Abstract
Purpose
Although uncommon, detection of BRAF V600E mutations in adult patients with glioblastoma has become increasingly relevant given the widespread application of molecular diagnostics and encouraging therapeutic activity of BRAF/MEK inhibitors.
Methods
We performed a retrospective study of adult glioblastoma patients treated at Dana-Farber Cancer Institute/Brigham and Women’s Hospital or Massachusetts General Hospital from January 2011 to July 2019 with an identified BRAF V600E mutation by either immunohistochemistry or molecular testing. Patient characteristics, molecular genomics, and preoperative MRI were analyzed.
Results
Nineteen glioblastoma patients were included, with median age at diagnosis of 41-years-old (range 22–69). Only 1/18 was IDH1/2-mutant; 10/17 had MGMT unmethylated tumors. The most common additional molecular alterations were CDKN2A/2B biallelic loss/loss-of-function (10/13, 76.9%), polysomy 7 (8/12, 66.7%), monosomy 10 (5/12, 41.7%), PTEN biallelic loss/loss-of-function (5/13, 38.5%) and TERT promoter mutations (5/15, 33.3%). Most tumors were well-circumscribed (11/14) and all were contrast-enhancing on MRI. Twelve patients eventually developed subependymal or leptomeningeal dissemination. Six patients were treated with BRAF/MEK inhibition following disease progression after standard of care therapy, with 4/6 patients showing partial response or stable disease as best response. Median time to progression after BRAF/MEK inhibition was 6.0 months (95% CI 1.2–11.8). Grade 1 skin rash was present in 2 patients, but no other adverse events were reported. Median OS for the entire cohort was 24.1 months (95% CI 15.7–38.9).
Conclusion
Understanding the natural history and features of BRAF V600E glioblastoma may help better identify patients for BRAF/MEK inhibition and select therapeutic strategies.
Keywords: Glioblastoma, BRAF, Molecular targeted therapy, MEK inhibitor, BRAF inhibitor
Introduction
Despite advances in our understanding of glioma biology, patients with glioblastoma continue to have dismal outcomes with 5-year survival rates of about 6% [1] and median survival of between 14 to 19 months [2, 3]. There has been increased implementation of molecular diagnostics in clinical care for identification of molecular markers, which in turn can clarify diagnosis and prognosis in CNS tumors. In addition, there is a high level of interest in the detection of potentially targetable oncogenic mutations in glioma.
Activating mutations in BRAF (v-raf murine viral oncogene homolog B1), resulting in the substitution of glutamic acid for valine at position 600 (V600E mutation), are commonly present in a number of cancers, in particular in melanoma and colorectal cancer. Among primary CNS tumors, BRAF V600 driver events have been frequently identified in pleomorphic xanthoastrocytoma (PXA), gangliogliomas, as well as in ~ 1.7% of adult glioblastoma [4], where they are particularly enriched in the epithelioid variant of glioblastoma [5].
Detection of BRAF V600E mutations in adults with glioblastoma, through increased availability of molecular diagnostics, could have potentially life-extending effects due to the encouraging therapeutic activity of BRAF and mitogen-activated protein kinase (MEK) inhibitors, their relatively high tolerability and encouraging safety profiles in melanoma [6]. There is therefore considerable interest in using BRAF/MEK inhibitors in patients with BRAF V600E mutant GBM. Several case studies have described success with combination BRAF/MEK inhibitors in treatment of high-grade gliomas [7, 8].
As the incidence of this mutation in GBM is low, the natural history of these tumors is not well described. While uncommon, there might be a higher representation of this subtype of GBM in young adults [9]. Survival characteristics are also lacking and recurrence of BRAF V600E-mutant tumors has previously been documented with leptomeningeal involvement, a relatively rare event in GBM (estimated 2–14%) [10, 11].
Given these promising therapeutic avenues, further recognition and characterization of this subtype of GBM, through clinical, imaging and other pathological features would be important. Of equal value would be the understanding of the natural history and prognosis of BRAF V600E mutant GBM. We investigated the demographics, clinical course, radiologic characteristics, and genetic mutations in an adult cohort of BRAF V600E mutant glioblastoma, which has not previously been well described.
Methods
Cohort selection
This retrospective study included adults (≥ 18-years-old) with a pathological diagnosis of newly diagnosed glioblastoma WHO grade IV, treated at Dana-Farber Cancer Institute/Brigham and Women’s Hospital or Massachusetts General Hospital from January 1, 2011 to July 31, 2019. Patients with BRAF V600E mutation on immunohistochemistry staining or molecular diagnostics were included after a review of an institutional pathology database.
Data analysis
All charts were reviewed and data were collected retrospectively for each patient, including: (1) demographic data; (2) clinical data (including all tumor-directed therapy); (3) radiological data; and (4) pathological and molecular information.
Imaging features
Qualitative MRI data was reviewed by three investigators (ML, KS, EG) using Visually Accessible Rembrandt Images (VASARI) feature set at the time of diagnosis. Leptomeningeal or subependymal dissemination was determined by imaging review or positive CSF cytology. For patients started on BRAF/MEK inhibition, response to treatment was assessed using the Response assessment in Neuro-oncology (RANO) criteria [2].
Pathological and molecular features
Histologic diagnosis was performed in accordance with the 2016 WHO classification [2]. Molecular features were evaluated, including IDH1/2 and BRAF V600E mutations, by IHC and/or NGS, and O6-methylguanine-DNA methyltransferase (MGMT) promoter methylation status. Targeted NGS sequencing was conducted using validated institutional platforms [12, 13] and analyzed for single nucleotide variants, indels, copy number variants, and structural variants. Copy number variants were further evaluated using micro-array-based comparative genomic hybridization.
