Abstract
Introduction
Little is known about the underlying biomechanical cause of low back pain (LBP). Recently, technological advances have made it possible to quantify biomechanical and neurophysiological measurements, potentially relevant factors in understanding LBP etiology. However, few studies have explored the relation between these factors. This study aims to quantify the correlation between biomechanical and neurophysiological outcomes in non-specific LBP and examine whether these correlations differ when considered regionally vs. segmentally.
Methods
This is a secondary cross-sectional analysis of 132 participants with persistent non-specific LBP. Biomechanical data included spinal stiffness (global stiffness) measured by a rolling indenter. Neurophysiological data included pain sensitivity (pressure pain threshold and heat pain threshold) measured by a pressure algometer and a thermode. Correlations were tested using Pearson’s product-moment correlation or Spearman’s rank correlation as appropriate. The association between these outcomes and the segmental level was tested using ANOVA with post-hoc Tukey corrected comparisons.
Results
A moderate positive correlation was found between spinal stiffness and pressure pain threshold, i.e., high degrees of stiffness were associated with high pressure pain thresholds. The correlation between spinal stiffness and heat pain threshold was poor and not statistically significant. Aside from a statistically significant minor association between the lower and the upper lumbar segments and stiffness, no other segmental relation was shown.
Conclusions
The moderate correlation between spinal stiffness and mechanical pain sensitivity was the opposite of expected, meaning higher degrees of stiffness was associated with higher pressure pain thresholds. No clinically relevant segmental association existed.
Keywords: Low back pain, Mechanics, Hyperalgesia, Central sensitization
Introduction
The lumbar spine is a complex anatomical structure, the chief function of which is biomechanical—to bear loads through various static and dynamic functions and provide protection for soft neural tissue [1]. However, it is not apparent from patient history, clinical examination, or diagnostic imaging when perturbations in biomechanical function are causal factors for developing low back pain (LBP), when they result from LBP, and when they are simply irrelevant normal variants [2]. Hence, LBP is often considered non-specific. In fact, only around 5–10% of LBP can be attributed to an explicit patho-anatomical issue. In the remaining 90–95%, there is no apparent structural issue [3]. In a clinical setting among manual therapy providers, it is common to attribute such non-specific LBP to permutations in biomechanical function, i.e., as a causal factor, although the evidence is lacking [2, 4, 5].
Much research has been conducted to understand LBP’s etiology better, but progress has been limited [6–8]. Arguably, this is the result of limitations in measurement, methodology, and population sampling. In measurement, recent technological advances have made it possible to collect data in new areas, which could shed light on LBP’s underlying causes. In particular, the development of new technologies to quantify spinal stiffness non-invasively [9], thus better quantifying what is thought to be an influential clinical factor in LBP [10]. Spinal stiffness has shown promise in that it may be associated with treatment-induced disability improvements. E.g., in patients with LBP, those who have immediate reductions in spinal stiffness after spinal manipulation have improvements in disability (≥ 30% reduction in The Oswestry Disability Index), and this change in stiffness does not occur for those who do not have improvements in disability [11, 12].
In parallel to exploring the mechanical aspect of LBP, researchers are also exploring the underlying mechanisms of the pain experience itself. This includes quantitative sensory testing (QST), which quantifies individual pain perception in response to controlled noxious stimuli [13]. Such experimental tests can differentiate LBP patients from healthy controls, and perturbations in pain modulation (sensitization of the somatosensory system e.g., leading to decreases of pain thresholds) appear to manifest in the sub-acute stage as pain turns persistent [14]. It is of interest that these perturbations, in a commonly noted mechanical syndrome such as LBP, extend beyond deep muscle mechanical pain sensitivity, i.e., pressure pain, to superficial skin measures, e.g., heat pain. Nevertheless, apparent differences have previously been reported for both these measurements between LBP patients and healthy controls [15, 16].
Furthermore, each new technology allows researchers to explore these properties at the segmental level [9, 15], an important consideration considering that back pain is often thought to be localized to specific anatomic areas pertaining to a given segmental level. While biomechanical testing has been used to evaluate the spine’s primary biomechanical function, and experimental QST has done the same for neurophysiological function, it seems unlikely that the two are not interconnected. We put forward that another factor that may hinder our understanding of LBP etiology is artificial segregation of biomechanics and neurophysiology. These two aspects of LBP are often discussed, studied, and treated in research and clinical settings as if they are distinct phenomena, when in reality, they may well be interconnected [17]. Studying biomechanical and neurophysiological systems together may provide important information to understand the etiology of LBP better. This is particularly important in the field of manual therapy, where the treatment site is often determined using a mixture of stiffness and pain locations [17]. However, we know little about the segmental interplay between stiffness and pain sensitivity.
Hence, this study will use an experimental test setup that mimics clinical practice to investigate the relationship between lumbar stiffness and mechanical and non-mechanical QSTs, two measurements that appear to correlate with each other [18]. Yet, whether a correlation also exists with mechanical spinal stiffness is unknown. Furthermore, all three experimental tests appear to be affected following manual therapy [12, 19, 20].
Therefore, the specific aims of this investigation are to (1) quantify the correlation between biomechanical and neurophysiological measurements (global stiffness, pressure pain threshold, and heat pain threshold) in LBP patients, and (2) examine if these correlations differ when considered regionally (the lumbar back) or segmentally (e.g., L4).
From a clinical standpoint, we hypothesize that stiffness and pain sensitivity are negatively associated, i.e., high degrees of stiffness and low pain threshold are correlated, and that this association may be greater for (1) a mechanical stimuli as opposed to a thermal stimuli and (2) segmentally versus regionally.
Methods
Design
Our study design was a secondary observational cross-sectional analysis of baseline data from a randomized trial of participants with persistent non-specific LBP [21]. The study was approved by the Regional Committee on Health Research Ethics for Southern Denmark (S-20160201). The manuscript was prepared in reference to the STROBE format.
Setting
A population sample of patients seen at the Spine Center of Southern Denmark, a public, regional hospital department specializing in spinal pain, was recruited consecutively between November 2017 and February 2019.
