Graphical abstract
Keywords: COVID-19, SARS-CoV-2, Nucleic acid-based vaccines, Viral vector vaccines, Protein-based vaccines, Inactivated vaccines
Abstract
>20 months has been passed since the detection of the first cases of SARS-CoV-2 infection named COVID-19 from Wuhan city of China. This novel coronavirus spread rapidly around the world and became a pandemic. Although different therapeutic options have been considered and approved for the management of COVID-19 infection in different stages of the disease, challenges in pharmacotherapy especially in patients with moderate to severe COVID-19 and with underlying diseases have still remained. Prevention of infection through public vaccination would be the only efficient strategy to control the morbidity and mortality caused by COVID-19. To date, several COVID-19 vaccines using different platforms including nucleic acid-based vaccines, adenovirus-based vaccines, protein-based vaccines, and inactivated vaccines have been introduced among which many have received approval for prevention against COVID-19. In this comprehensive review, available COVID-19 vaccines have been discussed. The mechanisms, safety, efficacy, dosage, dosing intervals, possible adverse reactions, storage, and coverage of these four different vaccine platforms against SARS-CoV-2 variants have been discussed in detail and summarized in tabular format for ease of comparison and conclusion. Although each COVID-19 vaccine has various advantages and disadvantages over the others, accessibility and affordability of approved vaccines by the official health organizations, especially in developing countries, would be essential to terminate this pandemic. The main limitation of this study was the lack of access to the clinical data on available COVID-19 vaccines developed in Eastern countries since the data on their efficacy, safety, and adverse reactions were limited.
1. Introduction
In late December 2019, a novel member of betacoronaviridae family called SARS-CoV-2 caused COVID-19 that emerged from the Wuhan City of China and soon spread worldwide becoming a pandemic [1]. Until September 12th 2021, over 219 million cases have been infected with SARS-CoV-2 and 4.55 million deaths had been reported. COVID-19 can induce different complications and affect different organs mainly the respiratory tract, gastrointestinal tract, central nervous system [2], liver, kidney, heart, etc. [1]. Older adults and patients with underlying diseases are highly prone to severe type of COVID-19 pneumonia that can be presented by acute respiratory distress syndrome (ARDS), cytokine release syndrome (CRS), acute kidney injury (AKI), septic shock, multi-organ failure (MOF), and cardiac arrest [1]. Different therapies including antiviral agents, anticoagulants, anti-inflammatory agents [140], immunomodulators, and miscellaneous agents including iron and zinc chelating agents [1], [3], [4], [5], [6], [7] have been considered in the management of different stages of COVID-19 infection. Although several therapeutic strategies are available and administered to infected patients, COVID-19 pharmacotherapy is still a challenge and many people all around the world still suffer from this disease and its related complications. Furthermore, several SARS-CoV-2 variants have been identified originating from different geographical areas that are the main cause of global viral dissemination [8]. Therefore, the best strategy to limit this pandemic would be preventive actions among which worldwide vaccination is most lucrative.
The conventional platform for vaccine production is whole inactivated viruses or live attenuated ones. The main concerns regarding this vaccine platform is the possibility of incomplete viral inactivation. Hence, validation of each batch is necessary to confirm complete inactivation of recruited pathogens. The other concern related to inactivated vaccines is the possibility of antibody disease enhancement (ADE) syndrome that can be attributed to the production of non-neutralizing antibodies and can result in increased lung pathology [9]. Another vaccine platform, the protein-based vaccines, are of great use, since they beneficially induce immune responses against viral infections [10], [11]. These vaccines can be produced by the purification of specific proteins from viruses as well as the production of recombinant proteins in the host cells. Protein-based vaccines have the potential of higher safety at the injection site with less side effects. However, adjuvants and vaccine delivery systems are required in order to boost the immune response to support their sufficient efficacy against COVID-19 [12]. Besides, the virus-like particles could be prepared by the arrangement of antigen proteins on a nanoparticle to mimic the natural protein structure. Although these classic vaccine platforms have shown great impact on the eradication of various life-threatening viral diseases, several factors have limited their fast development which has decelerated the rate of global vaccination [10], [13]. The main limitation of such vaccine platforms to control pandemics is the possibility of mass production and fast availability. In other words, the game-changer vaccines must be able to be produced for large populations in a short time. Such limitations have led researchers to seek novel platforms providing quick response with minimal biosafety concerns [14]. In this review, different platforms of COVID-19 vaccines have been thoroughly discussed. Also, the mechanisms, safety, and efficacy of available vaccines have been summarized. Finally, the dosage, dosing intervals, storage, reported adverse reactions, and the coverage of each available COVID-19 vaccine against different SARS-CoV-2 variants have been summarized in a Table.
2. Pathologic effects of COVID-19
SARS-CoV-2 infection can induce several complications including loss of tissue stem cells that can stop the epithelial cell repair process and induce inflammatory fibrosis. In this regard, SARS-CoV-2 can infect the gut enterocyte and result in enhanced intestinal viral pool. The consequence would be the experience of gastrointestinal presentations of COVID-19 including nausea, vomiting, diarrhea, and anorexia. The possible mechanism of intestinal involvement during COVID-19 infection, would be the presence of angiotensin converting enzyme 2 (ACE2) and transmembrane serine protease 2 (TMPRSS2) receptors that have an important role in viral entry to the cells [15]. Also, SARS-CoV-2 infection can result in severe respiratory distress and lung involvement due to epithelial cell damage and inflammatory responses. This process takes place due to the loss of lung stem cells and decline of lipid metabolism in epithelial cells after COVID-19 infection [15]. The other complication of COVID19 infection would be neural cell damage and neurological complications including cerebrovascular attack, seizure, and psychotic disorders. The possible mechanism would be the expression of ACE2 and TMPRSS2 receptors in brain that can induce viral entry to the brain cells. Also, SARSCoV-2 can damage the choroid plexus epithelium in the brain that can result in easy passage of different immune cells, cytokines, and pathogens. Furthermore, SARS-CoV-2 might have some direct neurotoxic potentials that can induce neurological complications after viral infection [2], [15]. The other consequence of COVID-19 is the occurrence of new onset acute kidney injury (AKI) after SARS-CoV-2 infection that can worsen patients’ prognosis. COVID-19-induced renal failure can be attributed to the high level of ACE2 and TMPRSS2 receptors expressed in renal cells that make the kidney organ as one of the prominent targets of SARS-CoV-2. Pro-inflammatory and pro-fibrotic processes after COVID-19 infection was ascribed to the ACE2 internalization, renin-angiotensin-aldosterone system (RAAS) imbalance and enhanced angiotensin II signaling [15]. Another common complication of COVID-19, especially in hospitalized patients, is hyper coagulopathy state and cardiovascular disorders that can be attributed to the inflammasome activation. COVID-19-induced cardiovascular disorders include acute coronary syndrome (ACS), venous thrombosis, arrhythmia, and myocarditis. The possible mechanism of these cardiac events might be attributed to the hyper inflammatory state related to NLRP3 inflammasome activation and ACE2 signaling pathway modulation [16]. In this regard, administration of prophylactic or therapeutic doses of anticoagulant agents are recommended in COVID-19 patients in special conditions.
The possible effects of COVID-19 on pregnant women would be the enhanced expression of von Willebrand factor and the reduced expression of Claudin-5 and vascular endothelial cadherin in endothelium of decidual cells and chorionic villus of placenta which were observed in severe cases of COVID-19 in pregnant women. Additionally, histopathological studies revealed that SARS-CoV-2 could affect placental vessels associated with thrombosis, infarction, and vascular remodeling [17]. The probable mechanism of COVID-19 severity in elderly patients in comparison to the younger adults would be the theory of thymic aging. The age related severity of COVID-19 can be attributed to the thymic function that can affect the immune system. In this regard, it has been shown that the aged thymic function in thymus can result in the reduced number of naïve T cells, increased number of self-reactive T cells, and the enhanced regulatory T cells production. Aged thymic function in lung can be associated with the reduced antiviral function of T cells and plasma cells. Also, cytokine storm related to the inflammatory processes and lung tissue fibrosis after inflammation would be expectable [18].
The possible effect of genetic predisposition on severity of COVID-19 infection has been studied [19]. Results revealed that polymorphisms on related genes including the encoding angiotensin converting enzyme 1 (ACE1) gene was associated with different comorbidities including obesity, diabetes, hypertension, cardiovascular disorders, and renal insufficiency disorders. In this regard, it has been reported that DD carriers of the ACE1 gene exhibited higher ACE1 activity. Also, lack of control over inflammatory mediators including IL-1ß, IL-6, IL-8, and TNF-ɑ was obvious in patients with congenital alpha 1 anti-trypsin deficiency disorders. It has been hypothesized that ACE1/ACE2 imbalance in these patients, is responsible for severe conditions after COVID-19 infection due to the inflammatory nature of these comorbidities and direct relation with RAAS [19].
3. Timelines and challenges of developing a vaccine for COVID-19
Vaccine development is a very time-consuming and expensive process that confronts different challenges. This process usually takes a period of 10–15 years in which most of the time is spent in clinical trials phases. However; this time can be shortened in lethal infectious pandemics such as the COVID-19 era [20]. Clinical trial phases can be divided into three phases. These phases are between pre-clinical experiments and vaccine approval licensure. Phase I clinical trial will be performed in a small group of healthy and immunocompetent volunteers who not been previously exposed to the intended pathogen. The main goal of this phase would be the assessment of vaccine safety and immunogenicity. In case phase I is successfully passed, clinical trial will pass to phase II . The main focus of phase II clinical trial is on vaccine immunogenicity and in a lower extent the vaccine efficacy. In this phase, the larger population will be included and the effect of gender, age, and ethnicity will be assessed. In phase III vaccine efficacy will be assessed in multi-centers. Phase III clinical trial will be conducted in an active outbreak in order to assess the efficacy and adverse reactions of the vaccine [20], [21].
The time of design and development of COVID-19 vaccine has been shortened to approximately one year. This shortened period is due to the emergency of the situation and the lethal nature of the ongoing pandemic.
4. Concerns about vaccine development from an ethical perspective
Different concerns regarding the ethical perspective of COVID-19 vaccine design and development can be divided into three categories including:
1) Concerns regarding vaccine design and experimental studies (“research ethics”)
2) Concerns regarding receivers of COVID-19 vaccine (“distributive justice public health ethics”),
3) Concerns regarding individual interests and public health autonomy (“clinical ethics”) [22].
4) Post-licensure assessment of vaccine safety and efficacy is another ethical concern that should be taken in to consideration.
5) Administration of placebo during controlled trial study for new vaccine. This concern might be overcome to some extent through the informed consent forms that should be signed by each volunteer participant [22].
Although different ethical concerns have been raised during vaccine design and development to protect against COVID-19, vaccination is still the best and the only way to manage lethal pandemics.
5. Design of vaccines in conjugation with nanotechnology intervention
Emerging need of accessible safe vector for antigen and gene delivery result in recruitment of different nanocarriers in order to design nucleic acid-based (mRNA and DNA vaccines) and subunit vaccines [138], [139]. Many of the novel vaccines have been designed by the encapsulation of nucleic acid or peptide/protein within the polymeric and lipid based nanoparticles [23]. In this regard, different COVID-19 vaccines have been designed and approved including Pfizer-BioNTech and Moderna vaccines considered as mRNA vaccines. Another nanotechnology-based platform is the protein-based vaccines including Novavax in which the recombinant S protein of the SARS-CoV-2 has been conjugated to the surface of the virus-like particles (VLP). During vaccine design and development, nanoparticles can be used not only as the carrier of the antigens but also as a co-deliver adjuvants for boosting immune response against desired antigens [23]. Co-encapsulation or conjugation of antigens and adjuvants can result in synchronized and targeted delivery to the antigen-presenting cells (APCs). This targeted co-delivery can reduce the need for required antigen dose to protect against COVID-19. Pfizer/BioNTech and Moderna vaccines that are among the lipid based nanoparticles have been designed in such a way to encapsulate mRNA viruses and provide protection against nuclease degradation [24], [25].
6. Types of COVID-19 vaccines
6.1. Nucleic acid-based vaccines
Nucleic acid-based therapeutics and vaccines can be considered as a novel platform for drug and vaccine delivery. They provide a great opportunity to design drugs for undruggable targets and prepare vaccine not only for infectious diseases but also for cancer [26], [27], [28]. Nucleic acid based vaccines can be prepared by plasmid DNAs as well as RNAs [29]. Although the potential of mRNA for vaccine development has been demonstrated since 1990 s, plasmid DNA gained more attention in the past decades. The main reason is that the plasmid DNA is more stable than mRNA in terms of intrinsic stability of DNA double helixes rather than the single strands of mRNA and the effect of degrading RNase in various tissues and organs [30].
Another main characteristic of nucleic acid vaccines is feasibility of their scale up procedure and providing great opportunity for their industrial production in a short time. The process of manufacturing of conventional vaccines such as killed or live-attenuated ones is a time-consuming process in addition to safety concerns of working with virulent viruses [31]. This process for the development of a new vaccine takes more than a decade to reach the market. Novel vaccine platforms have the great advantage of quick development process [32].
Nucleic acid-based vaccines have provided robust and efficient platform for the development of novel vaccines. However, there are some challenges which may limit their wide application. Stability can be considered as the main obstacle for future development of such vaccines particularly mRNA vaccines [33]. The term of stability refers to in-vivo stability following the administration and the stability during the shelf-life (storage). Vaccine stability must allow wide distribution of the vaccines globally. While Moderna and Pfizer/BioNTech vaccines (mRNA-based vaccines) are stored at −15 and −25 ◦C and between −60 and −90 ◦C, respectively, the wide application of such formulations need well-equipped distribution chain [33], [34]. Also, there is not enough available data to show the stability of these formulations for a long period of time in terms of colloidal stability and the extent of entrapped mRNA inside the lipid nanoparticles (LNPs).
6.1.1. DNA vaccines
As for COVID-19, there are several DNA and mRNA based vaccines in different clinical trial phases as well as pre-clinical studies [35]. For instance, plasmid DNA vaccine, INO-4800, developed by Inovio Pharmaceuticals/International Vaccine Institute contains a pGX001 expression vector to encode the SARS-CoV-2 Spike protein [36]. While the preclinical studies resulted in the induction of humoral and cellular responses in animal models, the clinical trial phase I was conducted with the intradermal (ID) administration of two doses. The results of these clinical trials demonstrated that overall expected responses were observed in individuals enrolled in the study [37]. The company obtained authorization to conduct phase III efficacy trial on its DNA vaccine on August 26th, 2021 [38]. One of the major advantages of this DNA based vaccine is its storage condition. According to the previous formulations developed by Inovio Pharmaceuticals/International Vaccine Institute, this vaccine can be stored at 25 ◦C and might be stable for years at 4 ◦C. Such storage condition can be considered as a great benefit for a wide worldwide distribution [36], [39]. Another DNA vaccine candidate for COVID-19 is AG0301-COVID19 developed by Osaka University/AnGes/ Takara Bio which is administered in two doses via IM injections [40]. The same candidates based on plasmid DNA is also developed by other companies. For example, ZyCoV-D and GX-19 are two vaccine candidates developed by Cadila Healthcare and Genexine Consortium, respectively [40]. These candidates can be used via ID or IM injections. On August 20th, 2021, the Indian drug regulator approved ZyCoV-D as the first plasmid DNA vaccine for restricted use. This vaccine is injected intradermally via jet injector [41]. According to the results of interim phase III clinical trial reported by Cadila Healthcare, the vaccine showed 66% efficacy in the prevention of symptomatic disease and 100% effectiveness in moderate to severe Covid-19 [42]. bacTLR-Spike is another DNA based vaccine developed by Symvivo company. Bifidobacterium longum has been widely used as a probiotic bacterium. The company designed a plasmid encoding S protein of SARS-CoV2 and transferred the plasmid into the B.lungum. This plasmid is able to be actively replicated. Therefore, the bacterium can be administered orally in a single dose as the first-in-human trial of an oral COVID-19 vaccine [40].
Another plasmid DNA candidate in clinical trial is CORVax12 which encodes the S protein of the virus. This vaccine candidate is administered intradermally by electroporation. CORVax12 can be used alone or in combination with another plasmid DNA encoding IL-12. Since the role of IL-12 in augmentation of the efficacy of immunotherapy has been shown in several studies, the company decided to add the plasmid encoding IL-12 in the vaccination regimen to augment anti SARS-CoV2 immune responses [43], [44], [45], [46], [47]. Early preclinical data demonstrated the induction of IgG responses against the S protein and the receptor-binding domain (RBD). Also, the titer of neutralizing antibodies increased. The vaccination regimen with IL-12 increased, at least transiently, the neutralizing antibodies [48].
In summary, there are various DNA-based COVID-19 vaccines in clinical trials and they are different in the storage condition as well as route of administration and the target protein for the induction of immune response [32].
