Abstract
Cancer immunotherapy, especially immune checkpoint blockade (ICB), has revolutionized oncology. However, only a limited number of patients benefit from immunotherapy, and some cancers that initially respond to immunotherapy can ultimately relapse and progress. Thus, some studies have investigated combining immunotherapy with other therapies to overcome resistance to monotherapy. Recently, multiple preclinical and clinical studies have shown that tumor vasculature is a determinant of whether immunotherapy will elicit an antitumor response; thus, vascular targeting may be a promising strategy to improve cancer immunotherapy outcomes. A successful antitumor immune response requires an intact “Cancer‐Immunity Cycle,” including T cell priming and activation, immune cell recruitment, and recognition and killing of cancer cells. Angiogenic inducers, especially vascular endothelial growth factor (VEGF), can interfere with activation, infiltration, and function of T cells, thus breaking the “Cancer‐Immunity Cycle.” Together with immunostimulation‐regulated tumor vessel remodeling, VEGF‐mediated immunosuppression provides a solid therapeutic rationale for combining immunotherapy with antiangiogenic agents to treat solid tumors. Following the successes of recent landmark phase III clinical trials, therapies combining immune checkpoint inhibitors (ICIs) with antiangiogenic agents have become first‐line treatments for multiple solid tumors, whereas the efficacy of such combinations in other solid tumors remains to be validated in ongoing studies. In this review, we discussed synergies between antiangiogenic agents and cancer immunotherapy based on results from preclinical and translational studies. Then, we discussed recent progress in randomized clinical trials. ICI‐containing combinations were the focus of this review because of their recent successes, but combinations containing other immunotherapies were also discussed. Finally, we attempted to define critical challenges in combining ICIs with antiangiogenic agents to promote coordination and stimulate collaboration within the research community.
Keywords: antiangiogenesis, bevacizumab, cancer, combination therapy, immune‐checkpoint inhibitor, immunotherapy, vascular endothelial growth factor
In this review, mechanisms of VEGF‐induced immunosuppression based on results from preclinical and translational studies and recent progress in randomized clinical trials were discussed. Key challenges associated with dual targeting of VEGF and PD‐1/PD‐L1 pathways were also defined in order to stimulate collaboration and help galvanize a broader effort.
Abbreviations
- PD‐1
programmed cell death protein 1
- PD‐L1
programmed death ligand 1
- NK
natural killer
- DC
dendritic cell
- TME
tumor microenvironment
- MDSC
myeloid‐derived suppressive cell
- ICI
immune‐checkpoint inhibitor
- FDA
the United States Food and Drug Administration
- VEGF
vascular endothelial growth factor
- VEGFR1
vascular endothelial growth factor receptor 1
- VEGFR2
vascular endothelial growth factor receptor 2
- CRC
colorectal cancer
- TKI
tyrosine kinase inhibitor
- MHC
major histocompatibility complex
- NF‐κB
nuclear factor‐κB
- Treg
regulatory T cell
- CXCL10
CXC‐chemokine ligand 10
- CXCL11
CXC‐chemokine ligand 11
- ICAM1
intracellular adhesion molecule 1
- VCAM1
vascular adhesion molecule 1
- TNF‐α
tumor necrosis factor α
- FASL
FAS antigen ligand
- CTL
cytotoxic T lymphocyte
- CTLA‐4
cytotoxic T‐lymphocyte‐associated protein 4
- LAG3
lymphocyte activation gene 3 protein
- TIM3
T cell immunoglobulin mucin receptor 3
- RCC
renal cell carcinoma
- STAT3
signal transducer and activator of transcription 3
- Th1
T helper‐1
- interferon‐γ
IFNγ
- HCC
hepatocellular carcinoma
- uHCC
unresectable hepatocellular carcinoma
- mPFS
median progression‐free survival
- mOS
median overall survival
- OS
overall survival
- ORR
objective response rate
- ASCO
American Society of Clinical Oncology
- NSCLC
non‐small cell lung cancer
- EGFR
epidermal growth factor receptor
- ALK
anaplastic lymphoma kinase
- PFS
progression‐free survival
- HR
hazard ratio
- MSI‐H
microsatellite instability high
- dMMR
DNA mismatch repair pathway deficient
- ABC
atezolizumab plus bevacizumab plus capecitabine
- ICB
immune checkpoint blockade
- TMB
tumor mutation burden
1. INTRODUCTION
1.1. Immune checkpoint inhibition
Immune checkpoints are evolutionarily conserved molecules including, but not limited to, programmed cell death protein 1 (PD‐1) and programmed death ligand 1 (PD‐L1), which was the first ligand of PD‐1 discovered. They are well‐established inhibitory regulators of antitumor immunity and play essential roles in fine‐tuning the immune response. PD‐1 is widely expressed on many immune cells, including peripherally activated T‐lymphocytes, natural killer (NK) cells, B‐lymphocytes, monocytes, and specific dendritic cells (DCs) [1]. In addition to tumor cells, PD‐L1 is also expressed on many types of cells in the tumor microenvironment (TME), such as DCs, macrophages, myeloid‐derived suppressive cells (MDSCs), T cells, endothelial cells, and fibroblasts [2]. The binding of the PD‐1/PD‐L1 complex inhibits the activation of immune cells via intracellular signaling pathways, resulting in a reduction of the secretion of antibodies and cytokines by immune cells, exhaustion of the T‐lymphocytes, and promotion of their apoptosis, which are responsible for cancer immune escape [1]. Immune‐checkpoint inhibitors (ICIs) are designed to block immune checkpoints to “unleash” strong T cell antitumor responses. Over the last decade, the usage of ICIs such as nivolumab and pembrolizumab (both are anti‐PD‐1 monoclonal antibodies) has revolutionized the treatment of multiple solid tumors by prolonging the survival of patients with recalcitrant tumors [3]. To date, 10 ICIs targeting the PD‐1/PD‐L1 signaling pathway have received approval from the United States Food and Drug Administration (FDA) for 19 different types of cancer, including a tissue‐agnostic indication [4]. The success of cancer immunotherapy is also reflected by the fact that the immuno‐oncology field is still growing despite coronavirus disease 2019 [5, 6].
Despite the unprecedented durable response rates observed with ICIs, primary resistance (cancer not responding to ICI) and acquired resistance (cancer initially responded to ICI but ultimately relapsed and progressed) prevent the majority of patients from benefiting from the treatment [7, 8]. According to an investigation, up to 87% of eligible patients have been estimated to not respond to FDA‐approved ICIs [9]. In several clinical trials, some common cancer types have shown a low frequency of response to ICIs (advanced breast, prostate, and liver cancers) [10, 11, 12, 13, 14, 15]. A critical cause for ICI resistance is that tumors manipulate alternative immunosuppressive mechanisms and thereby evade immune clearance [7]. To overcome monotherapy resistance, some researchers have investigated combining immunotherapy with other therapies. The shift from monotherapy to combination therapy has been significant, as indicated by 80% of 3674 active clinical trials testing combination regimens on evaluating inhibitors of the PD‐1/PD‐L1 signaling pathway [16, 17].
1.2. Angiogenesis and anti‐angiogenesis
Angiogenesis, the formation of new blood vessels from preexisting vessels, is critical for embryonic and adult development [18]. The identification of vascular endothelial growth factor (VEGF) in 1989 laid the foundations for understanding angiogenic mechanisms [19, 20]. VEGF, VEGFB, VEGFC, VEGFD, and placental growth factor belong to the VEGF family of growth factors. Among these proteins, VEGF is the master regulator of both normal and pathological angiogenesis and is produced by most cancer cells [21, 22]. VEGF binds to two complementary receptors, VEGF receptor 1 (VEGFR1) and VEGF receptor 2 (VEGFR2), of which the latter one is the primary regulator of angiogenesis and vessel permeability [23].
In 1971, Judah Folkman [24] proposed the concept of “anti‐angiogenesis,” i.e., preventing angiogenesis for clinical benefits. Bevacizumab, an anti‐VEGF monoclonal antibody, received FDA approval for the treatment of colorectal cancer (CRC) in 2004 following the success of a landmark phase III clinical trial [25]. The FDA has approved more than ten agents targeting the VEGF/VEGFR axis for treating a spectrum of cancers (Figure 1). These agents can be classified into two broad categories: protein inhibitors and multi‐targeted receptor tyrosine kinase inhibitors (TKIs). While effective antiangiogenic treatments have well‐established efficacies in halting tumor growth, under certain conditions, they cannot eradicate tumors via monotherapy due to compensatory mechanisms [18]. Hence, combining antiangiogenic agents with other therapeutic strategies might be necessary for effective tumor eradication [26, 27, 28, 29, 30, 31]; among these strategies, targeting the VEGF/VEGFR axis has been the most common combination treatment modality in 2020 [16].
FIGURE 1.
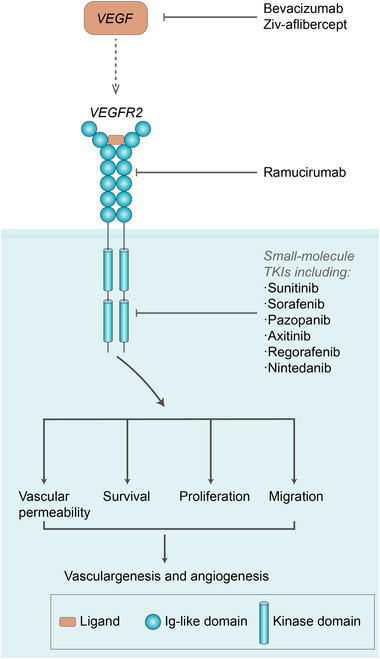
Protein inhibitors and multitargeted receptor tyrosine kinase inhibitors (TKIs) of the vascular endothelial growth factor (VEGF) pathway.
In this review, we discuss the mechanisms underpinning VEGF‐mediated immunosuppression because VEGF has been the focus of angiogenesis research and because the roles of other angiogenic factors, such as angiopoietins, on immune cells are less well understood. We provided an overview of results from recent randomized clinical trials combining ICIs with antiangiogenic agents. Further, despite the remarkable success achieved by the dual inhibition of the VEGF/VEGFR and PD‐1/PD‐L1 axes, we also discussed the remaining key challenges in combining ICIs with antiangiogenic agents.
2. ANTIANGIOGENIC TREATMENT HELPS TO OVERCOME RESISTANCE TO CANCER IMMUNOTHERAPY
A spectrum of step‐by‐step procedures must be initiated and iteratively expanded for a successful anticancer immune response to eradicate malignant cells. These steps are characterized by Daniel S Chen and Ira Mellman [32] as the “Cancer‐Immunity Cycle.” This seven‐step, self‐propagating cycle can be divided into three broad phases:
Tumor antigen presentation and T cell activation;
Trafficking and infiltration of immune effector cells; and
Recognition and killing of tumor cells.
As discussed later, by binding to receptors on the endothelium and immune cells, VEGF can interfere with the initiation and propagation of the “Cancer‐Immunity Cycle” and, thus, impair anticancer immunity (Figure 2). At the same time, agents targeting the VEGF/VEGFR axis can help build an immune‐supportive TME and increase antitumor immunity (Table 1). Therefore, antiangiogenic treatments could be investigated to improve immunotherapy outcomes.
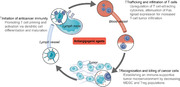
FIGURE 2.
