Summary
Coronaviruses are constantly circulating in humans, causing common colds and mild respiratory infections. In contrast, infection with the novel severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2), responsible for coronavirus disease‐2019 (COVID‐19), can cause additional severe complications, particularly in patients with obesity and associated metabolic disturbances. Obesity is a principal causative factor in the development of the metabolic syndrome; a series of physiological, biochemical, clinical, and metabolic factors that increase the risk of obesity‐associated diseases. “Metabolically unhealthy” obesity is, in addition to metabolic disturbances, also associated with immunological disturbances. As such, patients with obesity are more prone to develop serious complications from infections, including those from SARS‐CoV‐2. In this review, we first describe how obesity and related metabolic disturbances increase the risk of SARS‐CoV‐2 infection. Then, mechanisms contributing to COVID‐19 complications and poor prognosis in these patients are discussed. Finally, we discuss how obesity potentially reduces long‐term COVID‐19 vaccination efficacy. Despite encouraging COVID‐19 vaccination results in patients with obesity and related metabolic disturbances in the short‐term, it is becoming increasingly evident that long‐term COVID‐19 vaccination efficacy should be closely monitored in this vulnerable group.
Keywords: COVID‐19, metabolic syndrome, obesity, severe acute respiratory syndrome coronavirus 2

Abbreviations
- ACE2
angiotensin‐converting enzyme 2
- AID
activation‐induced cytidine deaminase
- ALP
alkaline phosphatase
- ALT
alanine aminotransferase
- AMPK
AMP‐activated protein kinase
- Ang II
angiotensin II
- Ang (1‐7)
angiotensin‐(1‐7)
- ASC
apoptosis‐associated speck‐like protein containing a CARD
- AST
aspartate aminotransferase
- AT1R
angiotensin 1 receptor
- AT2R
angiotensin 2 receptor
- Bm cells
memory B cells
- COPD
chronic obstructive pulmonary disease
- COVID‐19
coronavirus disease‐2019
- DIO
diet induced obesity
- ECs
endothelium cells
- EMA
European Medicine Agency
- FDA
US Food and Drug Administration
- HAI
hemagglutination inhibition
- HDL
high‐density lipoproteins
- HIF‐1α
hypoxia‐inducible factor‐1α
- ICU
intensive care unit
- IFN
interferons
- Ig
immunoglobulin
- IL‐6
interleukin 6
- MCP1
monocyte chemoattractant protein 1
- MERS‐CoV
Middle East respiratory syndrome coronavirus
- MetS
metabolic syndrome
- MGRA
MAS1 proto‐oncogene G protein‐coupled receptor
- mTORC
mammalian target of rapamycin complex 1
- mtROS
mitochondrial reactive oxygen species
- NAFLD
non‐alcohol fatty liver disease
- NASH
non‐alcoholic steatohepatitis
- NF‐kB
nuclear factor kappa‐light‐chain‐enhancer of activated B cells
- NLRP3
NLR family pyrin domain containing 3
- oxLDL
oxidized low‐density lipoproteins
- PAI‐1
plasminogen activator inhibitor‐1
- PD‐1
programmed cell death protein 1
- PD‐L1
programmed death‐ligand 1
- PERK
PKR‐like endoplasmic reticulum kinase
- PKR
protein kinase R
- R
reproductive
- RAAS
renin‐angiotensin aldosterone system
- SARS‐CoV
syndrome coronavirus
- SARS‐CoV‐2
severe acute respiratory syndrome coronavirus 2
- Tem cells
effector memory T cells
- TMPRSS2
Transmembrane Protease Serine 2
- TNFα
tumor necrosis factor α
- VOC
Variant of Concern 202012/01
1. INTRODUCTION
Coronavirus disease‐2019 (COVID‐19) caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) has taken the world by storm. Detected in more than 200 countries, with the number of confirmed cases already exceeding 114 million and deaths tallying at 2,5 million worldwide, it is one of the worst disasters of modern times. 1
Viruses are grouped based on the type of genetic material they carry. DNA viruses have integrated large host DNA sequences in their genome throughout their evolution, which consequently allows for exploitation of host cell metabolism in order to promote viral replication. In contrast, RNA viruses such as respiratory syncytial virus, (para)influenza virus, metapneumovirus, rhinovirus, and coronavirus have smaller genomes consisting of RNA sequences encoding only a few proteins which are unfamiliar to the host. Despite this, RNA viruses can still manipulate host cells through other mechanisms to induce viral replication. 2 Coronaviruses are responsible for 10%–30% of the common colds in humans. 3 For example, the HCoV‐229E and HCoV‐OC43 viruses, regularly flaring up in temperate climate countries during winter or early spring, cause respiratory infections with mild clinical symptoms such as nasal obstruction, rhinorrhea, sneezing, sore throat, and cough. 4 , 5 In contrast, other coronaviruses such as severe acute respiratory syndrome coronavirus (SARS‐CoV) and Middle East respiratory syndrome coronavirus (MERS‐CoV) frequently cause serious respiratory illness. 6 , 7 The clinical course of SARS and MERS is remarkably similar, although subtle differences do exist such as the incidence of acute respiratory distress syndrome and the percentage of patients with comorbidities being higher in MERS then in SARS. In line, the mortality rate of MERS (36%) is higher compared with SARS (10%). 8 Both, SARS‐CoV and MERS‐CoV are zoonotic coronaviruses that cross from animals to humans. Similarly, the novel highly pathogenic coronavirus, named SARS‐CoV‐2, is also zoonotic. 9 , 10 Sequencing data show a high resemblance between SARS‐CoV and SARS‐CoV‐2. 11 Yet the higher reproductive (R) number of SARS‐CoV‐2 (2.87–3.44) 12 compared with SARS‐CoV (1.7–1.9) 13 indicates the higher pandemic potential of SARS‐CoV‐2, as also reflected in the rapid global spread of SARS‐CoV‐2 numbering in millions, whereas the absolute number of SARS‐CoV cases was only 8096. 1 , 12 , 13 , 14 Alarmingly, more contagions mutated SARS‐CoV‐2 variants have been detected in several countries. For example, the novel SARS‐CoV‐2 lineage designated as Variant of Concern 202012/01 (VOC), originally detected in England, has a substantial transmission advantage compared with the non‐VOC lineage with an estimated difference in R‐number ranging between 0.4 and 0.7 in a period of high levels of social distancing. 15 In the earliest stages of the current COVID‐19 pandemic, it was thought that mostly the elderly, aged >60, were at risk of poor prognosis upon SARS‐CoV‐2 infection. However, it has become apparent that also patients aged <60 years are at risk of developing severe illness, particularly when they suffer from obesity. 16
Notably, 10%–25% of the individuals with obesity are not affected by metabolic disturbances, 17 , 18 , 19 whereas in the other individuals the abnormal or excessive fat accumulation presents a risk to health. Obesity is a principal causative factor in the development of the metabolic syndrome (MetS). MetS is defined as a series of physiological, biochemical, clinical, and metabolic factors that increase the risk of obesity‐associated diseases. These risk factors include central obesity with a focus on waist circumference, insulin resistance, hypertension, increased plasma triglycerides and reduced plasma high‐density lipoproteins (HDL). 20 Though other abnormalities have also been associated with MetS, the presence of three of the five previously mentioned components is considered sufficient for a diagnosis of this syndrome, especially when associated with visceral obesity. 21 Obesity, accompanied by the characteristics of MetS, constitutes the greatest threat to global health, affecting 20%–25% of the adult population. 21 These patients are at increased risk of developing cardiovascular diseases, type 2 diabetes, and non‐alcoholic steatohepatitis (NASH). 22 “Metabolically unhealthy” obesity is, in addition to metabolic disturbances, also associated with immunological disturbances, such as increased systemic leukocyte numbers and increased pro‐inflammatory plasma cytokine levels. This chronic low‐grade inflammatory state disrupts the immune response in patients with obesity. In that context, substantial evidence indicates a link between obesity and reduced host defense. For example, obesity increases the susceptibility for postoperative and nosocomial infections, and individuals with obesity are more prone to develop serious complications from common infections. 23 , 24 Moreover, during influenza pandemics (e.g., H1N1), individuals with obesity were overrepresented at the intensive care unit (ICU) and needed longer duration of mechanical ventilation compared with individuals with a healthy weight. 25 , 26 Diabetes, a frequently occurring obesity‐related metabolic comorbidity, actually tripled the risk of hospitalization after H1N1 infection and even quadrupled the risk of ICU admission once hospitalized. 27 Additionally, obesity is associated with a reduced immunogenicity in response to vaccination for hepatitis B, tetanus, and influenza. 28 , 29 In line with these data, it is not surprising that patients suffering from obesity and related metabolic disturbances have poorer prognoses upon infection with the highly pathogenic coronaviruses SARS‐CoV, 30 MERS‐CoV, 30 and SARS‐COV‐2. 31
In the current COVID‐19 pandemic, individuals with obesity have an increased risk of testing positive for SARS‐CoV‐2. 32 Additionally, a substantial amount of patients hospitalized with COVID‐19 suffer from comorbidities closely associated with obesity, such as diabetes and cardiovascular disorders, 31 presumably leading to the higher ICU admission rate and mortality rate of these patients with COVID‐19. Because most COVID‐19 cohort studies do not report whether patients with obesity also have metabolic disturbances related to MetS, although very likely, it is currently difficult to argue if and how MetS exacerbates the severity of COVID‐19 beyond the role of obesity. Proposed explanations for the strong association between obesity and severe COVID‐19 include pulmonary dysfunction, 33 hypertension, 34 upregulated angiotensin‐converting enzyme 2 (ACE2) expression, 35 , 36 hyperglycemia, 37 dyslipidemia, 38 insulin resistance, 39 , 40 chronic low‐grade inflammation, 33 a pre‐existing pro‐thrombotic environment, 41 and impairment of endothelial and gut barrier function. 42 , 43 Although in‐depth analysis of these mechanisms is beyond the scope of the current review, we refer to previously published mechanistic insights and other specific reviews when applicable. In this review, we aim to explain the molecular link between obesity and related metabolic disturbances and increased SARS‐CoV‐2 infection risk. We will focus specifically on upregulated TMPRSS2 and ACE2 expression (cell membrane proteins facilitating SARS‐CoV‐2 viral entry into host cells), hyperglycemia, and weakened immune surveillance. We then describe how several of the above‐mentioned mechanisms contribute to the poorer prognosis of COVID‐19 in this patient population. Lastly, the risk of reduced long‐term COVID‐19 vaccination efficacy in patients with obesity is discussed by focusing on potentially reduced memory T cell and memory B cell responses upon re‐infection.
2. OBESITY AND RELATED METABOLIC DISTURBANCES INCREASE THE RISK OF SARS‐COV‐2 INFECTION
Due to the variety of clinical symptoms associated with obesity, ranging from overexpression of proteins that facilitate viral entry into cells to hyperglycemia and hyperinsulinemia, these patients are at increased risk of acquiring infections, including SARS‐CoV‐2. In line, meta‐analysis data showed an association between individuals with obesity and the risk of testing positive for COVID‐19. 32 Here, several factors that contribute to the increased susceptibility of patients with obesity and related metabolic disturbances to get infected with SARS‐CoV‐2 are described.
