Abstract
Rutin is a unique antioxidant flavonoid that is mainly found in fruit, vegetables, cereals, and many other plant-based human diets. This review aims to highlight the in vitro anticancer properties of rutin including combination therapeutic strategies. Literature resources were gathered through PubMed, Scopus, Web of Science, and Google Scholar databases that cover the period of 1995–2021. Rutin is demonstrated to inhibit the proliferation of breast, colon, lung, and prostate cancers and other tumors. Furthermore, rutin alone or in combination with other therapeutic agents has been shown to regulate several signalling pathways involving the Ras/Raf and PI3K/Akt, MAPK, and TGF-β2/Smad2/3Akt/PTEN, etc., which are related to the processes of carcinogenesis and induction of apoptosis. The combination of rutin with other chemotherapy drugs may benefit on prevention of tumor cells by decreasing drug resistance and chemotherapy side effects. Moreover, rutin induces apoptosis synergistically with the therapeutic agent. More in vivo and clinical data are however needed to evaluate the true potential of rutin as an anticancer agent as an adjuvant. The present review highlights the effects of rutin which can be a promising candidate in combination with other antitumor drugs or alone for cancer treatment in vitro. Also, rutin can lead to decrease in drug resistance and chemotherapeutic side effects.
1. Introduction
1.1. Chemical Structure of Rutin as a Flavonoid
The 15-carbon basic structure of flavonoids consists of a three-ring system arranged as C6-C3-C6. This normally includes two aromatic rings (rings A and B) often in polyphenolic forms joined together with a three-carbon chain to form the third ring (ring C) in many flavonoids. Depending on the oxidation state, presence or absence of double bond, and the way ring C is connected with ring B, flavonoids are classified into different families such as flavonols, flavones, and flavanones [1]. Biosynthetically, the first group of flavonoids that are considered precursors are the chalcones. Naringenin chalcone is one of the first chalcone derivatives to be produced naturally and could give rise to flavanones such as naringenin (Figure 1). Flavones such as apigenin have low toxicity and multiple beneficial bioactivities. Rutin possess a double bond in ring C and further hydroxylation at the C-3 position gives rise to flavonols such as rutin and kaempferol (Figure 1). Quercetin is a derivative of flavonols with diorthohydroxyl group in the ring, while its ruinoside derivative at the C-3 position is called rutin (Figure 1) [2].
Figure 1.

The structure of rutin and related flavonoids.
Glycosyl flavonoids include the C-glycosides and O-glycosides depending on the sugar attachment bound as a C–C bond or C–O bond. Several C-glycoside flavones (e.g., at C-6 or C-8 position) such as rutin are more impressive in biological activities than O-glycosides flavone isomers [3, 4]. The various biological activities of flavonoids include therapeutic implications against diseases such as cancer, depression, diabetes, dementia, lupus, autism, and heart disease. They also exhibit strong antioxidative activity which is often considered as a mechanism of action for their healing functions in irregular biological systems such as cancer cells. Flavonoids such as rutin are found mainly in fruits, vegetables, grains, and many other human diets [5–7]. Several studies have shown that flavonoids as polyphenolic compounds possess antiproliferative and cytoprotective effects in addition to induction of apoptosis in different cancer cell lines [8].
Some flavonoids have been shown to regulate several signal proto-oncogene pathways. Proto-oncogenes have been shown to encode cell surface receptors, nuclear proteins, membrane proteins, growth factors, and phosphokinases. C-Fos, c-Jun, and c-Myc as three important proto-oncogenes are associated with both processes of carcinogenesis and inflammation. In the same way, rutin induced cytotoxicity through genotoxicity, mitochondrial apoptotic pathway, and relatively upstream apoptotic factors, including reduction of ROS generation and induction of antioxidant enzyme (AOE) (Figure 2) [9, 10].
Figure 2.
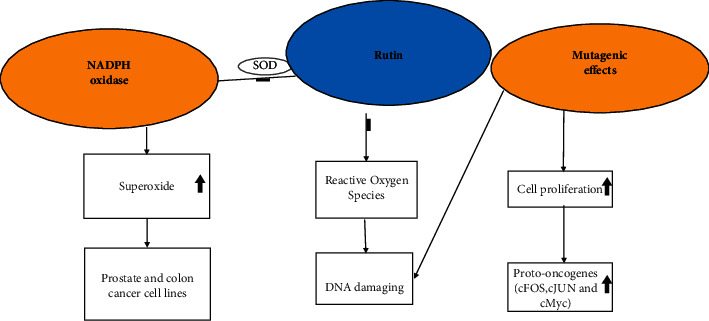
Regulation of activation of c-Fos and c-Jun proto-oncogenes on cancer cells.
Among the nuclear proteins coded by proto-oncogenes (c-Onc) are c-Myc, c-Fos, and c-Jun. These proteins have the potential to regulate other genes directly or indirectly, thereby influencing some gene expressions and apoptosis. This review aims to highlight the in vitro anticancer properties of rutin including combination therapeutic strategies: A Novel Treatment Strategy for Cancer Treatment.
1.2. Natural Occurrence and General Pharmacological Effects of Rutin
The metabolism, absorption, and antioxidant activity of flavonoids significantly differ from each other based on structural variations including the presence, nature, and position of glycosylation [11]. Among the phytochemicals with anticancer effects, rutin (3,3′,4′,5,7-pentahydroxyflavone-3-rhamnoglucoside: Figure 3) is one of the highly potent antioxidant agents, with some other biochemical activities in cancer prevention and treatment. Due to these biological effects, rutin is also known as vitamin P [5, 12]. More than 130 therapeutic drugs that have been existed are containing rutin in their structure. It is found in plants such as buckwheat, green tea, and apples and is widely used as medicine in China. Buckwheat is one of the major sources of rutin [13, 14]. The origin of rutin from the buckwheat plant can go back to the 1940s when buckwheat was used for medicinal purposes in the United States. Notably, the leaves and flowers of the plant were found to be in the highest concentration, nearly 2–10% of plant weight. The content of rutin at various parts of the plant considerably depends on its geographical source and genetic type [15, 16]. Moreover, it is demonstrated that more than 70 plant species are good sources of rutin and these include Ruta graveolens L. (Rutaceae) and Sophora japonica L. (Fabaceae), Strelitzia reginae Banks ex Aiton (Strelitziaceae), Maranta leuconeura (Marantaceae), Orchidantha maxillarioides (Lowiaceae), Eucalyptus spp. (Myrtaceae), Canna indica L. (Cannaceae), and Canna edulis Ker Gawl. (Cannaceae) [15, 17]. Up to 1.5% of rutin could also be extracted from tobacco leaves [18]. The biosynthesis of rutin is regulated by ultraviolet light and its accumulation serves as a protection. In this connection, when dill cell cultures were subjected to sunlight, the prominent flavonoid synthesized was shown to be quercetin-3-O-β-glucuronide. Both rutin and quercetin are major sources of pharmaceutical products for phytotherapy [19, 20].
