Abstract
Improving the quality of life (QoL) is crucial in the management of pediatric inflammatory bowel disease (IBD). We aimed to (1) Validate the IMPACT-III questionnaire in Italian IBD children; (2) explore factors associated to QoL in pediatric IBD. Internal consistency, concurrent validity, discriminant validity and reproducibility of the Italian version of the IMPACT-III questionnaire was measured in IBD children/adolescents in 8 centers. Associations between patient and disease characteristics and the IMPACT-III domains were analyzed through quantile regression analysis. The IMPACT-III questionnaire, collected in 282 children with IBD (median age: 14.8 years; IQR 12.4–16.4) showed a median total score of 76 (IQR 67–83). Female gender, active disease and age were negatively associated with the total IMPACT-III score. Specifically, female gender was negatively associated with the Bowel/Systemic Symptoms, Emotional and Treatment domain scores, while disease activity was significantly associated with Bowel Symptoms and Treatment/Interventions reported QoL. The IMPACT- III showed good internal consistency (Cronbach’s alpha coefficient = 0.87, 95% CI 0.85–0.89) and reproducibility (Concordance Correlation Coefficient = 0.66, 95% CI 0.57–0.74). In Italian children with IBD active disease, female gender and adolescence are associated to a worse QoL, indicating the need of more attention in this subgroup of young patients. IMPACT-III questionnaire is a reliable instrument to measure QoL in Italian children.
Subject terms: Gastroenterology, Health care
Introduction
Inflammatory bowel diseases (IBD) including Crohn’s disease (CD), Ulcerative Colitis (UC) and IBD unclassified (IBD-U), are chronic inflammatory conditions characterized by an unpredictable clinical course, alternating periods of remission and clinical relapses. About 25% of IBD cases are diagnosed in childhood (< 18 years) and the global incidence of pediatric IBD is increasing worldwide1. Children with IBD face a lifelong disabling condition, experiencing an increased risk of psychological distress and social disruption, and report impaired quality of life (QoL) in comparison to healthy controls2.
Improvement of patient-reported outcomes (PROs), such as QoL, has recently been recognized as an important target of IBD management3–5, therefore a robust, culturally adapted and reliable instrument is necessary to accurately measure QoL, both in clinical and research settings. The IMPACT III questionnaire is the most frequently used scale to assess disease specific QoL in children with IBD6,7. The original version has been translated in several languages but so far, a rigorous cultural validation has been conducted only in few European countries (UK, Switzerland and Croatia8–10).
Several factors, including individual and disease specific characteristics, have been reported to impact QoL in IBD. It has been described that disease activity is the main predictive factor of IBD-related QoL in adults2, however pediatric data are lacking. An accurate knowledge of the demographic and clinical variables affecting QoL in a specific geographical context can be useful in the interpretation of reported QoL, with the ultimate goal of improving care of pediatric patients with IBD.
The aim of the present study was to assess the validity of the IMPACT III questionnaire as a measure of QoL in Italian IBD children. Furthermore, we aimed to identify patient and disease characteristics associated with QoL of children with IBD.
Methods
Patients
Children and adolescents (age 8–18 years) with a confirmed diagnosis of IBD (according to the revised Porto criteria)11, established at least 6 months prior to enrolment, were invited to participate. Patients with cognitive impairment, severe co-morbidities and presence of an ostomy were not included, as their IBD related QoL was likely to be significantly influenced by factors different from the majority of IBD children who do not present these complications. Children were enrolled at 8 regional referral pediatric IBD centers, equally distributed across the Italian territory (Ancona, Brescia, Milano, Pesaro, Roma, San Giovanni Rotondo, Trieste and Monza), both in out-patient and in-patient setting, irrespective of disease activity. Informed consent was obtained by the parents or guardians. The study was approved by the coordinating center Ethics Committee (“Comitato Etico Regione Marche, CERM”, protocol number 2016–0359 OR).
At the time of enrolment, the following data were collected: age at diagnosis, diagnosis, location and phenotype according to Paris classification11, recent relapses (12 months), previous surgery, fecal calprotectin levels and growth. Body weight (BW) and height (H) were collected and body mass index (BMI) was calculated as weight/height2 (kg/m2). BW, H, and BMI z-scores were then calculated. Disease activity was measured using the weighted Pediatric Crohn’s Disease activity index (wPCDAI)12 or Pediatric Ulcerative Colitis Index (PUCAI)13 for CD and UC patients respectively plus Physician Global assessment (PGA). Physicians indicated whether patients had quiescent, mild, moderate, or severe PGA, according to the definitions of the ImproveCareNow network [https://improvecarenow.org]14. Fecal calprotectin level > 250 µg/g was considered as an indicator of active mucosal disease.
Quality of life questionnaires
Generic health related quality of life (HRQoL) was assessed using the generic HRQoL questionnaire for children, Pediatric Quality of Life Inventory (PedsQL, Italian version) designed by J. Varni15. PedsQL is a 24-items questionnaire with proven validity and reliability in pediatric patients, exploring 4 different domains (Physical, Emotional, Social and School Functioning). The Total score, the Physical Health Summary score and Psycho-Social Health summary score were calculated, according to the author guidelines.