6.1.2. mRNA vaccines
Beside DNA vaccines, mRNA vaccines attracted great attention to combat COVID-19. Moderna and Pfizer/BioNTech vaccines are the first formulations obtained Emergency Use Authorization (EUA) from FDA and conditional approval by EMA. mRNA-1273 developed by Moderna (US) is an LNP encapsulated mRNA to express SARS-CoV-2 full-length S protein and is administered via IM injections in two doses. The promising results of the clinical trials resulted in the approval of this vaccine for wide human application. Likewise, Pfizer/BioNTech developed LNP encapsulated mRNA vaccines (BNT162a1, b1, b2, c2). BNT162b1 encodes SARS-CoV2 RBD while BNT162b2 mRNA vaccine has been designed to encode full length S protein in the prefusion conformation. The results of clinical trial to compare these two vaccines demonstrated that both of them are able to enhance neutralizing antibodies. Since BNT162b2 mRNA vaccine led to less severe adverse events, the company decided to proceed this formulation for obtaining approval [25], [32], [49], [50]. While BNT162b2 obtained the approval from regulatory authorities worldwide, the results of phase I study of the BNT162b1 mRNA vaccine (Pfizer/BioNTech) showed promising safety profile and high levels of humoral and T cell responses. This mRNA vaccine has been designed to encode a trimerized, secreted version of the SARS-CoV-2 spike (S) glycoprotein RBD. Following the administration of two doses, CD4+ and CD8+ T-cell-mediated responses was increased; leading to the production of IFN-γ [51]. There are other mRNA based vaccine candidates in clinical trials developed by other companies. For example, CVnCoV vaccine which I developing by CureVac company is a LNP encapsulated mRNA vaccine encoding S protein and is administrated in two doses via IM injection.[32], [35]. It was expected that this vaccine could be cheaper and more stable rather than the other mRNA vaccines developed by Moderna and BioNTech/Pfizer. There are various explanations for the disappointing results of CureVac’s mRNA vaccine from the dose of the vaccine to the mRNA design. Although it is too early to draw conclusion, it seems that the application of uridine instead of pseudouridine in the mRNA design could be considered as the main reason for lower levels of immune responses produced by this vaccine. While BioNTech/Pfizer and Moderna mRNA vaccines employ modified nucleotide of pseudouridine to reduce human inflammatory responses against foreign mRNA, CureVac vaccine used normal uridine. Also, the differences between the non-coding regions of the CureVac vaccine and the other mRNA vaccines in addition to the differences of storage temperature could play roles in the obtained results [52], [53]. Also, ARCT-021(Lunar-COV19) and LNP-nCoVsaRNA vaccines are under investigation by Acturus/ Duke-NUS and Imperial College, London, respectively. These vaccines are replicating mRNA and supposed to be used via IM injections [32], [35].
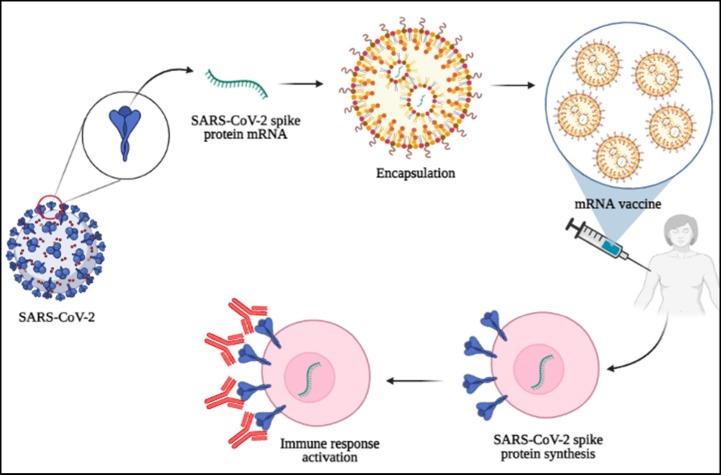
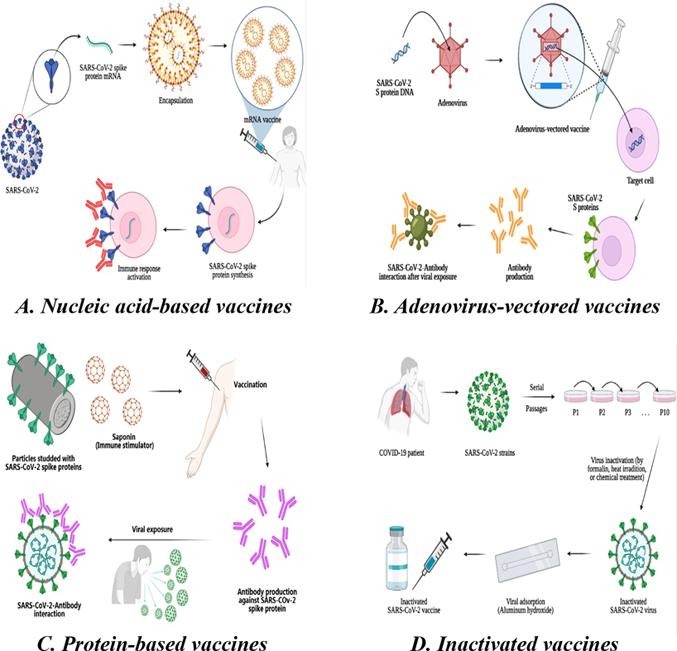
In summary, the development of nucleic acid vaccines has opened up new horizons not only for novel vaccine platforms but also will facilitate the development of nucleic acid-based therapeutics. The current phase IV clinical trials for LNP-encapsulated mRNA vaccines are the biggest trial for a nanomedicine. This can be considered as a new basis for the design and development of future nanotechnology-based therapeutics including gene or drug delivery approaches. A schematic view of the design and development of mRNA COVID-19 vaccines is shown in Fig. 1 .
Fig. 1.
A schematic view of the design and development of mRNA SARS-CoV-2 vaccines.
6.1.2.1. BNT162b2 (Pfizer/BioNTech)
LNPs have been used for the development of mRNA vaccines including BNT162b2. This carrier system is used to protect the nucleic acid materials from premature degradation and facilitate its delivery to the targeted cells. The promising results of the clinical trials resulted in the approval for EUA of this vaccine for wide human application. Likewise, Pfizer/BioNTech developed LNP encapsulated mRNA vaccines (BNT162a1, b1, b2, c2). Two of these candidates encode the S protein and two encode the optimized RBD [32]. BNT162b2 vaccine should be administered with a dosage of 0.3 mL containing 30 µg nucleosid-modified mRNA through IM route. This vaccine has been scheduled in 2 doses with 21-days interval. The BNT162b2 (Pfizer/BioNTech) vaccine showed an efficacy of 95% against COVID-19. This efficacy could be achieved at least 7 days after the second dose administration [54]. Also, it has been reported that the efficacy of 52% would be achieved after administration of the first dose [55]. On December 2020, the Food and Drug Administration (FDA) confirmed the EUA for Pfizer/BioNTech vaccine to prevent against COVID-19 in individuals of15 years and older [56]. Among vaccine recipients, several case reports have been declared about the severe allergic reactions including anaphylaxis after vaccination. Most of these reports were attributed to patients with previous history of allergies, allergic reactions, and anaphylaxis [56], [57]. One of the drawbacks in the design and development of mRNA vaccines would be their storage and stability considerations. Pfizer/BioNTech COVID-19 vaccine should be stored at −70˚C which might disrupt the feasibility and affordability of worldwide distribution especially by the developing countries [33], [34]. The American Academy of Physician Assistants (AAPA) and the National Association of Pediatric Nurse Practitioners (NAPNP) have considered COVID-19 vaccination for children and adolescents aged 18 years or younger [58]. On May 2021, the FDA issued the EUA for Pfizer/BioNtech vaccine administration in children of 12–15 years of age [59]. The neutralization effect of the Pfizer/BioNTech COVID-19 vaccine against Alpha, Beta, Gamma, and Epsilon SARS-CoV-2 variants was reduced by 2, 6.5, 6.7, and 4-folds, respectively in comparison to the wild type virus [60]. The efficacy of single-dose of Pfizer/BioNTech vaccine against Alpha and Delta variant were reported 30.7% and 48.7%, respectively. While fully vaccinated individuals showed the efficacy of 93.7% and 88% against Alpha and Delta SARS-CoV-2 variants, respectively after receiving the second dose [61]. The effectiveness of Pfizer/BioNTech COVID-19 vaccine on Delta variant was also investigated in a study conducted in the United Kingdom (UK). The results showed that 14 days following the second injection, the vaccine provides 92% protection against high viral load. However, its effectiveness reduces to 85% and 78% after two and three months, respectively [62]. Pfizer/BioNTech vaccine, also known as Comirnaty®, was approved on 23rd August 2021 by the FDA as the first COVID-19 vaccine to be administered in individuals of 16 years of age and older to protect against COVID-19 infection.
6.1.2.2. mRNA-1273 (Moderna)
The mRNA-1273 (Moderna) COVID-19 vaccine is a nucleoside modified mRNA vaccine in which mRNA is encapsulated in LNPs. This mRNA vaccine would express the pre-fusion SARS-CoV-2 spike proteins that can induce immune reactions [63]. Due to the weak response after administration of the first dose, Moderna vaccine has been scheduled to be given in 2 doses with a 28-day interval to boost the immune system with a stronger reaction [64]. Each dosage volume is 0.5 mL containing 100 µg mRNA that should be administered IM. Results of phase III clinical trials revealed the efficacy of 94.1%, 14 days after the second dose [63], [65]. Vaccine efficacy was measured in terms of prevention against symptomatic and laboratory data confirming infection by COVID-19 [63]. The most common adverse reactions after the first dose were mild to moderate reactogenicity signs and symptoms including systemic and local adverse reactions. Systemic adverse reactions were mostly common after administration of the second dose. These mild to moderate adverse reactions were occurred within the first to second days of vaccination mostly recovered after 2–3 days [63]. Several case reports of severe allergic reactions including anaphylaxis have been documented after the first dose as well. Anaphylactic reaction has occurred within minutes to hours (median time of 7.5 min) after vaccine injection. Most of these anaphylactic reactions were observed in patients with documented history of allergic reactions or previous history of anaphylactic reactions [66]. Also, some reports regarding the lymphadenopathy after mRNA COVID-19 vaccination (Pfizer/BioNTech and Moderna) have been documented [67]. Furthermore, there are some case series that reported the occurrence of a delayed localized hypersensitivity reaction after the first and second dose of Moderna vaccine. These pruritic hypersensitivity reactions with a median onset of 7 days were completely self-limited and not considered as contraindications for vaccination [68]. Finally, on December 2020, the FDA has approved the EUA of Moderna COVID-19 vaccine for adults of 18 years of age and older [63]. Results of a preliminary study revealed that maternal vaccination (with mRNA vaccines) during pregnancy will induce neonatal protection against COVID-19 due to trans-placental transfer of SARS-CoV-2 antibodies, but further larger longitudinal follow-up studies are required to assess the safety of these vaccines in pregnant women [69]. Reports are suggestive of decreased neutralization effects of Moderna COVID-19 vaccine against Alpha, Beta, Gamma, and Epsilon SARS-CoV-2 variants by 1.8, 8.6, 4.5, and 2.8-folds, respectively comparing to the wild type virus [60].
6.2. Viral vector vaccines
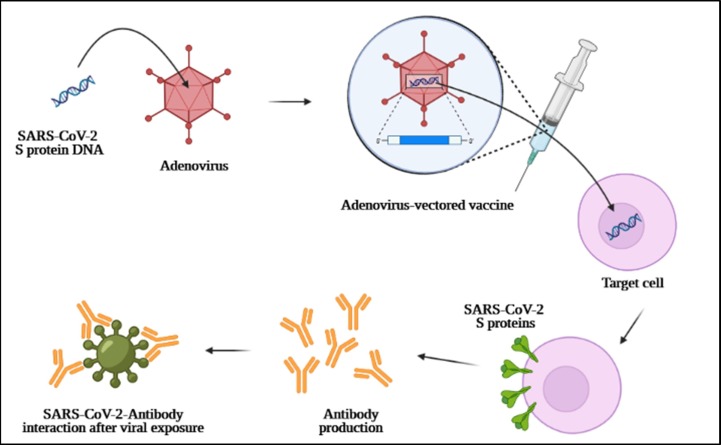
Adenovirus-vectored vaccines are promising prophylactic strategies against COVID-19 infections. Adenoviruses are non-enveloped dsDNA viruses. These viruses can be considered as the cause of non-severe and self-limiting human infections including ocular and respiratory tract infections. Adenovirus vectored vaccines are considered as high-tech vaccine platforms [70]. In recent years adenoviruses are used as suitable carriers in the field of nanotechnology for gene delivery purposes. In order to stop viral replication, the E1 and E3 viral genes should be omitted and replaced with desired antigens including SARS-CoV-2 S proteins for the purpose of vaccine design and development. The most important advantage of using adenovirus as vectors for drug and gene delivery and vaccine development is their inability to integrate into the human genome which warranted safety after administration [70]. These vaccines have the potential of targeted gene delivery to the cells that results in efficient gene transduction and immune response induction. Adenovirus-vectored vaccines can induce a high level of antigenic protein expression that leads to antibody production, cytotoxic T cell activation and viral elimination [71]. A schematic view of the design and development of adenovirus-vectored COVID-19 vaccines was shown in Fig. 2 .
Fig. 2.
A schematic view of the design and development of adenovirus-vectored SARS-CoV-2 vaccines.
6.2.1. ChAdOx1 nCoV-19 (AstraZeneca/Oxford)
AstraZeneca/Oxford vaccine (Vaxzevria®), also known as ChAdOx1 nCoV-19 recombinant adenovirus vaccine, is synthesized based on S glycoprotein. The AstraZeneca/Oxford vaccine can express SARS-CoV-2 spike protein (S-protein) using a chimpanzee adenovirus [72]. In this strategy, the sequence coding SARS-CoV-2 amino acids and the tissue plasminogen activator (tPA) leader at the 5′ end are encapsulated in a shuttle plasmid [71]. On December 2020, the UK regulatory authority, Medicines and Healthcare products Regulatory Agency (MHRA) approved the emergency use of the AstraZeneca/Oxford vaccine in Europe. At first, this vaccine was designed as a single-dose COVID-19 vaccine, but due to the suboptimal response after the first dose administration, the booster dose was suggested [73]. The first shot of AstraZeneca/Oxford vaccine administration was accompanied with 43% and 80% lower risk of emergency hospitalization and severe infection that cause hospitalization after COVID-19 infection, respectively [73]. Two full doses (standard doses) of this vaccine should be injected with an interval of at least 4 to 12 weeks. It has been reported that at least 14 days after the second dose administration to adults, it induces an efficacy of 70.4% against COVID-19 infection [74]. According to the results of clinical trials, dosing intervals of less than 6 weeks had an average efficacy of 55%, while administration of the second dose with an interval of 12 weeks could result in 81% efficacy. Therefore, delay in receiving the booster dose would be beneficial for the AstraZeneca/Oxford vaccine [73]. In general, 28 to 34 days after the first dose of AstraZeneca/Oxford COVID-19 vaccine, hospitalization was 94% reduced, while this rate was 81% in elderly individuals older than 80 years old [73]. The primary efficacy of this vaccine was assessed in participants from UK, Brazil, and South Africa as the seronegative symptomatic COVID-19 with PCR positive swab test at least 14 days after the second dose administration [75]. The secondary efficacy analysis was performed at least 22 days after the second shot and the exploratory outcome was antibody response measurement [76]. Although its efficacy in older adults (≥56 years old) was controversial, on 15th February 2021, the World Health Organization (WHO) added this vaccine to the Emergency Use Listing (EUL) in adults and elderly while acknowledging the limited data on its efficacy in the elderly group [77]. It has been reported that in participants with age 18–55 years old, extending the interval between the first and the second dose of AstraZeneca/Oxford vaccine to 12 weeks would be accompanied by higher efficacy and binding antibody response. These results can be attributed to the sufficient efficacy of the single-dose vaccine during the first 90 days of the administration that support the 12-week dose interval vaccination program [76]. The main advantage of this three-month dose interval vaccination is the accelerated COVID-19 pandemic control by early vaccination of the largest number of individuals in nations [76]. Till now, the AstraZeneca/Oxford vaccine has not been applied or approved for emergency use by the United States Food and Drug Administration (US FDA). Although this vaccine showed proper protection against the UK SARS-CoV-2 variant (Alpha variant), but failed to show good protection against the South African variant (Beta variant) with an efficacy of about 10% [77]. The neutralization titer against the Beta variant was about 9-folds lower in comparison to the wild-type virus [73]. The effectiveness of this vaccine after the first dose immunization against Alpha and Delta SARS-CoV-2 variants were 48.7% and 30.7%, respectively. The efficacy of the AstraZeneca/Oxford vaccine against the Delta variant after full vaccination (two-dose administration) was 67% while it was effective against the Alpha variant about 74.5% [61].