Figure 3.
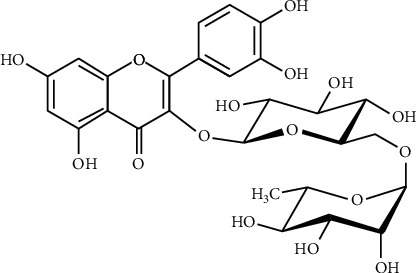
Chemical structure of rutin (84).
Many studies revealed rutin possesses anti-inflammatory, anticarcinogenic, neuroprotection, antiproliferative, antimetastatic, and antioxidative stress effects via inhibiting lipid peroxidation and amelioration of oxidative stress [21, 22]. Reactive oxygen species (ROS) can damage DNA, chromosomal mutations, unregulated gene expressions, cell division, and cell growth. Also, they could reduce the activities of some proteins involved in the antioxidant systems [23, 24]. Several studies have reported that ROS are associated with some tumors such as colon cancer, hepatocellular carcinoma, leukaemia, neuroblastoma, lung cancer, and breast cancer [12, 25–27]. Lipid peroxidation is the process by which oxygen combines with lipids to produce lipid hydroperoxides through the intermediate formation of peroxyl radicals and the cleavage of hydrogen. Unless checked by antioxidants such as rutin, lipid peroxidation can play a significant pathological role. Previous studies have shown that modified lipid composition could lead to cell apoptosis and other effects that could be targeted by rutin. Furthermore, rutin eliminating edema and reduction in sensitivity reduced ischemia-reperfusion damage. Rutin provides pharmacological information via platelet aggregation and cyclooxygenase-1 inhibition and its downstream pathway (Figure 4) [28, 29].
Figure 4.
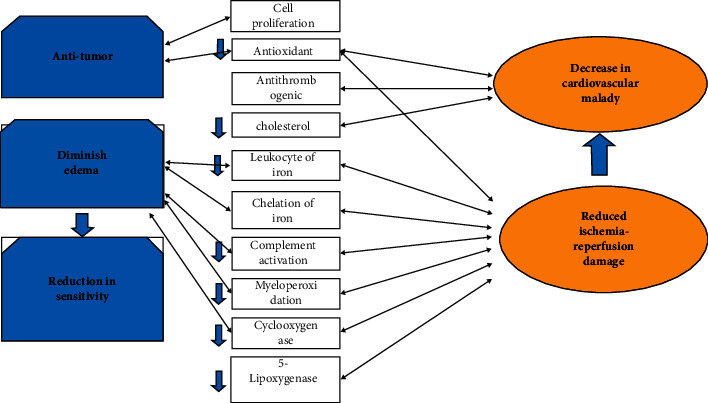
Rutin can reduce the expression of proinflammatory cytokines (TNF-α, IL-6, IL-1β, and COX-2) and downregulate inflammatory markers and antiproliferative impact. This anti-inflammatory activity is primarily related to the regulation on MAPKs and NF-κB signalling pathways by in vivo and in vitro data (66).
Therefore, rutin is considered a promising natural product in cancer prevention not only for its known efficacy but also accessibility and limited side effects.
2. Anticancer Properties of Rutin through Modulation of Signalling Pathways
Cancer is associated with the free radical generation and oxidative stress, fatty acid synthase (FAS) gene expression, and other mechanisms that lead to tumor via multiple modifications and mutations in cell proliferation and apoptosis genes controlling [1, 5]. Among the flavonoids with anticancer effects, rutin has one of the most widespread uses as an antioxidant and as an antitumor agent among the flavonoids because of its abundance in the human diet such as fruits and vegetables [30]. Here is a summary of its most important anticancer functions.
In some types of cancer, an oncogenic change induces an inflammatory microenvironment that promotes the development of cancers. The molecular pathways of this cancer-related inflammation are now being unravelled, resulting in the identification of new target molecules that could lead to improved diagnosis and treatment. There is also evidences to suggest that rutin exerts anti-inflammatory effects by downregulating the expression of cyclooxygenase-2, inducible nitric oxide synthase (iNOS), and suppression of lipid peroxidation. Also, rutin suppresses proinflammatory cytokines secretion [5, 31]. Nitric oxide is synthesized by a family of isoenzymes in normal cells. The high expression of stimulation nitric oxide synthase (NOS) via myeloid-derived suppressor cells (MDSCs) is a major signal of defence mechanism in cancer. NOS is also expressed by γδ T cells by contributing to their polarization to a protumor phenotype in cancer. Moreover, it has been indicated that rutin modulates activators of transcription, mitogen‐activated protein kinase (MAPK), PI3K/Akt, and Wnt/β‐catenin signalling cascades and Janus kinase/signal transducers in carcinogenic cells. The Ras/Raf and PI3K/Akt, MAPK, and TGF-β2/Smad2/3Akt/PTEN signalling pathways are stimulated through the epidermal growth factor (EGF) signalling pathway [14, 32, 33]. Rutin has been demonstrated to directly bind to the EGF receptor (EGFR) protein and arrest the downstream signalling factors [5]. They demonstrated that rutin reduces the expression of miR‐22‐5p an important microRNA in increasing the levels of RAP1/ERK signalling pathway‐related proteins that lead to apoptosis in in vitro and in vivo studies via Rap1a. Rap1a is one of the target genes of miR‐22‐5p [14, 34]. The present review highlights the anticancer potentials of rutin on cancer cell lines such as the colon, lung, breast, prostate, and HepG2 through various known molecular mechanisms including modulation of apoptosis genes in in vitro experimental models (Tables 1 and 2) [57].
Table 1.
