Abstract
Chronic obstructive lung disease (COPD) and lung cancer are both caused by smoking and often occur as comorbidity. The programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) axis is an important canonic immunoregulatory pathway, and antibodies that specifically block PD-1 or PD-L1 have demonstrated efficacy as therapeutic agents for non-small cell lung cancer. The role of the PD-1/PD-L1 axis in the pathogenesis of COPD is unknown. Here, we analyzed the function of the PD-1/PD-L1 axis in preclinical COPD models and evaluated the concentrations of PD-1 and PD-L1 in human serum and bronchoalveolar lavage (BAL) fluids as biomarkers for COPD. Anti-PD-1 treatment decreased lung damage and neutrophilic inflammation in mice chronically exposed to cigarette smoke (CS) or nontypeable Haemophilus influenzae (NTHi). Ex vivo stimulated macrophages obtained from anti-PD-1-treated mice released reduced amounts of inflammatory cytokines. PD-L1 concentrations correlated positively with PD-1 concentrations in human serum and BAL fluids. Lung sections obtained from patients with COPD stained positive for PD-L1. Our data indicate that the PD-1/PD-L1 axis is involved in developing inflammation and tissue destruction in COPD. Inflammation-induced activation of the PD-1 pathway may contribute to disease progression.
Keywords: COPD, inflammation, lung damage, macrophages, PD-1, PD-L1
INTRODUCTION
Chronic obstructive lung disease (COPD) and lung cancer are major disease entities caused by tobacco abuse and result in significant morbidity and mortality. Both diseases often occur as comorbidities (1, 2). Inflammation in COPD is characterized by increased numbers of inflammatory cells in the lung. Neutrophils are suggested to promote disease progression through diverse mechanisms, including the release of proteases and reactive oxygen species. Acute and chronic infections with gram-negative bacteria [e.g., nontypeable Haemophilus influenzae (NTHi)] amplify pulmonary inflammation and therefore contribute to tissue destruction and loss of lung function in COPD (3, 4).
The presence of COPD is a significant risk factor for lung cancer (5). Immune checkpoint inhibitors have been targeted for the treatment of non-small cell lung cancer. One group of these novel drugs that block the interaction of programmed cell death protein 1 (PD-1) with its ligand programmed cell death ligand 1 (PD-L1) has shown encouraging results in patients with lung cancer (6).
Recent studies indicate an increased expression of PD-1 in lymphocytes obtained from patients with COPD (7–9). However, it is mostly unknown whether or not the PD-1/PD-L1 axis has a function in the development and pathogenesis of COPD and whether therapeutic modulation of the PD-1/PD-L1 axis is beneficial or unfavorable for patients with COPD. As the activation of PD-1 may be a mechanism to avoid overstimulation by bacteria, it is possible that blocking of the PD-1/PD-L1 axis results in increased pulmonary inflammation and further exacerbates the disease (10, 11). However, therapeutic antibodies against PD-1/PD-L1 have the potential to improve immunity against bacteria or viruses by reversing the suppression of T cells (10). Thus, it is a matter of discussion whether and how therapeutic antibodies against PD-1 or PD-L1 affect the course of COPD (10, 11).
This study aimed to examine the function of the PD-1/PD-L1 axis in the development of COPD. In two preclinical models of COPD, we show that blocking of PD-1 results in reduced lung damage. We further demonstrate that PD-L1 concentrations correlate positively with PD-1 concentrations in serum and bronchoalveolar lavage (BAL) fluids collected from patients with COPD. Moreover, lung sections obtained from patients with COPD stain positive for PD-L1. Our data, therefore, suggest that activation of the PD-1 pathway may contribute to disease progression.
Some of the results of these studies have been previously reported in an abstract (12).
METHODS
Patient Study
The present study included patients from the PULMOHOM (Pulmology Homburg) cohort (approved by the ethics committee of the Landesaerztekammer des Saarlandes), which is a prospective cohort study on patients with inflammatory lung disease. All participants provided written informed consent. Patients were enrolled, if the following inclusion criteria were fulfilled: 1) aged 40 yr and older and 2) diagnosis of COPD [according to Global Initiative for Chronic Obstructive Lung (GOLD) criteria] from written documentation with pulmonary function available within an 8-wk time frame. Acute exacerbations of COPD (AECOPD) were defined as a condition with an acute increase of respiratory symptoms (dyspnea, cough, increased sputum production) and the exclusion of differential diagnoses. Clinical data on pulmonary function during a stable phase of the disease and quality of live life were determined.
PD-1 and PD-L1 concentrations were determined using Milliplex Human Immuno-Oncology Checkpoint Protein Panel MAP Kit (Merck KGaA, Darmstadt, Germany) on Luminex MAGPIX (Austin, TX). We retrospectively selected six lung explants with end-stage COPD, which were kindly provided by the Institute of Pathology (Saarland University, Homburg, Germany) and approved by the ethics committee of the Landesaerztekammer des Saarlandes. Formalin-fixed and paraffin-embedded (FFPE) tissue samples were used for immunohistochemistry (IHC) with a primary antibody for PD-L1 (R&D Systems, Germany).
Mouse Experiments
All animal experiments were approved by the Landesamt für Soziales, Gesundheit und Verbraucherschutz of the State of Saarland in agreement with the national guidelines for animal treatment. Six- to eight-week-old female C57BL/6 mice were obtained from Janvier Labs (France) and were acclimated for at least 2 wk. The mice were randomly chosen for the experimental groups with two to five mice per cage. For experiments, 8- to 10-wk-old mice were exposed to NTHi or smoke. At the end of the experiment, mice were anesthetized [intraperitoneal (ip) injection of 90 to 120 mg/kg of ketamine hydrochloride (Ketanest; Pfizer, Germany) and 10 to 12 mg/kg of xylazine hydrochloride (Rompun; Bayer, Germany)] and killed by bleeding with puncture of the ventricle. Smoke model: The mice were subjected to two smoking periods of 50 min per day with a resting phase of 2 h between the smoke exposures (3R4F; College of Agriculture, Reference Cigarette Program, University of Kentucky, Lexington, KY) in a Teague Enterprises (TE-10) smoking machine (Teague Enterprises, Woodland, CA) at 5 days per week. The cigarette smoke (CS) concentration was 400–500 mg/m3 of total suspended particles, and carbon monoxide (CO) concentrations were between 200 and 350 ppm during the smoking period. Mice were exposed to CS for 5 days and 4 wk for the analyses of pulmonary inflammation and for 12 wk for histology and lung function. NTHi-exposure model: The mice were exposed to a heat-inactivated and sonicated clinical isolate of NTHi (2.5 mg/mL protein in PBS) for 40 min per day in a plexi glass box connected to a Pari MASTER nebulizer (Pari GmbH, Starnberg, Germany) at days 1, 3, and 5 of the week. Mice were exposed to NTHi for 4 wk. A PD-1 blocking antibody (clone 29 F.1A12, BioXcell, West Lebanon, NH) that has been shown to affect tumor growth in murine cancer models (13–15) or isotype control (clone 2A3, BioXcell) were administrated by intraperitoneal injection (100 µg in PBS per dose) three times a week in the NTHi model and two times a week in the CS model (16).
