Abstract
Our prior work and the work of others have demonstrated that asthma increases the risk of a broad range of both respiratory (e.g., pneumonia and pertussis) and non-respiratory (e.g., zoster and appendicitis) infectious diseases as well as inflammatory diseases (e.g., celiac disease and myocardial infarction [MI]), suggesting the systemic disease nature of asthma and its impact beyond the airways. We call these conditions asthma-associated infectious and inflammatory multimorbidities (AIMs). At present, little is known about why some people with asthma are at high-risk of AIMs, and others are not, to the extent to which controlling asthma reduces the risk of AIMs and which specific therapies mitigate the risk of AIMs. These questions represent a significant knowledge gap in asthma research and unmet needs in asthma care, because there are no guidelines addressing the identification and management of AIMs. This is a systematic review on the association of asthma with the risk of AIMs and a case study to highlight that 1) AIMs are relatively under-recognized conditions, but pose major health threats to people with asthma; 2) AIMs provide insights into immunological and clinical features of asthma as a systemic inflammatory disease beyond a solely chronic airway disease; and 3) it is time to recognize AIMs as a distinctive asthma phenotype in order to advance asthma research and improve asthma care. An improved understanding of AIMs and their underlying mechanisms will bring valuable and new perspectives improving the practice, research, and public health related to asthma.
Keywords: Asthma, infection, inflammation, autoimmunity, disease, multimorbidity, comorbidity, risk, epidemiology
INTRODUCTION
Nearly 235 million people globally have asthma.1 Current evidence suggests that asthma poses a significant risk for a broad range of respiratory infections as summarized in Supplementary Table S1. In 2008, the Advisory Committee on Immunization Practices (ACIP) of the United States recognized asthma as an independent risk factor for invasive pneumococcal diseases (IPD) and recommended that a single dose of 23-valent pneumococcal polysaccharide vaccine (PPSV-23) be given to adults with asthma at ages 19–64 years.2 Similarly, the 2014 Canadian National Advisory Committee on Immunization recommends that adults with asthma receive a single dose PPSV-23.3
As summarized in Supplementary Table S1, the emerging literature suggests that the impact of asthma on susceptibility to infection goes beyond the airways as asthma has increased the risk of non-respiratory infections. Also, the observation on the association between asthma and risk of non-respiratory tract infection can be further extended to systemic inflammatory or autoimmune diseases, although the literature on the relationship has also been inconsistent. We term these conditions asthma-associated infectious and inflammatory multimorbidities (AIMs). These studies suggest that asthma may pose major (e.g., similar impact to diabetes mellitus),4,5 but relatively under-recognized health threats and also provide an insight into the nature of asthma as a systemic immunological disease beyond a solely chronic inflammatory airway disease.
Both the Guidelines on asthma by National Asthma Education and Prevention Program and the Global Initiative for Asthma have emphasized that the goals of asthma management are symptom control and risk reduction of exacerbations and known co-morbidities (e.g., allergic rhinitis or gastroesophageal reflux disease).6 However, the guidelines do not address how to assess and manage AIMs, although asthma is an independent risk factor for serious and common infections. Patients and their caregivers, as well as clinicians and researchers, are under-informed about the clinical impact of AIMs. Therefore, despite the major health threats to patients with asthma via AIMs, interventions to mitigate the risk of AIMs in asthma care are poorly understood. Better understanding of the nature of AIMs is an indispensable step toward the improvement of clinical practice and guidelines pertaining to AIMs to mitigate the risk and outcomes of AIMs in the future. Since the review on AIMs in 2014,7 the field of researching AIMs has made significant progress.
In this review, the authors discuss systematic review of the literature on asthma and its risk on AIMs. As an extension of this discussion and given the significantly limited literature on AIMs in children, we include the results for a recent population-based birth cohort study for AIMs in children as a case study. To further understand the systemic inflammatory nature of asthma, we extend the review to include both inflammatory and infectious diseases among people with asthma. The authors will discuss the potential mechanisms underlying AIMs and conclude this review with the implications of AIMs in research, clinical practice, and public health.
SYSTEMATIC REVIEW OF THE LITERATURE ON AIMs
To assess the current state of AIMs research, a comprehensive literature search of several databases from January 1, 1974 to February 24, 2021, English language, was conducted (Supplementary Data S1 for the detailed method for our systematic literature search and the summary of results of each study, Supplementary Table S1). A total of 47 papers on associations of asthma at risk of infections and inflammatory diseases were included in this systematic review by excluding abstracts, review papers, research protocols, unavailable full papers, and non-matching topics. Herein, we summarize the current state of AIMs research and discuss the key findings.
Asthma and the risk of respiratory infections
As summarized in Supplementary Table S1, asthma is associated with the increased risk of a broad range of respiratory infections including pneumococcal pneumonia, IPD, Streptococcus pyogenes upper respiratory infection (URI), recurrent or persistent otitis media, acute otitis media/myringitis, pertussis, viral URI, pneumonia, sinusitis, H1N1 influenza, and breakthrough varicella infection. These respiratory infections represent clinically both common and serious infections, microbiologically both bacterial (gram positive and gram negative) and viral infections, and anatomically both upper and lower airway infection/pneumonia. The most widely studied AIM is IPD, which led to the US and Canadian recommendation for PPSV-23 vaccine in adults with asthma at ages of 19–64 years, as discussed above.2,3 The association between asthma and the risk of IPD has been demonstrated in both adults and children, and the effect size and direction of the association were fairly consistent with a few exceptions. Some studies showed a dose-response relationship depending on the low-vs. high-risk asthma (severity).8,9 The effect sizes (risk ratios [RR] of from 1.4 to 19.3) for the associations between asthma and the risk of respiratory infections varied depending on the study design, study population as well as asthma and outcome definition and type of infections. At a population level, population attributable risk % (PAR%) can be assessed the basis of the reported data (e.g., 11%–17% for IPD8 or serious pneumococcal diseases [SPD, pneumococcal pneumonia and/or IPD10). For example, Juhn et al.10 reported that PAR% for asthma in adults was up to 17%, whereas the PAR% for all combined ACIP of the United States vaccine-eligible conditions in adults was 24%. These data suggest that asthma status alone increases the burden of SPD disproportionately at a population level. Apart from SPD and IPD, overall, the positive associations between asthma and the risk of respiratory infections were fairly consistent despite different respiratory infections.
Asthma and the risk of non-respiratory infections
Besides respiratory infections, previous studies reported the associations of asthma with the risk of non-respiratory infections, including herpes zoster, community-acquired Escherichia coli bloodstream infection, sepsis, urinary tract infection, appendicitis, skin infection, gingivostomatitis, and gastroenteritis with effect sizes of hazard ratios (HR) of 1.15 to odds ratio (OR) of 2.74. These infections involve the gastrointestinal (GI) tract, genitourinary (GU) tract, skin and nerve cells (ganglion) indicating the impact of asthma on the risk of non-respiratory infections. Similar to the GI tract, the GU tract and skin share a similar structural feature in terms of epithelial cells connected by tight junction, the associations between asthma and the risk of infections involving these systems may share a similar mechanism, such as ‘the epithelial barrier defect hypothesis,’11 discussed in a later section. One noteworthy observation (which may not be subject to the ‘epithelial barrier defect hypothesis’) is the association between asthma and the risk of herpes zoster that was first reported in asthmatic children by Kim et al.12 in 2013, and the association has been replicated in adults by our group and other groups.13,14,15 The recent meta-analysis corroborated the association, and the risk of herpes zoster was similar between patients with asthma and diabetes mellitus (RR, 1.25 vs. 1.30).5 Immunosenescence in cell-mediated immunity (CMI) is the currently proposed mechanism of the increased risk of herpes zoster infection as almost all (99.2%–99.6%) adults aged ≥ 40 years16,17 and 86%–93% of children aged 6–18 years (during the pre-varicella vaccine era) in the US have serologic evidence for previous varicella zoster virus infection.16 Along the lines with the observation on the increased risk of breakthrough varicella infection among asthmatic children,18 these observations imply a subgroup of asthmatic patients may have impaired adaptive immune function apart from the widely recognized impairment in innate immune function discussed in a later section. A recent Danish study reported that the impact of asthma on the risk of any infection (both respiratory and non-respiratory infections) they studied as measured by PAR% was similar to that of diabetes mellitus (2.2% vs. 2.6%).4 Therefore, the literature on the association between asthma and the risk of non-respiratory infections suggests that asthma may pose major, (similar impact to diabetes mellitus) albeit relatively under-recognized, health threats and also provide an important insight into the nature of asthma as a systemic immunological disease beyond a mere chronic inflammatory airway disease.
Asthma and the risk of inflammatory diseases
The literature suggests that asthma is associated with a broad range of inflammatory and autoimmune diseases including celiac disease, rheumatoid arthritis, diabetes mellitus, MI or coronary heart disease, systemic lupus erythematosus (SLE), Sjogren's syndrome, myasthenia gravis, psoriasis, pernicious anemia, inflammatory bowel disease (IBD), vitiligo, Addison disease, amyotrophic lateral sclerosis, chronic rheumatoid heart disease, Graves, immune thrombocytopenic purpura, polyarteritis nodosa, polymyalgia rheumatica, Sarcoidosis, systemic sclerosis, Wegener granulomatosis, and autoimmune thyroiditis. The most recent work on the association between asthma and the risk of autoimmune diseases is a retrospective cohort study by Krishna et al.19 that leveraged a UK primary care database and assessed the incidence of autoimmune diseases between asthmatic patients (n = 1,049,868) and matched non-asthmatic/allergic patients (n = 1,732,480) based on a longitudinal observation between 1990 and 2018. They found asthmatic patients had significantly increased risks of SLE, Sjögren's syndrome, vitiligo, rheumatoid arthritis, psoriasis, pernicious anemia, myasthenia gravis, IBD, celiac disease, and autoimmune thyroiditis with effect sizes ranging from adjusted RR of 1.23 to 1.61, but multiple sclerosis was not associated with asthma. As summarized in Supplementary Table S1, many autoimmune or inflammatory conditions were replicated by 2 or more independent studies. These observations on the association of asthma with increased risks of autoimmune diseases or inflammatory diseases pose challenges to the interpretation of results in both conceptual and methodological aspects. For example, unlike infections, the causal relationship between 2 chronic diseases (e.g., temporality) is difficult to discern. Also, it is difficult to conceptualize how an asthma-associated immune suppressive state as shown in the associations of asthma with the increased risks of both respiratory and non-respiratory infections and their underlying immune impairment (see the section for the mechanism) can coexist or predispose to a hyper-inflammatory state leading to autoimmune or inflammatory diseases. Overall, the literature suggests that asthma is associated with the risks of autoimmune or inflammatory diseases.