Disease specific QoL was assessed using the Italian version of the IMPACT-III questionnaire. The original English version of the IMPACT-III was forward- and backward-translated and culturally adapted according to the guidelines outlined by Beaton and colleagues16 in 2008 by other authors (data not published), therefore an Italian version certified by the developer (Anthony Otley) was already available and obtained with permission. The questionnaire contains 35 IBD specific items, exploring six different domains: Bowel Symptoms, Emotional functioning, Social functioning, Systemic symptoms, Body image and Treatment/Intervention. The responses for each question are on a five-point Likert scale (scores are 0–4, with 0 indicating the highest HRQoL and 4 indicating the lowest). As per author’s guidelines scores are then linearly and reversely transformed to a range of 0–100, with 0 indicating the lowest HRQoL and 100 indicating the highest HRQoL. Missing data on IMPACT-III scores were treated as indicated in the IMPACT-III User’s Guide, total and domain scores were computed as the mean of all completed items17.
Children were requested to fill in both PedsQL and IMPACT III without parental help, but could ask the administering physician for information.
To assess the test–retest reliability children were contacted by phone after 4–8 weeks. If the patient reported no change in clinical symptoms and no further treatments had been introduced since the enrolment, children were asked to compile a second copy of the IMPACT III and return it to the refering center.
Statistical analysis
Validation of the Italian version of the IMPACT III questionnaire: reliability and validity evaluation
The questionnaire’s validation was carried out using sample data. Internal reliability was assessed using Cronbach’s α coefficient, analyzing the consistency of each domain. The distributions of each domain score were asymmetric, hence a non-parametric approach was chosen. In order to evaluate the concurrent validity, the correlation between PedsQL and IMPACT III total score was analyzed. A correlation analysis was performed between each domain of IMPACT III and PedsQL’s Psychosocial Health Summary Score, Physical Health Summary Score by Spearman’s correlation coefficient and 95% Confidence Intervals (95% CI).
Discriminant validity was analyzed to determine whether the questionnaire (total score and domain scores) was able to discriminate patients in remission from patients with moderate/severe activity, according to the wPCDAI and PUCAI results. The Wilcoxon sum-rank test was used to evaluate differences between domain scores of the two groups.
Data collected from patients with a stable disease (no change in reported symptoms and no additional treatments), who had completed IMPACT-III questionnaire twice, were analyzed for testing the reproducibility, estimating the median of the differences in the domains of IMPACT III score and 95% Confidence Intervals (95% CI) and by means of Concordance Correlation Coefficient (CCC)18 and 95% Confidence Intervals (95% CI); CCC quantifies the agreement between two measurements of the same variable.
IMPACT-III questionnaire analysis
A descriptive analysis of the main characteristics of subjects was performed to compare CD and UC patients. Medians and interquartile ranges (IQR), absolute and percentage frequencies were used to summarize quantitative and qualitative variables respectively. Wilcoxon rank sum test or Chi-square/Fisher exact test were performed to evaluate differences between groups.
The distributions of scores of each IMPACT-III domain according to participating centres were summarized using medians and IQRs and comparisons between centres were performed using Kruskal–Wallis test.
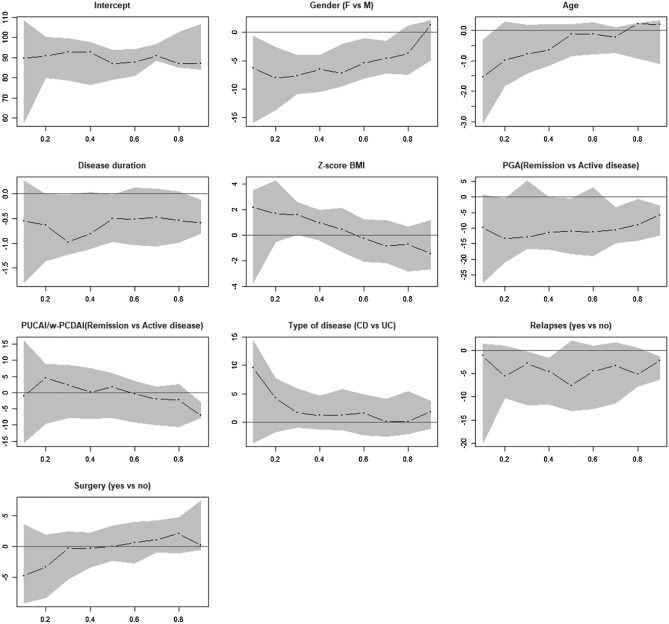
Multiple quantile regression analysis was used to evaluate the impact of subject characteristics and type of disease on the median value of each IMPACT-III domain distribution (dependent variable). The following independent variables were considered in each model: gender, age, disease duration, BMI Z-score, disease severity measured by PGA and PUCAI/wPCDAI (both dichotomized as remission and active disease), type of disease (CD versus UC), occurrence of relapses during the last year and surgery. Quantile regression is a non parametric method that does not make any model assumption that may not hold and provides a more complete view of the effect of each variable on the global distribution of IMPACT-III scores. Nine deciles of the distributions of scores were evaluated when the total score was analyzed. Results for IMPACT-III total score domain were showed in a plot where the x-axis indicated the 9 deciles of the response distribution (score) and y-axis exhibited the effect of each variable (regression coefficients) and 95% confidence bands (grey area). If the confidence interval included 0, the estimates could not be considered statistically different from 0.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the coordinating center Ethics Committee (“Comitato Etico Regione Marche, CERM”, protocol number 2016–0359 OR). Informed consent was obtained from all individual participants included in the study.
