Abstract
The outbreak of coronavirus disease 2019 (COVID‐19) has rapidly evolved into a global pandemic. A significant proportion of COVID‐19 patients develops severe symptoms, which may include acute respiratory distress syndrome and acute kidney injury as manifestations of multi‐organ failure. Acute kidney injury (AKI) necessitating renal replacement therapy (RRT) is increasingly prevalent among critically ill patients with COVID‐19. However, few studies have focused on AKI treated with RRT. Many questions are awaiting answers as regards AKI in the setting of COVID‐19; whether patients with COVID‐19 commonly develop AKI, what are the underlying pathophysiologic mechanisms? What is the best evidence regarding treatment approaches? Identification of the potential indications and the preferred modalities of RRT in this context, is based mainly on clinical experience. Here, we review the current approaches of RRT, required for management of critically ill patients with COVID‐19 complicated by severe AKI as well as the precautions that should be adopted by health care providers in dealing with these cases. Electronic search was conducted in MEDLINE, PubMed, ISI Web of Science, and Scopus scientific databases. We searched the terms relevant to this review to identify the relevant studies. We also searched the conference proceedings and ClinicalTrials.gov database.
Keywords: acute kidney injury, COVID‐19, critically ill patients, hemodialysis, peritoneal dialysis
1. INTRODUCTION
Coronaviruses are positive‐stranded RNA viruses with a crown‐like appearance under an electron microscope owing to a glycoprotein spike on the envelope. Genomic characterization has revealed that bats and rodents are the gene sources of alpha CoVs and betaCoVs [1]. The genome of the new HCoV, had 89% nucleotide identity with bat SARS‐like‐CoVZXC21 and 82% with that of human SARS‐CoV [2]. Accordingly, the new virus was termed SARS‐CoV‐2, which belongs to the betaCoVs category [3].
The first reported cases of the coronavirus disease 2019 (COVID‐19) were related to direct exposure to the Huanan Seafood Wholesale Market of Wuhan, for that reason, the animal‐to‐human transmission was supposed to be the main mechanism. However, subsequent cases were not associated with this exposure. Consequently, it was concluded that the virus could also be transmitted from human‐to‐human. Like other respiratory pathogens, the transmission is believed to occur via droplet infection as well as aerosol transmission. The incubation time could be generally within 3–7 days and up to 2 weeks [4].
Diagnosis [5]:
Detection of viral RNA through manual or automated nucleic acid amplification tests (NAAT), such as real time reverse transcription polymerase chain reaction (rRT‐PCR). Nucleic acid amplification tests (NAAT) are the gold standard for diagnosis of acute SARS‐CoV‐2 infection. Mutation‐detecting NAAT assays may be used as a screening tool for SARS‐CoV‐2 variants.
Detection of viral antigens through immunodiagnostic techniques, such as lateral flow assays (LFAs), also termed rapid diagnostic tests or Ag‐RDTs. Ag‐RDTs helps to increase the availability and speed of testing in appropriate scenarios.
Detection of host antibodies through serological techniques, such as LFAs, ELISAs, or chemiluminescent immunoassays (CLIAs). Antibody detection is not recommended for diagnosis of COVID‐19, but it helps in the detection of past infection for surveillance
Medical imaging: Chest CT scans may be helpful to diagnose COVID‐19 pneumonia, however, the complex implementation techniques and high‐cost CT scans are not appropriate as a frontline tool. Typical features on CT initially include bilateral multi‐lobar ground‐glass opacities and infiltrating shadows on bilateral lobules [6, 7].
2. AKI IN CRITICALLY ILL PATIENTS WITH COVID‐19
Although some studies reported that AKI is uncommon in COVID‐19, and that SARS‐CoV‐2 infection does not result in AKI [8], others reported that the incidence of AKI among COVID‐19 infected patients ranges from 0.5% to 23%, and up to 50% among non‐survivors [9, 10, 11, 12], with renal replacement therapy (RRT) required in 16%–23% of intensive care unit (ICU) or mechanically‐ventilated patients [9, 13]. This controverse could be attributed to the diversity of symptoms during the initial phase of COVID‐19 pandemic, lack of definitive diagnostic tools along with a shortage of infrastructure and training of medical staff in non‐specialty hospitals. This in addition to enrollment of milder cases with incomplete documentation of laboratory testing. In a cohort of 3099 critically ill patients with COVID‐19 across the United States, 20.6% developed AKI requiring RRT [14]. The overall incidence of AKI in COVID‐19 patients was even higher in other cohorts of 28% and reaching up to 66% in severe cases [15].
The development of AKI in patients infected with SARS‐CoV‐2 has been explained by two theories; a direct cytopathic effect on kidney tissues, and tissue hypoxia, in the setting of massive cytokine release as well as the systemic effects of virus [16]. It has been identified that SARS‐CoV‐2 binds to the host cell receptor angiotensin converting enzyme 2 (ACE2). Therefore, it is assumed that the cells with ACE2 expression such as renal tubular epithelial cells may be more susceptible to COVID‐19 infection and therefore the risk of AKI is increased, especially in case of high viraemia [16]. This coincides with earlier reports revealing that SARS‐CoV‐2 has been isolated from urine of infected patients [17].