Summary of the systematic review
The current literature suggests that asthma is associated with increased risks of a broad range of respiratory and non-respiratory infections as well as autoimmune/inflammatory diseases, namely AIMs. Overall, asthma may pose major health threats to people with asthma via AIMs. Specifically, the literature suggests that the magnitude of the impact of asthma on the risk of any infection may be similar to that for diabetes mellitus as described above. In addition, the literature provides an important insight into the systematic inflammatory or immunological nature of asthma beyond a mere chronic airway disease. The current literature is mostly based on retrospective studies which relied on structured codes for the ascertainment of asthma and outcomes of interest, and lacks data derived from prospective studies with immunological measures (endotyping). While it provides epidemiological evidence for the association, it is significantly limited in the characterization of asthma and outcomes in a way identifying which subgroups of asthmatics are at risk of AIMs, what mechanisms underlie the risk, and outcome of AIMs, and what factors mitigate the risk and outcome of AIMs. A population-based pediatric study can be valuable as it is less confounded by comorbidities with aging in adult populations, but currently the literature based on a population-based pediatric study is quite limited. Better understanding of the nature of AIMs is an indispensable step toward the improvement in clinical practice and guidelines pertaining to AIMs to mitigate the risk and outcomes of AIMs in the future.
A POPULATION-BASED STUDY OF AIMs IN CHILDREN: A CASE STUDY
The literature on a population-based pediatric study addressing the risk of AIMs using well-defined pediatric populations with asthma is scant, especially, birth cohort studies. We conducted a population-based retrospective birth cohort study in Olmsted County, Minnesota.
Study design and methods
This study was approved by the Mayo Clinic Institutional Review Board (IRB No. 14-009934). The study was designed as a retrospective birth cohort study which followed the 1997-2016 Mayo Clinic Birth Cohort born in Olmsted County, Minnesota. The study design and methods are described in detail in Supplementary Data S2.
Results
The characteristics of the study subjects are summarized in Table 1. Of the eligible children (n = 21,827), 3,116 (14.3%) had asthma, which is slightly higher than the previously reported prevalence (12.9%) of asthma diagnosis in a pediatric population in our community.20 We attempted to match each asthmatic subject to 3 children who had no history of asthma. Those children with asthma who were not able to be matched were excluded. However, if 1 or 2 matched controls were available, we included these sets. With exact matching with birth year, sex, and follow-up duration (within 2 years), all asthmatic patients except 76 were matched. Among the asthmatic subjects, 1,803 (59%) were males and 2,298 (76%) were white. The mean ages at the index date of asthma and the last follow-up date were 4.0 years and 11.0 years, respectively. We identified a total of 56 infectious and inflammatory conditions from electronic health records (EHRs) of all children with asthma (Supplementary Data S2 for details in the procedures for the identification of these conditions), and 23 conditions were clinical diagnostic codes which met 0.5% prevalence threshold and were included in the analysis.
Table 1. Baseline characteristics for children with asthma and their matched non-asthmatic children with regard to birth year, sex, and last follow-up date (i.e., similar follow-up duration).
| Characteristics | No asthma (n = 6,295) | Asthma (n = 3,040) | Total (n = 9,335) | P value | |
|---|---|---|---|---|---|
| Age at index | < 0.001 | ||||
| Mean ± SD | 3.72 ± 3.85 | 4.01 ± 3.89 | 3.82 ± 3.87 | ||
| Median (Q1, Q3) | 2.13 (0.93, 5.36) | 2.41 (1.13, 5.73) | 2.22 (1.01, 5.47) | ||
| Range | 0.08–20.14 | 0.03–20.56 | 0.03–20.56 | ||
| Male (sex) | 3,702 (58.8) | 1,803 (59.3) | 5,505 (59.0) | 0.645 | |
| Race | < 0.001 | ||||
| Black | 379 (6.0) | 301 (9.9) | 680 (7.3) | ||
| Asian | 338 (5.4) | 153 (5.0) | 491 (5.3) | ||
| Hawaiian/PacIsl | 15 (0.2) | 5 (0.2) | 20 (0.2) | ||
| Am. Indian | 12 (0.2) | 5 (0.2) | 17 (0.2) | ||
| Other/mixed | 402 (6.4) | 210 (6.9) | 612 (6.6) | ||
| White | 4,935 (78.4) | 2,298 (75.6) | 7,233 (77.5) | ||
| Refusal | 50 (0.8) | 20 (0.7) | 70 (0.7) | ||
| Unknown | 164 (2.6) | 48 (1.6) | 212 (2.3) | ||
| Ethnicity | 0.011 | ||||
| Non-hispanic | 5,686 (90.3) | 2,790 (91.8) | 8,476 (90.8) | ||
| Hispanic | 418 (6.6) | 189 (6.2) | 607 (6.5) | ||
| Unknown | 191 (3.0) | 61 (2.0) | 252 (2.7) | ||
| White | 4,935 (78.4) | 2,298 (75.6) | 7,233 (77.5) | 0.002 | |
| HOUSES Q at birth | 0.015 | ||||
| N-miss | 299 | 121 | 420 | ||
| 1 | 1,345 (22.4) | 743 (25.5) | 2,088 (23.4) | ||
| 2 | 1,508 (25.2) | 696 (23.8) | 2,204 (24.7) | ||
| 3 | 1,636 (27.3) | 756 (25.9) | 2,392 (26.8) | ||
| 4 | 1,507 (25.1) | 724 (24.8) | 2,231 (25.0) | ||
| Average number of clinic visits per year | < 0.001 | ||||
| Mean ± SD | 6.48 ± 4.59 | 8.60 ± 5.97 | 7.17 ± 5.18 | ||
| Median (Q1, Q3) | 5.33 (3.83, 7.57) | 7.18 (5.20, 10.20) | 5.87 (4.13, 8.45) | ||
| Range | 0.65–81.17 | 0.49–81.06 | 0.49–81.17 | ||
Data are shown as mean ± SD or number (%).
SD, standard deviation; Q, quartile; HOUsing-based Index of SocioEconomic Status.
The risk of AIMs in relation to asthma status in children
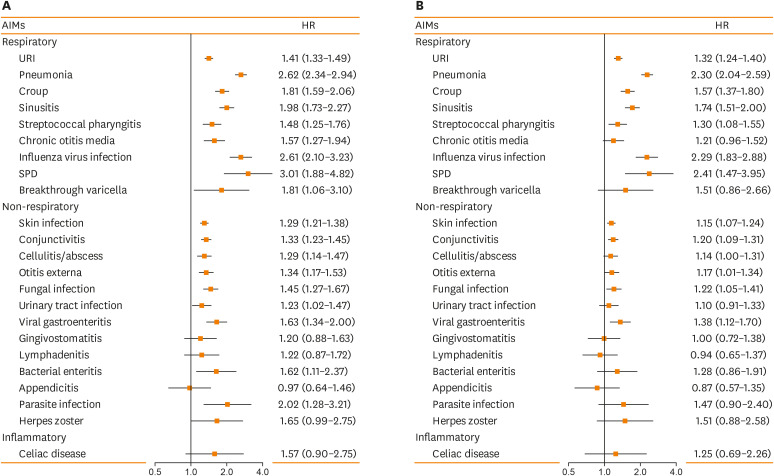
The main results on the association between asthma and the subsequent risk of AIMs are summarized in Figure using Forest plot (1A for unadjusted analysis and 1B for adjusted analysis). Based on univariate analysis, asthma was associated with 18 of 23 conditions examined. After adjusting for race, individual level socioeconomic status defined by HOUsing-based Index of SocioEconomic Status (HOUSES) index (a validated SES measure),21,22,23 and the average number of clinic visits as well as matching age, sex, and follow-up duration, asthma was associated with 13 of 23 conditions which include both respiratory infections (URI, pneumonia, croup, sinusitis, streptococcal pharyngitis, influenza virus infection, and SPDs) and non-respiratory infections (skin infection, conjunctivitis, cellulitis/abscess, otitis externa, fungal infection, and viral gastroenteritis). The effect sizes in adjusted HRs for those 13 conditions ranged from 1.14 (cellulitis/abscess) to 2.41 (SPD). Asthma was not associated with the risk of inflammatory diseases such as celiac disease, in part due to low incidence of inflammatory disease in this birth cohort. While these results largely corroborate previous reports on AIMs (IPD, Streptococcal pharyngitis, URI, pneumonia, influenza, skin infection, viral gastroenteritis, herpes zoster and SPDs), they also revealed potentially new infectious conditions associated with asthma (cellulitis/abscess, otitis externa, and fungal infection).
Figure. The risk of AIMs in relation to asthma status in children and (A) depicts unadjusted analysis and (B) for adjusted analysis. (A) Unadjusted Cox model for the association between asthma and the risk of AIMs. (B) Adjusted Cox model for the association between asthma and the risk of AIMs controlling race (white vs. non-white), HOUSES (socioeconomic status) at birth, and average number of clinic visits.
AIMs, asthma-associated infectious and inflammatory multimorbidities; HR, hazard ratio; URI, upper respiratory infection; SPD, serious pneumococcal diseases; HOUSES, HOUsing-based Index of SocioEconomic Status.
The associations of asthma and other atopic conditions at risk of AIMs
In order to assess the effects of other allergic diseases on the association between asthma and the risk of AIMs, we performed an additional analysis examining the interaction of asthma status with other allergic diseases. We divided the subjects into 4 groups (1. asthma only, 2. other atopic conditions only, 3. both asthma and other atopic conditions, and 4. neither asthma nor other atopic conditions as the reference group). To investigate the interaction of asthma and other atopic conditions, atopy was considered a time-dependent covariate (after the asthma index date). The adjustment and matching for the analysis were the same as above. The results are summarized in Table 2. Overall, we did not observe significant interactions between asthma and other atopic conditions in relation to the risk of AIMs except a few conditions with statistically significant interactions. Of the 13 AIMs significantly associated with asthma described above, 6 (URI, skin infection, pneumonia, cellulitis/abscess, sinusitis, and influenza) were associated with both asthma only and asthma AND other atopic conditions whereas 5 (conjunctivitis, croup, streptococcal pharyngitis, viral gastroenteritis, SPD, and chronic otitis media) were associated with asthma only and 2 (otitis externa and herpes zoster) were associated with asthma AND other atopic conditions. It is noteworthy that the risk of herpes zoster was significantly elevated among children with both asthma and other atopic conditions, compared to either asthma or other atopic condition alone.
