Abstract
Naringin (Nar) is one of the natural glycosides extracted from pomelo and other citrus fruits. It has various pharmacological activities, including anti-inflammatory, antioxidant, anti-proliferative and anti-cancer. However, the underlying mechanisms by which Nar regulates apoptosis and autophagy in gastric cancer remain unclear. Thus, the present study aimed to assess the therapeutic effect of Nar and the underlying mechanisms. SNU-1 cell proliferation was determined using Cell Counting Kit-8 assay. Cell morphological changes were observed under a phase-contrast microscope. The changes in the cell cycle were determined using flow cytometry analysis and the changes in cell apoptosis were determined using flow cytometry, Hoechst 33258 and TUNEL staining. The protein levels pertaining to the PI3K/AKT pathway and cell apoptosis and autophagy were monitored using western blot analysis. The results demonstrated that Nar significantly inhibited SNU-1 cell growth and induced cell cycle arrest in the G0/G1 phase and cell apoptosis. Further mechanistic studies demonstrated that Nar blocked the PI3K/AKT pathway, activated cell autophagy and stimulated the expression of apoptosis-associated protein cleaved caspase 3 and Bax, but decreased the expression of Bcl-2. Preincubating SNU-1 cells with 3-methyladenine, a cell-autophagy inhibitor, significantly alleviated the effects of Nar in promoting cell apoptosis and cleaved caspase 3 expression. It was concluded that Nar promoted SNU-1 cell apoptosis via blocking the PI3K/AKT signaling pathway and activating cell autophagy.
Keywords: apoptosis, cell autophagy, gastric carcinoma SNU-1 cells, naringin, PI3K/AKT
Introduction
Gastric cancer, one of the commonest malignancies, is the third leading cause of cancer-related deaths worldwide (1). The incidence and mortality rates of gastric cancer are declining every year due to the improved control of related risk factors and the development of screening techniques (2). However, the incidence rate of gastric cancer remains high due to the large population base of the world and the trend of population aging (3). The gastric cancer cases and deaths in China account for 50% of the total gastric cancer cases in the world. In China, the incidence rate of gastric cancer is higher in northwestern and eastern coastal areas than in southern regions (4,5). Gastric cancer not only damages the digestive system but also affects the liver, kidney and respiratory functions once metastasis occurs. In severe cases, gastric cancer may lead to cachexia and ultimately become life-threatening (6). Surgery is the main treatment method for gastric cancer. Numerous patients with gastric cancer have witnessed an improvement in their conditions following surgery combined with chemo- or radiotherapies. However, a number of patients still suffer from recurrences and metastases after initial treatment. The effects of treatment and the 5-year survival rate are unsatisfactory (7). Therefore, new drugs and therapies for the clinical treatment of gastric cancer need to be urgently developed.
A number of studies have demonstrated that Chinese herbal medicines can effectively kill cancer cells and possess fewer problems in drug-resistance and toxic side effects (8). Naringin (4′,5,7-trihydroxyflavanone-7-rhamnoside, Nar; Fig. 1A) is a natural glycoside, also called bioflavonoid, found in pomelo and other citrus fruits (9). Previous studies have shown that Nar has various pharmacological activities, including anti-inflammatory, antioxidant and cardioprotective effects and can regulate glucose and lipid metabolism (10,11). Accumulating studies have also confirmed the anti-cancer activities of Nar through eliminating free radicals via its antioxidant effects and inhibiting oncogene expression and then inhibiting cancer cell proliferation and inducing cancer cell apoptosis (12–14). Nar can induce apoptosis and inhibit cancer cell growth in colorectal cancer, cervical cancer, ovarian cancer, liver cancer and other types of cancer (15–18). However, the effect of Nar on gastric cancer SNU-1 cells and its mechanism remain unclear.
Figure 1.
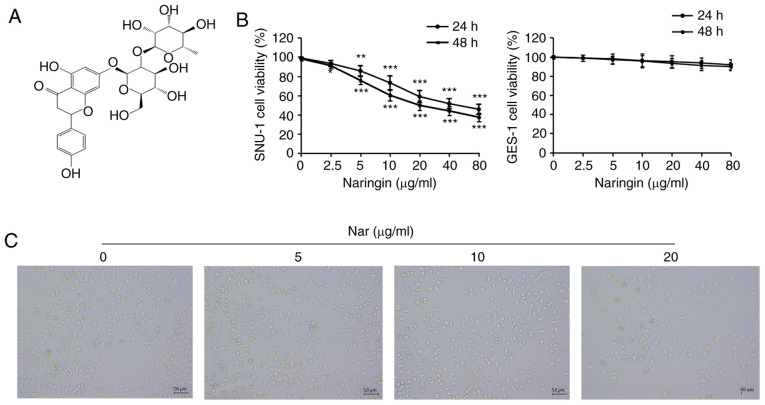
Naringin inhibits the proliferation of SNU-1 cells and induced cell cycle arrest. (A) Chemical structure of naringin; molecular formula: C27H32O14. (B) The viability of SNU-1 cells treated with different concentrations of costunolide for 24 and 48 h, as detected by CCK-8 assay. *P<0.05, **P<0.01, ***P<0.001 vs. the control group. (C) The morphology of SNU-1 cells was observed using an inverted phase contrast microscope (magnification, ×200).
The present study demonstrated the anti-cancer effect of Nar and its potential molecular mechanism through experimental research on gastric cancer cell line SNU-1, thus providing a new theoretical basis for further research on the anti-cancer effect of Nar and related molecular mechanisms.
