immunoglobulin-like receptors (KIR), HLA class I, and FCGR3A loci. < i > In</i><i > vitro</i > ADCC assay, co-incubating primary NK cells expressing specific KIR repertoire with multiple myeloma cell lines (MM cells) expressing selected HLA class I ligands, was utilized to confirm the identified genetic correlatives of clinical response. < i > Trial registration number: </i > NCT01749969 Results: Patients with KIR3DL2 + and its cognate-ligand HLA-A3/11 + had superior PFS than patients missing this combination (hazard ratio[HR] = 0.43; P = 0.02), while patients carrying KIR2DL1 + and HLA-C2C2 + compared to patients missing this pair showed short PFS (HR = 3.54; P = 0.05) (Fig.1A, B). Patients with KIR3DL2 + and HLA-A3/11 + plus high-affinity FCGR3A-158 V allele showed the most prolonged PFS (HR = 0.35; P = 0.007). Consistent with these clinical data, mechanistic experiments demonstrated that NK cells expressing KIR3DL2 trigger greater ADCC when MM cells express HLA-A3/11 (Fig.1C). Inversely, NK cells expressing KIR2DL1 do not kill if MM cells express the HLA-C2C2 ligand (Fig.1D). NK cells expressing high-affinity FCGR3A-158VV induced greater ADCC compared to those with low-affinity FCGR3A-158FF. Conclusion: Our results suggest that KIR3DL2 + and HLA-A3/11 + with FCGR3A-158 V markers lead to enhanced Isatuximab-dependent NK-mediated cytolysis against MM cells and results in improved PFS in RRMM patients treated by Isatuximab-Len-Dex. Moreover, the presence of KIR2DL1 + and HLA-C2C2 + identifies patients who may have a lower response to Isatuximab-Len-Dex therapy linked to a reduced NK-mediated ADCC. These biomarkers could potentially identify, via precision medicine, patients more likely to respond or resistant to Isatuximab-Len-Dex immunotherapy.
Fig. 1. The Kaplan-Meier curves showing the impact of specific KIR + HLA combinations on progression-free survival (PFS) of relapsed or refractory multiple myeloma (RRMM) patients treated by Isatuximab + Len + Dex (A, B). Mechanistic studies confirm the role of these KIR + HLA interactions in modulating isatuximab-dependent NK cytolysis of MM cells (C, D).
P50
Clinical research, Solid organ
Title: ASSOCIATION BETWEEN NON-HLA ANTIBODIES AND OUTCOMES IN KIDNEY TRANSPLANT
Authors: A.M. Sanfilippo, <u > J. Pena</u>, Pathology, Beth Israel Deaconess Medical Center, Boston, Massachusetts, UNITED STATES|P. Nissaisorakarn, F. Cardarelli, Medicine, Beth Israel Deaconess Medical Center, Boston, Massachusetts, UNITED STATES|A. Pandit, Medici
Aim: A major barrier to successful long-term kidney transplant function is HLA alloimmunization and rejection. However, there is growing evidence that antibodies against non-HLA antigens may independently, or in conjunction with HLA donor-specific antibodies (DSA), mediate allograft failure. In a subset of kidney transplant patients who do not meet criteria for antibody-mediated rejection, allograft dysfunction may be accompanied by abnormal biopsy findings, but without HLA DSA. We sought to determine if the presence of non-HLA antibodies is associated with poor allograft function. Methods: Patients who received a kidney transplant between 2015 and 2018 were retrospectively tested for non-HLA antibodies using One Lambda Labscreen® Autoantibody kits. Three groups of patients were included: patients who had a for-cause biopsy but did not have HLA DSA (n = 30, for-cause bx), patients who developed de novo HLA DSA (n = 14, <i > dn </i > HLA DSA) and patients with normal allograft function (n = 12, control). Allograft biopsies were done in the for-cause bx and dn DSA groups. Results: At one-year post-transplant, controls had the best allograft function compared to the < i > dn</i > HLA DSA or for-cause bx groups measured by eGFR in mL/min/1.73 m < sup > 2 </sup>(mean eGFR 67.7, SD 15.8, mean eGFR 64, SD 16.6, and mean eGFR 52.13, SD 21.63 respectively; <i > P</i> =0.04). The < i > dn</i > HLA DSA had generally better allograft function compared to the for-cause bx group (<i > P</i> =0.062). A similar finding was observed using the mean urine protein:creatinine ratio (in mg/mg) in controls (0.11, SD 0.03), <i > dn </i > HLA DSA (0.18, SD 0.14) and for-cause bx (0.64, SD 0.9) (<i > P=</i > 0.033). Again, <i > dn</i > HLA DSA had generally better allograft function compared to the for-cause bx group (<i > P</i>=0.013). Conclusion: At 1-year post-transplant, allograft function was best in the control group, followed by the < i > dn</i > HLA DSA group, and worst in patients who underwent for-cause biopsy. Patients in the for-cause bx group had increased non-HLA antibodies (PRKCH, REG3 and GAPDH) compared to patients with < i > dn</i > HLA DSA. These antibody targets have been implicated in non-kidney allograft rejection and may represent potential mediators of poor kidney allograft outcomes independent of HLA DSA. Additional studies are needed to further elucidate the role of non-HLA antibodies in kidney transplant.
P51
Clinical research, Solid organ
Title: EARLY ASSESSMENT OF THE IMPACT OF CHANGES IN UNOS DECEASED DONOR KIDNEY ALLOCATION FOR PATIENTS TRANSPLANTED AT UCLA
Authors: <u > J. Zhang</u>, M. Hickey, N.M. Valenzuela, C. Butler, R.A. Sosa, E. Reed, Pathology, UCLA Immunogenetics Center, Los Angeles, California, UNITED STATES|E. Lum, A. Basmah, J. Yabu, E. Kendrick, T. Pham, G. Danovitch, Division of Nephrology, David Geffen
Aim: On March 15, 2021, a new kidney transplant allocation policy was implemented to remove donor service areas and regions. The allocation is now based on geographical distance between donor hospital and recipient transplant center. The goal of this study is to understand the impact of the new allocation policy on deceased kidney transplantation at UCLA. Methods: One Legacy donorsare defined as “Local” donor; non-One Legacy donors identified within 250 nautical miles (NM) to UCLA is defined as “Local Import” donors; donors identified outside of 250NM circle from UCLA are defined as “National Import” donors. Donor OPO as well as patient cPRA were collected for patients who received a deceased donor transplant between 3/15 to 5/21 in 2019, 2020 and 2021. Results: There were 48, 28 and 81 patients who received deceased kidney transplantation during the study period in 2019, 2020 and 2021 at UCLA, respectively. There was a significant reduction in deceased donor transplantation in 2020 due to the COVID-19 pandemic. In 2021, 45/81 (55.6%) patients received deceased donor transplant from “local” donors, similar to 24/48 (50%) in 2019. Local import donor transplant represented 15/48 (31.3%) in 2019 and only 16/81(19.7%) in 2021 (Figure 1. n.s). However, there were a total of 22 non-sensitized patients transplanted out of 1443 non-sensitized waitlist patients (1.5%) in 2019 compared to 44/1172 (3.6%) in 2021 (p < 0.01, Table 1). Within 250NM of UCLA, 19/1443 (1.3%) non-sensitized received transplant in 2019 compared to 27/1216 (3.0%) patients in 2021 (p < 0.05). There was a significant increase in transplant of 100% cPRA patients in 2021 (6/29: 20.1%) compared to 2019 (2/39: 5.1%; p < 0.05). However, the number of patients in this group is relatively small, and significant effects could multifactorial. Conclusion: The early analysis of the impact of the new Kidney Allocation policy showed there was a slight reduction in deceased donor transplant from local import donors at UCLA. In addition, since the new policy allows candidates inside 250NM circle who located closest to the donor hospital receive 2 maximum proximity points. These extra points may be the cause led to increased transplantation of non-sensitized patients within ‘local’ and 250NM.
Fig. 1. UCLA Deceased Donor Transplants (March 15-May 31).
P52
Clinical research, Solid organ
Title: THE NEW KIDNEY DISTRIBUTION SYSTEM: HAS IT CHANGED ANYTHING?
Authors: <u > A. Comeaux</u>, I. Salvador, R. Vorhaben, A. Usmani, C. Lacelle, University of Texas Southwestern Medical Center, Dallas, Texas, UNITED STATES|
Aim: The new kidney distribution system implemented on March 15, 2021 allocates kidneys based on concentric circles of 250 and 500NM from the donor center rather than by local/regional distribution based on donor service area and OPTN regions. This single center study aims to investigate the impact of the new system on donor origination, KDPI, and DCD status. It also aims to determine whether the use of physical vs virtual crossmatching is affected by the new system. Methods: First, we performed a query in UNET to obtain a list of all deceased donor kidney transplant performed at our center between January 1 and May 16, 2021. Donor geographical location, KDPI, and DCD status were collected. Second, we calculated the distance between the donor and transplant center in NM and gathered crossmatch data from our information system. Results: A total of 77 deceased kidney transplants were performed at our center between January 1 and May 16, 2021. 31 transplants occurred prior to and 46 after the implementation of the new kidney distribution system. 58.1% of deceased kidney transplants originated from our OPO pre- vs 28.3% post- implementation. While a marked decreased in the number of kidneys coming from our OPO was observed, the distance from the donor to the transplant center only slightly increased with an average of 132 NM pre- vs 194NM post-implementation. The decrease in kidneys originating from our OPO was associated with an increase in kidney from neighboring OPOs within 250NM from our center. When looking at KDPI the average KDPI of donor was similar pre (59%) and post-implementation (61%). In contrast 29% of kidneys were from DCD donors pre- compared to 72.2% post-implementation of the new system. An increase in the number of transplants performed on prospective virtual crossmatch from 74.2% pre- to 84.8% post-implementation was also observed. Conclusion: The new kidney distribution system did not substantially change the distance travelled by kidneys however, a greater number of kidneys originated from outside our OPO. A substantial increase in the number of DCD donors transplanted at our center was observed post implementation and may be due to decrease usage of DCD kidneys in neighboring OPOs. A slight increase in the number of patients transplanted on virtual crossmatches was also observed and is likely due to the availability of blood for crossmatch pre-organ collection when donor originates from inside vs outside our OPO.
P53
Clinical research, HCT
Title: FREQUENCY OF HLA ANTIGENS IN THE DONOR POOL DOES NOT ADEQUATELY EXPLAIN WHY SOME PLATELET REFRACTORY PATIENTS HAVE TO BE SUPPORTED BY HLA INCOMPATIBLE PLATELETS
Authors: <u > J. Pena</u>, Pathology, Beth Israel Deaconess Medical Center, Boston, Massachusetts, UNITED STATES|J. Cone Sullivan, Pathology, Tufts Medical Center, Boston, Massachusetts, UNITED STATES|
Aim: Platelet transfusion support of HLA-alloimmunized platelet refractory patients with hematologic disease can be a challenge. When clinically indicated, we use HLA incompatible (HLAi) platelets, which are donor units which an intended recipient has donor HLA antigen-specific antibodies (DSA). We hypothesize that when we resort to HLAi transfusions, high-frequency HLA antigens are more likely targets by DSA than lower frequency antigens. Methods: We performed a 2-year retrospective review of > 700 platelet transfusions and analyzed d ata for HLA-alloimmune refractory patients. Antibodies were identified using single antigen assay with mean fluorescence intensity > 1000. We calculated the frequency of individual HLA antigens in the general donor pool using the organ procurement transplant network calculator: https://optn.transplant.hrsa.gov/resources/allocation-calculators/cpra-calculator/. Results: Donor HLA typing and recipient antibodies was available for 279 transfusion events with 38 (14%) requiring the use of HLAi platelets. In these HLAi transfusions, 76.3% had a single donor antigen targeted by DSA; 23.7% of such platelets were targeted by multiple DSA. There were 27 distinct HLA antigen targets represented a spectrum of common antigens (HLA-A2, -A3 and -B44) to less common antigens (e.g. HLA-B67 and B75). Conclusion: In our study of HLAi transfusions, the antigenic targets were a spectrum of frequent and less frequent antigens. Thus, the frequency of HLA antigenic targets by patient DSA alone is probably not a good predictor for determining which patients will be more difficult to provide HLA compatible platelets. Additional studies are needed to determine what factors predict difficulty in transfusion support of HLA refractory patients
P54
Clinical research, HCT
Title: CHARACTERIZATION OF B*15:220 CARRYING HAPLOTYPES IN SUBJECTS OF MULTIPLE ETHNICITIES LIVING IN QATAR
Authors: <u > S.A. Saleh</u>, Department of Laboratory Medicine and Pathology, Hamad Medical Corporation, Doha, QATAR|M. Askar, The Transplant Immunology Laboratory, Baylor University Medical Center, Dallas, Texas, UNITED STATES|
Aim: HLA-B*15:220 has only one non-synonym nucleotide substitution in exon 1 at position 47 (C > T) compared to its closest allele HLA-B*15:03:01 (-9 Ala > Val). In 2020, it has been recently added to the common, intermediate, and well-documented HLA alleles version 3.0. HLA-B*15:220 carrying haplotypes have been reported previously in populations from Brazil, Russia, and United Arab Emirates. In 2018, our laboratory at Hamad Medical Corporation, State of Qatar, validated the next-generation sequencing for HLA genotyping for solid organ and hematopoietic cell transplantation as well as non-transplant purposes. The population in the State of Qatar is close to 3 million and is multi-ethnic with close to 85% expatriates from almost all countries of the world. Methods: Review of our laboratory information system for subjects carrying HLA-B*15:220. We also reviewed the literature for HLA-B*15:220 carrying haplotypes. Results: We identified HLA-B*15:220 allele in 10 patients. Five of them, their haplotypes are similar with Brazil, Russia, and United Arab Emirates population. Two other probable haplotypes were identified in two families (3 and 2 family members, respectively). < br /> <br /> Description of HLA-B*15:220 carrying haplotypes with reference to previously reported similar haplotypes in other populations and new probable haplotype reported in Qatar are described in the table below. Conclusion: The wide utilization of NGS in HLA laboratories has facilitated characterization of haplotypes carrying recently well-documented alleles in different populations. In addition to the previously reported association with HLA-C*04:01 and C*12:03, we report an association between HLA-B*15:220 and HLA-C*07:01 observed in 2 Sudanese non-HLA identical siblings.
P55
Clinical research, Solid organ
Title: THE INCREASED NUMBER OF AUTOANTIBODY IS ASSOCIATED WITH REDUCED LONG-TERM SURVIVAL OF LIVER TRANSPLANT
Authors: <u > Q. Xu</u>, Pathology, University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, UNITED STATES|A. Sidahmed, S. Leckie, London Health Sciences Centre, London, Ontario, CANADA|V. McAlister, University of Western Ontario, London, Ontario, CANADA|
Aim: Recent studies indicated that autoantibodies are detrimental to the survival of solid organ transplants, usually in synergy with antibodies specific to donor mismatched HLA (DSA). Numerous autoantibodies have been reported in transplantation literature. Here we examine if the number of positive autoantibodies has any impact on graft survival after liver retransplantation. Methods: <br /> In this retrospective study, 91 patients who received a second liver transplant were included. Pre-retransplant sera were tested with Luminex-based solid-phase assay (Immucor Inc.) for IgG antibodies to 89 autoantigens. Raw MFI was used for data analysis. Three times of trimmed mean of 94 normal control sera were positive thresholds for each autoantibody. The primary outcome is survival of the second liver graft. Results: 47/91 (51.6%) patients had lost second liver regrafts, while 44/91 (48.4%) still had functioning regrafts. All except 8 patients had at least one positive autoantibodies (mean = 16.3, SD = 20.9, range = 0–87). The number of autoantibodies was higher in patients who lost liver regrafts [median (IQR): 9.0 (3.5–26.0)] than patients with functioning regraft [median (IQR): 5.5 (2.0–16.5)], but it’s not significant (p = 0.157). In univariate Cox regression, 6 autoantibodies (designated as “panel-6”) were associated with increased risk for graft loss (p < 0.10): Iodotyrosine deiodinase (HR = 1.79, p = 0.095), GAPDH (HR = 1.70, p = 0.078), Prelamin A/C (HR = 1.76, p = 0.07), Phospholipase A2 receptor 1 (HR = 1.64, p = 0.099), Angiotensin II Type I Receptor cell line-2 (HR = 1.80, p = 0.073), Tubulin-beta (HR = 1.81, p = 0.087). The number of panel-6 autoantibodies were significantly higher (p = 0.002) in patients who lost liver regrafts [median (IQR): 2.0 (0.5–3.5)] than patients with functioning regraft [median (IQR): 0.0 (0.0–1.5)]. The increased number of panel-6 autoantibodies was associated with a higher risk for graft loss in univariate analysis [HR = 1.17(1.02–1.33) per incremental number; p = 0.023]. In the multivariate model adjusting DSA, recipient male gender and donor age, number of panel-6 autoantibodies was still significant contributor for graft loss [HR = 1.16 (1.01–1.33), p = 0.037). Conclusion: An increased number of autoantibodies is associated with an elevated risk for liver graft loss. Multiple antigen targets are likely involved in autoimmunity after organ transplantation, and their effects are additive.
P56
Clinical research, HCT
Title: DISTRIBUTION OF HIGH-RESOLUTION HLA-A, -B, -C, -DPB1, -DQB1, AND – DRB1 ALLELES IN THE PROVINCE OF QUEBEC, CANADA
Authors: <u > W. Lemieux</u>, A. Lewin, Medical Affairs & Innovation, Héma-Québec, Montréal, Quebec, CANADA|<u > W. Lemieux</u>, R. Sapir-Pichhadze, Centre for Outcomes Research and Evaluation (CORE), Research Institute of McGill University Health Centre, Montreal, Qu
Aim: The province of Quebec has a rich and complex settlement history that impacts the genetic diversity of its inhabitants. We previously described the distribution of low-resolution HLA-A, -B, and -DRB1 genotypes in the province of Quebec. In this study, we aimed to improve upon this work by considering high-resolution sequencing. Methods: Similar to the aforementioned study, we used Héma-Quebec’s bone marrow donor registry as a proxy of the general population. High-resolution HLA typing data were obtained for 3850 individuals from the stem cell donor registry. These individuals were routinely typed at the two-field allele level for HLA-A, -B, -C, -DPB1, -DQB1, and -DRB1 loci as part of the admission to the registry. Statistical analysis was performed using the GENE[RATE] population analysis tools and Python programming language. Hardy-Weinberg equilibrium was assessed with a nested likelihood procedure. Allele and haplotype frequencies were estimated for each region and frequencies outliers were identified. Using information on participants’ geographical residence, we also assessed how the distribution of HLA alleles and haplotypes may vary by the 17 sociodemographic regions in Quebec. Three regions (Côte-Nord, Nord-du-Québec, and Gaspésie-Îles-de-la Madeleine) included too few participants to be analyzed. Results: We were able to study HLA distribution in 14 of the 17 regions in Quebec. Most of the 14 studied regions (10/14) were in a Hardy-Weinberg equilibrium for HLA class I loci, whereas most regions (10/14) were in disequilibrium for at least one of the HLA class II loci. This demonstrates the different behavior of classI and class II loci in the province of Quebec. High-resolution allele and haplotype frequencies were comparable to the low-resolution frequencies identified in the previous study. In addition, there seemed to be a correlation between the number of outliers observed in allele frequencies and low immigration influx in the different regions. Conclusion: Our study provides a better understanding of the demographic pressures that shape the province of Quebec. This information will help inform future recruitment strategies for potential stem cell donors by administrative region in the province of Quebec.
P57
Clinical research, Solid organ
Title: CHANGES IN AUTOIMMUNITY STATUS CAN EFFECT DECEASED DONOR KIDNEY OFFER ACCEPTANCE.
Authors: <u > P. Jindra</u>, J. Magana, N. Chitnis, B. Murthy, A. Awan, Baylor College of Medicine, Houston, Texas, UNITED STATES|B. Murthy, A. Awan, Baylor St. Lukes' Medical Center, Houston, Texas, UNITED STATES|
Aim: The aim of this study was to demonstrate how auto-crossmatch results can change over time and help to rule out false positive deceased donor crossmatches in renal transplantation. Methods: Auto Flow Cytometry (FXM) and Complement Dependent Cytotoxicity (CDCXM) crossmatches were performed at the initial renal evaluation. When a deceased donor (DD) FXM was positive at the time of DD offer without the presence of DSA an auto FXM was additionally performed for 15 patients. Auto XM shifts were subtracted from DD FXM shifts to calculated the overall XM interpretation. Patients were followed post- transplant and monitored for the presence of DSA, serum creatine, GFR and biopsy proven rejection. Results: Within the FXM positive group with the absence of DSA (N = 15) we observed 86% had an auto FXM shift indicating anti-IgG Ab binding to self T cells or B cells at the time of transplant. Differentiating positive auto FXM by cell type: 33% B cell, 20% T cell, and 27% both T and B cell. Taking into account the positive auto FXM results the overall FXM interpretation was resulted as negative and patient’s were transplanted. As alternative method a cohort of 5 patients with documented autoimmunity had their sera auto-absorbed with their own cells on the pre-txp sample and all saw a reversal of their DD allo-XM from positive to negative. Post-transplant follow-up, only one patient had a suspicion for TCMR on biopsy, that was considered not clinically relevant, and serum creatine had improved spontaneously. No other rejections were observed of a 6 month period. However, all patients have CKD 3 T with GFR below 60 (except one patient with GFR of 67) which might indicate some underlying immunopathology. Conclusion: AutoXM has traditionally been performed at initial patient workup. While listed for deceased donor XM a patient’s autoimmune status may change and at the time of deceased donor offer could be elevated and lead to false positive allo-crossmatches. Our data suggest patient’s with a history of autoimmunity and high PRA could have a false positive allo-crossmatches in the absence of DSA. Not testing a patient’s autoreactivity at the time of organ offer may falsely deny an individual’s access to kidney transplantation who may receive very limited number of DD offers.
P58
Clinical research, Solid organ
Title: NAVIGATING NAUTICAL MILES-A SINGLE CENTER EXPERIENCE WITH THE NEW KIDNEY ALLOCATION POLICY
Authors: <u > J.A. Houp</u>, D. Anderson, V. Hauptfeld-Dolejsek, Surgery, University of Alabama at Birmingham, Birmingham, Alabama, UNITED STATES|V. Kumar, Nephrology, University of Alabama at Birmingham, Birmingham, Alabama, UNITED STATES|
Aim: UNOS has recently implemented a policy change to the kidney and pancreas allocation process (KAS). The change involves the removal of allocation by UNOS Region and local DSA (Donor Service Area) and replaces it with a 250 nautical mile (nm) circle. The intent is to increase access to transplant for key groups. It has been reported anecdotally, that this change has impacted histocompatibility labs significantly and variably, with some labs seeing an increase in the work load and requests for virtual crossmatch. We sought to examine the impact of this new KAS at our single center. Methods: We compared deceased donor kidney transplants that occurred at our center (transplant rate, CPRA, 250 nm inclusion) in the 3 months just prior to the allocation policy change and the first 3 months post-implementation. Results: Our center evaluated over 100 potential donors from the new 250 nm catchment area in the first three months of the policy change. 50% of these kidneys from 250 nm donors were placed at our center. The overall transplant rate for deceased donor kidneys increased by 27%. The most significant increase (53%) was observed in CPRA group 80–97% followed closely by 98–100 %CPRA group with a 50% increase. 0 and 1–79% CPRA groups had only marginal increases Conclusion: Early results appear to show that the allocation reorganization has benefit for more highly sensitized (>80 CPRA) waitlist candidates at our center. This is a critical group that may have been disadvantaged by KAS sliding scale CPRA allocation as compared to the (98–100% CPRA) group. It has been published previously that highly sensitized patients' chance for transplant increases with a larger pool of potential donors. The additional prioritization for circles beyond 250 nm may be benefitting our most highly sensitized (98–100% CPRA) group as well. Although a positive benefit for our center, it remains to be seen if this will remain a sustained change. Additionally, it is important to assess the impact this will have logistically for the histocompatibility lab as it poses a significant increase in requests for virtual crossmatches, including STAT testing and confirmatory typing. Finally, it should be noted that an increase for one center likely means a decrease for another center and the equity of the adjustment should be reviewed carefully for unintended consequences.