The cytokine release syndrome (CRS) which characterizes COVID‐19 infection includes in part AKI because of intrarenal inflammation, increased vascular permeability, volume depletion, and cardiomyopathy. The predominant heart–kidney crosstalk could also contribute to AKI in patients with COVID‐19; for instance, CRS cardiomyopathy and acute viral myocarditis can both contribute to renal vein congestion, hypotension and renal hypoperfusion, leading to a reduction in glomerular filtration rate [18]. Additionally, rhabdomyolysis, metabolic acidosis, and hyperkalemia can also develop in critically ill patients with COVID‐19, compromising kidney injury [19].
Critically ill patients with COVID‐19 exhibited significantly higher in‐hospital mortality rate reaching up to 40%–60% when complicated by impairments of kidney function, including hematuria, proteinuria, elevated baseline serum creatinine or BUN, and AKI [12, 14, 20, 21]. This reflects the importance of anticipation, prevention, and early management of AKI in those vulnerable patients.
Management of those patients consists of ensuring suitable infection control, supportive care, and possible use of drugs with potential activity against severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) [22]. General management plans should include pharmacologic prophylaxis of venous thromboembolism for all hospitalized patients [23] The critically ill COVID‐19‐positive patients should be treated in isolation ICUs dedicated to COVID‐19.
3. INDICATIONS AND MODALITIES OF RRT IN CRITICALLY ILL PATIENTS WITH COVID‐19
The treatment of AKI, indications of renal replacement therapy, and the modality of RRT seem to be no different than in other settings. Extracorporeal therapies have also been studied as potential treatments for removal of cytokines in patients with sepsis, and therefore, potentially considered for prevention of CRS‐induced organ damage in critically ill patients with COVID‐19 [24].
The indications of RRT should be those generally accepted to treatment of AKI, such as restoration of the immune homeostasis, removal of inflammatory mediators which lead to acute respiratory distress syndrome (ARDS), and prevention of fluid overload which represents a known independent risk factor for ICU mortality [25]. Fluid overload may also compromise the respiratory system by inducing several complications including pulmonary edema [26]. Although, there is no sufficient data to support the early initiation of RRT in COVID‐19 patients with AKI, [27], early initiation of RRT in critically ill patients and patients with sepsis seem to attenuate the disease severity and improve outcomes [28, 29]. In COVID‐19 patients with severe AKI, the continuation of RRT has been shown as vital for their survival [27]. The coexistence of other comorbidities such as diabetes mellitus and hypertension, in addition to the severity of respiratory disease severity determine the treatment modality [13].
The currently used modalities of RRT include continuous renal replacement therapy (CRRT), prolonged intermittent hemodialysis (PIHD), conventional intermittent hemodialysis (IHD), and peritoneal dialysis (PD) (Figure 1). CRRT seems to be the most used modality in patients with critical illness. The potential use of these modalities in the context of COVID‐19 is studied as follows:
FIGURE 1.
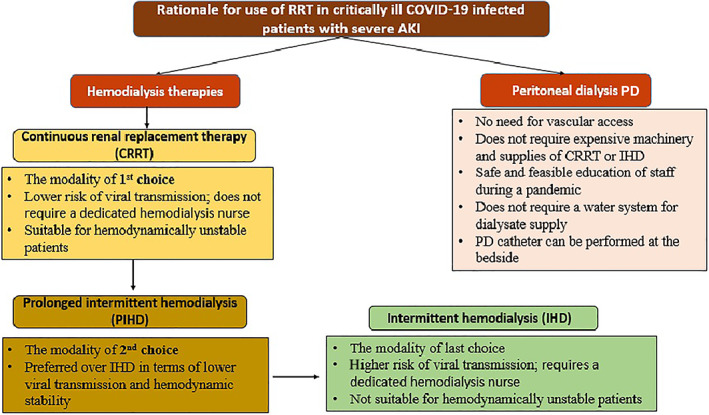
Rationale for use of renal replacement therapy in critically ill COVID‐19 infected patients with severe acute kidney injury (AKI)
3.1. Continuous renal replacement therapy
The main indication for selecting CRRT over IHD is hemodynamic instability and high catabolic states [27]. For COVID‐19 patients with AKI, CRRT is indicated in patients with stage 3 AKI (defined as either an increase in creatinine levels of a 3‐fold from baseline or creatinine ≥4.0 mg/dl or a decrease in the amount of urine output: <0.3 ml/kg/h for ≥24 h or anuria for ≥12 h) in patients hospitalized in ICU [24, 30].
In general, prior to the COVID‐19 pandemic, CRRT represented the modality of choice for hemodynamically unstable patients requiring RRT [31]. However, after the outbreak, it has become difficult to allocate hemodialysis machines to COVID‐19 patients with AKI due to infection control [32]. The current incidence of critically ill patients with COVID‐19 undergoing RRT in the form of CRRT ranges from 5% to 52% [33, 34].
In severe ARDS, the World Health Organization (WHO) guidance released a statement, in which they suggest referring patients with refractory hypoxemia despite lung‐protective ventilation to those settings with expertise in extracorporeal membrane oxygenation (ECMO) [35]; CRRT modalities can be used with ECMO [18].