Participants
Participants were included based on a diagnosis of non-specific LBP from a clinician at the Spine Center. A total of 132 participants were included using the following criteria [21]:
Persistent non-specific LBP.
No surgical indication or previous spinal surgery.
Daily oral opioid intake was limited to 40 mg of morphine at the time of inclusion.
Body mass index under 35 kg/m2.
Age between 18 and 60 years old.
Procedure
All testing was done at the Spine Center by one rater who gained experience with the test procedures through practical laboratory training, including pilot testing on 20 participants with persistent LBP not included in the present study.
The baseline test session was initiated by identifying each lumbar segment. Each spine process from S1 to T12 was marked superficially with a marker with the participant in the prone position. The segment identification was confirmed using ultrasonography (Sonosite Titan Linear, L38 probe) [22].
Afterward, we completed the protocol in the following standardized order for all participants, deep mechanical pain sensitivity, superficial thermal pain sensitivity, and spinal stiffness with sufficient rest time of approximately two minutes between each procedure, limiting potential interactions. Each procedure is described below in greater detail.
Spinal stiffness
Spinal stiffness was tested using the VerteTrack (VT). The device consists of two weighted probe wheels (3 cm apart) that slowly rolls along the lumbar paravertebrales guided by the surface markings of spinous process locations with the subject in a prone position. The landing site is centered around the S1 spine process and the lifting site at the T12 spine process. The resulting posterior-to-anterior displacement during rolling is measured by a string potentiometer, which can then be quantified as stiffness (applied mass/displacement or N/mm) with a sampling rate of 30 Hz. This process is then repeated with increasing loads of 10 N up to a maximum of 60 N. Before testing, each participant was instructed to exhale and hold their breath at around the residual air volume while completely relaxing their muscle until the trial was complete. If pain or discomfort were elicited, the procedure would be repeated one more time, and if the trial continued to produce discomfort, the procedure was discontinued. Only trials with no discomfort were used for the analysis as trials with discomfort could lead to muscle guarding and erroneous stiffness measures. The VT is a novel experimental device reported as comfortable and safe [23], has good reliability in asymptomatic subjects [9], and demonstrates high accuracy under bench-top conditions [24]—suggesting that the VT can be used to assess spinal stiffness in-vivo.
Deep mechanical pain sensitivity
Deep mechanical pain sensitivity was determined using a pressure algometer (Model 2, Somedic, Sweden) with a custom-made double-headed probe (2 × 1 cm2, 3 cm apart), which allowed for bilateral pressure at either side of the mid-line for each lumbar segment. The pressure was increased gradually with an approximate rate of 50 kPa/s until the participant reported the pressure as painful by pressing an indicator button. If no pain had been elicited by 1000 kPa, the test was discontinued, and 1000 kPa was recorded as the pressure pain threshold (PPT). The pressure algometer has excellent intra-rater reliability for patients with LBP [25].
Superficial thermal pain sensitivity
Superficial thermal pain sensitivity was assessed using a handheld thermode (Medoc TSA-II, Israel) with a single 30 × 30 mm probe placed in the midline for each lumbar segment centered at the spinous process ensuring complete contact between skin surface and probe. The baseline temperature was pre-set to 32 degrees Celsius (C). During testing, the temperature increased at a rate of 1 C/s until the participant reported the temperature as painful by pressing an indicator button. Maximum temperature was pre-set at 50 C, and if no pain had been elicited by then, this was recorded as the heat pain threshold (HPT). Using the thermode to indicate the HPT has good-to-excellent intra-rater reliability when tested at the spine of asymptomatic volunteers [26].
All QST tests (PPT and HPT) were performed, with the patient in the prone position, at each spinal segment three times. The segments were tested in a pre-determined, computer-generated, random order with 10-s rest intervals between each test. If no pain had been elicited by 1000 kPa after the first two trials, a third trial was not performed for PPT. Before data collection, one or two test trials of each pain threshold assessment were performed at the lower extremity and one test at the T12 segment to familiarize participants with the procedure.
Variables of interest
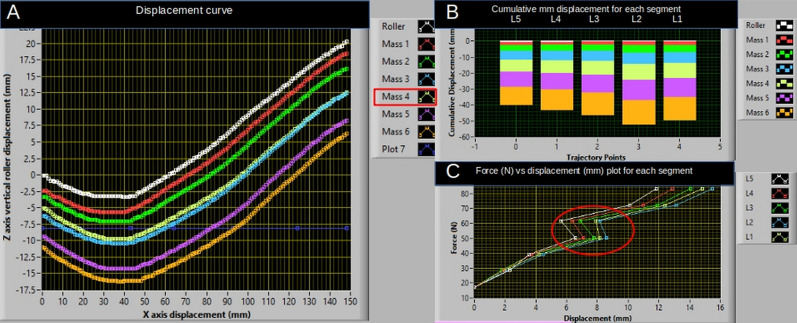
Lumbar stiffness: The VT data were smoothed (SVD algorithm, polynomial order of 2, least-squares method, and tolerance of 0.0001) and visualized (Labview 15.0f3 for Windows 10, National Instruments, Texas, USA) before being exported to a spreadsheet (LibreOffice, vers. 7, for Ubuntu 18.04) for further analysis. Global lumbar stiffness (GS) indicated stiffness throughout the available load and was calculated as the slope of force–displacement (N/mm) from the second load to the second highest load. Hence, the terminal loads were removed (e.g., 0 N and 60 N). As part of the data analysis, a subjective inspection of the smoothed data was completed before extracting the data. Some loads within the participant trial were affected by factors such as breathing, muscle guarding, or technical errors and were omitted. This process was guided by visual inspection of the displacement curve corresponding to each load. See Fig. 1 for a LabView output and an example of a removed trial. Global stiffness is a continuous parameter ranging from 0 to and was recorded for each segment [L1–L5].
Fig. 1.