2.1. ACE2 and TMPRSS2 overexpression
After binding to host receptors, viruses can use the endosomal or the non‐endosomal pathway to enter host cells. Viral cell entry through the endocytic route is usually by transport in clathrin‐coated vesicles or pits, whereas the non‐endocytic route of entry involves directly crossing the plasma membrane at neutral pH. 44 Generally, the endosomal pathway is advantageous for viruses: the endocytic vesicle supports intracellular transport of the virus, the endocytic vesicle contains specific proteases that provide necessary proteolytic activation of certain viruses, and upon endocytosis, no viral antigens remain on the cell membrane resulting in delayed detection by the immune system. 45 Depending on the host cell type, SARS‐CoV‐2 can use the endosomal and non‐endosomal pathways to establish viral cell entry. 46 In the non‐endosomal pathway of SARS‐CoV‐2, the spike protein (S) of virus binds to host receptor, angiotensin‐converting enzyme 2 (ACE2), a membrane bound aminopeptidase highly, but not exclusively, expressed in the lungs and cardiovascular system. 47 Upon binding, the host protease, transmembrane protease serine 2 (TMPRSS2), expressed on the cell membrane, cleaves S into the S1 and S2 subunits to activate S to facilitate virus‐host cell fusion. 48 In the endosomal pathway, SARS‐CoV‐2 also binds ACE2, but instead of activating TMPRSS2, SARS‐CoV‐2 is internalized via clathrin‐mediated endocytosis. 49 Thus, in both pathways, ACE2 plays an essential role to induce viral cell invasion. In normal physiological situation, ACE2 functions as key regulatory enzyme in the renin‐angiotensin aldosterone system (RAAS), a hormonal system regulating blood pressure and water balance. In the RAAS system, angiotensin II (Ang II) binds to the angiotensin 1 or 2 receptor (AT1R or AT2R). Binding of Ang II to AT1R induces a proinflammatory vasoconstrictive effect, whereas binding of Ang II to AT2R counteracts this effect by promoting vasodilatation and inhibiting inflammatory events. 50 ACE2 converts Ang II into its metabolite angiotensin‐(1‐7) (Ang (1‐7)). Following its conversion from Ang II, Ang (1‐7) then acts on the MAS1 proto‐oncogene G protein‐coupled receptor (MGRA) pathway, resulting in an anti‐inflammatory vasodilative response. 51 , 52 In the context of obesity and its related metabolic disturbances, elevated insulin levels, as observed in patient with insulin resistance and MetS, have been speculated to upregulate TMPRSS2 expression via phosphoinositide 3‐kinase/protein kinase B/androgen receptor signaling. 53 In addition to TMPRSS2 overexpression, two ex vivo studies comparing heart samples from patients with (n = 23 and n = 40, respectively) and without (n = 9 and n = 15, respectively) heart failure found that ACE2 was upregulated in the heart failure samples. 54 , 55 Moreover, a large genome‐wide association study (74,124 type 2 diabetes cases and 824,006 controls) reported a causal link between type 2 diabetes and elevated ACE2 expression in the lung. 56 A comprehensive description of altered ACE2 expression in comorbidities associated with severe COVID‐19 has recently been published. 57 Strikingly, in patients with obesity and related metabolic disturbances, ACE2 expression can be upregulated not only due to the disease physiology but also due to the use of medication to control disease. For example, angiotensin receptor blockers, used to reduce blood pressure, can increase ACE2 expression 35 , 36 , 58 and starting insulin therapy within 1 year of a diabetes diagnosis has been reported to be causally associated with increased ACE2 expression. 56 Taken together, due to upregulated ACE2 54 , 55 , 56 , 59 and TMPRSS2 53 expression in patients with obesity and related metabolic disturbances, SARS‐CoV‐2 cell invasion is facilitated, contributing to an increased risk to get infected with SARS‐CoV‐2 (Figure 1).
FIGURE 1.
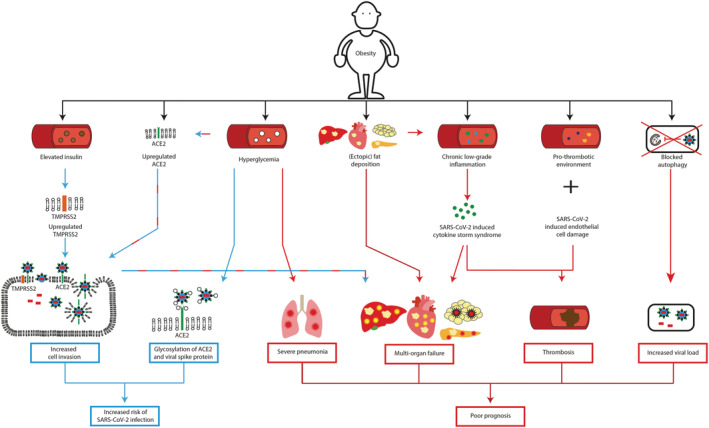
Obesity increases the risk of SARS‐CoV‐2 infection and leads to poorer prognosis. Blue arrows: Mechanisms contributing to increased SARS‐CoV‐2 infection risk in patients with obesity. Red arrows: Mechanisms contributing to poorer prognosis of COVID‐19 patients with obesity. In patients with obesity, elevated insulin levels upregulates TMPRSS2 expression, which in combination with overexpression of ACE2 increases SARS‐CoV‐2 cell invasion. Hyperglycemia increases ACE2 expression and potentially changes the glycosylation of ACE2 and the viral spike protein S, altering binding of S to ACE2. Together, these factors contribute to an increased risk of SARS‐CoV‐2 infection in patients with obesity. Hyperglycemia compromises the immune response in the lungs and may contribute to lung inflammation. Overexpression of ACE2 increases SARS‐CoV‐2 viral cell entry. Intracellularly, the virus induces cellular damage in adipose, heart, pancreas, and liver cells. In obesity, the organ function of the adipose tissue, heart, pancreas and liver is already affected due to (ectopic) fat deposition. SARS‐CoV‐2 can aggravate organ damage and thus contribute to multi‐organ failure. Chronic low‐grade inflammation, as observed in obesity, potentially facilitates the cytokine storm induced by SARS‐CoV‐2 consequently, extremely high levels of pro‐inflammatory cytokines, enhance cellular damage and eventually induce multi‐organ failure. Obesity is associated by a pro‐thrombotic environment. SARS‐CoV‐2 induces endothelial cell damage. In combination with the cytokine storm induced by SARS‐CoV‐2 this increases the risk of thrombosis in COVID‐19 patients with obesity. SARS‐CoV‐2 might block autophagy, resulting in decreased viral clearance. The disrupted autophagy process in obesity potentially further reduces SARS‐CoV‐2 clearance and by extension increases viral load. Together, these mechanisms lead to poorer prognosis of patients with obesity infected with SARS‐CoV‐2. ACE2, Angiotensin‐converting enzyme 2; TMPRSS2, transmembrane protease serine 2
2.2. Hyperglycemia
Hyperglycemia, a hallmark of MetS and type 2 diabetes (both strongly associated with obesity), increases the rate of infections. Despite strict glycemic control, patients with diabetes still have a 1.7‐fold probability of developing an ICU‐acquired bloodstream infection compared with subjects without diabetes. 60 Hyperglycemia potentially also contributes to an increased risk of SARS‐CoV‐2 infection.
Upon SARS‐CoV‐2 infection, lung epithelial cells start producing chemokines to recruit macrophages into the lung. 61 When these macrophages also become virally infected with SARS‐CoV‐2, mitochondrial reactive oxygen species (mtROS) production is increased inducing stabilization of hypoxia‐inducible factor‐1α (HIF‐1α). Following, HIF‐1α promotes metabolic reprogramming of these macrophages driving them toward glycolysis. The metabolically reprogrammed glycolytic SARS‐CoV‐2 infected macrophages acquire a pro‐inflammatory phenotype resulting in an augmented secretion of interferons (IFN), leading to enhanced ACE2 expression in those lung macrophages. 62 It is possible that due to elevated glucose levels, as frequently observed in obesity and MetS, the glycolysis‐mediated ACE2 upregulation in the lung macrophages of these patients subsequently increases SARS‐CoV‐2 cell invasion, contributing to an increased risk to get infected with SARS‐CoV‐2 (Figure 1).
In physiological conditions, glucose is not present in airway secretion. However, in a cohort study of patients admitted to a general ICU and expected to require intubation for more than 24 h (n = 60), a relationship appeared to be present between a high blood glucose concentration and the appearance of glucose in airway secretion. However, no correction was performed for confounding factors such as waist circumference or BMI. 63 It has been suggested that glucose in airway surface liquid possibly contributes to airway inflammation by impairing host immunity through glycosylation of innate or acquired immune proteins. 63 In the context of SARS‐CoV‐2 infection, it has also been proposed that hyperglycemia potentially changes the glycosylation of ACE2 and the viral spike protein S, altering binding of S to ACE2 and the degree of the immune response to the virus, 64 thereby contributing to increased susceptibility of patients with MetS and type 2 diabetes (both strongly associated with obesity) to get infected with SARS‐CoV‐2 (Figure 1).
2.3. Weakened immune surveillance
Immunological surveillance, a monitoring process of the immune system to detect and destroy virally infected and neoplastically transformed cells in the body, is severely weakened in patients with obesity and related metabolic disturbances (see Andersen et al. for more mechanistic details). 28 Obesity has been shown to be linked to a compromised B cell functioning, directly contributing to a weakened immune surveillance. For example, diet induced obesity (DIO) in mice lowers bone marrow B cell frequency, and in a murine infection study with influenza A viral infection, mice fed a western diet had suppressed antibody titers compared with mice receiving a control diet. 65 In line, a (small) human cohort study comparing four groups (young lean n = 8; young obese n = 6; elderly lean n = 8; and elderly obese n = 4) indicated that obesity is associated with reduced antibody titers after influenza vaccination, in both young and elderly patients. 66 In addition to a reduced humoral immune response, available evidence also indicates that impaired cellular immunity contributes to weakened immune surveillance in obesity. In that context, progressive obesity in mice is linked to inhibition of thymopoiesis, consequently restricting T cell repertoire diversity. This observation correlates with data in middle‐aged humans, which also indicated that obesity compromises thymic output. 23 Moreover, a human cohort study (comparing individuals with healthy weight n = 137, overweight n = 154, and obesity n = 164) demonstrated that individuals with obesity have decreased effector memory T (Tem) cell activation after ex vivo vaccine strain virus challenge compared with healthy weight individuals. 67 Remarkably, initial clinical trials show that COVID‐19 vaccines are effectively protecting patients with obesity and patients with obesity‐related metabolic disturbances (e.g., chronic pulmonary diseases, diabetes, hypertension, liver disease, and cardiovascular disease) in the short‐term. 68 , 69 , 70 , 71 These most recent data indicate that SARS‐CoV‐2 is well detected by immune surveillance mechanisms in patients with obesity and related metabolic disturbances. As such, these findings are suggesting that a potentially reduced long‐term COVID‐19 vaccination efficacy in obesity (cfr. infra) would not be due to defects in immune surveillance mechanisms in these vulnerable patients.
