Abstract
Background
Coastal wetlands are threatened by the increased salinity that may result from sea level rise. Salinity stress alters species zonation patterns through changes in competitive outcome between species differing in salinity tolerance. This study therefore aimed to understand how salinity and light affect two dominant and competing coastal wetland grasses that differ in salt tolerance, height and photosynthetic metabolism.
Methods
The C4 species Spartina anglica and the C3 species Phragmites australis were grown at five salinity levels (0, 7, 14, 21 and 28 ppt) and two light fluxes (100 % and 50 % of natural daylight) in an outdoor experimental setup for 102 d with full access to nutrients.
Key Results
Salinity reduced the biomass, height and shoot density of P. australis from 81.7 g dry weight (DW), 0.73 m and 37 shoots per pot at a salinity of 0 ppt to 16.8 gDW, 0.3 m and 14 shoots per pot at a salinity of 28 ppt. Biomass, height and shoot density of S. anglica did not respond or were only slightly reduced at the highest salinity of 28 ppt. High salinity also resulted in a higher tissue concentration of N and P in P. australis. Both species had low ability to acclimate to the lower light flux. Shade acclimation in S. anglica occurred via modest changes in specific leaf area, pigment content and biomass allocation.
Conclusions
High salinity reduced traits important for light competition and increased the nutrient concentration in P. australis leaf and root biomass, while this was overall unaffected in S. anglica. This is likely to reduce the competitive ability of P. australis over S. anglica for light because at high salinities the former cannot effectively shade the lower-growing S. anglica. Neither species effectively acclimates to shade, which could explain why S. anglica does not occur in the understorey of P. australis at low salinities.
Keywords: Salt tolerance, shade acclimation, Spartina anglica, Phragmites australis, light competition, C4 photosynthesis, ion regulation, sodium, salt exclusion
INTRODUCTION
Wetlands provide a wide range of ecosystem services including shoreline protection, nutrient retention, habitat provision, biomass production and carbon sequestration (Barbier et al., 2011, Kiviat, 2013). These services are threatened in salt marshes due to climate change effects such as sea level rise, which is expected to inundate salt marshes and reduce their area by up to 45 % by the end of the 21st century (Craft et al., 2009). More frequent extreme weather events and changes in precipitation are also expected (IPCC, 2014), altering salinity regimes in coastal wetlands. Increased precipitation will reduce soil salinity, whereas storms will deposit saline water further inland and, with reduced precipitation, increase soil salinity. Thus, it is very likely that soil salinity in many coastal wetlands will change in the future. High salinity reduces the primary productivity of salt marshes (Curco et al., 2002; Pennings et al., 2005), as maintaining ion homeostasis in salt marsh vegetation requires more energy when the relative amount of ions such as Na increases in the soil while others are diluted. In addition, under saline conditions more C and N is used for producing compatible osmolytes, leaving less C and N available for plant growth (Munns and Tester, 2008).
Coastal wetlands often have a conspicuous species zonation in the form of large monospecific stands due to interspecific difference in stress tolerance, resource competition or a combination of these where abiotic stress affects resource competition (Levine et al., 1998; Huckle et al., 2000; Emery et al., 2001; Engels and Jensen, 2010; Schoolmaster and Stagg, 2018). Nutrient addition experiments, aimed at understanding how anthropogenic eutrophication has affected nutrient competition, have shown that higher nutrient availability resulted in increased growth in some species but also subsequent loss of or decline in species where high nutrient availability had lower effects on growth (van Wijnen and Bakker, 1999; Emery et al., 2001; Crain, 2007). This loss or decline has been ascribed to shading effects from the species that experienced increased growth. It is likely that the ability to increase light capture via shade acclimation when growing next to a taller and more vigorous species will determine whether the shorter species can continue to coexist with the enhanced taller species (Hautier et al., 2009).
Murchie and Horton (1997) assessed the light acclimation potential in woodlands, which they defined as the magnitude of change in traits under high vs. low light flux. They found that the acclimation potential to differences in light fluxes was highest for herbaceous species found in both shaded woodlands and areas with higher light fluxes such as open grasslands. Species growing predominantly in shade or high light fluxes had lower acclimation potential. It is therefore likely that coastal wetland plants will have a low acclimation potential, as salt marshes are high light flux ecosystems due to the absence of tall vegetation such as trees. Salt marsh plants therefore usually experience high light fluxes. A few individuals at the ecotone could, however, be shaded by adjacent stands of a taller species as it attenuates light. Thus, at the transition zone both shade acclimation and light attenuation (i.e. light competition) could be important for the outcome between two neighbouring species (Esselink et al., 2000). However, in salt marshes much less is known about light competition and how it is affected by physical stress such as high salinity compared to competition for other resources (Craine and Dybzinski, 2013).
Functional traits can be used to predict the outcome of various biotic interactions including competition (Adler et al., 2013; Kunstler et al., 2016; Semchenko et al., 2018). In an attempt to distinguish the magnitude of below-ground competition from whole plant competition, various experimental designs have been used, but they are not able to fully separate above- and below-ground competition from each other (McPhee and Aarssen, 2001). As an example, the clipping technique, where above-ground biomass is removed to reduce light competition, also affects below-ground competition (Remison and Snaydon, 1980). In the present study, we therefore recognize the difficulties in separating above- and below-ground competition and do not directly attempt to compare the magnitude of below- vs. above-ground competition. Instead, we intend to understand how soil salinity interacts with light competition. To avoid below-ground competition, plants were grown in isolated pots, while shade cloth was used to produce light fluxes similar to those experienced below the canopy of a taller species (Hirtreiter and Potts, 2012). This experimental setup allowed us to infer light competition based on trait responses to salinity.
We chose two species with multiple ecophysiological contrasts that co-occur in northern European salt marshes (Esselink et al., 2000). The first is Spartina anglica, a grass with the C4 photosynthetic pathway, which is invasive in mainland Europe after being deliberately introduced for land reclamation in the early 20th century. It is an allotetraploid derived from the S. alterniflora × S. maritima hybrid S. × townsendii (Raybould et al., 1991). It has a high salt tolerance but it does not reach more than 0.5 m in height. For contrast, we chose Phragmites australis, a species with C3 photosynthesis and lower salt tolerance but also taller (1–2 m). Based on their ecophysiological contrasts we predict that S. anglica will produce more biomass under a combination of high light and high salinity but it will possess a low acclimation potential to shade due to its C4 photosynthesis, which is less plastic compared to C3 photosynthesis (Sage and McKown, 2006). In addition, we predict that high salinity will reduce traits important for light competition more in P. australis than in S. anglica. Elucidation of species-specific differences in salt and shading stresses in the these two important salt marsh species will improve our understanding of above-ground competition, and assist our predictions about how coastal wetlands in western Europe will respond to the hydrological changes they will inevitably experience. We therefore aim to provide insight into the longer-term above-ground competitive interactions emerging from these challenges.
