Abstract
Upper gastrointestinal bleeding from oesophageal or gastric varices is an important medical condition in patients with portal hypertension. Despite the emergence of a number of novel endoscopic and radiologic therapies for oesophagogastric varices, controversy exists regarding the indication, timing and modality of therapy. The aim of this review is to provide a concise and practical evidence-based overview of these issues.
Keywords: Upper gastrointestinal bleeding, Portal hypertension, Gastric varices, Variceal band ligation, Variceal obliteration, Sclerotherapy, Transjugular intrahepatic portosystemic shunt, Balloon-occlusion retrograde transvenous obliteration
Core Tip: Gastric varices are an uncommon source of bleeding in patients with portal hypertension. Although evidence supports acute bleeding treatment and secondary prophylaxis using interventional endoscopy or radiology, there is still lack of data to support primary prophylaxis for all patients. If treatment is required, both interventional endoscopy and radiological approaches should be considered. Interventional endoscopy using endoscopic ultrasound-guided combination coil and cyanoacrylate obliteration appears to be the optimal approach based on the current literature.
INTRODUCTION
Incidence
The incidence of gastric varices (GV) is 15%-20% from endoscopic epidemiological studies of portal hypertension[1,2]. Unlike oesophageal varices that tend to be present in the lamina propria mucosae and superficial submucosa, GV lie deep in the submucosa and as such can be difficult to differentiate from prominent gastric rugae with standard endoscopy. Endoscopic ultrasonography studies have demonstrated that a proportion of GV are undiagnosed on standard diagnostic endoscopy[3,4]. However, this may not be clinically significant, as the size of the varix is one of the characteristics that predict risk of haemorrhage, and larger GV are less likely to remain undetected by standard endoscopy.
Both GV and oesophageal varices develop as a consequence of portal hypertension. Portal hypertension may lead to reversal of flow through the portal circulation, with two common outlets - via the coronary (gastric) vein to the right and left gastric veins, and via the splenic vein to the short and posterior gastric veins[5]. The former supply the distal oesophagus and cardia of the stomach where transmitted pressures and increased flow lead to formation of oesophageal and cardio-oesophageal varices. The latter supply the fundus whereby increased pressures and flow through this system leads to the formation of fundal varices. In a haemodynamic study of oesophageal and and GV by Watanabe et al[5], 78% of patients with portal hypertension had the majority of collateral flow through the left and right gastric veins, likely accounting for the difference in incidence between oesophageal and GV.
In the same study, GV were demonstrated to bleed at lower portal pressures than oesophageal varices, largely due to the higher prevalence of gastro-renal shunts in those with GV. These shunts decompress the portal system. This finding has since been confirmed in further studies[6,7].
GV CLASSIFICATION
Sarin and Kumar[8] seminal paper on the anatomical classification of GV from 1989 remains the most widely accepted method for describing GV. They are divided into two groups, with further sub-classification (Figure 1): (1) Gastroesophageal varices (GOV) – are continuation of oesophageal varices that extend beyond the gastroesophageal junction. These are divided into: (a) Type 1 (GOV1) – those that extend along the lesser curve of the stomach. These account for 75% of all GV[2]; (b) Type 2 (GOV2) – those that extend along the greater curve of the stomach into the fundus; (2) Isolated GV (IGV) – occur in the absence of oesophageal varices and are sub-classified into: (a) Type 1 (IGV1) – located in the fundus and do not extend to the cardia. They are also called fundal varices; (b) Type 2 (IGV2) – can occur anywhere in the stomach (i.e., body, antrum, pylorus). These are rare, occurring in < 5% of those with GV.
Figure 1.

Classification of gastric varices according to Sarin et al[2]. Citation: Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: A long-term follow-up study in 568 portal hypertension patients. Hepatology 1992; 16: 1343-1349. Copyright © The Authors 1992. Published by John Wiley and Sons.
The use of this classification system has been shown to predict risk of bleeding and guides management. GOV1 varices behave similarly to oesophageal varices, and so the treatment paradigm for prophylaxis and acute variceal haemorrhage is the same for oesophageal varices. IGV1 and GOV2 varices are more difficult to control when they bleed compared with GOV1 varices and portend a poorer prognosis[2].
An alternate classification, published by Hashizume et al[9], is a more detailed examination of GV describing the form (tortuous, nodular or tumorous), location (anterior, posterior, lesser curve or greater curve of cardia, or fundic), and colour (red or white) of the varix (Figure 2). Similar to classification systems for oesophageal varices, this classification is aimed at stratifying patients at highest risk of bleeding. In a stepwise logistic regression analysis, those with varices in the anterior or greater curve of the cardia, nodular appearance (i.e., larger size), or red colour spot had the highest predicted risk for bleeding[9]. In another study, which focused on patients with fundic varices, increased size and presence of a red spot increased risk of haemorrhage[10]. Advanced liver disease, as determined by the Child-Turcotte-Pugh classification, was an additional risk factor for bleeding[10]. Other classification systems exist[11], however are used less frequently than those described by Sarin and Kumar[8], and Hashizume et al[9].
Figure 2.
