This randomized clinical trial compares the use of postoperative narcotics between patients who can select whether they want opioids prescribed after cervical endocrine surgery and those who receive standard postoperative prescription of opioids.
Key Points
Question
Does limiting opioid prescription to patients who request it (ie, opt-in program) decrease the quantity of opioids prescribed and consumed compared with patients given a routine prescription after outpatient cervical endocrine surgery?
Findings
In this randomized clinical trial of 102 adults undergoing thyroidectomy or parathyroidectomy, patients randomized to the opt-in program were prescribed less opioids without reporting worse postoperative pain scores or health-related quality of life. No patients who opted out required a rescue opioid prescription.
Meaning
In this randomized clinical trial, an opt-in strategy for opioid prescriptions after endocrine surgery was noninferior to use of routine prescriptions with respect to postoperative pain and reduced overprescription of opioids.
Abstract
Importance
Historically, opioid pain medications have been overprescribed following thyroid and parathyroid surgery. Many narcotic prescriptions are incompletely consumed, creating waste and opportunities for abuse.
Objective
To determine whether limiting opioid prescriptions after outpatient thyroid and parathyroid surgery to patients who opt in to narcotic treatment reduces opioid consumption without increasing postoperative pain compared with usual care (routine narcotic prescriptions).
Design, Setting, and Participants
A randomized clinical trial of Postoperative Opt-In Narcotic Treatment (POINT) or routine narcotic prescription (control) was conducted at a single tertiary referral center from June 1 to December 30, 2020. A total of 180 adults undergoing ambulatory cervical endocrine surgery, excluding patients currently receiving opioids, were assessed for eligibility. POINT patients received perioperative pain management counseling and were prescribed opioids only on patient request. Patients reported pain scores (0-10) and medication use through 7 daily postoperative surveys. Logistic regression was used to determine factors associated with opioid consumption.
Interventions
Patients in the POINT group were able to opt in or out of receiving prescriptions for opioid pain medication on discharge. Control patients received routine opioid prescriptions on discharge.
Main Outcomes and Measures
Daily peak pain score through postoperative day 7 was the primary outcome. Noninferiority was defined as a difference less than 2 on an 11-point numeric rating scale from 0 to 10. Analysis was conducted on the evaluable population.
Results
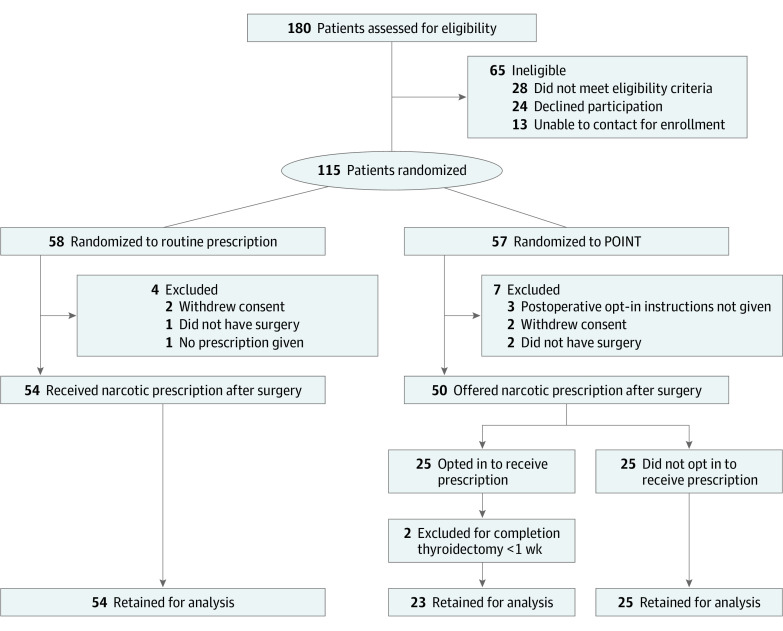
Of the 180 patients assessed for eligibility, the final study cohort comprised 102 patients: 48 randomized to POINT and 54 to control. Of these, 79 patients (77.5%) were women and median age was 52 (interquartile range, 43-62) years. A total of 550 opioid tablets were prescribed to the control group, and 230 tablets were prescribed to the POINT group, in which 23 patients (47.9%) opted in for an opioid prescription. None who opted out subsequently required rescue opioids. In the first postoperative week, 17 POINT patients (35.4% of survey responders in the POINT group) reported consuming opioids compared with 27 (50.0%) control patients (P = .16). Median peak outpatient pain scores were 6 (interquartile range, 4-8) in the control group vs 6 (interquartile range, 5-7) in the POINT group (P = .71). In multivariate analysis, patients with a history of narcotic use were 7.5 times more likely to opt in (95% CI, 1.61-50.11; P = .02) and 4.8 times more likely to consume opioids (95% CI, 1.04-1.52; P = .01). Higher body mass index (odds ratio, 1.11; 95% CI, 1.01-1.23; P = .03) and highest inpatient postoperative pain score (odds ratio, 1.24; 95% CI, 1.04-1.52; P = .02) were also associated with opioid consumption.
Conclusions and Relevance
In this trial, an opt-in strategy for postoperative narcotics reduced opioid prescription without increasing pain after cervical endocrine surgery.
Trial Registration
ClinicalTrials.gov Identifier: NCT04710069
Introduction
The opioid epidemic has increased scrutiny on the surgeon’s contribution to the overprescription of narcotic pain medications.1,2,3 Postoperative pain following cervical endocrine operations has generally been overestimated, and patients are prescribed far more opioids than they consume.4,5,6,7 Not only is overprescribing wasteful, it also places patients and their families at risk for addiction and opioid abuse.8 Studies have reported that 5% to 10% of patients prescribed postoperative narcotics had persistent opioid use beyond the perioperative period.9,10,11,12,13 In a recent prospective study of 216 patients who underwent outpatient cervical endocrine surgery, Ruffolo et al14 reported that most patients chose to forgo opioids after implementation of an opt-in program, but lack of follow-up pain assessment raised questions of adequate pain treatment after discharge.