MATERIALS AND METHODS
Growth conditions and plant material
Rhizomes of S. anglica and P. australis were collected at random points along a 300-m transect parallel to the shoreline at Alrø, Horsens Fjord, Denmark (55.85°N, 10.06°E) in January 2017 and initially propagated in a glasshouse at the Påskehøjgård research farm (56.23°N, 10.13°E), 10 km north of Aarhus, Denmark. Rhizomes were propagated in peat soil, fertilized once in January with 20 g of ‘OsmocotePlus NPK 15–5–11 + Mg and Micro’ (Scotts Miracle-Gro Company, OH, USA) per 12-L pot. Natural light was supplemented with artificial lighting in the period 0600–1800 h if ambient sunlight was <300 µmol m−2 s−1. Temperature was above 15 °C the entire day. Sky windows opened and heat reflective curtains were used whenever the temperature reached 22 °C. Glasshouse temperatures could, however, still increase above 22 °C on sunny, cloud-free days. On 22–23 May 2017 (23 May = Day 1 of the experiment), plants were carefully dug up, separated into 160 plantlets each with 3–5 tillers and approximately the same amount of below-ground biomass (visual assessment). Each individual was planted in a 12-L black plastic bucket (surface area of 616 cm2). The buckets were filled with 10 L of sand and had drainage holes at the bottom. The buckets were lined with a synthetic weed barrier that prevented roots from growing outside the pot and kept the sand inside. The 12-L bucket was placed inside a larger 20-L black plastic bucket, which retained water and ensured the 12-L pot was always flooded. Pots were randomly distributed in an outdoor experimental plot. Each plant received 10 L of water with 0.08 g L−1 of ‘Pioner Ironchelat 6 % EDDHA’ (Horticoop Scandinavia A/S, Denmark) and we ensured that the water level was just below the surface of the sand in the inner pot. Each pot was fertilized weekly with 10 mL of a stock solution which contained 10 % (w/v) ‘Grøn Pioner NPK 19–2–15 + Mg’ and 1 mL of ‘Mikro Plus with Iron’ (Brøste A/S, Denmark).
The salinity treatments were initiated at Day 1 and were increased in steps of 7 ppt (1 ppt = 1 g L−1), with 4–5 d between each step. In total, there were five salinity levels (0, 7, 14, 21 and 28 ppt). The 28 ppt salinity treatment was achieved on Day 15. A commercial sea salt (hw-Marinemix professional) was used to prepare the salt water. Water level and conductivity in the outer pot were checked regularly and water added as needed to maintain a total volume of 10 L. On two occasions (Days 29 and 57), it was necessary to replace the water because heavy rain had diluted the salt and water had spilled from the outer pots. The pots were all flushed twice with 10 L of saltwater of their respective salinity and then filled with 8 L of saltwater containing 0.08 g L−1 of ‘Pioner Ironchelat 6 % EDDHA’. At the second flushing, the water volume was reduced to 5 L. The reduction in water volume prevented overflow and inhibited algal growth, which otherwise quickly occurred whenever the sand surface was inundated.
The light treatments consisted of an unshaded plot receiving full sunlight (HL), and another plot which was shaded (LL) with green Hostalen shade cloth made of PE foil with a density of 125 g m−2 (Hartving ApS, Denmark). The shade cloth reduced the natural incident light flux to 50 % of HL and was mounted ~1.8 m above the ground on metal wires stretched between wooden poles. To prevent the plants in the shade treatment from receiving full sunlight when the sun was at a low angle, shade cloth was also mounted on the sides of the poles down to a height of 0.4–0.6 m, creating a ‘tunnel’ of shade cloth. All plants were placed at least 0.5 m apart to prevent them from shading each other. Each plot contained both species at all five salinity levels. The experimental design was 2 species × 2 light treatments × 5 salinity levels × 8 replicates, giving a total of 160 plants. On Day 36, all plants and the shade cloth were switched between the two plots to reduce block effects. Temperature loggers were randomly distributed in three pots in each light treatment to monitor whether the shade treatment affected soil temperature (mean ± s.d. HL = 18.3 ± 0.1 °C and LL = 17.1 ± 0.1 °C). A climate station (CR1000, Campbell Scientific, Logan, UT, USA) situated 50 m from the plots recorded incident light flux, relative air humidity and air temperature (Supplementary Data Fig. S1).
Morphological measurements
At weekly intervals, the height of the tallest shoot in each pot and total numbers of shoots per pot were recorded. Shoot height and density were measured from Days 4 to 58. For simplicity, we chose to report values from Day 58, as shoot production rate and height increment did not reveal additional information compared to shoot height and density at Day 58. Shoot density is presented as numbers of shoots per pot. The experiment was harvested over 3 d (Days 91–93). All plants were then separated into leaves, stems, rhizomes, roots, above-ground dead biomass and below-ground dead biomass. Leaves and roots were rinsed with tap water and blotted dry with paper for further tissue elemental analysis (see below). Plant fractions were oven dried at 60 °C for at least 48 h to obtain their dry weight (DW). Total biomass data were expressed as absolute DW per pot and the biomass fractions as proportion of total biomass.
Tissue analysis
The concentrations of Na, K, P, Ca, Mg, S, Fe, Mn and N and C : N ratio in leaves and roots of the two species were analysed. First, the entire fractions were ground to fine powder. Then, 2–5 mg DW was used to analyse total C and N with a CHNS analyser (Vario EL Cube, Elementar, Langenselbold, Germany). Another 0.5 g DW was acid-digested with 4 mL 65 % HNO3 and 2 mL 30 % H2O2 in a microwave digestion system (Multiwave 3000, Anton Paar GmbH, Graz, Austria) and the concentrations of Na, K, P, Ca, Mg, S, Fe and Mn in the digest were analysed by inductively coupled plasma optical emission spectroscopy (Optima 2000 DV, Perkin Elmer Instruments Inc., Shelton, CT, USA). The mass ratio between C and N (C : N) was calculated. The ratio between Na and K for leaf and roots (Na : K ratio) was calculated on a molar basis. For the 0, 14 and 28 ppt salinity treatments, the leaf and root concentrations of the four major cations Na, K, Ca and Mg were pooled.
Photosynthetic pigment analysis, specific leaf area (SLA) and leaf water content
On Days 63–76, a fully developed sun-exposed leaf was removed from each replicate for pigment analysis. Immediately after the leaf had been detached, it was weighed to obtain its fresh weight (FW). The leaf was then fixed in place under a glass plate and photographed perpendicular to the leaf surface for area determination using ImageJ v. 1.52i (Easlon and Bloom, 2014). All pictures were taken with the same distance to the leaf surface. The leaf was then frozen at −20 °C. Leaves were then placed in a dry shipper with liquid N2 and returned to the laboratory for further processing. Leaves were freeze-dried and weighed to obtain DW and then analysed for chlorophyll a (Chl a), chlorophyll b (Chl b), total chlorophyll (Chl a + b) and total carotenoids (Car) with a spectrophotometer (UV-1800, Shimadzu, Tokyo, Japan) according to Lichtenthaler (1987). The ratios between Chl a and b (Chl a : b) and Car and Chl a + b (Car : Chl a + b) were calculated, as well as the SLA (ratio between leaf area and DW of the same leaf) and the content of water in the leaf were determined (leaf water, calculated as ((FW − DW)/DW)). Pigment concentrations, SLA and leaf water were only analysed for the 0, 14 and 28 ppt salinity treatments.
Statistical analysis
Data were analysed as fully factorial using a generalized linear model (GLM) with normally distributed errors and identity as a link function. The significance level was 0.05 and the analysis was made with the software JMP 14.3.0 (SAS Institute, Cary, NC, USA). Species (S. anglica, SA; P. australis, PA), salinity (0, 7, 14, 21, 28 ppt or 0, 14, 28 ppt), light (HL, LL) and all interaction terms were used as explanatory variables. To meet the assumptions of the GLM, normality and homoscedasticity of the residuals were inspected with Q-Q and residuals vs. fitted values plots while 11 outliers were detected and removed according to Grubbs (1969). The following parameters were therefore transformed to meet the assumptions: leaf C : N, leaf Na : K (log-transformed), leaf Na, root Na (power-transformed), Chl a, Chl b and Chl a + b and root Ca (reciprocal transformation). Parameters are reported as means ± s.e. (standard error) of untransformed data.