The anticancer activities of rutin combination with other antioxidants or herbal ingredients.
| Compound (rutin with other agents) | Mechanism | Cancer cell line | Reference |
|---|---|---|---|
| Quercetin, myricetin | Protection of cancer cells against hydrogen peroxide | Caco-2 and HepG2 | [35, 36] |
| Quercetin | Decrease the activity of oxygen radical | Human hepatocellular carcinoma (HepG2) | [37–39] |
| AgNPs and natural bioactive compounds | Represent novel therapeutics fighting and synergistic, antimicrobial effects | Epithelioma papulosum cyprini (EPC) cell lines | [40] |
| Dietary polyphenols | Inducing cell cycle arrest or apoptosis, metastasis, and angiogenesis | Colorectal cancer cell lines | [41] |
| Fucoidan | Nuclear fragmentation, and mitochondrial potential loss, ROS generation | HaCaT (immortal noncancerous keratinocyte), HeLa (epithelial adenocarcinoma cells of the cervix) | [27] |
| Liquid crystalline nanoparticles | Inhibited the genes, namely, Nox2B and Nox4, which caused oxidative stress, upregulation in the expression of the antioxidant genes Gclc and Nqo-1 | Human bronchial epithelial cells (BEAS-2B) | [36, 42] |
| Quercetin | Reduce the enzymes precarcinogenic compounds such as CYP1A | Intestinal HCT‐8 Cells | [38, 42] |
| Diosmin and Tangeretin | Antimetastatic effects | Melanoma B16F10 cancer cells | [43] |
| Zinc(II) flavonoid-metal complex | Modulated expression of genes related to angiogenesis, cell cycle progression | Leukaemia (KG1, K562, and Jurkat), melanoma (B16F10 and SK-Mel-28), and multiple myeloma (RPMI8226) cell lines | [44] |
| Ionic liquids nanoparticles | Caused a significant increase in the sub-G1 population, a significant decrease in cell viability | Human renal cancer cell line (786-O) | [45] |
| Silibinin | Anticancer, apoptosis effects regulating the expressions of genes apoptosis, inflammation, and MAPK pathway proteins | Human colon cancer cell line | [46] |
| Poly (D, L-lactic-co-glycolic acid) (PLGA) nanospheres, Benzamide | Cell cycle disruption and apoptotic induction | MDA-MB-231 | [47] |
Table 2.
The anticancer activities of rutin combination with other therapeutically drugs as revealed by recent studies in vitro.
| Compound (rutin with other therapeutically drugs) | Mechanism | Cancer cell line | Reference |
|---|---|---|---|
| Apigenin, tamoxifen | Induce apoptosis through a p53-dependent pathway | Breast cancer cell line (MCF-7) | [48] |
| Cisplatin, isoquercetin | Inducing toxicity, could decrees cell count percentage in G0/G1, S, and G2/M phases in addition to increase it in sub G0 phase in comparison with the single doses | Gastric cancer | [49] |
| Hyperoside | Increase the expression of regulator positive apoptosis such as Bax, Caspase 3, 8, 9 | Colon cancer cells (HT-29) | [50] |
| 5-FU | Increase of p53 gene expression and decreases Bcl-2 protein expression | Prostate cancer cell (PC3) | [21] |
| Oxaliplatin | Induced apoptosis may be associated with the activation of the p38/caspase signal pathway | SGC-7901 gastric cancer cell line | [51] |
| Orlistat | Cytotoxic effects and promoting apoptosis | Breast cancer (MCF-7) and pancreatic cancer cell line (PANC-1) | [5] |
| Oxidovanadium(IV) | Inhibit superoxide, hydroxyl radicals and improved the antioxidant activity | Lung cancer A549 cell line | [52] |
| 5-FU and oxaliplatin | Antiproliferative effect and reduce possible adverse effects of these drugs | Caco-2 cancer cell line | [53] |
| Zein-co-acrylic acid hybrid hydrogels loaded 5-fluorouracil | Enhanced anticancer efficacy | MDA-MB-231 and MCF-7 breast cancer cell lines | [54, 55] |
| Doxorubicin (DOX) | Anticancer, protective effects | Human neuroblastoma cell line (IMR32) | [56] |
2.1. Rutin Induces Apoptosis and Arrests the Cell Cycle in Tumor Cells
Apoptosis is a process of eliminating damaged cells and is executed by a family of caspases. It is regulated by apoptotic gene expression such as p53 and Bax [58]. The p53 gene mutation is the most common genetic irregularities related to apoptosis in human tumors. Rutin (900 μm) was found to downregulate the expression of mutant p53 protein in human prostate cancer cell lines (DU-145, PC-3, and LNCaP) [21, 59].
The G1 and G2 phases of the cell cycle controlled by the p53 gene are a main regulatory mechanism in cellular proliferation [20]. Many studies demonstrated that rutin increases glutathione S-transferase P1 (GSTP1) polymorphism, Cyp1A1 cytochrome P450 (CYP1A1, CYP2D6) polymorphisms (inhibitory effect of antitumor genes), p53 (apoptosis gene expression), and Nqo-1 (antioxidant enzyme) leading to cell cycle arrest and consequently induction of apoptosis [21, 46, 60]. Also, it was reported that rutin could decrease Bcl-2 antiapoptosis gene expression and Bcl-2/Bax ratio along with a reduction in the levels of MYCN mRNA [61] (Figure 5). Moreover, other studies suggest that rutin can inhibit and regulate the cell cycle in the G2 and G1 phases. It stimulates apoptosis and decreases some of the oncogenic pathways including NF-κβ pathway and phosphorylation of the p38 MAP kinase [60, 62]. Therefore, these studies brought to light the unique roles of rutin in the prevention and treatment of cancer via the control of cellular proliferation and apoptosis induction [9].
Figure 5.
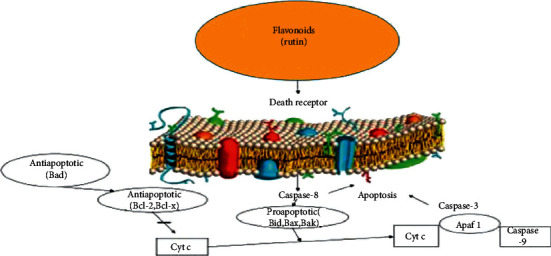
In a pathway independent of transcription, the mitochondrial p53 is bonded to Bcl-2 oncoprotein and Bcl-x proteins which reduce their inhibitory effect on the Bak and Bax apoptotic proteins. Bcl-2 promoted cell survival by blocking chemotherapy-induced apoptosis. Therefore, p53 leads to mitochondrial permeability and cytochrome C release consequently and activation apoptosis (21).