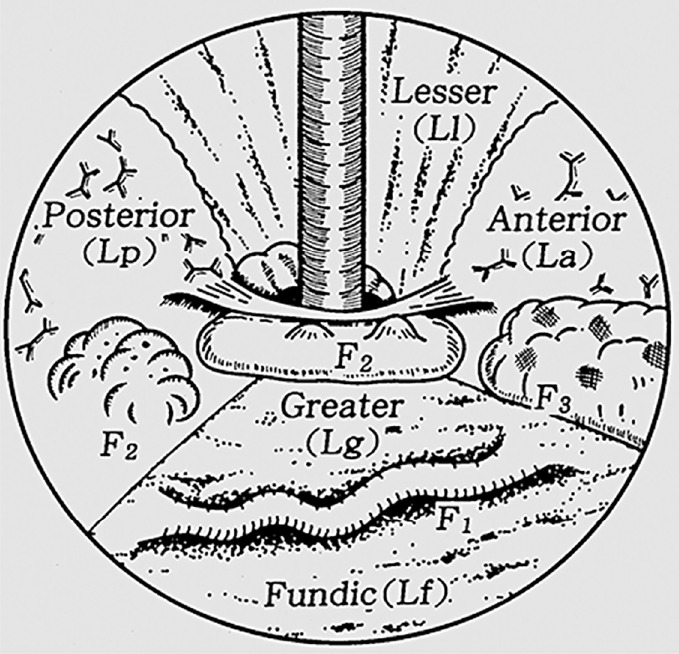
Classification of gastric varices according to Hashizume et al[9]. Citation: Hashizume M, Kitano S, Yamaga H, Koyanagi N, Sugimachi K. Endoscopic classification of gastric varices. Gastrointestinal Endoscopy 1990; 36: 276-280. Copyright © The Authors 1990. Published by Elsevier.
ACUTE GASTRIC VARICEAL HAEMORRHAGE
Bleeding from GV is considered definite if there is active spurting or oozing from the varix or an adherent clot or fibrin plug on the varix. GV bleeding should be considered the cause of upper gastrointestinal bleeding when a GV without high-risk stigmata is present in the absence of oesophageal varices or an alternate source of bleeding[12].
Pre-endoscopic management
Pre-endoscopic management follows that for oesophageal variceal bleeding, namely use of splanchnic vasoconstrictors (i.e., terlipressin or octreotide) to reduce portal pressures, prophylactic antibiotics (e.g., ceftriaxone), and a restrictive transfusion protocol[13,14]. The same medical treatment is instituted in patients with a presumed diagnosis of variceal haemorrhage with known portal hypertension who present with symptoms of upper gastrointestinal bleeding.
Endoscopic management
As outlined above, GOV1 varices are considered an extension of oesophageal varices and so at the time of haemorrhage should be treated in the same manner as bleeding oesophageal varices [i.e., endoscopic variceal band ligation (EVBL), Figure 3][2,12-14]. One small prospective randomized controlled trial (RCT) reported numerically higher haemostasis and lower re-bleeding rates in the subset of patients with GOV1 haemorrhage treated with endoscopic variceal obturation (EVO) rather than EVBL[15], although this did not reach statistical significance. Both treatments may be considered equally efficacious for GOV1 varices.
Figure 3.
Endoscopic variceal banding of gastroesophageal varices 1. A: Pre-banding forward view of varix; B: Pre-banding retroflexed view of varix; C: Immediately post-banding of varix.
Current guidelines support the use of cyanoacrylate injection – either as N-butyl-2-cyanoacrylate (e.g., Histoacryl®) or 2-octyl-cyanoacrylate (e.g., Dermabond) – for acutely bleeding fundic varices (GOV2 and IGV1), in a procedure termed EVO[13,14]. Cyanoacrylate is a tissue adhesive that rapidly polymerizes upon contact with water/blood, leading to a change in the liquid composition to one of a hard brittle acrylic plastic. Majority evidence for its use stems from uncontrolled retrospective and prospective studies[16], with one RCT demonstrating a statistically non-significant increased haemostasis rate when compared to alcohol-based sclerotherapy (89% vs 62%)[17]. Haemostasis rates for cyanoacrylate glue injection are 80%-100%, with re-bleeding rates of 10%-60%[16]. Complications are rare, with fever and pain being the most common, while the most feared is embolization of the glue into systemic beds that can lead to ischaemia in those tissues (e.g., stroke, myocardial infarction, splenic infarction, pulmonary embolus).
Thrombin injection has been utilized as an alternative to cyanoacrylate for EVO for almost three decades; however, like cyanoacrylate, evidence for its use is taken from small, uncontrolled studies[16]. It appears safe, with few adverse procedure-related outcomes, and haemostasis and re-bleeding rates similar to cyanoacrylate.
Sclerotherapy, the injection of a sclerosant agent into the varix, has gone out of favour for the treatment of gastric variceal haemorrhage due to unacceptably high re-bleeding rates of up to 90%-100%[16]. This is often due to ulceration at the point of injection resulting from the high volume of sclerosant required to obliterate GV, with a large amount of sclerosant flowing away from the variceal bed via co-existent gastro-renal shunts that occur with high prevalence in patients with GV[16]. Adverse events include fever, and retrosternal and abdominal pain.
IGV-2 varices are rare; hence little evidence exists as to the optimal endoscopic management. In general, it is accepted that they should be treated according to GOV2/IGV1 varices with EVO.
Salvage therapy
Balloon tamponade with Sengstaken-Blakemore, Minnesota or Linton-Nachlas tubes can be utilized in patients with bleeding from GOV1, GOV2 and IGV1 varices. They will not be effective for IGV2 varices, given the ectopic location of the culprit lesion. The Linton-Nachlas tube may be preferred in gastric variceal haemorrhage if available, as the gastric balloon has greater volume capacity[12].