P59
Clinical research, Solid organ
Title: NON-HLA ANTIBODIES ASSOCIATED WITH PATHOLOGICAL FEATURES OF RENAL ALLOGRAFT REJECTION
Authors: <u > C. Butler</u>, M. Hickey, Y. Zheng, G. Sunga, D. Gjertson, E. Reed, Immunogenetics Center, Department of Pathology & Laboratory Medicine, UCLA, Los Angeles, California, UNITED STATES|M. Pearl, P. Weng, Pediatric Nephrology, UCLA, Los Angeles, Californi
Aim: Non-HLA antibodies (Abs) have been associated with antibody mediated and cellular rejection in kidney transplantation. The pathological features related to these non-HLA Abs are poorly understood. Methods: A single center pediatric kidney transplant recipient cohort (n = 62 patients; 34 without rejection and 28 with rejection) with paired plasma and protocol biopsies (6, 12, 24 months) was retrospectively tested for non-HLA Abs (Immucor LIFECODES). Results: Hazard ratio analysis of a single-center pediatric kidney allograft recipient cohort (n = 62) identified 15 non-HLA Abs associated with the time to first rejection (Figure 1; p < 0.1). Seven of these (DEXI, CSF2, IL-8, LGALS3, SNPRN, STAT6 and LGALS8) are newly described to be associated with renal transplant rejection. A mixed effect ANOVA analysis (p < 0.05) of the graft pathological features identified that 4/15 non-HLA Abs associated with rejection were independently associated with IFTA after removing samples with confounding HLA DSA. 12/15 were independently associated with arteriolar hyalinosis thickening after samples with confounding HLA DSA were removed. CSF2 was associated with acute peritubular capillaritis (ptc) and acute intimal arteritis after removing samples with confounding HLA DSA. Conclusion: The data show an association between non-HLA Abs and pathological features of rejection. Assessment of non-HLA Abs is useful to elucidate the incidence and identity of non-HLA Abs associated with kidney allograft rejection.
P60
Clinical research, Solid organ
Title: VISUALIZATION OF LINKAGE DISEQUILIBRIUM PATTERNS OF FREQUENT MISMATCHED AMINO ACID POSITIONS ACROSS HLA CLASS II LOCI: IMPLICATIONS FOR KIDNEY TRANSPLANT OUTCOMES STUDIES
Authors: <u > G.L. Wager</u>, D.G. Biagini, L. Gragert, Pathology and Laboratory Medicine, Tulane University School of Medicine, New Orleans, Louisiana, UNITED STATES|M. Kamoun, R. Urbanowicz, M. Dribus, University of Pennsylvania, Philadelphia, Pennsylvania, UNITED
Aim: Mismatching of HLA amino acids in kidney transplant pairs can inform outcomes models to predict graft failure (GF). Previous single center studies have utilized eplet mismatch counts in validated models. Our aim is to utilize larger-scale datasets to stratify risk of amino acid (AA) mismatch categories. Here we explore the impact of multicollinearity by computing asymmetric linkage disequilibrium (ALD) values between commonly mismatched AAs for HLA Class II loci, and evaluate stepwise logistic regression model (SLRM) selection to manage epistatic interactions. Methods: High resolution HLA typing was imputed for all classical HLA loci from donor and recipient antigen-level HLA assignments in the Scientific Registry of Transplant Recipients (SRTR) kidney dataset. AA polymorphisms were assigned to HLA Class II loci (DRB1, DQA1, DQB1) by random realization of high resolution haplotypes for donor-recipient pairs. Mismatch frequency was calculated for each AA across HLA Class II. ALD values were visualized for AAs mismatched in >=25% of pairs, based on NMDP reference population haplotype frequencies. A univariate SLRM identified DRB1, DQA1, and DQB1 AAs that independently associate with kidney GF. Results: AA mismatch frequency, SLRM selection results, and univariate associations with GF are provided in Table 1. DRB1 AAs were favored for incorporation in the SLRM relative to DQA1 and DQB1 loci. Complex patterns of ALD within HLA Class II haplotypes are visualized in Figure 1. Notably, the SLRM did not sequentially incorporate highly-correlated AAs. Conclusion: This preliminary analysis of SRTR data indicates that HLA Class II AAs confer differing risks of kidney GF. ALD calculations and SLRM selection techniques are useful for managing multicollinearity. Multicenter studies incorporating imputation offer greater statistical power for discovery, however high resolution typed datasets are needed for cross-validation.
P61
Clinical research, Solid organ
Title: HLA PROFILE OF DECEASED DONORS OF SOLID ORGANS AND POTENTIAL RECIPIENTS REGISTERED ON THE WAITING LIST FOR KIDNEY TRANSPLANTATION
Authors: A. Neunfeld, S.C. Wagner, Federal University of Health Science of Porto Alegre, Porto Alegre, Rio Grande do Sul, BRAZIL|J. Boquett, Department of Genetics, Universidade Federal do Rio Grande do Sul, Porto Alegre, Rio Grande do Sul, BRAZIL|J. Boquett, Facu
Aim: This study aimed to determine and compare the allele and haplotype frequencies in 5,903 potential recipients registered on the waiting list for kidney transplantation and in 2,290 deceased donors of solid organs in the southern region of Brazil in the period from 2011 to 2018, as well as to compare the allele frequencies of the samples under study with population allele frequencies in the Brazilian Bone Marrow Donor Registry (REDOME). Methods: Allele and haplotype frequency estimations, tests of Hardy-Weinberg equilibrium and Prevosti’s genetic distances with multidimensional scaling representation were performed with the Gene[RATE] tools. The difference between the allele frequencies between donors and recipients was calculated by Pearson's chi-square test using the FDR (False Discovery Rate) control method proposed by Benjamini & Hochberg, using the PyHLA software. Results: The study identified 20 HLA-A, 33 HLA-B, 15 HLA-C, 13 HLA-DRB1 and 5 HLA-DQB1 allele groups. The most frequent allele groups in the study for each locus were HLA-A*02, -B*35, -C*07, -DRB1*04, and - DQB1*03 and the most frequent haplotype for the set of 4 loci was HLA-A*01 ∼ B*08 ∼ DRB1*03 ∼ DQB1*02. In most comparisons, no significant differences were found between the two study samples, except for the allele groups HLA-B*41 (P = 0.0319), -DRB1*01 and -DRB1*14 (P = 0.0383 in both). The comparison with the Canadian database used to calculate Cpra did not show any significant difference between these two populations. Conclusion: This is the first study to characterize groups of deceased donors of solid organs and potential recipients for kidney transplantation in the southern region of Brazil based on HLA genotyping data. No significant differences were observed among donors and recipients HLA profile, likely meaning that, in average, the waiting time is mainly influenced by the anti-HLA sensitization history and not by discrepancies among the HLA profile. This study will serve as a basis for a calculator to estimate the waiting time for a given patient at the moment of the entrance in the waiting list.
P62
Clinical research, Solid organ
Title: PROSPECTIVE MATCHING OF RENAL TRANSPLANT PATIENTS IN A HETEROGENEOUS POPULATION USING PIRCHE-II ALGORITHM
Authors: <u > K. Sherwood</u>, J. Tran, J. Lan, P.A. Keown, University of British Columbia, Vancouver, British Columbia, CANADA|O. Gunther, GUNTHER Analytics, Vancouver, British Columbia, CANADA|M. Niemann, PIRCHE AG, Berlin, GERMANY|R. Sapir-Pichhadze, McGill Unive
Aim: Minimizing epitope disparity between donor and recipient is a critical strategy to optimize graft outcomes. We have previously demonstrated the feasibility of within-centre matching for antibody-verified epitopes via HLAMatchmaker in a heterogeneous Canadian population. Here, we model organ allocation to determine the potential compatibility of T-cell epitope-matching via PIRCHE-II scores. Methods: PIRCHE-II (v3.3.36) scores were pre-calculated for 300,150 possible recipient/donor pairs, using NGS HLA data for 1,150 patients and 261 deceased donors. PIRCHE-II scores were calculated using 5 principal genes (HLA-A, B, C, DRB1, DQB1). Simulation models implemented in R (3.5.3) incorporated ABO identity, waitlist rank and optimal score for matching and employed Canadian provincial waitlist sizes for a 1 year simulation without burn-in period. Scenario analyses considered three previously published PIRCHE-II mismatch thresholds (up to- 9, −35–90). Results: Active donor/recipient matching markedly enhanced the probability of achieving lower PIRCHE-II scores compared with a Canadian base case (Fig.1). In the matched model using five combined gene loci, the average median PIRCHE-ll score was reduced to 29 over 10 simulations (95% of donor/recipient pairs had a score of 49 or less), compared with an average median score of 79.25 for the base case simulation (with a 95%-cumulative probability score of 170). For individual gene loci, the probability of achieving a PIRCHE-II score of 0 ranged from 0.57 (HLA-B) to 0.85 (HLA-DRQB1). Cumulative probability for allocation of deceased donors with a match score of 0 across a range of Canadian renal transplant waitlist sizes is shown in Fig. 2. Using donor to recipient waitlist ratios specific to each province (data not shown), scenario analysis showed that approximately 40% of patients could be donor matched to achieve a 0 DRB1 PIRCH-II score with a waitlist size of ∼ 300. Conclusion: Modeling shows that active donor/recipient matching substantially reduces the PIRCHE-ll score for renal transplants in Canada. The magnitude of improvement increases with the size of the waitlist, suggesting that regional or national organ sharing may enhance success.
P63
Clinical research, Solid organ
Title: PRECISION MEDICINE IN TRANSPLANTATION: EPITOPE IDENTITY OR MINIMAL MISMATCH AT HUMAN LEUKOCYTE ANTIGEN DQ OR DR REDUCES THE PROBABILITY OF DONOR-SPECIFIC ANTIBODY AND PROVIDES A PRACTICAL AND FEASIBLE STRATEGY FOR OPTIMIZING COMPATIBILITY
Authors: <u > J. Tran</u>, J. Lan, F. Fenninger, K. Sherwood, S. Dobrer, P.A. Keown, G. GCTC, Pathology and Laboratory Medicine, University of British Columbia, Vancouver, British Columbia, CANADA|J. Lan, J. Gill, J. Gill, P.A. Keown, Medicine, University of British
Aim: We have demonstrated that prospective epitope matching at cardinal human leukocyte antigen (HLA) class II genes is a practical and feasible strategy to optimize compatibility in deceased donor transplantation. Here, we confirm that identical or minimal mismatch at HLA-DQ and –DR is associated with reduced formation of < i > de novo</i > donor-specific antibody (<i > dn</i > DSA), reinforcing the opportunity to actively reduce the risk of antibody-mediated rejection (AMR). Methods: Next generation sequencing was performed for 11 HLA genes on sequential, ethnically heterogeneous renal patients and their deceased donors (1155 pairs) transplanted in Vancouver, Canada from 2008 to 2018. Epitopes were determined using HLAMatchmaker and patients were stratified according to donor-recipient epitope identity or quantitative mismatch including thresholds defined by Wiebe et al. 2013. Post-transplant antibody testing for < i > dn</i > DSA was performed for cause and the occurrence of < i > dn</i > DSA and graft failure were compared using Kaplan-Meier survival analysis and log-rank tests. Results: For-cause post-transplant < i > dn</i > DSA testing was performed in 248 of 1155 patients (21%) and was a significant predictor (p < 0.0001) of death-censored graft failure (DCGF) (Fig 1a). Stratifying the recipient cohort by epitope mismatch across all HLA genes did not predict the probability of < i > dn</i > DSA testing or DCGF. Stratification by HLA class I and DP mismatches alone also did not predict < i > dn</i > DSA, though there were trends to improved outcomes in those with lower mismatches. In contrast, none of the patients with zero DQ (n = 51) or DR (n = 39) mismatches developed < i > dn</i > DSA (7 patients with a zero DQ score and 8 patients with a zero DR score had post-transplant testing, all tests were negative), and quantitative epitope mismatches of < 10 DQ or < 18 DR were associated with significantly reduced development of donor-specific < i > dn</i > DSA (Fig1b). Conclusion: These data confirm that a zero or minimal mismatch at HLA-DR or DQ substantially reduces the risk of < i > dn</i > DSA. Combined with our previous report on modeling matching feasibility and proven strategies for rapid epitope assignment for deceased donors, prospective epitope matching offers a practical strategy to optimize donor-recipient histocompatibility and minimize the risk of AMR.
Fig. 1. Survival analyses of various clinical outcomes in 1155 renal transplant recipients from Vancouver, Canada transplanted from 2008 to 2018. Kaplan-Meier analysis was used for the curves, and were compared using log-rank tests. A. Patient cohort stratified by whether or not they received a post-transplant de novo donor-specific antibody (dnDSA) test in relation to death-censored graft failure. B. Patient cohort stratified by HLA-DQ (left) and -DR (right) thresholds defined by Wiebe et al., 2013, in relation to their gene-specific dnDSA-free survival.
P64
Clinical research, Cellular therapies
Title: PROVISION OF HUMAN LEUKOCYTE ANTIGEN (HLA) MATCHED PLATELETS – 10-YEAR EXPERIENCE OF A REGIONAL BLOOD CENTRE IN HONG KONG
Authors: <u > J. Kwok</u>, K.W. Tam, S.K. Cheung, J.C. Ho, Division of Transplantation and Immunogenetics, Department of Pathology, Queen Mary Hospital, Hong Kong, HONG KONG|L. Chan, Medicine & Geriatrics, Princess Margaret Hospital, Hong Kong, HONG KONG|C. Wong, J.
Aim: Human leukocyte antigen (HLA) matched platelet concentrates are frequently requested in patients with platelet transfusion refractoriness due to the presence of anti-HLA antibodies. A retrospective study was conducted from May 2010 to April 2020 to evaluate the provision of HLA-matched platelets in Hong Kong, a regional blood centre. Methods: In the study period, 1,080 units of HLA Class I matched apheresis platelets were given to 147 patients (male 54%, mean age: 48 ± 21 years). The degree of HLA matching and HLA antibody results, and 1-hour corrected count increments (CCIs) were reviewed. Further analysis was performed to identify the predictors of haematological response. Results: All donor-recipient pairs were matched at HLA-A and B loci, except one with one mismatch at the HLA-B locus. The mean 1-hour CCI for all transfusions was significantly improved to 16.9 ± 8.9 m < sup > 2</sup>/L compared to previous random donor platelet transfusions and 84% of the transfusions achieved 1-hour CCIs ≥ 7.5 m < sup > 2</sup>/L. ABO blood group compatibility did not alter the 1-hour CCIs. Of the 54 patients tested for anti-HLA Class I antibodies, 41 (76%) was positive but none of them received platelet products containing the respective HLA antigens. Conclusion: In this cohort, satisfactory 1-hour CCIs were achieved in most HLA-matched apheresis platelet transfusions. As these apheresis donors are recruited from the Hong Kong Bone Marrow Donor Registry which currently consisted of>130,000 donors, the chance of finding a Class I HLA-matched donor is very high, almost 100%. This approach can therefore, reduce the incidence of platelets transfusion refractoriness due to the presence of anti-HLA antibodies whereas the speed in identifying suitable donors from the registry is much faster. On the other hand, advocating judicious request for the product and thorough investigations of platelet transfusion refractoriness could also enhance the clinical management.
P65
COVID-19
Title: HLA-A*11:01:01:01, HLA*C*12:02:02:01-HLA-B*52:01:02:02, AGE AND SEX ARE ASSOCIATED WITH SEVERITY OF JAPANESE COVID-19 WITH RESPIRATORY FAILURE
Authors: <u > S. Khor</u>, Y. Omae, K. Tokunaga, Genome Medical Science Project , National Center for Global Health and Medicine, Tokyo, JAPAN|N. Kinoshita, T. Suzuki, M. Suzuki, N. Ohmagari, Disease Control and Prevention Center, National Center for Global Health a
Aim: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus causing coronavirus disease 2019 (COVID-19) was announced as an outbreak by the World Health Organization (WHO) in January 2020 and as a pandemic in March 2020. The majority of infected individuals have experienced no or only mild symptoms, ranging from fully asymptomatic cases to mild pneumonic disease. However, a minority of infected individuals develop severe respiratory symptoms. The objective of this study was to identify susceptible HLA alleles and clinical markers that can be used in risk prediction model for the early identification of severe COVID-19 among hospitalized COVID-19 patients. Methods: A total of 137 patients with mild COVID-19 (mCOVID-19) and 53 patients with severe COVID-19 (sCOVID-19) were recruited from the Center Hospital < b> </b > of the National Center for Global Health and Medicine (NCGM), Tokyo, Japan for the period of February–August 2020. High-resolution sequencing-based typing for eight HLA genes was performed using next-generation sequencing. Results: In the HLA association studies, HLA-A*11:01:01:01 [P < sub > c</sub> = 0.013, OR = 2.26 (1.27–3.91)] and HLA-C*12:02:02:01-HLA-B*52:01:01:02 [P < sub > c</sub> = 0.020, OR = 2.25 (1.24–3.92)] were found to be significantly associated with the severity of COVID-19. After multivariate analysis controlling for other confounding factors and comorbidities, HLA-A*11:01:01:01 [P = 3.34E-03, OR = 3.41 (1.50–7.73)], age at diagnosis [P = 1.29E-02, OR = 1.04 (1.01–1.07)] and sex at birth [P = 8.88E-03, OR = 2.92 (1.31–6.54)] remained significant. The area under the curve of the risk prediction model utilizing HLA-A*11:01:01:01, age at diagnosis, and sex at birth was 0.772, with sensitivity of 0.715 and specificity of 0.717. Conclusion: To the best of our knowledge, this is the first article which describes associations of HLA alleles with COVID-19 at the 4-field (highest) resolution level. Early identification of potential sCOVID-19 could help clinicians prioritize medical utility and significantly decrease mortality from COVID-19.
P66
COVID-19
Title: ABO AND COVID-19: TISSUE ABH GLYCANS, ABO ANTIBODIES AND SUSCEPTIBILITY TO SARS-COV-2 INFECTION
Authors: L.J. West, Dept of Laboratory Medicine and Pathology, University of Alberta, Edmonton, Alberta, CANADA|<u > A.M. Halpin</u>, B. Motyka, T. Ellis, J. Pearcey, R. Bascom, L.J. West, Pediatrics, University of Alberta, Edmonton, Alberta, CANADA|<u > A.M. Halpin</
Aim: Population-based and GWAS studies have reported an association between ABO blood group and SARS-CoV-2 susceptibility, with ABO blood group A (ABO-A) individuals relatively more susceptible than other ABO-groups, and ABO-O individuals relatively protected. We aimed to define possible roles of ABH glycans and ABO antibodies in susceptibility to SARS-CoV-2 infection. Methods: I) We examined the ABH glycan decoration of lung and nasal tissue from individuals of known ABO blood groups by immunohistochemistry using a panel of monoclonal antibodies to ABH glycan subtype antigens. < br /> II) We investigated ABH glycan binding properties of the SARS-CoV-2 receptor binding domain (RBD) protein using electrospray ionization mass spectrometry (ESI-MS). < br /> III) We measured ABO antibodies (isotype and ABH glycan subtype-specificity) in PCR + COVID-19 patients (n = 68) and healthy controls (n = 41) with a novel Luminex bead-based assay. Results: I) < i > Tissue ABH:</i > In ABO-A individuals, upper (sustentacular cells) and lower airway epithelial cells were found to be decorated with A-subtype II and III/IV glycans, but lung endothelium was decorated with only A-subtype II glycans. ABO-A-glycans on lung epithelial and endothelial cells were co-localized with ACE2 (<b > Figure 1</b > ). < br /> II) < i > Glycan binding:</i > A-glycans bound RBD protein (<b > Figure 2</b > ). < br /> III) < i > ABO antibodies: </i > In ABO-O individuals, IgG and IgM anti-A antibodies specific for subtypes II-IV were significantly lower in individuals with COVID-19 compared to healthy controls (<b > Figure 3</b > ). Conclusion: Expression of tissue A-glycans and their co-localization with ACE2 in airway epithelial cells of ABO-A individuals, together with their ability to bind spike RBD proteins, are consistent with a possible viral co-receptor role, as described for other viruses. This could enhance infection susceptibility in ABO-A individuals. We have previously shown that healthy ABO-O individuals have a wide range of anti-A antibody levels (IgG, IgM, IgA), thus cannot be categorized into one risk group. This heterogeneity is likely of key importance in understanding the population-level association between ABO-O blood group and lower risk of COVID-19 infection. Our preliminary data support the hypothesis that ABO-O individuals with lower levels of anti-A antibodies, perhaps of a particular subtype-specificity and/or isotype, may be at higher risk of infection than ABO-O individuals with more abundant anti-A antibodies.