The main advantage of the AstraZeneca/Oxford vaccine is its affordability in low/middle-income countries and nations [78]. Also, this vaccine can be stored at 2–8 ˚C which makes it a suitable candidate vaccine for global distribution [75]. Unfortunately, some reports on thromboembolic events with the AstraZeneca/Oxford vaccine has resulted in ceasing its use in many European countries in March 2021. European Medicines Agency (EMA) reported 30 cases with thromboembolic events, most of them developed venous thromboembolism (VTE), among 5 million vaccine recipients [79]. Results of a recent study revealed that the possible mechanism of thrombosis and thrombocytopenia associated with AstraZeneca/Oxford vaccine in recipients with the age of 32 to 54 years old would be the high levels of antibody against platelet factor 4 (PF4). Although these individuals had no previous exposure to heparin, they developed a rare “vaccine-induced immune thrombotic thrombocytopenia” that was similar to heparin-induced thrombocytopenia (HIT) [80]. Also, according to the results of a recent preprint study, transcription of wild-type and codon-optimized Spike open reading frames enables alternative splice events that lead to C-terminal truncated, soluble Spike protein variants. These soluble Spike variants may initiate severe side effects, including thrombotic events, when binding to ACE2-expressing endothelial cells in blood vessels. The underlying disease mechanism of vaccine-related thrombotic events is called “vaccine-induced COVID-19 mimicry” syndrome (VIC19M syndrome). Also, a recent case report study documented a new case of “isolated carotid arterial thrombosis” that occurred 8 days after receiving the first shot of AstraZeneca/Oxford vaccine in a 31 years old man who presented acute aphasia, headache, and hemiparesis [81]. Recently, the EMA declared that although there is a possible link between the rare risk of thromboembolic events and thrombocytopenia and AstraZeneca/Oxford vaccine within 2 weeks of administration, still the overall benefits of vaccination overweighs its risks. Therefore, due to reduced rate of hospitalization and death from COVID-19, its administration should proceed but patients should be informed of the possible adverse reactions including shortness of breath, chest pain, swelling in leg, persistent abdominal (belly) pain, neurological symptoms containing severe and persistent headaches or blurred vision, and tiny blood spots under the skin beyond the site of injection. It is noteworthy that EMA continues its study on the safety and efficacy of AstraZeneca/Oxford vaccine. On 8th September 2021, EMA released a COVID-19 vaccine safety update on Vaxzervia® vaccine. It has been reported that Guillain-Barré syndrome was documented in 833 cases out of 592 million doses of Vaxzervia® injection. Also, the incidence of thrombosis with thrombocytopenia syndrome (TTS) was 1,503 out of 592 million shots of Vaxzervia®. Other rare but major adverse reactions that have been reported after Vaxzervia® injection were capillary leak syndrome (CLS) and cerebral venous sinus thrombosis (CVST) without thrombocytopenia. Also, in this vaccine safety update, abdominal pain (stomachache), pain in extremities (leg and arm), and influenza-like symptoms (including chills, fever, rhinorrhea, cough and sore throat) have been added to the side effects of Vaxzervia® vaccine. However, there was no evidence regarding the casual relationship between the menstrual disorders and Vaxzervia® injection [82].
6.2.2. Ad26.COV2-S (Johnson & johnson; Janssen)
Johnson and Johnson vaccine, also known as Janssen, is a type of adenovirus-vectored vaccine. The SARS-CoV-2 spike protein gene was added to the adenovirus 26 DNA (Ad26.COV2-S). Administration of this modified adenovirus vaccine can enter the cell and release its viral DNA. Thereafter, the spike protein will be produced through the viral DNA, the immune system will be evoked and antibodies will be produced against these SARS-CoV-2 spike proteins. Therefore, vaccination may lead to prevention of SARS-CoV-2 infection after viral exposure [83]. Since this adenovirus that delivers SARS-CoV-2 spike protein DNA cannot multiply, infection will not occur after vaccination. Storage temperature of this vaccine is 2–8 ˚C due to the stability of SARS-CoV-2 DNA molecules which eases its distribution worldwide in comparison to the previously approved vaccines such as Pfizer/BioNTech and Moderna that need an ultra-cold storage conditions [83].
It has been reported that in 90% of vaccine recipients, antibody production was taken place after the first dose administration. However, the amount of produced antibodies were much higher after the second dose [84]. Data from Johnson and Johnson revealed that administration of a single dose of this vaccine would result in 66% efficacy in providing protection against moderate to severe COVID-19 and 100% efficacy against COVID-19-induced hospitalization and death [83]. Johnson & Johnson vaccine has received EUA by the US FDA. In January 2021, results of interim data on 44,325 participants reported that 28 days after the single-dose administration, the Janssen vaccine showed the efficacy of 66% and 85% against moderate-to-severe and severe COVID-19 infection, respectively [70]. There are several reports of splanchnic venous thrombosis, cerebral venous sinus thrombosis (CVST), and deep vein thrombosis [73]. After the development of blood clots in six cases out of 6.8 million vaccine recipients in the USA, administration of the Johnson and Johnson vaccine was suspended in USA. All of these 6 reports of blood clots occurred in women between the ages of 18 and 48 years old. Thrombosis and thrombocytopenia have occurred between 6 and 13 days post-vaccination. Still, the EMA and MHRA have declared that although AstraZeneca/Oxford and Johnson and Johnson COVID-19 vaccines might be associated with rare thrombocytopenia and thrombosis side effects, their benefits in current pandemic control will overweigh their possible risks. US FDA and the Centre for Disease Control (CDC) have warned that people who are vaccinated with Johnson and Johnson vaccine should be aware of the potential signs and symptoms of thrombosis occurrence within three weeks post-vaccination. These alarming signs and symptoms are severe headache, abdominal pain, leg pain, or shortness of breath [84]. The Janssen COVID-19 vaccine is effective against Alpha, Beta, Gamma, and Epsilon SARS-CoV-2 variants [85]. The Johnson & Johnson vaccine has received the EUA and now is going to be used in individuals of 18 years and older in the USA and many other countries [70].
6.2.3. Gam-COVID-Vac (Gamaleya’s Sputnik V)
Sputnik V, also known as Gam-COVID-Vac, is a type of heterologous recombinant adenovirus (rAd26 and rAd5)-vectored vaccine [70], [86]. Adenoviral vector-delivered antigens are capable to induce both cellular and humoral immunity after administration of the first dose. However, administration of the second dose of these vaccines would be accompanied by a long-term immune response [87]. The major drawback of these adenovirus-vectored vaccines would be the immunogenicity against vector components. This drawback can be minimized and overcome through the prime-boost heterologous vaccination approach using two different vectors for the first and second dose of vaccination. Sputnik V is a combined vector vaccine which consists of rAd26/ rAd5 that delivers SARS-CoV-2 DNA encoding spike proteins (rAd26-S and rAd5-S respectively). These heterologous recombinant adenoviruses (rAd26-S and rAd5-S) are injected in a 21-day interval dosage respectively. This vaccine has received early Russian approval after the successful results of the phase I/II clinical trials [87], [88]. Results of phase III clinical trials revealed that Gamaleya’s Sputnik V vaccine had an overall efficacy of 91.6% on day 21 after the first dose (the day of the second dose) administration. In this study, the starting point for vaccine efficacy measurement was considered as PCR-positive COVID-19 cases counting 21 days after the first dose (the day of the second dose) administration. Also, it showed a mean efficacy of 91.8% among participants older than 60 years old. Sputnik V vaccine’s efficacy against severe COVID-19 was 100% [87]. Also, this vaccine results in robust induction of both humoral and cellular immunity, 42 days and 28 days after first dose administration respectively. Humoral immune response was assessed in terms of neutralizing antibody titers and RBD-specific IgG titers while the cellular immune response was assessed as enhanced IFN-γ secretion among vaccine recipients [87]. Sputnik V vaccine was well tolerated in recipients and no major serious adverse reactions related to the vaccine administration were reported. The most common local and systemic adverse reactions related to the Sputnik V vaccine were pain at the injection site, hyperthermia, headache, asthenia, and muscle and joint pain [89]. This vaccine was formulated in two dosage forms with different storage stability considerations: The liquid form which was stored at −18 ˚C and the lyophilized powder which was stored at 2–8 ˚C. Ministry of Health of the Russian Federation approved the lyophilized powder form to obtain ease of global vaccine distribution [87]. Results of the phase III clinical trial of Sputnik V vaccine revealed promising efficacy and safety [90] and now this vaccine is approved in Russia and many other countries for public use. In April 2021, Sputnik V vaccine has received EUA from Russia and some other countries [70].
6.2.4. Ad5-nCoV (CanSino)
CanSino vaccine (Ad5-nCoV), also known as CanSinoBIO, is a type of adenovirus 5 (Ad5)-vectored vaccine that delivers the DNA to the cells which encode SARS-CoV-2 spike proteins [91]. Results of an early phase I, dose-escalation, open-label, non-randomized study in China revealed that CanSino vaccine was well-tolerated and could induce both humoral and cellular immunity 28 days and 14 days after single-dose administration respectively [92], [93]. This vaccine was tested in three dosing schedules including low dose (5 × 1010 viral particles), middle dose (1 × 1011), and high dose (1.5 × 1011). Although the high dose administration was more immunogenic in terms of B cell and T cell induction, the reactogenicity was also higher after this high dose vaccine administration in comparison to the low and middle doses. [92]. The most commonly reported local and systemic adverse reactions related to vaccination were pain at the injection site, fever, headache, fatigue, and muscle pain that were considered as mild to moderate in severity assessments. Also, the most common reactions after high dose administration (1.5 × 1011) was fever, dyspnea, joint pain, fatigue, and muscle pain with more severity. No serious adverse reactions were reported during 28 days post-vaccination [92]. Results of phase II clinical trial in a larger population revealed that single-dose CanSino vaccine administration with a dosage of 5 × 1010 viral particles would be considered safe and could efficiently induce both humoral and cellular immune systems. According to the results of this study, older adults might show lower post-vaccination immune response with CanSino in comparison with the younger population. So, it seems that a second booster dose might be necessary in older adults to induce better post-vaccination immunity [94].
Since COVID-19 is a serious respiratory infection, it has been hypothesized that mucosal vaccination in addition to the routine IM injection can induce better protection against COVID-19 challenges. Results of a recent animal study revealed that single-dose mucosal administration of the CanSino vaccine could protect against the upper and lower respiratory tract against SARS-CoV-2 infection. Furthermore, single-dose IM injection of the CanSino vaccine could protect mice lungs against SARS-CoV-2 infection and could significantly reduce viral replication after exposure.Hence, according to the results of this animal study, it seems that mucosal immunity in combination with systemic immunity can significantly protect against SARS-CoV-2 infection and reduce person-to-person transmission. However, controlled clinical trials and human studies are required to assess the safety and efficacy of this novel route of administration [95].
6.2.5. VIR-7831 (Medicago)
VIR-7831 is a plant-based, non-infectious, viral vector vaccine against COVID-19 that has been developed by Medicago Company in Canada. This plant-based vaccine has been designed through the utilization of virus-like particles of SARS-CoV-2 including S protein to mimic the structure and function of SARS-CoV-2 after injection [96]. Since these plant-based viruses lack the genetic materials of SARS-CoV-2, host infectivity would not be expected after vaccination. The virus-liked particles, used in the development of the COVID-19 vaccine, can be easily recognized by the immune system and induce sufficient immune response. Now, VIR-7831 is in phase III clinical trial. This vaccine is cheaper to produce and so it is more affordable to use in developing countries, which is the main advantage of this vaccine. Furthermore, this vaccine might better prevent new variants and emerging mutations [96].
6.3. Protein-based vaccines
Among the majority of anti-viral vaccines being licensed for human use are the protein-based vaccines. Protein-based vaccines are considered as classic vaccine platforms. Classical vaccine platforms have significantly contributed to global health breakthroughs such as eradication of smallpox. Protein-based vaccines can either be composed of a purified protein of the virus, recombinant protein, virus-infected cells or virus-like particles. Among the all types of developed vaccines, peptide-based vaccines are the safest, which is due to the exclusion of the epitopes that cause antibody-dependent infections. However, low immunogenicity of these vaccines might be considered as a disadvantage which is solved by adding the adjuvants that helps inducing a robust immune response [97]. The S protein of the SARS-CoV-2 is the most suitable antigen to induce the neutralizing antibodies against the pathogen [98]. Two protein-based COVID-19 vaccines that are currently approved for human use are NVX-CoV2373® (Novavax) and EpiVacCorona (Vector institute). A schematic view of the design and development of Novavax COVID-19 vaccine is shown in Fig. 3 .
Fig. 3.
A schematic view of the design and development of Novavax COVID-19 vaccine.
6.3.1. NVX-CoV2373® (Novavax)
NVX-CoV2373 is a recombinant rSARS-CoV-2 nanoparticle vaccine, developed by a biotechnology company, Novavax. NVX-CoV2373 is composed of trimeric full-length spike glycoproteins of SARS-CoV-2 and Matrix-M1 adjuvant [99]. The adjuvant is a saponin-based Matrix-M™ that has demonstrated potent as well as well-tolerated effects. It causes the antigen presenting cells to enter the injection site and enhance presentation of antigens in local lymph nodes resulting in evoked immune response. Spike glycoprotein of full-length wild-type SARS-CoV-2, which facilitates attachment of the virus to human angiotensin-converting enzyme 2 (hACE2) receptor in order to enter the cell, serves as a fundamental target for development of antibodies and vaccines [100], [101].
NVX-CoV2373 is being tested in two major Phase III trials, one taking part in the U.K that has shown a 96.4% efficacy against the wild virus strain and an overall efficacy of 89.7% and the PREVENT-19 trial in the U.S. and Mexico that has recently been started. Phase II trials are currently ongoing in South African and the USA as well as Australia. Novavax has announced successful results of its Phase IIb study conducted in South Africa. Phase IIb trial in South Africa has shown 48.6% efficacy against a newly emerged variant.
Preliminary data from clinical trials show that NVX-CoV2373 is 95.6% effective against the original variant of SARS-CoV-2 but also provides protection against the newer variants B.1.1.7 (85.6%) and B.1.351 (60%) [102]. Reports of a randomized controlled trial in South Africa indicate that NVX-CoV2373 vaccine was efficacious in preventing COVID-19 caused by the B.1.351 variant, while prior infection with the wild SARS-CoV-2 did not provide protection against this variant [103]. Among HIV-negative participants, NVX-CoV2373 has shown 60.1% efficacy against B.1.351 variant [102], [104].
Doses of vaccine and adjuvant in a clinical setting (5 and 25 µg rs SARS-CoV-2 adjuvant with 50 µg Matrix-M1) that were administered in 2 doses, ensued sterilizing immunity in both the lungs and nasal passage, suggestive of the protection provided by the vaccine against upper and lower respiratory tract infection caused by COVID-19 and has exhibited to inhibit transmission. The mentioned doses were evaluated in phase I and in 131 healthy adult participants with 18 to 59 years of age and in phase II of study in 750 to 1,500 participants of 18 to 84 years of age, including those with co-morbid diseases [105]. Although results of phase I is in support of both doses of SARS-CoV-2 rS/Matrix-M1 adjuvant regarding safety and immunology, the lower dose (5 µg) offers the advantage of dose-sparing [106]. Based on both nonclinical and Phase I data, the chosen dose for Phase III study is 5 µg SARS-CoV-2 rS/50 µg Matrix-M1 adjuvant being administered intramuscularly (IM) on Days 0 and 21.
A recent report also shows that two-dose regimens of 5 μg and 25 μg rSARS-CoV-2 with 50 μg Matrix-M1 adjuvant in participants were well tolerated. Following the first dose, both doses of NVX-CoV2373 were well tolerated but there was a trend toward a higher incidence of local reactogenicity with the higher dose in both younger and older adults but no apparent differences in anti-spike protein binding IgG levels and neutralizing antibody responses by dose level. Following the second dose, both dose levels of NVX-CoV2373 were well tolerated despite increased frequencies and intensities of local and systemic reactogenicity in both younger and older adults, and a trend toward higher incidences of local and systemic reactogenicity with the higher dose remained. NVX-CoV2373 induced robust levels of anti-spike protein binding. Both younger and older adults demonstrated no significant difference between the two dose levels with respect to IgG and neutralizing antibodies levels. Data on the antibody responses are in support of the low-dose and two vaccination regimens of 5-μg NVX-CoV2373 [107].