Histopathology and Lung Function
Histologic analysis and lung function were performed as described before (17, 18). In brief, lungs not subjected to BAL were fixed in 4% formalin in PBS under a constant hydrostatic pressure of 30 cm H2O for 15 min, embedded in agarose (1% in PBS), cut into slices of the same thickness, and embedded in paraffin. Double staining was performed using PolyStain DS Kit (Goat/Rabbit, NeoBiotech, Seoul, Korea). Primary antibodies for PD-1 (AF-1021; R&D, MN), PD-L1 (AF-1019), CD4 (ab183685; Abcam, Cambridge, UK), CD8 (ab217344), and CD68 (ab125212) were used for immunohistochemistry analysis blinded to the investigator. Randomly selected fields were evaluated for positive cells using an Olympus BX51 microscope. Paraffin sections were blinded to the investigator and stained with hematoxylin-eosin (H&E). The mean chord length (MCL) was calculated using the Visiopharm Integrator System (Visiopharm, Hoersholm, Denmark) on an Olympus BX51 microscope blinded to the investigator. Invasive lung function was determined in anesthetized mice using the FlexiVent system (Scireq Inc., Montreal, Canada).
Determination of Immune Cells and Cytokine Concentrations
BAL was performed through the cannulated trachea with 1 mL of PBS as described before (18). Numbers of total leukocytes in the blood and BAL fluids were determined using a Neubauer cell counter. Cytospin preparations of BAL fluids and blood smears were stained (Diff Quik Staining kit, Medion Diagnostics, Switzerland) to differentiate macrophages, neutrophils, and lymphocytes by light microscopy. Cytokine concentrations were measured by enzyme-linked immunosorbent assay (ELISA, R&D Systems, Germany).
Ex Vivo Stimulation of Macrophages
Mice were exposed to air or CS and treated with blocking antibodies for 4 wk as described above in Mouse Experiments. Alveolar macrophages were isolated by flushing the lungs five times with 1 mL of PBS supplemented with 0.5 mM EDTA 2 h after the final exposure to CS. BAL cells were centrifuged at 800 rpm for 5 min and recovered in RPMI 1640 (Invitrogen, Grand Island, NY) supplemented with 10% fetal calf serum (FCS, Invitrogen), 100 U/mL penicillin, and 100 U/mL streptomycin (PAA Laboratories GmbH, Pasching, Austria) and seeded at a density of 1 × 105/well in 48-well plates. After 2 h, nonadherent cells were removed by changing the medium. Adherent macrophages were stained (Diff-Quik Staining Kit) to confirm the presence of mononuclear cells. To analyze inflammatory responses, alveolar macrophages were activated with heat-inactivated and sonicated NTHi (2.5 mg/mL protein in PBS, diluted 1:50 in RPMI 1640 Media). Cytokines were determined after 24 h.
Statistical Analysis
The software Prism (GraphPad Software, San Diego, CA) was used for statistical analysis. Comparisons between groups were analyzed by two-way ANOVA with Bonferroni posttest or Mann–Whitney test as indicated in the figure legends. Correlation analysis was performed using nonparametric Spearman’s correlation test. The results were considered statistically significant for P < 0.05 (all Supplemental material is available at https://doi.org/10.6084/m9.figshare.14113577).
RESULTS
Blockade of PD-1 Decreases CS-Induced Neutrophilic Inflammation
As there is evidence that the activation of the PD-1/PD-L1 axis is associated with the pathogenesis of COPD (7–9), we examined the function of PD-1 in a preclinical model of CS-induced lung damage. C57BL/6 mice were exposed to CS for up to 12 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. There were no significant differences in the body weight and numbers of blood leukocytes between the different groups after 4 and 12 wk of CS exposure (Supplemental Fig. S1). There was also no difference in the subtype distribution and functionality of splenic CD8+T cells in mice exposed to CS for 4 wk. Splenic CD8+T cells isolated from CS-exposed isotype or PD-1 antibody-treated mice killed the target cancer cell lines P815 or EG7 at the same rate (data not shown).
Neutrophils are central in the pathogenesis of COPD (3). Differential cell count showed that numbers of total immune cells, neutrophils, and lymphocytes were significantly decreased in anti-PD-1-treated mice compared with isotype-treated mice after exposure to CS for 4 wk, whereas numbers of macrophages in BAL fluids were not affected by the blockade of PD-1 (Fig. 1, A–D, and Supplemental Fig. S2). We further determined the concentrations of cytokines involved in the recruitment of neutrophils. Concentrations of keratinocyte-derived chemokine (KC) and macrophage inflammatory protein 2 (MIP-2) in BAL fluids were significantly decreased in anti-PD-1-treated mice compared with isotype-treated mice after 5 days of exposure to CS (Fig. 1, E and F).
Figure 1.
Anti-PD-1 treatment decreases CS-induced neutrophilic inflammation. C57BL/6 mice were exposed to CS 5 days a week for 5 days and 4 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. Numbers of total immune cells (A), neutrophils (B), macrophages (C), and lymphocytes (D), and concentrations of (E) KC and (F) MIP-2 were determined in BAL fluids 24 h after the final exposure to CS; n = 4–8 mice per group. Two independent experiments for each group. Data were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. **P < 0.01 and ***P < 0.001. BAL, bronchoalveolar lavage; CS, cigarette smoke; PD-1, programmed cell death protein 1.