Statistical analysis
Statistical analyses were performed using STATA version 16.0 software. Time-to-event analyses for overall survival (OS) were calculated using the Kaplan–Meier method from time of glioblastoma diagnosis to date of death. Progression-free survival (PFS) was calculated as treatment start date to date of progression or death. Difference in survival was compared between groups using the log-rank test. Patients lost to follow up were censored at the date of last contact.
Results
Patient characteristics
BRAF V600E mutations were identified in 19 patients with newly-diagnosed GBM. Sixteen (84.2%) patients were identified based on NGS and 3 (15.8%) were identified based on immunohistochemistry alone (NGS not performed). Median age at diagnosis was 41 years (range 22–69) and 68.4% (13/19) were female (Table 1).
Table 1.
Patient characteristics for 19 patients with glioblastoma harboring BRAF V600E mutation
| Total, n = 19 (%) | |
|---|---|
| Median age at diagnosis, year (range) | 41.0 (22–69) |
| Female sex | 13 (68.4%) |
| Race | |
| Caucasian | 14 (73.6%) |
| African-American | 2 (10.5%) |
| Other | 3 (15.8%) |
| KPS at baseline ≥ 80 (n = 18) | 14 (77.8%) |
| IDH 1/2wildtypea (n = 18) | 17 (94.4%) |
| MGMT promoter(n = 17) | |
| Methylated | 6 (35.3%) |
| Partially methylated | 1 (5.9%) |
| Unmethylated | 10 (58.8%) |
| Anatomical location | |
| Supratentorial | 18 (94.7%) |
| Midline | 2 (22.2%) |
| Multifocal | 1 (5.3%) |
| Extent of resection (n = 17) | |
| Gross total resection | 10 (58.8%) |
| Subtotal resection | 5 (29.4%) |
| Biopsy | 2 (11.8%) |
| First-line Standard-of-care chemoradiation and adjuvant TMZ | 16 (84.2%) |
| BRAF/MEK inhibition as salvage treatment (n = 6) | |
| BRAF inhibition alone | 1 (16.7%) |
| BRAF/MEK inhibition | 5 (83.3%) |
| Radiological features at diagnosis (n = 14) | |
| Circumscribed | 11 (78.6%) |
| Superficial location | 6 (42.9%) |
| Cortical involvement | 8 (57.1%) |
| Ependymal/leptomeningeal extension | 2 (14.3%) |
| Heterogeneous enhancement | 13 (92.9%) |
| Cystic appearance | 10 (71.4%) |
| Hemorrhagic appearance | 4 (28.6%) |
IDH mutation was assessed by next generation sequencing and/or immunohistochemistry
KPS Karnofsky Performance Status; IDH Isocitrate dehydrogenase; MGMT O6-methylguanine-DNA methyltransferase; TMZ temozolomide; BRAF B-Raf Proto-Oncogene; MEK mitogen-activated protein kinase
Pathological and molecular characteristics
The tumors displayed a variety of histomorphological appearances, including: 5 that displayed prominent or focal epithelioid features (one of which additionally displayed areas of neuronal differentiation that were synaptophysin positive); two with prominent giant cell components; one gliosarcoma and one GBM with spindled fascicular pattern; one with regional rhabdoid cytomorphology; and a sparsely sampled case with histology reminiscent of ependymoma. In all cases the final integrated histomolecular neuropatho-logical diagnosis was glioblastoma, WHO grade IV, and included consensus diagnosis for cases with non-canonical glioblastoma histological appearances. Only 1/18 (5.6%) was IDH1 (R132S) mutant, which would now be reclassified as a Grade IV IDH-mutant astrocytoma [14], and 10/17 (58.8%) were MGMT promoter unmethylated. BRAF V600E IHC had been performed in addition to NGS on 7 patients, with 6 (71.4%) interpreted by neuropathology as showing positive staining (Supplemental Fig. 1) and the 7th patient’s initial non-diagnostic pathology consisted only of fragments of focally mildly hypercellular brain tissue with rare atypical cells, in which the BRAF V600E IHC was negative and the positive molecular testing was conducted on the subsequent diagnostic resection specimen. An additional 3 cases were diagnosed by positive BRAF V600E IHC alone. The intensity and quality of BRAF V600E immunopositivity was variable across cases, and on molecular testing, although the BRAF V600E variant was commonly present at an allelic frequency (median 34.7%, interquartile range 20.1–47.8) that was suggestive of a clonal mutation, the variant allelic frequency was as low as 8%. On molecular analysis, the most common alterations were CDKN2A/2B biallelic loss/loss-of-function (76.9%, 10/13), polysomy 7 (66.7%, 8/12), monosomy 10 (41.7%, 5/12), PTEN biallelic loss/loss-of-function (38.5%, 5/13) and TERT promoter mutations (33.3%, 5/15). No EGFR amplification, EGFRvIII variants, or H3F3A mutations were identified (Fig. 1).
Fig. 1.