Table 2. The associations of asthma and other atopic conditions with the risk of AIMs based on Cox proportional hazard models matched birth year, sex and last follow-up with adjusted with race (white vs. non-white), HOUSES, average number of clinic visits.
| Type of AIMs | Clinical conditions | No asthma or atopy | Other atopic conditions only | Asthma only | Asthma and other atopic conditions | P value* |
|---|---|---|---|---|---|---|
| Respiratory infection | Upper respiratory tract infection | 1 (reference) | 1.04 (0.94–1.15) P = 0.452 | 1.33 (1.24–1.42) P ≤ 0.001 | 1.32 (1.2–1.46) P ≤ 0.001 | 0.578 |
| Pneumonia | 1 (reference) | 1.01 (0.81–1.27) P = 0.920 | 2.49 (2.18–2.85) P ≤ 0.001 | 1.93 (1.62–2.31) P ≤ 0.001 | 0.068 | |
| Croup | 1 (reference) | 0.79 (0.6–1.04) P =0.098 | 1.72 (1.48–2) P ≤ 0.001 | 1.06 (0.84–1.33) P = 0.650 | 0.168 | |
| Sinusitis | 1 (reference) | 1.53 (1.23–1.91) P ≤ 0.001 | 1.90 (1.61–2.24) P ≤ 0.001 | 1.92 (1.57–2.35) P ≤ 0.001 | 0.007 | |
| Streptococcal pharyngitis | 1 (reference) | 1.07 (0.8–1.43) P = 0.664 | 1.35 (1.09–1.67) P = 0.006 | 1.24 (0.95–1.63) P = 0.109 | 0.483 | |
| Chronic otitis media | 1 (reference) | 1.08 (0.72–1.6) P = 0.717 | 1.41 (1.1–1.81) P = 0.006 | 0.77 (0.51–1.15) P = 0.197 | 0.017 | |
| Influenza virus infection | 1 (reference) | 0.95 (0.62–1.47) P = 0.818 | 2.15 (1.65–2.81) P ≤ 0.001 | 2.48 (1.84–3.36) P ≤ 0.001 | 0.470 | |
| Serious pneumococcal disease | 1 (reference) | 1.06 (0.4–2.79) P = 0.908 | 2.69 (1.54–4.68) P ≤ 0.001 | 1.93 (0.93–4) P = 0.078 | 0.519 | |
| Breakthrough varicella | 1 (reference) | 0.87 (0.3–2.52) P = 0.797 | 1.48 (0.78–2.82) P = 0.232 | 1.48 (0.65–3.39) P = 0.353 | 0.840 | |
| Non-respiratory infection | Skin infection | 1 (reference) | 1.27 (1.14–1.41) P ≤ 0.001 | 1.20 (1.11–1.31) P ≤ 0.001 | 1.2 (1.08–1.34) P ≤ 0.001 | 0.003 |
| Conjunctivitis | 1 (reference) | 0.96 (0.82–1.11) P = 0.547 | 1.25 (1.13–1.38) P ≤ 0.001 | 1.05 (0.91–1.21) P = 0.485 | 0.227 | |
| Cellulitis/abscess | 1 (reference) | 1.42 (1.18–1.73) P ≤ 0.001 | 1.19 (1.01–1.4) P = 0.033 | 1.3 (1.07–1.58) P = 0.008 | 0.062 | |
| Otitis externa | 1 (reference) | 1.19 (0.97–1.47) P = 0.098 | 1.16 (0.98–1.38) P = 0.080 | 1.29 (1.06–1.58) P = 0.012 | 0.626 | |
| Fungal infection | 1 (reference) | 0.88 (0.69–1.13) P = 0.313 | 1.17 (0.99–1.39) P = 0.065 | 1.23 (1–1.52) P = 0.053 | 0.297 | |
| Urinary tract infection | 1 (reference) | 0.91 (0.67–1.23) P = 0.541 | 1.07 (0.86–1.34) P = 0.546 | 1.09 (0.83–1.44) P = 0.532 | 0.599 | |
| Viral gastroenteritis | 1 (reference) | 0.89 (0.61–1.29) P = 0.531 | 1.48 (1.17–1.88) P = 0.001 | 1.1 (0.79–1.52) P = 0.588 | 0.483 | |
| Gingivostomatitis | 1 (reference) | 1.45 (0.93–2.26) P = 0.105 | 1.01 (0.68–1.5) P = 0.951 | 1.22 (0.77–1.94) P = 0.393 | 0.598 | |
| Lymphadenitis | 1 (reference) | 0.61 (0.31–1.18) P = 0.138 | 0.96 (0.63–1.45) P = 0.837 | 0.72 (0.4–1.29) P = 0.266 | 0.640 | |
| Bacterial enteritis | 1 (reference) | 1.2 (0.63–2.27) P = 0.581 | 1.42 (0.9–2.25) P = 0.135 | 1.15 (0.63–2.1) P = 0.656 | 0.382 | |
| Appendicitis | 1 (reference) | 1.11 (0.62–1.97) P = 0.734 | 0.65 (0.36–1.17) P = 0.152 | 1.29 (0.74–2.24) P = 0.372 | 0.199 | |
| Parasite infection | 1 (reference) | 0.96 (0.4–2.32) P = 0.928 | 1.66 (0.96–2.86) P = 0.070 | 1.09 (0.52–2.31) P = 0.821 | 0.516 | |
| Herpes zoster | 1 (reference) | 0.9 (0.35–2.36) P = 0.836 | 1.07 (0.54–2.14) P = 0.844 | 2.3 (1.17–4.53) P = 0.016 | 0.167 | |
| Inflammatory disease | Celiac disease | 1 (reference) | 1.39 (0.59–3.31) P = 0.452 | 1.65 (0.83–3.29) P = 0.150 | 0.9 (0.35–2.31) P = 0.823 | 0.149 |
Bolded are conditions with statistical significance.
AIMs, asthma-associated infectious and inflammatory multimorbidities; HOUSES, HOUsing-based Index of SocioEconomic Status.
*P value for interaction between asthma and atopic conditions.
Summary of the results
Overall, the results of this population-based birth cohort study support previously reported associations of asthma with a broad range of respiratory and non-respiratory infections among children. As analysis was adjusted for important confounding factors (e.g., individual level SES, health care access [number of clinic visits], and follow-up duration), the findings may not be attributed to detection bias or transfer bias. The associations of asthma with the risk of infections did not seem to be significantly modified by the presence of other atopic conditions. However, the analysis was challenged due to the limited statistical power to assess the association of asthma with conditions with low incidence such as celiac disease and appendicitis. Our study did not address an important study question on the influence of asthma control or severity status as well as therapies on the risk of AIMs which might be another important confounder.
POTENTIAL FACTORS AND MECHANISMS FOR THE ASSOCIATION BETWEEN ASTHMA AND THE RISK OF AIMs
Influence of inhaled corticosteroid (ICS) therapies on the risk of AIMs
The preponderance of studies has been on the effect of ICSs on the risk of pneumonia among patients with asthma, and the literature is inconsistent.24,25,26,27,28,29,30,31,32,33,34 For example, some studies reported no association of ICSs with the risk of pneumonia26,28,31 or even protective effect,28,29,33 while other studies (all retrospective studies) showed associations of ICS, use with the increased risk of pneumonia.24,25,27 A recent meta-analysis reported that the use of ICS was protective of pneumonia with an RR of 0.74 (95% confidence interval [CI], 0.57–0.95), based on clinical trials, while retrospective observational studies showed summed OR of 1.97 (95% CI, 1.87–2.07), suggesting an increased risk of pneumonia with use of ICSs in asthmatic patients.29 A cohort study, which compiled clinical trial data (n = 14,993), showed ICS use was not associated with the risk of hospitalized pneumonia (HR,1.30; 95% CI, 0.53–3.14) or reduced the risk of non-hospitalized pneumonia (HR, 0.51; 95% CI, 0.35–0.74) compared to placebo pills (short-acting beta-agonists), independent of ICS dose, type, and duration.28 Also, an earlier randomized clinical trial (RCT) showed the safety of ICSs on the risk of infection by comparing ICS treatment versus placebo treatment.35 The results, indeed, showed a significant decrease from before to after therapy in the percentage of days of upper respiratory tract infection (21%–10% ICS vs. 19%–16% placebo) and lower respiratory tract infection (30%–15% ICS vs. 27%–21% placebo).35 Another study in patients with asthma gave an estimated OR of 0.34 for the association between treatment with corticosteroids and the risk of mycoplasma or chlamydia infections (P = 0.07), which suggests that corticosteroid treatment may have a protective effect for the risk of these infections.36 However, case-control studies by others showed that ICSs increased the risk of pneumonia.25,27 For example, McKeever et al.25 reported an increased risk of pneumonia among ICSs users (OR, 1.58; 95% CI, 1.29–1.93) but only limited to fluticasone, not other ICS and the same group failed to show the same association in their recent clinical trial.26 This is suggestive of inadequate adjustment for confounders such as ICS-related asthma control status and severity, which might account for their discrepant study results. As discussed below, since poorly controlled asthma or type 2 inflammation (Th2) may impair immune functions, asthma control and severity status are likely to be important confounders to be adjusted for the analysis. In this respect, large retrospective studies derived from administrative database, which could not adequately address asthma control or severity status, tend to find positive associations, while prospective studies or RCT showed no associations or protective effect of ICS. Given the significant limitations of retrospective observational studies, based on the results derived from compiled clinical trials, we believe that ICS use is not associated with the risk of pneumonia or even protective effect in a subgroup of asthmatics. Therefore, ICS use is unlikely to account for the impact of asthma status on the risk of infectious diseases.
Asthma control status and prognosis in the long-term risk of AIMs
As discussed above, at present, little is known about whether and the extent to which adequate asthma control and severity status impacts the risk of AIMs because the risk of AIMs is not considered a typical outcome measure for asthma study.37 O'Byrne et al.28 reported the reduced risk of non-hospitalized pneumonia (HR, 0.51; 95% CI, 0.35–0.74) compared to a placebo. In their study, among baseline risk factors, patients with impaired lung function (FEV1) below 80% had an inverse association with the risk of pneumonia, suggesting poorly controlled asthma might pose an increased risk of pneumonia. Talbot et al.8 reported that high-risk asthma (i.e., poorly controlled asthma defined by frequent hospitalization, ED visit, and oral corticosteroids) posed a greater risk of IPD (OR, 2.6; 95% CI, 2.0–3.5) than low-risk asthma (i.e., not well-controlled asthma) (OR, 1.7; 95% CI, 0.99–3.0), and this observation was supported by another study.9 As a potential mechanism, while impairment of rhinovirus-induced type I and III interferon secretion and its subsequent increased replication of rhinovirus have been widely recognized,38 such phenomena were not observed in well-controlled asthma,39 but those with severe therapy-resistant atopic asthma.40 At present, the literature on the relationship between asthma control or severity and the risk of AIMs, especially risk of infections and its underlying mechanisms is significantly limited. Both epidemiological and mechanistic studies addressing these study questions are further needed.