While, CRRT dosing can be generally applied according to Kidney Disease: Improving Global Outcomes (KDIGO) guidelines [36], high‐volume hemofiltration at a dose of 6 L/h has been found as effective in improving the Sequential Organ Failure Assessment scores in patients with sepsis; suggesting a potential benefit of CRRT in patients with COVID‐19 and sepsis [37].
Hypercoagulability in patients with COVID‐19 is an important concern. Clotting of the CRRT membrane is a major limitation to care, as it leads to blood loss, inefficient dialysis, and exhausts limited resources (CRRT filters) [38]. Interruption of CRRT sessions as a result of circuit clotting can have a significant impact on the actual delivered dose [39]. Regional anticoagulation using unfractionated heparin or citrate, or systemic anticoagulation with unfractionated or low‐molecular‐weight heparin should be used during the procedure along with blood flow >120 ml/min to prevent premature clotting of the circuit [19, 40]. Based on low quality data, regional citrate anticoagulation appears to be less effective among COVID‐19 patients compared to systemic anticoagulation [41, 42].
The 25th Acute Disease Quality Initiative (ADQI) Workgroup stated that “the use of Continuous veno‐venous hemodialysis (CVVHD) or continuous veno‐venous hemodiafiltration (CVVHDF) modality and minimizing post‐filter replacement fluid in patients who are on CRRT will decrease the risk of circuit clotting.” This requires the use of CRRT at a delivered effluent flow rate of 20–25 ml/kg/h (prescribed dose of 25–30 ml/kg/h) [39].
3.2. Prolonged intermittent hemodialysis
PIHD, also termed as sustained low‐efficiency daily dialysis (SLEDD), describes an IHD but for an extended period (usually 6–18 h) and at least three times a week. The principle of PIHD includes combination of both convective (i.e., hemofiltration) and diffusion (i.e., HD) principles, based on the technique of solute removal [36]. PIHD is generally preferable over IHD as it allows higher ultrafiltration volume with lower risk of hemodynamic instability [43]. PIHD using high‐cutoff membranes might provide better clearance of middle molecules [44]. The feasible conduction of PIHD using the standard machines used for IHD or CRRT, and with use of standard tubing and hemodialyzers [44], together with the possible treatment of multiple patients at the same time by one nurse makes it a valuable therapeutic option in management of critically ill patients with COVID‐19 requiring dialysis. However, PIHD does not permit limited contact time between health care providers (HCPs) and COVID‐19 infected patients as compared to CRRT; it requires a designated HD nurse to be present for the whole duration of therapy time [27]. Anticoagulation in PIHD is essentially required in patients with COVID‐19, especially in terms of associated hypercoagulability. Treatment with either CRRT or PIHD is generally preferred in COVID‐19 infected patients with AKI over IHD.
3.3. Conventional IHD
The use of IHD in critically ill patients with COVID‐19 should be limited to circumstances of unavailable CRRT or PIHD; being suitable only for ICU patients with severe AKI who are hemodynamically stable. The higher flow rates needed to improve the efficacy of dialysis, requires close monitoring throughout the session to avoid hemodynamic instabilities. This requires an HCP to be in contact with the patient for the entire duration of session; making IHD the least ideal therapeutic modality for patients infected with COVID‐19 [45]. The use of IHD, under certain conditions, in COVID‐19 patients might follow the standard dosing and prescription in the general population. Further studies are needed to address the key points of IHD prescription for AKI patients in COVID‐19 pandemic.
3.4. Peritoneal dialysis
PD modality may be a feasible and safe alternative to HD in the unplanned setting [46]. PD has been long investigated in treatment of critically ill patients with AKI and has been well documented as a suitable treatment option in this setting [47, 48]. There are multiple advantages of using PD in COVID‐19 infected patients with severe AKI. First, there is no need for vascular access; which is also preferable to avoid in view of the coexisting coagulopathy [49]. Second, PD can be conducted with few resources, it does not require expensive machinery and supplies of CRRT or IHD [50]. Third, education of staff on performing PD is safe, feasible during a pandemic, and less technically challenging compared to other hemodialysis techniques [51]. Fourth, unlike hemodialysis, PD does not require a water system for dialysate supply, which represented a major obstacle in multiple ICU locations temporarily constructed in response to the COVID‐19 pandemic [50]. Fifth, PD catheter can be performed at the bedside [52, 53]. It has been revealed that an unplanned start to PD right after the insertion of PD catheter is an efficient and safe procedure [46, 54]. PD was more simply conducted in non‐critically ill patients outside the ICU. The implementation of an acute PD program for COVID‐19 patients with severe AKI requiring RRT was found to successfully mitigate the demand for RRT during the COVID‐19 pandemic's peak [50, 55]. Regarding the efficacy of PD in the context of severe AKI, PD has been found to be comparable to hemodialysis therapies in management of patients with AKI as regard metabolic control, mortality, and kidney function recovery rates [56]. A large body of evidence including previously published meta‐analyses also showed comparable clinical outcomes including mortality and renal recovery between patients who received RRT as PD or hemodialysis [57, 58]. Therefore, PD appears to be an effective and feasible method for treating critically ill COVID‐19 infected patients with AKI, especially in hospitals where more sophisticated technologies are not available.
