Abstract
Aims
Coronavirus disease 2019 (COVID‐19) has been recognised as a predominantly respiratory tract infection, but some patients manifest severe systemic symptoms/coagulation abnormalities. The aim of this study was to evaluate the impact of severe COVID‐19 infection on the gastrointestinal tract.
Methods and results
We examined clinicopathological findings in 28 resected ischaemic bowels from 22 patients with severe COVID‐19. Most patients required intubation preoperatively and presented with acute decompensation shortly before surgery. D‐dimer levels were markedly elevated in all measured cases (mean, 5394 ng/ml). Histologically, 25 cases (19 patients) showed evidence of acute ischaemia with necrosis. In this group, the most characteristic finding was the presence of small vessel fibrin thrombi (24 of 25 cases, 96%), which were numerous in 64% of cases. Patients with COVID‐19 were significantly more likely than a control cohort of 35 non‐COVID‐19‐associated acute ischaemic bowels to show isolated small intestine involvement (32% versus 6%, P < 0.001), small vessel fibrin thrombi (100% versus 43%, P < 0.001), submucosal vessels with fibrinous degeneration and perivascular neutrophils (90% versus 54%, P < 0.001), fibrin strands within submucosal vessels (58% versus 20%, P = 0.007), and histological evidence of pneumatosis (74% versus 34%, P = 0.010). Three cases in this cohort had histopathological findings normally seen in the setting of chronic ischaemia, notably prominent fibroblastic proliferation affecting the outer layer of the muscularis propria.
Conclusions
Herein, we describe the histopathological findings in COVID‐19‐associated ischaemic bowels and postulate a relationship with the hypercoagulable state seen in patients with severe COVID‐19 infection. Additional experience with these cases may further elucidate specific features or mechanisms of COVID‐19‐associated ischaemic enterocolitis.
Keywords: COVID‐19, fibrin thrombi, gastrointestinal, ischaemic enterocolitis, microthrombi
Introduction
Coronavirus disease 2019 (COVID‐19), which is caused by severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) and is currently a worldwide public health crisis, has been recognised as a predominantly respiratory tract infection. However, a subset of COVID‐19 patients develop severe systemic disease with manifestations such as acute respiratory failure, shock, and multiple organ dysfunction, leading to substantial mortality. 1 The reported frequencies of gastrointestinal (GI) symptoms in patients with COVID‐19 range widely, from 1% to 50%, with the most common symptoms being diarrhoea, nausea/vomiting, and abdominal pain. 2 , 3 , 4 , 5 , 6 , 7 The mechanism is not understood, but the SARS‐CoV‐2 viral receptor angiotensin‐converting enzyme 2 (ACE2) is expressed in epithelial cells throughout the GI tract, and viral RNA has been detected in stool samples from COVID‐19 patients, suggesting that SARS‐CoV‐2 can infect the GI tract. 2 , 3 , 4 , 6 , 7 , 8
Patients with severe COVID‐19 can also manifest coagulation abnormalities that have some similarities with other systemic coagulopathies associated with severe infections, such as disseminated intravascular coagulation (DIC) and thrombotic microangiopathy. Initial studies reported the incidence of thromboembolic complications in COVID‐19 patients to be 35–45%. 9 The typical findings in COVID‐19 patients with coagulopathy are a mildly prolonged prothrombin time (PT), mild‐to‐moderate thrombocytopenia, and markedly elevated D‐dimer levels. 10 , 11 , 12 , 13 , 14 Although this pattern is suggestive of DIC, it is distinct from the DIC associated with sepsis, in which the thrombocytopenia is more pronounced and the D‐dimer level is not elevated to nearly the dramatically high levels seen in COVID‐19 patients. 11 In COVID‐19 patients, coagulopathy was shown to be a strong predictor of mortality, with 71% of non‐survivors meeting the International Society on Thrombosis and Haemostasis criteria for DIC, as compared with only 0.4% of survivors. 12 Early series of COVID‐19 autopsies in the USA showed the presence of fibrin thrombi and CD61+ megakaryocytes/platelet aggregates within small‐to‐medium‐sized vessels of the pulmonary parenchyma and multiple other organ systems. 15 , 16 , 17
The impact of COVID‐19‐associated coagulopathy on the GI tract has not yet been investigated in detail. At our institution, a large academic tertiary‐care‐level centre, abdominal imaging was performed in 33% of inpatients with COVID‐19: 31% of computed tomography (CT) scans showed bowel wall abnormalities, and 20% of CTs in intensive care unit patients showed signs of ischaemia. 18 In addition, an analysis of 141 critically ill COVID‐19 patients over a 1‐month period at our institution showed that 74% of these patients had at least one GI complication during their admission; of those, ⁓5% of cases developed bowel ischaemia that necessitated surgical intervention. 19 Herein, we present the clinicopathological findings in this cohort of COVID‐19 patients with resected segments of ischaemic bowel, and discuss the pathological findings in the context of our understanding of COVID‐19 coagulopathy and other histopathological considerations in the differential diagnosis.
Materials and methods
STUDY POPULATION
The study was approved by the Massachusetts General Hospital Institutional Review Board. Twenty‐eight ischaemic bowel resection specimens from 22 COVID‐19 patients were identified from March 2020 to March 2021. Demographic information, clinical and radiological data and intraoperative and postoperative follow‐up findings were collected from patient records. In order to determine whether the changes in the COVID‐19 bowels were simply a reflection of ischaemic bowel in general, a group of control ischaemic bowel resection specimens from patients without COVID‐19 was compiled by searching the surgical files for ischaemic bowel resections in the surgical pathology archives from 2017 to June 2019. Of the initial cases identified, those secondary to venous outflow obstruction (volvulus, closed loop obstruction, etc.) or that were markedly haemorrhagic (precluding evaluation of specific small vessel pathological features) were excluded, resulting in a final control cohort of 35 cases.
PATHOLOGY EXAMINATION
All slides from each COVID‐19 case were reviewed and evaluated semiquantitatively by two to three pathologists (M.L.Z. and/or F.J. and J.M.) for numerous histopathological features, including: distribution, type and depth of necrosis; the presence, distribution and quantity of small vessel fibrin thrombi; small vessel changes; large‐vessel changes; the presence of pneumatosis; histological changes in the muscularis propria; and evidence of pre‐existing vascular disease. Histological evidence of pneumatosis included large empty spaces in the submucosa not lined by endothelium, often in areas of marked oedema of the surrounding tissue. ‘Few vessels’ was defined as vessels present only focally (e.g. one to two high‐power fields) on a single section or on up to a few histological sections. ‘Numerous vessels’ was defined as readily identified vessels, present in many fields on a single section and/or throughout many sections. Control cases were reviewed and evaluated for the same histopathological features as above. Any discrepancies were adjudicated by discussion at a multiheaded microscope.
IMMUNOHISTOCHEMISTRY AND IN‐SITU HYBRIDISATION (ISH)
Immunohistochemistry was performed with an automated stainer (Bond‐III; Leica Microsystems, Bannockburn, IL, USA) with SARS Nucleocapsid Protein Antibody [clone NB100‐56576 (1:300); Novus Biologicals, Littleton, CO, USA], in accordance with the manufacturer’s recommendations.
