Abstract
Purpose
The correlation between chemerin and diabetic retinopathy (DR) has been demonstrated previously. We aimed to investigate the potential inflammatory and angiogenic roles of chemerin in DR using rat primary retinal microvascular endothelial cells (RRMECs).
Methods
RRMECs were incubated in low- and high-glucose media, and stable chemerin receptor (ChemR23) knockdown in RRMECs was established by lentiviral infection. Real-time quantitative PCR (RT-qPCR), enzyme-linked immunosorbent assay (ELISA), and western blotting were employed to investigate the mRNA and protein expression of intercellular adhesion molecule-1 (ICAM-1), vascular endothelial growth factor (VEGF), tumor necrosis factor-α (TNF-α), and the interleukin-6 receptor (IL-6R) to explore the inflammatory and angiogenic effects of chemerin. A scratch assay was employed to evaluate the effect of chemerin on RRMEC migration.
Results
Chemerin and TNF-α markedly increased the mRNA and protein expression of ICAM-1 in RRMECs (p<0.001). ChemR23 knockdown may have decreased the ICAM-1 expression under low- and high-glucose conditions (p<0.001). Even in the ChemR23-knockdown group, TNF-α significantly increased the mRNA and protein levels of ICAM-1 under low- and high-glucose conditions (p<0.001). Chemerin promoted VEGF expression under low- and high-glucose conditions. ChemR23 knockdown markedly decreased VEGF levels under low- and high-glucose conditions (p<0.05) and significantly decreased RRMEC migration (p<0.001).
Conclusions
Chemerin promotes the expression of ICAM-1, the secretion of VEGF, and the migration of RRMECs via the activation of ChemR23.
Introduction
Diabetic retinopathy (DR) is a common complication in patients with diabetes mellitus. Almost all patients with type 1 diabetes and 60% of patients with type 2 diabetes develop DR 15–20 years after diabetes onset [1]. Globally, in 2010, 2.6% of cases of blindness were caused by DR, and more than 70% of these cases involved moderate or severe vision impairment [2]. Microvascular pathological changes can occur in the early stages of DR, including the prediabetic stage [3]. Mild DR was found in 8.2% of the prediabetic population [1].
The pathophysiological mechanism of DR is highly complex and involves increased vascular endothelial growth factor (VEGF) secretion, oxidative stress, and proinflammatory mediators [4]. Hyperglycemia can induce the expression of chemokines, cytokines, and proangiogenic and proinflammatory growth factors, as well as leukocyte accumulation around the retinal capillary walls, resulting in chronic inflammation; this damages the tight junctions between endothelial cells and leads to the breakdown of the blood–retinal barrier (BRB). Chronic inflammation can damage pericytes and increase serum leakage. The loss of pericytes, damage of tight junctions, and thickening of the endothelial basal membrane can increase vascular permeability, capillary occlusion, retinal ischemia, hypoxia, angiogenic factor release, and retinal neovascularization. The new fragile retinal blood vessels cause vitreous hemorrhage and retinal detachment [5,6]. The expression of many chemokines, inflammatory cytokines, and growth factors increases during the pathogenesis of DR. Hyperglycemia increases the expression of fibronectin (an extracellular matrix protein), collagen, and intercellular adhesion molecule-1 (ICAM-1) [7,8]. Chronic and persistent inflammation occurs during the early stages of diabetes mellitus. Proinflammatory cytokines, such as interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), have been found in the retinas of humans and animal models [9]. These cytokines and growth factors promote the pathological changes in DR.
Chemerin is an adipokine involved in adipogenesis, glucose metabolism, inflammation, and angiogenesis [10]. ChemR23 is found in human endothelial cells, and the chemerin/ChemR23 system is thought to be involved in endothelial angiogenesis [11]. Jiang et al. [12] found that chemerin/ChemR23 promotes the proliferation, adipogenesis, and angiogenesis of preadipocytes by activating the protein kinase B (AKT)/mammalian target of rapamycin (mTOR) and extracellular signal-regulated kinase (ERK) pathways. Kaur et al. [11] found that chemerin can promote the proliferation, differentiation, and migration of endothelial cells and the formation of capillary tubes; they also thought that chemerin regulated angiogenesis through phosphoinositide 3-kinase (PI3K)/Akt and mitogen-activated protein kinase (MAPK)/ERK signaling. Moreover, chemerin is associated with many cytokines and growth factors that are involved in the development of DR. The level of plasma chemerin is elevated in chronic inflammatory diseases, and the expression of chemerin is correlated with that of proinflammatory cytokines [13-15]. High expression of chemerin has been associated with tumor angiogenesis and VEGF upregulation [16,17]. Furthermore, chemerin regulates diabetic microvascular complications. In previous studies, increased serum chemerin levels were found in patients with diabetic nephropathy [18]. Increased vitreous levels of chemerin have also been found in patients with proliferative DR (PDR) [19]. The vitreous chemerin concentration has been positively correlated with the vitreous VEGF concentration. We think that chemerin plays a key role in the development of DR, but its role has not been fully elucidated. Thus, this study aimed to investigate the role of chemerin in DR development and the interplay between chemerin and DR-associated cytokines.
Methods
Cell culture
Rat primary retinal microvascular endothelial cells (RRMECs) were procured from Procell Life Science & Technology Co., Ltd. (catalog no. CP-R114; Wuhan, China) and cultured in non-essential amino acid (minimum essential medium-MEM) with 10% fetal bovine serum (FBS; Gibco BRL, Rockville, MD), 100 U/ml of penicillin G, and 0.1 mg/ml of streptomycin in low (5.5 mM) and high (30 mM) concentrations of glucose. Cells at passages 3–4 were used in this study. RRMECs were stimulated with chemerin (10 nM; Nearshore Protein Technology Co., LTD, Shanghai, China) for 24 h and with TNF-α (30 ng/ml) for 6 h.
ChemR23 knockdown by lentivirus-mediated shRNA
Stable knockdown of ChemR23 in RRMECs was generated using a lentiviral construct. The vector used in this study was pHBLV-U6-MCS-CMV-ZsGreen-PGK-PURO (Hanbio Biotechnology, Shanghai, China). In this study, the siRNA sequence was “T CGC CTG GTC AAT GCT CTA AGT GAA,” and the lentiviral shRNA sequence was as follows: Top strand: GAT CCG TCG CCT GGT CAA TGC TCT AAG TGA ATT CAA GAG ATT CAC TTA GAG CAT TGA CCA GGC GAT TTT TTG; Bottom strand: AAT TCA AAA AAT CGC CTG GTC AAT GCT CTA AGT GAA TCT CTT GAA TTC ACT TAG AGC ATT GAC CAG GCG ACG. The ChemR23-knockdown sequence was inserted into the lentiviral vector to knock down the expression of ChemR23. RRMECs were infected with viral supernatants containing ChemR23-knockdown lentiviral constructs and the corresponding negative control (NC) following the manufacturer’s instructions. Knockdown of ChemR23 in RRMECs was confirmed by western blotting using the corresponding antibody (1:1000; Abcam, Cambridge, MA). Cells were infected for 48 h before further treatment.