We conducted a randomized clinical trial comparing perioperative pain management counseling with opioid prescription only on patient request with usual care with routine opioid prescription. The purpose of the trial was to determine whether allowing patient choice could reduce opioid consumption without having a negative effect on patients’ postoperative pain levels and quality of life. The primary outcome was daily postoperative pain score through postoperative day 7. Secondary outcomes included quantity of opioids prescribed and consumed, health-related quality of life (HRQOL), and clinical factors associated with opioid request and consumption. We hypothesized that postoperative pain would be equivalent between opt-in and routine opioid prescribing strategies and that an opt-in strategy would result in reduced opioid consumption.
Methods
Study Setting and Design
We conducted a randomized clinical trial from June 1 to December 30, 2020, to compare pain scores and opioid consumption between an opt-in strategy and routine narcotic prescription following cervical endocrine surgery at a single institution. The protocol is available in Supplement 1. All adult (age, ≥18 years), English-speaking patients scheduled for outpatient surgery (<24 hours inpatient stay) were screened for eligibility. We excluded patients who were actively taking opioid pain medications. Patients were informed of the study during their preoperative clinic visit and subsequently enrolled over the telephone using a standardized script after informed consent was obtained. Participants received financial compensation if they completed all surveys and the HRQoL questionnaire (eAppendix 1 in Supplement 2). This study was approved by the University of California, Los Angeles Institutional Review Board and followed Consolidated Standards of Reporting Trials (CONSORT) reporting guideline for randomized clinical trials.
A random number–generated randomization schedule in a 1:1 allocation ratio was created before the start of the study by a research team member (D.T.N.) uninvolved in patient interactions. Treatment assignments were concealed in sealed, numbered envelopes until enrollment. Patients randomized to the intervention group were enrolled in the Postoperative Opt-In Narcotic Treatment (POINT) program. The program entailed a 5-minute preoperative pain counseling session using a standardized script (eAppendix 2 in Supplement 2), a 2-minute video review of the pain counseling session shown after surgery and before discharge (eAppendix 2 in the Supplement 2), and a discharge brochure for pain management strategies. The counseling sessions discussed the risks and adverse effects associated with opioids but did not advocate for or against opioid treatment after surgery. Patients were reassured of the option to call in for a rescue opioid prescription.
Patients in the control group received no counseling. All patients were administered the Patient Health Questionnaire-2 (PHQ-2), which has high sensitivity for the diagnosis of major depression, given that symptoms of depression have been associated with increased postoperative pain and opioid use.15,16,17
Intraoperatively, all patients received a bilateral superficial cervical plexus block with 10 mL of bupivacaine, 0.25%, bilaterally and a 5- to 10-mL subcutaneous injection of bupivacaine, 0.25%, with epinephrine 1:200 000 at the incision site before incision. Inpatient perioperative pain treatment was left to the discretion of the anesthesia and surgical teams. All opioids administered perioperatively were converted to oral morphine equivalents (OMEs) to adjust for differences in potency. Patients were observed in the postanesthesia recovery unit for at least 4 hours before discharge or admitted for overnight observation based on patient factors such as age, comorbidities, and distance from hospital to home. Patient pain levels were assessed every 1 to 2 hours in the postanesthesia recovery unit and every 4 hours on the ward.
Before discharge, patients randomized to the control group were routinely prescribed narcotics, in keeping with usual care. Nursing staff directed POINT patients to view the 2-minute pain counseling video and sign a form attesting to whether they chose to receive a narcotic prescription. In both groups, 10 tablets of hydrocodone, 5 mg, with acetaminophen, 325 mg (50 OMEs), was the standard prescription dispensed. Patients who had a history of poor tolerance for hydrocodone with acetaminophen were prescribed 10 tablets of codeine, 30 mg, with acetaminophen, 300 mg (45 OMEs), as a first-line alternative or 10 tablets of tramadol, 50 mg (50 OMEs), as a second-line alternative.
On postoperative days 1 through 7, all participants were administered daily surveys (eAppendix 3 in Supplement 2) to assess pain levels through PRIME for Patients (2020 UCLA Health), an iOS operating system–compatible, Health Insurance Portability and Accountability Act–compliant mobile application. Patients without iOS devices were administered surveys via an online password-protected data management and survey platform (REDCap, version 10.0.33; Vanderbilt University) or by telephone. An electronic gift card was offered to patients at the end of the study to encourage survey completion. Patients were asked to rate their peak pain score using the 11-point numerical rating scale over the previous 24 hours, identify sites of pain, and list the type, dose, and number of tablets of pain medication (both prescription and over-the-counter agents) used. Logged pain scores greater than or equal to 8 triggered an automated alert, which prompted a physician-initiated telephone call to the patient. On postoperative day 7, patients were administered an assessment of HRQOL over the first postoperative week.
Study Outcomes
The primary outcomes studied were patient-reported pain scores in the first postoperative week. Secondary outcomes studied were the quantity of opioid tablets prescribed and consumed in each group and the HRQOL assessment, which consisted of both the Patient-Reported Outcomes Measurement Information System 29-item profile (PROMIS-29 v2.0) profile, and the Secure Flourish Index. The PROMIS-29 v2.0 profile consists of 7 domains (physical function, anxiety, depression, fatigue, sleep disturbance, social function, and pain interference) standardized using a T-score metric with the mean (SD) of the US general population equal to 50 (10).18 A higher score in each domain may represent either a positive or negative health-related outcome (eg, higher physical function, higher pain interference). The Secure Flourish Index is a brief, 12-item holistic assessment of psychological well-being.19,20 Each question is scored on a scale of 0 to 10, with higher scores indicating more favorable response, creating a composite index score of 0 to 120.