Regarding adverse reactions reported from NVX-CoV2373 after receiving the first dose, pain at the injection site was the most frequent of all. Headache, fatigue and malaise were also among the main reported adverse reactions. Following the second dose, adverse reactions were more or less similar with mean duration of slightly higher which lasted less than 3 days. Local tenderness, fatigue and joint pain were observed after the second dose. Severe local adverse reactions were rare but occurred more often in the seronegative NVX-CoV2373 group. More frequent adverse reactions included headache (20–25%), muscle pain (17–20%), and fatigue (12–16%). Fever was reported in only one participant. All adverse reactions resolved in 2 days after injection. Considering laboratory data, hemoglobin level dropped in 6 patients which resolved 7–21 days after injection with no clinical significance. Rise in liver enzymes was observed in four individuals that resolved within 7–14 days after vaccination [106].
Taken together, the data released from clinical trials indicate that the rSARS-CoV-2/Matrix-M1 vaccine is highly immunogenic and well tolerated, however awaiting data from a phase IIa/b and two phase III ongoing studies which evaluates the efficacy and safety of the two-dose regimen of 5-µg NVX-CoV2373 in South Africa, the United Kingdom, the USA and Mexico are yet to be released.
6.3.2. EpiVacCorona (Vector Institute)
EpiVacCorona vaccine developed by the Vector institute, the State Research Center of Virology and Biotechnology, is a protein-based vaccine containing a chemically synthesized peptide immunogens of the S protein of SARS-CoV-2 conjugated to a carrier protein and adsorbed on aluminum hydroxide. Novel SARS-CoV-2 N protein chosen as the carrier protein in EpiVacCorona, augments peptide immunogenicity. This protein is conserved, induces no virus neutralizing antibodies, but contains virus-specific T-cell epitopes and is involved in memory T-cell production. Since peptide vaccines contain only short sequences of the viral protein, it adds to the safety profile of the vaccine which makes it suitable for use in immunocompromised patients. Alongside, EpiVacCorona is effective against antigenically variable strains due to containing conservative SARSCoV-2 epitopes. Additionally, ease of production and stability of the components of the vaccine allows the process of producing vaccine for large populations [108].
Phase I–II clinical trial of the EpiVacCorona vaccine is being conducted in Russia at present. Phase III is ongoing in Russia enrolling>3,000 participants with an age of 18 years and older. Participants will receive two separate doses of 225 ± 45 μg/0.5 mL IM with the interval of three to four weeks [104], [109].
Data from phase I–II clinical trials report that the only adverse reaction observed in participants after receiving the first dose is pain at the site of injection (14%). No systematic reactions such as headache, fever and myalgia were observed. Biochemical parameters as well as ECG of participants remained normal. Regarding heamatological indicators, changes in the level of monocytes were observed in 14% of patients. The most common adverse reaction in phase II, was also pain at injection site (14%). All local reactions were mild and transient and lasted less than 2 days. Increase in body temperature for 12 hrs was also observed in one participant. EpiVacCorona Vaccine demonstrated low reactogenicity and only 9.3% and 4.7% of participants experienced mild local reactions after the first and the second dose respectively [109].
6.3.3. ZF 2001 (Ahui Zhifei longcom Biopharmaceutical)
As mentioned earlier, protein-based vaccines, contain either purified or synthesized viral proteins. Although subunit vaccines are safer than other vaccine platforms, they entail adjuvants and booster shots [110]. ZF2001 vaccine is a dimeric RBD (Receptor-binding Domain) adjuvant with aluminum hydroxide that has been developed by Ahui Zhifei Langcom pharmaceutical. This vaccine targets SARS-CoV-2 S protein RBD which is accountable for engaging its cellular receptor, human hACE2 [111], [112]. ZF2001 is the first RBD-based protein subunit vaccine have reported clinical data. It has been approved for emergency use in Uzbekistan and China [26], [27]. ZF2001 provokes modest cellular as well as notable humoral immune response. ZF2001 has shown reasonable immunogenicity and is reported to be well-tolerated in phases I and II clinical trial. ZF2001 is now being evaluated in a phase III clinical trial (NCT04646590) [27].
There are no severe adverse reaction reports conveyed with ZF2001 and reported mild side effects resolved within four days of vaccination. Seroconversion rates for doses of 25 and 50 µg after two-weeks of the final dose were 76% and 72% respectively. On the other hand, seroconversion rate in groups that received three doses was increased to 97% (25 µg) and 93% (50 µg) at the same time point. Phase III clinical trial is enrolling with the three-doses of 25 µg schedule for large-scale safety and efficacy evaluation, since no evidence for a dose-dependent manner of enhancing immunogenicity was observed [113].
ZF 2001 has shown to greatly preserve neutralizing titres, against 501Y.V2 also known as B.1.351, which first emerged in South Africa, compared to the titres against the original SARS-CoV-2 and the currently circulating D614G virus [28].
6.4. Inactivated vaccines
A conventional method in vaccine development is using whole inactivated virus (WIV) Several WIV vaccines have been designed for SARS-CoV-2 prevention including Bharat Biotech (Covaxin®) from India and Sinovac and Sinopharm from China [114]. A schematic view of inactivated SARS-CoV-2 vaccine development is depicted in Fig. 4 . Besides many advantages attributes to this conventional vaccine platform, the main drawback of these inactivated vaccines is the moderate immunogenicity. So, concurrent administration of adjuvant is required to enhance immune response. Also, administration of the booster doses of inactivated vaccines are required to confirm their efficacy against COVID-19 infection [115].
Fig. 4.
A schematic view of the design and development of inactivated SARS-CoV-2 vaccines.
6.4.1. BBIBP-CorV (Sinopharm Beijing) & WIBP-CorV (Sinopharm Wuhan)
Sinopharm Beijing, also known as BBIBP-CorV, is a kind of inactivated SARS-CoV-2 vaccine. During vaccine design, different SARS-CoV-2 strains containing 19nCoV-CDC-Tan-HB02 (HB02), 19nCoV-CDC-Tan-Strain03 (CQ01), 19nCoV-CDC-Tan-Strain04 (QD01) were isolated from the bronchoalveolar lavage samples of the hospitalized COVID-19 patients [116]. Results revealed that the HB02 strain would be optimal due to the highest replication and virus yield generation in comparison to other strains. Therefore, the HB02 strain was selected for SARS-CoV-2 inactivated vaccine development. Thereafter, the HB02 strain was purified and passaged as viral stock. The viral stock was expanded on Vero cells. The original seed for inactivated SARS-CoV-2 vaccine production was obtained after 10 generation adaptation and passages with a sequence homology of 99.95%. In order to inactivate SARS-CoV-2 production, ß-propiolactone was mixed with the harvested virus sample. The viral inactivation process was accompanied by vial infectivity elimination and formulation stability. The stock vaccine solution showed protective antigens (SARS-CoV-2 spike proteins) in Western blot analysis. The results of stained electron microscopy revealed oval shape SARS-CoV-2 viruses (average diameter of 100 nm) with spike proteins on their surface [116]. Results of the animal studies revealed that two-dose immunization with inactivated Sinopahrm vaccine could induce high titers of neutralizing antibodies. Moreover, Sinopharm vaccine was genetically stable and considered safe in animal models [116]. Results of phase I/II clinical trial revealed that Sinopharm as an inactivated SARS-CoV-2 vaccine was safe and well-tolerated. Also, it was immunogenic in healthy vaccine recipients. This vaccine was injected with a different dosage of 2, 4, and 8 µg with an interval of 28 days. Sinopharm (BBIBP-CorV) was tested in two groups of recipients comprising 18–59 and ≥ 60 years old participants. The results revealed that the immunogenicity and neutralizing antibody production were obvious in 100% of vaccine recipients in both groups (18–59 and ≥ 60 years old) after two-dose administration. The most common adverse reactions reported were pain and fever. No major severe adverse reaction was documented among vaccine recipients [117]. Results of an interim analysis of the phase III clinical trial revealed that Sinopharm vaccine had an efficacy of 79.34% [72], [114]. These vaccine has received Chinese authority approval for public use. Results of a recent study declared that the B.1.1.7 variant showed little resistance to neutralization with Sinopharm and Sinovac vaccines and convalescent plasma while B.1.351 variant showed more resistance against Sinopharm and Sinovac vaccines and convalescent plasma in comparison to the wild type variant (Wuhan-1 reference strain) [118]. Sinopharm Beijing should be administered in 2 doses of 4 µg through IM injection 21 or 28 days apart and Sinopharm Wuhan should be administered in 2 doses of 5 µg through IM injection with a 21-day interval. The reported efficacy of Sinopharm Wuhan and Sinopharm Beijing vaccines are 72.51% and 79.34% respectively. The neutralization effect of Sinopharm COVID-19 vaccine against Beta SARS-CoV-2 variant was reduced by 1.6-fold in comparison to the wild type virus. Finally, on 7th May 2021, WHO has approved the emergency use of the Sinopharm Beijing vaccine.
6.4.2. CoronaVac (Sinovac)
Sinovac, also known as CoronaVac, is a kind of WIV vaccine that has been designed and manufactured in China [114]. In this regard, the CN02 strain of the SARS-CoV-2 vaccine was propagated in African green monkey kidney cells. Then, SARS-CoV-2 was harvested and inactivated using ß-propiolactone. Thereafter, the sample was concentrated and purified. Finally, it was adsorbed on aluminum hydroxide. Before the sterilization process, the aluminum hydroxide complex was diluted with water, sodium chloride, and phosphate-buffered saline (PBS) [119]. Data from phase I/II clinical trial revealed that Sinovac was well-tolerated among healthy participants and could moderately induce immunogenicity [120]. The most common adverse reaction was pain at the injection site. Also, results revealed that fever was less common with Sinovac in comparison with RNA vaccines and adenovirus vectored-vaccines [120]. According to the published data, Sinovac was safe and well-tolerated among older adults with age ≥ 60 years old and could induce sufficient titers of neutralizing antibody [119]. During double-blind, randomized, placebo-controlled phase III clinical trial, the efficacy and safety of the adsorbed inactivated COVID-19 vaccine, Sinovac was evaluated in participants with age 18–59 and ≥ 60 years old population after two-dose IM administration of 0.3 µg/0.5 mL inactivated SARS-CoV-2 vaccine with a 28-day interval [121]. The primary efficacy endpoint for Sinovac was considered as detection of PCR-positive COVID-19 cases two weeks after the second dose administration. Also, the safety endpoint was assessed by local and systemic adverse reactions monitoring within one week after vaccination [121]. Sinovac showed the efficacy of 50%, 65%, 78%, and 91% in different countries that were participated in phase III clinical trials [72]. The least efficacy was attributed to the Brazilian population with an average efficacy of 50.38% [114], [122]. Results of a recent study revealed that the B.1.1.7 variant showed little resistance to neutralization with the Sinovac vaccine while the B.1.351 variant was more resistant in comparison to the wild-type variant [118]. Sinovac has received Chinese approval for public administration.
6.4.3. BBV152 (Bharat Biotech)
Bharat Biotech COVAXIN, also known as BBV152, is an inactivated SARS-CoV-2 vaccine designed in India [123], [124]. COVAXIN is produced by inactivation of the NIV-2020–770 strain using ß-propiolactone. This strain contained the Asp614Gly mutation in their spike proteins [125]. Results of the phase I clinical trial revealed that COVAXIN was well-tolerated and safe among participants and could induce immunogenicity and enhance immune response (predominantly T-cell response) after vaccination [125]. The most common reported adverse reactions were pain at the site of injection, fever, fatigue, nausea, and vomiting being more prevalent after the first shot [126]. Results of phase II clinical trial on the efficacy and safety of two-dose administration of COVAXIN, with a 28-day interval, showed long-term humoral and cellular immunogenicity about 3 months after the second dose. Bharat Biotech COVAXIN is considered safe and cost-benefit [127]. COVAXIN, an immunogenic inactivated SARS-CoV-2 vaccine, can be stored at 2–8 ˚C [128] which eases its global distribution. Results of an interim analysis revealed the efficacy of 81% after two-dose COVAXIN injection with an interval of 28 days apart but further studies are required [126]. Also, COVAXIN showed sufficient efficacy in neutralizing antibody production against UK SARS-CoV-2 variant (B.1.1.7 variant) [129]. COVAXIN has received approval for emergency use by the Drugs Controller General of India (DCGI) [126].
7. Antibody therapy
The potential mechanism of convalescent plasma therapy would be the presence of neutralizing antibodies including IgG and IgM antibodies against SARS-CoV-2 that can prevent viremia [1]. The level of these antibodies would be higher after 2 to 3 weeks from the initiation of COVID-19 symptoms [130]. Immunotherapy would be a promising approach in the prevention and treatment of infectious diseases including COVID-19. Most of the monoclonal antibodies have been designed to target the SARS-CoV-2 S protein and RBD. These monoclonal antibodies can inhibit the interaction of RBD of S1 protein subunit with its desired receptor ie. ACE2 receptor in host cells. Some monoclonal antibodies can also act on the S2 subunit and block the virus from receptor binding. Besides these monoclonal antibodies, the human neutralizing antibodies are other promising therapeutic options in the management of COVID-19 infection. In this regard, B38, H4, and 47D11were introduced first as potential neutralizing antibodies to block SARS-CoV-2 [131]. Additionally, neutralizing antibodies that are isolated from convalescent plasma of recovered COVID-19 patients can act as a promising therapeutic agents in COVID-19 management. S309 is the most promising monoclonal antibody of all that has been designed against SARS-CoV-2 S protein RBD. Alongside, the three-dimensional (3D) structure alignment studies showed that S309 had the most neutralization potency against both SARS-CoV and SARS-CoV-2 S protein RBDs [132]. The main target site of these neutralizing antibodies is a segment in the RBD domain which has a 19 amino acid-length and is called N318-V510 that can induce passive immunization against COVID-19 infection [133].
According to the latest update on NIH COVID-19 Treatment Guideline, in non-hospitalized patients with mild to moderate COVID-19 infection and at higher risk of clinical signs and symptoms progression, the monoclonal antibodies including casirivimab plus imdevimab or sotrovimab can be administered. These monoclonal antibodies have received Emergency Use Authorization (EUA) in outpatient settings, however, they are not authorized to administer in hospitalized patients. It is to note that administration of the low-titer COVID-19 convalescent has no longer been authorized for treatment of COVID-19 patients. In immunocompetent hospitalized patients dependent on mechanical ventilation, the NIH guideline recommended against the use of convalescent plasma therapy. Furthermore, high-titer convalescent plasma therapy is no longer authorized for the treatment of immunocompetent hospitalized patients who are not on mechanical ventilation. However, data regarding high-titer convalescent plasma therapy in immunocompromised hospitalized patients and also in outpatient settings is not sufficient and we cannot either recommend nor reject the routine administration due to unknown safety and efficacy [134]. Because of these controversial results, it has been suggested that measuring antibody titers (IgM and IgG) before convalescent plasma transfusion would be beneficial [135].
8. Discussion and conclusion
Over 70% of the world population should be vaccinated in order to achieve a desirable community immunity. Although each COVID-19 vaccine has various advantages and disadvantages over the others, accessibility and affordability of vaccines approved by the official authorities of health organizations, especially in developing countries, would be essential to terminate this pandemic. This review emphasized on different vaccine platforms as well as related mechanisms, safety, and efficacy of available COVID-19 vaccines. A summary of characteristics of these vaccines including brand names, innovator company names, dosage, manufacturing technology, dosing intervals, storage, efficacy, and adverse reactions have been summarized in Table 1 . Also, characteristics of various COVID-19 vaccine platforms in different age groups have been summarized in Table 2 . Furthermore, the immunologic mechanisms of different vaccine platforms have been summarized in Table 3 .
Table 1.