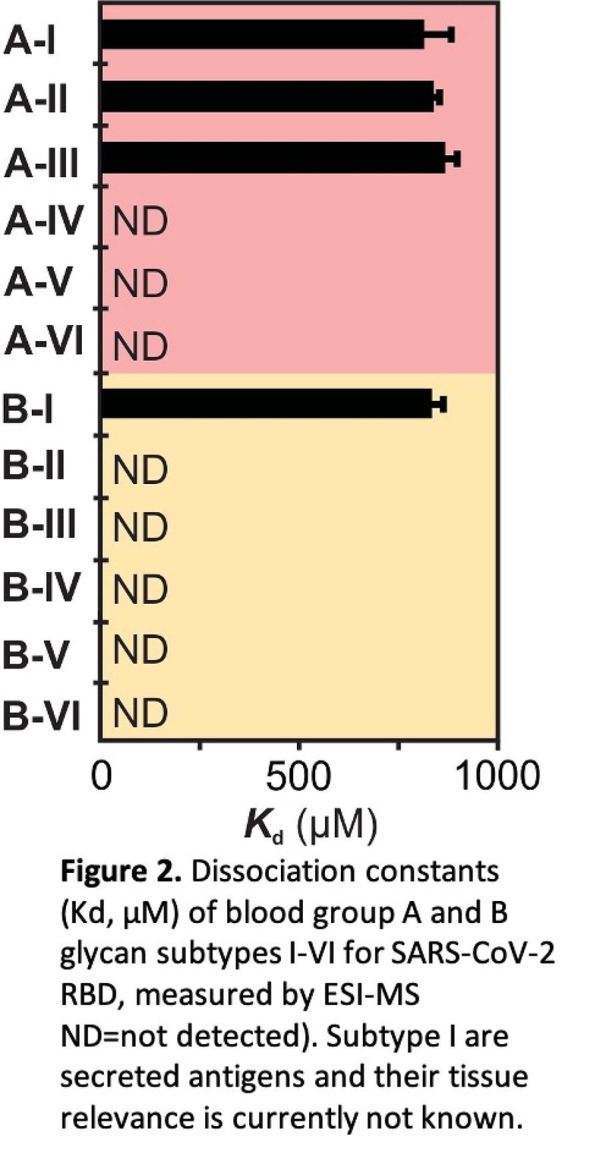
P67
COVID-19
Title: ABO/Rh MAY CONTRIBUTE TO THE DIFFERENT CLINICAL FORMS OF EXPRESSION OF SARS-COV-2-COVID-19 IN MEXICAN MESTIZOS FROM THE HIGHLANDS AND CENTER MEXICO
Authors: K.I. Rivera, H. Schabib, C.F. Mercado, C.J. Balderas, A. Del Olmo, M.A. Carmona, A. Florentino, A. Rodriguez, <u > C. Gorodezky</u>, Laboratory of Immunology and Immunogenetics, Fundación Comparte Vida, A.C., Mexico City, CDMX, MEXICO|
Aim: Over 173 million cases of COVID19 have been confirmed and 4 million deaths were reported (WHO, June 2021). Most people develop slight to moderate respiratory disease. Elder patients with chronic conditions are more susceptible to the illness and a group of people although infected, stays asymptomatic. The relationship between ABO and COVID-19 has been shown, since high titer isoagglutinin can avoid the transmission, thus anti-A, anti-B and Anti-AB antibodies may be partly responsible for the inhabitation of the infection. It was reported based in a meta-analysis of 105 countries, that A group people are at a high risk of infection and severity, whereas group O is related to low risk.The goal was to look for susceptibility/protection ABO/Rh association in Mexicans with COVID-19. Methods: We selected 281 Mestizos from the Center and Highlands Mexico (195 controls/86 COVID-19 patients); RNA extracted from nasopharyngeal swabs (Promega Corp.) was examined by RT-PCR (GeneFinder COVID 19 PLUS RealAmp kit, OSANG HealthCare Co.) on the ABI 7500 instrument. The v2.3 Software was used for analysis. ABO and Rh antigens were typed using direct tube hemagglutination with Anti-A, Anti-B, Anti-AB and Anti-D sera (kindly donated by Lafon Lab.). SPSS 25 Software, was used for statistics. The cases were divided in: 1)Asymptomatic; 2)Influenza type symptoms; 3)hospitalized with oxygen requirement; 4)Intensive care patients. Results: Of the 86 positive COVID-19 cases and 195 controls, ranging in age between 16 and 87 years, 24.4%, 3.5%, 1.2% and 70.9% were groups A, B, AB and O; 95.3%Rh + and 4.7 Rh- of the patients; 22.6%, 6.7%, 2.1, 68.7% of the controls were A, B, AB and O; 94.9% were Rh + and 5.1%, Rh-. Univariate analysis showed no significant difference among blood groups. However, the multivariate analysis, demonstrated that group O contributes to protection, since G1 Vs. controls had a pc = 0.02, G3 pc = 0.005, and G4 pc = 0.0001, being the 3 RR = 0.007. Conclusion: Our study supports group O as protective to COVID-19 infection. The association prevails protecting to a more serious disease as shown by the sub-group analysis. Studies specially done in Asians, demonstrate the same protective effect of O group. Although the mechanism is unknown, it is suggested that it may be mediated by natural anti-A antibodies or by the influence of plasma glycoproteins (FVIII and VWF).
P68
COVID-19
Title: IMPACT OF COVID-19 INFECTION ON ANTI-HLA ANTIBODY PROFILES OF KIDNEY TRANSPLANT RECIPIENTS
Authors: J. Ge, E. Caglar, E. Qama, N. Hayde, E. Akalin, <u > A. Colovai</u>, Montefiore Medical Center, Bronx, New York, UNITED STATES|
Aim: SARS-CoV-2 infection affected a large population of kidney transplant recipients, predominantly Hispanic and African American, followed at our center. This prompted significant changes in immunosuppression (IS) to allow for a more robust immune response against the virus. To determine the effects of SARS-CoV-2 infection and IS changes on HLA sensitization, we explored anti-HLA antibody profiles before and after COVID-19 infection in 82 transplant recipients. Methods: From March 16 < sup > th</sup > 2020 to June 1 < sup > st</sup > 2020, 132 kidney transplant recipients were diagnosed with COVID-19, as confirmed by RT-PCR. In 82 survivors, anti-HLA antibody testing was performed prior to and post- COVID-19 diagnosis at a median time of 4 months pre- and 8 months post-COVID, using One Lambda Single Antigen beads. Molecular HLA typing of patients and donors was available for the identification of donor specific antibodies (DSA). As most patients experienced severe disease, IS changes consisted of anti-metabolite withdrawal and calcineurin inhibitor dose reduction. Results: The characteristics of the patient population are presented in Table 1. Anti-HLA antibody results indicated that mean cPRA class I increased from 11% to 14% (p = 0.002, paired t-test), and cPRA class II increased from 23% to 26% (p < 0.001) after COVID-19 infection. The number of patients with detectable anti-HLA antibodies increased from 48% to 57% (Fig 1). Fifteen (18%) patients had DSA prior to COVID-19 diagnosis, primarily directed to DQ antigens (N = 13). In 11 out of 15 patients, DSA were also detected post-COVID infection, with no significant change in MFI values (mean MFI 10,013 pre- vs 11,491 post-COVD). Interestingly, 4 out of 15 patients lost their DSA after COVID infection. These DSA had MFI values lower than 5000. Five patients (6%) with cPRA 0% and no DSA before COVID infection developed DSA at a median time of 2 months post-COVID infection. All DSA were directed to DQ antigens (mean MFI 15240). Out of 5 patients, there was one graft loss and one death over 10 months of follow-up post-COVID infection. Among the remaining 77 patients, 3 lost their grafts and one patient died. Conclusion: These results indicate that HLA sensitization in kidney transplant recipients increased after COVID-19 infection. However, despite significant reduction in IS, development of new DSA occurred in<10% of patients, possibly due to virus induced immune dysregulation.
Fig. 1. cPRA distribution in 82 kidney transplant recipients pre- and post-COVID-19 infection.
P69
COVID-19
Title: ANALYSIS OF HLA ANTIBODIES AFTER COVID-19 MRNA VACCINATION IN POST-TRANSPLANT KIDNEY RECIPIENTS AND HEALTHY VOLUNTEERS
Authors: <u > M. McAuliffe</u>, M. Nevski, G. Wedermyer, C.L. Roark, B.M. Freed, ClinImmune Labs, Aurora, Colorado, UNITED STATES|
Aim: Standard protein vaccines, such as the influenza vaccine, have been shown to correlate with transient anti-HLA antibody production. With the first widespread use of mRNA vaccines to combat the novel Coronavirus (SARS-CoV-2), we evaluated if COVID mRNA vaccines would induce < i > de novo</i > HLA antibody or increase production of existing HLA antibodies. Methods: Study participants included post-transplant kidney recipients (n = 8) and healthy volunteers (n = 10). Study participants received two doses of either Pfizer or Moderna COVID mRNA vaccines and varied in age, sex, and race. Of the healthy volunteers, 4 had a history of sensitization through pregnancy. Healthy volunteers had a sample drawn before the first vaccine dose to establish a baseline. Post-transplant participants either had a consistent, multi-year PRA history or a sample within four months pre-vaccination. Some participants had samples drawn two weeks following their first dose but prior to receiving their second vaccine dose. All participants had samples drawn between two weeks to two months following the second vaccine administration. All samples were analyzed for class I and class II HLA antibodies using OneLambda Single Antigen beads on the Luminex platform. Samples from post second vaccine dose were compared to the pre-vaccine profile. A major sensitization was classified as an increase in MFI of HLA antibody>2000. A minor sensitization was classified as an increase in MFI between 1000 and 1999. Appearance of antibodies that One Lambda has specified as false positive when seen in isolation or antibodies that were negative on OneLambda Screen were not considered. Results: No major sensitizations were identified. Four minor sensitizations occurred. Two were seen in healthy volunteers, with one of the sensitizations affecting a participant without history of sensitization, and two in post-transplant patients. The cPRA did not increase in any of the sensitizations. Despite the presence of minor sensitizations, the changes identified were consistent with baseline changes expected for samples drawn weeks to months apart. Conclusion: Based on this data, the COVID mRNA vaccines do not appear to induce significant < i > de novo</i > HLA antibody or increase production of existing HLA antibodies. Further study with a larger sample size may be warranted as vaccine-induced HLA sensitization is a rare phenomenon.
P70
COVID-19
Title: SARS-COV-2 ANTIBODY RESPONSE AMONG LABORATORY STAFF AFTER VIRAL EXPOSURE AND VACCINATION
Authors: <u > N.A. Lemp</u>, L.M. Saquilayan, J.J. Chew, S.S. Musa, N. Kasahara, J.C. Cicciarelli, HLA Laboratory, VRL Eurofins, Los Angeles, California, UNITED STATES|R. Montes, B. Williams, J.C. Cicciarelli, HLA Laboratory, Sharp Healthcare, San Diego, California,
Aim: The COVID-19 pandemic presents particular risks to staff in the clinical laboratory setting. Here we sought to examine antibody responses against SARS-CoV-2 among laboratory staff after exposure to the virus and vaccination. Methods: Sera from the staff of two different labs (total n = 39 individuals) after vaccination with either Moderna or Pfizer vaccines (n = 12 with a prior history of COVID-19 preceding vaccination, designated as ‘COVID+’) were tested with the LABScreen COVID Plus kit for antibodies against whole SARS-CoV-2 spike protein, spike S1 and S2 subunits, S1 receptor binding domain (RBD), nucleocapsid (NC) protein, and S1 subunits from SARS, MERS, and four other HCoV. These sera were tested either 2 weeks after the 1st vaccine dose (Dose1), ≥2 weeks after the 2nd dose (Dose2), or after both doses (n = 5, of which n = 2 were COVID + ). Pre-pandemic sera (n = 3) from patients in 2019 served as controls. Results: Strong antibodies against SARS-CoV-2 spike protein were detected in all staff sera, with whole spike > RBD > S1≫S2 antibodies. Notably, antibodies detected in Dose1 sera were weaker than in Dose2 sera, particularly against S2. After both doses, COVID- sera showed an increase in all SARS-CoV-2 spike antibodies, particularly against S2, but COVID + sera showed little change. Pre-vaccination sera were not collected, so it was not possible to determine if either vaccine increased antibody levels in COVID + participants. Negligible NC antibodies were detected from COVID- sera, as expected since both mRNA vaccines encode only the spike protein, while NC antibodies were detected in all COVID + sera, as all viral proteins including NC are expressed during infection. Notably, antibodies against SARS S1 were also detected in all sera, with higher levels in COVID + than COVID-, and none in controls. Indeed, cross-reactivity between SARS antisera and SARS-CoV-2 spike was previously reported using the LABScreen COVID Plus kit. No antibodies against MERS S1 were detected. Antibodies against all four other HCoV S1 were detected at varying levels, with no significant difference between COVID-, COVID+, and control sera. Conclusion: Our results indicate that this is an informative method to assess anti-CoV antibodies, and that using this kit, cross-reactivity of post-vaccination sera is not observed with MERS S1, and appears unlikely with circulating HCoV, but cross-reactivity with SARS S1 cannot be ruled out.
P71
COVID-19
Title: THE IMPACT OF COVID-19 PANDEMIC ON HISTOCOMPATIBILITY LABORATORY
Authors: <u > R. Al attas</u>, 2. A.AlQahtani, M. Qahtani, M. Awaji, A. MOHAMMED, S. Zahrani, K. Ajlan, A. Aqool, R. Bamrdouf, S. Kebasi, histocompatibility and immunogenetics, king fahad specialist hospital Dammam, Dammam, SAUDI ARABIA|N. Quhaidan, Immunology / ser
Aim: One of the important role of HLA- laboratory is to test for HLA- sensitization (PRA)& assessing compatibility by performing crossmatch between patients and donors. A positive flow cytometry crossmatch (FCXM) is often a contraindication for transplantation however, many viral infections can trigger the formation of nonspecific antibodies that cause false positive crossmatch and make risk assessments difficult. < br /> We like other part in the world impacted by the pandemic of COVID-19. During the epidemic we have noticed frequent positive B-cells FcXM despite negative DSA , furthermore , we have noticed that some donor cells reacting even with negative control sera invalidating many tests (figure1). When we searched in some pairs, we found recent history of CVID-19 infection and or vaccination in either the donor or the patient Most of these positive reactions turned negative upon blocking lymphocytes with high protein sera . Therefor we decided to look for presence of HLA- antibodies in non-sensitized COVID-19 males , to see the impact SARS –CO-V2 positive sera on crossmatch and how to deal with such problem. We aim to share our experiences in dealing with COVID-19 samples tested for pretranspalnat workup. Methods: 40 male COVID-19 infected patients never transplanted or transfused who screened, positive for SARS-CoV-2 Ab (IgG/ IGM antibodies) where tested for HLA-Ab. HLA antibodies performed by LSM and SAB using One Lambda® on Luminex platform & analyzed using fusion software. A standard three-color FCXM was performed against 0.6 mg pronase treated separated cells & analysed on FACSCanto II. Blocking of nonspecific binding is done by incubation with 20% fetal sera (FBS). < br /> SARS coV2 antibody detected by LAISON SARS-CoV-2 S1/S2 IgG/I gM (DiaSorin) Results: The prevalence of HLA- antibodies was 75% in our tested population. The specificities included class I& II HLA- antibodies or both with some MFI reaching 10,000. FCXM of these sera with surrogate cells yielded variable results some positive results turned negative with adsorbing the sera or blocking the cells . Conclusion: Covid-19 had affected HLA- laboratories by creating false positive crossmatch results & emergence of HLA- antibodies in non-sensitized patients, accordingly each laboratory has to put this fact during interpretation of serological tests. Blocking nonspecific binding is an option to get rid of nonspecific binding but need to be validated in a large scale.
P72
COVID-19
Title: USE OF VIRTUAL CROSSMATCH IN SOLID ORGAN TRANSPLANT CANDIDATES AFTER COVID VACCINATION
Authors: <u > D. Wagenknecht</u>, S. Hale, D. Becker, V. Fudge, C. Martin, J. McKie, L. Wright, M. Lopez-Cepero, S. Resto-Ruiz, J. Lunz, Transplant Immunology Laboratory, LifeLink Foundation, Tampa, Florida, UNITED STATES|
Aim: Use of a virtual crossmatch (VXM) prior to organ transplantation can save time and resources. VXM eligible patients at our center have a tested sample collected within 30 days, no history of donor specific antibody (DSA) and no recent sensitizing event. With recent widespread COVID vaccination, we asked whether transplant candidates developed new anti-HLA antibodies after vaccination that might deny VXM eligibility. Methods: <br /> Thirty-nine patients received organ offers (33 kidney, 1 heart, 4 lung, 1 kidney/liver) after COVID vaccination. Fifteen patients were Caucasian, 12 Black, 7 Hispanic, 3 Asian and 2 “other”; 22 were male. HLA antibody analyses were performed by FlowPRA or LABScreen mixed HLA Class I & II antibody detection assays. Sera with a positive PRA were tested by LABScreen single antigen HLA Class I or II beads (SAB) to define specificity. Results: Prior to COVID vaccination, 24 patients were unsensitized; 13 patients had CI PRA (4% – 99%), 9 had CII PRA (5% – 99%), 7 had CI and CII PRA measured by FlowPRA. Three non-sensitized patients were transplanted before a post-vaccine PRA was tested and had physical crossmatches. Post-vaccination sera from 36 patients were tested for PRA changes. Twenty-one patients remained unsensitized. Generally, the FlowPRA for sensitized patients was unchanged by vaccination and no new specificities were detected by LABScreen SAB for the 6 patients tested post-vaccination. One patient’s PRA was 4/0 prior to vaccination, 12/0 12 days post- and 5/0 33 days post-vaccination. Another patient had 10/55 PRA pre- and 12/33 PRA post-vaccination. For both patients, the FlowPRA peak profiles were similar pre- and post-vaccination and the variations were likely due to subjectivity in FlowPRA analyses. Eight patients were not transplanted. Physical crossmatches were performed for 22 patients; 13 by protocol (9 living donors, 4 lung transplants), 4 had a history of DSA and 5 were < 28 days post-vaccine. Nine patients (29%) had a VXM prior to transplantation. Conclusion: Previously unsensitized patients did not develop new anti-HLA antibodies after COVID vaccination. In the sensitized patients, no new anti-HLA antibody specificities were found post-vaccination. Our data suggest that COVID vaccination may not significantly increase sensitization and VXM assessment eligibility. A larger patient cohort will further gauge the impact of COVID vaccination as a sensitizing event.
P73
COVID-19
Title: OPTIMIZATION AND DIVERSIFICATION OF THE LABSCREEN COVID PLUS ASSAY
Authors: E. Pringle, R. Duncan, Department of Microbiology and Immunology, Dalhousie University, Halifax, Nova Scotia, CANADA|<u > A. Greenshields</u>, R. Liwski, Department of Pathology, Dalhousie University, Halifax, Nova Scotia, CANADA|
Aim: Antibody responses to Spike are an important correlate of protection for SARS-CoV-2 vaccines. Less clear are the specific characteristics of antibody responses that mediate this protection. Recently, One Lambda released the novel high throughput LABScreen COVID Plus assay, which roughly maps IgG response to domains of the Spike protein of SARS2-CoV-2, nucleocapsid of SARS-COV-2, and the S1 domain of six other coronaviruses known to infect humans. This study aimed to decrease assay time by adapting the Rapid Optimized Single Antigen Bead (ROB) protocol, currently used for HLA specific IgG antibody identification, for the COVID Plus assay. To increase the diversity of antibodies interrogated by this assay, the ROB protocol was also adapted to detect IgM and IgA antibodies. In addition, we developed a C1q-binding assay using COVID Plus beads and the C1qScreen. Methods: Parallel testing of a panel of 20 convalescent sera from SARS-CoV-2 infected patients and 5 healthy controls was performed using the COVID Plus kit by the Standard Method (SM; 85 min assay time) and ROB-COVID Antibody method (ROB-CA; 25 min assay time). Methods were compared with respect to MFI. Adaptions to the SM included reduced primary and secondary antibody incubation times, reduced centrifugation time, and increased secondary antibody concentration. Results: Comparison between the SM and ROB-CA confirmed an excellent correlation in coronavirus specific antibody reactivity for all 25 sera (MFI r < sup > 2</sup> = 0.98), with no significant difference in antibody identification. Importantly, use of the ROB-CA method decreased assay time by 70%. IgA and IgM specific secondary antibodies allowed for the identification of varying levels of SARS-CoV-2 specific reactivity in convalescent sera. Limited IgM and IgA reactivity targeting other human corona virus S1 proteins was observed. C1q positivity was identified in convalescent sera, positivity was associated with an IgG MFI of over 20,000 for Spike or 10,000 for the SARS-CoV-2 nucleocapsid SABs. Conclusion: Here we optimized and diversified the potential application of the COVID Plus assay. Rapid assessment of functional antibody responses using minimal quantities of sera will be an important diagnostic tool as vaccination campaigns continue and next-generation vaccines are developed. We are currently using these tools to test a large panel of convalescent sera and correlating results with neutralizing antibody titers.
P74
Immunogenetics, Disease association
Title: IDENTIFICATION OF SEVEN PRIMARY FUNCTIONAL VARIANTS IN PRIMARY BILIARY CHOLANGITIS SUSCEPTIBILITY LOCI IN THE JAPANESE POPULATION
Authors: <u > Y. Hitomi</u>, M. Tsuiji, Hoshi University, Tokyo, JAPAN|Y. Aiba, M. Nakamura, National Hospital Organization (NHO) Nagasaki Medical Center, Omura, JAPAN|K. Ueno, Y. Kawai, N. Nishida, K. Tokunaga, National Center for Global Health and Medicine, Tokyo,
Aim: Primary biliary cirrhosis (PBC) is a chronic non-suppurative destructive cholangitis (CNSDC) that predominates in middle-aged and older women, and is considered an organ-specific autoimmune disease characterized by the destruction of intrahepatic small bile ducts. The higher monozygotic/dizygotic (MZ/DZ) ratio and the higher estimated relative sibling risk (λs) in PBC patients as compared to unaffected individuals indicates the involvement of strong genetic factors in the development of PBC. < br /> Our previous genome-wide association studies (GWASs) identified HLA class II and seven other loci as susceptibility loci for PBC in the Japanese population. However, candidate genes with well-known functions that are located in disease susceptibility loci are often selected as disease susceptibility genes in GWASs. The contribution of susceptibility loci to pathogenesis can be understood by the identification of effector genes regulated by primary functional variants. Methods: In order to analyze the mechanism conferring the disease susceptibility in these 8 susceptibility loci, we carried out high-density association mapping of disease susceptibility loci based on single nucleotide polymorphisms (SNPs) imputation using data from a whole-genome sequence reference panel of 2,049 Japanese individuals and our previous GWAS (case: n = 2,060; control: n = 1,985). <br /> Subsequent < i > in silico</i > and < i > in vitro</i > functional analyses were performed for the identification of primary functional variants in each locus. Results: <i > In silico</i > and < i > in vitro</i > functional analyses identified primary functional variants in each locus (<i > TNFSF15</i>, <i > PRKCB</i>, <i > NFKB1</i>, <i > MANBA</i>, <i > COLCA1</i>, <i > COLCA2</i>, chromosome 3q13.3, and chromosome 17q12-21) among candidate variants which showed stronger association with susceptibility to PBC (P < 1.0 × 10 < sup > -6</sup > ). < br /> These primary functional variants have been strongly associations with endogenous gene expression levels inside or outside the susceptibility loci by expression-quantitative trait loci (e-QTL) analyses. The effects of primary functional variants on gene expression levels were confirmed using genotype knock-in versions of cell lines constructed using the CRISPR/Cas9 system. Conclusion: Our series of studies have identified effector genes regulated by primary functional variant from GWAS data, and it is assumed that their effector genes contribute to the development of PBC.