An example of the LabView output. A The displacement curve, the y-axis represent vertical displacement for each trajectory point along the lumbar lordosis (x-axis). Each load trajectory is presented as a unique color (each trial with a different load). B The cumulative displacement (mm) for each segment across loads. C The force (N) versus displacement curve (mm) for each segment. In this example, we would omit the light-yellow 40 N line (mass 4) as the displacement recorded was less than that recorded with 30 N (mass 3), suggesting muscle guarding. This is also illustrated by the skewness of the plot in (C) (red circle). After data cleaning, the stiffest segment was L5 with a global stiffness score of 5.5, and the least stiff segment was L2 with a global stiffness score of 4.2 (B)
Deep muscle pain sensitivity: Pressure pain threshold is a continuous parameter ranging from 0 to 1000 kPa and was calculated for each segment [L1–L5] as the average of three trials.
Superficial skin pain sensitivity: Heat pain threshold is a continuous parameter ranging from 32 to 50 C and was calculated for each segment [L1–L5] as the average of three trials.
Statistical analysis
Descriptive statistics
Descriptive data, including the demographics and all outcomes, are presented as means, medians, standard deviations, and interquartile ranges. Normal distribution for each outcome was visualized using density and QQ-plots. Visual inspection for skewness and data shape was further conducted [27].
Correlation
The correlation between the three outcomes is presented visually as Loess slopes [28] plotted for each segment. Pearson’s product-moment correlation (ρ) was used for parametric data, and Spearman’s rank correlation (Rs) was used for non-parametric data. We omitted individual participants if they did not have data for both the parameters in question. The strength of the correlations was evaluated as poor (< 0.30), moderate (0.30 ≤ 0.50), good (0.51 ≤ 0.70) and strong (> 0.70) [29]. All correlations were examined as a single summarized value for all segments for each participant and individually for each segment. A p value < 0.05 was considered to be significant.
Segmental statistics
Segmental data are depicted as means and 95% confidence intervals. The association between outcomes was tested using a one-way analysis with the outcomes as the dependent variable and segment as the independent variable. The assumptions for the ANOVA were tested for (1) normality by plotting the residuals against predicted values and (2) homogeneity of variances using Levene’s test. A p value of less than 0.1 would indicate further post-hoc testing using Tukey multiple pairwise comparisons to investigate between-segment differences. The results for each outcome are presented as F-statistics, between-segment difference and adjusted p values.
Data analyses were completed using R [30], for Linux, v. 4.0 with R-studio v. 1.4. Data cleaning were performed using the Tidyverse [31].
Results
Participants characteristics
Of the 132 participants included, complete experimental baseline data were available for 128 (3 participants had missing HPT data, 1 participant had incomplete GS data). The sample consisted of 72 (55%) males. Complete demographic data are presented in Table 1.
Table 1.
A demographic profile of the 132 participants included in the analysis
| Parameter | Mean | SD | Median | IQR |
|---|---|---|---|---|
| Age | 45.1 | 9.7 | 46.0 | 14.2 |
| Body Mass Index | 26.2 | 3.9 | 25.6 | 4.9 |
| Low back pain intensity | 5.6 | 1.8 | 5.5 | 2.7 |
| Oswestry disability index | 27.8 | 11.6 | 27.3 | 18.5 |
| Pain duration (months) | 48.2 | 78.1 | 14.8 | 53.9 |
All data were normally distributed except for PPT. None of the variables were visually skewed and overall had consistent data shape across outcomes. All experimental outcomes are presented in Table 2 as a single average score for all segments and each segment. None of the participants reached the maximum of 1000 kPa or 50 °C.
Table 2.
The regional experimental outcome measurements of the 132 participants included in the analysis
| Outcome | Segment | Mean | SD | Median | IQR |
|---|---|---|---|---|---|
| Global stiffness | All | 4.14 | 0.88 | 4.05 | 1.13 |
| L1 | 4.01 | 0.88 | 3.89 | 0.94 | |
| L2 | 3.97 | 0.86 | 3.90 | 0.97 | |
| L3 | 4.03 | 0.84 | 3.93 | 0.92 | |
| L4 | 4.20 | 0.85 | 4.11 | 1.06 | |
| L5 | 4.48 | 0.90 | 4.40 | 1.07 | |
| Pressure pain threshold | All | 475 | 231 | 446 | 363 |
| L1 | 522 | 244 | 523 | 369 | |
| L2 | 482 | 228 | 465 | 357 | |
| L3 | 472 | 225 | 452 | 376 | |
| L4 | 455 | 225 | 437 | 346 | |
| L5 | 445 | 229 | 401 | 338 | |
| Heat pain threshold | All | 42.2 | 3.7 | 42.0 | 5.6 |
| L1 | 42.5 | 3.6 | 42.4 | 5.3 | |
| L2 | 42.3 | 3.8 | 42.4 | 6.3 | |
| L3 | 42.0 | 3.6 | 41.3 | 5.3 | |
| L4 | 42.1 | 3.8 | 41.6 | 5.6 | |
| L5 | 42.0 | 3.6 | 41.8 | 5.7 |
Correlation data
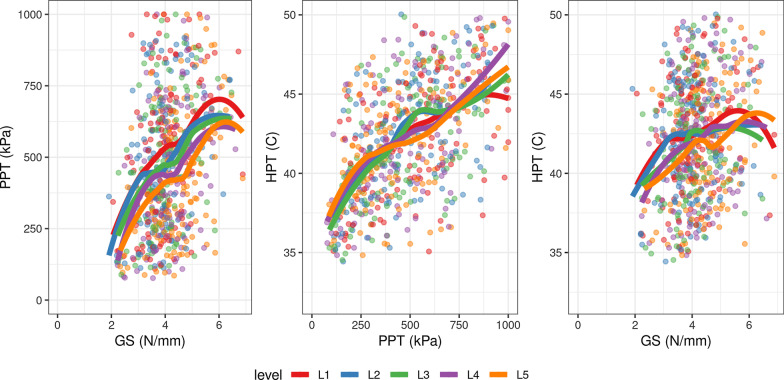
The relationship between segments and each outcome is depicted in Fig. 2. Visually the correlation does not differentiate between segments.