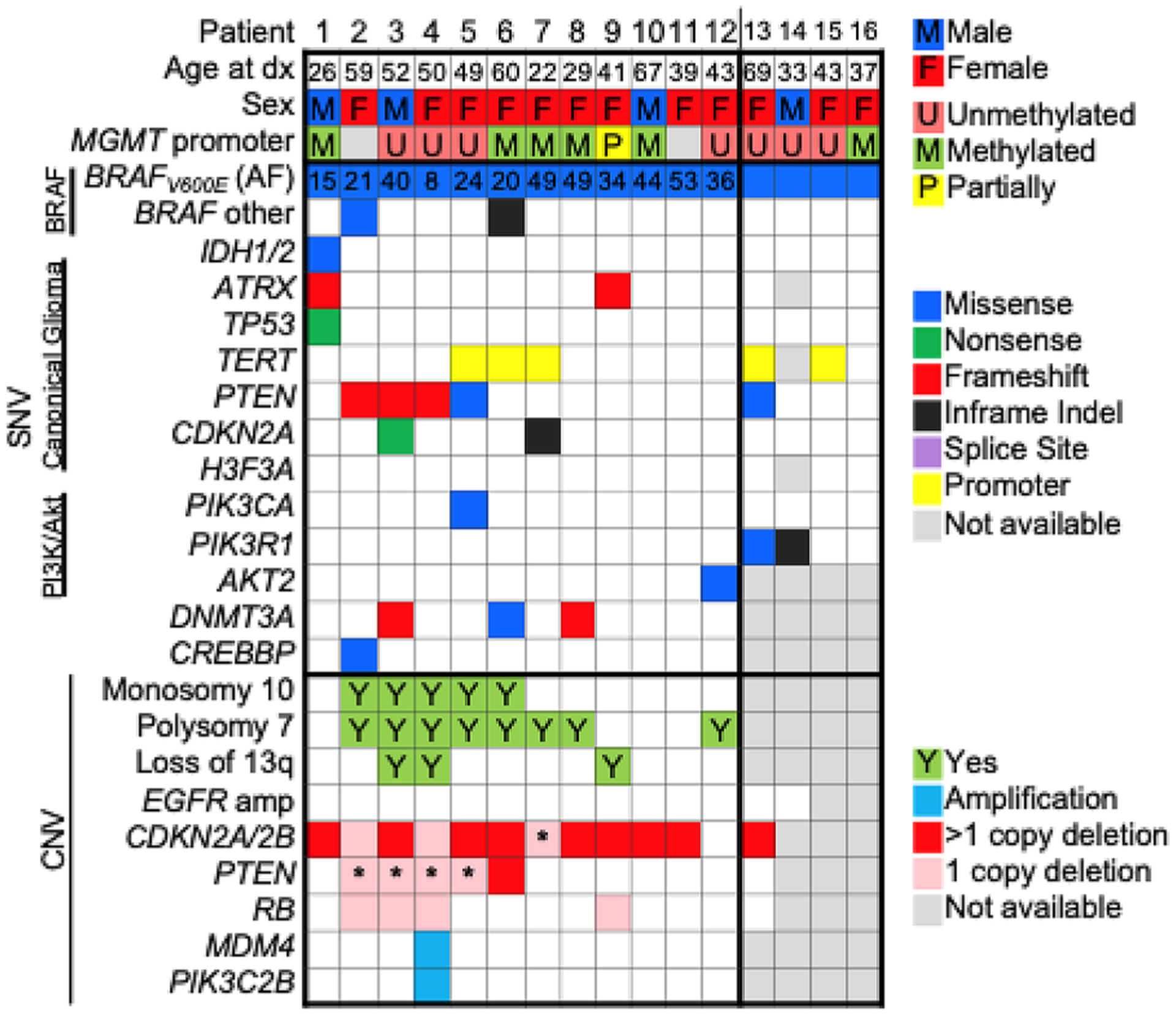
BRAF V600E glioblastoma patient demographics and molecular alterations. A comutation plot displaying the demographic and molecular characteristics of the 16 patients with BRAF V600E mutant glioblastoma that underwent next generation sequencing. The BRAF V600E variant was frequently present at a clonal allele frequency (AF; median 34.7%, interquartile range 20.1–47.8). No EGFR amplification, EGFRvIII variants, or H3F3A mutations were identified. *Single copy deletion concurrently with a loss-of-function single nucleotide variant is suggestive of biallelic inactivation
Radiological features and leptomeningeal metastasis
Fourteen patients were included for analysis of presurgical tumor imaging characteristics, summarized in Table 1. Most tumors were well circumscribed (78.6%, 11/14) and all were contrast-enhancing on MRI. While only 2 patients (14.3%) had leptomeningeal or ependymal involvement at diagnosis, 12/19 (63.2%) or 10 additional patients, developed subependymal or leptomeningeal dissemination at progression, including distal leptomeningeal metastasis to the spinal cord (n = 2) and the posterior fossa (n = 4). Two patients underwent a lumbar puncture for suspected leptomeningeal metastasis, and both had malignant cells present on cytology. Median number of months to leptomeningeal metastasis was 15.9 months (95% CI 3.13–35.20) and median OS after leptomeningeal metastasis was 4.7 months (95% CI 0.4–8.8). There was no significant difference in overall survival in the patients who developed leptomeningeal spread vs those who did not (24.1 months vs 21.1 months, p = 0.51) (Table 2).
Table 2.
