Abstract
Coronavirus disease of 2019 (COVID‐19) caused by severe acute respiratory syndrome virus type 2 (SARS‐CoV‐2) is an emerging severe acute respiratory disease affecting global human health. In this study, a large‐scale serological survey of antibodies against SARS‐CoV‐2 in dogs and cats was conducted during the first and second waves of COVID‐19 outbreaks in Thailand, from April to December 2020. A total of 3215 serum samples were collected from dogs (n = 2102) and cats (n = 1113) living in Bangkok and in the vicinities. Serum samples were tested for SARS‐CoV‐2 antibodies by using an indirect multispecies enzyme‐linked immunosorbent assay (ELISA). Positive and suspected samples were additionally tested for neutralizing antibodies by the surrogate virus neutralization test (sVNT). The indirect ELISA results showed that 1.66% (35 out of 2103) of dogs and 0.36% (four out of 1112) of cats were positive for SARS‐CoV‐2 antibodies. The sVNT results showed that all ELISA‐positive and suspected samples were negative for neutralizing antibodies. Positive serum samples (35 dogs and four cats) were obtained from clinically healthy animals and animals with mild respiratory signs aged <1–13 years living in Bangkok and Samutprakarn Provinces. In summary, a serological survey revealed evidence of anti‐N‐IgG antibodies suggesting SARS‐CoV‐2 exposure in both dogs and cats during the first and second COVID‐19 outbreaks in Thailand.
Keywords: cats, dogs, SARS‐CoV‐2, survey, Thailand
1. INTRODUCTION
Coronavirus disease of 2019 (COVID‐19) caused by severe acute respiratory syndrome virus type 2 (SARS‐CoV‐2) is an emerging disease that has caused outbreaks in human population worldwide. As of May 2021, more than 154 million confirmed cases have been reported with over 3.2 million deaths (WHO, 2021). A few reports of SARS‐CoV‐2 natural infections have been documented in non‐human mammals, including dogs, cats, tigers, lions, gorillas and minks (Abdel‐Moneim & Abdelwhab, 2020; Leroy et al., 2020; Newman et al., 2020; Ruiz‐Arrondo et al., 2021; Sailleau et al., 2020). There are reports of additional animal species, including ferrets, fruit bats, hamsters and nonhuman primates, that have been infected with SARS‐CoV‐2 under experimental conditions (Lu et al., 2020; Schlottau et al., 2020; Shi et al., 2020 ). Cats and large felids are susceptible to SARS‐CoV‐2 infection with mild to moderate respiratory symptoms; on the other hand, dogs are less likely to show clinical signs (McAloose et al., 2020; Sailleau et al., 2020; Segalés et al., 2020 ). Molecular detection of viral RNA in dogs and cats in close contact with SARS‐CoV‐2‐infected persons has been reported in Belgium, China, France, Hong Kong, Spain, the UK and the USA (Abdel‐Moneim & Abdelwhab, 2020; Newman et al., 2020; Ruiz‐Arrondo et al., 2021; Sailleau et al., 2020). With respect to antibody detection, a serological survey of SARS‐CoV‐2 in cats in China reported that 14.7% of cats were found to be positive by using commercial enzyme‐linked immunosorbent assays (ELISA) based on receptor binding domain (RBD) (Zhang et al., 2020). In Italy, a serological study of dogs and cats living in COVID‐19‐positive households showed that 3.4% and 3.9% of dogs and cats developed neutralizing antibodies against SARS‐CoV‐2, respectively (Patterson et al., 2020). In Germany, 0.69% (six out of 920) of cats were found to show antibodies against SARS‐CoV‐2 by ELISA and immunofluorescence tests (Michelitsch et al., 2020). These reports emphasized evidence of natural infection by SARS‐CoV‐2 in dogs and cats. In this study, we conducted a large‐scale serological survey of SARS‐CoV‐2 antibodies in 3215 serum samples from domestic dogs and cats in Bangkok and in the vicinity during the period encompassing the first and second waves of COVID‐19 outbreaks in Thailand, from April 2020 to December 2020.
2. METHODS
2.1. Serum samples from domestic dogs and cats
In this study, we collected 3215 serum samples from dogs (n = 2102) and cats (n = 1113) during routine health care visits at the Chulalongkorn University Small Animal Hospital between April and December 2020. These animals were from six zones of Bangkok and nearby provinces (Nakhon Pathom, Nonthaburi, Pathum Thani, Samut Sakhon and Samut Prakan). Data related to sex, age, breed, clinical signals and owner household registration of each animal were recorded. However, information on the risk of close contact with COVID‐19 patients or households was not available. Approximately 3 ml of blood was collected from each animal, and serum was separated by centrifugation and then stored at −20°C until use. Dog and cat sera (n = 50) collected from 2014–2019 (pre‐COVID‐19 cohort serum), sera from canine respiratory coronavirus (CRCoV)‐positive dogs (n = 3), sera from canine enteric coronavirus (CECoV)‐positive dogs (n = 3) and feline coronavirus (FCoV) positive cat sera (n = 4) were obtained from the serum bank of the Center of Excellence for Emerging and Re‐emerging Infectious Diseases in Animals. The study was conducted under the approval of the Institute for Animal Care and Use Committees, Faculty of Veterinary Sciences, Chulalongkorn University (IBC#2031022 and IACUC#2031050).
