Abstract
The traditional use of plants and their preparations in the treatment of diseases as a first medication in the past centuries indicates the presence of active components for specific targets in the natural material. Many of the tested plants in this study have been traditionally used in the treatment of Diabetes mellitus type 2 and associated symptoms in different cultural areas. Additionally, hypoglycemic effects, such as a decrease in blood glucose concentration, have been demonstrated in vivo for these plants. In order to determine the mode of action, the plants were prepared as methanolic and aqueous extracts and tested for their effects on intestinal glucose and fructose absorption in Caco2 cells. The results of this screening showed significant and reproducible inhibition of glucose uptake between 40 and 80% by methanolic extracts made from the fruits of Aronia melanocarpa, Cornus officinalis, Crataegus pinnatifida, Lycium chinense, and Vaccinium myrtillus; the leaves of Brassica oleracea, Juglans regia, and Peumus boldus; and the roots of Adenophora triphylla. Furthermore, glucose uptake was inhibited between 50 and 70% by aqueous extracts made from the bark of Eucommia ulmoides and the fruit skin of Malus domestica. The methanolic extracts of Juglans regia and Peumus boldus inhibited the fructose transport between 30 and 40% in Caco2 cells as well. These findings can be considered as fundamental work for further research regarding the treatment of obesity-correlated diseases, such as Diabetes mellitus type 2.
Keywords: plant extracts, traditional use, glucose, fructose, uptake inhibition, Diabetes mellitus, Caco2 cells
Introduction
In the last years, not only did the absolute number of people suffering from Diabetes mellitus increase from 108 Mio in 1980 to 422 Mio in 2014 but also the prevalence of this disease rose from 4.7 to 8.5% among adults during this time period worldwide (WHO, 2019). Apart from the estimated annual cost to the world of US$ 827 billion, Diabetes is associated with premature mortality and decreased quality of life (WHO, 2016). Hyperglycemia, as one of the main symptoms for diagnosis, results from decreased insulin secretion and/or reduced insulin action and can lead to long-term damage and dysfunction of different organs (American Diabetes Associa, 2014). Particularly, the most common kind of the disease, Diabetes mellitus type 2 (American Diabetes Associa, 2014), can be prevented or delayed in its progress by a lifestyle, which includes a healthy diet, regular exercise with moderate intensity, and no tobacco use (WHO, 2019).
With a focus on the diet, not only a decrease of energy intake to reduce obesity (Grundy, 1998) but also the selection of the consumed food can improve the health status. Supplements can be used to ensure the adequate intake of certain nutrients and support patient’s therapy (Ota and Ulrih, 2017). For example, the reduction of glucose, fructose, and saturated long-chain fatty acids and the increased intake of polyunsaturated, omega-3 fatty acids, such as eicosapentaenoic and docosahexaenoic acid, play a very important role in the prevention and improvement of metabolic disorders by alleviating inflammation processes (Sears and Perry, 2015). Regarding the consumption of plant-based food, secondary metabolites from plants showed hypoglycemic properties by affecting different targets, for example, modulation of intracellular insulin signaling pathways, increase in insulin secretion from β-cells, and inhibition of intestinal enzymes and transporters (Hanhineva et al., 2010; Bahadoran et al., 2013; Ota and Ulrih, 2017). In order to treat obesity as one of the main risk factors for Diabetes mellitus type 2 (Smith, 2007), the reduction of nutritional uptake into enterocytes as the last step before energy overload affects the body is an important therapeutic approach. Furthermore, the inhibition of enzymes and transporters leads to increased concentrations of nutrients in distal sections of the small intestine, which initiates the “ileal brake.” This complex mechanism is known to influence the digestive process and ingestive behavior resulting in reduced appetite and food intake (Maljaars et al., 2008). The glucose transport in the small intestine is maintained by the cotransporter SGLT1 (sodium-glucose linked transporter 1) and GLUT2 (glucose transporter 2), which belongs to the GLUT family and facilitates diffusion processes. The intestinal fructose uptake is performed by GLUT5 (glucose transporter 5) and GLUT2 as well (Schreck and Melzig, 2018). Medicinal plants traditionally used in the treatment of diseases have been applied as candidates for drug discovery in the last decades (Fabricant and Farnsworth, 2001). Aspirin, atropine, ephedrine, digoxin, and morphine are examples of effective substances, which owe their discovery to investigative studies of folk medicine (Gilani and Atta-ur-RahmanTrends, 2005). Ethnopharmacology studies show the continuing acceptance of traditionally used plant preparations as a therapy option due to their long-term experience and marginal side effects (Fabricant and Farnsworth, 2001; Gilani and Atta-ur-RahmanTrends, 2005). The plants tested in this study were chosen due to their traditional use in some cultural areas and countries during the last centuries and their hypoglycemic effects, which were discovered in animal or human studies, as listed in Table 1. For example, in Jordan, preparations of Allium sativum, Ceratonia siliqua, Cuminum cyminum, Juglans regia, Nigella sativa, Olea europaea, Sarcopoterium spinosum have been used in the treatment of Diabetes mellitus (Al-Aboudi and Afifi, 2011), whereas in Congo, Brassica oleracea and Citrus limon have been prepared as folk medicine to treat Diabetes mellitus (Katemo et al., 2012). The plants Adenophora triphylla, Cornus officinalis, Crataegus pinnatifida, Eucommia ulmoides, Lycium chinense, Pueraria lobata, and Rosa rugosa belong to the pharmacopoeia of the Traditional Chinese Medicine (Stöger, 2009). Although Adenophora triphylla and Crataegus pinnatifida showed hypoglycemic effects in studies, the traditional use as antidiabetic remedy was not confirmed in English-language literature as shown in Table 1. According to Table 1, some of the plants, such as Olea europaea and Brassica oleracea, were used in different areas of the world supporting the assumption of efficacy in the treatment of the metabolic disorder. The plants used in this study have been already tested in animal experiments and hypoglycemic properties such as a decrease in blood glucose were confirmed as shown in Table 1, but the exact mechanisms still need to be studied. In order to determine the mode of action, methanolic and aqueous plant extracts were screened for their inhibitory activity on intestinal transporters using Caco2 cells, which originally stem from a colorectal adenocarcinoma cell line. After confluence, the cells need 15–21 days to differentiate and fully express intestinal characteristics, such as a brush border membrane with enzymes and transporters. The adenocarcinoma cell line is widely used as a model for intestinal transport studies (Chantret et al., 1988; Hidalgo et al., 1989; Jumarie and Malo, 1991; Sambuy et al., 2005). We have determined the expression of the SGLT1, GLUT2, and GLUT5 transporters in the Caco2 cells during 21 days (Schreck and Melzig, 2019) and adapted the experimental conditions to ensure valuable results.
TABLE 1.
Selected plants, which are/were traditionally used in specific countries and their antidiabetic effects measured in studies and discussed active compounds (n.f. = not found in English-language literature for this indication; n.n. = not named).