Summary of treatment and objective response of BRAF/MEK inhibition in 6 patients
| Study ID number | BRAF/MEK inhibitor | Treatment Prior to BRAF/MEKi | Measurable tumor at initiation | Best response | Time to progression on inhibitor (months) | Overall survival (months) |
|---|---|---|---|---|---|---|
| 9 | Dabrafenib 150 mg BID, then 100 mg BID after 2 months/trametinib 2 mg QD, then 1.5 mg QD after 2 months | Concurrent RT/TMZ, 6 cycles of adjuvant TMZ | Yes | Stable disease | 4.7 | 26.7 |
| 10 | Vemurafenib 960 mg BID every other week, then 960 mg BID after 2 months | Concurrent RT/TMZ, 12 cycles of adjuvant TMZ | Yes | Stable disease | 1.7 | N/A |
| 12 | Dabrafenib150mg BID/trametinib 2 mg QD | Concurrent RT/TMZ, 5 cycles of adjuvant TMZ | Yes | Partial response | 6.6 | N/A |
| 13 | Dabrafenib 150 mg BID / trametinib 2 mg QD | Concurrent RT/TMZ, 6 cycles of adjuvant TMZ | Yes | Partial response | 10.9 (intermittently adherent) | 35.6 |
| 15 | Dabrafenib 150 mg BID/trametinib 2 mg QD (received for 13 days at 4 mg QD, then on hold due to decreased LVEF) | Concurrent RT/TMZ, 6 cycles of adjuvant TMZ, Avastin, Carboplatin, repeat RT | No | Progressive disease | 1.3 | 24.1 |
| 16 | Dabrafenib 150 mg BID/trametinib 2 mg QD | Concurrent RT/TMZ, 6 cycles of adjuvant TMZ | Yes | Partial response | 11.9 (intermittently adherent) | 39.7 |
BID twice daily, QD once daily, RT radiation therapy, TMZ temozolomide
BRAF/MEK inhibition
Six patients were treated with BRAF/MEK inhibition following progression after standard-of-care therapy. None received upfront treatment with BRAF or BRAF/MEK inhibition. Of these, 4 (66.7%) were treated on an “off-label” compassionate use basis, and 2 (33.3%) were enrolled in a clinical trial. One patient was on vemurafenib alone and 5 were on a combination of dabrafenib and trametinib. Half of the patients treated with BRAF/MEK inhibitor therapy (3/6) demonstrated a partial response (as illustrated in Fig. 2) and 1 experienced stable disease as their best response. Median time to progression after BRAF/MEK inhibitor therapy was 6.0 months (95% CI 1.3–11.8). There were no reported significant adverse events (AE) except for mild rash (CTCAE Grade 1) in 2 patients.
Fig. 2.
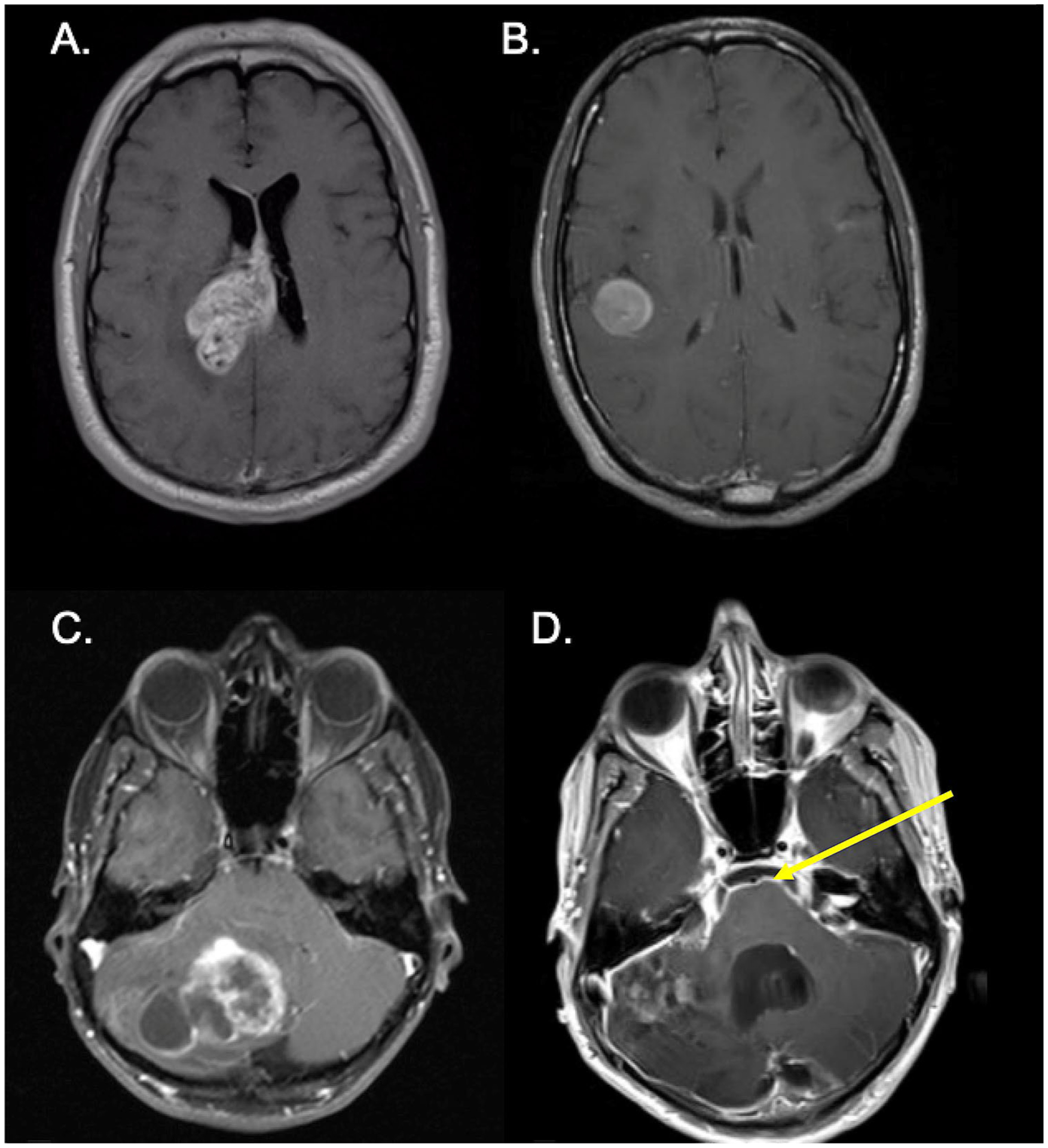
Radiological appearance of BRAF V600E mutant glioblastoma. MRI imaging of the brain showing subependymal (a) and subcortical (b) involvement of glioblastoma. Progression of initial cerebellar tumor (c) with leptomeningeal metastasis (yellow arrow; d)
Survival outcomes
Median OS for the entire cohort was 24.1 months (95% CI 15.7–38.9). Formal evaluation of the effect of BRAF/MEK inhibitor therapy on OS was limited by our small sample size but a non-statistically significant trend favoring longer OS was observed with BRAF/MEK inhibitor therapy; specifically, median OS for patients on BRAF/MEK inhibitor therapy was 35.6 months (95% CI 24.1–not estimable) compared to 17.0 months (95% CI 5.7–38.9) among patients who did not receive BRAF/MEK inhibitor therapy (p = 0.42) (Fig. 3).
