Abstract
Background/objective
Severe fever with thrombocytopenia syndrome (SFTS) cause encephalitis/encephalopathy, but few reports were available. We aimed to investigate the incidence of encephalitis/encephalopathy in SFTS patients and to summarize clinical characteristics, laboratory findings and imaging features.
Methods
We conducted a retrospective review of all patients with confirmed SFTS admitted to Nanjing Drum Tower Hospital, a tertiary hospital in Nanjing City, China, between January 2016 and July 2020. The patients were divided into two groups according to whether they had encephalitis/encephalopathy: encephalitis/encephalopathy group and non- encephalitis/encephalopathy group. Clinical data, laboratory findings, imaging characteristics, treatments and outcomes of these patients were collected and analyzed.
Results
A total of 109 SFTS patients with were included, of whom 30 (27.5 %) developed encephalitis/encephalopathy. In-hospital mortality (43.3 %) was higher in encephalitis/encephalopathy group than non-encephalitis/encephalopathy group (12.7 %). Univariate logistic regression showed that cough, wheezing, dyspnoea, respiratory failure, vasopressors use, bacteremia, invasive pulmonary aspergillosis (IPA) diagnoses, PCT > 0.5 ug/L, CRP > 8 mg/L, AST > 200 U/L and serum amylase level > 80 U/L were the risk factors for the development of encephalitis/encephalopathy for SFTS patients. Multivariate logistic regression analysis identified bacteremia, PCT > 0.5 mg/L and serum amylase level > 80 U/L as independent predictors of encephalitis/ encephalopathy development for SFTS patients.
Conclusions
SFTS-associated encephalitis/encephalopathy has high morbidity and mortality. it was necessary to strengthen the screening of CSF testing and brain imaging after admission for SFTS patients who had symptoms of encephalitis/encephalopathy. SFTS patients with bacteremia, PCT > 0.5 ug/L or serum amylase level > 80 U/L should be warned to progress to encephalopathy.
Keywords: Encephalitis, Encephalopathy, Severe fever with thrombocytopenia syndrome, Cerebrospinal fluid, Bacteremia, Vasopressor, Amylase
Background
Severe fever with Thrombocytopenia syndrome virus (SFTSV), a novel Banyangvirus genus in the Phenuiviridae family, was first discovered in China in 2009 [1]. Since then, the virus has also been isolated in South Korea, Japan, Vietnam, Taiwan, Myanmar, Thailand and Pakistan [2–7]. SFTS is a kind of viral hemorrhagic fever with high fatality rate and patients with SFTS show common symptoms such as a sudden fever, vomiting, stomachache, diarrhea, muscle soreness and some hemorrhagic symptoms [8, 9]. Severe cases can cause central nervous system (CNS) symptoms and are thought to be related to the severity of the disease[8] . The fatality rate of SFTS patients with encephalitis was reported as high as 44.7 % [10]. However, to date, few reports of CNS complications in SFTS patients were few and have a detailed CSF testing [10, 11]. Therefore, we investigated the clinical and laboratory findings in SFTS patients with confirmed encephalitis/encephalopathy.
Methods
Study population
We conducted a retrospective review of the patients with confirmed SFTS admitted to Nanjing Drum Tower Hospital, a tertiary hospital in Nanjing, China, between January 2016 and July 2020. The requirement for informed consent by individual patients was waived by Ethical Committee of Drum Tower Hospital affiliated with the Medical School of Nanjing University given the retrospective nature of the study.
Diagnostic criteria for SFTS and SFTS-associated encephalopathy/ encephalitis
The criteria for confirmed SFTS were defined by (a) acute fever, (b) thrombocytopaenia, (c) detection of SFTSV RNA using polymerase chain reaction, detection of IgM against SFTSV, or isolation and culture positive of SFTSV [12]. The criteria for clinical diagnosed encephalopathy/encephalitis was defined as meeting the following criteria: (1) major criterion (required): Patients presenting to medical attention with altered mental status (defined as decreased or altered level of consciousness, lethargy or personality change) lasting ≥ 24 h with no alternative cause identified. (2) minor criteria (2 required for possible encephalitis; ≥ 3 required for probable or confirmed encephalitis): ① documented fever ≥ 38° C (100.4 °F) within the 72 h before or after presentation; ② generalized or partial seizures not fully attributable to a preexisting seizure disorder; ③ new onset of focal neurologic findings; ④ CSF WBC count ≥ 5/cubic mm; ⑤ abnormality of brain parenchyma on neuroimaging suggestive of encephalitis that is either new from prior studies or appears acute in onset; ⑥ abnormality on electroencephalography that is consistent with encephalitis and not attributable to another cause [13]. The criteria for clinical diagnosed SFTS-associated encephalopathy/encephalitis: meet SFTS criteria and encephalopathy/encephalitis simultaneously.
Clinical data collection
Investigators collected clinical data through the electronic medical record system in Nanjing Drum Tower Hospital, a tertiary hospital in Nanjing, China, including demographic data, underling diseases, clinical symptoms and laboratory findings, comorbidity, treatment and overall prognosis were also recorded.
Statistical analysis
Data were analyzed with SPSS 21.0 (SPSS Inc., Chicago, USA) for all statistical analysis. Data are reported as percentage for categorical variables and as mean ± standard deviation (SD) or median with interquartile range (IQR), as appropriate, for continuous variables. Chi-square test or Fisher’s exact test were used for categorical variables. T-test was used for continuous variables, as propriate. All tests of significance were 2-sided, and p < 0.05 was considered statistically significant. Univariate and multivariate logistic regression analyses were undertaken to examine risk factors for the development of encephalitis/encephalopathy. The results are reported as adjusted odds ratio of death with corresponding 95 % confidence intervals.