We also performed subgroup analyses to determine patient and perioperative risk factors associated with a greater likelihood of requesting and/or consuming opioids. We analyzed inpatient pain scores reported as the maximum score during hospitalization, 4 hours postoperatively, and the last recorded score before discharge (reflective of patients’ pain level when deciding whether to receive an opioid prescription in the POINT group).21
Statistical Analysis
Patient demographic and baseline characteristics were compared between the 2 groups. The sample size was calculated based on a noninferiority design, and we defined a noninferiority margin as a difference of 2 points on the 11-point numerical rating scale for pain. To achieve a statistical power of 90% at a 1-sided α level of .05 and an SD of 2 points, 18 patients were needed in each group. In our interim analysis, we observed that half of the POINT patients opted in for a prescription. We expanded our enrollment with the aim to enroll 50 patients in each study arm, such that a projected 25 patients would each opt in and opt out of opioids in the POINT group to sufficiently power analyses of differences in pain scores observed between subgroups, while allowing for a 15% dropout margin. Analysis was conducted on the evaluable population.
We used the Wilcoxon rank sum test or 2-sided, unpaired t test for comparison of continuous variables and Fisher exact test for categorical variables, with statistical significance defined as P < .05. Mean differences in pain scores between groups were calculated with 95% CIs, and regression models were used to adjust for any covariates. Noninferiority was established if the upper bound of the 95% CI was less than 2 points on the numerical rating scale of 0 to 10. Mean differences in PROMIS-29 T scores and the Secure Flourish Index level were also calculated with 95% CIs, adjusted for covariates. Because minimal clinically important difference for PROMIS-29 and Secure Flourish have not been validated in patients undergoing endocrine surgery, we used the Norman and colleagues’22 definition of minimal clinically important difference as 0.5 SD, using the SD of the control group. Noninferiority was established in the Secure Flourish Index and the PROMIS-29 physical and social function domains if the lower bound of the 95% CI was within the minimal clinically important difference. Noninferiority was established in the remaining PROMIS-29 domains if the upper bound of the 95% CI was within the minimal clinically important difference. A superiority analysis of the POINT patients who opted in or out of opioid prescriptions was performed using the Wilcoxon rank sum test to determine whether there were differences in pain scores or HRQOL. Multivariate logistic regression was used to analyze independent factors for opting in for a prescription and opioid consumption after identifying factors that were statistically significant in univariate analysis. All analyses were performed in R, version 1.4.1103 (R Foundation for Statistical Computing).
Results
A total of 115 patients were enrolled, with 58 patients (50.4%) randomized to receive routine narcotic prescription (control), and 57 patients (49.5%) randomized to POINT (Figure 1). After exclusions, 102 patients remained in the study, with 54 (52.9%) in the control arm and 48 (47.1%) in the intervention arm. Participants included 79 women (77.5%) and 23 men (22.5%); median age was 52 (IQR, 43-62) years. Baseline characteristics were similar between groups with the exception of median (interquartile range [IQR]) age (control, 55 [IQR, 48-65] vs POINT, 46 [IQR, 39-53] years), distribution of procedure type (parathyroidectomy: control, 29 [53.7%] vs POINT, 16 [33.3%]), and operative time (median, control, 70 [IQR, 57.3-88] vs POINT, 79 [IQR, 64.5-108] minutes) (Table 1). Survey completion of 5 or more daily surveys was 97.1% (99 of all 102 patients). Outcomes data from 3 patients (2 control patients and 1 POINT patient who opted out of opioids) who completed fewer than 5 surveys were excluded from analysis.
Figure 1. Study Recruitment Flowchart.
POINT indicates postoperative opt-in narcotic treatment.
Table 1. Demographic and Baseline Characteristics.
| Characteristic | No. (%) | P value, standardized difference | |
|---|---|---|---|
| Control (n = 54) | POINT (n = 48) | ||
| Age, median (IQR), y | 55 (48-65) | 46 (39-53) | .01 |
| Sex | |||
| Female | 42 (77.8) | 37 (77.1) | >.99 |
| Male | 12 (22.2) | 11 (22.9) | |
| BMI, median (IQR) | 26.6 (24.4-31.4) | 25.5 (22.1-30) | .09 |
| History of narcotic use | |||
| Never | 15 (27.8) | 16 (33.3) | .73 |
| Previously after surgery | 30 (55.6) | 26 (54.2) | |
| Previously for a limited perioda | 6 (11.1) | 6 (12.5) | |
| Previously for chronic pain | 2 (3.7) | 0 | |
| Not answered | 1 (1.9) | 0 | |
| PHQ-2 screen | |||
| Positive | 21 (38.9) | 14 (29.2) | .54 |
| Negative | 30 (55.6) | 30 (62.5) | |
| Not answered | 3 (5.6) | 4 (8.3) | |
| Preoperative cancer diagnosis | |||
| Yes | 15 (27.8) | 14 (29.2) | >.99 |
| No | 39 (72.2) | 34 (70.8) | |
| Procedure | |||
| Parathyroidectomy | 29 (53.7) | 16 (33.3) | .11 |
| Thyroid lobectomy | 15 (27.8) | 20 (41.7) | |
| Total thyroidectomy | 12 (22.2) | 12 (25.0) | |
| Operative time, median (IQR), min | 70 (57.3-88) | 79 (64.5-108) | .03 |
| Overnight stay | |||
| Yes | 20 (37.0) | 20 (41.7) | .69 |
| No | 34 (63.0) | 28 (58.3) | |
| Prescription | |||
| Hydrocodone, 5 mg, with acetaminophen, 325 mg | 45 (83.3) | 22 (95.7) | .83 |
| Codeine, 30 mg, with acetaminophen, 300 mg, #3 | 6 (11.1) | 1 (4.3) | |
| Tramadol, 50 mg | 2 (3.7) | 0 (0.0) | |
| Oxycodone, 5 mg | 1 (1.9) | 0 (0.0) | |
| Survey administration type | |||
| PRIME for Patients mobile app | 41 (75.9) | 33 (68.8) | .22 |
| 9 (16.7) | 14 (29.2) | ||
| Telephone | 4 (7.4) | 1 (2.1) | |
Abbreviations: BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); IQR, interquartile range; PHQ-2, Patient Health Questionnaire-2; POINT, postoperative opt-in narcotic treatment.
