Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative pathogen of the coronavirus disease 2019 (COVID-19), has caused more than 179 million infections and 3.8 million deaths worldwide. Global health authorities working on the COVID-19 outbreak continue to explore methods to reduce the rate of its transmission to healthy individuals. Treatment protocols thus far have focused on social distancing and masking, treatment with antivirals early in infection, and steroids to reduce the inflammatory response. An alternative approach is therapy with low dose radiation (LDR), which has several advantages compared to the current drugs and medicines. To date more than 10 case reports and pilot clinical trial preliminary outcome are available from different countries. These reports cover a wide range of patient conditions and LDR treatment strategies. Although one report showed the failure to observe the improvement of COVID-19 patients after LDR therapy, the majority showed some clinical improvement, and demonstrated the safety of LDR for COVID-19 patients, particularly with 0.5 Gy. This review aims to summarize the potential rationales and mechanisms of LDR therapy for COVID-19 patients, and its current clinical status and potential use.
Keywords: Low dose radiation, Radiation hormesis, Radiotherapy, COVID-19, SARS-CoV-2
1. Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative pathogen of the coronavirus disease 2019 (COVID-19), has caused more than 179 million infections and 3.8 million deaths worldwide.1 Global health authorities working on the COVID-19 outbreak continue to explore methods to reduce the rate of its transmission to healthy individuals. Viruses typically have different sensitivities to physical and chemical treatments, such antiviral drugs, UV, and heat, ranging from extremely sensitive to extremely resistant. Incomplete treatment of individuals provides evolutionary pressure on the viral genome, leading to the generation of more resistant organisms. An alternative approach is therapy with low dose radiation (LDR). As suggested in by Ghadimi-Moghadam et al.,2 COVID-19 patients may receive a single dose of 100, 180 or 250 mGy X-rays, less than the maximum annual radiation dose of the residents of high background radiation areas of Ramsar (<260 mSv). In contrast with antiviral drugs, a single dose of either 100, 180 or 250 mSv of low LET X-rays may not exert a significant selective pressure on the SARS-CoV-2 and hence does not lead to directed accelerated evolution of these viruses.
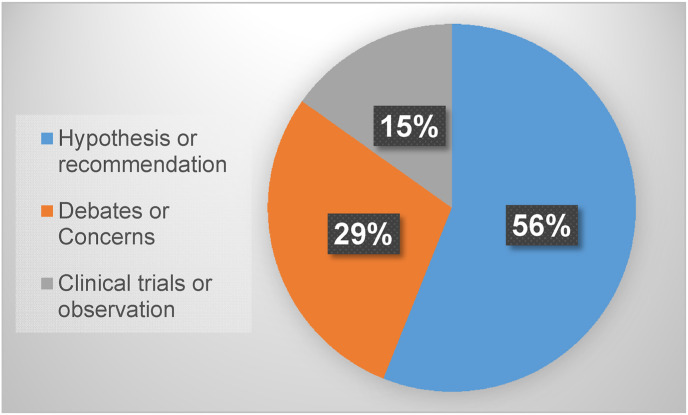
The concept of LDRT for COVID has been evaluated by several groups - a PubMed search with key words “low dose radiation therapy” and “COVID-19” on August 16, 2021 resulted in 66 publications related to the potential therapy for the patients with COVID-19. As outlined in Fig. 1 , 56% of these publications proposed various rationales and potential mechanisms for LDRT impacting COVID-19 patients, 29% belong to debates and concerns for the potential application of LDR therapy (LDRT) for COVID-19 patients, and only 15% are clinical observations or trials. This review aims to summarize the potential rationales or mechanisms, currently status of clinical outcome, and future directions.
Fig. 1.
Analysis of PubMed publications related to LDRT for the patients with COVID-19, based on search results on August 16, 2021.
2. LDR induced hormesis and adaptive response, and its application to treat phenomena historically
In the context of radiation protection and many fields of radiobiology, ‘low dose’ is understood to be 100 mGy or less. However, in the field of radiation oncology where the daily dose fraction is typically 2 Gy, and 6 weeks of therapy amounts to a total dose of 60 Gy (2 Gy/d × 5 d/week × 6 weeks), a single 1.0 Gy is regarded as a low dose.3 LDR has distinct effects from high dose radiation (HDR): the latter causes massive DNA damage, cell death, organ damage, and can even cause acute radiation sickness. In contrast, LDR, particularly at the 100 mGy or less, does not cause the above effect. LDR does cause some DNA damage, as well as stimulate DNA repair enzyme activity, anti-stress proteins and apoptotic cell death pathways, in an effort to reduce genomic instability4 , 5 as observed non-ionizing radiation genotoxic agent.6 This has led to two described impacts of LDR: “hormesis”, which is stimulating cell function and cellular signaling pathways, and “adaptive response” which is the development of resistance to subsequent chemical or physical stress.3, 4, 5
In the last seven decades, LDR hormetic and adaptive effects have been confirmed in a variety of animal (preclinical) models,7, 8, 9 and have also been gradually applied in clinics.10 For instance, a pilot trial of LDR via diagnostic Computed Tomography (CT, 40–60 mGy) to treat patient with Alzheimer's disease was been started in 2016,11, 12, 13 which has led to a NIH trial being approved for LDR therapy for patient with Alzheimer's disease (ClinicalTrials.gov Identifier: NCT03597360 for CT).