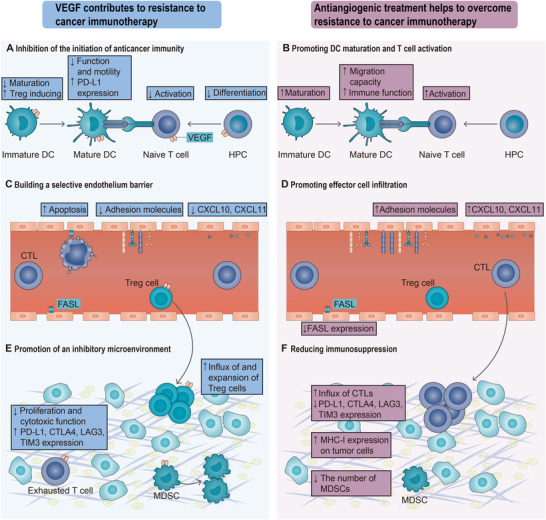
Antiangiogenic treatment helps to overcome resistance to cancer immunotherapy. By binding to receptors on multiple immune cells and endothelial cells, VEGF interferes with the whole cancer immunity cycle—from initiation of anticancer immunity to recruitment of T cells to recognition and killing of cancer cells. VEGF limits the supply of mature DCs and naïve T cells in lymph organs because they can inhibit the maturation and differentiation of their progenitor cells, respectively. In tumor vessels, VEGF induces apoptosis of CTLs and makes the endothelium favor tumor homing of immunosuppressive cells over CTLs. In the tumor microenvironment, VEGF promotes the proliferation and function of immunosuppressive cells and dampens cytotoxic function of CTLs by promoting the expression of inhibitory molecules. Abbreviations are as follows: CTL, cytotoxic T lymphocyte; CTLA‐4, cytotoxic T‐lymphocyte‐associated protein 4; CXCL, CXC‐chemokine ligand; DC, dendritic cells; FASL, FAS antigen ligand; MHC; major histocompatibility complex; HPC, hematopoietic progenitor cell; LAG3, lymphocyte activation gene 3 protein; MDSC, myeloid‐derived suppressive cells; PD‐L1, programmed death‐ligand 1; TIM3, T cell immunoglobulin mucin receptor 3; TME, tumor microenvironment; Treg cell, regulatory T cell; VEGF, vascular endothelial growth factor
TABLE 1.
Immunomodulatory effects of antiangiogenic treatments in preclinical and translational studies
| Antiangiogenic treatment | Cancer type | Key result | Reference |
|---|---|---|---|
| VEGF studies | |||
| Bevacizumab or sorafenib | NA |
Dendritic cell differentiation↑ HLA‐DR and CD86 expression↑ |
[31] |
| Bevacizumab | Renal cell carcinoma (Caki‐1) in nude mice | Circulating VEGFR1+ myeloid cells↓ | [67] |
| Bevacizumab | Multiple tumors in human patients |
Immature dendritic cells in peripheral blood↓ Dendritic cell population↑ Allostimulatory capacity of dendritic cells↑ T cell proliferation↑ |
[44] |
| VEGF‐specific antibody (clone G6‐31) + aspirin |
Ovarian cancer (ID8‐VEGF) Colon cancer (CT26) Renal cell cancer (Renca) Melanoma (B16) |
Influx of tumor‐rejecting CD8+ over FoxP3+ T cells↑ CD8‐dependent tumor growth suppression↑ |
[55] |
| Angiopoietin studies | |||
| AMG386 (Trebananib, an Ang‐1/2 neutralizing peptibody) | Glioblastoma (GL261) |
Number of F4/80+ macrophages↑ Microvessel coverage with desmin+ pericytes↑ |
[26] |
| MEDI3617 (an anti‐Ang‐2‐neutralizing antibody) + Cediranib | Glioblastoma (Gl261) | Reprogramming TAMs towards the M1 polarized subtype | [27] |
| A2V (Ang‐2/VEGF bispecific antibody) | Glioblastoma (GL261) | Reprogramming TAMs towards the M1 polarized subtype | [28] |
| A2V (Ang‐2/VEGF bispecific antibody) | Melanoma (B16‐OVA) |
Proportions of CTLs expressing an IFNγ+ or CD69+ phenotype↑ Proportion of intratumoral, OVA‐specific CTLs↑ Proportion of APCs that cross‐presented the OVA‐derived peptide↑ Phagocytic activity of APCs↑ |
[29] |
| AMG386 + MET kinase inhibitor | Clear cell renal cell carcinoma (RP‐R‐02LM) | The presence of TAMs in the tumor microenvironment↓ | [30] |
Abbreviations: Ang, angiopoietin; APC, antigen‐presenting cell; CTL, cytotoxic T lymphocyte; NA, not applicable; TAM, tumor‐associated macrophage; IFN, interferon; VEGF, vascular endothelial growth factor.
2.1. Antiangiogenic treatment promotes DC maturation and T cell activation
A robust anticancer immune response is reliant on effective cancer neoantigen presentation to prime and activate naïve T cells. The absence of cancer neoantigen presentation and the absence of tumor‐specific T cells are major factors contributing to resistance to cancer immunotherapy [7]. Successful priming and activation of T cells are affected by two independent factors: the function of antigen‐presenting cells and the availability of naïve T cells with tumor antigen‐specific T cell receptors.
DCs are the most effective antigen‐presenting cells and make a significant contribution to the initiation and amplification of antitumor immunity [33]. DCs can be divided into two functional states, “mature” and “immature,” mainly according to their ability to activate effector T cell responses against neoantigens in secondary lymph organs [33]. Upon immature DCs capture antigens in TME, they travel to tumor‐draining lymph nodes and present captured antigens on major histocompatibility complex (MHC) class I molecules to CD8+ T cells, leading to the priming and activation of antigen‐specific naïve T cells [34]. At the same time, immature DCs also gradually become mature, which is characterized by upregulated expression of co‐stimulatory molecules and secretion of proinflammatory cytokines [33]. One of the first reported immunosuppressive functions of antiangiogenic factors is that tumor‐derived VEGF inhibits the maturation of DCs from progenitor cells, which leads to reduced presentation of cancer neoantigens in tumor‐draining lymph nodes and thus contributes to immune evasion by tumors [35]. The mechanism of DC dysfunction involves VEGF‐induced inhibition of the nuclear factor‐κB (NF‐κB) signaling pathway in progenitor cells by limiting degradation of an NF‐κB inhibitor [36, 37]. Compared with mature DCs, immature DCs are more powerful Foxp3+ regulatory T cell (Treg) inducers [38]. VEGF has been recently reported to impair the immune function and migration capability of mature DCs through the VEGFR2‐RhoA‐cofilin1 pathway [39]. Dysfunctional circulating DCs and decreased mature DC populations are related to the existence of enhanced concentrations of VEGF in a number of cancers, especially metastatic malignancies (Figure 2A) [40–42]. Moreover, VEGF has been reported to upregulate the expression of PD‐L1 on myeloid DCs, which could impair DC‐mediated T cell priming and proliferation [43]. In vitro studies demonstrated that the function of DCs differentiated in the presence of VEGF can be recovered upon inhibition of the VEGF axis by bevacizumab or sorafenib [31]. Bevacizumab administration in cancer patients decreased the accumulation of immature precursors of DCs and induced an increase in the number of circulating DCs compared with samples taken before treatment (Table 1) [44]. Recently, blockade of VEGF using aflibercept, an FDA‐approved VEGF inhibitor, was reported to result in a more mature DC phenotype in mice with glioblastomas, as evidenced by upregulated expression of the co‐stimulatory molecules [45]. Thus, antiangiogenic inhibitor administration increases functional DCs necessary for T cell priming and activation in lymph nodes (Figure 2B).
VEGF has been shown to directly interfere with T‐cell antitumor function by impairing the development of CD4 or CD8 single‐positive T cells from blood progenitor cells in the thymus of mouse models [46]. This likely reduces the number of naïve T cells available for priming and activation and contributes to cancer‐derived immune evasion.
2.2. Antiangiogenic treatment promotes effector cell infiltration
Immune cells require a normal and functional tumor blood vessel network to infiltrate tumors. Effective trafficking of tumor‐specific T cells to the tumor site and infiltration of them into the tumor bed are necessary for response to cancer immunotherapy [47]. Consequently, poor T‐cell infiltration into the tumor before treatment is usually correlated with resistance to cancer immunotherapy [48, 49]. The “angiogenic switch” is governed by countervailing angiogenic inducers and inhibitors, such as VEGF and thrombospondin‐1, respectively. The hypoxic microenvironment within solid tumors leads to continued production of angiogenic factors; as such, the “angiogenic switch” is continually activated to meet oxygen and nutrients demand [50]. As a result, newly produced blood vessels are immature and aberrant and impair T cell extravasation (Figure 3).
FIGURE 3.
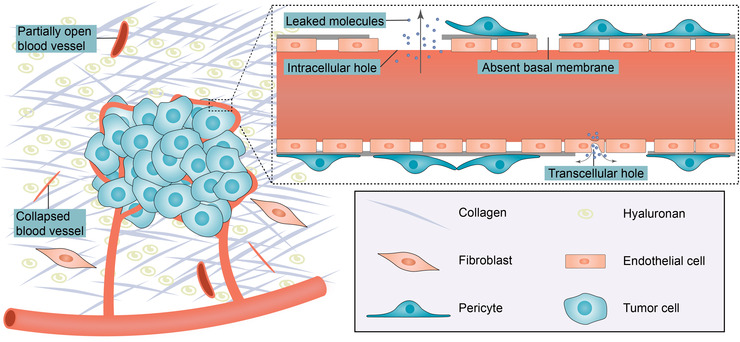
Leaky and compressed tumor blood vessels. Big transendothelial and interendothelial channels, discontinuous or absent basal membrane, and detached pericytes make tumor blood vessels hyperpermeable to circulating molecules. Stromal components such as fibroblasts, collagen and hyaluronan could compress blood vessels and make some vessels partially open or totally collapsed
One potential explanation for T cell exclusion is the downregulation of T cell‐attracting cytokines such as CXC‐chemokine ligand 10 (CXCL10) and CXC‐chemokine ligand 11 (CXCL11) secreted by endothelial cells (Figure 2C). VEGF has been reported to suppress T‐lymphocyte infiltration into solid tumors by limiting NF‐κB‐mediated endothelial activation and inhibiting VEGF signaling in a melanoma mouse model by sunitinib upregulated CXCL10 and CXCL11 expression in tumor vessels [51].
Another reason for the inability of effector cells to penetrate solid tumors could be the downregulation of adhesion molecules on dysfunctional tumor endothelium. T cell infiltration is dependent on adhesion molecules, such as intracellular adhesion molecule 1 (ICAM1), vascular adhesion molecule 1 (VCAM1), and CD34, expressed by the tumor endothelium. In vivo studies have shown that VEGF impairs leukocyte‐vessel wall interactions; this mechanism involves the VEGF‐induced downregulation of tumor necrosis factor α (TNF‐α)‐mediated expression of ICAM1 and VCAM1 on the endothelium [52]. Relatedly, the endothelial expression of CD34 is also downregulated by VEGF in human cancers [53]. In CRC and melanoma mouse models, treatment with antiangiogenic agents promoted the leucocyte‐vessel wall interaction and thus increased leukocyte infiltration by increasing the expression of adhesion molecules ICAM‐1 and VCAM‐1 (Figure 2D) [54].
VEGF has recently been reported to help build a selective wall on the tumor endothelium. Selective expression of FAS antigen ligand (FASL) has been detected on endothelial cells of human and murine tumors but not on healthy endothelium, which is induced by tumor‐derived VEGF, in combination with interleukin‐10 and prostaglandin E2 [55]. Endothelial FASL can cause apoptosis of tumor‐infiltrating cytotoxic T lymphocytes (CTLs), whereas Treg cells remain alive. Indeed, FASL expression has been linked with the “immune desert” (sparse CD8+ T cell presence) phenotype and the predominant existence of Treg cells [55]. This study demonstrated that the pharmacologic inhibition of VEGF plus aspirin (to prevent the expression of prostaglandin E2) produced a marked attenuation of FASL expression on the human microvascular endothelium, a significant increase in the influx of tumor‐killing CTLs over Tregs, and a marked enhancement of CTL‐dependent tumor cell cytotoxicity (Table 1).