RNA ISH was performed on formalin‐fixed paraffin‐embedded (FFPE) sections with two methods. Chromogenic RNA ISH was performed with the SARS‐CoV‐2 RNA‐specific probe RNAscope 2.5 LS Probe‐V‐nCoV2019‐S [Advanced Cell Diagnostics (ACD), Newark, CA, USA; 848568] and RNAscope 2.5 LS Reagent Kit‐RED (ACD; 322150) on an automated BondRx platform (Leica Biosystems, Danvers, MA, USA). 20 The probe is specific for the S gene encoding the spike protein. ISH was performed on 5‐µm‐thick FFPE sections. All of the steps from baking for 1 h at 60°C to counterstaining with haematoxylin were performed on the BondRx machine. RNA unmarking was performed with Bond Epitope Retrieval Solution 2 for 15 min at 95°C, and this was followed by protease treatment for 15 min and probe hybridisation for 2 h. Signals were amplified by a series of signal amplification steps, and this was followed by colour development in red with Bond Polymer Refine Red Detection (Leica Biosystems). The reactivity was visualised as red dots. All of the samples, including negative controls, showed uniform and robust staining, confirming well‐preserved RNA.
The second method was a single‐molecule florescence ISH (smFISH) approach. In the smFISH experiments, the RNAscope Multiplex Fluorescent Reagent Kit v2 (ACD) was applied with the following incubation time modifications: protease incubation for 30 min and manual target retrieval for 25 min. The utilised hybridisation probes were the SARS‐CoV‐2‐specific probe RNAscope probe V‐nCoV2019‐S (ACD; 848561) and Hs‐ACE2 (ACD; 848151). The probe is specific for the S gene encoding the spike protein. Slides were counterstained with 4′,6‐diamidino‐2‐phenylindole, mounted, and imaged by the use of confocal microscopy. The SARS‐CoV‐2 RNA sense (+) probe was validated by evaluating human lung samples that were known to be infected with SARS‐CoV‐2. Expression of SARS‐CoV‐2 RNA was clearly visible in lung macrophages and in pneumocytes.
STATISTICAL ANALYSIS
Fisher exact tests were used to compare categorical variables, and two‐sided t‐tests were used to compare continuous variables. P‐values of <0.05 were considered to be statistically significant.
Results
CLINICAL FEATURES OF THE COVID‐19 COHORT WITH BOWEL ISCHAEMIA
The 22 patients who constituted the study cohort had a mean age of 57 years (range, 32–78 years), and 14 (64%) were male (Table 1). Seventeen patients (77%) had pre‐existing medical conditions related to metabolic syndrome. Four patients (18%) had a history of prior thrombosis: one patient had a history of deep vein thrombosis (DVT)/pulmonary embolism with an inferior vena cava filter in place; the second had a history of DVT and antineutrophil cytoplasmic autoantibody vasculitis; the third had a history of splenic vein thrombosis while receiving warfarin; and the fourth had a history of thrombotic thrombocytopenic purpura (TTP).
Table 1.
Clinical features of 22 coronavirus disease 2019 (COVID‐19) patients with ischaemic enterocolitis
| Features | Values | Normal ranges |
|---|---|---|
| Clinical characteristics | ||
| Age (years), mean (range) | 57 (32–78) | – |
| Sex, n (%) | ||
| Male | 14 (64) | – |
| Female | 8 (36) | – |
| Body mass index (kg/m2), mean (range) | 33 (21–61) | – |
| Pre‐existing medical conditions, n (%) | ||
| Hypertension, type 2 diabetes, and/or obesity | 17 (77) | – |
| Thrombosis | 4 (18) | – |
| History of malignancy | 4 (18) | – |
| Coronary artery disease | 3 (14) | – |
| Thrombotic thrombocytopenic purpura | 1 (5) | – |
| No pre‐existing medical condition | 2 (9) | – |
| Preoperative clinical parameters | ||
| Time since positive COVID‐19 test (days), mean (range) | 20 (6–89) | – |
| Intubation prior to abdominal surgery, n (%) | 20 (91) | – |
| Duration of intubation (days), mean (range) (n = 19) | 12 (0–25) | – |
| Preceding clinical course | ||
| Worsening | 14 (64) | – |
| Stable | 4 (18) | – |
| Improving | 4 (18) | – |
| COVID‐19 treatments, n (%) | ||
| Antibiotics | 20 (91) | – |
| Remdesivir | 11 (50) | – |
| Dexamethasone | 8 (36) | – |
| Tocilizumab | 3 (14) | – |
| Hydroxychloroquine | 2 (9) | – |
| Convalescent plasma | 1 (5) | – |
| Anticoagulation, n (%) | ||
| Prophylactic | 14 (64) | – |
| Therapeutic | 4 (18) | – |
| None | 4 (18) | – |
| Radiological modalities, n (%) | ||
| Abdominal computed tomography | 13 (59) | – |
| Abdominal X‐ray | 10 (46) | – |
| Right upper quadrant ultrasound | 4 (18) | – |
| Radiological findings, n (%) | ||
| Intestinal pneumatosis | 14 (64) | – |
| Dilated/distended bowel | 13 (59) | – |
| Ischaemic changes | 12 (55) | – |
| Portal venous gas | 8 (36) | – |
| Abdominal free air | 3 (14) | |
| Preoperative laboratory values, mean (range) | ||
| White blood cells (109/l) | 18.3 (5.5–38.5) | 4.5–11.0 |
| Platelets (109/l) | 280 (21–568) | 150–400 |
| Activated partial thromboplastin time (s) | 40.1 (27.5–85.3) | 22–36 (70–100 with heparin) |
| Prothrombin time (s) | 16.0 (13.4–22.6) | 11.5–14.5 |
| D‐dimer (ng/ml) (n = 21) | 5394 (973–22 022) | <500 |
| Fibrinogen (mg/dl) (n = 20) | 571 (221–1096) | 150–400 |
| Patient outcome, n (%) | ||
| Discharged in stable condition | 11 (50) | – |
| Died during hospitalisation | 9 (41) | – |
| Remains hospitalised postoperatively | 2 (9) | – |
Although the number of days between a positive COVID‐19 test result and emergent operation for ischaemic bowel ranged widely, from 6 to 89 days (mean, 20 days), almost all patients presented to the emergency room with worsening shortness of breath and significant hypoxaemia requiring oxygen support upon admission. Most patients were intubated prior to surgery (20/22, 91%), and another two patients went from the emergency room directly to the operating room for suspected bowel ischaemia, where they were subsequently intubated. All but two patients were treated with prophylactic antibiotics; half received remdesivir, and eight patients (36%) received dexamethasone. The majority of patients were given prophylactic anticoagulation (enoxaparin) during their preoperative inpatient stay, and therapeutic heparin was given preoperatively to four patients because of a rapidly increasing D‐dimer level (n = 2), arterial line clotting (n = 1), and pulmonary embolism (n = 1).
In terms of pertinent preoperative laboratory values, the mean peak white blood cell (WBC) count within 24 h prior to surgery was elevated, at 18.3 × 109/l (range, 5.5–38.5 × 109/l), with 12 patients (55%) showing a >50% increase in the WBC count during the preceding 24 h. Coagulation studies showed mildly elevated partial thromboplastin times, PTs, and fibrinogen levels; however, D‐dimer levels were markedly elevated, with a mean of 5394 ng/ml (upper limit of normal: 500 ng/ml).
Clinical factors that raised suspicion for bowel ischaemia and led to emergent surgery included a worsening clinical course (i.e. increased vasopressor requirement, and abdominal distention) and abdominal radiology showing distended bowel segments, intestinal pneumatosis, ischaemic changes, portal venous gas, and/or abdominal free air concerning for perforation. Three patients presented with clinical/radiological features of intestinal pseudo‐obstruction, including one patient with marked dilatation of the ascending and transverse colon with a transition point at the splenic flexure. Mesenteric thrombosis was not identified on imaging studies. On follow‐up, 11 patients (50%) were discharged in a stable condition, and nine patients (41%) died during hospitalisation owing to postoperative complications and/or worsening respiratory failure.