4. EXTRACORPOREAL BLOOD PURIFICATION THERAPIES FOR CYTOKINES REMOVAL
The immune response to SARS‐CoV‐2 may lead to a pathophysiological condition of a “cytokine storm” which represents a severe CRS leading to multi‐organ failure [59].
The blood purification systems like therapeutic plasma exchange (TPE), adsorption, perfusion, blood/plasma filtration can remove inflammatory cytokines to an extent that it blocks CRS [1]. These therapies, while considered as “under scientific investigation,” still represent a therapeutic option for patients with severe CRS [59]. They include:
4.1. Continuous renal replacement therapy
CRRT plus hemoperfusion seem to be effective in CRS; it does not only support vital organ functions like heart, lungs, kidneys, and liver but also avoid organ damage by removing excess inflammatory mediators [60]. CRRT has been proven to effectively eliminate inflammatory cytokines such as C‐reactive protein (CRP), interleukin‐1 (IL‐1), and interleukin‐6 (IL‐6) [61]. High‐volume hemofiltration (>50 ml/kg/h) allows greater removal of hydrophilic middle molecular weight inflammatory molecules [62].
4.2. Hemoperfusion
Hemoperfusion is a blood purification technique which relies on the interaction between a sorbent and target molecules. Numerous kinds of hemoperfusion cartridges, are now available worldwide targeting cytokines or endotoxins, for example, oXiris and CytoSorb. The oXiris membrane is a heparin grafted membrane, principally designed for cytokine and endotoxin adsorption. The CytoSorb has been temporarily approved in April 2020 by the FDA for the management of CRS in patients with COVID‐19 [59]. Malard et al. showed a comparable efficacy between oXiris and CytoSorb in removal of endotoxin and inflammatory mediators and as compared to Toraymyxin [63]. Toraymyxin is polymyxin B‐immobilized fiber column, which is widely used in endotoxin removal and adsorption [64]. AN69 membrane is another highly negatively charged membrane with a hydrogel structure allowing cytokine adsorption [65].
4.3. Therapeutic plasma exchange
Plasmapheresis and plasma exchange have not been widely studied for this indication. There is no consensus on the routine use of TPE in patients with COVID‐19 related CRS considering the scarce evidence of small studies suggesting that TPE may decrease mortality in patients with sepsis [66, 67]. Lectin affinity plasmapheresis is another adjunctive potential extracorporeal therapy for coronavirus trapping based on the high affinity between the viral envelope and lectins so that it can reduce the viremia [68].
The extracorporeal blood purification techniques may represent promising future therapeutic tools for management of CRS, however, they can similarly remove essential drugs and antibiotics which should be also considered in patients with COVID‐19 [69].
5. SUMMARY OF INTERNATIONAL RECOMMENDATIONS AND GUIDELINES CONCERNING RRT IN CRITICALLY ILL PATIENTS WITH COVID‐19:
5.1. Wuhan expert statement
In general, front‐line experts in Wuhan, China fighting against the COVID‐19 pandemic developed an expert statement regarding the management of critically ill COVID‐19 patients [70]. They suggest the use of KDIGO criteria for the diagnosis of AKI in COVID‐19 patients [31]. They also suggested using CRRT for the critical cases complicated by severe AKI, or cytokine storm syndrome (expert opinion) [70].
5.2. American society of nephrology
The ASN released recommendations for care of hospitalized patients with COVID‐19 and kidney failure requiring RRT [45]; among which we highlight the following:
Indications to start RRT are the same as in other patients with AKI.
The preferred modality for RRT in critically ill patients is CRRT or prolonged intermittent renal replacement therapies (PIRRT).
CRRT machines are preferred over IHD in setting of biocontainment/isolation, as IHD requires 1:1 hemodialysis nursing support.
IHD can also be performed, if CRRT and PIRRT equipment are not available.
5.3. National institutes of health
The National Institutes of Health (NIH) stated the following recommendations regarding the management of severe AKI in COVID‐19 patients [71]:
For critically ill patients with COVID‐19 who have acute kidney injury (AKI) and who develop indications for renal replacement therapy (RRT), the COVID‐19 Treatment Guidelines Panel recommends CRRT, if available (BIII).
If CRRT is not available, the Panel recommends prolonged intermittent renal replacement therapy (PIRRT) rather than IHD (BIII).
The panel suggests using the same indications for RRT in patients with COVID‐19 as those used for other critically ill patients. Since RRT modalities have not been compared in COVID‐19 patients; the Panel's recommendations are motivated by the desire to minimize the risk of viral transmission to health care workers; therefore, CRRT is preferable as it does not require nursing staff to enter the patient's room to begin and end dialysis sessions. CRRT and PIRRT are both preferable to IHD because neither requires a dedicated hemodialysis nurse [71].