RESULTS
Morphology
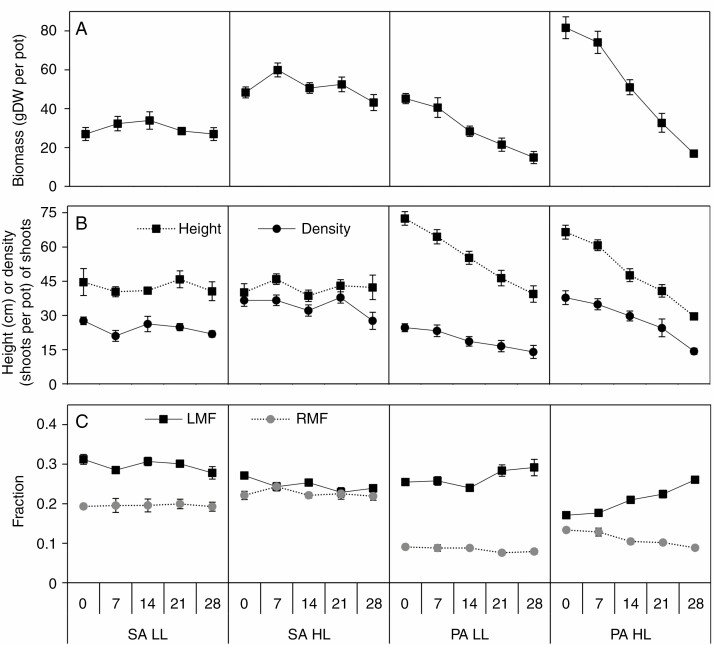
At 0 ppt salinity P. australis was twice as tall as S. anglica and produced twice as much biomass, while their shoot densities were similar (Fig. 1). Total biomass, shoot height and shoot density were affected by salinity and light (Table 1). High salinity resulted in lower total biomass, shoot height and density in P. australis but not in S. anglica (species × salinity, Table 1, Fig. 1A and B). The LL treatment resulted in lower total biomass and shoot density in both species. Total biomass was 41 % lower in LL compared to HL in S. anglica but similar between salinity treatments, while the biomass reduction due to shading depended on salinity in P. australis (species × salinity × light, Table 1, Fig. 1A). In LL at 28 ppt, P. australis was shorter, produced less biomass and had lower shoot density than S. anglica. The salinity-induced reduction in total biomass for P. australis was larger in HL compared to LL. In contrast to S. anglica, P. australis was taller in the LL treatment compared to the HL treatment (species × light, Table 1, Fig. 1B).
Fig. 1.
Changes in total biomass (A), shoot height (Height), number of shoots per pot (Density) (B) and the fraction of biomass allocated to roots (RMF) and leaves (LMF) (C) in S. anglica (SA) and P. australis (PA) to the two light fluxes (LL low light, 50 % full sunlight; HL high light, 100 % full sunlight) and five salinity levels (0, 7, 14, 21, 28 ppt). Values are mean ± s.e. (n = 8).
Table 1.
χ 2 ratios of GLM (d.f. total = 151–159) analyses of morphology in P. australis and S. anglica by species (Sp.), light (L) and salinity (S)
| Sp. d.f. = 1 | L d.f. = 1 | S d.f. = 4 | Sp. × L | Sp. × S | S × L | Sp. × L × S | |
|---|---|---|---|---|---|---|---|
| Biomass | 0.1 | 120.3*** | 110.5*** | 0.0 | 82.2*** | 23.0*** | 12.7* |
| Shoot height | 52.7*** | 6.9** | 75.0*** | 5.1* | 72.5*** | 2.0 | 1.1 |
| Shoot density | 24.6*** | 63.7*** | 50.6*** | 0.2 | 18.5** | 11.0* | 4.9 |
| LMF | 53.7*** | 105.8*** | 15.7** | 0.7 | 51.9*** | 7.9 | 9.8* |
| RMF | 282.9*** | 44.2*** | 10.6* | 0.2 | 6.2 | 5.5 | 1.8 |
| SMF | 176.7*** | 0.3 | 18.2** | 0.0 | 17.8** | 6.5 | 4.3 |
| RZMF | 47.1 | 9.9** | 6.4 | 0.3 | 25.8*** | 4.3 | 4.3 |
| BG dead | 0.5 | 0.6 | 6.0 | 1.6 | 4.9 | 5.6 | 3.4 |
| AG dead | 63.0*** | 59.0*** | 29.7*** | 0.1 | 22.2*** | 17.0** | 15.6** |
Number of shoots per pot (Shoot density), leaf mass fraction (LMF), root mass fraction (RMF), stem mass fraction (SMF), rhizome mass fraction (RZMF), below-ground dead biomass (BG dead), and above-ground dead biomass (AG dead).
Statistically significant values are in bold: *P < 0.05, **P < 0.01, ***P < 0.001.
Biomass allocation was affected by salinity and light (Table 1). The salinity response differed between species (species × salinity, Table 1). In S. anglica leaf mass fraction (LMF) was not affected by salinity while the LL treatment resulted in higher LMF compared to HL. In P. australis LMF was also higher in the LL treatment but only at low salinity. At high salinity, LMF did not differ between the light treatments in P. australis (species × light × salinity, Table 1, Fig. 1C). Root mass fraction (RMF) was affected by the three main factors and was higher in HL compared to LL (Table 1, Fig. 1C). The RMF reduction due to salinity was larger in P. australis compared to in S. anglica but this was not significant (no species × salinity interaction, Table 1, Fig. 1C). High salinity also affected shoot mass fraction (SMF) and rhizome mass fraction (RZMF) and the response differed between species (species × salinity, Table 1; Supplementary Data Fig. S2A). Finally, above-ground dead biomass was also affected, both by main factors and several interactions, while below-ground dead biomass was unaffected (Table 1; Fig. S2B and C). The amount of dead biomass responded similarly to total biomass, such that a large total biomass production resulted in high above-ground dead biomass (Fig. 1A; Fig. S2B).
