Abstract
Background
Current evidence regarding COVID‐19 convalescent plasma (CCP) transfusion practices is limited and heterogeneous. We aimed to determine the impact of the use of CCP transfusion in patients with previous circulating neutralizing antibodies (nAbs) in COVID‐19.
Methods
Prospective cohort including 102 patients with COVID‐19 transfused with ABO compatible CCP on days 0–2 after enrollment. Clinical status of patients was assessed using the adapted World Health Organization (WHO) ordinal scale on days 0, 5, and 14. The nAbs titration was performed using the cytopathic effect‐based virus neutralization test with SARS‐CoV‐2 (GenBank MT126808.1). The primary outcome was clinical improvement on day 14, defined as a reduction of at least two points on the adapted WHO ordinal scale. Secondary outcomes were the number of intensive care unit (ICU)‐free days and the number of invasive mechanical ventilation‐free days.
Results
Both nAbs of CCP units transfused (p < 0.001) and nAbs of patients before CCP transfusions (p = 0.028) were associated with clinical improvements by day 14. No significant associations between nAbs of patients or CCP units transfused were observed in the number of ICU or mechanical ventilation‐free days. Administration of CCP units after 10 days of symptom onset resulted in a decrease in ICU‐free days (p < 0.001) and mechanical ventilation‐free days (p < 0.001).
Conclusion
Transfusion of high titer nAbs CCP units may be a determinant in clinical strategies against COVID‐19. We consider these data as useful parameters to guide future CCP transfusion practices.
Keywords: convalescent plasma transfusion, COVID‐19, neutralizing antibodies, SARS‐CoV‐2
1. INTRODUCTION
COVID‐19 convalescent plasma (CCP) transfusion has emerged early in the pandemic as a safe alternative approach to treat pneumonia cases of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS‐CoV‐2).1, 2, 3 Several specific therapies are still under development, with limited evidence concerning efficacy and survival benefits.4, 5, 6 Historically, convalescent plasma has been used as an emergent strategy in many outbreaks, such as Spanish influenza7 and the Severe Acute Respiratory Syndrome for Coronavirus (SARS‐CoV‐1),8 with conflicting results about clinical benefit.7, 9
The rationale for CCP transfusion relies on the fact that convalescent individuals have circulating neutralizing antibodies (nAbs) that are able to suppress the infection.10 Other immunomodulatory mechanisms, such as complement activation, antibody‐dependent cytotoxicity, or phagocytosis are potential pathways through which CCP might alleviate systemic inflammation. Also, non‐nAbs that are bound to the virus, despite not interfering with replication capacity, might contribute to recovery.11 Previous studies have demonstrated that CCP transfusion was able to inhibit SARS‐CoV‐2 replication12 and its efficacy was associated with the concentration of nAbs in the plasma of convalescent donors.13, 14, 15 However, data about the clinical impact of nAbs on COVID‐19 outcomes are still sparse.
Current evidence regarding characteristics of the plasma product, the titer of nAbs in the transfused units, time to onset of intervention, and impact of nAbs produced by the patient are limited and heterogeneous, which precludes consistent conclusions from the available data. In this study, we describe the results of 102 patients with severe pneumonia due to SARS‐CoV‐2 transfused with CCP at three medical centers in Brazil.
2. MATERIALS AND METHODS
This was a single‐arm prospective study conducted at three referral medical centers in the cities of São Paulo and Brasilia, Brazil. The study was approved by the Brazilian National Commission for Research Ethics—CONEP, CAAE #30922420.6.2002.0071 and by the Ethical Committees of participating institutions. Informed consent was obtained from all participants of the study or from their legal representatives.
2.1. Convalescent plasma collection
CCP donors with laboratory‐confirmed COVID‐19 diagnosis by reverse transcriptase‐polymerase chain reaction (RT‐PCR), fully recovered and asymptomatic for at least 14 days were recruited at participating sites. Donor screening considered the Brazilian regular criteria for blood donation and negative RT‐PCR from blood and naso‐oropharyngeal swabs that were collected on the day of the donation. Serum for determination of SARS‐CoV‐2 nAbs from each donor was also collected on the day of the donation for further analysis. Only men, nulliparous women, or women with history of up to two gestations and who had negative HLA screening (Lifecodes Lifescreen Deluxe—Immucor) were eligible for donation of CCP. Convalescent plasma products were obtained from whole blood or plasmapheresis donations.16
2.2. Patients
From April 11 to October 25, we assessed for eligibility 191 consecutive patients admitted to participating sites with COVID‐19 confirmed diagnosis by RT‐PCR, aged ≥18 years old and with criteria for severe pneumonia (defined by respiratory distress: oxygen saturation of 93% or less on room air, respiratory rate >30 breaths/min and/or arterial partial pressure of oxygen [PaO2]/fraction of inspired oxygen [FiO2] of 300 or less). Patients with preexisting history of anaphylactic transfusion reaction, pregnant or lactating women were excluded for safety reasons. A total of 102 patients comprised the final sample.
2.3. Intervention
All enrolled patients were transfused with 1 or 2 doses of 200–300 ml of ABO compatible CCP in days 0–2 after enrolment, with close monitoring. Criteria for a second dose of CCP included mechanical ventilation and absence of clinical improvement within 2 days from the first CCP transfusion. CCP units with nAbs titers ≥160 were transfused, except for the first 11 patients included in the first 2 weeks of the study, given that nAbs from CCP units were not available before transfusion at that time. These patients received 16 CCP doses with nAb geometric mean titers ranging from 20 to 640. Titration of nAbs was performed using the cytopathic effect‐based virus neutralization test (CPE‐based VNT) with SARS‐CoV‐2 (GenBank MT126808.1)17 with patient samples on the day of enrollment (day 0) (nAbs P0) and on day 5 (nAbs P5) after CCP transfusions. Samples were kept frozen at ‐80°C for further analysis. Circulating nAbs produced by the patients prior to transfusion were not available because of the turnaround time of this assay. The clinical status of patients was assessed according to severity organ failure assessment (SOFA) score on day 0 and the evolution was assessed using the adapted World Health Organization (WHO) ordinal scale18 on days 0, 5, and 14. Intensive care unit (ICU)‐free days, mechanical ventilation‐free days, and disease evolution were recorded.