In conclusion, VEGF‐induced dysfunctional tumor vasculature interferes with the trafficking and infiltration of T cells and represents a critical obstacle to cancer immunotherapies. Current insights into the relationship between cancer and host immune systems have already shown that adaptive immune cell infiltration has prognostic value; this lays the foundations for rationally‐guided classifications of patients and therapeutic strategies [56]. In fact, increasing the infiltration of T cells to convert an “immune desert” tumor to an inflamed tumor is the focus of active research. Not surprisingly, cancer immunotherapies have achieved limited benefits in tumors with low intratumoral microvascular density, such as pancreatic adenocarcinoma, which has a desmoplastic stroma with scarce T cell infiltration [57].
2.3. Antiangiogenic treatment reduces immunosuppression
For later steps (identification and killing of cancer cells) to take place, the balance between effector cells and immunosuppressive cells should be skewed toward an immune‐supportive TME. Immunosuppression in TME is a significant factor contributing to resistance to cancer immunotherapy, and multiple immunosuppression‐related mechanisms can inhibit the generation of anticancer response [7]. Tumor cell‐intrinsic mechanisms include (but are not limited to) alterations in antigen processing machinery and downregulation of the expression of MHC class I molecules. Tumor‐extrinsic mechanisms include (but are also not limited to) the expression of inhibitory immune checkpoints and the existence of immunosuppressive cells [7]. There are multiple studies supporting that VEGF skews the immune microenvironment toward an immunosuppressive one (Figure 2E).
Preclinical studies have shown that VEGF promotes CTL exhaustion by upregulating the production of multiple immune checkpoint molecules, including PD‐1, cytotoxic T‐lymphocyte‐associated protein 4 (CTLA‐4), lymphocyte activation gene 3 protein (LAG3), and T cell immunoglobulin mucin receptor 3 (TIM3) [58]. In other preclinical studies, VEGF has been reported to suppress the function of CTLs by inhibiting their proliferation and cytotoxic function [59]. In CRC mouse models, anti‐VEGF treatment reverted the expression of inhibitory molecules PD‐1, CTLA‐4, LAG3, and TIM3, which are related to T cell exhaustion [58]. In renal cell carcinoma (RCC) mouse models, bevacizumab monotherapy increased the number of intratumoral CTLs and upregulated the expression of MHC class I molecules on tumor cells (Figure 2F) [60].
In contrast to inhibiting effector T cell development, VEGF binding to VEGFR2 on Treg cells induces their proliferation [46, 61]. Indeed, in patients with CRC, VEGF binding to VEGFR2 has been associated with more Treg cells in the blood, and targeting the VEGF/VEGFR2 axis reduced peripheral Treg cell numbers [62]. Furthermore, ablation of the VEGFR2 effect on T cells strikingly inhibited the infiltration of Treg cells into solid tumors [63].
MDSCs are well‐established immunosuppressive cells that have been recently reported to paralyze cytotoxic T lymphocytes through the cell‐cell transfer of the metabolite methylglyoxal [64, 65]. VEGF promotes the expansion of MDSCs, and the mechanism involves the activation of signal transducer and activator of transcription 3 (STAT3) [66]. In agreement with this finding, preclinical studies have shown that bevacizumab reduced the number of MDSCs in an RCC mouse model [67]. In patients with RCC, neoadjuvant sunitinib increased tumor‑infiltrating lymphocytes, which is linked with a reduction in intratumoral MDSCs [68]. Another study reported that sunitinib reduced intratumoral MDSCs in an RCC mouse model and circulating MDSCs in patients with RCC [69].
3. SYNERGIES BETWEEN ANTIANGIOGENIC AGENTS AND CANCER IMMUNOTHERAPY
Aberrant tumor blood vessels reduce blood flow, compromise the delivery of therapeutic agents, worsen hypoxia, and interfere with the recruitment of immune cells, particularly CD8+ T cells, to solid tumors [70]. Therefore, improvement to tumor vascular function, i.e., an event termed “vascular normalization,” has the potential to enhance the delivery and efficacy of therapeutic agents and reverse immunosuppressive TME [70, 71]. Jain RK [72] proposed the concept of “vascular normalization” in 2001. Since then, a spectrum of preclinical and clinical studies reported that judicious use of antiangiogenic drugs can normalize tumor blood vessels and improve their function [73].
Although the vascular normalization response alleviates immunosuppression, its regulation is poorly understood. In 2017, Tian et al. [74] reported that ICIs can lead to remodeling of the tumor vasculature. In their study, PD‐1 and CTLA‐4 blockades improved vessel perfusion, reduced tumor vascular density, and alleviated hypoxia in the TME; these are hallmarks of tumor vessel normalization. Using genetically modified mice lacking CD4+ T cells (CD4–/–) or CD8+ T cells (CD8–/–), Tian et al. [74] showed that activated CD4+ T helper‐1 (Th1) cells, rather than CD8+ T cells, induced vascular normalization effects in breast cancer via secretion of interferon‐γ (IFNγ).
Alternatively, in breast cancer and CRC models, Zheng et al. [75] reported that CD4+ T cells were not enough to induce vessel normalization effects. In contrast, the ablation of CD4+ T cells led to the accumulation of CTLs, unregulated secretion of the proinflammatory cytokine IFNγ, and a vascular remodeling effect. Thus, the authors concluded that ICIs can stimulate CTLs to secrete IFNγ to remodel the tumor vasculature [75].
To date, the exact role of effector T cells in normalizing tumor vasculature in the setting of checkpoint blockade remains unknown. However, the studies discussed above strongly suggest that CD4+ and CD8+ T cells activated by ICIs can produce and secrete IFNγ, which interacts with IFNγ receptors on pericytes and endothelial cells, and ultimately normalizes tumor blood vessels. This intertwined relationship between vascular remodeling and immunostimulation provides a new rationale for combining vascular targeting therapy with immunotherapies.
A spectrum of preclinical studies evaluating combinations of cancer immunotherapies and antiangiogenic drugs showed promising results [58, 76‐78]. For example, in colon cancer mouse models, treatment with anti‐PD‐1 monoclonal antibody and sunitinib decreased the number of intratumoral PD‐1+CD8+ T cells. The efficacy of this combination was revealed by the fact that the anti–VEGF and anti–PD‐1 combination showed significant antitumor efficacy compared with anti‐VEGF monotherapy or anti‐PD‐1 monotherapy [58]. Basing on the mutual regulation between vascular normalization and cancer immunotherapy, we propose an immunostimulatory vascular‐modulating cycle to explain synergies between antiangiogenic agents and cancer immunotherapy (Figure 4). On the one hand, antiangiogenic agents promote vascular remodeling and relieve immunosuppression in the TME. Normalized blood vessels promote the infiltration of effector immune cells and improve their function, leading to the regression of tumors. On the other hand, immunotherapeutics activate effector T cells, which in turn promote the remodeling of the tumor vasculature system via IFNγ‐mediated vascular remodeling. This feedback loop between immunostimulation and remodeling of tumor blood vessels reinforces itself, ultimately leads to enhanced tumor regression.
FIGURE 4.
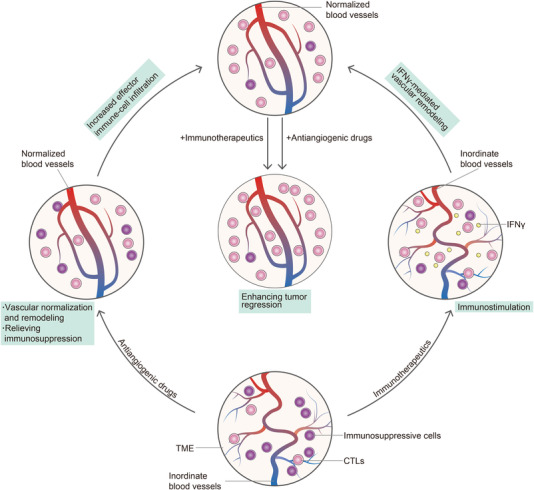
Synergies between antiangiogenic agents and cancer immunotherapy. Abbreviations are as follows: CTL, cytotoxic T lymphocyte; IFNγ, interferon‐γ; TME, tumor microenvironment
4. COMBINED THERAPY FROM BENCH TO BEDSIDE: CLINICAL PROGRESS
A spectrum of clinical trials evaluating ICI and antiangiogenic agent combined therapy was conducted in recent years because of overwhelming biological evidence (Table 2). In the first three quarters of 2020, up to 154 new trials testing the combination of PD1/PD‐L1 inhibitors and VEGF axis inhibitors were initiated [16].
TABLE 2.
Completed phase III clinical trials testing the combination of PD1/PD‐L1 inhibitors and VEGF axis inhibitors
| PFS | OS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Clinical trial | Cancer type | No. of patients | Primary endpoints | Treatment | Median PFS (months) | Comparison | Median OS (months) | Comparison | References |
| IMbrave150 | HCC | 501 | PFS and OS in ITT population | Atezolizumab + bevacizumab | 6.8 | HR = 0.59; P <0.001 | NE | HR = 0.58; P <0.001 | [81] |
| Sorafenib | 4.3 | 13.2 | |||||||
| IMpower150 | NSCLC | 1202 | PFS in both ITT‐WT and Teff‐high WT populations; OS in ITT‐WT population | Atezolizumab + bevacizumab + carboplatin + paclitaxel |
ITT‐WT: 8.3 Teff‐high WT: 11.3 |
ITT‐WT: HR = 0.62; P <0.001 Teff‐high WT: HR = 0.51; P <0.001 |
ITT‐WT: 19.2 | HR = 0.78; P = 0.02 | [90] |
| Bevacizumab + carboplatin + paclitaxel |
ITT‐WT: 6.8 Teff‐high WT: 6.8 |
ITT‐WT: 14.7 | |||||||
| IMmotion151 | RCC | 915 | PFS in PD‐L1+ population and OS in ITT population | Atezolizumab + bevacizumab | PD‐L1+: 11.2 | HR = 0.74; P = 0·0217 | ITT: 33.6 | HR = 0.93; P = 0·4751 | [92] |
| Sunitinib | PD‐L1+: 7.7 | ITT: 34.9 | |||||||
| JAVELIN Renal 101 | RCC | 886 | PFS and OS in PD‐L1+ population | Avelumab + axitinib | PD‐L1+: 13.8 | HR = 0.62; P <0.0001 | PD‐L1+: NE | HR = 0.83; P = 0.1301 | [94] |
| Sunitinib | PD‐L1+: 7.0 | PD‐L1+: 28.6 (27.4‐NE) | |||||||
| KEYNOTE‐426 | RCC | 1062 | OS and PFS in ITT population | Pembrolizumab + axitinib | ITT: 15.1 | HR = 0.69; P <0.001 | Data immature | Data immature | [93] |
| Sunitinib | ITT: 11.1 | ||||||||
Abbreviations: HCC, hepatocellular carcinoma; HR, hazard ratio; ITT, intention‐to‐treat; NE, could not be estimated; NSCLC, non‐small cell lung cancer; OS, overall survival; PD‐L1, programmed cell death 1 ligand 1; PFS, progression‐free survival; RCC, renal cell cancer; WT, wild‐type.
4.1. Clinical progress in hepatocellular carcinoma (HCC)
HCC has an immunosuppressive nature as evidenced by the failure of nivolumab or pembrolizumab monotherapy as first‐line or second‐line treatments, respectively [14, 15]. These studies indicated that adding other immunomodulatory agents, such as antiangiogenic agents, might be critical for reversing the immunosuppressive TME in HCC.
Lenvatinib, a VEGFR‐associated TKI, is an FDA‐approved first‐line treatment option for advanced‐stage HCC patients [79]. Recently, a phase Ib trial tested lenvatinib plus pembrolizumab in a first‐line setting of unresectable HCC (uHCC). Research has shown that the combination of lenvatinib plus pembrolizumab has positive antitumor activity with acceptable safety and tolerability, a median progression‐free survival (mPFS) of 8.6 months, and a median overall survival (mOS) of 22.0 months [80]. Lenvatinib combined with pembrolizumab was granted a breakthrough therapy designation in 2019. An ongoing phase III clinical trial (NCT03006926) further tests the usage of lenvatinib plus pembrolizumab compared with lenvatinib monotherapy as a first‐line treatment for uHCC.