RT-qPCR
Total RNA was extracted from the RRMECs using an RNA purification kit (SBS, Beijing, China) according to the manufacturer’s protocol. Add 1ml of Redzol (SBS) to RRMECs, dispersing the pellet and mixing well. Incubate samples at 15 to 30 °C for 5 min. Add 0.2 ml of chloroform (SBS) to the lysate, shake and mix well and incubate for 5 min. Centrifuge at 12,000 × g for 15 min at 4 °C. Transfer the aqueous phase to a fresh tube. Use 0.5 ml of isopropyl alcohol (SBS) per 1 ml of Redzol used for the initial homogenization. Incubate samples at 15 to 30 °C for 10 min and centrifuge at 12,000 xg for 10 min at 4 °C. Remove the supernatant. Wash the RNA pellet once with 75% ethanol. Mix the sample and centrifuge at 7,500 xg for 5 min at 4 °C. Air-dry the RNA pellet for 5-10 min. Dissolve the RNA in 50 μl H2O treated with diethypyrocarbonate (SBS) and incubate at 55-60 °C for 10 min to completely dissolve the RNA. RNA was used to synthesize cDNA using moloney murine leukemia virus (M-MLV) reverse transcriptase (Promega, Madison, WI). The relative expressions of chemerin, ChemR23, TNF-α receptor (TNFR), IL-6 receptor (IL-6R), ICAM-1, VEGF, and pigment epithelium-derived factor (PEDF) were measured using SYBR® Premix Ex TaqTM (Takara, Dalian, China) following the manufacturer’s instructions. A real-time quantitative PCR (RT-qPCR) system (Applied Biosystems, Foster City, CA) was used. The specific primers used in this study are listed in Table 1. Actin was used as the internal control. Relative fold changes of each targeted mRNA were calculated using the 2−ΔΔCt method.
Table 1. The primers used for the real-time quantitative polymerase chain reaction.
| Gene symbol | Gene ID | Forward primers (5′-3′) | Reverse primers (5′-3′) |
|---|---|---|---|
| Actin |
81,822 |
GTGTTGTCCCTGTATGCCTCTGGT |
CTCTTTAATGTCACGCACGATTTCC |
| Chemerin |
297,073 |
TACAATCAAACCAAATGGGAGGAA |
AGGGAAGAAGTAGATGCGGGAGT |
| ChemR23 |
60,669 |
AGGAGACACGCCTGGAGCCTTTG |
TGGAGACTGTGCAGCATGTTTGG |
| TNFR |
25,625 |
TGCGATGCTGTATGCTGTGGTGG |
GGACGAGTGGGCAGGGCTTTCTA |
| IL-6R |
24,499 |
GCGAGGAGTAAAGCATGTGGTGC |
TAGCCAAGGAGTGCCTGTGACCT |
| PEDF |
287,526 |
TATCACCTTAACCGACCGTTCATCT |
TTTCTGTAGCGCACTGGGACATT |
| VEGF |
83,785 |
GAGGAGACTCTTCGAGGAGCACTTT |
TGTCTACAGGAATCCCAGAAACAAAA |
| ICAM-1 | 3383 | AAGTCTGTCAAACGGGAGATGAATGG | TAAATGGACGCCACGATCACGAA |
Western blotting
The RRMECs were lysed using radioimmunoprecipitation assay (RIPA) buffer (Beyotime, Shanghai, China), and the protein concentration was detected using a bicinchoninic acid (BCA) protein assay kit (Thermo, Rockford, USA). The proteins were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) before being transferred to a nitrocellulose membrane (Millipore, Bedford, MA). After blocking with 5% nonfat dry milk, the membranes were incubated with the primary antibodies chemerin (Abcam), ChemR23 (Abcam), TNF-α (Abcam), ICAM-1 (Proteintech, Rosemont, IL), NF-κB (Proteintech), inhibitor of nuclear factor kappa-B alpha (Iκ-Bα; Proteintech), and IL-6R (Proteintech) overnight at 4 °C. The nitrocellulose membranes were incubated with secondary antibodies for 2 h at room temperature. Densitometry was used to detect the protein bands. Tubulin was used as an internal control.
ELISA
VEGF levels in low and high concentrations of glucose were measured using a rat VEGF enzyme-linked immunosorbent assay (ELISA) kit (Kenuodi biotechnology Co., Ltd, Quanzhou, China) according to the manufacturer’s protocol. A microplate reader (Rayto RT6100, Shenzhen, China) was used to determine the optical density at 450 nm.
Cell migration assay
The effect of chemerin on cell migration was evaluated using a scratch test. RRMECs were seeded in a 24-well plate (2×105 cells/well). RRMECs were grown to 90% confluence and transfected with the vector and shChemR23 for 48 h. A 10 μl sterile pipette tip was used to scratch a linear cell-free zone along the diameter of the well. The 24-well plate was washed three times with PBS (1X; 20 mM KCl, 120 mM NaCl, 5 mM KH2PO4, 10 mM Na2HPO4, pH 7.4), and 500 μl of MEM was added. Cell-free zones were photographed after 0 h and 24 h. The migrated area was measured using ImageJ software (Media Cybernetics). Cell migration assays were performed in triplicate.
Statistical analyses
All data are presented as the mean ± standard deviation (SD). SPSS version 20.0 (IBM Corp., Chicago, IL) for Windows was used for statistical analyses. One-way analysis of variance (ANOVA) followed by Tukey’s post hoc analysis was performed to compare multiple groups. Statistical significance was set at p<0.05.
Results
Expression of ChemR23 and receptors of inflammatory factors in RRMECs after high glucose stimulation
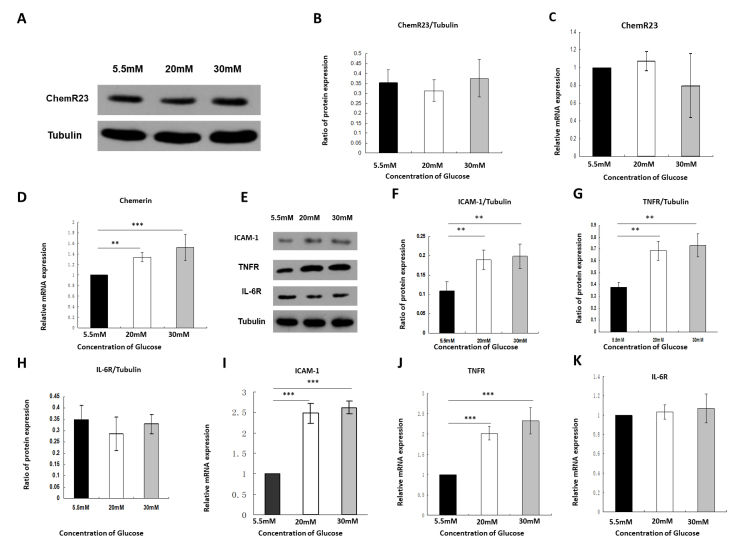
We investigated the expression of ChemR23 in RRMECs under low- and high-glucose conditions. No significant difference was found in the expression of ChemR23 between low- and high-glucose conditions by western blotting (F = 3.268, p = 0.066; Figure 1A,B) and RT-qPCR (F = 2.641, p = 0.104; Figure 1C). High-glucose conditions significantly increased the mRNA expression of chemerin in RRMECs; stimulation with 20 nM and 30 nM glucose increased chemerin mRNA expression by 1.34 ± 0.09-fold and 1.52 ± 0.24-fold, respectively (F = 18.752, p<0.001; Figure 1D). High-glucose conditions markedly increased the protein (F = 19.588, p<0.001) and mRNA (F = 168.75, p<0.001) expression of ICAM-1 (Figure 1E,F,I). TNF-α and IL-6 play key roles in the inflammatory process in DR. We found that the high-glucose conditions significantly increased the protein (F = 36.839, p<0.001) and mRNA (F = 63.869, p<0.001) levels of TNFR (Figure 1E,G,J); stimulation with 20 nM and 30 nM glucose increased the mRNA expression of TNFR by 2.02 ± 0.17-fold and 2.32 ± 0.33-fold, respectively (Figure 1J). However, no significant change was observed in the protein (F = 1.423, p = 0.272) or mRNA (F = 0.797, p = 0.469) levels of IL-6R (Figure 1E,H,K).