3. COVID‐19 LEADS TO POORER PROGNOSIS IN PATIENTS WITH OBESITY AND RELATED METABOLIC DISTURBANCES
In the current COVID‐19 pandemic, patients suffering from diabetes, hypertension, and cardiovascular disorders are overly represented in the hospital. 31 , 72 This observation is indicative of these patients being a vulnerable population with increased risk of complications and poorer prognosis upon SARS‐CoV‐2 infection.
3.1. Pneumonia
The most frequent serious complication of COVID‐19 is pneumonia. Upon hospital admission (almost) all patients suffer from unilateral or bilateral pneumonia. 9 , 73 Upon SARS‐CoV‐2 infection, lung macrophages undergo a HIF‐1α mediated metabolic reprogramming toward glycolysis, inducing a pro‐inflammatory phenotype. 62 These glycolytic macrophages secrete pro‐inflammatory cytokines, such as TNFα and IL‐6, which have been suggested to contribute to epithelial cell death in the lungs of patients with COVID‐19. 62 It is possible that due to elevated glucose levels, as observed in obesity, MetS, and type 2 diabetes, the glycolytic macrophages secrete enhanced levels of pro‐inflammatory cytokines. As such, high blood glucose levels, may increase lung inflammation, predisposing patients with obesity‐related metabolic disturbances to become more susceptible to develop severe pneumonia upon SARS‐CoV‐2 infection, which potentially partially explains the poorer prognosis of these patients upon SARS‐CoV‐2 infection (Figure 1).
3.2. Multi‐organ failure
Apart from the lungs, SARS‐CoV‐2 also affects other organs. COVID‐19 has been shown to be associated with gastrointestinal injury, reduced kidney function, and liver injury (see Renu et al. for more details). 74 Here, we focus specifically on adipose tissue, heart, liver, and pancreas dysfunction in COVID‐19 patients with obesity and related metabolic disturbances.
3.2.1. Multi‐organ failure (adipose tissue)
Central or visceral obesity is defined as increased adipose tissue surrounding the intra‐abdominal organs. Ironically, despite the increased health risk associated with visceral obesity (e.g., development of cardiovascular diseases, insulin resistance and diabetes), patients with obesity may have prognostic benefits in some diseases such as heart failure, 75 chronic obstructive pulmonary disease (COPD), 76 and pneumonia, 77 being expressed as the “obesity paradox.” However, recent meta‐analysis data reveal no evidence for an “obesity paradox” in the context of COVID‐19. 78 As extensively reviewed by Kruglikov et al. 79 and Goossens et al., 80 one of the main reasons for poor prognosis of COVID‐19 patients with visceral obesity is an increased number of adipocytes in combination with upregulated ACE2 expression. This turns the visceral adipose tissue into a viral reservoir for SARS‐CoV‐2, eventually leading to visceral adipose tissue dysfunction and systemic inflammation, 79 , 81 presumably contributing to the poorer prognosis COVID‐19 patients with obesity (Figure 1).
3.2.2. Multi‐organ failure (heart)
ACE2, the receptor through which SARS‐CoV‐2 enters host cells, is upregulated in the heart of patients with cardiovascular disorders 54 , 55 (frequently observed in patients with obesity). This makes the heart a potential target organ for SARS‐CoV‐2 infection. Tissue distribution analysis of ACE2 in human donor hearts through single‐cell RNA sequencing revealed that ACE2 is highly expressed in pericytes, whereas cardiomyocytes only have a low expression. Furthermore, cell–cell interaction analysis between pericytes and other cell types indicated that neuron‐like cells and endothelium cells (ECs) have the closest crosstalk interaction with pericytes. Therefore, it is suggested that SARS‐CoV‐2 attacks pericytes and causes capillary ECs dysfunction, leading to micro‐circulation disorders. 55 Moreover, cardiac pericyte dysfunction may increase the propensity for atrial fibrillation via increased myocardial inflammation, fibrosis, increased tissue edema, and interstitial hydrostatic pressure. 82
Because in visceral adipocytes of patients with central obesity ACE2 is upregulated 79 , 81 and meta‐analysis data showed a strong positive correlation between epicardial adipose tissue thickness and MetS, 83 ACE2 expression in epicardial fat cells from patients with MetS has also been explored. Data are limited, but one study analyzing epicardial biopsies from patients who underwent open‐heart surgery demonstrated that, similar to visceral adipocytes, epicardial fat cells highly express ACE2. Relevantly, highest ACE2 expression was found in biopsies from patient suffering from obesity and diabetes. 84 Therefore, the thickened epicardial adipose tissue, observed in patients with obesity‐related insulin resistance, 85 diabetes, 84 and MetS 83 might be considered another viral reservoir for SARS‐CoV‐2. Moreover, SARS‐CoV‐2 internalizes ACE2 upon binding, resulting in loss of cell surface ACE2. 43 Because loss of ACE2 expression in epicardial adipose tissue has been associated with increased epicardial adipose tissue inflammation, 86 a mechanistic link with COVID‐19 related myocarditis 87 might be present. Furthermore, it is tempting to speculate that fat deposition, presence of perivascular adipocytes in the heart, and adipocyte infiltration into the myocardium contribute to COVID‐19‐related heart damage in patient with obesity or related metabolic disturbances by inducing pro‐inflammatory responses; however, there are currently no solid data available to support this.
Overall, because patients with obesity or related metabolic disturbances often already have a reduced heart function, accumulative heart damage induced by cardiac pericyte dysfunction and increased myocardial inflammation may well contribute to poorer prognosis of these patients upon SARS‐CoV‐2 infection (Figure 1).
3.2.3. Multi‐organ failure (liver)
Results from a recent (relatively small) cohort study comparing patients with obesity (n = 20) and patients with obesity and NASH (n = 17) demonstrated that ACE2 and TMPRSS2 are upregulated in the liver of patients with NASH, 88 making the liver another potential target organ for SARS‐CoV‐2 infection. In line with these observations, single‐cell RNA sequencing of healthy liver tissue to investigate the tissue distribution of hepatic ACE2 demonstrated high ACE2 expression specifically in the cholangiocytes, whereas low or no ACE2 expression is observed in the hepatocytes, immune cells, and stromal cells. Therefore, it can be speculated that SARS‐CoV‐2 uses ACE2 as host receptor to induce direct damage of the bile ducts, leading to liver damage and reduced liver function. 89 For example, a cohort study with patients hospitalized with COVID‐19 (n = 1099) demonstrated that around 20% of the patients has elevated serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT), and around 10% has elevated serum total bilirubin upon hospital admission. 90 Similarly, another cohort study (n = 148) reported that 37.2% of the patients hospitalized with COVID‐19 have an abnormal liver function, indicated by elevated ALT, AST, g‐glutamyltransferase, alkaline phosphatase (ALP), and total bilirubin. 91 Elevated serum ALP levels are indicative for bile duct damage, supporting the suggestion that SARS‐CoV‐2 uses ACE2 on the cholangiocytes to induce direct damage of the bile ducts, leading to reduced liver function. Yet, in both these cohort studies, it is not clear whether the COVID‐19 patients with liver damage upon hospital admission were obese or had obesity‐related metabolic disturbances. Also, in two most recent meta‐analysis, which confirm that COVID‐19 affects liver function, a split group analysis of patients with and without obesity is missing. 92 , 93 Moreover, these meta‐analysis do not distinguish between direct damage of the hepatocytes consequently reducing liver function or SARS‐CoV‐2 induced cholangiocytes damage eventually affecting liver function.
Given that COVID‐19 negatively affects liver function, and obesity‐associated NASH is also characterized by reduced liver function, the latter group potentially forms a high‐risk group with poor prognosis upon SARS Cov‐2 infection. In line, preliminary data from a multicenter cohort study (n = 153) reported that pre‐existing chronic liver disease (22.4% non‐alcoholic fatty liver disease [NAFLD], 19.7% alcohol, 11.8% hepatitis B, 10.5% hepatitis C, 35.6% other/combination) appears to be an independent risk factor for poor outcome in COVID‐19 patients, also after correction for BMI in multiple logistic regression analysis. 94 One of the universal mechanisms affecting liver function in patients with obesity is excessive ectopic fat accumulation in the liver. Excessive lipid deposition in the liver exacerbates hepatic insulin resistance and promotes inflammation. Relevantly, an ongoing prospective COVID‐19 cohort study (currently n = 201, 18% hospitalized) reported that ectopic fat in the liver was higher in individuals hospitalized with COVID‐19 compared with non‐hospitalized individuals, 95 indicating a potential link between hepatic steatosis and severe COVID‐19. Presumably, the low‐grade chronic inflammatory state in patients with obesity‐associated NAFLD or NASH aggravates the immunogenic response induced upon SARS‐CoV‐2 infection. Overall, elevated ACE2 levels in the liver of patients with obesity and related metabolic disturbances, together with exacerbated pro‐inflammatory responses as a consequence of hepatic steatosis, may well explain the poorer prognosis of these patients upon SARS‐CoV‐2 infection (Figure 1).
3.2.4. Multi‐organ failure (pancreas)
ACE2 is expressed in the pancreas, particularly in the exocrine glands and the islets of the pancreas, as demonstrated by a tissue distribution study utilizing single‐cell RNA sequencing. 96 In line, SARS‐CoV‐2 exposure to ex vivo cultured human pancreatic islets isolated from human donors, resulted in viral SARS‐CoV‐2 replication, and affected glucose‐dependent insulin secretion in the pancreatic islets. 97 Although clinical data are limited, case reports of new‐onset type 1 diabetes after SARS‐CoV‐2 infection have been described, 98 , 99 and results from an early cohort study with severely ill COVID‐19 patients (n = 121) demonstrated that 17% (13 out of 121) of those patients has pancreatic injury, although four of these patients had been treated with glucocorticoids during hospitalization, which may be associated with drug‐induced pancreatitis. 96 In addition, two COVID‐19 cohort studies (n = 218 and n = 50, respectively) indicated that a selection of patients with type 2 diabetes, who presented with diabetic ketoacidosis upon hospital admission, showed high mortality rates. 100 , 101 Together, these data suggest that SARS‐CoV‐2 indeed potentially induces pancreatic damage, which might have detrimental consequences in patients with pre‐existing pancreatic diseases.
Obesity is characterized by excessive lipid deposition in the pancreas, which is associated with impaired insulin secretion 102 and inflammation. 103 Crucially, the same prospective cohort study (currently n = 201, 18% hospitalized) comparing ectopic fat in the liver between hospitalized and non‐hospitalized COVID‐19 patients also reported that ectopic fat in the pancreas was higher in individuals hospitalized with COVID‐19 compared with non‐hospitalized individuals, 95 indicating a potential link between excessive ectopic fat deposition in the pancreas and severe COVID‐19. The chronic low‐grade inflammatory state in patients with obesity or related metabolic disturbances may exacerbate the immunogenic response induced upon SARS‐CoV‐2, consequently aggravating pre‐existing pancreatic inflammation, thereby also contributing to the poorer prognosis of patients with obesity upon SARS‐CoV‐2 infection (Figure 1).