5.4. Consensus report of the 25th adqi workgroup 39
The panel of the 25th ADQI Workgroup indicated that there is no specific evidence to suggest that COVID‐19 AKI should be managed differently from other causes of AKI in critically ill patients. However, the following recommendations have been stated:
The use of ultrasound for insertion of vascular access and RRT dose delivery remain based on KDIGO AKI guidelines (evidence level: 1A).
Timing of RRT initiation, vascular access site and modality of acute RRT should be based on patient needs, local expertise and the availability of staff and equipment (not graded).
As COVID‐19 often induces a hypercoagulable state, if using CRRT, the panel suggested use of continuous veno‐venous hemodialysis or continuous veno‐venous hemodiafiltration to decrease filtration fraction and reduce the risk of circuit clotting (evidence level: 2C).
A large body of evidence is daily growing throughout the current pandemic. However, further research is needed to identify patients at risk of AKI and to guide management strategies. International collaborative research is needed to develop new approaches to management.
The precautions that should be adopted by HCPs conducting dialysis treatment to critically ill patients with COVID‐19:
Prevention of transmission of infection to HCPs conducting dialysis sessions is mandatory. Dialysis staff should follow standard contact instructions, including isolation gowns, gloves, N‐95 or higher‐level respirator or facemask masks, and eye protection (shields or goggles) [72].
It is perfect to limit the time of contact between dialysis staff, and COVID‐19 infected patients in the ICU. Nephrologists should contact ICU teams to rely on physical examination findings to minimize patient contact. The facility should provide telemedicine for monitoring the patient through a glass door, or camera. Ideally, in facilities, where ICU nurses are trained on the use of CRRT, hemodialysis nurses should bring and set up the CRRT machine outside the isolation ICU. Then, the ICU nurse will take the machine into the ICU and connect the patient in the room in order to minimize exposure of medical staff, and use of personal protective equipment (PPEs) [72].
Disinfection protocols for the dialysis setting should be implemented. This should include wipes for the dialysis machine, chair, dialysis station surfaces located within 6 feet of patients, and all non‐dedicated, non‐disposable medical equipment used for dialysis treatment including blood pressure cuffs and stethoscopes. Cleaning and disinfection is performed with a disinfectant made of sodium hypochlorite solution containing 1000 ppm of active chlorine. [73, 74].
Dialysis effluent from COVID‐19 patients can be disposed of per standard facility protocols. The genomic material of SARS‐CoV2 was detected in CRRT effluent in an earlier report [75]. Therefore, HCP caring for the patient in the ICU or disposing of effluent from the CRRT machine should wear recommended PPE.
It is recommended that the PD effluent should be mixed with 500 mg/L chlorine‐containing solution for 1 h and poured into the toilet [76].
Adequate rest for HCP is mandatory to concentrate fully at work to prevent accidental contamination. The mental and physical health of the medical staff is very important and should be monitored continuously for any disturbances [76].
6. CONCLUSION
Acute kidney injury in patients infected with coronavirus disease 2019 is associated with high mortality rates in intensive care unit. The treatment of acute kidney injury, indications of renal replacement therapy, and the modality of renal replacement therapy seem to be no different than in other settings. Continuous renal replacement therapy and prolonged intermittent hemodialysis therapies are preferred over intermittent hemodialysis in terms of minimizing viral transmission to health care providers. Peritoneal dialysis may represent a valuable therapeutic option in the current pandemic especially in hospitals where more sophisticated technologies are not available. Further studies concerning renal replacement therapy in acute kidney injury patients with coronavirus disease 2019 are highly warranted to address the targeted management protocols.
CONFLICT OF INTEREST
No conflict of interest has been declared by the authors.
ACKNOWLEDGMENTS
We acknowledge Professor Ahmed Halawa at Sheffield Teaching Hospitals NHS Foundation Trust for his online contribution with us regarding the topic COVID‐19 and kidney.
Shemies RS, Nagy E, Younis D, Sheashaa H. Renal replacement therapy for critically ill patients with COVID‐19‐associated acute kidney injury: A review of current knowledge. Ther Apher Dial. 2022;26:15–23. 10.1111/1744-9987.13723
REFERENCES
- 1. Chan JF‐W, To KK‐W, Tse H, Jin D‐Y, Yuen K‐Y. Interspecies transmission and emergence of novel viruses: lessons from bats and birds. Trends Microbiol. 2013;21:544–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Chen Y, Liu Q, Guo D. Emerging coronaviruses: genome structure, replication, and pathogenesis. J Med Virol. 2020;92:418–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Cascella M, Rajnik M, Cuomo A, Dulebohn SC, Di Napoli R. Features, evaluation and treatment coronavirus (COVID‐19). StatPearls Publishing; 2020. Statpearls [Internet]. https://www.statpearls.com/ [PubMed] [Google Scholar]
- 4. Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus‐infected pneumonia. N Engl J Med. 2020;382:1199–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. World Health Organization . Recommendations for national SARS‐CoV‐2 testing strategies and diagnostic capacities: interim guidance, 25 June 2021. World Health Organization; 2021.