Defined as less than 90 consecutive days.
Outcomes
Patients in both groups received similar perioperative quantities of acetaminophen and opioid analgesics (Table 2). Few patients received additional nonopioid analgesics, such as gabapentin (n = 6) and ketorolac (n = 1). Postoperative pain scores before discharge were similar between groups; for example, the highest inpatient median score was 6 (IQR, 4-7) in the control group vs 6 (IQR, 3-7) in the POINT group.
Table 2. Perioperative Pain Characteristics, Pain Assessment, and Opioid Consumption.
| Characteristic | Control (n = 54) | POINT (n = 48) | P value |
|---|---|---|---|
| Perioperative pain characteristics and treatment | |||
| Perioperative pain treatment | |||
| Acetaminophen administered, No. (%) | 44 (81.5) | 42 (87.5) | .43 |
| Acetaminophen total dosage, median (IQR), mg | 1000 (1000-1319) | 1000 (1000-1650) | .20 |
| Total OMEs, median (IQR) | 54.8 (30.0-88.3) | 52.5 (37.9-77.8) | .61 |
| Inpatient postoperative pain scores, median (IQR) | |||
| 4-h Postoperative pain score | 0 (0-3) | 2 (0-4) | .11 |
| Highest postoperative pain score | 6 (4-7) | 6 (3-7) | .23 |
| Last recorded pain score before discharge | 0 (0-3) | 1.5 (0-4) | .17 |
| Patient self-reported pain assessment and opioid consumption | |||
| Patients with ≥5 surveys completed, No. (%) | 52 (96.3) | 47 (97.9) | |
| Opioid consumption | |||
| Tablets dispensed for the group, No. | 550 | 230 | <.001 |
| Tablets dispensed per person, mean (SD) | 10.1 (0.7) | 4.8 (5.1) | |
| Patients who took opioids, No. (%) | 27 (51.9) | 17 (36.2) | .16 |
| Tablets consumed for the group, No. (% of dispensed tablets) | 135.5 (24.6) | 66 (28.7) | |
| Tablets consumed per person, mean (SD) | 2.6 (3.8) | 1.4 (2.5) | .07 |
| Total OMEs consumed over the week, median (IQR) | 5 (0-20) | 0 (0-10) | .09 |
| Total OMEs consumed over the week for those taking opioids, median (IQR) | 20 (10-34) | 15 (10-30) | .49 |
| OTC use, No. (%) | |||
| Acetaminophen | 38 (73.1) | 39 (83.0) | .33 |
| Ibuprofen | 11 (21.2) | 18 (38.3) | .08 |
| Naproxen | 4 (7.7) | 3 (6.4) | >.99 |
| Postoperative pain scores, median (IQR) | |||
| Maximum reported pain score | 6 (4-8) | 6 (5-7) | .71 |
| Daily peak pain score averaged across the week | 3 (2-4) | 3 (2-4) | .85 |
| No. of patients with alert triggered for pain score ≥8, No. (%) | 17 (31.5) | 11 (22.9) | .27 |
| Total number of physician-initiated telephone calls for the group triggered by survey alert, No. | 28 | 14 | |
| No. of physician-initiated telephone calls per person, mean (SD) | 0.5 (0.9) | 0.3 (0.6) | .13 |
| Refill requests, No. | 1 | 0 | NA |
Abbreviations: IQR, interquartile range; NA, not applicable; OME, oral morphine equivalents; POINT, postoperative opt-in narcotic treatment.