GROSS FINDINGS OF COVID‐19‐ASSOCIATED ISCHAEMIC BOWELS
A total of 28 resection specimens from 22 patients were examined. Four patients had two specimens each from separate operations, and one patient had three sequential specimens from operations on subsequent days. Resected bowel segments included 15 cases (54%) of the small intestine and colon (12 cases designated as the terminal ileum), nine cases (32%) of the small intestine only (three cases designated as the terminal ileum), and four cases (14%) of the large intestine only (Table 2). The most common external finding was serosal discolouration (25 cases, 89%), and approximately one‐third of specimens showed a curious tan–yellow discolouration that was noted intraoperatively (Figure 1). In some of those cases, the surgeon noted a pattern of discontinuous discolouration involving the antimesenteric border of the bowel. In addition, nine (32%) specimens showed significant bowel dilatation, four (14%) had palpable pneumatosis, and three (11%) had a grossly identified perforation. Upon opening of the bowel, 26 specimens (93%) showed mucosal discolouration and six (21%) had a visible mucosal exudate.
Table 2.
Intraoperative and gross pathology findings of 28 specimens from 22 patients with coronavirus disease 2019 (COVID‐19)‐associated ischaemic enterocolitis
| Findings | n (%) |
|---|---|
| Specimen type (N = 28) | |
| Small intestine only | 9 (32) |
| Small intestine and colon | 15 (54) |
| Colon/rectum only | 4 (14) |
| External findings | |
| Significant dilatation | 9 (32) |
| Perforation | 3 (11) |
| Pneumatosis | 4 (14) |
| Serosal adhesions | 9 (32) |
| Serosal exudate | 8 (29) |
| Serosal discolouration | 25 (89) |
| Dusky red/grey–brown | 17 (61) |
| Tan–yellow | 9 (32) |
| Grey–green | 3 (11) |
| Internal findings | |
| Mucosal exudate | 6 (21) |
| Mucosal discolouration | 26 (93) |
| Dusky red–purple/black | 13 (46) |
| Tan–yellow/green | 12 (46) |
| Grey–brown | 2 (7) |
Figure 1.
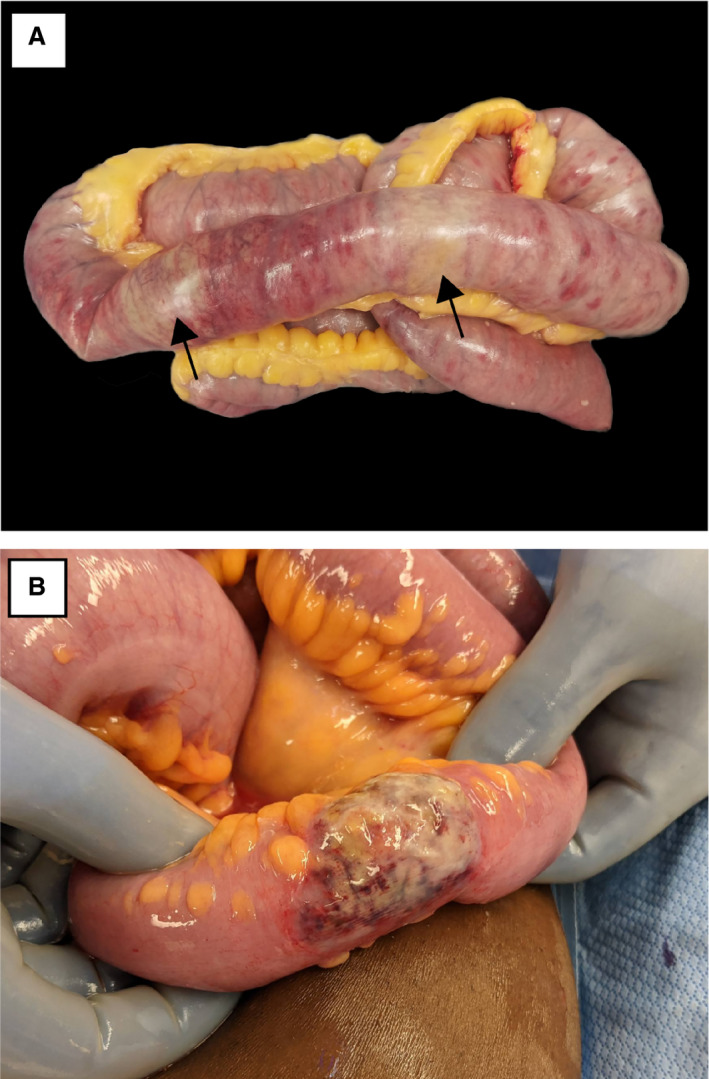
Gross appearance of coronavirus disease 2019‐associated ischaemic bowel. A, Small intestine showing extensive full‐thickness and antimesenteric necrosis with patchy yellow discolouration (arrows). B, Small intestine showing a well‐demarcated area of antimesenteric necrosis with yellowish discolouration.
HISTOPATHOLOGICAL FINDINGS OF COVID‐19‐ASSOCIATED ISCHAEMIC BOWELS
Histologically, 25 cases (19 patients) showed evidence of acute ischaemia. In this group, the pattern of mucosal necrosis varied from coagulative to haemorrhagic to purulent (including a pseudomembranous pattern), with most cases showing all three patterns. In a little over half of cases, the necrosis extended to involve the muscularis propria, either partially or transmurally (Table 3). Thirteen cases (52%) showed patchy/multifocal mucosal necrosis, often with sharply demarcated alternating necrotic and viable areas juxtaposed on the same slide (Figure 2A). Histological evidence suggestive of pneumatosis was seen in 15 cases (60%) (Figure 2B). The most characteristic histopathological finding was the presence of small vessel fibrin thrombi in 24 of 25 cases (96%), which were numerous in 64% of cases (Figure 3A,B). These fibrin thrombi were found in the submucosa (23 cases, 92%) and mucosa (19 cases, 76%), and were almost always in areas beneath necrotic mucosa. Phosphotungstic acid haematoxylin staining was performed in 12 cases, and highlighted fibrin thrombi in all of those cases (Figure 3C). In addition, in 23 (92%) cases, the submucosal small vessels in the markedly oedematous areas beneath necrotic mucosa showed fibrinous degeneration of the wall, with perivascular neutrophils, reminiscent of leukocytoclastic vasculitis (Figure 4A,B). In one case, the ischaemic changes were accompanied by unusual hyalinisation of the submucosal vessels (Figure 4C); a Congo red stain was negative. In 14 (56%) cases, medium‐sized arteries in the submucosa showed fibrin strands within the vessel lumen, in serum or admixed with red blood cells (Figure 5). No well‐formed fibrin thrombi or thromboemboli were seen within mesenteric vessels.
Table 3.