2.2. Indirect ELISA assay for the detection of SARS‐CoV‐2 antibodies
To detect SARS‐CoV‐2 antibodies in serum samples, the ID Screen® SARS‐CoV‐2 Double Antigen Multi‐species ELISA kit (ID VET, Montpellier, France) was used. This indirect ELISA was based on the detection of anti‐SARS‐CoV‐2 nucleocapsid antibodies (IgG) in the tested animal serum (Sailleau et al., 2020). Indirect ELISA tests were performed according to the manufacturer's instructions. Briefly, 25 μl of each serum sample and positive and negative control samples were transferred to separate wells, diluted with 25 μl of dilution buffer and incubated at 37°C for 45 min. Each well was washed with 300 μl of washing buffer. After washing, 100 μl of horseradish peroxidase (HRP)‐conjugated N protein recombinant antigen was added and incubated at 25°C for 30 min. Then, each well was washed three times with 300 μl washing buffer. After washing, 100 μl of the substrate was added to each well, and samples were incubated at 25°C for 20 min. Then, 100 μl of stop solution was added to stop the reaction. After the addition of stop solution, the optical densities (ODs) at 450 nm were read. The OD of each sample was calculated as the S/P percentage (S/P%). Serum with S/P% > 60% was defined as positive, while serum with S/P% 50−60% was considered ‘suspect’. Indirect ELISA with the ID Screen® SARS‐CoV‐2 Double Antigen Multi‐species ELISA kit shows 100% sensitivity (10 positive samples tested) and 100% specificity (47 negative samples tested) which can be used with individual samples from susceptible species, including dogs and cats.
2.3. Virus neutralization test (VNT) for the detection of neutralizing antibodies against SARS‐CoV‐2
To detect the presence of neutralizing SARS‐CoV‐2 antibodies, in addition to the ELISA tests, both positive and suspect sera (n = 50) together with randomly selected sera (n = 15) were subjected to surrogate virus neutralization tests (sVNT). The VNT was performed by using the cPass™ SARS‐CoV‐2 Neutralization Antibody Detection Kit (GenScript Biotech, Jiangsu, China) which detects neutralizing antibodies against the interaction between the virus RBD and the ACE2 cell surface receptor (Tan et al., 2020). In brief, 50 μl of each 1:10 diluted serum sample was mixed with 50 μl HRP conjugated to the SARS‐CoV‐2 spike RBD (HRP‐RBD) and incubated at 37°C for 30 min. After that, each mixture was added to a plate that was precoated with ACE2 protein and incubated at 37°C for 15 min. Then, 260 μl of washing buffer was added to each well, and they were washed four times. After washing, 3,3',5,5'‐tetramethylbenzidine solution was added, and the plate was incubated at 25°C for 15 min. Then, the ODs at 450 nm were read by using a microplate reader. The OD of each sample was calculated as the inhibition percentage (% inhibition); serum with % inhibition above 20% was considered as positive, and serum within % inhibition not exceeding 20% was considered negative (Meyer et al., 2020). The cPass™ SARS‐CoV‐2 Neutralization Antibody Detection Kit features 95–100% sensitivity and 99.93% specificity (Tan et al., 2020).
2.4. Statistical analysis
The chi‐square test was used for statistical analysis of the variation in SARS‐CoV‐2 seropositivity with demographic variables (sex, age and location of animals) by using Prism version 9.0 (GraphPad, https://www.graphpad.com).
3. RESULTS
In this study, we collected 3125 serum samples from dogs and cats during visits to the Chulalongkorn University Small Animal Hospital, Bangkok, Thailand, from April to December 2020. The sample collection period covered the first and second waves of COVID‐19 outbreaks in Thailand. Serum samples were collected from dogs (n = 2102) and cats (n = 1113) from all zones (six zones) of Bangkok (dogs 1733, cats 940) and in the vicinity (five provinces) (dogs 370, cats 172) (Figure 1). Serum samples were collected from males (dogs 1069, cats 545) and females (dogs 987, cats 512) with ages ranging from <1 year to >13 years old. Our results showed that 1.21% (39 out of 3215) of animals tested positive by indirect ELISA for antibodies against the N protein of SARS‐CoV‐2, while 0.34% (11 out of 3215) were suspected. In detail, 1.66% (35 out of 2103) of dogs and 0.36% (four out of 1112) of cats showed positive SARS‐CoV‐2 antibody levels, with %S/P ranging from 63.49−383.40% (average 132.24%) (Figure 2 and Table S1). Positive samples from dogs were found every month from April to December 2020, while positive samples from cats were found only in April and May 2020 (Table 1). Of all animals tested, 1.82% (18 out of 987) and 1.50% (16 out of 1069) of female and male dogs were positive, compared with 0.78% (four out of 512) and 0% (zero out of 545) of female and male cats, respectively, testing positive (statistical significance, p = .0387). All age group (<1 year to >13 years old) of animals included positive cases, with the highest percentages for dogs 1–4 years old (2.20%) and cats >13 years old (3.26%) (statistical significance, p = .0001) (Table S2). In terms of location, we found positive dogs in all zones of Bangkok and in one province (Samut Prakarn), while cats were found to show low positivity in zones 1, 2 and 5 (Table 2). It should also be noted that among animals with SARS‐CoV‐2 antibodies, only five animals (five out of 39 positive animals) showed mild respiratory symptoms at the time of sample collection, such as serous nasal discharge, increased lung sound or cough. Unfortunately, the information on SARS‐CoV‐2 exposure in the positive animals was not available.