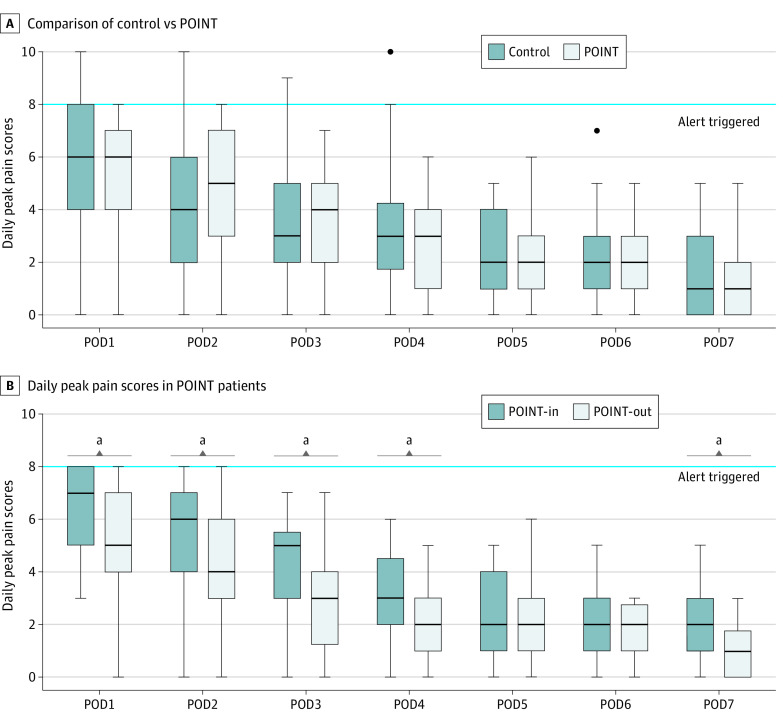
The highest patient-reported pain score in the first postoperative week was a median of 6 (IQR, 4-8) for the control group and 6 (IQR, 5-7) for the POINT group (P = .71). The upper boundary of the 95% CI was within the noninferiority margin of 2 points on the numerical rating scale for each day of the first postoperative week (eFigure in Supplement 2). The most common primary sources of pain were incisional pain (36.5%) and sore throat (35.8%), followed by shoulder pain (8.1%), headache (6.9%), and back pain (3.9%). The proportion of patients who triggered an automated alert by reporting a pain score of 8 or higher was also similar in the control vs POINT groups (32.7% vs 23.4%; P = .27) (Table 2). A total of 28 physician-initiated telephone calls resulted from the alerts in the control group (mean [SD], 0.5 [0.9] calls per person) and 14 in the POINT group (mean, 0.3 [0.6] calls per person; P = .13).
In a subgroup analysis of POINT patients, those who opted in for opioid medication on discharge (POINT-In) experienced significantly greater pain than those who opted out (POINT-Out) through postoperative day 3 (Figure 2; eTable 2 in Supplement 2). The highest reported pain score was a median of 7 (IQR, 6-8) in the POINT-In group vs a median of 5 (IQR, 4-7) in the POINT-Out group (P = .02).
Figure 2. Patient-Reported Daily Peak Pain Scores Using the 11-Point Numeric Rating Scale Through Postoperative Day (POD) 7.
A, Comparison of control and Postoperative Opt-In Narcotic Treatment (POINT) patients did not show any significant difference in pain scores. Few patients reported severe pain (score ≥8) in the first 4 postoperative days that triggered an automated survey alert at nearly twice the frequency in the control group as in the POINT group (28 alerts triggered for 54 control patients vs 14 alerts triggered for 48 POINT patients). B, Subgroup analysis of patient-reported daily peak pain scores in POINT patients. Pain scores were significantly higher in the patients who opted in (POINT-In) for a prescription compared with those who opted out (POINT-Out). A clinically significant difference of 2 points on the numeric rating scale was observed on POD1 to POD3. Boxes indicate the 25th percentile, median, and 75th percentile. Dots represent outliers. Whiskers show the minimum and maximum ranges.
aSignificant at P < .05.
The control group was prescribed 550 opioid tablets, amounting to a total of 2690 OMEs. Of patients completing 5 or more daily surveys, 27 control patients (50.0%) reported using opioids, collectively consuming a total of 135.5 tablets (24.6% of tablets dispensed). The POINT group was prescribed a total 230 tablets to 23 patients (47.9%) who opted in for a prescription. Of those, 17 POINT patients (73.9% of POINT-In, 35.4% of POINT group) reported consuming a total of 66 tablets (28.7% of tablets dispensed). Only 10% of all patients required more than 6 tablets. Of patients who used opioids, the median OMEs consumed over the week was similar between groups (control, 20 [IQR, 10-34] vs POINT, 15 [IQR, 10-30]; P = .49). The groups did not differ significantly in the use of over-the-counter pain medication. No POINT-Out patients required a rescue prescription.
The HRQOL scores at postoperative day 7 in the POINT group were noninferior to scores in the control group in the Secure Flourish Index and all PROMIS-29 domains, with the exception of the anxiety domain (eTable 1 in Supplement 2); however, anxiety T scores were not significantly different between groups (median T score, 51.4 [IQR, 40.3-57.7] vs 53.8 [IQR, 44.2-58.6]; P = .26). Compared with POINT-Out patients, POINT-In patients had significantly more anxiety (median T score, 56.2 [IQR, 54-63.2] vs 49.8 [IQR, 40.3-53.8]; P = .001) and pain interference (median T score of 55.7 [IQR, 55.7-61.5] vs 54.6 [IQR, 41.6-57]; P = .01), but the latter did not exceed the threshold for minimal clinically important difference.
Factors Associated With Opoid Consumption
We performed an analysis of all study patients to determine factors associated with opioid consumption (Table 3). In univariate analysis of opioids consumed vs not consumed, body mass index (median [IQR], 28.8 [24.6-31.5] vs 25.0 [21.7-29.5]; P = .003) (calculated as weight in kilograms divided by height in meters squared), history of narcotic use (38 [86.4%] vs 30 [54.5%]; P = .001), surgical procedure (eg, total thyroidectomy: 15 [34.1%] vs 9 [16.4%]; P = .04), incision length (median, 4 [range, 3-4] vs 3 [range, 2.5-5] cm; P = .02), and higher inpatient postoperative pain scores (median [IQR], 7 [6-8] vs 4 [2-6]; P < .001) were factors associated with opioid consumption. We included only peak pain scores in the multivariate regression model, as opposed to median or last recorded pain score, because the peak scores had the strongest association with opioid use. In multivariate analysis, higher body mass index (odds ratio, 1.11; 95% CI, 1.01-1.23; P = .03), highest postoperative pain score (odds ratio, 1.24; 95% CI, 1.04-1.52; P = .02), and history of narcotic use (odds ratio, 4.80; 95% CI, 1.51-18.45; P = .01) were identified as independent factors associated with consuming opioids.