3.3. Cytokine storm syndrome
Chronic low‐grade inflammation is a well‐established characteristic in patients with “metabolically unhealthy” obesity. Mechanisms contributing to this chronic low‐grade inflammatory state include increased activation of the RAAS system, 80 in which specifically Ang II has been shown to induce metabolic inflammation 104 and altered adipose tissue functioning results in an amplified release of pro‐inflammatory adipocytokines. 105 In that context, elevated leptin levels (which might reduce long‐term COVID‐19 vaccination efficacy [cfr. infra]), the driving force for obesity‐related metabolic disorders, 106 can contribute to chronic low‐grade inflammation. 107 Moreover, lipid metabolism is severely dysregulated in obesity. This dysregulated lipid metabolism, in combination with elevated oxidative stress levels (which potentially also affects long‐term COVID‐19 vaccination efficacy in patients with obesity [cfr. Infra]), leads to an increased production of oxidized low‐density lipoproteins (oxLDL). Subsequently, increased oxLDL contributes to chronic low‐grade inflammation by interacting with immune cells and disturbing cholesterol trafficking. 108 As such, increased oxLDL levels strongly correlate with obesity‐related metabolic disturbances. 108 , 109 Similarly to obesity, COVID‐19 is also associated with an excess production of pro‐inflammatory cytokines. 110 SARS‐CoV‐2 infection of epithelial cells mediates mitochondrial ROS production, consequently stimulating NLR family pyrin domain containing 3 (NLRP3) and nuclear factor kappa‐light‐chain‐enhancer of activated B cells (NF‐kB) synthesis, which triggers excessive cytokine release by immune cells. 111 Moreover, SARS‐CoV‐2 enhances this cytokine secretion by internalizing ACE2 upon binding, due to which Ang II cannot be converted anymore, leading to more cytokine production. 43 , 80 Crucially, there seems to be a direct link between obesity‐mediated chronic low‐grade inflammation and the cytokine storm development upon SARS‐CoV‐2 infection observed in these patients: upon SARS‐CoV‐2 infection, macrophages chronically exposed to oxLDL are suggested to potentiate pre‐existing chronic low‐grade inflammation through apoptosis‐associated speck‐like protein containing a CARD (ASC) mediated caspase‐1 activation, leading to hyper‐inflammation and excessive cytokine secretion. 112 In addition, SARS‐CoV‐2 can interact with protein kinase R (PKR) and PKR‐like endoplasmic reticulum kinase (PERK), 113 potentially downregulating the insulin signaling pathway through serine phosphorylation of insulin receptor substrates, eventually enhancing insulin resistance. 39 Insulin resistance in adipocytes results in production of monocyte chemoattractant protein 1 (MCP1), which recruits pro‐inflammatory macrophages, creating a pro‐inflammatory environment. 114 Because insulin resistance is associated with obesity, and creates a pro‐inflammatory environment, it might be possible that the cytokine storm observed in COVID‐19 patients with obesity is aggravated via SARS‐CoV‐2‐induced insulin resistance. 39 Overall, chronic low‐grade inflammation and insulin resistance potentially facilitate the cytokine storm induced by SARS‐CoV‐2. Consequently, extremely high levels of pro‐inflammatory cytokines enhance cellular damage and eventually induce multi‐organ failure, thereby contributing to poorer prognosis of this COVID‐19 infected patient population (Figure 1).
3.4. Thromboembolism
Upon entry of SARS‐CoV‐2 into endothelial cells, inflammatory responses are induced thereby generating a pro‐thrombotic environment. 115 Extensive meta‐analysis has demonstrated that COVID‐19 patients with thromboembolism have a higher ICU admission rate and mortality rate compared with patients without thromboembolism. 116 In the context of obesity and related metabolic disturbances, several correlates exist between increased fibrinogen, factor VIII, von Willebrand factor, plasminogen activator inhibitor‐1 (PAI‐1), and decreased antithrombin III with metabolic features such as waist circumference, BMI, liver tests, and parameters of lipid and glucose metabolism as reported in a cohort of patients with NAFLD (n = 273). After multiple regression analysis, hepatic steatosis remained an independent predictor of PAI‐1 levels in this cohort. 117 Also, other cohort studies (n = 49 and n = 60) are clearly pointing toward an increased risk of thrombosis in COVID‐19 patients with obesity. For instance, anti‐thrombin levels were reported to be significantly lower in COVID‐19 patients with central obesity compared with patients without obesity, 118 whereas D‐dimer levels were significantly higher. Elevated D‐dimer was independently associated with ALT elevation, 119 indicating that microvascular thrombosis might be induced by liver inflammation in these patients. In line, another cohort of COVID‐19 patients with (n = 75) and without (n = 125) NALFD reported that NAFLD was associated with elevated D‐dimer levels at the time of ICU admission, and crucially, within this cohort, the incidence of deep vein thrombosis was higher in COVID‐19 patients with NAFLD. 120 Previously described pro‐inflammatory mechanisms (e.g., excessive ectopic fat deposition in the liver and the cytokine storm syndrome) contribute to extremely high levels of pro‐inflammatory cytokines, which consequently, can activate the coagulation system. 115 , 121 Presumably, this response enhances the generation of the pro‐thrombotic environment induced upon SARS‐CoV‐2 entry into endothelial cells, subsequently increasing the risk of thrombosis in COVID‐19 patients with MetS and potentially contributing to the higher ICU treatment rate and mortality rate of these patients upon SARS‐CoV‐2 infection (Figure 1).
3.5. Viral load
In normal physiological conditions, autophagy contributes to the defense against viral infections. Double membrane vesicles containing viral particles fuse with lysosomes where the virus is degraded. The innoxious viral antigens are used for antigen presentation to T cells to coordinate a powerful adaptive immune response against the virus. Additionally, autophagy initiates an innate immune response by activating pattern recognition receptor signaling resulting in IFN production. 122 However, in patients with obesity, MetS, cardiovascular disease, or NASH, the autophagy process shows apparent abnormalities. 123 , 124 For instance, in MetS, autophagy is attenuated in the liver, whereas in adipose tissue, the autophagic activity is induced. 124 One of the factors contributing to attenuation of the autophagy activity in NASH is an elevation of plasma cholesterol. Cholesterol accumulation inside lysosomes leads to lysosomal dysfunction and inhibition of autophagy. A possible explanation for the blocked autophagy is that lipid accumulation alters the membrane structure, consequently leading to improper fusion between lysosomes and autophagosomes. 125 , 126 In the context of host defense against SARS‐CoV‐2, this blocked autophagy process potentially contributes to decreased SARS‐CoV‐2 clearance, consequently leading to increased viral load. Additionally, many viruses have acquired properties to manipulate autophagy for their own benefit. 122 , 127 In line, SARS‐CoV‐2 has been shown to reprogram host cell metabolism to limit AMP‐activated protein kinase/mammalian target of rapamycin complex 1 (AMPK/mTORC1) activation and autophagy. 128 Mechanistical details on how disturbed autophagy potentially contribute to the close correlation between obesity, and the severe clinical manifestations of COVID‐19 have recently been reviewed elsewhere. 129 Overall, in patients with MetS and NASH the disrupted autophagy process potentially reduces SARS‐CoV‐2 clearance further and by extension increases viral load, leading to more severe COVID‐19 symptoms and poor prognosis upon SARS‐CoV‐2 infection (Figure 1).
4. OBESITY POTENTIALLY REDUCES LONG‐TERM COVID‐19 VACCINATION EFFICACY
Although clinical care for patients with COVID‐19 has significantly improved last few months, there are currently no therapies proven to be effective to cure COVID‐19. Thus, there remains an urgent need for vaccines to protect vulnerable populations, including patients with obesity and associated metabolic comorbidities. The European Medicine Agency (EMA) and The US Food and Drug Administration (FDA) have both authorized two mRNA‐based COVID‐19 vaccines for all adult individuals. 68 , 69 , 130 In addition to these two mRNA‐based COVID‐19 vaccines, the FDA 130 and EMA 70 have recently authorized other adenovirus based COVID‐19 vaccines, although these vaccines are currently on hold in several countries due to thrombosis related complications in some patients. Moreover, other potential COVID‐19 vaccine candidates are currently in development. 131 Obesity is associated with a reduced immunogenicity in response to vaccination for hepatitis B, tetanus, and influenza. 23 , 24 , 28 , 29 These vaccination studies hardly report whether patients with obesity also have MetS‐related metabolic disturbance. Therefore, although very likely, we cannot argue how MetS reduces immunogenicity after vaccination beyond the role of obesity. Here, we discuss how patients with obesity potentially develop a reduced immunogenicity in response to COVID‐19 vaccination by focusing on memory T cell and memory B cell responses in these patients.
4.1. Memory T cells
The cytotoxic T cell response is essential for the viral infection clearance, and effector memory T (Tem) cells play a crucial role in providing long‐term immunity. 132 A recent cohort study (n = 36) indicated that after 17 years, blood collected from individuals recovered from SARS still contains long‐lived Tem cells reactive against SARS‐CoV peptides. Remarkably, these long‐lived Tem cells also cross‐react to proteins of SARS‐CoV‐2. 133 Other cohort studies have found SARS‐CoV‐2 specific T cells, 134 , 135 and recently, SARS‐CoV‐2‐specific Tem cells 136 could be isolated from blood of patients recovered from COVID‐19 (n = 20, n = 25, and n = 235, respectively). These SARS‐CoV‐2 specific Tem cells, which are generated upon primary infection, or upon injection with a COVID‐19 vaccine, 137 , 138 are needed for long‐term protection against SARS‐CoV‐2. Unfortunately, several animal studies indicate that Tem cell responsiveness is severely hampered in obesity. For instance, after primary influenza infection, both lean and DIO mice have a population of influenza specific Tem cells in the lung, but obese mice show a greater percent loss of Tem cells over time, resulting in significantly decreased Tem cell numbers in the lung of obese mice post infection. 139 It has also been shown that after a secondary influenza virus challenge, although DIO mice had similar absolute percentages of Tem cells in the lung compared with lean mice, the percentage of influenza specific Tem cells responding to the challenge by producing IFNy was significantly reduced. 140 Also, after secondary influenza infection, Tem cells from DIO mice have an altered cellular metabolism, largely characterized by increased oxygen consumption, which was not reversed with weight loss. 141 However, because other mice studies did not find an effect of obesity on Tem cell development and function after influenza infection or vaccination, 142 , 143 controversy exists about the impact of obesity on Tem cell responsiveness after infection or vaccination. Yet a human cohort study comparing individuals with normal weight (n = 137) and obesity (n = 164) vaccinated against influenza demonstrated that individuals with obesity have decreased Tem cell activation after ex vivo vaccine strain virus challenge. 67 Overall, these data seem to suggest that patients with obesity potentially have a reduced Tem cell responsiveness, even after weight loss. The underlying mechanisms of the observed reduced Tem cell responsiveness after infection or vaccination is still unknown, but shorter telomere length of T cells might offer a possible explanation. 144 In a cohort of 22 elderly (>70 years) vaccinated against influenza, patients with long T cell telomeres had a slight increase in the percentage of influenza‐specific T cells in their blood compared with elderly with a shorter T cell telomere length, 84 days post vaccination. 145 Because obesity is associated with shorter telomeres due to chronic low‐grade inflammation and oxidative stress, 146 , 147 it is tempting to speculate that this affects Tem cell proliferation and development upon infection and vaccination. Another explanation might be, similar as in cancer, that obesity increases the expression of programmed cell death protein 1 (PD‐1) and programmed death‐ligand 1 (PD‐L1) on Tem cells inducing T cell exhaustion and thus reducing Tem cell responsiveness, 148 which by extension potentially diminishes long‐term protection against re‐infections. In the context of COVID‐19, initial clinical trials show that both mRNA‐based COVID‐19 vaccines and the adenovirus based COVID‐19 vaccines are effectively protecting humans in short‐term, also when suffering from obesity or associated metabolic disturbances. 68 , 69 , 71 Nonetheless, for all vaccines, it is currently not known yet how long this protection will last. Because obesity is associated with reduced Tem cell responsiveness, long‐term protection against re‐infections is also diminished. Therefore, despite COVID‐19 vaccination, patients with obesity may still be more vulnerable for re‐infection with SARS‐CoV‐2 (Figure 2).