A summary of different characteristics of available COVID-19 vaccines.
| Vaccine trade name | Innovator company | Technology of production | Final status | Dosage | Number of shots | Interval | Storage | Efficacy | Common adverse reactions | Major adverse reactions |
|---|---|---|---|---|---|---|---|---|---|---|
| BNT162b2 mRNA | Pfizer/BioNTech | RNA vaccine | Emergency use in US FDA | 0.3 mL (30 µg nucleosid-modified mRNA) IM | 2 | 21 days | −70 ˚C | 95% | Pain, swelling, redness, fever, fatigue, headache, chills, vomiting, diarrhea, muscle pain, joint pain, lymphadenopathy, shoulder injury, right axillary lymphadenopathy, and right leg paresthesia. | Allergic reactions including anaphylaxis, paroxysmal ventricular arrhythmia, and syncope. Multisystem inflammatory syndrome (MIS). |
| mRNA-1273 | Moderna | RNA vaccine | Emergency use in US FDA | 0.5 mL (100 µg mRNA) IM | 2 | 28 days | −20 ˚C | 94.5% | Pain, swelling, redness at the site of injection, fever, fatigue, headache, chills, vomiting, arthralgia, myalgia, urticaria. (These clinical symptoms were mild to moderate after the first dose of vaccine and moderate to severe after the second dose of vaccine). | Allergic reactions including anaphylaxis, facial swelling, and Bell’s palsy |
| ChAdOx1 nCoV-19 | AstraZeneca/Oxford | Adenovirus-vectored vaccines | Emergency use in UK, WHO’s Emergency Use Listing | 0.5 mL (5 × 1010 viral particles) IM | 2 | 4–12 weeks | 2–8 ˚C | 70% | Headache, nausea, vomiting, diarrhea, myalgia, arthralgia,, injection site tenderness, pain, warmness, pruritus, bruising, swelling, and erythema, fatigue, malaise, chills, and fever. | Thrombosis with thrombocytopenia syndrome (TTS), Guillain-Barré syndrome, capillary leak syndrome (CLS), cerebral venous sinus thrombosis (CVST) without thrombocytopenia. |
| Ad26.COV2.S (Janssen) | Johnson & Johnson | Adenovirus-vectored vaccines | Emergency use in US FDA | 0.5 mL (5 × 1010 viral particles) IM | 1 | – | 2–8 ˚C | 66.3% | Fever | Venous thromboembolism |
| Gam- COVID- Vac (Sputnik V) | Gamaleya Research Institute | Adenovirus-vectored vaccines | Early use in Russia | 0.5 mL (1 × 1011 viral particles rAd26-S, followed by 1 × 1011 viral particles rAd5-S) IM | 2 | 21 days | −18 ˚C | 92% | Flu-like illness, injection site pain, headache, and asthenia. | Renal colic, deep vein thrombosis, and extremity abscess was observed in patients older than 60 years old. But no association was found between serious adverse events and COVID-19 vaccine administration. |
| Ad5- nCoV | CanSino | Adenovirus-vectored vaccines | Phase III clinical trials, Chinese approval | 0.5 mL (5 × 1010 viral particles) IM | 1 | – | −20 ˚C | 65.7% | Injection site pain, soreness, fatigue, and mild fever. | No serious adverse events reported. |
| NVX- CoV2373 | Novavax | Protein-subunit vaccine | Phase III clinical trials | 0.5 mL (5 µg SARS-CoV-2 rS/50 µg Matrix-M1 adjuvant) IM | 2 | 21 days | −20 ˚C | 89.3% | Headache, fatigue and malaise. | No serious adverse events reported. |
| EpiVacCorona | Vector Institute | Protein-subunit vaccine | Early use in Russia | 0.5 mL (225 ± 45 μg) IM | 2 | 21 days | 2–8 ˚C | NA* | Headache, fever and myalgia | No serious adverse events reported. |
| BBIBP- CorV | Sinopharm (Beijing) | Inactivated vaccine | WHO’s Emergency use approval, Chinese approval | 0.5 mL (4 µg in aluminum adjuvant) IM | 2 | 21 to 28 days | 2–8 ˚C | 79% | Pain and fever. | No serious adverse events reported. |
| WIBP- CorV | Sinopharm (Wuhan) | Inactivated vaccine | Chinese approval | 0.5 mL (5 µg in aluminum adjuvant) IM | 2 | 14 to 21 days | 2–8 ˚C | 72.5% | Pain and fever. | No serious adverse events reported. |
| CoronaVac | Sinovac Biotech | Inactivated vaccine | Phase III clinical trials, Chinese approval | 0.5 mL (3 µg in aluminum adjuvant) IM | 2 | 28 days | 2–8 ° C | 50.65% to 83.5% | Injection site pain | No serious adverse events reported. |
| BBV152 | Bharat Biotech | Inactivated vaccine | Phase III clinical trials, Emergency use in India | 0.5 mL (6 µg in Aluminum hydroxiquim-II adjuvant) IM | 2 | 28 days | 2–8 ˚C | 81% | Injection site pain, fever, fatigue, nausea, and vomiting. | No serious adverse events reported. |
*NA: Data not available.
Table 2.
Characteristics of various COVID-19 vaccine platforms in different age groups.
| Vaccine type | Vaccine name | Age group (years) | Approval status | Efficacy | Safety |
|---|---|---|---|---|---|
| Nucleic acid vaccines | BNT162b2 mRNA (Pfizer/BioNTech) | Children (less than12) | Not approved | – | – |
| Adolescents (12–15) | FDA1 EUA2 | 100% | Mild to moderate reactogenicity including local injection site pain and systemic adverse reactions including | ||
| Young adults (>15) | FDA approved | 95% | Mild to moderate reactogenicity and adverse reactions. Rare major adverse reactions. |
||
| Elderly | FDA approved | >90% | Mild to moderate adverse reactions. | ||
| mRNA-1273 (Moderna) | Children | Not approved | – | – | |
| Adolescents (12–17) | EMA3 approval | Ongoing study | |||
| Young adults (≥18) | FDA EUA | 94.5% | Mild to moderate reactogenicity and adverse reactions. Rare major adverse reactions. |
||
| Elderly | FDA EUA | >90% | Mild to moderate local reactions including injection site pain and systemic reactions including lethargy. | ||
| Adenovirus-vectored vaccines | ChAdOx1 nCoV-19 (AstraZeneca/Oxford) | Young adults (≥18-69) | WHO4 EUA | 70% | Safe and well-tolerated. Mild to moderate adverse reactions with rare major reactions including thrombosis. |
| Elderly (70–84) | WHO EUA | Sufficient neutralizing antibody production | Lower adverse effects than in younger adults. Low reactogenicity. |
||
| Ad26.COV2.S (Janssen) | Younger adult (18–65) | FDA EUA | 66.3% | Safe and well-tolerated. Mild to moderate adverse reactions with rare major reactions including thrombosis. |
|
| Older adults (>65) | FDA EUA | NA | Lower adverse effects than in younger adults. | ||
| Gam-COVID-Vac (Gamaleya’s Sputnik V) | NA | Early use in Russia | 92% | Well-tolerated with no serious adverse reaction. | |
| Ad5-nCoV (CanSino) | Younger adult (18–55) | Phase III clinical trials, Chinese approval | 65.7% | Well-tolerated with no serious adverse reaction. | |
| Older adults (>55) | Phase III clinical trials, Chinese approval | Lower neutralizing antibody production in comparison to the younger adults. | |||
| Protein-based vaccines | NVX-CoV2373(Novavax) | Younger adult (18–65) | Phase III clinical trials | 89.3% | Well-tolerated with no serious adverse reaction. |
| Older adults (65–84) | Phase III clinical trials | Sufficient neutralizing antibody production | |||
| EpiVacCorona | NA | Early use in Russia | NA | Well-tolerated with no serious adverse reaction. | |
| Inactivated vaccines | BBIB-CorV (Sinopharm) | NA | WHO EUA | 79% | Well-tolerated with no serious adverse reaction. |
| WIBP-CorV (Sinopharm) | NA | Chinese approval | 72.5% | Well-tolerated with no serious adverse reaction. | |
| CoronaVac (Sinovac) | Children & adolescents (3–17) | Under clinical trials investigations | Good immunogenicity | Good safety and tolerability. Mild to moderate adverse reactions including injection site pain. |
|
| Younger adults (18–59) | Early use in Russia | 50.6–83.5% | low reactogenicity and mild local reactions | ||
| Younger adults (≥60) | NA | NA | NA | ||
| BBV152 (Bharat Biotech) | NA | Phase III clinical trials, EUA in India | 81% | Well-tolerated with no serious adverse reaction. |
1Food and Drug Administration
2Emergency Use Authorization
3European Medicines Agency
4World Health Organization
3Data not available regarding the efficacy in different age groups
Table 3.
The immunologic mechanisms of different COVID-19 vaccine platforms.
| Vaccine name | Humoral responses | Cellular responses |
|---|---|---|
| BNT162b2 mRNA (Pfizer/BioNTech) | Protein S1-binding antibody production after the first and second dose of vaccination. NAb1 production after the second dose. |
Enhancement of antigen-specific CD4+ and CD8+ T cells secreting INFγ2 and IL-23 after the second dose. |
| mRNA-1273 (Moderna) | Protein S-binding antibody production after the first and second dose. Significant production of NAb after the second dose. |
Enhancement of CD4+ T cells secreting TNF4 and INFγ after the second dose. |
| ChAdOx1 nCoV-19 (AstraZeneca/Oxford) | Protein S-binding antibody production (specifically IgG3 and IgG1) after the first and second dose. NAb production after the first and second dose. |
Enhanced CD4+ T cells secreting TNF and INFγ after the first and second dose. |
| Ad26.COV2.S (Janssen) | Protein S-binding antibody production after the single dose. NAb production after the single dose. |
Enhancement of antigen-specific CD4+ and CD8+ T cells secreting INFγ and IL-2 after the single dose. |
| Gam-COVID-Vac (Gamaleya’s Sputnik V) | Protein S-binding antibody production after the first and second dose. NAb production after the first and second dose. |
Enhanced CD4+ and CD8+ T cells secreting INFγ after the first and second dose. |
| Ad5-nCoV (CanSino) | RBD5-binding antibodies, anti-RBD binding antibodies, and NAb production after the single dose. | Enhanced T cells secreting INFγ after the single dose |
| NVX-CoV2373 (Novavax) | Protein S-binding antibody production after the first and second dose. NAb production after the first and second dose. |
Enhanced CD4+ T cells secreting INFγ, TNF, and IL-2 after the second dose. |
| CoronaVac (Sinovac) | RBD-specific binding antibody and NAb production after the second dose. | No cellular immunity |
| BBIB-CorV (Sinopharm) | Binding antibodies against whole inactivated SARS-CoV-2 and NAb production after the second dose. | No cellular immunity |
| WIBP-CorV (Sinopharm) | Binding antibodies against whole inactivated SARS-CoV-2 and NAb production after the second dose. | No cellular immunity |
| BBV152 (Bharat Biotech) | Anti-S binding antibodies production after the first and second dose. NAb production after the first and second dose. |
Enhancement of CD4+, CD45RO+ T cells secreting INFγ and TNF after the second dose. |
1 Neutralizing antibodies
2Interferone Gamma
3Interleukin 2
4Tumor necrosis factor
5Receptor-binding domain
The coverage of different COVID-19 vaccines against various SARS-CoV-2 variants that have been reported from different sources has been summarized in Table 4 .
Table 4.
A summary of different SARS-CoV-2 variants and suitable vaccines for each variant.
| SARS-CoV-2 variants | WHO label | Source of detection | Suitable vaccines |
|---|---|---|---|
| D614G (wild type) | – | Wuhan city of China |
|
| B.1.1.7 (20I/501Y.V1) | Alpha | United Kingdom | |
| B.1.351 (20H/501Y.V2) | Beta | South Africa |
|
| P1 (20 J/501Y.V3) | Gamma | Brazil & Japan | |
| P2 | Zeta | Brazil | NA* |
| P3 | Theta | Philippines | NA* |
| B.1.427 / B.1.429 (CAL20.C) | Epsilon | California | |
| B.1.525 | Eta | Multiple countries | NA* |
| B.1.526 | Lota | New York | NA* |
| B.1.617.1 | Kappa | India | NA* |
| B.1.617.2 | Delta | India | |
| C.37 | Lambda | Peru | NA* |
| B.1.621 | Mu | Colombia | NA* |
*NA: Data not available.
Although several vaccines with different manufacturing technologies have been approved for COVID-19, with their own specific characteristics regarding their efficacy and side effects, due to urgent need for at least 11 billion doses of vaccine for effective vaccination of the at least 70% of the world population, accessibility and affordability of vaccines is an important issue that the health service authorities around the world faces. Although various vaccines with different efficacies are available, fast vaccination of the world population is recommended to prevent the emergence of the new variants of the virus that may be resistant to developed vaccines.
9. Final viewpoint on vaccine development and usage
Based on CDC recommendation and due to the SARS-CoV-2 Delta variant pandemic, moderately to severely immunocompromised patients should receive an additional booster dose of mRNA vaccines after completion of the initial 2 dose shots. This booster dose should be administered at least 28 days after the second dose of Pfizer/BioNTech or Moderna COVID-19 vaccines. Since the level of immunity after 2 doses administration in these immunocompromised patients would not be equal to the immunocompetent individuals, the booster dose is required in order to improve initial response. Also, in immunocompetent individuals administration of an additional booster dose after the second dose would be promising to achieve better protection against COVID-19 which is now being applied in some countries including USA, Israel, Turkey, and the United Arab Emirates. However, at this time, CDC does not recommend additional booster doses in any population other than moderately to severely immunocompromised patients.
The new COVID-19 cases in many countries have increased significantly in both vaccinated and non-vaccinated populations. Although public vaccination has been initiated and completed in many countries including USA, some people were not convinced to be vaccinated. Therefore, the nations and their health policies should encourage all people to receive vaccine to prevent further viral spread among vaccinated and non-vaccinated populations. Also, the other reason for COVID-19 infection after vaccination can be attributed to the fact that the available vaccines do not have 100% efficacy and therefore infection and death are still possible after vaccination. However; the rate of mortality and severe infection has significantly reduced after massive vaccination. Furthermore, the new SARS-CoV-2 variants including the Delta variant, with a faster transmission rate would be the other cause of increased number of cases after public vaccination in some nations.
In order to prevent new ongoing SARS-CoV-2 variants, massive vaccination of people around the world should be accomplished as soon as possible. Also, based on the published data, it seems that mRNA vaccines including Pfizer/BioNTech and Moderna have shown higher efficacy and protection against COVID-19 infection. Additionally, these vaccines might have a longer duration of action in comparison to others. Furthermore, using mRNA vaccines, the new SARS-CoV-2 variants can be targeted specifically and their high efficacy against COVID-19 infection can be preserved.
According to the details provided in this review, comparison among different available COVID-19 vaccines and decision-making about the suitability of each vaccine for different nations can be easily accessible for clinicians, pharmacists, and researchers in this field.
10. Study limitation
The main limitation of this study was the lack of access to the clinical data on available COVID-19 vaccines developed in Eastern countries. The data on their efficacy, safety, and adverse reactions were limited. Also, the long-term adverse reactions of available COVID-19 vaccines around the world are not clear yet and they will be announced during phase IV clinical trials and post-marketing.
11. Future scope/direction of the present review
Further clinical data from available COVID-19 vaccines especially vaccines developed in Eastern countries will be published in the future. Also, many vaccines in clinical trial phases will be passed hopefully and become available for nations to accelerate massive vaccination. Furthermore, exact planning on the rate of vaccine production is necessary to provide massive vaccination in a short period of time. In the end, the rare and also long-term adverse reactions of available COVID-19 vaccines with different platforms will be announced during the phase IV clinical trials and post-marketing period.