Fig. 3.
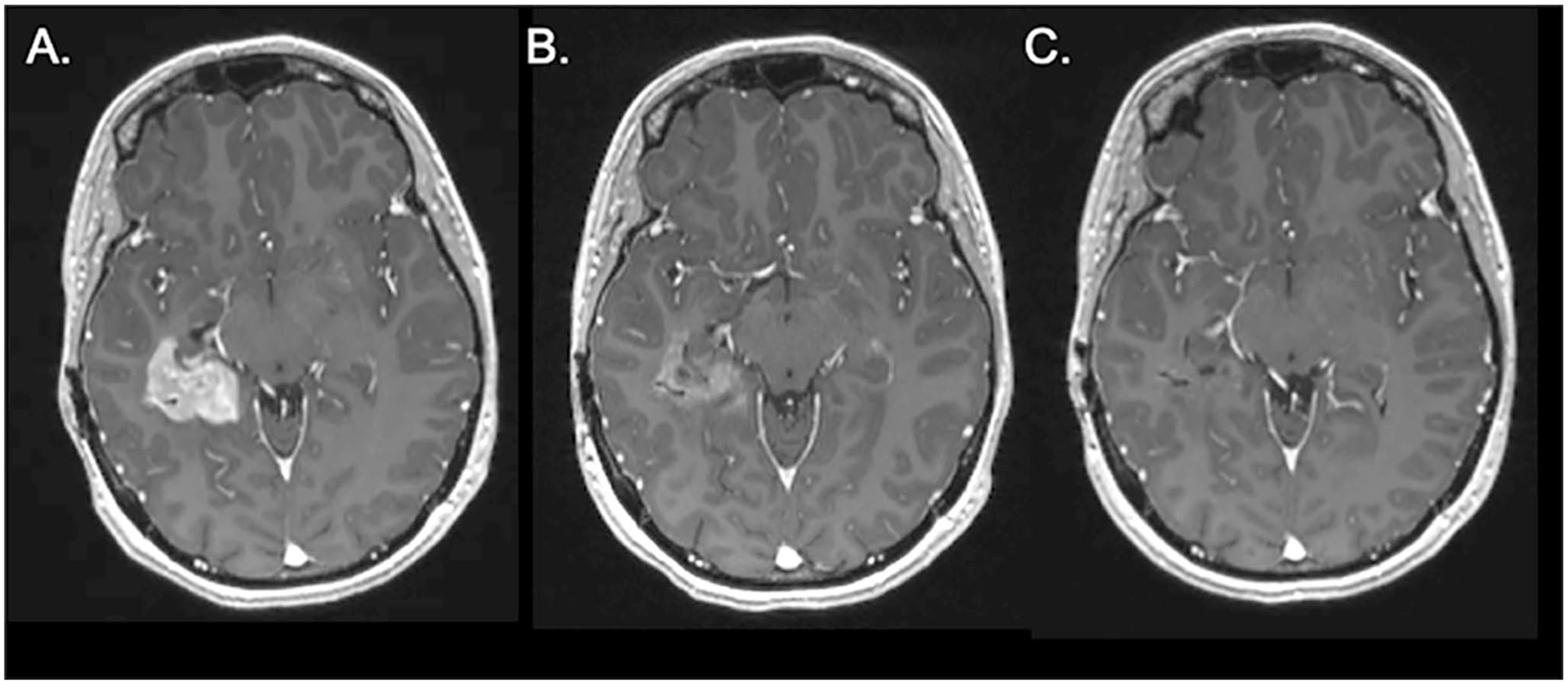
Radiographic responses to BRAF/MEK inhibitors for BRAF V600E mutant glioblastomas. An axial MRI post-contrast T1 sequence of a patient with BRAF V600E mutant glioblastoma at initiation of dabrafenib/trametinib (A). Follow-up scan shows partial response at 1 month (B) and near complete response at 3 months (C)
Discussion
Compared to the general glioblastoma patient population, which has a median age at diagnosis of 65 years and a median OS of 14.6 months [2], our cohort had a median age at diagnosis of 41 years and a median OS of 24.1 months. BRAF V600E mutations have been described in younger patients with glioma, although the majority are low grade, IDHwt gliomas [9, 15, 16]. BRAF V600E mutations are also particularly enriched (50–67%) in epithelioid GBM [5], which tend to affect younger patients with a reported median age of 25–30 [17, 18].