Results
A total of 282 children with IBD (median age 14.8 years, IQR 12.4–16.4) were consecutively enrolled over 19 months, after informed consent was obtained. One-hundred and fifty-five children had CD (54.9%), 123 had UC (43.6%) and 4 had IBD-U (1.4%). At the time of enrolment, the majority of subjects was in clinical remission (68.7%; 95% CI 61.1–75.2), with only 5 patients (3 with UC) having severe disease. Table 1 shows the clinical characteristics of enrolled subjects. No significant difference between CD and UC subjects was found, except a higher prevalence of males in CD subjects (p = 0.010) and a higher number of relapses during the last year in the UC group (p = 0.012). The IMPACT-III questionnaire was completely filled in by 272 children (97.8% of the enrolled population), with 6 children missing only one response.
Table 1.
Demographic and clinical characteristics of subjects according to type of disease.
| CD (n = 155) | UC (n = 123) | p | |
|---|---|---|---|
| Gender [M, n (%)] | 97 (62.6%) | 57 (46.3%) | 0.010a |
| Age [years, median (IQR)]) | 15.2 (12.6; 16.7) | 14.6 (12.5; 16.5) | 0.258b |
| Disease duration [years, median (IQR)] | 2.9 (1.4; 5.2) | 3.8 (1.7; 6.2) | 0.052b |
| Z-score weight [median (IQR)] | − 0.5 (− 1.2; 0.3) | − 0.4 (− 0.9; 0.2) | 0.663b |
| Z-score BMI [median (IQR)] | − 0.48 (− 1.1; 0.2) | − 0.4 (− 1.19; 0.2) | 0.795b |
| Disease activity | |||
| N. of relapses [median (IQR)] | 0 (0; 3) | 0 (0; 4) | 0.012b |
| PUCAI/w-PCDAI [n (%)] | 0.492c | ||
| Remission | 111 (73.5%) | 90 (75%) | |
| Mild | 33 (21.9%) | 20 (16.7%) | |
| Moderate | 5 (3.3%) | 7 (5.8%) | |
| Severe | 2 (1.3%) | 3 (2.5%) | |
| PGA [n (%)] | 0.138c | ||
| Remission | 106 (70.2%) | 87 (75.5%) | |
| Mild | 37 (24.5%) | 17 (14.8%) | |
| Moderate | 7 (4.6%) | 9 (7.8%) | |
| Severe | 1 (0.7%) | ||
| Surgery [yes, n (%)] | 9 (11.25%) | 6 (8.45%) | 0.439a |
| Fecal calprotectin > 250 ug/g [yes, n (%)] | 32 (36.4%) | 30 (44.1%) | 0.800a |
| Fecal calprotectin [ug/g; median (IQR)] | 118 (37; 427) | 193 (32; 707) | 0.364b |
p-values refer to: aChi-square test; bWilcoxon test; cFisher Exact test.
IQR interquartile range.
The number of patients and IMPACT-III domain scores for each participating centre is shown in Supplementary Table S1. On average, 35 subjects were recruited in each centre, with two centres (Pesaro, Monza) with the lowest numbers of participants (6 and 10 respectively) and Trieste with the highest number of participants. There were no significant differences in the total score and in the subdomains of the questionnaire between participating centres, except for a significant difference in the Bowel Symptoms domain score between San Giovanni Rotondo and Trieste.
IMPACT-III validation
Internal consistency and concurrent validity
Internal consistency and concurrent validity results are shown in Tables 2 and 3, respectively. The overall Cronbach's alpha coefficient showed a very high consistency among all the items of the questionnaire (α = 0.87, 95% CI 0.85–0.89). Cronbach's alpha coefficients showed high internal consistency for the Emotional Functioning and Systemic Symptoms domains; a good internal consistency for Bowel Symptoms, Social Functioning and Body Image domains. Cronbach’s alpha of the Treatment Intervention domain was 0.52, showing a fair internal consistency.
Table 2.
Internal consistency.
| IMPACT III domains | Cronbach's alpha | |
|---|---|---|
| α | 95% CI | |
| Bowel symptoms | 0.73 | (0.69; 0.78) |
| Emotional functioning | 0.83 | (0.8; 0.86) |
| Social functioning | 0.72 | (0.67; 0.77) |
| Systemic simptoms | 0.8 | (0.76; 0.84) |
| Body imgae | 0.65 | (0.58; 0.72) |
| Treatment Interventions | 0.52 | (0.42; 0.62) |
95% CI 95% Confidence Interval.