Potential mechanisms underlying the risk of AIMs
Previous review articles covered the topic on the potential mechanisms underlying the risk of infections: impairment of both innate and adaptive immunity in people with asthma by potentially Th2 high inflammation (cytokines) associated with asthma is the currently proposed mechanism for the association between asthma and the risk of infections, primarily respiratory infections.7,38 Therefore, in this review, we discuss new information on this topic not covered by previous reviews and explore the mechanisms underlying the associations of asthma with the risk of non-respiratory infections and inflammatory diseases.
The heterogeneity of asthma has been widely recognized as a spectrum disorder.41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66 For example, numerous cluster analyses have been reported to characterize children with asthma,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64 but none of the studies have assessed AIMs as a distinctive phenotype of asthma in defining the heterogeneity of asthma. For example, neither current guidelines nor expert opinions recognize AIMs as an outcome of interest in asthma studies.37 Thus, the current literature is limited in identifying a subgroup of people with asthma who are at high-risk of AIMs. We believe that only a subgroup of asthmatic patients are likely to be at risk of AIMs, not necessarily all asthmatic patients, and that the heterogeneity of asthma is likely to be an important factor for understanding the mechanisms underlying the risk of AIMs. For example, as summarized in Supplementary Table S1, while previous studies reported that asthma was associated with the risk of non-respiratory infections and inflammatory diseases involving GI tract such as appendicitis67,68 and celiac disease,69 our group showed that only a subgroup of children with active asthma (i.e., evidence of asthma in the past 12 months) are at increased risk of appendicitis70 and celiac disease,71 suggesting the sub-phenotype of asthma plays an important role in the mechanisms for AIMs. Specifically, how can we explain a shared mechanism underlying the association of asthma with the risk of distinctively different diseases, such as respiratory infection, non-respiratory infections, and autoinflammatory diseases? A few plausible mechanisms can be contemplated.
There is emerging literature on ‘epithelial barrier defect hypothesis’ as a potential mechanism for the association of asthma with a broad range of infections and autoimmune diseases.11,72 For details of the hypothesis and its immunological pathways, we refer the readers to the recent review.11 Conceptually, alteration in environmental exposure over time might have disrupted the physical integrity of the barrier by degrading the intercellular barrier proteins at tight junctions, triggering epithelial alarmin cytokine responses such as interleukin (IL)-25, IL-33, and thymic stromal lymphopoietin, and increasing the epithelial barrier permeability (“leaky epithelium”) which may lead to infection or activation of host immune responses resulting in a broad range of Th2 and Th1 inflammatory diseases including asthma, chronic rhinosinusitis, allergic rhinitis, atopic dermatitis, eosinophilic esophagitis, and celiac and IBD.11 Therefore, under this hypothesis, various up-stream and down-stream immunological pathways underlying chronic airway and systemic inflammation of asthma might also be a shared mechanism for various AIMs even if they are distinctively different respiratory and non-respiratory infections as well as autoinflammatory diseases (e.g., appendicitis, eosinophilic esophagitis, celiac disease and other autoimmune diseases, and MI). However, it is noteworthy that for asthmatic patients with inflammatory or autoimmune disease features or recurrent febrile illnesses, one condition to be considered as a differential diagnosis is autoinflammatory disease, a disease of innate immune system, although it is infrequent.73
Alternatively, a recent genome-wide association study (GWAS) identified a gene with pleiotropic effects on asthma and obesity.74 Along these lines, although it is inconsistent,75,76,77 several GWAS showed potential pleiotropic effects of genes (either risk or protective effect) underlying the association of asthma with the risk of multiple autoimmune diseases.75,76 It needs to be elucidated how the pleiotropic effect of a specific gene can account for a broad range of AIMs including respiratory, non-respiratory, and inflammatory/autoimmune diseases.
Various immunological effects derived from the up- and down-stream immunological pathways for asthma as a result of genetic and environmental factors might determine the heterogeneity of asthma which subsequently affects the risk of AIMs depending on the nature of AIMs. Conceptually, the interplay among asthma, infection, and inflammatory diseases is complex,78 and at present, little is known about the unified mechanisms accounting for the risk of a broad range of clinically distinctive infectious and inflammatory diseases in addition to the mechanism specific to each infectious and inflammatory disease as described above. Also, little is known about how innate and adaptive immune functions are affected in a way increasing certain non-respiratory infections. Adaptive immune dysfunction will be an important mechanism underlying certain non-respiratory AIMs such as herpes zoster.12,13 Many research groups, including ours, are actively researching this under-studied area.
IMPLICATIONS
Clinical practice
Since the last review on AIMs in 2014,7 there has been burgeoning literature pertaining to AIMs. A few implications in clinical practice can be considered. First, given the significant proportion of children and adults affected by asthma and their increased risk of AIMs, clinicians might need to be cognizant of the presence of AIMs and consider more careful evaluation of those with serious or common infections to discern whether a patient has undetected clinical asthma or other atopic conditions, instead of solely seeking primary immunodeficiency, especially when a patient had multiple AIMs. Also, they will need to educate their patients and their caregivers about the presence, nature, and implications of AIMs in asthma management because serious threats to health from AIMs in children and adults with asthma are largely under-recognized by patients and their caregivers as well as clinicians. Secondly, as discussed in the Introduction section, no guidelines currently address the identification and management of AIMs. However, clinicians and caregivers of patients with asthma often run into situations where they need some guidance in assessing and managing AIMs. Thus, using current evidence, examples of potential strategies for AIMs in asthma care are summarized in Table 3 and have been provided in asthma practice of authors' institution (AskMayoExpert). These strategies can be further developed and revised as understanding about AIMs advances. Thirdly, while it may not be feasible to fully unravel the mechanisms underlying AIMs in the near future, some research approaches can be directly beneficial and relevant to asthma management as discussed in the next section.
Table 3. Potential management strategies for a high-risk group of asthmatics for AIMs7,123 .
| Clinical scenarios in asthma csymptomare | Current management/potential unmet needs | Potential management strategies and rationales for AIMs |
|---|---|---|
| Uncontrolled cough | Step-up asthma medication or oral corticosteroid/missing or delaying identification of pertussis permits continuing transmission of pertussis124 | -Check for pertussis as pertussis is an AIMs |
| -Check serologic titer for DPT and boost dose if immunity is waned | ||
| Serious or frequent common infections | Assess PID/patient receives no counseling for the role of asthma in accounting for such infections | -If no PID is present, discern whether a patient has undetected clinical asthma among undiagnosed asthmatics |
| -Counsel about the risk of infections for asthmatics and develop a management plan for asthma after diagnosis | ||
| Concerns about risk of pneumonia from ICS | May not adequately address concern/potential under-treatment of ICS or poor control of asthma | -Use ICS or other controller medications per asthma guidelines |
| -No evidence to suggest that ICS poses an increased risk of pneumonia in asthmatics | ||
| -Better control of asthma may reduce the risk of pneumonia | ||
| Declining routine vaccinations | Often patients choose opt out option/increased risk of vaccine-preventable AIMs | -Inform patients of their increased susceptibility to vaccine-preventable AIMs and the role of vaccinations |
| Current vaccinations | While addressing the need for influenza vaccination, they may not properly address the patient's unique needs or susceptibility to other vaccine preventable AIMs among asthmatics | -PPSV23 regardless of asthma severity or control status |
| -Check vaccine titer to determine if vaccine preventable disease or frequent infections may occur | ||
| -Recommend zoster vaccine for asthmatics age 50 or older | ||
| -Consider replacing Td with Tdap for asthmatics | ||
| Identification of serious AIMs (appendicitis, Celiac disease) | Relying on traditional criteria, risk factor/potential delay of identification and therapy in affected asthmatic patients | -Define asthma as an unrecognized new risk factor for AIMs |
| -This allows timely identification (e.g., population surveillance for celiac symptoms and targeted testing for a high-risk group for celiac disease) and therapy (e.g., diet for celiac disease) | ||
| Prediction for uncontrolled asthma | Relying on traditional risk factors or biomarkers for reactive approach after exacerbation vs. proactive | -Predictive analytics to predict a future risk of asthma exacerbation or poorly controlled asthma125,126 as a risk factor for AIMs103 |
AIMs, asthma-associated infectious and inflammatory multimorbidities; DPT, diphtheria, pertussis, and tetanus; PID, primary immunodeficiency; ICS, inhaled corticosteroid; PPSV23, 23-valent pneumococcal polysaccharide vaccine; Td, tetanus and diphtheria; Tdap, tetanus, diphtheria, and acellular pertussis.
Research
In researching asthma in terms of phenotypes, it might be timely to consider the presence of AIMs as a distinctive asthma phenotype to be considered in asthma studies which typically limit to asthma control or exacerbation, quality of life, and impaired lung function. Currently, guidelines and consensus report do not address AIMs as a distinctive clinical and immunological phenotype in asthma.37,79 How we define the phenotype of asthma in research (“moving target”) leads us in very different directions and frequently to inconsistent results of the literature which is a major reason for deterring translation of research findings into clinical practice. Specifically, as discussed above, the effects of asthma or its associated inflammatory responses may not be limited to the airways, but rather may be systemic as asthma may not be a mere chronic airway disease (a traditional and current paradigm for understanding asthma). As whether we define asthma as a mere airway disease vs. a systemic inflammatory disease may impact both conceptual and methodological directions in future asthma research, this review prompts the research community to recognize under-recognized health threats to people with asthma via AIMs as an asthma phenotype and to reframe asthma as a systemic inflammatory and immunological disease beyond an airway disease.
In this respect, research effort for improving phenotyping asthma via innovative approaches will be crucial. Specifically, reducing methodological heterogeneity in phenotyping asthma (e.g., 60 different definitions of asthma in the literature)80 is crucially important as it often obscures true biological heterogeneity and results in inconsistent results deterring translation of scientific findings into practice. Inconsistent results have been reported in GWAS,81,82,83 clinical trials,84,85 studies addressing heterogeneity of asthma41,46,,86,87 and studies concerning the coronavirus disease 2019 (COVID-19) pandemic.88,89,90,91,92,93,94,95,96,97,98,99,100,101,102 Some sources of variability in the above studies include inconsistent asthma criteria, ascertainment processes, and sampling frames which may obscure a better understanding of heterogeneity of asthma. This inconsistency may be responsible for the heterogeneity of study results in a greater degree than the true phenotypic heterogeneity of asthma itself. In this respect, discerning the complex interactions among clinical asthma, their underlying biological pathways, and the nature of AIMs as an asthma phenotype requires new approaches mining large-scale EHRs such as artificial intelligence (AI)-augmented approaches in data curation and analysis beyond traditional approaches (e.g., self-report or manual review and statistical analysis). For example, our group recently demonstrated AI-augmented phenotyping of asthma status by applying 2 existing asthma criteria successfully identified a subgroup of asthmatic children with distinctive immunological and clinical characteristics including Th2 immune response, poor asthma outcomes, and the risk of various AIMs.103,104,105 AI-augmented phenotyping approaches can address many limitations of traditional approaches in leveraging longitudinal EHRs such as scalability, precision, accuracy, and efficiency. We refer the readers to the recent review paper on this topic, AI approaches for advancing EHR-based clinical research in allergy, asthma, and immunology.105
While research community innovates approaches for asthma phenotyping and study the mechanistic pathways underlying AIMs, it is crucially important to identify and characterize a subgroup of asthmatic patients at risk of AIMs because identification of this subgroup of asthmatic patients is an indispensable step to develop strategies mitigating the risk and outcomes of AIMs in the future and impacts future guidelines for optimal asthma management for AIMs. The basic questions, why some asthmatics are at high-risk of AIMs, and others are not, specifically, what is the impact of asthma control and asthma therapies on the risk of AIMs, need to be answered to improve asthma care. Successful identification of a subgroup of asthmatic patients at risk of COVID-19 or AIMs will ultimately help us unravel the mechanisms underlying the risk of AIMs and develop strategies mitigating the risk and outcomes of AIMs in asthma management.