Results
Clinical characteristics
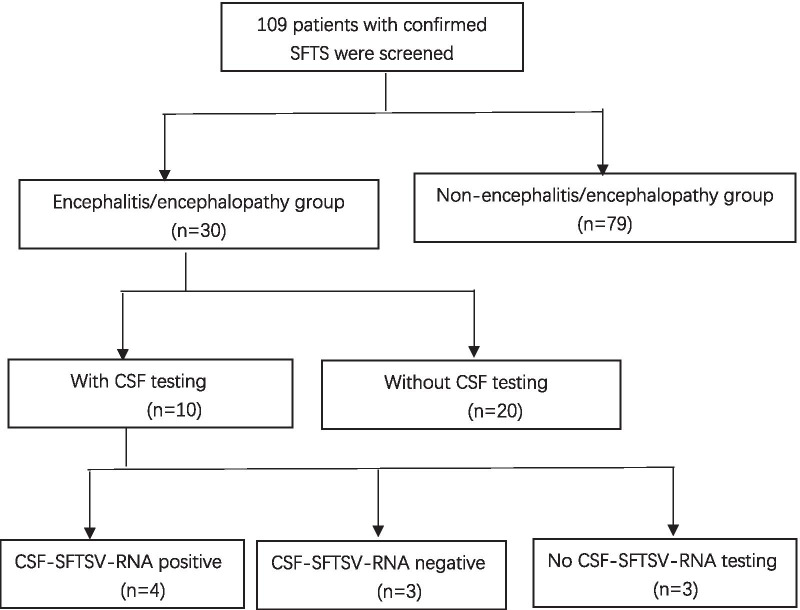
Between January 2016 and July 2020, a total 109 cases were confirmed with SFTS diagnosed by positive detection of SFTSV through polymerase chain reaction (PCR) for RNA. Of these 109 patients, 30 (27.5 %) cases were diagnosed with SFTSV-associated encephalitis/encephalopathy (Fig. 1). Among them, 10 cases performed cerebrospinal fluid (CSF) examination (CSF SFTSV-RNA test was positive in 4 cases, negative in 3 cases, and not available in 3 cases).
Fig. 1.
Schematic flow chart of patient enrolment. SFTS severe fever with thrombocytopenia syndrome, CSF cerebrospinal fluid
Table 1 presents the differences in demographic characteristics, underlying diseases, clinical manifestations, laboratory finding at admission, comorbidity, treatment and outcome parameters between encephalitis/ encephalopathy and non-encephalitis/encephalopathy patients. 45/109 (41.3 %) cases were male and there were more male patients in encephalitis /encephalopathy group than non-encephalitis/encephalopathy group (p = 0.028). The mean age was 61 years in all patients and no differences between the two groups.
Table 1.
Clinical characteristics of patients with SFTS-associated encephalitis/encephalopathy
| All SFTS patients (N = 109) |
Encephalitis/encephalopathy patients (N = 30) |
Non-encephalitis/encephalopathy patients (N = 79) |
P | |
|---|---|---|---|---|
| Demographic characteristics | ||||
| Age, years | 60.69 ± 12.53 | 64.07 ± 10.21 | 59.34 ± 13.16 | 0.086 |
| Sex, male (%) | 45 (41.3) | 18 (60) | 27(34.2) | 0.028 |
| Days prior to admission (days) | 6.95 ± 2.42 | 6.73 ± 2.30 | 7.04 ± 2.57 | 0.559 |
| Underlying diseases, n (%) | ||||
| Hypertension | 24 (22.2) | 9 (30) | 15 (19) | 0.163 |
| Diabetes | 12 (11) | 6 (20) | 6 (7.6) | 0.071 |
| Clinical manifestation, n (%) | ||||
| Fever | 109 (100) | 30 (100) | 79 (100) | – |
| Headache | 44(40.4) | 30 (100) | 14 (17.7) | < 0.001 |
| Cough | 36 (33.3) | 15 (50) | 21 (26.6) | 0.014 |
| Wheezing | 33 (30.3) | 16 (53.3) | 17 (21.5) | 0.002 |
| Dyspnoea | 10 (9.23) | 6 (20) | 4 (5.1) | 0.013 |
| Vomiting/diarrhoea | 10 (9.2) | 1 (0.9) | 9 (8.3) | 0.002 |
| Laboratory findings at admission | ||||
|
Leukocytes (*109/L) (normal range, 4–10) |
2.85 (1.70–4.63) | 2.80 (1.78–4.55) | 3 (1.63–4.68) | 0.763 |
| Percentage of neutrophils (%) (normal range, 50–70) | 55.10 (39.58–72.60) | 58.8 (48.9-74.78) | 52.15 (38.03–72.60) | 0.167 |
|
Percentage of lymphocytes (%) (normal range, 20–40) |
34.05 (20.70-45.35) | 30.1 (21.35–41.83) | 35.8 (20.7-47.23) | 0.309 |
|
Haemoglobin (g/L) (normal range, 110–160) |
127.50 (115–142) | 130.5 (118–143) | 126.5(114.75-141.25) | 0.433 |
|
Platelets (*109/L) (normal range, 100–300) |
47.50 (32–66.25) | 42.5 (29.5-64.25) | 49 (33.25–69.75) | 0.208 |
|
PCT (µg/L) (normal range,0-0.5) |
0.30 (10.12–0.86) | 0.58 (0.24–3.17) | 0.22 (0.1–0.73) | 0.005 |
|
CRP (mg/L) (normal range,0–8) |
6.8 (3.15–15.2) | 11.55 (4.15–38.75) | 5.4 (3–12) | 0.016 |
|
ALT (U/L) (normal range, 0–40) |
73.2 (51.05–137.2) | 83 (60.58-168.43) | 71.1 (148.8-120.7) | 0.167 |
|
AST (U/L) (normal range, 0–40) |
159.05 (89.55–264.40) | 231.90 (129.93-547.73) | 122.3 (88.28–234.75) | 0.008 |
|
LDH(U/L) (normal range, 109–245) |
1679 (1326–2595) | 1683 (1245–2788) | 1430 (1125–2846) | 0.623 |
|
CK(U/L) (normal range, 25–200) |
1027 (506–1528) | 1110 (392–2020) | 993 (465–1720) | 0.912 |
|
Amylase (U/L) (normal range, 25–115) |
119 (61–195) | 185 (87–266.5) | 105 (47–163) | 0.003 |
| Comorbidity, n (%) | ||||
| Respiratory failure | 10 (9.2) | 6 (20) | 4 (5.1) | 0.013 |
| Vasopressors use | 27 (24.8) | 13 (43.3) | 14 (17.7) | 0.001 |
| Acute kidney injury | 17 (15.6) | 8 (26.7) | 9 (11.4) | 0.074 |
| Bacteraemia | 12 (11) | 8 (26.7) | 4 (5.1) | < 0.001 |
| Probable or proven-IPA | 38 (34.9) | 17 (56.7) | 21 (26.6) | 0.004 |
| Treatment, n (%) | ||||
| Immunoglobulin | 35 (32.1) | 14 (46.7) | 21 (26.6) | < 0.001 |
| Ribavirin | 102 (93.6) | 30 (100) | 72 (91.1) | 0.097 |
| Methylprednisolone | 22 (20.2) | 10 (33.3) | 12 (15.2) | < 0.001 |
| Outcome | ||||
| ICU transfer, n (%) | 16 (14.7) | 11 (36.7) | 5 (6.3) | < 0.001 |
| Length of hospital stay (days) | 9 (6.5–13) | 9 (6–11.25) | 10 (7–13) | 0.272 |
| In-hospital mortality, n (%) | 23 (21.1) | 13 (43.3) | 10 (12.7) | < 0.001 |