Transjugular intrahepatic portosystemic shunt (TIPS) (Figure 4) is an effective salvage therapy for patients with endoscopically-uncontrollable bleeding oesophageal varices, however its utility in refractory gastric variceal haemorrhage is less clear[12]. Although haemostasis rates exceed 90%, re-bleeding is reported to occur in 15%-30%[18-20] and concerns remain over post-TIPS encephalopathy, which can be recalcitrant to standard medical therapy and necessitate revision of the TIPS. Several hypotheses have been proposed to explain the risk of re-bleeding[21,22]; ‘Proximity’ theory – feeding vessels to GV lie further away from a TIPS shunt than feeding vessels to oesophageal varices, hence the shunt is less effective in decompressing GV; ‘Throughput’ theory – gastro-renal shunts that occur in high frequency in association with bleeding GV compete with the TIPS for portal flow and can continue to feed the gastric variceal bed; ‘Recruitment’ theory – development of new feeder vessels after proximal embolization of a GV.
Figure 4.
Transjugular intrahepatic portosystemic shunt decompressing gastric varix. A: Pre-transjugular intrahepatic portosystemic shunt (TIPS) with contrast toward gastric variceal bed; B: Post-TIPS with no contrast flow toward gastric variceal bed.
In retrospective comparison studies, Mahadeva et al[23] found TIPS to be more effective in preventing re-bleeding when compared with EVO in acute gastric variceal haemorrhage, whilst Procaccini et al[24] found no difference between the two modalities.
Balloon-occlusion retrograde transvenous obliteration (BRTO) and its modifications (coil-assisted or plug-assisted retrograde transvenous obliteration) aim to sclerose a varix without treating portal hypertension. BRTO is often reserved for use in patients with anatomy not amenable for TIPS or where TIPS is contraindicated (i.e., past history of hepatic encephalopathy or advanced synthetic liver dysfunction), and is reliant on the presence of a gastro-renal shunt for technical feasibility. Similar to TIPS, it is highly effective in achieving haemostasis with success rates > 90%[21,25]. A meta-analysis of uncontrolled studies reported a clinical success rate of 97%, defined as no GV recurrence or re-bleed of acutely bleeding GV or no bleed in the case of at-risk GV that have never-bled[26]. The main sclerosant used for BRTO in reported studies was ethalonamine oleate, in 94% of cases, and the most common side effect was haematuria, occurring in 70%[26]. Given ethalonamine oleate is a known cause of haemolysis, the common occurrence of haematuria is somewhat expected from consequent haemoglobinuria. The antidote for this is parenteral administration of haptoglobin. Although this review by Park et al[26] included patients who underwent BRTO for acute treatment of variceal haemorrhage, primary prophylaxis and secondary prophylaxis, a breakdown of indication was not provided and subgroup analysis for this purpose not available. Another, more recently published meta-analysis[27] found that there was no significant difference in immediate haemostasis rates between the two procedures, but a higher re-bleeding rate post-TIPS [relative risk (RR) 2.61, 95% confidence interval (CI) 1.75–3.90, P < 0.01], higher post-procedural hepatic encephalopathy rate post-TIPS (RR 16.11, 95%CI: 7.13–36.37, P < 0.01), statistically non-significant higher rate of ascites in the BRTO group and statistically non-significant worsening in Child-Pugh status in those who received TIPS. Apart from a small pilot study out of Seoul, Korea[28] that randomly assigned 14 patients with acutely bleeding GV to up-front TIPS or BRTO, there have not been any head-to-head RCTs to ascertain the difference in safety and efficacy between these procedures in patients with acutely bleeding GV. A disadvantage of BRTO is that it can lead to the development or worsening of non-GV (oesophageal or ectopic) as portal blood flow is diverted through alternate pathways, or exacerbation or new development of ascites due to raised portal pressures. This is not an issue post-TIPS, which effectively decompresses the portal system.
SECONDARY PROPHYLAXIS
Non-selective beta-blockers
No controlled study has demonstrated efficacy of non-selective beta-blockers (NSBB) for secondary prophylaxis following gastric variceal bleeding. In a RCT by Mishra et al[29], EVO was far more effective in preventing re-bleeding than propranolol, with a relative risk reduction of 80% and absolute risk reduction of 35%. Hung et al[30] and Chen et al[31] explored the adjunct use of propranolol or carvedilol, respectively, to 3-4 weekly EVO alone following gastric variceal haemorrhage in an RCT setting, albeit with no placebo arm. Neither found a difference in gastric variceal re-bleeding rates between the two groups. Of note is that both studies were conducted in the same institution with similar inclusion and exclusion criteria, except the more recent study by Chen et al[31] included all patients with any form of gastric variceal bleeding, whilst Hung et al[30] only included patients with fundic variceal haemorrhage (GOV2 or IGV1). The study by Chen et al[31] did demonstrate a significant reduction in all-cause upper gastrointestinal re-bleeding in the group assigned to carvedilol (28% vs 48%, P = 0.03), driven by a reduction in bleeding from portal hypertensive gastropathy, and oesophageal and gastric ulcers. However, the authors also reported on a higher incidence of adverse events in the carvedilol group (53% vs 15%, P < 0.001), due to more frequent dizziness and exertional dyspnoea. Despite this, no patient in the carvedilol group discontinued therapy. The lack of efficacy with NSBB following gastric variceal haemorrhage is postulated to be due to the fact that GVs bleed at lower portal pressures, with NSBB having little effect on preventing flow of blood to the culprit variceal bed via co-existent gastro-renal shunt[30,31].