Finally, this review paper focused on non-respiratory infection and inflammatory diseases as AIMs. However, little is known about what constitutes AIMs beyond conditions mentioned above. A recent report from Korea suggests that the incidence of cancer increased following the diagnosis of asthma among adults participating in a Korean population-based cohort using national administrative data.106 They reported that adults with asthma had a 75% greater risk of incident cancer overall (although the effects vary for different cancers). The excess risk for incident cancer was greatest during the first 2 years after asthma diagnosis, and this risk remained elevated throughout the follow-up. Patients with nonatopic asthma had a greater risk of overall cancer, than those with atopic asthma. A high cumulative dose of ICS among asthma patients was associated with a 56% reduced risk of lung cancer but had no effect on the risk of overall cancer. For example, our group recently reported that asthma may potentially reduce the risk of glioma.107 While the relationship between asthma and the risk of cancer has been a longstanding epidemiological question in the field,108,109,110,111,112,113,114 it needs to be studied under a conceptual framework incorporating mechanisms for asthma and other AIMs. Also, as discussed above, it would be worthy of investigating similarities and differences between asthma-associated cancers and AIMs discussed in this review paper in relation to the clinical and immunogenetic characteristics of subgroups of asthmatic patients affected by these conditions. To investigate these research questions, it is necessary to leverage higher quality data sources such as EHR free texts than along with administrative data with inherent limitations and more cutting-edge technology-driven approaches such as AI-augmented machine learning or deep learning as discussed above. Ultimately, the improved knowledge on these issues will help us unravel the mechanisms and better mitigate the risk and even outcomes of AIMs. While the epithelial barrier dysfunction or disruption hypothesis draws significant scientific interest as a potential mechanism underlying AIMs, it needs to elucidate how the up-stream and down-stream immunological pathways interplay in a way impacting or resulting in the risk of specific individual AIMs in the context of interface between innate and adaptive immune functions. In this respect, research efforts understanding the causes and effects of asthma must be juxtaposed to each other because they are likely to guide each other.
Public health
The National Institute of Allergy and Infectious Diseases of the United States identified emerging and re-emerging infectious diseases which have significant public health threats at a population level, including measles, mumps, and pertussis.115,116 We demonstrated a significantly increased risk of vaccine preventable diseases such as pertussis and breakthrough varicella infection among individuals with asthma.18,117 As a potential mechanism, we reported more rapid waning of adaptive immunity over time in children with asthma who had been immunized with one-dose MMR vaccine at approximately 15 months of age.118 Measles antibody waned over time more rapidly in asthmatics (a decrease of −0.114 unit per year) than non-asthmatics (a decrease of −0.046 unit per year; P value for interaction = 0.010). This differential waning rate resulted in a lower mean measles antibody level, leading to a lower seropositivity rate in asthmatics compared to non-asthmatics (73% vs. 84%, P = 0.038) starting around 9.3 years after the initial measles vaccination. Importantly, children without asthma at the time of antibody measurement who subsequently developed asthma (genetic predisposition to asthma) had a lower antibody level than non-asthmatics and lower seropositivity (66%) (P = 0.024). Thus, asthma status is associated with the waning kinetics of measles antibody (humoral immunity) among children, which may have an important public health implication on the epidemiology of emerging and re-emerging infectious diseases by asthma epidemiology. In the context of the unprecedented impact of the COVID-19 pandemic on morbidity and mortality worldwide, given the large proportion of people affected by asthma, it is important to assess the relationship between asthma and the risk/outcomes of COVID-19 including potential differential waning of antibody against COVID-19 between asthmatics and non-asthmatics. While the reported relationship between asthma and the risk of COVID-19 has been inconsistent,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102 given the combination of methodological and biological heterogeneity of asthma as discussed above, these discrepancies are not surprising. While the effect of Th2 cytokines and asthma status on the expression of ACE2 receptors has also been inconsistent,119,120,121 identifying a subgroup of asthmatics at an increased risk of COVID-19 or its-related poor outcomes is clinically and scientifically important, especially in the context of the impact of asthma control status and therapies on the risk of COVID-19 (e.g., ICS therapy was associated with lower expression of ACE2 and TMPRSS2 receptor genes). Along these lines, beyond the currently recognized AIMs, it is worthy of investigating the impact of asthma epidemiology on cancer risk as this poses an important public health implication.106,122
In conclusion, our systematic review and case study on the association of asthma with the risk of AIMs suggest that while AIMs are relatively under-recognized conditions, they pose major health threats to people with asthma. AIMs provide new insights into immunological and clinical features of asthma as a systemic inflammatory disease beyond a solely chronic airway disease. It is time to recognize AIMs as a distinctive asthma phenotype in order to advance asthma research and improve asthma care.
ACKNOWLEDGMENTS
The presented work was supported by the NIH (R01 HL 126667 and R21 AI 142702).
Footnotes
Disclosure: There are no financial or other issues that might lead to conflict of interest.
SUPPLEMENTARY MATERIALS
Systematic review of the literature on AIMs and literature search strategy
Description for a population-based birth cohort study
Systematic review for AIMs from the literature
Twenty-three infectious and inflammatory diseases and their designated ICD codes used for retrieval of each condition
References
- 1.World Health Organization. Fact sheet for asthma [Internet] Geneva: World Health Organization; 2017. [cited 2018 Jan 23]. Available from: http://www.who.int/mediacentre/factsheets/fs307/en/index.html. [Google Scholar]
- 2.Centers for Disease Control and Prevention (CDC); Advisory Committee on Immunization Practices. Updated recommendations for prevention of invasive pneumococcal disease among adults using the 23-valent pneumococcal polysaccharide vaccine (PPSV23) MMWR Morb Mortal Wkly Rep. 2010;59:1102–1106. [PubMed] [Google Scholar]
- 3.Canada PHAo. An Advisory Committee Statement (ACS) National Advisory Committee On Immunization (NACI): update on the use of pneumococcal vaccines: addition of asthma as a high-risk condition. Ottawa: Canada PHAo; 2014. [Google Scholar]
- 4.Helby J, Nordestgaard BG, Benfield T, Bojesen SE. Asthma, other atopic conditions and risk of infections in 105 519 general population never and ever smokers. J Intern Med. 2017;282:254–267. doi: 10.1111/joim.12635. [DOI] [PubMed] [Google Scholar]
- 5.Kawai K, Yawn BP. Risk factors for herpes zoster: a systematic review and meta-analysis. Mayo Clin Proc. 2017;92:1806–1821. doi: 10.1016/j.mayocp.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 6.Deschildre A, Pin I, El Abd K, Belmin-Larrar S, El Mourad S, Thumerelle C, et al. Asthma control assessment in a pediatric population: comparison between GINA/NAEPP guidelines, Childhood Asthma Control Test (C-ACT), and physician's rating. Allergy. 2014;69:784–790. doi: 10.1111/all.12402. [DOI] [PubMed] [Google Scholar]
- 7.Juhn YJ. Risks for infection in patients with asthma (or other atopic conditions): is asthma more than a chronic airway disease? J Allergy Clin Immunol. 2014;134:247–257. doi: 10.1016/j.jaci.2014.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Talbot TR, Hartert TV, Mitchel E, Halasa NB, Arbogast PG, Poehling KA, et al. Asthma as a risk factor for invasive pneumococcal disease. N Engl J Med. 2005;352:2082–2090. doi: 10.1056/NEJMoa044113. [DOI] [PubMed] [Google Scholar]
- 9.Klemets P, Lyytikäinen O, Ruutu P, Ollgren J, Kaijalainen T, Leinonen M, et al. Risk of invasive pneumococcal infections among working age adults with asthma. Thorax. 2010;65:698–702. doi: 10.1136/thx.2009.132670. [DOI] [PubMed] [Google Scholar]
- 10.Juhn YJ, Kita H, Yawn BP, Boyce TG, Yoo KH, McGree ME, et al. Increased risk of serious pneumococcal disease in patients with asthma. J Allergy Clin Immunol. 2008;122:719–723. doi: 10.1016/j.jaci.2008.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Celebi Sözener Z, Cevhertas L, Nadeau K, Akdis M, Akdis CA. Environmental factors in epithelial barrier dysfunction. J Allergy Clin Immunol. 2020;145:1517–1528. doi: 10.1016/j.jaci.2020.04.024. [DOI] [PubMed] [Google Scholar]
- 12.Kim BS, Mehra S, Yawn B, Grose C, Tarrell R, Lahr B, et al. Increased risk of herpes zoster in children with asthma: a population-based case-control study. J Pediatr. 2013;163:816–821. doi: 10.1016/j.jpeds.2013.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kwon HJ, Bang DW, Kim EN, Wi CI, Yawn BP, Wollan PC, et al. Asthma as a risk factor for zoster in adults: a population-based case-control study. J Allergy Clin Immunol. 2016;137:1406–1412. doi: 10.1016/j.jaci.2015.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Forbes HJ, Bhaskaran K, Thomas SL, Smeeth L, Clayton T, Langan SM. Quantification of risk factors for herpes zoster: population based case-control study. BMJ. 2014;348:g2911. doi: 10.1136/bmj.g2911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Esteban-Vasallo MD, Domínguez-Berjón MF, Gil-Prieto R, Astray-Mochales J, Gil de Miguel A. Sociodemographic characteristics and chronic medical conditions as risk factors for herpes zoster: a population-based study from primary care in Madrid (Spain) Hum Vaccin Immunother. 2014;10:1650–1660. doi: 10.4161/hv.28620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kilgore PE, Kruszon-Moran D, Seward JF, Jumaan A, Van Loon FP, Forghani B, et al. Varicella in Americans from NHANES III: implications for control through routine immunization. J Med Virol. 2003;70(Suppl 1):S111–8. doi: 10.1002/jmv.10364. [DOI] [PubMed] [Google Scholar]