Dr. Calabrese has offered a nice review regarding the historical application of X-ray therapy of pneumonia during the first half of the 20th century. He has collected 15 studies with approximately 700 cases of bacterial (lobar and bronchopneumonia), sulfanilamide non-responsive, interstitial, and atypical pneumonia, showing the effective treatment by low dose (LD) X-rays, based on clinical symptoms, objective disease biomarkers, and mortality incidence. He found the capacity of the X-ray treatment to reduce mortality was similar to serum therapy and sulfonamide treatment during that time period. In his review, he also provided four experimental animal models (mice, guinea pig, cat, and dog) with bacterial and viral pneumonia to confirm the clinical observation and outcomes. X-ray treatment of these pneumonia is hypothesized to induce an anti-inflammatory phenotype to rapidly reverse clinical symptoms and facilitate disease resolution. This experience with both bacterial and viral pneumonias in the past suggests the possibility that LDRT could have an impact on COVID-19 as well.14
3. LDRT for the patients with COVID-19: its potential rationales and mechanisms
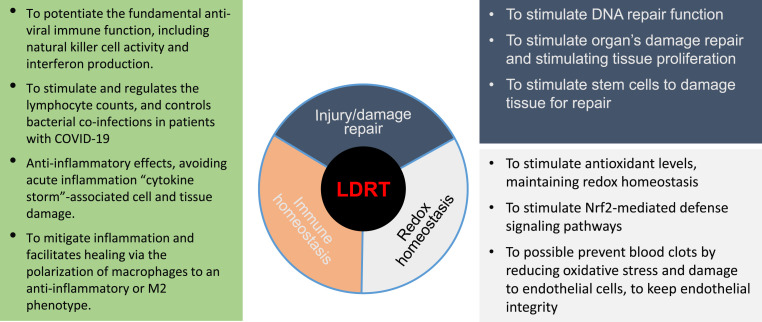
SARS-CoV-2 infects the lower respiratory system resulting in the recruitment of macrophages and monocytes to the lung as inflammation that further causes widespread damage to the lung's airways.1 , 15 Similar to other beta-corona viruses, SARS-CoV-2 requires the angiotensin-converting enzyme 2 (ACE2) as a receptor for entering the cells. ACE2 receptor expresses most cells in our body, including vascular endothelial cells, the renal tubular epithelium, Leydig cells in the testes, gastrointestinal tract, lung, and heart. However, SARS-CoV-2 most commonly affects the lungs of the patients with COVID-19, 20% of which becomes severe and results in ARDS or sepsis due to a cytokine storm. It is well-known that there remains insufficient therapy for the patients with COVID-19, making it highly lethal. Even though vaccinations are available from several countries, this prevalence and mortality remain high and new variants of SARS-CoV-2 are appearing rapidly, all these will continually spread it and threaten the world.1 , 15 Thus, alternative efficient approaches to prevent and treat the patients with COVID-19 remain urgently needed, among which LDRT was one proposed with several reasons, as illustrated in Fig. 2 .
Fig. 2.
Potential rationales and mechanisms responsible for the application of low-dose radiation therapy (LDRT) for COVID-19 patients.
As early as March 20, 2020, a preprint from Mohan Doss suggested application of LDRT for the patients with COVID-19 (https://osf.io/6zy9u/),16 following which, other two hypotheses immediately appeared on April 1, 2020 from Ghadimi-Moghadam et al.,2 and on April 6 as the early online version from Kirkby & Mackenzie.17 Doss remarked that the SARS-CoV-2 infection can lead to pneumonia and death, for which the weakness of the immune system would be the major contributing factors; therefore, it is necessary for some pilot clinical trials to explore LDRT with dose of 300 mGy for the lungs to boot systemic immune function and anti-inflammation.16 Ghadimi-Moghadam et al.2 also introduced the similar propose for COVID-19 patients to receive a single dose of 100, 180 or 250 mSv X-ray radiation since that is less than the maximum annual radiation dose of the residents of high background radiation areas of Ramsar that is up to 260 mSv.
Subsequently there were several publications suggesting the application of LDRT for the patients with COVID-19 with different reasons and historical evidence. Fig. 2 summarized the potential rationales or mechanisms responsible for the LDRT for these patients.
3.1. LDR and its anti-inflammation and immuno-stimulation
One goal of the therapy of COVID-19 patients is to stimulate the patient's immune function to kill the virus, but suppress the cytokine storm that leads to severe pulmonary injury.18 Although multiple drugs are being tested to treat COVID-19 patients, there are no drugs that have been approved by FDA to successfully treat severe cases of COVID-19. LDR has historically demonstrated robust effectiveness on viral pneumonia.14 Immunologically LDRT is able to induce IL-10 and TGF-β to enhance the immuno-suppressive effects and is also able to increase CD4 and CD8 T-cell counts. Hypothetically LDRT appears to negate the effects of cytokine release syndrome through immunomodulation.
The cytokine storm associated with severe COVID 19 patients has been considered as the cause of death in these sick patients.18 , 19 Based on Kumar et al.,18 Table 1 lists the typical changes of cytokines in the patients with COVID-19, including decreases in CD3, CD4, CD8, NK cells and B-cells, decrease in IL-10 and IFN-γ, but increases in IL-4, IL-6 and TNF-α. In contrast, LDR-induced responses include an increase in CD3, CD4, and CD8 T cells, increases in IL-10 and TGF-β, decreases in IL-6 and TNF-α, and M2 macrophage phenotype activation.18 Genard et al.20 also summarized literature to point out that LDR at <1.0 Gy maintaining high level of M2 microphage compared to radiation at medium dose (2–10 Gy) showing predominant M1 macrophage.
Table 1.
A few examples of immune cells and cytokines in COVID-19 patients & LDR-irradiated mice.