P75
Immunogenetics, Autoimmunity
Title: DISSECTING THE RESTRICTION OF A DIABETOGENIC T CELL CLONE FROM AN ENTIRE GENOTYPE USING CLONED HLA MOLECULES
Authors: E.R. Sunderhaus, <u > B. Freed</u>, C.L. Roark, Medicine, Univ. of Colorado AMC, Aurora, Colorado, UNITED STATES|A.C. Hohenstein, R. Baker, Barbara Davis Center, Univ. of Colorado, Aurora, Colorado, UNITED STATES|
Aim: The risk of developing Type 1 Diabetes (T1D) is strongly correlated to the inheritance of the HLA-DR3 and -DR4 haplotypes. The disease is caused by the destruction of insulin-secreting β cells in the pancreas mediated by the activation of CD4 < sup>+</sup > inflammatory T cells stimulated by native antigens and modified epitopes such as Hybrid Insulin Peptides (HIPs). < br /> HLA restriction of CD4 < sup>+</sup > T cells isolated from patient samples is often determined using antibodies to HLA-DR or -DQ to interfere with peptide presentation. The specific allele involved in the etiology of the disease is then assumed to be a known risk allele identified in the genotype of the patient. In the age of personalized medicine, this general assumption may no longer be sufficient, such as while developing small molecules that can interfere with presentation of diabetogenic epitopes. < br /> Here we evaluated whether T2 cell lines expressing a single HLA class II molecule could be used to identify the specific allele responsible for the presentation of a Chromogranin A-derived HIP using peptide binding and CD25 stimulation assays. Methods: The HLA-DR and DQ alleles (<i > cis</i > and < i > trans</i > combinations) from a T1D patient were individually cloned into T2 lines. In parallel, a T cell clone (clone D11) reactive to HIP1 (a fusion peptide between the C-peptide of proinsulin and WE14 of Chromogranin A) was isolated from this patient. Peptide binding of biotinylated HIP1 was measured by flow cytometry. The T2 lines expressing individual HLA and the patient’s autologous EBV-transformed B cell line were then used to stimulate the D11 T cell clone to determine HLA restriction. Results: HLA-DRB1*11:03, DQA1*03:01/DQB1*03:01, and DQA1*03:01/DQB1*03:02 all bound the HIP1 peptide, with DRB1*11:03 binding the strongest (Figure A). Stimulation of the HIP1 restricted D11 T cell clone showed an increase in CD25 expression only when peptide was presented by DRB1*11:03 and not the HLA-DQ alleles confirming its restriction to DRB1*11:03. Also, stimulation of the D11 T cell clone by the DRB1*11:03-expressing T2 cell was comparable to using the patient’s EBV line (Figure B). Conclusion: HLA-Class II T2 lines can predict and confirm the restriction of a CD4 < sup>+</sup > T cell clone through peptide binding and stimulation assays. These cell lines are powerful tools that can be used in diverse ways to further study antigen presentation in the context of various autoimmune diseases.
P76
Immunogenetics, Disease association
Title: GEOMX DIGITAL SPATIAL PROFILING OF VESSELS FROM HUMAN CARDIAC ALLOGRAFT VASCULOPATHY REJECTED GRAFTS
Authors: <u > J. Nevarez-Mejia</u>, H.C. Pickering, R.A. Sosa, R.P. Lau, G.A. Fishbein, E. Reed, Pathology and Laboratory Medicine, UCLA, Los Angeles, California, UNITED STATES|
Aim: Cardiac allograft vasculopathy (CAV) limits long-term graft and patient survival. The production of donor specific antibodies (DSA) remains a major risk factor contributing to CAV. DSA elicit alloimmune responses against the graft vasculature leading to endothelial cell activation and leukocyte recruitment. Chronic inflammation results in thickening of arterial intima and vessel occlusion. The aim of this study is to identify gene and protein expression profiles indicative of vascular and immune cell activation within CAV + vessels using GeoMx digital spatial profiling (DSP). Methods: Cardiac allograft rejected tissues from CAV + DSA + patients (N = 3; 2 females, 1 male) were subjected to DSP analysis. Regions of interest (ROIs) were selected based on immunofluorescent staining to identify macrophages (CD68 + CD163 + ) localized within vessels (CD34 + ) as well as H&E to identify CAV lesions. In total, 33 vessel ROIs were selected. Vessels were scored based on the level of CAV progression/neointimal thickening: minimal +/-, mild 1+, moderate 2+, significant 3+, and very significant 4 + . All ROIs were subjected to whole genome sequencing and a 78-protein panel for immune-related proteins and signaling activation. Results: Protein expression results indicated that 41 proteins were upregulated across all vessel ROIs. These included immune-infiltrates such as monocytes/macrophages, T cells, B cells, and NK cells. Markers related to immune inflammation (HLA-DR, CTLA4, CD44, and STING), cell death (Caspase 9, BAD, and p53) and phosphorylated proteins in MAPK and PI3K/AKT signaling were also detected. Vessels with low neointimal scores (+/- and 1 + ) exhibited higher T cell and antigen presentation markers (OX40L, CD27, and 4-1BB), and PI3K/AKT signaling compared to vessels with high neointima (2+, 3+, and 4 + ). Top genes identified in all vessel ROIs included < i > ADAM15, CCN2, IGFBP7, ITGB1, </i > and < i > TMEM106C</i > . Vessels with low neointimal scores mainly increased < i > VWF</i > while high neointimal vessels increased < i > PDGFRA</i > . Conclusion: We utilized DSP to identify protein markers and gene expression profiles of DSA + CAV + vessels. Moreover, we identified that vessels with low neointima exhibit high inflammatory markers while high neointima vessels contain a pro-fibrotic phenotype. These findings will help guide the discovery for better biomarkers for CAV diagnosis and prognostication.
P77
Immunogenetics, Disease association
Title: RARE ALLELE HAPLOTYPES
Authors: <u > P. CANO</u>, M. O'Leary, A.J. Yousefi Cohen, J. Wendel, E. Cavero, G. Roman, K. Rozar, C. Campbell, C. Iozzino, MOFFITT CANCER CENTER, Wesley Chapel, Florida, UNITED STATES|
Aim: One of the most valuable methods to confirm allele assignment is linkage disequilibrium and haplotype analysis. With rare alleles these tools are not readily available. The purpose of this study is to provide such tools. If one finds a rare allele, one must take additional steps to confirm the allele assignment. Having knowledge of the genetic environment in which an allele appears provides the proper context for this confirmation process. Methods: An HLA allele is a variant of the DNA sequence, either coding or non-coding, in one of the HLA genes. A rare allele is an allele with a gene frequency of<0.001 in any population. < br /> Typically, we have only observed 1 or 2 cases with a given rare allele. This is not enough information to evaluate linkage disequilibrium, but taking into account their very low gene frequency, any coincidence with the same or similar haplotypes identified in other labs must be considered informative. We compared our haplotype analysis with the phenotype of IMGT cells for each of the rare alleles identified. < br /> The matching of haplotypes of the isolated rare cases in which these alleles are observed has enough weight to make reliable conjectures about which haplotypes are associated with rare alleles. < br /> We use Immucor Mia Fora NGS for HLA typing with OneLambda single-antigen-bead SSO for complementary typing in cases of rare alleles or difficult allele assignment. Results: The table of rare allele haplotypes is too large to be presented here and it is available at:<br /> https://jupiterproject.org/ASHI_2021/HLA_Haplotypes_Rare_Alleles.xlsx < br /> Haplotypes in this table may only include the strong linkage disequilibrium blocks B-C and DR-DQ, as there is often too much recombination with the other loci to have stable extended haplotypes. Conclusion: Access to data regarding the genetic context in which rare allele appears has proven one of the most valuable quality control tools in HLA typing. Nevertheless, it must be remembered that whenever a rare allele is identified, such identification must be confirmed by another method, or by further sequence analysis of the NGS results. < br /> A rare allele that appears in all the cases reported in the same haplotype must be considered to have a definitive punctual origin. In the absence of a common haplotype, the rare allele must be considered the result of isolated random mutations. This only happens in mutation hotspots, like in the case of repeated random C insertions in codons 183–185 of exon 4 of HLA-A.
P78
Immunogenetics, Disease association
Title: FORMAL AND INFORMAL METHODS TO GIVE AN ACCOUNT OF LINKAGE DISEQUILIBRIUM IN HLA
Authors: <u > P. CANO</u>, M. O'Leary, C. Iozzino, C. Campbell, K. Rozar, G. Roman, E. Cavero, J. Wendel, A.J. Yousefi Cohen, MOFFITT CANCER CENTER, Wesley Chapel, Florida, UNITED STATES|
Aim: In interpreting HLA typing results it is important to have access to the level of LD promiscuity to anticipate what deviation from the well-established haplotypes is allowed without initiating and extensive review. Methods: From the point of view of linkage disequilibrium there are two paradigmatic alleles: ‘promiscuous’ alleles that are present in many different haplotypes, and ‘anchor’ alleles that form the core of well-established haplotypes. Results: Examples of anchor alleles:<br /> <b><u > DQA1*05:01:01:01 / DQA1*05:01:01:02 haplotypes</u></b><br /> One of the most common haplotypes: [A*01:01:01:01 + B*08:01:01:01 + C*07:01:01:01 + DRB1*03:01:01:01 + DRB3*01:01:02:01 + DQA1*05:01:01:02/01 + DQB1*02:01:01:01] < br /> DRB1 is always DRB1*03:01:01:01 < br /> DQB1 is always DQB1*02:01:01:01 < br /> DRB345 is either DRB3*01:01:02:01 or DRB3*02:02:01:01 < br /> DRB3*01:01:02:01 can go with either DQA1*05:01:01:01 or DQA1*05:01:01:02 < br /> DRB3*02:02:01:01 only goes with DQA1*05:01:01:01 < br /> [B*07:02:01:01 + C*07:02:01:01] and [B*08:01:01:01 + C*07:01:01:01] both go with DRB3*01:01:02:01, but not with DRB3*02:02:01:01 < br /> DRB3*02:02:01:01 goes with other B-C blocks, including [B*18:01:01:01 + C*05:01:01:01], [B*18:01:01:06 + C*05:01:01:01], and [B*14:02:01:01 + C*08:02:01:01] < br /> <b><u > DRB4*01:03:01:02 N haplotypes</u></b><br /> Very stable DR-DQ block:<br /> [DRB1*07:01:01:01 + DRB4*01:03:01:02 N + DQA1*02:01:01:01 + DQB1*03:03:02:01] < br /> Strong association with [B*57:01:01:01 + C*06:02:01:01] < br /> <b><u > DRB1*15:01:01:01 haplotypes</u></b><br /> Very stable DR-DQ block:<br /> [DRB1*15:01:01:01 + DRB5*01:01:01:01 + DQA1*01:02:01:01 + DQB1*06:02:01:01] < br /> with some variation: DQA1*01:02:01:01 vs DQA1*01:02:01:10 < br /> Most common B-C block association is with [B*07:02:01:01 + C*07:02:01:01/03], but there is variation with other B-C blocks. < br /> <b><u > DQB1*05:01:01:03/01 haplotypes</u></b><br /> Very stable DR-DQ block:<br /> [DRB1*01:01:01:01 + DRB345:DEL + DQA1*01:01:01:01 + DQB1*05:01:01:03/01] < br /> DRB1 variation: DRB1*01:02:01:01, DRB1*01:03:01, DRB1*01:02:13, DRB1*10:01:01:01, etc. < br /> DQA1*01:02:01:04 seen with [DRB1*13:02:01:02 + DRB3*03:01:01:01] Conclusion: Although formal methods of giving an account of linkage disequilibrium have been developed mainly using 2x2 contingency tables, informal descriptions are also useful and both formal methods and informal descriptions should be used in conjunction. Such combined approach has proven very useful in education and training.
P79
Immunogenetics, Disease association
Title: HIGH-RESOLUTION LINKAGE DISEQUILIBRIUM OF HLA-B-C BLOCKS
Authors: <u > P. CANO</u>, M. O'Leary, J. Wendel, C. Iozzino, E. Cavero, G. Roman, K. Rozar, A.J. Yousefi Cohen, C. Campbell, MOFFITT CANCER CENTER, Wesley Chapel, Florida, UNITED STATES|
Aim: The purpose of this study is to update linkage disequilibrium data between HLA-B and HLA-C based on high-resolution NGS HLA typing. Methods: Linkage disequilibrium between each B-C pair was analysis by means of 2x2 contingency tables, from which several association measures were calculated, like delta, mutual information, chi-square, etc. In the data provided here only the chi-square is included. Data is only provided for B-C pairs with significant association. In the data set provided 2x2 contingency tables are included for each B-C pair, where ‘a’ is the number of cases where Bx and Cx are present, ‘b’ is the number of cases where Bx is present and Cx is not, ‘c’ is the number of cases where Cx is present and Bx is not, and ‘d’ is the number of cases where neither Bx nor Cx are present. ‘a = b’ is the number of cases with Bx and ‘a + c’ is the number of cases with Cx. (Bx and Cx are the particular alleles in each B-C pair.) < br /> Typing performed with Immucor Mia Fora NGS. Both coding and non-coding regions were analysed in detail to determine exact four-field allele assignment. Results: 9646 haplotypes were used in this study. Haplotype analysis was performed with every single case based on family segregation analysis and prior knowledge of linkage disequilibrium with multiple reiterations to correct previous haplotype definitions based on new typing data. This reiterative haplotype analysis serves not only to correct inappropriate previous haplotype assignments, but also review previous typing and make necessary corrections on historical records. < br /> Data set can be retrieved at:<br /> https://jupiterproject.org/ASHI_2021/LD-A_BC.xlsx Conclusion: This study shows that linkage disequilibrium reaches a much higher level of resolution when both the coding and non-coding regions are analysed. Many associations are only revealed by differences in non-coding regions. < br /> This higher resolution in linkage disequilibrium enhances the value of LD tables to question and confirm HLA typing results. < br /> One of the problems in HLA typing laboratories is that old and legacy data cannot be used as reference because of the poor quality resulting from obsolete typing methods. I our laboratory the HLA typing enterprise is organised as a constant reiterative process, where old data is constantly review in the light of new data available, resulting in a high-quality cumulative data set.
P80
Immunogenetics, Disease association
Title: HIGH-RESOLUTION LINKAGE DISEQUILIBRIUM (LD) OF HLA-DR-DQ BLOCKS
Authors: <u > P. CANO</u>, M. O'Leary, J. Wendel, C. Iozzino, E. Cavero, G. Roman, K. Rozar, A.J. Yousefi Cohen, C. Campbell, MOFFITT CANCER CENTER, Wesley Chapel, Florida, UNITED STATES|
Aim: The purpose of this study is to update LD data in the DR-DQ blocks based on high-resolution NGS HLA typing. Methods: LD is done between pairs of genes or gene blocks. It is possible to do association studies of a dimensionality higher than 2, but it complicates things too much. We limit ourselves to associations between pairs. The DR-DQ block can be evaluated in different ways: 1) DRB1 versus DRB345-DQA1-DQB1, 2) DRB1-DRB345 versus DQA1-DQB1, 3) DRB345 versus DRB1-DQA1-DQB1, 4) DQA1 versus DRB1-DRB345-DQB1, 5) DQB1 versus DRB1-DRB345-DQA1, 6) DRB1 versus DRB345, 7) DQA1 versus DQB1. As can be seen, linkage disequilibrium is best studied dynamically with constantly updated data. The same gene block can be evaluated from the perspective of each gene as required by the question at hand in the review of a particular genotype with specific challenges. We have implemented such a dynamic system with the possibility of ad hoc inquiries as an aid in the interpretation of typing results with significant detection and solution of typing problems. < br /> Typing performed with Immucor Mia Fora NGS. Both coding and non-coding regions were analysed in detail to determine exact four-field allele assignment. Results: Although the real value of LD studies are dynamically executed to address the challenges of a particular allele assignment, here we present static data with the most relevant associations of DRB1 versus DRB345-DQA1-DQB1 as an example of the granularity achieved by NGS typing with both the coding and non-coding regions are analysed. < br /> 9638 haplotypes were used in this study. This data set can be retrieved at:<br /> https://jupiterproject.org/ASHI_2021/LD-A_DRDQ.xlsx Conclusion: This study shows that low-granularity linkage disequilibrium reaches a much higher level of detail. Many associations are only revealed by differences in non-coding regions. < br /> Nevertheless, high-granularity low-detail studies are also very valuable, like in the association between DQA1 and DQB1, where only at a high-granularity level it becomes clear that DQA1*01:01 alleles are associated with DQB1*05 alleles, DQA1*01:02 with DQB1*06:02, DQA1*01:03 with DQB1*06:01, DQA1*02 with DQB1*02:02 and DQB1*03:03, DQA1*03 with DQB1*03:02, DQA1*04 with DQB1*04, DQA1*05 with DQB1*02:01, DQA1*06 with DQB1*03:01. < br /> Therefore, the proper study of linkage disequilibrium requires moving back and forth from low granularity to high granularity.
P81
Immunogenetics, Disease association
Title: HLA ALLELES CANNOT BE DEFINED BASED ON UNSTABLE DNA REGIONS
Authors: <u > P. CANO</u>, M. O'Leary, J. Wendel, C. Iozzino, E. Cavero, G. Roman, K. Rozar, A.J. Yousefi Cohen, C. Campbell, MOFFITT CANCER CENTER, Wesley Chapel, Florida, UNITED STATES|
Aim: ‘Variable number of tandem repeats’ (VNTRs) are unstable DNA regions with very high mutation rates. They are short nucleotides sequences repeated multiple times. HLA definitions based on VNTRs are difficult to analyse as current analytical methods are not optimised for these regions − nor should they be. VNTR mutations are not only clinically irrelevant, but they are also properties of individuals and not of populations, providing no information from the point of view of population genetics. < br /> The purpose of this study is to identify VNTR regions in the HLA genome and to distinguish those alleles based on these regions. Methods: VNTRs are easy to detect just by looking at intron sequence alignments of HLA alleles. Each VNTR region defines a VNTR classification in which each class is defined by the equivalence relation of having the same sequence outside the VNTR region. VNTR classes are identified by a class representative, an allele that stands for all the alleles in the class regardless of VNTR differences. Results: Examples of VNTRs are the following:<br /> - DRB1 - intron 2 - ∼5730 < br /> - DPB1 – intron 2 - ∼8870 < br /> - DQA1 - intron 1 - ∼3110 < br /> - DQA1 - intron 1 - ∼3360 < br /> - DQA1 - intron 3 - ∼4840 < br /> <br /> Examples of VNTR HLA-DQA1 classes:<br /> - DQA1*01:01:01:01, :02, :03, :05, :06, :08, :09 < br /> - DQA1*01:04:01:01, :02, :04 < br /> - DQA1*01:02:01:01, :03, :05 < br /> - DQA1*02:01:01:01, :02 < br /> - DQA1*04:01:02:01, :02, :03 < br /> - DQA1*05:05:01:01, :02, :04, :07, :09, :23, :25, :26 < br /> - DQA1*06:01:01:01, :02, :04 < br /> (Examples in other loci will be provided.) Conclusion: Taking into account VNTR-bases alleles in the allele-assignment process is misleading for two reasons because they neither provide clinically relevant information nor they reveal any genetic feature of any population. < br /> The identification of VNTR classes is a useful tool in allele assignment. Allele assignment in HLA typing should avoid identifying members within a VNTR class. Whenever any allele in a class is detected, the class representative should be used in allele assignment. < br /> In identifying new alleles, those that belong to the same VNTR class as previously identified alleles should be ignored. < br /> Alleles in VNTR HLA classes should be removed from the HLA allele catalogue, leaving only the class representative allele. Leaving them leads to recurrent errors in allele assignment.
P82
Immunogenetics, Non-classical HLA
Title: MATCHING ACROSS THE BLOCKS: HLA CLASS 1b and MICA/MICB GENOTYOPING REVEAL ADDITIONAL MHC MISMATCHES IN CONVENTIALLY HLA MATCHED HCT DONOR/RECIPIENT PAIRS
Authors: <u > D.C. Sayer</u>, CareDx, Fremantle, Western Australia, AUSTRALIA|M. Anderson, Versiti Diagnostic Laboratories, Milwaukee, Wisconsin, UNITED STATES|
Aim: HCT from HLA matched unrelated donors has been reported to result in worse outcomes than HCT from matched sibling donors suggesting that differences at other MHC loci are important. Recombination events at specific sites within the MHC create a “block” structure of linked genomic subregions. Conventionally typed HLA loci provide information for 3 of at least 7 MHC genomic blocks. < i > HLA-A</i > is in the alpha block, <i > HLA-B</i > and < i > -C</i > are in the beta block and < i > DRB1</i > and < i > -DQB1</i > are in the delta block. We performed HLA typing on unrelated HCT donor / patient pairs, including the HLA Class 1b loci < i > HLA-F</i>, <i > -G</i>, <i > -H</i>, -E and < i > MICA</i > and < i > MICB</i > to determine if these genes contribute additional genomic matching. HLA-F, -E and MICB are in unique MHC genomic blocks. HLA-G,-H are in the alpha block and MICA is in the beta block Methods: 157 unrelated donor/patient pairs were typed with AlloSeq™ Tx 17 kits and AlloSeq™ Assign® sequence analysis software. AlloSeq Tx 17 is a 17 locus typing assay that uses hybrid capture for target enrichment. 4 field resolution was obtained for for HLA class Ia and Ib and full coding resolution was obtained for MICA, MICB and HLA class II. Given other studies have shown the importance of matching at HLA non-coding sequence – any sequence difference between donors and patients was considered a mismatch Results: 56 of the 157 pairs were matched for < i > HLA-A, -B, -C,</i> <i > -DRB1, -DRB3/4/5, -DQA1 and -DQB1. </i > Of the 56 pairs matched at these conventional loci only 14 pairs (25%) were matched for < i > HLA-F, -G, -H, -E</i>, <i > MICA</i > and < i > MICB</i > . < i > HLA-E</i>, <i > -F</i > and < i > MICB</i > were the most frequently mismatched (26, 20, and 11 pairs respectively) with fewer mismatches at < i > HLA-G</i>, <i > -H</i > and < i > MICA</i> (9, 4, and 2 pairs respectively). Conclusion: Genotyping HCT pairs with the additional content in the AlloSeq Tx 17 assay reveals a surprisingly high degree of mismatching especially in genomic blocks that aren’t currently represented by conventional HLA typing. The fewer mismatches at HLA-G/H and MICA reflect linkage with HLA-A and HLA-B/C respectively but still indicate these genes add additional genomic block diversity. Additional studies are underway to determine the impact of these mismatches on transplant outcome. Such studies may lead to identification of the MHC genomic blocks that contain the additional sequences that contribute to adverse outcomes in unrelated HCT
P83
Immunogenetics, Non-classical HLA
Title: HLA-G, -H TYPING PROVIDES INSIGHTS INTO MHC ALPHA BLOCK DIVERSITY: IMPLICATIONS FOR UNRELATED HCT, MHC DISEASE ASSOCIATION AND MHC EVOLUTION.