2.2. Anticancer Effect of Rutin: Single Therapy on Cancer Cell Lines
There are numerous in vitro studies that investigate the effect of rutin on the proliferation of cancer cell lines, such as the breast, colon, prostate, and lung. Rutin caused growth inhibition in human glioblastoma cell lines through induction of apoptosis and G2/M phase cell cycle arrest, as well as regulation of expression of the pro- and antiapoptotic genes (Bcl-2, Cas-3, Bax, and TP53). It also reduced mitochondrial membrane potential [62]. Rutin (400–700 mM/ml) has shown in vitro antiangiogenic properties on SW480 (human colon adenocarcinoma cell line) through cell cycle arrest at G1 phase and regulation of microRNAs (miRNAs), long noncoding RNAs (lncRNAs), messenger RNAs (mRNAs), and transcription factors (TFs) [26, 31]. Rutin as well as other antioxidants such as gallic acid, chlorogenic acid, coumaric acid, quercetin, and ferulic acid also induced cell apoptosis via a caspase-independent pathway [63]. In another cancer, osteosarcoma, it was further reported that rutin can stimulate osteoblast in human MG-63 cells [31, 64]. Other studies demonstrate that rutin exerts a notable reduction in tumor extent on human leukaemia (HL-60) and hepatic cells [65–67]. Also, rutin significantly inhibited the growth of LAN-5 and neuroblastoma cells through induction of apoptosis and cycle arrest at the G2/M phase [61]. Some studies indicated that rutin (810 μm) can cause a decrease in viability and consequently induce apoptosis in human liver cells [66, 68]. In breast cancer MDA-MB-231 cells, MCF‐7 cell line, and HTC hepatic cells, rutin has been shown to modulate the Wnt, JAK-STAT, EGF signalling, AP-1, NF-κB, and Akt [69, 70]. Therapeutically controlling cell signalling using different antioxidants has been demonstrated to have promise in cancer though it still needs more research [69]. In studies conducted by Karakurt on human hepatocytes (HepG2), rutin showed a regulatory role in the expression of cytochrome P450 and antioxidant enzymes [60]. Another study indicated a defensive effect of rutin on LLC‐PK1 cells (a porcine renal tubular epithelial cell line) that caused cell death and production of intracellular ROS [5, 71]. It also exerts in vitro cytotoxic effects on prostate cancer cells via increased caspase activity and apoptosis [72]. Moreover, rutin could be a candidate therapeutic agent for the treatment of human lung (A549) and colon (HT-29 and Caco-2) cancer cell lines by decreasing cell viability [53, 73]. Rutin treatment could significantly inhibit endothelial cell proliferation in vitro with the implication of angiogenesis inhibition [56]. So, rutin seems to be useful as an adjuvant in cancer therapy and the evidence shows that it alone has some anticancer potentials.
2.3. Rutin as a Combination Therapy with Antioxidants or Herbal Ingredients in Cancer Cells In Vitro
Combination therapy, the main treatment strategy that combines two or more therapeutic agents, is recently used by many investigators for cancer therapy. It has been shown that the mixture of antioxidants can promote antiproliferative, anticarcinogenic effects when compared to the monotherapy approach in cancer [48, 74, 75] (Table 1). The results have shown that rutin prenanoemulsion could inhibit cancer cells such as lung cancer cell line (A549) and colon cancer cell line. Also, the effects of rutin with antioxidants prevented protein modifications by lipid peroxidation products. Further, it induced rutin-protein adduct formation, which supports intra/extracellular signalling and the Nrf2/ARE antioxidant pathway, contributing to the protective effects against UV-induced oxidative stress [76].
Regulation of cellular signalling pathways such as Wnt/β‐catenin, p53‐independent pathway, apoptosis, JAK/STAT, PI3K/Akt, and MAPK as well as NF‐κB signalling pathways helps to mediate the anticancer effects of rutin. Exploration of these antitumor mechanisms can facilitate the development of this beneficial compound for its application in the treatment of cancers [77–79].
2.4. Rutin in Combination with Chemotherapy Drugs: In Vitro Studies
Randomized clinical trials have shown that the administration of antioxidants with chemotherapeutic drugs offers the possibility of better tumor protection and survival [74, 80]. Also, previous studies have demonstrated that rutin in combination with drugs can reduce the proliferation of many cancer cells through activation of apoptosis and arrest the cell cycle and many other mechanisms (Table 2). Therefore, the present review aimed to show the synergistic effects of rutin with anticancer drugs in cancer cell lines (Table 2). Rutin was demonstrated to target various inflammatory, apoptotic, autophagic, and angiogenic signalling mediators, including nuclear factor-κB, tumor necrosis factor-α, interleukins, light chain 3/Beclin, B cell lymphoma 2 (Bcl-2), Bcl-2 associated X protein, caspases, and vascular endothelial growth factor [40, 81]. A critical analysis of the anticancer potential of rutin and associated molecular targets among various cancer types has not been performed previously. Moreover, combination therapy can lead to a decrease in drug resistance and chemotherapeutic side effects [40, 78–82].
3. Applications of Novel Drugs and Limitations Associated with the Use of Some Flavonoids Such as Rutin
Great advances have been made on the development of novel drug combination systems for plant actives. The variety of novel herbal formulations like Rutin as a promising candidate in combination with other antitumor drugs or alone has been reported for cancer treatment by decreasing the exposure in normal tissues exploiting enhanced permeability and retention effect phenomenon by utilizing targeting strategies. Also, improve the efficacy of herbal cosmetic formulations on the human body. The mentioned formulations demonstrated that they have remarkable advantages of rutin which include enhancement of solubility, protection from toxicity, bioavailability enhancement of pharmacological activity, and stability. These criteria have improved tissue macrophages distribution and protection from physical and chemical degradation. Also, reduce medicinal doses and improve the absorbency of herbal medicines compared with single therapy [83–86].
Clinical studies have supported effects as a cardioprotective and neuroprotective agent. The combination of polyphenols with existing drugs also shows promising results and significantly reduces their toxicity. The major limitation to the use of these plants is the side effects associated with their use in high doses due to their neurotoxic side effect and cardiac function, apoptosis, and necroptosis. During the clinical study, we reported a mixture of rutin, diosmin, troxerutin, hesperidin, and quercetin in the treatment of I–III degree hemorrhoidal disease in different time can effectively mean managing bleeding from hemorrhoidal disease and minimal adverse events [84, 87, 88].