2.4. Outcomes
The primary outcome was clinical improvement on day 14, defined as a reduction of at least two points on the adapted WHO ordinal scale. Secondary outcomes were ICU‐free days and invasive mechanical ventilation‐free days during the first 30 days of the study and progression to mechanical ventilation.
2.5. Statistical analysis
Baseline characteristics were described as counts and percentages for categorical variables or mean and standard deviation (SD) for continuous variables with a normal distribution. Median, 1st and 3rd quartiles, that is, interquartile range (IQR), were used to describe continuous variables with asymmetrical distribution. Univariate and multivariate saturated regression models were adjusted to assess the outcomes: clinical improvement on the day 14 (binary), ICU‐free days, mechanical ventilation‐free days on the day 30 of the study, and progression to mechanical ventilation. Explanatory variables considered were mean geometric titer of nabs from CP units transfused (nAbsCP), neutralizing antibody titers from patients before transfusion (nAbsP0), neutralizing antibody titers from patients on day 5 (nAbsP5), age, sex, weight, ABO group, severity organ failure assessment score on the admission day (SOFA D0), use of other supportive therapies for COVID‐19 (azithromycin, hydroxychloroquine, steroids, tocilizumab, human immunoglobulin, antiviral therapies), and comorbidities. To analyze if earlier CP transfusion had an impact on outcomes, we also analyzed whether CP transfusion was performed up to 10 days of the disease onset or later. For each outcome, we performed a generalized estimating equation model (GEE) to account for the dependence of data collected from each of the three institutions according to a compound symmetry correlation structure. For mechanical ventilation‐free days during the first 30 days of the study, we used the negative binomial distribution, and for ICU‐free days we adopted the gamma distribution, both models with log link functions. Clinical improvement on day 14 was assessed with a binomial distribution and logit link function. Results were presented as mean ratios (MR) or odds ratios (OR) with 95% confidence intervals and p values; p < 0.05 was considered statistically significant. For the analysis, we used IBM SPSS Statistics for Windows, Version 24.0. 2016 and the R package.19 Clinically relevant variables selected for the univariate models were all considered in multivariate models to control the effects of the explanatory variables in the saturated models, and no variable selection was applied. Only neutralizing antibody titers from patients prior to enrolment and on day 5 of the study (nAbsP0 and nAbsP5) were not considered in the same model to avoid multicollinearity. Interactions were not investigated due to the restricted sample size for the multivariate models.
3. RESULTS
Table 1 shows baseline characteristics of the patients and CCP transfusion data. The mean age of the patients was 64 years old (SD: 15 years), 72 patients (70.6%) were men. Ninety‐one (89.2%) had different comorbidities, and arterial hypertension was present in 51% of the individuals, diabetes mellitus in 29.4%, and 35.6% were overweight (body mass index ≥25) or obese (body mass index of 30 or higher). Patients' mean body weight was 86 kg (SD: 19 kg). Regarding the ABO blood group, 46 (45.1%) were A individuals, 44 (43.1%) were O, 10 were B (9.8%) and 2(2.0%) were AB. Most of the patients (73.5%%) have also received other therapies (azithromycin, hydroxychloroquine, tocilizumab and/or combinations). Ninety‐eight patients (96%) were treated with steroids. Thirty‐two patients (31.4%) were already intubated prior to the enrollment at the study, and 18 (25.7%) progressed to mechanical ventilation, totalizing 50 patients (49.0%) requiring invasive mechanical ventilation. NAbs from patients on day 0 (immediately before CCP transfusions—nAbsP0) ranged from 10 to 10,240 [1st Q 640–3rd Q 5120]. To consider the dose‐effect of nAbs on each outcome, the total amount of nAbs were calculated using the geometric mean nAbs titer from CCP units (nAbsCP), which varied from 20 to 3044 [IQR 254–640]. Two patients received doses of 200 ml (2.0%), 19 patients received doses of 300 ml (18.6%), 22 patients received 400 ml (21.6%), and 59 received 600 ml (57.8%). CCP transfusions were well‐tolerated and five (4.8%) mild to moderate transfusion reactions occurred: three febrile non‐hemolytic reactions and two allergic reactions. Two suspected cases were screened for TRALI (donor screening: Lifecodes Lifescreen Deluxe—Immucor) with negative results. No severe adverse events were observed. Six (5.7%) had thromboembolic events (TE) after CCP infusion, even under thrombotic prophylaxis or on full anticoagulation due to TE diagnosed before the enrollment.
TABLE 1.
Demographic and clinical data of studied patients (n = 102)
| Sex | |
| Female | 30 (29.4%) |
| Male | 72 (70.6%) |
| Age (years) (n = 102) | |
| Median (SD) | 64 (15) |
| Min–Max | 31–95 |
| Body weight (kg),(n = 102) | |
| Median | 86 (19) |
| Min–Max | 42–144 |
| ABO | |
| A | 46 (45.1%) |
| AB | 2 (2.0%) |
| B | 10 (9.8%) |
| O | 44 (43.1%) |
| Comorbidities | |
| Hypertension | 52 (51%) |
| Diabetes | 30 (29.4%) |
| Obesity | 36 (35.6%) |
| OMS ordinal scale—D0 | |
| Median (IQR) | 6 (5; 7) |
| Min–Max | 4–9 |
| SOFA score D0 (n = 96) | |
| Median (IQR) | 2 (1; 4) |
| Min–Max | 0–12 |
| Use of other therapies | 75 (73.5%) |
| Hydroxychloroquin | 35 (34.3%) |
| Azythromicin | 73 (71.6%) |
| Immuneglobulin | 3 (2.9%) |
| Monoclonal antibodies | 8 (7.8%) |
| NAbs P0 (n = 101) | |
| Median (IQR) | 160 (20; 640) |
| Min–Max | 10–10,240 |
| NAbs P5 (n = 91) | |
| Median (IQR) | 5120 (640; 5120) |
| Min–Max | 10–5120 |
| Volume transfused (ml) | |
| 200 | 2 (2.0%) |
| 300 | 19 (18.6%) |
| 400 | 22 (21.6%) |
| 600 | 59 (57.8%) |
| Total | 102 (100.0%) |
| NAbsCP (N = 102) | |
| Median (IQR) | 403 (254; 640) |
| Min–Max | 20–3044 |
| Invasive mechanical ventilation | 50 (49.0%) |
| Number of ventilation‐free days over the first 30 days of the study | |
| Median (IQR) | 30 (17; 30) |
| Min–Max | 0–30 |
| ICU admission | 93 (91.2%) |
| Number of ICU‐free days over the first 30 days of the study | |
| Median (IQR) | 18 (7; 25) |
| Min–Max | 0–30 |
| Death | 11 (10.8%) |
| Hospital length of stay | |
| Median (IQR) | 18 (11; 32) |
| Min–Max | 3–128 |
Abbreviations: nAbs P0, Neutralizing antibody titers from patient samples before convalescent plasma transfusion; nAbs CP, mean geometric titer of nAbs from CP units transfused.