- 6. Lee EY, Ng M‐Y, Khong P‐L. COVID‐19 pneumonia: what has CT taught us? Lancet Infect Dis. 2020;20:384–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wang C, Wang Z, Wang G, Lau JY‐N, Zhang K, Li W. COVID‐19 in early 2021: current status and looking forward. Signal Transduct Target Ther. 2021;6:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wang L, Li X, Chen H, Yan S, Li D, Li Y, et al. Coronavirus disease 19 infection does not result in acute kidney injury: an analysis of 116 hospitalized patients from Wuhan, China. Am J Nephrol. 2020;51:343–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. W‐j G, Ni Z‐Y, Hu Y, Liang W‐H, Ou C‐Q, He J‐X, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Diao B, Feng Z, Wang C, Wang H, Liu L, Wang C, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infection. MedRxiv. 2020;12(1):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, et al. Kidney disease is associated with in‐hospital death of patients with COVID‐19. Kidney Int. 2020;97:829–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hirsch JS, Ng JH, Ross DW, Sharma P, Shah HH, Barnett RL, et al. Acute kidney injury in patients hospitalized with COVID‐19. Kidney Int. 2020;98:209–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Gupta S, Coca SG, Chan L, Melamed ML, Brenner SK, Hayek SS, et al. AKI treated with renal replacement therapy in critically ill patients with COVID‐19. J Am Soc Nephrol. 2021;32:161–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Li Z, Wu M, Yao J, Guo J, Liao X, Song S, et al. Caution on kidney dysfunctions of COVID‐19 patients; 2020.
- 16. Pan X‐W, Da Xu HZ, Zhou W, Wang L‐H, Cui X‐G. Identification of a potential mechanism of acute kidney injury during the COVID‐19 outbreak: a study based on single‐cell transcriptome analysis. Intensive Care Med. 2020;1:1114–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Peng L, Liu J, Xu W, Luo Q, Deng K, Lin B, et al. 2019 novel coronavirus can be detected in urine, blood, anal swabs and oropharyngeal swabs samples. MedRxiv. 2020. 10.1101/2020.02.21.20026179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ronco C, Navalesi P, Vincent JL. Coronavirus epidemic: preparing for extracorporeal organ support in intensive care. Lancet Respir Med. 2020;8:240–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ronco C, Reis T. Kidney involvement in COVID‐19 and rationale for extracorporeal therapies. Nat Rev Nephrol. 2020;16(6):308–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Zhang B, Zhou X, Qiu Y, Feng F, Feng J, Jia Y, et al. Clinical characteristics of 82 death cases with COVID‐19. MedRxiv. 2020;15(7):e0235458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wilbers TJ, Koning MV. Renal replacement therapy in critically ill patients with COVID‐19: a retrospective study investigating mortality, renal recovery and filter lifetime. J Crit Care. 2020;60:103–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. World Health Organization . Protocol for assessment of potential risk factors for coronavirus disease 2019 (COVID‐19) among health workers in a health care setting, 23 March 2020. World Health Organization; 2020.
- 23. Thachil J, Tang N, Gando S, Falanga A, Cattaneo M, Levi M, et al. ISTH interim guidance on recognition and management of coagulopathy in COVID‐19. J Thromb Haemost. 2020;18:1023–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ronco C, Reis T, De Rosa S. Coronavirus epidemic and extracorporeal therapies in intensive care: si vis pacem para bellum. Blood Purif. 2020;49:255–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Garzotto F, Ostermann M, Martin‐Langerwerf D, Sanchez‐Sanchez M, Teng J, Robert R, et al. The dose response multicentre investigation on fluid assessment (DoReMIFA) in critically ill patients. Crit Care. 2016;20:196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Nalesso F, Garzotto F, Cattarin L, Gobbi L, Qassim L, Sgarabotto L, et al. A continuous renal replacement therapy protocol for patients with acute kidney injury in intensive care unit with COVID‐19. J Clin Med. 2020;9:1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Adapa S, Aeddula NR, Konala VM, Chenna A, Naramala S, Madhira BR, et al. COVID‐19 and renal failure: challenges in the delivery of renal replacement therapy. J Clin Med Res. 2020;12:276–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Ronco C, Reis T, Husain‐Syed F. Management of acute kidney injury in patients with COVID‐19. Lancet Respir Med. 2020;8:738–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. CARL DE, GROSSMAN C, BEHNKE M, SESSLER CN, GEHR TWB. Effect of timing of dialysis on mortality in critically ill, septic patients with acute renal failure. Hemodial Int. 2010;14:11–7. [DOI] [PubMed] [Google Scholar]
- 30. Ad‐hoc Working Group of ERBP , Fliser D, Laville M, Covic A, Fouque D, Vanholder R, et al. A European Renal Best Practice (ERBP) position statement on the kidney disease improving global outcomes (KDIGO) clinical practice guidelines on acute kidney injury: part 1: definitions, conservative management and contrast‐induced nephropathy. Nephrol Dial Transplant. 2012;27:4263–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kdigo A. Work group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2:1–138. [Google Scholar]
- 32. Goldfarb DS, Benstein JA, Zhdanova O, Hammer E, Block CA, Caplin NJ, et al. Impending shortages of kidney replacement therapy for COVID‐19 patients. Clin J Am Soc Nephrol. 2020;15:880–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus‐infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. World Health Organization . Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected, Interim guidance, 13 March 2020; 2020.