Histological findings of 25 specimens from 19 patients with acute coronavirus disease 2019 (COVID‐19)‐associated ischaemic enterocolitis
| Histological features (N = 25) | n (%) |
|---|---|
| Depth of necrosis | |
| Mucosa/submucosa | 11 (44) |
| Mural/transmural | 14 (56) |
| Distribution of necrosis | |
| Focal | 1 (4) |
| Multifocal/patchy | 13 (52) |
| Diffuse | 11 (44) |
| Histological evidence of pneumatosis | |
| Not present | 10 (40) |
| Present | 15 (60) |
| Quantity of small vessel fibrin thrombi | |
| None | 1 (4) |
| Rare/few | 8 (32) |
| Numerous | 16 (64) |
| Location of fibrin thrombi | |
| Mucosa | 19 (76) |
| Submucosa | 23 (92) |
| Muscularis propria | 2 (8) |
| Subserosa | 1 (4) |
| Small vessel fibrinous degeneration with perivascular neutrophils | |
| None | 2 (8) |
| Few vessels | 12 (48) |
| Numerous vessels | 11 (44) |
| Fibrin strands in submucosal vessels | |
| None | 11 (44) |
| Few vessels | 12 (48) |
| Numerous vessels | 2 (8) |
Figure 2.
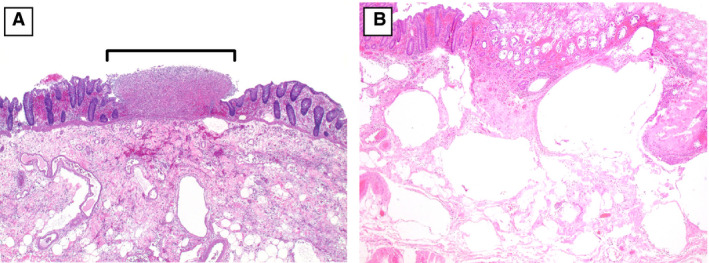
A, A segment of colon with a discrete, punctate area of necrosis (bracket) adjacent to viable mucosa [haematoxylin and eosin (H&E)]. B, Markedly oedematous submucosa with numerous large empty spaces, consistent with pneumatosis intestinalis (H&E).
Figure 3.
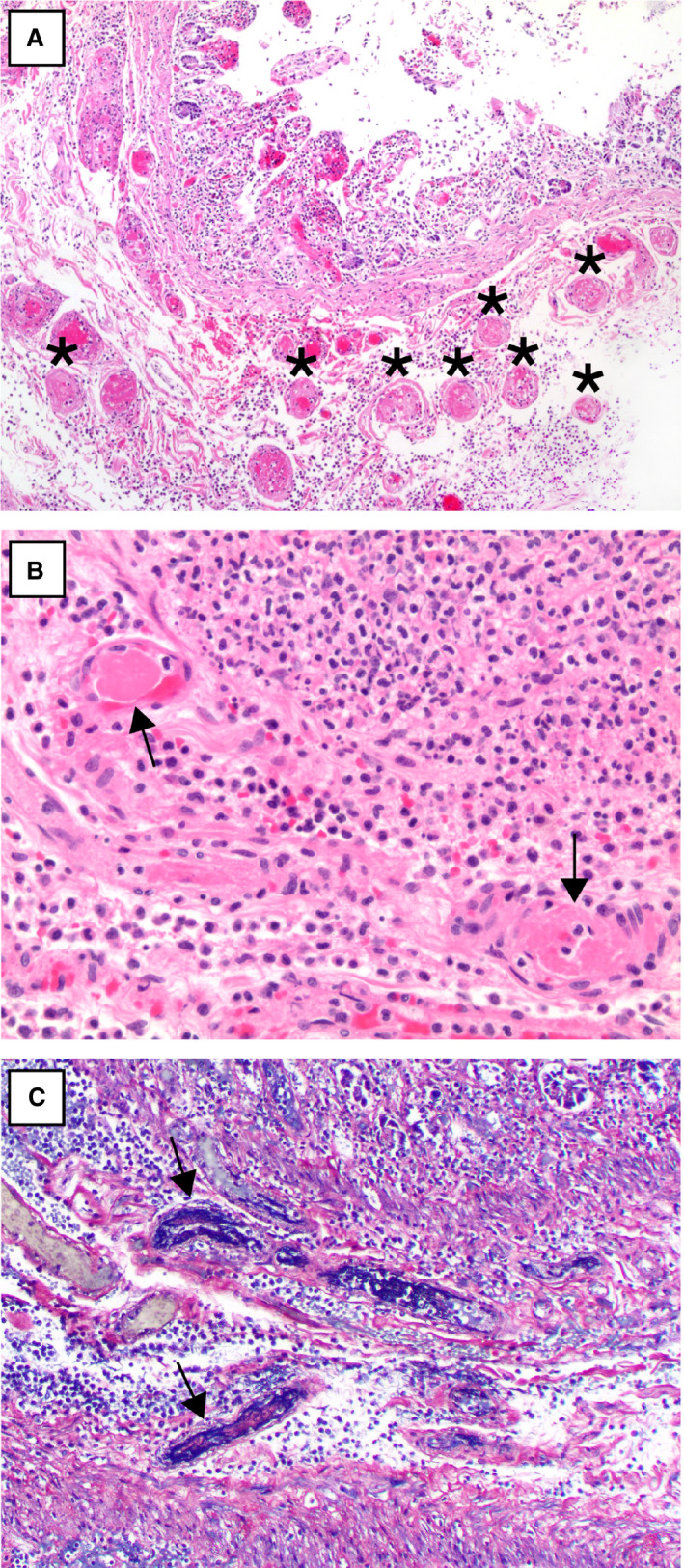
A, A segment of small intestine with numerous submucosal small vessel fibrin thrombi (asterisks) beneath an area of necrotic mucosa [haematoxylin and eosin (H&E)]. B, A higher‐power view of two well‐formed submucosal fibrin thrombi (arrows) (H&E). C, Phosphotungstic acid haematoxylin staining highlights fibrin thrombi in small vessels (arrows).[Colour figure can be viewed at wileyonlinelibrary.com]
Figure 4.
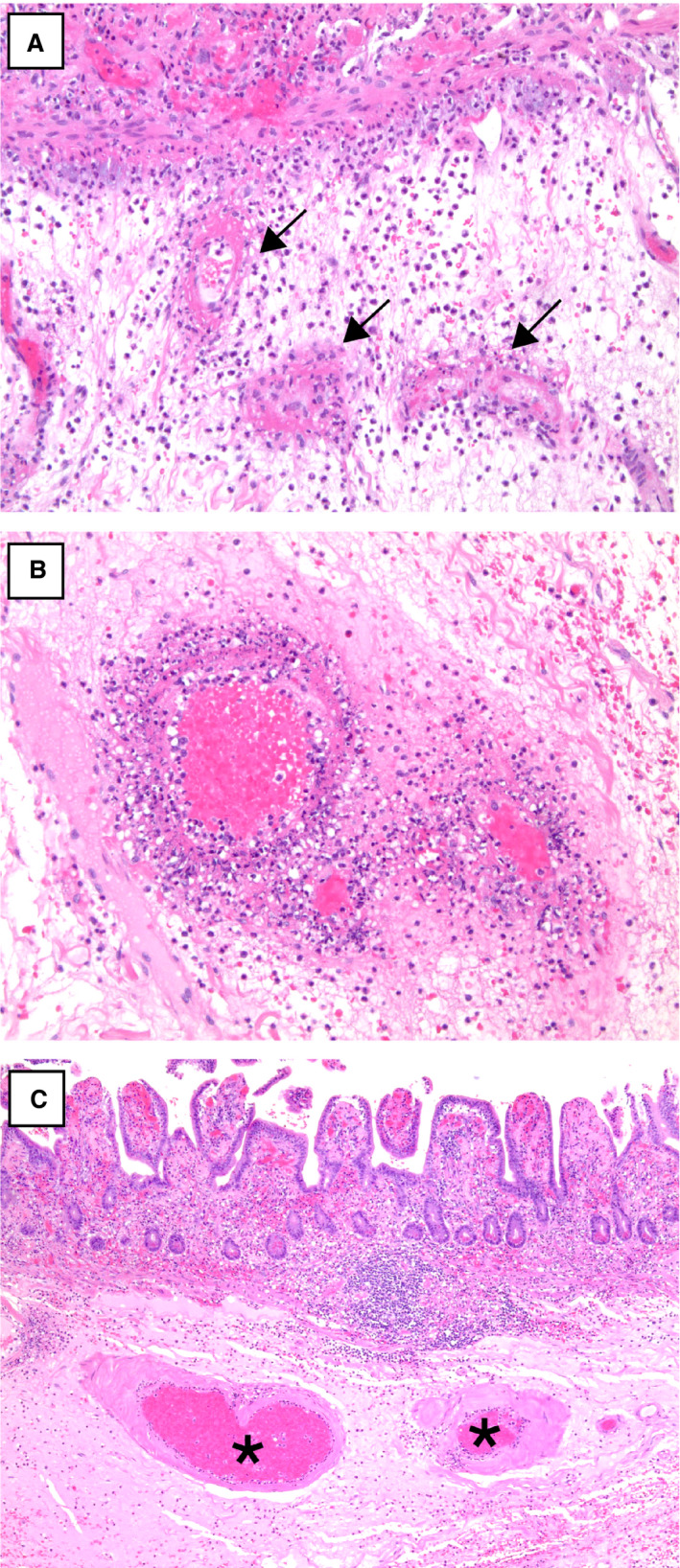
A,B, Submucosal small vessels with fibrinous degeneration of the vessel wall and perivascular neutrophils (arrows) [haematoxylin and eosin (H&E)]. C, Submucosal vessels with perivascular hyalinisation in one case of coronavirus disease 2019‐associated ischaemic enterocolitis (asterisks) (H&E). [Colour figure can be viewed at wileyonlinelibrary.com]
Figure 5.