4. Conclusion
This review summarizes that rutin can be a promising candidate in combination with drugs for cancer treatment in vitro. It has been shown that the antitumor effect of rutin is related to interactions with signalling processes such as mixed-lineage protein kinase 3, Wnt, and mitogen-activated protein kinase. Furthermore, it demonstrated that it inhibits cell proliferation and regulates apoptosis and cell cycle in cancer cell lines and helps to design the best therapeutic strategies. However, more future studies are needed to understand the proper mechanism of action of rutin on cancer cell lines. Also, further investigations are needed to assess rutin in combination or alone on the expression of anti- or proapoptotic pathways and genes and other ingredients.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Abotaleb M., Samuel S. M., Varghese E., et al. Flavonoids in cancer and apoptosis. Cancers. 2019;11(1):p. 28. doi: 10.3390/cancers11010028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li H., Pan S., Xu X. Structure characteristics of flavonoids for cyclooxygenase-2 mRNA inhibition in lipopolysaccharide-induced inflammatory macrophages. European Journal of Pharmacology. 2019;856(1) doi: 10.1016/j.ejphar.2019.172416.172416 [DOI] [PubMed] [Google Scholar]
- 3.Chander M. Anticancer efficacy of some plant phenolics—a recent scenario. International Journal of Current Microbiology and Applied Sciences. 2018;7(10):1746–1768. doi: 10.20546/ijcmas.2018.710.200. [DOI] [Google Scholar]
- 4.Nijveldt R. J., van Nood E., Boelens P. G., van Norren K., van Leeuwen P. A. Flavonoids: a review of probable mechanisms of action and potential applications. American Journal of Clinical Nutrition. 2001;4(74):418–425. doi: 10.1093/ajcn/74.4.418. [DOI] [PubMed] [Google Scholar]
- 5.Nasrabadi P. N., Zali M. R. A detailed image of rutin underlying intracellular signaling pathways in human SW480 colorectal cancer cells based on miRNAs—lncRNAs—mRNAs—TFs interactions. Journal of Cellular Physiology. 2019;234(9):15570–15580. doi: 10.1002/jcp.28204. [DOI] [PubMed] [Google Scholar]
- 6.Albizia L. Isolation of flavonoids and pharmacognostical study of Iraqi. Journal of Pharmaceutical Sciences and Research. 2019;11(1):239–242. [Google Scholar]
- 7.Muller A. G., Sarker S. D., Saleem I. Y., Hutcheon G. A. Delivery of natural phenolic compounds for the potential treatment of lung cancer. DARU Journal of Pharmaceutical Sciences. 2019;27:433–449. doi: 10.1007/s40199-019-00267-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gęgotek A., Ambrożewicz E., Jastrząb A., Jarocka I., Elżbieta K. Rutin and ascorbic acid cooperation in antioxidant and antiapoptotic effect on human skin keratinocytes and fibroblasts exposed to UVA and UVB radiation. Archives of Dermatological Research. 2019;311(3):203–219. doi: 10.1007/s00403-019-01898-w. [DOI] [PubMed] [Google Scholar]
- 9.Chen G. The Molecular Basis for Tumor Growth Suppression by tRNA Methyltransferase 9-Like (TRM9L) Cambridge, MA, USA: MIT Libraries; 2017. [Google Scholar]
- 10.García-Lafuente A., Guillamo E., Villares A., Rostagno M. A., Martínez J. A. Flavonoids as anti-inflammatory agents: implications in cancer and cardiovascular disease. Inflammation Research. 2009;58(9):537–552. doi: 10.1007/s00011-009-0037-3. [DOI] [PubMed] [Google Scholar]
- 11.Mishra B., Priyadarsini K. I., Kumar M. S., Unnikrishnan M. K. Effect of O-glycosilation on the antioxidant activity and free radical reactions of a plant flavonoid chrysoeriol. Bioorganic & Medicinal Chemistry. 2003;11:2677–2685. doi: 10.1016/s0968-0896(03)00232-3. [DOI] [PubMed] [Google Scholar]
- 12.Ganeshpurkar A., Saluja A. K. The pharmacological potential of rutin. Saudi Pharmaceutical Journal. 2017;25(2):149–164. doi: 10.1016/j.jsps.2016.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Luk L., Ko T., Melini V., Acquistucci R., Germ M., Kreft I. Rutin and quercetin transformation during preparation of buckwheat sourdough bread. Journal of Cereal Science. 2016;69:71–76. doi: 10.1016/j.jcs.2016.02.011. [DOI] [Google Scholar]
- 14.Li X., Liu Z., Gu Y., Lv Z., Chen Y., Gao H. Expression of NF-kappaB and p38 under intervention of rutin in lung cancer therapy. Biomedical Research. 2017;28(5):2344–2347. [Google Scholar]
- 15.Suan L. A review on plant-based rutin extraction methods and its pharmacological activities. Journal of Ethnopharmacology. 2013;150(3):805–817. doi: 10.1016/j.jep.2013.10.036. [DOI] [PubMed] [Google Scholar]
- 16.Carraresi L., Thiele B., Groher T., Reimer J. J., Bro S. Tomato’s green gold: bioeconomy potential of residual tomato leaf biomass as a novel source for the secondary metabolite rutin. ACS Omega. 2019;4(21):19071–19080. doi: 10.1021/acsomega.9b01462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Prasad R., Prasad S. B. A review on the chemistry and biological properties of rutin, a promising nutraceutical agent. Asian Journal of Pharmaceutics. 2019;6:p. 3. [Google Scholar]
- 18.Al-Majmaie S., Nahar L., Sharples G. P., Wadi K., Sarker S. isolation and antimicrobial activity of rutin and its derivatives from ruta chalepensis (rutaceae) growing in Iraq. Records of Natural Products. 2019;13(1):1307–6167. doi: 10.25135/rnp.74.18.03.250. [DOI] [Google Scholar]
- 19.Mazzafera P. Rutin synthase in fava d’anta: purification and influence of stressors. Canadian Journal of Plant Science. 2009;89(5):895–902. doi: 10.4141/CJPS09001. [DOI] [Google Scholar]
- 20.Baghel S. S., Shrivastava N., Baghel R. S. A review of quercetin: antioxidant and anticancer properties a review of quercetin. Canadian Journal of Plant Science. 2009 [Google Scholar]
- 21.Satari A., Amini S. A., Raeisi E., Lemoigne Y., Hiedarian E. Synergetic impact of combined 5-fluorouracil and rutin on apoptosis in PC3 cancer cells through the modulation of P53 gene expression. Advanced Pharmaceutical Bulletin. 2019;9(2):462–469. doi: 10.15171/apb.2019.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang C., Shang S., Zheng X., et al. Fluorescent sensors based on Cu-doped carbon quantum dots for the detection of rutin. Journal of the Brazilian Chemical Society. 2019;30(5):988–996. doi: 10.21577/0103-5053.20180245. [DOI] [Google Scholar]