FIGURE 1.
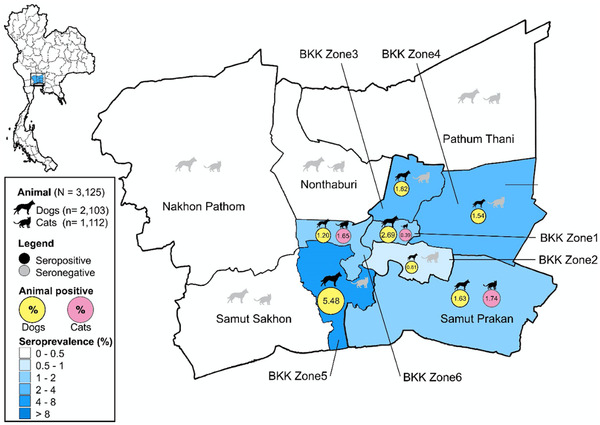
Map of Bangkok and vicinity and distribution of SARS‐CoV‐2‐positive sera in dogs and cats by indirect ELISA. The pictures of dogs and cats represent the locations and numbers of positive samples (black) and negative samples (grey) in this study
FIGURE 2.
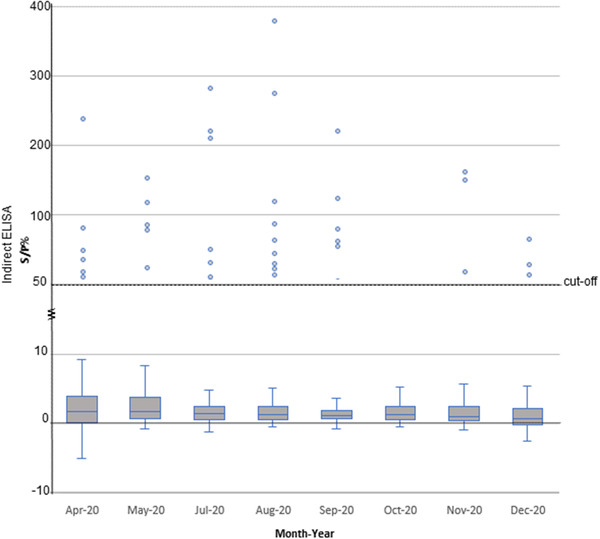
Graph depicting %S/P of indirect ELISA results. Dots represent positive samples by indirect ELISA (S/P% > 60%). Bar charts represent %SP with standard deviations. For the cut‐off values, S/P% > 60% is positive, S/P% 50−60% is suspect and < 50% is negative
TABLE 1.
Occurrence of SARS‐CoV‐2 antibodies in dogs and cats in Thailand by month (April 2020–December 2020)
| Dog | Cat | ||||
|---|---|---|---|---|---|
| Month‐Year | Sample | ELISAa positive [suspected]/test (% positive) | sVNTb positive/test (% positive) | ELISAa positive [suspected]/test (% positive) | sVNTb positive/test (% positive) |
| Apr‐20 | 504 | 4[3]/311 (1.29%) | 0/7 | 1[1]/193 (0.52%) | 0/2 |
| May‐20 | 412 | 3[1]/213 (1.41%) | 0/4 | 3[0]/199 (1.51%) | 0/3 |
| Jun‐20 | NA | NA | NA | NA | NA |
| Jul‐20 | 459 | 6[2]/278 (2.16%) | 0/8 | 0/181 | 0 |
| Aug‐20 | 523 | 12[2]/374 (3.21%) | 0/14 | 0/149 | 0 |
| Sep‐20 | 581 | 5[0]/401 (1.25%) | 0/5 | 0/180 | 0 |
| Oct‐20 | 175 | 0/115 | 0 | 0/60 | 0 |
| Nov‐20 | 175 | 2[1]/120 (1.67%) | 0/3 | 0/55 | 0 |
| Dec‐20 | 386 | 3[0]/291 (1.03%) | 0/3 | 0[1]/95 | 0/1 |
| 3215 | 35[9]/2103 (1.66%) | 0/44 | 4[2]/1112 (0.36%) | 0/6 | |
aIndirect ELISA: ID Screen® SARS‐CoV‐2 Double Antigen Multi‐species ELISA kit (ID VET, Montpellier, France). For the cut‐off values, S/P% > 60% is positive, S/P% 50−60% is suspect and < 50% is negative.
bsVNT: cPass™ SARS‐CoV‐2 Neutralization Antibody Detection Kit (GenScript Biotech, Jiangsu, China). The cut‐off values were defined as follows: positive of % inhibition > 20% and negative otherwise.
TABLE 2.