CRediT authorship contribution statement
Parisa Ghasemiyeh: Writing – original draft, Writing – review & editing. Soliman Mohammadi-Samani: Conceptualization, Project administration, Supervision, Writing – original draft, Writing – review & editing. Negar Firouzabadi: Conceptualization, Writing – original draft, Writing – review & editing. Ali Dehshahri: Conceptualization, Writing – original draft, Writing – review & editing. Afsaneh Vazin: Conceptualization, Writing – original draft, Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.intimp.2021.108162.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- 1.Ghasemiyeh P., Mohammadi-Samani S. COVID-19 outbreak: Challenges in pharmacotherapy based on pharmacokinetic and pharmacodynamic aspects of drug therapy in patients with moderate to severe infection. Heart Lung. 2020;49(6):763–773. doi: 10.1016/j.hrtlng.2020.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ghasemiyeh P., Borhani-Haghighi A., Karimzadeh I., Mohammadi-Samani S., Vazin A., Safari A., et al. Major neurologic adverse drug reactions, potential drug–drug interactions and pharmacokinetic aspects of drugs used in covid-19 patients with stroke: A narrative review. Ther. Clin. Risk Manag. 2020;16:595. doi: 10.2147/TCRM.S259152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ghasemiyeh P., Mohammadi-Samani S., Vazin A. Micronutrients Supplementation in Pregnant Women during COVID-19 Pan-demy: Pros and Cons. Trends. Pharma. Sci. 2021;7(3) [Google Scholar]
- 4.Mirjalili M., Shafiekhani M., Vazin A. Coronavirus disease 2019 (COVID-19) and transplantation: pharmacotherapeutic management of immunosuppression regimen. Ther. Clin. Risk Manag. 2020;16:617. doi: 10.2147/TCRM.S256246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zamai L. Upregulation of the Renin-Angiotensin System Pathways and SARS-CoV-2 Infection: The Rationale for the Administration of Zinc-Chelating Agents in COVID-19 Patients. Cells. 2021;10(3):506. doi: 10.3390/cells10030506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ghasemiyeh P., Mohammadi-Samani S. Iron Chelating Agents: Promising Supportive Therapies in Severe Cases of COVID-19? Trends Pharma. Sci. 2020;6(2):65–66. [Google Scholar]
- 7.Ghasemiyeh P, Mohammadi-Samani S. The necessity of early anti-inflammatory therapy initiation in cases with mild-to-moderate COVID-19: A personal experience from an attending pharmacist and his resident. Acta Bio-medica: Atenei Parmensis. 2021;92(3):e2021250-e. [DOI] [PMC free article] [PubMed]
- 8.Anand U., Jakhmola S., Indari O., Chandra Jha H., Chen Z.-S., Tripathi V., et al. Potential therapeutic targets and vaccine development for COVID-19 management: A review on the recent update. Front. Immunol. 2021;12:2454. doi: 10.3389/fimmu.2021.658519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Palatnik-de-Sousa C.B. What Would Jenner and Pasteur Have Done About COVID-19 Coronavirus? The Urges of a Vaccinologist. Front. Immunol. 2020;11:2173. doi: 10.3389/fimmu.2020.02173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Greenwood B. The contribution of vaccination to global health: Past, present and future. Philos. Trans. R Soc. B Biol. Sci. 2014;369(1645):20130433. doi: 10.1098/rstb.2013.0433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Negahdaripour M., Shafiekhani M., Moezzi S.M.I., Amiri S., Rasekh S., Bagheri A., Mosaddeghi P., Vazin A. Administration of COVID-19 Vaccines in ImmunocompromisedPatients. Int. Immunopharmacol. 2021;99:108021. doi: 10.1016/j.intimp.2021.108021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao J., Zhao S., Ou J., Zhang J., Lan W., Guan W., et al. COVID-19: Vaccine Development Updates. Front. Immunol. 2020;11:3435. doi: 10.3389/fimmu.2020.602256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patel C, Brotherton JM, Pillsbury A, Jayasinghe S, Donovan B, Macartney K, et al. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: what additional disease burden will a nonavalent vaccine prevent? 2018;23(41):1700737. [DOI] [PMC free article] [PubMed]
- 14.DeFrancesco L. The 'anti-hype' vaccine. Nat Biotechnol. 2017;35(3):193–197. doi: 10.1038/nbt.3812. [DOI] [PubMed] [Google Scholar]
- 15.Chugh R.M., Bhanja P., Norris A., Saha S. Experimental models to study COVID-19 effect in stem cells. Cells. 2021;10(1):91. doi: 10.3390/cells10010091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gedefaw L., Ullah S., Leung P.H., Cai Y., Yip S.-P., Huang C.-L. Inflammasome Activation-Induced Hypercoagulopathy: Impact on Cardiovascular Dysfunction Triggered in COVID-19 Patients. Cells. 2021;10(4):916. doi: 10.3390/cells10040916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Flores-Pliego A., Miranda J., Vega-Torreblanca S., Valdespino-Vázquez Y., Helguera-Repetto C., Espejel-Nuñez A., Borboa-Olivares H., Espino y Sosa S., Mateu-Rogell P., León-Juárez M., Ramírez-Santes V., Cardona-Pérez A., Villegas-Mota I., Torres-Torres J., Juárez-Reyes Á., Rizo-Pica T., González R.O., González-Mariscal L., Estrada-Gutierrez G. Molecular Insights into the Thrombotic and Microvascular Injury in Placental Endothelium of Women with Mild or Severe COVID-19. Cells. 2021;10(2):364. doi: 10.3390/cells10020364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang W., Thomas R., Oh J., Su D.-M. Thymic Aging May Be Associated with COVID-19 Pathophysiology in the Elderly. Cells. 2021;10(3):628. doi: 10.3390/cells10030628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yamamoto N., Yamamoto R., Ariumi Y., Mizokami M., Shimotohno K., Yoshikura H. Does Genetic Predisposition Contribute to the Exacerbation of COVID-19 Symptoms in Individuals with Comorbidities and Explain the Huge Mortality Disparity between the East and the West? Int. J. Mol. Sci. 2021;22(9):5000. doi: 10.3390/ijms22095000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sharma O., Sultan A.A., Ding H., Triggle C.R. A Review of the Progress and Challenges of Developing a Vaccine for COVID-19. Front. Immunol. 2020;11:2413. doi: 10.3389/fimmu.2020.585354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sharpe H.R., Gilbride C., Allen E., Belij‐Rammerstorfer S., Bissett C., Ewer K., Lambe T. The early landscape of coronavirus disease 2019 vaccine development in the UK and rest of the world. Immunology. 2020;160(3):223–232. doi: 10.1111/imm.13222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Raus K., Mortier E., Eeckloo K. Ethical reflections on Covid-19 vaccines. Acta Clin. Belg. 2021;1–6 doi: 10.1080/17843286.2021.1925027. [DOI] [PubMed] [Google Scholar]
- 23.Chung Y.H., Beiss V., Fiering S.N., Steinmetz N.F. COVID-19 vaccine frontrunners and their nanotechnology design. ACS Nano. 2020;14(10):12522–12537. doi: 10.1021/acsnano.0c07197. [DOI] [PubMed] [Google Scholar]
- 24.Jackson L.A., Anderson E.J., Rouphael N.G., Roberts P.C., Makhene M., Coler R.N., et al. An mRNA vaccine against SARS-CoV-2—preliminary report. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2022483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mulligan M.J., Lyke K.E., Kitchin N., Absalon J., Gurtman A., Lockhart S., et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature. 2020;586(7830):589–593. doi: 10.1038/s41586-020-2639-4. [DOI] [PubMed] [Google Scholar]
- 26.Dai L., Zheng T., Xu K., Han Y., Xu L., Huang E., An Y., Cheng Y., Li S., Liu M., Yang M.i., Li Y., Cheng H., Yuan Y., Zhang W., Ke C., Wong G., Qi J., Qin C., Yan J., Gao G.F. A universal design of betacoronavirus vaccines against COVID-19, MERS, and SARS. Cell. 2020;182(3):722–733.e11. doi: 10.1016/j.cell.2020.06.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang S., Li Y., Dai L., Wang J., He P., Li C., Fang X., Wang C., Zhao X., Huang E., Wu C., Zhong Z., Wang F., Duan X., Tian S., Wu L., Liu Y., Luo Y.i., Chen Z., Li F., Li J., Yu X., Ren H., Liu L., Meng S., Yan J., Hu Z., Gao L., Gao G.F. Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: two randomised, double-blind, placebo-controlled, phase 1 and 2 trials. Lancet. Infect. Dis. 2021;21(8):1107–1119. doi: 10.1016/S1473-3099(21)00127-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang B., Dai L., Wang H., Hu Z., Yang X., Tan W., Gao G.F. Serum sample neutralisation of BBIBP-CorV and ZF2001 vaccines to SARS-CoV-2 501Y. V2. Lancet Microbe. 2021;2(7):e285. doi: 10.1016/S2666-5247(21)00082-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pardi N., Hogan M.J., Porter F.W., Weissman D. mRNA vaccines-a new era in vaccinology. Nat. Rev. Drug. Discov. 2018;17(4):261–279. doi: 10.1038/nrd.2017.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Houseley J, Tollervey DJC. The many pathways of RNA degradation. 2009;136(4):763-76. [DOI] [PubMed]
- 31.Brisse M., Vrba S.M., Kirk N., Liang Y., Ly H. Emerging Concepts and Technologies in Vaccine Development. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.583077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pushparajah D., Jimenez S., Wong S., Alattas H., Nafissi N., Slavcev R.A. Advances in gene-based vaccine platforms to address the COVID-19 pandemic. Adv. Drug Deliv. Rev. 2021;170:113–141. doi: 10.1016/j.addr.2021.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schoenmaker L., Witzigmann D., Kulkarni J.A., Verbeke R., Kersten G., Jiskoot W., Crommelin D.J.A. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int. J. Pharm. 2021;601:120586. doi: 10.1016/j.ijpharm.2021.120586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sahin U., Karikó K., Türeci Ö. MRNA-based therapeutics-developing a new class of drugs. Nat. Rev. Drug Discov. 2014;13(10):759–780. doi: 10.1038/nrd4278. [DOI] [PubMed] [Google Scholar]
- 35.van Riel D., de Wit E. Next-generation vaccine platforms for COVID-19. Nat. Mater. 2020;19(8):810–812. doi: 10.1038/s41563-020-0746-0. [DOI] [PubMed] [Google Scholar]
- 36.Smith T.R.F., Patel A., Ramos S., Elwood D., Zhu X., Yan J., Gary E.N., Walker S.N., Schultheis K., Purwar M., Xu Z., Walters J., Bhojnagarwala P., Yang M., Chokkalingam N., Pezzoli P., Parzych E., Reuschel E.L., Doan A., Tursi N., Vasquez M., Choi J., Tello-Ruiz E., Maricic I., Bah M.A., Wu Y., Amante D., Park D.H., Dia Y., Ali A.R., Zaidi F.I., Generotti A., Kim K.Y., Herring T.A., Reeder S., Andrade V.M., Buttigieg K., Zhao G., Wu J.-M., Li D., Bao L., Liu J., Deng W., Qin C., Brown A.S., Khoshnejad M., Wang N., Chu J., Wrapp D., McLellan J.S., Muthumani K., Wang B., Carroll M.W., Kim J.J., Boyer J., Kulp D.W., Humeau L.M.P.F., Weiner D.B., Broderick K.E. Immunogenicity of a DNA vaccine candidate for COVID-19. Nat. Commun. 2020;11(1) doi: 10.1038/s41467-020-16505-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.INOVIO, Announces Positive Interim Phase 1 Data For INO-4800 Vaccine for COVID-19 [August 2021]. Available from: https://ir.inovio.com/news-releases/news-releases-details/2020/INOVIO-Announces-Positive-Interim-Phase-1-Data-For-INO-4800-Vaccine-for-COVID-19/default.aspx%202020/.
- 38.INOVIO Receives Authorization to Conduct Phase 3 Efficacy Trial of its COVID-19 DNA Vaccine Candidate, INO-4800 [Sepember 2021]. Available from: https://ir.inovio.com/news-releases/news-releases-details/2021/INOVIO-Receives-Authorization-to-Conduct-Phase-3-Efficacy-Trial-of-its-COVID-19-DNA-Vaccine-Candidate-INO-4800/default.aspx.
- 39.Tebas P, Kraynyak KA, Patel A, Maslow JN, Morrow MP, Sylvester AJ, et al. Intradermal SynCon® Ebola GP DNA Vaccine Is Temperature Stable and Safely Demonstrates Cellular and Humoral Immunogenicity Advantages in Healthy Volunteers. J Infect Dis. 2019;220(3):400-10. [DOI] [PubMed]
- 40.Silveira M.M., Moreira G.M.S.G., Mendonça M. DNA vaccines against COVID-19. Perspect. Challenges. 2021;267:118919. doi: 10.1016/j.lfs.2020.118919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Momin T., Kansagra K., Patel H., Sharma S., Sharma B., Patel J., Mittal R., Sanmukhani J., Maithal K., Dey A., Chandra H., Rajanathan C.TM., Pericherla H.PR., Kumar P., Narkhede A., Parmar D. Safety and Immunogenicity of a DNA SARS-CoV-2 vaccine (ZyCoV-D): Results of an open-label, non-randomized phase I part of phase I/II clinical study by intradermal route in healthy subjects in India. EClin. Med. 2021;38:101020. doi: 10.1016/j.eclinm.2021.101020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zydus applies to the DCGI for EUA to launch ZyCoV-D, the world’s first Plasmid DNA vaccine for COVID-19 [September 2021]. Available from: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwi1vK6YjvryAhVGzBoKHd2IAx4QFnoECAIQAQ&url=https%3A%2F%2Fwww.zyduscadila.com%2Fpublic%2Fpdf%2Fpressrelease%2FZyCoV_D_Press_Release_1_7_2021.pdf&usg=AOvVaw3lC-N3TTAclRV7Q4kIstIW.
- 43.Gringeri M., Mosini G., Battini V., Cammarata G., Guarnieri G., Carnovale C., Clementi E., Radice S. Preliminary evidence on the safety profile of BNT162b2 (Comirnaty): new insights from data analysis in EudraVigilance and adverse reaction reports from an Italian health facility. Human Vaccines & Immunother. 2021;17(9):2969–2971. doi: 10.1080/21645515.2021.1917236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gurajala S. A Sneak Peek into COVID-19 Vaccines-Present Status. J. Clin. Diagnos. Res. 2021;15(3) [Google Scholar]
- 45.Khalvati B., Sheikhsaran F., Sharifzadeh S., Kalantari T., Behzad Behbahani A., Jamshidzadeh A., Dehshahri A. Delivery of plasmid encoding interleukin-12 gene into hepatocytes by conjugated polyethylenimine-based nanoparticles. Artif. Cells Nanomed. Biotechnol. 2017;45(5):1036–1044. doi: 10.1080/21691401.2016.1202256. [DOI] [PubMed] [Google Scholar]
- 46.Misra S.K., Pathak K., Pathak D., Yadav R. Current updates on COVID-19 vaccines. Asian J. Pharma. Clin. Res. 2021:17–23. [Google Scholar]
- 47.Sadeghpour H., Khalvati B., Entezar-Almahdi E., Savadi N., Alhashemi S.H., Raoufi M., et al. Double domain polyethylenimine-based nanoparticles for integrin receptor mediated delivery of plasmid DNA. Sci. Rep. 2018;8(1):1–12. doi: 10.1038/s41598-018-25277-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jensen S, Twitty C, Paustian C, Laws M, McDonald G, Wegmann K, et al., editors. Preliminary evaluation of a novel coronavirus vaccine (CORVax) using electroporation of plasmid DNA encoding a stabilized prefusion SARS-CoV-2 spike protein alone or with transfection of plasmid IL-12. Journal for Immunotherapy of Cancer; 2020: BMJ PUBLISHING GROUP BRITISH MED ASSOC HOUSE, TAVISTOCK SQUARE, LONDON WC1H …..
- 49.Walsh E.E., Frenck R.W., Falsey A.R., Kitchin N., Absalon J., Gurtman A., Lockhart S., Neuzil K., Mulligan M.J., Bailey R., Swanson K.A., Li P., Koury K., Kalina W., Cooper D., Fontes-Garfias C., Shi P.-Y., Türeci Ö., Tompkins K.R., Lyke K.E., Raabe V., Dormitzer P.R., Jansen K.U., Şahin U., Gruber W.C. Safety and immunogenicity of two RNA-based Covid-19 vaccine candidates. N. Engl. J. Med. 2020;383(25):2439–2450. doi: 10.1056/NEJMoa2027906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sahin U., Muik A., Derhovanessian E., Vogler I., Kranz L.M., Vormehr M., Baum A., Pascal K., Quandt J., Maurus D., Brachtendorf S., Lörks V., Sikorski J., Hilker R., Becker D., Eller A.-K., Grützner J., Boesler C., Rosenbaum C., Kühnle M.-C., Luxemburger U., Kemmer-Brück A., Langer D., Bexon M., Bolte S., Karikó K., Palanche T., Fischer B., Schultz A., Shi P.-Y., Fontes-Garfias C., Perez J.L., Swanson K.A., Loschko J., Scully I.L., Cutler M., Kalina W., Kyratsous C.A., Cooper D., Dormitzer P.R., Jansen K.U., Türeci Ö. COVID-19 vaccine BNT162b1 elicits human antibody and TH 1 T cell responses. Nature. 2020;586(7830):594–599. doi: 10.1038/s41586-020-2814-7. [DOI] [PubMed] [Google Scholar]
- 51.Li J, Hui A, Zhang X, Yang Y, Tang R, Ye H, et al. Safety and immunogenicity of the SARS-CoV-2 BNT162b1 mRNA vaccine in younger and older Chinese adults: a randomized, placebo-controlled, double-blind phase 1 study. 2021:1-9. [DOI] [PubMed]
- 52.Kremsner P, Mann P, Bosch J, Fendel R, Gabor J, Kreidenweiss A, et al. Phase 1 Assessment of the safety and immunogenicity of an mRNA-lipid nanoparticle vaccine candidate against SARS-CoV-2 in human volunteers. medRxiv 2020.11. 09.20228551 [Preprint]. 9 November 2020. Preprint]. 2020;9.