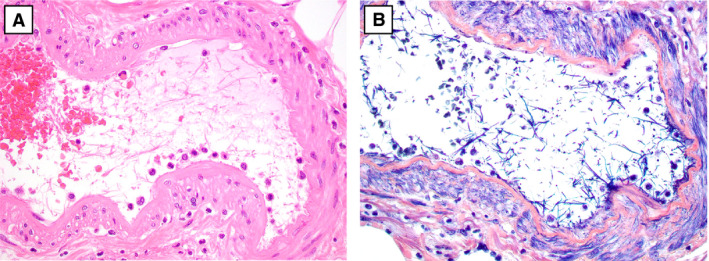
A, Fibrin strands within medium‐sized submucosal arteries (haematoxylin and eosin). B, Phosphotungstic acid haematoxylin highlights the fibrin strands in submucosal arteries. [Colour figure can be viewed at wileyonlinelibrary.com]
Three of the 28 cases (11%) showed features of acute and chronic ischaemia, including the case of pseudo‐obstruction with dilatation of the ascending and transverse colon. These patients presented in an acute manner and had clinical courses similar to those of the rest of the cohort; none had known predisposing risk factors for chronic bowel ischaemia. The acute features were either mild or focal, ranging from submucosal oedema to focal ulceration. However, all three cases had varying degrees of fibrosis of the external layer of the muscularis propria (Figure 6). The mildest case showed a segment of colon with submucosal oedema and focal fibrosis of the external muscularis propria layer (Figure 6A). A second case showed submucosal oedema with necrosis of the external muscularis propria that was focally replaced by exuberant reactive fibroblasts (Figure 6B). Finally, the case of pseudo‐obstruction showed dilatation of the colon with ulceration at the ileocaecal valve, with areas of dense fibrous replacement of the external layer and, focally, both layers of the muscularis propria (Figure 6C,D). Where the fibrosis was most developed, myenteric ganglion cells were reduced in number. No small vessel fibrin thrombi were seen in these three cases.
Figure 6.
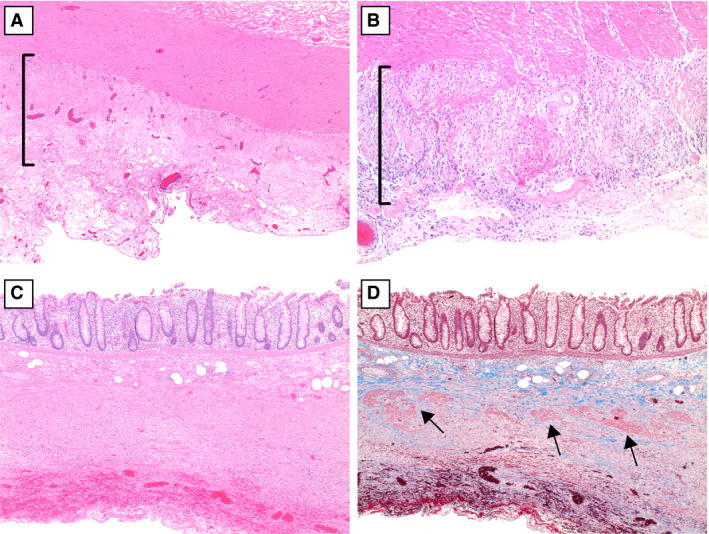
Varying degrees of fibrosis of the muscularis propria in coronavirus disease 2019‐associated ischaemic enterocolitis. A, Focal fibrosis of the external muscularis propria (bracket) [haematoxylin and eosin (H&E)]. B, Replacement of the external muscularis propria by an exuberant fibroblastic reaction (bracket) (H&E). C, The muscularis propria is replaced by dense fibrosis. D, A trichrome stain highlights the replacement of the external layer of the muscular wall by dense fibrosis, with residual bundles of smooth muscle in the internal layer of the muscularis propria (arrows). [Colour figure can be viewed at wileyonlinelibrary.com]
SARS‐COV‐2 IMMUNOHISTOCHEMISTRY AND ISH RESULTS
Immunohistochemistry for SARS‐CoV‐2 was performed in two cases and gave negative results in both cases. Chromogenic ISH for SARS‐CoV‐2 was performed on 5‐µm‐thick FFPE sections in seven cases and gave negative results in all cases. In two of these seven cases, ISH for SARS‐CoV‐2 was also performed with the smFISH approach. Whereas histological evaluation of the bowel samples revealed high levels of ACE2 expression as determined with smFISH, SARS‐CoV‐2 RNA could not be detected in either sample (Figure 7).
Figure 7.