| Scientific plant name | Countries/areas with traditional use of plant preparations as antidiabetic remedy and adjuncts to conventional treatments in the therapy of Diabetes mellitus (reference) | Selected, antidiabetic effects of traditionally used plants measured in studies | Discussed active compounds | Reference for antidiabetic effects and discussed active compounds |
|---|---|---|---|---|
| Adenophora triphylla (thunb.) ADC. | n.f. | Decrease of blood glucose, inhibition of intestinal glucose absorption in rats and mice | n.n. | Sanae et al. (1996) |
| Allium sativum L. | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020), India Rizvi and Mishra (2013), United Kingdom Swanston-Flatt et al. (1991) | Decrease of serum glucose and triglycerides, increase of serum insulin levels in diabetic rats | Allicin-type compounds | Eidi et al. (2006) |
| Aronia melanocarpa michx. elliott | n.f. | Decrease of serum glucose and lipids, reduced α-glucosidase activity | Anthocyanins | Banjari et al. (2017) |
| Artemisia dracunculus L. | United Kingdom Swanston-Flatt et al. (1991) | Decrease of elevated blood glucose level and blood insulin concentrations in diabetic mice | Flavonoids (luteolin, apigenin), coumarins (scopoletin), sesquiterpenoid lactones (costunolide), cinnamates | Ribnicky et al. (2006) |
| Brassica oleracea L. | Congo Katemo et al. (2012), Morocco Idm’hand et al. (2020), United Kingdom Swanston-Flatt et al. (1991) | Decrease of blood glucose in diabetic rats, decrease of blood lipids, and restoration of renal function in rats | n.n. | Kataya and Hamza (2008); Assad et al. (2014); Shah et al. (2016) |
| Camellia sinensis(L.) kuntze (Assam) | Morocco Idm’hand et al. (2020) | Decrease of blood glucose level in rats/mice, reduced α-glucosidase, α-amylase, and lipase activity | Catechins, flavanols, polysaccharides | Gomes et al. (1995); Han et al. (2011); Wang et al. (2017) |
| Camellia sinensis(L.) kuntze (Darjeeling) | ||||
| Camellia sinensis(L.) kuntze (gunpowder) | ||||
| Camellia sinensis(L.) kuntze (Sencha) | ||||
| Ceratonia siliqua L | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020) | Decrease of blood glucose in rats, reduced α-glucosidase, α-amylase activity, inhibition of intestinal glucose transport | Polyphenolic compounds | Rtibi et al. (2017) |
| Citrus limon (L.) osbeck | Congo Katemo et al. (2012), United Kingdom Swanston-Flatt et al. (1991) | Decrease of blood glucose in rats | n.n. | Naim et al. (2012) |
| Coffea arabica L. | n.f. | Decrease of blood glucose in rats, reduced glucose uptake, stimulated insulin secretion | Caffeine, polyphenolic compounds: chlorogenic acid, quinolactones | Campos-Florian et al. (2013) |
| Cornus officinalissiebold and zucc. | China Li et al. (2004); Ma et al. (2014) | Decrease of blood glucose in mice, reduced α-glucosidase activity, increased glucose uptake in HepG2 | Morroniside, loganin, ursolic acid | He et al. (2016) |
| Crataegus pinnatifida bunge | n.f. | Decrease of hyperglycemia in rats, modulation of insulin regulation, anti-obesity effect, anti-hyperlipidemia effect, reduced rats’ α-glucosidase activity | Flavonoids, hyperosid, chlorogenic acid, tripenic acids | Dehghani et al. (2019) |
| Cuminum cyminum L. | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020) | Decrease of hyperglycemia, oxidative stress, and AGE formation in rats, reduced α-glucosidase activity | Cuminaldehyde, flavonoids | Jagtap and Patil (2010) |
| Cynara cardunculus L. | Morocco Idm’hand et al. (2020); Tahraoui et al. (2007) | Decrease in blood glucose and serum lipid levels in humans and rats | Flavonoids | Nazni et al. (2006); Heidarian and Soofiniya (2011) |
| Eucommia ulmoides oliv. | China, Japan, Korea He et al. (2014) | Decrease in blood glucose and increase in plasma insulin in rats, decreased plasma lipid levels in mice, reduced glycation | Flavonoids | Kim et al. (2004); Lee et al. (2005); Park et al. (2006) |
| Hibiscus sabdariffa L. | Morocco Idm’hand et al. (2020) | Decreased hyperglycemia, hyperinsulinemia, serum lipids, and AGE formations in rats | Polyphenolic compounds | Peng et al. (2011) |
| Ilex paraguariensis a. st.-hil | South America de Freitas Junior and de Almeida (2017) | Decrease of serum lipids and glucose in mice, modulation of food intake | Polyphenolic compounds, methylxanthines, saponins | Kang et al. (2012) |
| Juglans regia L. | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020) | Decrease of fasting blood glucose, HbA1c, and fasting blood lipids in humans | Phenolic acids and flavonoids: 3- and 5-caffeoylquinic acids, quercetin-3-galactoside, quercetin-3-arabinoside | Hosseini et al. (2014) |
| Lycium chinense mill. | China Li et al. (2004) | Decrease of blood glucose and attenuation of dyslipidemia in rats | Polyphenolic compounds | Olatunji et al. (2017) |
| Malus domestica borkh. | Morocco Idm’hand et al. (2020) | Decrease of postprandial blood glucose in mice and humans, decrease of glucose absorption in mouse intestine, inhibition of human SGLT1 in X. laevis oocytes, inhibition of lipase in vitro, and decrease of plasma triglycerides in mice and humans | Polyphenolic compounds: quercetin, phlorizin, procyanidins | Sugiyama et al. (2007); Schulze et al. (2014) |
| Melissa officinalis L. | Iran, Turkey Shakeri et al. (2016) | Decrease of plasma glucose levels in rats, reduced α-glucosidase, α-amylase activity, decrease of HbA1c, serum triglyceride and fasting blood glucose levels in humans | Polyphenolic compounds (flavonoids), essential oils | Hasanein and Riahi (2015); Asadi et al. (2019) |
| Mentha aquatica L. | n.f. | Decrease of fasting blood glucose and lipid levels, nephroprotective, reduced HbA1c, and increase of insulin levels in rats | Polyphenolic compounds (flavonoids, tannins), saponins, volatile oils | Yellanur Konda et al. (2020) |
| Momordica charantia L. | Asia, South America, East Africa Rizvi and Mishra (2013), United Kingdom Swanston-Flatt et al. (1991) | Decrease of blood glucose and glycosylated haemoglobin and increase of plasma insulin in animal studies, inhibition of intestinal transporters | Charantin, polypeptide-p, momordin ic, oleanolic acid 3-O-monodesmoside, and oleanolic acid 3-O-glucuronide | Grover and Yadav (2004) |
| Nigella sativa L. | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020) | Decrease of fasting blood glucose, reduced insulin resistance, and improved β-cell function | n.n. | Bamosa et al. (2010) |
| Olea europaea L. | Jordan Al-Aboudi and Afifi (2011), Morocco Idm’hand et al. (2020), Portugal Neves et al. (2009) | Decrease of serum blood glucose, triglycerides, and cholesterol, increase of serum insulin levels | Oleuropeoside | Eidi et al. (2009) |
| Origanum creticum L. | Morocco Idm’hand et al. (2020) | Decrease of blood glucose, lipids, and HbA1c in rats, reduced α-amylase activity | n.n. | Prasanna et al. (2017) |
| Panax ginseng c.a. mey. | Asia Park et al. (2019) | Decrease of 75 g-OGTT-plasma-glucose indices, fasting plasma insulin, and 75 g-OGTT-plasma-insulin indices in humans | Ginsenosides: PPT, (20R)-PPD, Rg1, Rc, Rd, Re, Rf, Rg2, Rh1, Rb1, and Rb2; peptidoglycan: panaxan B | Vuksan et al. (2008) |
| Peumus boldus molina | n.f. | Decrease of plasma glucose level, inhibition of α-amylase and lipase | Boldine | Jang et al. (2000); Buchholz and Melzig (2016) |
| Potentilla aurea L. | n.f. | Inhibition of α-amylase and lipase | n.n. | Buchholz and Melzig (2016) |
| Pueraria lobata (willd.) ohwi | China Li et al. (2004) | Decrease of fasting blood glucose, improved glucose tolerance and insulin sensitivity in mice | Isoflavones: puerarin | Prasain et al. (2012) |
| Punica granatumL. (peel) | Morocco Idm’hand et al. (2020); Tahraoui et al. (2007) | Decrease of fasting blood glucose and serum lipids in rats | Phenolic compounds | Bagri et al. (2009) |
| Rosa rugosa thunb. | Korea Lee et al. (2008) | Decrease of blood glucose, serum insulin, serum lipids, increased insulin sensitivity in rats, reduced α-glucosidase activity | Polyphenolic compounds | Liu et al. (2017) |
| Rosmarinus officinalis L. | Morocco Idm’hand et al. (2020); Tahraoui et al. (2007) | Decrease of blood glucose and increase of serum insulin in rabbits, reduced α-glucosidase activity | Volatile oils | Bakırel et al. (2008) |
| Salvia officinalis L. | Morocco Idm’hand et al. (2020); Tahraoui et al. (2007), United Kingdom Swanston-Flatt et al. (1991) | Decrease in 2-h-postprandial blood glucose and cholesterol in humans, decrease in serum glucose and lipids in rats | Flavonoids (rosmarinic acid, phenolic acids, carnosic compounds) | Eidi and Eidi (2009); Behradmanesh et al. (2013) |
| Sarcopoterium spinosum (L.) spach | Jordan Al-Aboudi and Afifi (2011), Israel Bachrach (2007) | Improved glucose tolerance in mice, insulin-like effects, increased insulin secretion in vitro, reduced α-glucosidase and α-amylase activity | Polyphenolic compounds: catechin, epicatechin | Smirin et al. (2010); Elyasiyan et al. (2017) |