Figure 1.
High concentrations of glucose increased chemerin expression, intercellular adhesion molecule-1 (ICAM-1), and tumor necrosis factor-α receptor (TNFR), whereas they had no effect on ChemR23 or interleukin-6 receptor (IL-6R). A,B: Western blot analysis of the expression of ChemR23 under different glucose concentrations. C: Real-time quantitative PCR (RT-qPCR) showed the mRNA expression of ChemR23 under different glucose conditions. D: RT-qPCR showed the mRNA expression of chemerin under different glucose conditions. E-H: Western blot analysis of ICAM-1, TNFR, and IL-6R proteins, normalized to tubulin. I: The expression of ICAM-1 in rat primary retinal microvascular endothelial cells (RRMECs) detected by RT-qPCR. J: The expression of TNFR in RRMECs detected by RT-qPCR. K: The expression of IL-6R in RRMECs detected by RT-qPCR. Data are shown as mean ± standard deviation (SD) of six replicates. **p<0.01, ***p<0.001.
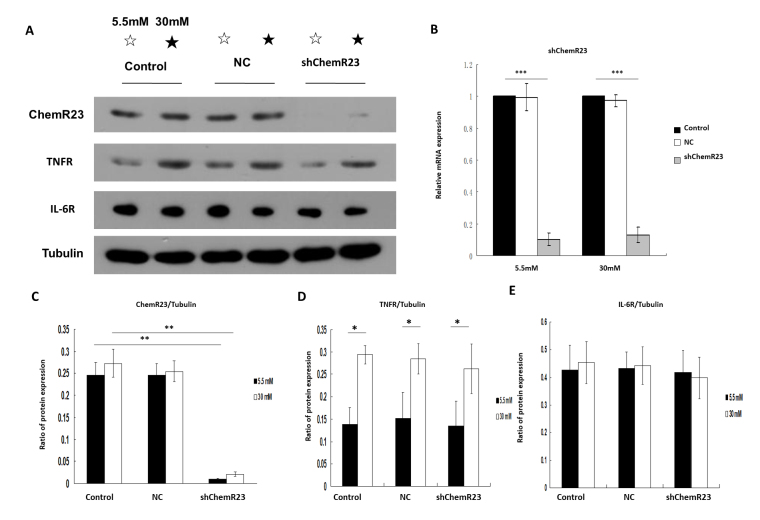
Knockdown of ChemR23 has no effect on the expression of TNFR and IL-6R
The knockdown of ChemR23 was confirmed by western blotting (F = 156.03, p<0.001; Figure 2A,C) and RT-qPCR (Figure 2B) under low- (F = 537.35, p<0.001) and high-glucose conditions (F = 1175.34, p<0.001). The mRNA levels of ChemR23 were markedly decreased under low- and high-glucose conditions in the ChemR23-knockdown group (Flow glucose = 537.35, Plow glucose<0.001; Fhigh glucose = 1175.34, Phigh glucose<0.001; Figure 2B). Significant differences were noted in the protein levels of TNFR between low- and high-glucose in the control, NC, and ChemR23-knockdown groups (p<0.05; Figure 2D). No significant difference was observed in the protein levels of IL-6R under low- and high-glucose conditions (p>0.05; Figure 2E).
Figure 2.
The expression of tumor necrosis factor-α receptor (TNFR) and interleukin-6 receptor (IL-6R) was not affected by ChemR23 knockdown in rat primary retinal microvascular endothelial cells (RRMECs). A and C-E: Western blot analysis of ChemR23, TNFR, and IL-6R proteins, normalized to tubulin under low- (5.5 mM) and high-glucose (30 mM) conditions. B: The expression of ChemR23 in RRMECs detected by real-time quantitative PCR (RT-qPCR). Data are shown as mean ± standard deviation (SD) of six replicates. *p<0.05, **p<0.01, ***p<0.001. Abbreviations: NC, RRMECs transfected with empty vector; shChemR23, RRMECs transfected with shRNA targeting ChemR23.
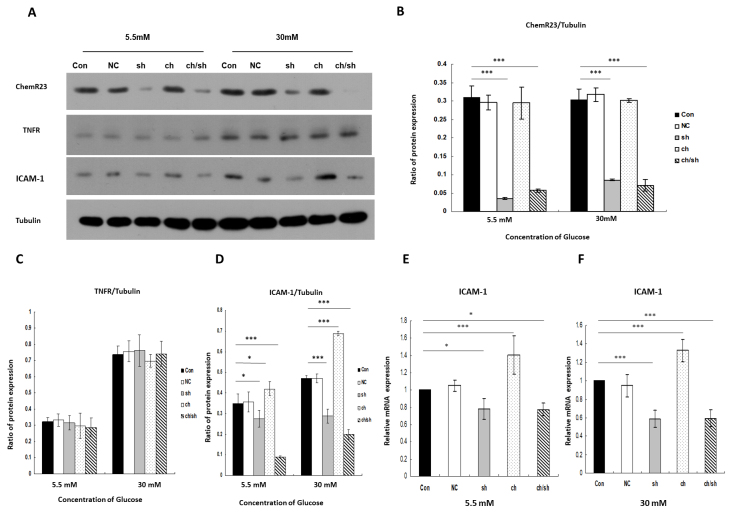
Chemerin and TNF-α increased the expression of ICAM-1 via the activation of ChemR23 and NF-κB
To investigate the role of the chemerin/ChemR23 pathway in DR, we used a lentiviral construct to block the chemerin/ChemR23 pathway. RRMECs were cultured with chemerin for 24 h in two groups—the ch group, where RRMECs were stimulated with chemerin (10 nM) for 24 h before the start of the experiment, and the ch/sh group, where RRMECs were transfected with shRNA targeting ChemR23 and were stimulated with chemerin (10 nM) for 24 h before the start of the experiment. As shown in Figure 3A,D, chemerin significantly increased the protein levels of ICAM-1 under low- and high-glucose conditions (Flow glucose = 60.94, Plow glucose<0.001; Fhigh glucose = 259.65, Phigh glucose<0.001). The mRNA levels of ICAM-1 increased by 1.41 ± 0.22-fold and 1.33 ± 0.12-fold with chemerin stimulation (10 nM for 24 h) under low- and high-glucose conditions, respective, compared with the control (Flow glucose = 23.37, Plow glucose<0.001; Fhigh glucose = 69.93, Phigh glucose<0.001; Figure 3E,F). However, inhibition of the chemerin/ChemR23 pathway markedly decreased the protein levels of ICAM-1 under low- and high-glucose conditions. In the ChemR23-knockdown group with chemerin stimulation (10 nM for 24 h), the protein of ICAM-1 decreased significantly under low- and high-glucose conditions (Flow glucose = 60.94, Plow glucose<0.001; Fhigh glucose = 259.65, Phigh glucose<0.001). In the ChemR23-knockdown group with chemerin stimulation, the mRNA levels of ICAM-1 decreased by 0.78 ± 0.07-fold (p = 0.019) and 0.59 ± 0.09-fold (p<0.001) under low- and high-glucose conditions, respectively (Figure 3E,F). Chemerin did not affect TNFR expression under low- and high-glucose conditions (p>0.05; Figure 3A,C).