PCT procalcitonin, CRP C-reactive protein, AST aspartate aminotransferase, ALT alanine aminotransferase, LDH lactic dehydrogenase, CK creatine kinase, IPA invasive pulmonary aspergillosis
The most common underlying diseases in SFTS patients were hypertension (22.2 %) and diabetes (11 %), and no differences between the two groups. All patients presented with a high fever (100 %), other common symptoms included headache (40.4 %), cough (33.3 %), wheeze (30.3 %), dyspnea (9.23 %), and vomiting/diarrhoea (9.2 %). Among them, patients with cough, wheezing, dyspnea and vomiting/diarrhoea in encephalitis/ encephalopathy group were significantly higher than those in non- encephalitis/ encephalopathy group (p all < 0.05).
Laboratory findings at admission were also showed in Table 1. The leukocyte count with median 2.85 *109/L decreased significantly but the ratio of neutrophils and lymphocytes remained within the normal range. All cases had decreased platelet count with median 34.05 *109/L. C-reactive protein (CRP) and procalcitonin (PCT) in encephalitis/encephalopathy group were both slightly increased, which was statistically different from that in the non- encephalitis/encephalopathy group (p < 0.05). The median value of serum aspartate aminotransferase (AST) and aspartate aminotransferase (ALT) levels were elevated almost in all cases and AST in encephalitis/encephalopathy group obviously raised than non-encephalitis/encephalopathy group (p = 0.008). The serum amylase level with median 119 U/L was increase remarkably in SFTS patients and higher in encephalitis/encephalopathy group than non-encephalitis/encephalopathy group (p = 0.003).
Comorbidity include respiratory failure (10/109, 9.2 %), vasopressors use (27/109, 24.8 %), acute kidney injury (17/109, 15.6 %), bacteremia (12/109, 11 %), and probable or proven-invasive pulmonary aspergillosis (IPA) (38/109, 34.9 %). The rate of respiratory failure, vasopressors use, bacteremia and IPA in encephalitis/encephalopathy group was significantly higher than that in non-encephalitis/encephalopathy group (p all < 0.05).
Immunoglobulin and methylprednisolone were used in 35/109 (32.1 %) and 22/109 (20.2 %) cases, respectively, and the rate of encephalitis/ encephalopathy group was also significantly higher than those in non-encephalitis/encephalopathy group (p < 0.001). 102 (93.6 %) patients were treated with ribavirin as antiviral treatment in in-hospital period and had no difference between the two groups.
16 (14.7 %) cases were transferred to intensive care unit (ICU) and encephalitis/encephalopathy group had higher rate of ICU transfer (p < 0.001), but the length of hospital stay was no differences in two groups. In-hospital mortality totally was 21.1 % and higher in encephalitis/encephalopathy patients (43.3 %) than non-encephalitis/encephalopathy group (12.7 %, p < 0.001).
Clinical data of SFTS-associated encephalitis/encephalopathy with cerebrospinal fluid (CSF) testing
As shown in Table 2 and 10 cases of confirmed SFTS-associated encephalitis/encephalopathy had performed CSF testing. 2 (20 %) cases were elevated in count of white blood cell (WBC) and 7 cases (70 %) of neutrophil percentage were raised in CSF testing. The protein level in CSF of 9 cases went up dramatically and case 9 is within normal range. 4 cases (40 %) were elevated and 2 cases (20 %) were decreased in glucose level of CSF. There were no obvious abnormalities in CSF chlorine levels in 10 cases. Elevated IgG level of 2 cases was observed. Among the 10 cases, CSF-SFTSV-RNA load of 7 cases were available and 4 cases were positive and 3 cases negative. Compared to blood-SFTSV-RNA load, CSF-SFTSV-RNA load of the 4 positive cases were all obviously lower. Of 10 cases, brain imaging of 9 cases was available and all were abnormal. Among them, the most common brain CT findings were lacunar infarction (6/9), and the most brain MRI findings were hypoxic changes in white matter (3/4). In case 7, intracerebral hemorrhage (ICH) was shown on brain CT scan, and multiple abnormal signals were shown on brain MRI scan, simultaneously. Combined with confirmed IPA diagnosis in this patient, the possibility of intracranial fungal infection was considered (Fig. 2). Of the 10 cases, 8 were co-infected, 7 were IPA, 5 were bacteremia, 4 were combined with IPA and bacteremia. Nine of the 10 cases died (Table 2).