Materials and methods
Experimental reagents
Gastric carcinoma cell lines (SNU-1; cat. no. CL-0474), normal human gastric epithelial cells (GES-1; cat. no. CL-0563) and fetal bovine serum (FBS) were purchased from Procell Life Science & Technology Co., Ltd. RPMI-1640 medium was purchased from Gibco (Thermo Fisher Scientific, Inc.). The Hoechst 33258 staining solution (C1017), TUNEL Apoptosis Assay kit (C1088) and BCA Protein Assay Kit (P0012) were procured from Beyotime Institute of Biotechnology. The rabbit anti-human cysteinyl aspartate specific proteinase (caspase 3) antibody (1:1,000; cat. no. ab179517), rabbit anti-human Bax (1:1,000; cat. no. ab32503), rabbit anti-human Bcl-2 (1:2,000; cat. no. ab182858), rabbit anti-human microtubule-associated protein 1 light chain 3β (LC3B; 1:3,000; cat. no. ab51520), rabbit anti-human Beclin (1:2,000; cat. no. ab207612), rabbit anti-human p62 (1:500; cat. no. ab155686), rabbit anti-human phosphorylated (p-)-PI3K (1:1,000; cat. no. ab138364), rabbit anti-human PI3K (1:1,000; cat. no. ab32089), rabbit anti-human AKT antibody (1:500; cat. no. ab8805), rabbit anti-human p-AKT antibody (1:500; cat. no. ab38449) and rabbit anti-human GAPDH antibody (1:2,500; cat. no. ab9485) were obtained from Abcam. Horseradish peroxidase (HRP)-labeled goat anti-rabbit immunoglobulin G (IgG) antibody (1:2,000; cat. no. CW0103) was purchased from Cwbio. Naringin (cat. no. HY-N0153) and 3-methyladenine (3-MA; cat. no. HY-19312) were purchased from MedChemExpress. Annexin V-fluorescein isothiocyanate (FITC) apoptosis detection kits (cat. no. KGA106) and cell cycle detection kits (cat. no. KGA511) were purchased from Keygentec. Radio-Immunoprecipitation Assay (RIPA) solution (cat. no. P0013) was obtained from Beyotime Institute of Biotechnology.
Cell culture
All cells were maintained in the RPMI-1640 medium supplemented with 10% FBS and 100 U/ml penicillin and 100 µg/ml streptomycin in an incubator at 37°C with a 5% CO2 atmosphere. In the present study, SNU-1 cells without costunolide (Cos) treatment (0 µg/ml) group served as the control group.
Cell Counting Kit-8 (CKK-8) assay
Cell proliferation was analyzed using CCK-8 solution. SNU-1 and GES-1 cells (100 µl/well, 1.5×105 cells/ml) were seeded in a 96-well plate and incubated in a CO2 incubator for 24 h at 37°C. After aspiration, the cells were incubated with different concentrations (0, 2.5, 5, 10, 20, 40 and 80 µg/ml) of Nar in FBS-free RPMI 1640 for 24 h and then CCK-8 assay was used according to the manufacturer's protocol. Optical density values were measured with a microplate reader at 450 nm.
Observation of cell morphology
The SNU-1 cells were treated with different concentrations (0, 5, 10 and 20 µg/ml) of Nar in FBS-free RPMI 1640 for 24 h and observed and images captured using an inverted light microscope (magnification, ×100; Olympus Corporation).
Hoechst 33258 staining
Cell apoptosis was analyzed using Hoechst 33258 staining. SNU-1 cells were seeded into 12-well plates and treated with different concentrations (0, 5, 10 and 20 µg/ml) of Nar for 24 h. The adherent cells were washed twice with phosphate-buffered saline. The cells were then stained with Hoechst 33258 for 5 min at room temperature and washed twice. The blue-stained nuclei was observed under the BX41 fluorescence microscope (magnification, ×100; Olympus Corporation). Images were captured and used to quantitatively analyze the apoptosis of cells using Image-Pro Plus analysis software 6.0 (Media Cybernetics, Inc.).
TUNEL staining
Cell apoptosis was analyzed using TUNEL staining. SNU-1 cells were plated onto 12-well plates for 24 h and treated with different concentrations (0, 5, 10 and 20 µg/ml) of Nar for 24 h. Then, apoptosis was evaluated using the TUNEL apoptosis assay kit and observed under the BX41 fluorescence microscope (magnification, ×100; Olympus Corporation). Images were captured and used to quantitatively analyze the apoptosis of cells using Image-Pro Plus analysis software 6.0 (Media Cybernetics, Inc.).
Flow cytometry
Cell cycle and apoptosis were measured by flow cytometry. The cell cycle was analyzed using an Annexin V-FITC/propidium iodide (PI) apoptosis kit. The six-well plate was seeded with SNU-1 cells (2.0 ml/well, 1.0×106 cells/ml) cultured for 24 h. After aspiration, the cells were incubated with 2.0 ml of different concentrations (0, 5, 10 and 20 µg/ml) of Nar for 24 h, or treated with Nar before pretreatment with 3-MA, following which the collected cells were fixed with 75% ethanol at 4°C overnight. The cell cycle detection kit was used following the manufacturer's protocols. The cell samples were mixed with PI for 15 min at 37°C in the dark and apoptotic cells were examined with a BD FACSaria Fusion flow cytometer (BD Biosciences) and ModFit software version 3.2 (BD Biosciences). The apoptotic rate was calculated as the percentage of early and late apoptotic cells.
Western blot analysis
The changes in apoptosis-related proteins (caspase 3, Bax and Bcl2), signaling pathway-related proteins (AKT, phosphorylated (p-)AKT, PI3K and p-PI3K) and autophagy-related proteins (LC3B, Beclin-1 and p62) in SNU-1 cells were analyzed using western blot analysis. After incubation with Nar, the cells were collected and proteins were extracted on ice with RIPA lysis buffer containing protease inhibitors. Proteins were quantified with the BCA Protein Assay kit. The collected lysate samples (20 µg/well) were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) on 12% gels and transferred to nitrocellulose membranes blocked with 50 g/l skimmed milk for 2 h at room temperature. Following overnight incubation with the primary antibodies of caspase 3, Bax, Bcl-2, AKT, p-AKT, PI3K, p-PI3K, LC3B, Beclin-1, p62 and GAPDH at 4°C, the membranes were incubated with an HRP-labeled goat anti-rabbit IgG antibody for 2 h at room temperature. In a darkroom, the SuperSignal ELISA Femto Substrate was added onto the membranes which were subsequently exposed to x-ray films. Protein bands were imaged using an Alpha Innotech FluorChem FC2 Imaging System (ProteinSimple). The densitometric analysis was performed using ImageJ software v1.46 (National Institutes of Health) and GAPDH expression was used to normalize the data.
Wound-healing assay
SNU-1 cells (2.0 ml/well, 1×106 cells/ml) were seeded to confluence in 6-well plates; when the confluence of the cells reached ~90% wounds were made by a 200-µl pipette tip, then the cells were incubated in a serum-free medium with different concentrations (0, 10, 20, 40 µg/ml) of Cos for 24 h, observed and images captured using an inverted light microscope (magnification, ×100; Olympus Corporation). Quantitation of wound healing assay results were analyzed by Image-Pro Plus software v 6.0 (Media Cybernetics, Inc.).