FIGURE 2.
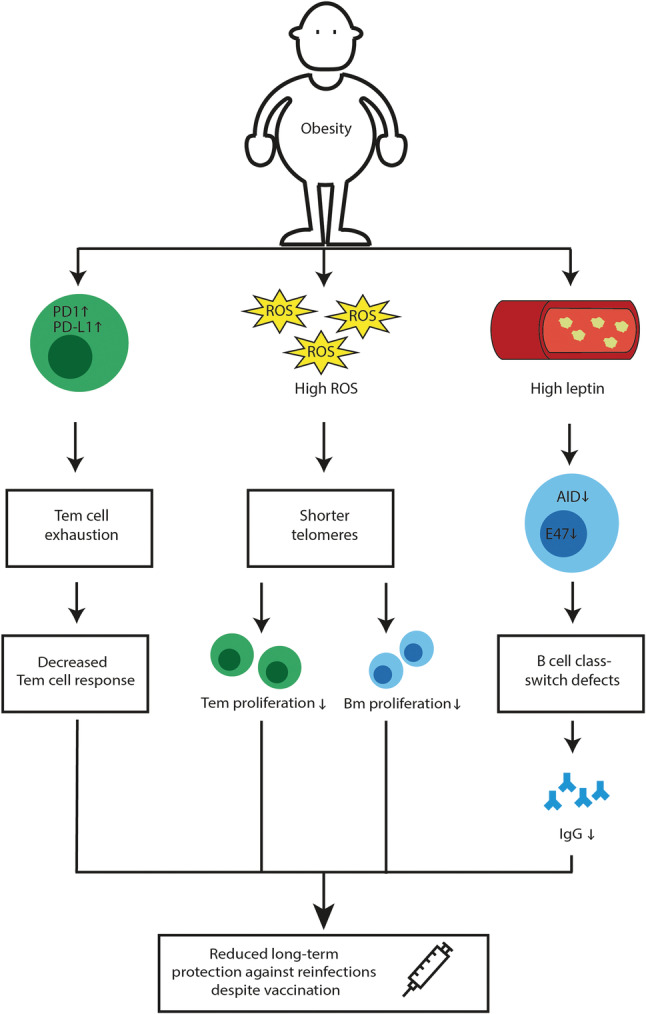
Obesity potentially reduces long‐term COVID‐19 vaccination efficacy. Obesity alters PD1 and PD‐L1 expression on Tem cells, weakening stem cell responsiveness. Also, obesity is associated with elevated systemic ROS causing shorter telomere length of immune cells, leading to decreased Tem and Bm proliferation. Moreover, high serum leptin levels, as observed in obesity, reduces AID and E47 expression in B cells, inducing B cell class‐switching defects, potentially leading to decreased SARS‐CoV‐2‐specific‐IgG production. These factors together reduce long‐term protection against re‐infections. Therefore, despite COVID‐19 vaccination, patients with obesity may still be vulnerable for re‐infection with SARS‐CoV‐2. AID, activation‐in induced cytidine deaminase; Bm, B memory cells; IgG, immunoglobulin G; PD1, programmed cell death protein 1; PD‐L1, programmed death‐ligand 1; ROS, reactive oxygen species; Tem effector memory T cells
4.2. Memory B cells
The humoral B cell immune response is crucial for clearance of viral infections and long‐term immunity. 149 The SARS‐CoV‐2 virus elicits a robust humoral B cell response as evidenced by a high production of virus specific antibodies found in blood of several COVID‐19 cohorts (n = 518 and n = 607). 150 , 151 A recent study showed that most individuals reach a neutralizing antibody peak after an average of 23.1 days post onset of symptoms. In the follow‐up period of this study, it was found that IgM and IgA antibodies decline rapidly after 20–30 days post onset of symptoms, whereas IgG antibodies lasted longer (max study follow‐up of 95 days). 152 Relevantly, a longitudinal cohort study (n = 254) showed that during the early phase, SARS‐CoV‐2 specific class‐switched IgG and IgM memory B (Bm) cells are similarly present in high amounts in the blood of patients recovered from COVID‐19, whereas the population of class‐switched IgA Bm cells was low. Over time, the class‐switched IgM Bm cell population declines and the class‐switched IgG Bm cells become the dominant population. 153 Notably, these SARS‐CoV‐2 specific class‐switched Bm cells, either induced by primary infection or upon vaccination, 154 , 155 are involved in regulating the immune response against SARS‐CoV‐2 upon re‐infection. However, similar as Tem cell responsiveness, several studies indicate that humoral Bm cell responsiveness is also reduced in obesity. For example, compared with lean mice, DIO mice revealed lower hemagglutination inhibition (HAI) titers (standard assay used to determine antibody levels to influenza virus) after influenza infection 65 , 143 or adjuvant influenza vaccination. 156 A similar result was found in a human cohort study (consisting of four groups, young lean n = 8; young obese n = 6; elderly lean n = 8 and elderly obese n = 4) comparing individuals with normal weight and obesity vaccinated against influenza, in which both the young and elderly individuals with obesity had a decreased percentage of class‐switched Bm cells and an increased percentage of exhausted Bm cells in their blood, compared with respective healthy weight individuals of the same age. 66 These observations potentially partially explain, the increased risk of acquiring influenza infection, despite vaccination, of individuals with obesity compared with individuals with a healthy weight. The data may be explained by increased leptin levels (a pro‐inflammatory adipokine) observed in individuals with obesity. 157 In the presence of leptin, ex vivo cultured human B cells showed class‐switching defects, potentially regulated via leptin‐induced downregulation of activation‐induced cytidine deaminase (AID) (the enzyme necessary for class switch recombination, somatic hypermutation and IgG production) and its transcriptional regulator E47. Therefore, leptin might decrease influenza‐vaccine‐specific‐IgG production in individuals with obesity. 158 In contrast, other cohort studies comparing individuals with a normal weight and obesity vaccinated against seasonal influenza (n = 34, 50% obese) 67 or tick‐borne encephalitis (n = 73, 50% obese), 159 indicated that individuals with obesity at first develop stronger antibody responses but have a steeper decline over time. In addition, individuals with obesity vaccinated against tick‐borne encephalitis had more naïve B cells in their blood and less expansion to Bm cells upon booster vaccination. 159 Similarly, as discussed above, this decreased Bm cell expansion might also be explained by the shorter telomere length in obesity. 145 , 146 , 147 In the context of COVID‐19, initial clinical trials show that al authorized COVID‐19 vaccines are effectively protecting patients with obesity and related metabolic disturbances in the short‐term. 68 , 69 , 70 , 71 Nonetheless, for these vaccines, it is currently not known yet how long this protection lasts in healthy individuals as well as in patients with obesity. Because obesity is associated with reduced humoral Bm cell responsiveness, long‐term protection against re‐infections is also diminished. Therefore, despite COVID‐19 vaccination, patients with obesity may still be vulnerable for re‐infection with SARS‐CoV‐2 (Figure 2).
5. CONCLUSION AND FUTURE DIRECTIONS
Overall, due to increased expression of proteins facilitating viral entry into cells and hyper‐glycosylation of those proteins, patients with obesity and related metabolic disturbances have an increased risk of becoming infected with SARS‐CoV‐2. In addition, due to a compromised immune response in the lungs, hyper‐inflammatory systemic responses, increased risk of thrombosis and increased viral load, patients with obesity and related metabolic disturbances also develop severe complications upon SARS‐CoV‐2 infection, leading to higher morbidity and mortality risks upon COVID‐19. Although clinical care for patients with COVID‐19 has significantly improved last few months, there are currently no therapies proven to be effective to cure COVID‐19. Therefore, there remains an urgent need for vaccines to protect vulnerable populations such as patients with obesity and related metabolic disturbances. Initial clinical trials show that currently authorized COVID‐19 vaccines are effectively protecting these patients, nonetheless, it is currently not known yet how long this protection lasts. 68 , 69 Obesity is associated with reduced memory immune responses leading to diminished long‐term protection against re‐infections. Therefore, despite encouraging COVID‐19 vaccination results, patients with obesity may still be vulnerable for re‐infection with SARS‐CoV‐2 in the long run. This may affect herd immunity and impact SARS‐CoV‐2 elimination. In conclusion, to limit further impact of COVID‐19 on patients with obesity and related metabolic disturbances, and society, long‐term COVID‐19 vaccine efficacy should be closely monitored in these patients.
CONFLICT OF INTERESTS
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent‐licensing arrangements), or non‐financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
ACKNOWLEDGMENTS
We acknowledge the contribution of all authors cited. We thank all people from the Shiri‐Sverdlov and Theys research groups for their contribution. This work was funded by TKI‐LSH (grant no. 40‐41200‐98‐9306 to RS‐S) and VCK (grant no. Swu16.0057‐VT to RS‐S). The funders had no role in the collection and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
Westheim AJF, Bitorina AV, Theys J, Shiri‐Sverdlov R. COVID‐19 infection, progression, and vaccination: Focus on obesity and related metabolic disturbances. Obesity Reviews. 2021;22(10):e13313. 10.1111/obr.13313
Funding information VCK, Grant/Award Number: Swu16.0057‐VT; TKI‐LSH, Grant/Award Number: 40‐41200‐98‐9306
REFERENCES
- 1. WHO . https://www.who.int/emergencies/diseases/novel‐coronavirus‐2019?gclid=EAIaIQobChMIxKaNnuLW6QIV2fhRCh3NBwGEEAAYASAAEgK1jPD_BwE. Accessed March 3, 2021.