- 17.Reynolds MA, Kruszon-Moran D, Jumaan A, Schmid DS, McQuillan GM. Varicella seroprevalence in the U.S.: data from the National Health and Nutrition Examination Survey, 1999–2004. Public Health Rep. 2010;125:860–869. doi: 10.1177/003335491012500613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Umaretiya PJ, Swanson JB, Kwon HJ, Grose C, Lohse CM, Juhn YJ. Asthma and risk of breakthrough varicella infection in children. Allergy Asthma Proc. 2016;37:207–215. doi: 10.2500/aap.2016.37.3951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Krishna MT, Subramanian A, Adderley NJ, Zemedikun DT, Gkoutos GV, Nirantharakumar K. Allergic diseases and long-term risk of autoimmune disorders: longitudinal cohort study and cluster analysis. Eur Respir J. 2019;54:1900476. doi: 10.1183/13993003.00476-2019. [DOI] [PubMed] [Google Scholar]
- 20.Yawn BP, Wollan P, Kurland M, Scanlon P. A longitudinal study of the prevalence of asthma in a community population of school-age children. J Pediatr. 2002;140:576–581. doi: 10.1067/mpd.2002.123764. [DOI] [PubMed] [Google Scholar]
- 21.Juhn YJ, Beebe TJ, Finnie DM, Sloan J, Wheeler PH, Yawn B, et al. Development and initial testing of a new socioeconomic status measure based on housing data. J Urban Health. 2011;88:933–944. doi: 10.1007/s11524-011-9572-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wi CI, St Sauver JL, Jacobson DJ, Pendegraft RS, Lahr BD, Ryu E, et al. Ethnicity, socioeconomic status, and health disparities in a mixed rural-urban US Community-Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:612–622. doi: 10.1016/j.mayocp.2016.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bjur KA, Wi CI, Ryu E, Derauf C, Crow SS, King KS, et al. Socioeconomic status, race/ethnicity, and health disparities in children and adolescents in a mixed rural-urban community-Olmsted County, Minnesota. Mayo Clin Proc. 2019;94:44–53. doi: 10.1016/j.mayocp.2018.06.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim MH, Rhee CK, Shim JS, Park SY, Yoo KH, Kim BY, et al. Inhaled corticosteroids in asthma and the risk of pneumonia. Allergy Asthma Immunol Res. 2019;11:795–805. doi: 10.4168/aair.2019.11.6.795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McKeever T, Harrison TW, Hubbard R, Shaw D. Inhaled corticosteroids and the risk of pneumonia in people with asthma: a case-control study. Chest. 2013;144:1788–1794. doi: 10.1378/chest.13-0871. [DOI] [PubMed] [Google Scholar]
- 26.McKeever T, Mortimer K, Wilson A, Walker S, Brightling C, Skeggs A, et al. Quadrupling inhaled glucocorticoid dose to abort asthma exacerbations. N Engl J Med. 2018;378:902–910. doi: 10.1056/NEJMoa1714257. [DOI] [PubMed] [Google Scholar]
- 27.Qian CJ, Coulombe J, Suissa S, Ernst P. Pneumonia risk in asthma patients using inhaled corticosteroids: a quasi-cohort study. Br J Clin Pharmacol. 2017;83:2077–2086. doi: 10.1111/bcp.13295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.O’Byrne PM, Pedersen S, Carlsson LG, Radner F, Thorén A, Peterson S, et al. Risks of pneumonia in patients with asthma taking inhaled corticosteroids. Am J Respir Crit Care Med. 2011;183:589–595. doi: 10.1164/rccm.201005-0694OC. [DOI] [PubMed] [Google Scholar]
- 29.Bansal V, Mangi MA, Johnson MM, Festic E. Inhaled corticosteroids and incident pneumonia in patients with asthma: Systematic review and meta-analysis. Acta Med Acad. 2015;44:135–158. doi: 10.5644/ama2006-124.141. [DOI] [PubMed] [Google Scholar]
- 30.Festic E, Bansal V, Gajic O, Lee AS, the United States Critical Illness and InjuryTrials Group: Lung Injury Prevention Study Investigators (USCIITG-LIPS) Prehospital use of inhaled corticosteroids and point prevalence of pneumonia at the time of hospital admission: secondary analysis of a multicenter cohort study. Mayo Clin Proc. 2014;89:154–162. doi: 10.1016/j.mayocp.2013.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Almirall J, Bolíbar I, Serra-Prat M, Palomera E, Roig J, Hospital I, et al. Inhaled drugs as risk factors for community-acquired pneumonia. Eur Respir J. 2010;36:1080–1087. doi: 10.1183/09031936.00022909. [DOI] [PubMed] [Google Scholar]
- 32.Htun ZM, Aldawudi I, Katwal PC, Jirjees S, Khan S. Inhaled corticosteroids as an associated risk factor for asthmatic pneumonia: a literature review. Cureus. 2020;12:e8717–8717. doi: 10.7759/cureus.8717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sheffer AL, Silverman M, Woolcock AJ, Díaz PV, Lindberg B, Lindmark B. Long-term safety of once-daily budesonide in patients with early-onset mild persistent asthma: results of the inhaled steroid treatment as regular therapy in early asthma (START) study. Ann Allergy Asthma Immunol. 2005;94:48–54. doi: 10.1016/S1081-1206(10)61285-9. [DOI] [PubMed] [Google Scholar]
- 34.Woodcock A, Bateman ED, Busse WW, Lötvall J, Snowise NG, Forth R, et al. Efficacy in asthma of once-daily treatment with fluticasone furoate: a randomized, placebo-controlled trial. Respir Res. 2011;12:132–132. doi: 10.1186/1465-9921-12-132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Doull IJ, Lampe FC, Smith S, Schreiber J, Freezer NJ, Holgate ST. Effect of inhaled corticosteroids on episodes of wheezing associated with viral infection in school age children: randomised double blind placebo controlled trial. BMJ. 1997;315:858–862. doi: 10.1136/bmj.315.7112.858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Martin RJ, Kraft M, Chu HW, Berns EA, Cassell GH. A link between chronic asthma and chronic infection. J Allergy Clin Immunol. 2001;107:595–601. doi: 10.1067/mai.2001.113563. [DOI] [PubMed] [Google Scholar]
- 37.Busse WW, Morgan WJ, Taggart V, Togias A. Asthma outcomes workshop: overview. J Allergy Clin Immunol. 2012;129:S1–8. doi: 10.1016/j.jaci.2011.12.985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Edwards MR, Strong K, Cameron A, Walton RP, Jackson DJ, Johnston SL. Viral infections in allergy and immunology: how allergic inflammation influences viral infections and illness. J Allergy Clin Immunol. 2017;140:909–920. doi: 10.1016/j.jaci.2017.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sykes A, Macintyre J, Edwards MR, Del Rosario A, Haas J, Gielen V, et al. Rhinovirus-induced interferon production is not deficient in well controlled asthma. Thorax. 2014;69:240–246. doi: 10.1136/thoraxjnl-2012-202909. [DOI] [PubMed] [Google Scholar]
- 40.Edwards MR, Regamey N, Vareille M, Kieninger E, Gupta A, Shoemark A, et al. Impaired innate interferon induction in severe therapy resistant atopic asthmatic children. Mucosal Immunol. 2013;6:797–806. doi: 10.1038/mi.2012.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fitzpatrick AM, Teague WG, Meyers DA, Peters SP, Li X, Li H, et al. Heterogeneity of severe asthma in childhood: confirmation by cluster analysis of children in the National Institutes of Health/National Heart, Lung, and Blood Institute Severe Asthma Research Program. J Allergy Clin Immunol. 2011;127:382–389.e1-13. doi: 10.1016/j.jaci.2010.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chang TS, Lemanske RF, Jr, Mauger DT, Fitzpatrick AM, Sorkness CA, Szefler SJ, et al. Childhood asthma clusters and response to therapy in clinical trials. J Allergy Clin Immunol. 2014;133:363–369. doi: 10.1016/j.jaci.2013.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Howrylak JA, Fuhlbrigge AL, Strunk RC, Zeiger RS, Weiss ST, Raby BA, et al. Classification of childhood asthma phenotypes and long-term clinical responses to inhaled anti-inflammatory medications. J Allergy Clin Immunol. 2014;133:1289–1300. 1300.e1–1300.e12. doi: 10.1016/j.jaci.2014.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Havstad S, Johnson CC, Kim H, Levin AM, Zoratti EM, Joseph CL, et al. Atopic phenotypes identified with latent class analyses at age 2 years. J Allergy Clin Immunol. 2014;134:722–727.e2. doi: 10.1016/j.jaci.2014.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Herr M, Just J, Nikasinovic L, Foucault C, Le Marec AM, Giordanella JP, et al. Risk factors and characteristics of respiratory and allergic phenotypes in early childhood. J Allergy Clin Immunol. 2012;130:389–396.e4. doi: 10.1016/j.jaci.2012.05.054. [DOI] [PubMed] [Google Scholar]
- 46.Lazic N, Roberts G, Custovic A, Belgrave D, Bishop CM, Winn J, et al. Multiple atopy phenotypes and their associations with asthma: similar findings from two birth cohorts. Allergy. 2013;68:764–770. doi: 10.1111/all.12134. [DOI] [PubMed] [Google Scholar]
- 47.Kurukulaaratchy RJ, Zhang H, Raza A, Patil V, Karmaus W, Ewart S, et al. The diversity of young adult wheeze: a cluster analysis in a longitudinal birth cohort. Clin Exp Allergy. 2014;44:724–735. doi: 10.1111/cea.12306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Just J, Gouvis-Echraghi R, Rouve S, Wanin S, Moreau D, Annesi-Maesano I. Two novel, severe asthma phenotypes identified during childhood using a clustering approach. Eur Respir J. 2012;40:55–60. doi: 10.1183/09031936.00123411. [DOI] [PubMed] [Google Scholar]
- 49.Zoratti EM, Krouse RZ, Babineau DC, Pongracic JA, O'Connor GT, Wood RA, et al. Asthma phenotypes in inner-city children. J Allergy Clin Immunol. 2016;138:1016–1029. doi: 10.1016/j.jaci.2016.06.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Spycher BD, Silverman M, Brooke AM, Minder CE, Kuehni CE. Distinguishing phenotypes of childhood wheeze and cough using latent class analysis. Eur Respir J. 2008;31:974–981. doi: 10.1183/09031936.00153507. [DOI] [PubMed] [Google Scholar]