| Syzygium aromaticum(L.) merr. and l.m.perry | Morocco Idm’hand et al. (2020) | Decrease of postprandial hyperglycemia in rats, reduced α-glucosidase and α-amylase activity, downregulation of intestinal transporters | Triterpenes (oleanolic acid, maslinic acid) | Khathi et al. (2013) |
| Thymus vulgaris L. | Morocco Idm’hand et al. (2020), United Kingdom Swanston-Flatt et al. (1991) | Reduced blood glucose and serum lipids in rats | Flavonoids | Ekoh et al. (2014) |
| Vaccinium myrtillus L. | Europe Helmstaedter (2007) | Decrease of blood glucose and lipids in rats | Polyphenolic compounds | Sidorova et al. (2017) |
| Vitis vinifera L. | Morocco Idm’hand et al. (2020) | Decrease of postprandial hyperglycemia in mice, reduced α-glucosidase activity | Polyphenolic compounds: flavonoids, anthocyanins | Hogan et al. (2010) |
Materials and Methods
Plant Extract Preparations
The methanolic and aqueous extract preparations were performed according to Buchholz (2017) with modifications. The parts of the plants, which were used to prepare the extracts, are listed in Tables 2, 3. Except for the fresh fruits of Malus domestica, the botanicals were obtained as dried plant material from the companies. The bulbs of Allium sativum , fruits of Aronia melanocarpa and Vaccinium myrtillus, fruit skin of Punica granatum, the herbal part of Cynara cardunculus and Potentilla aurea , leaves of Mentha aquatica and Olea europaea , roots of Panax ginseng and Sarcopoterium spinosum, and seeds of Nigella sativa were purchased from Kräuter Schulte aktiv Kräuter Drogerie e.K. (Gernsbach, Germany). The following herbal drugs were obtained from Alfred Galke GmbH (Gittelde, Germany): flowers of Hibiscus sabdariffa and Syzygium aromaticum ; fruits of Ceratonia siliqua; fruit skin of Citrus limon; leaves of Brassica oleracea , Camellia sinensis, Ilex paraguariensis, Juglans regia, Melissa officinalis, Origanum creticum, Rosmarinus officinalis, and Salvia officinalis; and seeds of Cuminum cyminum and Vitis vinifera (pomace). The plant materials used in Traditional Chinese Medicine, such as the bark of Eucommia ulmoides ; flowers of Rosa rugosa; fruits of Cornus officinalis , Crataegus pinnatifida, and Lycium chinense; and the roots of Adenophora triphylla and Pueraria lobata were acquired from Zieten Apotheke (Berlin, Germany). The fruits of the cucurbitaceae Momordica charantia were kindly given by Dr. Serhat Sezai Çiçek (Institute of Pharmacy of Christian-Albrechts-Universitaet Kiel, Germany). The green seeds of Coffea arabica were purchased from Ridders Kaffeerosterei (Berlin, Germany). The leaves of Peumus boldus were gained from Heinrich Klenk GmbH & Co. KG (Schwebheim, Germany), whereas the leaves of Artemisia dracunculus and the fresh fruits of Malus domestica were obtained from a local supermarket. The fresh fruits of Malus domestica were washed with water. The fruit’s skin was cut into small pieces, dried in a drying cabinet at 40°C for 3 days, and freeze-dried for 48 h. The dried plant material was ground with a M20 mill from IKA®-Werke GmbH & Co. KG (Staufen, Germany). 10.0 g of the plant’s powder, adjusted to the drug extract ratio as listed in Tables 2, 3, was extracted in 100 ml. For the preparation of the aqueous extracts, plant material was extracted in water obtained from the ultrapure water system LaboStar from Siemens AG Wasseraufbereitung (Barsbüttel, Germany) for 15 min at 40°C and filtered with a Buechner filter. In some cases, extracts were centrifuged at 4°C first with an Allegra X-30R centrifuge from Beckman Coulter GmbH (Krefeld, Germany) to facilitate the filtration process. The extracts were freeze-dried for at least 48 h with the lyophilization machine Alpha 2-4 LSCplus from Martin Christ Gefriertrocknungsanlagen GmbH (Osterode am Harz, Germany). The powder was stored at −20°C until use. For the preparation of the methanolic extracts, plant material was extracted in methanol purchased from VWR International S.A.S. (Fontenay-sous-Bois, France) for 60 min at 40°C and filtered. In order to obtain a dry powder, they were evaporated to 5 ml with the rotary evaporator RV 10 basic from IKA®-Werke GmbH & Co. KG (Staufen, Germany) and dried in a Petri dish from VWR International GmbH (Darmstadt, Germany) under the exhaust hood overnight. Subsequently, the extracts were freeze-dried to lose residual water and stored at −20°C until use. For application in cell experiments, the aqueous extracts were solved in DPBS (Dulbecco’s phosphate-buffered saline) purchased from Lonza Cologne AG (Cologne, Germany), whereas the extracts made with methanol were first dissolved in DMSO (0.3% V/V final well concentration), which was acquired from Fisher Scientific GmbH (Schwerte, Germany), and filled with DPBS until final volume. Extract solutions were filtered with a 0.8 µm and a 0.2 µm sterile filter purchased from Carl Roth GmbH + Co. KG (Karlsruhe, Germany) and stored at −20°C until use. Three different extract concentrations (1.0, 0.1, or 0.01 mg/ml) were tested in MTT assay [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] as described in the Supplementary Material and only the highest concentration, which did not affect cell viability, was used in uptake studies. The GLUT2 inhibitor phloretin, which was purchased from Cayman Chemical (Michigan, United States), and the SGLT1 inhibitor phlorizin applied from Carl Roth GmbH + Co. KG (Karlsruhe, Germany) were used with a final well concentration of 100 µM as a positive control for uptake inhibition of glucose.
TABLE 2.
Results of glucose and fructose uptake studies with methanolic extracts, the used plant part, drug extract ratio, and tested concentration, which did not show any influence on cells in MTT assay. Only the plants that showed the strongest inhibition rates on glucose uptake were tested for fructose uptake inhibition. Regarding methanolic and aqueous extracts, the extract of a plant that showed the stronger inhibition on glucose absorption was repeated and tested for its impact on fructose uptake; mean value ± SD; *significant difference in U-test (n = 2 with 12 replicates each; two-sided; α = 0.05); n.d. = not determined.
| Scientific plant name | Used part of the plant | Drug extract ratio for methanolic extracts | Used methanolic extract concentration in mg/mL | Uptake inhibition of glucose in % | Uptake inhibition of fructose in % |
|---|---|---|---|---|---|
| Adenophora triphylla (thunb.) ADC. | Root | n.d. | 1 | 64.6* ± 4.5 | <25 |
| Allium sativum L. | Bulb | 20:1 | 0.01 | <25 | n.d. |
| Aronia melanocarpa michx. elliott | Fruit | n.d. | 1 | 57.1* ± 7.2 | <25 |
| Artemisia dracunculus L. | Leaf | 9:1 | 0.1 | <25 | n.d. |
| Brassica oleraceaL. “Capitata alba” | Leaf | 3:1 | 1 | 82.8* ± 4.3 | <25 |
| Camellia sinensis(L.) kuntze (Assam) | Leaf | 11:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (Darjeeling) | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (gunpowder) | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (Sencha) | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Ceratonia siliqua L. | Fruit | 4:1 | 1 | <25 | n.d. |
| Citrus limon (L.) osbeck | Fruit skin | 7:1 | 0.1 | <25 | n.d. |
| Coffea arabica L. | Green seed | 7:1 | 1 | <25 | n.d. |
| Cornus officinalissiebold and zucc. | Fruit | 3:1 | 1 | 73.8* ± 6.7 | <25 |
| Crataegus pinnatifida bunge | Fruit | n.d. | 1 | 73.7* ± 4.2 | <25 |
| Cynara cardunculus L. | Herb | n.d. | 0.1 | <25 | n.d. |
| Eucommia ulmoides oliv. | Bark | n.d. | 0.1 | <30 | n.d. |
| Hibiscus sabdariffa L. | Flower | 4:1 | 0.1 | <25 | n.d. |
| Ilex paraguariensis a. st.-hil | Leaf | 7:1 | 0.1 | <25 | n.d. |
| Juglans regia L. | Leaf | 12:1 | 1 | 52.4* ± 1.7 | 30.2* ± 11.7 |
| Lycium chinense mill. | Fruit | n.d. | 1 | 75.2* ± 0.5 | <25 |
| Melissa officinalis L. | Leaf | n.d. | 0.1 | <25 | n.d. |
| Mentha aquatica L. | Leaf | 16:1 | 0.1 | <25 | n.d. |
| Momordica charantia L. | Fruit | 13:1 | 0.1 | <25 | n.d. |
| Nigella sativa L. | Seed | 14:1 | 0.1 | <25 | n.d. |
| Olea europaea L. | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Origanum creticum L. | Leaf | 16:1 | 0.1 | <25 | n.d. |
| Panax ginseng c.a. mey | Root | 9:1 | 0.1 | <25 | n.d. |
| Peumus boldus molina | Leaf | 5:1 | 1 | 47.5* ± 7.5 | 32.6* ± 26.1 |
| Potentilla aurea L | Herb | 15:1 | 1 | <25 | n.d. |
| Pueraria lobata (willd.) ohwi | Root | 6:1 | 0.1 | <25 | n.d. |
| Rosa rugosa thunb. | Flower | 6:1 | 0.1 | <25 | n.d. |
| Rosmarinus officinalis L. | Leaf | 6:1 | 0.1 | <25 | n.d. |
| Salvia officinalis L. | Leaf | 9:1 | 0.1 | <25 | n.d. |
| Sarcopoterium spinosum (L.) spach | Root | 14:1 | 0.1 | <25 | n.d. |
| Syzygium aromaticum(L.) merr. and l.m.perry | Flower | 17:1 | 0.1 | <25 | n.d. |
| Thymus vulgaris L. | Herb | 14:1 | 0.1 | <25 | n.d. |
| Vaccinium myrtillus L. | Fruit | 2:1 | 1 | 79.9* ± 1.1 | <25 |
| Vitis vinifera L. | Seed (pomace) | 15:1 | 1 | <25 | n.d. |
TABLE 3.
