Abstract
The purpose of this study was to evaluate the SARS‐CoV‐2 immunoglobulin M/immunoglobulin G (IgM/IgG) rapid antibody test results in symptomatic patients with COVID‐19 and their chest computed tomography (CT) data. A total of 320 patients admitted to our hospital for different durations due to COVID‐19 were included in the study. Serum samples were obtained within 0–7 days from COVID‐19 patients confirmed by reverse‐transcription polymerase chain reaction (RT‐PCR) and chest CT scan. According to the SARS‐CoV‐2 RT‐PCR results, the patients included in the study were divided into two groups: PCR positive group (n = 46) and PCR negative group (n = 274). The relationship between chest CT and rapid antibody test results were compared statistically. Of the 320 COVID‐19 serum samples, IgM, IgG, and IgM/IgG were detected in 8.4%, 0.3%, and 11.6% within 1 week, respectively. IgG/IgM antibodies were not detected in 79.7% of the patients. In the study, 249 (77.8%) of 320 patients had positive chest CT scans. Four (5.6%) of 71 patients with negative chest CT scans had IgM and two (2.8%) were both IgM/IgG positive. IgM was detected in 23 (9.2%), IgG in one (0.4%), and IgM/IgG in 35 (14%) of chest CT scan positive patients. The rate of CT findings in patients with antibody positivity was found to be significantly higher than those with antibody negativity. The results of the present study show the accurate and equivalent performance of serological antibody assays and chest CT in detecting SARS‐CoV‐2 within 0–7 days from the onset of COVID19 symptoms. When RT‐PCR is not available, we believe that the combination of immunochromatographic test and chest CT scan can increase diagnostic sensitivity for COVID‐19.
Keywords: chest CT, COVID‐19, lateral flow, rapid diagnosis, RT‐PCR, SARS‐CoV‐2
1. INTRODUCTION
2019 Coronavirus disease (COVID‐19), originated in Hubei Province, China, in December 2019. 1 COVID‐19 disease, caused by Severe Acute Respiratory Syndrome Coronavirus‐2 (SARS‐CoV‐2), is an infectious disease that has started to spread rapidly and has turned into a pandemic within three months from its first detection. 2 As there is no specific treatment or vaccine for COVID‐19, early diagnosis and subsequent isolation of infected patients are important for disease control. Today, the reverse‐transcription polymerase chain reaction (RT‐PCR) test for SARS‐CoV‐2 is considered the gold standard for the diagnosis of COVID‐19. However, as RT‐PCR is time‐consuming, and requires special equipment and trained laboratory technicians, it is not widely used in the clinical setting. As the RT‐PCR test is performed on the upper and lower respiratory tract samples, it increases the risk of exposure to viral droplets during the collection and RNA isolation process. Therefore, alternative diagnostic tests to RT‐PCR are required for the clinical management of COVID‐19. Detection of antibodies against SARS‐CoV‐2 can play a complementary role with the RT‐PCR test in the diagnosis of COVID‐19 and in evaluating the immune status of individuals. Also, serological data will enable the gathering of important epidemiological information about the spread of the epidemic, and its morbidity and mortality. Additionally, the detection of antibodies against SARS‐CoV‐2 will play an important role in determining vaccine development strategies. 1 , 3 , 4
As a routine imaging tool in the diagnosis of COVID‐19, chest computed tomography (CT) scan is a relatively easy, fast, and noninvasive radiological diagnostic tool. Chest CT analysis plays an important role not only in early detection and diagnosis especially in false‐negative RT‐PCR tests but also in monitoring the clinical course and evaluating the severity of the disease. 5 , 6 It has been reported that the RT‐PCR test is only 30%–60% sensitive in the diagnosis of COVID‐19, and is less sensitive than chest CT during the early course of the disease. For example, five suspected cases were shown to have ground‐glass opacity and/or mixed consolidation on chest CT scan, but the initial RT‐PCR test was negative. A few days later, all five cases tested for the second or third examinations were confirmed as positive for SARS‐CoV‐2. 6 , 7
In cases where the RT‐PCR test of symptomatic and/or asymptomatic cases is negative, serological tests and chest CT scanning can be considered complementary tests for the RT‐PCR technique. It may also be useful in the diagnosis of SARS‐Cov‐2 infection by providing promising clinical findings. Findings to be obtained from antibody testing will bring benefits for epidemiological studies for the COVID‐19 pandemic in Nigde, Turkey. Thus, it will provide advantages such as increasing the reliability of test results and preventing epidemics. This study aimed to evaluate the SARS‐CoV‐2 immunoglobulin M/immunoglobulin G (IgM/IgG) rapid antibody test results from serum samples taken from admitted patients with suspected COVID‐19 and their chest CT data.