Table 3.
Concurrent validity.
| IMPACT III domains | PEDSQL domains | |||
|---|---|---|---|---|
| Psychosocial health summary score | Physical health summary score | |||
| r (95%CI) | p | r (95% CI) | p | |
| Bowel symptoms | 0.52 (0.43; 0.61) | < 0.001 | 0.64 (0.56; 0.70) | < 0.001 |
| Emotional functioning | 0.61 (0.53; 0.68) | < 0.001 | 0.54 (0.45; 0.62) | < 0.001 |
| Social functioning | 0.53 (0.44; 0.61) | < 0.001 | 0.44 (0.34; 0.53) | < 0.001 |
| Systemic simptoms | 0.63 (0.55; 0.70) | < 0.001 | 0.67 (0.60; 0.73) | < 0.001 |
| Body imgae | 0.42 (0.32; 0.51) | < 0.001 | 0.40 (0.30; 0.49) | < 0.001 |
| Treatment Interventions | 0.47 (0.37; 0.55) | < 0.001 | 0.39 (0.29; 0.49) | < 0.001 |
r Spearman correlation coefficient, 95% CI 95% Confidence interval, p refers to Spearman correlation test.
IMPACT-III and PedsQL scores showed a good correlation (r = 0.75, 95% CI 0.69–0.80). All the IMPACT-III domains exhibited positive correlation coefficients with PedsQL domains (Table 3). Physical Health Summary Score domain on PedsQL was strongly correlated with Bowel Symptoms domain (r = 0.64) and with the Systemic Symptoms domain (r = 0.67) on IMPACT III. Psychosocial Health Summary Score was moderately correlated to all IMPACT-III domains, ranging from 0.42 to 0.63.
Discriminant validity and reproducibility
Discriminant validity performed using PUCAI/wPCDAI indices showed significant differences in all the IMPACT-III domains (Fig. 1). IMPACT-III questionnaire discriminated the severity of disease’s activity, i.e. patients with moderate-severe activity had significantly lower QoL scores in all IMPACT-III domains compared to patients in remission.
Figure 1.
Discriminant validity for IMPACT-III questionnaire.
Reliability analysis was based on 176 subjects that compiled the questionnaire twice and had a stable disease. The 95% CI of the medians of the differences in IMPACT III scores between the two measurements always included zero for each domain, indicating no significant difference between the two time points (Supplementary Fig. S1). The coefficients were greater than 0.5 in all the domains highlighting a good level of reproducibility (Supplementary Table S2).
IMPACT-III questionnaire analysis
A median total IMPACT-III score of 76 (IQR 67–83) was observed, with no difference between CD and UC children (median scores: 78, IQR 68–84 and 76, IQR 66–82, respectively). Table 4 shows the comparison of IMPACT-III domain scores according to the type of disease. Bowel symptoms domain score was significantly higher in subjects affected by CD, while no significant difference between the two groups was found in the other domains.
Table 4.
Health-related quality of life according to type of disease.
| IMPACT-III domains | CD (n = 155) | UC (n = 123) | p | ||
|---|---|---|---|---|---|
| Min; max | Median (IQR) | Min; max | Median (IQR) | ||
| Bowel symptoms | 32; 100 | 89 (79; 96) | 21; 89 | 82 (68; 89) | 0.001 |
| Emotional functioning | 14; 100 | 75 (64; 86) | 7; 100 | 71 (56; 89) | 0.158 |
| Social functioning | 44; 94 | 77 (71; 82) | 42; 92 | 75 (71; 81) | 0.31 |
| Systemic simptoms | 17; 100 | 75 (67; 83) | 8; 100 | 75 (58; 83) | 0.168 |
| Body image | 17; 100 | 67 (58; 83) | 33; 100 | 67 (58; 83) | 0.196 |
| Treatment interventions | 17; 100 | 75 (67; 92) | 17; 100 | 75 (67; 83) | 0.347 |
| Total score | 48; 95 | 78 (68; 84) | 34; 96 | 76 (66; 82) | 0.119 |
The total and single domain scores range from 0 to100, with 0 indicating the lowest HRQoL and 100 indicating the highest HRQoL. p-value refers to Wilcoxon rank sum test. IQR interquartile range, min minimum score value, max maximum score value.
Quantile regression analysis
Table 5 shows the results of the multiple quantile regression analysis at median level for each of the IMPACT-III domains. Female gender, lower BMI z-scores and relapses during the previous year were negatively associated to the Bowel Symptoms domain score. Remission (according to PGA) and CD diagnosis were associated with better QoL related to bowel symptoms (subjects with CD with a median score higher of 5 points compared to patients with UC).
Table 5.