| Measurements | COVID-19 | Responses to LDR |
|---|---|---|
| T Cells (Cd3, CD4, CD8, NK) | Decreased | Increased |
| IL-6, TNF-α | Increased | Decreased |
| IFN-γ | Increased | Decreased |
| Types of increased microphages | M1 | M218,20 |
| MnSOD level or activity | Decreased28 | Increased34,35 |
Note: Information cited in this table predominantly from the review by Kumer et al.,18 otherwise was indicated in the table. M1 & M2 are two types of macrophages: M1 macrophages are recognized as classically activated macrophages and show enhanced ability to phagocyte pathogens while M2 macrophages are requested in infection-free healing circumstances and have pro-tumoral functions. The polarization of macrophages can be driven by different micro-environmental molecules and leads to the production by the macrophages of different cytokines and chemokines.20
Consistent with the report of Kumar et al.,18 the stimulating effect of LDR in animal models has been reported before,21 , 22 which demonstrated that LDR can stimulate proliferation of normal cells and activate their defense systems. The preclinical and clinical studies of the specific effects of LDR on the immune system indicate potential efficacy in the treatment of some immune-related diseases, especially autoimmune diseases and malignant tumors.22 Most recently, Meziani et al.23 also conducted a comprehensive study in mouse model and human tissue samples. In this study, the authors used airways-instilled lipopolysaccharide (LPS) and influenza virus (H1N1) as murine models of pneumonia, and Toll-like receptor (TLR)-3 stimulation in human lung macrophages. LDRT (0.5 or 1 Gy) decreased LPS induced pneumonia, and increased the percentage of nerve- and airway-associated Macrophages producing IL-10. LDR treatment decreased H1N1 viral infection-induced lung tissue damage and immune cell infiltration in animals and LDRT also increased IL-10 production by infiltrating immune cells into the lung. Exposure of TLR-3 ligand-stimulated human lung macrophages ex vivo to LDR increased IL-10 secretion and decreased IFNγ and IL-6 production. Therefore, these results also support the concept that LDR-mediated immune homeostasis with reduction of inflammatory response is most likely the mechanisms by which LDRT afford the therapy for the patients with COVID-19.23
3.2. LDR induced adaptive or hormetic responses showing the resistance of multiple organs to virus-infection caused damages
Except for the above anti-inflammatory and immuno-stimulatory effects, LDRT may also induce systemic tissues' adaptive or hormetic response, making the tissues resistant to the challenge by virus. Oxidative stress is actively involved in the pathogenic process of respiratory viral infections, therefore, preventing the cytokine storm that rapidly generates extra amounts of reactive oxygen species would be an important step to prevent multi-organ damage.1 , 15 Antioxidant therapy has been proven to most probably increase the effectiveness of the response against SARS-CoV-2.24 Recent studies have clearly shown the upregulation of oxidative stress gene markers during SARS-COV-2 viral infection and is under consideration as a predictor for COVID-19 severity and the duration of ICU stay.25 , 26 Specifically, the COVID-19-associated oxidative damage to red blood cells has been reported in 9-year-old boy with COVID-19 symptom and positive for SARS-CoV-2 RNA.27 In samples from the lung, heart, and liver in a recent prospective cohort of 15 postmortem patients who died from COVID-19, manganese superoxide dismutase (MnSOD) was shown in both mitochondria and cytosol, suggesting that this enzyme may not sufficiently detoxify the reactive oxygen or nitrogen species in mitochondria, leading to an increased oxidation of mitochondrial proteins.28 The NRF2 antioxidant gene expression pathway is suppressed in biopsies obtained from COVID-19 patients29; leading to the hypothesis that activation of NRF2 has potential for treating COVID-19.30
Calabrese et al.31 recently have addressed NRF2 activation as the responsible etiology for the clinical benefits of LDRT in COVID-19 pneumonia. In our early studies we have provided evidence for the induction of NRF232 , 33 and its down-stream antioxidants, including SOD,34 in various tissues of animals by LDR, which prevented diabetes-induced organ's damages. LDR-induced NRF2 and its downstream antioxidants not only offers the prevention of oxidative stress and damage caused by SARS-CoV2 infections, but also favorably rebalances the skewed oxidative redox homeostasis of immunological cells, driving them toward anti-inflammatory phenotypes.31 At the cellular and subcellular levels, hormetic doses of LDR (<1.0 Gy) induce polarization shifts in the predominant population of lung macrophages, from an M1 pro-inflammatory to an M2 anti-inflammatory phenotype.20 , 31 Intracellularly in response to LDR at 0.1 Gy, cytosolic CDK4 relocates to mitochondria where it directly phosphorylates MnSOD at serine-106 (S106) to activate MnSOD enzymatic activity and mitochondrial respiration.35 This LDR-mediated mitochondrial MnSOD activation and respiration make the cells more resistant to second high-dose radiation-induced damage in vitro and in vivo. 35 Therefore, LDRT may protect patients with COVID-19 from SARS-CoV-2-induced cytotoxicity not only by activating NRF2-mediated transcriptional function for increasing its downstream antioxidants, but also by enhancing antioxidant activities through stimulating posttranscriptional modification of these anti-oxidative enzymes. Together, the NRF2-mediated antioxidant responses and the subsequent shifts to anti-inflammatory phenotypes would have the capacity to suppress cytokine storms, resolve inflammation, promote tissue repair, and prevent COVID-19-related mortality.31
3.3. Stimulating stem cell proliferation to repair the damage organs
Infection of cells with SARS-CoV2 leading to the host cell damage and death.1 , 15 The repairing of these damaged or lost cells is an important step to recover from the infection-mediated ARDS; accordingly, therapeutic strategies with different kinds of stem cells have been explored.1 , 36 , 37 The capacity for LDR at 75 mGy to 0.5 Gy to stimulate bone marrow progenitor cells has been demonstrated in our early works,38 , 39 and its therapeutic effects on damaged tissues also were reported from our group,40 and others.41 , 42
Mesenchymal stromal/stem cells (MSCs) possess potent and broad-ranging immunomodulatory activities.43 As summarized by Rogers et al.,43 MSCs function via several mechanisms relevant to acute lung injury: when MSCs are administered intravenously, these cells sequester in the lung, but are immune-modulatory, anti-inflammatory, antibacterial, and antiviral. They additionally are anti-lung fibrosis, anti-apoptosis of injured cells, and stimulate alveolar fluid clearance and lung tissue regeneration. These functions of MSCs have been confirmed by multiple in vivo studies in animal models and ex vivo human lung models, and furthermore, the in vivo safety of local and intravenous administration of MSCs has been demonstrated in multiple human clinical trials, including studies of ARDS.43
ADSCs, an abundant type of MSCs, are proposed as a therapeutic option for the treatment of COVID-19 patients in order to reduce morbidity and mortality.43 , 44 We and others have indicated that LDR ≤0.1 Gy was able enhance the proliferation of several human normal cell lines and animal bone-marrow stem cells in vivo, but not that of tumor cells, establishing a precedent for using LDR to stimulate stem cell therapies.45, 46, 47, 48 A recent elegant study with human ADSCs isolated from 10 donors has shown that LDR (50 mGy) promoted the proliferation and survival of ADSCs. LDR at dose of ≤1.0 Gy did not have either genomic or cytotoxic effects detectable, but at greater doses (0.1–2.0 Gy) induced residual double-strand breaks and reduced the long-term survival and proliferation rate of ADSCs.44 These studies suggest that LDR could be a possible mediator to improve stem cells in potential therapy for the patients with COVID-19.
