Abstract
Background and study aims Two scores have been implemented to standardize capsule endoscopic (CE) findings in patients with Crohn’s disease (CD): Lewis score (LS) and Capsule Endoscopy Crohnʼs Disease Activity Index (CECDAI). Both have limitations and are not well validated in the pediatric population. The aim of our study was to assess a new score (capsule endoscopy – Crohn’s disease index, CE-CD) in pediatric patients with CD and to compare it to preexisting scores.
Patients and methods This was a double-center, retrospective study involving pediatric subjects with CD who underwent CE. Correlation analyses between CE-CD, endoscopy scores and noninvasive markers of disease activities were performed. The ability of different CE scores to predict clinical and endoscopic outcomes was evaluated with regression and survival analyses.
Results A total of 312 subjects were analyzed. The CE-CD score showed a moderate (Pearson’s r = 0.581, P < 0.001) and strong (r = 0.909, P < 0.001) association with LS and CECDAI, respectively. CE-CD was a statistically significant predictor of hospitalization (hazard ratio [HR]1.061), treatment escalation (HR 1.062), steroid therapy (HR 1.082), clinical (HR 1.064) and endoscopic (HR 1.060) relapse over the twenty-four months ( P < 0.001). Subjects with mucosal inflammation according to CE-CD (CE-CD ≥ 9) had worse outcomes compared to patients without inflammation (CE-CD < 9) (Log rang test < 0.001).
Conclusions The CE-CD score is a simple, reliable, reproducible, and predictive score for evaluation of small bowel inflammation in pediatric patients with CD. Prospective validation is needed to confirm the applicability of this new index in clinical practice.
Introduction
Crohn’s disease (CD) is a chronic relapsing inflammatory disorder characterized by discontinuous and transmural inflammation. It can affect any part of the intestine, with small bowel (SB) involved in at least 40 % of children, even if this prevalence may be remarkably underestimated 1 . Capsule endoscopy (CE) is a useful, noninvasive tool for the study of SB in patients with CD, both at diagnosis and follow-up, with high sensitivity and negative predictive value 2 3 . CE assesses mucosal lesions with higher sensitivity compared to magnetic resonance enterography, which better evaluates transmural and perienteric inflammation 4 5 . Despite the high sensitivity, one of the issues with CE examination has been the lower specificity 2 4 .
Several endoscopic scoring systems have been implemented to standardize assessment of endoscopic findings. Two main scores have been developed for CD: the Lewis score (LS) and CE Crohn’s Disease Activity Index (CECDAI) 6 7 . Both indices have been used in small pediatric series, but remarkable discrepancies between the two were reported, with CECDAI better reflecting intestinal inflammation than LS 8 . LS is currently the most widespread and known CE score with well-defined cutoff values for disease activity. The LS total value is largely driven by stenosis and also includes villous edema, which is not considered a major feature of CD and it leads to risk of errors in assessment of mucosal healing (MH).
MH has become the foremost therapeutic goal of CD as its achievement had been associated with better long-term outcomes, such as surgery and hospitalization reduction. Unfortunately, MH has yet to be precisely defined 9 10 11 because the ideal situation with a complete lack of lesions may be almost unachievable over a sustained period.
Finally, many endoscopists are not familiar with the current available CE scores, because they mostly use scoring for standard colonoscopy, which favors different items in assessing inflammation 12 . This difference usually makes objective evaluation of CE lesions by using available scores more difficult.
Considering all these factors, a novel CE score was developed: the Capsule Endoscopy – Crohn’s Disease (CE-CD) index. CE-CD was devised adapting the Simple Endoscopic Score for Crohn’s Disease (SES-CD), a well validated and accepted score for ileocolonoscopic findings 13 . Similar to SES-CD, CE-CD considers ulcers as elemental lesions of CD and takes into account the number of ulcers, size of the largest ulcer, percentage of affected surface, and the presence of stenosis. The question is whether the CE-CD, while aligned with the SES-CD, can provide a more accurate measure of CD activity in the small intestine than the existing endoscopy scores. Moreover, with the advent of pan-intestinal endoscopy, this new score might allow small intestinal and colonic evaluation at the same time, as is the case with current ileocolonoscopy scores. The aim of our study was to compare CE-CD to preexisting scores on SB lesions and to evaluate its reliability in the setting of pediatric patients with CD.
Patients and methods
The primary objective was to correlate the new CE-CD score (either as a total or sub-score) with the LS and CECDAI in defining disease activity in pediatric CD. Secondary objectives were to correlate the CE-CD score with FC, PCDAI and laboratory parameters and to compare it with the correlation of the LS and CECDAI with same parameters; assess the reproducibility of each score by calculating the inter-observer agreement; determine disease activity thresholds for CE-CD by comparison with LS; and evaluate the ability of the CE-CD score in predicting disease outcomes over 24 months (i. e. hospitalization, clinical and endoscopic relapse, treatment escalation, steroid therapy, surgery).
Study characteristics and subjects
This was a two-center retrospective study. Participant centers were the Pediatric Gastroenterology and Liver Unit at Sapienza – University of Rome (Rome, Italy) and the Children's Center for Digestive Health Care (Atlanta, Georgia). A search of the CE database (October 2006 to November 2013) was performed to identify consecutive pediatric subjects with CD who underwent capsule endoscopy. Inclusion criteria were: age 6 to 18 years, suspected or known CD of the small bowel diagnosed according with clinical, laboratory, radiological, endoscopic, and histopathological criteria. Exclusion criteria were patients with clinical or imaging evidence of bowel stenosis or occlusion and nonsteroidal anti-inflammatory drug intake within 4 weeks prior to capsule endoscopy. All cases were fully read by two investigators with over 10 years’ experience in CE and were assessed for disease activity at the time of their clinical care, according to the pediatric Crohnʼs Disease Activity Index (PCDAI): clinical remission was defined as PCDAI < 10, mild disease activity as PCDAI 10–30, and moderate to severe disease activity as PCDAI > 30 14 . In addition, clinical information, including disease duration, laboratory tests (hemoglobin, c-reactive protein (CRP), serum albumin, fecal calprotectin (FCP), and concurrent medications, were recorded ( Table 1 ).