Demographic description of serum samples collected from dogs and cats and SARS‐CoV‐2 antibody detection in this study
| Dogs | Cats | ||||||
|---|---|---|---|---|---|---|---|
| Total# | Pos. [Susp.]/test | (%) | Total# | Pos. [Susp.]/test | (%) | ||
| Sex (n = 2103) | Sex (n = 1112) | ||||||
| Female (n = 987) | 18[3]/987 | (1.82%) | Female (n = 512) | 4[2]/512 | (0.78%) | ||
| Intact | 579 | 11[0]/579 | (1.90%) | Intact | 256 | 2[1]/256 | (0.78%) |
| Spayed | 408 | 7[3]/408 | (1.72%) | Spayed | 256 | 2[1]/256 | (0.78%) |
| Male (n = 1069) | 16[6]/1069 | (1.50%) | Male (n = 545) | 0/545 | 0 | ||
| Intact | 770 | 12[6]/770 | (1.56%) | Intact | 264 | 0/264 | 0 |
| Neutered | 299 | 4[0]/299 | (1.34%) | Neutered | 281 | 0/281 | 0 |
| NA (n = 47) | 47 | 1[0]/47 | (2.13%) | NA (n = 55) | 55 | 0/55 | 0 |
| Age (n = 2103) | Age (n = 1112) | ||||||
| <1 | 98 | 1[0]/98 | (1.02%) | <1 | 203 | 0[1]/203 | 0 |
| 1–4 | 409 | 9[0]/409 | (2.20%) | 1–4 | 448 | 1[0]/448 | (0.22%) |
| 5–8 | 498 | 10[4]/498 | (2.01%) | 5–8 | 214 | 0/214 | 0 |
| 9–12 | 630 | 7[2]/630 | (1.11%) | 9–12 | 145 | 0/145 | 0 |
| >13 | 437 | 8[2]/437 | (1.83%) | >13 | 92 | 3[1]/92 | (3.26%) |
| NA | 31 | 1[0]/31 | (3.23%) | NA | 10 | 0/10 | 0 |
| Location (n = 2103) | Location (n = 1112) | ||||||
| Bangkok (n = 1733) | Bangkok (n = 940) | ||||||
| Zone 1 (Central Bangkok) | 297 | 8[2]/297 | (2.69%) | Zone 1 (Central Bangkok) | 259 | 1[0]/259 | (0.39%) |
| Zone 2 (Southern Bangkok) | 742 | 6[3]/742 | (0.81%) | Zone 2 (Southern Bangkok) | 390 | 0[1]/390 | 0 |
| Zone 3 (Northern Bangkok) | 55 | 1[2]/55 | (1.82%) | Zone 3 (Northern Bangkok) | 29 | 0/29 | 0 |
| Zone 4 (Eastern Bangkok) | 65 | 1[1]/65 | (1.54%) | Zone 4 (Eastern Bangkok) | 32 | 0/32 | 0 |
| Zone 5 (Northern Thonburi) | 251 | 3[1]/251 | (1.20%) | Zone 5 (Northern Thonburi) | 121 | 2[0]/121 | (1.65%) |
| Zone 6 (Southern Thonburi) | 219 | 12[0]/219 | (5.48%) | Zone 6 (Southern Thonburi) | 77 | 0/77 | 0 |
| NA (Bangkok) | 104 | 0/104 | 0 | NA (Bangkok) | 32 | 0/32 | 0 |
| Vicinity (n = 370) | Vicinity (n = 172) | ||||||
| Nakhon Pathom | 9 | 0/9 | 0 | Nakhon Pathom | 4 | 0/4 | 0 |
| Nonthaburi | 68 | 0/68 | 0 | Nonthaburi | 34 | 0/34 | 0 |
| Pathum Thani | 27 | 0/27 | 0 | Pathum Thani | 12 | 0/12 | 0 |
| Samut Prakan | 246 | 4[0]/246 | (1.63%) | Samut Prakan | 115 | 2[0]/115 | (1.74%) |
| Samut Sakhon | 20 | 0/20 | 0 | Samut Sakhon | 7 | 0/7 | 0 |
In this study, the archived serum samples (24 dogs, and 26 cats) from the pre‐COVID‐19 cohort from 2014 to 2019, together with serum from four cats with FCoV infection, three dogs with canine respiratory coronavirus and three dogs with canine enteric virus infection, were also included in the indirect ELISA tests. None of these serum samples tested positive for antibodies against the N protein of SARS‐CoV‐2, suggesting specificity of the assay or no potential cross‐reactive antibodies (Table S1). In contrast to the ELISA results, all serum samples that were positive for IgG antibodies against SARS‐CoV‐2 showed negative results for neutralizing antibodies by sVNT. Fifty positive and suspected serum samples from dogs (n = 44) and cats (n = 6) showed % inhibition lower than 20% (0.08−19.37%, average 7.82%), indicating a lack of neutralizing antibodies against the SARS‐CoV‐2 virus based on the RBD.