Fig. 2.
Correlation between lumbar stiffness, mechanical pain sensitivity, and superficial pain sensitivity, presented as Loess slopes for each segment. GS global stiffness, PPT pressure pain threshold, HPT heat pain threshold
Table 3 lists the correlations and p values for all outcomes. All correlations were statistically significant, except for the correlation between GS and HPT at the L2 segment (p value = 0.12). All correlations had a positive direction. The correlation between GS and PPT for all segments was moderate (ρ = 0.38). Conversely, the correlation between GS and HPT was poor (Rs = 0.23) and not significant. The correlations between PPT and HPT were good for all segments (ρ = 0.53).
Table 3.
Correlation for all and every segment between lumbar stiffness, pressure pain, and superficial pain sensitivity
| Global stiffness versus Pressure pain threshold | Global stiffness versus Heat pain threshold | Pressure pain threshold versus Heat pain threshold | ||||
|---|---|---|---|---|---|---|
| Segment | Rs | p value | ρ | p value | Rs | p value |
| All | 0.38 | < 0.01 | 0.23 | < 0.05 | 0.58 | < 0.01 |
| L1 | 0.36 | < 0.01 | 0.19 | 0.03 | 0.53 | < 0.01 |
| L2 | 0.33 | < 0.01 | 0.14 | 0.12 | 0.55 | < 0.01 |
| L3 | 0.34 | < 0.01 | 0.21 | < 0.05 | 0.57 | < 0.01 |
| L4 | 0.33 | < 0.01 | 0.22 | < 0.05 | 0.56 | < 0.01 |
| L5 | 0.38 | < 0.01 | 0.28 | < 0.01 | 0.50 | < 0.01 |
Presented as chi2 scores and p values
Rs = Spearman’s rank correlation, ρ = Pearson’s product-moment correlation
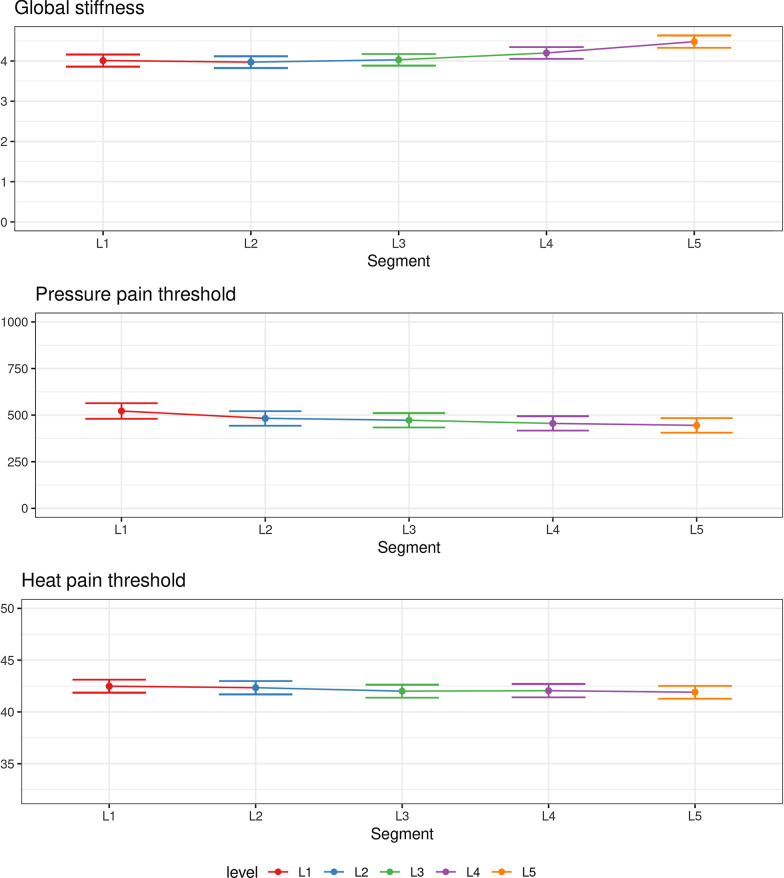
Segmental data
All outcomes are presented visually in Fig. 3. A segmental pattern can be observed from L1 to L5. Global stiffness increased while PPT and HPT decreased caudally. This indicates that L5 is the stiffest segment and the most sensitive segment for both pressure and heat pain thresholds. The between-segment difference in GS and HPT for the upper segments is almost negligible. All model assumptions were upheld for the ANOVA. The ANOVA revealed the following: There was a statistically significant effect of spinal segmental level on GS (F4,650 = 7.7, p < 0.01). Whereas, we found no significant effect on PPT (F4,655 = 2.2, p = 0.07) or HPT (F4,640 = 7.7, p = 0.68). This indicates that a statistically significant association was apparent for GS, while PPT had a p value of just above 0.05. Heat pain threshold was clearly not significant and exceeded the cut point (p value = 0.1). Post-hoc testing of GS and PPT were, consequently, indicated.
Fig. 3.
The segmental value for lumbar stiffness (global stiffness—N/mm), deep pressure pain (pressure pain threshold—kPa), and superficial pain (heat pain threshold—°C). Connected means with error bars indicating the 95% confidence interval
For GS, a significant adjusted difference was observed in the Tukey multiple comparisons for L5 against L1, L2, and L3. No segments differed significantly for the PPT scores when considering the adjusted scores. However, as Fig. 3 demonstrates, L5 and L4 have the lowest PPT scores compared to L1. All values are available in Table 4.
Table 4.