Table 1. General characteristics of the population (n:312).
| Sex (M/F) | 133/179 (44.5 %/55.5 %) | |
| Age (years) | 13 (11–16) | |
| Disease duration (months) | 12 (6–18) | |
| PCDAI | 12.5 (5–22.5) | |
| PCDAI < 10 (number) | 132 (42.3 %) | |
| PCDAI 10–30 (number) | 155 (49.7 %) | |
| PCDAI > 30 (number) | 25 (8 %) | |
| Hemoglobin (g/dL) | 12 (11.1–12.5) | |
| Serum albumin (g/dL) | 3.9 (3.4–4.2) | |
| C-reactive protein (mg/L) | 5 (2–9) | |
| Fecal calprotectin (μg/g) | 250 (100–750) | |
| Lewis score | 600 (0–1350) | |
| CECDAI | 9 (0–15) | |
| CE-CD score | 14 (0–24) | |
| Location (number) | Before CE | After CE |
|
55 (17.6 %) | 25 (8 %) |
|
0 (0 %) | 29 (9.3 %) |
|
39 (12.5 %) | 23 (7.3 %) |
|
8 (2.6 %) | 23 (7.3 %) |
|
80 (25.6 %) | 37 (12 %) |
|
45 (14.4 %) | 77 (24.7 %) |
|
62 (19.9 %) | 98 (31.4 %) |
|
23 (7.5 %) | 0 (0 %) |
| Therapy (single or in combination) (number) | N | |
|
20 | |
|
27 | |
|
100 | |
|
33 | |
|
99 | |
|
27 | |
|
23 | |
Data are expressed as number (percentage) or median (P25–75).
Location was assessed according to Paris classification.
PCDAI, Pediatric Crohn’s Disease Activity Index; CECDAI, Capsule Endoscopy Crohnʼs Disease Activity Index; CE-CD, capsule endoscopic Crohn’s disease.
In all subjects, data about first hospitalization, surgery related to CD, treatment escalation, steroidal therapy, clinical or endoscopic relapse of CD were collected for twenty-four months following the CE procedure.
Capsule endoscopy
CE was performed with the PillCam SB1/SB2/SB3 (Given Imaging Ltd, Yoqneam, Israel) capsule endoscopes using the predefined procedure’s protocol for each unit (fasting for 12 hours prior to the procedure; and only at the Rome center, polyethylene glycol 50 mL/kg up to 2 L the day prior the exam and simethicone 30 minutes before ingestion). Pediatric gastroenterologists (SO, SC) with extensive experience in IBD, CE interpretation, and LS calculation who were blinded to the clinical and laboratory information reviewed all the CE sequences for LS, CECDAI, and CE-CD calculation. The sequence review was performed using RAPID software (version 7 or 8). The CE video sequences were not de-identified.
Capsule endoscopic scores
Lewis score was calculated as previously reported 6 . The small bowel (SB) was divided into tertiles according to transit time and two parameters (villous edema, ulcers) were independently evaluated in each segment while a third parameter (stenosis) was evaluated for the entire SB. LS corresponded to maximum tertile score (resulting of edema + ulcers) plus score of stenosis. Conventionally, LS < 135 was considered suggestive of absent or insignificant inflammation, ≥ 135 to < 790: mild inflammation, ≥ 790: moderate to severe inflammation. LS calculation was performed with the help of RAPID software (version 7 or 8).
CECDAI consists of three parameters: inflammation (A: 0–5), extent of disease (B: 0–3) and strictures (C: 0–3) 15 . SB was divided in two segments according to transit time and for each segment a partial CECDAI was calculated as follow: A × B + C, with definitive CECDAI resulting as the sum of the two partial scores.
CECDAI score ranges from 0 to 36 but, unlike LS, it does not provide cutoff values even if higher CECDAI scores correspond to higher mucosal inflammation.
For the new CE-CD score, the four endoscopic variables selected were number of ulcers, size of ulcers, proportion of the surface with any sign of inflammation, and stenosis. Each variable was scored from 0 to 3 in each tertile: ulcers were scored according to the number (none; 1–3; 4–10; > 10), size (none; aphthous, < 1/4 image size, > 1/4 image size); proportion of affected surface according to the extent (unaffected segment, ≤ 10 %, 11–50 %, > 50 %), and stenosis as none, single or multiple, and whether the capsule could pass through the narrowed lumen. Total CE-CD score ranges from 0 to 34. Table 2 details specific features and calculation of each CE score.
Table 2. Comparisons of features and calculation of CE scores.
| Lewis score | Capsule Endoscopy Crohnʼs Disease Activity Index | Capsule Endoscopy – Crohnʼs Disease score | |||||
| Parameter | Number | Longitudinal extent | Descriptors | Parameter | Score | Parameter | Score |
| Villous appearance (worst-affected tertile) |
|
|
|
Inflammation |
|
Number of ulcers |
|
| Ulcer (worst-affected tertile) |
|
|
|
Extent of the disease |
|
Size of largest ulcer |
|
| Stenosis (whole study) |
|
|
|
Narrowing (stricture) |
|
Surface involved |
|
| Stenosis |
|
||||||
Lewis score = Score of the worst-affected tertile [(villous parameter x extent x descriptor) + (ulcer parameter x extent x descriptor)] + (stenosis number x ulcerated x traversed).
Capsule Endoscopy Crohnʼs Disease Activity Index = proximal (inflammation x extent + narrowing) + distal (Inflammation x extent + narrowing).
Capsule Endoscopy – Crohn’s Disease score = 1st + 2nd + 3 rd tertile (number of ulcer + size + surface + stenosis).