4. DISCUSSION
Thailand has faced at least three waves of COVID‐19 outbreaks. The first wave took place from March to May 2020, and the second wave lasted from December 2020 to February 2021. The current third wave of the outbreak started in late March 2021, and the number of confirmed SARS‐CoV‐2 cases is still rising (Figure S1). Although SARS‐CoV‐2 mainly circulates in the human population, causing a significant impact on human health and socioeconomic conditions, there are some reports of SARS‐CoV‐2 infection in domestic animals. A study on host receptors for SARS‐CoV‐2 showed that many animal species, including cats, ferrets, orangutans and monkeys, have angiotensin‐converting enzyme‐2 receptors identical or similar to those of humans. Therefore, the transmission of SARS‐CoV‐2 from humans to those animals is theoretically possible (Hossain et al., 2020). A field survey on domestic animals in known COVID‐19‐positive areas and households showed that natural infection with SARS‐CoV‐2 can occur in dogs and cats living in these locations, resulting in seroconversion against SAR‐CoV‐2 in the animals (Patterson et al., 2020). In this study, our results showed that domestic dogs (1.66%) and cats (0.36%) in Bangkok and vicinity tested positive for anti‐N‐IgG antibodies against SARS‐CoV‐2. The seropositivity in this study is comparable to that observed in several countries. For example, in Germany, only 0.69% (six out of 920) of cat sera from a veterinary diagnostic laboratory were seropositive by indirect multispecies ELISA and indirect immunofluorescence tests (Michelitsch et al., 2020). In Italy, 3.3% of dogs (15 out of 451) and 5.8% of cats (11 out of 191) from health care visits were seropositive by the plaque reduction neutralization test. According to COVID‐19‐positive households, higher seropositivity was observed at 12.8% (six out of 47) in dogs and 4.5% (one out of 22) in cats (Patterson et al., 2020). On the other hand, in Wuhan, China, serum samples were collected from pet hospitals, abandoned animals and COVID‐19 patient households and showed high level of seropositivity for SARS‐CoV‐2 antibodies of 14.70% for cats (15 out of 102) and 1.69% for dogs (16 out of 946) (Zhang et al., 2020; Zhao et al., 2022 ). Similarly, in France, 15.4% of dogs (two out of 13) and 23.5% of cats (eight out of 34) from COVID‐19‐positive households exhibited high seropositivity by microsphere immunoassay and neutralization tests (Fritz et al., 2021).
Serological diagnosis is an essential tool to determine viral exposure in populations. To date, several serological assays have been developed and employed to facilitate diagnosis of SARS‐CoV‐2 infections. The spike (S) and nucleocapsid (N) structural proteins of SARS‐CoV‐2 display high immunogenic activity, making these proteins attractive candidates for the development of serological diagnostic assays (Liu et al., 2020). Both ELISA and neutralization assays are typically considered the gold standards for the detection of antibodies against SARS‐CoV‐2 (Liu et al., 2020; Tan et al., 2020; Tehrani et al., 2020 ). The ELISA and sVNT assays used in this study feature high sensitivity and specificity (Tan et al., 2020). Spike‐based assay, especially with the S1 subunit, is reportedly the most specific for the diagnosis of COVID‐19, whereas N‐based assays offer a higher sensitivity (Kontou et al., 2020; Liu et al., 2020; Okba et al., 2020; Tehrani et al., 2020 ). Nevertheless, a comparable sensitivity and specificity of at least 98% were reported for both the S‐ and N‐based commercially available assays in a recent head‐to‐head comparative study (National, 2020). Even though similar diagnostic performance was observed for both assays, detection of antibodies against S protein is advantageous as it correlates well with neutralization test (Folegatti et al., 2020; Ni et al., 2020; Okba et al., 2020 ). Therefore, it was not unexpected that the 50 sera tested positive on N‐based ELISA in this study lacked neutralizing activity. One limitation of our study is the discrepancy between the results of indirect ELISA and sVNT, which deserves further investigation. This discrepancy is consistent with findings of some previous studies. For example, a serological study of cats in Germany showed six positive serum samples by indirect multispecies ELISA, and only two out of six were VNT positive (Michelitsch et al., 2020). In China, a serological study in dogs and cats showed discrepancies between results of ELISA and VNT assays, which might have been related to the background of the animals. The animals from pet hospitals and stray dogs and cats had low neutralizing antibody levels when compared with animals owned by COVID‐19 patients (Zhang et al., 2020; Zhao et al., 2022 ). Another explanation for the discrepancy is that the timing of animal infection and serum sample collection may affect the ELISA and VNT test results. Antibody response to SARS‐COV‐2 has been found to be relatively high within 10 days of infection, but the duration of the peak titre was short (Zhang et al., 2020). N is immunodominant, inducing an earlier and higher immune response (Bond et al., 2020; Hachim et al., 2020 ). While the neutralizing antibody response develops later and peaks at 7–13 days after infection (Bosco‐Lauth et al., 2020; Schlottau et al., 2020; Shi et al., 2020 ). It should be noted that the structure of the nucleocapsid protein is conserved, cross‐reactivity with antibodies against other betacoronavirues, such as OC43 and HKU1, is another concern regarding the use of N‐based ELISA in human cases. However, CRCoV, a betacoronavirus, shares only 45.8–46.2% nucleotide similarity with SARS‐CoV‐2. Also, FCoV and CECoV, the most common coronavirus in cats and dogs, belong to Alphacoronavirus‐1 and considerably different from SARS‐CoV‐2 (Sharun et al., 2020). Thus, cross‐reactivity with other CoVs by ELISA is possible, even less likely, and requires further investigation.