2. MATERIALS AND METHODS
2.1. Patients and data sources
A total of 320 inpatient COVID‐19 cases, admitted to Niğde Ömer Halisdemir University Training and Research Hospital between March 11 and June 30, 2020, were enrolled in this study. Serum samples were obtained within 0–7 days from COVID‐19 patients confirmed by RT‐PCR and chest CT scan. The date of symptoms onset, clinical classification, RT‐PCR testing results, hospitalization period, and demographic information was obtained from the clinical records of our hospital. In the study, serum samples were taken from patients before the COVID‐19 pandemic were used as negative controls to evaluate the analytical sensitivity and specificity of the antibody test.
Combined throat/nasal swabs were obtained from the patients included in the study and RT‐PCR analysis was performed. The vNAT Transfer Tube (Bio‐speedy) was used for RNA isolation from the swab samples. RNA samples were stored at −20°C until the amplification process. A COVID‐19 RT‐PCR detection kit (Bio‐Speedy® SARS‐CoV‐2 double gene RT‐qPCR) was used to obtain complementary DNA and for amplification. RT‐PCR assay targeting the nucleocapsid protein (N) and open reading frame 1 ab (ORF1ab) genes were performed according to the manufacturer's instructions.
According to the SARS‐CoV‐2 RT‐PCR results, the symptomatic patients included in the study were divided into two groups: PCR positive group (n = 46) and PCR negative group (n = 274). RT‐PCR negative patients were clinically diagnosed with SARS‐CoV‐2 infection according to the guideline for the adult patient treatment of COVID‐19 (SARS‐CoV‐2 infection) published by the National Health Commission of the Ministry of Health of the Republic of Turkey. 8 The clinical diagnosis according to the guidelines above means that cases are negative by RT‐PCR testing but present viral pneumonia by the chest CT.
Chest CT imaging was performed in the supine position with the arms raised and at the deep‐inspiration breath‐hold. All chest CT findings of the patients were classified as positive or negative for COVID‐19. Chest CT findings (bilateral or multilobar involvement, ground‐glass opacity, subpleural involvement, pleural effusion) of patients enrolled in this study were evaluated by an Infection and Clinical Microbiologist to determine clinical features caused by COVID‐19.
2.2. Collection of serum samples
Blood samples taken from the patients (5–6 ml) were placed in tubes containing separator gel (SST) and centrifuged at 10,000g for 5 min. Patient sera were stored at −80°C until the day of the study. All risks related to the storage condition and transporting of the samples were applied following the conditions specified by the Turkish Republic Ministry of Health and Microbiology Laboratory guidelines. 8
2.3. Detection of SARS‐CoV‐2 IgM/IgG antibodies
Qualitative detection of antibodies was performed using the COVID‐19 IgM/IgG rapid test kit developed for SARS‐CoV‐2 (colloidal gold) based on the Lateral flow method (Singclean®) according to the manufacturer's instructions. Briefly, 10 μl of blood or serum was added to the sample pad of the antibody test. Two drops of sample buffer (phosphate buffer, NaCl, tween‐20) were then added to the same sample pad, and the results were interpreted after a 10‐min incubation period. The presence of only the control line is negative, the presence of the control line and IgM line is IgM positive, the presence of the IgG line together with the control line is IgG positive, and the presence of IgM and IgG lines together with the control line are evaluated as both IgM and IgG positive.
2.4. Statistical analysis
SPSS 20.0 was used for statistical analysis. χ 2 test was used in statistical analysis. Pearson correlation test was used to see the level of agreement between the rapid antibodies test and chest CT scan. Significance was set at a p < 0.05. Cohen's κ statistics were used to evaluate the correlation between rapid antibody testing and chest CT methods.
3. RESULTS
In this retrospective study, a total of 320 patients including 162 (50.6%) male and 158 (49.4%) female were included. The patients were between the ages of 18–90 (mean age 54.14). The clinical signs of COVID‐19 patients are shown in Table 1. The most common symptoms in patients were; fever (75.3%), headache (70.6%), myalgia (67.5%), cough (67.8%), dyspnea (25%), loss of taste and smell (19.7%), and diarrhea (4.0%). RT‐PCR results of 320 patients were determined as 46 (13.2%) positive and 274 (85.6%) negative.