Pigments, SLA and leaf water
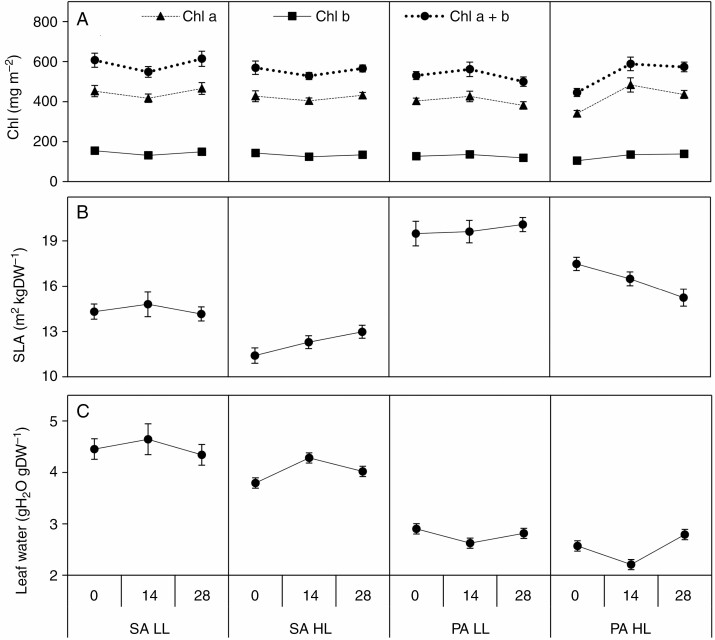
Chl a, Chl b and Chl a + b showed a three-way interaction (Table 2). This was due to lower concentrations at 0 ppt and HL in P. australis while the remaining concentrations were higher and the differences between the other treatments were small (7–13 %) (Fig. 2A). Because Chl a and Chl b did not change by an equal magnitude, this affected the Chl a : b ratio (Table 2). High salinity resulted in a higher Chl a : b ratio in S. anglica but not in P. australis (species × salinity, Table 2; Supplementary Data Fig. S3A). The light treatment also affected Chl a : b but the changes were small (>7 %). Car:Chl a + b was higher in S. anglica while the response to salinity differed between species (species × salinity, Table 2; Fig. S3B). Both species responded to LL with higher SLA, while salinity had the largest effect on SLA in HL (Fig. 2B). In HL the effect of salinity on SLA differed between species, as high salinity resulted in lower SLA in P. australis but higher SLA in S. anglica (species × light × salinity, Table 2, Fig. 2B). Finally, leaf water content was also affected by both light and salinity and differed between species (Table 2, Fig. 2C).
Table 2.
χ 2 ratios of GLM (d.f. total = 79–82) analyses of pigments per leaf area, specific leaf area (SLA) and leaf water content in P. australis and S. anglica by species (Sp.), light (L) and salinity (S)
| Sp. d.f. = 1 | L d.f. = 1 | S d.f. = 2 | Sp. × L | Sp. × S | S × L | Sp. × L × S | |
|---|---|---|---|---|---|---|---|
| SLA | 113.9*** | 57.9*** | 0.3 | 3.4 | 4.6 | 0.6 | 9.6** |
| Chl aarea | 4.4* | 0.3 | 5.1 | 1.4 | 13.8** | 8.2* | 6.4* |
| Chl barea | 15.6*** | 3.1 | 1.5 | 0.9 | 21.1*** | 9.4** | 9.9** |
| Chl a + barea | 5.4* | 0.3 | 4.3 | 1.7 | 16.8** | 8.8* | 7.4* |
| Chl a : b | 11.4*** | 5.2* | 11.6** | 0.0 | 13.5** | 2.4 | 3.1 |
| Car:Chl a + b | 49.8*** | 2.2 | 3.5 | 0.6 | 8.6* | 0.6 | 0.5 |
| Leaf water content | 1314*** | 7.2** | 7.1* | 0.5 | 17.8*** | 1.9 | 0.5 |
Chlorophyll a (Chl aarea), chlorophyll b (Chl barea), total chlorophyll (Chl a + barea), chlorophyll a and b ratio (Chl a : b), ratio between total carotenoids and total chlorophyll (Car : Chl a + b) and degrees of freedom (d.f.).
Statistically significant values are in bold: *P < 0.05, **P < 0.01, ***P < 0.001.
Fig. 2.
Response of chlorophyll pigments (Chl) (A), specific leaf area (SLA) (B) and water in the leaf (Leaf water) (C) in S. anglica (SA) and P. australis (PA) to changes in light fluxes (LL low light, 50 % full sunlight; HL high light, 100 % full sunlight) and salinity (0, 14, 28 ppt). Values are mean ± s.e. (n = 5–7).
Leaf mineral nutrient concentrations
In general, mineral nutrient concentrations of leaves were significantly affected by all three main factors and several interactions (Table 3). The two species differed in mineral concentrations and responded differently to salinity (species × salinity, Table 3). The LL treatment produced a general pattern of higher leaf element concentrations in both species except for leaf Na. Specifically, salinity resulted in higher leaf N and the salinity-induced increase in leaf N was larger in P. australis than in S. anglica (species × salinity, Table 3, Fig. 3A). Plants with low total biomass had high leaf N and vice versa (S. anglica y = 3.4−0.01x, R2 = 0.74, P ≤ 0.001; P. australis y = 4.8−0.006x, R2 = 0.86, P ≤ 0.001). Additionally, leaf C : N was also affected by all three main factors and interactions (Table 3; Supplementary Data Fig. S4A). Leaf P was affected similar to leaf N but the salinity-induced increase in leaf P was lower than the increase in leaf N (Fig. 3).
Table 3.
χ 2 ratios of GLM (d.f. total = 156–159) analyses of leaf tissue elements in P. australis and S. anglica by species (Sp.), light (L) and salinity (S)
| Sp. d.f. = 1 | L d.f. = 1 | S d.f. = 4 | Sp. × L | Sp. × S | S × L | Sp. × L × S | |
|---|---|---|---|---|---|---|---|
| C : NLeaf | 387.8*** | 201.1*** | 136.4*** | 17.1*** | 53.1*** | 17.7** | 3.5 |
| NLeaf | 392.0*** | 176.5*** | 153.8*** | 0.0 | 93.4*** | 9.5* | 7.6 |
| PLeaf | 2.1 | 83.9*** | 51.8*** | 4.0* | 10.0* | 6.6 | 3.5 |
| CaLeaf | 89.7*** | 7.6** | 247.7*** | 0.2 | 60.9*** | 7.3 | 1.7 |
| KLeaf | 95.6*** | 122.7*** | 56.0*** | 0.7 | 14.3** | 7.5 | 1.2 |
| MgLeaf | 85.3*** | 47.6*** | 129.3*** | 2.0 | 6.6 | 4.3 | 2.9 |
| NaLeaf | 533.0*** | 0.6 | 314.0*** | 31.9*** | 89.3*** | 5.6 | 7.9 |
| SLeaf | 226.9*** | 173.6*** | 42.5*** | 8.9** | 92.8*** | 7.0 | 1.3 |
| FeLeaf | 109.8*** | 11.7*** | 3.3 | 0.3 | 9.9* | 14.3** | 5.9 |
| MnLeaf | 157.7*** | 45.1*** | 34.4*** | 1.2 | 123.3*** | 8.3 | 4.9 |
| Na : KLeaf | 487.2*** | 44.9*** | 256.4*** | 25.1*** | 2.3 | 9.5* | 11.1* |
| CationsLeaf | 255.6*** | 104.8*** | 151.9*** | 8.1** | 80.4*** | 2.5 | 1.2 |
Ratio between carbon and nitrogen (C : NLeaf), concentrations of N, P, Ca, K, Mg, Na, S, Fe and Mn per unit leaf dry weight (NLeaf, PLeaf, CaLeaf, KLeaf, MgLeaf, NaLeaf, SLeaf, FeLeaf, MnLeaf), pooled content of CaLeaf, KLeaf, MgLeaf, NaLeaf (CationsLeaf) and degrees of freedom (d.f.).
Statistically significant values are in bold: *P < 0.05, **P < 0.01, ***P < 0.001.
Fig. 3.
Changes in percentage N (A) and percentage P (B) per biomass in root and leaf tissue in S. anglica (SA) and P. australis (PA) to the two light fluxes (LL low light, 50 % full sunlight; HL high light, 100 % full sunlight) and five salinity levels (0, 7, 14, 21, 28 ppt). Values are mean ± s.e. (n = 8).