Endoscopic therapy
Although endoscopic ultrasound (EUS) guided injection of cyanoacrylate reduces embolic complication rates, as a result of reduced volume of cyanoacrylate injected, its use during acute gastric variceal haemorrhage is limited in most centres due to access to endoscopists with expertise in EUS. However, it is an emerging therapy for secondary prophylaxis. A two-part observational comparative study by Lee et al[32] compared fortnightly EUS-guided injection of cyanoacrylate in patients presenting with acute bleeding from any type of GV with “on demand” therapy, whereby standard endoscopy and injection of cyanoacrylate was only undertaken at the time of re-bleeding. A significant reduction in re-bleeding was demonstrated in the active endoscopic treatment group (35% vs 70%, P = 0.0006). There was no impact on mortality, likely due to the small number of patients in the study. In a similar cohort trial design, Bick et al[33] found that there was a lower gastric variceal and all-cause upper gastrointestinal re-bleeding rate (9% vs 24%, P = 0.045 and 19% vs 50%, P < 0.001, respectively) in those managed with EUS-guided cyanoacrylate injection compared with standard endoscopy guided injection. It is important to note that the standard endoscopy cohort had a higher mean MELD (17 vs 13, P = 0.004) and lower incidence of IGV1 varices (8% vs 47%), with the latter likely accounted for by the greater sensitive of EUS for the detection of GV.
A novel endoscopic method that is gaining popularity globally is EUS-guided coiling of GV, which involves injection of metal embolization micro-coils coated with synthetic stainless steel-fibres, leading to turbulent blood flow and intravariceal clot formation to obliterate the varix[34]. This can be combined with injection of cyanoacrylate glue (Figure 5), in a procedure that aims to prevent systemic embolization of the cyanoacrylate, as the coils may provide a scaffold for polymerization, as well as requiring less volume of glue injection as a result of precise delivery into the target tributary[35]. The additional benefit of EUS over standard endoscopy is the ability for immediate post-treatment Doppler evaluation of the variceal bed and its afferent tributaries, to ensure complete obliteration[34,35]. In a retrospective, multicentre study by Romero-Castro et al[36] comparing outcomes of 30 patients who underwent EUS-guided coil (n = 11) with those who underwent EUS-guided cyanoacrylate injection (n = 19) into GVs that had previously bled (n = 23) or never bled (n = 7), both methods were highly effective in obliterating the varices (96.7% cumulatively) without a difference in re-bleeding rate. The cyanoacrylate group required more sessions to achieve obliteration (29 sessions vs 14 sessions, P = 0.29) and had lesser proportion of patients achieving variceal obliteration after a single endoscopic session (18% vs 82%), whilst also having a higher reported adverse event rate (58% vs 9%, P < 0.01). However, the majority of adverse events were asymptomatic pulmonary emboli detected on routine computed tomography (CT) of the chest of patients post-procedure, with no difference noted in the symptomatic adverse event rate between groups. It is also noteworthy that a statistically significant higher proportion of patients with bleeding varices and Child-Pugh C status cirrhosis were represented within the cyanoacrylate group in this study.
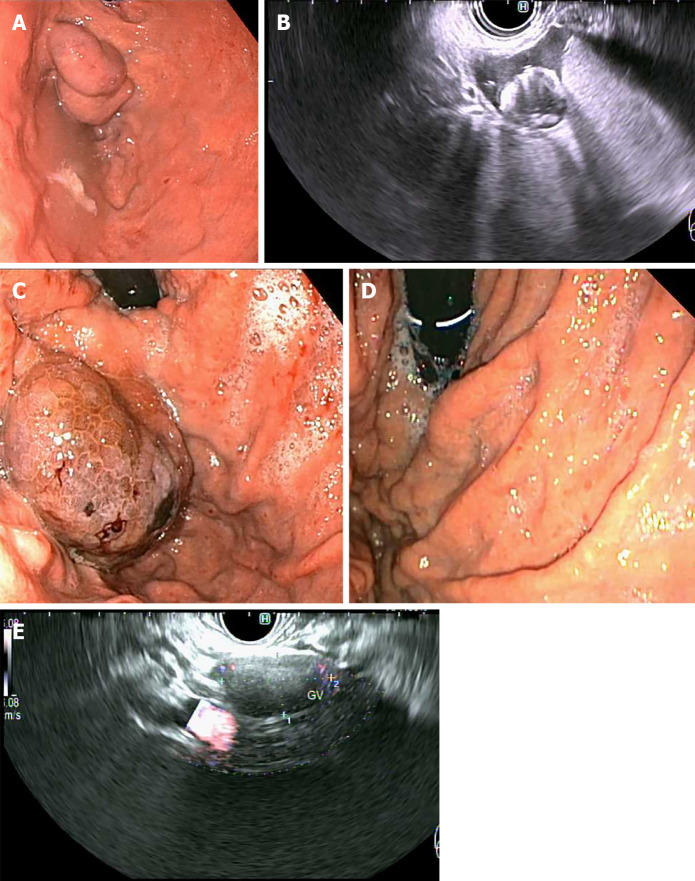
Figure 5.
Primary prophylaxis of isolated gastric varices 1 with five 0.18 10 mm micronester coils in combination 1 mL cyanoacrylate glue. A: Pre-treatment endoscopic appearance; B: Endoscopic ultrasound (EUS) appearance immediately post-deployment of coils; C: Endoscopic appearance 1-mo post-treatment; D: Complete obliteration 4-mo post-treatment; E: EUS appearance of varix with no Doppler flow 4-mo post-treatment.
Binmoeller et al[37] were the first to publish on the efficacy and safety of combined EUS-guided coil and cyanoacrylate injection for GV, predominantly in patients who had recovered from an acute gastric variceal haemorrhage (n = 28/30). The same group reported on a more extensive patient cohort (n = 152) some years later, with high obliteration rate (93%) at follow-up endoscopy, low re-bleeding rate (16%, with 50% re-bleeding events non-variceal in origin), and few procedure-related adverse events (7%; 4/9 patients with abdominal pain, 1/9 patients with pulmonary embolus)[38]. Of note, 26% of patients in this study underwent treatment as primary prophylaxis, somewhat unique in the GV treatment evidence base. A single randomized trial has been performed evaluating combination coiling and cyanoacrylate injection with coiling alone, reporting a higher variceal disappearance rate on immediate post-procedure endoscopy (87% vs 13%, P < 0.001), and lower re-bleeding rate (3.3% vs 20%, P = 0.04), variceal reappearance rate on follow-up endoscopy at 3-mo (13% vs 47%, P < 0.001), and re-intervention rate (17% vs 40%, P = 0.045) in the arm allocated to combination therapy[39]. The cumulative mortality rate of 28% from this study despite relatively preserved liver function in participants (90% Child Pugh A, median MELD 9.5 at enrolment) is of concern and somewhat unexplained, particularly given 10/17 patients died from uncontrolled haemorrhage and 9/10 of these were variceal in nature.