Figure 3.
Chemerin/ChemR23 pathway–mediated intercellular adhesion molecule-1 (ICAM-1) expression. A-D: Western blot analysis of ChemR23, tumor necrosis factor-α receptor (TNFR), and ICAM-1 proteins with/without chemerin stimulation (10 nM for 24 h) or ChemR23 knockdown under low- and high-glucose conditions, normalized to tubulin. E: The expression of ICAM-1 in RRMECs detected by real-time quantitative PCR (RT-qPCR) under low-glucose condition (5.5 mM). F: The expression of ICAM-1 in rat primary retinal microvascular endothelial cells (RRMECs) detected by RT-qPCR under high-glucose conditions (30 mM). Data are shown as mean ± standard deviation (SD) of six replicates. *p<0.05, ***p<0.001. Abbreviations: NC, RRMECs transfected with empty vector; sh, RRMECs transfected with shRNA targeting ChemR23; ch, with chemerin stimulation (10 nM for 24 h); ch/sh, RRMECs transfected with shRNA targeting ChemR23 and chemerin stimulation (10 nM for 24 h).
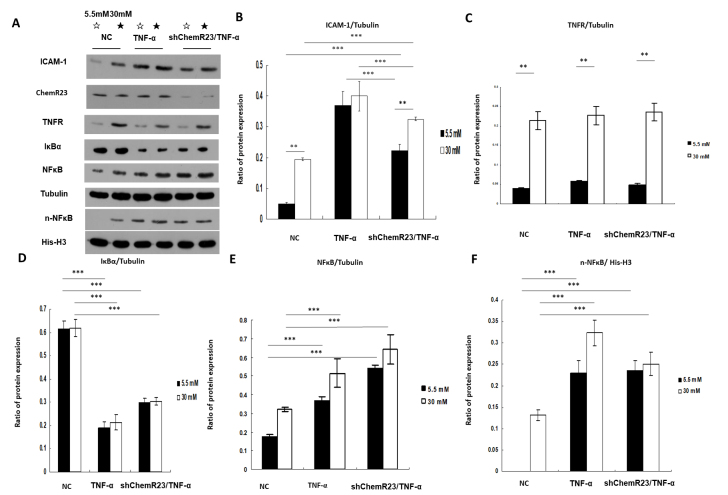
To investigate the role of the TNF-α/TNFR pathway in DR, RRMECs were cultured with TNF-α (30 ng/ml) for 6 h. TNF-α significantly increased the protein levels of ICAM-1 under low- and high-glucose conditions compared with that in the NC group (p<0.001; Figure 4A,B). In the ChemR23-knockdown group, the protein levels of ICAM-1 markedly decreased under low- and high-glucose conditions compared with that in the TNF-α group (Plow glucose<0.001, Phigh glucose = 0.001); however, the ICAM-1 protein levels were higher in the ChemR23-knockdown group than they were in the NC group (Plow glucose<0.001, Phigh glucose<0.001; Figure 4A,B). TNFR expression was upregulated under high-glucose conditions in the three groups (p<0.001; Figure 4A,C).
Figure 4.
Chemerin and tumor necrosis factor-α (TNF-α) both increased intercellular adhesion molecule-1 (ICAM-1) expression via the activation of ChemR23 and nuclear factor-κB (NF-κB), respectively. A-C: Western blot analysis of TNF-α receptor (TNFR) and ICAM-1 proteins with/without TNF-α (30 ng/ml for 6 h) stimulation or ChemR23 knockdown under low- and high-glucose conditions, normalized to tubulin. A,D,E,F: Western blot analysis of inhibitor of nuclear factor kappa-B alpha (Iκ-Bα) and intranuclear NF-κB (n-NF-κB) proteins with/without TNF-α stimulation or ChemR23 knockdown under low- and high-glucose conditions. The protein expression of Iκ-Bα and NF-κB was normalized to tubulin, whereas the protein expression of n-NF-κB was normalized to His-H3. Data are shown as mean ± standard deviation (SD) of six replicates. **p<0.01, ***p<0.001. Abbreviations: NC, RRMECs transfected with empty vector; shChemR23, RRMECs transfected with shRNA targeting ChemR23.
The expression of Iκ-Bα, NF-κB, and intranuclear NF-κB (n-NF-κB) was evaluated using western blotting to investigate the biologic function of TNF-α. Compared with the NC group, TNF-α stimulation decreased the expression of Iκ-Bα (p<0.001; Figure 4A,D) and significantly increased the expression of NF-κB (p<0.001; Figure 4A,E) in the ChemR23-knockdown group compared with the NC group. TNF-α also increased the expression of n-NF-κB in the ChemR23-knockdown group compared with that in the NC group (p<0.001; Figure 4A,F).
Effect of chemerin on VEGF in RRMECs
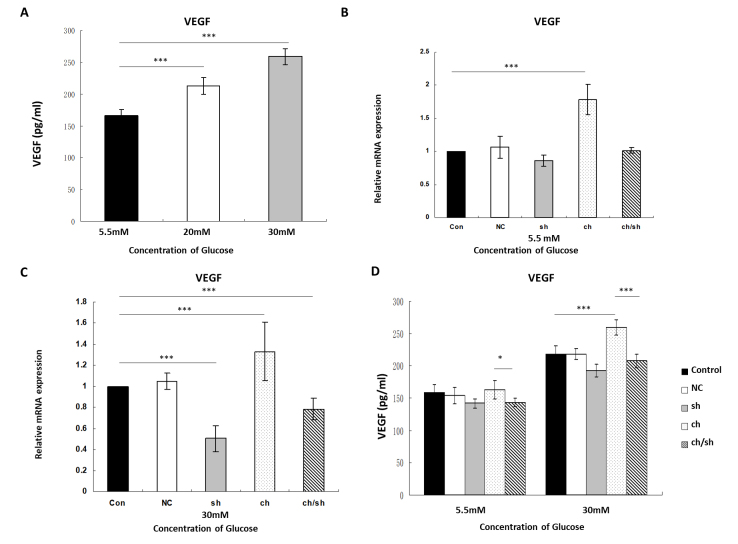
RRMECs were cultured with chemerin (10 nM) for 24 h in the ch and ch/sh groups, as described above. High-glucose levels upregulated VEGF expression (Figure 5A). The RT-qPCR results showed that in the presence of chemerin, the mRNA levels of VEGF were upregulated by 1.78 ± 0.23-fold and 1.33 ± 0.28-fold under low- and high-glucose conditions, respectively (Plow glucose<0.001, Phigh glucose<0.001). After ChemR23 knockdown, the mRNA levels of VEGF were downregulated by 1.01 ± 0.042-fold and 0.78 ± 0.10-fold under low- and high-glucose conditions, respectively (Plow glucose>0.05, Phigh glucose = 0.008; Figure 5B,C).