In summary, there are multiple potential mechanisms by which LDRT may provide beneficial effects in patients with COVID-19. Clinical trials are needed to assess if these theoretical benefits will translate into improved outcomes.
4. Current status of LDRT for COVID-19 patients
As of August 16, 2021, 17 different groups have registered LDRT trials for COVID-19 in ClinicalTrials.gov (Search of: low dose radiation, therapy | Covid19 - List Results - ClinicalTrials.gov). Among these clinical trials, four have competed, two are the status of “Active not recruiting” and the rest are “Recruiting”. One of the four completed clinical trials is the only clinical trial to explore “Inhalation Low Dose Radionuclide Therapy in Treatment COVID-19 (NCT04724538)”, for which Inhalation of 99Tcm-pertechnetate aerosol from commercial gas generator that was loaded by 4,165 MBq of 99Tcm -pertechnetate. Computed tomography (CT) scans and blood tests were conducted at 1 d before, 7 and 14 d after inhalation procedure for both 14 treated COVID-19 patients and 11 non-treated COVID-19 patients. The final analyzed results are expected soon.
Within the literature, there are 9 publications for the case report or clinical trials available regarding the use of LDRT with COVID-19, which are summarized in Table 2 .
Table 2.
Detail of clinical report for the use of LDRT to treat COVID-19 patients.
| Authors (Country) | Online time (Published time) | No. of COVID-19 cases | LDRT doses (Gy) | Conclusions |
|---|---|---|---|---|
| Ameri et al.49 (Iran) | July 21, 2020 (Dec. 1, 2020) | 5 | 0.5 | With a response rate of 80%, whole-lung irradiation in a single fraction of 0.5 Gy had encouraging results in oxygen-dependent patients with COVID-19 pneumonia. |
| Del Castillo et al.51 (Peru; Germany) | Aug. 20, 2020 (Dec. 2020) | 1 | 1.0 | Whole-lung low-dose radiotherapy seems to be a promising approach for avoiding or delaying invasive respiratory support. Delivered low doses are far from meeting toxicity ranges |
| Hess et al.52 (USA) | Sept. 28, 2020 (Dec. 1, 2020) | 9 | 1.5 | In a pilot trial of 5 oxygen-dependent elderly patients with COVID-19 pneumonia, low-dose whole-lung radiation led to rapid improvements in clinical status, encephalopathy, and radiographic consolidation without acute toxicity. Low-dose whole-lung radiation appears to be safe, shows early promise of efficacy, and warrants further study. |
| Sanmamed et al.54 (Spain) | Nov. 26, 2020 (March 15, 2021) | 9 | 1.0 | LDRT was a feasible and well-tolerated treatment, with potential clinical improvement. |
| Ameri et al.50 (Iran) | Dec. 2, 2020 (March 15, 2021) | 10 | 0.5/1.0 | LD-WLI with a single fraction of 0.5 Gy or 1 Gy is feasible. |
| Hess et al.53 (USA) | Dec. 16, 2020 (March 15, 2021) | 10 | 1.5 | Low-dose, whole-lung radiation for patients with COVID-19-related pneumonia appears safe and may be an effective immunomodulatory treatment. |
| Papachristofilou et al.58 (Switzerland) | March 15, 2021 (Aug 1, 2021) | 11 | 1.0 | Whole-lung LDRT failed to improve clinical outcomes in critically ill patients requiring mechanical ventilation for COVID-19 pneumonia |
| Bonet et al.56 (Spain) | May 19, 2021 | 10 | 0.5 | LD-RT is not only an encouraging option for COVID-19 pneumonia patients, but a convenient and feasible procedure if performed in a coordinated way by reducing procedure times. |
| Arenas et al.57 (Spain) | July 6, 2021 | 36 | 0.5 | LD-RT appears to be a feasible and safe option in a population with COVID-19 bilateral interstitial pneumonia in the presence of significant comorbidities |
| Sharma et al.55 (India) | Nov. 18, 2020 | 10 | 0.7 | Results of 90% response rate suggest the feasibility and clinical effectiveness of LDRT in COVID-19 patients having moderate to severe risk disease. |
As the first report for the use of LDRT to the patients with COVID-19, Ameri et al. published a pilot study in 2020,49 and second study in 2021.50 In the first study, they treated five patients with COVID-19 aged over 60 years with whole lung exposure to 0.5 Gy. They reported the improvement of four patients within a few days after irradiation, with an average discharging time of 6 days and no obvious radiation toxicity. In the subsequent study, they treated 10 COVID-19 patients with different dose patterns: 5, 1 and 4 patients with a single 0.5 Gy as did in first study, two 0.5 Gy with interval of 48–72 h, and a single dose of 1.0 Gy, respectively. The mean improvements in blood oxygen level at 1 and 2 d after LDRT were 2.4% and 3.6%, respectively. Nine patients were treated after 1 d. Overall, the response rate (RR, defined as an increase in blood oxygen level) of these patients and clinical recovery (CR) were 63.6% and 55.5%, respectively.50
In August 2020 Del Castillo et al.51 reported treatment of a 64-year old patient with COVID-19 with 1.0 Gy whole lungs exposure and found the significant improvement at 3 d post-LDRT, discharged to home at 7 d post-LDRT.