Results of glucose and fructose uptake studies with aqueous extracts, the used plant part, drug extract ratio, and tested concentration, which did not show any influence on cells in MTT assay. Only the plants that showed the strongest inhibition rates on glucose uptake were tested for fructose uptake inhibition. Regarding methanolic and aqueous extracts, the extract of a plant that showed the stronger inhibition on glucose absorption was repeated and tested for its impact on fructose uptake; mean value ± SD; *significant difference in U-test (n = 2–3; 12 replicates each; two-sided; α = 0.05); n.d. = not determined.
| Scientific plant name | Used part of the plant | Drug extract ratio for aqueous extracts | Used aqueous extract concentration in mg/mL | Uptake inhibition of glucose in % | Uptake inhibition of fructose in % |
|---|---|---|---|---|---|
| Adenophora triphylla (thunb.) ADC. | Root | n.d. | 1 | <40 | n.d. |
| Allium sativum L. | Bulb | 2:1 | 1 | <50 | n.d. |
| Aronia melanocarpa michx. elliott | Fruit | 3:1 | 1 | <60 | n.d. |
| Artemisia dracunculus L. | Leaf | 5:1 | 1 | <25 | n.d. |
| Brassica oleraceaL. “Capitata alba” | Leaf | 2:1 | 0.1 | <60 | n.d. |
| Camellia sinensis(L.) kuntze (Assam) | Leaf | 6:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (Darjeeling) | Leaf | 7:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (gunpowder) | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Camellia sinensis(L.) kuntze (sencha) | Leaf | 6:1 | 0.1 | <25 | n.d. |
| Ceratonia siliqua L. | Fruit | 3:1 | 1 | <25 | n.d. |
| Citrus limon (L.) osbeck | Fruit skin | 4:1 | 1 | <40 | n.d. |
| Coffea arabica L. | Green seed | 5:1 | 1 | <25 | n.d. |
| Cornus officinalissiebold and zucc. | Fruit | n.d. | 1 | <65 | n.d. |
| Crataegus pinnatifida bunge | Fruit | n.d. | 1 | <60 | n.d. |
| Cuminum cyminum L. | Seed | 5:1 | 0.1 | <25 | n.d. |
| Cynara cardunculus L. | Herb | 3:1 | 0.1 | <30 | n.d. |
| Eucommia ulmoides oliv. | Bark | 16:1 | 1 | 68.7* ± 13.2 | <25 |
| Hibiscus sabdariffa L. | Flower | 2:1 | 0.1 | <25 | n.d. |
| Ilex paraguariensis a. st.-hil | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Juglans regia L. | Leaf | 7:1 | 1 | <25 | n.d. |
| Lycium chinense mill. | Fruit | 3:1 | 1 | <75 | n.d. |
| Malus domesticaborkh. “Gold en delicious” | Fruit skin | 3:1 | 1 | 57.5* ± 9.2 | <25 |
| Melissa officinalis L. | Leaf | 4:1 | 0.01 | <25 | n.d. |
| Mentha aquatica L. | Leaf | 6:1 | 1 | <40 | n.d. |
| Momordica charantia L. | Fruit | 6:1 | 1 | <25 | n.d. |
| Nigella sativa L. | Seed | 4:1 | 0.1 | <25 | n.d. |
| Olea europaea L. | Leaf | 5:1 | 1 | <25 | n.d. |
| Origanum creticum L. | Leaf | 4:1 | 0.1 | <25 | n.d. |
| Panax ginseng c.a. mey | Root | 3:1 | 1 | <25 | n.d. |
| Peumus boldus molina | Leaf | n.d | 1 | <40 | n.d. |
| Potentilla aurea L. | Herb | 8:1 | 0.1 | <25 | n.d. |
| Pueraria lobata (willd.) ohwi | Root | 4:1 | 1 | <50 | n.d. |
| Punica granatum L. | Fruit skin | 3:1 | 0.1 | <25 | n.d. |
| Rosa rugosa thunb. | Flower | n.d | 0.1 | <30 | n.d. |
| Rosmarinus officinalis L. | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Salvia officinalis L. | Leaf | 5:1 | 0.1 | <25 | n.d. |
| Sarcopoterium spinosum (L.) spach | Root | 28:1 | 1 | <25 | n.d. |
| Syzygium aromaticum(L.) merr. and l.m.perry | Flower | n.d. | 0.1 | <25 | n.d. |
| Thymus vulgaris L. | Herb | 6:1 | 1 | <25 | n.d. |
| Vaccinium myrtillus L. | Fruit | 2:1 | 1 | <80 | n.d. |
| Vitis vinifera L. | Seed (pomace) | 27:1 | 1 | <40 | n.d. |
General Handling of the Cell Line
Caco2 cells were purchased from Leibniz-Institut DSMZ-Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH (Braunschweig, Germany) and seeded in 24-well plates, which were acquired from Greiner Bio-One GmbH (Frickenhausen, Germany), for every experiment. As general medium DMEM (Dulbecco’s modified eagle medium) without phenol red containing 1% UltraGlutamine 1, both acquired from Lonza Cologne AG (Cologne, Germany) and 5 or 20% FBS derived from Bio&SELL GmbH (Nürnberg, Germany) for experiment or mother line, respectively, was used for the handling with Caco2 cells. Cells were washed once with PBS from Lonza Cologne AG (Cologne, Germany) for every medium change and washed twice before Trypsin (TrypLE™ Express without phenol red) from Gibco Life Technologies corporation (NY, United States) was applied to detach the cells for seeding. The cells were incubated at 37°C and 5% CO2-supply during the whole time. Passages 20–35 were used in the experiments. The buffers and solutions for cell culture were prepared with water obtained from the ultrapure water system LaboStar from Siemens AG Wasseraufbereitung (Barsbüttel, Germany).
Glucose and Fructose Uptake in Caco2 Cells
For uptake studies, cells were seeded in a density of 400,000 cells per well. Confluence appeared 24 h after seeding and was controlled visually. The radiolabeled substrates 1 mCi/ml glucose (specific activity: 30 Ci/mmol) and 1 mCi/ml fructose (specific activity: 5 Ci/mmol) were obtained from Biotrend Chemikalien GmbH (Köln, Germany). Although fructose absorption into cells was determined by using 54 nM solution of radiolabeled substrate as final well concentration, the 2 nM radiolabeled glucose solution was spiked with 0.001 µM non-radiolabeled glucose, which was purchased from Merck KGaA (Darmstadt, Germany). For uptake studies with extracts, cells were used at day three and day fifteen after confluence for glucose and fructose, respectively. On the day of the experiment, cells were washed once with 2 ml buffer containing 20 mM Hepes [4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid], which was obtained from Carl Roth GmbH + Co. KG (Karlsruhe, Germany) and 150 mM NaCl purchased from Fisher Scientific GmbH (Schwerte, Germany). The pH of the buffer was adjusted with sodium hydroxide from Merck KGaA (Darmstadt, Germany) using a 766 Laboratory pH meter from Knick Elektronische Messgeraete GmbH & Co. KG (Berlin, Germany) to 7.4. After 1 h starving incubation with Hepes-NaCl buffer at 37°C, substrate or substrate-extract solution were added to the cells and incubated at 37°C. After 1 h, the buffer was discarded, the cells were washed once with 1 ml ice-cold Hepes-NaCl buffer, and 500 µL Hepes-NaCl solution containing 1% Triton X 100 was added to each well. The samples were taken from the well plates, added to a sufficient volume of scintillation cocktail in scintillation tubes, both purchased from Hidex (Mainz, Germany), mixed for 20 s using a vortexer from Heidolph Instruments GmbH & Co. KG (Schwabach, Germany), and measured with a β-Counter from Hidex (Mainz, Germany). The plant extracts, which showed the strongest inhibition on intestinal glucose transporters, were repeated and tested for their influence on fructose uptake with a focus on methanolic extracts. Randomly, well plates, which were cultivated and treated under the same conditions as cells used for uptake studies, were tested in Hoechst Assay as described in the Supplementary Material to determine variations depending on the procedure.
Statistical Evaluation and Use of Software
All statistical tests were performed using Microsoft Excel. Normal distribution was established via Shapiro–Wilk test manually before using Excel’s tool “Data analysis” for provision of the F-test and the t-tests. If the data was not normally distributed, Mann–Whitney U test was applied for further statistical analysis. IC50 data was calculated with GraphPad Prism 5.0.
Results
Uptake Studies with Glucose and Fructose
In order to prevent decreased metabolic activity due to the use of toxic extracts, methanolic and aqueous plant preparations were tested in MTT assay and the concentration, which did not reduce cell viability significantly, was used for uptake studies as shown in Tables 2, 3. According to the results obtained from Hoechst Assay that showed an average coefficient of variation of 21.7% within 14 randomly tested well plates, uptake inhibition of plant extracts was distinguishable from general fluctuations in the procedure for values >25%. In every experiment, a control without extract was running and served as 100% uptake control. After correction of values by subtracting the blank, uptake inhibition was calculated as the difference between control and sample. The obtained values of monosaccharide uptake inhibition in Caco2 cells treated with methanolic and aqueous extracts are reported in Tables 2, 3, respectively. Only the herbal drugs that showed the strongest inhibition rates on glucose uptake were tested for fructose uptake inhibition. For some of the plants, methanolic and aqueous extracts decreased glucose uptake significantly. However, only the extract of a plant that showed stronger inhibition on glucose absorption was repeated and tested for its impact on fructose uptake as described for the following plants. Additionally, the focus for further studies was on methanolic extracts as they naturally contain fewer monosaccharides, which could interfere with the measurement. The methanolic extracts made from the fruits of Aronia melanocarpa (uptake inhibition: 57.1%), Cornus officinalis (uptake inhibition: 73.8%), Crataegus pinnatifida (uptake inhibition: 73.7%), Lycium chinense (uptake inhibition: 75.2%), and Vaccinium myrtillus (uptake inhibition: 79.9%); the leaves of Brassica oleracea (uptake inhibition: 82.8%), Juglans regia (uptake inhibition: 52.4%), and Peumus boldus (uptake inhibition: 47.5%); and the roots of Adenophora triphylla (uptake inhibition: 64.6%) indicated a strong and reproducible inhibitory potential between 40 and 80% on intestinal glucose transporters as visualized in Figure 1. Seven of these extracts showed higher uptake inhibition rates than the two controls phloretin (uptake inhibition: 60.4%) and phlorizin (uptake inhibition: 55.1%). A queous extracts made from the bark of Eucommia ulmoides (uptake inhibition: 68.7%) and the fruits of Malus domestica (uptake inhibition: 57.5%) inhibited glucose uptake significantly and reproducibly. Additionally, for the methanolic extract of Brassica oleracea and the aqueous extract of Eucommia ulmoides, IC50 was calculated as 0.0904 and 0.4285 mg/ml, respectively. These extracts decreased glucose uptake in a dose-dependent manner. The methanolic extracts of Juglans regia (uptake inhibition: 30.2%) and Peumus boldus (uptake inhibition: 32.6%) inhibited the fructose uptake significantly.