In 2020, the investigators from the IMbrave150 study reported that in patients with untreated uHCC, atezolizumab (an anti‐PD‐L1 monoclonal antibody) plus bevacizumab was significantly superior to standard‐of‐care sorafenib monotherapy (Table 2) [81]. At the time of the primary analysis (August 29, 2019), the results of the study showed that the combination of atezolizumab plus bevacizumab significantly improved overall survival (OS) (hazard ratio [HR] = 0.58; P = 0.0006) and mPFS (6.8 vs. 4.3 months; HR = 0.59; P < 0.001) and more than doubled objective response rate (ORR; 27% vs. 12%, P < 0.0001) compared with standard‐of‐care. Notably, the safety profile of the combination of atezolizumab plus bevacizumab was shown to be very tolerable [81].
More recently, data from the phase Ib clinical trial GO30140 provide further support for the results from the IMbrave150 trial [82]. The GO30140 study compared atezolizumab plus bevacizumab with atezolizumab monotherapy among patients with uHCC. The investigators from the GO30140 study reported that atezolizumab plus bevacizumab significantly prolonged mPFS compared with atezolizumab monotherapy (5.6 vs. 3.4 months; HR = 0.55; P = 0.011), with an acceptable toxicity profile [82]. These two studies together demonstrate that a therapy combining an ICI with an antiangiogenic agent is superior to either of them as a monotherapy.
Before the advent of the combination of atezolizumab plus bevacizumab, the TKI sorafenib had been the unchallenged gold‐standard first‐line treatment option for advanced HCC since 2007 [83]. Currently, atezolizumab plus bevacizumab has received approval from health authorities as a first‐line treatment option for advanced‐stage HCC. In a recently updated guideline, the American Society of Clinical Oncology (ASCO) recommended the combination of atezolizumab plus bevacizumab as the prior treatment option for most patients with newly diagnosed advanced‐stage HCC [79].
4.2. Clinical progress in non‐small cell lung cancer (NSCLC)
With the development of biomarker‐directed therapies, NSCLC is no longer a cancer. While up to 70% of newly diagnosed patients are eligible for ICI‐containing regimens, patients with genomic alterations, such as epidermal growth factor receptor (EGFR) mutations or anaplastic lymphoma kinase (ALK) rearrangements, were excluded from many clinical trials evaluating ICI‐containing regimens due to disappointing immune efficacy results in previous studies [84, 85, 86, 87, 88, 89]. In contrast, the Impower150 clinical trial enrolled these patients [90].
The Impower150 trial enrolled a total of 1,202 chemotherapy‐naive patients with advanced‐stage NSCLC. Patients who had EGFR or ALK alterations were also permitted to enroll if they had not formerly received chemotherapy (Table 2) [90]. They were randomly assigned to one of the following arms to be treated with either: ABCP (atezolizumab, bevacizumab, carboplatin, and paclitaxel), ACP (without bevacizumab), or BCP (without atezolizumab). In wild‐type (defined as no EGFR or ALK alternation) population, the ABCP quadruplet regimen significantly prolonged not only mPFS (8.3 vs. 6.8 months; HR = 0.62; P < 0.001) but also mOS (19.2 vs. 14.7 months; HR = 0.78; P = 0.02) in comparison to standard‐of‐care BCP therapy, leading to FDA approval of ABCP quadruplet therapy for the treatment of this kind of patient [90]. Subsequent subgroup analyses demonstrated that compared with BCP, ABCP increased progression‐free survival (PFS) regardless of PD‐L1 status, existence of baseline liver metastases, and EGFR or ALK genetic alteration status [91]. For the first time, this study has provided a promising regimen for patients with EGFR mutations after the failure of TKI therapy.
4.3. Clinical progress in renal cell carcinoma (RCC)
In addition to ICIs, antiangiogenic drugs are routinely used to treat advanced RCC. Over the past few years, the combination of these two treatment modalities has been extensively tested to determine any potential synergistic effects (Table 2).
IMmotion151 is a phase III clinical trial comparing atezolizumab plus bevacizumab with sunitinib as a first‐line treatment option for advanced‐stage RCC. Notably, both sarcomatoid and clear cell histology were included. Primary PFS analyses revealed that PD‐L1+ patients receiving atezolizumab plus bevacizumab had a superior mPFS over patients receiving sunitinib monotherapy. Interestingly, the PFS data demonstrated the benefit of atezolizumab plus bevacizumab in all subgroups regardless of PD‐L1 status. However, the combination of atezolizumab plus bevacizumab failed to obtain FDA approval because it did not improve mOS [92].
KEYNOTE‐426 is a phase III clinical trial comparing pembrolizumab plus axitinib (a VEGFR‐associated TKI) with the standard‐treatment sunitinib for treating patients with newly‐diagnosed advanced‐stage clear‐cell RCC. In the primary analysis (August 24, 2018), pembrolizumab plus axitinib remarkably prolonged mPFS compared with sunitinib among intention‐to‐treat patients. This study confirmed the benefit of combining pembrolizumab with axitinib across any risk category and different PD‐L1 statuses. It is noteworthy that this research was the first of the combination clinical trials to show significantly improved OS (HR = 0.53; P < 0.0001) in comparison with standard‐of‐care sunitinib in RCC [93]. The combination of pembrolizumab plus axitinib has received FDA approval for previously untreated advanced‐stage RCC.
JA VELIN Renal 101 is a contemporary phase III clinical trial comparing avelumab (an anti‐PD‐L1 monoclonal antibody) plus axitinib with sunitinib in a first‐line setting of advanced‐stage RCC. As of the second analysis (January 28, 2019), the combination arm showed longer mPFS in not only the PD‐L1+ subgroup (HR = 0.62; P < 0.0001) but also in all patients (HR = 0.69; P < 0.0001). Even though OS results were unavailable, the combination of avelumab plus axitinib has received FDA approval as a first‐line treatment option for metastatic RCC [94].
Although these three phase III studies are different with respect to patient enrollment, definition of PD‐L1 expression levels, and endpoints, all showed that the combinations of ICIs and vascular‐targeting agents improved PFS regardless of PD‐L1 status or prognostic subgroups without significant increases in toxicities [92, 93, 94]. Emerging data from other phase III clinical trials, such as the CheckMate 9ER trial and the CLEAR trial, could further demonstrate the benefits of the combinations of ICIs and antiangiogenic agents [95, 96].
4.4. Clinical progress in other cancers
Up to 70% of endometrial cancers are not microsatellite instability high (MSI‐H) or DNA mismatch repair pathway deficient (dMMR) and have limited response rates to single‐agent ICIs [97]. Thus, an effective combination regimen is urgently needed to augment anticancer immune responses. The single‐arm phase Ib/II clinical trial NCT02501096 enrolled patients with advanced‐stage endometrial cancer who had received no more than two prior chemotherapies to evaluate the combination of lenvatinib plus pembrolizumab. In the final analysis, the combination therapy showed promising efficacy in patients with tumors that were neither MSI‐H nor dMMR with an ORR of 36.2% at week 24 and a mPFS of 7.4 months [98]. In 2019, the FDA granted accelerated approval to the combination of lenvatinib plus pembrolizumab for the treatment of patients with advanced‐stage endometrial tumors that are neither MSI‐H nor dMMR and have progressed after previous treatments. Two phase III clinical trials (NCT03517449 and NCT03884101) are further testing the combination of lenvatinib plus pembrolizumab .
Relapsed ovarian cancer represents another type of disease largely unresponsive to single‐agent ICIs. A recent phase II clinical trial (NCT02873962) assessed nivolumab plus bevacizumab for recurrent ovarian cancer. The combination regimen showed activity in some enrolled patients with an ORR of 28.9% and a total clinical benefit rate of 55.3% [99]. Another similar trial (NCT02853318) tested nivolumab plus bevacizumab with cyclophosphamide for this hard‐to‐treat cancer. The investigators reported a high ORR of 37.5%, an encouraging 6‐month PFS rate of 70%, and a very tolerable toxicity profile [100].
BACCI is a phase II clinical trial evaluating the efficacy and safety of atezolizumab plus bevacizumab plus capecitabine (ABC) for the treatment of advanced‐stage CRC. ABC significantly prolonged mPFS compared with atezolizumab plus bevacizumab plus placebo [101]. In the era of advanced‐stage CRC, this was the first trial reporting a positive result when combing an ICI with an antiangiogenic agent.
These preliminary results strongly indicate the benefit of the combinations of ICIs and antiangiogenic agents in multiple solid tumors. A spectrum of ongoing clinical trials will further reveal the benefit of such combinations on a broader scale (Table 3).
TABLE 3.
Ongoing phase II or III clinical trials combining immune checkpoint inhibitors with VEGF axis inhibitors
| Clinical trial ID | Combination arm | ||||
|---|---|---|---|---|---|
| NCT03172754 | VEGF inhibitors and ICIs | Other agents | Cancer type | Phase | Status at time of search |
| NCT02636725 | Axitinib + nivolumab | NA | Advanced RCC | I/II | Recruiting |
| NCT03764293 | Axitinib + pembrolizumab | NA | Soft tissue sarcomas | II | Active, not recruiting |
| NCT02724878 | Apatinib + SHR‐1210 | NA | Advanced HCC | III | Recruiting |
| NCT03181100 | Bevacizumab + atezolizumab | NA | Advanced non‐clear cell RCC | II | Active, not recruiting |
| NCT02982694 | Bevacizumab + atezolizumab | NA | Anaplastic or poorly differentiated thyroid cancer | II | Recruiting |
| NCT03526432 | Bevacizumab + atezolizumab | NA | Chemotherapy resistant, MSI‐like CRC | II | Recruiting |
| NCT03133390 | Bevacizumab + atezolizumab | NA | Endometrial cancer | II | Recruiting |
| NCT03074513 | Bevacizumab + atezolizumab | NA | Metastatic/unresectable urothelial cancer | II | Recruiting |
| NCT02921269 | Bevacizumab + atezolizumab | NA | Rare solid tumors | II | Active, not recruiting |
| NCT02715531 | Bevacizumab + atezolizumab | NA | Recurrent, persistent, or metastatic cervical cancer | II | Active, not recruiting |
| NCT03175432 | Bevacizumab + atezolizumab | NA | Solid tumors | Ib | Active, not recruiting |
| NCT03272217 | Bevacizumab + atezolizumab | NA | Untreated melanoma brain metastases | II | Recruiting |
| NCT04102098 | Bevacizumab + atezolizumab | NA | Urothelial carcinoma | II | Recruiting |
| NCT02659384 | Bevacizumab + atezolizumab | NA | HCC | III | Recruiting |
| NCT02891824 | Bevacizumab + atezolizumab | Aspirin | Recurrent platinum resistant ovarian cancer | II | Active, not recruiting |
| NCT03414983 | Bevacizumab + atezolizumab | Platinum‐based chemotherapy | Late relapse ovarian cancer | III | Active, not recruiting |
| NCT03353831 | Bevacizumab + atezolizumab | Oxaliplatin + leucovorin + fluorouracil | Metastatic CRC | II/III | Active, not recruiting |
| NCT03024437 | Bevacizumab + atezolizumab | Aclitaxel/PLD | Recurrent ovarian cancer | III | Recruiting |
| NCT03395899 | Bevacizumab + atezolizumab | Entinostat | Advanced RCC | I/II | Recruiting |
| NCT03038100 | Bevacizumab + atezolizumab | Ipatasertib | Breast cancer | II | Recruiting |
| NCT03556839 | Bevacizumab + atezolizumab | Paclitaxel + carboplatin | Ovarian, fallopian tube, or primary peritoneal cancer | III | Active, not recruiting |
| NCT02839707 | Bevacizumab + atezolizumab | Cisplatin/carboplatin + paclitaxel | Metastatic cervical carcinoma | III | Recruiting |
| NCT03721653 | Bevacizumab + atezolizumab | PLD | Relapsed ovarian, fallopian tube or peritoneal cancer | II/III | Recruiting |
| NCT03786692 | Bevacizumab + atezolizumab | FOLFOXIRI | Advanced CRC | II | Active, not recruiting |
| NCT03762018 | Bevacizumab + atezolizumab | Carboplatin + pemetrexed | Advanced NSCLC | II | Recruiting |
| NCT02336165 | Bevacizumab + atezolizumab | Carboplatin + pemetrexed | Pleural mesothelioma | III | Recruiting |
| NCT03847428 | Bevacizumab + durvalumab | NA | Glioblastoma | II | Active, not recruiting |
| NCT02519348 | Bevacizumab + durvalumab | NA | HCC | III | Recruiting |
| NCT03737643 | Bevacizumab + durvalumab | NA | Advanced HCC | II | Active, not recruiting |
| NCT03778957 | Bevacizumab + durvalumab | Olaparib + carboplatin + paclitaxel | Advanced ovarian cancer | III | Recruiting |
| NCT01950390 | Bevacizumab + durvalumab | Transarterial chemoembolization | Intermediate‐stage HCC | III | Recruiting |
| NCT03117049 | Bevacizumab + ipilimumab | NA | Unresectable stage III or IV melanoma | II | Active, not recruiting |
| NCT03452579 | Bevacizumab + nivolumab | Carboplatin + paclitaxel | NSCLC | III | Active, not recruiting |
| NCT02681549 | Bevacizumab + nivolumab | NA | Glioblastoma | II | Active, not recruiting |
| NCT03396926 | Bevacizumab + pembrolizumab | NA | Melanoma or NSCLC with brain metastases | II | Recruiting |
| NCT03635567 | Bevacizumab + pembrolizumab | Capecitabine | Advanced‐stage MSS CRC | II | Recruiting |
| NCT02039674 | Bevacizumab + pembrolizumab | Carboplatin + cisplatin + paclitaxel | Advanced cervical cancer | III | Active, not recruiting |
| NCT03755791 | Bevacizumab + pembrolizumab | Paclitaxel + carboplatin | NSCLC | I/II | Active, not recruiting |
| NCT03141177 | Cabozantinib + atezolizumab | NA | Advanced HCC | III | Recruiting |
| NCT03793166 | Cabozantinib + nivolumab | NA | Locally advanced or metastatic RCC | III | Active, not recruiting |
| NCT03937219 | Cabozantinib + nivolumab | Ipilimumab | Advanced kidney cancer | III | Recruiting |
| NCT02811861 | Cabozantinib + nivolumab | Ipilimumab | Advanced RCC | III | Recruiting |
| NCT03884101 | Lenvatinib + pembrolizumab | NA | Advanced RCC | III | Active, not recruiting |
| NCT03713593 | Lenvatinib + pembrolizumab | NA | Endometrial carcinoma | III | Recruiting |
| NCT03517449 | Lenvatinib + pembrolizumab | NA | Advanced HCC | III | Active, not recruiting |
| NCT03172754 | Lenvatinib + pembrolizumab | NA | Advanced endometrial cancer | III | Active, not recruiting |
The results were obtained from http://clinicaltrials.gov/.