In September 2020, the first pilot study from Hess et al.52 was available, in which they treated five patients with COVID-19 with a single dose (1.5 Gy) whole lungs exposure. They found that the respiratory condition of four patients improved in the first 24 h after exposure with an average recovery time as 1.5 d and discharge time of 12 d. There were no obvious acute radiation toxicities. By comparing the blood tests and imaging examinations the effectiveness of LDRT for patients COVID-19 is estimated about 80%.52 Then they treated another 10 patients with 1.5 Gy whole lung exposure. By comparing with 10 control patients with and without other COVID-19 drugs, three key variables including the median times for clinical recovery and hospital discharge, and intubation rates were 3 vs. 12 d, 12 vs. 20 d, 10% vs 40% (LDRT vs. Control), respectively.53
Sanmamed et al.54 treated 9 COVID-19 patients with whole lung exposure to 1.0 Gy and demonstrated that SatO2/FiO2 index significantly improved 3 d post-LDRT. Compared to COVID-19 patients without LDRT, the median days of hospitalization of patients who received LDRT was reduced by approximately 20%. Seven patients were discharged and two patients died in the hospital; the reasons for death were sepsis and severe baseline chronic obstructive pulmonary disease.
Sharma et al. performed LDRT to treat 10 patients with COVID-19 with an age range of 38–63 years. Both lungs of these patients were irradiated by a single-dose fraction (0.7 Gy). All patients completed the prescribed treatment. Nine patients achieved CR within a period ranging from 3 to 7 d, and discharged from hospital with the range of 10–24 d after LDRT. One patient died 24 d after LDRT. No radiation toxicity was observed in these patients. Overall, RR of these patients was 90%.55
Under the same clinical trial, IPACOVID (NCT04380818),56 , 57 Bonet et al.56 have first optimized the procedure of LDRT for the patients with COVID-19. They were able to perform LDRT in a median of 38 min, suggesting that LDRT could be performed safely and easily. Then they treated 36 patients with COVID-19 pneumonia (a mean age of 84 years, 19 men and 17 women) with 0.5 Gy LDRT along with dexamethasone treatment. Significant improvements compared to before treatment were achieved, as shown by improvements in oxygen saturations and amount of supplemental oxygen needed. In those who survived (23/36, 64%), a significant improvement was observed in the percentage of lung involvement in the CT scan at 1 week after LD-RT. No adverse effects related to radiation treatment have been reported. Through these two studies, they conclude that LDRT is a feasible and safe option in a population with COVID-19 in the presence of significant comorbidities.57
The above clinic studies on LDRT for the patients with COVID-19 generally showed the improved and safe outcomes (Table 2); however, the outcome of Papachristofilou et al.58 is different from the above clinical outcomes. In this study, 22 patients were randomized to either whole-lung LDRT or sham-radiation therapy. They did not observe a difference in 15-d ventilator-free days or overall survival at 28 d between the two arms. In this study, whole-lung LDRT failed to improve clinical outcomes in critically ill patients requiring mechanical ventilation for COVID-19 pneumonia.58 This discrepancy was discussed for the potential confounders or issues.58, 59
In summary, performing LDRT is a new approach to treat pneumonia resulting from COVID-19 disease.60 The completed clinical trials that have performed LDRT to treat COVID-19 showed that the effectiveness of LDRT in treating COVID-19 was up to 90%. The vast majority of primary and secondary outcomes of clinical trial investigations regarding LDRT in treating COVID-19 found that LDRT can be considered a feasible treatment to improve mortality of COVID-19 patients.
5. Perspectives
In the past century there has been substantial evidence from studies of intended and unintended irradiation of animals and humans demonstrating the harmful effects of radiation at high doses. However, LDR at 100 mGy or lower is not only harmless, but can be beneficial with decreased rates of cancer and increased longevity.4 , 10 , 21 , 22 Historically, X-rays have been used to successfully treat bacterial and atypical (including viral) pneumonia.19 , 31 As summarized by Calabrese and Dhawan,31 a single treatment with LD X-rays quickly relieved respiratory distress and markedly reduced the risk of mortality in patients with pneumonia, especially when given early in the disease course. The reported total cure rate reached 83.1% among a total of 863 pneumonia patients treated by X-ray therapy, including 85 virus-induced pneumonia; the reduced pneumonia mortality was similar to those treated by immune serum and sulfonamide. From the limited clinical trials (Table 2), LDRT in the most of cases appears to reduce or significantly relieve the course of severe pneumonia.
With regard to the biologic effects of LDR, Torres Royo et al.61 summarized the previous studies on LDRT for benign pathologies with the conclusion that the net effect at doses of 0.1–0.3 Gy are anti-inflammatory actions. This is consistent with another comprehensive review, stating that low (<0.1 Gy) to moderate doses (0.1–1.0 Gy) of low and high LET radiation have anti-inflammatory effects on individuals with local inflammatory conditions, and may result in improvement in a variety of clinical symptoms and parameters.62 Therefore, LDRT would offer a low-risk, noninvasive, therapeutic alternative for elderly patients, with low toxicity. Currently it is only considered as a rescue treatment for the patients with COVID-19 when standard management has failed.61
There remain several questions need to be further addressed in the future clinical trials. The first question is the dose level of LDRT for the patients with COVID-19. Several clinical trials showed improvement for the therapy of COVID-19 patients with dose range of 1.5 to 0.5 Gy (Table 2). Although the observation of Emory group indicated the improvement of 1.5 Gy-treated patients, the safety of this dose over the long-term outcome remains a concern. Based on recent estimation of the risks of radiation-induced cancer (RIC) and cardiac disease, Arruda et al.63 pointed out that the lung is at the highest risk for RIC. Lifetime attributable risks for lung cancer with a radiotherapy dose of 1 Gy was elevated for women >60 years of age and men <40 years of age. Only 0.5 Gy had an acceptable risk of exposure-induced death. Therefore, Arruda et al. suggested that radiotherapy dose ≤0.5 Gy provides an acceptable LAR estimate (≤1%) for RIC and REID, irrespective of sex and age. The current ongoing trials should initially use doses ≤0.5 Gy to maintain the risks at an acceptable level and include only patients who fail or do not have any other treatment option.63
Although there more than 10 clinical studies that have carried out to assess the role of LDRT in COVID-19 patients, most of these studies (Table 2) were with small participant numbers of 1–10 patients, were not controlled, and the results were complicated by treatment by other mechanisms such as remdesivir and dexamethasone.64
Finally, the risk of radiation exposure in younger patients is less well understood. As such, we do not endorse the use of LDRT for teenagers or young adults with COVID-19 to ensure a lack of any potential side-effects for these radiation-susceptible populations. Further studies are needed to determine the optimal patient populations for consideration of LDRT.