Interventional radiology
Only a single RCT has evaluated EVO with up-front TIPS for secondary prophylaxis of gastric variceal haemorrhage[40], and revealed a 71% relative risk reduction over 3 years in gastric variceal re-bleeding rate in the TIPS arm, albeit with 26% of TIPS patients suffering from hepatic encephalopathy. This study pre-dates the era of EUS-guided coils and very few patients in this study had IGV1 varices. TIPS may be an attractive option in patients with concurrent ascites and/or presence of other non-GV, but less so in those with a history of prior encephalopathy or advanced synthetic liver dysfunction.
A single prospective, non-randomized study[41] and two retrospective, observational studies[42,43] have demonstrated a lower re-bleeding rate in patients treated with BRTO rather than EVO for secondary prophylaxis of variceal haemorrhage (3%-15% vs 22%-71%). They each had differing inclusion criteria, with the prospective study including patients with GOV1, GOV2 and IGV1 varices, whilst the two retrospective studies both excluded patients with GOV1 varices, and one only included patients with IGV1 varices[42].
Contemporary case series have begun exploring the feasibility and safety of combined interventional radiological procedures, namely TIPS with balloon-occluded transvenous obliteration (whether in an antegrade or retrograde fashion)[44-46]. Purported benefits from retrospective audits of combined procedures are reduced re-bleeding and post-procedure encephalopathy rates, stable or improved liver function, and prevention or improvement of ascites. Finally, percutaneous transhepatic obliteration is an alternate route to obliteration of a gastric varix in those without a gastro-renal shunt and who may have contraindication to TIPS[47].
Given the wide array of therapeutic options available are reliant on specific anatomical features, such as feeding vessels into the variceal bed or presence of a gastro-renal shunt, appropriate imaging of the portomesenteric circulation with CT should be attained in patients with GV to allow the most anatomically suitable intervention to be chosen.
PRIMARY PROPHYLAXIS
Whilst there is a modest evidence-base for secondary prophylactic measures for bleeding GV, there is a paucity of data examining the role of primary prophylaxis. Few trials have recruited patients with the intention to treat GV prior to bleeding, and those that have[36,38,39] have done so in low numbers which prevents any meaningful subgroup analysis.
One RCT by Mishra et al[48] randomized patients with never-bled GOV2 or IGV1 ≥ 10 mm in size to cyanoacrylate injection, propranolol or no therapy in a 1:1:1 ratio. This demonstrated a significant reduction in GV bleeding in those treated with cyanoacrylate compared to those with propranolol or no therapy (10% vs 38% vs 53%, P = 0.003), as well as a significant reduction in bleed-related mortality between those receiving endoscopic therapy and those who received no specific therapy (0% vs 20%, P = 0.025). There was no statistical difference in overall mortality between the groups (7% vs 17% vs 26%, P = 0.113), nor therapy-related complications (3% vs 3% vs 7%, P = 1.0). This suggests endoscopic cyanoacrylate therapy could be recommended in patients with GV larger than 10mm in size, and NSBB therapy considered in those with contraindication to, or declining, cyanoacrylate injection.
In subgroup analysis of another study by Bhat et al[38], 93% of patients undergoing combined EUS-guided coiling with cyanoacrylate for primary prophylaxis had no GV bleeding over a mean follow-up time of 449 d.
To date, there are no head-to-head trials comparing endoscopic therapy with radiologic interventions for primary prophylaxis, nor any specific trials to compare various endoscopic therapies (coil vs cyanoacrylate or combination therapy vs monotherapy).
CONCLUSION
Future directions
The majority of evidence for the treatment of GV stems from retrospective studies, and so there is a need for further prospective and randomized trials to better guide management. In particular, there is a paucity of data on primary prophylaxis of GV, the risk of treating small (< 10 mm) or low-risk GV, and on the optimal approach to secondary prophylaxis (endoscopic, radiologic or combined) since the advent of EUS-based combination therapy. Furthermore, little is known regarding the ideal timeframe for surveillance of GV, whether treated or untreated.
Footnotes
Conflict-of-interest statement: The authors confirm there are no conflicts of interest to declare
Manuscript source: Invited manuscript
Peer-review started: February 24, 2021
First decision: May 3, 2021
Article in press: July 29, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Australia
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kreisel W S-Editor: Liu M L-Editor: A P-Editor: Guo X
Contributor Information
Karl Vaz, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia. karl.vaz@austin.org.au.
Marios Efthymiou, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia; University of Melbourne, Parkville 3052, Victoria, Australia.
Rhys Vaughan, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia; University of Melbourne, Parkville 3052, Victoria, Australia.
Adam G Testro, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia.
Hin-Boon Lew, Department of Radiology, Austin Health, Heidelberg 3084, Victoria, Australia.
Leonardo Zorron Cheng Tao Pu, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia.
Sujievvan Chandran, Department of Gastroenterology and Hepatology, Austin Health, Heidelberg 3084, Victoria, Australia; University of Melbourne, Parkville 3052, Victoria, Australia.