The leaf Ca response to salinity was species-specific as the reduction in leaf Ca with increasing salinity was larger in S. anglica than in P. australis (species × salinity, Table 3; Supplementary data Fig. S5A). In contrast to leaf Ca, leaf Mg was higher at high salinities and both species responded similarly to salinity (Table 3; Fig. S5A). Leaf K was highest at high salinity and the response to salinity differed between species (species × salinity, Table 3, Fig. 4A). More importantly, leaf K was highest in P. australis in all instances. The leaf Na response in S. anglica was in sharp contrast to that in P. australis as leaf Na never exceeded 0.08 mol kgDW−1 while it was up to 7 times higher in S. anglica (species × salinity, Table 3, Fig. 4A). The only exception to the general response of higher leaf element concentrations in LL was noted for leaf Na, which was lower in LL but only in P. australis, while leaf Na followed the general response and was higher in LL in S. anglica (species × light, Table 3, Fig. 4A). Higher leaf S was observed in LL for both species while the salinity effect on leaf S resulted in lower leaf S in S. anglica but higher leaf S in P. australis (species × salinity, Table 3; Fig. S5C). The sum of changes in different cations affected the leaf cation pool (Table 3, Fig. 4C) while the responses of leaf Na and K affected leaf Na : K (Fig. S4B). Finally, leaf Fe and Mn were also affected by salinity and light (Table 3, Fig. S6A).
Fig. 4.
Comparison of K and Na concentration in the leaf (A) and root (B) tissue and the total concentration of Ca, K, Mg and Na in the leaf and root tissue (C) of S. anglica (SA) and P. australis (PA) to the two light fluxes (LL low light, 50 % full sunlight; HL high light, 100 % full sunlight) and five salinity levels (0, 7, 14, 21, 28 ppt). Values are mean ± s.e. (n = 8).
Root mineral nutrient concentrations
Root mineral nutrient concentrations in several instances showed species × salinity interactions (Table 4). Species × salinity interactions were observed for root N, P and C : N as they were not affected by salinity in S. anglica in contrast to P. australis (Table 4). At low salinity, root N and P were lowest and root C : N highest in P. australis but at higher salinity this changed, such that root N and P were lowest and root C : N highest in S. anglica (Fig. 3A; Supplementary Data Fig. S4A). In addition, root N increased with biomass but, unlike leaf N, there was no species difference in the root N and biomass relationship (y = 1.87−0.011x, R2 = 0.74, P ≤ 0.001). Despite the three-way interaction on root Ca, the overall response was similar (Table 4; Fig. S5B). Again, despite the interactions, root Mg was not overall differently affected by light or salinity in the two species (Fig. S5B). Root K and Na were higher at high salinity and in LL compared to HL (Table 4, Fig. 4B). Phragmites australis had higher root Na than S. anglica while root K did not differ between species. For root Na : K, we detected a species × salinity × light interaction (Table 4; Fig. S4B). The net results of variation in these four cations affected the cation pool but the response did not differ due to species-specific salinity or light responses (no interactions, Table 4, Fig. 4C). Finally, salinity and light also affected root S, Fe and Mn (Table 4; Figs S5C and S6).
Table 4.
χ 2 ratios of GLM (d.f. total = 156–159) analyses of root tissue elements in P. australis and S. anglica by species (Sp.), light (L) and salinity (S)
| Sp. d.f. = 1 | L d.f. = 1 | S d.f. = 4 | Sp. × L | Sp. × S | S × L | Sp. × L × S | |
|---|---|---|---|---|---|---|---|
| C : N Root | 1.4 | 150.0*** | 95.5*** | 0.7 | 68.9*** | 10.5* | 11.9* |
| N Root | 0.0 | 114.0*** | 73.6*** | 0.0 | 56.1*** | 2.0 | 2.4 |
| P Root | 45.5*** | 56.2*** | 82.5*** | 3.4 | 84.3*** | 2.7 | 11.9* |
| Ca Root | 6.5* | 14.3*** | 44.8*** | 2.3 | 3.1 | 2.5 | 18.5** |
| K Root | 0.6 | 23.4*** | 80.8*** | 1.6 | 7.6 | 1.5 | 5.1 |
| Mg Root | 26.3*** | 16.2*** | 297.2*** | 5.1* | 27.0*** | 13.8** | 2.3 |
| Na Root | 185.1*** | 12.7*** | 243.2*** | 1.7 | 4.3 | 1.8 | 8.9 |
| S Root | 14.5*** | 0.3 | 14.6** | 4.0* | 1.1 | 4.8 | 2.3 |
| Fe Root | 11.3*** | 17.2*** | 88.6*** | 0.4 | 23.2*** | 1.6 | 6.3 |
| Mn Root | 122.8*** | 63.0*** | 10.1* | 0.0 | 33.6*** | 2.5 | 1.4 |
| Na : KRoot | 182.2*** | 11.8** | 116.1*** | 12.7** | 17.7*** | 6.6 | 10.4* |
| Cations Root | 62.4*** | 22.8*** | 177.4*** | 0.7 | 0.8 | 1.5 | 2.4 |
Ratio between carbon and nitrogen (C : NRoot), concentrations of N, P, Ca, K, Mg, Na, S, Fe and Mn per unit root dry weight (NRoot, PRoot, CaRoot, KRoot, MgRoot, NaRoot, SRoot, FeRoot, MnRoot), pooled content of CaRoot, KRoot, MgRoot, NaRoot (CationsRoot) and degrees of freedom (d.f.).
Statistically significant values are in bold: *P < 0.05, **P < 0.01, ***P < 0.001.
DISCUSSION
Our results have demonstrated substantial differences in growth and tissue properties between S. anglica and P. australis in response to light and salinity. Our first hypothesis was that S. anglica would have maximum total biomass under a combination of high light and high salinity, which our results confirmed. Light flux affected P. australis less at high compared to low salinity. This indicates that the ability of P. australis to use light effectively for biomass production is lower at high salinity, while this was not true for S. anglica as the differences in biomass due to the light treatment were sustained at all salinities. That the light treatment affected P. australis less at low compared to high salinity could be due to the larger amount of water transpired in the HL treatment compared to the LL treatment, and due to its lower salt tolerance compared to S. anglica. This indicates that low light fluxes could reduce some of the adverse effects from high salinity in P. australis. Although air temperature could also have been higher in the HL than in the LL treatment, as it can be difficult to reduce light fluxes in outdoor experiments without affecting temperature, this is likely to reflect natural conditions as shaded areas have lower temperature (Belsky et al., 1993). High transpiration rates result in higher mass flow of water containing dissolved Na towards the roots thereby concentrating Na in the rhizosphere (Oliver and Barber, 1966), but it can also result in lower leaf water content (Lichtenthaler et al., 1981) in accordance with our observations. A reduction in water content lowers the amount of ions needed to obtain a given water potential such that the lower leaf water content could be associated with higher leaf ion concentrations as we observed in HL (Glenn, 1987). The changes in leaf morphology that reduce transpiration rates will probably reduce mass flow water with dissolved ions and accordingly we observed lower SLA in P. australis due to salinity but only in the HL treatment (Wright et al., 1994). Low SLA leaves are, however, more costly to produce in terms of biomass (Villar and Merino, 2001). High rhizosphere Na concentrations increase the amount of energy needed for ion exclusion as this raises the influx of Na into the tissue. Phragmites australis actively transports most Na back into the rhizosphere before it reaches the shoot (Matsushita and Matoh, 1992), increasing the possibility of futile Na cycling in P. australis. Futile Na cycling is an energy-requiring process where the same Na ion is taken up and excluded several times (Britto and Kronzucker, 2006). These well-documented salt effects on P. australis are all consistent with its greater salt sensitivity in HL than LL in our study, as they would be exacerbated by the greater transpiration rate in HL.