Factors associated with IMPACT-III domains.
| Variables | Bowel symptoms | Emotional functioning | Social functioning | Systemic symptoms | Body image | Treatment/intervention | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| b | 95% CI | b | 95% CI | b | 95% CI | b | 95% CI | b | 95% CI | b | 95% CI | |
| Gender (F vs M) | − 5.8 | (− 8.3; − 2.4) | − 12.5 | (− 16.3; − 3.7) | − 0.8 | (− 4.4; 2.6) | − 11.5 | (− 16; − 2.5) | − 8.3 | (− 14.2; 1.6) | − 6.9 | (− 13.7; − 1.0) |
| Age | − 0.0 | (− 0.6; 0.5) | − 0.1 | (− 1.2; 1.2) | − 0.2 | (− 0.7; 0.4) | − 2.0 | (− 3.2; − 1.1) | − 2.7 | (− 3.7; − 1.1) | − 0.1 | (− 1.2; 1.9) |
| Disease duration | − 0.2 | (− 0.6; 0.3) | − 0.1 | (− 1.6; 0.3) | − 0.5 | (− 1.1; − 0.1) | − 0.9 | (− 1.2; 0.4) | − 1.1 | (− 2.1; 0.4) | − 1.0 | (− 1.5; − 0.2) |
| Z-score BMI | 1.5 | (0.1; 2.8) | 1.5 | (− 3.2; 4.5) | − 0.6 | (− 1.8; 1.5) | 0.0 | (− 2.4; 1.5) | − 0.3 | (− 4.8; 1.3) | 1.4 | (− 2.6; 2.3) |
| PGA (active disease vs Remission) | − 10.2 | (− 19.3; − 5.5) | − 20.6 | (− 22.6; 1.8) | − 4.0 | (− 5.9; 3.5) | − 8.0 | (− 22.3; 17.1) | − 6.8 | (− 21.3; 3.1) | − 22.7 | (− 28.3; − 0.3) |
| PUCAI/w-PCDAI (Active disease vs Remission) | − 8.6 | (− 10.6; 3.5) | 10.6 | (− 0.6; 14.4) | 0.2 | (− 7.0; 4.7) | − 7.7 | (− 30.9; 8.7) | − 3.2 | (− 17.0; 12) | 11.7 | (− 7.0; 16.0) |
| Type of disease (CD vs UC) | 5.4 | (1.9; 9.3) | 2.9 | (− 5.7; 7.3) | 3.1 | (− 1.9; 5.7) | 4.8 | (− 0.8; 15.4) | − 8.4 | (− 15.2; 2.1) | − 1.9 | (− 6.5; 5.7) |
| Relapses (yes vs no) | − 3.7 | (− 10.1; − 0.1) | − 9.9 | (− 22.1; − 6.1) | − 0.3 | (− 5.8; 2.3) | − 5.4 | (− 15.5; − 0.3) | 4.3 | (− 5.0; 11) | − 7.4 | (− 16.5; 0.3) |
| Surgery (yes vs no) | 0.6 | (− 2.3; 2.9) | 3.7 | (− 2.2; 5.0) | − 1.3 | (− 3.3; 2.3) | − 4.6 | (− 6.3; 2.6) | 5.2 | (− 0.9; 10.6) | 4.6 | (− 1.5; 8.8) |
Results of quantile regression analysis.
b quantile regression coefficient estimate, 95%CI 95% confidence interval. Significant coefficients are marked in bold.
Both gender (females reporting 12 points less than males) and relapses had negative significant association with the Emotional Functioning domain.
Social Functioning domain was significantly associated with disease duration, a longer history of disease being associated to a worse social functioning.
Female gender, older age, longer disease duration and UC diagnosis were negatively associated with the QoL related to systemic symptoms.
Body Image score significantly decreased with increasing age (in median of three point for each year of age).
Treatment/Intervention score was significantly lower in females than males. The domain score significantly decreased with longer disease duration and in presence of active disease, as evaluated by clinicians (PGA).
Figure 2 shows the effect of each variable on the IMPACT-III total score. Gender had a significant negative effect in the first 7 deciles of the distribution, while age was significantly associated with the total score in the first decile (subjects reporting the lowest QoL). Active disease, measured by PGA index, decreased the quality of life in the last three deciles of the score distribution (subjects with the highest QoL).
Figure 2.
Effects of different variables on the IMPACT-III total score. The x-axis indicates the 9 deciles of the response distribution (score) and y-axis exhibits the effect of each variable (regression coefficients) and 95% confidence bands (grey area). If the confidence interval includes 0, the estimates cannot be considered statistically different from 0.
Discussion
QoL is an important multidimensional issue in IBD care that needs to be addressed by the healthcare professionals involved in management of IBD. The knowledge of specific factors affecting or associated with QoL in a given cultural setting allows the prompt recognition of patients at risk for a low QoL and finally helps to improve the quality of care. This is the first multicenter study to assess factors associated with the HRQoL in a large cohort of Italian children and adolescents with IBD.