FIGURE 1.
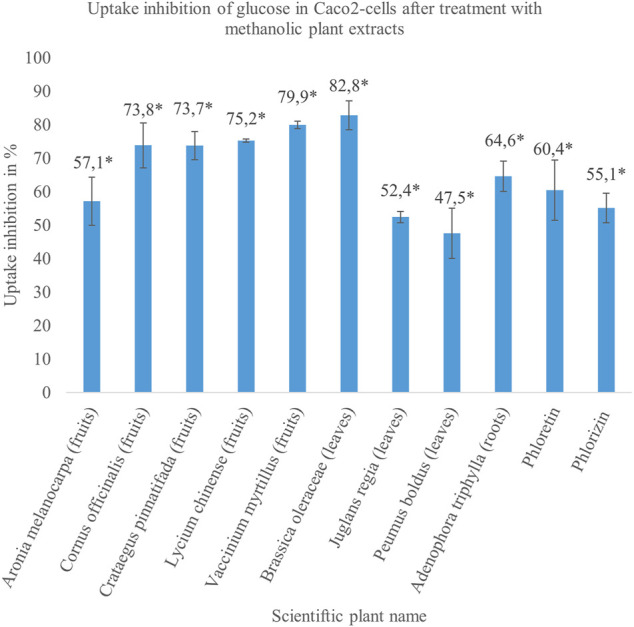
The results of the methanolic plant extracts, which reduced glucose uptake between 40 and 80% in treated cells significantly compared to untreated cells in a concentration of 1 mg/ml. The extracts were made from different parts of the plants enclosed in brackets. The solutions of 100 µM phloretin and 100 µM phlorizin were used as positive controls and inhibited glucose uptake in Caco2 cells by approximately 60%. The illustrated results refer to the data in Table 2. Mean value ± SD. * Significant difference in U-test (n = 2 with 12 replicates each; two-sided, α = 0.05).
Discussion
According to the results of the present study, the observed hypoglycemic effects in animal and/or human studies after treatment with preparations of Adenophora triphylla , Aronia melanocarpa, Brassica oleracea, Cornus officinalis , Crataegus pinnatifida, Eucommia ulmoides, Juglans regia , Lycium chinense, Malus domestica, Peumus boldus, and Vaccinium myrtillus as listed in Table 1 possibly relate to the inhibition of intestinal transporters. The methanolic plant extracts made from the leaves of Juglans regia and Peumus boldus decreased glucose as well as fructose uptake possibly related to inhibition of GLUT2, which is able to transport both monosaccharides. These findings show that the traditional use of Brassica oleracea, Cornus officinalis, Eucommia ulmoides, Juglans regia, Lycium chinense, Malus domestica, and Vaccinium myrtillus and their preparations as antidiabetic remedy and adjuncts to conventional treatments in the therapy of Diabetes mellitus is reasonable. Extracts of these plants show inhibitory activity on intestinal glucose and fructose transporters resulting in lower blood concentrations of the monosaccharides and reduced concomitant health problems.
In literature, there is not much comparable data available, because the inhibition of intestinal transporters by crude plant extracts in Caco2 cells has not been investigated very well (Schreck and Melzig, 2018). Kim et al. determined a moderate inhibition of glucose uptake in Caco2 cells for methanolic extracts of Adenophora triphylla (uptake inhibition: 13.4%) and Cornus officinalis (uptake inhibition: 16.4%), but other results as for Crataegus pinnatifida (no uptake inhibition) are not consistent with our findings (Kim et al., 2011). Zhang et al. confirmed an uptake inhibition of 26.3% for Eucommia ulmoides (Zhang et al., 2015). Manzano et al. only tested specific polyphenol-rich fractions of Malus domestica (Manzano and Williamson, 2010), which showed inhibitory activity on intestinal glucose transporters, but it is not directly comparable with our work. Discrepancies between results from different studies probably depend on different preparations of the extracts, the use of modified substrates, and/or an insufficient validation of the cell model. We determined the expression of relevant transporters over three weeks in Caco2 cells and adapted our experimental settings in accordance with the ensured presence of functional transporters (Schreck and Melzig, 2019). Buchholz et al. examined whether extracts of the traditionally used plants inhibit cleavage of polysaccharides and lipids by affecting lipase and α-amylase activity (Prasain et al., 2012). The methanolic extracts of Aronia melanocarpa, Peumus boldus, and Vaccinium myrtillus showed strong lipase and α-amylase inhibition as well as inhibition of intestinal monosaccharide transporters in our studies. This multi-target effect of the mentioned plant preparations describes a partial, but multiple drug action and is proposed to exceed drugs with single-target effects. Based on synergistic mechanisms, the plants that affect various targets to alleviate monosaccharide uptake show therapeutic advantages concerning higher intended and lower adverse effects (Csermely et al., 2005). Since the plants, especially the fruits, contain sugars themselves, it cannot be totally excluded that a part of the inhibition is a “pseudo-inhibition” and comes from the displacement of the radioactive substrates by non-radioactive monosaccharides during absorption processes. For this reason, the focus of this work was on methanolic extracts that naturally contain fewer monosaccharides. Commonly, the discussed constituents of the plants with inhibitory effects on intestinal transporters are polyphenolic compounds as shown in Table 1. Particularly, the chalcones phlorizin and phloretin (Debnam and Levin, 1975; Zheng et al., 2012; Raja and Kinne, 2015), which are often used as positive controls in transport studies, the flavonoids quercetin, fisetin, and myricetin (Kwon et al., 2007) and the tannins (-)-epigallocatechin gallate, (-)-epigallocatechin, and (-)-epicatechin gallate seem to act as inhibitors of intestinal, monosaccharide transporters (Johnston et al., 2005). Further studies are planned to investigate which fraction of the extracts, or more specifically, which group of compounds are responsible for the inhibition of the intestinal glucose and fructose transporters in this study.
Acknowledgments
We would like to thank Yi Gu for screening the plants in the MTT assay according to our protocol. We acknowledge the support from the Open Access Publication Fund of the Freie Universitaet Berlin.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, and further inquiries can be directed to the corresponding author.