Figure 5.
Chemerin promotes vascular endothelial growth factor (VEGF) expression via the activation of ChemR23 in RRMECs. A: The expression of VEGF detected by enzyme-linked immunosorbent assay (ELISA) under different glucose conditions (5.5, 20, and 30 mM). B,C: RT-qPCR detected VEGF expression with/without chemerin stimulation (10 nM for 24 h) or ChemR23 knockdown under low- (5.5 mM) and high-glucose (30 mM) glucose conditions. D: VEGF expression detected by ELISA with/without chemerin stimulation (10 nM for 24 h) or ChemR23 knockdown under low- (5.5 mM) and high-glucose (30 mM) conditions. Data were shown as mean ± standard deviation (SD) of six replicates. *p<0.05, **p<0.01, ***p<0.001. Abbreviations: NC, RRMECs transfected with empty vector; sh, RRMECs transfected with shRNA targeting ChemR23; ch, with chemerin stimulation (10 nM for 24 h); ch/sh, RRMECs transfected with shRNA targeting ChemR23 and chemerin stimulation (10 nM for 24 h).
To examine whether the change in VEGF transcription led to the change in translation, ELISA was used to measure the protein expression of VEGF. RRMECs were cultured with chemerin (10 nM) for 24 h in the ch and ch/sh groups, as described above, before ELISA was conducted. Chemerin treatment increased the secretion of VEGF under low-glucose conditions (VEGFlow glucose chemerin = 162.90 ± 14.38 pg/ml, VEGFlow glucose control = 158.47 ± 12.18 pg/ml), and no significant difference was found between the ch group and the control group (p = 0.955). However, under high glucose conditions, VEGF expression was elevated in the presence of chemerin compared to that in the control (VEGFhigh glucose chemerin = 259.50 ± 11.69 pg/ml, VEGFhigh glucose control = 217.91 ± 13.46 pg/ml, p<0.001). ChemR23 knockdown decreased the expression of VEGF under low-glucose conditions compared with that in the chemerin group (VEGFlow glucose chemerin = 162.90 ± 14.38 pg/ml, VEGFlow glucose ChemR23 knockdown group = 143.41 ± 6.42 pg/ml, p = 0.038). Under high-glucose conditions, ChemR23 knockdown significantly decreased the expression of VEGF compared with that in the chemerin group (VEGFhigh glucose chemerin = 259.50 ± 11.69 pg/ml, VEGFhigh glucose control = 207.72 ± 10.11 pg/ml, p<0.001; Figure 5D).
Effect of chemerin on cell migration
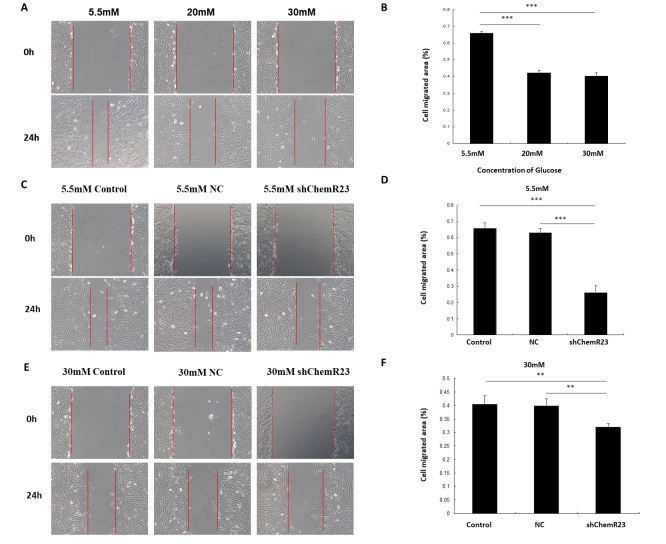
A scratch test was used to investigate the effect of the chemerin/ChemR23 pathway on the migration of RRMECs. High-glucose conditions significantly inhibited cell migration. RRMECs migrated markedly faster under low-glucose conditions than they did under high-glucose conditions (Migrated area5.5mM = 66.02% ± 0.010%, Migrated area20mM = 42.23% ± 0.18%, Migrated area30mM = 40.31% ± 0.20%, p<0.001; Figure 6A,B). Under low-glucose conditions, ChemR23 knockdown inhibited the migration of RRMECs. Cell migration was significantly lower in the ChemR23-knockdown group compared with the control and NC groups (Migrated areaControl = 65.49% ± 0.036%, Migrated areaNC = 62.92% ± 0.027%, Migrated areashChemR23 = 25.96% ± 0.045%, p<0.001; Figure 6C,D). The same result was observed under high-glucose conditions (Migrated areaControl = 40.44% ± 0.031%, Migrated areaNC = 39.80% ± 0.027%, Migrated areashChemR23 = 31.96% ± 0.013%, PControl VS. shChemR23 = 0.006, PNC VS. shChemR23 = 0.009; Figure 6E,F). Cell migration was significantly decreased after ChemR23 knockdown.
Figure 6.
Chemerin promotes the migration of rat primary retinal microvascular endothelial cells (RRMECs) via activation of ChemR23. A,B: The effects of different glucose concentrations (5.5, 20, and 30 mM) on RRMEC migration. C,D: The effect of the chemerin/ChemR23 pathway on RRMEC migration under low-glucose condition (5.5 mM). E,F: The effect of the chemerin/ChemR23 pathway on RRMEC migration under high-glucose condition (30 mM). The representative images were photographed and quantitative analysis of the scraping area was performed at 0 and 24 h after scraping. The experiment was conducted in triplicate. **p<0.01, ***p<0.001. Abbreviations: NC, RRMECs transfected with empty vector; shChemR23, RRMECs transfected with shRNA targeting ChemR23.
Discussion
In the present study, chemerin significantly increased the expression of ICAM-1 via the activation of the chemerin/ChemR23 pathway. Leukostasis plays a key role in microvascular injury in DR [20]. ICAM-1 has been associated with the adherence of activated leukocytes to the retinas of patients with diabetes and in rat models. The ICAM-1 antibody has been found to markedly inhibit leukostasis in rats [8,21,22]. Moreover, elevated chemerin expression is positively correlated with renal inflammation and fibrosis [23,24]. Chemerin can increase the expression of inflammatory factors, such as TNF-α, IL-6, and IL-1β, in nucleus pulposus cells [25]. Our previous study observed elevated serum and vitreous levels of chemerin in patients with diabetes and PDR [19]. In a study by Lu et al. [26], chemerin was positively associated with ICAM-1 expression in patients with diabetes. In this study, chemerin was shown to increase ICAM-1 expression via the chemerin/ChemR23 pathway during the processes of microvascular injury and inflammation in DR.
We found that blocking the chemerin/ChemR23 pathway markedly decreased the expression of ICAM-1, even with TNF-α stimulation. TNF-α is a biomarker of inflammation, which is associated with vascular complications in patients with early stage DR [27,28]. TNF-α induces the adhesion of leukocytes to the endothelial cells and increases reactive oxygen species and BRB breakdown. Previous studies have shown that TNF-α increases the expression of ICAM-1 in endothelial cells [29-31]. However, chemerin levels were positively associated with ICAM-1 in rat models of diabetes and in patients with type 2 diabetes [26,32]. A recent study showed that chemerin upregulated the expression of ICAM-1 via the chemerin/ChemR23 signaling pathway in human umbilical vein endothelial cells [33]. Based on these results, both TNF-α and chemerin may participate in regulating ICAM-1 expression.