A study of pediatric patients with secondary BRAF V600E mutant high grade gliomas suggest that these tumors initially had increased time to malignant transformation to high grade glioma and improved survival [19]. Although our cohort included only newly diagnosed glioblastoma, BRAF V600E mutation status may be a better marker of prognosis in the younger adult IDHwt GBM population compared to histological grading. Indeed, historic cohorts have shown that IDHwt diffuse gliomas in pediatric and young adult cohorts with a BRAF V600E mutation, FGFR alterations or MYB or MYBL1 rearrangement tend to have a better prognosis and good overall survival, prompting unique considerations and characterization highlighted by the cIMPACT-NOW update 4 [20]. This prognostic difference highlights the importance of BRAF V600E mutation testing in young adults with IDHwt high grade gliomas. In terms of histopathology, although a substantial plurality of our cases exhibited epithelioid morphology, we found that BRAF V600E mutations were present in a wide variety of histological variants of GBM. Only one patient had an IDH mutant tumor, with a non-canonical IDH mutation, IDH1 R132S, which has been recurrently reported as a pathogenic variant in infiltrative gliomas (COSMIC). Patients with BRAF V600E mutations represent a distinct biological cohort from those harboring canonical IDH mutation in pediatric low grade gliomas, as these represent different oncogenic pathways [21]. A review of the available cohorts on cBio-Portal regrouping the TCGA and MSKCC glioma datasets also confirms that these driver events appear to be mutually exclusive in high grade glioma, although non-canonical IDH mutations are occasionally associated with BRAF V600E mutations. Compared to the pediatric patients where this appears to be more distinct, the presence of several driver events may be present in older patients with gliomas. The true significance of this co-mutation in our cohort remains unclear given the small sample size and our selection of only Grade IV gliomas.
In our study, seven patients (40%) had BRAF V600E IHC in addition to molecular testing, in which all diagnostic cases exhibited concordance between IHC and NGS panel testing. In one case, the IHC on the initial non-diagnostic tissue was negative, whereas the molecular testing of the diagnostic resection specimen was positive. The intensity and quality of staining varied across cases, and may reflect the BRAF V600E variant allele frequency. Taken together, the findings from our cohort and others’ studies support that BRAF V600E testing be considered for IDHwt GBM patients that are younger or exhibit epithelioid histology. For most institutions that do not routinely conduct targeted NGS panel testing of brain tumors, BRAF V600E IHC can be an inexpensive and widely-available screening tool, which if equivocal or negative, should prompt for molecular testing. The latter should preferably include targeted NGS panel testing, so as to also identify any additional potential therapeutic targets in young GBM patients or epithelioid GBMs.
Overall, the radiographic appearance of BRAF V600E mutations at diagnosis tended to be heterogeneous, although the majority were (1) well circumscribed, (2) heterogeneously enhancing and (3) had a cystic component. While none of these baseline features necessarily distinguish the BRAF V600E mutant tumors from other IDHwt GBMs, we observed that the majority of these patients had imaging evidence of leptomeningeal spread and in some, distant metastasis.
Leptomeningeal dissemination is a relatively rare phenomenon in glioblastoma with estimated rates of less than 5% [10]. In our cohort, 63% of patients exhibited evidence of subependymal or leptomeningeal dissemination on imaging, most frequently at the time of recurrence, suggesting a particular propensity for such phenomena among BRAF V600E mutant glioblastomas. Four patients in our cohort harbored co-existing TERT promoter and CDKN2A/CDKN2B mutations—genetic features commonly associated with BRAF V600E mutant gliomas laying on the spectrum between PXA and epithelioid glioblastoma [17], which have often been associated with leptomeningeal dissemination [22]. While early studies suggested that leptomeningeal dissemination might be a feature of disease duration, this is unlikely to be the only factor as the time to antemortem diagnosis of leptomeningeal spread in GBM is quite wide [23]. Intrinsic tumor characteristics rather than disease duration may also predict the propensity for leptomeningeal dissemination. Our study suggests that BRAF V600E may be a predisposing genetic factor. In this context, while whole spine imaging and CSF analyses for malignant cells are not usually part of routine screening in GBM patients, the threshold for doing so should be lower in patients harboring a BRAF V600E mutation.
Our cohort had an objective response rate of 50% to BRAF/MEK inhibition, all of which were partial responses. Two patients had recurrent disease with leptomeningeal spread at the time of initiation and both demonstrated a partial response to therapy. A prior case series had also described dramatic responses to dabrafenib in 3 patients with BRAF V600E recurrent gliomas and leptomeningeal disease [24]. BRAF/MEK inhibitors are widely available therapies, having demonstrated substantial effectiveness for patients with BRAF V600 mutant metastatic melanoma, including those with brain involvement [25]. Combined BRAF/MEK inhibition has been showed to improve both PFS and OS in metastatic melanoma [26]. This combination is thought to overcome the development of resistance through reactivation of the MAPK pathway. Notably, among 11 patients with malignant diffuse glioma enrolled in a basket trial of vemurafenib alone, one demonstrated a partial response and 5 patients experienced stable disease [27]. Additionally, some case series have observed similarly encouraging responses to BRAF/MEK targeted therapy in recurrent BRAF V600E mutant adult glioma [8], including a case of prolonged complete response with combination therapy after initial failure with BRAF-inhibition alone [28]. Encouragingly, preliminary results from an open-label basket trial of combination therapy with dabrafenib and trametinib (NCT02034110), which enrolled 37 patients with BRAF V600E mutant HGG, demonstrated an overall response rate of 26% including 1 complete response, and most responders displayed prolonged durations of response [29]. BRAF/MEK inhibiton was safely tolerated, with less than a 10% rate of serious adverse events (i.e. CTCAE Grade 3 or more). A number of trials are ongoing both in adult and pediatric patients with newly diagnosed and recurrent glioma (e.g. NCT03919071, NCT03973918, NCT02684058) to further evaluate the impact of BRAF/MEK inhibitor therapy among patients with BRAF V600E mutant gliomas.