In contrast, in C4 species such as S. anglica morphological changes that improve water use, such as lower SLA, are less advantageous. C4 species have lower transpiration rates than C3 species, so transport less Na by mass flow and the concentration is probably lower in the root zone. This often negates the need for the SLA and water content acclimations seen in C3 species; in fact, SLA in S. anglica increased rather than decreased at high salinity. Additionally, Na is not transported back to the root zone in S. anglica, but is taken up and used for osmoregulation in the leaf tissue (Bradley and Morris, 1991), which can lower the soil salinity further in the root zone (Ke-Fu, 1991). Spartina anglica secretes excess Na in the leaf tissue onto the leaf surface thereby forming small crystals, which we also observed. Salt crystals reflect light, reducing the amount of light absorbed by the leaf (Esteban et al., 2013). The higher SLA observed in S. anglica at high salinity and HL accommodates more room for salt glands and crystals, which can occupy a large proportion of the leaf surface area (Semenova et al., 2009), and may explain why SLA was higher and not lower as often reported (Lopez-Hoffman et al., 2006; Munns and Tester, 2008; Lum and Barton, 2020).
High soil salinity not only resulted in lower biomass, as it also affected the tissue concentration of leaf elements, especially of N and S. The negative relationship between N and biomass indicates that despite the application of fertilizer in our experiment, there were still nutrient limitations. This could be due to higher N requirements at high salinity (Bradley and Morris, 1992). In halophytes, N is used to produce compatible osmolytes such as proline (Slama et al., 2015); S. anglica, like most Spartina spp., uses S to produce dimethylsulphoniopropionate (DMSP). This could give S. anglica an advantage, as it would save N, yet we observed that leaf S was lower at high salinity in S. anglica. The allocation of S allocated to DMSP in halophytes varies from 40 % to 86 % (Slama et al., 2015; Bazzano and Elmer, 2017), so our results do not support the notion that DMSP was acting as a compatible osmolyte in S. anglica in our study. This is in accordance with previous work indicating that DMSP is generally a constitutive osmolyte in Spartina spp. in which the tissue concentration is independent of external salinity (Otte and Morris, 1994; Colmer et al., 1996; Slama et al., 2015), possibly conferring salt tolerance instead by intracellular transport and compartmentation (Otte and Morris, 1994; Otte et al., 2004).
A typical effect of high soil salinity is reduced stomatal conductance, limiting photosynthesis and growth (Munns and Tester, 2008). Lower stomatal conductance is likely to reduce photosynthesis, but the observed higher leaf N at high salinity could reduce the limiting effect of lower stomatal conductance on photosynthesis, by providing higher enzyme concentrations in leaves (Wright et al., 2001; Hinojo-Hinojo et al., 2018). At similar stomatal conductance and internal CO2 concentration, higher leaf N results in a larger drawdown of CO2 thereby improving water use. Pagter et al. (2009) showed for P. australis that photosynthesis did not significantly decrease, despite stomatal conductance doing so, when it was exposed to high soil salinity. Similar to N, P requirements can also increase at high salinity due to salinity-induced changes in membrane properties, where P is associated with membrane lipids or changes in energy fixation (Awad et al., 1990). High nutrient availability is also likely to reduce this higher P demand at high salinity, just as it apparently ameliorates stomatal-related photosynthetic limitation via enhanced N supply.
Our results, when related to expected increases in soil salinity, show that the competitive outcome between S. anglica and P. australis in the transition zone, and in other species with similar contrasts, could change in the future. This is because P. australis is able to suppress vegetation by attenuating light fluxes in its understorey (Hirtreiter and Potts, 2012) whereas in saline conditions lower biomass, height and shoot density reduce the ability to compete for light (Gaudet and Keddy, 1988; Craine and Dybzinski, 2013), while higher nutrient requirements probably add to this (Berendse, 1985). Interactions between high salinity and other factors that affect transpiration, such as high light fluxes, are likely to favour C4 species and other salt-tolerant species with low transpirational demands. Climate change will affect the temporal variation in soil salinity (Alvarez-Rogel et al., 1997; IPCC, 2014) and it is likely that this will result in high salinity periods in which S. anglica establishment is favoured, similar to how salt marsh plants have invaded upland forests (Woodruff et al., 2013). As all P. australis survived 28 ppt salinity, albeit being lower in stature than S. anglica and producing less biomass, P. australis may survive even higher salinity due to shading effects from S. anglica alleviating some of the adverse effects from high soil salinity on P. australis (Remorini et al., 2009).
Species growing below a dense canopy are able to survive low light fluxes, but this depends on their acclimation potential. The reduction in biomass compared to the reduction in light can serve as a broad, general assessment of all morphological and biochemical acclimation processes to the low light conditions. Assuming that the light flux is below or at the photosynthetic saturation level, shade cloth that reduced light fluxes by 50 % should lead to a similar reduction in biomass without enhanced light capture (Sterck and Schieving, 2011). The biomass reduction in S. anglica was 41 %, which indicates that S. anglica has little ability to enhance light capture in shaded conditions. For comparison, other species can maintain a higher degree of their biomass in low light conditions due to changes in leaf morphology and biochemistry (Kennedy et al., 2006; Guenni et al., 2018). Our observed changes in traits that increase light capture are small compared with other studies (Murchie and Horton, 1997; Poorter et al., 2012; Poorter et al., 2019). This indicates that S. anglica is an obligate sun species, which maintains sun-adapted photosynthetic traits to a high degree even at low light fluxes (Lambers et al., 2008) and explaining why it does not occur in the understorey of taller species. Total height, a trait important for light competition (Craine and Dybzinski, 2013), was not affected by the light treatments in S. anglica. This could be due to a trade-off as taller S. anglica would break easily due to waves in the pioneer zone (Vuik et al., 2018). Unlike S. anglica, P. australis was taller in shaded conditions than in full light, but responses in SLA, LMF and Chl to LL did not indicate that it had substantially greater acclimation potential than S. anglica. These results could also explain why S. anglica is not found in the understorey of P. australis, or other taller species with dense canopies, at low salinity (Hirtreiter and Potts, 2012).
In conclusion, our results have shown that the combination of high salinity reduced traits important for light competition but also increased the nutrient costs to produce tissue in P. australis, whereas this was more or less unaffected in S. anglica. This could constitute an advantage in the transition zone and lead to increased presence of S. anglica in European coastal wetlands under future climate change, as its low acclimation potential to light indicates that it is currently supressed from growing in the understorey of taller species due to shading effects. Compared to S. anglica, P. australis had similar low acclimation potential but this is probably less relevant for the competitive outcome due to it being taller at low salinity. Increased presence of S. anglica could be exacerbated if transpirational demands result in higher soil salinity.