Table 2.
Clinical presentation, CSF findings, SFTS-DNA testing and imaging findings in patients with SFTS-associated encephalopathy/encephalitis who underwent CSF testing (n = 7)
| Case no. | Age /sex |
Days to neurological presentation | Neurological presentation | CSF findings | Blood SFTSV- RNA load (copies/ml) |
Brain CT | Brain MRI | Co-infection | Outcome | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WBC (*106/L) |
Neutrophils (%) |
Lymphocyte (%) |
Protein (mg/L) | Glucose (mmol/L) |
Chlorine (mmol/L) |
IgG (mg/L) | SFTSV-RNA load (copies/ml) | |||||||||
| Normal range | 0–8 | 0–6 | 40–80 | 140–450 | 2.5–4.5 | 120–132 | 4.8–58.6 | Negative | Negative | |||||||
| 1 | 61/M | 6th day | Unconsciousness, seizure | 9.0 | 35 | 65 | 736 | 5.14 | 130.9 | 187 | 103/L | 106/L | Lacunar infarction | Chronic hypoxia changes in white matter | IPA | Died |
| 2 | 71/M | 4th day | Unconsciousness | 6.0 | 66.6 | 33.4 | 976 | 2.41 | 123 | 148 | 103/L | 104/L | Lacunar infarction | Chronic hypoxia changes in white matter | IPA, bacteremia | Died |
| 3 | 71/F | 4th day | Altered mental status, disorientation | 5.0 | 20 | 80 | 469 | 3.22 | 130 | NA | 103/L | 106/L | Lacunar infarction | NA | IPA | Died |
| 4 | 64/M | 4th day | Unconsciousness, seizure | 1.0 | 0 | 100 | 572 | 3.97 | 126.2 | 53.1 | 103/L | 106/L | Lacunar infarction | NA | Bacteremia | Died |
| 5 | 45/M | 7th day | Headache, altered mental status | 1.0 | 0 | 100 | 542 | 3.26 | 133 | 29.5 | Negative | 106/L | Lacunar infarction | Chronic hypoxia changes in white matter | No | Survived |
| 6 | 76/F | 3rd day | Unconsciousness | 0 | 0 | 0 | 914 | 5.35 | 135.3 | 61 | Negative | 108/L | NA | NA | No | Died |
| 7 | 28/M | 5th day | Headache, altered mental status | 50 | 36 | 54 | 1226 | 1.33 | 129 | NA | Negative | 108/L | ICH | Multiple abnormal signal shadows | IPA | Died |
| 8 | 61/F | 4th day | Headache, altered mental status | 3 | 33.3 | 66.7 | 498 | 9.77 | 119 | NA | NA | 106/L | Normal | NA | IPA, bacteremia | Died |
| 9 | 49/M | 6th day | Headache, altered mental status | 4 | 25 | 75 | 416 | 4.02 | 126 | NA | NA | 106/L | NA | NA |
IPA, bacteremia |
Died |
| 10 | 75/M | 4th day | Headache, altered mental status | 7 | 50 | 50 | 904 | 6.16 | 138 | NA | NA | 104/L | Lacunar infarction | NA | IPA, bacteremia | Died |
SFTS severe fever with thrombocytopenia syndrome, CSF cerebrospinal fluid, WBC white blood cell, ICH intracranial haemorrhage, NA not available
Fig. 2.
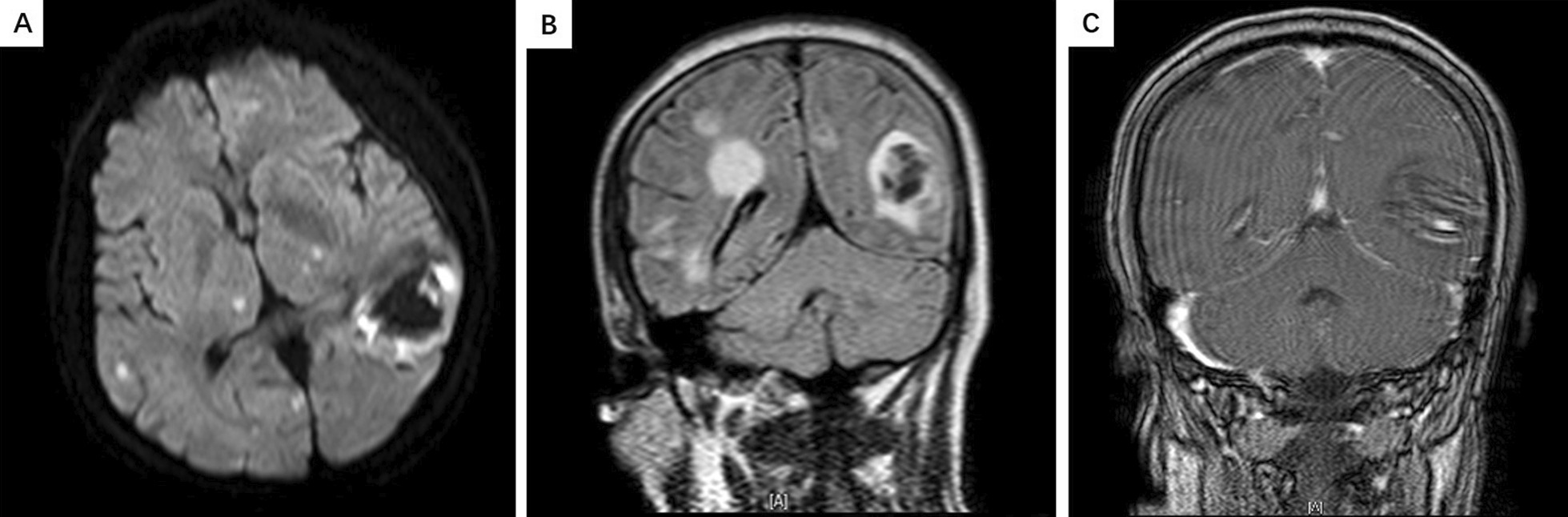
A Brain MRI showed patchy mixed signal shadows in the left occipital lobe, with a diameter of approximately 3.9 cm. B, C Short T1 signal shadows were visible, and patchy enhancement was observed. Multiple patchy slightly longer T2 signal shadows can be seen in both cerebral hemispheres. DWI phase shows high signal intensity, surrounding areas of oedema, and enhanced patchy enhancement
Univariate and multivariate analyses of risk factors for the development of encephalitis/encephalopathy for SFTS patients
Univariate logistic regression showed that cough, wheezing, dyspnoea, respiratory failure, vasopressors use, bacteremia, IPA diagnoses, PCT > 0.5 ug/L, CRP > 8 mg/L, AST > 200 U/L and serum amylase level > 80 U/L were the risk factors for the development of encephalitis/encephalopathy for SFTS patients. Multivariate logistic regression analysis identified bacteremia, PCT > 0.5 ug/L and serum amylase level > 80 U/L as independent predictors of encephalitis/encephalopathy development for SFTS patients (Table 3).