Our findings suggest that BRAF V600E screening should be considered for IDHwt GBM patients who are young adults, have leptomeningeal dissemination, or display epithelioid histomorphology, in order to potentially identify patients for targeted BRAF V600E therapy.
Supplementary Material
Funding
No funding was received in support of this study.
Conflict of interest
M.J. Lim-Fat; K.W. Song; B.M. Andersen; D.A. Forst; E.R. Gerstner and I. Arrillaga-Romany report no disclosures relevant to the manuscript; J.B. Iorgulescu received support from the NIH (NCI K12CA090354) and Conquer Cancer Foundation/Sontag Foundation Young Investigator Award; J.T. Jordan received honoraria from Elsevier and Consulting for Navio Theragnostics, Recursion Pharmaceuticals, CereXis, Inc, and Health2047, Inc; D.A. Reardon received Research support (paid to DFCI) fromAcerta Phamaceuticals; Agenus; Celldex; EMD Serono; Incyte; Inovio; Midatech; Omniox; Tragara, advisory/consultation (paid to Dr. Reardon): Abbvie; Advantagene; Agenus; Amgen; Bayer; Bristol-Myers Squibb; Celldex; Del-Mar; EMD Serono; Genentech/Roche; Imvax; Inovio; Merck; Merck KGaA; Monteris; Novocure; Oncorus; Oxigene; Regeneron; Stemline; Taiho Oncology, Inc., Honoraria (paid to Dr. Reardon): Abbvie; Advantagene; Agenus; Bristol-Myers Squibb; Celldex; EMD Serono; Genentech/Roche; Imvax; Inovio; Merck; Merck KGaA; Monteris; Novocure; Oncorus; Oxigene; Regeneron; Stemline; Taiho Oncology, Inc; P.Y. Wen received honoraria from Agios, Astra Zeneca/Medimmune, Beigene, Celgene, Eli Lily, Genentech/Roche, Kazia, MediciNova, Merck, Novartis, Oncoceutics, Vascular Biogenics, VBI Vaccines (research support); Agios, Astra Zeneca, Bayer, Blue Earth Diagnostics, CNS Pharmaceuticals, Immunomic Therapeutics, Karyopharm, Kiyatec, Puma, Taiho, Vascular Biogenics, Deciphera, VBI Vaccines, Tocagen, Voyager, QED, Imvax, Elevate Bio (advisory board) and Merck, Prime Oncology (speaker honoraria).
Footnotes
Supplementary Information The online version contains supplementary material available at https://doi.org/10.1007/s11060-021-03719-5.
Ethical approval This study was conducted under a Dana-Farber/Harvard Cancer Center Institutional Review Board protocol. A waiver was granted for informed consent due to its retrospective nature.
References
- 1.Ostrom QT, Patil N, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS (2020) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013–2017. Neuro Oncol 22:iv. 10.1093/neuonc/noaa200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wen PY, Weller M, Lee EQ et al. (2020) Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol 22(8):1073–1113. 10.1093/neuonc/noaa106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lamba N, Chukwueke UN, Smith TR et al. (2020) Socioeconomic disparities associated with MGMT promoter methylation testing for patients with glioblastoma. JAMA Oncol 6(12):1972–1974. 10.1001/jamaoncol.2020.4937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brennan CW, Verhaak RGW, McKenna A et al. (2013) The somatic genomic landscape of glioblastoma. Cell 155(2):462. 10.1016/j.cell.2013.09.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kleinschmidt-Demasters BK, Aisner DL, Birks DK, Foreman NK (2013) Epithelioid GBMs show a high percentage of BRAF V600E mutation. Am J Surg Pathol 37(5):685–698. 10.1097/PAS.0b013e31827f9c5e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Long GV, Stroyakovskiy D, Gogas H et al. (2014) Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med 371(20):1877–1888. 10.1056/NEJMoa1406037 [DOI] [PubMed] [Google Scholar]
- 7.Johanns TM, Ferguson CJ, Grierson PM, Dahiya S, Ansstas G (2018) Rapid clinical and radiographic response with combined dabrafenib and trametinib in adults with BRAF-mutated high-grade glioma. JNCCN J Natl Compr Cancer Netw 16(1):4–10. 10.6004/jnccn.2017.7032 [DOI] [PubMed] [Google Scholar]
- 8.Schreck KC, Guajardo A, Lin DDM, Eberhart CG, Grossman SA (2018) Concurrent BRAF/MEK inhibitors in BRAF V600-Mutant high-grade primary brain tumors. JNCCN J Natl Compr Cancer Netw 16(4):343–347. 10.6004/jnccn.2017.7052 [DOI] [PubMed] [Google Scholar]
- 9.Dahiya S, Emnett RJ, Haydon DH et al. (2014) BRAF-V600E mutation in pediatric and adult glioblastoma. Neuro Oncol 16(2):318–319. 10.1093/neuonc/not146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Andersen BM, Miranda C, Hatzoglou V, Deangelis LM, Miller AM (2019) Leptomeningeal metastases in glioma: the Memorial Sloan Kettering Cancer Center experience. Neurology 92(21):e2483–e2491. 10.1212/WNL.0000000000007529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vertosick FT, Selker RG (1990) Brain stem and spinal metastases of supratentorial glioblastoma multiforme. Neurosurgery 27(4):516. 10.1097/00006123-199010000-00002 [DOI] [PubMed] [Google Scholar]