Fecal calprotectin measurements
A stool sample was obtained within 7 days before the CE procedure. In both centers, FC was measured using CALPRO Calprotectin ELISA (enzyme-linked monoclonal – polyclonal antibody combination immunosorbent assay).
Statistical analysis
A Kolmogorov-Smirnov test was performed to check data distribution. Data were presented as median and 25th to 75th percentile (P25–75). A Kruskal-Wallis test was used to compare medians among three CE-CD severity groups. Differences between proportions were calculated with a chi-square test. The relationship between continuous variables was analyzed through Pearson’s linear correlation and a two-tailed probability P < 0.05 was considered to be statistically significant. Interobserver
agreement between investigators for each scoring system was computed by using Cohen’s k. Receiver operating characteristic (ROC) curves and area under the curves (AUCs) were generated for fecal calprotectin and PCDAI to study accuracy in diagnosing endoscopic inflammation and sensitivity, specificity, and positive and negative predictive values and diagnostic accuracy were calculated.
Cox proportional hazard regression analyses were performed between different CE scores (independent variables) and outcomes (dependent variables) over for a follow-up period of twenty-four months. The equation generated by linear regression among LS and CE-CD score was used to identify CE-CD values corresponding to LS cutoff values (LS:135 and LS: 790).
Survival analyses, using the log-rank test in the Kaplan-Maier model, were performed to evaluate the differences in outcome over time in CD subjects according to their CE-CD score. Statistical analyses were performed with SPSS v.21.0 (IBM Corp., Armonk, New York, United States).
Ethics
This study was carried out in accordance with the World Medical Association Declaration of Helsinki. After review by the local ethics committee of each participating center (ref. 4340, serial number 520/17, date of approval: January 16, 2017), further specific ethical review and approval were not required, as the study was considered a retrospective audit work using data obtained as part of regular patient care.
Results
The database searches identified 312 patients (133 males) who had a CE performed for CD in the two centers in the aforementioned period ( Fig. 1 , Table 1 ). Of them, 252 had a complete assessment including laboratory and FCP results. The median age of the subjects was 13 years (11–16) with a median disease duration of 12 months (6–18). Eighty CEs were performed with PillCam SB1, 212 with SB2, and 20 with SB3. Two hundred and ninety-six (n = 296; 95 %) SBCEs were complete to the cecum; only 17 capsules were delivered endoscopically in the duodenum. Despite using different preparation regimens, no significant difference was observed for bowel cleansing between the two participant sites. Median PCDAI and FC were 12.5 (5–22.5) and 250 μg/g (100–750), respectively.
Fig. 1.
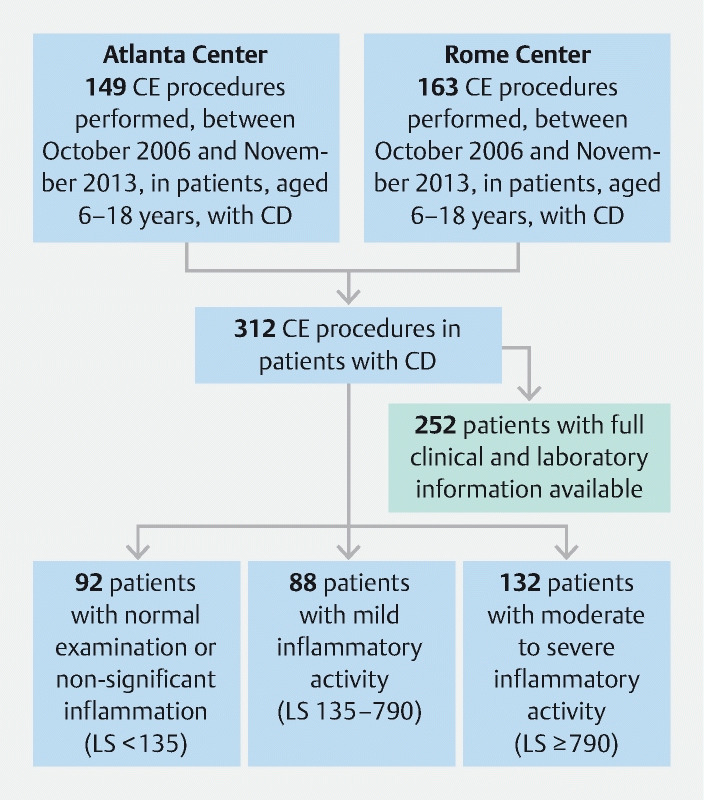
Flowchart of enrolled patients.
According to LS, 88 subjects (28.2 %) presented with mild inflammatory activity (LS: 135–790), 132 (42.3 %) with moderate to severe inflammation (LS: ≥ 790), while 92 subjects (29.5 %) had a normal examination or insignificant inflammation (LS: < 135).
Comparison of CE scores
In the entire population, median CE-CD, LS, and CECDAI were: 14 (0–24), 600 (0–1350), and 9 (0–15), respectively. In subjects with isolated SB involvement, CE-CD, LS and CECDAI were 13 (5–24), 675 (135–1350), and nine (3–15).
In correlation analysis, CE-CD score showed a moderate association with LS (Pearson’s r = 0.581, P < 0.001) and a strong association with CECDAI (r = 0.909, P < 0.001). LS and CECDAI displayed a significant positive association between them (r = 0.724, P < 0.001). Fig. 2 shows correlations of different scores in each SB tertile.
Fig. 2.