Statistical analysis
All data were shown as mean ± standard error of mean. The intergroup deviations were evaluated using one-way analysis of variance (ANOVA) implemented in the GraphPad Prism 6.0 software. Comparison between groups was performed using one-way ANOVA followed by Tukey's test. P<0.05 was considered to indicate a statistically significant difference.
Results
Effects of Nar on SNU-1 cell proliferation
The changes in cell proliferation were determined using CCK-8 assay to demonstrate the effects of Nar on SNU-1 cell proliferation. As shown in Fig. 1B, Nar could significantly inhibit the proliferation of SNU-1 cells in a dose-dependent manner, but the effect of Nar on normal gastric cells (GES-1 cells) was not as sensitive as that on SNU-1 cells (Fig. 1B). The half-maximal inhibitory concentration (IC50) for both 24 and 48 h was ~20 µg/ml. Therefore, the concentrations of 0, 5, 10 and 20 µg/ml were used for subsequent in vitro assays. The results of observing the cell morphology with a phase-contrast microscope showed that Nar induced shrinkage, nuclear lysis and rupture of SNU-1 cells (Fig. 1C).
Nar induces SNU-1 cell cycle arrest
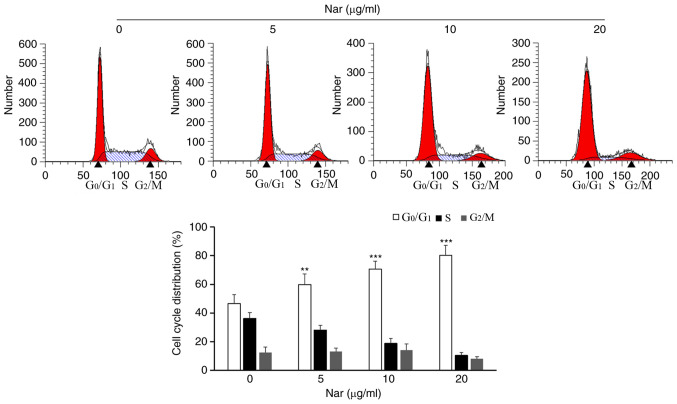
Based on the results of anti-proliferative assays, SNU-1 cells were treated with 0, 5, 10 and 20 µg/ml Nar to reveal the effect of Nar on the cell cycle. The flow cytometry results showed that Nar could significantly induce cell cycle arrest in the G0/G1 phase in SNU-1 cells dose-dependently (Fig. 2).
Figure 2.
Effects of Nar on the SNU-1 cell cycle were determined by flow cytometry. **P<0.01, ***P<0.001 vs. the control group. Nar, naringin.
Nar induces the apoptosis of SNU-1 cells
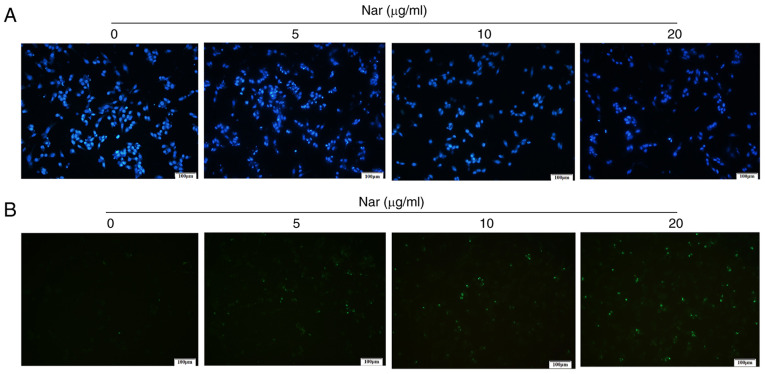
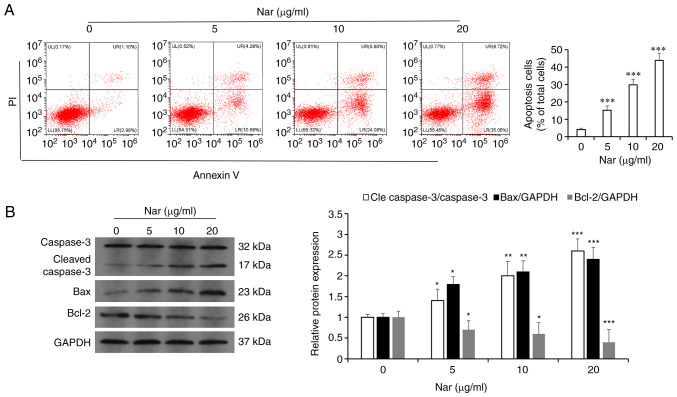
Hoechst 33258 and TUNEL staining demonstrated that the rate of apoptosis increased with an increase in Nar concentration (Fig. 3A and B). The flow cytometry results showed that Nar could dose-dependently induce the apoptosis of SNU-1 cells (Fig. 4A). Western blot analysis results also demonstrated that Nar upregulated the expression of caspase 3 and Bax, but downregulated the expression of Bcl-2 in a dose-dependent manner (Fig. 4B).
Figure 3.
Nar induces apoptosis of SNU-1 cells. (A) Hoechst 33258 staining (magnification, ×100). (B) TUNEL staining (magnification, ×100). Nar, naringin.
Figure 4.
SNU-1 cell apoptosis as determined by flow cytometry and western blot analysis. (A) The apoptosis of Nar induced in SNU-1 cells was analyzed by flow cytometry. (B) The apoptosis of Nar induced in SNU-1 cells was analyzed by western blot analysis. GAPDH expression was used to normalize the data. *P<0.05, **P<0.01, ***P<0.001 vs. the control group. Nar, naringin.