Table 3. Univariate and Multivariate Analyses of Factors Associated With Opioid Consumption Among All Patients.
| Variable | Opioids consumed (n = 44) | No opioids consumed (n = 55) | Univariate P value | Multivariate OR (95% CI) | Multivariate P value |
|---|---|---|---|---|---|
| Age, median (IQR), y | 49 (39-63) | 46 (41-61) | .19 | NA | NA |
| BMI, median (IQR) | 28.8 (24.6-31.5) | 25.0 (21.7-29.5) | .003 | 1.11 (1.01-1.23) | .03 |
| Male sex, No. (%) | 9 (20.5) | 11 (20.0) | .96 | NA | NA |
| History of narcotic use, No. (%) | 38 (86.4) | 30 (54.5) | .001 | 4.80 (1.51-18.45) | .01 |
| Positive PHQ-2, No. (%) | 16 (36.4) | 18 (32.7) | .39 | NA | NA |
| Preoperative cancer diagnosis, No. (%) | 14 (31.8) | 14 (25.5) | .49 | NA | NA |
| Surgical procedure, No. (%) | |||||
| Parathyroidectomya | 15 (34.1) | 27 (49.1) | NA | NA | NA |
| Thyroid lobectomy | 14 (31.8) | 19 (34.5) | .55 | 0.65 (0.15-4.14) | .55 |
| Total thyroidectomy | 15 (34.1) | 9 (16.4) | .04 | 1.46 (0.28-7.57) | .65 |
| Operative time, median (IQR), min | 77 (59.8-98) | 73 (57.5-93) | .20 | NA | NA |
| Incision length, cm | 4 (3-4) | 3 (2.5-4) | .02 | 1.83 (0.84-4.14) | .13 |
| Overnight stay, No. (%) | 19 (43.2) | 21 (38.2) | .62 | NA | NA |
| Inpatient OME given, median (IQR) | 68 (49.3-91.1) | 45 (30-60) | .02 | NA | NA |
| Inpatient postoperative pain score, median (IQR) | |||||
| 4-h Postoperative pain | 3 (0-4) | 0 (0-3) | .07 | NA | NA |
| Last recorded pain | 2 (0-4) | 0 (0-2) | .01 | NA | NA |
| Highest postoperative pain | 7 (6-8) | 4 (2-6) | <.001 | 1.24 (1.04-1.52) | .02 |
Abbreviations: BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); IQR, interquartile range; NA, not applicable; OME, oral morphine equivalents; OR, odds ratio; PHQ-2, Patient Health Questionnaire-2.
Reference variable.
We also performed a subgroup analysis of POINT patients to identify factors associated with opting in for postoperative opioids (eTable 3 in Supplement 2). In multivariate analysis, every 1-point increase in last recorded pain score was associated with a 1.7-fold increase in the likelihood of requesting an opioid prescription (95% CI, 1.15-2.68; P = .01). Patients who previously used opioids were 7.5 times more likely than opioid-naive patients to opt in compared with opioid-naive patients (95% CI, 1.61-50.11; P = .02).
Discussion
We found that patients randomized to an opt-in strategy for postoperative narcotic treatment following cervical endocrine surgery were prescribed fewer narcotics than those randomized to receive routine narcotic prescription without experiencing increased pain levels. Randomization to the POINT program also did not have a negative effect on HRQOL. The cumulative amount of opioids consumed by the control group was twice that consumed by the POINT group, which is consistent with other studies that found an association between prescription availability and higher opioid consumption.23,24 No patients required a rescue opioid prescription if they opted out at the time of discharge, which indicates that the patients were able to predict their own postoperative opioid requirements. Despite having access to opioids, POINT-In patients had higher postoperative pain levels and worse HRQOL in the anxiety and pain assessments than POINT-Out patients. These findings suggest the subjective perception of pain may be more influenced by patient- and surgery-specific factors; a certain level of postoperative discomfort appears unavoidable and is unaffected by additional opioids.
Other contemporary studies have reported that many patients do not require opioids following cervical endocrine operations; when prescribed opioids after endocrine operations, 34% to 52% of patients do not take opioid medication.4,25 In our study, 53% of POINT patients chose not to receive a prescription for opioids, and there were no calls for rescue prescriptions. We routinely perform preincision bilateral cervical plexus block, which may lower postoperative opioid requirement.26,27 Our findings support the opt-in strategy as a means of reducing overprescription of opioids and also indicate that patients who choose to forgo opioids are not experiencing increased pain at home but not reporting it.
The proportion of POINT patients who chose to receive an opioid prescription was 5- to 10-fold greater than in prior studies of an opt-in strategy for opioid prescriptions.14,28 The larger number of opioid requests may reflect the equipoise we purposefully maintained during patient counseling. Through a patient-centered approach to decision-making, we encouraged participants to carefully consider the risks and benefits of using opioids but did not discourage prescription requests. In addition, patients randomized to POINT understood that those in the control group were receiving opioids as part of our previous standard practice, in contrast to Ruffolo et al14 and Kwan et al,28 who presented an opt-in strategy as the new standard of care. Most patients in both the control and POINT groups had used opioids after surgery previously, which normalized narcotic treatment as part of routine postoperative care.