Figure 1 shows the timeline of clinical evolution after the onset of illness for the 102 patients. The median time from onset of illness to hospital admission was 6.6 days [IQR 4.0–9.0], while the median time from onset of illness to CCP transfusion was 10 days [IQR 8.0–13.0].
FIGURE 1.
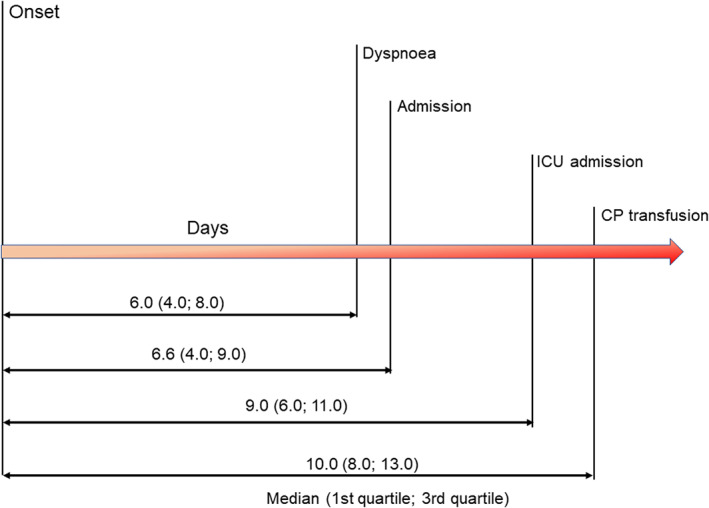
Timeline of COVID‐19 patients included in the study [Color figure can be viewed at wileyonlinelibrary.com]
Clinical improvement was assessed using the adapted WHO scale score on days 0, 5 and, 14 (Figure 2). By day 5, 19 (18.6%) patients have achieved clinical improvement. On day 14, the proportion of patients who had improved raised to 45.1%, with 40.1% of patients with mild disease (based on WHO ordinal scale between 0 and 4), and 38 (37.25%) had been discharged. There were 11 deaths (10.8%) and one event happened before day 7 after CCP transfusion.
FIGURE 2.
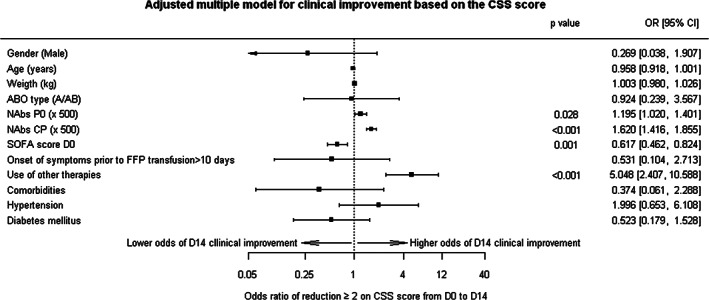
Clinical improvement on day 14 post‐CCP transfusion. nAbs P0 = Neutralizing antibody titers from patient samples before convalescent plasma transfusion, nAbs CP = mean geometric titer of nAbs from CP units transfused. Variables without a p value mean that p was not statistically significant
Multivariate analysis showed that the level of patient nAb titers prior to transfusion (nAbsP0) (Odds ratio (OR) = 1.195; 95% CI: 1.020–1.401, p = 0.028), the mean geometric nAbs titer from units transfused (nAbsCP) (OR = 1.620; 95% CI 1.416–1.855, p < 0.001) and the use of other supportive therapies for COVID‐19 (OR = 5.048; 95% CI 2.407–10.558, p < 0.001) were associated with higher odds of clinical improvement on day 14. However, the SOFA score on day 0 (OR = 0.617; 95% CI 0.462–0.824, p = 0.001) was associated with lower chances of clinical improvement. Age, sex, ABO group, body weight, time from onset of illness until CCP transfusion and comorbidities had no impact on clinical improvement on day 14.
We sought to determine predictive variables for the duration of mechanical ventilation. To better predict the impact of CCP transfusion on mechanical ventilation (Table 2), we analyzed ventilation‐free days over the first 30 days of the study for all patients included. Fifty (49%) patients required mechanical ventilation support, intubation periods varied from 2 to 61 days and the median duration of this period was 11 days [IQR 5–16]. The median number of ventilation‐free days was 30 [IQR 17–30]. GEE models were performed to identify associated variables. NAbsP0 (MR = 1.014 95% CI 0.995–1.034 p 0.147) and nAbsCP (MR = 0.918 95% CI 0.780–1.080, p 0.300) had no association with duration of mechanical ventilation in our study. However, CCP transfusion after 10 days of symptom onset (Mean ratio (MR) 0.737; 95% CI 0.611–0.890, p = 0.001) was associated with shorter periods of ventilation‐free days, that is, longer intubation periods. Patients who received CCP transfusions before 10 days of symptom onset had 26.3% shorter intubation periods compared with patients transfused after 10 days of illness onset. Male sex (MR = 0.776, 95% CI 0.721–0.836, p < 0.001), SOFA score on day 0 (MR = 0.805 95% CI 0.747–0.867, p < 0.001) and diabetes (MR 0.722, 95% CI 0.592–0.879, p 0.001) were also statistically associated with shorter periods of mechanical ventilation‐free days. Conversely, the use of other supportive therapies was associated with longer ventilation‐free days periods (MR = 1.136, 95% CI 1.021–1.263, p 0.019). Age, weight, ABO group and presence of comorbidities also failed to provide any associations with length of mechanical ventilation support.