Our results showed that stimulation with a high concentration of glucose increased the expression of TNFR and NF-κB and promoted the nuclear translocation of NF-κB in RRMECs. Previous studies have also shown that high concentrations of glucose increase the expression of TNF-α [34]. The binding of TNF-α to TNFR promotes the phosphorylation of Iκ-Bα and the nuclear translocation of NF-κB, which upregulates the expression of E-selectin, VCAM-1, and ICAM-1 [35-37]. Hyperglycemia increases the activity of the TNFR-mediated NF-κB pathway and increases ICAM-1 expression.
The role of chemerin in cell migration has not been fully elucidated. In the present study, we found that chemerin promoted the migration of RRMECs via the activation of ChemR23. Nakamura et al. [38] found that chemerin could promote the differentiation of human umbilical vein endothelial cells into capillary-like structures and induce their migration by activating the Akt and ERK pathways. Knockdown of chemokine-like receptor 1 (CMKLR1) completely suppressed the migration of human umbilical vein endothelial cells. Another study showed that CMKLR1 siRNA effectively inhibited the chemerin-induced migration of vascular smooth muscle cells [39]. Carlino et al. [40] also found that chemerin could stimulate the migration of human natural killer cells through activation of the ERK pathway. However, Li et al. [41] found that chemerin suppressed the migration of hepatocellular carcinoma cells through the downregulation of phospho-protein kinase B (p-Akt) via the CMKLR1-phosphatase and tensin homolog deleted on chromosome ten (PTEN) pathway. Together, these findings necessitate further studies in different cells and tissues.
VEGF plays a key role in the process of angiogenesis in DR. In this study, chemerin stimulation upregulated the expression of VEGF in RRMECs. Elevated levels of VEGF increase vascular permeability and promote BRB breakdown and retinal neovascularization [42,43]. Karthikkeyan et al. [44] found that hyperglycemia can increase VEGF expression in human retinal endothelial cells. Lin et al. [17] revealed that the expression of both chemerin and VEGF increased in rats with diabetic nephropathy and that chemerin and VEGF were positively correlated with inflammatory factors and renal function. Du et al. [45] found that VEGF was positively correlated with the serum levels of chemerin in patients with diabetes. Our previous study also showed that the vitreous chemerin concentration was positively correlated with the vitreous VEGF concentration in patients with PDR [19]. Recent studies have found that chemerin upregulates VEGF through a different pathway. Huang et al. found that chemerin promoted the expression of VEGF via the maternally expressed gene 3- microRNA 217- Dickkopf-related protein 3 (Meg3/miR-217/Dkk3) axis in three T3-L1 preadipocytes [46]. Jiang et al. [12] found that chemerin increased VEGF levels through the Akt/mTOR and ERK1/2 signaling pathways in preadipocytes. Wang et al. found that chemerin-induced VEGF protein expression via the mitogen-activated protein kinase (MEK)/ERK pathway in gastric cancer cells [47]. Therefore, further studies are required.
The roles of the chemerin/ChemR23 pathway in RRMECs identified in this study are summarized in Figure 7. Retinal microvessels are composed of pericytes and endothelial cells. DR is characterized by damage of the tight junctions between endothelial cells, pericyte apoptosis, and BRB breakdown. Secretion of proinflammatory mediators and oxidative stress are also involved in the pathogenic process of DR [4,5]. Only RRMECs were used in this study; since in vitro cell experiments cannot perfectly simulate the complicated in vivo microenvironment associated with DR, further studies using animal models are required.
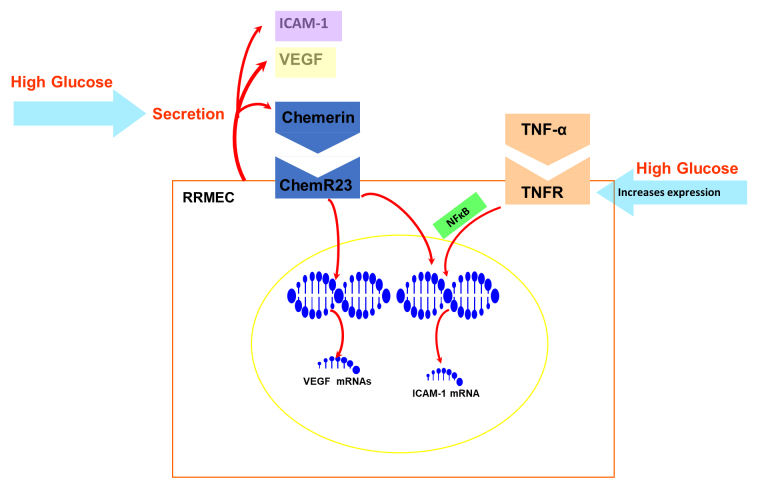
Figure 7.
Schematic summary of chemerin/ChemR23 pathway in rat primary retinal microvascular endothelial cells (RRMECs). A high concentration of glucose increases the secretion of vascular endothelial growth factor (VEGF) and chemerin in RRMECs. High concentration of glucose increases the expression of tumor necrosis factor-α (TNF-α) receptor (TNFR) but has no effect on the expression of ChemR23. Chemerin increases the mRNA and protein expression of VEGF and intercellular adhesion molecule-1 (ICAM-1) via chemerin/ChemR23 pathway. TNF-α can also increase the mRNA and protein expression of ICAM-1 via activation of TNFR and nuclear translocation of nuclear factor-κB (NF-κB). TNF-α and chemerin both regulate ICAM-1 expression.
In conclusion, our study found that chemerin regulates the expression of ICAM-1 via the chemerin/ChemR23 pathway. Both TNF-α and chemerin regulate ICAM-1 expression. Chemerin promoted the migration of RRMECs and VEGF secretion. Therefore, we think that chemerin may promote the angiogenic process in DR by regulating the expression of VEGF. These results provide novel insights into the pathophysiological mechanisms of DR.
Acknowledgments
Song Hui (songh221@hotmail.com) and Li Jun (mike5492@sina.com) are both the co-corresponding authors to this study. This study was supported by Tianjin eye hospital science and technology fund (YKYB2002) and Tianjin health science and technology project (ZC20189). The authors declare no conflict of interest.