Table 3.
Univariate and multivariate logistic regression analyses of risk factors for the development of encephalitis/encephalopathy in patients with SFTS
| Univariate | Multivariate | |||
|---|---|---|---|---|
| OR (95 % CI) | P value | OR (95 % CI) | P value | |
| Sex, male | 2.091 (0.888–4.923) | 0.091 | ||
| Hypertension | 1.829 (0.699–4.786) | 0.219 | ||
| Diabetes | 3.042 (0.896 ~ 10.323) | 0.074 | ||
| Cough | 2.762 (1.154 ~ 6.609) | 0.022 | ||
| Wheezing | 4.168 (1.702 ~ 10.209) | 0.002 | ||
| Dyspnoea | 3.700 (1036–13.215) | 0.044 | ||
| Vomiting/diarrhoea | 3.729 (0.452 ~ 30.780) | 0.222 | ||
| Respiratory failure | 5.707 (1.533 ~ 21.240) | 0.009 | ||
| Vasopressor use | 5.306 (2.113 ~ 13.326) | < 0.001 | ||
| Bacteraemia | 9.375 (2.659 ~ 33.053) | < 0.001 | 7.211 (1.459–35.635) | 0.015 |
| Probable or proven-IPA | 3.612 (1.501 ~ 8.689) | 0.004 | ||
| PCT > 0.5 µg/L | 2.581 (1.072 ~ 6.213) | 0.034 | 4.008 (1.110–14.479) | 0.034 |
| CRP > 8 mg/L | 3.437 (1.435 ~ 8.236) | 0.006 | ||
| AST > 200 U/L | 4.771 (1.950 ~ 11.672) | 0.001 | ||
| Amylase > 80 U/L | 7.929 (2.222 ~ 28.290) | 0.001 | 9.094 (1.761–46.935) | 0.008 |
PCT procalcitonin, CRP C-reactive protein, AST aspartate aminotransferase
Discussion
This retrospective study showed that 27.5 % of SFTS patients admitted to our hospital developed encephalitis/encephalopathy in our hospital from January 2016 to July 2020, while 13–34 % in other studies [10, 11, 14]. The fatal outcome of the encephalitis/encephalopathy patients was high as 43.3 % in our study while 44.7 % reported in others [10].
There are few data on the CSF data of patients with SFTS-associated encephalitis/encephalopathy. Cui et al. reported that 103 (19 %) of 538 SFTS patients developed encephalitis, and evidence of SFTSV was found in CSF in two patients [10]. The protein and glucose levels in the CSF of the two patients with positive-SFTSV were slightly increased. Park et al. also reported that 14 (34 %) of 41 SFTS patients developed encephalitis but revealed normal protein and glucose levels but plecytosis in all the six patients with positive-SFTSV [11]. The copy number of SFTS virus in CSF in the six cases was lower than that in serum. However, Kim et al. found that on the 12th day of the course of disease, the copy number of virus in CSF was higher than that in serum, indicating that SFTSV was neurotropic [15]. In our study, among the 4 patients with positive-SFTSV in CSF, only one patient had plecytosis, but all the 4 patients had mildly elevated protein level and 2 patients with elevated IgG in CSF. The SFTSV-RNA viral load in CSF was also lower than that in serum. These results indicate that SFTSV direct infection is one of the important mechanisms of SFTSV-associated encephalitis/encephalopathy.
Some studies showed that SFTS patients occasionally suffered from multiple organ failure, with abnormal serum levels of various pro-inflammatory cytokines, which were related to disease severity and mortality in the acute stage of infection [16, 17]. In Nakamura et al. study, the levels of interleukin-10 (IL-10), interferon-gamma (IFN-γ) and interferon induced protein 10 (IP-10) were significantly increased in patients with acute stage of SFTV infection [18]. In other studies, hemophagocytes were found in CSF of patients with SFTSV-associated encephalitis, suggesting that cytokine and chemokine storms may indirectly lead to the disturbance of consciousness in SFTS patients [19, 20]. CNS involvement is closely related to high cytokine and viral load in serum. The high cytokine level caused by viral infection leads to increased vascular permeability, and SFTSV can enter the nervous system through the blood-brain barrier and cause intracranial infection, which may be an important cause of SFTS-associated encephalitis/encephalopathy.