The PD-1/PD-L1 Axis Has a Function in the Development of CS-Induced Lung Damage
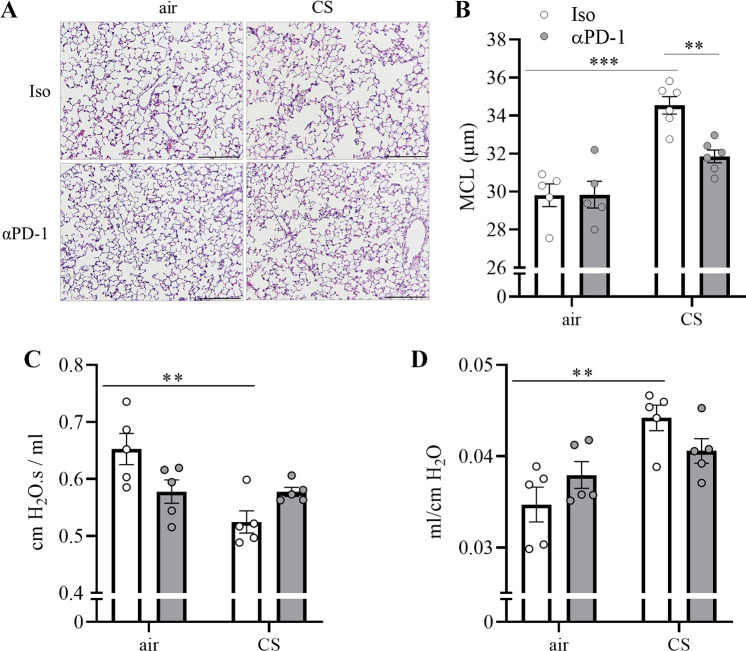
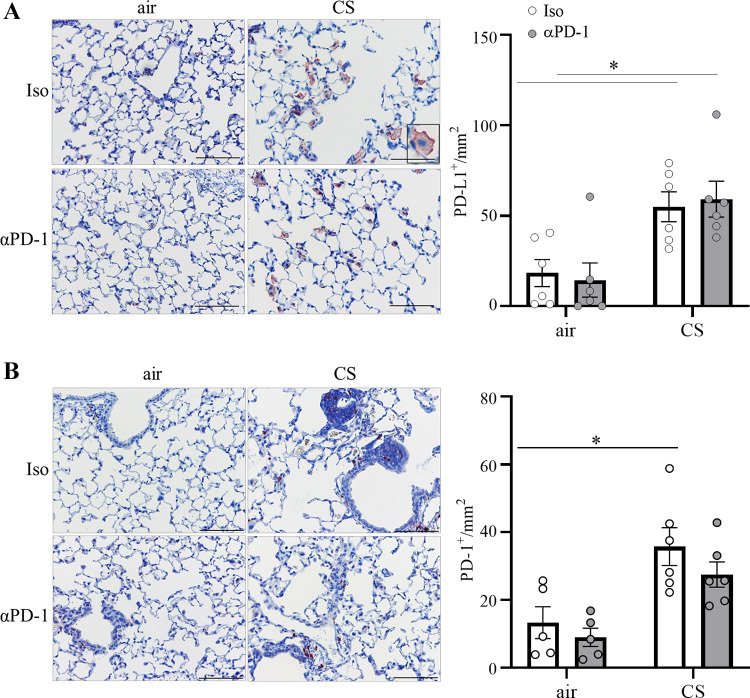
Histopathologic and stereologic analysis of the alveolar parenchyma showed that the blockade of PD-1 attenuated the CS-induced lung damage. Exposure to CS for 12 wk resulted in an increased mean chord length (MCL), which was significantly reduced in anti-PD-1-treated mice (Fig. 2, A and B). Invasive lung function measurements revealed that anti-PD-1 treatment also affected pulmonary mechanics. Exposure to CS for 12 wk resulted in a significantly decreased respiratory resistance (Fig. 2C) and in a significantly increased respiratory system compliance (Fig. 2D) in isotype-treated mice, whereas there were no significant differences in the resistance and compliance between air- and CS-exposed anti-PD-1-treated mice. Immunohistochemistry (IHC) revealed a significantly increased membrane expression of PD-L1 and significantly increased numbers of PD-1+ cells in the lungs of mice exposed to CS for 12 wk compared with control mice (Fig. 3). We further performed IHC double staining to identify the expression of PD-1 in CD4- and CD8-positive cells. Exposure to CS resulted in increased numbers of PD-1-expressing CD4+ cells and to a lesser extend of PD-1-expressing CD8+ cells (Fig. 4, A and B, and Supplemental Fig. S3). Moreover, PD-L1 was strongly expressed in CD68+ cells, a marker for mononuclear phagocytes (Fig. 4C and Supplemental Fig. S3). These data indicate that exposure to CS activates the PD-1/PD-L1-axis in the lung and that the blockade of PD-1 attenuates the CS-induced loss of lung structure and function.
Figure 2.
Anti-PD-1 treatment decreases CS-induced lung damage. C57BL/6 mice were exposed to CS 5 days a week for 12 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. A: lung histology (representative hematoxylin-eosin staining, scale bar = 100 µm), B: mean chord length (MCL), C: respiratory resistance, and D: respiratory system compliance were determined 24 h after the final exposure to CS; n = 5 or 6 mice per group. Two independent experiments for each group. Data were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. **P < 0.01 and ***P < 0.001. CS, cigarette smoke; PD-1, programmed cell death protein 1.
Figure 3.
Numbers of PD-L1+ and PD-1+ cells are increased in the lungs of CS-exposed mice. C57BL/6 mice were exposed to CS 5 days a week for 12 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. Immunohistochemical staining (representative histology, scale bar = 50 µm) for (A) PD-L1 and (B) PD-1 and quantification of the PD-L1+ and PD-1+ cells in lung parenchyma; n = 5 or 6 mice per group. Two independent experiments for each group. Data were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. *P < 0.05. CS, cigarette smoke; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1.