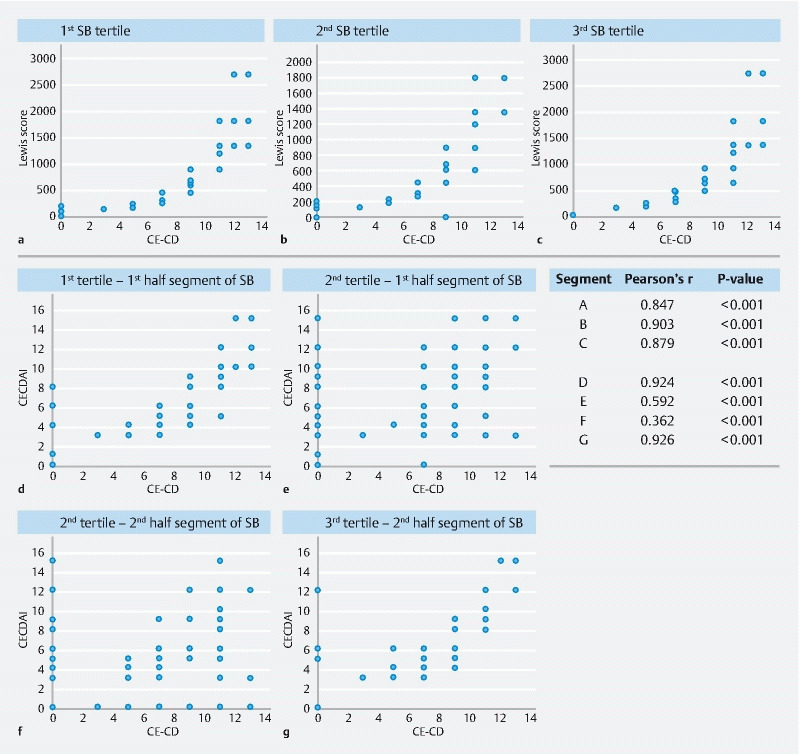
Correlations between CE-CD score, Lewis score, and CECDAI in different small bowel sections.
In subjects with isolated SB involvement: CE-CD score showed a moderate association with LS (Pearson’s r = 0.610, P < 0.001) and a strong association with cumulative-LS (r = 0.809, P < 0.001) CECDAI (r = 0.906, P < 0.001). LS and CECDAI were significantly positively associated (r = 0.772, P < 0.001).
All three scores showed similar correlation with PCDAI. CECDAI revealed the highest correlation with FC (r = 0.642, P < 0.001) while CE-CD had stronger correlations with CRP (r = 0.685, P < 0.001), albumin (r = –0.702, P < 0.001), and hemoglobin (r = –0.751, P < 0.001) compared to other scores. Similar results were found in subjects with isolated SB involvement ( Supplementary Table 1 ).
The interobserver agreement between two investigators was moderate for Lewis score (0.59, CI: 0.43–0.78) and CECDAI (0.56, CI: 0.39–0.71), and substantial for CE-CD (0.74, CI: 0.51–0.87) ( P < 0.05).
Disease activity categories
Linear regression between LS and CE-CD identified nine and 13 as CE-CD values corresponding to conventional LS cutoff scores of 135 and 790. The same cut-off scores were identified in patients with SB involvement.
Subjects with moderate to severe inflammation (CE-CD ≥ 13) showed higher values of fecal calprotectin (median value: 550 μg/g; 250–1200, P < 0.001) compared to those with mild (CE-CD 9–13) (150 μg/g: 100–200) or no relevant inflammation (CE-CD < 9) (90 μg/g; 50–150). Similarly, a progressive increase in CRP was observed among the three groups ( Fig. 3 ).
Fig. 3.
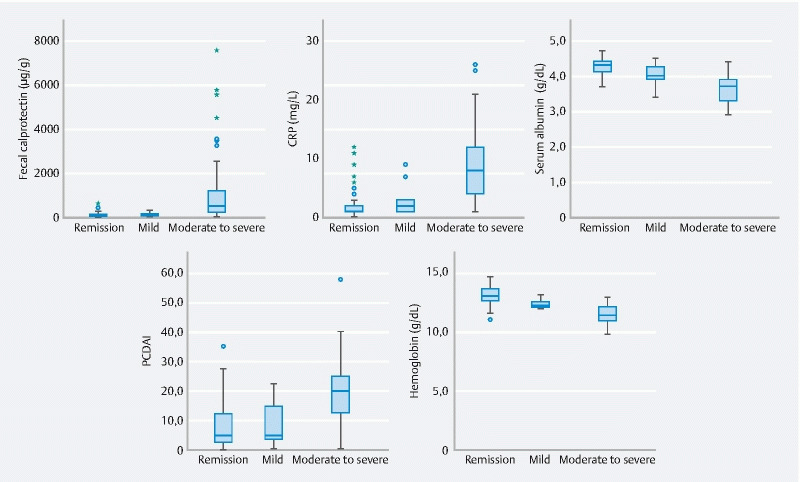
Comparisons of clinical and laboratory markers of small bowel inflammation among different CE-CD groups.
In the subgroup of children with SB disease, Subjects with moderate to severe inflammation (CE-CD ≥ 13) showed higher values for fecal calprotectin (median value:750 μg/g; 300–1350, P < 0.001) compared to those with mild (CE-CD 9–13) (200 μg/g: 137–237) or no relevant inflammation (CE-CD < 9) (90 μg/g; 50–100).
Analysis of subjects’ proportion distribution according to PCDAI and CE-CD revealed that 23 of 25 patients (92 %) with PCDAI > 30 had a concurrent CE-CD ≥ 13. On the other hand, 35 of 132 (26.5 %) with a PCDAI < 10 had severe inflammatory activity ( Table 3 , Supplementary Fig. 1 ).