TABLE 2.
Number of ventilation‐free days over the first 30 days of the study
| Univariate analysis | Univariate analysis | |||
|---|---|---|---|---|
| Mean ratio (95% CI) | p Value | Mean ratio (95% CI) | p Value | |
| Sex | ||||
| Female | Reference | Reference | ||
| Male | 1.002 (0.965; 1.042) | 0.904 | 0.776 (0.721; 0.836) | <0.001 |
| Age (years) | 0.991 (0.989; 0.993) | <0.001 | 0.998 (0.992; 1.004) | 0.501 |
| Body weight (kg) | 1.001 (1.000; 1.002) | 0.004 | 1.002 (0.996; 1.009) | 0.469 |
| ABO | ||||
| B/O | Reference | Reference | ||
| A/AB | 0.989 (0.878; 1.114) | 0.855 | 1.324 (0.799; 2.194) | 0.276 |
| NAbs P0a | 1.017 (1.011; 1.022) | <0.001 | 1.014 (0.995; 1.034) | 0.147 |
| NAbs P5a | 0.994 (0.979; 1.010) | 0.454 | ||
| NAbsCP | 1.024 (0.932; 1.125) | 0.621 | 0.918 (0.780; 1.080) | 0.300 |
| SOFA score D0 | 0.845 (0.779; 0.916) | <0.001 | 0.805 (0.747; 0.867) | <0.001 |
| Time from symptom onset prior to CCP | ||||
| Up to 10 days | Reference | Reference | ||
| >10 days | 1.002 (0.806; 1.245) | 0.987 | 0.737 (0.611; 0.890) | 0.001 |
| Use of other COVID‐19 supportive therapies | ||||
| No | Reference | Reference | ||
| Yes | 0.808 (0.785; 0.831) | <0.001 | 1.136 (1.021; 1.263) | 0.019 |
| Comorbidities | ||||
| No | Reference | Reference | ||
| Yes | 0.738 (0.675; 0.807) | <0.001 | 1.088 (0.742; 1.593) | 0.667 |
| Hypertension | ||||
| No | Reference | Reference | ||
| Yes | 1.015 (0.846; 1.218) | 0.873 | 1.067 (0.861; 1.323) | 0.551 |
| Diabetes | ||||
| No | Reference | Reference | ||
| Yes | 0.782 (0.670; 0.913) | 0.002 | 0.722 (0.592; 0.879) | 0.001 |
500 units increase; nAbsP0, Neutralizing antibody titers from patient samples before convalescent plasma transfusion; nAbsP5, nAb titers from patient samples collected at day 5 after CCP transfusion; nAbsCP, geometric mean titer of CCP units transfused.
Note: Bold values are statistically significant, that is, p< 0.05.
Table 3 shows predictive variables for ICU length of stay (ICU LOS). We considered number of ICU free‐ days over the first 30 days of CCP transfusion for all patients on the multivariate GEE model. Neither level of patient nAbs titer prior to transfusion (nAbs P0) (MR 1.020, 95% CI 0.998–1.043, p 0.069), neither nAbs from units transfused (nAbsCP) (MR 0.981 95% CI 0.826–1.166, p 0.829) showed statistical significance to the number of ICU‐free days. Administration of CCP transfusion after 10 days of disease onset was associated with an estimated 31.7% shorter ICU free‐days period, that is, earlier CCP transfusions were associated with a reduction on ICU LOS (MR = 0.683, 95% CI 0.575–0.810, p < 0.001). Age (MR = 0.990, 95% CI 0.987–0.992, p < 0.001), SOFA score on day 0 (MR = 0.817 95% CI 0.747–0.894 p < 0.001) and diabetes (MR 0.808 95% CI 0.666–0.981 p < 0.031) were also associated with shorter ICU‐ free days. Sex, ABO group, exposure to other supportive therapies and comorbidities had no evidence of association with the ICU LOS.
TABLE 3.
Number of ICU‐free days over the first 30 days of the study
| Univariate models | Multivariate model | |||
|---|---|---|---|---|
| Mean ratio (95% CI) | p Value | Mean ratio (95% CI) | p Value | |
| Sex | ||||
| Female | Reference | Reference | ||
| Male | 1.258 (1.105; 1.434) | 0.001 | 0.904 (0.710; 1.152) | 0.416 |
| Age (years) | 0.983 (0.981; 0.985) | <0.001 | 0.990 (0.987; 0.992) | <0.001 |
| Body weight (kg) | 1.004 (1.002; 1.006) | <0.001 | 0.997 (0.993; 1.002) | 0.270 |
| ABO | ||||
| B/O | Reference | Reference | ||
| A/AB | 0.955 (0.848; 1.075) | 0.446 | 1.124 (0.815; 1.552) | 0.476 |
| NAbs P0a | 1.024 (1.021; 1.027) | <0.001 | 1.020 (0.998; 1.043) | 0.069 |
| Nabs P5a | 1.008 (0.991; 1.026) | 0.366 | ||
| NAbs CPa | 1.056 (0.932; 1.197) | 0.394 | 0.981 (0.826; 1.166) | 0.829 |
| SOFA D0 | 0.807 (0.717; 0.907) | <0.001 | 0.817 (0.747; 0.894) | <0.001 |
| Time from symptom onset prior to CCP | ||||
| Up to 10 days | Reference | Reference | ||
| >10 days | 0.928 (0.783; 1.099) | 0.385 | 0.683 (0.575; 0.810) | <0.001 |
| Use of other COVID‐19 supportive therapies | ||||
| No | Reference | Reference | ||
| Yes | 0.649 (0.534; 0.789) | <0.001 | 0.986 (0.854; 1.138) | 0.846 |
| Comorbidities | ||||
| No | Reference | Reference | ||
| Yes | 0.590 (0.489; 0.712) | <0.001 | 0.802 (0.572; 1.126) | 0.202 |
| Hypertension | ||||
| No | Reference | Reference | ||
| Yes | 0.970 (0.700; 1.345) | 0.857 | 1.258 (1.158; 1.368) | <0.001 |
| Diabetes | ||||
| No | Reference | Reference | ||
| Yes | 0.786 (0.635; 0.973) | 0.027 | 0.808 (0.666; 0.981) | 0.031 |
500 units increase; nAbsP0: Neutralizing antibody titers from patient samples before convalescent plasma transfusion; nAbsP5, nAb titers from patient samples collected at day 5 after CCP transfusion; nAbsCP, geometric mean titer of CCP units transfused.