Figure 4.
Numbers of PD-1-expressing CD4+ cells and PD-L1-expressing CD68+ cells are increased in the lungs of CS-exposed mice. C57BL/6 mice were exposed to CS 5 days a week for 12 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. Double immunohistochemical staining (representative histology, scale bar = 20 µm) for (A) CD4 (red)/PD-1 (green), (B) CD8 (red)/PD-1 (green), and (C) CD68 (red)/PD-L1 (green), and quantification of the cells in lung parenchyma; n = 3 or 4 mice per group. Results are presented for each mouse, were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. *P < 0.05, **P < 0.01, and ***P < 0.001. CS, cigarette smoke; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1.
PD-1 Blockade Attenuates NTHi-Induced Loss of Lung Structure
In a separate study, we showed that treatment with a PD-1 blocking antibody results in a reduced NTHi-induced expression of pulmonary cytokines and a decreased recruitment of neutrophils in a Kras-dependent lung cancer model (16). To examine whether the blockade of PD-1 reduces loss of lung structure in an NTHi-dependent preclinical model of COPD-like inflammation, we exposed C57BL/6 mice to heat-inactivated NTHi for 4 wk and treated the mice with a PD-1-blocking antibody or an isotype antibody three times a week. This model of NTHi-induced COPD-like inflammation is comparable with well-established elastase- and LPS-dependent and LPS-dependent COPD models and induces numerous characteristics of COPD [e.g., robust neutrophilic lung inflammation, emphysema (19–22)]. There was no significant difference in the weight and numbers of blood leukocytes between the different groups after 4 wk of NTHi exposure (Supplemental Fig. S4). IHC showed a strong membrane expression of PD-L1 and increased numbers of PD-1+ cells in the lungs of mice chronically exposed to NTHi (Fig. 5, A and B). IHC double staining further showed that exposure to NTHi resulted in increased numbers of CD4+ and CD8+ cells expressing PD-1 as well as in increased numbers of CD68+ cells expressing PD-L1 (Supplemental Fig. S5). Histopathologic and stereologic analysis of alveolar parenchyma showed that the blockade of PD-1 attenuated the NTHi-induced lung damage, as seen in the CS model. The MCL was significantly decreased in anti-PD-1-treated mice compared with isotype-treated mice after exposure to NTHi for 4 wk (Fig. 5, C and D). These data indicate that chronic exposure to bacterial components activates the PD-1/PD-L1-axis in the lung and that the blockade of PD-1 attenuates the bacterial-induced loss of lung structure.
Figure 5.
Anti-PD-1 treatment decreases NTHi-induced lung damage. C57BL/6 mice were exposed to NTHi 3 days a week for 4 wk and treated with a PD-1-blocking antibody or an isotype antibody three times a week. Immunohistochemical staining (representative histology, scale bar = 50 µm) for (A) PD-L1, (B) PD-1, (C) lung histology (representative hematoxylin-eosin staining, scale bar = 100 µm), and (D) MCL were determined 24 h after the final exposure to NTHi; n = 4 or 5 mice per group. Two independent experiments for each group. Data were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. ***P < 0.001. MCL, mean chord length; NTHi, nontypeable Haemophilus influenza; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1.
Anti-PD-1 Treatment Modulates the Inflammatory Response of Alveolar Macrophages
To examine whether the blockade of PD-1 affects alveolar macrophages inflammatory responses, mice were exposed to CS for 4 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. Macrophages were isolated 2 h after the final exposure to CS and ex vivo stimulated with inactivated NTHi for 24 h. Figure 6 shows that macrophages obtained from CS-exposed mice released substantially higher amounts of all measured cytokines in response to the stimulation with NTHi compared with macrophage obtained from air-exposed mice. Anti-PD-1 treatment resulted in a significantly decreased release of IL-6, TNF-α, granulocyte colony stimulating factor (G-CSF), and KC in macrophages of CS-exposed mice (Fig. 6, A–D). Moreover, the release of IL-6 was significantly reduced in NTHi-stimulated macrophages of air-exposed mice treated with the anti-PD-1 body (Fig. 6A). Concentrations of cytokines in supernatants of alveolar macrophages that were not stimulated with NTHi were below the detection limit [8.25 pg/mL (IL-6), 0.74 pg/mL (TNF-α), 3.96 pg/mL (G-CSF), 8.83 pg/mL (KC), and 2.25 pg/mL (IL-10)].
Figure 6.
Anti-PD-1 treatment modulates the inflammatory response of alveolar macrophages. C57BL/6 mice were exposed to CS 5 days a week for 4 wk and treated with a PD-1-blocking antibody or an isotype antibody two times a week. Alveolar macrophages were isolated 2 h after the final exposure to CS and stimulated with inactivated NTHi or control media for 24 h. The concentrations of (A) IL-6, (B) TNF-α, (C) G-CSF, (D) KC, and (E) IL-10 were measured in supernatants. Two independent experiments for each group (n = 6 replicates per group). Data were compared by two-way ANOVA with Bonferroni posttest and are shown as means ± SE. *P < 0.05, **P < 0.01, ***P < 0.001. CS, cigarette smoke; NTHi, nontypeable Haemophilus influenza; PD-1, programmed cell death protein 1.
PD-1 and PD-L1 Levels in Serum and BAL Fluids of Patients with COPD
We measured the concentration of PD-L1 and PD-1 in serum collected from patients with stable COPD and patients during AECOPD. The characteristics of the patients are summarized in Table 1. PD-L1 and PD-1 were detectable in the serum of patients with COPD regardless of the exacerbation status. PD-L1 concentrations correlated positively with PD-1 concentrations in patients with stable COPD and during AECOPD (Fig. 7A). Patients with AECOPD had significantly increased levels of PD-L1 in their blood compared with patients with stable COPD (Fig. 7B). We also measured PD-L1 and PD-1 concentrations in BAL fluids of patients with COPD during stable disease. The characteristics of the patients are summarized in Table 2. PD-1 concentrations correlated positively with PD-L1 concentrations (Fig. 7C). Subgroup analysis did not reveal any significant differences in the concentrations of PD-L1 or PD-1 during advanced COPD (GOLD III/IV) compared with moderate (GOLD I/II) COPD (Fig. 7D). Tissue expression PD-L1 was examined in lung section obtained from six patients with end-stage COPD by immunohistochemistry. As shown in Fig. 7E, PD-L1 was present in cells with a macrophage-like appearance.