Authors: <u > L. Olafson</u>, J. Edwards, C. Ellis, D.C. Sayer, CareDx Pty Ltd, Fremantle, Western Australia, AUSTRALIA|
Aim: Many studies suggest matching MHC sequences in addition to conventionally typed HLA are required for best outcomes in unrelated HCT. But these sequences remain unknown. Similarly, despite MHC disease associations being first described over 40 years ago the nature of these associations is not understood. One of the reasons for the lack of progress in both areas maybe that focus has been on the diversity of HLA itself rather than genetic diversity of MHC genomic blocks. In this study we have analysed HLA-G, -H and HLA-A to help understand the degree of diversity within the MHC alpha block. Methods: We analysed over 2000 samples typed with AlloSeq™ Tx 17 kits and AlloSeq™ Assign®. AlloSeq Tx 17 performs target enrichment by hybrid capture for the HLA Class 1a genes HLA-A, -B, -C, the Class 1b genes HLA-E, -F, -G, -H, MICA and MICB and the HLA class 2 genes HLA-DRB1, -DRB3/4/5, -DQA1, -DQB1, -DPA1 and -DPB1. AlloSeq Tx 17 enables typing of additional genomic blocks (containing HLA-F, -E, MICB) as well as providing additional markers for MHC blocks already typed (HLA-G, -H in the alpha block and MICA in the beta block). We used a combined observational approach and analysis in Arlequin v3.0 to confirm HLA-G, -H and -A haplotypes. Results: In general, HLA-G and -H formed haplotypes with HLA-A alleles within broad serological groups suggesting HLA-A has evolved from an ancestral sequence where HLA-G and HLA-H have remained remarkabley conserved. However, there were exceptions: 1) Some common HLA-A alleles (A*01:01:01:01, A*24:02:01:01, A*11:01:01:01) were “splt” according to HLA-G, -H suggesting that HLA-A alone is insufficient as a marker of alpha block diversity. Specifically, the alpha block of the disease risk haplotype 8.1 is unique compared with other haplotypes marked by A*01:01:01:01, and 2) HLA-A alleles of the same serological lineage, eg HLA-A*02 could be grouped according to different G-H haplotypes. This may have implications for selecting HLA mismatched donors Conclusion: HLA-G and -H ,in additional to HLA-A, contribute to alpha block diversity. Further studies are required to understand implications for HCT. HLA-G, -H typing in disease associations studies should be performed to assist in identifying the disease risk sequences on the 8.1 ancestral haplotype.
P84
Immunogenetics, Disease association
Title: HLA GENOTYPES AND IMMUNE RESPONSE TO COVID- 19
Authors: <u > R. Al attas</u>, M. ALZAHRANI, D. abduladherem, S. Zahrani, A. MOHAMMED, S. Kebasi, M. Awaji, H. harbi, S. Ghandorah, histocompatibility and immunogenetics, king fahad specialist hospital Dammam, Dammam, SAUDI ARABIA|<u > R. Al attas</u>, Saudi society o
Aim: Human leukocyte antigen (HLA) molecules play an important role in the development of a specific immune response to viral infections by presenting viral antigens at the cell surface to T- lymphocytes & therefore affect both the susceptibility and the severity of viral infections. Among common infections where severity had been associated with certain HLA antigens are malaria, HIV, and viral hepatitis. Information on the interconnection of HLA genotypes and severity of the course of COVID-19 caused by SARS-CoV- 2 is sparse.We searched for possible connections between HLA- genotypes in a cohort of COVID-19 infected patients with varialble disease spectra . Methods: <br /> 134 adult patients with PCR confirmed SARS-CoV- 2 infection were HLA- typed. The recruited patients had been divided into 4 groups based on symptoms :(asymptomatic (23) mildly symptomatic (52), moderately (32) to severely symptomatic requiring ICU admission (32)). patients had no reported obvoius comorbidity & were a subgroup from a cohort of an approved study on antibody responses to SARS-Cov-2 infection from centers in Eastern Saudi Arabia < br /> <b > Statistical Analysis: </b > Allele frequencies were estimated by dividing the number of occurrences of a given allele by doubled total number of individuals (i.e. homozygous individuals were counted as two occurrences). Fisher analysis of variance (ANOVA) is used to determine statistical significance of association between any certain allele & the disease spectrum in the 4 categories. Results: All alleles in 6 loci (A, B.C, DR, DQ, DP) found in each categories were recorded & any allele occurrence of more than one was calculated & distributed among the four groups in a bar graph illustrated in Figure 1–5 .Other alleles that occurred only once were all among the CWD alleles , were excluded from the graph for simplicity. Statistical tool found no significant association of any reported allele with certain disease spectrum. Conclusion: Despite that our preliminary work did not find significant correlation between HLA genotypes & severity of COVID-19, there is still scope for further study into the role of HLA in COVID-19 in a larger poulation , as well as other non HLA- genes as determinants of immune response.
Fig. 1: Distribution of HLA-A* alleles found in the four groups of COVID-19 patients
Fig. 2. Distribution of HLA- B* alleles in the 4 groups of COVID-19 patients
Fig. 3. Distribution of HLA- C* alleles in the 4 groups of COVID-19 patients.
Fig. 4. Distribution of HLA- DRB1* alleles in the 4 groups of COVID-19 patients
Fig. 5. Distribution of HLA- DQB1* alleles in the 4 groups of COVID-19 patients
P85
Immunogenetics, Non-classical HLA
Title: HIGH-RESOLUTION ANALYSIS OF 35 HLA LOCI IN UNIQUE SOUTH AMERICAN POPULATIONS IDENTIFIES POSSIBLE SIGNATURES OF SELECTION IN HLA-G
Authors: B. Ho, R. Francisco, J. Hollenbach, D.G. Augusto, Neurology, University of California, San Francisco, San Francisco, California, UNITED STATES|H.C. Issler, <u > L. B. Vargas</u>, M. Petzl-Erler, D.G. Augusto, Genética, Universidade Federal do Paraná, Curiti
Aim: Although substantial previous work has characterized < i > HLA</i > diversity in global populations, most studies focused on classical loci only, with very limited population studies for non-classical genes and pseudogenes. Here, we performed high-resolution characterization of all < i > HLA</i > loci in ancestrally diverse South American populations, including: five Amerindian populations from Brazil, one Amerindian population from Paraguay, and one predominantly European admixed population from Curitiba, Brazil. Methods: We applied our custom capture-based next-generation sequencing method in 601 individuals from seven populations. Sequencing of 35 < i > HLA</i > loci was performed on the Illumina platform and raw reads were analyzed with the software HLA Explore®. Results: We observed 375 < i > HLA</i > variants at third-field resolution, of which 50 were found in the pseudogenes. < i > HLA-H</i > was the pseudogene exhibiting the largest number of variants (13). Overall, we found 45 < i > HLA</i > alleles in Amerindians that were not observed in the Euro-descendant population. Some of these variants are possibly exclusive to Amerindian populations, e.g. < i > HLA-B*35:11:01</i>, observed only in the Guarani populations (0.04 > <i > f </i≫ 0.10). We observed a remarkably high frequency of < i > HLA-G*01:03</i > in Kaingang (0.68 to 0.86) and < i > HLA-G</i > was the gene that showed most differentiation among the study populations (F < sub > ST </sub>= 0.29). Besides, we observed a significantly increased rate of synonymous compared to non-synonymous substitutions in < i > HLA-G</i> (dN-dS < 0; 0.007 > <i > p </i≫ 0.02) in multiple populations, which may indicate purifying selection shaping the diversity of this gene in Amerindians. Finally, we integrated our results with previously generated allelic data for < i > KIR2DL4</i > in the same individuals. We identified that the frequencies of HLA-G and KIR2DL4 pairs are not randomly distributed in Kaingang (<i > p </i>= 2x10 < sup > -4</sup > ) and Guarani (<i > p </i>= 3x10 < sup > -6</sup > ), indicating that selection might be favoring specific receptor-ligand combinations. Conclusion: Our unprecedented high-resolution characterization of 35 < i > HLA</i > loci in unique human populations identified signatures that could indicate purifying selection on < i > HLA-G</i>, which is known to be implicated in human reproduction. The study < i> </i > of these medically important loci in ancestrally diverse populations provides the basis to understand the evolutionary mechanisms shaping the < i > HLA</i > diversity and disease association.
P86
Laboratory improvement, Cellular assays
Title: LOGICLE SCALING FOR FLOW CYTOMETRIC CROSSMATCHING INCREASES EVENTS OF SCARCE B CELLS
Authors: <u > N. Brown</u>, B. Ngo, T. Bui, Pathology & Laboratory Medicine, Penn Medicine, Philadelphia, Pennsylvania, UNITED STATES|
Aim: Multi-color flow cytometry often uses fluorochromes with overlapping emission spectra, requiring compensation to ensure fluorochrome signals do not interfere with each other. Compensation involves subtracting a signal from one channel based on the value of another channel, which, due to data scatter, can result in negative channel values. Traditionally, flow cytometry data is plotted on a logarithmic (log) scale, which cannot plot negative values. Modern flow cytometers use digital signal processing which can employ a biexponential scale, known as logicle, capable of plotting both linear and logarithmic values. We reasoned that cell events in flow cytometric crossmatch (FCXM) would be hidden using traditional plotting, while these cells could be analyzed using logicle scaling. We tested this on B cells, which, due to their scarcity in peripheral blood, are often the limiting factor for FCXM acquisition. Methods: Three-color FCXM using anti-CD3-PerCP, CD19-PE, and human IgG-FITC was acquired by a CytoFLEX Flow Cytometer (Beckman Coulter). Gating for CD3 < sup>+</sup > T cells and CD19 < sup>+</sup > B cells used rectangular gates on a traditional 4.5 log scale (Fig. 1A). After analysis, scales were converted to logicle (Fig. 1B), and gates were expanded to include all T and B cells (Fig. 1C). Peripheral blood samples with low B cell counts were analyzed. Results: Seven crossmatches, representing a total of 40 control or test samples, were analyzed. All samples had low B cells upon initial run, defined as cells where the sample was exhausted before 2,500B cell events were acquired. 24 samples had B cell events below 500, the cutoff for a valid run. In all cases, re-gating B cells on logical scales increased the B cell events, with a minimum of 46, maximum of 679, and a mean increase of 33% (Fig. 2A). In 8 of the samples, the B cell events increased to above 500, resulting in one previously invalid crossmatch becoming valid with logicle gating. Importantly, the B cell crossmatch ratio, the ultimate result of the assay, was not significantly affected by the inclusion of the extra cells (Fig. 2B). Conclusion: A full third of precious B cells are acquired during FCXM, but cannot be analyzed due to plotting compensated flow cytometry data on log scales. We show here that modern flow cytometers can overcome this limitation using the logicle scale, resulting in reduced invalid crossmatches due to insufficient B cell events.
P87
Laboratory improvement, QA/QC
Title: HOW EFFECTIVE IS HIGH CONCENTRATION DITHIOTHREITOL SERUM TREATMENT IN UNCOVERING PROZONE?
Authors: <u > K. Gvozdjan</u>, J. Patel, Z. Du, Pathology and Laboratory Medicine, Loyola University Medical Center, Maywood, Illinois, UNITED STATES|
Aim: Laboratories use different serum treatment reagents, including dithiothreitol (DTT) and/or ethylenediaminetetraacetic acid (EDTA), and reagent concentrations prior to HLA antibody testing in order to avoid false negative results due to prozone effect that may be IgM and/or C1 mediated. EDTA has been reported as more effective in removing prozone than DTT. The purpose of our study was to determine if the current method of 10 µM DTT treatment is effective compared to the commonly employed EDTA (5 mM) method in removing prozone for HLA antibody testing. Methods: 13 patient sera with confirmed prozone effect (MFI increase ≥ 3000) on LabScreen single antigen bead (SAB) testing for HLA Class I and II antibodies after treatment with 10 µM DTT and/or 5 mM EDTA were included in the study. HLA antibody identification was performed using SAB kits (ThermoFisher One Lambda, Inc., West Hills, CA), and the Luminex system (Austin, TX). Results: There was a strong correlation between Class I (n = 1261) and II (n = 1235) SAB MFI values from DTT and EDTA-treated samples (Pearson correlation coefficient (r) = 0.976 and 0.994, respectively). DTT treatment uncovered prozone at greater proportion of Class I loci (284/1261, 22.5%) compared to EDTA (263/1261, 20.9%). EDTA treatment uncovered prozone at greater proportion of Class II loci (188/1235, 15.2%) compared to DTT (176/1235, 14.3%). Discordant results (prozone criteria met after either DTT or EDTA treatment) involved a total of 47 Class I and 15 Class II allele specificities in 11/13 and 5/13 cases, respectively. Only DTT treatment in 1/13 cases uncovered prozone at multiple HLA-A, B, and C allele specificities to the degree that resulted in clinically significant change in antibody strength classification compared to EDTA. Conclusion: 10 µM DTT treatment method is overall comparable to 5 mM EDTA in effectiveness at removing prozone for clinical purposes. In selected cases, when predominantly IgM-mediated prozone effect is suspected yet not evident after EDTA treatment, repeat testing after DTT treatment is suggested.
P88
Laboratory improvement, QA/QC
Title: MINIMIZING INTER-TECHNOLOGIST VARIABILITY FOR CLASS I SINGLE ANTIGEN ANTIBODY TESTING
Authors: <u > H.C. Sullivan</u>, R. Toddings, M. Muchhala, H.M. Gebel, R.A. Bray, Pathology and Laboratory Medicine, Emory Univesity, Atlanta, Georgia, UNITED STATES|
Aim: Given the clinical relevance of SAB testing in determining unacceptable antigens in the pre-transplant setting and identifying donor specific antibody in the post-transplant setting, reproducibility among technologists is important. As part of annual internal quality control (iQC), inter-technologist variability was evaluated by comparing mean fluorescence intensity (MFI) values of SAB testing. Herein, we set out to identify and address the areas of variability in the SAB testing protocol to reduce inter-technologist variability. Methods: Fifteen technologists participated in Class I iQC assessment (LABScreen, One Lambda) using the same test serum. The percent coefficient of variation (%CV) was calculated using the MFI means and standard deviations of each bead among the technologists’ runs. The assay protocol and workflow were reviewed with the technologists to identify variability. The protocol was updated and reviewed once more prior to repeat Class I iQC testing. The repeat %CV values were then compared to the original run. iQC was repeated one year after protocol review and revision. Results: The mean MFI %CV prior to protocol review and revision was 19.4% (range: 7.6 – 69.8%). Protocol review revealed several areas of variability among the technologist (Table). Following protocol review and revision, the mean MFI %CV among the technologists was 9.0% (range: 4.3 – 25.8%), a significant improvement from the original iQC (P = 0.0001); and all %CVs were below 30%. Highest variability pre- and post-intervention was among beads with lower MFI values. One year later, the mean MFI %CV crept up to 14.6% (range: 4.5 – 49.8%), significantly higher than the repeat testing immediately after the intervention (P = 0.002). Conclusion: Repeat iQC Class I SAB testing demonstrated significant improvement, reducing %CV to below 30% for all beads and aligning with published standardization targets. The intervention of review and clarification of the testing protocol with the technologists was simple, inexpensive, and effective. iQC testing will continue to be performed annually, a practice that is supported by the %CV increase in testing a year after the intervention. Maintaining test reproducibility is essential to provide consistency in results for the care of our transplant candidates and recipients.
P89
Laboratory improvement, New assays
Title: QUANTITATIVE IMPACT OF MFI-LIMITER SOFTWARE REMOVAL ON SAB OUTPUTS FROM LUMINEX 3D INSTRUMENTS
Authors: <u > P. Kluz</u>, J. Burger, S.W. Stanley, L.G. Hidalgo, HLA laboratory, U. Wisc. - Madison, Madison, Wisconsin, UNITED STATES|L.G. Hidalgo, Surgery - Transplant Division, U. Wisc. - Madison, Madison, Wisconsin, UNITED STATES|
Aim: <br /> HLA Fusion < sup > TM</sup > single antigen bead (SAB) analysis software decreases MFI data obtained on Luminex 3D instruments using software calculations aimed to retain historic MFI trends obtained using previous generation Luminex instruments. This neutralizes the enhanced sensitivity properties of the Luminex 3D instrument. We sought to quantify the effects of removing the software MFI limiter on SAB testing. Methods: Pre- and post-transplant patient samples were pre-treated with EDTA, tested by LABScreen < sup > TM</sup > Class I and Class II SABs and read on the Luminex 3D instrument. Samples were acquired with the MFI limiter software ON then spun down, washed, and reacquired with the MFI limiter software OFF. MFI values were compared between acquisitions with MFI limiter ON vs. with MFI limiter OFF and examined the data across groups divided into MFI ranges in increments 5 K MFI with a negative threshold of 500. Data was analyzed and graphed using RStudio. Results: <br /> Patient samples positive for HLA Class I (N = 530) and Class II antibodies (N = 531) were acquired and compared. As expected, removal of the MFI limiter led to overall increases in MFI values but these were not consistent across all MFI ranges. Class I MFI values with MFI limiter OFF increased 26 % at 500 – 5 K MFI; 21 % at 5 K – 10 K MFI; and then gradually increased between 42 and 48% between 10 K and 30 K MFI (Figure 1). Overall Class II MFI values increased with MFI limiter OFF in a similar pattern to Class I values (Figure 2). Importantly, the patterns of bead reactivity were not affected with MFI limiter OFF setting with correlations between ON vs. OFF: Class I – R = 0.998, Class II – R = 0.999. Conclusion: <br /> Our study defines the level of MFI increases observed when SABs are acquired on a Luminex 3D instrument without manipulation from the Fusion < sup > TM</sup > software MFI limiter. The data was acquired using data from a large number of sensitized patient samples assessed in our clinical laboratory that serves a large, multi-organ transplant center and therefore has direct clinical application. Our data provides the groundwork to help laboratories assess whether to access the increased sensitivity potential of the Luminex 3D instrument and what to expect with regards to relation to historic MFI data.
P90
Laboratory improvement, Evaluations
Title: OPTIMIZING TEST UTILIZATION IN HIGHLY SENSITIZED PATIENTS
Authors: <u > H.C. Sullivan</u>, A. Taylor, F. Hennig, H.M. Gebel, R.A. Bray, Pathology and Laboratory Medicine, Emory Univesity, Atlanta, Georgia, UNITED STATES|
Aim: Monitoring antibody profile changes in highly sensitized renal transplant candidates is important as fluctuations in CPRA could affect access. Proper test utilization can be difficult to optimize: Underutilization could miss changes in antibody status while overutilization can impact laboratory resources. Our HLA testing algorithm for patients with > 98% CPRA includes monthly FlowPRA and quarterly single antigen bead (SAB) testing. The aim herein was to assess the validity of the current algorithm in attempt to optimize testing intervals. < br /> . Methods: HLA antibody testing performed in 2020 on renal transplant candidates with CPRA > 98% was retrospectively reviewed. Results from the first of the year were compared to results from the end of the year to examine changes in SAB testing (LABScreen, One Lambda) and FlowPRA (One Lambda). Unacceptable antigens were used to compute CPRA to evaluate for changes in fine CPRA groups (CPRA > 2 decimal places). HLA class I and II FlowPRA percentages and histograms were also compared. Results: Results from 44 highly sensitized candidates were evaluated. The first test of the year identified an average of 3 unique antibody specificities that were not reported on the last test of the year; whereas, the last test reported on average 1 unique specificity that was not present on the first test. Unique specificities had an average of 5210 MFI. CPRA did not vary significantly between the two time points (Table). In 29/44(65%) cases, the variances did not change the patient’s fine CPRA group, and of the remaining cases, the majority (11/15) only changed one fine CPRA group. FlowPRA also did not differ significantly between the time points (Table). Most cases for class I (42/44) and II FlowPRA (40/44) had no change or varied by < u>≪/u > 10%. In terms of histogram architecture, 27 (61%) had no change; and only 5 cases had a notable change suspicious for an underlying difference in alloimmune status. Conclusion: Data presented indicate that the current algorithm for patients with CPRA > 98% denotes test overutilization. The antibody profiles between the beginning and end of the year were not significantly different and would appear to have little impact on offer rates. Most unique specificities were at MFI values close to positive thresholds. Thus, modifying the current algorithm by decreasing the testing frequency, the HLA laboratory could save time, reduce costs, and conserve resources without compromising patient care.
P91
Laboratory improvement, Cellular assays
Title: 2ND B CELL POPULATION IN FLOW CROSSMATCH IS CAUSED BY IGG B CELL RECEPTOR
Authors: <u > F. Li</u>, X. Zhang, comprehensive transplant center-HLA Lab, Cedars Sinai medical center, Los Angeles, California, UNITED STATES|
Aim: The median channel shift in the flow crossmatch (XM) assay is used to evaluate the compatibility between the donor and recipient. The median channel shift is calculated as fluorescence channel numbers of the patient sample minus the channel numbers of the negative control. However, it is not uncommon, especially for cells from spleen of deceased donors, that the negative control displays two populations (double peaks), which increases the background and lowers the test sensitivity. B cells express B Cell Receptors (BCR), the membrane bound forms of IgG, IgM, IgA, IgE, or IgD, on the cell surface. We hypothesize that the 2 < sup > nd</sup > B cell population observed in flow crossmatches is caused by binding of the IgG BCR by FITC-anti-human IgG antibody used in the flow crossmatch. Methods: Lymphocytes were first stained with PE-mouse anti-human IgG F(ab). Then cells were incubated with patient sera or controls, and stained with PerCP-CD3, APC-CD19, FITC- F(ab’) < sub > 2 </sub > -rabbit -anti-human IgG. To determine if IgG BCR positive cells are memory cells, cells were also stained with memory B cell marker CD27. Results: In the negative controls, IgG BCR negative B cells overlapped with the first population in B cell flow XM, while IgG BCR positive B cells corresponded to the 2 < sup > nd</sup > population (Fig 1). Furthermore, we found that CD27 negative cells presented in the first peak, while CD27 + cells existed in both populations. Conclusion: The double peaks are caused by binding of FITC-secondary antibody to IgG BCR on B cells. Gating on IgG-BCR negative cells can remove the double peaks phenomenon in B cell flow XM.
Fig. 1. The 2nd B cell population presented in untreated- and AB-serum -treated PBMC is CD19+ B cells carried with IgG-BCR. A. the gating of B and T cells. B. two B cell peaks present in untreated cells when stained with F(ab’)2 anti-human IgG second antibody (Ab). C. Gating of IgG-BCR- and IgG-BCR+ B cells by pre-stained cells with PE-anti-human-IgG-F(ab). D. the 2nd peak in B cell flow XM is contributed by B cells carrying IgG-BCR. IgG-BCR- B cells overlap with the 1st peak in B cell flow XM, while IgG-BCR+ B cells harmonize with the 2nd peak. Histograms of FITC- F(ab’)2 anti-human IgG for IgG-BCR- and IgG-BCR+ B cells are shown on the top 2 graphs. Overlay of these four gated B cell populations is shown on the bottom. These graphs are representative data of 4 individual patient samples.