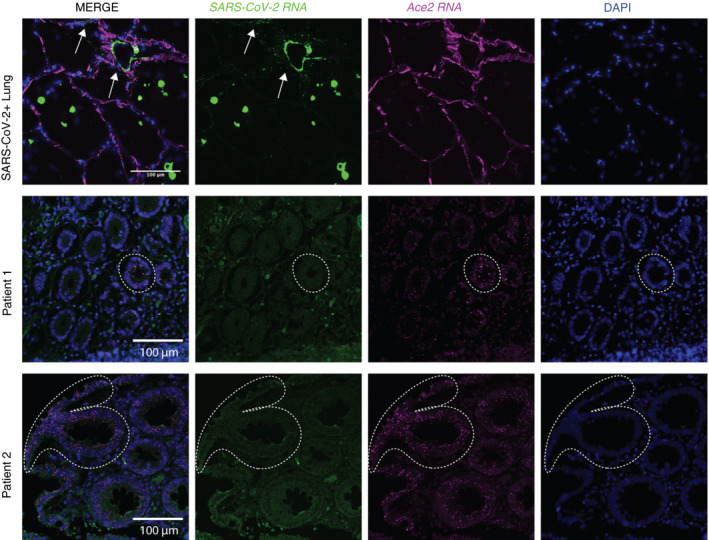
Absence of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) viral RNA in coronavirus disease 2019 (COVID‐19)‐associated ischaemic bowels. SARS‐CoV‐2 and angiotensin‐converting enzyme 2 (ACE2) single‐molecule florescence in‐situ hybridisation (smFISH) was performed on lung and ischaemic bowel samples. Top: SARS‐CoV‐2‐positive control smFISH of a human lung sample shows viral RNA localisation to lung macrophages and pneumocytes. Middle and bottom: SARS‐CoV‐2 and ACE2 RNA in‐situ hybridisation of ischaemic bowel cases from two different patients who tested positive for SARS‐CoV‐2. Arrows highlight regions of SARS‐CoV‐2 viral RNA positivity. Dotted lines outline positive ACE2 RNA signals in ischaemic bowel samples. [Colour figure can be viewed at wileyonlinelibrary.com]
COMPARISON OF COVID‐19‐ASSOCIATED WITH CONTROL ISCHAEMIC BOWEL CASES
The control cohort had a mean age of 65 years (range, 28–92 years), which was significantly higher than that of the COVID‐19 cohort (P = 0.032). There was no significant different in sex between the COVID‐19 cohort and the control cohort, in which 24 (69%) patients were male (P = 0.772). Resected bowel segments included 18 cases (51%) of the small intestine and colon (five cases designated as the terminal ileum and right colon), 15 cases (43%) of the large intestine only (four cases each designated as the right colon and left colon/rectum, respectively), and two cases (6%) of the small intestine only. The anatomical distribution of specimens differed significantly between the COVID‐19 cohort and the control cohort (P < 0.001), with the COVID‐19 cohort including a greater proportion of small‐intestine‐only resections (32% versus 6%) and a smaller proportion of colon‐only resections (14% versus 43%) (Table 4).
Table 4.
Comparison of key histological features of coronavirus disease 2019 (COVID‐19) patients with those of control cases with acute ischaemic enterocolitis
| Histological features | COVID‐19 patients (N = 19), n (%) | Control cases (N = 35), n (%) | P‐value |
|---|---|---|---|
| Depth of necrosis | 0.155 | ||
| Mucosa/submucosa | 6 (32) | 19 (54) | |
| Mural/transmural | 13 (68) | 16 (46) | |
| Distribution of necrosis | 1.000 | ||
| Focal/multifocal | 8 (42) | 16 (46) | |
| Diffuse | 11 (58) | 19 (54) | |
| Histological evidence of pneumatosis | 0.010 | ||
| Absent | 5 (26) | 23 (66) | |
| Present | 14 (74) | 12 (34) | |
| Small vessel fibrin thrombi | <0.001 | ||
| Absent | 0 (0) | 20 (57) | |
| Few | 5 (26) | 10 (29) | |
| Numerous | 14 (74) | 5 (14) | |
| Small vessel fibrinous degeneration with perivascular neutrophils | <0.001 | ||
| Absent | 2 (11) | 16 (46) | |
| Few vessels | 7 (37) | 16 (46) | |
| Numerous vessels | 10 (53) | 3 (9) | |
| Fibrin strands in submucosal vessels | 0.007 | ||
| Absent | 8 (42) | 28 (80) | |
| Few vessels | 9 (47) | 7 (20) | |
| Numerous vessels | 2 (11) | 0 (0) |
Patients with COVID‐19‐associated ischaemic bowels were significantly more likely to show small vessel fibrin thrombi (100% versus 43%, P < 0.001), submucosal vessels with fibrinous degeneration and perivascular neutrophils (90% versus 54%, P < 0.001), fibrin strands within submucosal vessels (58% versus 20%, P = 0.007), and histological evidence of pneumatosis (74% versus 34%, P = 0.010). Notably, 14 of 19 patients (74%) had specimens showing numerous fibrin thrombi, as compared with 14% of control cases (P < 0.001). Other histopathological findings, including pattern of necrosis, depth of necrosis, distribution of necrosis, and location of fibrin thrombi, when present, did not differ significantly between the two groups.
Discussion
In this cohort of critically ill COVID‐19 patients who developed bowel ischaemia, we found the following constellation of histological findings that characterised these cases: submucosal and mucosal small vessel fibrin thrombi, fibrinous degeneration of submucosal vessels with perivascular neutrophils, fibrin strands in the lumen of submucosal medium‐sized arteries, and histological evidence of pneumatosis. Otherwise, the histological findings resembled those of the usual ischaemic bowel, with serosal and mucosal discolouration and bowel dilatation. The depth of necrosis and its distribution did not differ significantly from those of ischaemic bowel resulting from other causes. Three cases had a pattern of evolving fibrosis of the outer layer of the muscularis propria, including focal necrosis and fibrosis, to exuberant fibroblastic reaction, and ultimately to marked fibrosis of the entire wall, with reduced numbers of myenteric ganglion cells. Immunohistochemistry and ISH for COVID‐19 gave negative results in all cases in which these studies were performed.
Classically, ischaemic bowel disease is most commonly seen in older patients with associated cardiac or vascular disease. Other common precipitating causes include drugs (e.g. medical vasoconstrictors and illicit cocaine), infection (e.g. cytomegalovirus and Escherichia coli O157:H7), hypercoagulable states, oral contraceptive use, shock, and systemic vasculitides, among other aetiologies. 21 In the literature, rare and mild cases of COVID‐19‐associated GI injury have primarily been reported. Ischaemic GI complications have been reported in only one case report 22 and in one case in a recent larger series, 23 and are uncommon even in our institutional cohort of critically ill COVID‐19 patients (5% of patients). A systematic review summarising histopathological findings in COVID‐19 patients described the following intestinal changes but no ischaemia: stenosis and segmental dilatation, lymphoplasmacytic infiltration, and lamina propria oedema. 24 Carvalho et al. reported one case of SARS‐CoV‐2 infection causing haemorrhagic colitis; however, endoscopic evaluation revealed focal erythema without ulceration in the descending colon, sigmoid colon, and rectum, and histologically unremarkable biopsies without evidence of infectious colitis, ischaemic colitis, or inflammatory bowel disease. 4 Xiao et al. investigated COVID‐19 patients with SARS‐CoV‐2 RNA detected in the stool and found no significant mucosal damage to the oesophagus, stomach, duodenum, and rectum. 25 A large series of autopsies performed on 67 COVID‐19‐positive patients in New York City evaluated the GI tract in 20 patients, 11 of which had GI symptoms. 17 Histological examination of the oesophagus (four cases), stomach (11 cases), small intestine (16 cases) and colon (17 cases) revealed no histological abnormalities.
More recently, Westerhoff et al. reported histological findings of GI specimens from 25 COVID‐19 patients, one of which was an ischaemic colon resection. 23 Similarly to the patients in our study, this patient presented with a markedly elevated D‐dimer level. Grossly, no yellow serosal discolouration was described, but a sharp demarcation between the necrotic portion and background viable tissue was noted. Microscopically, the specimen showed findings consistent with those that we identified in this larger cohort—including diffuse mucosal haemorrhagic necrosis, areas resembling pseudomembranous colitis, and submucosal vein thrombi—although no further details regarding the distribution or frequency of small vessel thrombi were provided. ISH and next‐generation sequencing for SARS‐CoV‐2 also gave negative results in this case.