Post hoc Tukey comparison after ANOVA testing of lumbar stiffness and pressure pain threshold between each segment
| Segment | Global stiffness | Pressure pain threshold | ||
|---|---|---|---|---|
| Difference (95% CI) | Adj. p value | Difference (95% CI) | Adj. p value | |
| L2–L1 | − 0.04 (− 0.33 to 0.25) | ~ 1 | − 39.5 (− 117.2 to 38.3) | 0.63 |
| L3–L1 | 0.02 (− 0.27 to 0.31) | ~ 1 | − 49.6 (− 127.3 to 28.2) | 0.41 |
| L4–L1 | 0.19 (− 0.10 to 0.48) | 0.40 | − 66.7 (− 144.5 to 11.0) | 0.13 |
| L5–L1 | 0.47 (0.18 to 0.76) | < 0.01 | − 77.0 (− 154.8 to 0.7) | 0.05 |
| L3–L2 | 0.06 (− 0.23 to 0.35) | 0.98 | − 10.10 (− 87.9 to 67.7) | ~ 1 |
| L4–L2 | 0.23 (− 0.06 to 0.52) | 0.21 | − 27.24 (− 105.0 to 50.5) | 0.87 |
| L5–L2 | 0.51 (0.22 to 0.80) | < 0.01 | − 37.6 (− 115.3 to 40.2) | 0.68 |
| L4–L3 | 0.17 (− 0.12 to 0.46) | 0.50 | − 17.14 (− 94.9 to 60.6) | 0.97 |
| L5–L3 | 0.45 (0.16 to 0.74) | < 0.01 | − 27.46 (− 105.2 to 50.3) | 0.87 |
| L5–L4 | 0.28 (− 0.01 to 0.57) | 0.07 | − 10.32 (− 88.1 to 67.4) | ~ 1 |
Bold indicates a significant adjusted statistical difference between segments
Discussion
Summary of the results
Our results demonstrated a moderate positive correlation between GS and PPT; higher stiffness scores were associated with higher deep mechanical pain thresholds. In contrast, the correlation between GS and HPT was poor, while, as expected, a good correlation was observed between deep and superficial pain thresholds. When examining stiffness and pain sensitivity across segments, we only observed a statistically significant difference in stiffness between higher and lower lumbar segments.
Correlation findings
Surprisingly, the correlation observed between spinal stiffness and pressure pain threshold was opposite than expected: Participants with higher degrees of spinal stiffness also had higher pressure pain thresholds (i.e., lower pain sensitivity). Three different postulations could view this relation: (1) The increased lumbar stiffness might be explained as part of an adaptive mechanical protection system [32] that decreases nociceptive activity in the lumbar region and therefore increases PPT. This is consistent with similar research. When inducing pain at the low back in two asymptomatic populations, higher degrees of stiffness were observed [33], whereas the PPT score did not change [34]. Possibly, and opposed to pain sensitivity, stiffness could be viewed as a continuum where high degrees of stiffness can be advantageous for the locomotor system. (2) This could be reversed, so higher pain thresholds increase stiffness again as a protective adaption. (3) Possibly, as a perceptual influence—a stiffer spine may be perceived as more resilient to applied forces. Given that pain is considered a protective response, a stiffer spine might require less protection, resulting in an increased ability to tolerate force (i.e., higher PPTs). In contrast, in a less stiff spine, the spine may be perceived as less resilient to applied forces, with low PPT scores through psychological mediation (increased protection needed). However, the authors are not aware of any research that has investigated this previously for LBP. Nevertheless, there is evidence of this connection between mind and body in prior work from other fields [35].
A good correlation observed between PPT and HPT is congruent with previous findings [18]. This correlation may simply reflect shared modulation of both central as well as peripheral pain mechanisms. This is an interesting finding as these measurements differ within the aspect of pain processing. A mechanical pressure involves activating deep tissue afferent fibers and thermal stimuli involving peripheral skin activation [36].
Segmental findings
Figure 3 revealed an apparent minor association between all the outcomes, which reached statistical significance for lumbar stiffness. When comparing outcomes between segments, a difference in mean stiffness was observed between the higher and the lower lumbar segments. However, the largest mean difference in stiffness observed was between L5 and L2 and corresponded to 11%, which is only marginally higher than the mean manual detectable threshold of change in stiffness at 8% [37]. The small difference between the largest values suggests it is nearly impossible to palpate differences in stiffness between closer or adjacent segments. Furthermore, this difference is possibly even smaller as the current analysis did not consider the standard error of measurement for the VT [38].
For the QST, no differences were found between segments. This is consistent with a prior study conducted at our laboratory [15]. Arguably, this is due to the sample’s chronicity, indicating that the original nociceptive input has developed into a generalized peripheral sensitization [39]. Furthermore, changes at the supra-spinal level could also lead to the generalized effect observed for both thresholds. Current research indicates that persistent LBP patients often have perturbations in neurological mapping of the somatosensory system and cortical homunculus or “cortical smudging” [40]. Potentially, such cortical smudging could lead to difficulty identifying the different stimuli at the nearby segments. This argument is supported by previous findings of distorted body images [41] and difficulty identifying the midline of the trunk under painful sensorimotor manipulation in persistent LBP patients [42]. Also, another study investigated the neural activity of the hemisphere, and when testing PPT at L1 and L5 segments in healthy subjects, an activity overlap of 76% was observed at the right hemisphere and 59% at the left hemisphere [43]. Theoretically, this area of activity is likely to increase with pain chronicity, as persistent LBP patients also have difficulty extending beyond 2-point-discrimination to decreased graphesthesia at the lower back compared to healthy controls [44]. It is also possible that the spatial resolution of painful sensory input is too poor in the lumbar region to differentiate one segment from another.
The previously described cortical smudging could also affect movement behaviors such as postural control [45], lumbopelvic motor control [46], and thoracolumbar dissociation [40]. This indicates that patients with persistent LBP are probably less able to perceive lumbar stiffness reliably. This is highlighted by the findings of Stanton et al., who reported that patients with persistent LBP felt significantly stiffer compared to healthy controls. However, when measuring lumbar stiffness using mechanical indentation, no between-group difference was observed [47]. Additionally, in a similar cohort of LBP patients, self-reported stiffness was not associated with two different stiffness measures obtained from the VT [48]. Suggesting that perceived stiffness may genuinely be a perceptual influence. Whether this finding is present in acute LBP is unknown.