One of the major findings of our study is the negative association of female gender with most of the domains scores (Bowel Symptoms, Emotional Functioning, Systemic Symptoms and Treatment/Intervention) and with the total IMPACT-III score. While a similar effect has been well described in adult studies19–21, pediatric data are less consistent22,23. Previous studies reported a lower score in the “body image domain” for females which might be correlated with impaired QOL, a finding that is not confirmed by our study. It is possible that fears of body change and attractiveness is much more relevant in women compared to young females, while possible explanations of the reduced QoL in younger girls include a different perception of illness compared to boys, a greater concern on having symptoms or relapse or on taking long term medications. Furthermore, in comparison to men, women with IBD are more exposed to the risk of psychological disturbances (particularly anxiety)24–26 and social difficulties (e.g. in work productivity)27,28 and this can at least partially explain their reduced QoL as adults.
Another vulnerable category of patients are adolescents, as shown by the lower QoL reported by teens compared to younger children. Specifically, age was associated with a reduced scores in the Systemic Symptoms (2 points for each year) and the Body Image domains (2.7 points for each year), in line with previous studies22,29. The latter finding can be associated to a different perception and fear of body changes (related to cosmetic effects of drugs or to the diseases itself) in adolescents, with a reflection on QoL. Older children can sometimes deny or under report symptoms to minimize their disease and this can lead to an under estimation of disease activity (based on clinical scores) and to a delay in treatment intensification, with a subsequent disease flare. Both the twisted coping strategy and the worsening symptoms can ultimately affect their IBD related QoL. However, we found the effect of age on the total QoL score limited to subjects with the lowest QoL (first decile). This suggest that it is a specific subgroup of adolescents, where other factors contribute, to be particularly vulnerable to a reduced QoL. Our data warn about an early impairment of the QoL of girls and teenagers with IBD compared to boys, independent from other factors (such as social or work factors), and suggest the need of a careful monitoring of the QoL especially in female patients.
Our large multicenter experience does not support a significant association between the duration of disease and the total IMPACT-III score, therefore it seems appropriate to attribute the impaired QoL of older children to the vulnerable phase of development they are facing, rather than to the effect of the longer disease duration. It’s well described also in other chronic conditions (e.g. celiac disease)30 that accepting and coping with a chronic and social limiting disease can be particularly challenging for adolescents, with a negative impact on the QoL. However, the interpretation of these findings should also consider the cross-sectional design of our study that did not allow a longitudinal evaluation of possible temporal changes in QoL.
We did not find any significant difference in the QoL between CD and UC patients, except in the Bowel Symptoms domain score, which was significantly higher in CD subjects. This is at variance with reports in adults, that generally indicate a lower QoL in patients with CD compared to UC31. Pediatric data from Swiss, Asian and Greek cohorts do not confirm this finding29,32,33, e.g. the only difference in the Greek study was a higher emotional QoL in subjects with CD compared to UC. Inconsistencies between pediatric and adult data could be associated with the age-related increase of disease-complications and surgery (generally more frequent in patients with CD). It is also important to notice that in our study children with UC experienced a higher number of relapses in the previous year, perhaps correlating with their worse HRQoL on the Bowel Symptoms. Furthermore females were less represented in our CD group and, considering the strong association between female gender and a worse QoL, this finding should at least partially explains the comparable QoL recorded in all the other domains between UC and CD patients.
Finally, our data confirm that disease activity significantly influences the patient-reported QoL, as shown by several findings: (a) the univariate analysis shows that patients in remission reported higher total and subdomains scores, (b) in the quantile analysis, remission was associated to better QoL related to bowel symptoms and treatment/interventions and to a higher total QoL score. Furthermore, experience of relapses in the previous year significantly affected the Bowel Symptoms, the Emotional Functioning and the Systemic Symptoms QoL. Several previous studies indicated disease activity as a major factor associated with low QoL both in children6,8 and in adults; adult studies have shown the negative effect of disease activity on QoL, irrespective of the type of questionnaire (generic or disease specific)31. Controlling disease activity is one of the main objectives in treating IBD patients and this leads not only to remission of symptoms and achievement of mucosal healing, but also to improvement of psycho-social aspects and eventually of health-related QoL. Nevertheless it is important to notice how, in our experience, some domain scores were not associated to measures of disease activity (e.g. Emotional and Social Functioning), indicating that some aspects of QoL need to be specifically investigated and addressed by physician, regardless the control of symptoms and disease activity. Administration of a questionnaire investigating QoL and collection of the results prior to a hospital visit could be a strategy to focus on specific aspects of QoL, frequently underinvestigated during a medical interview, and to support the patient with different health professionals, such as a psychologist or a social assistant.
In line with previous pediatric studies7–10, our results confirm the good internal consistency of all the different domains of the IMPACT-III questionnaire and an appropriate concurrent validity (as shown by a comparison with the generic PEDsQL questionnaire). Furthermore, the test demonstrated good reproducibility and high discriminant validity, as shown by the capacity of separating patients according to disease’s activity (Fig. 1). Lastly, the questionnaire was easy to understand and appropriately completed by children and adolescents, as demonstrated by the low number of missing responses. Our findings definitively demonstrate, in a large multicenter setting, that the Italian version of the IMPACT-III questionnaire is a reliable instrument to assess QoL in IBD pediatric patients, supporting the use of this tool for clinical and research purposes.