Table 1.
Patient characteristics
| Parameter | Stable | AECOPD | P value |
|---|---|---|---|
| N | 53 | 19 | |
| Sex, female/male | 21/31 | 7/12 | |
| Age, yr | 63.03 ± 8.483 | 64.9 ± 8.672 | ns |
| Height, m | 1.676 ± 0.109 | 1.698 ± 0.091 | ns |
| Weight, kg | 69.67 ± 17.87 | 76.46 ± 21.75 | ns |
| FEV1 | 0.864 ± 0.467 | 1.136 ± 0.599 | ns |
| FEV1, %predicted | 31.01 ± 12.36 | 39.66 ± 20.88 | ns |
| FEV1/FVC | 46.69 ± 14.47 | 58.37 ± 13.08 | 0.0195 |
Values are means ± SD. ns, not significant; GOLD, Global Initiative for Chronic Obstructive Lung Disease.
Figure 7.
PD-1 and PD-L1 in serum and BAL fluids collected from patients with COPD. A: association of PD-L1 and PD-1 concentrations in serum collected from patients with stable COPD and patients during AECOPD. B: PD-L1 and PD-1 concentrations in serum collected from patients with stable COPD and during AECOPD. C: association of PD-L1 and PD-1 concentrations in BAL fluids collected from stable COPD patients. D: PD-L1 and PD-1 concentrations in BAL fluids collected from stable GOLD I/II and GOLD III/IV patients with COPD. Data were compared by Mann–Whitney and are shown as means ± SE. *P < 0.05. Correlation analysis was performed using the nonparametric Spearman’s correlation test. E: PD-L1 was detected by immunohistochemistry in formalin histological-fixed paraffin-embedded human lung samples from patients with end-stage COPD (representative histology, scale bar = 50 µm). BAL, bronchoalveolar lavage; COPD, chronic obstructive lung disease; GOLD, Global Initiative for Chronic Obstructive Lung Disease; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1.
Table 2.
Patient characteristics
| Parameter | GOLD I/II | GOLD III/IV | P value |
|---|---|---|---|
| N | 19 | 12 | |
| Sex, female/male | 2/17 | 3/9 | |
| Age, yr | 67.32 ± 9.9 | 68.25 ± 6.5 | ns |
| Height, m | 1.732 ± 0.079 | 1.686 ± 0.118 | ns |
| Weight, kg | 84.99 ± 18.80 | 72.72 ± 19.92 | ns |
| FEV1 | 1.91 ± 0.65 | 0.872 ± 0.35 | <0.0001 |
| FEV1, %predicted | 65.67 ± 15.93 | 32.263 ± 12.38 | <0.0001 |
| FEV1/FVC | 0.685 ± 0.097 | 0.422 ± 0.0103 | <0.0001 |
Values are means ± SD. ns, not significant; GOLD, Global Initiative for Chronic Obstructive Lung Disease.
DISCUSSION
In the present study, we examined the role of the PD-1/PD-L1 axis in the progression of COPD. We demonstrated in preclinical models that CS-induced and bacterial-induced inflammation associates with increased numbers of pulmonary CD4+ and CD8+ cells expressing PD-1 and PD-L1 positive mononuclear phagocytes. We showed that blockade of PD-1 results in reduced lung damage and neutrophilic inflammation. We found increased concentrations of PD-L1 in serum collected during AECOPD. PD-L1 concentrations correlated positively with PD-1 concentrations in serum and BAL fluids.
COPD is often associated with lung cancer and COPD is an independent risk factor for lung cancer (5). Immune checkpoint inhibitors against PD-1 or PD-L1 are promising in patients with lung cancer and there is evidence that COPD-associated inflammation increases the sensitivity of lung cancer patients to targeted therapy against PD-1. Clinical studies suggest that patients with non-small cell lung cancer (NSCLC) with a coexisting COPD qualify for anti-PD-1 therapies (8, 23, 24). There is also evidence that the activation of the PD-1/PD-L1 axis is implicated in the pathogenesis of COPD (7–9). Thus, it is a matter of discussion whether the PD-1 pathway contributes to the development of COPD and whether therapeutic antibodies against PD-1 are effective in patients with COPD, even though the suggested mechanisms by which anti-PD-1/PD-L1 therapies result in decreased tumor burden, such as neoantigen-specific T cell responses, do not seem to be relevant in cancer-free patients with COPD (9, 10, 25). Therefore, and as experimental studies examining the role of PD-1/PD-L1 axis in preclinical COPD models are lacking, we decided to study the role of PD-1 in CS- and NTHi-dependent preclinical models. In both of our COPD models, treatment with a PD-1 blocking antibody resulted in significantly reduced lung damage. Moreover, the blockade of PD-1 was associated with decreased neutrophilic lung inflammation. Thus, in our experimental setups, activation of the PD-1 pathway drives lung inflammation and lung damage. Importantly, there were no significant differences in the subtype distribution and functionality of splenic CD8+T cells, body weight, and numbers of blood leukocytes between anti-PD-1-treated and isotype-treated mice, nor signs of intestinal inflammation after anti-PD-1 treatment. Thus, the improved outcome in our COPD models does not seem to be a result of systemic effects of our anti-PD-1 treatment regime.