References
- 1.Hosking SW, Johnson AG. Gastric varices: a proposed classification leading to management. Br J Surg. 1988;75:195–196. doi: 10.1002/bjs.1800750303. [DOI] [PubMed] [Google Scholar]
- 2.Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992;16:1343–1349. doi: 10.1002/hep.1840160607. [DOI] [PubMed] [Google Scholar]
- 3.Lo GH, Lai KH, Cheng JS, Huang RL, Wang SJ, Chiang HT. Prevalence of paraesophageal varices and gastric varices in patients achieving variceal obliteration by banding ligation and by injection sclerotherapy. Gastrointest Endosc. 1999;49:428–436. doi: 10.1016/s0016-5107(99)70038-6. [DOI] [PubMed] [Google Scholar]
- 4.Caletti G, Brocchi E, Baraldini M, Ferrari A, Gibilaro M, Barbara L. Assessment of portal hypertension by endoscopic ultrasonography. Gastrointest Endosc. 1990;36:S21–S27. doi: 10.1016/s0016-5107(90)71011-5. [DOI] [PubMed] [Google Scholar]
- 5.Watanabe K, Kimura K, Matsutani S, Ohto M, Okuda K. Portal hemodynamics in patients with gastric varices. A study in 230 patients with esophageal and/or gastric varices using portal vein catheterization. Gastroenterology. 1988;95:434–440. doi: 10.1016/0016-5085(88)90501-x. [DOI] [PubMed] [Google Scholar]
- 6.Tripathi D, Therapondos G, Jackson E, Redhead DN, Hayes PC. The role of the transjugular intrahepatic portosystemic stent shunt (TIPSS) in the management of bleeding gastric varices: clinical and haemodynamic correlations. Gut. 2002;51:270–274. doi: 10.1136/gut.51.2.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sanyal AJ, Freedman AM, Luketic VA, Purdum PP 3rd, Shiffman ML, DeMeo J, Cole PE, Tisnado J. The natural history of portal hypertension after transjugular intrahepatic portosystemic shunts. Gastroenterology. 1997;112:889–898. doi: 10.1053/gast.1997.v112.pm9041251. [DOI] [PubMed] [Google Scholar]
- 8.Sarin SK, Kumar A. Gastric varices: profile, classification, and management. Am J Gastroenterol. 1989;84:1244–1249. [PubMed] [Google Scholar]
- 9.Hashizume M, Kitano S, Yamaga H, Koyanagi N, Sugimachi K. Endoscopic classification of gastric varices. Gastrointest Endosc. 1990;36:276–280. doi: 10.1016/s0016-5107(90)71023-1. [DOI] [PubMed] [Google Scholar]
- 10.Kim T, Shijo H, Kokawa H, Tokumitsu H, Kubara K, Ota K, Akiyoshi N, Iida T, Yokoyama M, Okumura M. Risk factors for hemorrhage from gastric fundal varices. Hepatology. 1997;25:307–312. doi: 10.1053/jhep.1997.v25.pm0009021939. [DOI] [PubMed] [Google Scholar]
- 11.Abby Philips C, Sahney A. Oesophageal and gastric varices: historical aspects, classification and grading: everything in one place. Gastroenterol Rep (Oxf) 2016;4:186–195. doi: 10.1093/gastro/gow018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ryan BM, Stockbrugger RW, Ryan JM. A pathophysiologic, gastroenterologic, and radiologic approach to the management of gastric varices. Gastroenterology. 2004;126:1175–1189. doi: 10.1053/j.gastro.2004.01.058. [DOI] [PubMed] [Google Scholar]
- 13.Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310–335. doi: 10.1002/hep.28906. [DOI] [PubMed] [Google Scholar]
- 14.European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406–460. doi: 10.1016/j.jhep.2018.03.024. [DOI] [PubMed] [Google Scholar]
- 15.Lo GH, Lai KH, Cheng JS, Chen MH, Chiang HT. A prospective, randomized trial of butyl cyanoacrylate injection vs band ligation in the management of bleeding gastric varices. Hepatology. 2001;33:1060–1064. doi: 10.1053/jhep.2001.24116. [DOI] [PubMed] [Google Scholar]
- 16.Sarin SK, Mishra SR. Endoscopic therapy for gastric varices. Clin Liver Dis. 2010;14:263–279. doi: 10.1016/j.cld.2010.03.007. [DOI] [PubMed] [Google Scholar]
- 17.Sarin SK, Jain AK, Jain M, Gupta R. A randomized controlled trial of cyanoacrylate vs alcohol injection in patients with isolated fundic varices. Am J Gastroenterol. 2002;97:1010–1015. doi: 10.1111/j.1572-0241.2002.05622.x. [DOI] [PubMed] [Google Scholar]
- 18.Barange K, Péron JM, Imani K, Otal P, Payen JL, Rousseau H, Pascal JP, Joffre F, Vinel JP. Transjugular intrahepatic portosystemic shunt in the treatment of refractory bleeding from ruptured gastric varices. Hepatology. 1999;30:1139–1143. doi: 10.1002/hep.510300523. [DOI] [PubMed] [Google Scholar]
- 19.Chau TN, Patch D, Chan YW, Nagral A, Dick R, Burroughs AK. "Salvage" transjugular intrahepatic portosystemic shunts: gastric fundal compared with esophageal variceal bleeding. Gastroenterology. 1998;114:981–987. doi: 10.1016/s0016-5085(98)00640-4. [DOI] [PubMed] [Google Scholar]