Strengths of our study are the multi-center involvement and the large size of the sample, compared to previous reports. We were able to test the validity of the questionnaire with different approaches, including the test–retest reliability, by using an adequate period of reassessment. We are also aware that the cross-sectional design of our study limits the identification of longitudinal factors impacting QoL, nevertheless it allows to estimate factors associated with QoL.
Conclusion
This study confirms the validity of the IMPACT III questionnaire in assessing QoL in a large sample of Italian children with IBD. We identified some characteristics potentially associated to QoL, suggesting the need of a special attention to a more vulnerable subgroup of IBD children (females, older children, UC subgroup and particularly patients with active disease and growth impairment).
Supplementary Information
Author contributions
S.G., G.D.B. and C.C. designed the study. S.G., G.D.B., G.C., M.A., M.B., G.D.A., E.F., M.F., S.L., M.P., R.P. and F.P. enrolled and followed up patients and collected data. A.F. and R.G. analyzed data and performed the statistical analysis. S.G., A.F., R.G. and C.C. interpreted data. S.G., A.F. and R.G. drafted the manuscript. M.B. and C.C. reviewed the manuscript. All the authors approved the final version of the article.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-97661-1.
References
- 1.Sýkora J, Pomahačová R, Kreslová M, Cvalínová D, Štych P, Schwarz J. Current global trends in the incidence of pediatric-onset inflammatory bowel disease. World J. Gastroenterol. 2018;24(25):2741–2763. doi: 10.3748/wjg.v24.i25.2741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Knowles SR, Graff LA, Wilding H, Hewitt C, Keefer L, Mikocka-Walus A. Quality of life in inflammatory bowel disease: A systematic review and meta-analyses—part I. Inflamm. Bowel. Dis. 2018;24(4):742–751. doi: 10.1093/ibd/izx100. [DOI] [PubMed] [Google Scholar]
- 3.Peyrin-Biroulet L, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): Determining therapeutic goals for treat-to-target. Am. J. Gastroenterol. 2015;110:1324–1338. doi: 10.1038/ajg.2015.233. [DOI] [PubMed] [Google Scholar]
- 4.Ryan JL, et al. The clinical utility of health-related quality of life screening in a pediatric inflammatory bowel disease clinic. Inflamm. Bowel. Dis. 2013;19:2666–2672. doi: 10.1097/MIB.0b013e3182a82b15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.El-Matary W. Patient-reported outcome measures in inflammatory bowel disease. Can. J. Gastroenterol. Hepatol. 2014;28:536–542. doi: 10.1155/2014/792386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Otley A, et al. The IMPACT questionnaire: A valid measure of health related quality of life in pediatric inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2002;35:557–563. doi: 10.1097/00005176-200210000-00018. [DOI] [PubMed] [Google Scholar]
- 7.Otley AR, et al. IMPACT-III is a valid, reliable and responsive measure of health-related quality of life in pediatric Crohn’s disease. J. Pediatr. Gastroenterol. Nutr. 2006;43(suppl 2):S49. doi: 10.1097/01.mpg.0000256260.38116.4a. [DOI] [Google Scholar]
- 8.Abdovic S, et al. The IMPACT-III (HR) questionnaire: A valid measure of health-related quality of life in Croatian children with inflammatory bowel disease. J. Crohns Colitis. 2013;7:908–915. doi: 10.1016/j.crohns.2012.12.010. [DOI] [PubMed] [Google Scholar]
- 9.Ogden CA, Akobeng AK, Abbott J, Aggett P, Sood MR, Thomas AG. Validation of an instrument to measure quality of life in British children with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2011;53:280–286. doi: 10.1097/MPG.0b013e3182165d10. [DOI] [PubMed] [Google Scholar]
- 10.Wernera H. Validation of the IMPACT-III quality of life questionnaire in Swiss children with inflammatory bowel disease. J. Crohns Colitis. 2014;8:641–648. doi: 10.1016/j.crohns.2013.11.025. [DOI] [PubMed] [Google Scholar]
- 11.Levine A, et al. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2014;58:795–806. doi: 10.1097/MPG.0000000000000239. [DOI] [PubMed] [Google Scholar]
- 12.Turner D, et al. Mathematical weighting of the pediatric crohn's disease activity index (PCDAI) and comparison with its other short versions. Inflamm. Bowel. Dis. 2012;18(1):55–62. doi: 10.1002/ibd.21649. [DOI] [PubMed] [Google Scholar]
- 13.Turner D, et al. Development and evaluation of a pediatric ulcerative colitis activity index (PUCAI): A prospective multicenter study. Gastroenterology. 2007;133:423–432. doi: 10.1053/j.gastro.2007.05.029. [DOI] [PubMed] [Google Scholar]
- 14.Samson CM, et al. Improved outcomes with quality improvement interventions in pediatric inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2012;55:679–688. doi: 10.1097/MPG.0b013e318262de16. [DOI] [PubMed] [Google Scholar]
- 15.Varni JW, Seid M, Kurtin PS. PedsQL 4.0: Reliability and validity of the PediatricQuality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med. Care. 2001;39:800–812. doi: 10.1097/00005650-200108000-00006. [DOI] [PubMed] [Google Scholar]
- 16.Beaton DE, Bombardier C, Guillemin F, Ferraz MB. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine. 2000;25:3186–3191. doi: 10.1097/00007632-200012150-00014. [DOI] [PubMed] [Google Scholar]
- 17.Otley, A. IMPACT-III: User's Guide (2005).