P92
Laboratory improvement, Cellular assays
Title: IMPROVEMENT OF T AND B CELL CDC CROSSMATCH ASSAY BY USING FLOW CYTOMETRY
Authors: <u > F. Li</u>, X. Zhang, comprehensive transplant center-HLA Lab, Cedars Sinai medical center, Los Angeles, California, UNITED STATES|
Aim: Complement dependent cytotoxicity (CDC) crossmatch (XM) is a classical assay to evaluate donor/recipient compatibility. In our center, we use CDC XM as tie breaker to transplant highly sensitized patients. However, reading the classical CDC XM is very subjective. The aim of this study is to use flow cytometry to quantify cell death and reduce the background caused by unhealthy cells. Methods: Lymphocytes were enriched by EasySep and pre-stained with To-Pro3 to gate out dead cells. Cells were then incubated with patient sera, and complements. Next, cells were stained with CD3-PerCP for T cells, CD19-PE for B cells, and Live/Dead < sup > TM</sup > green for dead cells. T and B cell death were evaluated by the percentage of dead cells in the parental population. 20% of cell death was used as the cutoff for positivity. Results: Treatment of lymphocytes with positive controls CD3 antibody and pan HLA-DR antibody leaded to a positive flow-based CDC-XM for T cells and B cells, respectively. In addition, flow-based CDC XM of lymphocyte specific IgM incubation were positive for both T and B cells, and DTT pretreatment rendered them negative. Using serial dilution of broad-reactive serum sera, flow-based CDC XM produced a concentration-dependent effect In T and B cells, indicating flow-based CDC XM provide a quantitative measurement for CDC XM. We further performed 14 flow-based CDC crossmatches on proficient test samples from CAP2017 and 2018. The flow-based CDC XM results were consistent with the consensus reported by CAP (Table 1). Conclusion: Pre-staining cells with To-Pro3 to exclude dead cells before crossmatching reduces the background and improves test sensitivity. Evaluation of cell death by the flow-based XM provides easier and objective interpretation of results.
Table 1. Comparison of T cell XM results between flow-based CDC XM and T cell crossmatching survey for CAP 2017 and 2018 samples.
P93
Laboratory improvement, New assays
Title: ISOLATION OF CD3 + T CELLS AND CD19 + B CELLS FOR SEROLOGY-BASED CYTOTOXICITY ASSAYS
Authors: <u > K. McQueen</u>, C. Barr, R. Dos Remedios, V. Posarac, A. Kokaji, A.C. Eaves, S.A. Louis, Research and Development, STEMCELL Technologies, Vancouver, British Columbia, CANADA|
Aim: Serology-based complement dependent cytotoxicity (CDC) assays are used to test for compatibility of donor and recipient for solid organ transplantation as they are quick, reproducible, and cost effective. Routine diagnostic laboratories must test for HLA class I and HLA class II compatibility, so isolation of the appropriate cells is critical. We have developed kits to positively select viable CD3 + T and CD19 + B cells directly from patient peripheral blood for use in CDC assays. The isolation procedure can also be automated on RoboSep™-S. Methods: Each kit was used to isolate either T or B cells from peripheral blood from healthy donors 24 to 48 h post-draw. Manual or automated cell isolation was carried out directly from unprocessed blood. Desired cells are labelled with an antibody cocktail and magnetic particles. Samples are then placed in a magnet, where desired cells are held in the tube and unwanted cells are simply poured off. Magnetic particles remain bound to the cell surface and can be used to settle cells when placed on a plate magnet as part of the CDC assay. Purity and viability of CD3 + T cells or CD19 + B cells was determined by flow cytometry. Results: After manual EasySep™ isolation, the purities of CD3 + and CD19 + cells were 99.1 ± 0.5% (mean ± SD; n = 10) and 93.8 ± 4.2% (n = 33), respectively. Viability of CD3 + cells was 99.9 ± 0.1%; viability of CD19 + cells was 98.9 ± 2.2%. 1.29 ± 1.18 × 10e6 CD3 + cells and 1.74 ± 1.0 × 10e5 CD19 + cells were obtained per mL of blood. After isolation using the automated procedure on RoboSep™-S, the purity of CD3 + cells was 97.0 ± 4.3% (n = 5); the purity of CD19 + cells was 94.0 ± 2.8% (n = 9). Viability of CD3 + and CD19 + cells was 99.7 ± 0.5% and 99.4 ± 1.5%, respectively. 5.81 ± 3.31 × 10e5 CD3 + cells and 9.7 ± 6.2 × 10e4 CD19 + cells were obtained per mL of blood. Conclusion: The EasySep™ Serology Whole Blood CD3 and CD19 Positive Selection Kits isolate highly viable T and B cells with high purity and yield that are compatible for use in CDC assays. The positive selection protocols can be automated on RoboSep™-S for standardized isolation procedures with minimal technologist hands-on time.
P94
Laboratory improvement, Cellular assays
Title: ANGIOTENSIN II RECEPTOR TYPE 1 NOT DETECTED ON HUMAN LYMPHOCYTES
Authors: <u > M. Li</u>, F. McDougan, P. Kimball, Surgery, VCU, Richmond, Virginia, UNITED STATES|
Aim: Angiotensin II receptor type 1 (AT1R) is a class of G protein-coupled membrane receptor with angiotensin II as its ligand. AT1R is expressed in kidney, adrenal glands, heart, brain and blood vessels. Kidney transplant recipients who have AT1R antibodies are at higher risk for allograft rejection and poor long-term graft outcome. Lymphocytes from healthy living donors or deceased donors are often used for flow cytometric crossmatch tests before organ transplants to decide whether the donors are acceptable for the recipients. Can AT1R antibodies cause positive flow cytometric crossmatch with lymphocytes? In the current study, we examined whether human lymphocytes express AT1R. Methods: Lymphocytes from peripheral blood of healthy living donors and spleen of deceased donors were negatively selected with a lymphocyte separation kit (Stem Cell Technologies). Lymphocytes were stained with PE-conjugated anti-CD3, APC-conjugated anti-CD19 and FITC-conjugated monoclonal mouse anti-human AT1R antibodies (R&D Systems) or isotype antibodies for 30 min at 4 °C. Lymphocytes from some donors were also stained with polyclonal rabbit anti-human AT1R antibodies (Invitrogen) or isotype antibodies, and FITC-conjugated goat anti-rabbit IgG. The stained cells were collected with BD FACSLyric Flow Cytometer. The percentage of positive cells was determined by subtracting the result of isotype antibody staining from the result of AT1R antibody staining. ≥ 1% was set for positive cut-off value. Results: The purity of enriched lymphocytes was about 95% (Mean = 95%, SD = 2%). The percentages of AT1R-positive T cells and B cells from peripheral blood of healthy living donors were 0.2% (Mean = 0.2%, SD = 0.2%) and 0.4% (Mean = 0.4%, SD = 0.4%), respectively. The percentages of AT1R-positive T cells and B cells from spleen of deceased donors were 0.3% (Mean = 0.3%, SD = 0.4%) and 0.2% (Mean = 0.2%, SD = 0.3%), respectively. Conclusion: AT1R protein is not expressed on cell surfaces of human lymphocytes from peripheral blood of healthy living persons and spleen of deceased donors.
P95
Laboratory improvement, Evaluations
Title: COMPARISON OF DIFFERENT VENDOR’S SINGLE ANTIGEN BEAD ASSAYS: DETECTION OF DONOR SPECIFIC ANTIBODY AND FLOW CYTOMETRY CROSS MATCH RESULTS
Authors: <u > A.J. Norin</u>, B. Das, Medicine, SUNY Downstate Health Sciences University, Brooklyn, New York, UNITED STATES|M. Ravindranath, Children's Hospital, Los Angeles, California, UNITED STATES|E. Filippone, Medicine, Sidney Kimmel Medical College at Thomas
Aim: Cell surface native class I HLA consists of trimers, i.e. polymorphic alpha-chain, beta 2 microglobulin, and associated peptide, termed Closed Conformers (CC). Before assembly in the ER, HLA Class 1 alpha-chains exist as monomers, referred to as Open Conformers (OC) or denatured HLA (dHLA). Using highly specific Mab in previous publications, we and others have shown that LabScreen (LS) Single Antigen Beads (SABs) contain a mixture of trimeric and OC HLA, whereas Lifecodes (LC) SABs have bound native CC-HLA only. We hypothesized that antibody against HLA-OCs on SABs detected by the LS assay but not detected by LC SABs would only minimally react with HLA-s since the latter molecules would be in a native CC configuration. Alternatively, antibody directed against HLA-CCs detected in the LC-SAB assay would react with native HLA on the cell surface.To test the above hypotheses we performed FCXM assays on representative sera that reacted strongly in the LS-SAB but weakly in LC-SAB assays. Also tested by FCXM were representative sera that reacted strongly in LC-SAB assays but weakly in LS-SAB assays. Methods: SAB assays were performed according to the manufacturer’s instructions. FCXMs were performed on a Cytoflex flow cytometer) as previously described with a 3SD positive cutoff over the negative control serum. Results: A monoclonal antibody that reacts with OC on SABs was positive for LS beads but non – reactive with LC beads indicating the presence of OC HLA on the current lot of LS SABs. Representative sera that reacted strongly in LS-SAB assays (>13,000 MFI) but weakly with LC-SABs (<1,000 MFI) gave negative T and B cell FCXMs. In contrast, representative sera that reacted strongly with LC-SABs (>13,000 MFI) but weakly with LS-SABs (<2100 MFI) exhibited positive T and B cell FCXMs. Conclusion: HLA antibodies directed solely against OCs on SAB assays, as determined by correlation with negative FCXMs, may lead to inappropriate listing of unacceptable antigens in UNet, inappropriate pre or post - desensitization procedures and/or decision not to XM. One approach to address this issue, other than changing manufacturing procedures to avoid beads with OCs, is to determine if the antibody reacts with known native HLA epitopes (www.epregistry.com.br) and is therefore of clinical consequence.
P96
Laboratory improvement, Evaluations
Title: PARADIGM SHIFT IN DECEASED DONOR WORKUPS DUE TO RECENT CHANGES IN KIDNEY ALLOCATION
Authors: <u > M. Rewinski</u>, P. Doyle, J. Devivo, S. Vicki, D. Durkin, R. Caruk, L. Bow, Transplant Immunology, Hartford Healthcare, Hartford, Connecticut, UNITED STATES|
Aim: We monitored two months of data following implementation of the new UNOS kidney allocation policy on 3/15/21 and compared it to the same two months from 2019. Our aim was to assess our approach to current deceased donor (DD) workups and address how to effectively implement changes, if needed. Methods: The UNOS OPTN Kidney Transplantation Committee has recently adjusted the algorithm for kidney allocation, essentially eliminating our Donor Service Area and consequently the old Region 1, when determining kidney placement for transplant. While other waiting list factors such as candidate age, time on dialysis, etc., have remained unchanged, the new criteria now incorporates a geographical range of 250 nautical miles from the donor hospital for kidney allocation. Points are assigned according to the candidate’s proximity to the donor hospital, “to prevent a kidney being transported further away when there is a candidate of similar priority closer to the donor hospital” (OPTN Proposal Overview, 2019). Results: In April and May of 2019, our lab received samples from 18 DD and performed 94 crossmatches (XMs) for an average of 5.2 XMs per donor, resulting in 20 patients receiving kidney transplants. Aside from offers outside of our region, all XMs were performed on patients from sequence 1–12 on the UNET kidney match run. 61% (11/18) of these DD originated from local hospitals within our DSA/Region 1. In April and May of 2021, our lab received samples from 21 DD but performed only 43 XMs for an average of 2.0 XMs per donor, resulting in 10 kidney transplants. The match run sequences for these patients ranged from 4 to 1381 and overall were much higher (i.e. our program’s patients appeared further down the list) than those in 2019. Only 5% (1/21) of these DD would have been considered ‘local’ donors under the old allocation system. Conclusion: While the overall number of DD in the comparative time frames remained essentially the same, the main impact of the new kidney allocation, as expected, has been fewer DD from local hospitals, fewer patients to XM and consequently fewer overall DD transplants. Since many DD are now originating from distant donor hospitals, we place greater reliance on virtual XMs and have appropriately prepared for accurate, timely virtual XMs by more frequent and robust antibody testing.
P97
Laboratory improvement, Evaluations
Title: HLA ANTIBODY IDENTIFICATION BY LUMINEX SINGLE ANTIGEN BEAD ASSAY – INTRALABORATORY COMPARISON
Authors: <u > A.F. Locke</u>, Y. Zheng, J. Zhang, E. Reed, D. Gjertson, Pathology and Laboratory Medicine, UCLA Immunogenetics Center, Los Angeles, California, UNITED STATES|
Aim: Widely used for histocompatibility testing, the Luminex single antigen bead (SAB) test measures the binding of HLA antibodies in patient sera by correlating median fluorescence intensity (MFI) with HLA antibody concentration. The aim of this study is to compare the SAB data of individual labs to examine variability in the reporting of antibodies and antibody strength. Methods: Labs participating in the UCLA Single Antigen, Flow and Virtual Crossmatch Exchange tested sera with a broad range of class I and/or class II specificities. The study collected and analyzed Luminex SAB data from sera tested 2x across consecutive studies from 2016 − 2020. Data collected included antibody specificities, MFI and positive cutoff values, vendor/lot information, and pretreatment data. The mean MFI of the dominate antibody reported by an individual lab for each locus was calculated to determine the change (delta) in the reporting of antibody strength across consecutive studies. Results: A total of 12 samples were tested 2x across 13 studies (Exchanges 6 – 18). Positive cutoff values among labs ranged from 700 to 15,000 for class I and class II loci. Overall, the reporting of antibodies and antibody strength was consistent for individual labs. For example, each point in the figures below represents the change in the MFI reported by individual labs for a serum examined as sample nos. 102 and 105. The position of the points between the 2 boundaries shows MFI for peak antibodies tested a second time to fall within a variation of 50% of the mean value. When the mean MFI was < 10000, points were found to be beyond these boundaries compared to relative positions of points when the mean MFI was > 15000. Although it is not shown, data from the study also revealed no significant difference in the reporting of DSA strength between kit lots. Deviations in MFI reporting across repeat serum were 10% – 15% less for treated sera compared to untreated sera. Conclusion: The data showed that labs using the Luminex SAB test were consistent in the reporting of antibodies and antibody strength for repeat serum. Results were consistent across different lots and consistent among labs with pretreatment protocols. However, factors such as tech to tech differences, reagents used for treating sera, and handling/storage of sera may still affect test outcome. Therefore, standards for testing are still needed to ensure the accuracy of the test.
P98
Laboratory improvement, Evaluations
Title: MONITORING HLA ANTIBODY TITERS DURING ANTIBODY MEDIATED REJECTION
Authors: <u > C. Maguire</u>, R. Das, A. Shetty, M. Abundis, A.R. Tambur, Northwestern University, Chicago, Illinois, UNITED STATES|
Aim: HLA antibody titer can serve as a prognostic tool to assess candidacy and measure efficacy of antibody removal therapies, superior to MFI values of the single antigen bead (SAB) assay. Further, the amount of antibody removed during desensitization with plasmapheresis/intravenous immunoglobulin (PP/IVIG) correlates with the number of PP/IVIG cycles, for the initial 5 cycles. This pilot study investigated whether a similar correlation exists between titer and antibody removal therapies in patients (pts) undergoing antibody mediated rejection (ABMR) treatment. Methods: 14 adult kidney transplant recipients (57%M; 50% Cauc/21% Hisp/14% AA/7% Asian; age: 41 +/- 16.4) with biopsy-confirmed ABMR and HLA Class II antibodies with titers > 256 were studied. All pts had at least 2 serum samples tested: pre- and end-of-treatment. New dilution studies and C3d SAB assays were performed using LIFECODES LSA Class II beads generously donated by Immucor for the purpose of this study; all samples per patient were tested simultaneously, on the same day, by a single technologist to minimize assay variability. Results: Biopsy results demonstrated ABMR as sole diagnosis in 50% of pts, 29% had mixed T-cell mediated rejection/ABMR, and 21% had ABMR with other histologic findings. All pts received PP/IVIG ranging from 4 to 20 cycles (mean 10.6 +/- 4.9). eGFR at the start of ABMR treatment was 34.14 +/-13.28. DSA titer at the time of diagnosis ranged 1:8–8192 (median 2048). All pts exhibited a decrease in antibody titer following treatment, with a median decrease of 2.99 titers (0.67–5.44) for all positive antibodies and 3 titers (0–6) for DSA. A good correlation between titer and C3d positivity was observed for titers>1:64 (PPV: 95.5%). Conclusion: No correlation between HLA antibody titer and number of PP/IVIG cycles was found in this cohort with high titer DSA and ABMR, comparing pre- and end of treatment antibody levels. Of note, we did not have mid-treatment serum samples to determine if the initial response was proportional to number of cycle (as we have seen for desensitization) but lost in pts that received higher numbers of cycles. Study limitations include small cohort size with variable treatment protocols in addition to PP/IVIG. Additionally, only pts with very strong HLA antibodies were included. Those with lower levels of antibody may respond more favorably to PP/IVIG such that measuring titers could be informative.
P99
Laboratory improvement, Evaluations
Title: SSO PROBE PATTERN CODING ERROR FOR SOME HLA ALLELES REPORTED AT THE FOURTH FIELD LEVEL
Authors: <u > I. Salvador</u>, A. Comeaux, R. Vorhaben, A. Usmani, C. Lacelle, University of Texas Southwestern Medical Center, Dallas, Texas, UNITED STATES|
Aim: This study documents errors in the coding of SSO probe pattern reported at the fourth field level resulting in the software calling erroneous allele pair possibilities. Methods: Samples were tested using LABTYPE XR kit and analyzed using Fusion 4.6 (One Lambda). Results: We first noticed the issue in a patient’s HLA-B typing results. For this sample, Fusion 4.6 identified the possible G1 typing (common on both allele) B*42:01:01:01,-, but listed under G2 (common on one allele) the possible allele combination B*07:02:01:22, 42:01:01:01. The presence of the HLA-B*07:02:01:22 allele in one of the allele pair made us question the call and wonder if the common B*07:02:01:01 allele might be present given the differences between the two alleles exist in non-coding regions that are not targeted by probes in the LABTYPE XR HLA-B kit. If the B*07:01:01:22 could not be ruled out by the software, the B*07:02:01:01 should not be excluded by the software. Upon further investigation to determine why all HLA-B*07:02:01:XX except the B*07:02:01:22 allele were ruled out, we discovered that the coding for the B*07:02:01:22 allele was erroneous. Several probes that are coded positive for all HLA-B*07:02:01:XX alleles including probe 530, 556, 557 and 570 were missed when the coding for the HLA-B*07:02:01:22 allele was entered causing this allele to show up in a possible allele pair while other alleles identical for the three first fields were excluded. Upon identification of this case, we went back and looked at probe coding for the LABTYPE XR products and identified several alleles including A*02:05:01:07, 02:01:01:52, 01:01:01:24 and 03:01:01:52 that were miscoded. All miscoded alleles identified in our laboratory were alleles with 4 fields. Verifying the probe coding for the other fields is substantially more time consuming as they cannot be identified easily without performing sequence alignment and verifying probe sequence information. Conclusion: The case presented is representative of others we have encountered and reinforces the importance of software validation in the laboratory. It also questions the use of 4 field alleles for SSO typing platforms that do not assess for DNA differences in non-coding regions. Due to the increasing number of 4 field alleles called using NGS, the use of 2 fields typing by intermediate/limited high resolution SSO software should be considered to ensure greater quality of the product.
P100
Laboratory improvement, Evaluations
Title: OVERUTILIZATION OF HLA TESTING FOR CELIAC DISEASE SCREENING
Authors: <u > F. Davidovic</u>, P. Dokouhaki, T. Pearce, A. Mostafa, Saskatchewan Health Authority, Saskatoon, Saskatchewan, CANADA|
Aim: Although testing for HLA-DQ2 and DQ8 could be valuable for diagnosis of celiac disease (CD), it is not recommended for primary screening due to its low positive predictive value. Inappropriate use of HLA typing could not only be misleading but also lead to unnecessary downstream investigations with potential harm to patients and extra burden for health care system. By analyzing the pattern of HLA test ordering in Saskatchewan-Canada, we aim to determine if the increased rates of testing are due to clinically inappropriate utilization. Methods: Utilization data for HLA-DQ2/DQ8 ordered in Saskatchewan from Jan 1 < sup > st</sup> – Dec 31 < sup > st</sup > 2019 were retrieved. We then compared this against the list of patients for whom anti-Tissue Transglutaminase (TTG) test, the recommended screening test for CD, have been requested in 2019. Next, based on the available clinical guidelines, we determined the rate of inappropriate utilization of HLA-DQ2/DQ8 testing in our health care system. Results: In early 2020, Saskatchewan's population was estimated at 1,181,987 with allele frequency of DQ2 (18%), and DQ8 (8.6%). This indicates approximately 150,000 of Saskatchewan population carry either allele associated with celiac disease. In 2019, the HLA laboratory in Saskatchewan received 171 samples for HLA-DQ2 and DQ8 genotype testing. Out of the 171 tests, 18 (10.5%) were positive for DQ2 homozygous, 61 (35.7%) for DQ2 heterozygous, 17 (9.9%) for DQ2/DQ8 and 19 (11.1%) positive for DQ8 only. Almost half of the patients (46.2%) for whom HLA typing had been requested, also were tested for anti-TTG serology, with only 3 (4%) yielded positive results. Conclusion: Our preliminary findings suggest a significant portion of HLA testing for CD was clinically inappropriate, which may have led to unnecessary diagnoses, tests, procedures, treatments, and burden on the healthcare system.
P101
Laboratory improvement, New assays
Title: RAPID ENHANCED FLOW CYTOMETRIC CROSSMATCH ASSAY
Authors: <u > Y. Yin</u>, P. Takemura, G. Aguas, M. Hickey, N.M. Valenzuela, R.A. Sosa, C. Butler, J. Zhang, E. Reed, UCLA Immunogenetics Center, Department of Pathology & Laboratory Medicine, Los Angeles, California, UNITED STATES|
Aim: To reduce turnaround time of the flow cytometric crossmatch (FCXM) assay, we optimized a high-throughput 96-well plate FCXM protocol. The optimized protocol reduces the overall time to complete the assay, in part, by using an automated lymphocyte isolation. Methods: For conventional lymphocyte isolation, cells were isolated using RosetteSep (Stemcell Technologies) by density gradient centrifugation following manufacturer's instructions. In the automated lymphocyte isolation protocol, cells were isolated on RoboSep™-S platform (Stemcell Technologies). Cells were left untreated or treated with Pronase (Sigma) and mouse-anti-human CD20 (Southern Biotech) to reduce CD20 and FcγRs on the cell surface. Then, cells were incubated with selected sera. The optimization of the FCXM includes a 96-well plate platform rather than individual tubes. Cells were stained with anti-CD3-PE, anti-CD19-APC and anti-human IgG-FITC to identify T cells, B cells and IgG bound to the surface of the cells. Fluorescence was analyzed on a multicolor flow cytometer (BD Biosciences). Results: We developed a rapid FCXM procedure, which reduces assay turnaround time from lymphocyte isolation to fluorescence acquisition by > 30% over conventional method, and simplifies the set-up without compromising quality or sensitivity. For the two isolation methods (RoboSep vs. RosetteSep) with 50 donors (PBL-LD n = 20, PBL-DD n = 20, and spleen n = 10), the average lymphocyte yield on RoboSep platform increases 26.8% (P < 0.0001) and 30% (P = 0.0002) for PBL-LD, -DD, respectively, and even doubles (103%, P = 0.0006) for spleen. Importantly, linear regression analysis of the median channel shift (MCS) values of untreated FCXM revealed excellent concordance (R < sup > 2</sup > of 0.97 for T cell, and 0.93 for B cell) between optimized and conventional FCXM protocol (Figure 1 A and B). Upon further investigation, the sensitivity and specificity of optimized assay for T cell are both 100%, while 100% and 98.1% for B cell, respectively. Additionally, the FCXM results for 17 Pronase treated lymphocytes (PBL-LD n = 5, PBL-DD n = 10, and spleen n = 2) are in 100% agreement for both of T and B cells (R < sup > 2</sup > of 0.99, Figure 1C and D). Overall, the accuracy for the optimized FCXM assay is 99.5%. Conclusion: Optimized FCXM assay is more efficient in generating highly correlated results compared to conventional method. Implementation of the rapid optimized FCXM assay will expedite pre-transplantation work-up and improve patient care.