In our cohort of patients with severe COVID‐19 infection, the gross and microscopic appearances of the ischaemic segment were distinct in some ways from the typical picture of ischaemic enterocolitis. Intraoperatively, the ischaemic/necrotic bowel usually has a dark purple–black discolouration; however, approximately one‐third of the cases in our cohort showed yellow discolouration of the necrotic segments, with patchy discrete foci of necrosis along the antimesenteric side of the bowel and clear demarcation from adjacent viable areas. 19 , 26 The significance of this yellow discolouration is unclear, as the more recent cases in our cohort showed discolouration more typical of ischaemic bowels, and we did not identify a histological correlate.
The typical histopathological features of acute bowel ischaemia include segmental/patchy mucosal and mural/transmural necrosis, degeneration and sloughing of the surface epithelium and superficial glands/crypts (which are most vulnerable to ischaemic injury), haemorrhage and/or hyalinisation of the lamina propria, congestion of mucosal capillaries and submucosal veins (which may contain thrombi), thickening of the bowel wall due to submucosal oedema, and serositis with fibrinopurulent exudates. Occasionally, bacterial superinfection may induce pseudomembrane formation resembling Clostridium difficile colitis or lead to the formation of gas within the bowel wall (i.e. pneumatosis intestinalis). In these COVID‐19‐associated ischaemic bowels, some combination of the above features was seen. Although we considered that the small vessel disease in COVID‐19 cases would lead to less frequent transmural necrosis, we did not find that the distribution and depth of necrosis differed significantly between COVID‐19‐associated and control ischaemic bowels. We also hypothesised that the discrete areas of necrosis and foci of antimesenteric necrosis would be more frequent in these bowels than in controls; however, we were unable to establish significance for these variables, a reflection of the general practice of prosectors to sample areas of ischaemic bowel without reference to precise anatomical location. However, other small vessel pathologies were observed in COVID‐19‐associated ischaemic bowels, including the presence of frequent small vessel fibrin thrombi, fibrinous degeneration of submucosal vessels with perivascular neutrophils, and fibrin strands in the lumens of submucosal vessels. We also frequently noted histological pneumatosis intestinalis. No well‐formed fibrin thrombi were identified in large vessels and, notably, the mesenteric vasculature was patent intraoperatively.
The presence of fibrin thrombi in ischaemic segments of the intestine was once described as the pathological hallmark of DIC, but has since been found to be a non‐specific feature of necrosis. 27 In fact, they have also been seen in patients with occlusive and non‐occlusive ischaemic bowel disease, E. coli O157:H7‐associated haemorrhagic colitis, C. difficile pseudomembranous colitis, amoebic colitis, staphylococcal enteritis, inflammatory bowel disease, acute appendicitis, and cocaine use. 27 , 28 , 29 , 30 TTP has also been reported to present with intestinal ischaemia characterised by numerous fibrin thrombi in the microvasculature of the intestines. 31 In a series of 11 E. coli O157:H7‐associated colitis cases, Griffin et al. found that seven cases had small fibrin/platelet thrombi within mucosal capillaries, whereas submucosal and mesenteric vessels were unremarkable. 29 They concluded that the combination of ischaemic (focal necrosis and haemorrhage) and infectious (neutrophilic cryptitis) patterns of injury, especially in conjunction with capillary microthrombi and a clinical presentation of bloody diarrhoea, was suggestive of E. coli O157:H7‐associated colitis. Thus, the presence of fibrin thrombi may not be specific for DIC, and may occur as a result of, rather than as a cause of, bowel ischaemia.
In practice, when pathologists encounter fibrin thrombi in segments of ischaemic bowel, they often have difficulty in determining their relevance, as it is unclear whether they constitute the cause of ischaemia or merely a reflection of stasis in the ischaemic and necrotic segment. In fact, in most cases, they are considered to constitute merely a reflection of stasis in these vessels, rather than a sign of hypercoagulability. In this regard, the presence of fibrin thrombi outside the areas of necrosis is frequently viewed as more significant for determining causality than the presence of fibrin thrombi immediately in areas of mucosal necrosis. However, in our cases, nearly all of the thrombi were within areas of necrosis, making it difficult to determine whether they were contributing to the ischaemia, particularly in the initial cases before we had gained some experience with these cases; however, as we gained experience, we noted that the frequency and number of fibrin thrombi in these cases suggested that they were relevant beyond the effects of stasis. We found that patients with COVID‐19‐associated ischaemic bowels were significantly more likely to have fibrin thrombi than the control cases (100% of the COVID‐19 group versus 43% of the control group), and more cases had numerous fibrin thrombi (74% of the COVID‐19 group versus 14% of the control group). This finding is consistent with ischaemic bowel resulting from a hypercoagulable state in COVID‐19 patients. Given the inability to find virus by the use of several methods, the thrombi are unlikely to constitute a direct viral effect on small vessels. The significance of the small vessel degeneration with neutrophils is uncertain, as we did not see convincing vasculitis outside areas of necrosis. The fibrin strands within arteries may be a reflection of coagulation within the vasculature, but additional studies are needed.
The mechanism of small vessel ischaemia in patients with severe COVID‐19 infection may be due to DIC (given the markedly elevated D‐dimer levels) and/or secondary to the systemic hypercoagulable state that has been reported in these patients. 32 Furthermore, these patients receive intensive treatment (in sometimes novel combinations) and are often intubated/mechanically ventilated for a long period of time prior to surgery (mean, 12 days; range, 0–25 days), which inherently predisposes patients to bowel ischaemia. The increased frequency with which we found pneumatosis intestinalis may be a reflection of ventilation, although it may be due to factors related to the small vessel thrombosis. It is worth noting that no significant gross or microscopic differences (including frequency of fibrin thrombi) were identified between the following subgroups and the rest of the cohort: the four patients with a history of thrombosis, the four patients who did not receive prophylactic or therapeutic anticoagulation, and the 11 patients who survived to discharge (versus the nine patients who died during hospitalisation). Direct involvement by the virus was not demonstrated by immunohistochemistry or ISH, suggesting that the bowel ischaemia is related to the hypercoagulable state and aggressive treatment of these patients, rather than being a viral pathogenic effect.
Three patients in this cohort had unusual histopathological findings that are normally attributed to chronic ischaemia. These patients were not known to have a predisposing vascular condition, and, other than one patient who presented with colonic pseudo‐obstruction, they presented with a clinical course similar to that of the other patients in this cohort (including a similar time from initial symptom onset to acute clinical decompensation followed by emergent surgery). It is unclear why these cases appear to be histologically distinct from the rest of the cohort and whether the ischaemic process arose via a different mechanism, but the similarity of findings among these three cases suggests a common pathogenesis for this second pattern. Whether patients who survive COVID‐19 will be predisposed to chronic ischaemia or pseudo‐obstruction remains to be seen.
In conclusion, we describe the histological findings in COVID‐19‐associated ischaemic bowels. We note that this disorder is characterised by frequent association with submucosal and mucosal small vessel fibrin thrombi, degenerated submucosal vessels with perivascular neutrophils, fibrin strands in the submucosal vessels, and pneumatosis intestinalis. A second rare pattern of injury involves the outer layer of the muscularis propria, which can be necrotic or replaced by a fibroblastic proliferation. We were unable to find virus by the use of either immunohistochemistry or ISH in these cases, suggesting that the pattern of ischaemia is related to the hypercoagulable state induced by the virus rather than being a direct effect of the virus itself.
Conflicts of interest
The authors state that they have no conflicts of interest.
Funding
No funding was received for this work.