- 53.Dolgin E. CureVac COVID vaccine let-down spotlights mRNA design challenges. Nature. 2021;594(7864):483. doi: 10.1038/d41586-021-01661-0. [DOI] [PubMed] [Google Scholar]
- 54.Chagla Z. The BNT162b2 (BioNTech/Pfizer) vaccine had 95% efficacy against COVID-19≥ 7 days after the 2nd dose. Ann. Intern. Med. 2021;174(2):JC15. doi: 10.7326/ACPJ202102160-015. [DOI] [PubMed] [Google Scholar]
- 55.Mahase E. Covid-19: Pfizer vaccine efficacy was 52% after first dose and 95% after second dose, paper shows. British Medical Journal Publishing Group; 2020. [DOI] [PubMed]
- 56.Covid C, Team R Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine—United States, December 14–23, 2020. Morb. Mortal. Wkly Rep. 2021;70(2):46. doi: 10.15585/mmwr.mm7002e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shimabukuro T. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine—United States, December 14–23, 2020. Am. J. Transplant. 2021;21(3):1332–1337. doi: 10.1111/ajt.16516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wodi A.P., Ault K., Hunter P., McNally V., Szilagyi P.G., Bernstein H. Advisory Committee on Immunization Practices Recommended Immunization Schedule for Children and Adolescents Aged 18 Years or Younger—United States, 2021. Morb. Mortal. Wkly Rep. 2021;70(6):189–192. doi: 10.15585/mmwr.mm7006a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wallace M. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine in Adolescents Aged 12–15 Years—United States, May 2021. MMWR Morbidity and Mortality Weekly Report. 2021;70. [DOI] [PMC free article] [PubMed]
- 60.Abdool Karim S.S., de Oliveira T. New SARS-CoV-2 variants—clinical, public health, and vaccine implications. N. Engl. J. Med. 2021;384(19):1866–1868. doi: 10.1056/NEJMc2100362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lopez Bernal J, Andrews N, Gower C, Gallagher E, Simmons R, Thelwall S, et al. Effectiveness of Covid-19 vaccines against the B. 1.617. 2 (Delta) variant. New England Journal of Medicine. 2021. [DOI] [PMC free article] [PubMed]
- 62.Pouwels K.B., Pritchard E., Matthews P., Stoesser N.B., Eyre D.W., Vihta K.-D., et al. Impact of Delta on viral burden and vaccine effectiveness against new SARS-CoV-2 infections in the UK. medRxiv. 2021 doi: 10.1038/s41591-021-01548-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Oliver S.E. MMWR Morbidity and mortality weekly report. December 2020. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Moderna COVID-19 Vaccine—United States; p. 2020;69.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Livingston E.H. Necessity of 2 doses of the Pfizer and Moderna COVID-19 vaccines. JAMA. 2021;325(9):898. doi: 10.1001/jama.2021.1375. [DOI] [PubMed] [Google Scholar]
- 65.Chagla Z. In high-risk adults, the Moderna vaccine had 94% efficacy against COVID-19≥ 14 d after the 2nd dose. Ann. Intern. Med. 2021;174(3):JC28. doi: 10.7326/ACPJ202103160-028. [DOI] [PubMed] [Google Scholar]
- 66.Covid C, Team R Allergic reactions including anaphylaxis after receipt of the first dose of Moderna COVID-19 Vaccine—United States, December 21, 2020–January 10, 2021. Morb. Mortal. Wkly Rep. 2021;70(4):125. doi: 10.15585/mmwr.mm7004e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Özütemiz C., Krystosek L.A., Church A.L., Chauhan A., Ellermann J.M., Domingo-Musibay E., et al. Lymphadenopathy in COVID-19 vaccine recipients: diagnostic dilemma in oncology patients. Radiology. 2021;210275 doi: 10.1148/radiol.2021210275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Johnston M.S., Galan A., Watsky K.L., Little A.J. Delayed Localized Hypersensitivity Reactions to the Moderna COVID-19 Vaccine: A Case Series. JAMA Dermatol. 2021;157(6):716. doi: 10.1001/jamadermatol.2021.1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Shimabukuro T.T., Kim S.Y., Myers T.R., Moro P.L., Oduyebo T., Panagiotakopoulos L., et al. Preliminary findings of mrna covid-19 vaccine safety in pregnant persons. N. Engl. J. Med. 2021 doi: 10.1056/NEJMoa2104983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mendonça S.A., Lorincz R., Boucher P., Curiel D.T. Adenoviral vector vaccine platforms in the SARS-CoV-2 pandemic. npj Vaccines. 2021;6(1):1–14. doi: 10.1038/s41541-021-00356-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kaur S.P., Gupta V. COVID-19 Vaccine: A comprehensive status report. Virus Res. 2020;288:198114. doi: 10.1016/j.virusres.2020.198114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kim J.H., Marks F., Clemens J.D. Looking beyond COVID-19 vaccine phase 3 trials. Nat. Med. 2021;27(2):205–211. doi: 10.1038/s41591-021-01230-y. [DOI] [PubMed] [Google Scholar]
- 73.Brüssow H. COVID-19: vaccination problems. Environ. Microbiol. 2021 doi: 10.1111/1462-2920.15549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chagla Z. In adults, the Oxford/AstraZeneca vaccine had 70% efficacy against COVID-19> 14 d after the 2nd dose. Ann. Intern. Med. 2021;174(3):JC29. doi: 10.7326/ACPJ202103160-029. [DOI] [PubMed] [Google Scholar]
- 75.Voysey M., Clemens S.A.C., Madhi S.A., Weckx L.Y., Folegatti P.M., Aley P.K., Angus B., Baillie V.L., Barnabas S.L., Bhorat Q.E., Bibi S., Briner C., Cicconi P., Collins A.M., Colin-Jones R., Cutland C.L., Darton T.C., Dheda K., Duncan C.J.A., Emary K.R.W., Ewer K.J., Fairlie L., Faust S.N., Feng S., Ferreira D.M., Finn A., Goodman A.L., Green C.M., Green C.A., Heath P.T., Hill C., Hill H., Hirsch I., Hodgson S.H.C., Izu A., Jackson S., Jenkin D., Joe C.C.D., Kerridge S., Koen A., Kwatra G., Lazarus R., Lawrie A.M., Lelliott A., Libri V., Lillie P.J., Mallory R., Mendes A.V.A., Milan E.P., Minassian A.M., McGregor A., Morrison H., Mujadidi Y.F., Nana A., O’Reilly P.J., Padayachee S.D., Pittella A., Plested E., Pollock K.M., Ramasamy M.N., Rhead S., Schwarzbold A.V., Singh N., Smith A., Song R., Snape M.D., Sprinz E., Sutherland R.K., Tarrant R., Thomson E.C., Török M.E., Toshner M., Turner D.P.J., Vekemans J., Villafana T.L., Watson M.E.E., Williams C.J., Douglas A.D., Hill A.V.S., Lambe T., Gilbert S.C., Pollard A.J., Aban M., Abayomi F., Abeyskera K., Aboagye J., Adam M., Adams K., Adamson J., Adelaja Y.A., Adewetan G., Adlou S., Ahmed K., Akhalwaya Y., Akhalwaya S., Alcock A., Ali A., Allen E.R., Allen L., Almeida T.C.D.S.C., Alves M.P.S., Amorim F., Andritsou F., Anslow R., Appleby M., Arbe-Barnes E.H., Ariaans M.P., Arns B., Arruda L., Azi P., Azi L., Babbage G., Bailey C., Baker K.F., Baker M., Baker N., Baker P., Baldwin L., Baleanu I., Bandeira D., Bara A., Barbosa M.A.S., Barker D., Barlow G.D., Barnes E., Barr A.S., Barrett J.R., Barrett J., Bates L., Batten A., Beadon K., Beales E., Beckley R., Belij-Rammerstorfer S., Bell J., Bellamy D., Bellei N., Belton S., Berg A., Bermejo L., Berrie E., Berry L., Berzenyi D., Beveridge A., Bewley K.R., Bexhell H., Bhikha S., Bhorat A.E., Bhorat Z.E., Bijker E., Birch G., Birch S., Bird A., Bird O., Bisnauthsing K., Bittaye M., Blackstone K., Blackwell L., Bletchly H., Blundell C.L., Blundell S.R., Bodalia P., Boettger B.C., Bolam E., Boland E., Bormans D., Borthwick N., Bowring F., Boyd A., Bradley P., Brenner T., Brown P., Brown C., Brown-O'Sullivan C., Bruce S., Brunt E., Buchan R., Budd W., Bulbulia Y.A., Bull M., Burbage J., Burhan H., Burn A., Buttigieg K.R., Byard N., Cabera Puig I., Calderon G., Calvert A., Camara S., Cao M., Cappuccini F., Cardoso J.R., Carr M., Carroll M.W., Carson-Stevens A., Carvalho Y.d.M., Carvalho J.A.M., Casey H.R., Cashen P., Castro T., Castro L.C., Cathie K., Cavey A., Cerbino-Neto J., Chadwick J., Chapman D., Charlton S., Chelysheva I., Chester O., Chita S., Cho J.-S., Cifuentes L., Clark E., Clark M., Clarke A., Clutterbuck E.A., Collins S.L.K., Conlon C.P., Connarty S., Coombes N., Cooper C., Cooper R., Cornelissen L., Corrah T., Cosgrove C., Cox T., Crocker W.E.M., Crosbie S., Cullen L., Cullen D., Cunha D.R.M.F., Cunningham C., Cuthbertson F.C., Da Guarda S.N.F., da Silva L.P., Damratoski B.E., Danos Z., Dantas M.T.D.C., Darroch P., Datoo M.S., Datta C., Davids M., Davies S.L., Davies H., Davis E., Davis J., Davis J., De Nobrega M.M.D., De Oliveira Kalid L.M., Dearlove D., Demissie T., Desai A., Di Marco S., Di Maso C., Dinelli M.I.S., Dinesh T., Docksey C., Dold C., Dong T., Donnellan F.R., Dos Santos T., dos Santos T.G., Dos Santos E.P., Douglas N., Downing C., Drake J., Drake-Brockman R., Driver K., Drury R., Dunachie S.J., Durham B.S., Dutra L., Easom N.J.W., van Eck S., Edwards M., Edwards N.J., El Muhanna O.M., Elias S.C., Elmore M., English M., Esmail A., Essack Y.M., Farmer E., Farooq M., Farrar M., Farrugia L., Faulkner B., Fedosyuk S., Felle S., Feng S., Ferreira Da Silva C., Field S., Fisher R., Flaxman A., Fletcher J., Fofie H., Fok H., Ford K.J., Fowler J., Fraiman P.H.A., Francis E., Franco M.M., Frater J., Freire M.S.M., Fry S.H., Fudge S., Furze J., Fuskova M., Galian-Rubio P., Galiza E., Garlant H., Gavrila M., Geddes A., Gibbons K.A., Gilbride C., Gill H., Glynn S., Godwin K., Gokani K., Goldoni U.C., Goncalves M., Gonzalez I.G.S., Goodwin J., Goondiwala A., Gordon-Quayle K., Gorini G., Grab J., Gracie L., Greenland M., Greenwood N., Greffrath J., Groenewald M.M., Grossi L., Gupta G., Hackett M., Hallis B., Hamaluba M., Hamilton E., Hamlyn J., Hammersley D., Hanrath A.T., Hanumunthadu B., Harris S.A., Harris C., Harris T., Harrison T.D., Harrison D., Hart T.C., Hartnell B., Hassan S., Haughney J., Hawkins S., Hay J., Head I., Henry J., Hermosin Herrera M., Hettle D.B., Hill J., Hodges G., Horne E., Hou M.M., Houlihan C., Howe E., Howell N., Humphreys J., Humphries H.E., Hurley K., Huson C., Hyder-Wright A., Hyams C., Ikram S., Ishwarbhai A., Ivan M., Iveson P., Iyer V., Jackson F., De Jager J., Jaumdally S., Jeffers H., Jesudason N., Jones B., Jones K., Jones E., Jones C., Jorge M.R., Jose A., Joshi A., Júnior E.A.M.S., Kadziola J., Kailath R., Kana F., Karampatsas K., Kasanyinga M., Keen J., Kelly E.J., Kelly D.M., Kelly D., Kelly S., Kerr D., Kfouri R.d.Á., Khan L., Khozoee B., Kidd S., Killen A., Kinch J., Kinch P., King L.D.W., King T.B., Kingham L., Klenerman P., Knapper F., Knight J.C., Knott D., Koleva S., Lang M., Lang G., Larkworthy C.W., Larwood J.P.J., Law R., Lazarus E.M., Leach A., Lees E.A., Lemm N.-M., Lessa A., Leung S., Li Y., Lias A.M., Liatsikos K., Linder A., Lipworth S., Liu S., Liu X., Lloyd A., Lloyd S., Loew L., Lopez Ramon R., Lora L., Lowthorpe V., Luz K., MacDonald J.C., MacGregor G., Madhavan M., Mainwaring D.O., Makambwa E., Makinson R., Malahleha M., Malamatsho R., Mallett G., Mansatta K., Maoko T., Mapetla K., Marchevsky N.G., Marinou S., Marlow E., Marques G.N., Marriott P., Marshall R.P., Marshall J.L., Martins F.J., Masenya M., Masilela M., Masters S.K., Mathew M., Matlebjane H., Matshidiso K., Mazur O., Mazzella A., McCaughan H., McEwan J., McGlashan J., McInroy L., McIntyre Z., McLenaghan D., McRobert N., McSwiggan S., Megson C., Mehdipour S., Meijs W., Mendonça R.N.Á., Mentzer A.J., Mirtorabi N., Mitton C., Mnyakeni S., Moghaddas F., Molapo K., Moloi M., Moore M., Moraes-Pinto M.I., Moran M., Morey E., Morgans R., Morris S., Morris S., Morris H.C., Morselli F., Morshead G., Morter R., Mottal L., Moultrie A., Moya N., Mpelembue M., Msomi S., Mugodi Y., Mukhopadhyay E., Muller J., Munro A., Munro C., Murphy S., Mweu P., Myasaki C.H., Naik G., Naker K., Nastouli E., Nazir A., Ndlovu B., Neffa F., Njenga C., Noal H., Noé A., Novaes G., Nugent F.L., Nunes G., O'Brien K., O'Connor D., Odam M., Oelofse S., Oguti B., Olchawski V., Oldfield N.J., Oliveira M.G., Oliveira C., Oosthuizen A., O'Reilly P., Osborne P., Owen D.R.J., Owen L., Owens D., Owino N., Pacurar M., Paiva B.V.B., Palhares E.M.F., Palmer S., Parkinson S., Parracho H.M.R.T., Parsons K., Patel D., Patel B., Patel F., Patel K., Patrick-Smith M., Payne R.O., Peng Y., Penn E.J., Pennington A., Peralta Alvarez M.P., Perring J., Perry N., Perumal R., Petkar S., Philip T., Phillips D.J., Phillips J., Phohu M.K., Pickup L., Pieterse S., Piper J.o., Pipini D., Plank M., Du Plessis J., Pollard S., Pooley J., Pooran A., Poulton I., Powers C., Presa F.B., Price D.A., Price V., Primeira M., Proud P.C., Provstgaard-Morys S., Pueschel S., Pulido D., Quaid S., Rabara R., Radford A., Radia K., Rajapaska D., Rajeswaran T., Ramos A.S.F., Ramos Lopez F., Rampling T., Rand J., Ratcliffe H., Rawlinson T., Rea D., Rees B., Reiné J., Resuello-Dauti M., Reyes Pabon E., Ribiero C.M., Ricamara M., Richter A., Ritchie N., Ritchie A.J., Robbins A.J., Roberts H., Robinson R.E., Robinson H., Rocchetti T.T., Rocha B.P., Roche S., Rollier C., Rose L., Ross Russell A.L., Rossouw L., Royal S., Rudiansyah I., Ruiz S., Saich S., Sala C., Sale J., Salman A.M., Salvador N., Salvador S., Sampaio M., Samson A.D., Sanchez-Gonzalez A., Sanders H., Sanders K., Santos E., Santos Guerra M.F.S., Satti I., Saunders J.E., Saunders C., Sayed A., Schim van der Loeff I., Schmid A.B., Schofield E., Screaton G., Seddiqi S., Segireddy R.R., Senger R., Serrano S., Shah R., Shaik I., Sharpe H.E., Sharrocks K., Shaw R., Shea A., Shepherd A., Shepherd J.G., Shiham F., Sidhom E., Silk S.E., da Silva Moraes A.C., Silva-Junior G., Silva-Reyes L., Silveira A.D., Silveira M.B.V., Sinha J., Skelly D.T., Smith D.C., Smith N., Smith H.E., Smith D.J., Smith C.C., Soares A., Soares T., Solórzano C., Sorio G.L., Sorley K., Sosa-Rodriguez T., Souza C.M.C.D.L., Souza B.S.D.F., Souza A.R., Spencer A.J., Spina F., Spoors L., Stafford L., Stamford I., Starinskij I., Stein R., Steven J., Stockdale L., Stockwell L.V., Strickland L.H., Stuart A.C., Sturdy A., Sutton N., Szigeti A., Tahiri-Alaoui A., Tanner R., Taoushanis C., Tarr A.W., Taylor K., Taylor U., Taylor I.J., Taylor J., te Water Naude R., Themistocleous Y., Themistocleous A., Thomas M., Thomas K., Thomas T.M., Thombrayil A., Thompson F., Thompson A., Thompson K., Thompson A., Thomson J., Thornton-Jones V., Tighe P.J., Tinoco L.A., Tiongson G., Tladinyane B., Tomasicchio M., Tomic A., Tonks S., Towner J., Tran N., Tree J., Trillana G., Trinham C., Trivett R., Truby A., Tsheko B.L., Turabi A., Turner R., Turner C., Ulaszewska M., Underwood B.R., Varughese R., Verbart D., Verheul M., Vichos I., Vieira T., Waddington C.S., Walker L., Wallis E., Wand M., Warbick D., Wardell T., Warimwe G., Warren S.C., Watkins B., Watson E., Webb S., Webb-Bridges A., Webster A., Welch J., Wells J., West A., White C., White R., Williams P., Williams R.L., Winslow R., Woodyer M., Worth A.T., Wright D., Wroblewska M., Yao A., Zimmer R., Zizi D., Zuidewind P. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. The Lancet. 2021;397(10269):99–111. doi: 10.1016/S0140-6736(20)32661-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Voysey M., Clemens S.A.C., Madhi S.A., Weckx L.Y., Folegatti P.M., Aley P.K., et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials. The Lancet. 2021;397(10277):881–891. doi: 10.1016/S0140-6736(21)00432-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Dal-Ré R. Will AstraZeneca be able to provide clinical trial data on its COVID-19 vaccine efficacy in older adults? Br. J. Clin. Pharmacol. 2021 doi: 10.1111/bcp.14828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Khan Sharun R.S., Dhama K. Oxford-AstraZeneca COVID-19 vaccine (AZD1222) is ideal for resource-constrained low-and middle-income countries. Ann. Med. Surg. 2021 doi: 10.1016/j.amsu.2021.102264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Østergaard S.D., Schmidt M., Horváth-Puhó E., Thomsen R.W., Sørensen H.T. England); London: 2021. Thromboembolism and the Oxford–AstraZeneca COVID-19 vaccine: side-effect or coincidence? Lancet. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Schultz N.H., Sørvoll I.H., Michelsen A.E., Munthe L.A., Lund-Johansen F., Ahlen M.T., et al. Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. 2021 doi: 10.1056/NEJMoa2104882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Walter U., Fuchs M., Grossmann A., Walter M., Thiele T., Storch A., et al. Adenovirus-Vectored COVID-19 Vaccine-Induced Immune Thrombosis of Carotid Artery: A Case Report. Neurology. 2021 doi: 10.1212/WNL.0000000000012576. [DOI] [PubMed] [Google Scholar]
- 82.COVID-19 vaccine safety update, VAXZEVRIA (AstraZeneca AB) [September 2021]. Available from: https://www.ema.europa.eu/en/medicines/human/EPAR/vaxzevria-previously-covid-19-vaccine-astrazeneca#safety-updates-section.