Table 1.
The demographic and clinical characteristics of the patients
| Parameter | Value (n/%) |
|---|---|
| The average age | 54.14 (18–90 years old) |
| Gender | Male (162/50.6%), female (158/49.4%) |
| Symptoms | |
| Fever | 241 (75.3%) |
| Headache | 226 (70.6%) |
| Myalgia | 216 (67.5%) |
| Cough | 217 (67.8%) |
| Dyspnea | 80 (25%) |
| Loss of smell and taste | 63 (19.7%) |
| Diarrhea | 13 (4.0%) |
| C t range for RT‐PCR | 16–40 |
Abbreviations: C t, threshold cycle; RT‐PCR, reverse‐transcription polymerase chain reaction; RT‐PCR positive (C t ≤ 38), RT‐PCR negative (C t > 38).
3.1. Rapid IgM/IgG antibody test
The 320 serum samples collected for the study were evaluated by rapid card test based on the lateral flow method of IgM/IgG antibodies. The results of the antibody test against SARS‐CoV‐2 are shown in Table 2. In a total of 320 inpatients, IgM was detected in 27 (8.4%), IgG in 1 (0.3%), and both IgM and IgG were found positive in 37 (11.6%) of the serum samples included in the study. IgG/IgM antibodies were not detected in 255 (79.7%) of the patient samples. The sensitivity and specificity of the rapid IgM/IgG antibody test were 20.3% and 95%, respectively.
Table 2.
Detection of IgM/IgG antibody in serum samples of patients with COVID‐19
| Presence of antibodies | Number of patients (n = 320) | Control group (n = 40) |
|---|---|---|
| IgM | 27 | 0 |
| IgG | 1 | 2 |
| Both IgM/IgG | 37 | 0 |
| Sensitivity | 20.3% | – |
| Specificity | – | 95% |
Abbreviation: IgM/IgG, immunoglobulin M/immunoglobulin G.
When the presence of antibodies according to the RT‐PCR results were evaluated, 46 patients with positive RT‐PCR results had two (4.3%) IgM, and six (13.0%) IgM and IgG positive. In patients with negative RT‐PCR results (n = 274), IgG was detected in one patient (0.36%), IgM in 25 patients (9.1%), and IgM/IgG in 31 patients (11.3%). No antibodies were detected in the remaining patients with positive (38/46) and/or negative (217/274) RT‐PCR scans.
In the negative control patient group (n = 40), only two serum samples were found IgG positive. We think it is likely due to the imperfect performance of existing tests and cross‐reaction with seasonal CoVs.
3.2. Chest CT findings
Chest CT scans of symptomatic patients were performed on the day of admission to the hospital. The percentage of findings that are compatible with the clinical features of COVID‐19 in the chest CT scans of the patients is given in Figure 1. Mostly, ground‐glass opacity (68.5%) findings were detected in the chest CT scans of the patients. When clinical findings consistent with COVID‐19 were evaluated collectively, it was determined that 249 (77.8%) of 320 patients had positive and 71 patients (22.2%) had negative chest CT scans. The sensitivity rate of chest CT scans was calculated as 77.8% (249/320).
Figure 1.
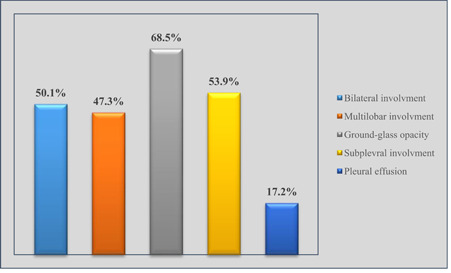
Distribution of chest CT findings of COVID‐19 patients. CT, computed tomography
In the present study, the presence of antibodies was evaluated according to the chest CT scan results. Four (5.6%) of those with negative chest CT scans (n = 71) had IgM positive and two (2.8%) were positive for both IgM and IgG. In patients with positive chest CT scan (n = 249) 23 (9.2%) IgM, 1 (0.4%) IgG, and 35 (14.0%) both IgM/IgG were positive. No antibodies were found in the serum of remaining patients with positive (190/249) and/or negative (65/71) chest CT scans.