SUPPLEMENTARY DATA
Supplementary data are available online at https://academic.oup.com/aob and consist of the following. Fig. S1: Average diel cycles in solar radiation, temperature and relative humidity. Fig. S2: The fraction of biomass allocated to stems and rhizomes and comparison of above- and below-ground dead biomass. Fig. S3: Response of chlorophyll a to b ratio and the ratio between carotenoids and total chlorophyll. Fig. S4: Changes in the C : N and Na : K ratio in leaf and root tissue. Fig. S5: Changes in the concentration of Ca, Mg and S in the leaf and root tissue. Fig. S6: Changes in the concentration of Fe and Mn in leaf and root tissue.
ACKNOWLEDGEMENTS
We thank the staff at Påskehøjgård research farm for assistance with plant propagation, the technical staff in our laboratory for support with plant culture and analysis, and the anonymous referees for their valuable comments. The authors declare no conflicts of interest.
FUNDING
The Sino-Danish Center for Education and Research funded E.J. and G.H.S., while the Carlsberg Foundation (grant number CF15-0330) funded F.E.
LITERATURE CITED
- Adler PB, Fajardo A, Kleinhesselink AR, Kraft NJB. 2013. Trait-based tests of coexistence mechanisms. Ecology Letters 16: 1294–1306. [DOI] [PubMed] [Google Scholar]
- Alvarez-Rogel J, Hernandez J, Silla RO, Alcaraz F. 1997. Patterns of spatial and temporal variations in soil salinity: Example of a salt marsh in a semiarid climate. Arid Soil Research and Rehabilitation 11: 315–329. [Google Scholar]
- Awad AS, Edwards DG, Campbell LC. 1990. Phosphorus enhancement of salt tolerance of tomato. Crop Science 30: 123–128. [Google Scholar]
- Barbier EB, Hacker SD, Kennedy C, Koch EW, Stier AC, Silliman BR. 2011. The value of estuarine and coastal ecosystem services. Ecological Monographs 81: 169–193. [Google Scholar]
- Bazzano M, Elmer W. 2017. Interactions and consequences of silicon, nitrogen, and Fusarium palustre on herbivory and DMSP levels of Spartina alterniflora. Estuarine Coastal and Shelf Science 198: 106–113. [Google Scholar]
- Belsky AJ, Mwonga SM, Amundson RG, Duxbury JM, Ali AR. 1993. Comparative effects of isolated trees on their undercanopy environments in high- and low-rainfall Savannas. Journal of Applied Ecology 30: 143–155. [Google Scholar]
- Berendse F. 1985. The effect of grazing on the outcome of competition between plant species with different nutrient requirements. Oikos 44: 35–39. [Google Scholar]
- Bradley PM, Morris JT. 1991. Relative importance of ion exclusion, secretion and accumulation in Spartina alterniflora Loisel. Journal of Experimental Botany 42: 1525–1532. [Google Scholar]
- Bradley PM, Morris JT. 1992. Effect of salinity on the critical nitrogen concentration of Spartina alterniflora Loisel. Aquatic Botany 43: 149–161. [Google Scholar]
- Britto DT, Kronzucker HJ. 2006. Futile cycling at the plasma membrane: a hallmark of low-affinity nutrient transport. Trends in Plant Science 11: 529–534. [DOI] [PubMed] [Google Scholar]
- Colmer TD, Teresa W-MF, Läuchli A, Higashi RM. 1996. Interactive effects of salinity, nitrogen and sulphur on the organic solutes in Spartina alterniflora leaf blades. Journal of Experimental Botany 47: 369–375. [Google Scholar]
- Craft C, Clough J, Ehman J, et al. 2009. Forecasting the effects of accelerated sea-level rise on tidal marsh ecosystem services. Frontiers in Ecology and the Environment 7: 73–78. [Google Scholar]
- Crain CM. 2007. Shifting nutrient limitation and eutrophication effects in marsh vegetation across estuarine salinity gradients. Estuaries and Coasts 30: 26–34. [Google Scholar]
- Craine JM, Dybzinski R. 2013. Mechanisms of plant competition for nutrients, water and light. Functional Ecology 27: 833–840. [Google Scholar]
- Curco A, Ibanez C, Day JW, Prat N. 2002. Net primary production and decomposition of salt marshes of the Ebre delta (Catalonia, Spain). Estuaries 25: 309–324. [Google Scholar]
- Easlon HM, Bloom AJ. 2014. Easy Leaf Area: automated digital image analysis for rapid and accurate measurement of leaf area. Applications in Plant Sciences 2: 1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emery NC, Ewanchuk PJ, Bertness MD. 2001. Competition and salt-marsh plant zonation: Stress tolerators may be dominant competitors. Ecology 82: 2471–2485. [Google Scholar]
- Engels JG, Jensen K. 2010. Role of biotic interactions and physical factors in determining the distribution of marsh species along an estuarine salinity gradient. Oikos 119: 679–685. [Google Scholar]
- Esselink P, Zijlstra W, Dijkema KS, van Diggelen R. 2000. The effects of decreased management on plant-species distribution patterns in a salt marsh nature reserve in the Wadden Sea. Biological Conservation 93: 61–76. [Google Scholar]
- Esteban R, Fernández-Marín B, Hernandez A, et al. 2013. Salt crystal deposition as a reversible mechanism to enhance photoprotection in black mangrove. Trees 27: 229–237. [Google Scholar]
- Gaudet CL, Keddy PA. 1988. A comparative approach to predicting competitive ability from plant traits. Nature 334: 242–243. [Google Scholar]
- Glenn EP. 1987. Relationship between cation accumulation and water content of salt-tolerant grasses and a sedge. Plant, Cell & Environment 10: 205–212. [Google Scholar]
- Grubbs FE. 1969. Procedures for detecting outlying observations in samples. Technometrics 11: 1–21. [Google Scholar]
- Guenni O, Romero E, Guedez Y, de Guenni LB, Pittermann J. 2018. Influence of low light intensity on growth and biomass allocation, leaf photosynthesis and canopy radiation interception and use in two forage species of Centrosema (DC.) Benth. Grass and Forage Science 73: 967–978. [Google Scholar]
- Hautier Y, Niklaus PA, Hector A. 2009. Competition for light causes plant biodiversity loss after eutrophication. Science 324: 636–638. [DOI] [PubMed] [Google Scholar]
- Hinojo-Hinojo C, Castellanos AE, Llano-Sotelo J, Penuelas J, Vargas R, Romo-Leon JR. 2018. High V-cmax, J(max) and photosynthetic rates of Sonoran Desert species: using nitrogen and specific leaf area traits as predictors in biochemical models. Journal of Arid Environments 156: 1–8. [Google Scholar]
- Hirtreiter JN, Potts DL. 2012. Canopy structure, photosynthetic capacity and nitrogen distribution in adjacent mixed and monospecific stands of Phragmites australis and Typha latifolia. Plant Ecology 213: 821–829. [Google Scholar]
- Huckle JM, Potter JA, Marrs RH. 2000. Influence of environmental factors on the growth and interactions between salt marsh plants: effects of salinity, sediment and waterlogging. Journal of Ecology 88: 492–505. [Google Scholar]
- IPCC. 2014. Climate Change 2014: Synthesis Report. In: Pachauri RK, Meyer LA, Core Writing Team , eds. Contribution of Working Groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. Geneva, Switzerland: IPCC, 151. [Google Scholar]