In conclusion, our results demonstrated that dogs and cats living in COVID‐19‐affected areas in Thailand with unknown SARS‐CoV‐2 exposure developed IgG antibodies against the virus. The role of dogs and cats in SARS‐CoV‐2 maintenance and transmission within species or across species from animals to humans remains unclear. Serosurveys for SARS‐CoV‐2 infection in animals should be routinely conducted. Therefore, awareness should be raised of SARS‐CoV‐2 infection in pets, and recommendations on the potential risks of the human–pet interface should be provided for high‐risk occupations and the general public.
ETHICS STATEMENT
This study was conducted under the approval of the Institute for Animal Care and Use Committee protocol of the Faculty of Veterinary Sciences, Chulalongkorn University (IBC#2031022 and IACUC#2031050).
CONFLICT OF INTEREST
All authors declare no conflict of interest.
Supporting information
SUPPORTING INFORMATION
ACKNOWLEDGEMENTS
We would like to thank the staff at the Chulalongkorn University Small Animal Hospital for collaboration in sample collection. Chulalongkorn University supported the Center of Excellence for Emerging and Re‐emerging Infectious Diseases in Animals (CUEIDAs) and the One Health Research Cluster (764002‐HE02). The Thailand Research Fund (TRF) supported a grant for senior scholar to the corresponding author (Alongkorn Amonsin) (RTA6080012). This research is funded by Chulalongkorn University's TSRI Fund (CU_FRB640001_01_31_1), the Agricultural Research Development Agency (ARDA) fund (PRP6405031220) and the Office of National Higher Education Science Research and Innovation Policy Council (NXPO‐PMU‐B) (B17F640011).
Udom, K. , Jairak, W. , Chamsai, E. , Charoenkul, K. , Boonyapisitsopa, S. , Bunpapong, N. , Techakriengkrai, N. , & Amonsin, A. (2022). Serological survey of antibodies against SARS‐CoV‐2 in dogs and cats, Thailand. Transboundary and Emerging Diseases, 69, 2140–2147. 10.1111/tbed.14208
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available in the supplementary material for this article.
REFERENCES
- Abdel‐Moneim, A. S. , & Abdelwhab, E. M. (2020). Evidence for SARS‐CoV‐2 infection of animal hosts. Pathogens, 9(7). 10.3390/pathogens9070529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bond, K. , Williams, E. , Howden, B. P. , & Williamson, D. A. (2020). Serological tests for COVID‐19. Medical Journal of Australia, 213(9), 397–399 e391. 10.5694/mja2.50766 [DOI] [PubMed] [Google Scholar]
- Bosco‐Lauth, A. M. , Hartwig, A. E. , Porter, S. M. , Gordy, P. W. , Nehring, M. , Byas, A. D. , Vandewoude, S. , Ragan, I. K. , Maison, R. M. , & Bowen, R. A. (2020). Experimental infection of domestic dogs and cats with SARS‐CoV‐2: Pathogenesis, transmission, and response to reexposure in cats. Proceedings of the National Academy of Sciences of the United States of America, 117(42), 26382–26388. 10.1073/pnas.2013102117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folegatti, P. M. , Ewer, K. J. , Aley, P. K. , Angus, B. , Becker, S. , Belij‐Rammerstorfer, S. , Bellamy, D. , Bibi, S. , Bittaye, M. , Clutterbuck, E. A. , Dold, C. , Faust, S. N. , Finn, A. , Flaxman, A. L. , Hallis, B. , Heath, P. , Jenkin, D. , Lazarus, R. , Makinson, R. , … Oxford, C. V. T. G. (2020). Safety and immunogenicity of the ChAdOx1 nCoV‐19 vaccine against SARS‐CoV‐2: A preliminary report of a phase 1/2, single‐blind, randomised controlled trial. Lancet, 396(10249), 467–478. 10.1016/S0140-6736(20)31604-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritz, M. , Rosolen, B. , Krafft, E. , Becquart, P. , Elguero, E. , Vratskikh, O. , Denolly, S. , Boson, B. , Vanhomwegen, J. , Gouilh, M. A. , Kodjo, A. , Chirouze, C. , Rosolen, S. G. , Legros, V. , … Leroy, E. M. (2021). High prevalence of SARS‐CoV‐2 antibodies in pets from COVID‐19+ households. One Health, 11, 100192. 10.1016/j.onehlt.2020.100192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachim, A. , Kavian, N. , Cohen, C. A. , Chin, A. W. H. , Chu, D. K. W. , Mok, C. K. P. , Tsang, O. T. Y. , Yeung, Y. C. , Perera, R. A. P. M. , Poon, L. L. M. , Peiris, J. S. M. , & Valkenburg, S. A. (2020). ORF8 and ORF3b antibodies are accurate serological markers of early and late SARS‐CoV‐2 infection. Nature Immunology, 21(10), 1293–1301. 10.1038/s41590-020-0773-7 [DOI] [PubMed] [Google Scholar]