Regarding chest CT findings according to RT‐PCR results, it was found to be positive in 28 (61.0%) of the RT‐PCR positive patients (n = 46). The rest of the positive RT‐PCR patients (18/46) were without lesions at chest CT. However, in patients with negative RT‐PCR (n = 274) 221 patients (80.7%) had positive chest CT scans. In contrast, typical radiological features were not detected in 53 (19.3%) patients who were RT‐PCR negative.
When the rapid antibody test and chest CT scan results were evaluated together (Table 3), the rate of CT findings in patients with antibody positivity (n = 65) was found to be significantly higher (90.8%) than those with antibody negativity (74.5%) (p = 0.005).
Table 3.
Rapid antibody test and chest CT findings for patients with COVID‐19
| Parameters | CT findings | Total | |||
|---|---|---|---|---|---|
| Positive | Negative | ||||
| SARS‐CoV‐2 IgM/IgG | Negative | n | 190 | 65 | 255 |
| Within SARS‐CoV‐2 IgM/IgG (%) | 74.5% | 25.5% | 100.0% | ||
| Positive | n | 59 | 6 | 65 | |
| Within SARS‐CoV‐2 IgM/IgG (%) | 90.8% | 9.2% | 100.0% | ||
| Total | n | 249 | 71 | 320 | |
| Within SARS‐CoV‐2 IgM/IgG (%) | 77.8% | 22.2% | 100.0% | ||
| PPV | 23.69% | ||||
| NPV | 91.55% | ||||
| Cohen's κ coefficient | 0.08 (95% CI, 0.024–0.13) | ||||
Abbreviations: CI, confidence interval; IgM/IgG, immunoglobulin M/immunoglobulin G; NPV, negative predictive value; PPV, positive predictive value.
Cohen's κ coefficient test was adopted for the analysis in the study. The Cohen's κ coefficient value was 0.08 (95% confidence interval, 0.024–0.13, p = 0.005), which represented the slight agreement between the two diagnostic methods.
The positive predictive value and negative predictive value of rapid antibody test and chest CT findings were 23.69%, and 91.55%, respectively.
4. DISCUSSION
Early diagnosis of COVID‐19 is very important to treat and control the disease. An accurate, fast, and cost‐effective diagnostic method is required for the diagnosis of the disease during the pandemic process. Nowadays molecular methods, serological tests, and chest CT scans are used for this purpose. 9 , 10
In the diagnosis of COVID‐19, the RT‐PCR test is used as a gold standard to detect viral RNA in respiratory tract samples. However, the RT‐PCR test leads to false‐negative results due to problems in sample collection and/or detection. Also, it is a time‐consuming and expensive diagnostic method. 11 , 12
Serological testing is universally recognized as an accurate and appropriate diagnostic method for identifying asymptomatic and symptomatic persons and monitoring the immune status of people recovering from acute COVID‐19 infection. 13 Although several countries have purchased one or more of the rapid antibody tests, inconsistencies have been reported about the sensitivity and specificity rates of these tests in studies. 14
Compared to other diagnostic tests (e.g., RT‐PCR), chest CT imaging is a more reliable, practical, and fast method to diagnose and evaluate COVID‐19, especially during the epidemic period. 15 Studies have shown that the sensitivity of chest CT in symptomatic patients is high (74.3%–97%). The clinical performance of chest CT depends on factors such as patient population differences, disease severity, and accessibility to chest CT scans. 16 , 17 , 18
In this study, the antibody response in symptomatic COVID‐19 patients was investigated with the rapid IgM/IgG card test. The serological data obtained were evaluated with the chest CT scan data of the same patients. The rapid antibody test used in this study was found to be less sensitive (20.3%, 65/320) for serum samples collected within 0–7 days from the onset of symptoms. Test performance features (sensitivity 95.7% and specificity 97.3%) provided by the manufacturer were found higher than those observed in our study. In the present study, IgM/IgG antibodies were not detected in 255 (79.7%) of the patient samples. This is likely due to a variety of factors, such as the state of the patient's immune system, quality and type of specimen, sample collection time, antibody level, and sample processing techniques.
In our study, the chest CT scan was positive in 249 of the symptomatic patients. The chest CT scans were found to be positive in 90.8% of the patients who were antibody positive and 74.5% in those who were antibody negative (Table 3).
A study evaluating rapid antibody test and chest CT scan method was performed by Imai et al. 12 They examined 139 serum samples collected from 112 patients with COVID‐19. In the study, 90 COVID‐19 serum specimens, IgM, IgG, and IgM/IgG were detected in 27.8%, 3.3%, and 27.8%, respectively (in total 58.9%) of the specimens collected within 1 week. The diagnostic sensitivity of the chest CT scan was 74.3% (55/74) for the symptomatic patients.