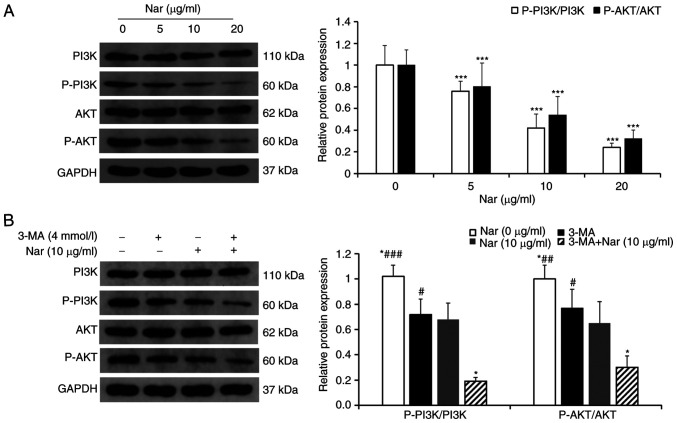
Nar induces apoptosis of SNU-1 cells via inhibiting the PI3K/AKT pathway
The changes in PI3K/AKT pathway-related proteins treated with different concentrations of Nar were detected using western blot analysis. The results showed that the ratios p-PI3K/PI3K and p-AKT/AKT were downregulated in a dose-dependent manner (Fig. 5A). After pretreatment with 3-MA, an inhibitor of PI3K, the ratios of p-PI3K/PI3K and p-AKT/AKT in cells co-treated with 3-MA and Nar were markedly downregulated compared with those in cells treated with Nar or 3-MA alone (Fig. 5B).
Figure 5.
Nar induces apoptosis and autophagy of SNU-1 cells by inhibiting the PI3K/AKT signaling pathway. (A) Changes in cellular PI3K, p-PI3K, AKT and p-AKT levels were determined by western blot analysis. GAPDH expression was used to normalize the data. (B) Changes in PI3K, p-PI3K, AKT and p-AKT levels in SNU-1 cells following Nar (10 µg/ml), 3-MA and Nar (10 µg/ml) plus 3-MA treatment. GAPDH expression was used to normalize the data. *P<0.05, ***P<0.001 vs. Nar (10 µg/ml) group; #P<0.05, ##P<0.01, ###P<0.001 vs. Nar (10 µg/ml) plus 3-MA group. Nar, naringin; p-, phosphorylated.
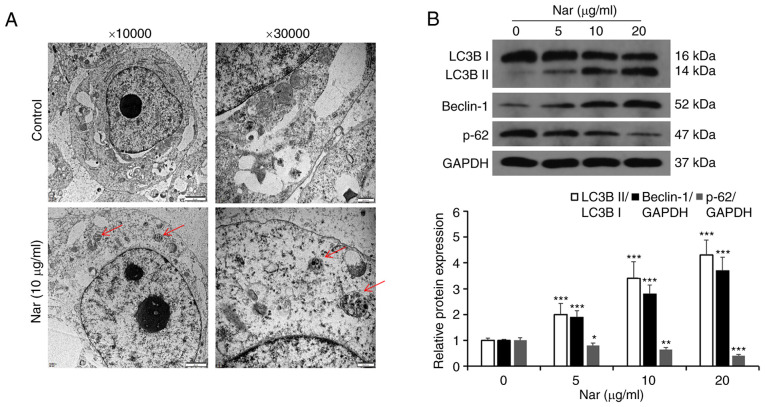
Nar induces apoptosis via activating autophagy in SNU-1 cells
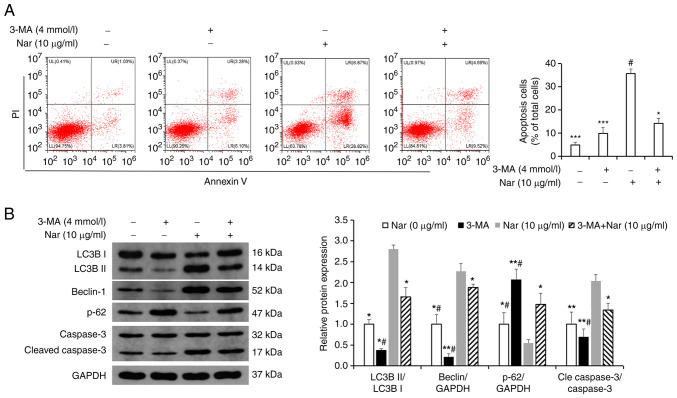
Transmission electron microscopy results showed the formation of autophagic vacuoles in SNU-1 cells following treatment with Nar (Fig. 6A). Western blot analysis results showed that Nar could promote the expression of Beclin-1, increase the LC3BII/LC3BI ratio and inhibit the expression of p62 (Fig. 6B). SNU-1 cells were treated with 10 µg/ml Nar for 24 h before incubating with 4 mmol/l 3-MA (an autophagy inhibitor) for 1 h and the apoptosis were measured by flow cytometry. The results showed that Nar-induced apoptosis was significantly attenuated in the 3-MA- and Nar-co-treated group compared with the Nar-treated group (Fig. 7A). Western blot analysis results also showed that the Beclin-1 expression level and the LC3BII/LC3BI ratio were markedly downregulated and the expression of p62 was upregulated in 3-MA-treated SNU-1 cells. However, when cells were co-stimulated with 3-MA and Nar, Beclin-1 expression level and LC3BII/LC3BI ratio significantly increased and the p62 degradation was rescued compared with those in cells treated with 3-MA alone (Fig. 7B).
Figure 6.
Nar induces autophagy in SNU-1 cells. (A) The autophagic microstructures were observed by transmission electron microscopy [red arrow: Autophagic vacuole (autophagosome or autophagosome)]. (B) The levels of cell autophagy-associated proteins were determined by western blot analysis. GAPDH expression was used to normalize the data. *P<0.05, **P<0.01, ***P<0.001 vs. the control group. Nar, naringin; LC3B, microtubule-associated protein 1 light chain 3 β.
Figure 7.
Changes in SNU-1 cell apoptosis after inhibiting autophagy with 3-MA. (A) Changes in the apoptosis of SNU-1 cells after Nar (10 µg/ml), 3-MA and Nar (10 µg/ml) plus 3-MA treatment were analyzed by flow cytometry analysis. (B) Changes in autophagy-related proteins and caspase 3 of SNU-1 cells after Nar (10 µg/ml), 3-MA and Nar (10 µg/ml) plus 3-MA treatment were analyzed by western blot analysis. GAPDH expression was used to normalize the data. *P<0.05, **P<0.01, ***P<0.001 vs. Nar (10 µg/ml) group; #P<0.05 vs. Nar (10 µg/ml) plus 3-MA group. Nar, naringin; LC3B, microtubule-associated protein 1 light chain 3 β; p-, phosphorylated.