- 2. Durmus S, Ulgen KO. Comparative interactomics for virus‐human protein‐protein interactions: DNA viruses versus RNA viruses. FEBS Open Bio. 2017;7(1):96‐107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Hodinka RL. Respiratory RNA viruses. Microbiol Spectr. 2016;4(4):1‐33. [DOI] [PubMed] [Google Scholar]
- 4. Jean A, Quach C, Yung A, Semret M. Severity and outcome associated with human coronavirus OC43 infections among children. Pediatr Infect Dis J. 2013;32(4):325‐329. [DOI] [PubMed] [Google Scholar]
- 5. Gaunt ER, Hardie A, Claas EC, Simmonds P, Templeton KE. Epidemiology and clinical presentations of the four human coronaviruses 229E, HKU1, NL63, and OC43 detected over 3 years using a novel multiplex real‐time PCR method. J Clin Microbiol. 2010;48(8):2940‐2947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lee N, Hui D, Wu A, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348(20):1986‐1994. [DOI] [PubMed] [Google Scholar]
- 7. Habib AMG, Ali MAE, Zouaoui BR, Taha MAH, Mohammed BS, Saquib N. Clinical outcomes among hospital patients with Middle East respiratory syndrome coronavirus (MERS‐CoV) infection. BMC Infect Dis. 2019;19(1):870‐875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. de Wit E, van Doremalen N, Falzarano D, Munster VJ. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14(8):523‐534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395(10223):497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. The Lancet. 2020;395(10223):470‐473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Zhou P, Yang XL, Wang XG, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270‐273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Billah MA, Miah MM, Khan MN. Reproductive number of coronavirus: a systematic review and meta‐analysis based on global level evidence. PLoS ONE. 2020;15(11):1‐17, e0242128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Petrosillo N, Viceconte G, Ergonul O, Ippolito G, Petersen E. COVID‐19, SARS and MERS: are they closely related? Clin Microbiol Infect. 2020;26(6):729‐734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Park SE. Epidemiology, virology, and clinical features of severe acute respiratory syndrome‐coronavirus‐2 (SARS‐CoV‐2; Coronavirus Disease‐19). Clin Exp Pediatr. 2020;63(4):119‐124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Erik Volz SM, Chand M, Barrett JC, Johnson R, Geidelberg L, Hinsley WR, Laydon DJ, Dabrera G, O'Toole Á, Amato R, Ragonnet‐Cronin M, Harrison I, Jackson B, Ariani CV, Boyd O, Loman NJ, McCrone JT, Gonçalves S, Jorgensen D, Myers R, Hill V, Jackson DK, Gaythorpe K, Groves N, Sillitoe J, Kwiatkowski DP, The COVID‐19 Genomics UK (COG‐UK) consortium, Seth Flaxman, Oliver Ratmann, Samir Bhatt, Susan Hopkins, Axel Gandy, Andrew Rambaut, Neil M Ferguson . Transmission of SARS‐CoV‐2 Lineage B.1.1.7 in England: insights from linking epidemiological and genetic data. medRxiv preprint. 2021.
- 16. Jennifer Lighter MP, Hochman S, Sterling S, Johnson D, Francois F, Stachel A. Predictors of refractory coronavirus disease (COVID‐19) pneumonia. Clin Infect Dis. 2020;71:895‐896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bluher M. The distinction of metabolically ‘healthy’ from ‘unhealthy’ obese individuals. Curr Opin Lipidol. 2010;21(1):38‐43. [DOI] [PubMed] [Google Scholar]
- 18. Soverini V, Moscatiello S, Villanova N, Ragni E, Di Domizio S, Marchesini G. Metabolic syndrome and insulin resistance in subjects with morbid obesity. Obes Surg. 2010;20(3):295‐301. [DOI] [PubMed] [Google Scholar]
- 19. Wildman RP, Muntner P, Reynolds K, et al. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999‐2004). Arch Intern Med. 2008;168(15):1617‐1624. [DOI] [PubMed] [Google Scholar]
- 20. Eckel RH, Alberti KG, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet. 2010;375(9710):181‐183. [DOI] [PubMed] [Google Scholar]
- 21. Grundy SM, Cleeman JI, Daniels SR, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735‐2752. [DOI] [PubMed] [Google Scholar]
- 22. https://www.idf.org. Accessed April 22, 2021.
- 23. Yang H, Youm YH, Vandanmagsar B, et al. Obesity accelerates thymic aging. Blood. 2009;114(18):3803‐3812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6(7):438‐446. [DOI] [PubMed] [Google Scholar]
- 25. Hanslik T, Boelle PY, Flahault A. Preliminary estimation of risk factors for admission to intensive care units and for death in patients infected with A(H1N1)2009 influenza virus, France, 2009‐2010. PLoS Curr. 2010;2(2):1‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Morgan OW, Bramley A, Fowlkes A, et al. Morbid obesity as a risk factor for hospitalization and death due to 2009 pandemic influenza A(H1N1) disease. PLoS ONE. 2010;5(3):9694‐9699, e9694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Allard R, Leclerc P, Tremblay C, Tannenbaum TN. Diabetes and the severity of pandemic influenza A (H1N1) infection. Diabetes Care. 2010;33(7):1491‐1493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Andersen CJ, Murphy KE, Fernandez ML. Impact of obesity and metabolic syndrome on immunity. Adv Nutr. 2016;7(1):66‐75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Milner JJ, Beck MA. The impact of obesity on the immune response to infection. Proc Nutr Soc. 2012;71(2):298‐306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Halim AA, Alsayed B, Embarak S, Yaseen T, Dabbous S. Clinical characteristics and outcome of ICU admitted MERS corona virus infected patients. Egypt J Chest Dis Tuberc. 2016;65(1):81‐87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Hu J, Wang Y. The clinical characteristics and risk factors of severe COVID‐19. Gerontology. 2021;1‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Popkin BM, Du S, Green WD, et al. Individuals with obesity and COVID‐19: a global perspective on the epidemiology and biological relationships. Obes Rev. 2020;21(11):1‐17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Aghili SMM, Ebrahimpur M, Arjmand B, et al. Obesity in COVID‐19 era, implications for mechanisms, comorbidities, and prognosis: a review and meta‐analysis. Int J Obes (Lond). 2021;45(5):998‐1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Tadic M, Saeed S, Grassi G, Taddei S, Mancia G, Cuspidi C. Hypertension and COVID‐19: ongoing Controversies. Front Cardiovasc Med. 2021;8:1‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Zheng YY, Ma YT, Zhang JY, Xie X. COVID‐19 and the cardiovascular system. Nat Rev Cardiol. 2020;17(5):259‐260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Zheng YY, Ma YT, Zhang JY, Xie X. Reply to: ‘Interaction between RAAS inhibitors and ACE2 in the context of COVID‐19’. Nat Rev Cardiol. 2020;17(5):313‐314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Wang J, Meng W. COVID‐19 and diabetes: the contributions of hyperglycemia. J Mol Cell Biol. 2021;12(12):958‐962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Casari I, Manfredi M, Metharom P, Falasca M. Dissecting lipid metabolism alterations in SARS‐CoV‐2. Prog Lipid Res. 2021;1‐17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Santos A, Magro DO, Evangelista‐Poderoso R, Saad MJA. Diabetes, obesity, and insulin resistance in COVID‐19: molecular interrelationship and therapeutic implications. Diabetol Metab Syndr. 2021;13(1):23‐36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Govender N, Khaliq OP, Moodley J, Naicker T. Insulin resistance in COVID‐19 and diabetes. Prim Care Diabetes. 2021;1035‐1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. McGonagle D, O'Donnell JS, Sharif K, Emery P, Bridgewood C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID‐19 pneumonia. Lancet Rheumatol. 2020;2(7):e437‐e445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Kruglikov IL, Scherer PE. Preexisting and inducible endotoxemia as crucial contributors to the severity of COVID‐19 outcomes. PLoS Pathog. 2021;17(2):1‐5, e1009306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kruglikov IL, Shah M, Scherer PE. Obesity and diabetes as comorbidities for COVID‐19: underlying mechanisms and the role of viral‐bacterial interactions. Elife. 2020;9:1‐21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Dimitrov DS. Virus entry: molecular mechanisms and biomedical applications. Nat Rev Microbiol. 2004;2(2):109‐122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Mercer J, Schelhaas M, Helenius A. Virus entry by endocytosis. Annu Rev Biochem. 2010;79(1):803‐833. [DOI] [PubMed] [Google Scholar]
- 46. Nabavi SF, Habtemariam S, Berindan‐Neagoe I, et al. Rationale for effective prophylaxis against COVID‐19 through simultaneous blockade of both endosomal and non‐endosomal SARS‐CoV‐2 entry into host cell. Clin Transl Sci. 2021;14(2):431‐433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Li MY, Li L, Zhang Y, Wang XS. Expression of the SARS‐CoV‐2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty. 2020;9(1):45‐51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Hoffmann M, Kleine‐Weber H, Schroeder S, et al. SARS‐CoV‐2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271‐280 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Luquain‐Costaz C, Rabia M, Hullin‐Matsuda F, Delton I. Bis (monoacylglycero)phosphate, an important actor in the host endocytic machinery hijacked by SARS‐CoV‐2 and related viruses. Biochimie. 2020;179:247‐256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Berk BC. Angiotensin type 2 receptor (AT2R): a challenging twin. Sci STKE. 2003;2003(181):16‐18. [DOI] [PubMed] [Google Scholar]
- 51. Benter IF, Ferrario CM, Morris M, Diz DI. Antihypertensive actions of angiotensin‐(1‐7) in spontaneously hypertensive rats. Am J Physiol. 1995;269(1 Pt 2):H313‐H319. [DOI] [PubMed] [Google Scholar]
- 52. Santos RA, Simoes e Silva AC, Maric C, et al. Angiotensin‐(1–7) is an endogenous ligand for the G protein‐coupled receptor Mas. Proc Natl Acad Sci U S A. 2003;100:8258‐8263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Mishra D, Dey CS. Type‐2 diabetes, a co‐morbidity in Covid‐19: does insulin signaling matter? Biochem Soc Trans. 2021;49(2):987‐995. [DOI] [PubMed] [Google Scholar]
- 54. Goulter AB, Goddard MJ, Allen JC, Clark KL. ACE2 gene expression is up‐regulated in the human failing heart. BMC Med. 2004;2(1):19‐25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Chen L, Li X, Chen M, Feng Y, Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS‐CoV‐2. Cardiovasc Res. 2020;116(6):1097‐1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Rao S, Lau A, So HC. Exploring diseases/traits and blood proteins causally related to expression of ACE2, the putative receptor of SARS‐CoV‐2: a Mendelian randomization analysis highlights tentative relevance of diabetes‐related traits. Diabetes Care. 2020;43(7):1416‐1426. [DOI] [PubMed] [Google Scholar]
- 57. Zamorano Cuervo N, Grandvaux N. ACE2: evidence of role as entry receptor for SARS‐CoV‐2 and implications in comorbidities. Elife. 2020;9:1‐25, e61390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Mourad JJ, Levy BI. Interaction between RAAS inhibitors and ACE2 in the context of COVID‐19. Nat Rev Cardiol. 2020;17(5):313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Paizis G, Tikellis C, Cooper ME, et al. Chronic liver injury in rats and humans upregulates the novel enzyme angiotensin converting enzyme 2. Gut. 2005;54(12):1790‐1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Michalia M, Kompoti M, Koutsikou A, et al. Diabetes mellitus is an independent risk factor for ICU‐acquired bloodstream infections. Intensive Care Med. 2009;35(3):448‐454. [DOI] [PubMed] [Google Scholar]
- 61. Chen H, Liu W, Liu D, Zhao L, Yu J. SARS‐CoV‐2 activates lung epithelia cell proinflammatory signaling and leads to immune dysregulation in COVID‐19 patients by single‐cell sequencing. medRxiv preprint. 2020. [DOI] [PMC free article] [PubMed]
- 62. Codo AC, Davanzo GG, Monteiro LB, et al. Elevated glucose levels favor SARS‐CoV‐2 infection and monocyte response through a HIF‐1alpha/glycolysis‐dependent axis. Cell Metab. 2020;32(3):437‐446 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Philips BJ, Meguer JX, Redman J, Baker EH. Factors determining the appearance of glucose in upper and lower respiratory tract secretions. Intensive Care Med. 2003;29(12):2204‐2210. [DOI] [PubMed] [Google Scholar]
- 64. Brufsky A. Hyperglycemia, hydroxychloroquine, and the COVID‐19 pandemic. J Med Virol. 2020;92(7):770‐775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Kosaraju R, Guesdon W, Crouch MJ, et al. B cell activity is impaired in human and mouse obesity and is responsive to an essential fatty acid upon murine influenza infection. J Immunol. 2017;198(12):4738‐4752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Frasca D, Ferracci F, Diaz A, Romero M, Lechner S, Blomberg BB. Obesity decreases B cell responses in young and elderly individuals. Obesity (Silver Spring). 2016;24(3):615‐625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Sheridan PA, Paich HA, Handy J, et al. Obesity is associated with impaired immune response to influenza vaccination in humans. Int J Obes (Lond). 2012;36:1072‐1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Agency EM . Assessment report Comirnaty Common name: COVID‐19 mRNA vaccine (nucleoside‐modified) Procedure No. EMEA/H/C/005735/0000. 707383/2020 Committee for Medicinal Products for Human Use (CHMP). 2020.