- 20.Stanley AJ, Jalan R, Ireland HM, Redhead DN, Bouchier IA, Hayes PC. A comparison between gastric and oesophageal variceal haemorrhage treated with transjugular intrahepatic portosystemic stent shunt (TIPSS) Aliment Pharmacol Ther. 1997;11:171–176. doi: 10.1046/j.1365-2036.1997.106277000.x. [DOI] [PubMed] [Google Scholar]
- 21.Philips CA, Ahamed R, Rajesh S, George T, Mohanan M, Augustine P. Beyond the scope and the glue: update on evaluation and management of gastric varices. BMC Gastroenterol. 2020;20:361. doi: 10.1186/s12876-020-01513-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rajesh S, George T, Philips CA, Ahamed R, Kumbar S, Mohan N, Mohanan M, Augustine P. Transjugular intrahepatic portosystemic shunt in cirrhosis: An exhaustive critical update. World J Gastroenterol. 2020;26:5561–5596. doi: 10.3748/wjg.v26.i37.5561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mahadeva S, Bellamy MC, Kessel D, Davies MH, Millson CE. Cost-effectiveness of N-butyl-2-cyanoacrylate (histoacryl) glue injections vs transjugular intrahepatic portosystemic shunt in the management of acute gastric variceal bleeding. Am J Gastroenterol. 2003;98:2688–2693. doi: 10.1111/j.1572-0241.2003.08769.x. [DOI] [PubMed] [Google Scholar]
- 24.Procaccini NJ, Al-Osaimi AM, Northup P, Argo C, Caldwell SH. Endoscopic cyanoacrylate vs transjugular intrahepatic portosystemic shunt for gastric variceal bleeding: a single-center U.S. analysis. Gastrointest Endosc. 2009;70:881–887. doi: 10.1016/j.gie.2009.03.1169. [DOI] [PubMed] [Google Scholar]
- 25.Saad WE, Darcy MD. Transjugular Intrahepatic Portosystemic Shunt (TIPS) vs Balloon-occluded Retrograde Transvenous Obliteration (BRTO) for the Management of Gastric Varices. Semin Intervent Radiol. 2011;28:339–349. doi: 10.1055/s-0031-1284461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Park JK, Saab S, Kee ST, Busuttil RW, Kim HJ, Durazo F, Cho SK, Lee EW. Balloon-Occluded Retrograde Transvenous Obliteration (BRTO) for Treatment of Gastric Varices: Review and Meta-Analysis. Dig Dis Sci. 2015;60:1543–1553. doi: 10.1007/s10620-014-3485-8. [DOI] [PubMed] [Google Scholar]
- 27.Wang ZW, Liu JC, Zhao F, Zhang WG, Duan XH, Chen PF, Yang SF, Li HW, Chen FW, Shi HS, Ren JZ. Comparison of the Effects of TIPS vs BRTO on Bleeding Gastric Varices: A Meta-Analysis. Can J Gastroenterol Hepatol. 2020;2020:5143013. doi: 10.1155/2020/5143013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Choi YH, Yoon CJ, Park JH, Chung JW, Kwon JW, Choi GM. Balloon-occluded retrograde transvenous obliteration for gastric variceal bleeding: its feasibility compared with transjugular intrahepatic portosystemic shunt. Korean J Radiol. 2003;4:109–116. doi: 10.3348/kjr.2003.4.2.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mishra SR, Chander Sharma B, Kumar A, Sarin SK. Endoscopic cyanoacrylate injection vs beta-blocker for secondary prophylaxis of gastric variceal bleed: a randomised controlled trial. Gut. 2010;59:729–735. doi: 10.1136/gut.2009.192039. [DOI] [PubMed] [Google Scholar]
- 30.Hung HH, Chang CJ, Hou MC, Liao WC, Chan CC, Huang HC, Lin HC, Lee FY, Lee SD. Efficacy of non-selective β-blockers as adjunct to endoscopic prophylactic treatment for gastric variceal bleeding: a randomized controlled trial. J Hepatol. 2012;56:1025–1032. doi: 10.1016/j.jhep.2011.12.021. [DOI] [PubMed] [Google Scholar]
- 31.Chen WC, Hsin IF, Chen PH, Hsu PI, Wang YP, Cheng JS, Lin HS, Hou MC, Lee FY. Addition of Carvedilol to Gastric Variceal Obturation Does Not Decrease Recurrence of Gastric Variceal Bleeding in Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2019;17:2356–2363. doi: 10.1016/j.cgh.2019.02.021. [DOI] [PubMed] [Google Scholar]
- 32.Lee YT, Chan FK, Ng EK, Leung VK, Law KB, Yung MY, Chung SC, Sung JJ. EUS-guided injection of cyanoacrylate for bleeding gastric varices. Gastrointest Endosc. 2000;52:168–174. doi: 10.1067/mge.2000.107911. [DOI] [PubMed] [Google Scholar]
- 33.Bick BL, Al-Haddad M, Liangpunsakul S, Ghabril MS, DeWitt JM. EUS-guided fine needle injection is superior to direct endoscopic injection of 2-octyl cyanoacrylate for the treatment of gastric variceal bleeding. Surg Endosc. 2019;33:1837–1845. doi: 10.1007/s00464-018-6462-z. [DOI] [PubMed] [Google Scholar]
- 34.Binmoeller KF. Endoscopic Ultrasound-Guided Coil and Glue Injection for Gastric Variceal Bleeding. Gastroenterol Hepatol (NY) 2018;14:123–126. [PMC free article] [PubMed] [Google Scholar]
- 35.Khoury T, Nadella D, Wiles A, Marshall C, Kumar M, Shapira G, Ludvik N, Schumann A, Mahamid M, Mizrahi M. A review article on gastric varices with focus on the emerging role of endoscopic ultrasound-guided angiotherapy. Eur J Gastroenterol Hepatol. 2018;30:1411–1415. doi: 10.1097/MEG.0000000000001200. [DOI] [PubMed] [Google Scholar]