Previous narcotic use or substance abuse, depression or anxiety, and postoperative pain scores have been identified as factors associated with opioid consumption.14,29,30,31 We screened patients with the PHQ-2, which has been validated with similar or higher sensitivity compared with the PHQ-9 to identify patients with major depression,15 but PHQ-2 positivity was not predictive of either opting in or opioid consumption. Our factors for opioid consumption were higher body mass index, previous narcotic use, and higher inpatient postoperative pain scores. Body mass index as an independent predictor of opioid consumption may reflect greater positioning-related discomfort and/or pain from more forceful retraction during surgery. Previous narcotic use was the strongest predictor for both opting in and consuming opioids. Most prior opioid experience was in the context of postprocedural treatment and not chronic use. These findings have 2 main implications: every effort should be made to avoid exposing opioid-naive patients to opioids, because opioid naivety predicts against future use, and patients with higher immediate postoperative pain levels and previous opioid exposure may benefit from additional nonopioid adjuncts for postoperative pain control to minimize opioid consumption. Patients reported postoperative pain as frequently from sore throat as incisional pain, suggesting that efforts to minimize postintubation discomfort may lessen opioid consumption. Regardless of individual risk factors, patients can reliably determine their own need for opioids after surgery, demonstrated by the 76% of POINT patients who opted in to receive opioids and subsequently used the medication. Despite this level, only 29% of total dispensed tablets in the POINT-In group were consumed, which suggests reducing the standard prescription quantity would adequately treat most patients requesting a narcotic prescription after cervical endocrine surgery.
Limitations
Our study has limitations. First, this trial was performed at a single institution, which limits the generalizability of our findings to dissimilar practice settings, operative techniques, and perioperative pain management practices. Second, we used self-reported data on opioids consumed, which are less reliable than pill counts. Third, we did not administer a preoperative HRQOL assessment and assumed patients in both groups shared a similar baseline quality of life.
Conclusions
In this randomized clinical trial, preoperative counseling combined with a patient-choice strategy for prescribing opioid analgesics reduced the number of opioids prescribed without worsening postoperative pain or HRQOL. Patients should be able to choose whether to include opioids in their pain management regimen after cervical endocrine surgery to prevent undertreating those with a postoperative opioid requirement but reduce the risk of opioid exposure in those who require minimal analgesics. Most patients who request postoperative opioids do not require more than 30 OMEs.
Trial Protocol
eFigure 1. Noninferiority Analysis of Patient-Reported Pain Scores Using the Numeric Rating Scale
eTable 1. Noninferiority Analysis for Postoperative Day 7 Health-Related Quality of Life Assessment
eTable 2. Subgroup Superiority Analysis of daily Patient-Reported Postoperative Pain Scores and Health-Related Quality of Life Assessment in POINT Patients
eTable 3. Univariate and Multivariate Analyses of Factors Associated With Opting in for Postoperative Opioids Amongst POINT Patients
eAppendix 1. Phone Enrollment and Consent Script
eAppendix 2. POINT Program Discharge Video Script
eAppendix 3. Daily Postoperative Survey Questions
Data Sharing Statement
References
- 1.Wetzel M, Hockenberry J, Raval MV. Interventions for postsurgical opioid prescribing: a systematic review. JAMA Surg. 2018;153(10):948-954. doi: 10.1001/jamasurg.2018.2730 [DOI] [PubMed] [Google Scholar]
- 2.American College of Surgeons . Statement on the opioid abuse epidemic. August 2, 2017. Accessed February 18, 2021. https://www.facs.org/About-ACS/Statements/100-opioid-abuse
- 3.Habermann EB. Are opioids overprescribed following elective surgery? Adv Surg. 2018;52(1):247-256. doi: 10.1016/j.yasu.2018.03.003 [DOI] [PubMed] [Google Scholar]
- 4.Lancaster E, Inglis-Arkell C, Hirose K, et al. Variability in opioid-prescribing patterns in endocrine surgery and discordance with patient use. JAMA Surg. 2019;154(11):1069-1070. doi: 10.1001/jamasurg.2019.2518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shindo M, Lim J, Leon E, Moneta L, Li R, Quintanilla-Dieck L. Opioid prescribing practice and needs in thyroid and parathyroid surgery. JAMA Otolaryngol Head Neck Surg. 2018;144(12):1098-1103. doi: 10.1001/jamaoto.2018.2427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lou I, Chennell TB, Schaefer SC, et al. Optimizing outpatient pain management after thyroid and parathyroid surgery: a two-institution experience. Ann Surg Oncol. 2017;24(7):1951-1957. doi: 10.1245/s10434-017-5781-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McCrary HC, Newberry CI, Casazza GC, Cannon RB, Ramirez AL, Meier JD. Evaluation of opioid prescription patterns among patients undergoing thyroid surgery. Head Neck. 2021;43(3):903-908. doi: 10.1002/hed.26551 [DOI] [PubMed] [Google Scholar]
- 8.Lipari HA. How People Obtain the Prescription Pain Relievers They Misuse: The CBHSQ Report. Center for Behavioral Health Statistics and Quality, Substances and Metal Health Services Administration; 2013. [PubMed] [Google Scholar]
- 9.Alam A, Gomes T, Zheng H, Mamdani MM, Juurlink DN, Bell CM. Long-term analgesic use after low-risk surgery: a retrospective cohort study. Arch Intern Med. 2012;172(5):425-430. doi: 10.1001/archinternmed.2011.1827 [DOI] [PubMed] [Google Scholar]