Author Contributions
KS designed the studies, performed experiments, analyzed the data, and drafted the manuscript. MM supervised the performance of the experiments and interpretation of the data. MM proofread the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2021.692566/full#supplementary-material
References
- Al-Aboudi A., Afifi F. U. (2011). Plants Used for the Treatment of Diabetes in jordan: A Review of Scientific Evidence. Pharm. Biol. 49, 221–239. 10.3109/13880209.2010.501802 [DOI] [PubMed] [Google Scholar]
- American Diabetes Association (2014). Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 37, 81–90. 23959568 [Google Scholar]
- Asadi A., Shidfar F., Safari M., Hosseini A. F., Fallah Huseini H., Heidari I., et al. (2019). Efficacy of Melissa Officinalis L. (Lemon Balm) Extract on Glycemic Control and Cardiovascular Risk Factors in Individuals with Type 2 Diabetes: A Randomized, Double-Blind, Clinical Trial. Phytother Res. 33, 651–659. 10.1002/ptr.6254 [DOI] [PubMed] [Google Scholar]
- Assad T., Khan R. A., Feroz Z. (2014). Evaluation of Hypoglycemic and Hypolipidemic Activity of Methanol Extract of brassica Oleracea. Chin. J. Nat. Medicines 12, 648–653. 10.1016/s1875-5364(14)60099-6 [DOI] [PubMed] [Google Scholar]
- Bachrach Z. Y. (2007). Ethnobotanical Studies of Sarcopoterium Spinosum in israel. Isr. J. Plant Sci. 55, 111–114. 10.1560/ijps.55.1.111 [DOI] [Google Scholar]
- Bagri P., Ali M., Aeri V., Bhowmik M., Sultana S. (2009). Antidiabetic Effect of Punica Granatum Flowers: Effect on Hyperlipidemia, Pancreatic Cells Lipid Peroxidation and Antioxidant Enzymes in Experimental Diabetes. Food Chem. Toxicol. 47, 50–54. 10.1016/j.fct.2008.09.058 [DOI] [PubMed] [Google Scholar]
- Bahadoran Z., Mirmiran P., Azizi F. (2013). Dietary Polyphenols as Potential Nutraceuticals in Management of Diabetes: A Review. J. Diabetes Metab. Disord. 12, 43. 10.1186/2251-6581-12-43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakırel T., Bakırel U., Keleş O. Ü., Ülgen S. G., Yardibi H. (2008). In Vivo assessment of Antidiabetic and Antioxidant Activities of Rosemary (Rosmarinus Officinalis) in Alloxan-Diabetic Rabbits. J. Ethnopharmacology 116, 64–73. [DOI] [PubMed] [Google Scholar]
- Bamosa A. O., Kaatabi H., Lebdaa F. M., Elq A. M., Al-Sultanb A. (2010). Effect of Nigella Sativa Seeds on the Glycemic Control of Patients with Type 2 Diabetes Mellitus. Indian J. Physiol. Pharmacol. 54, 344–354. [PubMed] [Google Scholar]
- Banjari I., Misir A., Šavikin K., Jokić S., Molnar M., De Zoysa H. K. S., et al. (2017). Antidiabetic Effects of Aronia Melanocarpa and its Other Therapeutic Properties. Front. Nutr. 4, 53. 10.3389/fnut.2017.00053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behradmanesh S., Derees F., Rafieian-Kopaei M. (2013). Effect of Salvia Officinalis on Diabetic Patients. J. Ren. Inj Prev 2, 51–54. 10.12861/jrip.2013.18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz T. (2017). “Secondary Metabolites as Inhibitors of Selected Digestive Enzymes,” (Freie Universitaet Berlin; ). Thesis. [Google Scholar]
- Buchholz T., Melzig M. F. (2016). Medicinal Plants Traditionally Used for Treatment of Obesity and Diabetes Mellitus - Screening for Pancreatic Lipase and α-Amylase Inhibition. Phytother. Res. 30, 260–266. 10.1002/ptr.5525 [DOI] [PubMed] [Google Scholar]
- Campos-Florian J., Bardales-Valdivia J., Caruajulca-Guevara L., Cueva-Llanos D. (2013). Anti-diabetic Effect of Coffea Arabica, in Alloxan-Induced Diabetic Rats. Emirates J. Food Agric. 25, 772. [Google Scholar]
- Chantret I., Barbat A., Dussaulx E., Brattain M. G., Zweibaum A. (1988). Epithelial Polarity, Villin Expression, and Enterocytic Differentiation of Cultured Human colon Carcinoma Cells: A Survey of Twenty Cell Lines. Cancer Res. 48, 1936–1942. [PubMed] [Google Scholar]
- Csermely P., Ágoston V., Pongor S. (2005). The Efficiency of Multi-Target Drugs: The Network Approach Might Help Drug Design. Trends Pharmacol. Sci. 26, 178–182. 10.1016/j.tips.2005.02.007 [DOI] [PubMed] [Google Scholar]
- de Freitas Junior L. M., de Almeida E. B., Jr. (2017). Medicinal Plants for the Treatment of Obesity: Ethnopharmacological Approach and Chemical and Biological Studies. Am. J. Transl Res. 9, 2050–2064. [PMC free article] [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. (1975). An Experimental Method of Identifying and Quantifying the Active Transfer Electrogenic Component from the Diffusive Component during Sugar Absorption Measured In Vivo . J. Physiol. 246, 181–196. 10.1113/jphysiol.1975.sp010885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dehghani S., Mehri S., Hosseinzadeh H. (2019). The Effects of Crataegus Pinnatifida (Chinese Hawthorn) on Metabolic Syndrome: A Review. Iran J. Basic Med. Sci. 22, 460–468. 10.22038/IJBMS.2019.31964.7678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eidi A., Eidi M. (2009). Antidiabetic Effects of Sage (Salvia Officinalis l.) Leaves in normal and Streptozotocin-Induced Diabetic Rats. Diabetes Metab. Syndr. Clin. Res. Rev. 3, 40–44. 10.1016/j.dsx.2008.10.007 [DOI] [Google Scholar]
- Eidi A., Eidi M., Darzi R. (2009). Antidiabetic Effect ofOlea europaeaL. In normal and Diabetic Rats. Phytother. Res. 23, 347–350. 10.1002/ptr.2629 [DOI] [PubMed] [Google Scholar]
- Eidi A., Eidi M., Esmaeili E. (2006). Antidiabetic Effect of Garlic (Allium Sativum l.) in normal and Streptozotocin-Induced Diabetic Rats. Phytomedicine 13, 624–629. 10.1016/j.phymed.2005.09.010 [DOI] [PubMed] [Google Scholar]
- Ekoh S. N., Akubugwo I. E., Ude V. C., Edwin N. (2014). Anti-hyperglycemic and Anti-hyperlipidemic Effect of Spices (Thymus Vulgaris, Murraya Koenigii, Ocimum Gratissimum and Piper Guineense) in Alloxan-Induced Diabetic Rats. Int. J. Biosciences 4, 179–187. [Google Scholar]
- Elyasiyan U., Nudel A., Skalka N., Rozenberg K., Drori E., Oppenheimer R., et al. (2017). Anti-diabetic Activity of Aerial Parts of Sarcopoterium Spinosum. BMC Complement. Altern. Med. 17, 356. 10.1186/s12906-017-1860-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabricant D. S., Farnsworth N. R. (2001). The Value of Plants Used in Traditional Medicine for Drug Discovery. Environ. Health Perspect. 109, 69–75. 10.1289/ehp.01109s169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilani A. H., Rahman U.-A., Trends R. (2005). Trends in Ethnopharmacology. J. Ethnopharmacology 100, 43–49. 10.1016/j.jep.2005.06.001 [DOI] [PubMed] [Google Scholar]
- Gomes A., Vedasiromoni J. R., Das M., Sharma R. M., Ganguly D. K. (1995). Anti-hyperglycemic Effect of Black tea (Camellia Sinensis) in Rat. J. Ethnopharmacology 45, 223–226. 10.1016/0378-8741(95)01223-z [DOI] [PubMed] [Google Scholar]
- Grover J. K., Yadav S. P. (2004). Pharmacological Actions and Potential Uses of Momordica Charantia: A Review. J. Ethnopharmacology 93, 123–132. 10.1016/j.jep.2004.03.035 [DOI] [PubMed] [Google Scholar]
- Grundy S. M. (1998). Multifactorial Causation of Obesity: Implications for Prevention. Am. J. Clin. Nutr. 67, 563S–572S. 10.1093/ajcn/67.3.563s [DOI] [PubMed] [Google Scholar]
- Han Q., Yu Q.-Y., Shi J., Xiong C.-Y., Ling Z.-J., He P.-M. (2011). Molecular Characterization and Hypoglycemic Activity of a Novel Water-Soluble Polysaccharide from tea (Camellia Sinensis) Flower. Carbohydr. Polym. 86, 797–805. 10.1016/j.carbpol.2011.05.039 [DOI] [Google Scholar]
- Hanhineva K., Törrönen R., Bondia-Pons I., Pekkinen J., Kolehmainen M., Mykkänen H., et al. (2010). Impact of Dietary Polyphenols on Carbohydrate Metabolism. Int. J. Mol. Sci. 11, 1365–1402. 10.3390/ijms11041365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasanein P., Riahi H. (2015). Antinociceptive and Antihyperglycemic Effects of Melissa Officinalis Essential Oil in an Experimental Model of Diabetes. Med. Princ Pract. 24, 47–52. 10.1159/000368755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He K., Song S., Zou Z., Feng M., Wang D., Wang Y., et al. (2016). The Hypoglycemic and Synergistic Effect of Loganin, Morroniside, and Ursolic Acid Isolated from the Fruits of Cornus Officinalis. Phytother. Res. 30, 283–291. 10.1002/ptr.5529 [DOI] [PubMed] [Google Scholar]