Table 3. Population comparison according to CE-CD categories.
|
CE-CD: < 9
n = 112 (35.9 %) |
CE-CD: 9–13
n = 27 (8.7 %) |
CE-CD: ≥ 13
n = 173 (55.4 %) |
P value | |
| CE-CD | 0 (0–3) | 9 (9–10) | 22 (18–27) | < 0.001 |
| PCDAI | 5 (2.5–12.5) | 5 (0–5) | 17.5 (12–25) | < 0.001 |
| FCP (μg/g) | 90 (50–150) | 150 (100–200) | 550 (250–1200) | < 0.001 |
| Hemoglobin (g/dL) | 12.9 (12.5–13.5) | 12.1 (11.9–12.4) | 11.3 (10.8–12) | < 0.001 |
| Serum albumin (g/dL) | 4.3 (4.1–4.4) | 4 (3.9–4.3) | 3.7 (3.3–3.9) | < 0.001 |
| C-reactive protein (mg/L) | 1 (1–2) | 2 (1–3) | 8 (4–12) | < 0.001 |
| PCDAI < 10 (n: 132) | 79 (59.9 %) | 18 (13.6 %) | 35 (26.5 %) | < 0.001 |
| PCDAI 10–30 (n: 155) | 31 (20 %) | 9 (5.8 %) | 115 (74.2 %) | |
| PCDAI > 30 (n: 25) | 2 (8 %) | 0 (0 %) | 23 (92 %) |
Data are expressed as median (P25–75) or number (percentages).
CE-CD, capsule endoscopic-Crohn’s Disease; PCDAI, Pediatric Crohn’s Disease Activity Index.
Similarly, in subjects with SB involvement, 16 of 16 subjects (100 %) with PCDAI > 30 had a concurrent CE-CD ≥ 13; 21 of 73 (28.8 %) with a PCDAI < 10 had severe inflammatory activity.
CE-CD and outcomes by total score
In the 24 months following the CE procedure, 42 subjects (13.5 %) underwent surgery related to CD, 233 (74.7 %) had one or more admissions to the hospital; 73 patients (23.4 %) needed to escalate treatment, and 275 (88.1 %) received steroid medications. Finally, 296 (94.9 %) and 277 subjects (88.8 %) experienced a clinical and/or endoscopic relapse of CD. Mean clinical and endoscopic relapse times were 10 ± 6.23 and 11.9 ± 5.85 months, respectively.
In regression analyses, CE-CD was a statistically significant predictor of hospitalization (hazard ratio [HR] = 1.061, P < 0.001), treatment escalation (HR = 1.062, P < 0.001), steroid therapy (HR = 1.082, P = 0.001), clinical (HR = 1.064, P < 0.001), and endoscopic (HR = 1.060, P < 0.001) relapse. LS and CECDAI produced similar results for the same outcomes while only LS was able to predict surgery risk (HR 1.001, P = 0.027) ( Table 4 ).
Table 4. Cox regression analyses between scores or fecal calprotectin (independent variables) and main outcomes (dependent variables).
| CE-CD | Lewis score | CECDAI | FCP | |||||
| Hazard ratio (95 % CI) | P value | Hazard ratio (95 % CI) | P value | Hazard ratio (95 % CI) | P value | Hazard ratio (95 % CI) | P value | |
| Surgery | 1.014 (0.977–1.053) | 0.449 | 1.001 (1.000–1.001) | 0.027 | 1.042 (0.993–1.092) | 0.093 | 1.00003 (0.999–1.0002) | 0.646 |
| Hospitalization | 1.061 (1.048–1.075) | < 0.001 | 1.001 (1.000–1.001) | < 0.001 | 1.096 (1.076–1-117) | < 0.001 | 1.0003 (1.0002–1.0004) | < 0.001 |
| Treatment escalation | 1.062 (1.050–1.074) | < 0.001 | 1.001 (1.000–1.001) | < 0.001 | 1.093 (1.076–1.112) | < 0.001 | 1.0003 (1.0002–1.0004) | < 0.001 |
| Steroids | 1.082 (1.007–1.096) | < 0.001 | 1.001 (1.000–1.001) | < 0.001 | 1.112 (1.093–1.132) | < 0.001 | 1.0004 (1.0003–1.0005) | < 0.001 |
| Clinical relapse | 1.064 (1.052–1.076) | < 0.001 | 1.001 (1.000–1.001) | < 0.001 | 1.102 (1.084–1.121) | < 0.001 | 1.0003 (1.0002–1.0004) | < 0.001 |
| Endoscopic relapse | 1.060 (1.048–1.072) | < 0.001 | 1.001 (1.000–1.001) | < 0.001 | 1.099 (1.080–1.118) | < 0.001 | 1.0003 (1.0002–1.0004) | < 0.001 |
Similarly, in subjects with isolated SB involvement, CE-CD was able to predict the risk of hospitalization (HR = 1.061, P < 0.001), treatment escalation (HR = 1.064, P < 0.001), steroid therapy (HR = 1.088, P = 0.001), and clinical (HR = 1.066, P < 0.001) and endoscopic (HR = 1.058, P < 0.001) relapse.
In subjects with isolated colonic disease, CE-CD was a significant predictor of treatment escalation (HR = 1.095, P < 0.001), steroid therapy (1.068 P = 0.026), and clinical relapse (HR = 1.068, P < 0.026), but not of hospitalization or endoscopic relapse.
CE-CD outcomes by disease severity categories
Survival analyses of outcomes according to the presence (CE-CD ≥ 9) or absence (CE-CD < 9) of mucosal inflammation are shown in Fig. 4 . Subjects with active inflammation had a higher cumulative probability of hospitalization, treatment escalation, steroid therapy, and clinical or endoscopic relapse (Log rank test < 0.001). Also, in the subgroup of children in clinical remission (PCDAI < 10), the presence of mucosal inflammation (CE-CD ≥ 9) was statistically associated with worse outcomes (Log rank test < 0.001). As a consequence, subjects in clinical remission with mucosal inflammation tended to develop early clinical or endoscopic relapses compared to subjects without mucosal inflammation ( Supplementary Table 2 ).
Fig. 4.