Figure 1. Optimized and conventional FCXM results show excellent concordance. A) and B): Untreated lymphocyte (patient sera n = 106). C) and D): Pronase treated lymphocyte (patient sera n = 27). The lymphocytes were performed in parallel using optimized (RoboSep) and conventional (RosetteSep) FCXM protocol, and the data are expressed as MCS.
P102
Laboratory improvement, QA/QC
Title: IMPROVING DNA QUALITY FROM SUBOPTIMAL SAMPLES FOR HLA TYPING USING ION TORRENT NEXT GENERATION SEQUENCING
Authors: <u > J. Glover</u>, M. Baron, A. Sum, S.J. Taylor, C. Carozza, E. Pimpinella, W. Dong, M.C. Philogene, Histocompatibility Laboratory Services, American National Red Cross, Philadelphia, Pennsylvania, UNITED STATES|
Aim: Hematopoietic stem cell transplantation and clinical research studies require allele-level HLA typing. DNA concentration of 25 ng/µl and A260/A230 ratio > 1.7 is ideal for HLA typing by Next-Generation Sequencing (NGS). However, samples collected from buccal swabs, frozen cells or whole blood from transplant candidates, their donors or for research studies may not always produce optimal DNA. Our aim was to improve DNA quality from rare and valuable samples to reduce failed amplification reactions which lead to repeat testing or failure to obtain acceptable HLA typing data. Validation of a cleaning protocol was completed in our laboratory in December 2020. Methods: Genomic DNA was extracted using the MagMAX DNA Ultra 2.0 kit (ThermoFisher Scientific) on the KingFisher™ Flex and DNA concentrations measured with the NanoDrop™ 2000. PCR amplification was performed using the AllType NGS 11-Loci Amplification Kit (One Lambda, Inc). DNA samples that failed to amplify were cleaned using AMPure XP Beads (Beckman Coulter) with two washes of 75% ethanol. Testing was then repeated according to manufacturer instructions using AllType™ NGS reagents and Ion 520™& Ion 530™ExT kit (One Lambda, Inc) on the Ion S5™ System and Ion Chef™ workstation. Results: Between January 2021 and May 2021, 1116 DNA samples were extracted for NGS testing. 109 DNA samples did not meet required quality standards for NGS and were cleaned using validated protocol. HLA typing was successful at all 11 loci for 107 (98%) of the DNA samples which were deemed to have poor quality (Figure 1). Conclusion: Samples with A260/A230 < 1.5 are now cleaned prior to amplification for NGS. We have successfully reduced our NGS failure rate due to poor sample quality to < 1.8%. AmPure XP beads can be used as an effective method to purify genomic DNA with an unacceptable A260/A230 ratio for amplification.
Figure 1: A representative sampling of pre cleaning and post cleaning results
P103
Laboratory improvement, QA/QC
Title: ROUTINE USE OF HUMANIZED ANTI-HLA MONOCLONAL ANTIBODIES AS POSITIVE CONTROL SERA
Authors: <u > H. Sanders</u>, V. Bravo-Egana, Histocompatibility/Immunology Laboratory, Medical College of Georgia/Augusta University, Augusta, Georgia, UNITED STATES|
Aim: Historically, HLA laboratories have pooled highly reactive patient samples to create positive control sera. However, this is not always a simple process. Some patient serum requires treatment such as heat inactivation or adsorption. Addition of serum with particular specificities must be balanced against the dilution effect of added volume. Each lot of pooled positive control must be extensively validated. And, most alarmingly, it is still possible – even probable – that the process will conclude with a positive control that has gaps in reactivity. This begs the question: is there a better way? Methods: The major barrier to developing HLA control sera with predictable, comprehensive reactivity is the need to use human antibody, as commercial assay reagents bind to human IgG. Research published in 2013 described one solution: hybrid human/murine antibodies. Murine monoclonal antibodies (MoAb) W6/32 and F3.3 were selected for their availability and reactivity with public epitopes of class I and class II HLA, respectively, and engineered to chimerize with human constant portions C-gamma1 and C-kappa. In the original publication, these recombinant chimeric MoAb were shown to have consistent positive reactivity in flow crossmatching and both serological and Luminex-based antibody screening. Results: Our laboratory validated the MoAb controls, now commercially available, for flow crossmatching, flow antibody screening, and Luminex antibody panels. We determined optimal dilutions of MoAb positive controls for each assay and confirmed that results were stable and reproducible. Conclusion: Our laboratory now has six years of experience with MoAb positive controls. Their use has significantly improved the consistency of positive shifts in flow crossmatch controls as well as creating reliable, reactive positive controls for Luminex panels. In addition, the reactivity of bioengineered MoAb is highly conserved in each new lot and shipment, greatly simplifying the process of validation and quality control. For consistency and ease of use, positive control using humanized MoAb has been a great improvement over pooled patient serum.
P104
Laboratory improvement, Evaluations
Title: DISCORDANT DPB1 TYPING REVEALS TESTING PLATFORM DEFICIENCIES
Authors: <u > M. Najor</u>, A. Thai, J. Jang, S. Murphy, S. Longwe, D. Antisdel, S. Rosen-Bronson, O. Timofeeva, Laboratory Medicine and Pathology, Medstar Georgetown University Hospital, Springfield, Virginia, UNITED STATES|S. Rosen-Bronson, Georgetown University,
Aim: Accurate HLA typing is quintessential to successful solid organ transplantation. We present a case study of a potential kidney candidate evaluated at MGUH that observed multiple different DPB1 genotypes using five different assay platforms. These results have ultimately led to a laboratory workflow process improvement when rare typing results are detected. Methods: HLA DPB1 typing was performed on gDNA extracted from whole blood using rSSO-XR, NGS MX11-3 kit, high resolution SSP, and qPCR using QTYPE and Linkseq kits. Results: The patient was originally HLA typed by rSSO, which resulted in DPB1*03:01:01G and DPB1*682:01 alleles. The sample was reflexed to NGS due to the rare allele detection and resulted in a different genotype of DPB1*04:01:01G and DPB1*111:01. High Resolution SSP was then administered due to the discordant results between platforms and surprisingly, resulted in two rare allele calls (Table 1), ruling out both genotypes due to false negative reaction. qPCR-based typing using Linkseq was not able to assign a genotype for DPB1, while QTYPE resulted in DPB1*03:01 and DPB1*04:01 alleles. LinkSeq ruled out DPB1*03:01 and neither qPCR assay could rule out the DPB1*111:01. Based on the exon 2 sequence, DPB1*111:01 is the only allele known in IMGT/HLA database 3.43.0 that has 0 mismatches with our results. DPB1*03:01:01:01 differs from DPB1*111:01 by a single nucleotide in exon 2 (Figure 1), resulting in a different amino acid at position 32 (arginine vs. proline). Furthermore, rSSO and SSP do not have probes or primers to distinguish these alleles. Since the remaining exon sequences are unknown for DPB1*111:01, it leads to problematic genotype assignment using rSSO and SSP platforms. Conclusion: Ultimately the patient’s results were reported based on the findings from NGS, which presented a clear typing of DPB1*04:01:01G and DPB1*111:01 alleles. The MGUH lab has implemented a process improvement, whereby all rare HLA typing detected using non-NGS platforms will be reflexed to NGS. It is important to note that many laboratories do not have NGS technology and/or multiple platforms to reflex typing discrepancies. An inaccurate typing result can lead to major ramifications, such as potential AMR and potential graft loss because the patient may have donor specific antibody to an undetected HLA allele in the donor’s typing.
DPB1*03:01 and DPB1*111:01 Exon 2 Sequence Alignment
P105
Laboratory improvement, QA/QC
Title: NEW UNOS ALLOCATION SYSTEM – IMPACT ON LABORATORY OPERATIONS
Authors: <u > P. Rao</u>, M. Marchioni, D. King, I. Okere, J. Napoleon, Laboratory, NJ Sharing Network, New Providence, New Jersey, UNITED STATES|
Aim: March 15 < sup > th</sup > of 2021, UNOS replaced the donation service area (DSA) with a 250 nautical mile fixed circle. As New Jersey borders the Atlantic Ocean, this new allocation system had a significant impact on transplant laboratory operations. Methods: We reviewed the following data in 30-day intervals leading up to the allocation change and after:<br /> - patients crossmatched with local donors < br /> - patients crossmatched with import donors < br /> - virtual crossmatch requests < br /> - local donors < br /> - import donors Results: Post allocation change, there was a 3.6-fold decrease in the average number of patients crossmatched with local donors, a 1.6-fold increase in patients crossmatched with import donors, a 4.1-fold increase in virtual crossmatch requests, a 1.3-fold decrease in local donors crossmatched, and a 1.8-fold increase in import donors crossmatched. Conclusion: Of the changes we observed, the most significant were the increase in virtual crossmatch requests and the decrease in patients crossmatched with local donors. < br /> Of note was the need for extra staff to handle the increase in virtual crossmatch and import demand, the impact on the balance of our routine workload versus stat donor testing requests, and associated stress on the staff. < br /> Lastly, we believe that the new allocation has resulted in patients within our DSA receiving less local donor organs and more import donor organs.
P106
Laboratory improvement, Evaluations
Title: EXPLAINING THE UNEXPECTED CROSSMATCH RESULT USING AUTOMATION
Authors: <u > M.C. Montgomery</u>, C. Cornaby, J. Schmitz, E. Weimer, Immunology, Histocompatibility and Immunogenetics Laboratories, UNC Health, Chapel Hill, North Carolina, UNITED STATES|J. Schmitz, E. Weimer, Department of Pathology & Laboratory Medicine, Univers
Aim: In an effort to reduce variability, our lab has begun to validate the single antigen bead (SAB) assay using automation. Comparison of the mean fluorescence intensity (MFI) results indicated higher values using automation compared to manual testing. < br /> <br /> The higher MFI values with automation were investigated using unexplained positive flow cytometric crossmatches (FCXM). We hypothesized that improved serum washing using automation provided a more accurate antibody profile and would explain the positive FCXM. Methods: Three potential recipients of deceased donor kidneys were evaluated via manual SAB testing (One Lambda LABScreen Single Antigen Class I and II) with EDTA treated serum. Virtual crossmatch (VXMs) predictions and FXCM results are in Table 1. Sera were then tested with SABs on the Tecan FreedomEvo200 platform with and without EDTA treatment. Results: Donor specific antibody MFI were higher (average of 138% increase) when run on the Tecan compared to manual. Negative and positive control sera were within the range observed with the manual assay. Not all MFIs showed this increase (Table 2). Taken together, these observations suggest the MFI increase is due to antibodies present in the serum and not a global increase in reactivity. < br /> <br /> The observed higher MFIs with Tecan explain the FCXM results. Had those data been used in a VXM, a positive FCXM would have been predicted. More importantly, had the Tecan SAB data been available, none of the potential deceased donors would have come up on a match list for these patients. Conclusion: Automated SAB testing better explained FCXM results. It is likely the Tecan provided better SAB washing compared to the manual method, reducing interfering substances that inhibit antibody binding or fluorescence detection. Implementation of the Tecan may provide a more accurate representation of the patients’ antibody profile and in turn provide better data in order to select the proper donor for transplantation.
Table 2. MFI values for manual and automated SAB tests
P107
Laboratory improvement, Cellular assays
Title: RESOLVING DUBIOUS FLOW CYTOMETRIC CROSSMATCH RESULTS DUE TO THE PRESENCE OF GAMMA-DELTA T CELLS
Authors: <u > S.K. Cheung</u>, J.C. Ho, K.W. Tam, E. Leung, J. Kwok, Division of Transplantation and Immunogenetics, Pathology, Queen Mary Hospital, Pokfulam, HONG KONG|
Aim: Flow cytometric crossmatch (FCXM) is a routine cell-based assay for evaluation of histocompatibility between potential kidney transplant donor and recipient. It has high sensitivity to detect the presence of preformed donor-specific IgG antibodies (DSA) which may lead to graft rejection. False negative or positive FCXM results due to Fc receptor, pronase treatment or Rituximab have been reported. We here reported a simple method to resolve dubious FCXM results due to the presence of gamma-delta T (gdT) cells. Methods: Standard FCXM assay was performed for living related kidney transplant workup. Donor peripheral blood mononuclear cells were incubated with recipient’s or control sera, followed by staining with CD3-PerCP, CD19-PE and anti-human IgG-FITC antibodies. Analysis was performed in a flow cytometer and the binding of DSA antibodies was determined by mean channel shift (MCS) with pre-defined cut-offs. Anti-TCRgd-PC7 antibodies were added in the staining step when significant double peaks were detected in T-cell FCXM. Results: FCXM was performed with anti-TCRgd antibodies in 3 transplant workups with significant double peaks in T-cell FCXM. Over 30% of the total CD3 + T cells were TCRgd positive and these gdT cells have a significant stronger IgG binding affinity which contributed solely to the second peak with higher MFI. Negative T-cell FCXM results in both TCRgd- and TCRgd + T cells were found in 2 out of the 3 cases. A weak positive T-cell FCXM result was detected in 1 pair which was likely due to the presence of anti-A isohemagglutinins in the patient serum against the potential donor of ABOi transplant work up. Conclusion: Gamma-delta T cells express low level of Fc gamma receptor which leads to the non-specific binding of IgG. The presence of high gdT cell count may interfere with the FCXM interpretation. Resolving the TCRgd + and TCRgd- cells populations allow calculation of MCS separately and provide more accurate interpretation of crossmatch results.
Figure 1. Detection of TCRgd- and TCRgd + T cell populations in lymphocytes.
Figure 2. Differential IgG binding affinity of TCRgd- and TCRgd + T cell populations.
P108
New Technologies, Bioinformatics
Title: A WEB-BASED CPRA CALCULATOR BASED ON A COMPREHENSIVE HIGH RESOLUTION HLA GENOTYPE PANEL OF US STEM CELL DONOR
Authors: M. Maiers, National Marrow Donor Program / Be The Match, Minneapolis, Minnesota, UNITED STATES|<u > L. Gragert</u>, D.G. Biagini, Pathology and Laboratory Medicine, Tulane University School of P, New Orleans, Louisiana, UNITED STATES|C. Jett, K. Lindblad, U
Aim: Organ transplant candidate HLA antibody sensitization limits the pool of compatible donors. To provide equity in access to transplantation for the kidney allocation system (KAS), sensitized candidates are given escalating allocation points and priority based on their calculated panel reactive antibody (CPRA) values, the percentage of the donor pool incompatible based on the candidate’s unacceptable antigens. The current OPTN panel for computing CPRA has limitations due to antigen-level typing resolution, lack of coverage of the DQA1, DPA1, and DPB1 loci, and small sample size for minority populations. Our aim was to develop an accurate and comprehensive CPRA calculator based on high resolution genotypes from the US stem cell donor registry. Methods: Population-specific high resolution haplotype frequencies across all classical HLA loci were computed based on a panel of over 10 million US stem cell donors typed by DNA-based methods from the Be The Match Registry. For each donor, high resolution HLA genotypes were assigned using random realization of possible haplotype pairs. Based on proportions of previous US deceased donors across all organ types, we computed ethnic-weighted average frequencies for each unique genotype. CPRA values are computed based on summing the ethnic-weighted frequency of genotypes that contain any of the input list of unacceptable HLA antigens. Results: We previously found stem cell donors applicable for computing antigen-level CPRA values for A, B, C, DR, and DQ based on HLA haplotype data, and have now expanded coverage to all classical HLA loci at high resolution based on genotypes. Antigen categories, two-field HLA alleles, DPB1 hypervariable region epitopes, single amino acid polymorphisms, Class II heterodimers, and eplet categories are supported. A web-based CPRA calculator implemented in Python using the Django web framework is available at https://transplanttoolbox.org/nmdp_cpra/. Conclusion: This calculator is serving as a testbed for development and validation of a new OPTN CPRA calculator for the US allocation system, expected for 2022 with kidney/pancreas. After implementation, candidate CPRA values will begin to reflect donor HLA frequencies for allele-specific unacceptable antigens as well as DQA1, DPA1, and DPB1 locus HLA specificities. Our approach be applied to other global populations with suitable HLA data.
P109
New Technologies, Bioinformatics
Title: FROM ZERO TO HERO: USE OF THE STATISTICAL PROGRAMMING LANGUAGE R TO MANAGE CLINICAL HLA LABORATORY DATA BY A LAB WITH LITTLE PROGRAMMING EXPERIENCE
Authors: <u > N. Brown</u>, T. Bui, Pathology & Laboratory Medicine, Penn Medicine, Philadelphia, Pennsylvania, UNITED STATES|H. Gao, Surgery, Penn Medicine, Philadelphia, Pennsylvania, UNITED STATES|
Aim: Laboratory information systems (LIS) are optimized for managing and reporting clinical results but are not designed to facilitate data analysis outside of normal clinical operations. Programming languages used to manipulate data often require specialized skills. The statistical programming language R has many features for data management, and its code offers a gentle learning curve for new users. To determine if an HLA lab with little programming experience could use R to manage clinical data, our LIS was searched to find cases for the “HLA Haplotypes in Families by NGS” project from the 18 < sup > th</sup > International HLA & Immunogenetics Workshop (IHIW). Methods: R (version 4.1.0) and a user-friendly code visualization program (RStudio version 1.4.1717), both free of charge, were run on a desktop computer. The packages “obdc,” “DBI,” “tidyverse,” and “openxlsx,” which extend the functionality of R, were installed in RStudio. The Patient, RelatedPatient, PatientCase, Sample and Test tables were extracted from our LIS (HistoTrac version 2.52.8, SystemLink), which stores data in an SQL server. Results: The IHIW families project requires nuclear families ≥ 3, containing at least a parent and child. Fig. 1 diagrams the approach used to find such families in our database. Recipients and donors were separated to facilitate subsequent steps, and then joined using the PatientCase and RelatedPatient tables, preserving only patients with donors. Of the 75 different donor types found in our database, only those comprising a nuclear family (mother, wife, sister, daughter, etc.) were kept, and further simplified to one of 4 categories: child, parent, sibling or spouse. Individuals were linked to the Sample and Test tables to filter for those with NGS typing. Finally, families were filtered for those matching one of 4 possible groups meeting IHIW requirements (Fig. 2), identifying 164 families. The majority, 137 families, consisted of a patient and multiple children. The code used in this project was deposited here: github.com/k96nb01/R-HistoTrac-IHIW-Families. Conclusion: With no direct experience with R, our HLA laboratory found it and the code visualization program RStudio to be easy to use, facilitating automatic identification of families for an IHIW project. These tools offer great promise for lab management, quality assurance, and research purposes.
P110
New Technologies, NGS and TGS
Title: UNDERSTANDING THE GENOMIC HLA DIFFERENCES OF MEXICAN MESTIZOS ACCORDING TO THEIR GEOGRPHIC LOCATIONS: OBREGÓN CITY, SONORA, NORTHWEST MEXICO, MEXICO CITY (CDMX) AND OAXACA IN THE SOUTHWEST
Authors: A. Del Olmo, K.I. Rivera, A. Rodriguez, A. Florentino, C.F. Mercado, M.A. Carmona, C.J. Balderas, D. Castillo, <u > C. Gorodezky</u>, Laboratory of Immunology and Immunogenetics, Fundación Comparte Vida, A.C., Mexico City, CDMX, MEXICO|A. Ramos, Laboratorio
Aim: Molecular information reveals the structure and history of admixed populations. Gradients of African heritage were discovered from the center of Mexico out to the coasts, and of European heritage, from the south towards the north. The ethnic and genetic composition of the different regions in Mexico differs greatly, because of the distinct Indian groups settled and the different Europeans established. CDMX resulted from the admixture of Nahuas, Spaniards, African slaves and Sephardi Jews. Obregón City, called “Cajeme”, honoring its Indian Yaqui leader, fought against the Germans, Irish and North-Americans. Oaxacan Mestizos resulted from the admixture of Zapotecans, Africans and Spaniards. We compared individuals from these very distinct areas, to unravel their HLA diversity and origins. Methods: We included 77 Mestizos, born in CDMX, 108 Oaxacan and 76 from Sonora. DNA was isolated with the Maxwell16 instrument and NGS typing was done for 11 HLA loci; we included (A*,C*,B*,DRB1*,DQA1*, DQB1*loci only). The MIA FORA NGS Kit (Immucor, Inc.) was used on an Illumina MySeq platform; MIA FORA FLEX v3.0 was used for analysis. Results: Mediterranean, and Semitic alleles are prevalent in CDMX: <i > A*02:01,C*04:01,C*07:02,B*35:01,B*39:05, DRB1*08:02, DRB1*04:07, DQB1*03:01, DQB1*03:02</i > . while in Obregón prevail: <i > A*01:01, A*02:01, C*04:01,C*05:01,B*40:02,B*44:03,DRB1*04:07,DRB1*07:01, DQB1*03:02,DQB1*02:02.</i > In Oaxaca some alleles are unique to Mexicans < i>: A*68:03,A*68:05,C*03:04,C*15:09,B*39:05,B*35:14, B*35:12, B*35:43, B*39:05</i > and < i > B*35:17 which</i > exists In Natives, Hispanics and Asians. Two haplotypes were prevalent in Oaxaca Vs. CDMX < i > A*24:02-B*39:06-C*07:02-DRB1*14:06-DQA1*05:05-DQB1*03:01</i>(p = 0.01) present in Amerindians(AMI), Hispanics, Philipino, and Africans;<i > A*68:03-B*39:05-C*07:02-DRB1*04:07-DQA1*03:01-DQB1*03:02</i>(p = 0.01) exists in AMI, Africans, Hispanics, Caribbeans. In Obregón is significantly higher: <i > A*03:01-B*07:02-C*07:02-DRB1*15:01-DQA1*01:02-DQB1*06:02 (p = 0.03).</i > Conclusion: The Native, Semitic and Mediterranean backgrounds prevail in CDMX < i > . </i > Sonora composed by North/Center European genes, reflects the Irish and German ancestry, and some alleles from the Yaqui. Mestizos from Oaxaca have haplotypes that differentiated at multiple loci due to recombination or convergent evolution, showing Native, and Asian Ancestry.