- He X., Wang J., Li M., Hao D., Yang Y., Zhang C., et al. (2014). Eucommia Ulmoides oliv.: Ethnopharmacology, Phytochemistry and Pharmacology of an Important Traditional Chinese Medicine. J. Ethnopharmacology 151, 78–92. 10.1016/j.jep.2013.11.023 [DOI] [PubMed] [Google Scholar]
- Heidarian E., Soofiniya Y. (2011). Hypolipidemic and Hypoglycemic Effects of Aerial Part of Cynara Scolymus in Streptozotocin-Induced Diabetic Rats. J. Med. Plants Res. 5, 2717–2723. [Google Scholar]
- Helmstaedter A. (2007). Antidiabetic Drugs Used in Europe Prior to the Discovery of Insulin. Die Pharmazie 62, 717–720. [PubMed] [Google Scholar]
- Hidalgo I. J., Raub T. J., Borchardt R. T. (1989). Characterization of the Human colon Carcinoma Cell Line (Caco-2) as a Model System for Intestinal Epithelial Permeability. Gastroenterology 96, 736–749. 10.1016/s0016-5085(89)80072-1 [DOI] [PubMed] [Google Scholar]
- Hogan S., Zhang L., Li J., Sun S., Canning C., Zhou K. (2010). Antioxidant Rich Grape Pomace Extract Suppresses Postprandial Hyperglycemia in Diabetic Mice by Specifically Inhibiting Alpha-Glucosidase. Nutr. Metab. 7, 71. 10.1186/1743-7075-7-71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosseini S., Jamshidi L., Mehrzadi S., Mohammad K., Najmizadeh A. R., Alimoradi H., et al. (2014). Effects of Juglans Regia L. Leaf Extract on Hyperglycemia and Lipid Profiles in Type Two Diabetic Patients: A Randomized Double-Blind, Placebo-Controlled Clinical Trial. J. Ethnopharmacology 152, 451–456. 10.1016/j.jep.2014.01.012 [DOI] [PubMed] [Google Scholar]
- Idm’hand E., Msanda F., Cherifi K. (2020). Ethnopharmacological Review of Medicinal Plants Used to Manage Diabetes in morocco. Clin. Phytoscience 6, 18. [Google Scholar]
- Jagtap A. G., Patil P. B. (2010). Antihyperglycemic Activity and Inhibition of Advanced Glycation End Product Formation by Cuminum Cyminum in Streptozotocin Induced Diabetic Rats. Food Chem. Toxicol. 48, 2030–2036. 10.1016/j.fct.2010.04.048 [DOI] [PubMed] [Google Scholar]
- Jang Y. Y., Song J. H., Shin Y. K., Han E. S., Lee C. S. (2000). Protective Effect of Boldine on Oxidative Mitochondrial Damage in Streptozotocin-Induced Diabetic Rats. Pharmacol. Res. 42, 361–371. 10.1006/phrs.2000.0705 [DOI] [PubMed] [Google Scholar]
- Johnston K., Sharp P., Clifford M., Morgan L. (2005). Dietary Polyphenols Decrease Glucose Uptake by Human Intestinal Caco-2 Cells. FEBS Lett. 579, 1653–1657. 10.1016/j.febslet.2004.12.099 [DOI] [PubMed] [Google Scholar]
- Jumarie C., Malo C. (1991). Caco-2 Cells Cultured in Serum-free Medium as a Model for the Study of Enterocytic Differentiation In Vitro . J. Cel. Physiol. 149, 24–33. 10.1002/jcp.1041490105 [DOI] [PubMed] [Google Scholar]
- Kang Y.-R., Lee H.-Y., Kim J.-H., Moon D.-I., Seo M.-Y., Park S.-H., et al. (2012). Anti-obesity and Anti-diabetic Effects of Yerba Mate (ilex Paraguariensis) in C57bl/6j Mice Fed a High-Fat Diet. Lab. Anim. Res. 28, 23–29. 10.5625/lar.2012.28.1.23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kataya H. A. H., Hamza A. A. (2008). Red Cabbage (brassica Oleracea) Ameliorates Diabetic Nephropathy in Rats. Evidence-Based Complement. Altern. Med. 5, 281–287. 10.1093/ecam/nem029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katemo M., Mpiana P. T., Mbala B. M., Mihigo S. O., Ngbolua K. N., Tshibangu D. S. T., et al. (2012). Ethnopharmacological Survey of Plants Used against Diabetes in Kisangani City (Dr congo). J. Ethnopharmacology 144, 39–43. 10.1016/j.jep.2012.08.022 [DOI] [PubMed] [Google Scholar]
- Khathi A., Serumula M. R., Myburg R. B., Van Heerden F. R., Musabayane C. T. (2013). Effects of Syzygium Aromaticum-Derived Triterpenes on Postprandial Blood Glucose in Streptozotocin-Induced Diabetic Rats Following Carbohydrate challenge. PloS one 8, e81632. 10.1371/journal.pone.0081632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H. K., Baek S.-S., Cho H.-Y. (2011). Inhibitory Effect of Pomegranate on Intestinal Sodium Dependent Glucose Uptake. Am. J. Chin. Med. 39, 1015–1027. 10.1142/s0192415x11009378 [DOI] [PubMed] [Google Scholar]
- Kim H. Y., Moon B. H., Lee H. J., Choi D. H. (2004). Flavonol Glycosides from the Leaves of Eucommia Ulmoides O. With Glycation Inhibitory Activity. J. ethnopharmacology 93, 227–230. 10.1016/j.jep.2004.03.047 [DOI] [PubMed] [Google Scholar]
- Kwon O., Eck P., Chen S., Corpe C. P., Lee J. H., Kruhlak M., et al. (2007). Inhibition of the Intestinal Glucose Transporter Glut2 by Flavonoids. FASEB j. 21, 366–377. 10.1096/fj.06-6620com [DOI] [PubMed] [Google Scholar]
- Lee M.-K., Kim M.-J., Cho S.-Y., Park S. A., Park K.-K., Jung U. J., et al. (2005). Hypoglycemic Effect of Du-Zhong (Eucommia Ulmoides oliv.) Leaves in Streptozotocin-Induced Diabetic Rats. Diabetes Res. Clin. Pract. 67, 22–28. 10.1016/j.diabres.2004.05.013 [DOI] [PubMed] [Google Scholar]
- Lee Y.-H., Jung M. G., Kang H. B., Choi K.-C., Haam S., Jun W., et al. (2008). Effect of Anti-histone Acetyltransferase Activity from Rosa Rugosa Thunb. (Rosaceae) Extracts on Androgen Receptor-Mediated Transcriptional Regulation. J. Ethnopharmacology 118, 412–417. 10.1016/j.jep.2008.05.006 [DOI] [PubMed] [Google Scholar]
- Li W. L., Zheng H. C., Bukuru J., De Kimpe N. (2004). Natural Medicines Used in the Traditional Chinese Medical System for Therapy of Diabetes Mellitus. J. Ethnopharmacology 92, 1–21. 10.1016/j.jep.2003.12.031 [DOI] [PubMed] [Google Scholar]
- Liu L., Tang D., Zhao H., Xin X., Aisa H. A. (2017). Hypoglycemic Effect of the Polyphenols Rich Extract from Rose Rugosa Thunb on High Fat Diet and Stz Induced Diabetic Rats. J. Ethnopharmacology 200, 174–181. 10.1016/j.jep.2017.02.022 [DOI] [PubMed] [Google Scholar]
- Ma W., Wang K.-J., Cheng C.-S., Yan G.-q., Lu W.-L., Ge J.-F., et al. (2014). Bioactive Compounds from Cornus Officinalis Fruits and Their Effects on Diabetic Nephropathy. J. Ethnopharmacology 153, 840–845. 10.1016/j.jep.2014.03.051 [DOI] [PubMed] [Google Scholar]
- Maljaars P. W. J., Peters H. P. F., Mela D. J., Masclee A. A. M. (2008). Ileal Brake: A Sensible Food Target for Appetite Control. A Review. Physiol. Behav. 95, 271–281. 10.1016/j.physbeh.2008.07.018 [DOI] [PubMed] [Google Scholar]
- Manzano S., Williamson G. (2010). Polyphenols and Phenolic Acids from Strawberry and Apple Decrease Glucose Uptake and Transport by Human Intestinal Caco-2 Cells. Mol. Nutr. Food Res. 54, 1773–1780. 10.1002/mnfr.201000019 [DOI] [PubMed] [Google Scholar]
- Naim M., Amjad F. M., Sultana S., Islam S. N., Hossain M. A., Begum R., et al. (2012). Comparative Study of Antidiabetic Activity of Hexane-Extract of Lemon Peel (limon Citrus) and Glimepiride in Alloxan-Induced Diabetic Rats. Bangla Pharma J. 15, 131–134. 10.3329/bpj.v15i2.12577 [DOI] [Google Scholar]
- Nazni P., Vijayakumar T. P., Alagianambi P., Amirthaveni M. (2006). Hypoglycemic and Hypolipidemic Effect of Cynara Scolymus Among Selected Type 2 Diabetic Individuals. Pakistan J. Nutr. 5, 147–151. [Google Scholar]
- Neves J. M., Matos C., Moutinho C., Queiroz G., Gomes L. R. (2009). Ethnopharmacological Notes about Ancient Uses of Medicinal Plants in Trás-Os-Montes (Northern of Portugal). J. Ethnopharmacology 124, 270–283. 10.1016/j.jep.2009.04.041 [DOI] [PubMed] [Google Scholar]
- Olatunji O. J., Chen H., Zhou Y. (2017). Effect of the Polyphenol Rich Ethyl Acetate Fraction from the Leaves of Lycium Chinensemill. On Oxidative Stress, Dyslipidemia, and Diabetes Mellitus in Streptozotocin-Nicotinamide Induced Diabetic Rats. Chem. biodiversity 14, e1700277. 10.1002/cbdv.201700277 [DOI] [PubMed] [Google Scholar]