- 69. Agency EM . COVID‐19 vaccine Moderna COVID‐19 mRNA vaccine (nucleoside modified). 2021; EMA/704372/2020 EMEA/H/C/005735.
- 70. Agency EM . COVID‐19 vaccine AstraZeneca. COVID‐19 Vaccine (ChAdOx1‐S [recombinant]). 29 January 2021 EMA/57617/2021 EMEA/h/c/005675 2021.
- 71. Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV‐19 vaccine (AZD1222) against SARS‐CoV‐2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99‐111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Bae S, Kim SR, Kim MN, Shim WJ, Park SM. Impact of cardiovascular disease and risk factors on fatal outcomes in patients with COVID‐19 according to age: a systematic review and meta‐analysis. Heart. 2021;107(5):373‐380. [DOI] [PubMed] [Google Scholar]
- 73. Chen J, Qi T, Liu L, et al. Clinical progression of patients with COVID‐19 in Shanghai, China. J Infect. 2020;80(5):e1‐e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Renu K, Prasanna PL, Abilash VG. Coronaviruses pathogenesis, comorbidities and multi‐organ damage—a review. Life Sci. 2020;225:1‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Mahajan R, Stokes M, Elliott A, et al. Complex interaction of obesity, intentional weight loss and heart failure: a systematic review and meta‐analysis. Heart. 2020;106(1):58‐68. [DOI] [PubMed] [Google Scholar]
- 76. Sun Y, Milne S, Jaw JE, et al. BMI is associated with FEV1 decline in chronic obstructive pulmonary disease: a meta‐analysis of clinical trials. Respir Res. 2019;20(1):236‐245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Nie W, Zhang Y, Jee SH, Jung KJ, Li B, Xiu Q. Obesity survival paradox in pneumonia: a meta‐analysis. BMC Med. 2014;12(1):61‐69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Abumayyaleh M, Nunez Gil IJ, El‐Battrawy I, et al. Does there exist an obesity paradox in COVID‐19? Insights of the international HOPE‐COVID‐19‐registry. Obes Res Clin Pract. 2021;15(3):275‐280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Kruglikov IL, Scherer PE. The role of adipocytes and adipocyte‐like cells in the severity of COVID‐19 infections. Obesity (Silver Spring). 2020;28(7):1187‐1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Goossens GH, Dicker D, Farpour‐Lambert NJ, et al. Obesity and COVID‐19: a perspective from the European Association for the Study of Obesity on immunological perturbations, therapeutic challenges, and opportunities in obesity. Obes Facts. 2020;13(4):439‐452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Dicker D, Bettini S, Farpour‐Lambert N, et al. Obesity and COVID‐19: the two sides of the coin. Obes Facts. 2020;13(4):430‐438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Stone E, Kiat H, McLachlan CS. Atrial fibrillation in COVID‐19: a review of possible mechanisms. FASEB J. 2020;34(9):11347‐11354. [DOI] [PubMed] [Google Scholar]
- 83. Rabkin SW. The relationship between epicardial fat and indices of obesity and the metabolic syndrome: a systematic review and meta‐analysis. Metab Syndr Relat Disord. 2014;12(1):31‐42. [DOI] [PubMed] [Google Scholar]
- 84. Couselo‐Seijas M, Almengló C, Agra‐Bermejo R, et al. Higher ACE2 expression levels in epicardial cells than subcutaneous stromal cells from patients with cardiovascular disease: diabetes and obesity as possible enhancer. Eur J Clin Invest. 2020;51(5):1‐10, e13463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Iacobellis G, Leonetti F. Epicardial adipose tissue and insulin resistance in obese subjects. J Clin Endocrinol Metab. 2005;90(11):6300‐6302. [DOI] [PubMed] [Google Scholar]
- 86. Patel VB, Mori J, McLean BA, et al. ACE2 deficiency worsens epicardial adipose tissue inflammation and cardiac dysfunction in response to diet‐induced obesity. Diabetes. 2016;65(1):85‐95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Sawalha K, Abozenah M, Kadado AJ, et al. Systematic review of COVID‐19 related myocarditis: insights on management and outcome. Cardiovasc Revasc Med. 2021;23:107‐113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Fondevila MF, Mercado‐Gomez M, Rodriguez A, et al. Obese patients with NASH have increased hepatic expression of SARS‐CoV‐2 critical entry points. J Hepatol. 2021;74(2):469‐471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Chai X, Hu L, Zhang Y, et al. Specific ACE2 expression in cholangiocytes may cause liver damage after 2019‐nCoV infection. BioRxiv, the Preprint Server for Biology. 2020;1‐13. [Google Scholar]
- 90. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708‐1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Fan Z, Chen L, Li J, et al. Clinical features of COVID‐19‐related liver functional abnormality. Clin Gastroenterol Hepatol. 2020;18(7):1561‐1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Diaz LA, Idalsoaga F, Cannistra M, et al. High prevalence of hepatic steatosis and vascular thrombosis in COVID‐19: a systematic review and meta‐analysis of autopsy data. World J Gastroenterol. 2020;26(48):7693‐7706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Del Zompo F, De Siena M, Ianiro G, Gasbarrini A, Pompili M, Ponziani FR. Prevalence of liver injury and correlation with clinical outcomes in patients with COVID‐19: systematic review with meta‐analysis. Eur Rev Med Pharmacol Sci. 2020;24(24):13072‐13088. [DOI] [PubMed] [Google Scholar]
- 94. Moon AM, Webb GJ, Aloman C, et al. High mortality rates for SARS‐CoV‐2 infection in patients with pre‐existing chronic liver disease and cirrhosis: preliminary results from an international registry. J Hepatol. 2020;73(3):705‐708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Andrea Dennis MW, Kapur S, Alberts J, Badley AD, Decker GA, Rizza SA, Banerjee R, Amitava Banerjee D, COVERSCAN study investigators . Multi‐organ impairment in low‐risk individuals with long COVID. medRxiv preprint. 2021.