- Hossain, M. G. , Javed, A. , Akter, S. , & Saha, S. (2020). SARS‐CoV‐2 host diversity: An update of natural infections and experimental evidence. Journal of Microbiology, Immunology and Infection, 54(2), 175–181 10.1016/j.jmii.2020.06.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kontou, P. I. , Braliou, G. G. , Dimou, N. L. , Nikolopoulos, G. , & Bagos, P. G. (2020). Antibody tests in detecting SARS‐CoV‐2 infection: A meta‐analysis. Diagnostics (Basel), 10(5),. 10.3390/diagnostics10050319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leroy, E. M. , Ar Gouilh, M. , & Brugere‐Picoux, J. (2020). The risk of SARS‐CoV‐2 transmission to pets and other wild and domestic animals strongly mandates a one‐health strategy to control the COVID‐19 pandemic. One Health, 10, 100133. 10.1016/j.onehlt.2020.100133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, W. , Liu, L. , Kou, G. , Zheng, Y. , Ding, Y. , Ni, W. , Wang, Q. , Tan, L. , Wu, W. , Tang, S. , & Xiong, Z. (2020). Evaluation of nucleocapsid and spike protein‐based enzyme‐linked immunosorbent assays for detecting antibodies against SARS‐CoV‐2. Journal of Clinical Microbiology, 58(6),. 10.1128/JCM.00461-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu, S. , Zhao, Y. , Yu, W. , Yang, Y. , Gao, J. , Wang, J. , Kuang, D. , Yang, M. , Yang, J. , Ma, C. , Xu, J. , Qian, X. , Li, H. , Zhao, S. , Li, J. , Wang, H. , Long, H. , Zhou, J. , Luo, F. , … Peng, X. (2020). Comparison of nonhuman primates identified the suitable model for COVID‐19. Signal Transduction and Targeted Therapy, 5(1), 157. 10.1038/s41392-020-00269-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAloose, D. , Laverack, M. , Wang, L. , Killian, M. L. , Caserta, L. C. , Yuan, F. , Mitchell, P. K. , Queen, K. , Mauldin, M. R. , Cronk, B. D. , Bartlett, S. L. , Sykes, J. M. , Zec, S. , Stokol, T. , Ingerman, K. , Delaney, M. A. , Fredrickson, R. , Ivančić, M. , Jenkins‐Moore, M. , … Cronk, B. D. (2020). From people to Panthera: Natural SARS‐CoV‐2 infection in tigers and lions at the Bronx Zoo. Mbio, 11((5)),. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer, B. , Reimerink, J. , Torriani, G. , Brouwer, F. , Godeke, G.‐J. , Yerly, S. , Hoogerwerf, M. , Vuilleumier, N. , Kaiser, L. , Eckerle, I. , & Reusken, C. (2020). Validation and clinical evaluation of a SARS‐CoV‐2 surrogate virus neutralisation test (sVNT). Emerging Microbes & Infections, 9(1), 2394–2403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michelitsch, A. , Hoffmann, D. , Wernike, K. , & Beer, M. (2020). Occurrence of antibodies against SARS‐CoV‐2 in the domestic cat population of Germany. Vaccines (Basel), 8(4). 10.3390/vaccines8040772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- National, S.‐C.‐S. A. E. G. (2020). Performance characteristics of five immunoassays for SARS‐CoV‐2: A head‐to‐head benchmark comparison. The Lancet Infectious Diseases, 20(12), 1390–1400. 10.1016/S1473-3099(20)30634-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman, A. , Smith, D. , Ghai, R. R. , Wallace, R. M. , Torchetti, M. K. , Loiacono, C. , Murrell, L. S. , Carpenter, A. , Moroff, S. , Rooney, J. A. , & Barton Behravesh, C. (2020). First reported cases of SARS‐CoV‐2 infection in companion animals ‐ New York, March‐April 2020. Morbidity and Mortality Weekly Report, 69(23), 710–713. 10.15585/mmwr.mm6923e3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni, L. , Ye, F. , Cheng, M.‐L. , Feng, Y. , Deng, Y.‐Q. , Zhao, H. , Wei, P. , Ge, J. , Gou, M. , Li, X. , Sun, L. , Cao, T. , Wang, P. , Zhou, C. , Zhang, R. , Liang, P. , Guo, H. , Wang, X. , Qin, C.‐F. , … Dong, C. (2020). Detection of SARS‐CoV‐2‐specific humoral and cellular immunity in COVID‐19 convalescent individuals. Immunity, 52(6), 971–977 e973. 10.1016/j.immuni.2020.04.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okba, N. M. A. , Müller, M. A. , Li, W. , Wang, C. , Geurtsvankessel, C. H. , Corman, V. M. , Lamers, M. M. , Sikkema, R. S. , De Bruin, E. , Chandler, F. D. , Yazdanpanah, Y. , Le Hingrat, Q. , Descamps, D. , Houhou‐Fidouh, N. , Reusken, C. B. E. M. , Bosch, B.‐J. , Drosten, C. , Koopmans, M. P. G. , & Haagmans, B. L. (2020). Severe Acute Respiratory Syndrome Coronavirus 2‐specific antibody responses in coronavirus disease patients. Emerging Infectious Diseases, 26(7), 1478–1488. 