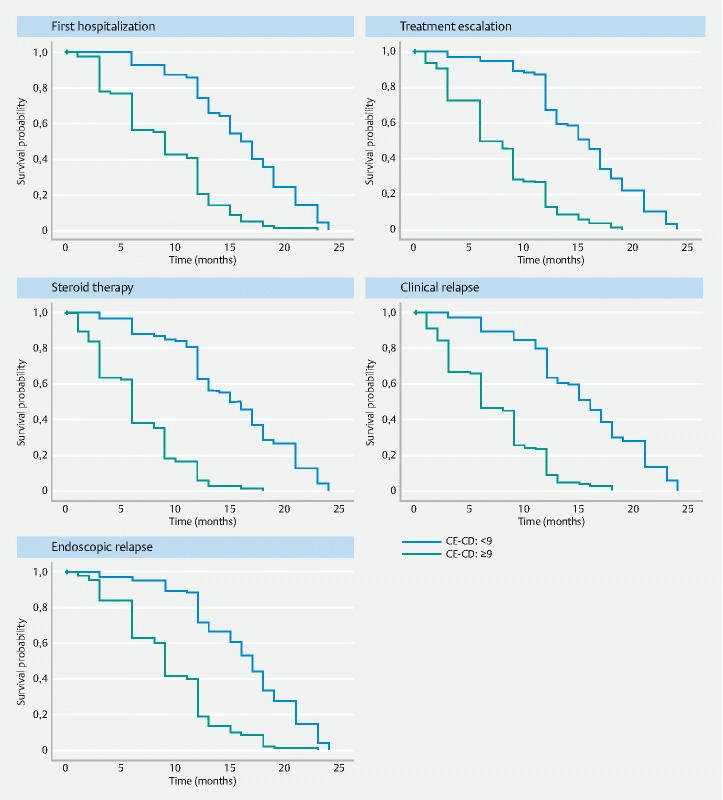
Survival analyses of outcomes according to the presence (CE-CD ≥ 9) or absence (CE-CD < 9) of mucosal inflammation.
Survival analyses were not performed for the surgical outcome as only one of 42 subjects who underwent surgery had a CE-CD < 9.
Accuracy of FC and PCDAI with CE-CD ≥ 9
ROC analysis revealed AUCs of 0.912 and 0.779 for the accuracy of FC and PCDAI to identify the presence of SB inflammation (CE-CD ≥ 9) ( Supplementary Fig. 2 ). FC ≥ 110 μg/g had a sensitivity of 89.0 % and a specificity of 69.6 % while PCDAI ≥ 15 had a sensitivity of 60.5 % and a specificity of 91.9 %.
Lower thresholds for both markers were associated with an increase in sensitivity (up to 97.7 % for FC and 73.5 for PCDAI) with a concomitant reduction in specificity ( Supplementary Table 3 ).
In subjects with SB disease, ROC analysis showed similar AUCs for accuracy of FC (0.945) and PCDAI (0.798) in identification of presence of SB inflammation. In this cohort, FC ≥ 115 μg/g had a sensitivity of 91.7 % and a specificity of 77.5 % while PCDAI ≥ 7.5 had a sensitivity of 69 % and a specificity of 75 %.
Discussion
This study aimed to evaluate the applicability, effectiveness, and reproducibility of a novel capsule endoscopy score (CE-CD) and to compare it to the preexisting scores by retrospectively assessing three scores in a cohort of 312 pediatric patients with CD from two different centers (Italy, United States).
CE-CD showed very strong concordance with CECDAI (r = 0.909) but only moderate with LS (r = 0.508). There are two possible explanations of this difference. The first is that CE-CD and CECDAI are both cumulative scores, while LS considers only the worst affected segment. Indeed, by comparing each LS tertile with the corresponding CE-CD tertile, the correlation considerably increased ( Fig. 2 ). The second possible explanation is that, if present, strictures account for a large portion of the LS final score, whereas CE-CD and CECDAI final scores are less affected by this single parameter.
CE-CD, CECDAI, and LS showed a moderate correlation with noninvasive, surrogate markers of SB inflammation (CRP, albumin and fecal calprotectin), although CE-CD was more closely associated with CRP and albumin (r = 0.685, r = –0.702) and CECDAI with fecal calprotectin (r = 0.642). Our data are analogous to those previously reported demonstrating only moderate correlations between laboratory markers of disease activity and SB findings at CE in IBD patients 16 17 18 .
In our study, we observed a progressive increase in median FC in the three severity groups according to novel CE-CD cutoff values. Moreover, we confirmed that FC is a valid surrogate marker of SB inflammation (CE-CD ≥ 9), with a sensitivity of 89 % and a specificity of 69.6 % for FC ≥ 110 μg/g (AUC: 0.912). A meta-analysis conducted in 2018 concluded that FC has an overall sensitivity and specificity of 82.4 % and 72.1 %, respectively, in CD 19 . Monteiro et al. investigated FC’s accuracy for identifying SB inflammation according to Lewis score (LS ≥ 135) and found a sensitivity of 78.6 % and a specificity of 87.9 % for FC ≥ 100 μg/g 20 .
We found a good correlation between PCDAI and CE-CD (r = 0.624), LS (r = 0.633) and CECDAI (r = 0.651). PCDAI appears to be a moderately accurate classifier of SB inflammation (CE-CD ≥ 9; AUC = 0.779) with a high specificity (90.1 % for PCDAI ≥ 15) and low sensitivity (60.5 %). In keeping with this, we observed that 35 of 132 subjects (26.5 %) in clinical remission (PCDAI < 10) had surprisingly severe endoscopic patterns (CE-CD > 13), suggesting that CE-CD might be a useful preclinical predictor of CD exacerbations rather than overestimating disease severity. Indeed, it has been reported that SB inflammation is considerably underestimated, especially in patients in clinical remission 21 22 23 .
To support this observation, we found that CE-CD was able to predict CD outcomes during follow-up and increases of one unit in CE-CD were mirrored by gains in risk of 6.2 %, 6.4 %, and 6 %, in treatment escalation, clinical and endoscopic relapse, respectively ( Table 4 ). Moreover, survival analysis revealed that subjects in clinical remission (PCDAI < 10) with mucosal inflammation according to CE-CD tended to develop the worst outcomes over the years, compared to those with normal mucosa.