In the current study, IgM, IgG, and both IgG/IgM were detected in 8.4%, 0.3%, and 11.6%, respectively (in total 20.3%) in the serum samples collected from symptomatic patients within one week. The sensitivity rate of chest CT scans was 77.8% (249/320) for the symptomatic patients.
In our study, patients with high suspicion of COVID‐19 who were negative for RT‐PCR (n = 274) but had radiographic findings were considered “clinically diagnosed” patients. In patients with negative RT‐PCR tests, a combination of exposure history, clinical symptoms, and typical CT imaging features should be used to identify COVID‐19 with higher sensitivity.
The present study showed that with the rapid antibody card test, SARS‐CoV‐2 IgM/IgG antibodies were found less frequently (65/320) in RT‐PCR positive and negative symptomatic patients at significant levels. In contrast, chest CT scans of patients with positive and/or negative RT‐PCR were found to be positive (249/320). Considering the results obtained from the rapid antibody test and chest CT scan, the chest CT screening test for the early diagnosis of COVID‐19 in symptomatic patients stands out with a sensitivity rate of 77.8%. Also, specific radiological features of COVID‐19 were not detected in 22.2% of symptomatic patients.
According to the results of this study, it is highly recommended to use symptoms, rapid antibody tests, and other diagnostic methods for diagnosing COVID‐19.
Our study has some limitations; only symptomatic patients were included in the study. Therefore, it is not known whether the antibody response is similar in asymptomatic individuals. Serum samples were collected for antibody testing within 1 week from the onset of symptoms. Therefore, it is necessary to take samples/scans at different times to obtain clear information about the sensitivity of these tests. Besides this, more detailed studies with different methods (such as enzyme‐linked immunosorbent assay) on consecutive samples are needed. Multi‐center studies should be conducted to determine the usefulness of both tests for the diagnosis of COVID‐19.
5. CONCLUSION
When the data obtained in our study were examined, the sensitivity of rapid antibody tests alone was not found to be sufficient in the diagnosis of COVID‐19 within 1 week from the onset of COVID‐19 symptoms. This study shows the accurate and equivalent performance of serological antibody assays and chest CT in diagnosing COVID‐19 within 0–7 days from the onset of COVID19 symptoms. Therefore, to properly manage the pandemic process, we believe that the combination of immunochromatographic test and chest CT scan can serve as a rapid diagnostic test of RT‐PCR‐negative suspected patients and screening of SARS CoV‐2 carriers. These tests/methods cannot take the place of RT‐PCR but may increase the diagnostic sensitivity for COVID‐19.
CONFLICT OF INTERESTS
The authors declare that there are no conflicts of interest.
ETHICS STATEMENT
This study was reviewed and approved by the Ethics Committee of Nigde Omer Halisdemir University Faculty of Medicine (Approval no. 2020/21).
AUTHOR CONTRIBUTIONS
Ali Ozturk designed the study. Ali Ozturk and Taylan Bozok conducted the laboratory tests. Ali Ozturk, Taylan Bozok and Tugce Simsek Bozok collected and analyzed the data. Ali Ozturk prepared the manuscript. Ali Ozturk, Taylan Bozok and Tugce Simsek Bozok edited and reviewed the manuscript. All the authors critically reviewed and revised the manuscript draft, and approved the final version of the manuscript.
Ozturk A, Bozok T, Simsek Bozok T. Evaluation of rapid antibody test and chest computed tomography results of COVID‐19 patients: A retrospective study. J Med Virol. 2021;93:6582‐6587. 10.1002/jmv.27209
Contributor Information
Ali Ozturk, Email: ali.81ozturk@gmail.com.
Taylan Bozok, Email: taylanbozok@hotmail.com.
Tugce Simsek Bozok, Email: tugce_0103@hotmail.com.