10.3201/eid2607.200841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patterson, E. I. , Elia, G. , Grassi, A. , Giordano, A. , Desario, C. , Medardo, M. , Smith, S. L. , Anderson, E. R. , Prince, T. , Patterson, G. T. , Lorusso, E. , Lucente, M. S. , Lanave, G. , Lauzi, S. , Bonfanti, U. , Stranieri, A. , Martella, V. , Basano, F. S. , Barrs, V. R. , … Decaro, N. (2020). Evidence of exposure to SARS‐CoV‐2 in cats and dogs from households in Italy. bioRxiv, 10.1101/2020.07.21.214346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz‐Arrondo, I. , Portillo, A. , Palomar, A. M. , Santibanez, S. , Santibanez, P. , Cervera, C. , & Oteo, J. A. (2021). Detection of SARS‐CoV‐2 in pets living with COVID‐19 owners diagnosed during the COVID‐19 lockdown in Spain: A case of an asymptomatic cat with SARS‐CoV‐2 in Europe. Transboundary and Emerging Diseases, 68(2), 973–976. 10.1111/tbed.13803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sailleau, C. , Dumarest, M. , Vanhomwegen, J. , Delaplace, M. , Caro, V. , Kwasiborski, A. , Hourdel, V. , Chevaillier, P. , Barbarino, A. , Comtet, L. , Pourquier, P. , Klonjkowski, B. , Manuguerra, J.‐C. , Zientara, S. , & Le Poder, S. (2020). First detection and genome sequencing of SARS‐CoV‐2 in an infected cat in France. Transboundary and Emerging Diseases, 67(6), 2324–2328. 10.1111/tbed.13659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlottau, K. , Rissmann, M. , Graaf, A. , Schön, J. , Sehl, J. , Wylezich, C. , Höper, D. , Mettenleiter, T. C. , Balkema‐Buschmann, A. , Harder, T. , Grund, C. , Hoffmann, D. , Breithaupt, A. , & Beer, M. (2020). SARS‐CoV‐2 in fruit bats, ferrets, pigs, and chickens: An experimental transmission study. Lancet Microbe, 1(5), e218–e225. 10.1016/S2666-5247(20)30089-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segalés, J. , Puig, M. , Rodon, J. , Avila‐Nieto, C. , Carrillo, J. , Cantero, G. , Terrón, M. T. , Cruz, S. , Parera, M. , Noguera‐Julián, M. , Izquierdo‐Useros, N. , Guallar, V. , Vidal, E. , Valencia, A. , Blanco, I. , Blanco, J. , Clotet, B. , & Vergara‐Alert, J. (2020). Detection of SARS‐CoV‐2 in a cat owned by a COVID‐19− affected patient in Spain. Proceedings of the National Academy of Sciences of the United States of America, 117(40), 24790–24793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharun, K. , Sircar, S. , Malik, Y. S. , Singh, R. K. , & Dhama, K. (2020). How close is SARS‐CoV‐2 to canine and feline coronaviruses? Journal of Small Animal Practice, 61(8), 523–526. 10.1111/jsap.13207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi, J. , Wen, Z. , Zhong, G. , Yang, H. , Wang, C. , Huang, B. , Liu, R. , He, X. , Shuai, L. , Sun, Z. , Zhao, Y. , Liu, P. , Liang, L. , Cui, P. , Wang, J. , Zhang, X. , Guan, Y. , Tan, W. , Wu, G. , … Bu, Z. (2020). Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS‐coronavirus 2. Science, 368(6494), 1016–1020. 10.1126/science.abb7015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan, C. W. , Chia, W. N. , Qin, X. , Liu, P. , Chen, M. I.‐C. , Tiu, C. , Hu, Z. , Chen, V. C.‐W. , Young, B. E. , Sia, W. R. , Tan, Y.‐J. , Foo, R. , Yi, Y. , Lye, D. C. , Anderson, D. E. , & Wang, L. F. (2020). A SARS‐CoV‐2 surrogate virus neutralization test based on antibody‐mediated blockage of ACE2‐spike protein‐protein interaction. Nature Biotechnology, 38(9), 1073–1078. 10.1038/s41587-020-0631-z [DOI] [PubMed] [Google Scholar]
- Tehrani, Z. R. , Saadat, S. , Saleh, E. , Ouyang, X. , Constantine, N. , DeVico, A. L. , Harris, A. D. , Lewis, G. K. , Kottilil, S. , & Sajadi, M. M. (2020). Specificity and performance of nucleocapsid and spike‐based SARS‐CoV‐2 serologic assays. medRxiv, 10.1101/2020.08.05.20168476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO . (2021). WHO Coronavirus (COVID‐19) Dashboard. Retrieved 7 May 2021. https://covid19.who.int
- Zhang, Q. , Zhang, H. , Gao, J. , Huang, K. , Yang, Y. , Hui, X. , He, X. , Li, C. , Gong, W. , Zhang, Y. , Zhao, Y. , Peng, C. , Gao, X. , Chen, H. , Zou, Z. , Shi, Z.‐L. , & Jin, M. (2020). A serological survey of SARS‐CoV‐2 in cat in Wuhan. Emerging Microbes & Infection, 9(1), 2013–2019. 10.1080/22221751.2020.1817796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao, Y. , Yang, Y. , Gao, J. , Huang, K. , Hu, C. , Hui, X. , He, X. , Li, C. , Gong, W. , Lv, C. , Zhang, Y. , Chen, H. , Zou, Z. , Zhang, Q. , & Jin, M. (2022). A serological survey of severe acute respiratory syndrome coronavirus 2 in dogs in Wuhan. Transboundary and Emerging Diseases, 69(2), 591–597. 10.1111/tbed.14024 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
SUPPORTING INFORMATION
Data Availability Statement
The data that support the findings of this study are available in the supplementary material for this article.