Note: Bold values are statistically significant, that is, p< 0.05.
Among the non‐intubated patients prior to enrollment, we sought to determine predictive variables for progression to mechanical ventilation (Table 4). Seventy patients were not on mechanical ventilation before CCP transfusion. Of these, 18 (25.7%) required mechanical ventilation. GEE models were performed to identify associated variables. Also, multivariate analysis showed that transfusion of CCP after 10 days of symptom onset doubles the odds of progression to mechanical ventilation (OR = 2.045 95% CI 1.495–2.798, p < 0.001). Nevertheless, nAbs titers of patients prior to enrolment (nAbs P0) were associated with 20% increased odds of progression to mechanical ventilation (OR = 1.201 95% CI 1.130–1.275, p < 0.001). Age, body weight, and the use of other supportive therapies were also associated with the progression to mechanical ventilation. No other associations were identified.
TABLE 4.
Progression to mechanical ventilation
| Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|
| Odds ratio (95% CI) | p Value | Odds ratio (95% CI) | p Value | |
| Sex | ||||
| Female | Reference | Reference | ||
| Male | 0.469 (0.389; 0.566) | <0.001 | 0.801 (0.326; 1.969) | 0.629 |
| Age (years) | 1.032 (1.008; 1.056) | 0.010 | 1.056 (1.014; 1.099) | 0.008 |
| Body weight (kg) | 0.998 (0.960; 1.038) | 0.937 | 1.044 (1.008; 1.082) | 0.018 |
| ABO | ||||
| B/O | Reference | Reference | ||
| A/AB | 1.138 (0.634; 2.040) | 0.665 | 0.483 (0.065; 3.594) | 0.477 |
| NAbs P0a | 0.974 (0.861; 1.103) | 0.682 | 1.201 (1.130; 1.275) | <0.001 |
| NAbs P5a | 1.099 (1.022; 1.182) | 0.011 | ||
| NAbs CPa | 1.172 (1.002; 1.371) | 0.047 | 1.420 (0.889; 2.267) | 0.142 |
| SOFA D0 | 1.553 (0.847; 2.847) | 0.155 | 1.646 (0.957; 2.830) | 0.072 |
| Time from symptom onset prior to CCP transfusion | ||||
| Up to 10 days | Reference | Reference | ||
| >10 days | 1.184 (0.753; 1.863) | 0.465 | 2.045 (1.495; 2.798) | <0.001 |
| Use of other supportive therapies | ||||
| No | Reference | Reference | ||
| Yes | 28.78 (3.22; 257.19) | 0.003 | 7.15 (1.89; 27.12) | 0.004 |
| Comorbidities | ||||
| No | Reference | Reference | ||
| Yes | 2.452 (0.403; 14.910) | 0.330 | 1.081 (0.024; 49.274) | 0.968 |
| Hypertension | ||||
| No | Reference | Reference | ||
| Yes | 0.785 (0.578; 1.065) | 0.119 | 0.218 (0.038; 1.250) | 0.087 |
| Diabetes | ||||
| No | Reference | Reference | ||
| Yes | 2.583 (0.550; 12.123) | 0.229 | 3.451 (0.743; 16.028) | 0.114 |
500 units increase; nAbsP0: Neutralizing antibody titers from patient samples before convalescent plasma transfusion; nAbsP5, nAb titers from patient samples collected at day 5 after CCP transfusion; nAbsCP, geometric mean titer of CCP units transfused.
Note: Bold values are statistically significant, that is, p< 0.05.
4. DISCUSSION
We report our experience with CCP transfusion in 102 critically ill patients with COVID‐19. The findings suggest that CCP is safe and no major hazard effects were observed in this group of severe patients. CCP transfusion is based on the fact that it provides nAbs that are able to reduce the viral burden and prevent systemic manifestations in susceptible patients.20 Although several observational studies reported data favoring the safety of convalescent plasma,21, 22, 23 a systematic review analyzing CCP and hyperimmune immunoglobulin failed to provide compelling evidence regarding the clinical efficacy of both products.24 In addition, randomized clinical trials showed conflicting evidence regarding optimal intervention strategies. While no benefit on mortality1, 25, 26 or clinical improvement27 was demonstrated with CCP transfusions within 1025, 26, 27 to 30 days of symptom onset,1 a post‐hoc analysis highlighted potential benefits on subspecific groups of patients transfused with high titer nAbs CCP units,14 suggesting the need for further research on this field.
We analyzed predictive factors for clinical improvement on day 14. Forty‐six patients achieved clinical improvement and the multivariate analysis showed that both nAbs T (p < 0.001) and nAbs P0 (p = 0.028) were statistically associated with higher odds of clinical improvement. In each −500 unit increase in nAbs CP, the odds of clinical improvement raised by 62%. Our findings are in accordance with a retrospective study by Joyner et al.14 that also emphasized a dose‐dependent benefit of high titer CCP units. Plasma transfusion with higher SARS‐CoV‐2 IgG antibody levels was associated with lower risk of death than transfusion of plasma with lower antibody levels on non‐intubated patients. Similar findings reported by Libster et al15 showed that early administration of high‐titer CCP reduced progression to severe respiratory disease in mildly ill infected older adults. Both studies suggested that the selection of high‐titer units and early intervention would provide better outcomes.