Abbreviations: CRC, colorectal cancer; FOLFOXIRI, l‐leucovorin + 5‐fluorouracil + oxaliplatin + irinotecan; HCC, hepatocellular carcinoma; ICI, immune‐checkpoint inhibitor; MSI, microsatellite instable; MSS, microsatellite stable; NA, not applicable; NSCLC, non‐small cell lung cancer; PLD, pegylated liposomal doxorubicin hydrochloride; RCC, renal cell cancer.
4.5. Clinical trials combining other immunotherapies with antiangiogenic agents
All of the landmark phase III clinical trials we discussed above combined ICIs with antiangiogenic agents, but cancer immunotherapy does not limit to immune checkpoint blockade (ICB) but also includes cell therapies, cancer vaccines, oncolytic viruses, and immunostimulatory cytokines. There are multiple preclinical studies reporting synergies between these immunotherapies and antiangiogenic agents [102, 103, 104, 105, 106]. However, clinical trials combining these immunotherapies with antiangiogenic agents are largely unsuccessful so far (Table 4) [107‐109]. For example, a phase III clinical trial (NCT02562755) to determine whether treatment with Pexa‐Vec (an oncolytic vaccinia virus) followed by sorafenib increases survival compared to standard‐of‐care sorafenib monotherapy in patients with advanced HCC was terminated recently due to disappointing preliminary results [110]. There are many steps to create an anticancer immune response, and the failure of these combinations strongly suggests the existence of immunosuppressive mechanisms that they cannot overcome.
TABLE 4.
Clinical trials combining other immunotherapies with antiangiogenic agents
| Clinical trial ID (reference) | Phase | Cancer type | No. of patients | Combinational arm | Status and results at time of search |
|---|---|---|---|---|---|
| Immunostimulatory cytokine studies | |||||
| NA [107] | III | Advanced RCC | 732 | Bevacizumab + IFNα | mOS: 18.3 months (combinational arm) vs. 17.4 months (IFNα arm), P = 0.069 |
| NA [108] | III | Advanced RCC | 649 | Bevacizumab + IFNα2A | mOS: 23.3 months (combinational arm) vs. 21.3 months (IFNα2A arm), P = 0.1291 |
| Cell therapy studies | |||||
| NCT00678119 [109] | II | Advanced RCC | 21 | Sunitinib + autologous DC immunotherapy | mPFS: 11.2 months; mOS: 30.2 months |
| NCT01582672 (NA) | III | Advanced RCC | 462 | Sunitinib + autologous DC immunotherapy | Terminated due to lack of efficacy |
| NCT02857920 (NA) | I/II | Solid tumors | 45 | Bevacizumab + allogeneic NK immunotherapy | Completed; no results posted |
| Oncolytic virus studies | |||||
| NCT02562755 [110] | III | Advanced HCC | 459 | Sorafenib + Pexa‐Vec | Terminated due to disappointing preliminary results |
| Vaccination studies | |||||
| NCT02616185 (NA) | I | Prostate cancer | 91 | Sunitinib + PF‐06755990 (vaccine) | Completed; no results posted |
Abbreviations: IFN, interferon; mOS, median overall survival; NA, not applicable; NK, natural killer cell; RCC, renal cell cancer.
5. CHALLENGES AND OPPORTUNITIES
Considering the enormity of the ongoing combination effort, coordination and collaboration are necessary for the research community to fulfill the goal of “curing cancer.” Here, we attempt to define essential challenges in combining ICIs with antiangiogenic agents.
5.1. Choosing an appropriate antiangiogenic agent
A critical concern is identifying an appropriate antiangiogenic agent for combination with ICIs. This combination modality has been investigated with either antibodies against VEGF/VEGFR or multitargeted TKIs. However, a resultant commensurate benefit of these two strategies in improving antitumor immunity remains unclear.
Small‐molecule TKIs inhibit multiple receptor tyrosine kinase signaling pathways and do not solely target the proangiogenic VEGF/VEGFR axis, whereas antibodies are designed against VEGF or its receptor. Thus, multitargeted TKIs might possess more antitumor abilities than antibodies because they have broader bioactivity. For example, sunitinib is a well‐defined antiangiogenic TKI but has also been reported to induce growth arrest and programmed cell death of mouse‐derived RCC tumor cells in vivo [111]. Likewise, the ability of sunitinib to decreased MDSC accumulation in a mouse model is supposedly caused by inhibition of mast/stem cell growth factor receptor c‐Kit in MDSCs rather than inhibition of VEGF [112]. Other contradictory studies have suggested that sunitinib leads to higher infiltration of Treg cells, upregulated PD‐L1 expression, and is related to disappointing outcomes in RCC patients [113].
It is noteworthy that many preclinical and clinical studies have used small‐molecule TKIs such as sunitinib or sorafenib. Considering the possibility of the existence of additional immune‐promoting mechanisms, the conclusions of such studies should be taken with caution before the contribution of non‐VEGF kinases to TME immunomodulation is delineated. Alternative experiments with genetic‐manipulation approaches or inhibitory antibodies are beneficial, if not essential, to determine the contribution of non‐VEGF kinases.
5.2. Biomarker development for better patient selection
Biomarker development is challenging in the era of combining ICIs and antiangiogenic agents. While predictive biomarkers for molecularly targeted drugs are usually a unique genetic aberration described as a binary assay, biomarkers for ICIs are often continuous variables that are gradually associated with clinical endpoints. The search for validated and sensitive biomarkers to better identify patients eligible for therapy combining ICIs and antiangiogenic agents is ongoing. Many biomarkers have been reported, but none has been formally accepted for routine clinical use.
The IMmotion150 trial, a phase II trial testing atezolizumab plus bevacizumab in RCC, included exploratory biomarker analyses to study three biological axes: angiogenesis, T‐effector/IFNγ response, and myeloid immune suppression [114]. Tumor mutation and neoantigen burden were not related to PFS, whereas angiogenesis, preexisting immune response, and myeloid inflammatory gene expression signatures were strongly and differentially related to PFS, not only within but also across treatment groups. Specifically, the combination of atezolizumab plus bevacizumab improved PFS in patients with preexisting immunity and myeloid inflammatory gene expression signatures, whereas sunitinib was more efficient than the combination therapy in highly angiogenic tumors [114].
More recently, Zhu et al. [115] reported data from a randomized phase Ib clinical trial that evaluated atezolizumab plus bevacizumab versus atezolizumab alone in uHCC. Similar to the IMmotion150 study, this genomic correlative study evaluated immunological biomarker subgroups divided depending on gene signatures. In the arm evaluating atezolizumab plus bevacizumab, there was no association between tumor mutation burden (TMB) and response to atezolizumab plus bevacizumab or PFS. In contrast, baseline tumor gene expression analyses showed that preexisting immunity was associated with more response and longer PFS. In the randomized arm comparing atezolizumab plus bevacizumab with atezolizumab, the PFS benefit of combination therapy versus monotherapy was remarkably obvious in patients with high expression of the following candidate biomarkers: VEGFR2, Treg, and myeloid inflammation signatures. Reduced levels of VEGFR2 and Treg signatures were further confirmed by analyses of 12 serial biopsy pairs after treatment with atezolizumab plus bevacizumab [115].
While the results from the two studies described above require validation, they strongly suggest that myeloid‐mediated immunosuppression is a principal contributor to resistance to ICIs and that such resistance can be therapeutically overcome by adding antiangiogenic agents to ICIs. Although there is a lack of relevant clinical trials, it is evident that identifying a universally accurate biomarker irrespective of tumor type or combined regimen might be difficult, if not impossible. To select tumors addicted to angiogenic signaling, molecular profiling of the TME components seems to be a promising strategy.
5.3. Tailoring the dose and sequence of treatments
Both antiangiogenic agents and ICIs lead to complicated biological responses. Hypertension requiring medical intervention is a typical side effect of bevacizumab, which can be observed in 11% of bevacizumab‐treated patients, and some unusual side effects like proteinuria and bleeding can also be observed [116]. Small molecule TKIs are possibly more toxic as they inhibit multiple signaling pathways. Typical side effects of TKIs include hypertension, diarrhea, and cardiac ischemia [117, 118]. The development of combination regimens could further enhance the complexity and increase the risk of toxicities. Therefore, identifying the optimal dose of agents and sequencing of treatment are critical for optimizing the effectiveness, toxicity, and tolerability of therapies combining antiangiogenic agents with ICIs in the clinic.