- 23.Nissanka N., Moraes C. T. Mitochondrial DNA damage and reactive oxygen species in neurodegenerative disease. FEBS Letters. 2018;592:728–742. doi: 10.1002/1873-3468.12956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kumari S., Badana A. K., Mohan G. M., Shailender G., Malla R. Reactive oxygen species: a key constituent in cancer survival. Biomarker Insights. 2018;6 doi: 10.1177/1177271918755391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Annapurna A., Reddy C. S., Akondi R. B., Rao S. R. C. Cardioprotective actions of two bioflavonoids, quercetin and rutin, in experimental myocardial infarction in both normal and streptozotocin-induced type I diabetic rats. Journal of Pharmacy and Pharmacology. 2009;61(10):1365–1374. doi: 10.1211/jpp/61.10.0014. [DOI] [PubMed] [Google Scholar]
- 26.Alonso-Castro A. J., Dom F., Garc A. Rutin exerts antitumor effects on nude mice bearing SW480 tumor. Archives of Medical Research. 2013;44:346–351. doi: 10.1016/j.arcmed.2013.06.002. [DOI] [PubMed] [Google Scholar]
- 27.Sathiya M., Thangam R., Salammal T., Sasirekha R., Sivasubramanian S., Dinesh M. Biomedicine & pharmacotherapy original article a novel rutin-fucoidan complex based phytotherapy for cervical cancer through achieving enhanced bioavailability and cancer cell apoptosis. Biomedicine & Pharmacotherapy. 2019;109:1181–1195. doi: 10.1016/j.biopha.2018.10.178. [DOI] [PubMed] [Google Scholar]
- 28.Agmon E., Solon J., Patricia B., Stockwell B. R. Modeling the effects of lipid peroxidation during ferroptosis on membrane properties. Scientic Report. 2018;8(1):p. 5155. doi: 10.1038/s41598-018-23408-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gutteridge J. M. C. Lipid peroxidation and antioxidants as biomarkers of tissue damage. Clinical Chemistry. 1995;41(12):1819–1828. doi: 10.1093/clinchem/41.12.1819. [DOI] [PubMed] [Google Scholar]
- 30.Patel K., Patel D. K. The beneficial role of rutin, a naturally occurring flavonoid in health promotion and disease prevention: a systematic review and update. In: Watson R. R., Preedy V. R., editors. Bioactive Food as Dietary Interventions for Arthritis and Related Inflammatory Diseases. Second. Cambridge, MA, USA: Academic Press; 2019. pp. 457–479. [DOI] [Google Scholar]
- 31.Al-Dhabi N. A., Arasu M. V., Park C. H., Park S. U. an up-to-date review of rutin and its biological and pharmacological activities. EXCLI Journal. 2015;14:59–63. doi: 10.17179/excli2014-663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Oberg H., Wesch D., Kalyan S., Kabelitz D. Regulatory interactions between neutrophils, tumor cells and T cells. Frontiers in Immunology. 2019;10:p. 1690. doi: 10.3389/fimmu.2019.01690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Italo C., Paolo C., Jacopo M., et al. Flavonoid mixture (diosmin, troxerutin, rutin, hesperidin, quercetin) in the treatment of I–III degree hemorroidal disease: a double-blind multicenter prospective comparative study. International Journal of Colorectal Disease. 2018;33(11):1595–1600. doi: 10.1007/s00384-018-3102-y. [DOI] [PubMed] [Google Scholar]
- 34.Ivanova I. A., Stoyanova D., Nenova E., Kostadinova A., Staneva A. Antimicrobial and cytotoxic properties of metal nanoparticles and graphene materials. Journal of Chemical Technology and Metallurgy. 2020;55(2):239–250. [Google Scholar]
- 35.Aherne S. A., O’Brien N. M. Protection by the flavonoids myricetin, quercetin, and rutin against hydrogen peroxide-induced DNA damage in caco-2 and hep G2 cells. Nutrition and Cancer. 1999;34(2):160–166. doi: 10.1207/s15327914nc3402_6. [DOI] [PubMed] [Google Scholar]
- 36.Grey C., Widén C., Adlercreutz P., Rumpunen K., Duan R. Antiproliferative effects of sea buckthorn (Hippophae rhamnoides L.) extracts on human colon and liver cancer cell lines. Food Chemistry. 2010;120(4):1004–1010. doi: 10.1016/j.foodchem.2009.11.039. [DOI] [Google Scholar]
- 37.Alía M., Mateos R., Ramos S., Lecumberri E., Bravo L., Goya L. Influence of quercetin and rutin on growth and antioxidant defense system of a human hepatoma cell line (HepG2) European Journal of Nutrition. 2006;45(1):19–20. doi: 10.1007/s00394-005-0558-7. [DOI] [PubMed] [Google Scholar]
- 38.Kim G., Jang H. Protective mechanism of quercetin and rutin using glutathione metabolism on H2O 2-induced oxidative stress in HepG2 cells. Annals of the New York Academy of Sciences. 2009;537:530–537. doi: 10.1111/j.1749-6632.2009.04690.x. [DOI] [PubMed] [Google Scholar]
- 39.Ramos A. A., Lima C. F., Pereira M. L., Fernandes-Ferreira M., Pereira-Wilson C. Antigenotoxic effects of quercetin, rutin and ursolic acid on HepG2 cells: evaluation by the comet assay. Toxicology Letters. 2008;177:66–73. doi: 10.1016/j.toxlet.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 40.Essawy E., Abdelfattah M. S., El-Matbouli M., Saleh M. Synergistic effect of biosynthesized silver nanoparticles and natural phenolic compounds against drug-resistant fish pathogens and their cytotoxicity: an in vitro study. Marine Drugs. 2021;19(1):p. 22. doi: 10.3390/md19010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Araújo J. R., Gonçalves P., Martel F. Chemopreventive effect of dietary polyphenols in colorectal cancer cell lines. Nutrition Research. 2011;31(2):77–87. doi: 10.1016/j.nutres.2011.01.006. [DOI] [PubMed] [Google Scholar]
- 42.Volková M. Modulatory effects of quercetin and rutin on the activity, expression and inducibility of CYP1A1 in intestinal HCT-8 cells. Phytotherapy Research. 2013;1893:1889–1893. doi: 10.1002/ptr.4992. [DOI] [PubMed] [Google Scholar]
- 43.Martínez Conesa C., Vicente Ortega V., Yáñez Gascón M. J., et al. Treatment of metastatic melanoma B16F10 by the flavonoids tangeretin, rutin, and diosmin. Journal of Agricultural and Food Chemistry. 2005;53(17):6791–6797. doi: 10.1021/jf058050g. [DOI] [PubMed] [Google Scholar]