- 36. Edrees F, Li T, Vijayan A. Prolonged intermittent renal replacement therapy. Adv Chronic Kidney Dis. 2016;23:195–202. [DOI] [PubMed] [Google Scholar]
- 37. Ghani RA, Zainudin S, Ctkong N, Rahman AFA, Wafa SRWSH, Mohamad M, et al. Serum IL‐6 and IL‐1‐ra with sequential organ failure assessment scores in septic patients receiving high‐volume haemofiltration and continuous venovenous haemofiltration. Nephrol Ther. 2006;11:386–93. [DOI] [PubMed] [Google Scholar]
- 38. Endres P, Rosovsky R, Zhao S, Krinsky S, Percy S, Kamal O, et al. Filter clotting with continuous renal replacement therapy in COVID‐19. J Thromb Thrombolysis. 2021;51:966–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Nadim MK, Forni LG, Mehta RL, Connor MJ, Liu KD, Ostermann M, et al. COVID‐19‐associated acute kidney injury: consensus report of the 25th Acute Disease Quality Initiative (ADQI) Workgroup. Nat Rev Nephrol. 2020;16:747–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Zhou Y, Fu B, Zheng X, Wang D, Zhao C, Qi Y, et al. Pathogenic T‐cells and inflammatory monocytes incite inflammatory storms in severe COVID‐19 patients. Natl Sci Rev. 2020;7(6):998–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Helms J, Tacquard C, Severac F, Leonard‐Lorant I, Ohana M, Delabranche X, et al. High risk of thrombosis in patients with severe SARS‐CoV‐2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Shankaranarayanan D, Muthukumar T, Barbar T, Bhasin A, Gerardine S, Lamba P, et al. Anticoagulation strategies and filter life in COVID‐19 patients receiving continuous renal replacement therapy: a single‐center experience. Clin J Am Soc Nephrol. 2021;16:124–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kielstein JT, Kretschmer U, Ernst T, Hafer C, Bahr MJ, Haller H, et al. Efficacy and cardiovascular tolerability of extended dialysis in critically ill patients: a randomized controlled study. Am J Kidney Dis. 2004;43:342–9. [DOI] [PubMed] [Google Scholar]
- 44. Schmidt JJ, Hafer C, Clajus C, Hadem J, Beutel G, Schmidt BM, et al. New high‐cutoff dialyzer allows improved middle molecule clearance without an increase in albumin loss: a clinical crossover comparison in extended dialysis. Blood Purif. 2012;34:246–52. [DOI] [PubMed] [Google Scholar]
- 45. American Society of Nephrology . Recommendations on the care of hospitalized patients with COVID‐19 and kidney failure requiring renal replacement therapy; 2020.
- 46. Dias DB, Banin V, Mendes ML, Barretti P, Ponce D. Peritoneal dialysis as an option for unplanned initiation of chronic dialysis. Hemodial Int. 2016;20:631–3. [DOI] [PubMed] [Google Scholar]
- 47. Al‐Hwiesh A, Abdul‐Rahman I, Finkelstein F, Divino‐Filho J, Qutub H, Al‐Audah N, et al. Acute kidney injury in critically ill patients: a prospective randomized study of tidal peritoneal dialysis versus continuous renal replacement therapy. Ther Apher Dial. 2018;22:371–9. [DOI] [PubMed] [Google Scholar]
- 48. Dias DB, Mendes ML, Caramori JT, Falbo dos Reis P, Ponce D. Urgent‐start dialysis: comparison of complications and outcomes between peritoneal dialysis and haemodialysis. Perit Dial Int. 2020;41(2):244–52. [DOI] [PubMed] [Google Scholar]
- 49. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Sourial MY, Sourial MH, Dalsan R, Graham J, Ross M, Chen W, et al. Urgent peritoneal dialysis in patients with COVID‐19 and acute kidney injury: a single‐center experience in a time of crisis in the United States. Am J Kidney Dis. 2020;76:401–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. El Shamy O, Sharma S, Winston J, Uribarri J. Peritoneal dialysis during the coronavirus 2019 (COVID‐19) pandemic: acute inpatient and maintenance outpatient experiences. Kidney Med. 2020;2(4):377–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Nagatomo M, Yamada H, Shinozuka K, Shimoto M, Yunoki T, Ohtsuru S. Peritoneal dialysis for COVID‐19‐associated acute kidney injury. Crit Care. 2020;24:1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Cruz MV, Bellorin O, Srivatana V, Afaneh C. Safety and efficacy of bedside peritoneal dialysis catheter placement in the COVID‐19 era: initial experience at a New York city hospital. World J Surg. 2004;44(10):3584–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Povlsen JV. Unplanned start on assisted peritoneal dialysis. Peritoneal dialysis—from basic concepts to clinical excellence. Volume 163, Basel: Karger Publishers; 2009. p. 261–3. [DOI] [PubMed] [Google Scholar]
- 55. El Shamy O, Patel N, Abdelbaset MH, Chenet L, Tokita J, Lookstein R, et al. Acute start peritoneal dialysis during the COVID‐19 pandemic: outcomes and experiences. J Am Soc Nephrol. 2020;31(8):1680–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Gabriel DP, Caramori J, Martim L, Barretti P, Balbi AL. High volume peritoneal dialysis vs daily hemodialysis: a randomized, controlled trial in patients with acute kidney injury. Kidney Int. 2008;73:S87–93. [DOI] [PubMed] [Google Scholar]