SFTS-associated encephalitis/encephalopathy is also related to the immune state of patients. In some studies SFTSV was not detected in CSF or even in some autopsy cases [12, 19, 21, 22]. CD4 + T cells decreased significantly in blood of patients in the acute stage of SFTSV infection, while CD8 + T cells increased or did not change significantly, accompanied by dramatic decrease in NK cells, while the number of CD4 + and CD8 + T cells further decreased in blood, while the number of NK cells further increased. When the cellular immune function is impaired, mainly mediated by CD4 + and CD8 + T cells, the immune transition response caused by the high activation of NK cells is an important pathological basis for multiple organ injury including CNS in severe SFTS patients. Furthermore, the increase of B cells in blood of severe SFTS patients is more obvious, and B cells can further differentiate into plasma cells [23]. The recent study found that within 3 weeks after the onset of SFTS, the proportion of plasma cells in non-survivals to B cells was higher than survivors, but they were non-functional plasma cells [24]. Apoptosis of monocytes in the early stage of SFTSV infection in the non-survivors reduced the antigen presentation of DC and affected the differentiation and function of CD4 + T cells, which was an important cause of virus-specific humoral immunodeficiency in non-survivors.
In our study, multivariate analysis identified 3 variables (bacteremia, PCT > 0.5ug/L and serum amylase level > 80U/L levels) as independent predictors for the development of encephalitis/encephalopathy in patients with SFTS. However, no other published manuscripts have found that these three factors associated with the incidence of SFTS-associated encephalitis/encephalopathy, but according to the pathogenesis of SFTS-associated encephalitis/encephalopathy, cytokine and chemokine storms lead to the increase of capillary permeability, secondary infection such as gut-origin sepsis, so bacteremia is independent risk factors for the development of SFTS-associated encephalitis/encephalopathy. The mechanism of hyperamylasemia in SFTS-associated encephalitis/encephalopathy patients is not clear. Studies have shown that cytokine storm played a key role in the development of the occurrence of acute pancreatitis, main show is hyperamylasemia, therefore, may be higher serum amylase level may be associated with cytokine storm [25, 26], Other studies have also found that the application of ribavirin may also lead to hyperamylasemia as a side effect [27, 28]. Therefore, to a certain extent, these three independent risk factors (bacteremia, PCT > 0.5 ug/L and serum amylase level > 80U/L levels) for the development of SFTS-associated encephalitis/encephalopathy reflect the severity of cytokine storm. The more severe the cytokine storm, the greater the chance of SFTS-associated encephalitis/encephalopathy. These three variables were routinely evaluated in clinical practice, thus yielding a highly predictive value for discriminating the patients at higher risk of encephalitis/encephalopathy, who should get more attention in treatment.
The specific treatment of SFTS is still unknown. In 2012, the Ministry of Health of China recommended ribavirin intravenous therapy (500 mg per day) in SFTS Treatment Guidelines. Ribavirin is a synthetic nucleoside broad-spectrum antiviral drug that has inhibitory activity against both DNA and RNA viruses [29]. In recent years, more and more clinical data have shown that ribavirin does not increase platelet count nor reduce serum viral load [8, 14, 30]. The in vitro experimental study conducted by Shimojima et al. compared the antiviral efficacy of ribavirin before and after 3 days of SFTSV inoculation, and found that ribavirin used before inoculation could significantly inhibit the proliferation of the virus, while the inhibitory ability of antiviral drugs decreased significantly after inoculation. Therefore, it is speculated that ribavirin is more suitable for the post-exposure prophylaxis of SFTS [31]. Because the virus into the blood is still in the incubation period, the patient has no clinical symptoms, no treatment measures. After 5–14 days of incubation and onset, the virus has already multiplied in large numbers in cells, and ribavirin can no longer reverse virus replication. It is inferred that the poor treatment effect of ribavirin may be due to missing the optimal treatment time.
Recently, studies have conducted some new treatment attempts for SFTS patients. More attempt to treat SFTS-associated encephalitis/ encephalopathy patients is corticosteroid pulse therapy. Corticosteroid pulse therapy can inhibit the overproduction of cytokines, thus reducing organ failure [32]. However, it is worth noting that steroid therapy may not always be beneficial for secondary infection complications (such as IPA) of SFTS patients [33]. However, the number of lymphocytes in the early stage is significantly reduced in SFTS patients [34], and the use of steroid can lead to further reduction of lymphocytes, which is not benefit to the body’s immune function. Gamma-globulin has been shown to effectively inhibit macrophage activation and cytokine storms in Crimean Congo haemorrhagic fever virus infection [35]. The application of gamma globulin in the treatment of severe SFTS patients has been reported [36]. The mechanism is to block virus replication by complementing non-specific anti-virus, anti-bacterial and anti-other pathogens IgG antibody in the body, thus playing a role of neutralizing toxin. Further evidence on the application of gamma globulin in the treatment of SFTS patients needs to be accumulated. Another attempt of plasmapheresis for SFTS patients has been reported recently [18, 37–39]. Studies have shown that plasmapheresis can reestablish homeostasis and improve the coagulation status of patients. To date, steroids are mostly used in combination with plasma exchange, immunoglobulin and antiviral drugs, which makes it difficult to distinguish the effects of these therapies for SFTS patients.
Our study has several limitations. First, there were a relatively small number of cases because of the relatively low incidence of SFTS-associated encephalitis/encephalopathy. Second, the study was a retrospective study, CSF-SFTSV-RNA screening tests were not available for all patients. Finally, the study is a single-center study and the results may be limited.
Conclusions
This study showed that there was a high incidence and mortality of encephalitis/encephalopathy in SFTS patients, it is necessary to strengthen the treatment of SFTS patients with neurological symptoms. SFTS patients with bacteremia, PCT > 0.5 ug/L or serum amylase level > 80U/L should be warned to progress to encephalitis/encephalopathy.