- 44.Douguet L., Bod L., Labarthe L., et al. Inflammation drives nitric oxide synthase 2 expression by γδ T cells and affects the balance between melanoma and vitiligo associated melanoma. OncoImmunology. 2018;7(9):1–9. doi: 10.1080/2162402X.2018.1484979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Caparica R., Ana J., Eduarda M., et al. Anticancer activity of rutin and its combination with ionic liquids on renal cells. Biomolecules. 2020;10(2):p. 233. doi: 10.3390/biom10020233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nafees S., Mehdi S. H., Zafaryab M., Zeya B., Sarwar T. Synergistic interaction of rutin and silibinin on human colon cancer cell line. Archives of Medical Research. 2018;49(4):226–234. doi: 10.1016/j.arcmed.2018.09.008. [DOI] [PubMed] [Google Scholar]
- 47.Deepika M. S., Thangam R., Sheena T. S., et al. Dual drug loaded PLGA nanospheres for synergistic efficacy in breast cancer therapy. Materials Science and Engineering: C. 2019;103 doi: 10.1016/j.msec.2019.05.001.109716 [DOI] [PubMed] [Google Scholar]
- 48.Castillo R. R., Colilla M., Vallet-Regí M. Advances in mesoporous silica-based nanocarriers for co-delivery and combination therapy against cancer. Expert Opinion on Drug Delivery. 2017;14(2):229–243. doi: 10.1080/17425247.2016.1211637. [DOI] [PubMed] [Google Scholar]
- 49.Ghavami G., Muhammadnejad S., Amanpour S., Sardari S. Bioactivity screening of mulberry leaf extracts and two related flavonoids in combination with cisplatin on human gastric adenocarcinoma cells. Iranian Journal of Pharmaceutical Research. 2020;19(2):371–382. doi: 10.22037/ijpr.2020.1101087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Guon T. E., Chung H. S. Hyperoside and rutin of Nelumbo nucifera induce mitochondrial apoptosis through a caspase-dependent mechanism in HT-29 human colon cancer cells. Oncology Letters. 2016;11(4):2463–2470. doi: 10.3892/ol.2016.4247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li Q., Ren L., Zhang Y., et al. P38 signal transduction pathway has more cofactors on apoptosis of SGC-7901 gastric cancer cells induced by combination of rutin and oxaliplatin. BioMed Research International. 2019;2019:9. doi: 10.1155/2019/6407210.6407210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Goitia H., Quispe P., Naso L. G., et al. Interactions of rutin with the oxidovanadium (iv) cation. anticancer improvement effects of glycosylated flavonoids. New Journal of Chemistry. 2019;43(45):17636–17646. doi: 10.1039/c9nj01039d. [DOI] [Google Scholar]
- 53.Nasiri F., Kismali G., Alpay M., Kosova F., Cakir D. U., Sel T. Abstract 2177: rutin enhances the antiproliferative effect of 5-FU and oxaliplatin in colon cancer cells. Cancer Research. 2016;76(14):p. 2177. doi: 10.1158/1538-7445.am2016-2177. [DOI] [Google Scholar]
- 54.Kunjiappan S., Theivendran P., Baskararaj S., Sankaranarayanan B. Modeling a pH—sensitive zein—co—acrylic acid hybrid hydrogels loaded 5—fluorouracil and rutin for enhanced anticancer efficacy by oral delivery. Three biotech. 2019;9(5):1–20. doi: 10.1007/s13205-019-1720-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ramalingayya G. V., Cheruku S. P., Nayak P. G., et al. Rutin protects against neuronal damage in vitro and ameliorates doxorubicin-induced memory deficits in vivo in wistar rats. Drug Design, Development and Therapy. 2017;11:1011–1026. doi: 10.2147/DDDT.S103511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Guruvayoorappan C., Girija K. Antiangiogenic effect of rutin and its regulatory effect on the production of VEGF, IL-lß and TNF-0£ in tumor associated macrophages. Journal of Biomedical Science. 2007;8:1511–1519. doi: 10.3923/jbs.2007.1511.1519. [DOI] [Google Scholar]
- 57.You H. J., Ahn H. J., Ji G. E. Transformation of rutin to antiproliferative quercetin-3-glucoside by aspergillus Niger. Journal of Agricultural and Food Chemistry. 2010 27;58(20):10886–10892. doi: 10.1021/jf102871g. [DOI] [PubMed] [Google Scholar]
- 58.Kim-Campbell N., Gomez H., Bayir H. Cell death pathways: apoptosis and regulated necrosis. In: Ronco C., Bellomo R., Kellum J. A., Ricci Z., editors. Critical Care Nephrology. Third. Amsterdam, Netherlands: Elseiver; 2019. [Google Scholar]
- 59.Yamada K., Yoshida K. Mechanical insights into the regulation of programmed cell death by p53 via mitochondria. BBA—Molecular Cell Research. 2019;1866(5):839–848. doi: 10.1016/j.bbamcr.2019.02.009. [DOI] [PubMed] [Google Scholar]
- 60.Karakurt S. Modulatory effects of rutin on the expression of cytochrome P450s and antioxidant enzymes in human hepatoma cells. Acta Pharmaceutica. 2016;66:491–502. doi: 10.1515/acph-2016-0046. [DOI] [PubMed] [Google Scholar]
- 61.Chen W., Liu Y., Li M., et al. Anti-tumor effect of a-pinene on human hepatoma cell lines through inducing G2/M cell cycle arrest. Journal of Pharmacological Sciences. 2015;127(3):332–338. doi: 10.1016/j.jphs.2015.01.008. [DOI] [PubMed] [Google Scholar]
- 62.Kowalczyk T., Sitarek P., Skała E., Toma M. Induction of apoptosis by in vitro and in vivo plant extracts derived from Menyanthes trifoliata L in human cancer cells. Cytotechnology. 2019;9:165–180. doi: 10.1007/s10616-018-0274-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Frusciante L., Barone A., Maria D., Manuela M. Antioxidant bioactive compounds in tomato fruits at different ripening stages and their effects on normal and cancer cells. Journal of Functional Foods. 2015;18:83–94. doi: 10.1016/j.jff.2015.06.060. [DOI] [Google Scholar]
- 64.Kumar S. P., Choi S. J., Hoi Y., et al. Rutin from dendropanax morbifera leveille protects human dopaminergic cells against rotenone induced cell injury through inhibiting JNK and p38 MAPK signaling. Neurochemical Research. 2014;39(4):707–718. doi: 10.1007/s11064-014-1259-5. [DOI] [PubMed] [Google Scholar]
- 65.Yuan J., Li W., Tian Y., Wang X. I. A. Anti-proliferative effect of flos albiziae flavonoids on the human gastric cancer SGC-7901 cell line. Experimental and Therapeutic Medicine. 2013;5(1):51–56. doi: 10.3892/etm.2012.771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wu C., Lin M., Wang H., Yang M., Jou M., Wang C. Rutin inhibits oleic acid induced lipid accumulation via reducing lipogenesis and oxidative stress in hepatocarcinoma cells. Food Science. 2011;76:p. 2. doi: 10.1111/j.1750-3841.2010.02033.x. [DOI] [PubMed] [Google Scholar]