Methodological considerations
A strength of the study was the large sample size compared to our previous topography study [15], albeit the measures were limited to the midline. All tests were conducted by the same rater limiting the intra-rater variability. While the VT is computer-controlled, further minimizing the risk of rater-based errors, this was not the case for the QST. We endeavored to reduce the risk of bias through multiple training sessions, allowing the participants up to three test stimuli to familiarize themselves with the procedures. Furthermore, both QST measurements are commonly used and, thus, vigorously described and tested in the literature [13, 49, 50].
However, the stiffness measurement was a single plane indentation and possibly not a clinically relevant measure of stiffness. It is not yet known if mechanical measurements of stiffness are clinically relevant beyond a few studies [11, 12, 21]. Manual palpation has some benefits over the experimental testing used in this study. It is possible to direct the pressure for stiffness and pain in multiple planes, examine trophic changes of the skin and muscles, perform joint-play, locate non-verbal reflectory muscle guarding and tender points using verbal feedback. The evidence for these factors is arguably sparse, and manual palpation carries with a considerable risk of bias [51]. Other biomechanical factors may better reflect spinal biomechanics such as local muscle activity [52], multifidus thickness, or disc diffusion [12]. Finally, the VT has only been deemed reliable in an asymptomatic population, and we currently do not have evidence that demonstrates the same measurement properties apply to LBP patients. However, the reliability score is consistent with the single indentation device, a similar technique, examined on LBP patients [38].
Another limitation is the lack of data on the most symptomatic clinical segment. While not apparent in this analysis, this localized point could potentially provide a more meaningful correlation, which we currently miss in the averaged data. Finally, this was a cross-sectional study that did not compare to other LBP groups or healthy controls. Thus the results are, therefore, only applicable for secondary care persistent non-specific LBP patients.
Conclusion
The a-priori hypothesis could not be confirmed. We found moderate correlations between spinal stiffness and mechanical pain sensitivity to be the opposite of what we expected; higher degrees of stiffness were associated with higher pressure pain threshold. As suspected, pressure and heat pain thresholds had a good correlation, while stiffness and heat sensitivity were poorly correlated. We observed no clinically relevant lumbar between-segment association for any of the outcomes in this population sample of persistent non-specific low back pain patients.
Acknowledgements
We would like to thank all the participants who participated in the study, the clinicians who helped with the inclusion, and Dr. Henrik Wulff Christensen for discussing the results. Furthermore, we want to acknowledge the reviewers for providing valuable feedback that helped improve the manuscript.
Abbreviations
- LBP
Low back pain
- QST
Quantitative sensory test
- VT
VerteTrack
- PPT
Pressure pain threshold
- C
Celsius
- HPT
Heat pain threshold
- GS
Global lumbar stiffness
- ρ
Pearson’s product-moment correlation
- Rs
Spearman’s rank correlation
Authors' contributions
CGN: Collected the data and wrote the initial draft of the manuscript. CGN, AGG, LK: Completed the data analysis. All authors contributed to the design and interpretation of the data. All authors read and approved the final manuscript.
Funding
CGN received funding from The Danish Chiropractic Fund for Research and Post Graduate Research, Hospital Lillebaelt—Research counsel, and The Danish Rheumatism Association.
Availability of data and materials
Data is available upon reasonable request. Please contact casper.nim@rsyd.dk.
Declarations
Ethics approval and consent to participate
All participants gave oral and written informed consent for the study, which was approved by the Regional Committees on Health Research Ethics for Southern Denmark (ID: S-20160201).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Netter FH. Atlas Der Anatomie. 5. Elsevier; 2011. [Google Scholar]
- 2.Hartvigsen J, Hancock MJ, Kongsted A, Louw Q, Ferreira ML, Genevay S, et al. What low back pain is and why we need to pay attention. Lancet. 2018;391(10137):2356–2367. doi: 10.1016/S0140-6736(18)30480-X. [DOI] [PubMed] [Google Scholar]
- 3.Bardin LD, King P, Maher CG. Diagnostic triage for low back pain: a practical approach for primary care. Med J Aust. 2017;206(6):268–273. doi: 10.5694/mja16.00828. [DOI] [PubMed] [Google Scholar]
- 4.Kent P, Keating J. Do primary-care clinicians think that nonspecific low back pain is one condition? Spine. 2004;29(9):1022–1031. doi: 10.1097/00007632-200405010-00015. [DOI] [PubMed] [Google Scholar]
- 5.Hancock MJ, Maher CG, Latimer J, Spindler MF, McAuley JH, Laslett M, et al. Systematic review of tests to identify the disc, SIJ or facet joint as the source of low back pain. Eur Spine J. 2007;16(10):1539–1550. doi: 10.1007/s00586-007-0391-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McGregor AH, Hukins DWL. Lower limb involvement in spinal function and low back pain. J Back Musculoskelet Rehabil. 2009;22(4):219–222. doi: 10.3233/BMR-2009-0239. [DOI] [PubMed] [Google Scholar]