Given the rapid rise and spread of new variants, it may be the time now to consider using LDR to fight COVID-19, either as a therapeutic procedure alone, or as an adjunct to other forms of therapy being tested.64
Declaration of conflicting interest
No.
Acknowledgments
The author of the present review greatly thanks to the authors whom the publications are cited from. The authors also apologize to those who could not be cited due to space constraints.
References
- 1.Forchette L., Sebastian W., Liu T. A comprehensive review of COVID-19 virology, vaccines, variants, and therapeutics. Curr Med Sci. 2021;9:1–15. doi: 10.1007/s11596-021-2395-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ghadimi-Moghadam A., Haghani M., Bevelacqua J.J., et al. COVID-19 tragic Pandemic: concerns over unintentional "Directed accelerated evolution" of novel coronavirus (SARS-CoV-2) and introducing a modified treatment method for ARDS. J Biomed Phys Eng. 2020;10(2):241–246. doi: 10.31661/jbpe.v0i0.2003-1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Calabrese E.J., Dhawan G., Kapoor R., et al. Radiotherapy treatment of human inflammatory diseases and conditions: optimal dose. Hum Exp Toxicol. 2019;38(8):888–898. doi: 10.1177/0960327119846925. [DOI] [PubMed] [Google Scholar]
- 4.Luckey T.D. Physiological benefits from low levels of ionizing radiation. Health Phys. 1982;43(6):771–789. doi: 10.1097/00004032-198212000-00001. [DOI] [PubMed] [Google Scholar]
- 5.Olivieri G., Bodycote J., Wolff S. Adaptive response of human lymphocytes to low concentrations of radioactive thymidine. Science. 1984;223(4636):594–597. doi: 10.1126/science.6695170. [DOI] [PubMed] [Google Scholar]
- 6.Samson L., Cairns J. A new pathway for DNA repair in Escherichia coli. Nature. 1977;267(5608):281–283. doi: 10.1128/JB.181.9.2878-2882.1999. [DOI] [PubMed] [Google Scholar]
- 7.Cai L., Jiang J., Wang B., et al. Induction of an adaptive response to dominant lethality and to chromosome damage of mouse germ cells by low dose radiation. Mutat Res. 1993;303(4):157–161. doi: 10.1016/0165-7992(93)90017-p. [DOI] [PubMed] [Google Scholar]
- 8.Cai L., Liu S.Z. Effect of cycloheximide on the adaptive response induced by low dose radiation. Biomed Environ Sci. 1992;5(1):46–52. [PubMed] [Google Scholar]
- 9.Cai L., Liu S.Z. Study on the mechanism of cytogenetic adaptive response induced by low dose radiation. Chin. Med. J. (Engl). 1992;105(4):277–283. [PubMed] [Google Scholar]
- 10.Chung M., Rhee H.Y., Chung W.K. Clinical approach of low-dose whole-brain ionizing radiation treatment in Alzheimer's disease dementia patients. J Alzheimers Dis. 2021;80(3):941–947. doi: 10.3233/JAD-210042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cuttler J.M., Moore E.R., Hosfeld V.D., et al. Treatment of Alzheimer disease with CT scans: a case report. Dose Response. 2016;14(2) doi: 10.1177/1559325816640073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cuttler J.M., Moore E.R., Hosfeld V.D., et al. Second update on a patient with Alzheimer disease treated by CT scans. Dose Response. 2018;16(1) doi: 10.1177/1559325818756461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cuttler J.M., Abdellah E., Goldberg Y., et al. Low doses of ionizing radiation as a treatment for alzheimer's disease: a pilot study. J Alzheimers Dis. 2021;80(3):1119–1128. doi: 10.3233/JAD-200620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Calabrese E.J., Dhawan G. How radiotherapy was historically used to treat pneumonia: could it be useful today? Yale J Biol Med. 2013;86(4):555–570. [PMC free article] [PubMed] [Google Scholar]
- 15.Tay M.Z., Poh C.M., Rénia L., et al. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20(6):363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Doss M. Treatment of COVID-19 with individualized immune boosting interventions. 2020. https://osf.io/6zy9u/ pdfVersion: Created: March 20, 2020, Preprint DOI:
- 17.Kirkby C., Mackenzie M. Is low dose radiation therapy a potential treatment for COVID-19 pneumonia? Radiother Oncol. 2020;147:221. doi: 10.1016/j.radonc.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kumar R., Haresh K.P., Sharma D.N., et al. Low-dose radiotherapy for COVID 19: a radioimmunological perspective. J Canc Res Therapeut. 2021;17(2):295–302. doi: 10.4103/jcrt.JCRT_1045_20. [DOI] [PubMed] [Google Scholar]
- 19.Li J.J. Mitigating coronavirus-induced acute respiratory distress syndrome by radiotherapy. iScience. 2020;23(6):101215. doi: 10.1016/j.isci.2020.101215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Genard G., Lucas S., Michiels C. Reprogramming of tumor-associated macrophages with anticancer therapies: radiotherapy versus chemo- and immunotherapies. Front Immunol. 2017;8:828. doi: 10.3389/fimmu.2017.00828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang G., Li W., Jiang H., et al. Low-dose radiation may be a novel approach to enhance the effectiveness of cancer therapeutics. Int J Canc. 2016;139(10):2157–2168. doi: 10.1002/ijc.30235. [DOI] [PubMed] [Google Scholar]