- 96. Liu F, Long X, Zhang B, Zhang W, Chen X, Zhang Z. ACE2 expression in pancreas may cause pancreatic damage after SARS‐CoV‐2 infection. Clin Gastroenterol Hepatol. 2020;18(9):2128‐2130.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Muller JA, Gross R, Conzelmann C, et al. SARS‐CoV‐2 infects and replicates in cells of the human endocrine and exocrine pancreas. Nat Metab. 2021;3(2):149‐165. [DOI] [PubMed] [Google Scholar]
- 98. Marchand L, Pecquet M, Luyton C. Type 1 diabetes onset triggered by COVID‐19. Acta Diabetol. 2020;57(10):1265‐1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Hollstein T, Schulte DM, Schulz J, et al. Autoantibody‐negative insulin‐dependent diabetes mellitus after SARS‐CoV‐2 infection: a case report. Nat Metab. 2020;2(10):1021‐1024. [DOI] [PubMed] [Google Scholar]
- 100. Goldman N, Fink D, Cai J, Lee YN, Davies Z. High prevalence of COVID‐19‐associated diabetic ketoacidosis in UK secondary care. Diabetes Res Clin Pract. 2020;166:1‐4, 108291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Chamorro‐Pareja N, Parthasarathy S, Annam J, Hoffman J, Coyle C, Kishore P. Letter to the editor: unexpected high mortality in COVID‐19 and diabetic ketoacidosis. Metabolism. 2020;110:1‐4, 154301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Wagner R, Jaghutriz BA, Gerst F, et al. Pancreatic steatosis associates with impaired insulin secretion in genetically predisposed individuals. J Clin Endocrinol Metab. 2020;105(11):3518‐3525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Horii T, Fujita Y, Ishibashi C, et al. Islet inflammation is associated with pancreatic fatty infiltration and hyperglycemia in type 2 diabetes. BMJ Open Diabetes Res Care. 2020;8(1):e001508_1‐e001508_8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Jia L, Li Y, Xiao C, Du J. Angiotensin II induces inflammation leading to cardiac remodeling. Front Biosci (Landmark Ed). 2012;17:221‐231. [DOI] [PubMed] [Google Scholar]
- 105. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860‐867. [DOI] [PubMed] [Google Scholar]
- 106. Zhao S, Zhu Y, Schultz RD, et al. Partial leptin reduction as an insulin sensitization and weight loss strategy. Cell Metab. 2019;30(4):706‐719 e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Iikuni N, Lam QL, Lu L, Matarese G, La Cava A. Leptin and Inflammation. Curr Immunol Rev. 2008;4(2):70‐79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Walenbergh SM, Koek GH, Bieghs V, Shiri‐Sverdlov R. Non‐alcoholic steatohepatitis: the role of oxidized low‐density lipoproteins. J Hepatol. 2013;58(4):801‐810. [DOI] [PubMed] [Google Scholar]
- 109. Jeurissen MLJ, Walenbergh SMA, Houben T, et al. Prevention of oxLDL uptake leads to decreased atherosclerosis in hematopoietic NPC1‐deficient Ldlr(−/−) mice. Atherosclerosis. 2016;255:59‐65. [DOI] [PubMed] [Google Scholar]
- 110. Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID‐19: consider cytokine storm syndromes and immunosuppression. The Lancet. 2020;395(10229):1033‐1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Bhaskar S, Sinha A, Banach M, et al. Cytokine storm in COVID‐19‐immunopathological mechanisms, clinical considerations, and therapeutic approaches: the REPROGRAM consortium position paper. Front Immunol. 2020;11:1‐16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Erol A. Role of oxidized LDL‐induced “trained macrophages” in the pathogenesis of COVID‐19 and benefits of pioglitazone: a hypothesis. Diabetes Metab Syndr Clin Res Rev. 2020;14(4):713‐714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Tiwari R, Mishra AR, Gupta A, Nayak D. Structural similarity‐based prediction of host factors associated with SARS‐CoV‐2 infection and pathogenesis. J Biomol Struct Dyn. 2021;1‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Shimobayashi M, Albert V, Woelnerhanssen B, et al. Insulin resistance causes inflammation in adipose tissue. J Clin Invest. 2018;128(4):1538‐1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Teuwen LA, Geldhof V, Pasut A, Carmeliet P. COVID‐19: the vasculature unleashed. Nat Rev Immunol. 2020;20(7):389‐391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Wang C, Zhang H, Zhou M, et al. Prognosis of COVID‐19 in patients with vein thrombosis: a systematic review and meta‐analysis. Eur Rev Med Pharmacol Sci. 2020;24(19):10279‐10285. [DOI] [PubMed] [Google Scholar]
- 117. Verrijken A, Francque S, Mertens I, et al. Prothrombotic factors in histologically proven nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology. 2014;59(1):121‐129. [DOI] [PubMed] [Google Scholar]
- 118. Gazzaruso C, Paolozzi E, Valenti C, et al. Association between antithrombin and mortality in patients with COVID‐19. A possible link with obesity. Nutr Metab Cardiovasc Dis. 2020;30(11):1914‐1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Tsutsumi T, Saito M, Nagai H, et al. Association of coagulopathy with liver dysfunction in patients with COVID‐19. Hepatol Res. 2021;51(2):227‐232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Ji D, Zhang M, Qin E, et al. Letter to the Editor: obesity, diabetes, non‐alcoholic fatty liver disease and metabolic dysfunction associated fatty liver disease are proinflammatory hypercoagulable states associated with severe disease and thrombosis in Covid‐19. Metabolism. 2021;115:154437‐154439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Bevilacqua MP, Pober JS, Majeau GR, Fiers W, Cotran RS, Gimbrone MA Jr. Recombinant tumor necrosis factor induces procoagulant activity in cultured human vascular endothelium: characterization and comparison with the actions of interleukin 1. Proc Natl Acad Sci U S A. 1986;83(12):4533‐4537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Choi Y, Bowman JW, Jung JU. Autophagy during viral infection—a double‐edged sword. Nat Rev Microbiol. 2018;16(6):341‐354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Lim YM, Lim H, Hur KY, et al. Systemic autophagy insufficiency compromises adaptation to metabolic stress and facilitates progression from obesity to diabetes. Nat Commun. 2014;5(1):4934‐4947. [DOI] [PubMed] [Google Scholar]
- 124. Menikdiwela KR, Ramalingam L, Rasha F, et al. Autophagy in metabolic syndrome: breaking the wheel by targeting the renin‐angiotensin system. Cell Death Dis. 2020;11(2):87‐103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Bieghs V, Hendrikx T, van Gorp PJ, et al. The cholesterol derivative 27‐hydroxycholesterol reduces steatohepatitis in mice. Gastroenterology. 2013;144(1):167‐178 e1. [DOI] [PubMed] [Google Scholar]
- 126. Hendrikx T, Walenbergh SM, Hofker MH, Shiri‐Sverdlov R. Lysosomal cholesterol accumulation: driver on the road to inflammation during atherosclerosis and non‐alcoholic steatohepatitis. Obes Rev. 2014;15(5):424‐433. [DOI] [PubMed] [Google Scholar]
- 127. Maier HJ, Britton P. Involvement of autophagy in coronavirus replication. Viruses. 2012;4(12):3440‐3451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Gassen NC, Papies J, Bajaj T, Dethloff F, Emanuel J, Weckmann K, et al. Analysis of SARS‐CoV‐2‐controlled autophagy reveals spermidine, MK‐2206, and niclosamide as 2 putative antiviral therapeutics. medRxiv preprint. 2020.
- 129. Garcia‐Perez BE, Gonzalez‐Rojas JA, Salazar MI, Torres‐Torres C, Castrejon‐Jimenez NS. Taming the autophagy as a strategy for treating COVID‐19. Cell. 2020;9(12):2679‐2706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. FDA . https://www.fda.gov/emergency‐preparedness‐and‐response/coronavirus‐disease‐2019‐covid‐19/covid‐19‐vaccines. Accessed April 22, 2021.
- 131. WHO . https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines. Accessed April 22, 2021.
- 132. Schmidt ME, Varga SM. The CD8 T cell response to respiratory virus infections. Front Immunol. 2018;9:678‐689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Le Bert N, Tan AT, Kunasegaran K, et al. SARS‐CoV‐2‐specific T cell immunity in cases of COVID‐19 and SARS, and uninfected controls. Nature. 2020;584(7821):457‐462. [DOI] [PubMed] [Google Scholar]
- 134. Grifoni A, Weiskopf D, Ramirez SI, et al. Targets of T cell responses to SARS‐CoV‐2 coronavirus in humans with COVID‐19 disease and unexposed individuals. Cell. 2020;181(7):1489‐1501 e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Braun J, Loyal L, Frentsch M, et al. SARS‐CoV‐2‐reactive T cells in healthy donors and patients with COVID‐19. Nature. 2020;587(7833):270‐274. [DOI] [PubMed] [Google Scholar]
- 136. Rha MS, Jeong HW, Ko JH, et al. PD‐1‐expressing SARS‐CoV‐2‐specific CD8(+) T cells are not exhausted, but functional in patients with COVID‐19. Immunity. 2021;54(1):44‐52 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Sahin U, Muik A, Vogler I, Derhovanessian E, Kranz LM, Vormehr M, et al. BNT162b2 induces SARS‐CoV‐2‐neutralising antibodies and T cells in humans. MedRxiv Preprint 2020.
- 138. Silva‐Cayetano A, Foster WS, Innocentin S, et al. A booster dose enhances immunogenicity of the COVID‐19 vaccine candidate ChAdOx1 nCoV‐19 in aged mice. Med (N Y). 2020;2(3):243‐262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Karlsson EA, Sheridan PA, Beck MA. Diet‐induced obesity in mice reduces the maintenance of influenza‐specific CD8+ memory T cells. J Nutr. 2010;140(9):1691‐1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Karlsson EA, Sheridan PA, Beck MA. Diet‐induced obesity impairs the T cell memory response to influenza virus infection. J Immunol. 2010;184(6):3127‐3133. [DOI] [PubMed] [Google Scholar]
- 141. Rebeles J, Green WD, Alwarawrah Y, et al. Obesity‐induced changes in T‐Cell metabolism are associated with impaired memory T‐cell response to influenza and are not reversed with weight loss. J Infect Dis. 2019;219(10):1652‐1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Khan SH, Hemann EA, Legge KL, Norian LA, Badovinac VP. Diet‐induced obesity does not impact the generation and maintenance of primary memory CD8 T cells. J Immunol. 2014;193(12):5873‐5882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Milner JJ, Sheridan PA, Karlsson EA, Schultz‐Cherry S, Shi Q, Beck MA. Diet‐induced obese mice exhibit altered heterologous immunity during a secondary 2009 pandemic H1N1 infection. J Immunol. 2013;191(5):2474‐2485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Liu F, Guo Z, Dong C. Influences of obesity on the immunogenicity of Hepatitis B vaccine. Hum Vaccin Immunother. 2017;13(5):1014‐1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Najarro K, Nguyen H, Chen G, et al. telomere length as an indicator of the robustness of B‐ and T‐cell response to influenza in older adults. J Infect Dis. 2015;212(8):1261‐1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Njajou OT, Cawthon RM, Blackburn EH, et al. Shorter telomeres are associated with obesity and weight gain in the elderly. Int J Obes (Lond). 2012;36:1176‐1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Duong MT, Sahin E. RAP1: protector of telomeres, defender against obesity. Cell Rep. 2013;3(6):1757‐1758. [DOI] [PubMed] [Google Scholar]
- 148. Wang Z, Aguilar EG, Luna JI, et al. Paradoxical effects of obesity on T cell function during tumor progression and PD‐1 checkpoint blockade. Nat Med. 2019;25(1):141‐151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Dorner T, Radbruch A. Antibodies and B cell memory in viral immunity. Immunity. 2007;27(3):384‐392. [DOI] [PubMed] [Google Scholar]
- 150. Ladhani SN, Jeffery‐Smith A, Patel M, et al. High prevalence of SARS‐CoV‐2 antibodies in care homes affected by COVID‐19: prospective cohort study, England. E Clinical Medicine. 2020;28:1‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Trieu MC, Bansal A, Madsen A, et al. SARS‐CoV‐2‐specific neutralizing antibody responses in Norwegian healthcare workers after the first wave of COVID‐19 pandemic: a prospective cohort study. J Infect Dis. 2020;223(4):589‐599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Jeffrey Seow CG, Merrick B, Acors S, et al. Longitudinal evaluation and decline of antibody responses in SARS‐CoV‐2 infection. medRxiv preprint 2020. July.
- 153. Dan JM, Mateus J, Kato Y, et al. Immunological memory to SARS‐CoV‐2 assessed for up to 8 months after infection. Science. 2021;371(6529):1‐22, eabf4063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Ewer KJ, Barrett JR, Belij‐Rammerstorfer S, et al. T cell and antibody responses induced by a single dose of ChAdOx1 nCoV‐19 (AZD1222) vaccine in a phase 1/2 clinical trial. Nat Med. 2021;27(2):270‐278. [DOI] [PubMed] [Google Scholar]
- 155. Wang Z, Schmidt F, Weisblum Y, et al. mRNA vaccine‐elicited antibodies to SARS‐CoV‐2 and circulating variants. bioRxiv. 2021;1‐51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Karlsson EA, Hertz T, Johnson C, Mehle A, Krammer F, Schultz‐Cherry S. Obesity outweighs protection conferred by adjuvanted influenza vaccination. MBio. 2016;7(4):e01144‐16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Liuzzi A, Savia G, Tagliaferri M, et al. Serum leptin concentration in moderate and severe obesity: relationship with clinical, anthropometric and metabolic factors. Int J Obes Relat Metab Disord. 1999;23(10):1066‐1073. [DOI] [PubMed] [Google Scholar]
- 158. Frasca D, Diaz A, Romero M, Blomberg BB. Leptin induces immunosenescence in human B cells. Cell Immunol. 2020;348:1‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Garner‐Spitzer E, Poellabauer EM, Wagner A, et al. Obesity and sex affect the immune responses to tick‐borne encephalitis booster vaccination. Front Immunol. 2020;11:860‐875. [DOI] [PMC free article] [PubMed] [Google Scholar]