In our analysis, considering ICU LOS and length of mechanical ventilation, neither nAbs P0, nor nAbsCP had an impact on the reduction of ICU‐free days or ventilation‐free days. Our findings agree with unpublished observations from a large randomized study that showed in an interim analysis that there is a very low probability (2.2%) that CCP would reduce the number of days requiring ICU support or death.28 Moreover, another randomized controlled trial that evaluated 5795 patients showed no benefit of high titer CCP transfusion in 28 day mortality (p = 0.95), or progression to invasive mechanical ventilation (p = 0.79) in hospitalized patients with COVID‐1929 (2021). Their median time from symptom onset to randomization was 9 days (IQR 6–12), which was similar to our intervention. Other studies1, 27, 30 have shown that CCP transfusions late in the course of the disease might not add any benefit. Our hypothesis is that CCP transfusions may alleviate systemic inflammatory response syndrome in early stages of COVID‐19, when viral injury can be restraint by CCP passive immunotherapy. The ICU admission and initiation of mechanical ventilation support tend to be late events in the course of the disease, when CCP therapy seems to have no impact on the outcomes. Hence, it is unlikely that critically ill intubated patients would benefit from this intervention.
We also observed that the severity of the disease at day 0 (SOFA D0), age and diabetes were associated with increases in the ICU LOS. Male sex, diabetes and SOFA D0 were associated with longer periods of mechanical ventilation. Our data partially agree with Gamberini et al, who demonstrated that age, SOFA score at ICU admission, PaO2/FiO2, renal and cardiovascular complications and late‐onset ventilation acquired pneumonia were all independent risk factors for prolonged mechanical ventilation in patients with COVID‐19.31 Differences found may be due to the low number of patients analyzed and the different methodologies implemented.
Among the non‐intubated patients, we investigated factors associated with progression to mechanical ventilation. Despite the limited number of patients analyzed (18 had progressed to mechanical ventilation among 70 who were not intubated prior to enrollment), the multivariate analysis showed that nAbs CP (p = 0.142) had no impact on the progression to mechanical ventilation. Similarly, data from Horby et al also found no difference in progression to mechanical ventilation among patients allocated to CCP transfusions when compared with usual care.31 We also observed that nAbs P0 were associated with higher odds (OR = 2.045 95% CI 1.495–2.798, p < 0.001) of progression to mechanical ventilation. It is important to consider that patients with more severe disease develop solid neutralizing antibody response,32 so that higher titer nAbs are expected in these scenarios and such finding might only be an association with no causative relation. Age (p 0.008), body weight (p 0.018), the use of other supportive therapies (p 0.004) were also statistically associated with higher odds of progression to mechanical ventilation.
Our findings also suggest that the timely administration of CCP is relevant for clinical outcomes. Despite the potential bias of such analysis without a control group, we found that administration of CCP after 10 days of symptom onset was associated with increases in the ICU LOS and duration of mechanical ventilation in a statistically significant manner. In addition, CCP transfusion after 10 days of disease onset was associated with higher odds of progression to mechanical ventilation. Antibody responses to SARS‐CoV‐2 seem to appear between 2 and 3 weeks after initiation of symptoms33 and nAbs specifically reach their peak within 10–15 days after the disease onset.34 In our study, the median time from onset of illness to CCP transfusion was 10 days [IQR 8.0–13.0]. Timely intervention with CCP transfusion has been covered by observational and randomized clinical trials.2, 14, 15, 22, 30, 35, 36 Our findings reinforce the fact that earlier initiation of passive immunotherapy might provide better outcomes. These data may be interpreted with a great deal of caution, since no control group was used in the analysis of the design of our study, which precludes definitive conclusions.
Our study had some limitations. First, this is a one‐arm observational study. Second, nAbs P0 were not available before CCP transfusion. It has been described that a significant proportion of patients already have high nAbs at hospital admission.25 Virus neutralization (VN) screening before CCP transfusion could be useful to identify individuals who could benefit from this passive therapy. Despite VN assays are considered the gold standard to measure antiviral activity of antibodies, some limitations (long turnaround times, specific biosafety laboratory environment and highly trained personnel) limit their usage in clinical practice. Recently, serological methods using IgG antibodies (anti‐spike ectodomain and anti‐receptor binding domain) are a plausible alternative for overcoming the aforementioned logistics restraints of VN assays, since strong correlations between levels of binding antibodies and VN titers were established.37, 38 Our findings that both nAbsCP and nAbsP0 were associated with higher odds of clinical improvement by day 14 reinforce the relevance of patients' nAbs baseline evaluation and the selection of high titer units for CCP transfusion. Further studies may incorporate binding antibodies or VN titer assays or for CCP donor qualification and the baseline status of patients eligible to this therapy.
We underscore that the potential efficacy of CCP transfusion depends on the specific nAbs directed against the infecting virus variant in the recipient. Reductions in neutralizability capacity of nAbs39, 40 have been recently reported after new variants were described ((B1.1.7, B1.325, and P.1).41, 42, 43 So far, no changes in the efficacy of CCP transfusion have been observed before and after the new variant in the United Kingdom.31 More studies are needed in order to clarify this point.
Our analysis suggests that neutralizing antibodies from CCP units and circulating nAbs produced by the patient prior to intervention are associated with improvements in clinical outcomes by day 14. The higher the nAbs, the higher the odds of clinical improvement. These results reinforce the fact that nAbs should be used to qualify eligible donors and characterize the baseline status of COVID‐19 patients. We consider these results as useful parameters to guide future CPP transfusion strategies against COVID‐19.
CONFLICT OF INTEREST
The authors have disclosed no conflicts of interest.
AUTHOR CONTRIBUTIONS
Conceptualization: Jose M. Kutner, Ana Paula H. Yokoyama, Carolina Bonet‐Bub, Silvano Wendel, Roberta M. Fachini; investigation: Jose M. Kutner, Ana Paula H. Yokoyama, Carolina Bonet‐Bub, Silvano Wendel, Ana Paula F Dametto, Roberta M. Fachini, Gabriela Candelaria, Patrícia Scuracchio, Ruth Achkar, Mirian Dal Bem, Rafael Rahal Guaragna Machado, Danielle Bastos Araujo, Edison Luiz Durigon, Fernando Blumm, Valeria F Dutra, Araci M Sakashita, Rita Fontão‐WEndel, Nelson Hamerschlak, Murillo Santucci Cesar Assunção, Victor Nudelman, Laerte Pastore, João R R Pinho, Roberto Kalil Filho, Mariane T Amano, Esper Kallás, Alfredo Salim Helito, Carlos Roberto Ribeiro de Carvalho, Anamaria A Camargo; formal analysis: Jose M. Kutner, Ana Paula H. Yokoyama, Silvano Wendel, Carolina Bonet‐Bub, l, Roberta M. Fachini, Alexandre R. Marra; resources: Luiz F. L. Reis, Luiz V. Rizzo, writing: Ana Paula H. Yokoyama, Silvano Wendel, Carolina Bonet‐Bub, Jose M Kutner.