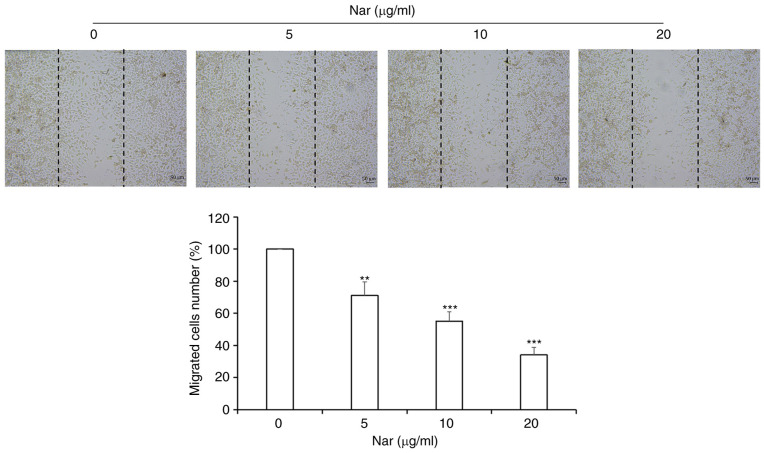
Nar inhibits migration of SNU-1 cells
Wound-healing assay results demonstrated that Nar inhibited the migration of SNU-1 cells in a dose-dependent manner (Fig. 8).
Figure 8.
Nar inhibits migration of SNU-1 cells (magnification, ×100). **P<0.01, ***P<0.001 vs. the control group. Nar, naringin.
Discussion
Gastric cancer is one of the commonest and most malignant tumors in the digestive system (19). Although the treatment of gastric cancer has improved to a certain level with the advancement of medical technology, the survival rate of patients with gastric cancer remains poor. Therefore, seeking more effective adjuvant treatment to improve the therapeutic effect on GC is urgently required for patients undergoing surgery and chemotherapy. Some studies have shown that ~60% of cancer patients in the United States take high-dose complex antioxidant nutrients while undergoing conventional treatment to improve the effect of conventional anti-cancer treatment and reduce the side effects of radiotherapy and chemotherapy (20,21). Oxidative nutrients (antioxidants) refer to a class of nutrients that have antioxidant capacity, inhibit the generation of free radicals, accelerate the elimination of free radicals and inhibit the oxidative damage of biological macromolecules caused by free radicals (20). Green vegetables such as cauliflower are rich sources of antioxidant nutrients, such as vitamin A, vitamin C, vitamin E and their derivatives, while citrus fruits are rich in flavonoids, isoflavones and saponins (9). At present, a large number of cell culture experiments, animal experiments and some clinical trials have confirmed that compound antioxidant nutrients have positive effects in anti-cancer treatment (20). In addition, studies have found that a number of plant-derived molecules can exert their anti-cancer effects by targeting specific signaling pathways (22,23). Therefore, extracting pure natural drugs from plants to develop more effective and nontoxic anti-cancer agents is an ideal and promising avenue. The Nar used in the present study is a dihydroflavonoid compound extracted from citrus fruits. Its biologically active substances are not only are plentiful in content but various, mainly including flavonoids, limonoids, carotenoids, coumarins, essential oils, dietary fiber and pectin (24). Nar has various biological activities, such as anti-inflammatory, anti-viral, anti-cancer, anti-mutation, anti-allergic, anti-ulcer, analgesic and antihypertensive. Gastric cancer has a number of risk factors, including gastric ulcer, atrophic gastritis and Helicobacter pylori infection. Nar can suppress these risk factors, which is particularly important in the prevention and adjuvant treatment of gastric cancer. In recent years, some anti-cancer active ingredients in Nar have become research targets in the food and medical fields (10,25). They can inhibit the growth of cancer cells via different molecular mechanisms, such as triggering cell cycle arrest, apoptosis, necrosis and autophagy (26). Studies have found that Nar initiates the release of TNF by inducing lipopolysaccharides, reduces the incidence of liver cancer, induces cancer cell apoptosis and inhibits oncogene expression through anti-oxidative and anti-free radical effects (27,28). However, the effect of Nar on gastric cancer SNU-1 cells and the related mechanism have been seldom studied. The present study demonstrated that Nar inhibited SNU-1 cell proliferation in a dose- and time-dependent manner. The inhibition of cell proliferation is considered to be specific to gastric cancer because Nar has no obvious inhibitory effect on normal gastric mucosal GES-1 cells, indicating that it has no cytotoxicity on human normal gastric cells. In addition, the present study found that Nar had an anti-proliferative effect on SNU-1 cells at low concentrations, as observed under an inverted microscope. This indicated that if Nar is supplemented continuously for a period of time, it would be effective even at lower concentrations. Nar has an anti-cancer effect on esophageal cancer stem cell xenotransplanted tumor mouse models without altering the body and liver weight and the combination of Nar and doxorubicin can reduce the side effects of doxorubicin (29). Nar is both effective and safe as an anti-cancer drug (30). The present study found that Nar arrested SNU-1 cells in the G0/G1 phase, promoting apoptosis in a dose-dependent manner. Further mechanistic studies demonstrated that Nar significantly blocked the PI3K/AKT signaling pathway and activated autophagy. 3-MA pretreatment significantly attenuated Nar-induced apoptosis. The results suggested the potential relevance of consuming Nar-rich foods or nutrients in reducing the development of gastric cancer.
Tumors are characterized by abnormal cell proliferation. Various anti-tumor drugs induce tumor cell cycle arrest and apoptosis, thereby inhibiting the abnormal proliferation of tumor cells and exerting anti-cancer activities (31–33). Nar can induce cell cycle arrest and apoptosis in human breast cancer (34). In cervical cancer cells, Nar induces G0/G1 phase arrest and activated endoplasmic reticulum-mediated apoptosis (35). It also induces G0/G1 phase arrest and apoptosis in human osteosarcoma MG63 and U2OS cells (36). The results confirmed that Nar significantly inhibited SNU-1 cells and induced G0/G1 phase arrest and apoptosis in a concentration-dependent manner. In addition, the results confirmed that Nar promoted the generation of cleaved caspase 3 in SNU-1 cells. The aforementioned results indicated that Nar inhibited SNU-1 cell proliferation by promoting the G0/G1 phase arrest and apoptosis. This was consistent with the results of previous studies showing that flavonoids, such as apigenin, luteolin and myricetin, induced exogenous apoptosis in different cancer cell lines (37–39).