P111
New Technologies, Bioinformatics
Title: SEARCH SUMMARY SCORE: THE NEXT GENERATION OF RAPID ASSESSMENT FOR UNRELATED BONE MARROW DONOR SEARCHES
Authors: <u > S. Fingerson</u>, Y. Bolon, Center for International Blood and Marrow Transplant Research, National Marrow Donor Program/Be The Match, Minneapolis, Minnesota, UNITED STATES|N. Garrett, K. Wadsworth, M. Maiers, National Marrow Donor Program/Be The Match
Aim: Existing measures such as the NMDP Search Prognosis and MSK Search Prognosis are able to provide high level assessments on the potential success of unrelated donor searches using known HLA genotype frequencies or the Haplogic search list but do not account for donor availability. We created a Search Summary Score that predicts the success of finding an available suitably matched donor in order to maximize the efficiency of the search process for the patient. Methods: We developed a novel score that utilizes both the Haplogic search list and the Donor Readiness Score to calculate the probability of a patient finding an available donor within a given match category. To validate this new scoring system, we analyzed 6,072 previous searches where the patient progressed to transplant and compared our Search Summary Score predictions for donors on search result lists at 8-of-8 and 7-of-8 HLA match categories to the match category of the actual selected donor. Donor lists for each patient include any donor on the list that has a 7-of-8 match prediction > 1% and a donor readiness score. Results: We found that we were able to successfully predict the outcome of searches resulting in an 8-of-8 match as well as a 7-of-8 match. We correctly predicted 5,814 results with the prediction threshold set at 50% which gives Brier Scores of 0.031 (8-of-8) and 0.181 (7-of-8) and F1 Scores of 0.974 (8-of-8) and 0.881 (7-of-8). Conclusion: The Search Summary Score can help provide a high-level upfront expectation of donor search outcomes, which will assist in decision making for whether or not to pursue multiple treatment path options. This can be especially helpful for non-white or multi-race patients who can often have shorter donors lists and see lower donor availability. With the implementation of the unified global Haplogic donor list and upcoming updates to Donor Readiness Score, we will be able to provide a global Search Summary Score that includes NMDP, WMDA, as well as other Co-op registries. We plan to continue this analysis to include additional match categories such as 10-of-10 and 9-of-10, compare results with existing scoring systems, and evaluate the utility of the Search Summary Score in borderline scenarios to impact overall outcomes.
P112
New Technologies, Bioinformatics
Title: EVALUATION OF COMPUTERIZED VIRTUAL CROSSMATCH WITH HL7 INTERFACING AND ANTIGEN MATCHING CAPABILITIES
Authors: M. Reardon, D. Saini, <u > C. Brack</u>, HLA Data Systems, Houston, Texas, UNITED STATES|N. Soe, M. Marschner, M. Sexton, S. Corliss, Tissue Typing Laboratory, AdventHealth, Orlando, Florida, UNITED STATES|
Aim: Our aim is to evaluate the accuracy, interoperability and antigen matching capabilities of a computerized virtual crossmatch within the AdventHealth Tissue Typing Laboratory. Our lab currently performs virtual crossmatch (vXM) assessments and has evaluated a computerized vXM (cVXM) to streamline on-demand vXMs for donor offers, confirmatory crossmatch testing and the replacement of our manual resulting process into the electronic medical record (EMR). Our manual virtual crossmatch (mVXM) consists of retrieving antibody profiles via analysis system Fusion (OneLambda) and lab management system mTilda (HLA Data Systems) along with the potential donor’s typing and matchrun via UNET (UNOS). An assessment form is then prepared which identifies DSA for recent and historical sera, cPRA cutoffs for AB, DR, DQ of 2000 and CW, DP of 4000. We aim to replace this mVXM process with the cVXM system VxMatch 2.0 (HLA Data System) which can electronically retrieve required data and produce custom paginated reports with discrete-data results to an EMR via HL7. Methods: We validated VxMatch functionality and accuracy against 60 mVXM cases which were a combination of graded survey samples (UCLA) and actual patient assessments. A comparison of the VxMatch antigen matching routine was performed to ensure that the cVXM could account for different nomenclatures, match alleles to antibodies based on resolution and serological equivalence and perform a comparison of the donor’s typing to the recipient’s antibody history to identify typing mismatches. Results: cVXM functionality and HL7 capabilities were validated and provisioned for deployment and the antigen matching routine was validated for in-lab production use. Preliminary cVXM results were found to meet the UCLA survey standard minimum of 90% concordance for identification of DSA. Conclusion: The cVXM workflow provided by VxMatch reduced assessment time and streamlined our processes by facilitating: one donor to many recipients analysis, the ability to save and resume sessions, patient screening history with antibodies previously not called per MFI criteria and risk factor review for sensitizing events, history of previous crossmatches and previous transplant mismatches. We conclude that replacing a mVXM with an EMR-integrated cVXM will reduce transcription error, save time and provide more analysis capabilities when performing VXM assessments.
P113
New Technologies, NGS and TGS
Title: A MULTIPLEX WHOLE-GENE HLA TYPING STRATEGY EVALUATION ON VARIOUS PLATFORMS: BOTH LONG READS (PACBIO, ONT), AND SHORT READS (ILLUMINA, ION TORRENT, MGI)
Authors: D. Gao, S. van Wageningen, P. van der Weele, H. de Bruin, E.H. Rozemuller, <u > M. Penning</u>, GenDx, Utrecht, NETHERLANDS|
Aim: HLA genotyping can be performed with multiple techniques. High resolution HLA typing can only be achieved by Next Generation Sequencing (NGS). However, multiple NGS platforms are available. To investigate which platforms are suitable for high resolution HLA typing, we performed a platform comparison study. Methods: HLA amplicons were generated from a 58-sample panel (GeT-RM, HLA58) from the Coriell institute. Using an 11-gene multiplex amplification, whole gene amplicons were generated for HLA-A, HLA-B, HLA-C, -DRB1, -DQB1, -DPA1, -DPB1, - DQA, -DRB3, DRB5 and for HLA-DRB4 a single amplicon containing exons 2 and 3 (NGSgo-MX11-3; GenDx). Amplicon pools were checked by gel electrophoresis and aliquoted to be used as input for library preparation for the different platforms. Libraries were generated for the following platforms; Illumina Miseq, Ion Torrent S5, Pacbio Sequel IIe and MGI DNBseq G50/G400 and ONT MinION. Data analysis was performed using NGSengine software (GenDx). Results: All platforms were capable of generating concordant HLA-typing results. In most cases high quality sequencing data was generated that could be aligned to the correct HLA loci. Here we present the comparison of the sequencing quality metrics of the different platforms, focusing on mappability, sequencing noise levels, gene coverage and phasing. We also demonstrate the promises and challenges of the different platforms for whole gene HLA sequencing. Conclusion: The Illumina Miseq, Ion Torrent S5, Pacbio Sequel IIe and MGI DNBseq G50/G400 sequencers all generated good quality sequencing data. Differences are observed in the data quality between different platforms, regarding noise levels and phasing. Overall, all platforms are proficient for high-resolution HLA typing. and selection of an NGS sequencing platform may depend mostly on other factors like sample throughput.
P114
New Technologies, NGS and TGS
Title: NGS-BASED CHIMERISM MONITORING: ANALYSIS OF 154 SAMPLE TYPINGS
Authors: P. van der Weele, J. Geerligs, J. Kooij, J. Crins, R. Welleweerd, L.A. van de Pasch, <u > M. Penning</u>, E.H. Rozemuller, GenDx, Utrecht, NETHERLANDS|
Aim: Post-transplant monitoring of the recipient chimeric state is essential for the early detection of relapse after stem cell transplantation. Performing chimerism monitoring by next-generation sequencing (NGS) allows for detection of markers at the microchimerism level, without requiring pre-transplant DNA for each monitoring experiment. Here, a novel method to facilitate chimerism monitoring by NGS (NGStrack) including software for analysis (TRKengine) is applied to a panel of 154 samples to define marker informativity and assay sensitivity. Methods: A novel method (NGStrack and TRKengine) targeting 32 hypervariable biallelic indel sites is developed and applied to genotype 154 samples from different origins. Homozygous and heterozygous typing results will be assessed and processed to identify assay detection limits. Genotyping results will be assessed for marker informativity potential using the Hardy-Weinberg equilibrium. Results: The genotyping results indicate high informativity potential for most markers. Homozygous typing results showed minimal noise levels, allowing at least 0.5% detection in 95% of cases for all markers. In many cases, sensitivity could be as low as 0.1% of the minor fraction. < br /> The average lowest allele frequency for all 32 markers combined was 0.36. Per marker, this implies a 25–30% chance of informativity. Chances of finding 2 informative markers between unrelated donors and recipients are > 99.9%. Conclusion: Based on the genotyping of 154 samples and quantifying artificial chimeric samples for 32 indel markers, NGStrack and TRKengine allow for consistent identification of informative markers with a sensitivity of at least 0.5%, potentially going as low as 0.1%. Marker informativity potential was very high, implying a high chance of identifying ≥ 2 informative markers between unrelated donors and recipients.
P115
New Technologies, NGS and TGS
Title: ALLELE-SPECIFIC QUANTIFICATION OF HLA TRANSCRIPT ISOFORMS BY NANOPORE LONG-READ SEQUENCING
Authors: <u > A.E. Hughes</u>, C. Liu, Department of Pathology and Immunology, Washington University School of Medicine, St. Louis, Missouri, UNITED STATES|M.C. Montgomery, E. Weimer, Molecular Immunology Laboratory, McLendon Clinical Laboratories, University of Nor
Aim: HLA typing plays a critical role in the evaluation of donor-recipient compatibility before hematopoietic stem cell and solid organ transplantation. While the current standard of care focuses on genomic DNA sequencing, the frequency and functional impact of HLA splice variants are not well understood. Here, we use nanopore long-read sequencing of full-length transcripts from primary human lymphocytes to determine the repertoire of HLA splice variants in a cohort of healthy donors. Methods: cDNA libraries were prepared from mRNA extracted from peripheral lymphocytes from 12 healthy donors (21 libraries total) and sequenced on MinION flow cells (R9.4.1) using MinKNOW (v3.1.13) to a median of 1.6x10 < sup > 6</sup > reads per library. Processed reads were aligned to the IPD-IMGT/HLA database (v3.41.0) using minimap2 (v2.12). Reads that mapped to individual HLA loci were realigned to allele-specific genomic references using HLA types determined by Athlon (v1.0) or Illumina sequencing. Alleles were excluded from further analysis if any exon was covered by<20 (class I) or 10 (class II) reads (24 of 330 alleles). Transcript isoforms were quantified by calculating the proportion of reads containing known exons. Results: Splice variants were uncommon among class I HLA genes (Figure 1), which had median exon retention rates ranging from 99% to 100%, except for exon 5 of < i > HLA-C</i>, which was spliced out of up to 15% of reads. Similarly, splice variants were uncommon among class II genes except for < i > HLA-DQB1 </i>(Figure 1). Exon 5 of < i > HLA-DQB1 </i > was spliced out of 90% to 100% of reads for 31 alleles with non-functional splice acceptor sites within intron 4, which is consistent with previous work. Interestingly, for the other 10 < i > HLA-DQB1</i > alleles with intact splice acceptor sites (e.g., DQB1*05:03), exon 5 was also spliced out of 8% to 44% of reads. Conclusion: Nanopore cDNA sequencing yields reads spanning full-length transcripts, which can be used to identify and quantify HLA splice variants, including known allele-specific variants involving exon 5 of < i > HLA-DQB1</i > . Further work is needed to validate novel isoforms and characterize the landscape of HLA transcripts in a larger cohort including broader tissue types.
Figure 1: Rate of allele-specific exon retention quantified by nanopore long-read sequencing. Open circles represent the proportion of reads containing known exons for each allele in each sample. Lines indicate medians.
P116
New Technologies, Bioinformatics
Title: PREDICTING HLA SEROLOGIC SPECIFICITIES WITH RANDOM FOREST MACHINE LEARNING MODELS
Authors: <u > D.G. Biagini</u>, L. Gragert, Pathology and Laboratory Medicine, Tulane University School of Medicine, River Ridge, Louisiana, UNITED STATES|M. Maiers, National Marrow Donor Program / Be The Match, Minneapolis, Minnesota, UNITED STATES|
Aim: To assess HLA compatibility in virtual crossmatch, a donor’s HLA specificities are compared to a patient’s anti-HLA antibody specificities. Compatibility assessments are complicated by lack of experimentally determined serology for many HLA alleles. Single-antigen bead antibody assays also have only a limited panel of HLA alleles. Even in the era of molecular typing for HLA, for virtual crossmatch cases there remains a need to predict serological categories for undefined alleles. A neural network (NN) was developed to predict serologic specificities as part of the HLA Dictionary efforts. As this NN was built with a now obsolete software library, our aim is to apply a new machine learning method that could yield human-interpretable predictions. Methods: For each serological category within each locus (A, B, C, DQB1, DRB1), we have created a random forest classifier using the Python scikit-learn package to predict assignments of alleles based on amino acid sequences from the IPD-IMGT/HLA database. The random forest is trained with the combined training and validation data of the original NN – a selection of expert-curated alleles that are representative of each serological category. Evaluation of our model compares predictions for a set of alleles that were experimentally characterized by serologic methods, as catalogued in the 2008 HLA Dictionary. We also compare predictions on a set of alleles for concordance between both models. Results: The random forest predicted serology with 85%+ concordance to the original NN on the concordance set across all loci, and had equal or slightly improved accuracy to the original NN on the accuracy set across all loci (Table 1). The NN was also replicated in a modern framework, PyTorch, achieving similar performance. Conclusion: In future work we will employ Local Interpretable Model-agnostic Explanations (LIME), to understand the rules leading to different predictions from the random forest. Extracting decision trees with LIME will provide insight into the basis for serological assignments, which will help to improve serological classification. We have developed a new query of the NMDP registry to identify locus typings assessed by both molecular and serologic methods, which will greatly expand our validation set. Based on imputation results, subjects with ambiguous molecular typings likely to have rare alleles could be selected for typing by NGS.
Figure 1. A theoretical example of a single decision tree in a random forest. In this case, the decision tree illustrates a set of amino acids that could distinguish a A2 antigen from other antigens.
P117
New Technologies, NGS and TGS
Title: EVALUATION OF NGS-BASED POST TRANSPLANT ENGRAFTMENT MONITORING
Authors: <u > L. chi</u>, P. Arnold, St. Jude Children's Research Hospital, Memphis, Tennessee, UNITED STATES|E. Gomes, C. Lind, CareDx, South San Francisco, California, UNITED STATES|
Aim: Monitoring the engraftment of donor cells in a recipient after hematopoietic cell transplantation (HCT) is vital for assessing the success of a transplant. Early detection of mixed chimerism may significantly impact treatment aimed at preventing relapse; thus, the sensitivity and accuracy of chimerism assessment become critically important. Chimerism by short tandem repeats (STR) is the gold standard but lacks sensitivity. Quantitative PCR (qPCR) is very sensitive but lacks accuracy. Here we evaluate AlloSeq HCT, an NGS-based reagent and software system that targets 202 single nucleotide polymorphisms (SNPs) to quantitate the amount of donor and recipient-derived DNA present in a post-transplant sample. Methods: Each AlloSeqHCT reagent kit contains all the reagents for multiplex amplification of 202 SNP loci across 24 different samples. Sequencer-ready amplicon is generated in a single amplification step. After sample library pooling and clean-up, sequencing is performed by the Illumina MiSeq. Automated analysis of the sequencing reads in the AlloSeq HCT software uses a proprietary algorithm to determine the contribution of recipient and up to 2 donor genomes in a sample. Results: Mixed chimerism in artificial dilutions was detected at levels as low as 0.2%, making NGS chimerism less sensitive than qPCR, but more sensitive than STR. Proficiency samples tested by NGS were highly concordant (R2 > 0.99) with mean consensus values, despite reflecting a majority of laboratories using STR. Comparisons of post-transplant patient samples tested by NGS to STR and qPCR in the expected sensitivity ranges of the individual methods showed close correlation, confirming accuracy and sensitivity in clinical samples. Intra-assay and inter-assay NGS chimerism results revealed high precision, with mean coefficient of variation (CV) <1% within the reportable range. On average, the number of informative markers in the post samples tested was 106 per sample, a number much higher than analyzed by conventional chimerism methods. Conclusion: The AlloSeq HCT reagent and software analysis system offer highly sensitive, accurate, and precise NGS-based chimerism quantitation over a large range of recipient and donor percentages, not possible with STR or qPCR methods alone. Simple workflow and automated analysis make this method a viable option for labs who desire to transition to NGS for engraftment monitoring.
P118
New Technologies, NGS and TGS
Title: UNIQUE MOLECULAR IDENTIFIER ENHANCED HLA TYPING AND TRANSCRIPT QUANTITATION USING NANOPORE TECHNOLOGY
Authors: <u > E. Weimer</u>, Pathology & Laboratory Medicine, UNC Chapel Hill, Chapel Hill, North Carolina, UNITED STATES|M.C. Montgomery, Molecular Immunology Laboratory, UNC Health, Chapel Hill, North Carolina, UNITED STATES|C. Liu, Pathology & Immunology, Washing
Aim: There is increasing evidence on the importance of HLA expression on transplant outcomes. However, there isn’t a good tool for determining HLA expression within the time constraints for deceased donor kidney allocation. Our group developed an assay for HLA typing and transcript quantitation. Methods: Total RNA was extracted from thirteen samples and first-strand reverse transcription was performed. Unique molecular identifiers (UMI) were added to HLA-specific cDNA during a two-cycle PCR. Additional PCRs were performed to enrich the HLA regions. Libraries were prepared using the native ligation kit and sequenced using R10.3 flowcells with high-accuracy basecalling. Sequence analysis involved adapter and barcode trimming followed by UMI grouping. Consensus sequences were generated using Medaka and HLA typing was performed at 2-field level Athlon2. UMI counts were log2 normalized for the sample and locus UMI count as well as sequencing depth. HLA transcript expression quantitation was based on normalized UMI counts. Results: UMI groups of 1 (min1) or 10 (min10) reads were evaluated. Using min10 generated a median consensus accuracy of 99.9% compared to 93.0% for min1. While the read accuracy was higher with min10, min1 consensus accuracy ranged from 89 to 95%. Min1 reads also enabled simultaneous PCR deduplication and consensus sequence generation. < br /> <br /> Using an average of 96,000 reads, 98.3% (227/231) HLA alleles were accurately typed. The alleles incorrectly typed were 2 were < i > DQA1</i>, 1 was < i > HLA-A</i>, and 1 was < i > DRB3</i > . The discordant < i > DRB3 </i > allele was within the same P group as the reference typing. < br /> <br /> Since the number of unique HLA alleles is low, HLA transcript counts were based on HLA antigen groups. Not surprisingly, HLA class I antigens were expressed 1.2-fold higher than class II. The rank order of expression was < i > A</i≫<i > B</i>=<i > C</i > . The highest class II loci were < i > DRB1 </i > and < i > DPB1</i > . Finally, the entire assay can be completed within approximately 7 h. Conclusion: We have described a novel assay that provides accurate HLA typing and transcript quantitation. The majority of the typing was accurate; however, the 3 inaccurate typing results were from a likely sample contamination. Importantly, the observed HLA expression was consistent with published literature and represents a new tool for assessing the impact of HLA expression. More studies are needed to understand the role of HLA transcript expression on transplant outcomes.
P119
New Technologies, Population genetics
Title: STEM CELL DONOR-DERIVED CPRA IMPROVES ACCESS FOR HIGHLY-SENSITIZED CANDIDATES
Authors: <u > K. Lindblad</u>, D. Stewart, United Network for Organ Sharing, Richmond, Virginia, UNITED STATES|L. Gragert, Tulane University School of Medicine, New Orleans, Louisiana, UNITED STATES|
Aim: The OPTN Histocompatibility Committee proposes replacing the CPRA metric used in organ allocation, which is based on HLA haplotype frequencies from ∼ 14,000 deceased kidney donors, with a genotype-based CPRA derived from over ten million stem cell donors. We examined the impact changing the CPRA calculation would have on kidney candidates’ CPRA, as well as the effect of changing from a calculation based on four ethnic groups to one using seven ethnic groups. Methods: We used OPTN data on unacceptable antigens (UAs) for 98,455 kidney candidates waiting on December 31, 2020 to calculate CPRA three ways: using deceased-donor frequencies and the current OPTN calculation; using genotype frequencies based on stem cell donors and four ethnic groups; and using genotype frequencies and seven ethnic groups. Results: While CPRA changed little for the majority of candidates (Fig 1), it increased up to 97% for those with UAs against loci (e.g. DPB) and alleles (e.g. A*24:03) not in the current CPRA. Many candidates would move into the highest sensitization categories: 271 to 98%, 384 to 99%, and 609 to 100% under the CPRA using seven ethnic groups. The maximum difference in CPRA between the four and seven ethnic group metrics for any one candidate was 0.39% (Fig 2). Conclusion: Switching to the proposed metric would have little impact for the majority of kidney candidates, but would allow those with UAs currently lacking frequencies to receive allocation points appropriate to their level of sensitization. Under the proposed metric, over 1,000 candidates would move into the highest CPRA allocation categories and receive greater access to transplant. Additionally, the similarity of the four- and seven-ethnic-category CPRAs indicates that there would be no substantial drawback to using a CPRA including frequencies from a more diverse population of donors.
P120
New Technologies, Population genetics
Title: STEM CELL DONOR-DERIVED CPRA BETTER PREDICTS TRANSPLANT ACCESS DISPARITIES THAN CURRENT CPRA
Authors: <u > K. Lindblad</u>, D. Stewart, United Network for Organ Sharing, Richmond, Virginia, UNITED STATES|L. Gragert, Tulane University School of Medicine, New Orleans, Louisiana, UNITED STATES|
Aim: The OPTN Histocompatibility Committee proposes replacing the CPRA metric used in organ allocation, which is based on HLA haplotype frequencies from ∼ 14,000 deceased kidney donors, with a genotype-based CPRA derived from over ten million stem cell donors. We explored whether the proposed CPRA would more accurately reflect transplant access disparities among kidney candidates. Methods: We used OPTN unacceptable antigen (UA) and offer data for all kidney candidates ever waiting 1/1/2018–12/31/2020. Within each kidney CPRA allocation category we fit Poisson models predicting offer rate based on wait time and one of three CPRA metrics: the current OPTN CPRA or the proposed CPRA using ethnic weights for either four or seven ethnic groups. Model fits were evaluated based on the AIC, with lower AIC indicating that the model better described the data. Results: The models using the proposed CPRA had slightly better AICs than those using the current CPRA for all categories except 100% (Fig 1). When considering only candidates with UAs against loci included in the proposed CPRA but not the current CPRA (e.g. DPB), the AIC for the proposed CPRA was better in all categories (Fig 2). There was little difference between the metric that used four ethnic groups and the metric that used seven ethnic groups. Conclusion: The proposed CPRA is more predictive of offer rates than the current CPRA for most kidney candidates and is more predictive for all candidates with UAs against loci not included in the current CPRA. Offer rate indicates level of transplant access; the proposed CPRA therefore more accurately reflects candidates’ access to transplant and is more appropriate for use in allocation than the current CPRA. A more predictive CPRA also provides a stronger basis for developing new allocation point scales based on sensitization, such as the CPRA scale proposed in the OPTN Lung Committee’s continuous distribution project.