Our finding that the blockade of PD-1 results in reduced lung damage is surprising given that the activation of PD-1 is a mechanism to avoid overwhelming adaptive immune responses (26). However, there is also evidence that the activation of the PD-1/PD-L1 axis results in harmful inflammation under pathological conditions. A recent study by Celada et al. (27) revealed that the activation of the PD-1/PD-L1 axis contributes to the progression of lung diseases independent of lung cancer. Celada et al. (27) could demonstrate that numbers of PD-1-expressing CD4+ cells are increased in sarcoidosis and idiopathic pulmonary fibrosis (IPF). These cells are mostly positive for IL-17A. The blockade of PD-1 resulted in a decreased expression of IL-17A ex vivo and to reduced disease symptoms in a model of bleomycin-induced lung fibrosis. We found that exposure to CS and NTHi resulted in increased numbers of PD-1-expressing CD4+ cells in the lung parenchyma. Further studies need to clarify whether these PD-1 expressing CD4+ cells drive harmful inflammation in our models of experimental CODP. In addition, we demonstrate that COPD-like inflammation also results in increased numbers of PD-1-expressing CD8+ cells. A study by McKendry et al. (8) demonstrated that COPD associates with dysregulation of the CD8+ population in lung parenchyma with an increased proportion of lung CD8+ cells in COPD. Additional studies are need to clarify whether PD-1-expressing CD8+ cells drive harmful inflammation in COPD and whether the blockade of the PD-1-immuncheckpoint affects the response to viral infection in these models.
It has been shown that the PD-1 pathway regulates the phagocytic capacity of macrophages and that the PD-1 blockade results in increased phagocytosis and intracellular killing of bacteria (28–30). We found that ex vivo activated alveolar macrophages from anti-PD-1-treated mice release reduced amounts of inflammatory cytokines. Thus, a reduced release of cytokines along with a restored phagocytic capacity of macrophages resulting in an improved removal of particles and apoptotic cells (e.g., neutrophils) may account for the observed response to the PD-1 blockade in our COPD models. Additional studies are needed to gain mechanistic insight into the role of the PD-1/PD-L1 pathway in the progression of COPD and its relevance for patient treatment.
PD-1 and PD-L1 have soluble forms which likely originate from the cleavage of membrane-bound proteins or splice variants (31). To the best of our knowledge, concentrations of PD-L1 and PD-1 have not been measured in BAL fluids from patients with stable COPD. We found that PD-1 concentrations were closely correlated with PD-L1 concentrations. It should be noted that only a relatively small number of BAL fluids was analyzed and that 17 out of 31 patients were diagnosed with lung cancer. Thus, larger cohorts are needed to examine whether concentrations of PD-L1 and PD-1 correlate with disease progression in patients with stable COPD independent of coexisting lung cancer. However, PD-L1 and PD-1 could be detected in all BALs tested, and it is of interest whether soluble forms of PD-L1 and PD-1 contribute to the pathogenesis of COPD. A function of soluble PD-L1 was shown in cancer-related research. Mahoney et al. (32) described a splice variant of PD-L1 resulting in a secreted form of PD-L1 that inhibits lymphocyte function in vitro. Frigola et al. (33) also identified a soluble form of PD-L1 with immunosuppressive activity. Thus, further studies are needed to understand whether soluble PD-L1 and PD-1 have a physiological function or reflect the presence of PD-L1 and PD-1 expressing inflammatory cells in the lungs of patients with COPD.
PD-L1 correlated positively with PD-1 in the serum of patients with stable COPD and during AECOPD. A positive correlation of PD-1 with PD-L1 has also been observed in other diseases, such as pancreatic cancer (34) and indicates a common regulation of PD-L1 and PD-1. Moreover, PD-L1 serum levels were significantly increased during exacerbation. Therefore, our data suggest that pulmonary inflammation results in increased systemic levels of potentially active soluble PD-L1. Whether this is of physiological relevance is unclear. However, soluble PD-L1 may affect the immune response to viral infections, which often associate with AECOPD. A limitation here is again the relative number of patients. Moreover, patients with AECOPD and stable COPD are from different samples which do not allow an intraindividual analysis of PD-L1 as a biomarker.
In conclusion, our data indicate a function of the PD-1/PD-L1 axis at the early stages of COPD. Inflammation-induced activation of the PD-1 pathway may contribute to disease progression. Further studies are needed to evaluate whether therapeutic interventions targeting the PD-1/PD-L1 axis are beneficial in stable COPD and during acute AECOPD. It needs to be evaluated whether the blockade of PD-1 affects the production of autoantibodies that likely contribute to the progression of COPD (35, 36) as well as the response to viral infections.
GRANTS
This study was supported by grants from Dr. Rolf M. Schwiete Stiftung (Nr. 11/2019) (to R.B.) and Deutsche Forschungsgemeinschaft (DFG) LI 1750/4-2 (to A.L.).
DISCLOSURES
No conflicts of interest, financial or otherwise are declared by the authors.
AUTHOR CONTRIBUTIONS
F.R., K.B., M.H., A.L., R.M.B., R.B., and C.B. conceived and designed research; F.R., K.B., G.V., P.C., A.A., C.H., and C.B. performed experiments; F.R., C.H., A.L., R.B., and C.B. analyzed data; F.R., M.H., and C.B. interpreted results of experiments; F.R. and C.B. prepared figures; F.R., C.H., M.D.M., M.H., A.L., R.M.B., R.B., and C.B. drafted manuscript; C.H., M.D.M., M.H., A.L., R.M.B., R.B., and C.B. edited and revised manuscript; F.R., K.B., G.V., P.C., A.A., C.H., M.D.M., M.H., A.L., R.M.B., R.B., and C.B. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank Andreas Kamyschnikow and Anja Honecker for excellent technical assistance.