Historically, antiangiogenic agents have been prescribed at maximum tolerated doses until cancer progression. However, traditional high‐dose and/or long‐term antiangiogenic treatments have now been well established to lead to excessive vessel pruning and increased immunosuppression in the TME [119, 120, 121]. A retrospective study concluded that low‐dose bevacizumab (<3.6 mg/kg per week) for progressive/recurrent glioblastoma, which is much lower than the FDA‐approved dose (10 mg/kg every two weeks), confers a more significant survival benefit than a higher dose [122]. This aligns with an article that systematically reviewed the immunomodulatory functions of antiangiogenic TKIs in preclinical studies, which showed that high‐dose TKIs suppressed antitumor immune response, whereas lower‐dose TKIs were immune‐promoting [123]. Of note, all recent phase III trials in HCC, NSCLC, and RCC used antiangiogenic agents according to standard FDA‐approved doses [81, 90, 92, 93, 94]. The optimal doses for antiangiogenic drugs and their importance when combined with ICIs require further investigation in prospective studies and well‐controlled clinical trials using low‐dose antiangiogenic agents.
The kinetics of recently defined immunostimulation‐induced tumor vasculature normalization is largely unknown. Delineating the possible mechanism to find the optimal sequence of the administration of antiangiogenic agents and ICIs is necessary to prolong normalization windows and maximize clinical benefits. Immune‐related adverse effects caused by ICIs can often resolve after discontinuing treatment or reducing drug doses [124]. Because antiangiogenic treatments can induce tumor vessel normalization, which can enhance the delivery of both ICIs and immune cells to solid tumors, therapies combining antiangiogenic agents with ICIs might not require traditional doses of ICIs to confer immunostimulation; this may subsequently reduce the risk of adverse effects.
6. OUTLOOK
As discussed, there are three rationales for combining antiangiogenic therapy with ICIs. First, both antiangiogenic agents and ICIs possess anticancer activity. Second, in addition to their well‐defined antiangiogenic function, antiangiogenic agents also have immunomodulatory effects. Third, these two therapies could reinforce each other through the immunostimulatory vascular‐modulating cycle.
Encouraging results from multiple clinical trials have shown that combining antiangiogenic treatments with ICIs significantly prolonged survival in comparison with standards‐of‐care in RCC, NSCLC, and HCC. It remains to be seen whether results from future clinical studies will advocate the usage of combinations of antiangiogenic agents plus ICIs in other tumors. It is plausible that such success might be a small part of possible combinations of ICIs and agents targeting specific components of the TME. The convergence of innovation will likely lead to rapid advances in tumor treatments, possibly making it more complex to determine the appropriate therapy for a specific malignancy.
In the future, efforts to delineate the mechanisms underlying response and develop predictive biomarkers for combining antiangiogenic treatments with ICIs will be facilitated by translational studies that include either neoadjuvant approaches or paired serial biopsies. The knowledge learned from such studies will provide rationales for the development of next‐generation combination strategies, in which agents modulating specific aspects of the immunosuppressive TME are added to an ICI backbone to improve antitumor immunity. The combinations of antiangiogenic agents and ICIs should be designed with caution to minimize or reduce the risk of intolerable adverse effects that might lead to termination of treatment. In this respect, antibody‐based agents are possibly superior to multitarget TKIs, which usually inhibit multiple signaling pathways and are very toxic (even though toxicity risks may be controlled by factors, such as the choice of the TKI and the dose and schedule of administration). With evidence from well‐designed clinical trials, physicians will be able to select optimal combination regimens and determine appropriate doses and sequences to enhance the efficacy and reduce the toxicity.
DECLARATIONS
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
COMPETING INTERESTS
The authors declare that they have no competing interests.
AUTHORS' CONTRIBUTIONS
Conception/design: Z.S. and S.L. Manuscript preparing and writing: S.L. and J.C. Preparation of tables and figures: S.L. Manuscript writing and editing: S.L. and J.C. All authors read and approved the final manuscript.
ACKNOWLEDGEMENTS
This work was financially supported by National Key Research and Development Program (2017YFSF090107), National Natural Science Foundation of China (82072996 and 81874131), Hubei Province Natural Science Funds for Distinguished Young Scholar (2017CFA062), and Innovative research team of high‐level local universities in Shanghai (ZLCX20180500).
Li S‐J, Chen J‐X, Sun Z‐J. Improving antitumor immunity using antiangiogenic agents: mechanistic insights, current progress, and clinical challenges. Cancer Commun. 2021;41:830–850. 10.1002/cac2.12183
REFERENCES
- 1.Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, et al. Role of the tumor microenvironment in PD‐L1/PD‐1‐mediated tumor immune escape. Mol Cancer. 2019;18(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zou W, Wolchok JD, Chen L. PD‐L1 (B7‐H1) and PD‐1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci Transl Med. 2016;8(328). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat Rev Immunol. 2020;20(11):651–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hack SP, Zhu AX, Wang Y. Augmenting anticancer immunity through combined targeting of angiogenic and PD‐1/PD‐L1 pathways: Challenges and opportunities. Front Immunol. 2020;11:598877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Upadhaya S, Hubbard‐Lucey VM, Yu JX. Immuno‐oncology drug development forges on despite COVID‐19. Nat Rev Drug Discov. 2020;19(11):751–2. [DOI] [PubMed] [Google Scholar]
- 6.Wu Q, Chu Q, Zhang H, Yang B, He X, Zhong Y, et al. Clinical outcomes of coronavirus disease 2019 (COVID‐19) in cancer patients with prior exposure to immune checkpoint inhibitors. Cancer Commun (Lond). 2020;40(8):374–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sharma P, Hu‐Lieskovan S, Wargo JA, Primary Ribas A., Adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lin Z, Zhang Y, Zhang L. Construction of an integrated prognostic classifier model for predicting the efficacy of immune checkpoint inhibitor therapy in non‐small cell lung cancer. Cancer Commun (Lond). 2020;40(8):370–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Haslam A, Prasad V. Estimation of the percentage of US patients with cancer who are eligible for and respond to checkpoint inhibitor immunotherapy drugs. JAMA Netw Open. 2019;2(5):e192535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Adams S, Schmid P, Rugo HS, Winer EP, Loirat D, Awada A, et al. Pembrolizumab monotherapy for previously treated metastatic triple‐negative breast cancer: Cohort A of the phase II KEYNOTE‐086 study. Ann Oncol. 2019;30(3):397–404. [DOI] [PubMed] [Google Scholar]
- 11.Rugo HS, Delord JP, Im SA, Ott PA, Piha‐Paul SA, Bedard PL, et al. Safety and antitumor activity of pembrolizumab in patients with estrogen receptor‐positive/human epidermal growth factor receptor 2‐negative advanced breast cancer. Clin Cancer Res. 2018;24(12):2804–11. [DOI] [PubMed] [Google Scholar]
- 12.Bono JSD, Goh JC, Ojamaa K, Rodriguez JMP, Drake CG, Hoimes CJ, et al. KEYNOTE‐199: Pembrolizumab (pembro) for docetaxel‐refractory metastatic castration‐resistant prostate cancer (mCRPC). 2018;36(15_suppl):5007‐. [Google Scholar]
- 13.Antonarakis ES, Piulats JM, Gross‐Goupil M, Goh J, Ojamaa K, Hoimes CJ, et al. Pembrolizumab for treatment‐refractory metastatic castration‐resistant prostate cancer: Multicohort, open‐label phase II KEYNOTE‐199 study. J Clin Oncol. 2020;38(5):395–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yau T, Park JW, Finn RS, Cheng AL, Mathurin P, Edeline J, et al. CheckMate 459: A randomized, multi‐center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first‐line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2019;30:v874–v5. [Google Scholar]
- 15.Finn RS, Ryoo BY, Merle P, Kudo M, Bouattour M, Lim HY, et al. Pembrolizumab as second‐line therapy in patients with advanced hepatocellular carcinoma in KEYNOTE‐240: A randomized, double‐blind, phase III trial. J Clin Oncol. 2020;38(3):193–202. [DOI] [PubMed] [Google Scholar]
- 16.Upadhaya S, Neftelino ST, Hodge JP, Oliva C, Campbell JR, Yu JX. Combinations take centre stage in PD1/PDL1 inhibitor clinical trials. Nat Rev Drug Discov. 2021;20(3):168–9. [DOI] [PubMed] [Google Scholar]
- 17.Zhang B, Qi L, Wang X, Xu J, Liu Y, Mu L, et al. Phase II clinical trial using camrelizumab combined with apatinib and chemotherapy as the first‐line treatment of advanced esophageal squamous cell carcinoma. Cancer Commun (Lond). 2020;40(12):711–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rajabi M, Mousa S. The Role of Angiogenesis in Cancer Treatment. Biomedicines. 2017;5(2):34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Keck PJ, Hauser SD, Krivi G, Sanzo K, Warren T, Feder J, et al. Vascular permeability factor, an endothelial cell mitogen related to PDGF. Science. 1989;246(4935):1309–12. [DOI] [PubMed] [Google Scholar]
- 20.Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science. 1989;246(4935):1306–9. [DOI] [PubMed] [Google Scholar]
- 21.Millauer B, Shawver LK, Plate KH, Risau W, Ullrich A. Glioblastoma growth inhibited in vivo by a dominant‐negative Flk‐1 mutant. Nature. 1994;367(6463):576–9. [DOI] [PubMed] [Google Scholar]
- 22.Fong GH, Rossant J, Gertsenstein M, Breitman ML. Role of the Flt‐1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature. 1995;376(6535):66–70. [DOI] [PubMed] [Google Scholar]
- 23.Ferrara N, Adamis AP. Ten years of anti‐vascular endothelial growth factor therapy. Nat Rev Drug Discov. 2016;15(6):385–403. [DOI] [PubMed] [Google Scholar]
- 24.Folkman J. Tumor angiogenesis: Therapeutic implications. N Engl J Med. 1971;285(21):1182–6. [DOI] [PubMed] [Google Scholar]
- 25.Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350(23):2335–42. [DOI] [PubMed] [Google Scholar]
- 26.Scholz A, Harter PN, Cremer S, Yalcin BH, Gurnik S, Yamaji M, et al. Endothelial cell‐derived angiopoietin‐2 is a therapeutic target in treatment‐naive and bevacizumab‐resistant glioblastoma. EMBO Mol Med. 2016;8(1):39–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Peterson TE, Kirkpatrick ND, Huang Y, Farrar CT, Marijt KA, Kloepper J, et al. Dual inhibition of Ang‐2 and VEGF receptors normalizes tumor vasculature and prolongs survival in glioblastoma by altering macrophages. Proc Natl Acad Sci USA. 2016;113(16):4470–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kloepper J, Riedemann L, Amoozgar Z, Seano G, Susek K, Yu V, et al. Ang‐2/VEGF bispecific antibody reprograms macrophages and resident microglia to anti‐tumor phenotype and prolongs glioblastoma survival. Proc Natl Acad Sci USA. 2016;113(16):4476–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schmittnaegel M, Rigamonti N, Kadioglu E, Cassara A, Wyser Rmili C, Kiialainen A, et al. Dual angiopoietin‐2 and VEGFA inhibition elicits antitumor immunity that is enhanced by PD‐1 checkpoint blockade. Sci Transl Med. 2017;9(385). [DOI] [PubMed] [Google Scholar]