- 67.Carolina K., Araújo F., Maria E., Costa D. M. B., Pazini F., Campos M. Bioconversion of quercetin and rutin and the cytotoxicity activities of the transformed products. Food and Chemical Toxicology. 2013;51:93–96. doi: 10.1016/j.fct.2012.09.015. [DOI] [PubMed] [Google Scholar]
- 68.Marcarini J. C., Tsuboy M. S. F., Luiz R. C., Ribeiro L. R., Hoffmann-Campo C. B., Mantovani M. S. Investigation of cytotoxic, apoptosis-inducing, genotoxic and protective effects of the flavonoid rutin in HTC hepatic cells. Experimental & Toxicologic Pathology. 2011;63(5):459–465. doi: 10.1016/j.etp.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 69.Perk A. A., Shatynska-Mytsyk I., Gerçek Y. C., Bozta K., Yazgan M., Fayyaz S. Rutin mediated targeting of signaling machinery in cancer cells. Cancer Cell International. 2014;14:p. 124. doi: 10.1186/s12935-014-0124-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Iriti M., Kubina R., Cochis A., et al. Rutin, a quercetin glycoside, restores chemosensitivity in human breast cancer cells. Phytotherapy Research. 2017;1538(6):1529–1538. doi: 10.1002/ptr.5878. [DOI] [PubMed] [Google Scholar]
- 71.Qu S., Wang C., Wang H. Rutin attenuates vancomycin—induced renal tubular cell apoptosis via suppression of apoptosis, mitochondrial dysfunction, and oxidative stress. Phytotheraphy Research. 2019;33(8):2056–2063. doi: 10.1002/ptr.6391. [DOI] [PubMed] [Google Scholar]
- 72.Turan I., Demir S., Kilinc K., Arslan N. Antiproliferative and apoptotic effect of morus nigra extract on human prostate cancer cells. Saudi Pharmaceutical Journal. 2017;25(2):241–248. doi: 10.1016/j.jsps.2016.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ben Sghaier M., Pagano A., Mousslim M., Ammari Y., Kovacic H., Luis J. Rutin inhibits proliferation, attenuates superoxide production and decreases adhesion and migration of human cancerous cells. Biomedicine & Pharmacotherapy. 2016;84:1972–1978. doi: 10.1016/j.biopha.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 74.Khurana R. K., Jain A., Jain A., Sharma T., Singh B., Kesharwani P. Administration of antioxidants in cancer: debate of the decade. Drug Discovery Today. 2018;23(4):763–770. doi: 10.1016/j.drudis.2018.01.021. [DOI] [PubMed] [Google Scholar]
- 75.Mokhtari R. B., Homayouni T. S., Baluch N. Therapy in combating cancer. Oncotarget. 2017;8(23):38022–38043. doi: 10.18632/oncotarget.16723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hoai T. T., Yen P. T., Thi T., Dao B., Long L. H. Evaluation of the cytotoxic effect of rutin prenanoemulsion in lung and colon cancer cell lines. Journal of Materials. 2020;2020:11. doi: 10.1155/2020/8867669.8867669 [DOI] [Google Scholar]
- 77.Imani A., Maleki N., Bohlouli S., Sharifi S., Dizaj S. M. Molecular mechanisms of anticancer effect of rutin. Phytotherapy Research. 2020;(8):1–14. doi: 10.1002/ptr.6977. [DOI] [PubMed] [Google Scholar]
- 78.Ming Z. J., Hu Y., Qiu Y. H., Cao L., Zhang X. G. Phytomedicine synergistic effects of b-aescin and 5-fluorouracil in human hepatocellular carcinoma SMMC-7721 cells. Phytomedicine. 2010;17(8-9):575–580. doi: 10.1016/j.phymed.2009.12.009. [DOI] [PubMed] [Google Scholar]
- 79.Kang K. A., Lee J. H., Zhang R., Piao M. J., Chung H. S., Hyun J. W. Oryzadine, a new alkaloid of Oryza sativa cv. heugjinjubyeo, attenuates oxidative stress-induced cell damage via a radical scavenging effect. Food Chemistry. 2010;119(3):1135–1142. doi: 10.1016/j.foodchem.2009.08.026. [DOI] [Google Scholar]
- 80.Sjöström J., Blomqvist C., Mouridsen H., et al. Docetaxel compared with sequential methotrexate and 5-fluorouracil in patients with advanced breast cancer after anthracycline failure: a randomised phase III study with crossover on progression by the Scandinavian breast group. European Journal of Cancer. 1999;35(8):1194–1201. doi: 10.1016/s0959-8049(99)00122-7. [DOI] [PubMed] [Google Scholar]
- 81.Ryu M. J., Chung H. S. [10]-gingerol induces mitochondrial apoptosis through activation of MAPK pathway in HCT116 human colon cancer cells. In Vitro Cellular & Developmental Biology Animal. 2015;51(1):92–101. doi: 10.1007/s11626-014-9806-6. [DOI] [PubMed] [Google Scholar]
- 82.Nouri Z., Fakhri S., Nouri K., Wallace C. E., Farzaei M. H., Bishayee A. Targeting multiple signaling pathways in cancer : the rutin therapeutic approach. Cancers. 2020;12(8):p. 2276. doi: 10.3390/cancers12082276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Agrawal A. D. Pharmacological activities of flavonoids : a review. International Journal of Molecular Sciences. 2011;4(2):1394–1398. doi: 10.37285/ijpsn.2011.4.2.3. [DOI] [Google Scholar]
- 84.Pinzaru I., Tanase A., Enatescu V., et al. Proniosomal gel for topical delivery of rutin : preparation , physicochemical characterization and in vitro toxicological profile using 3D reconstructed human epidermis tissue and. Antioxidants. 2021;10(1):p. 85. doi: 10.3390/antiox10010085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Effects A. Polyphenols: extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules. 2016;21(7):p. 901. doi: 10.3390/molecules21070901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Saraf S. Fitoterapia applications of novel drug delivery system for herbal formulations. Fitoterapia. 2010;81(7):680–689. doi: 10.1016/j.fitote.2010.05.001. [DOI] [PubMed] [Google Scholar]
- 87.Shen C., Wang C., Han S., et al. Biochimica et biophysica acta aldehyde dehydrogenase 2 deficiency negates chronic low-to-moderate alcohol consumption-induced cardioprotecion possibly via ROS-dependent apoptosis and RIP1/RIP3/MLKL-mediated necroptosis. BBA—Molecular Basis of Disease. 2017;1863(8):1912–1918. doi: 10.1016/j.bbadis.2016.11.016. [DOI] [PubMed] [Google Scholar]
- 88.Ijeh I. I., Omodamiro O. D., Nwanna I. J. A study of aqueous and ethanolic extracts of Ocimum gratissimum and xylopia aethiopica and their antimicrobial activities. International Scholars Journal. 2021;9(1):1–4. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.