- 22.Cui J., Yang G., Pan Z., et al. Hormetic response to low-dose radiation: focus on the immune system and its clinical implications. Int J Mol Sci. 2017 Jan 27;18(2):280. doi: 10.3390/ijms18020280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meziani L., Robert C., Classe M., et al. Low doses of radiation increase the immunosuppressive profile of lung macrophages during viral infection and pneumonia. Int J Radiat Oncol Biol Phys. 2021;110(5):1283–1294. doi: 10.1016/j.ijrobp.2021.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Darenskaya M., Kolesnikova L., Kolesnikov S. The association of respiratory viruses with oxidative stress and antioxidants: implications for the COVID-19 pandemic. Curr Pharmaceut Des. 2021;27(13):1618–1627. doi: 10.2174/1381612827666210222113351. [DOI] [PubMed] [Google Scholar]
- 25.Sharif-Askari N.S., Sharif-Askari F.S., Mdkhana B., et al. Upregulation of oxidative stress gene markers during SARS-COV-2 viral infection. Free Radic Biol Med. 2021;172:688–698. doi: 10.1016/j.freeradbiomed.2021.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cekerevac I., Turnic T.N., Draginic N., et al. Predicting severity and intrahospital mortality in COVID-19: the place and role of oxidative stress. Oxid Med Cell Longev. 2021;2021:6615787. doi: 10.1155/2021/6615787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Khakwani M., Horgan C., Ewing J. COVID-19-associated oxidative damage to red blood cells. Br J Haematol. 2021;193(3):481. doi: 10.1111/bjh.17317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soria-Castro E., Soto M.E., Guarner-Lans V., et al. The kidnapping of mitochondrial function associated with the SARS-CoV-2 infection. Histol Histopathol. 2021;16:18354. doi: 10.14670/HH-18-354. [DOI] [PubMed] [Google Scholar]
- 29.Olagnier D., Farahani E., Thyrsted J., et al. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat Commun. 2020;11(1):4938. doi: 10.1038/s41467-020-18764-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cuadrado A., Pajares M., Benito C., et al. Can activation of NRF2 be a strategy against COVID-19? Trends Pharmacol Sci. 2020;41(9):598–610. doi: 10.1016/j.tips.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Calabrese E.J., Kozumbo W.J., Kapoor R., et al. Nrf2 activation putatively mediates clinical benefits of low-dose radiotherapy in COVID-19 pneumonia and acute respiratory distress syndrome (ARDS): novel mechanistic considerations. Radiother Oncol. 2021;160:125–131. doi: 10.1016/j.radonc.2021.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xing X., Zhang C., Shao M., et al. Low-dose radiation activates Akt and Nrf2 in the kidney of diabetic mice: a potential mechanism to prevent diabetic nephropathy. Oxid Med Cell Longev. 2012:291087. doi: 10.1155/2012/291087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhao Y., Kong C., Chen X., et al. Repetitive exposure to low-dose X-irradiation attenuates testicular apoptosis in type 2 diabetic rats, likely via Akt-mediated Nrf2 activation. Mol Cell Endocrinol. 2016;422:203–210. doi: 10.1016/j.mce.2015.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang C., Xing X., Zhang F., et al. Low-dose radiation induces renal SOD1 expression and activity in type 1 diabetic mice. Int J Radiat Biol. 2014;90(3):224–230. doi: 10.3109/09553002.2014.877174. [DOI] [PubMed] [Google Scholar]
- 35.Jin C., Qin L., Shi Y., et al. CDK4-mediated MnSOD activation and mitochondrial homeostasis in radioadaptive protection. Free Radic Biol Med. 2015;81:77–87. doi: 10.1016/j.freeradbiomed.2014.12.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang W., Lei W., Jiang L., et al. Therapeutic mechanisms of mesenchymal stem cells in acute respiratory distress syndrome reveal potentials for Covid-19 treatment. J Transl Med. 2021;19(1):198. doi: 10.1186/s12967-021-02862-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kouroupis D., Lanzoni G., Linetsky E., et al. Umbilical cord-derived mesenchymal stem cells modulate TNF and soluble TNF receptor 2 (sTNFR2) in COVID-19 ARDS patients. Eur Rev Med Pharmacol Sci. 2021;25(12):4435–4438. doi: 10.26355/eurrev_202106_26156. [DOI] [PubMed] [Google Scholar]
- 38.Wang G.J., Cai L. Induction of cell-proliferation hormesis and cell-survival adaptive response in mouse hematopoietic cells by whole-body low-dose radiation. Toxicol Sci. 2000;53(2):369–376. doi: 10.1093/toxsci/53.2.369. [DOI] [PubMed] [Google Scholar]
- 39.Li W., Wang G., Cui J., et al. Low-dose radiation (LDR) induces hematopoietic hormesis: LDR-induced mobilization of hematopoietic progenitor cells into peripheral blood circulation. Exp Hematol. 2004;32(11):1088–1096. doi: 10.1016/j.exphem.2004.07.015. [DOI] [PubMed] [Google Scholar]
- 40.Guo W.Y., Wang G.J., Wang P., et al. Acceleration of diabetic wound healing by low-dose radiation is associated with peripheral mobilization of bone marrow stem cells. Radiat Res. 2010;174(4):467–479. doi: 10.1667/RR1980.1. [DOI] [PubMed] [Google Scholar]
- 41.Wei L.C., Ding Y.X., Liu Y.H., et al. Low-dose radiation stimulates Wnt/β-catenin signaling, neural stem cell proliferation and neurogenesis of the mouse hippocampus in vitro and in vivo. Curr Alzheimer Res. 2012;9(3):278–289. doi: 10.2174/156720512800107627. [DOI] [PubMed] [Google Scholar]
- 42.Ministro A., de Oliveira P., Nunes R.J., et al. Low-dose ionizing radiation induces therapeutic neovascularization in a pre-clinical model of hindlimb ischemia. Cardiovasc Res. 2017;113(7):783–794. doi: 10.1093/cvr/cvx065. [DOI] [PubMed] [Google Scholar]