A meta-analysis by Niv et al. highlighted that mucosal healing assessed by CE was significantly associated with improvements in outcome during different follow-up periods (12 weeks to 24 months) 24 . A recent study retrospectively evaluated 125 adults with CD who underwent SBCE and demonstrated that LS at a cutoff value of 264 could predict risk of CD-related emergency hospitalization within 1 year after CE 25 . Furthermore, subjects with LS < 264 had higher clinical relapse-free and hospitalization-free rates. The same observation was recently reported in another adult study with a LS value < 350. This confirms the idea that quantifying SB inflammation helps provide useful information about the disease course 26 . In our cohort, CE-CD ≥ 9 was associated with development of the worst outcomes, compared to absent or insignificant inflammation (CE-CD < 9) over 24 months.
CE-CD, therefore, is a valid score for evaluation of children with CD with an overall accuracy similar to CECDAI and LS. In our opinion, the main advantage of novel CE-CD compared to other scores are the ease of calculating it, which requires no software analysis. This was confirmed by the better interobserver agreement we found in comparison to LS and CECDAI. The CE-CD with a cumulative nature, reflecting inflammation of the entire small bowel, and the similarity with the SES-CD, permits a consistent and unified understanding of the small and large intestine. This capacity may be further enhanced and clinically appealing, if the CE-CD is validated in the colon using pan-enteric capsule endoscopy, allowing for a single, noninvasive procedure. Another useful characteristic of CE-CD is that ulcers are the main mucosal CE findings in CD. This is a major difference with LS and CECDAI that also takes into account edema and erythema, two parameters that are difficult to assess quantitatively and which have limited specificity in evaluation of CD inflammation 27 .
This study had several limitations. It was retrospective and confined to pediatric subjects with CD selected from two centers very experienced with IBD and CE. Therefore, future prospective studies are needed to validate the score and refine cutoff values for less experienced investigators. CE-CD cutoff values were indirectly determined by the LS values even though CE-CD better correlated with CECDAI. However, LS is the only formally validated score for disease activity cut-offs.
Inherent in the performance of this analysis on a cohort with known CD is that those undergoing the procedure are essentially doing so because of an exacerbation of symptoms that requires evaluation. This undoubtedly led to the high incidence of relapse that was seen as these patients were followed.
Another limitation is that our population included subjects with colonic involvement (L2 and L3 sec. Paris classification), which may have affected the overall values of surrogate markers of inflammation. In the future, it would be interesting to evaluate the same score on both SB and colon by using new pan-enteric capsule models.
Conclusions
In conclusion, we developed and assessed a novel capsule endoscopy score (CE-CD), which has proven to be simple, reliable, and reproducible for evaluation of SB inflammation in pediatric patients with CD. This score also seems to predict disease outcomes over time. Further studies are needed before use of this novel instrument can be implemented in clinical practice, including prospective validation in adults and likely on pan-enteric capsule endoscopy.
Footnotes
Competing interests Dr. Oliva is a consultant for Medtronic and Ocean Farma. Dr. Cohen is a consultant for Medtronic, Janssen and receives research support from Medtronic Janssen, Abbvie, Takeda, Arena, and Allergan Pharmaceuticals.
Supplementary material :
References
- 1.Rosen M J, Dhawan A, Saeed S A. Inflammatory bowel disease in children and adolescents. JAMA Pediatr. 2015;169:1053–1060. doi: 10.1001/jamapediatrics.2015.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Oliva S, Thomson M, de Ridder L et al. Endoscopy in pediatric inflammatory bowel disease: A position paper on behalf of the Porto IBD Group of the European Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2018;67:414–530. doi: 10.1097/MPG.0000000000002092. [DOI] [PubMed] [Google Scholar]
- 3.Pennazio M, Spada C, Eliakim R et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2015;47:352–376. doi: 10.1055/s-0034-1391855. [DOI] [PubMed] [Google Scholar]
- 4.Jensen M D, Nathan T, Rafaelsen S R et al. Diagnostic accuracy of capsule endoscopy for small bowel Crohnʼs disease is superior to that of MR enterography or CT enterography. Clin Gastroenterol Hepatol. 2011;9:124–129. doi: 10.1016/j.cgh.2010.10.019. [DOI] [PubMed] [Google Scholar]
- 5.Greener T, Klang E, Yablecovitch D et al. The impact of magnetic resonance enterography and capsule endoscopy on the re-classification of disease in patients with known Crohnʼs disease: A prospective Israeli IBD Research Nucleus (IIRN) study. J Crohns Colitis. 2016;10:525–531. doi: 10.1093/ecco-jcc/jjw006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gralnek I M, Defranchis R, Seidman E et al. Development of a capsule endoscopy scoring index for small bowel mucosal inflammatory change. Aliment Pharmacol Ther. 2008;27:146–154. doi: 10.1111/j.1365-2036.2007.03556.x. [DOI] [PubMed] [Google Scholar]
- 7.Niv Y, Ilani S, Levi Z et al. Validation of the capsule endoscopy Crohnʼs disease activity index (CECDAI or Niv score): a multicenter prospective study. Endoscopy. 2012;44:21–26. doi: 10.1055/s-0031-1291385. [DOI] [PubMed] [Google Scholar]