- 7.Song AY, Jo HJ, Sung PS, Kim YH. Three-dimensional kinematic analysis of pelvic and lower extremity differences during trunk rotation in subjects with and without chronic low back pain. Physiotherapy. 2012;98(2):160–166. doi: 10.1016/j.physio.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 8.Papi E, Bull AMJ, McGregor AH. Is there evidence to use kinematic/kinetic measures clinically in low back pain patients? A systematic review. Clin Biomech. 2018;55:53–64. doi: 10.1016/j.clinbiomech.2018.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hadizadeh M, Kawchuk GN, Parent E. Reliability of a new loaded rolling wheel system for measuring spinal stiffness in asymptomatic participants. BMC Musculoskelet Disord. 2019 doi: 10.1186/s12891-019-2543-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wong AYL, Kawchuk GN. The clinical value of assessing lumbar posteroanterior segmental stiffness: a narrative review of manual and instrumented methods. PM&R. 2017;9(8):816–830. doi: 10.1016/j.pmrj.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 11.Fritz JM, Koppenhaver SL, Kawchuk GN, Teyhen DS, Hebert JJ, Childs JD. Preliminary investigation of the mechanisms underlying the effects of manipulation: exploration of a multivariate model including spinal stiffness, multifidus recruitment, and clinical findings. Spine. 2011;36(21):1772–1781. doi: 10.1097/BRS.0b013e318216337d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wong AYL, Parent EC, Dhillon SS, Prasad N, Kawchuk GN. Do participants with low back pain who respond to spinal manipulative therapy differ biomechanically from nonresponders, untreated controls or asymptomatic controls? Spine. 2015;40(17):1329–1337. doi: 10.1097/BRS.0000000000000981. [DOI] [PubMed] [Google Scholar]
- 13.Graven-Nielsen T, Arendt-Nielsen L. Assessment of mechanisms in localized and widespread musculoskeletal pain. Nat Rev Rheumatol. 2010;6(10):599–606. doi: 10.1038/nrrheum.2010.107. [DOI] [PubMed] [Google Scholar]
- 14.Marcuzzi A, Wrigley PJ, Dean CM, Graham PL, Hush JM. From acute to persistent low back pain: a longitudinal investigation of somatosensory changes using quantitative sensory testing—an exploratory study. PAIN Rep. 2018;3(2):e641. doi: 10.1097/PR9.0000000000000641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.O’Neill S, Larsen JB, Nim C, Arendt-Nielsen L. Topographic mapping of pain sensitivity of the lower back—a comparison of healthy controls and patients with chronic non-specific low back pain. Scand J Pain. 2019;19(1):25–37. doi: 10.1515/sjpain-2018-0113. [DOI] [PubMed] [Google Scholar]
- 16.Alfieri FM, Lima ARS, Battistella LR, Silva NCOV. Superficial temperature and pain tolerance in patients with chronic low back pain. J Bodyw Mov Ther. 2019;23(3):583–587. doi: 10.1016/j.jbmt.2019.05.001. [DOI] [PubMed] [Google Scholar]
- 17.Triano JJ, Budgell B, Bagnulo A, Roffey B, Bergmann T, Cooperstein R, et al. Review of methods used by chiropractors to determine the site for applying manipulation. Chiropr Man Ther. 2013;21(1):36. doi: 10.1186/2045-709X-21-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Neziri AY, Curatolo M, Nüesch E, Scaramozzino P, Andersen OK, Arendt-Nielsen L, et al. Factor analysis of responses to thermal, electrical, and mechanical painful stimuli supports the importance of multi-modal pain assessment. Pain. 2011;152(5):1146–1155. doi: 10.1016/j.pain.2011.01.047. [DOI] [PubMed] [Google Scholar]
- 19.George SZ, Bishop MD, Bialosky JE, Zeppieri G, Robinson ME. Immediate effects of spinal manipulation on thermal pain sensitivity: an experimental study. BMC Musculoskelet Disord. 2006;7(1):68. doi: 10.1186/1471-2474-7-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Honoré M, Leboeuf-Yde C, Gagey O. The regional effect of spinal manipulation on the pressure pain threshold in asymptomatic subjects: a systematic literature review. Chiropr Man Ther. 2018;26(1):11. doi: 10.1186/s12998-018-0181-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nim CG, Kawchuk GN, Schiøttz-Christensen B, O’Neill S. The effect on clinical outcomes when targeting spinal manipulation at stiffness or pain sensitivity: a randomized trial. Sci Rep. 2020;10(1):14615. doi: 10.1038/s41598-020-71557-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mieritz RM, Kawchuk GN. The accuracy of locating lumbar vertebrae when using palpation versus ultrasonography. J Manipulative Physiol Ther. 2016;39(6):387–392. doi: 10.1016/j.jmpt.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 23.Brown BT, Blacke A, Carroll V, Graham PL, Kawchuk G, Downie A, et al. The comfort and safety of a novel rolling mechanical indentation device for the measurement of lumbar trunk stiffness in young adults. Chiropr Man Ther. 2017;25(1):21. doi: 10.1186/s12998-017-0153-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Young A, Swain MS, Kawchuk GN, Wong AYL, Downie AS. The bench-top accuracy of the VerteTrack spinal stiffness assessment device. Chiropr Man Ther. 2020;28(1):42. doi: 10.1186/s12998-020-00331-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Paungmali A, Sitilertpisan P, Taneyhill K, Pirunsan U, Uthaikhup S. Intrarater reliability of pain intensity, tissue blood flow, thermal pain threshold, pressure pain threshold and lumbo-pelvic stability tests in subjects with low back pain. Asian J Sports Med. 2012;3(1):8–14. doi: 10.5812/asjsm.34718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Knutti IA, Suter MR, Opsommer E. Test–retest reliability of thermal quantitative sensory testing on two sites within the L5 dermatome of the lumbar spine and lower extremity. Neurosci Lett. 2014;579:157–162. doi: 10.1016/j.neulet.2014.07.023. [DOI] [PubMed] [Google Scholar]
- 27.Joanes DN, Gill CA. Comparing measures of sample skewness and kurtosis. J R Stat Soc Ser D (Stat) 1998;47(1):183–189. doi: 10.1111/1467-9884.00122. [DOI] [Google Scholar]
- 28.Wilcox R. The regression smoother LOWESS: a confidence band that allows heteroscedasticity and has some specified simultaneous probability coverage. J Mod Appl Stat Methods. 2017;16(2):29–38. doi: 10.22237/jmasm/1509494580. [DOI] [Google Scholar]
- 29.Hazra A, Gogtay N. Biostatistics series module 6: correlation and linear regression. Indian J Dermatol. 2016;61(6):593–601. doi: 10.4103/0019-5154.193662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.R Development Core Team . R: a language and environment for statistical computing. R Foundation for Statistical Computing; 2009. [Google Scholar]