- 51.Henderson J, Granell R, Heron J, Sherriff A, Simpson A, Woodcock A, et al. Associations of wheezing phenotypes in the first 6 years of life with atopy, lung function and airway responsiveness in mid-childhood. Thorax. 2008;63:974–980. doi: 10.1136/thx.2007.093187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Savenije OE, Granell R, Caudri D, Koppelman GH, Smit HA, Wijga A, et al. Comparison of childhood wheezing phenotypes in 2 birth cohorts: ALSPAC and PIAMA. J Allergy Clin Immunol. 2011;127:1505–1512.e14. doi: 10.1016/j.jaci.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 53.Benton AS, Wang Z, Lerner J, Foerster M, Teach SJ, Freishtat RJ. Overcoming heterogeneity in pediatric asthma: tobacco smoke and asthma characteristics within phenotypic clusters in an African American cohort. J Asthma. 2010;47:728–734. doi: 10.3109/02770903.2010.491142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Carraro S, Giordano G, Reniero F, Carpi D, Stocchero M, Sterk PJ, et al. Asthma severity in childhood and metabolomic profiling of breath condensate. Allergy. 2013;68:110–117. doi: 10.1111/all.12063. [DOI] [PubMed] [Google Scholar]
- 55.Weinmayr G, Keller F, Kleiner A, du Prel JB, Garcia-Marcos L, Batllés-Garrido J, et al. Asthma phenotypes identified by latent class analysis in the ISAAC phase II Spain study. Clin Exp Allergy. 2013;43:223–232. doi: 10.1111/cea.12035. [DOI] [PubMed] [Google Scholar]
- 56.Simpson A, Tan VY, Winn J, Svensén M, Bishop CM, Heckerman DE, et al. Beyond atopy: multiple patterns of sensitization in relation to asthma in a birth cohort study. Am J Respir Crit Care Med. 2010;181:1200–1206. doi: 10.1164/rccm.200907-1101OC. [DOI] [PubMed] [Google Scholar]
- 57.Granell R, Henderson AJ, Sterne JA. Associations of wheezing phenotypes with late asthma outcomes in the Avon Longitudinal Study of Parents and Children: a population-based birth cohort. J Allergy Clin Immunol. 2016;138:1060–1070.e11. doi: 10.1016/j.jaci.2016.01.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee E, Lee SH, Kim YH, Cho HJ, Yoon J, Yang SI, et al. Association of atopy phenotypes with new development of asthma and bronchial hyperresponsiveness in school-aged children. Ann Allergy Asthma Immunol. 2017;118:542–550.e1. doi: 10.1016/j.anai.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 59.Guiddir T, Saint-Pierre P, Purenne-Denis E, Lambert N, Laoudi Y, Couderc R, et al. Neutrophilic steroid-refractory recurrent wheeze and eosinophilic steroid-refractory asthma in children. J Allergy Clin Immunol Pract. 2017;5:1351–1361.e2. doi: 10.1016/j.jaip.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 60.Lee E, Lee SH, Kwon JW, Kim YH, Yoon J, Cho HJ, et al. Persistent asthma phenotype related with late-onset, high atopy, and low socioeconomic status in school-aged Korean children. BMC Pulm Med. 2017;17:45. doi: 10.1186/s12890-017-0387-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cabral ALB, Sousa AW, Mendes FAR, Carvalho CRFd. Phenotypes of asthma in low-income children and adolescents: cluster analysis. J Bras Pneumol. 2017;43:44–50. doi: 10.1590/S1806-37562016000000039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Deliu M, Yavuz TS, Sperrin M, Belgrave D, Sahiner UM, Sackesen C, et al. Features of asthma which provide meaningful insights for understanding the disease heterogeneity. Clin Exp Allergy. 2018;48:39–47. doi: 10.1111/cea.13014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hose AJ, Depner M, Illi S, Lau S, Keil T, Wahn U, et al. Latent class analysis reveals clinically relevant atopy phenotypes in 2 birth cohorts. J Allergy Clin Immunol. 2017;139:1935–1945.e12. doi: 10.1016/j.jaci.2016.08.046. [DOI] [PubMed] [Google Scholar]
- 64.Chen Q, Zhong X, Acosta L, Divjan A, Rundle A, Goldstein IF, et al. Allergic sensitization patterns identified through latent class analysis among children with and without asthma. Ann Allergy Asthma Immunol. 2016;116:212–218. doi: 10.1016/j.anai.2016.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Deliu M, Belgrave D, Sperrin M, Buchan I, Custovic A. Asthma phenotypes in childhood. Expert Rev Clin Immunol. 2017;13:705–713. doi: 10.1080/1744666X.2017.1257940. [DOI] [PubMed] [Google Scholar]
- 66.Custovic A. “Asthma” or “asthma spectrum disorder”? J Allergy Clin Immunol Pract. 2020;8:2628–2629. doi: 10.1016/j.jaip.2020.06.005. [DOI] [PubMed] [Google Scholar]
- 67.Harlak A, Gulec M, Mentes O, Kilbas Z, Onguru O, Acikel C, et al. Atopy is a risk factor for acute appendicitis? A prospective clinical study. J Gastrointest Surg. 2008;12:1251–1256. doi: 10.1007/s11605-008-0511-1. [DOI] [PubMed] [Google Scholar]
- 68.Kim SY, Oh DJ, Park B, Park IS, Choi HG. Increased risk of appendectomy in patients with asthma: A nested case-control study using a national sample cohort. Medicine (Baltimore) 2019;98:e17203. doi: 10.1097/MD.0000000000017203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ludvigsson JF, Hemminki K, Wahlström J, Almqvist C. Celiac disease confers a 1.6-fold increased risk of asthma: a nationwide population-based cohort study. J Allergy Clin Immunol. 2011;127:1071–1073. doi: 10.1016/j.jaci.2010.12.1076. [DOI] [PubMed] [Google Scholar]
- 70.Hasassri ME, Jackson ER, Ghawi H, Ryoo E, Wi CI, Bartlett MG, et al. Asthma and risk of appendicitis in children: a population-based case-control study. Acad Pediatr. 2017;17:205–211. doi: 10.1016/j.acap.2016.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Patel B, Wi CI, Hasassri ME, Divekar R, Absah I, Almallouhi E, et al. Heterogeneity of asthma and the risk of celiac disease in children. Allergy Asthma Proc. 2018;39:51–58. doi: 10.2500/aap.2018.39.4100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pothoven KL, Schleimer RP. The barrier hypothesis and Oncostatin M: restoration of epithelial barrier function as a novel therapeutic strategy for the treatment of type 2 inflammatory disease. Tissue Barriers. 2017;5:e1341367. doi: 10.1080/21688370.2017.1341367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Broderick L, Hoffman HM. Pediatric recurrent fever and autoinflammation from the perspective of an allergist/immunologist. J Allergy Clin Immunol. 2020;146:960–966.e2. doi: 10.1016/j.jaci.2020.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Salinas YD, Wang Z, DeWan AT. Discovery and mediation analysis of cross-phenotype associations between asthma and body mass index in 12q13.2. Am J Epidemiol. 2021;190:85–94. doi: 10.1093/aje/kwaa144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kreiner E, Waage J, Standl M, Brix S, Pers TH, Couto Alves A, et al. Shared genetic variants suggest common pathways in allergy and autoimmune diseases. J Allergy Clin Immunol. 2017;140:771–781. doi: 10.1016/j.jaci.2016.10.055. [DOI] [PubMed] [Google Scholar]
- 76.Li X, Ampleford EJ, Howard TD, Moore WC, Torgerson DG, Li H, et al. Genome-wide association studies of asthma indicate opposite immunopathogenesis direction from autoimmune diseases. J Allergy Clin Immunol. 2012;130:861–868.e7. doi: 10.1016/j.jaci.2012.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Maier LM, Howson JM, Walker N, Spickett GP, Jones RW, Ring SM, et al. Association of IL13 with total IgE: evidence against an inverse association of atopy and diabetes. J Allergy Clin Immunol. 2006;117:1306–1313. doi: 10.1016/j.jaci.2005.12.1354. [DOI] [PubMed] [Google Scholar]
- 78.Bach JF. The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med. 2002;347:911–920. doi: 10.1056/NEJMra020100. [DOI] [PubMed] [Google Scholar]
- 79.Lötvall J, Akdis CA, Bacharier LB, Bjermer L, Casale TB, Custovic A, et al. Asthma endotypes: a new approach to classification of disease entities within the asthma syndrome. J Allergy Clin Immunol. 2011;127:355–360. doi: 10.1016/j.jaci.2010.11.037. [DOI] [PubMed] [Google Scholar]
- 80.Van Wonderen KE, Van Der Mark LB, Mohrs J, Bindels PJ, Van Aalderen WM, Ter Riet G. Different definitions in childhood asthma: how dependable is the dependent variable? Eur Respir J. 2010;36:48–56. doi: 10.1183/09031936.00154409. [DOI] [PubMed] [Google Scholar]
- 81.Li X, Howard TD, Zheng SL, Haselkorn T, Peters SP, Meyers DA, et al. Genome-wide association study of asthma identifies RAD50-IL13 and HLA-DR/DQ regions. J Allergy Clin Immunol. 2010;125:328–335.e11. doi: 10.1016/j.jaci.2009.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ferreira MA, Matheson MC, Duffy DL, Marks GB, Hui J, Le Souëf P, et al. Identification of IL6R and chromosome 11q13.5 as risk loci for asthma. Lancet. 2011;378:1006–1014. doi: 10.1016/S0140-6736(11)60874-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Meyers DA. Genetics of asthma and allergy: what have we learned? J Allergy Clin Immunol. 2010;126:439–446. doi: 10.1016/j.jaci.2010.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ducharme FM, Lemire C, Noya FJ, Davis GM, Alos N, Leblond H, et al. Preemptive use of high-dose fluticasone for virus-induced wheezing in young children. N Engl J Med. 2009;360:339–353. doi: 10.1056/NEJMoa0808907. [DOI] [PubMed] [Google Scholar]
- 85.Panickar J, Lakhanpaul M, Lambert PC, Kenia P, Stephenson T, Smyth A, et al. Oral prednisolone for preschool children with acute virus-induced wheezing. N Engl J Med. 2009;360:329–338. doi: 10.1056/NEJMoa0804897. [DOI] [PubMed] [Google Scholar]