The PI3K/AKT pathway is a classic signaling pathway involved in regulating a variety of cellular processes, including proliferation, migration, differentiation and apoptosis (40). Previous studies have identified anti-cancer drugs inducing apoptosis by blocking the PI3K/AKT signaling pathway (41,42). In colorectal cancer, Nar inhibits cell growth and induces apoptosis via blocking the PI3K/AKT signaling pathway (15). By targeting this pathway, Nar inhibits the proliferation of thyroid cancer cells and induces apoptosis (43). The mechanism may be that activated Akt can promote the Ser184 phosphorylation of Bax, which can negatively regulate the pro-apoptotic function and could also inactivate caspase-9 Ser196 phosphorylation and inhibit apoptosis (32,33). The results of the present study showed that Nar could significantly induce SNU-1 cell apoptosis by inhibiting the PI3K/AKT signaling pathway in SNU-1 cells.
Autophagy is a lysosomal degradation pathway of the cell. It is characterized by an increase in the number of acidic vesicle organelles associated with autophagosomes, which is often dysregulated in cancer as another important form of programmed cell death (44). Autophagy can both promote cell death and inhibit cell death. In tumor cells, the effect of autophagy often depends on the cell type (45). Previous studies have confirmed autophagy as an important signal downstream of the PI3K/AKT pathway; it is involved in drug-induced cancer cell apoptosis (46,47). Recent studies have confirmed that Nar activates autophagy by inhibiting PI3K/AKT signal, thereby inhibiting the growth of gastric cancer cells (48). The results of the present study confirmed that Nar significantly activated autophagy, featured by the expression of autophagy-related proteins LC3BII and Beclin 1 increased, while the expression of p62 decreased in a dose-dependent manner. This was inconsistent with reports that apigenin (which is a flavonoid) could induce autophagy and promote the increase in p62 expression. The p62 protein is located on the autophagosome by LC3 binding and it is degraded by autophagy (49). The overexpression of p62 can activate caspase 8 and promote cell apoptosis, which is related to the ubiquitin-associated domain at the C terminal (50). This indicates that besides being a marker of autophagy activation, p62 protein also served as an important regulator of apoptosis. Inhibiting autophagy significantly attenuated the generation of caspase 3 spliceosome induced by Nar. Based on these results, it was confirmed that PI3K/AKT signaling and autophagy were involved in the process of Nar-induced apoptosis in SNU-1 cells. Finally, the anti-gastric cancer metastatic effect of Nar was investigated using a wound healing test. The drug inhibited the migration of SNU-1 cells, further confirming that Nar had an anti-gastric cancer effect. However, its mechanism and whether Nar has the potential to overcome drug resistance in gastric cancer in the same manner as S-adenosyl-1-methionine needs to be studied in follow-up experiments (50). In addition, the experiments were performed only in a specific cancer cell line, SNU-1. At least one key finding should be reproduced in a different cancer cell line. Therefore, the use of one cell line as a limitation of the present study will be improved in the future studies. These future studies intend to use single-cell sequencing to screen out the targets of Nar acting on gastric cancer cells and further reveal its mechanism of action. In addition, the combination of Nar and other Chinese medicine monomers is being currently studied, to find the best combination for the treatment of gastric cancer.
In summary, Nar significantly inhibited the growth of SNU-1 cells, inducing G0/G1 phase arrest and apoptosis. Moreover, it induced SNU-1 cell apoptosis by inhibiting the PI3K/AKT signaling and activating autophagy. The present study was based on the potential application of Nar as a protective nutrient and chemopreventive and therapeutic molecule, confirming that Nar was a potential drug for the treatment of gastric cancer.
Our research group has conducted research on the anti-tumor mechanisms of luteolin, costanolactone and oleandrin. The combined effect and mechanism of several drugs will be further explored in detail. The findings are expected to lay the foundation for the development of anti-cancer plant nutrition sources in food science.
Acknowledgements
Not applicable.
Funding Statement
The present study was supported by the Shaanxi Science and Technology Innovation Team (grant no. 2017KCT-28), the Shaanxi Province Key R&D Project (grant no. 2019ZDLSF02-09-01) and the National Natural Science Foundation of China (grant no. 81900686).
Funding
The present study was supported by the Shaanxi Science and Technology Innovation Team (grant no. 2017KCT-28), the Shaanxi Province Key R&D Project (grant no. 2019ZDLSF02-09-01) and the National Natural Science Foundation of China (grant no. 81900686).
Availability of data and materials
All data generated or analyzed in the present study are included in this published article.
Authors' contributions
CX, XH, JW and XD conceived and designed the experiments. CX, XH, YH, XL and MW performed the experiments. CX, XH and JW analyzed the data. CX, JW and XD performed data interpretation and made critical manuscript revisions. CX and XH wrote the manuscript and confirmed the authenticity of all the raw data. All authors reviewed and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Strand MS, Lockhart AC, Fields RC. Genetics of gastric cancer. Surg Clin North Am. 2017;97:345–370. doi: 10.1016/j.suc.2016.11.009. [DOI] [PubMed] [Google Scholar]
- 3.Hamashima C. Current issues and future perspectives of gastric cancer screening. World J Gastroenterol. 2014;20:13767–13774. doi: 10.3748/wjg.v20.i38.13767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zong L, Abe M, Seto Y, Ji J. The challenge of screening for early gastric cancer in China. Lancet. 2016;388:2606. doi: 10.1016/S0140-6736(16)32226-7. [DOI] [PubMed] [Google Scholar]