- 36.Romero-Castro R, Ellrichmann M, Ortiz-Moyano C, Subtil-Inigo JC, Junquera-Florez F, Gornals JB, Repiso-Ortega A, Vila-Costas J, Marcos-Sanchez F, Muñoz-Navas M, Romero-Gomez M, Brullet-Benedi E, Romero-Vazquez J, Caunedo-Alvarez A, Pellicer-Bautista F, Herrerias-Gutierrez JM, Fritscher-Ravens A. EUS-guided coil vs cyanoacrylate therapy for the treatment of gastric varices: a multicenter study (with videos) Gastrointest Endosc. 2013;78:711–721. doi: 10.1016/j.gie.2013.05.009. [DOI] [PubMed] [Google Scholar]
- 37.Binmoeller KF, Weilert F, Shah JN, Kim J. EUS-guided transesophageal treatment of gastric fundal varices with combined coiling and cyanoacrylate glue injection (with videos) Gastrointest Endosc. 2011;74:1019–1025. doi: 10.1016/j.gie.2011.06.030. [DOI] [PubMed] [Google Scholar]
- 38.Bhat YM, Weilert F, Fredrick RT, Kane SD, Shah JN, Hamerski CM, Binmoeller KF. EUS-guided treatment of gastric fundal varices with combined injection of coils and cyanoacrylate glue: a large U.S. experience over 6 years (with video) Gastrointest Endosc. 2016;83:1164–1172. doi: 10.1016/j.gie.2015.09.040. [DOI] [PubMed] [Google Scholar]
- 39.Robles-Medranda C, Oleas R, Valero M, Puga-Tejada M, Baquerizo-Burgos J, Ospina J, Pitanga-Lukashok H. Endoscopic ultrasonography-guided deployment of embolization coils and cyanoacrylate injection in gastric varices vs coiling alone: a randomized trial. Endoscopy. 2020;52:268–275. doi: 10.1055/a-1123-9054. [DOI] [PubMed] [Google Scholar]
- 40.Lo GH, Liang HL, Chen WC, Chen MH, Lai KH, Hsu PI, Lin CK, Chan HH, Pan HB. A prospective, randomized controlled trial of transjugular intrahepatic portosystemic shunt vs cyanoacrylate injection in the prevention of gastric variceal rebleeding. Endoscopy. 2007;39:679–685. doi: 10.1055/s-2007-966591. [DOI] [PubMed] [Google Scholar]
- 41.Hong CH, Kim HJ, Park JH, Park DI, Cho YK, Sohn CI, Jeon WK, Kim BI, Hong HP, Shin JH. Treatment of patients with gastric variceal hemorrhage: endoscopic N-butyl-2-cyanoacrylate injection vs balloon-occluded retrograde transvenous obliteration. J Gastroenterol Hepatol. 2009;24:372–378. doi: 10.1111/j.1440-1746.2008.05651.x. [DOI] [PubMed] [Google Scholar]
- 42.Akahoshi T, Tomikawa M, Kamori M, Tsutsumi N, Nagao Y, Hashizume M, Maehara Y. Impact of balloon-occluded retrograde transvenous obliteration on management of isolated fundal gastric variceal bleeding. Hepatol Res. 2012;42:385–393. doi: 10.1111/j.1872-034X.2011.00939.x. [DOI] [PubMed] [Google Scholar]
- 43.Stein DJ, Salinas C, Sabri S, Onyeali R, Caldwell S, Henry Z. Balloon Retrograde Transvenous Obliteration Versus Endoscopic Cyanoacrylate in Bleeding Gastric Varices: Comparison of Rebleeding and Mortality with Extended Follow-up. J Vasc Interv Radiol. 2019;30:187–194. doi: 10.1016/j.jvir.2018.12.008. [DOI] [PubMed] [Google Scholar]
- 44.Saad WE, Wagner CC, Lippert A, Al-Osaimi A, Davies MG, Matsumoto AH, Angle JF, Caldwell S. Protective value of TIPS against the development of hydrothorax/ascites and upper gastrointestinal bleeding after balloon-occluded retrograde transvenous obliteration (BRTO) Am J Gastroenterol. 2013;108:1612–1619. doi: 10.1038/ajg.2013.232. [DOI] [PubMed] [Google Scholar]
- 45.Liu J, Yang C, Huang S, Zhou C, Shi Q, Qian K, Song S, Xiong B. The combination of balloon-assisted antegrade transvenous obliteration and transjugular intrahepatic portosystemic shunt for the management of cardiofundal varices hemorrhage. Eur J Gastroenterol Hepatol. 2020;32:656–662. doi: 10.1097/MEG.0000000000001705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Saad WE. Combining Transjugular Intrahepatic Portosystemic Shunt with Balloon-Occluded Retrograde Transvenous Obliteration or Augmenting TIPS with Variceal Embolization for the Management of Gastric Varices: An Evolving Middle Ground? Semin Intervent Radiol. 2014;31:266–268. doi: 10.1055/s-0034-1382796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Saad WE, Kitanosono T, Koizumi J. Balloon-occluded antegrade transvenous obliteration with or without balloon-occluded retrograde transvenous obliteration for the management of gastric varices: concept and technical applications. Tech Vasc Interv Radiol. 2012;15:203–225. doi: 10.1053/j.tvir.2012.07.004. [DOI] [PubMed] [Google Scholar]
- 48.Mishra SR, Sharma BC, Kumar A, Sarin SK. Primary prophylaxis of gastric variceal bleeding comparing cyanoacrylate injection and beta-blockers: a randomized controlled trial. J Hepatol. 2011;54:1161–1167. doi: 10.1016/j.jhep.2010.09.031. [DOI] [PubMed] [Google Scholar]