- Ke-Fu Z. 1991. Desalinization of saline soils by Suaeda salsa. Plant and Soil 135: 303–305. [Google Scholar]
- Kennedy S, Black K, O’Reilly C, Ní Dhubháin Á. 2006. The impact of shade on morphology, growth and biomass allocation in Picea sitchensis, Larix × eurolepis and Thuja plicata. New Forests 33: 139. [Google Scholar]
- Kiviat E. 2013. Ecosystem services of Phragmites in North America with emphasis on habitat functions. Aob Plants 5: 1–29. [Google Scholar]
- Kunstler G, Falster D, Coomes DA, et al. 2016. Plant functional traits have globally consistent effects on competition. Nature 529: 204–207. [DOI] [PubMed] [Google Scholar]
- Lambers H, Chapin SF III, Pons TL. 2008. Plant physiological ecology, 2nd edn. New York: Springer Science. [Google Scholar]
- Levine JM, Brewer JS, Bertness MD. 1998. Nutrients, competition and plant zonation in a New England salt marsh. Journal of Ecology 86: 285–292. [Google Scholar]
- Lichtenthaler HK. 1987. Chlorophylls and carotenoids – pigments of photosynthetic biomembranes. Methods in Enzymology 148: 350–382. [Google Scholar]
- Lichtenthaler HK, Buschmann C, Döll M, Fietz HJ, Bach T, Kozel U, Meier D, Rahmsdorf U. 1981. Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves. Photosynthesis Research 2: 115–141. [DOI] [PubMed] [Google Scholar]
- Lopez-Hoffman L, DeNoyer JL, Monroe IE, et al. 2006. Mangrove seedling net photosynthesis, growth, and survivorship are interactively affected by salinity and light. Biotropica 38: 606–616. [Google Scholar]
- Lum TD, Barton KE. 2020. Ontogenetic variation in salinity tolerance and ecophysiology of coastal dune plants. Annals of Botany 125: 301–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsushita N, Matoh T. 1992. Function of the shoot base of salt-tolerant reed (Phragmites communis Trinius) plants for Na+ exclusion from the shoots. Soil Science and Plant Nutrition 38: 565–571. [Google Scholar]
- McPhee CS, Aarssen LW. 2001. The separation of above- and below-ground competition in plants a review and critique of methodology. Plant Ecology 152: 119–136. [Google Scholar]
- Munns R, Tester M. 2008. Mechanisms of salinity tolerance. Annual Review of Plant Biology 59: 651–681. [DOI] [PubMed] [Google Scholar]
- Murchie EH, Horton P. 1997. Acclimation of photosynthesis to irradiance and spectral quality in British plant species: Chlorophyll content, photosynthetic capacity and habitat preference. Plant Cell and Environment 20: 438–448. [Google Scholar]
- Oliver S, Barber SA. 1966. An evaluation of the mechanisms governing the supply of Ca, Mg, K, and Na to soybean roots (Glycine max). Soil Science Society of America Journal 30: 82–86. [Google Scholar]
- Otte ML, Morris JT. 1994. Dimethylsulphoniopropionate (DMSP) in Spartina aterniflora Loisel. Aquatic Botany 48: 239–259. [Google Scholar]
- Otte ML, Wilson G, Morris JT, Moran BM. 2004. Dimethylsulphoniopropionate (DMSP) and related compounds in higher plants. Journal of Experimental Botany 55: 1919–1925. [DOI] [PubMed] [Google Scholar]
- Pagter M, Bragato C, Malagoli M, Brix H. 2009. Osmotic and ionic effects of NaCl and Na2SO4 salinity on Phragmites australis. Aquatic Botany 90: 43–51. [Google Scholar]
- Pennings SC, Grant MB, Bertness MD. 2005. Plant zonation in low-latitude salt marshes: disentangling the roles of flooding, salinity and competition. Journal of Ecology 93: 159–167. [Google Scholar]
- Poorter H, Niinemets U, Ntagkas N, et al. 2019. A meta-analysis of plant responses to light intensity for 70 traits ranging from molecules to whole plant performance. New Phytologist 223: 1073–1105. [DOI] [PubMed] [Google Scholar]
- Poorter H, Niklas KJ, Reich PB, Oleksyn J, Poot P, Mommer L. 2012. Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytologist 193: 30–50. [DOI] [PubMed] [Google Scholar]
- Raybould AF, Gray AJ, Lawrence MJ, Marshall DF. 1991. The evolution of Spartina anglica Hubbard, C.E. (Gramineae): origin and genetic variability. Biological Journal of the Linnean Society 44: 369–380. [Google Scholar]
- Remison SU, Snaydon RW. 1980. Effects of defoliation and fertilizers on root competition between Dactylis glomerata and Lolium perenne. Grass and Forage Science 35: 81–93. [Google Scholar]
- Remorini D, Melgar JC, Guidi L, et al. 2009. Interaction effects of root-zone salinity and solar irradiance on the physiology and biochemistry of Olea europaea. Environmental and Experimental Botany 65: 210–219. [Google Scholar]
- Sage RF, McKown AD. 2006. Is C4 photosynthesis less phenotypically plastic than C3 photosynthesis? Journal of Experimental Botany 57: 303–317. [DOI] [PubMed] [Google Scholar]
- Schoolmaster DR Jr. , Stagg CL. 2018. Resource competition model predicts zonation and increasing nutrient use efficiency along a wetland salinity gradient. Ecology 99: 670–680. [DOI] [PubMed] [Google Scholar]
- Semchenko M, Lepik A, Abakumova M, Zobel K. 2018. Different sets of belowground traits predict the ability of plant species to suppress and tolerate their competitors. Plant and Soil 424: 157–169. [Google Scholar]
- Semenova G, Fomina I, Biel K. 2009. Structural features of the salt glands of the leaf of Distichlis spicata ‘Yensen 4a’ (Poaceae). Protoplasma 240: 75–82. [DOI] [PubMed] [Google Scholar]
- Slama I, Abdelly C, Bouchereau A, Flowers T, Savouré A. 2015. Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Annals of Botany 115: 433–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterck F, Schieving F. 2011. Modelling functional trait acclimation for trees of different height in a forest light gradient: emergent patterns driven by carbon gain maximization. Tree Physiology 31: 1024–1037. [DOI] [PubMed] [Google Scholar]
- Villar R, Merino J. 2001. Comparison of leaf construction costs in woody species with differing leaf life-spans in contrasting ecosystems. New Phytologist 151: 213–226. [DOI] [PubMed] [Google Scholar]
- Vuik V, Suh Heo HY, Zhu Z, Borsje BW, Jonkman SN. 2018. Stem breakage of salt marsh vegetation under wave forcing: A field and model study. Estuarine, Coastal and Shelf Science 200: 41–58. [Google Scholar]
- Woodruff JD, Irish JL, Camargo SJ. 2013. Coastal flooding by tropical cyclones and sea-level rise. Nature 504: 44–52. [DOI] [PubMed] [Google Scholar]
- van Wijnen HJ, Bakker JP. 1999. Nitrogen and phosphorus limitation in a coastal barrier salt marsh: the implications for vegetation succession. Journal of Ecology 87: 265–272. [Google Scholar]
- Wright GC, Rao RCN, Farquhar GD. 1994. Water-use efficiency and carbon isotope discrimination in peanut under water deficit conditions. Crop Science 34: 92–97. [Google Scholar]
- Wright IJ, Reich PB, Westoby M. 2001. Strategy shifts in leaf physiology, structure and nutrient content between species of high- and low-rainfall and high- and low-nutrient habitats. Functional Ecology 15: 423–434. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.