- 12.Dias-Santagata D, Akhavanfard S, David SS et al. (2010) Rapid targeted mutational analysis of human tumours: a clinical platform to guide personalized cancer medicine. EMBO Mol Med 2(5):146–158. 10.1002/emmm.201000070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garcia EP, Minkovsky A, Jia Y et al. (2017) Validation of oncopanel a targeted next-generation sequencing assay for the detection of somatic variants in cancer. Arch Pathol Lab Med 141(6):751–758. 10.5858/arpa.2016-0527-OA [DOI] [PubMed] [Google Scholar]
- 14.Brat DJ, Aldape K, Colman H et al. (2020) cIMPACT-NOW update 5: recommended grading criteria and terminologies for IDH-mutant astrocytomas. Acta Neuropathol 139(3):603–608. 10.1007/s00401-020-02127-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Behling F, Barrantes-Freer A, Skardelly M et al. (2016) Frequency of BRAF V600E mutations in 969 central nervous system neoplasms. Diagn Pathol 10.1186/s13000-016-0506-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ryall S, Zapotocky M, Fukuoka K et al. (2020) Integrated molecular and clinical analysis of 1,000 pediatric low-grade gliomas. Cancer Cell 37(4):569–583.e5. 10.1016/j.ccell.2020.03.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Korshunov A, Chavez L, Sharma T et al. (2018) Epithelioid glioblastomas stratify into established diagnostic subsets upon integrated molecular analysis. Brain Pathol 28(5):656–662. 10.1111/bpa.12566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lu VM, George ND, Brown DA et al. (2019) Confirming diagnosis and effective treatment for rare epithelioid glioblastoma variant: an integrated survival analysis of the literature. World Neuroncol. 10.1016/j.wneu.2019.08.007 [DOI] [PubMed] [Google Scholar]
- 19.Mistry M, Zhukova N, Merico D et al. (2015) BRAF mutation and CDKN2A deletion define a clinically distinct subgroup of childhood secondary high-grade glioma. J Clin Oncol 9:1015–1022. 10.1200/JCO.2014.58.3922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ellison DW, Hawkins C, Jones DTW et al. (2019) cIMPACT-NOW update 4: diffuse gliomas characterized by MYB, MYBL1, or FGFR1 alterations or BRAFV600E mutation. Acta Neuropathol 137(4):683–687. 10.1007/s00401-019-01987-0 [DOI] [PubMed] [Google Scholar]
- 21.Ryall S, Zapotocky M, Fukuoka K, Ellison DW, Tabori U, Hawkins Correspondence C (2020) Integrated molecular and clinical analysis of 1,000 pediatric low-grade gliomas. Cancer Cell. 10.1016/j.ccell.2020.03.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Broniscer A, Tatevossian RG, Sabin ND et al. (2014) Clinical, radiological, histological and molecular characteristics of paediatric epithelioid glioblastoma. Neuropathol Appl Neurobiol 40(3):327–336. 10.1111/nan.12093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arita N, Taneda M, Hayakawa T (1994) Leptomeningeal dissemination of malignant gliomas. Incidence, diagnosis and outcome. Acta Neurochir (Wien) 126(24):84–92. 10.1007/BF01476415 [DOI] [PubMed] [Google Scholar]
- 24.Burger MC, Ronellenfitsch MW, Lorenz NI et al. (2017) Dabrafenib in patients with recurrent, BRAF V600E mutated malignant glioma and leptomeningeal disease. Oncol Rep 38(6):3291–3296. 10.3892/or.2017.6013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hauschild A, Grob JJ, Demidov LV et al. (2012) Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380(9839):358–365. 10.1016/S0140-6736(12)60868-X [DOI] [PubMed] [Google Scholar]
- 26.Robert C, Grob JJ, Stroyakovskiy D et al. (2019) Five-year outcomes with dabrafenib plus trametinib in metastatic melanoma. N Engl J Med 381(7):626–636. 10.1056/NEJMoa1904059 [DOI] [PubMed] [Google Scholar]
- 27.Kaley T, Touat M, Subbiah V et al. (2018) BRAF inhibition in BRAFV600-mutant gliomas: results from the VE-BASKET study. J Clin Oncol 36:3477–3484. 10.1200/JCO.2018.78.9990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kushnirsky M, Feun LG, Gultekin SH, de la Fuente MI (2020) Prolonged complete response with combined dabrafenib and trametinib after BRAF inhibitor failure in BRAF-mutant glioblastoma. JCO Precis Oncol 4:44–50. 10.1200/po.19.00272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wen P, Alexander S, Yung-Jue B et al. (2018) Rare-09. Efficacy and safety of dabrafenib + trametinib in patients with recurrent/refractory braf v600e–mutated high-grade gliomA (HGG). Neuro Oncol 20:238. 10.1093/neuonc/noy148.986 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