REFERENCES
- 1. Chen Y, Chan KH, Kang Y, et al. A sensitive and specific antigen detection assay for Middle East respiratory syndrome coronavirus. Emerg Microbes Infect. 2015;4(4):e26. 10.1038/emi.2015.26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Amanat F, Stadlbauer D, Strohmeier S, et al. A serological assay to detect SARS‐CoV‐2 seroconversion in humans. Nat Med. 2020;26(7):1033‐1036. 10.1038/s41591-020-0913-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Xiang F, Wang X, He X, et al. Antibody detection and dynamic characteristics in patients with coronavirus disease 2019. Clin Infect Dis. 2020;5;71(8):1930‐1934. 10.1093/cid/ciaa461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Zhao J, Yuan Q, Wang H, et al. Antibody responses to sars‐cov‐2 in patients with novel coronavirus disease 2019. Clin Infect Dis. 2020;71(16):2027‐2034. 10.1093/cid/ciaa344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Long QX, Liu BZ, Deng HJ, et al. Antibody responses to SARS‐CoV‐2 in patients with COVID‐19. Nat Med. 2020;26(6):845‐848. 10.1038/s41591-020-0897-1 [DOI] [PubMed] [Google Scholar]
- 6. Xie J, Ding C, Li J, et al. Characteristics of patients with coronavirus disease (COVID‐19) confirmed using an IgM‐IgG antibody test. J Med Virol. 2020;92(10):2004‐2010. 10.1002/jmv.25930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wang W, Wang H, Deng Y, et al. Characterization of anti‐MERS‐CoV antibodies against various recombinant structural antigens of MERS‐CoV in an importedcase in China. Emerg Microbes Infect. 2016;5(11):e113. 10.1038/emi.2016.114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Republic of Turkey Ministry of Health . COVID‐19 (SARS‐CoV2 infection) guide. 2020. 1–31. https://saglik.gov.tr/
- 9. Xiang F, Wang X, He X, et al. Antibody detection and dynamic characteristics in patients with coronavirus disease 2019. Clin Infect Dis. 2020;71(8):1930‐1934. 10.1093/cid/ciaa461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Vashist SK. In vitro diagnostic assays for COVID‐19: recent advances and emerging trends. Diagnostics (Basel). 2020;10(4):202. 10.3390/diagnostics10040202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chaimayo C, Kaewnaphan B, Tanlieng N, et al. Rapid SARS‐CoV‐2 antigen detection assay in comparison with real‐time RT‐PCR assay for laboratory diagnosis of COVID‐19 in Thailand. Virol J. 2020;17(1):177. 10.1186/s12985-020-01452-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Imai K, Tabata S, Ikeda M, et al. Clinical evaluation of an immunochromatographic IgM/IgG antibody assay and chest computed tomography for the diagnosis of COVID‐19. J Clin Virol. 2020;128:104393. 10.1016/j.jcv.2020.104393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ortiz‐Prado E, Simbaña‐Rivera K, Gómez‐ Barreno L, et al. Clinical, molecular, and epidemiological characterization of the SARS‐CoV‐2 virus and the coronavirus disease 2019 (COVID‐19), a comprehensive literature review. Diagn Microbiol Infect Dis. 2020;98(1):115094. 10.1016/j.diagmicrobio.2020.115094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ong DSY, de Man SJ, Lindeboom FA, Koeleman J. Comparison of diagnostic accuracies of rapid serological tests and ELISA to molecular diagnostics in patients with suspected coronavirus disease 2019 presenting to the hospital. Clin Microbiol Infect. 2020;26(8):1094.e7‐1094.e10. 10.1016/j.cmi.2020.05.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Mukhtar B, Mukhtar MO, Malik B. Radiological changes observed in COVID 19 pneumonia and utilization of CT scan as a screening tool along with a real‐time reverse transcriptase‐polymerase chain reaction (rRT‐PCR) for effective diagnosis. MSD. 2020;7(8):578‐583. 10.36472/msd.v7i8.411 [DOI] [Google Scholar]
- 16. Power SP, Moloney F, Twomey M, James K, O'Connor OJ, Maher MM. Computed tomography and patient risk: facts, perceptions and uncertainties. World J Radiol. 2016;8(12):902‐915. 10.4329/wjr.v8.i12.902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Parekh M, Donuru A, Balasubramanya R, Kapur S. Review of the chest CT differential diagnosis of ground‐glass opacities in the COVID era. Radiology. 2020;297:202504(3):E289‐E302. 10.1148/radiol.2020202504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ai T, Yang Z, Hou H, et al. Correlation of chest ct and RT‐PCR testing for coronavirus disease 2019 (COVID‐19) in China: a report of 1014 cases. Radiology. 2020;296(2):32‐40. 10.1148/radiol.2020200642 [DOI] [PMC free article] [PubMed] [Google Scholar]