- 31.Wickham H, Averick M, Bryan J, Chang W, McGowan L, François R, et al. Welcome to the Tidyverse. J Open Source Softw. 2019;4(43):1686. doi: 10.21105/joss.01686. [DOI] [Google Scholar]
- 32.Millican CR, Lam PH, Murrell GAC. Shoulder stiffness after rotator cuff repair: the fate of stiff shoulders up to 9 years after rotator cuff repair. J Shoulder Elbow Surg. 2020;29:1323–1331. doi: 10.1016/j.jse.2019.11.020. [DOI] [PubMed] [Google Scholar]
- 33.Wong AYL, Parent EC, Prasad N, Huang C, Chan KM, Kawchuk GN. Does experimental low back pain change posteroanterior lumbar spinal stiffness and trunk muscle activity? A randomized crossover study. Clin Biomech. 2016;34:45–52. doi: 10.1016/j.clinbiomech.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 34.Sa O, Ta G-N, Cb M, La A-N. Ultrasound guided, painful electrical stimulation of lumbar facet joint structures: an experimental model of acute low back pain. Pain. 2009;144(1–2):76–83. doi: 10.1016/j.pain.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 35.Wiech K, Lin C, Brodersen KH, Bingel U, Ploner M, Tracey I. Anterior insula integrates information about salience into perceptual decisions about pain. J Neurosci. 2010;30(48):16324–16331. doi: 10.1523/JNEUROSCI.2087-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arendt-Nielsen L, Yarnitsky D. Experimental and clinical applications of quantitative sensory testing applied to skin, muscles and viscera. J Pain. 2009;10(6):556–572. doi: 10.1016/j.jpain.2009.02.002. [DOI] [PubMed] [Google Scholar]
- 37.Kawchuk GN, Miazga S, Pagé I, Swain M, De Carvalho D, Funabashi M, et al. Clinicians’ ability to detect a palpable difference in spinal stiffness compared with a mechanical device. J Manip Physiol Ther. 2019;42(2):89–95. doi: 10.1016/j.jmpt.2019.02.002. [DOI] [PubMed] [Google Scholar]
- 38.Wong AYL, Kawchuk G, Parent E, Prasad N. Within- and between-day reliability of spinal stiffness measurements obtained using a computer controlled mechanical indenter in individuals with and without low back pain. Man Ther. 2013;18(5):395–402. doi: 10.1016/j.math.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 39.Arendt-Nielsen L, Graven-Nielsen T. Translational musculoskeletal pain research. Best Pract Res Clin Rheumatol. 2011;25(2):209–226. doi: 10.1016/j.berh.2010.01.013. [DOI] [PubMed] [Google Scholar]
- 40.Elgueta-Cancino E, Schabrun S, Hodges P. Is the organisation of the primary motor cortex in low back pain related to pain, movement and/or sensation? Clin J Pain. 2017;34:207–216. doi: 10.1097/AJP.0000000000000535. [DOI] [PubMed] [Google Scholar]
- 41.Moseley LG. I can’t find it! Distorted body image and tactile dysfunction in patients with chronic back pain. Pain. 2008;140(1):239–243. doi: 10.1016/j.pain.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 42.Bouffard J, Gagné M, Mercier C. Effect of painful and non-painful sensorimotor manipulations on subjective body midline. Front Hum Neurosci. 2013;7:77. doi: 10.3389/fnhum.2013.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Boendermaker B, Meier ML, Luechinger R, Humphreys BK, Hotz-Boendermaker S. The cortical and cerebellar representation of the lumbar spine: the neural representation of the lumbar spine. Hum Brain Mapp. 2014;35(8):3962–3971. doi: 10.1002/hbm.22451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wand BM, Di Pietro F, George P, O’Connell NE. Tactile thresholds are preserved yet complex sensory function is impaired over the lumbar spine of chronic non-specific low back pain patients: a preliminary investigation. Physiotherapy. 2010;96(4):317–323. doi: 10.1016/j.physio.2010.02.005. [DOI] [PubMed] [Google Scholar]
- 45.Tsao H, Galea MP, Hodges PW. Reorganization of the motor cortex is associated with postural control deficits in recurrent low back pain. Brain. 2008;131(8):2161–2171. doi: 10.1093/brain/awn154. [DOI] [PubMed] [Google Scholar]
- 46.Luomajoki H, Moseley GL. Tactile acuity and lumbopelvic motor control in patients with back pain and healthy controls. Br J Sports Med. 2011;45(5):437–440. doi: 10.1136/bjsm.2009.060731. [DOI] [PubMed] [Google Scholar]
- 47.Stanton TR, Moseley GL, Wong AYL, Kawchuk GN. Feeling stiffness in the back: a protective perceptual inference in chronic back pain. Sci Rep. 2017;7:9681. doi: 10.1038/s41598-017-09429-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Harsted S, Nyirö L, Downie A, Kawchuk GN, O’Neill S, Holm L, et al. Posterior to anterior spinal stiffness measured in a sample of 127 secondary care low back pain patients. Clin Biomech. 2021;87:105408. doi: 10.1016/j.clinbiomech.2021.105408. [DOI] [PubMed] [Google Scholar]
- 49.Rolke R, Baron R, Maier C, Tölle TR, Treede DR, Beyer A, et al. Quantitative sensory testing in the German research network on neuropathic pain (DFNS): standardized protocol and reference values. Pain. 2006;123(3):231–243. doi: 10.1016/j.pain.2006.01.041. [DOI] [PubMed] [Google Scholar]
- 50.O’Neill S, O’Neill L. Improving QST reliability—more raters, tests, or occasions? A multivariate generalizability study. J Pain. 2015;16(5):454–462. doi: 10.1016/j.jpain.2015.01.476. [DOI] [PubMed] [Google Scholar]
- 51.Stochkendahl MJ, Christensen HW, Hartvigsen J, Vach W, Haas M, Hestbaek L, et al. Manual examination of the spine: a systematic critical literature review of reproducibility. J Manipulative Physiol Ther. 2006;29(6):475–485.e10. doi: 10.1016/j.jmpt.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 52.Jiang N, Falla D, d’Avella A, Graimann B, Farina D. Myoelectric control in neurorehabilitation. Crit Rev Biomed Eng. 2010;38(4):381–391. doi: 10.1615/CritRevBiomedEng.v38.i4.30. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data is available upon reasonable request. Please contact casper.nim@rsyd.dk.