Authors' contributions
All work has been approved by all co-authors. YX and QG made substantial contributions to the conception and design; the acquisition of data was performed YX; the analysis and interpretation of data were performed by NL, DJD and JT; YX and QG wrote the draft of the article and revised it critically for intellectual content. MRS re-evaluated and added content on the brain imaging outcomes. No conflicts of interest exist in the submission of this manuscript. I would like to declare on behalf of my co-authors that the work described was original research that has not been published previously and is not under consideration for publication elsewhere, in whole or in part. All authors read and approved the final manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due individual privacy of patients could be compromised, but are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Ethical Committee of Drum Tower Hospital affiliated with the Medical School of Nanjing University. The data used in this study was anonymised before its use and were collected during routine procedures, which did not pose any additional risk to the patients. The requirement for informed consent by individual patients was waived by Ethical Committee of Drum Tower Hospital affiliated with the Medical School of Nanjing University given the retrospective nature of the study.
Consent for publication
Not applicable.
Competing interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Yu XJ, Liang MF, Zhang SY, et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med. 2011;21(16):1523–1532. doi: 10.1056/NEJMoa1010095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kim WY, Choi W, Park SW, et al. Nosocomial transmission of severe fever with thrombocytopenia syndrome in Korea. Clin Infect Dis. 2015;1(11):1681–1683. doi: 10.1093/cid/civ128. [DOI] [PubMed] [Google Scholar]
- 3.Kobayashi Y, Kato H, Yamagishi T, et al. Severe fever with thrombocytopenia syndrome, Japan, 2013–2017. Emerg Infect Dis. 2020;26(4):692–699. doi: 10.3201/eid2604.191011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tran XC, Yun Y, Van An L, et al. Endemic Severe Fever with Thrombocytopenia Syndrome, Vietnam. Emerg Infect Dis. 2019;25(5):1029–1031. doi: 10.3201/eid2505.181463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lin TL, Ou SC, Maeda K, et al. The first discovery of severe fever with thrombocytopenia syndrome virus in Taiwan. Emerg Microbes Infect. 2020;9(1):148–151. doi: 10.1080/22221751.2019.1710436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Win AM, Nguyen YTH, Kim Y, et al. Genotypic heterogeneity of Orientia tsutsugamushi in scrub typhus Patients and thrombocytopenia syndrome co-infection, Myanmar. Emerg Infect Dis. 2020;26(8):1878–1881. doi: 10.3201/eid2608.200135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zohaib A, Zhang J, Saqib M, et al. Serologic Evidence of Severe Fever with Thrombocytopenia Syndrome Virus and Related Viruses in Pakistan. Emerg Infect Dis. 2020;26(7):1513–1516. doi: 10.3201/eid2607.190611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gai ZT, Zhang Y, Liang MF, et al. Clinical progress and risk factors for death in severe fever with thrombocytopenia syndrome patients. J Infect Dis. 2012;1(7):1095–1102. doi: 10.1093/infdis/jis472. [DOI] [PubMed] [Google Scholar]
- 9.Saijo M. Pathophysiology of severe fever with thrombocytopenia syndrome and development of specific antiviral therapy. J Infect Chemother. 2018;24(10):773–781. doi: 10.1016/j.jiac.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 10.Cui N, Liu R, Lu QB, et al. Severe fever with thrombocytopenia syndrome bunyavirus-related human encephalitis. J Infect. 2015;70(1):52–59. doi: 10.1016/j.jinf.2014.08.001. [DOI] [PubMed] [Google Scholar]
- 11.Park SY, Kwon JS, Kim JY, et al. Severe fever with thrombocytopenia syndrome-associated encephalopathy/encephalitis. Clin Microbiol Infect. 2018;24(4):432. doi: 10.1016/j.cmi.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 12.Yoshikawa T, Fukushi S, Tani H, et al. Sensitive and specific PCR systems for detection of both Chinese and Japanese severe fever with thrombocytopenia syndrome virus strains and prediction of patient survival based on viral load. J Clin Microbiol. 2014;52(9):3325–3333. doi: 10.1128/JCM.00742-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Venkatesan A, Tunkel AR, Bloch KC, et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: consensus statement of the international encephalitis consortium. Clin Infect Dis. 2013;57(8):1114–1128. doi: 10.1093/cid/cit458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Deng B, Zhou B, Zhang S, et al. Clinical features and factors associated with severity and fatality among patients with severe fever with thrombocytopenia syndrome Bunyavirus infection in Northeast China. PloS One. 2013;8(11):e80802. doi: 10.1371/journal.pone.0080802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim UJ, Kim DM, Kim SE, et al. Case report: detection of the identical virus in a patient presenting with severe fever with thrombocytopenia syndrome encephalopathy and the tick that bit her. BMC Infect Dis. 2018;17(1):181. doi: 10.1186/s12879-018-3092-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sun Y, Jin C, Zhan F, et al. Host cytokine storm is associated with disease severity of severe fever with thrombocytopenia syndrome. J Infect Dis. 2012;1(7):1085–1094. doi: 10.1093/infdis/jis452. [DOI] [PubMed] [Google Scholar]