- 83.Livingston E.H., Malani P.N., Creech C.B. The Johnson & Johnson Vaccine for COVID-19. JAMA. 2021 doi: 10.1001/jama.2021.2927. [DOI] [PubMed] [Google Scholar]
- 84.Mahase E. Covid-19: US suspends Johnson and Johnson vaccine rollout over blood clots. British Medical Journal Publishing Group; 2021. [DOI] [PubMed]
- 85.Alter G., Yu J., Liu J., Chandrashekar A., Borducchi E.N., Tostanoski L.H., et al. Immunogenicity of Ad26. COV2. S vaccine against SARS-CoV-2 variants in humans. Nature. 2021:1–9. doi: 10.1038/s41586-021-03681-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Balakrishnan V.S. The arrival of Sputnik V. Lancet Infect. Dis. 2020;20(10):1128. doi: 10.1016/S1473-3099(20)30709-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Logunov D.Y., Dolzhikova I.V., Shcheblyakov D.V., Tukhvatulin A.I., Zubkova O.V., Dzharullaeva A.S., et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. The Lancet. 2021;397(10275):671–681. doi: 10.1016/S0140-6736(21)00234-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mahase E. Covid-19: Russia approves vaccine without large scale testing or published results. BMJ: British Med. J. (Online) 2020;370 doi: 10.1136/bmj.m3205. [DOI] [PubMed] [Google Scholar]
- 89.Logunov D.Y., Dolzhikova I.V., Zubkova O.V., Tukhvatullin A.I., Shcheblyakov D.V., Dzharullaeva A.S., et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: two open, non-randomised phase 1/2 studies from Russia. The Lancet. 2020;396(10255):887–897. doi: 10.1016/S0140-6736(20)31866-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jones I., Roy P. Sputnik V COVID-19 vaccine candidate appears safe and effective. The Lancet. 2021;397(10275):642–643. doi: 10.1016/S0140-6736(21)00191-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Le T.T., Andreadakis Z., Kumar A., Román R.G., Tollefsen S., Saville M., et al. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020;19(5):305–306. doi: 10.1038/d41573-020-00073-5. [DOI] [PubMed] [Google Scholar]
- 92.Zhu F.-C., Li Y.-H., Guan X.-H., Hou L.-H., Wang W.-J., Li J.-X., et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. The Lancet. 2020;395(10240):1845–1854. doi: 10.1016/S0140-6736(20)31208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Yamey G., Schäferhoff M., Hatchett R., Pate M., Zhao F., McDade K.K. Ensuring global access to COVID-19 vaccines. The Lancet. 2020;395(10234):1405–1406. doi: 10.1016/S0140-6736(20)30763-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zhu F.-C., Guan X.-H., Li Y.-H., Huang J.-Y., Jiang T., Hou L.-H., et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomised, double-blind, placebo-controlled, phase 2 trial. The Lancet. 2020;396(10249):479–488. doi: 10.1016/S0140-6736(20)31605-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wu S., Zhong G., Zhang J., Shuai L., Zhang Z., Wen Z., et al. A single dose of an adenovirus-vectored vaccine provides protection against SARS-CoV-2 challenge. Nat. Commun. 2020;11(1):1–7. doi: 10.1038/s41467-020-17972-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Forchette L., Sebastian W., Liu T. A comprehensive review of COVID-19 virology, vaccines, variants, and therapeutics. Current. Med. Sci. 2021:1–15. doi: 10.1007/s11596-021-2395-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Azmi F., Ahmad Fuaad A.A.H., Skwarczynski M., Toth I. Recent progress in adjuvant discovery for peptide-based subunit vaccines. Human Vaccines Immunother. 2014;10(3):778–796. doi: 10.4161/hv.27332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ou X., Liu Y., Lei X., Li P., Mi D., Ren L., et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020;11(1):1–12. doi: 10.1038/s41467-020-15562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bengtsson K.L., Song H., Stertman L., Liu Y., Flyer D.C., Massare M.J., et al. Matrix-M adjuvant enhances antibody, cellular and protective immune responses of a Zaire Ebola/Makona virus glycoprotein (GP) nanoparticle vaccine in mice. Vaccine. 2016;34(16):1927–1935. doi: 10.1016/j.vaccine.2016.02.033. [DOI] [PubMed] [Google Scholar]
- 100.Tai W., He L., Zhang X., Pu J., Voronin D., Jiang S., et al. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell. Mol. Immunol. 2020;17(6):613–620. doi: 10.1038/s41423-020-0400-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Du L., He Y., Zhou Y., Liu S., Zheng B.-J., Jiang S. The spike protein of SARS-CoV—a target for vaccine and therapeutic development. Nat. Rev. Microbiol. 2009;7(3):226–236. doi: 10.1038/nrmicro2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mahase E. Covid-19: Novavax vaccine efficacy is 86% against UK variant and 60% against South African variant. British Medical Journal Publishing Group; 2021. [DOI] [PubMed]
- 103.Shinde V., Bhikha S., Hossain Z., Archary M., Bhorat Q., Fairlie L., et al. Preliminary Efficacy of the NVX-CoV2373 Covid-19 Vaccine Against the B. 1.351 Variant. medRxiv. 2021 doi: 10.1056/NEJMoa2103055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.[June 2021]. Available from: https://ir.novavax.com/news-releases/news-release-details/novavax-covid-19-vaccine.
- 105.Guebre-Xabier M., Patel N., Tian J.-H., Zhou B., Maciejewski S., Lam K., et al. NVX-CoV2373 vaccine protects cynomolgus macaque upper and lower airways against SARS-CoV-2 challenge. Vaccine. 2020;38(50):7892–7896. doi: 10.1016/j.vaccine.2020.10.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Keech C., Albert G., Cho I., Robertson A., Reed P., Neal S., et al. Phase 1–2 trial of a SARS-CoV-2 recombinant spike protein nanoparticle vaccine. N. Engl. J. Med. 2020;383(24):2320–2332. doi: 10.1056/NEJMoa2026920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Formica N., Mallory R., Albert G., Robinson M., Plested J., Cho I., et al. Evaluation of a SARS-CoV-2 Vaccine NVX-CoV2373 in Younger and Older Adults. medRxiv. 2021 doi: 10.1371/journal.pmed.1003769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ryzhikov A.B., Ryzhikov E.A., Bogryantseva M.P., Danilenko E.D., Imatdinov I.R., Nechaeva E.A., et al. Immunogenicity and protectivity of the peptide candidate vaccine against SARS-CoV-2. Ann. Russian Acad. Med. Sci. 2021;76(1):5–19. [Google Scholar]
- 109.Ryzhikov A., Ryzhikov E., Bogryantseva M., Usova S., Danilenko E., Nechaeva E., et al. A single blind, placebo-controlled randomized study of the safety, reactogenicity and immunogenicity of the “EpiVacCorona” Vaccine for the prevention of COVID-19, in volunteers aged 18–60 years (phase I-II) Russian J. Infec. Immunity. 2021;11(2):283–296. [Google Scholar]
- 110.Premkumar L., Segovia-Chumbez B., Jadi R., Martinez D.R., Raut R., Markmann A., et al. The receptor binding domain of the viral spike protein is an immunodominant and highly specific target of antibodies in SARS-CoV-2 patients. Sci. Immunol. 2020;5(48) doi: 10.1126/sciimmunol.abc8413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wang N., Shang J., Jiang S., Du L. Subunit vaccines against emerging pathogenic human coronaviruses. Front. Microbiol. 2020;11:298. doi: 10.3389/fmicb.2020.00298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang Q, Zhang Y, Wu L, Niu S, Song C, Zhang Z, et al. Structural and functional basis of SARS-CoV-2 entry by using human ACE2. Cell. 2020;181(4):894-904. e9. [DOI] [PMC free article] [PubMed]
- 113.A Phase III Clinical Trial to Determine the Safety and Efficacy of ZF2001 for Prevention of COVID-19 [September 2021]. Available from: https://clinicaltrials.gov/ct2/show/NCT04646590.
- 114.Hotez P.J., Nuzhath T., Callaghan T., Colwell B. COVID-19 Vaccine Decisions: Considering the Choices and Opportunities. Microbes Infect. 2021;104811 doi: 10.1016/j.micinf.2021.104811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.He Q., Mao Q., Zhang J., Bian L., Gao F., Wang J., et al. COVID-19 Vaccines: Current Understanding on Immunogenicity, Safety, and Further Considerations. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.669339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wang H, Zhang Y, Huang B, Deng W, Quan Y, Wang W, et al. Development of an inactivated vaccine candidate, BBIBP-CorV, with potent protection against SARS-CoV-2. Cell. 2020;182(3):713-21. e9. [DOI] [PMC free article] [PubMed]
- 117.Xia S., Zhang Y., Wang Y., Wang H., Yang Y., Gao G.F., et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: a randomised, double-blind, placebo-controlled, phase 1/2 trial. Lancet. Infect. Dis. 2021;21(1):39–51. doi: 10.1016/S1473-3099(20)30831-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang G.-L., Wang Z.-Y., Duan L.-J., Meng Q.-C., Jiang M.-D., Cao J., et al. Susceptibility of Circulating SARS-CoV-2 Variants to Neutralization. N. Engl. J. Med. 2021 doi: 10.1056/NEJMc2103022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wu Z., Hu Y., Xu M., Chen Z., Yang W., Jiang Z., et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet. Infect. Dis. 2021 doi: 10.1016/S1473-3099(20)30987-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Zhang Y., Zeng G., Pan H., Li C., Hu Y., Chu K., et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet. Infect. Dis. 2021;21(2):181–192. doi: 10.1016/S1473-3099(20)30843-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Palacios R., Patiño E.G., de Oliveira P.R., Conde M.T.R.P., Batista A.P., Zeng G., et al. Double-Blind, Randomized, Placebo-Controlled Phase III Clinical Trial to Evaluate the Efficacy and Safety of treating Healthcare Professionals with the Adsorbed COVID-19 (Inactivated) Vaccine Manufactured by Sinovac–PROFISCOV: A structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21(1):1–3. doi: 10.1186/s13063-020-04775-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cordero D.A., Jr Respecting freedom in the absence of compassion: realistic behavior by public officials for the COVID-19 vaccination program. J. Public Health (Oxford, England). 2021 doi: 10.1093/pubmed/fdab019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Bhuyan A. India begins COVID-19 vaccination amid trial allegations. The Lancet. 2021;397(10271):264. doi: 10.1016/S0140-6736(21)00145-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Bagcchi S. The world's largest COVID-19 vaccination campaign. Lancet. Infect. Dis. 2021;21(3):323. doi: 10.1016/S1473-3099(21)00081-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Ella R., Vadrevu K.M., Jogdand H., Prasad S., Reddy S., Sarangi V., et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: a double-blind, randomised, phase 1 trial. Lancet. Infect. Dis. 2021 doi: 10.1016/S1473-3099(20)30942-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.R Srivastava R, Ish P, COVID S. The initial experience of COVID-19 vaccination from a tertiary care centre of India. Monaldi Archives for Chest Disease. 2021. [DOI] [PubMed]
- 127.Chakraborty C., Agoramoorthy G. India’s cost-effective COVID-19 vaccine development initiatives. Vaccine. 2020;38(50):7883. doi: 10.1016/j.vaccine.2020.10.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ella R., Reddy S., Jogdand H., Sarangi V., Ganneru B., Prasad S., et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: interim results from a double-blind, randomised, multicentre, phase 2 trial, and 3-month follow-up of a double-blind, randomised phase 1 trial. Lancet. Infect. Dis. 2021 doi: 10.1016/S1473-3099(21)00070-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Sapkal G.N., Yadav P.D., Ella R., Deshpande G.R., Sahay R.R., Gupta N., et al. Inactivated COVID-19 vaccine BBV152/COVAXIN effectively neutralizes recently emerged B 1.1. 7 variant of SARS-CoV-2. J. Travel Med. 2021 doi: 10.1093/jtm/taab051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wei L., Shang Y., Liu X., Li X., Chen G., Liang S., et al. Antibody Responses and the Effects of Clinical Drugs in COVID-19 Patients. Front. Immunol. 2021;12:2158. doi: 10.3389/fimmu.2021.580989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Jahanshahlu L., Rezaei N. Monoclonal antibody as a potential anti-COVID-19. Biomed. Pharmacother. 2020;129:110337. doi: 10.1016/j.biopha.2020.110337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chen J., Gao K., Wang R., Nguyen D.D., Wei G.-W. Review of COVID-19 antibody therapies. Annu. Rev. Biophys. 2021;50:1–30. doi: 10.1146/annurev-biophys-062920-063711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Siracusano G., Pastori C., Lopalco L. Humoral immune responses in COVID-19 patients: a window on the state of the art. Front. Immunol. 2020;11:1049. doi: 10.3389/fimmu.2020.01049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Fleming A.B., Raabe V. Current studies of convalescent plasma therapy for COVID-19 may underestimate risk of antibody-dependent enhancement. J. Clin. Virol. 2020;127:104388. doi: 10.1016/j.jcv.2020.104388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Chvatal-Medina M., Mendez-Cortina Y., Patiño P.J., Velilla P.A., Rugeles M.T. Antibody responses in COVID-19: a review. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.633184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.McCallum M, Bassi J, De Marco A, Chen A, Walls AC, Di Iulio J, et al. SARS-CoV-2 immune evasion by variant B. 1.427/B. 1.429. bioRxiv. 2021. [DOI] [PMC free article] [PubMed]
- 137.Del Rio C., Malani P.N., Omer S.B. Confronting the Delta Variant of SARS-CoV-2, Summer 2021. JAMA. 2021 doi: 10.1001/jama.2021.14811. [DOI] [PubMed] [Google Scholar]
- 138.Danesh-Bahreini M.A., Shokri J., Samiei A., Kamali-Sarvestani E., Barzegar-Jalali M., Mohammadi-Samani S. Nanovaccine for leishmaniasis: preparation of chitosan nanoparticles containing Leishmania superoxide dismutase and evaluation of its immunogenicity in BALB/c mice. Int J Nanomedicine. 2011;6:835–842. doi: 10.2147/IJN.S16805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Sabahi Z., Samani S.M., Dehshahri A. Conjugation of poly (amidoamine) dendrimers with various acrylates for improved delivery of plasmid encoding interleukin-12 gene. J Biomater Appl. 2015;29(7):941–953. doi: 10.1177/0885328214551010. [DOI] [PubMed] [Google Scholar]
- 140.Ghasemiyeh P., Mohammadi-Samani S. The necessity of early anti-inflammatory therapy initiation in cases with mild-to-moderate COVID-19: A personal experience from an attending pharmacist and his resident. Acta Biomed. 2021;92(3) doi: 10.23750/abm.v92i3.11683. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.