- 18.Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45(1):255–268. doi: 10.2307/2532051. [DOI] [PubMed] [Google Scholar]
- 19.Bernklev T, et al. Health-related quality of life in patients with inflammatory bowel disease five years after the initial diagnosis. Scand. J. Gastroenterol. 2004;39:365–373. doi: 10.1080/00365520310008386. [DOI] [PubMed] [Google Scholar]
- 20.Haapamaki J, Turunen U, Roine RP, Färkkilä MA, Arkkila PET. Impact of demographic factors, medication and symptoms on disease-specific quality of life in inflammatory bowel disease. Qual. Life Res. 2009;18:961–969. doi: 10.1007/s11136-009-9514-y. [DOI] [PubMed] [Google Scholar]
- 21.Casellas F, Lopez-Vivancos J, Casado A, Malagelada JR. Factors affecting health related quality of life of patients with inflammatory bowel disease. Qual. Life Res. 2002;11:775–781. doi: 10.1023/A:1020841601110. [DOI] [PubMed] [Google Scholar]
- 22.Hill RJ, et al. Quality of life in children with Crohn’s disease. J. Pediatr. Gastroenterol. Nutr. 2010;51:35–40. doi: 10.1097/MPG.0b013e3181c2c0ef. [DOI] [PubMed] [Google Scholar]
- 23.Kim S, et al. Can we estimate quality of life in pediatric inflammatory bowel disease patients? An Asian multicenter study. J. Pediatr. Gastroenterol. Nutr. 2019;68:45–49. doi: 10.1097/MPG.0000000000002119. [DOI] [PubMed] [Google Scholar]
- 24.Byrne G, Rosenfeld G, Leung Y, Qian H, Raudzus J, Nunez C, Bressler B. Prevalence of anxiety and depression in patients with inflammatory bowel disease. Can. J. Gastroenterol. Hepatol. 2017;2017:6496727. doi: 10.1155/2017/6496727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fuller-Thomson E, Lateef R, Sulman J. Robust association between inflammatory bowel disease and generalized anxiety disorder: Findings from a Nationally Representative Canadian Study. Inflamm. Bowel. Dis. 2015;21:2341–2348. doi: 10.1097/MIB.0000000000000551. [DOI] [PubMed] [Google Scholar]
- 26.Nahon S, et al. Risk factors of anxiety and depression in inflammatory bowel disease. Inflamm Bowel. Dis. 2012;18(11):2086–2091. doi: 10.1002/ibd.22888. [DOI] [PubMed] [Google Scholar]
- 27.Parra RS, et al. Quality of life, work productivity impairment and healthcare resources in inflammatory bowel diseases in Brazil. World. J. Gastroenterol. 2019;25(38):5862–5882. doi: 10.3748/wjg.v25.i38.5862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chhibba T, et al. Workplace accommodation for persons with IBD: What is needed and what is accessed. Clin. Gastroenterol. Hepatol. 2017;15(10):1589–1595. doi: 10.1016/j.cgh.2017.05.046. [DOI] [PubMed] [Google Scholar]
- 29.Loonen HJ, Grootenhuis MA, Last BF, Koopman HM, Derkx HHF. Quality of life in paediatric inflammatory bowel disease measured by a generic and a disease-specific questionnaire. Acta Paediatr. 2002;91:348–354. doi: 10.1111/j.1651-2227.2002.tb01727.x. [DOI] [PubMed] [Google Scholar]
- 30.Biagetti C, Gesuita R, Gatti S, Catassi C. Quality of life in children with celiac disease: A paediatric cross-sectional study. Dig. Liver Dis. 2015;47(11):927–932. doi: 10.1016/j.dld.2015.07.009. [DOI] [PubMed] [Google Scholar]
- 31.Knowles SR, Keefer L, Wilding H, Hewitt C, Graff LA, Mikocka-Walus A. Quality of life in inflammatory bowel disease: A systematic review and meta-analyses—part II. Inflamm. Bowel. Dis. 2018;24(5):966–976. doi: 10.1093/ibd/izy015. [DOI] [PubMed] [Google Scholar]
- 32.Rogler D, et al. Coping is excellent in Swiss children with inflammatory bowel disease: results from the Swiss IBD cohort study. J. Crohns Colitis. 2014;8:409–420. doi: 10.1016/j.crohns.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 33.Chouliaras G, Margoni D, Dimakou K, Fessatou S, Panayiotou I, Roma-Giannikou E. Disease impact on the quality of life of children with inflammatory bowel disease. World. J. Gastroenterol. 2017;23(6):1067–1075. doi: 10.3748/wjg.v23.i6.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.