- 43.Rogers C.J., Harman R.J., Bunnell B.A., et al. Rationale for the clinical use of adipose-derived mesenchymal stem cells for COVID-19 patients. J Transl Med. 2020;18(1):203. doi: 10.1186/s12967-020-02380-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Schröder A., Kriesen S., Hildebrandt G., et al. First insights into the effect of low-dose X-ray irradiation in adipose-derived stem cells. Int J Mol Sci. 2019;20(23):6075. doi: 10.3390/ijms20236075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jiang H., Xu Y., Li W., et al. Low-dose radiation does not induce proliferation in tumor cells in vitro and in vivo. Radiat Res. 2008;170(4):477–487. doi: 10.1667/rr1132.1. [DOI] [PubMed] [Google Scholar]
- 46.Liang X., Gu J., Yu D., et al. Low-dose radiation induces cell proliferation in human embryonic lung fibroblasts but not in lung cancer cells: importance of ERK1/2 and AKT signaling pathways. Dose Response. 2016;14(1) doi: 10.1177/1559325815622174. 1559325815622174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liang X., So Y.H., Cui J., et al. The low-dose ionizing radiation stimulates cell proliferation via activation of the MAPK/ERK pathway in rat cultured mesenchymal stem cells. J Radiat Res. 2011;52(3):380–386. doi: 10.1269/jrr.10121. [DOI] [PubMed] [Google Scholar]
- 48.Yang L., Liu Z., Chen C., et al. Low-dose radiation modulates human mesenchymal stem cell proliferation through regulating CDK and Rb. Am J Transl Res. 2017;9(4):1914–1921. [PMC free article] [PubMed] [Google Scholar]
- 49.Ameri A., Rahnama N., Bozorgmehr R., et al. Low-dose whole-lung irradiation for COVID-19 pneumonia: short course results. Int J Radiat Oncol Biol Phys. 2020;108(5):1134–1139. doi: 10.1016/j.ijrobp.2020.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ameri A., Ameri P., Rahnama N., et al. Low-dose whole-lung irradiation for COVID-19 pneumonia: final results of a pilot study. Int J Radiat Oncol Biol Phys. 2021;109(4):859–866. doi: 10.1016/j.ijrobp.2020.11.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Del Castillo R., Martinez D., Sarria G.J., et al. Low-dose radiotherapy for COVID-19 pneumonia treatment: case report, procedure, and literature review. Strahlenther Onkol. 2020;196(12):1086–1093. doi: 10.1007/s00066-020-01675-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hess C.B., Buchwald Z.S., Stokes W., et al. Low-dose whole-lung radiation for COVID-19 pneumonia: planned day 7 interim analysis of a registered clinical trial. Cancer. 2020;126(23):5109–5113. doi: 10.1002/cncr.33130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hess C.B., Nasti T.H., Dhere V.R., et al. Immunomodulatory low-dose whole-lung radiation for patients with coronavirus disease 2019-related pneumonia. Int J Radiat Oncol Biol Phys. 2021;109(4):867–879. doi: 10.1016/j.ijrobp.2020.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sanmamed N., Alcantara P., Cerezo E., et al. Low-dose radiation therapy in the management of coronavirus disease 2019 (COVID-19) pneumonia (LOWRAD-Cov19): preliminary report. Int J Radiat Oncol Biol Phys. 2021;109(4):880–885. doi: 10.1016/j.ijrobp.2020.11.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sharma D.N., Guleria R., Wig N., et al. Low dose radiation therapy for COVID-19 pneumonia: a pilot study. 2020. medRxiv preprint. [DOI] [PMC free article] [PubMed]
- 56.Bonet M., Vázquez S., García E., et al. Saving time in the radiotherapy procedures for COVID-19 pneumonia treatment. A single-institution experience. Clin Transl Oncol. 2021:1–6. doi: 10.1007/s12094-021-02634-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Arenas M., Algara M., De Febrer G., et al. Could pulmonary low-dose radiation therapy be an alternative treatment for patients with COVID-19 pneumonia? Preliminary results of a multicenter SEOR-GICOR nonrandomized prospective trial (IPACOVID trial) Strahlenther Onkol. 2021:1–11. doi: 10.1007/s00066-021-01803-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Papachristofilou A., Finazzi T., Blum A., et al. Low-dose radiation therapy for severe COVID-19 pneumonia: a randomized double-blind study. Int J Radiat Oncol Biol Phys. 2021;110(5):1274–1282. doi: 10.1016/j.ijrobp.2021.02.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bevelacqua J.J., Welsh J.S., Mortazavi S.M.J. Regarding: "the risk of induced cancer and ischemic heart disease following low dose lung irradiation for COVID-19: estimation based on a virtual case. Int J Radiat Biol. 2021;97(3):313–314. doi: 10.1080/09553002.2021.1864502. [DOI] [PubMed] [Google Scholar]
- 60.Ghaznavi H. Effectiveness of low-dose radiation therapy to improve mortality of COVID-19. J Canc Res Clin Oncol. 2021:1–4. doi: 10.1007/s00432-021-03707-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Torres Royo L., Antelo Redondo G., Árquez Pianetta M., et al. Low-dose radiation therapy for benign pathologies. Rep Practical Oncol Radiother. 2020;25(2):250–254. doi: 10.1016/j.rpor.2020.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lumniczky K., Impens N., Armengol G., et al. Low dose ionizing radiation effects on the immune system. Environ Int. 2021;149:106212. doi: 10.1016/j.envint.2020.106212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Arruda G.V., Weber R.R.D.S., Bruno A.C., et al. The risk of induced cancer and ischemic heart disease following low dose lung irradiation for COVID-19: estimation based on a virtual case. Int J Radiat Biol. 2021;97(2):120–125. doi: 10.1080/09553002.2021.1846818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kapoor R., Welsh J.S., Dhawan V., et al. Low-dose radiation therapy (LDRT) for COVID-19 and its deadlier variants. Arch Toxicol. 2021:1–8. doi: 10.1007/s00204-021-03124-x. [DOI] [PMC free article] [PubMed] [Google Scholar]