- 86.Haldar P, Pavord ID, Shaw DE, Berry MA, Thomas M, Brightling CE, et al. Cluster analysis and clinical asthma phenotypes. Am J Respir Crit Care Med. 2008;178:218–224. doi: 10.1164/rccm.200711-1754OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Moore WC, Meyers DA, Wenzel SE, Teague WG, Li H, Li X, et al. Identification of asthma phenotypes using cluster analysis in the Severe Asthma Research Program. Am J Respir Crit Care Med. 2010;181:315–323. doi: 10.1164/rccm.200906-0896OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Antonicelli L, Tontini C, Manzotti G, Ronchi L, Vaghi A, Bini F, et al. Severe asthma in adults does not significantly affect the outcome of COVID-19 disease: results from the Italian Severe Asthma Registry. Allergy. 2021;76:902–905. doi: 10.1111/all.14558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Broadhurst R, Peterson R, Wisnivesky JP, Federman A, Zimmer SM, Sharma S, et al. Asthma in COVID-19 hospitalizations: an overestimated risk factor? Ann Am Thorac Soc. 2020;17:1645–1648. doi: 10.1513/AnnalsATS.202006-613RL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cunningham JW, Vaduganathan M, Claggett BL, Jering KS, Bhatt AS, Rosenthal N, et al. Clinical outcomes in young US adults hospitalized with COVID-19. JAMA Intern Med. 2020;181:379–381. doi: 10.1001/jamainternmed.2020.5313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Garcia-Pachon E, Zamora-Molina L, Soler-Sempere MJ, Baeza-Martinez C, Grau-Delgado J, Canto-Reig V, et al. Asthma prevalence in patients with SARS-CoV-2 infection detected by RT-PCR not requiring hospitalization. Respir Med. 2020;171:106084. doi: 10.1016/j.rmed.2020.106084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gupta S, Hayek SS, Wang W, Chan L, Mathews KS, Melamed ML, et al. Factors associated with death in critically Ill patients with coronavirus disease 2019 in the US. JAMA Intern Med. 2020;180:1436–1447. doi: 10.1001/jamainternmed.2020.3596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Haroun-Díaz E, Vázquez de la Torre M, Ruano FJ, Somoza Álvarez ML, Alzate DP, González PL, et al. Severe asthma during the COVID-19 pandemic: clinical observations. J Allergy Clin Immunol Pract. 2020;8:2787–2789. doi: 10.1016/j.jaip.2020.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Heffler E, Detoraki A, Contoli M, Papi A, Paoletti G, Malipiero G, et al. COVID-19 in Severe Asthma Network in Italy (SANI) patients: Clinical features, impact of comorbidities and treatments. Allergy. 2021;76:887–892. doi: 10.1111/all.14532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Keswani A, Dhana K, Rosenthal JA, Moore D, Mahdavinia M. Atopy is predictive of a decreased need for hospitalization for coronavirus disease 2019. Ann Allergy Asthma Immunol. 2020;125:479–481. doi: 10.1016/j.anai.2020.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lombardi C, Roca E, Bigni B, Cottini M, Passalacqua G. Clinical course and outcomes of patients with asthma hospitalized for severe acute respiratory syndrome coronavirus 2 pneumonia: a single-center, retrospective study. Ann Allergy Asthma Immunol. 2020;125:707–709. doi: 10.1016/j.anai.2020.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lovinsky-Desir S, Deshpande DR, De A, Murray L, Stingone JA, Chan A, et al. Asthma among hospitalized patients with COVID-19 and related outcomes. J Allergy Clin Immunol. 2020;146:1027–1034.e4. doi: 10.1016/j.jaci.2020.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Song J, Zeng M, Wang H, Qin C, Hou HY, Sun ZY, et al. Distinct effects of asthma and COPD comorbidity on disease expression and outcome in patients with COVID-19. Allergy. 2021;76:483–496. doi: 10.1111/all.14517. [DOI] [PubMed] [Google Scholar]
- 99.Ssentongo P, Ssentongo AE, Heilbrunn ES, Ba DM, Chinchilli VM. Association of cardiovascular disease and 10 other pre-existing comorbidities with COVID-19 mortality: a systematic review and meta-analysis. PLoS One. 2020;15:e0238215. doi: 10.1371/journal.pone.0238215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wang L, Foer D, Bates DW, Boyce JA, Zhou L. Risk factors for hospitalization, intensive care, and mortality among patients with asthma and COVID-19. J Allergy Clin Immunol. 2020;146:808–812. doi: 10.1016/j.jaci.2020.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yang JM, Koh HY, Moon SY, Yoo IK, Ha EK, You S, et al. Allergic disorders and susceptibility to and severity of COVID-19: a nationwide cohort study. J Allergy Clin Immunol. 2020;146:790–798. doi: 10.1016/j.jaci.2020.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Seol HY, Rolfes MC, Chung W, Sohn S, Ryu E, Park MA, et al. Expert artificial intelligence-based natural language processing characterises childhood asthma. BMJ Open Respir Res. 2020;7:e000524. doi: 10.1136/bmjresp-2019-000524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Seol HY, Sohn S, Liu H, Wi CI, Ryu E, Park MA, et al. Early identification of childhood asthma: the role of informatics in an era of electronic health records. Front Pediatr. 2019;7:113. doi: 10.3389/fped.2019.00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Juhn Y, Liu H. Artificial intelligence approaches using natural language processing to advance EHR-based clinical research. J Allergy Clin Immunol. 2020;145:463–469. doi: 10.1016/j.jaci.2019.12.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Woo A, Lee SW, Koh HY, Kim MA, Han MY, Yon DK. Incidence of cancer after asthma development: 2 independent population-based cohort studies. J Allergy Clin Immunol. 2021;147:135–143. doi: 10.1016/j.jaci.2020.04.041. [DOI] [PubMed] [Google Scholar]
- 107.Kaur H, Lachance DH, Ryan CS, Sheen YH, Seol HY, Wi CI, et al. Asthma and risk of glioma: a population-based case-control study. BMJ Open. 2019;9:e025746. doi: 10.1136/bmjopen-2018-025746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.García Sanz MT, González Barcala FJ, Alvarez Dobaño JM, Valdés Cuadrado L. Asthma and risk of lung cancer. Clin Transl Oncol. 2011;13:728–730. doi: 10.1007/s12094-011-0723-9. [DOI] [PubMed] [Google Scholar]
- 109.Ivansson EL, Rasmussen F, Gyllensten UB, Magnusson PK. Reduced incidence of cervical cancer in mothers of sons with allergic rhinoconjunctivitis, asthma or eczema. Int J Cancer. 2006;119:1994–1998. doi: 10.1002/ijc.22070. [DOI] [PubMed] [Google Scholar]
- 110.Lowcock EC, Cotterchio M, Ahmad N. Association between allergies, asthma, and breast cancer risk among women in Ontario, Canada. Cancer Causes Control. 2013;24:1053–1056. doi: 10.1007/s10552-013-0177-1. [DOI] [PubMed] [Google Scholar]
- 111.Wang H, Diepgen TL. Is atopy a protective or a risk factor for cancer? A review of epidemiological studies. Allergy. 2005;60:1098–1111. doi: 10.1111/j.1398-9995.2005.00813.x. [DOI] [PubMed] [Google Scholar]
- 112.Vajdic CM, Falster MO, de Sanjose S, Martínez-Maza O, Becker N, Bracci PM, et al. Atopic disease and risk of non-Hodgkin lymphoma: an InterLymph pooled analysis. Cancer Res. 2009;69:6482–6489. doi: 10.1158/0008-5472.CAN-08-4372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Tambe NA, Wilkens LR, Wan P, Stram DO, Gilliland F, Park SL, et al. Atopic allergic conditions and colorectal cancer risk in the multiethnic cohort study. Am J Epidemiol. 2015;181:889–897. doi: 10.1093/aje/kwu361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Taranova AG, Maldonado D, 3rd, Vachon CM, Jacobsen EA, Abdala-Valencia H, McGarry MP, et al. Allergic pulmonary inflammation promotes the recruitment of circulating tumor cells to the lung. Cancer Res. 2008;68:8582–8589. doi: 10.1158/0008-5472.CAN-08-1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Morens DM, Folkers GK, Fauci AS. The challenge of emerging and re-emerging infectious diseases. Nature. 2004;430:242–249. doi: 10.1038/nature02759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.National Institute of Allergy and Infectious Diseases. NIAID emerging diseases/pathogens [Internet] Rockville (MD): National Institute of Allergy and Infectious Diseases; 2016. [cited 2017 Dec 21]. Available from: https://www.niaid.nih.gov/research/emerging-infectious-diseases-pathogens. [Google Scholar]
- 117.Capili CR, Hettinger A, Rigelman-Hedberg N, Fink L, Boyce T, Lahr B, et al. Increased risk of pertussis in patients with asthma. J Allergy Clin Immunol. 2012;129:957–963. doi: 10.1016/j.jaci.2011.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Yoo KH, Jacobson RM, Poland GA, Weaver A, Lee L, Chang T, et al. Asthma status and waning of measles antibody concentrations after measles immunization. Pediatr Infect Dis J. 2014;33:1016–1022. doi: 10.1097/INF.0000000000000375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bradding P, Richardson M, Hinks TSC, Howarth PH, Choy DF, Arron JR, et al. ACE2, TMPRSS2, and furin gene expression in the airways of people with asthma-implications for COVID-19. J Allergy Clin Immunol. 2020;146:208–211. doi: 10.1016/j.jaci.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kimura H, Francisco D, Conway M, Martinez FD, Vercelli D, Polverino F, et al. Type 2 inflammation modulates ACE2 and TMPRSS2 in airway epithelial cells. J Allergy Clin Immunol. 2020;146:80–88.e8. doi: 10.1016/j.jaci.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Peters MC, Sajuthi S, Deford P, Christenson S, Rios CL, Montgomery MT, et al. COVID-19-related genes in sputum cells in asthma. relationship to demographic features and corticosteroids. Am J Respir Crit Care Med. 2020;202:83–90. doi: 10.1164/rccm.202003-0821OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Gergen PJ. Adult-onset asthma and cancer: causal or coincidental? J Allergy Clin Immunol. 2021;147:52–53. doi: 10.1016/j.jaci.2020.10.028. [DOI] [PubMed] [Google Scholar]
- 123.Juhn YJ. The health threats to people with asthma through asthma-associated infectious disease comorbidities are largely under-recognized. J Intern Med. 2017;282:268–271. doi: 10.1111/joim.12650. [DOI] [PubMed] [Google Scholar]
- 124.Wi CI, Wheeler PH, Kaur H, Ryu E, Kim D, Juhn Y. Spatio-temporal comparison of pertussis outbreaks in Olmsted County, Minnesota, 2004–2005 and 2012: a population-based study. BMJ Open. 2019;9:e025521. doi: 10.1136/bmjopen-2018-025521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Finkelstein J, Jeong IC. Machine learning approaches to personalize early prediction of asthma exacerbations. Ann N Y Acad Sci. 2017;1387:153–165. doi: 10.1111/nyas.13218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ryu E, Wi CI, Sohn S, Moon S, Liu H, Green JA, et al. Application of data-driven approaches for identifying asthmatic children with suboptimal asthma care. J Allergy Clin Immunol. 2016;139:AB102. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Systematic review of the literature on AIMs and literature search strategy
Description for a population-based birth cohort study
Systematic review for AIMs from the literature
Twenty-three infectious and inflammatory diseases and their designated ICD codes used for retrieval of each condition