- 5.Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 6.Roder DM. The epidemiology of gastric cancer. Gastric Cancer. 2002;5(Suppl 1):S5–S11. doi: 10.1007/s10120-002-0203-6. [DOI] [PubMed] [Google Scholar]
- 7.Ang TL, Fock KM. Clinical epidemiology of gastric cancer. Singapore Med J. 2014;55:621–628. doi: 10.11622/smedj.2014174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kuo YT, Chang TT, Muo CH, Wu MY, Sun MF, Yeh CC, Yen HR. Use of complementary traditional Chinese medicines by adult cancer patients in Taiwan: A nationwide population-based study. Integr Cancer Ther. 2018;17:531–541. doi: 10.1177/1534735417722224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Singh N, Bansal Y, Bhandari R, Marwaha L, Singh R, Chopra K, Kuhad A. Naringin reverses neurobehavioral and biochemical alterations in intracerebroventricular collagenase-induced intracerebral hemorrhage in rats. Pharmacology. 2017;100:172–187. doi: 10.1159/000453580. [DOI] [PubMed] [Google Scholar]
- 10.Chen R, Qi QL, Wang MT, Li QY. Therapeutic potential of naringin: An overview. Pharm Biol. 2016;54:3203–3210. doi: 10.1080/13880209.2016.1216131. [DOI] [PubMed] [Google Scholar]
- 11.Bharti S, Rani N, Krishnamurthy B, Arya DS. Preclinical evidence for the pharmacological actions of naringin: A review. Planta Med. 2014;80:437–451. doi: 10.1055/s-0034-1368351. [DOI] [PubMed] [Google Scholar]
- 12.Jeon SM, Bok SH, Jang MK, Kim YH, Nam KT, Jeong TS, Park YB, Choi MS. Comparison of antioxidant effects of naringin and probucol in cholesterol-fed rabbits. Clin Chim Acta. 2002;317:181–190. doi: 10.1016/S0009-8981(01)00778-1. [DOI] [PubMed] [Google Scholar]
- 13.Jagetia GC, Reddy TK. Modulation of radiation-induced alteration in the antioxidant status of mice by naringin. Life Sci. 2005;77:780–794. doi: 10.1016/j.lfs.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 14.Rajadurai M, Stanely Mainzen Prince P. Preventive effect of naringin on lipid peroxides and antioxidants in isoproterenol-induced cardiotoxicity in Wistar rats: Biochemical and histopathological evidences. Toxicology. 2006;228:259–268. doi: 10.1016/j.tox.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 15.Cheng H, Jiang X, Zhang Q, Ma J, Cheng R, Yong H, Shi H, Zhou X, Ge L, Gao G. Naringin inhibits colorectal cancer cell growth by repressing the PI3K/AKT/mTOR signaling pathway. Exp Ther Med. 2020;19:3798–3804. doi: 10.3892/etm.2020.8649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ramesh E, Alshatwi AA. Naringin induces death receptor and mitochondria-mediated apoptosis in human cervical cancer (SiHa) cells. Food Chem Toxicol. 2013;51:97–105. doi: 10.1016/j.fct.2012.07.033. [DOI] [PubMed] [Google Scholar]
- 17.Cai L, Wu H, Tu C, Wen X, Zhou B. Naringin inhibits ovarian tumor growth by promoting apoptosis: An in vivo study. Oncol Lett. 2018;16:59–64. doi: 10.3892/ol.2018.8611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Banjerdpongchai R, Wudtiwai B, Khawon P. Induction of human hepatocellular carcinoma HepG2 cell apoptosis by naringin. Asian Pac J Cancer Prev. 2016;17:3289–3294. [PubMed] [Google Scholar]
- 19.Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635–648. doi: 10.1016/S0140-6736(20)31288-5. [DOI] [PubMed] [Google Scholar]
- 20.Ilghami R, Barzegari A, Mashayekhi MR, Letourneur D, Crepin M, Pavon-Djavid G. The conundrum of dietary antioxidants in cancer chemotherapy. Nutr Rev. 2020;78:65–76. doi: 10.1093/nutrit/nuz027. [DOI] [PubMed] [Google Scholar]
- 21.McDermott JH. Antioxidant nutrients: Current dietary recommendations and research update. J Am Pharm Assoc (Wash) 2000;40:785–799. doi: 10.1016/S1086-5802(16)31126-3. [DOI] [PubMed] [Google Scholar]
- 22.Nijveldt RJ, van Nood E, van Hoorn DE, Boelens PG, van Norren K, van Leeuwen PA. Flavonoids: A review of probable mechanisms of action and potential applications. Am J Clin Nutr. 2001;74:418–425. doi: 10.1093/ajcn/74.4.418. [DOI] [PubMed] [Google Scholar]
- 23.Courdavault V, O'Connor SE, Oudin A, Besseau S, Papon N. Towards the microbial production of plant-derived anticancer drugs. Trends Cancer. 2020;6:444–448. doi: 10.1016/j.trecan.2020.02.004. [DOI] [PubMed] [Google Scholar]
- 24.Wang X, Ouyang Y, Liu J, Zhu M, Zhao G, Bao W, Hu FB. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ. 2014;349:g4490. doi: 10.1136/bmj.g4490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Alam MA, Subhan N, Rahman MM, Uddin SJ, Reza HM, Sarker SD. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv Nutr. 2014;5:404–417. doi: 10.3945/an.113.005603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mani JS, Johnson JB, Hosking H, Ashwath N, Walsh KB, Neilsen PM, Broszczak DA, Naiker M. Antioxidative and therapeutic potential of selected Australian plants: A review. J Ethnopharmacol. 2021;268:113580. doi: 10.1016/j.jep.2020.113580. [DOI] [PubMed] [Google Scholar]
- 27.Blankson H, Grotterød EM, Seglen PO. Prevention of toxin-induced cytoskeletal disruption and apoptotic liver cell death by the grapefruit flavonoid, naringin. Cell Death Differ. 2000;7:739–746. doi: 10.1038/sj.cdd.4400705. [DOI] [PubMed] [Google Scholar]
- 28.Kawaguchi K, Kikuchi S, Hasegawa H, Maruyama H, Morita H, Kumazawa Y. Suppression of lipopolysaccharide-induced tumor necrosis factor-release and liver injury in mice by naringin. Eur J Pharmacol. 1999;368:245–250. doi: 10.1016/S0014-2999(98)00867-X. [DOI] [PubMed] [Google Scholar]
- 29.Tajaldini M, Samadi F, Khosravi A, Ghasemnejad A, Asadi J. Protective and anticancer effects of orange peel extract and naringin in doxorubicin treated esophageal cancer stem cell xenograft tumor mouse model. Biomed Pharmacother. 2020;121:109594. doi: 10.1016/j.biopha.2019.109594. [DOI] [PubMed] [Google Scholar]