ACKNOWLEDGMENTS
The authors would like to thank ES Victor for the statistical analysis and advise, also the following physicians for referring their patients to this study: ALP Albuquerque, AC Nicodemo, D Deheinzelin, E Negri, LFL Carvalho, LF Cardoso, MAR Cuoco, P Seferian‐Júnior, R Kairalla, T Pfiffer, Y Novis, H Souza, T Zinsly, A Pacheco, G Johanson, FT Moreira, S Santoro, B Normandia, RM Carraro, E Meyer, A Lichtentstein, F Bacal, M Amar, CAF Santos, D Goldenberg, A Jaime, M Rodrigues‐Junior, S Filizola, H Bacha, H Bogossian, C Hoelz, M Erlichman, E Pfefferman, R Kondo, MH Kuwakino, R Peres, GL Bub, DS Levi, and FG Menezes. Funding was provided by CAPES‐Coordenação de Aperfeiçoamento de Pessoal de Nível Superior CAPES[88887.131387/2016–00 to DBA]; Fundação de Amparo à Pesquisa do Estado de São Paulo [2016/20045–7 and 2020/06409–1 to ELD]; and the initiative “Todos Pela Saúde”—Fundação Itaú para Educação e Cultura.
Yokoyama APH, Wendel S, Bonet‐Bub C, et al. COVID‐19 convalescent plasma cohort study: Evaluation of the association between both donor and recipient neutralizing antibody titers and patient outcomes. Transfusion. 2021;61:2295–2306. 10.1111/trf.16573
Funding information Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Grant/Award Number: 88887.131387/2016‐00; Fundação de Amparo à Pesquisa do Estado de São Paulo, Grant/Award Numbers: 2016/20045‐7, 2020/06409‐1; Todos pela Saúde ‐ Fundação Itaú para Educação e Cultura
REFERENCES
- 1.Li L, Zhang W, Hu Y, et al. Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life‐threatening COVID‐19: a randomized clinical trial. JAMA. 2020;324:460–470. 10.1001/jama.2020.10044 Erratum in: JAMA. 2020;324(5):519. PMID: 32492084; PMCID: PMC7270883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu STH, Lin H‐M, Baine I, et al. Convalescent plasma treatment of severe COVID‐19: a propensity score‐matched control study. Nat Med. 2020;26:1708‐1713. 10.1038/s41591-020-1088-9 [DOI] [PubMed] [Google Scholar]
- 3.Joyner MJ, Wright RS, Fairweather D, et al. Early safety indicators of COVID‐19 convalescent plasma in 5000 patients. J Clin Invest. 2020;2020:140200. 10.1172/JCI140200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cao B, Wang Y, Wen D, et al. A trial of Lopinavir‐ritonavir in adults hospitalized with severe Covid‐19. N Engl J Med. 2020;382:1787–1799. 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beigel JH, Tomashek KM, Dodd LE, et al. ACTT‐1 study group members. Remdesivir for the treatment of Covid‐19 ‐ final report. N Engl J Med. 2020;383:1813–1826. 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hung IF, Lung KC, Tso EY, et al. Triple combination of interferon beta‐1b, lopinavir‐ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID‐19: an open‐label, randomised, phase 2 trial. Lancet. 2020;395:1695–1704. 10.1016/S0140-6736(20)31042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Luke TC, Kilbane EM, Jackson JL, Hoffman SL. Meta‐analysis: convalescent blood products for Spanish influenza pneumonia: a future H5N1 treatment? Ann Intern Med. 2006;145:599–609. 10.7326/0003-4819-145-8-200610170-00139. [DOI] [PubMed] [Google Scholar]
- 8.Cheng Y, Wong R, Soo YO, et al. Use of convalescent plasma therapy in SARS patients in Hong Kong. Eur J Clin Microbiol Infect Dis. 2005;24:44–46. 10.1007/s10096-004-1271-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Devasenapathy N, Ye Z, Loeb M, et al. Efficacy and safety of convalescent plasma for severe COVID‐19 based on evidence in other severe respiratory viral infections: a systematic review and meta‐analysis. CMAJ. 2020;192:E745–E755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Casadevall A, Pirofski LA. The convalescent sera option for containing COVID‐19. J Clin Invest. 2020;130:1545–1548. 10.1172/JCI138003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rojas M, Rodríguez Y, Monsalve DM, et al. Convalescent plasma in Covid‐19: possible mechanisms of action. Autoimmun Rev. 2020;19:102554. 10.1016/j.autrev.2020.102554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhou G, Zhao Q. Perspectives on therapeutic neutralizing antibodies against the novel coronavirus SARS‐CoV‐2. Int J Biol. 2020;16:1718–1723. 10.7150/ijbs.45123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van Griensven J, Edwards T, de Lamballerie X, et al. Evaluation of convalescent 377 plasma for Ebola virus disease in Guinea. N Engl J Med. 2016;374:33–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Joyner MJ, Carter RE, Senefeld JE, et al. Convalescent plasma antibody levels and the risk of death from Covid‐19. N Engl J Med. 2021;384(11):1015‐1027. 10.1056/NEJMoa2031893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Libster R, Pérez Marc G, Wappner D, et al. Early high‐titer plasma therapy to prevent severe Covid‐19 in older adults. N Engl J Med. 2021;384:610–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wendel S, Kutner JM, Machado R, et al. Screening for SARS‐CoV‐2 antibodies in convalescent plasma in Brazil: preliminary lessons from a voluntary convalescent donor program. Transfusion. 2020;60:2938‐2951. 10.1111/trf.16065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Araujo DB, Machado RRG, Amgarten DE, et al. SARS‐CoV‐2 isolation from the first reported patients in Brazil and establishment of a coordinated task network. Mem Inst Oswaldo Cruz. 2020;115:e200342. 10.1590/0074-02760200342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.COVID‐19 Therapeutic Trial Synopsis © World Health Organization; 2020. https://www.who.int/publications/i/item/covid-19-therapeutic-trial-synopsis.