- 10.Marcusa DP, Mann RA, Cron DC, et al. Prescription opioid use among opioid-naive women undergoing immediate breast reconstruction. Plast Reconstr Surg. 2017;140(6):1081-1090. doi: 10.1097/PRS.0000000000003832 [DOI] [PubMed] [Google Scholar]
- 11.Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152(6):e170504-e170504. doi: 10.1001/jamasurg.2017.0504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sun EC, Darnall BD, Baker LC, Mackey S. Incidence of and risk factors for chronic opioid use among opioid-naive patients in the postoperative period. JAMA Intern Med. 2016;176(9):1286-1293. doi: 10.1001/jamainternmed.2016.3298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Olds C, Spataro E, Li K, Kandathil C, Most SP. Assessment of persistent and prolonged postoperative opioid use among patients undergoing plastic and reconstructive surgery. JAMA Facial Plast Surg. 2019;21(4):286-291. doi: 10.1001/jamafacial.2018.2035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ruffolo LI, Jackson KM, Juviler P, et al. Narcotic free cervical endocrine surgery: a shift in paradigm. Ann Surg. 2021;274(2):e143-e149. [DOI] [PubMed] [Google Scholar]
- 15.Arroll B, Goodyear-Smith F, Crengle S, et al. Validation of PHQ-2 and PHQ-9 to screen for major depression in the primary care population. Ann Fam Med. 2010;8(4):348-353. doi: 10.1370/afm.1139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Larach DB, Sahara MJ, As-Sanie S, et al. Patient factors associated with opioid consumption in the month following major surgery. Ann Surg. 2021;273(3):507-515. doi: 10.1097/SLA.0000000000003509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ip HY, Abrishami A, Peng PW, Wong J, Chung F. Predictors of postoperative pain and analgesic consumption: a qualitative systematic review. Anesthesiology. 2009;111(3):657-677. doi: 10.1097/ALN.0b013e3181aae87a [DOI] [PubMed] [Google Scholar]
- 18.Cella D, Riley W, Stone A, et al. ; PROMIS Cooperative Group . The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008. J Clin Epidemiol. 2010;63(11):1179-1194. doi: 10.1016/j.jclinepi.2010.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.VanderWeele TJ, McNeely E, Koh HK. Reimagining health-flourishing. JAMA. 2019;321(17):1667-1668. doi: 10.1001/jama.2019.3035 [DOI] [PubMed] [Google Scholar]
- 20.Diener E, Wirtz D, Tov W, et al. New well-being measures: short scales to assess flourishing and positive and negative feelings. Social Indicators Research. 2010;97(2):143-156. doi: 10.1007/s11205-009-9493-y [DOI] [Google Scholar]
- 21.Kahneman D. Thinking, Fast and Slow. Farrar, Straus and Giroux; 2011. [Google Scholar]
- 22.Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41(5):582-592. doi: 10.1097/01.MLR.0000062554.74615.4C [DOI] [PubMed] [Google Scholar]
- 23.Howard R, Fry B, Gunaseelan V, et al. Association of opioid prescribing with opioid consumption after surgery in Michigan. JAMA Surg. 2019;154(1):e184234-e184234. doi: 10.1001/jamasurg.2018.4234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Harbaugh CM, Lee JS, Chua K-P, et al. Association between long-term opioid use in family members and persistent opioid use after surgery among adolescents and young adults. JAMA Surg. 2019;154(4):e185838-e185838. doi: 10.1001/jamasurg.2018.5838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen Y, Nwaogu I, Chomsky-Higgins K, et al. Postoperative pain and opioid use after thyroid and parathyroid surgery—a pilot, prospective SMS-based survey. J Surg Res. 2019;240:236-240. doi: 10.1016/j.jss.2019.03.016 [DOI] [PubMed] [Google Scholar]
- 26.Cai H-D, Lin C-Z, Yu C-X, Lin X-Z. Bilateral superficial cervical plexus block reduces postoperative nausea and vomiting and early postoperative pain after thyroidectomy. J Int Med Res. 2012;40(4):1390-1398. doi: 10.1177/147323001204000417 [DOI] [PubMed] [Google Scholar]
- 27.Karthikeyan VS, Sistla SC, Badhe AS, et al. Randomized controlled trial on the efficacy of bilateral superficial cervical plexus block in thyroidectomy. Pain Pract. 2013;13(7):539-546. doi: 10.1111/papr.12022 [DOI] [PubMed] [Google Scholar]
- 28.Kwan SY, Lancaster E, Dixit A, et al. Reducing opioid use in endocrine surgery through patient education and provider prescribing patterns. J Surg Res. 2020;256:303-310. doi: 10.1016/j.jss.2020.06.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Oyler DR, Randle RW, Lee CY, Jenkins G, Chang PK, Sloan DA. Implementation of opioid-free thyroid and parathyroid procedures: a single center experience. J Surg Res. 2020;252:169-173. doi: 10.1016/j.jss.2020.03.010 [DOI] [PubMed] [Google Scholar]
- 30.Rhon DI, Snodgrass SJ, Cleland JA, Sissel CD, Cook CE. Predictors of chronic prescription opioid use after orthopedic surgery: derivation of a clinical prediction rule. Perioper Med (Lond). 2018;7(1):25. doi: 10.1186/s13741-018-0105-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.O’Connell C, Azad TD, Mittal V, et al. Preoperative depression, lumbar fusion, and opioid use: an assessment of postoperative prescription, quality, and economic outcomes. Neurosurg Focus. 2018;44(1):E5. doi: 10.3171/2017.10.FOCUS17563 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Trial Protocol
eFigure 1. Noninferiority Analysis of Patient-Reported Pain Scores Using the Numeric Rating Scale
eTable 1. Noninferiority Analysis for Postoperative Day 7 Health-Related Quality of Life Assessment
eTable 2. Subgroup Superiority Analysis of daily Patient-Reported Postoperative Pain Scores and Health-Related Quality of Life Assessment in POINT Patients
eTable 3. Univariate and Multivariate Analyses of Factors Associated With Opting in for Postoperative Opioids Amongst POINT Patients
eAppendix 1. Phone Enrollment and Consent Script
eAppendix 2. POINT Program Discharge Video Script
eAppendix 3. Daily Postoperative Survey Questions
Data Sharing Statement