REFERENCES
- 1.Decramer M, Janssens W. Chronic obstructive pulmonary disease and comorbidities. Lancet Respir Med 1: 73–83, 2013. doi: 10.1016/S2213-2600(12)70060-7. [DOI] [PubMed] [Google Scholar]
- 2.Lopez-Campos JL, Tan W, Soriano JB. Global burden of COPD. Respirology 21: 14–23, 2016. doi: 10.1111/resp.12660. [DOI] [PubMed] [Google Scholar]
- 3.Hoenderdos K, Condliffe A. The neutrophil in chronic obstructive pulmonary disease. Am J Respir Cell Mol Biol 48: 531–539, 2013. doi: 10.1165/rcmb.2012-0492TR. [DOI] [PubMed] [Google Scholar]
- 4.Sethi S. Infection as a comorbidity of COPD. Eur Respir J 35: 1209–1215, 2010. doi: 10.1183/09031936.00081409. [DOI] [PubMed] [Google Scholar]
- 5.Durham AL, Adcock IM. The relationship between COPD and lung cancer. Lung Cancer 90: 121–127, 2015. doi: 10.1016/j.lungcan.2015.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.He J, Hu Y, Hu M, Li B. Development of PD-1/PD-L1 pathway in tumor immune microenvironment and treatment for non-small cell lung cancer. Sci Rep 5: 13110, 2015. doi: 10.1038/srep13110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kalathil SG, Lugade AA, Pradhan V, Miller A, Parameswaran GI, Sethi S, Thanavala Y. T-regulatory cells and programmed death 1+ T cells contribute to effector T-cell dysfunction in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 190: 40–50, 2014. doi: 10.1164/rccm.201312-2293OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McKendry RT, Spalluto CM, Burke H, Nicholas B, Cellura D, Al-Shamkhani A, Staples KJ, Wilkinson TM. Dysregulation of antiviral function of CD8(+) T cells in the chronic obstructive pulmonary disease lung. Role of the PD-1-PD-L1 axis. Am J Respir Crit Care Med 193: 642–651, 2016. doi: 10.1164/rccm.201504-0782OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stoll P, Ulrich M, Bratke K, Garbe K, Virchow JC, Lommatzsch M. Imbalance of dendritic cell co-stimulation in COPD. Respir Res 16: 19, 2015. doi: 10.1186/s12931-015-0174-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Staples KJ, Wilkinson TM. Reply: The PD-1-PD-L1 axis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 194: 644–645, 2016. doi: 10.1164/rccm.201604-0863LE. [DOI] [PubMed] [Google Scholar]
- 11.Stoll P, Virchow JC, Lommatzsch M. The PD-1-PD-L1 axis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 194: 644, 2016. doi: 10.1164/rccm.201604-0752LE. [DOI] [PubMed] [Google Scholar]
- 12.Beisswenger C, Ritzmann F, Vella G, Lis A, Herr C, Bals R. The PD-1 immune checkpoint regulates COPD-like inflammation in mice (Abstract). Am J Respir Crit Care Med 199: A3783, 2019. doi: 10.1164/ajrccm-conference.2019.199.1_MeetingAbstracts.A3783. [DOI] [Google Scholar]
- 13.Akbay EA, Koyama S, Carretero J, Altabef A, Tchaicha JH, Christensen CL, Mikse OR, Cherniack AD, Beauchamp EM, Pugh TJ, Wilkerson MD, Fecci PE, Butaney M, Reibel JB, Soucheray M, Cohoon TJ, Janne PA, Meyerson M, Hayes DN, Shapiro GI, Shimamura T, Sholl LM, Rodig SJ, Freeman GJ, Hammerman PS, Dranoff G, Wong KK. Activation of the PD-1 pathway contributes to immune escape in EGFR-driven lung tumors. Cancer Discov 3: 1355–1363, 2013. doi: 10.1158/2159-8290.CD-13-0310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Akbay EA, Koyama S, Liu Y, Dries R, Bufe LE, Silkes M, Alam MM, Magee DM, Jones R, Jinushi M, Kulkarni M, Carretero J, Wang X, Warner-Hatten T, Cavanaugh JD, Osa A, Kumanogoh A, Freeman GJ, Awad MM, Christiani DC, Bueno R, Hammerman PS, Dranoff G, Wong KK. Interleukin-17A promotes lung tumor progression through neutrophil attraction to tumor sites and mediating resistance to PD-1 blockade. J Thorac Oncol 12: 1268–1279, 2017. doi: 10.1016/j.jtho.2017.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang Y, Kurupati R, Liu L, Zhou XY, Zhang G, Hudaihed A, Filisio F, Giles-Davis W, Xu X, Karakousis GC, Schuchter LM, Xu W, Amaravadi R, Xiao M, Sadek N, Krepler C, Herlyn M, Freeman GJ, Rabinowitz JD, Ertl HCJ. Enhancing CD8(+) T cell fatty acid catabolism within a metabolically challenging tumor microenvironment increases the efficacy of melanoma immunotherapy. Cancer Cell 32: 377–391.e9,2017. doi: 10.1016/j.ccell.2017.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ritzmann F, Jungnickel C, Vella G, Kamyschnikow A, Herr C, Li D, Menger MM, Angenendt A, Hoth M, Lis A, Bals R, Beisswenger C. IL-17C-mediated innate inflammation decreases the response to PD-1 blockade in a model of Kras-driven lung cancer. Sci Rep 9: 10353, 2019. doi: 10.1038/s41598-019-46759-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jungnickel C, Schmidt LH, Bittigkoffer L, Wolf L, Wolf A, Ritzmann F, Kamyschnikow A, Herr C, Menger MD, Spieker T, Wiewrodt R, Bals R, Beisswenger C. IL-17C mediates the recruitment of tumor-associated neutrophils and lung tumor growth. Oncogene 36: 4182–4190, 2017. doi: 10.1038/onc.2017.28. [DOI] [PubMed] [Google Scholar]
- 18.Voss M, Wolf L, Kamyschnikow A, Wonnenberg B, Honecker A, Herr C, Lepper PM, Wegmann M, Menger MD, Bals R, Beisswenger C. Il-17A contributes to maintenance of pulmonary homeostasis in a murine model of cigarette smoke-induced emphysema. Am J Physiol Lung Cell Mol Physiol 309: L188–L195, 2015. doi: 10.1152/ajplung.00388.2014. [DOI] [PubMed] [Google Scholar]