- 17.Deng B, Zhang S, Geng Y, et al. Cytokine and chemokine levels in patients with severe fever with thrombocytopenia syndrome virus. PloS One. 2012;7(7):e41365. doi: 10.1371/journal.pone.0041365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nakamura S, Iwanaga N, Hara S, et al. Viral load and inflammatory cytokine dynamics associated with the prognosis of severe fever with thrombocytopenia syndrome virus infection: an autopsy case. J Infect Chemother. 2019;25(6):480–484. doi: 10.1016/j.jiac.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 19.Kawaguchi T, Matsuda M, Takajo I, et al. Severe fever with thrombocytopenia syndrome with myocardial dysfunction and encephalopathy: A case report. J Infect Chemother. 2016;22(9):633–637. doi: 10.1016/j.jiac.2016.01.022. [DOI] [PubMed] [Google Scholar]
- 20.Kaneko M, Shikata H, Matsukage S, et al. A patient with severe fever with thrombocytopenia syndrome and hemophagocytic lymphohistiocytosis-associated involvement of the central nervous system. J Infect Chemother. 2018;24(4):292–297. doi: 10.1016/j.jiac.2017.10.016. [DOI] [PubMed] [Google Scholar]
- 21.Uehara N, Yano T, Ishihara A, Saijou M, Suzuki T. Fatal severe fever with thrombocytopenia syndrome: an autopsy case report. Intern Med. 2016;55(7):831–838. doi: 10.2169/internalmedicine.55.5262. [DOI] [PubMed] [Google Scholar]
- 22.Hiraki T, Yoshimitsu M, Suzuki T, et al. Two autopsy cases of severe fever with thrombocytopenia syndrome (SFTS) in Japan: a pathognomonic histological feature and unique complication of SFTS. Pathol Int. 2014;64(11):569–575. doi: 10.1111/pin.12207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sun L, Hu Y, Niyonsaba A, et al. Detection and evaluation of immunofunction of patients with severe fever with thrombocytopenia syndrome. Clin Exp Med. 2014;14(4):389–395. doi: 10.1007/s10238-013-0259-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Takahashi T, Suzuki T, Hiroshige S, et al. Transient Appearance of Plasmablasts in the Peripheral Blood of Japanese Patients With Severe Fever With Thrombocytopenia Syndrome. J Infect Dis. 2019;5(1):23–27. doi: 10.1093/infdis/jiz054. [DOI] [PubMed] [Google Scholar]
- 25.Malleo G, Mazzon E, Siriwardena AK, Cuzzocrea S. Role of tumor necrosis factor-alpha in acute pancreatitis: from biological basis to clinical evidence. Shock. 2007;28(2):130–140. doi: 10.1097/shk.0b013e3180487ba1. [DOI] [PubMed] [Google Scholar]
- 26.Sah RP, Dawra RK, Saluja AK. New insights into the pathogenesis of pancreatitis. Curr Opin Gastroenterol. 2013;29(5):523–530. doi: 10.1097/MOG.0b013e328363e399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lu QB, Zhang SY, Cui N, et al. Common adverse events associated with ribavirin therapy for Severe Fever with Thrombocytopenia Syndrome. Antiviral Res. 2015;119:19–22. doi: 10.1016/j.antiviral.2015.04.006. [DOI] [PubMed] [Google Scholar]
- 28.Liu W, Lu QB, Cui N, et al. Case-fatality ratio and effectiveness of ribavirin therapy among hospitalized patients in China who had severe fever with thrombocytopenia syndrome. Clin Infect Dis. 2013;57(9):1292–1299. doi: 10.1093/cid/cit530. [DOI] [PubMed] [Google Scholar]
- 29.Graci JD, Cameron CE. Mechanisms of action of ribavirin against distinct viruses. Rev Med Virol. 2006;16(1):37–48. doi: 10.1002/rmv.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shimojima M, Fukushi S, Tani H, et al. Effects of ribavirin on severe fever with thrombocytopenia syndrome virus in vitro. Jpn J Infect Dis. 2014;67(6):423–427. doi: 10.7883/yoken.67.423. [DOI] [PubMed] [Google Scholar]
- 31.Lee MJ, Kim KH, Yi J, et al. In vitro antiviral activity of ribavirin against severe fever with thrombocytopenia syndrome virus. Korean J Intern Med. 2017;32(4):731–737. doi: 10.3904/kjim.2016.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hayden A, Park S, Giustini D, Lee AY, Chen LY. Hemophagocytic syndromes (HPSs) including hemophagocytic lymphohistiocytosis (HLH) in adults: a systematic scoping review. Blood Rev. 2016;30(6):411–420. doi: 10.1016/j.blre.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 33.Bae S, Hwang HJ, Kim MY, et al. Invasive Pulmonary Aspergillosis in Patients With Severe Fever With Thrombocytopenia Syndrome. Clin Infect Dis. 2020;17(7):1491–1494. doi: 10.1093/cid/ciz673. [DOI] [PubMed] [Google Scholar]
- 34.Winship AL, Koga K, Menkhorst E, et al. Interleukin-11 alters placentation and causes preeclampsia features in mice. Proc Natl Acad Sci USA. 2015;29(52):15928–15933. doi: 10.1073/pnas.1515076112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Erduran E, Bahadir A, Palanci N, Gedik Y. The treatment of crimean-congo hemorrhagic fever with high-dose methylprednisolone, intravenous immunoglobulin, and fresh frozen plasma. J Pediatr Hematol/Oncol. 2013;35(1):e19-24. doi: 10.1097/MPH.0b013e3182706444. [DOI] [PubMed] [Google Scholar]
- 36.Kim UJ, Kim DM, Ahn JH, et al. Successful treatment of rapidly progressing severe fever with thrombocytopenia syndrome with neurological complications using intravenous immunoglobulin and corticosteroid. Antiviral Therapy. 2016;21(7):637–640. doi: 10.3851/IMP3036. [DOI] [PubMed] [Google Scholar]
- 37.Yoo JR, Kim SH, Kim YR, Lee KH, Oh WS, Heo ST. Application of therapeutic plasma exchange in patients having severe fever with thrombocytopenia syndrome. Korean J Internal Med. 2019;34(4):902–909. doi: 10.3904/kjim.2016.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Park I, Kim HI, Kwon KT. Two Treatment Cases of Severe Fever and Thrombocytopenia Syndrome with Oral Ribavirin and Plasma Exchange. Infect Chemother. 2017;49(1):72–77. doi: 10.3947/ic.2017.49.1.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Oh WS, Yoo JR, Kwon KT, et al. Effect of Early Plasma Exchange on Survival in Patients with Severe Fever with Thrombocytopenia Syndrome: A Multicenter Study. Yonsei Med J. 2017;58(4):867–871. doi: 10.3349/ymj.2017.58.4.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due individual privacy of patients could be compromised, but are available from the corresponding author on reasonable request.