- 30.Elbanna M, Orillion AR, Damayanti NP, Adelaiye‐Ogala R, Shen L, Miles KM, et al. Dual inhibition of angiopoietin‐TIE2 and MET alters the tumor microenvironment and prolongs survival in a metastatic model of renal cell carcinoma. Mol Cancer Ther. 2020;19(1):147–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alfaro C, Suarez N, Gonzalez A, Solano S, Erro L, Dubrot J, et al. Influence of bevacizumab, sunitinib and sorafenib as single agents or in combination on the inhibitory effects of VEGF on human dendritic cell differentiation from monocytes. Br J Cancer. 2009;100(7):1111–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen DS, Mellman I. Oncology meets immunology: The cancer‐immunity cycle. Immunity. 2013;39(1):1–10. [DOI] [PubMed] [Google Scholar]
- 33.Patente TA, Pinho MP, Oliveira AA, Evangelista GCM, Bergami‐Santos PC, Barbuto JAM. Human dendritic cells: Their heterogeneity and clinical application potential in cancer immunotherapy. Front Immunol. 2018;9:3176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang Y, Xiang Y, Xin VW, Wang XW, Peng XC, Liu XQ, et al. Dendritic cell biology and its role in tumor immunotherapy. J Hematol Oncol. 2020;13(1):107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gabrilovich DI, Chen HL, Girgis KR, Cunningham HT, Meny GM, Nadaf S, et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2(10):1096–103. [DOI] [PubMed] [Google Scholar]
- 36.Mimura K, Kono K, Takahashi A, Kawaguchi Y, Fujii H. Vascular endothelial growth factor inhibits the function of human mature dendritic cells mediated by VEGF receptor‐2. Cancer Immunol Immunother. 2007;56(6):761–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oyama T, Ran S, Ishida T, Nadaf S, Kerr L, Carbone DP, et al. Vascular endothelial growth factor affects dendritic cell maturation through the inhibition of nuclear factor‐kappa B activation in hemopoietic progenitor cells. J Immunol. 1998;160(3):1224–32. [PubMed] [Google Scholar]
- 38.Belkaid Y, Oldenhove G. Tuning microenvironments: Induction of regulatory T cells by dendritic cells. Immunity. 2008;29(3):362–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Long J, Hu Z, Xue H, Wang Y, Chen J, Tang F, et al. Vascular endothelial growth factor (VEGF) impairs the motility and immune function of human mature dendritic cells through the VEGF receptor 2‐RhoA‐cofilin1 pathway. Cancer Sci. 2019;110(8):2357–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lissoni P, Malugani F, Bonfanti A, Bucovec R, Secondino S, Brivio F, et al. Abnormally enhanced blood concentrations of vascular endothelial growth factor (VEGF) in metastatic cancer patients and their relation to circulating dendritic cells, IL‐12 and endothelin‐1. J Biol Regul Homeost Agents. 2001;15(2):140–4. [PubMed] [Google Scholar]
- 41.Boissel N, Rousselot P, Raffoux E, Cayuela JM, Maarek O, Charron D, et al. Defective blood dendritic cells in chronic myeloid leukemia correlate with high plasmatic VEGF and are not normalized by imatinib mesylate. Leukemia. 2004;18(10):1656–61. [DOI] [PubMed] [Google Scholar]
- 42.Strauss L, Volland D, Kunkel M, Reichert TE. Dual role of VEGF family members in the pathogenesis of head and neck cancer (HNSCC): Possible link between angiogenesis and immune tolerance. Med Sci Monit. 2005;11(8):Br280–92. [PubMed] [Google Scholar]
- 43.Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, et al. Blockade of B7‐H1 improves myeloid dendritic cell‐mediated antitumor immunity. Nat Med. 2003;9(5):562–7. [DOI] [PubMed] [Google Scholar]
- 44.Osada T, Chong G, Tansik R, Hong T, Spector N, Kumar R, et al. The effect of anti‐VEGF therapy on immature myeloid cell and dendritic cells in cancer patients. Cancer Immunol Immunother. 2008;57(8):1115–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Malo CS, Khadka RH, Ayasoufi K, Jin F, AbouChehade JE, Hansen MJ, et al. Immunomodulation mediated by anti‐angiogenic therapy improves CD8 T cell immunity against experimental glioma. Front Oncol. 2018;8:320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ohm JE, Gabrilovich DI, Sempowski GD, Kisseleva E, Parman KS, Nadaf S, et al. VEGF inhibits T‐cell development and may contribute to tumor‐induced immune suppression. Blood. 2003;101(12):4878–86. [DOI] [PubMed] [Google Scholar]
- 47.Georganaki M, van Hooren L, Dimberg A. Vascular targeting to increase the efficiency of immune checkpoint blockade in cancer. Front Immunol. 2018;9:3081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hegde PS, Chen DS. Top 10 challenges in cancer immunotherapy. Immunity. 2020;52(1):17–35. [DOI] [PubMed] [Google Scholar]
- 49.Hegde PS, Karanikas V, Evers S. The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clin Cancer Res. 2016;22(8):1865–74. [DOI] [PubMed] [Google Scholar]
- 50.Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144(5):646–74. [DOI] [PubMed] [Google Scholar]
- 51.Huang H, Langenkamp E, Georganaki M, Loskog A, Fuchs PF, Dieterich LC, et al. VEGF suppresses T‐lymphocyte infiltration in the tumor microenvironment through inhibition of NF‐kappaB‐induced endothelial activation. FASEB J. 2015;29(1):227–38. [DOI] [PubMed] [Google Scholar]
- 52.Dirkx AE, Oude Egbrink MG, Kuijpers MJ, van der Niet ST, Heijnen VV, Bouma‐ter Steege JC, et al. Tumor angiogenesis modulates leukocyte‐vessel wall interactions in vivo by reducing endothelial adhesion molecule expression. Cancer Res. 2003;63(9):2322–9. [PubMed] [Google Scholar]
- 53.Hellwig SM, Damen CA, van Adrichem NP, Blijham GH, Groenewegen G, Griffioen AW. Endothelial CD34 is suppressed in human malignancies: Role of angiogenic factors. Cancer Lett. 1997;120(2):203–11. [DOI] [PubMed] [Google Scholar]
- 54.Dirkx AE, oude Egbrink MG, Castermans K, van der Schaft DW, Thijssen VL, Dings RP, et al. Anti‐angiogenesis therapy can overcome endothelial cell anergy and promote leukocyte‐endothelium interactions and infiltration in tumors. Faseb j. 2006;20(6):621–30. [DOI] [PubMed] [Google Scholar]
- 55.Motz GT, Santoro SP, Wang LP, Garrabrant T, Lastra RR, Hagemann IS, et al. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat Med. 2014;20(6):607–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Galon J, Bruni D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat Rev Drug Discov. 2019;18(3):197–218. [DOI] [PubMed] [Google Scholar]
- 57.Jiang H, Hegde S, Knolhoff BL, Zhu Y, Herndon JM, Meyer MA, et al. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nat Med. 2016;22(8):851–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Voron T, Colussi O, Marcheteau E, Pernot S, Nizard M, Pointet AL, et al. VEGF‐A modulates expression of inhibitory checkpoints on CD8+ T cells in tumors. J Exp Med. 2015;212(2):139–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mulligan JK, Day TA, Gillespie MB, Rosenzweig SA, Young MR. Secretion of vascular endothelial growth factor by oral squamous cell carcinoma cells skews endothelial cells to suppress T‐cell functions. Hum Immunol. 2009;70(6):375–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wallin JJ, Bendell JC, Funke R, Sznol M, Korski K, Jones S, et al. Atezolizumab in combination with bevacizumab enhances antigen‐specific T‐cell migration in metastatic renal cell carcinoma. 2016;7(1):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Terme M, Pernot S, Marcheteau E, Sandoval F, Benhamouda N, Colussi O, et al. VEGFA‐VEGFR pathway blockade inhibits tumor‐induced regulatory T‐cell proliferation in colorectal cancer. Cancer Res. 2013;73(2):539–49. [DOI] [PubMed] [Google Scholar]
- 62.Wada J, Suzuki H, Fuchino R, Yamasaki A, Nagai S, Yanai K, et al. The contribution of vascular endothelial growth factor to the induction of regulatory T‐cells in malignant effusions. Anticancer Res. 2009;29(3):881–8. [PubMed] [Google Scholar]
- 63.Zhu P, Hu C, Hui K, Jiang X. The role and significance of VEGFR2(+) regulatory T cells in tumor immunity. Onco Targets Ther. 2017;10:4315–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tesi RJ. MDSC; the most important cell you have never heard of. Trends Pharmacol Sci. 2019;40(1):4–7. [DOI] [PubMed] [Google Scholar]
- 65.Baumann T, Dunkel A, Schmid C, Schmitt S, Hiltensperger M, Lohr K, et al. Regulatory myeloid cells paralyze T cells through cell‐cell transfer of the metabolite methylglyoxal. Nat Immunol. 2020;21(5):555–66. [DOI] [PubMed] [Google Scholar]
- 66.Kortylewski M, Kujawski M, Wang T, Wei S, Zhang S, Pilon‐Thomas S, et al. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat Med. 2005;11(12):1314–21. [DOI] [PubMed] [Google Scholar]
- 67.Kusmartsev S, Eruslanov E, Kubler H, Tseng T, Sakai Y, Su Z, et al. Oxidative stress regulates expression of VEGFR1 in myeloid cells: Link to tumor‐induced immune suppression in renal cell carcinoma. J Immunol. 2008;181(1):346–53. [DOI] [PubMed] [Google Scholar]
- 68.Guislain A, Gadiot J, Kaiser A, Jordanova ES, Broeks A, Sanders J, et al. Sunitinib pretreatment improves tumor‐infiltrating lymphocyte expansion by reduction in intratumoral content of myeloid‐derived suppressor cells in human renal cell carcinoma. Cancer Immunol Immunother. 2015;64(10):1241–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Finke J, Ko J, Rini B, Rayman P, Ireland J, Cohen P. MDSC as a mechanism of tumor escape from sunitinib mediated anti‐angiogenic therapy. Int Immunopharmacol. 2011;11(7):856–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jain RK. Antiangiogenesis strategies revisited: From starving tumors to alleviating hypoxia. Cancer Cell. 2014;26(5):605–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Huang Y, Goel S, Duda DG, Fukumura D, Jain RK. Vascular normalization as an emerging strategy to enhance cancer immunotherapy. Cancer Res. 2013;73(10):2943–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jain RK. Normalizing tumor vasculature with anti‐angiogenic therapy: A new paradigm for combination therapy. Nat Med. 2001;7(9):987–9. [DOI] [PubMed] [Google Scholar]
- 73.Martin JD, Seano G, Jain RK. Normalizing function of tumor vessels: Progress, opportunities, and challenges. Annu Rev Physiol. 2019;81:505–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tian L, Goldstein A, Wang H, Ching Lo H, Sun Kim I, Welte T, et al. Mutual regulation of tumour vessel normalization and immunostimulatory reprogramming. Nature. 2017;544(7649):250–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zheng X, Fang Z, Liu X, Deng S, Zhou P, Wang X, et al. Increased vessel perfusion predicts the efficacy of immune checkpoint blockade. J Clin Invest. 2018;128(5):2104–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yasuda S, Sho M, Yamato I, Yoshiji H, Wakatsuki K, Nishiwada S, et al. Simultaneous blockade of programmed death 1 and vascular endothelial growth factor receptor 2 (VEGFR2) induces synergistic anti‐tumour effect in vivo. Clin Exp Immunol. 2013;172(3):500–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Allen E, Jabouille A, Rivera LB, Lodewijckx I, Missiaen R, Steri V, et al. Combined antiangiogenic and anti‐PD‐L1 therapy stimulates tumor immunity through HEV formation. Sci Transl Med. 2017;9(385). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wu FT, Man S, Xu P, Chow A, Paez‐Ribes M, Lee CR, et al. Efficacy of cotargeting angiopoietin‐2 and the VEGF pathway in the adjuvant postsurgical setting for early breast, colorectal, and renal cancers. Cancer Res. 2016;76(23):6988–7000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Gordan JD, Kennedy EB, Abou‐Alfa GK, Beg MS, Brower ST, Gade TP, et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline. J Clin Oncol. 2020;38(36):4317–45. [DOI] [PubMed] [Google Scholar]