- 30.Tarun EI, Kurchenko VP, Metelitsa DI. Flavonoids as effective protectors of urease from ultrasonic inactivation in solutions. Bioorg Khim. 2006;32:391–398. doi: 10.1134/s1068162006040054. (In Russian) [DOI] [PubMed] [Google Scholar]
- 31.Liu Y, Kang X, Niu G, He S, Zhang T, Bai Y, Li Y, Hao H, Chen C, Shou Z, Li B. Shikonin induces apoptosis and prosurvival autophagy in human melanoma A375 cells via ROS-mediated ER stress and p38 pathways. Artif Cells Nanomed Biotechnol. 2019;47:626–635. doi: 10.1080/21691401.2019.1575229. [DOI] [PubMed] [Google Scholar]
- 32.Kang X, Wang H, Li Y, Xiao Y, Zhao L, Zhang T, Zhou S, Zhou X, Li Y, Shou Z, et al. Alantolactone induces apoptosis through ROS-mediated AKT pathway and inhibition of PINK1-mediated mitophagy in human HepG2 cells. Artif Cells Nanomed Biotechnol. 2019;47:1961–1970. doi: 10.1080/21691401.2019.1593854. [DOI] [PubMed] [Google Scholar]
- 33.Qiu C, Zhang T, Zhang W, Zhou L, Yu B, Wang W, Yang Z, Liu Z, Zou P, Liang G. Licochalcone a inhibits the proliferation of human lung cancer cell lines A549 and H460 by inducing G2/M cell cycle arrest and ER stress. Int J Mol Sci. 2017;18:1761. doi: 10.3390/ijms18081761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kabala-Dzik A, Rzepecka-Stojko A, Kubina R, Iriti M, Wojtyczka RD, Buszman E, Stojko J. Flavonoids, bioactive components of propolis, exhibit cytotoxic activity and induce cell cycle arrest and apoptosis in human breast cancer cells MDA-MB-231 and MCF-7-a comparative study. Cell Mol Biol (Noisy-le-grand) 2018;64:1–10. doi: 10.14715/cmb/2018.64.8.1. [DOI] [PubMed] [Google Scholar]
- 35.Lin R, Hu X, Chen S, Shi Q, Chen H. Naringin induces endoplasmic reticulum stress-mediated apoptosis, inhibits β-catenin pathway and arrests cell cycle in cervical cancer cells. Acta Biochim Pol. 2020;67:181–188. doi: 10.18388/abp.2020_5182. [DOI] [PubMed] [Google Scholar]
- 36.Ming H, Chuang Q, Jiashi W, Bin L, Guangbin W, Xianglu J. Naringin targets Zeb1 to suppress osteosarcoma cell proliferation and metastasis. Aging (Albany NY) 2018;10:4141–4151. doi: 10.18632/aging.101710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sang Eun H, Seong Min K, Ho Jeong L, Vetrivel P, Venkatarame Gowda Saralamma V, Jeong Doo H, Eun Hee K, Sang Joon L, Gon Sup K. Scutellarein induces Fas-mediated extrinsic apoptosis and G2/M cell cycle arrest in Hep3B hepatocellular carcinoma cells. Nutrients. 2019;11:263. doi: 10.3390/nu11020263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhang Y, Xu X, Li W, Miao H, Huang S, Zhou Y, Sun Y, Li Z, Guo Q, Zhao L. Activation of endoplasmic reticulum stress and the extrinsic apoptotic pathway in human lung cancer cells by the new synthetic flavonoid, LZ-205. Oncotarget. 2016;7:87257–87270. doi: 10.18632/oncotarget.13535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tavsan Z, Kayali HA. Flavonoids showed anticancer effects on the ovarian cancer cells: Involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomed Pharmacother. 2019;116:109004. doi: 10.1016/j.biopha.2019.109004. [DOI] [PubMed] [Google Scholar]
- 40.Porta C, Paglino C, Mosca A. Targeting PI3K/Akt/mTOR signaling in cancer. Front Oncol. 2014;4:64. doi: 10.3389/fonc.2014.00064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yang J, Ren X, Zhang L, Li Y, Cheng B, Xia J. Oridonin inhibits oral cancer growth and PI3K/Akt signaling pathway. Biomed Pharmacother. 2018;100:226–232. doi: 10.1016/j.biopha.2018.02.011. [DOI] [PubMed] [Google Scholar]
- 42.Zhu ML, Zhang PM, Jiang M, Yu SW, Wang L. Myricetin induces apoptosis and autophagy by inhibiting PI3K/Akt/mTOR signalling in human colon cancer cells. BMC Complement Med Ther. 2020;20:209. doi: 10.1186/s12906-020-02965-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhou J, Xia L, Zhang Y. Naringin inhibits thyroid cancer cell proliferation and induces cell apoptosis through repressing PI3K/AKT pathway. Pathol Res Pract. 2019;215:152707. doi: 10.1016/j.prp.2019.152707. [DOI] [PubMed] [Google Scholar]
- 44.Yun CW, Lee SH. The roles of autophagy in cancer. Int J Mol Sci. 2018;19:3466. doi: 10.3390/ijms19113466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Towers CG, Wodetzki D, Thorburn A. Autophagy and cancer: Modulation of cell death pathways and cancer cell adaptations. J Cell Biol. 2020;219:e201909033. doi: 10.1083/jcb.201909033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wei M, Wu Y, Liu H, Xie C. Genipin induces autophagy and suppresses cell growth of oral squamous cell carcinoma via PI3K/AKT/MTOR pathway. Drug Des Devel Ther. 2020;14:395–405. doi: 10.2147/DDDT.S222694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhou J, Jiang YY, Chen H, Wu YC, Zhang L. Tanshinone I attenuates the malignant biological properties of ovarian cancer by inducing apoptosis and autophagy via the inactivation of PI3K/AKT/mTOR pathway. Cell Prolif. 2020;53:e12739. doi: 10.1111/cpr.12739. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 48.Raha S, Yumnam S, Hong GE, Lee HJ, Saralamma VV, Park HS, Heo JD, Lee SJ, Kim EH, Kim JA, Kim GS. Naringin induces autophagy-mediated growth inhibition by downregulating the PI3K/Akt/mTOR cascade via activation of MAPK pathways in AGS cancer cells. Int J Oncol. 2015;47:1061–1069. doi: 10.3892/ijo.2015.3095. [DOI] [PubMed] [Google Scholar]
- 49.Liu WJ, Ye L, Huang WF, Guo LJ, Xu ZG, Wu HL, Yang C, Liu HF. p62 links the autophagy pathway and the ubiqutin-proteasome system upon ubiquitinated protein degradation. Cell Mol Biol Lett. 2016;21:29. doi: 10.1186/s11658-016-0031-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang YB, Gong JL, Xing TY, Zheng SP, Ding W. Autophagy protein p62/SQSTM1 is involved in HAMLET-induced cell death by modulating apotosis in U87MG cells. Cell Death Dis. 2013;4:e550. doi: 10.1038/cddis.2013.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed in the present study are included in this published article.