- 57. Chionh CY, Soni SS, Finkelstein FO, Ronco C, Cruz DN. Use of peritoneal dialysis in AKI: a systematic review. Clin J Am Soc Nephrol. 2013;8:1649–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Liu L, Zhang L, Liu GJ, Fu P. Peritoneal dialysis for acute kidney injury. Cochrane Database Syst Rev. 2017;12:CD011457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Ronco C, Bagshaw SM, Bellomo R, Clark WR, Husain‐Syed F, Kellum JA, et al. Extracorporeal blood purification and organ support in the critically ill patient during COVID‐19 pandemic: expert review and recommendation. Blood Purif. 2021;50:17–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. MacLaren G, Fisher D, Brodie D. Preparing for the most critically ill patients with COVID‐19: the potential role of extracorporeal membrane oxygenation. JAMA. 2020;323:1245–6. [DOI] [PubMed] [Google Scholar]
- 61. Venkataraman R, Subramanian S, Kellum JA. Clinical review: extracorporeal blood purification in severe sepsis. Crit Care. 2003;7:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Kellum JA, Johnson JP, Kramer D, Palevsky P, Brady JJ, Pinsky MR. Diffusive vs. convective therapy: effects on mediators of inflammation in patients with severe systemic inflammatory response syndrome. Crit Care Med. 1998;26:1995–2000. [DOI] [PubMed] [Google Scholar]
- 63. Malard B, Lambert C, Kellum JA. In vitro comparison of the adsorption of inflammatory mediators by blood purification devices. Intensive Care Med Exp. 2018;6:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Chang T, Tu Y‐K, Lee C‐T, Chao A, Huang C‐H, Wang M‐J, et al. Effects of polymyxin B hemoperfusion on mortality in patients with severe sepsis and septic shock: a systemic review, meta‐analysis update, and disease severity subgroup meta‐analysis. Crit Care Med. 2017;45:e858–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Kobashi S, Maruhashi T, Nakamura T, Hatabayashi E, Kon A. The 28‐day survival rates of two cytokine‐adsorbing hemofilters for continuous renal replacement therapy: a single‐center retrospective comparative study. Acute Med Surg. 2019;6:60–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Busund R, Koukline V, Utrobin U, Nedashkovsky E. Plasmapheresis in severe sepsis and septic shock: a prospective, randomised, controlled trial. Intensive Care Med. 2002;28:1434–9. [DOI] [PubMed] [Google Scholar]
- 67. Temel S, Sungur M. Blood purification treatments in COVID‐19. J Crit Intensive Care. 2020;11(Suppl):32. [Google Scholar]
- 68. Koch B, Schult‐Dietrich P, Büttner S, Dilmaghani B, Lohmann D, Baer PC, et al. Lectin affinity plasmapheresis for middle east respiratory syndrome‐coronavirus and Marburg virus glycoprotein elimination. Blood Purif. 2018;46:126–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Reiter K, Bordoni V, Dall'Olio G, Ricatti MG, Soli M, Ruperti S, et al. In vitro removal of therapeutic drugs with a novel adsorbent system. Blood Purif. 2002;20:380–8. [DOI] [PubMed] [Google Scholar]
- 70. Shang Y, Pan C, Yang XH, Zhong M, Shang XL, Wu ZX, et al. Management of critically ill patients with COVID‐19 in ICU: statement from front‐line intensive care experts in Wuhan, China. Ann Intensive Care. 2020;10(1):1–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. National Institutes of Health . COVID‐19 treatment guidelines panel. Coronavirus disease 2019 (COVID‐19) treatment guidelines; 2020. [cited 2020 29.05]. [PubMed]
- 72. Sick IYA, Extra PWN. Interim additional guidance for infection prevention and control recommendations for patients with suspected or confirmed COVID‐19 in outpatient hemodialysis facilities.
- 73. Arenas MD, Villar J, González C, Cao H, Collado S, Crespo M, et al. Management of the SARS‐CoV‐2 (Covid 19) coronavirus epidemic in hemodialysis units. Nefrología (English Edition). 2020;40:258–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Shimada N, Shimada H, Itaya Y, Tomino Y. Novel coronavirus disease in patients with end‐stage kidney disease. Ther Apher Dial. 2020;1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Katagiri D, Ishikane M, Ogawa T, Kinoshita N, Katano H, Suzuki T, et al. Continuous renal replacement therapy for a patient with severe COVID‐19. Blood Purif. 2021;50:129–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Shen Q, Wang M, Che R, Li Q, Zhou J, Wang F, et al. Consensus recommendations for the care of children receiving chronic dialysis in association with the COVID‐19 epidemic. Pediatr Nephrol. 2020;35(7):1351–7. [DOI] [PMC free article] [PubMed] [Google Scholar]