- 80.Zhu AX, Finn RS, Ikeda M, Sung MW, Baron AD, Kudo M, et al. A phase Ib study of lenvatinib (LEN) plus pembrolizumab (PEMBRO) in unresectable hepatocellular carcinoma (uHCC). J Clin Oncol. 2020;38(15_suppl):4519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894–905. [DOI] [PubMed] [Google Scholar]
- 82.Lee MS, Ryoo B‐Y, Hsu C‐H, Numata K, Stein S, Verret W, et al. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): An open‐label, multicentre, phase 1b study. Lancet Oncol. 2020;21(6):808–20. [DOI] [PubMed] [Google Scholar]
- 83.Vogel A, Saborowski A. Current strategies for the treatment of intermediate and advanced hepatocellular carcinoma. Cancer Treat Rev. 2020;82:101946. [DOI] [PubMed] [Google Scholar]
- 84.Arbour KC, Riely GJ. Systemic therapy for locally advanced and metastatic non‐small cell lung cancer: A review. JAMA. 2019;322(8):764–74. [DOI] [PubMed] [Google Scholar]
- 85.Reck M, Rodriguez‐Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A, et al. Pembrolizumab versus chemotherapy for PD‐L1‐positive non‐small‐cell lung cancer. N Engl J Med. 2016;375(19):1823–33. [DOI] [PubMed] [Google Scholar]
- 86.Gandhi L, Rodriguez‐Abreu D, Gadgeel S, Esteban E, Felip E, De Angelis F, et al. Pembrolizumab plus chemotherapy in metastatic non‐small‐cell lung cancer. N Engl J Med. 2018;378(22):2078–92. [DOI] [PubMed] [Google Scholar]
- 87.Lee CK, Man J, Lord S, Links M, Gebski V, Mok T, et al. Checkpoint inhibitors in metastatic EGFR‐mutated non‐small cell lung cancer‐A meta‐analysis. J Thorac Oncol. 2017;12(2):403–7. [DOI] [PubMed] [Google Scholar]
- 88.Garassino MC, Cho B‐C, Kim J‐H, Mazières J, Vansteenkiste J, Lena H, et al. Durvalumab as third‐line or later treatment for advanced non‐small‐cell lung cancer (ATLANTIC): An open‐label, single‐arm, phase 2 study. Lancet Oncol. 2018;19(4):521–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gainor JF, Shaw AT, Sequist LV, Fu X, Azzoli CG, Piotrowska Z, et al. EGFR mutations and ALK rearrangements are associated with low response rates to PD‐1 pathway blockade in non‐small cell lung cancer: A retrospective analysis. Clin Cancer Res. 2016;22(18):4585–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, et al. Atezolizumab for first‐line treatment of metastatic nonsquamous NSCLC. N Engl J Med. 2018;378(24):2288–301. [DOI] [PubMed] [Google Scholar]
- 91.Reck M, Mok TSK, Nishio M, Jotte RM, Cappuzzo F, Orlandi F, et al. Atezolizumab plus bevacizumab and chemotherapy in non‐small‐cell lung cancer (IMpower150): Key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomised, open‐label phase 3 trial. The Lancet Respiratory Medicine. 2019;7(5):387–401. [DOI] [PubMed] [Google Scholar]
- 92.Rini BI, Powles T, Atkins MB, Escudier B, McDermott DF, Suarez C, et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): A multicentre, open‐label, phase 3, randomised controlled trial. Lancet North Am Ed. 2019;393(10189):2404–15. [DOI] [PubMed] [Google Scholar]
- 93.Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal‐cell carcinoma. N Engl J Med. 2019;380(12):1116–27. [DOI] [PubMed] [Google Scholar]
- 94.Choueiri TK, Motzer RJ, Rini BI, Haanen J, Campbell MT, Venugopal B, et al. Updated efficacy results from the JAVELIN Renal 101 trial: First‐line avelumab plus axitinib versus sunitinib in patients with advanced renal cell carcinoma. Ann Oncol. 2020;31(8):1030–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Motzer RJ, Grünwald V, Hutson TE, Porta C, Powles T, Eto M, et al. A phase 3 trial to compare efficacy and safety of lenvatinib in combination with everolimus or pembrolizumab versus sunitinib alone in first‐line treatment of patients with metastatic renal cell carcinoma. J Clin Oncol. 2018;36(6_suppl):TPS706. [Google Scholar]
- 96.Choueiri TK, Apolo AB, Powles T, Escudier B, Aren OR, Shah A, et al. A phase 3, randomized, open‐label study of nivolumab combined with cabozantinib vs sunitinib in patients with previously untreated advanced or metastatic renal cell carcinoma (RCC; CheckMate 9ER). J Clin Oncol. 2018;36(15_suppl):TPS4598. [Google Scholar]
- 97.Green AK, Feinberg J, Makker V. A review of immune checkpoint blockade therapy in endometrial cancer. Am Soc Clin Oncol Educ Book. 2020;40:1–7. [DOI] [PubMed] [Google Scholar]
- 98.Makker V, Taylor MH, Aghajanian C, Oaknin A, Mier J, Cohn AL, et al. Lenvatinib (LEN) plus pembrolizumab (PEMBRO) for early‐line treatment of advanced/recurrent endometrial cancer (EC). J Clin Oncol. 2020;38(15_suppl):6083. [Google Scholar]
- 99.Liu JF, Herold C, Gray KP, Penson RT, Horowitz N, Konstantinopoulos PA, et al. Assessment of combined nivolumab and bevacizumab in relapsed ovarian cancer: A phase 2 clinical trial. JAMA Oncol. 2019;5(12):1731–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zsiros E, Frederick PJ, Akers SN, Attwood K, Wang K, Lele SB, et al. A phase II trial of pembrolizumab in combination with bevacizumab and oral metronomic cyclophosphamide for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer. Gynecol Oncol. 2019;154:23. [Google Scholar]
- 101.Mettu NB, Twohy E, Ou FS, Halfdanarson TR, Lenz HJ, Breakstone R, et al. BACCI: A phase II randomized, double‐blind, multicenter, placebo‐controlled study of capecitabine (C) bevacizumab (B) plus atezolizumab (A) or placebo (P) in refractory metastatic colorectal cancer (mCRC): An ACCRU network study. Ann Oncol. 2019;30:v203. [Google Scholar]
- 102.Kottke T, Hall G, Pulido J, Diaz RM, Thompson J, Chong H, et al. Antiangiogenic cancer therapy combined with oncolytic virotherapy leads to regression of established tumors in mice. J Clin Invest. 2010;120(5):1551–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Currier MA, Eshun FK, Sholl A, Chernoguz A, Crawford K, Divanovic S, et al. VEGF blockade enables oncolytic cancer virotherapy in part by modulating intratumoral myeloid cells. Mol Ther. 2013;21(5):1014–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Bocca P, Di Carlo E, Caruana I, Emionite L, Cilli M, De Angelis B, et al. Bevacizumab‐mediated tumor vasculature remodelling improves tumor infiltration and antitumor efficacy of GD2‐CAR T cells in a human neuroblastoma preclinical model. Oncoimmunology. 2017;7(1):e1378843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Renner DN, Malo CS, Jin F, Parney IF, Pavelko KD, Johnson AJ. Improved treatment efficacy of antiangiogenic therapy when combined with picornavirus vaccination in the GL261 glioma model. Neurotherapeutics. 2016;13(1):226–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Huang X, Wong MK, Yi H, Watkins S, Laird AD, Wolf SF, et al. Combined therapy of local and metastatic 4T1 breast tumor in mice using SU6668, an inhibitor of angiogenic receptor tyrosine kinases, and the immunostimulator B7.2‐IgG fusion protein. Cancer Res. 2002;62(20):5727–35. [PubMed] [Google Scholar]
- 107.Rini BI, Halabi S, Rosenberg JE, Stadler WM, Vaena DA, Archer L, et al. Phase III trial of bevacizumab plus interferon alfa versus interferon alfa monotherapy in patients with metastatic renal cell carcinoma: Final results of CALGB 90206. J Clin Oncol. 2010;28(13):2137–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Escudier B, Bellmunt J, Negrier S, Bajetta E, Melichar B, Bracarda S, et al. Phase III trial of bevacizumab plus interferon alfa‐2a in patients with metastatic renal cell carcinoma (AVOREN): Final analysis of overall survival. J Clin Oncol. 2010;28(13):2144–50. [DOI] [PubMed] [Google Scholar]
- 109.Amin A, Dudek AZ, Logan TF, Lance RS, Holzbeierlein JM, Knox JJ, et al. Survival with AGS‐003, an autologous dendritic cell‐based immunotherapy, in combination with sunitinib in unfavorable risk patients with advanced renal cell carcinoma (RCC): Phase 2 study results. J Immunother Cancer. 2015;3(1):14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Forster V. Phase 3 trial for oncolytic viral therapy pexa‐vec in advanced liver cancer terminated early. [May 3, 2021]. Available from: https://www.cancertherapyadvisor.com/home/cancer‐topics/general‐oncology/phase‐3‐trial‐for‐oncolytic‐viral‐therapy‐pexa‐vec‐in‐advanced‐liver‐cancer‐terminated‐early/.
- 111.Garcia J, Hurwitz HI, Sandler AB, Miles D, Coleman RL, Deurloo R, et al. Bevacizumab (Avastin(R)) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat Rev. 2020;86:102017. [DOI] [PubMed] [Google Scholar]
- 112.Ozao‐Choy J, Ma G, Kao J, Wang GX, Meseck M, Sung M, et al. The novel role of tyrosine kinase inhibitor in the reversal of immune suppression and modulation of tumor microenvironment for immune‐based cancer therapies. Cancer Res. 2009;69(6):2514–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Liu XD, Hoang A, Zhou L, Kalra S, Yetil A, Sun M, et al. Resistance to antiangiogenic therapy is associated with an immunosuppressive tumor microenvironment in metastatic renal cell carcinoma. Cancer Immunol Res. 2015;3(9):1017–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.McDermott DF, Huseni MA, Atkins MB, Motzer RJ, Rini BI, Escudier B, et al. Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat Med. 2018;24(6):749–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhu AX, Guan Y, Abbas AR, Koeppen H, Lu S, Hsu C‐H, et al. Genomic correlates of clinical benefits from atezolizumab combined with bevacizumab vs. atezolizumab alone in patients with advanced hepatocellular carcinoma (HCC). Cancer Res. 2020;80(16 Suppl):Abstract nr CT044. [Google Scholar]
- 116.Ferrara N, Mass RD, Campa C, Kim R. Targeting VEGF‐A to treat cancer and age‐related macular degeneration. Annu Rev Med. 2007;58:491–504. [DOI] [PubMed] [Google Scholar]
- 117.Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear‐cell renal‐cell carcinoma. N Engl J Med. 2007;356(2):125–34. [DOI] [PubMed] [Google Scholar]
- 118.Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, et al. Sunitinib versus interferon alfa in metastatic renal‐cell carcinoma. N Engl J Med. 2007;356(2):115–24. [DOI] [PubMed] [Google Scholar]
- 119.Jung K, Heishi T, Incio J, Huang Y, Beech EY, Pinter M, et al. Targeting CXCR4‐dependent immunosuppressive Ly6C(low) monocytes improves antiangiogenic therapy in colorectal cancer. Proc Natl Acad Sci USA. 2017;114(39):10455–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Jung K, Heishi T, Khan OF, Kowalski PS, Incio J, Rahbari NN, et al. Ly6Clo monocytes drive immunosuppression and confer resistance to anti‐VEGFR2 cancer therapy. J Clin Invest. 2017;127(8):3039–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Rahbari NN, Kedrin D, Incio J, Liu H, Ho WW, Nia HT, et al. Anti‐VEGF therapy induces ECM remodeling and mechanical barriers to therapy in colorectal cancer liver metastases. Sci Transl Med. 2016;8(360). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Levin VA, Mendelssohn ND, Chan J, Stovall MC, Peak SJ, Yee JL, et al. Impact of bevacizumab administered dose on overall survival of patients with progressive glioblastoma. J Neurooncol. 2015;122(1):145–50. [DOI] [PubMed] [Google Scholar]
- 123.Lin YY, Tan CT, Chen CW, Ou DL, Cheng AL, Hsu C. Immunomodulatory effects of current targeted therapies on hepatocellular carcinoma: Implication for the future of immunotherapy. Semin Liver Dis. 2018;38(4):379–88. [DOI] [PubMed] [Google Scholar]
- 124.Nishino M, Ramaiya NH, Hatabu H, Hodi FS. Monitoring immune‐checkpoint blockade: Response evaluation and biomarker development. Nat Rev Clin Oncol. 2017;14(11):655–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.