- 19.Brass DM, Hollingsworth JW, Cinque M, Li Z, Potts E, Toloza E, Foster WM, Schwartz DA. Chronic LPS inhalation causes emphysema-like changes in mouse lung that are associated with apoptosis. Am J Respir Cell Mol Biol 39: 584–590, 2008. doi: 10.1165/rcmb.2007-0448OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ghorani V, Boskabady MH, Khazdair MR, Kianmeher M. Experimental animal models for COPD: a methodological review. Tob Induc Dis 15: 25, 2017. doi: 10.1186/s12971-017-0130-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Herr C, Han G, Li D, Tschernig T, Dinh QT, Beißwenger C, Bals R. Combined exposure to bacteria and cigarette smoke resembles characteristic phenotypes of human COPD in a murine disease model. Exp Toxicol Pathol 67: 261–269, 2015. doi: 10.1016/j.etp.2015.01.002. [DOI] [PubMed] [Google Scholar]
- 22.Vella G, Ritzmann F, Wolf L, Kamyschnikov A, Stodden H, Herr C, Slevogt H, Bals R, Beisswenger C. IL-17C contributes to NTHi-induced inflammation and lung damage in experimental COPD and is present in sputum during acute exacerbations. PLoS One 16: e0243484, 2021. doi: 10.1371/journal.pone.0243484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Biton J, Ouakrim H, Dechartres A, Alifano M, Mansuet-Lupo A, Si H, Halpin R, Creasy T, Bantsimba-Malanda C, Arrondeau J, Goldwasser F, Boudou-Rouquette P, Fournel L, Roche N, Burgel PR, Goc J, Devi-Marulkar P, Germain C, Dieu-Nosjean MC, Cremer I, Herbst R, Damotte D. Impaired tumor-infiltrating T cells in patients with chronic obstructive pulmonary disease impact lung cancer response to PD-1 blockade. Am J Respir Crit Care Med 198: 928–940, 2018. doi: 10.1164/rccm.201706-1110OC. [DOI] [PubMed] [Google Scholar]
- 24.Mark NM, Kargl J, Busch SE, Yang GHY, Metz HE, Zhang H, Hubbard JJ, Pipavath SNJ, Madtes DK, Houghton AM. Chronic obstructive pulmonary disease alters immune cell composition and immune checkpoint inhibitor efficacy in non-small cell lung cancer. Am J Respir Crit Care Med 197: 325–336, 2018. doi: 10.1164/rccm.201704-0795OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med 8: 328rv4, 2016. doi: 10.1126/scitranslmed.aad7118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fife BT, Pauken KE. The role of the PD-1 pathway in autoimmunity and peripheral tolerance. Ann NY Acad Sci 1217: 45–59, 2011. doi: 10.1111/j.1749-6632.2010.05919.x. [DOI] [PubMed] [Google Scholar]
- 27.Celada LJ, Kropski JA, Herazo-Maya JD, Luo W, Creecy A, Abad AT, Chioma OS, Lee G, Hassell NE, Shaginurova GI, Wang Y, Johnson JE, Kerrigan A, Mason WR, Baughman RP, Ayers GD, Bernard GR, Culver DA, Montgomery CG, Maher TM, Molyneaux PL, Noth I, Mutsaers SE, Prele CM, PeeblesRS , Jr., Newcomb DC, Kaminski N, Blackwell TS, Van Kaer L, Drake WP. PD-1 up-regulation on CD4(+) T cells promotes pulmonary fibrosis through STAT3-mediated IL-17A and TGF-beta1 production. Sci Transl Med 10: eaar8356, 2018. doi: 10.1126/scitranslmed.aar8356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gordon SR, Maute RL, Dulken BW, Hutter G, George BM, McCracken MN, Gupta R, Tsai JM, Sinha R, Corey D, Ring AM, Connolly AJ, Weissman IL. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 545: 495–499, 2017. doi: 10.1038/nature22396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huang X, Venet F, Wang YL, Lepape A, Yuan Z, Chen Y, Swan R, Kherouf H, Monneret G, Chung CS, Ayala A. PD-1 expression by macrophages plays a pathologic role in altering microbial clearance and the innate inflammatory response to sepsis. Proc Natl Acad Sci USA 106: 6303–6308, 2009. doi: 10.1073/pnas.0809422106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shen L, Gao Y, Liu Y, Zhang B, Liu Q, Wu J, Fan L, Ou Q, Zhang W, Shao L. PD-1/PD-L pathway inhibits M.tb-specific CD4(+) T-cell functions and phagocytosis of macrophages in active tuberculosis. Sci Rep 6: 38362, 2016. doi: 10.1038/srep38362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhu X, Lang J. Soluble PD-1 and PD-L1: predictive and prognostic significance in cancer. Oncotarget 8: 97671–97682, 2017. doi: 10.18632/oncotarget.18311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mahoney KM, Shukla SA, Patsoukis N, Chaudhri A, Browne EP, Arazi A, Eisenhaure TM, PendergraftWF , 3rd, Hua P, Pham HC, Bu X, Zhu B, Hacohen N, Fritsch EF, Boussiotis VA, Wu CJ, Freeman GJ. A secreted PD-L1 splice variant that covalently dimerizes and mediates immunosuppression. Cancer Immunol Immunother 68: 421–432, 2019. doi: 10.1007/s00262-018-2282-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Frigola X, Inman BA, Lohse CM, Krco CJ, Cheville JC, Thompson RH, Leibovich B, Blute ML, Dong H, Kwon ED. Identification of a soluble form of B7-H1 that retains immunosuppressive activity and is associated with aggressive renal cell carcinoma. Clin Cancer Res 17: 1915–1923, 2011. doi: 10.1158/1078-0432.CCR-10-0250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kruger S, Legenstein ML, Rosgen V, Haas M, Modest DP, Westphalen CB, Ormanns S, Kirchner T, Heinemann V, Holdenrieder S, Boeck S. Serum levels of soluble programmed death protein 1 (sPD-1) and soluble programmed death ligand 1 (sPD-L1) in advanced pancreatic cancer. Oncoimmunology 6: e1310358, 2017. doi: 10.1080/2162402X.2017.1310358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lee SH, Goswami S, Grudo A, Song LZ, Bandi V, Goodnight-White S, Green L, Hacken-Bitar J, Huh J, Bakaeen F, Coxson HO, Cogswell S, Storness-Bliss C, Corry DB, Kheradmand F. Antielastin autoimmunity in tobacco smoking-induced emphysema. Nat Med 13: 567–569, 2007. doi: 10.1038/nm1583. [DOI] [PubMed] [Google Scholar]
- 36.Wen L, Krauss-Etschmann S, Petersen F, Yu X. Autoantibodies in chronic obstructive pulmonary disease. Front Immunol 9: 66, 2018. doi: 10.3389/fimmu.2018.00066. [DOI] [PMC free article] [PubMed] [Google Scholar]