- Ota A., Ulrih N. P. (2017). An Overview of Herbal Products and Secondary Metabolites Used for Management of Type Two Diabetes. Front. Pharmacol. 8, 436. 10.3389/fphar.2017.00436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park S. A., Choi M.-S., Kim M.-J., Jung U. J., Kim H.-J., Park K.-K., et al. (2006). Hypoglycemic and Hypolipidemic Action of Du-Zhong (Eucommia Ulmoides Oliver) Leaves Water Extract in C57bl/ksj-Db/db Mice. J. Ethnopharmacology 107, 412–417. 10.1016/j.jep.2006.03.034 [DOI] [PubMed] [Google Scholar]
- Park S. J., Nam J., Ahn C. W., Kim Y. (2019). Anti-diabetic Properties of Different Fractions of Korean Red Ginseng. J. Ethnopharmacology 236, 220–230. 10.1016/j.jep.2019.01.044 [DOI] [PubMed] [Google Scholar]
- Peng C.-H., Chyau C.-C., Chan K.-C., Chan T.-H., Wang C.-J., Huang C.-N. (2011). Hibiscus sabdariffaPolyphenolic Extract Inhibits Hyperglycemia, Hyperlipidemia, and Glycation-Oxidative Stress while Improving Insulin Resistance. J. Agric. Food Chem. 59, 9901–9909. 10.1021/jf2022379 [DOI] [PubMed] [Google Scholar]
- Prasain J. K., Peng N., Rajbhandari R., Wyss J. M. (2012). The Chinese Pueraria Root Extract (Pueraria Lobata) Ameliorates Impaired Glucose and Lipid Metabolism in Obese Mice. Phytomedicine 20, 17–23. 10.1016/j.phymed.2012.09.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasanna R., Ashraf E. A., Essam M. A. (2017). Chamomile and Oregano Extracts Synergistically Exhibit Antihyperglycemic, Antihyperlipidemic, and Renal Protective Effects in Alloxan-Induced Diabetic Rats. Can. J. Physiol. Pharmacol. 95, 84–92. 10.1139/cjpp-2016-0189 [DOI] [PubMed] [Google Scholar]
- Raja M., Kinne R. K. H. (2015). Identification of Phlorizin Binding Domains in Sodium-Glucose Cotransporter Family: Sglt1 as a Unique Model System. Biochimie 115, 187–193. 10.1016/j.biochi.2015.06.003 [DOI] [PubMed] [Google Scholar]
- Ribnicky D. M., Poulev A., Watford M., Cefalu W. T., Raskin I. (2006). Antihyperglycemic Activity of Tarralin, an Ethanolic Extract of Artemisia Dracunculus L. Phytomedicine 13, 550–557. 10.1016/j.phymed.2005.09.007 [DOI] [PubMed] [Google Scholar]
- Rizvi S. I., Mishra N. (2013). Traditional Indian Medicines Used for the Management of Diabetes Mellitus. J. Diabetes Res. 2013, 712092. 10.1155/2013/712092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rtibi K., Selmi S., Grami D., Amri M., Eto B., El-benna J., et al. (2017). Chemical Constituents and Pharmacological Actions of Carob Pods and Leaves (Ceratonia Siliqua l.) on the Gastrointestinal Tract: A Review. Biomed. Pharmacother. 93, 522–528. 10.1016/j.biopha.2017.06.088 [DOI] [PubMed] [Google Scholar]
- Sambuy Y., De Angelis I., Ranaldi G., Scarino M. L., Stammati A., Zucco F. (2005). The Caco-2 Cell Line as a Model of the Intestinal Barrier: Influence of Cell and Culture-Related Factors on Caco-2 Cell Functional Characteristics. Cell Biol Toxicol 21, 1–26. 10.1007/s10565-005-0085-6 [DOI] [PubMed] [Google Scholar]
- Sanae F., Miyamoto K.-i., Sawanishi H., Kizu H., Tomimori Y., Sun J.-n., et al. (1996). Hypoglycaemic Effects ofShokatsu-Cha(Xiao-Ke-Ca) in Streptozotocin-Induced Diabetes Mellitus. Phytother. Res. 10, 127–130. [DOI] [Google Scholar]
- Schreck K., Melzig M. F. (2018). Intestinal Saturated Long-Chain Fatty Acid, Glucose and Fructose Transporters and Their Inhibition by Natural Plant Extracts in Caco-2 Cells. Molecules (Basel, Switzerland) 23, 2544. 10.3390/molecules23102544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreck K., Melzig M. F. (2019). The Expression of Selected Intestinal Glucose, Fructose and Long-Chain Fatty Acid Transporters Investigated in Caco-2 Cells. Planta Med. 85, 491. 30754052 [Google Scholar]
- Schulze C., Bangert A., Kottra G., Geillinger K. E., Schwanck B., Vollert H., et al. (2014). Inhibition of the Intestinal Sodium-Coupled Glucose Transporter 1 (Sglt1) by Extracts and Polyphenols from Apple Reduces Postprandial Blood Glucose Levels in Mice and Humans. Mol. Nutr. Food Res. 58, 1795–1808. 10.1002/mnfr.201400016 [DOI] [PubMed] [Google Scholar]
- Sears B., Perry M. (2015). The Role of Fatty Acids in Insulin Resistance. Lipids Health Dis. 14, 121. 10.1186/s12944-015-0123-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah M. A., Sarker D. M. M. R., Inamdar M. (2016). Antidiabetic Potential of brassica Oleracea Var. Italica in Type 2 Diabetic sprague Dawley (Sd) Rats. Int. J. Pharmacognosy Phytochem. Res. 8, 462–469. [Google Scholar]
- Shakeri A., Sahebkar A., JavadiMelissa officinalis B. l. (2016). Melissa Officinalis L. - A Review of its Traditional Uses, Phytochemistry and Pharmacology. J. Ethnopharmacology 188, 204–228. 10.1016/j.jep.2016.05.010 [DOI] [PubMed] [Google Scholar]
- Sidorova Y., Shipelin V., Mazo V., Zorin S., Petrov N., Kochetkova A. (2017). Hypoglycemic and Hypolipidemic Effect of Vaccinium Myrtillus L. Leaf and Phaseolus Vulgaris L. Seed Coat Extracts in Diabetic Rats. Nutrition 41, 107–112. 10.1016/j.nut.2017.04.010 [DOI] [PubMed] [Google Scholar]
- Smirin P., Taler D., Abitbol G., Brutman-Barazani T., Kerem Z., Sampson S. R., et al. (2010). Sarcopoterium Spinosum Extract as an Antidiabetic Agent: In Vitro and In Vivo Study. J. Ethnopharmacology 129, 10–17. 10.1016/j.jep.2010.02.021 [DOI] [PubMed] [Google Scholar]
- Smith S. C. (2007). Multiple Risk Factors for Cardiovascular Disease and Diabetes Mellitus. Am. J. Med. 120, S3–S11. 10.1016/j.amjmed.2007.01.002 [DOI] [PubMed] [Google Scholar]
- Stöger E. A. (2009). Arzneibuch der chinesischen medizin, monographien des arzneibuchs der volksrepublik china 2000 und 2005. Germany: Deutscher Apotheker Verlag Stuttgart. [Google Scholar]
- Sugiyama H., Akazome Y., Shoji T., Yamaguchi A., Yasue M., Kanda T., et al. (2007). Oligomeric Procyanidins in Apple Polyphenol Are Main Active Components for Inhibition of Pancreatic Lipase and Triglyceride Absorption. J. Agric. Food Chem. 55, 4604–4609. 10.1021/jf070569k [DOI] [PubMed] [Google Scholar]
- Swanston-Flatt S. K., Flatt P. R., Day C., Bailey C. J. (1991). Traditional Dietary Adjuncts for the Treatment of Diabetes Mellitus. Proc. Nutr. Soc. 50, 641–651. 10.1079/pns19910077 [DOI] [PubMed] [Google Scholar]
- Tahraoui A., El-Hilaly J., Israili Z. H., Lyoussi B. (2007). Ethnopharmacological Survey of Plants Used in the Traditional Treatment of Hypertension and Diabetes in South-Eastern morocco (Errachidia Province). J. Ethnopharmacology 110, 105–117. 10.1016/j.jep.2006.09.011 [DOI] [PubMed] [Google Scholar]
- Vuksan V., Sung M.-K., Sievenpiper J. L., Stavro P. M., Jenkins A. L., Di Buono M., et al. (2008). Korean Red Ginseng (Panax Ginseng) Improves Glucose and Insulin Regulation in Well-Controlled, Type 2 Diabetes: Results of a Randomized, Double-Blind, Placebo-Controlled Study of Efficacy and Safety. Nutr. Metab. Cardiovasc. Dis. 18, 46–56. 10.1016/j.numecd.2006.04.003 [DOI] [PubMed] [Google Scholar]
- Wang X., Liu Q., Zhu H., Wang H., Kang J., Shen Z., et al. (2017). Flavanols from the Camellia Sinensis Var. Assamica and Their Hypoglycemic and Hypolipidemic Activities. Acta Pharmaceutica Sinica B 7, 342–346. 10.1016/j.apsb.2016.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (2019). Diabetes. Available at: https://www.who.int/en/news-room/fact-sheets/detail/diabetes (Accessed April, , 20).
- WHO. Global Report on Diabetes; 2016. [Google Scholar]
- Yellanur Konda P., Egi J. Y., Dasari S., Katepogu R., Jaiswal K. K., Nagarajan P. (2020). Ameliorative Effects of Mentha Aquatica on Diabetic and Nephroprotective Potential Activities in Stz-Induced Renal Injury. Comp. Clin. Pathol. 29, 189–199. 10.1007/s00580-019-03042-6 [DOI] [Google Scholar]
- Zhang Y., Zhang H., Wang F., Yang D., Ding K., Fan J. (2015). The Ethanol Extract of Eucommia Ulmoides Oliv. Leaves Inhibits Disaccharidase and Glucose Transport in Caco-2 Cells. J. Ethnopharmacology 163, 99–105. 10.1016/j.jep.2015.01.015 [DOI] [PubMed] [Google Scholar]
- Zheng Y., Scow J. S., Duenes J. A., Sarr M. G. (2012). Mechanisms of Glucose Uptake in Intestinal Cell Lines: Role of Glut2. Surgery 151, 13–25. 10.1016/j.surg.2011.07.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, and further inquiries can be directed to the corresponding author.