References
- 1.Lamparter J, Raum P, Pfeiffer N, Peto T, Höhn R, Elflein H, Wild P, Schulz A, Schneider A, Mirshahi A. Prevalence and associations of diabetic retinopathy in a large cohort of prediabetic subjects: the Gutenberg Health Study. J Diabetes Complications. 2014;28:482–7. doi: 10.1016/j.jdiacomp.2014.02.008. [DOI] [PubMed] [Google Scholar]
- 2.Leasher JL, Bourne RR, Flaxman SR, Jonas JB, Keeffe J, Naidoo K, Pesudovs K, Price H, White RA, Wong TY, Resnikoff S, Taylor HR. Global Estimates on the Number of People Blind or Visually Impaired by Diabetic Retinopathy: A Meta-analysis From 1990 to 2010. Diabetes Care. 2016;39:1643–9. doi: 10.2337/dc15-2171. [DOI] [PubMed] [Google Scholar]
- 3.Ford ES, Zhao G, Li C. Pre-diabetes and the risk for cardiovascular disease: a systematic review of the evidence. J Am Coll Cardiol. 2010;55:1310–7. doi: 10.1016/j.jacc.2009.10.060. [DOI] [PubMed] [Google Scholar]
- 4.Pusparajah P, Lee LH, Abdul Kadir K. Molecular Markers of Diabetic Retinopathy: Potential Screening Tool of the Future? Front Physiol. 2016;7:200. doi: 10.3389/fphys.2016.00200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Semeraro F, Cancarini A, dell’Omo R, Rezzola S, Romano MR, Costagliola C. Diabetic Retinopathy: Vascular and Inflammatory Disease. J Diabetes Res. 2015;2015:582060. doi: 10.1155/2015/582060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Semeraro F, Morescalchi F, Cancarini A, Russo A, Rezzola S, Costagliola C. Diabetic retinopathy, a vascular and inflammatory disease: Therapeutic implications. Diabetes Metab. 2019;45:517–27. doi: 10.1016/j.diabet.2019.04.002. [DOI] [PubMed] [Google Scholar]
- 7.Roy S, Ha J, Trudeau K, Beglova E. Vascular basement membrane thickening in diabetic retinopathy. Curr Eye Res. 2010;35:1045–56. doi: 10.3109/02713683.2010.514659. [DOI] [PubMed] [Google Scholar]
- 8.Miyamoto K, Khosrof S, Bursell SE, Rohan R, Murata T, Clermont AC, Aiello LP, Ogura Y, Adamis AP. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci USA. 1999;96:10836–41. doi: 10.1073/pnas.96.19.10836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Krady JK, Basu A, Allen CM, Xu Y, LaNoue KF, Gardner TW, Levison SW. Minocycline reduces proinflammatory cytokine expression, microglial activation, and caspase-3 activation in a rodent model of diabetic retinopathy. Diabetes. 2005;54:1559–65. doi: 10.2337/diabetes.54.5.1559. [DOI] [PubMed] [Google Scholar]
- 10.Helfer G, Wu QF. Chemerin: a multifaceted adipokine involved in metabolic disorders. J Endocrinol. 2018;238:R79–r94. doi: 10.1530/JOE-18-0174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaur J, Adya R, Tan BK, Chen J, Randeva HS. Identification of chemerin receptor (ChemR23) in human endothelial cells: chemerin-induced endothelial angiogenesis. Biochem Biophys Res Commun. 2010;391:1762–8. doi: 10.1016/j.bbrc.2009.12.150. [DOI] [PubMed] [Google Scholar]
- 12.Jiang Y, Liu P, Jiao W, Meng J, Feng J. Gax suppresses chemerin/CMKLR1-induced preadipocyte biofunctions through the inhibition of Akt/mTOR and ERK signaling pathways. J Cell Physiol. 2018;233:572–86. doi: 10.1002/jcp.25918. [DOI] [PubMed] [Google Scholar]
- 13.Weigert J, Neumeier M, Wanninger J, Filarsky M, Bauer S, Wiest R, Farkas S, Scherer MN, Schäffler A, Aslanidis C, Schölmerich J, Buechler C. Systemic chemerin is related to inflammation rather than obesity in type 2 diabetes. Clin Endocrinol (Oxf) 2010;72:342–8. doi: 10.1111/j.1365-2265.2009.03664.x. [DOI] [PubMed] [Google Scholar]
- 14.Lehrke M, Becker A, Greif M, Stark R, Laubender RP, von Ziegler F, Lebherz C, Tittus J, Reiser M, Becker C, Göke B, Leber AW, Parhofer KG, Broedl UC. Chemerin is associated with markers of inflammation and components of the metabolic syndrome but does not predict coronary atherosclerosis. Eur J Endocrinol. 2009;161:339–44. doi: 10.1530/EJE-09-0380. [DOI] [PubMed] [Google Scholar]
- 15.Neves KB, Nguyen Dinh Cat A, Lopes RA, Rios FJ, Anagnostopoulou A, Lobato NS, de Oliveira AM, Tostes RC, Montezano AC, Touyz RM. Chemerin Regulates Crosstalk Between Adipocytes and Vascular Cells Through Nox. Hypertension. Dallas. 2015;66:657–66. doi: 10.1161/HYPERTENSIONAHA.115.05616. [DOI] [PubMed] [Google Scholar]
- 16.Wang N, Wang QJ, Feng YY, Shang W, Cai M. Overexpression of chemerin was associated with tumor angiogenesis and poor clinical outcome in squamous cell carcinoma of the oral tongue. Clin Oral Investig. 2014;18:997–1004. doi: 10.1007/s00784-013-1046-8. [DOI] [PubMed] [Google Scholar]
- 17.Lin S, Teng J, Li J, Sun F, Yuan D, Chang J. Association of Chemerin and Vascular Endothelial Growth Factor (VEGF) with Diabetic Nephropathy. Med Sci Monit. 2016;22:3209–14. doi: 10.12659/MSM.896781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hu W, Feng P. Elevated serum chemerin concentrations are associated with renal dysfunction in type 2 diabetic patients. Diabetes Res Clin Pract. 2011;91:159–63. doi: 10.1016/j.diabres.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 19.Li J, Hu WC, Song H, Lin JN, Tang X. Increased Vitreous Chemerin Levels Are Associated with Proliferative Diabetic Retinopathy. Ophthalmologica. 2016;236:61–6. doi: 10.1159/000447752. [DOI] [PubMed] [Google Scholar]
- 20.Lutty GA. Effects of diabetes on the eye. Invest Ophthalmol Vis Sci. 2013;54:Orsf81–7. doi: 10.1167/iovs.13-12979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McLeod DS, Lefer DJ, Merges C, Lutty GA. Enhanced expression of intracellular adhesion molecule-1 and P-selectin in the diabetic human retina and choroid. Am J Pathol. 1995;147:642–53. [PMC free article] [PubMed] [Google Scholar]
- 22.Nozaki M, Ogura Y, Hirabayashi Y, Saishin Y, Shimada S. Enhanced expression of adhesion molecules of the retinal vascular endothelium in spontaneous diabetic rats. Ophthalmic Res. 2002;34:158–64. doi: 10.1159/000063660. [DOI] [PubMed] [Google Scholar]
- 23.Zylla S, Pietzner M, Kühn JP, Völzke H, Dörr M, Nauck M, Friedrich N. Serum chemerin is associated with inflammatory and metabolic parameters-results of a population-based study. Obesity (Silver Spring) 2017;25:468–75. doi: 10.1002/oby.21735. [DOI] [PubMed] [Google Scholar]