- 8.Omori T, Kambayashi H, Murasugi S et al. Comparison of Lewis score and capsule endoscopy Crohnʼs disease activity index in patients with Crohnʼs disease. Dig Dis Sci. 2020;65:1180–1188. doi: 10.1007/s10620-019-05837-7. [DOI] [PubMed] [Google Scholar]
- 9.Shah S C, Colombel J F, Sands B E et al. Systematic review with meta-analysis: mucosal healing is associated with improved long-term outcomes in Crohnʼs disease. Aliment Pharmacol Ther. 2016;43:317–333. doi: 10.1111/apt.13475. [DOI] [PubMed] [Google Scholar]
- 10.Vaughn B P, Shah S, Cheifetz A S. The role of mucosal healing in the treatment of patients with inflammatory bowel disease. Curr Treat Options Gastroenterol. 2014;12:103–117. doi: 10.1007/s11938-013-0008-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Orlando A, Guglielmi F W, Cottone M et al. Clinical implications of mucosal healing in the management of patients with inflammatory bowel disease. Dig Liver Dis. 2013;45:986–991. doi: 10.1016/j.dld.2013.07.005. [DOI] [PubMed] [Google Scholar]
- 12.Daperno M, Comberlato M, Bossa F et al. Training programs on endoscopic scoring systems for inflammatory bowel disease lead to a significant increase in interobserver agreement among community gastroenterologists. J Crohns Colitis. 2017;11:556–561. doi: 10.1093/ecco-jcc/jjw181. [DOI] [PubMed] [Google Scholar]
- 13.Daperno M, D'Haens G, Van Assche G et al. Development and validation of a new, simplified endoscopic activity score for Crohnʼs disease: the SES-CD. Gastrointest Endosc. 2004;60:505–512. doi: 10.1016/s0016-5107(04)01878-4. [DOI] [PubMed] [Google Scholar]
- 14.Hyams J S, Ferry G D, Mandel F S et al. Development and validation of a pediatric Crohnʼs disease activity index. J Pediatr Gastroenterol Nutr. 1991;12:439–447. [PubMed] [Google Scholar]
- 15.Gal E, Geller A, Fraser G et al. Assessment and validation of the new capsule endoscopy Crohnʼs disease activity index (CECDAI) Dig Dis Sci. 2008;53:1933–1937. doi: 10.1007/s10620-007-0084-y. [DOI] [PubMed] [Google Scholar]
- 16.Koulaouzidis A, Nemeth A, Johansson G W et al. Dissecting Lewis score under the light of fecal calprotectin; an analysis of correlation of score components with calprotectin levels in capsule endoscopy. Ann Gastroenterol. 2015;28:259–264. [PMC free article] [PubMed] [Google Scholar]
- 17.Koulaouzidis A, Sipponen T, Nemeth A et al. Association between fecal calprotectin levels and small-bowel inflammation score in capsule endoscopy: a multicenter retrospective study. Dig Dis Sci. 2016;61:2033–2040. doi: 10.1007/s10620-016-4104-7. [DOI] [PubMed] [Google Scholar]
- 18.Yablecovitch D, Lahat A, Neuman S. The Lewis score or the capsule endoscopy Crohnʼs disease activity index: which one is better for the assessment of small bowel inflammation in established Crohnʼs disease? Therap Adv Gastroenterol. 2018;11 doi: 10.1177/1756283X17747780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rokkas T, Portincasa P, Koutroubakis I E. Fecal calprotectin in assessing inflammatory bowel disease endoscopic activity: a diagnostic accuracy meta-analysis. J Gastrointestin Liver Dis. 2018;27:299–306. doi: 10.15403/jgld.2014.1121.273.pti. [DOI] [PubMed] [Google Scholar]
- 20.Monteiro S, Barbosa M, Cúrdia Gonçalves T et al. Fecal calprotectin as a selection tool for small bowel capsule endoscopy in suspected Crohnʼs disease. Inflamm Bowel Dis. 2018;24:2033–2038. doi: 10.1093/ibd/izy098. [DOI] [PubMed] [Google Scholar]
- 21.Sorrentino D, Nguyen V Q. Clinically significant small bowel Crohnʼs disease might only be detected by capsule endoscopy. Inflamm Bowel Dis. 2018;24:1566–1574. doi: 10.1093/ibd/izy048. [DOI] [PubMed] [Google Scholar]
- 22.Hansel S L, McCurdy J D, Barlow J M et al. Clinical benefit of capsule endoscopy in crohnʼs disease: impact on patient management and prevalence of proximal small bowel involvement. Inflamm Bowel Dis. 2018;24:1582–1588. doi: 10.1093/ibd/izy050. [DOI] [PubMed] [Google Scholar]
- 23.Kopylov U, Yablecovitch D, Lahat A et al. Detection of small bowel mucosal healing and deep remission in patients with known small bowel Crohnʼs disease using biomarkers, capsule endoscopy, and imaging. Am J Gastroenterol. 2015;110:1316–1323. doi: 10.1038/ajg.2015.221. [DOI] [PubMed] [Google Scholar]
- 24.Niv Y. Small-bowel mucosal healing assessment by capsule endoscopy as a predictor of long-term clinical remission in patients with Crohnʼs disease: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2017;29:844–848. doi: 10.1097/MEG.0000000000000881. [DOI] [PubMed] [Google Scholar]
- 25.Nishikawa T, Nakamura M, Yamamura T et al. Lewis Score on capsule endoscopy as a predictor of the risk for Crohnʼs disease-related emergency hospitalization and clinical relapse in patients with small bowel Crohnʼs Disease. Gastroenterol Res Pract. 2019;2019:4.274257E6. doi: 10.1155/2019/4274257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ben-Horin S, Lahat A, Amitai M M et al. Assessment of small bowel mucosal healing by video capsule endoscopy for the prediction of short-term and long-term risk of Crohnʼs disease flare: a prospective cohort study. Lancet Gastroenterol Hepatol. 2019;4:519–528. doi: 10.1016/S2468-1253(19)30088-3. [DOI] [PubMed] [Google Scholar]
- 27.Mow W S, Lo S K, Targan S R et al. Initial experience with wireless capsule enteroscopy in the diagnosis and management of inflammatory bowel disease. Clin Gastroenterol Hepatol. 2004;2:31–40. doi: 10.1016/s1542-3565(03)00289-1. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