Author contributions
M. L. Zhang and J. Misdraji designed the study. M. L. Zhang, F. Jacobsen, B. J. Pepe‐Mooney, M. Mino‐Kenudson, V. Deshpande, A. R. Shih, A. R. Mattia, W. Goessling, J. O. Hwabejire, G. C. Velmahos and J. Misdraji performed data acquisition. M. L. Zhang, F. Jacobsen, B. J. Pepe‐Mooney and J. Misdraji performed statistical analysis and data interpretation. M. L. Zhang and J. Misdraji wrote the manuscript. All authors read and approved the manuscript.
Zhang M L, Jacobsen F, Pepe‐Mooney B J, Mino‐Kenudson M, Deshpande V, Shih A R, Mattia A R, Goessling W, Hwabejire J O, Velmahos G C & Misdraji J (2021) Histopathology 79, 1004–1017. 10.1111/his.14457 Clinicopathological findings in patients with COVID‐19‐associated ischaemic enterocolitis
References
- 1. Bhatraju PK, Ghassemieh BJ, Nichols M et al. Covid‐19 in critically ill patients in the Seattle region—case series. N. Engl. J. Med. 2020; 382; 2012–2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wong SH, Lui RN, Sung JJ. Covid‐19 and the digestive system. J. Gastroenterol. Hepatol 2020; 35; 744–748. [DOI] [PubMed] [Google Scholar]
- 3. Ng SC, Tilg H. COVID‐19 and the gastrointestinal tract: more than meets the eye. Gut 2020; 69; 973–974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Carvalho A, Alqusairi R, Adams A et al. SARS‐CoV‐2 gastrointestinal infection causing hemorrhagic colitis: implications for detection and transmission of COVID‐19 disease. Am. J. Gastroenterol. 2020; 115; 942–946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Jin X, Lian J‐S, Hu J‐H et al. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus‐infected disease 2019 (COVID‐19) with gastrointestinal symptoms. Gut 2020; 69; 1002–1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lin LU, Jiang X, Zhang Z et al. Gastrointestinal symptoms of 95 cases with SARS‐CoV‐2 infection. Gut 2020; 69; 997–1001. [DOI] [PubMed] [Google Scholar]
- 7. Tian Y, Rong L, Nian W, He Y. Gastrointestinal features in COVID‐19 and the possibility of faecal transmission. Aliment. Pharmacol. Ther 2020; 51; 843–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cheung KS, Hung IFN, Chan PPY et al. Gastrointestinal manifestations of SARS‐CoV‐2 infection and virus load in fecal samples from a Hong Kong cohort: systematic review and meta‐analysis. Gastroenterology 2020; 159; 81–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Klok FA, Kruip M, van der Meer NJM et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID‐19: an updated analysis. Thromb. Res. 2020; 191; 148–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Levi M, Thachil J, Iba T, Levy JH. Coagulation abnormalities and thrombosis in patients with COVID‐19. Lancet Haematol. 2020; 7; e438–e440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Connors JM, Levy JH. COVID‐19 and its implications for thrombosis and anticoagulation. Blood 2020; 135; 2033–2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020; 18; 844–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Xiong M, Liang X, Wei Y‐D. Changes in blood coagulation in patients with severe coronavirus disease 2019 (COVID‐19): a meta‐analysis. Br. J. Haematol. 2020; 189; 1050–1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Han H, Yang L, Liu R et al. Prominent changes in blood coagulation of patients with SARS‐CoV‐2 infection. Clin. Chem. Lab. Med. 2020; 58; 1116–1120. [DOI] [PubMed] [Google Scholar]
- 15. Fox SE, Akmatbekov A, Harbert JL, Li G, Brown JQ, Heide RSV. Pulmonary and cardiac pathology in COVID‐19: the first autopsy series from New Orleans. Lancet Respir. Med. 2020; 8; 681–686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S. COVID‐19 Autopsies, Oklahoma, USA. Am. J. Clin. Pathol. 2020; 153; 725–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bryce C, Grimes Z, Pujadas E et al. Pathophysiology of SARS‐CoV‐2: targeting of endothelial cells renders a complex disease with thrombotic microangiopathy and aberrant immune response. The Mount Sinai COVID‐19 autopsy experience. medRxiv 2020.
- 18. Bhayana R, Som A, Li MD et al. Abdominal imaging findings in COVID‐19: preliminary observations. Radiology 2020; 297; E207–E215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kaafarani HMA, El Moheb M, Hwabejire JO et al. Gastrointestinal complications in critically ill patients with COVID‐19. Ann. Surg. 2020; 272; e61–e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Massoth LR, Desai N, Szabolcs A et al. Comparison of RNA in situ hybridization and immunohistochemistry techniques for the detection and localization of SARS‐CoV‐2 in human tissues. Am. J. Surg. Pathol. 2021; 45; 14–24. [DOI] [PubMed] [Google Scholar]
- 21. Odze RD, Goldblum JR. Odze and Goldblum surgical pathology of the GI tract, liver, biliary tract, and pancreas. 3rd ed. Philadephia: Saunders/Elsevier, 2014. [Google Scholar]
- 22. Norsa L, Valle C, Morotti D, Bonaffini PA, Indriolo A, Sonzogni A. Intestinal ischemia in the COVID‐19 era. Dig. Liver Dis. 2020; 52; 1090–1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Westerhoff M, Jones D, Hrycaj SM et al. Gastrointestinal pathology in samples from coronavirus disease 2019 (COVID‐19)‐positive patients. Arch. Pathol. Lab. Med. 2021. E‐pub ahead of print, 7 May. [DOI] [PubMed] [Google Scholar]
- 24. Deshmukh V, Motwani R, Kumar A, Kumari C, Raza K. Histopathological observations in COVID‐19: a systematic review. J. Clin. Pathol. 2021; 74; 76–83. [DOI] [PubMed] [Google Scholar]
- 25. Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of SARS‐CoV‐2. Gastroenterology 2020; 158; 1831–1833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Gartland RM, Velmahos GC. Bowel necrosis in the setting of COVID‐19. J. Gastrointest. Surg. 2020; 24; 2888–2889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Brandt LJ, Gomery P, Mitsudo SM, Chandler P, Boley SJ. Disseminated intravascular coagulation in nonocclusive mesenteric ischemia: the lack of specificity of fibrin thrombi in intestinal infarction. Gastroenterology 1976; 71; 954–957. [PubMed] [Google Scholar]
- 28. Qizilbash AH. The nonspecific nature of fibrin thrombi in ischemic bowel disease. Can. Med. Assoc. J. 1978; 118; 807–810. [PMC free article] [PubMed] [Google Scholar]
- 29. Griffin PM, Olmstead LC, Petras RE. Escherichia coli 0157:H7‐associated colitis: a clinical and histological study of 11 cases. Gastroenterology 1990; 99; 142–149. [DOI] [PubMed] [Google Scholar]
- 30. Hoang MP, Lee EL, Anand A. Histologic spectrum of arterial and arteriolar lesions in acute and chronic cocaine‐induced mesenteric ischemia: report of three cases and literature review. Am. J. Surg. Pathol. 1998; 22; 1404–1410. [DOI] [PubMed] [Google Scholar]
- 31. Zhou Y, Reddy V, Reilly SD, Litovsky SH, Marques MB. Gastrointestinal ischemia as the initial presentation of thrombotic thrombocytopenic purpura. Am. J. Clin. Pathol. 2012; 138(Suppl. 2); A258. [Google Scholar]
- 32. Spiezia L, Boscolo A, Poletto F et al. COVID‐19‐related severe hypercoagulability in patients admitted to intensive care unit for acute respiratory failure. Thromb. Haemost. 2020; 120; 998–1000. [DOI] [PMC free article] [PubMed] [Google Scholar]