- 24.Mocker A, Hilgers KF, Cordasic N, Wachtveitl R, Menendez-Castro C, Woelfle J, Hartner A, Fahlbusch FB. Renal Chemerin Expression is Induced in Models of Hypertensive Nephropathy and Glomerulonephritis and Correlates with Markers of Inflammation and Fibrosis. Int J Mol Sci. 2019;20:6240. doi: 10.3390/ijms20246240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hu S, Shao Z, Zhang C, Chen L, Mamun AA, Zhao N, Cai J, Lou Z, Wang X, Chen J. Chemerin facilitates intervertebral disc degeneration via TLR4 and CMKLR1 and activation of NF-kB signaling pathway. Aging. 2020;12:11732–53. doi: 10.18632/aging.103339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lu B, Zhao M, Jiang W, Ma J, Yang C, Shao J, Gu P. Independent Association of Circulating Level of Chemerin With Functional and Early Morphological Vascular Changes in Newly Diagnosed Type 2 Diabetic Patients. Medicine. 2015;94:e1990. doi: 10.1097/MD.0000000000001990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schram MT, Chaturvedi N, Schalkwijk CG, Fuller JH, Stehouwer CD. Markers of inflammation are cross-sectionally associated with microvascular complications and cardiovascular disease in type 1 diabetes–the EURODIAB Prospective Complications Study. Diabetologia. 2005;48:370–8. doi: 10.1007/s00125-004-1628-8. [DOI] [PubMed] [Google Scholar]
- 28.Ibrahim AS, El-Remessy AB, Matragoon S, Zhang W, Patel Y, Khan S, Al-Gayyar MM, El-Shishtawy MM, Liou GI. Retinal microglial activation and inflammation induced by amadori-glycated albumin in a rat model of diabetes. Diabetes. 2011;60:1122–33. doi: 10.2337/db10-1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Aveleira CA, Lin CM, Abcouwer SF, Ambrósio AF, Antonetti DA. TNF-α signals through PKCζ/NF-κB to alter the tight junction complex and increase retinal endothelial cell permeability. Diabetes. 2010;59:2872–82. doi: 10.2337/db09-1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kany S, Vollrath JT, Relja B. Cytokines in Inflammatory Disease. Int J Mol Sci. 2019;20:6008. doi: 10.3390/ijms20236008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xue M, Qiqige C, Zhang Q, Zhao H, Su L, Sun P, Zhao P. Effects of Tumor Necrosis Factor α (TNF-α) and Interleukina 10 (IL-10) on Intercellular Cell Adhesion Molecule-1 (ICAM-1) and Cluster of Differentiation 31 (CD31) in Human Coronary Artery Endothelial Cells. Med Sci Monit. 2018;24:4433–9. doi: 10.12659/MSM.906838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hu W, Yu Q, Zhang J, Liu D. Rosiglitazone ameliorates diabetic nephropathy by reducing the expression of Chemerin and ChemR23 in the kidney of streptozotocin-induced diabetic rats. Inflammation. 2012;35:1287–93. doi: 10.1007/s10753-012-9440-y. [DOI] [PubMed] [Google Scholar]
- 33.Zhang RL, Wang QQ, Yang LJ. Chemerin induced by Treponema pallidum predicted membrane protein Tp0965 mediates the activation of endothelial cell via MAPK signaling pathway. J Cell Biochem. 2019;120:19621–34. doi: 10.1002/jcb.29269. [DOI] [PubMed] [Google Scholar]
- 34.Wu D, Jin L, Xu H. The Effects of the CXCR4 Antagonist, AMD3465, on Human Retinal Vascular Endothelial Cells (hRVECs) in a High Glucose Model of Diabetic Retinopathy. Med Sci Monit. 2019;25:6946–54. doi: 10.12659/MSM.917186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu H, Tang J, Lee CA, Kern TS. Metanx and early stages of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2015;56:647–53. doi: 10.1167/iovs.14-15220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kong DH, Kim YK, Kim MR, Jang JH, Lee S. Emerging Roles of Vascular Cell Adhesion Molecule-1 (VCAM-1) in Immunological Disorders and Cancer. Int J Mol Sci. 2018;19:1057. doi: 10.3390/ijms19041057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhou Z, Connell MC, MacEwan DJ. TNFR1-induced NF-kappaB, but not ERK, p38MAPK or JNK activation, mediates TNF-induced ICAM-1 and VCAM-1 expression on endothelial cells. Cell Signal. 2007;19:1238–48. doi: 10.1016/j.cellsig.2006.12.013. [DOI] [PubMed] [Google Scholar]
- 38.Nakamura N, Naruse K, Kobayashi Y, Miyabe M, Saiki T, Enomoto A, Takahashi M, Matsubara T. Chemerin promotes angiogenesis in vivo. Physiol Rep. 2018;6:e13962. doi: 10.14814/phy2.13962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kunimoto H, Kazama K, Takai M, Oda M, Okada M, Yamawaki H. Chemerin promotes the proliferation and migration of vascular smooth muscle and increases mouse blood pressure. Am J Physiol Heart Circ Physiol. 2015;309:H1017–28. doi: 10.1152/ajpheart.00820.2014. [DOI] [PubMed] [Google Scholar]
- 40.Carlino C, Trotta E, Stabile H, Morrone S, Bulla R, Soriani A, Iannitto ML, Agostinis C, Mocci C, Minozzi M, Aragona C, Perniola G, Tedesco F, Sozzani S, Santoni A, Gismondi A. Chemerin regulates NK cell accumulation and endothelial cell morphogenesis in the decidua during early pregnancy. J Clin Endocrinol Metab. 2012;97:3603–12. doi: 10.1210/jc.2012-1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li JJ, Yin HK, Guan DX, Zhao JS, Feng YX, Deng YZ, Wang X, Li N, Wang XF, Cheng SQ, Bao Y, Xie D. Chemerin suppresses hepatocellular carcinoma metastasis through CMKLR1-PTEN-Akt axis. Br J Cancer. 2018;118:1337–48. doi: 10.1038/s41416-018-0077-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Whitehead M, Wickremasinghe S, Osborne A, Van Wijngaarden P, Martin KR. Diabetic retinopathy: a complex pathophysiology requiring novel therapeutic strategies. Expert Opin Biol Ther. 2018;18:1257–70. doi: 10.1080/14712598.2018.1545836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rodríguez ML, Pérez S, Mena-Mollá S, Desco MC, Ortega ÁL. Oxidative Stress and Microvascular Alterations in Diabetic Retinopathy: Future Therapies. Oxid Med Cell Longev. 2019;2019:4940825. doi: 10.1155/2019/4940825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Karthikkeyan G, Nareshkumar RN, Aberami S, Sulochana KN, Vedantham S, Coral K. Hyperglycemia induced early growth response-1 regulates vascular dysfunction in human retinal endothelial cells. Microvasc Res. 2018;117:37–43. doi: 10.1016/j.mvr.2018.01.002. [DOI] [PubMed] [Google Scholar]
- 45.Du J, Li R, Xu L, Ma R, Liu J, Cheng J, Zhang Z, Sun H. Increased Serum Chemerin Levels in Diabetic Retinopathy of Type 2 Diabetic Patients. Curr Eye Res. 2016;41:114–20. doi: 10.3109/02713683.2015.1004718. [DOI] [PubMed] [Google Scholar]
- 46.Huang X, Fu C, Liu W, Liang Y, Li P, Liu Z, Sheng Q, Liu P. Chemerin-induced angiogenesis and adipogenesis in 3 T3–L1 preadipocytes is mediated by lncRNA Meg3 through regulating Dickkopf-3 by sponging miR-217. Toxicol Appl Pharmacol. 2019;385:114815. doi: 10.1016/j.taap.2019.114815. [DOI] [PubMed] [Google Scholar]
- 47.Wang C, Wu WK, Liu X, To KF, Chen GG, Yu J, Ng EK. Increased serum chemerin level promotes cellular invasiveness in gastric cancer: a clinical and experimental study. Peptides. 2014;51:131–8. doi: 10.1016/j.peptides.2013.10.009. [DOI] [PubMed] [Google Scholar]