- 19.R Foundation for Statistical Computing . R: A Language and Environment for Statistical Computing; 2018. https://www.R-project.org/.
- 20.Imai M, Iwatsuki‐Horimoto K, Hatta M, et al. Syrian hamsters as a small animal model for SARS‐CoV‐2 infection and countermeasure development. Proc Natl Acad Sci U S A. 2020;117:16587–16595. 10.1073/pnas.2009799117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Joyner MJ, Bruno KA, Klassen SA, et al. Safety update: COVID‐19 convalescent plasma in 20,000 hospitalized patients. Mayo Clin Proc. 2020. Sep;95:1888–1897. 10.1016/j.mayocp.2020.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duan K, Liu B, Li C, et al. The feasibility of convalescent plasma therapy in severe COVID‐19 patients: a pilot study. MedRxiv. 2020. [Preprint] 10.1101/2020.03.16.20036145 [DOI] [Google Scholar]
- 23.Rajendran K, Krishnasamy N, Rangarajan J, et al. Convalescent plasma transfusion for the treatment of COVID‐19: systematic review. J Med Virol. 2020;92:1475–1483. 10.1002/jmv.25961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chai KL, Valk SJ, Piechotta V, et al. Convalescent plasma or hyperimmune immunoglobulin for people with COVID‐19: a living systematic review. Cochrane Database Syst Rev. 2020;10:CD013600. 10.1002/14651858.CD013600.pub3. [DOI] [PubMed] [Google Scholar]
- 25.Gharbharan JCCE, Geurtsvankessel C, et al. Convalescent plasma for COVID‐19. A randomized clinical trial. medRxiv 2020:2020.07.01.20139857. [Preprint.] 10.1101/2020.07.01.20139857v1 [DOI] [Google Scholar]
- 26.Agarwal A, Mukherjee A, Kumar G, PLACID Trial Collaborators , et al. Convalescent plasma in the management of moderate covid‐19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID trial). BMJ. 2020;371:m3939. 10.1136/bmj.m3939 Erratum in: BMJ. 2020 Nov 3;371:m4232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Simonovich VA, Burgos Pratx LD, PlasmAr Study Group , et al. A randomized trial of convalescent plasma in Covid‐19 severe pneumonia. N Engl J Med. 2021;384(7):619‐629. 10.1056/NEJMoa2031304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wise J. Covid‐19: convalescent plasma may cut deaths in patients not on ventilation, study indicates. BMJ. 2021;372:n130. 10.1136/bmj.n130. [DOI] [PubMed] [Google Scholar]
- 29.The RECOVERY Collaborative Group . Convalescent plasma in patients admitted to hospital with COVID‐19 (RECOVERY): a randomized, controlled, opne‐label, platform trial. Lancet. 2021;397(10289):2049‐2059. 10.1016/S0140-6736(21)00897-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Salazar E, Christensen PA, Graviss EA, et al. Treatment of coronavirus disease 2019 patients with convalescent plasma reveals a signal of significantly decreased mortality. Am J Pathol. 2020;190:2290–2303. 10.1016/j.ajpath.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gamberini L, Tonetti T, Spadaro S, ICU‐RER COVID‐19 Collaboration , et al. Factors influencing liberation from mechanical ventilation in coronavirus disease 2019: multicenter observational study in fifteen Italian ICUs. J Intensive Care. 2020;8:80. 10.1186/s40560-020-00499-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang X, Guo X, Xin Q, et al. Neutralizing antibody responses to severe acute respiratory syndrome coronavirus 2 in coronavirus disease 2019 inpatients and convalescent patients. Clin Infect Dis. 2020;71(10):2688‐2694. 10.1093/cid/ciaa721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Callow KA, Parry HF, Sergeant M, Tyrrell DA. The time course of the immune response to experimental coronavirus infection of man. Epidemiol Infect. 1990;105:435–446. 10.1017/s0950268800048019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wu F, Wang AJ, Liu M, et al. Neutralizing antibody responses to SARS‐CoV‐2 in a COVID‐19 recovered patient cohort and their implications. medRxiv. 2020. [Preprint] 10.1101/2020.03.30.2004736. [DOI] [Google Scholar]
- 35.Mair‐Jenkins J, Saavedra‐Campos M, Baillie JK, et al. The effectiveness of convalescent plasma and hyperimmune immunoglobulin for the treatment of severe acute respiratory infections of viral etiology: a systematic review and exploratory meta‐analysis. J Infect Dis. 2015;211:80–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hung IFN, To KKW , Lee CK, et al. Hyperimmune IV immunoglobulin treatment: a multicenter double‐blind randomized controlled trial for patients with severe 2009 influenza a(H1N1) infection. Chest. 2013;144:464–473. [DOI] [PubMed] [Google Scholar]
- 37.Salazar E, Kuchipudi SV, Christensen PA, et al. Convalescent plasma anti‐SARSCoV‐2 spike protein ectodomain and receptor‐binding domain IgG correlate with virus neutralization. J Clin Invest. 2020;130:6728–6738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Luchsinger LL, Ransegnola BP, Jin DK, et al. Serological assays estimate highly variable SARS‐CoV‐2 neutralizing antibody activity in recovered COVID‐19 patients. J Clin Microbiol. 2020;58:e02005–e02020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Supasa P, Zhou D, Dejnirattisai W, et al. Reduced neutralization of SARS‐CoV‐2 619 B.1.1.7 variant by convalescent and vaccine sera. Cell. 2021;184(8):2201‐2211.e7. 10.1016/j.cell.2021.02.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Souza WM, Amorim MR, Sesti‐Costa R et al. Levels of SARS‐CoV‐2 lineage P.1 neutralization by antibodies elicited after natural infection and vaccination. [Preprint] https://ssrn.com/abstract=3793486.
- 41.Tegally H, Wilkinson E, Giovanetti M, et al. Emergence and rapid spread of a new severe acute respiratory syndrome‐related coronavirus 2(SARS‐CoV‐2) lineage with multiple spike mutations in South Africa. medRxiv. 2020. [Preprint] 10.1101/2020.12.21.20248640 [DOI] [Google Scholar]
- 42.Volz E, Mishra S, Chand M, et al. Transmission of 413 SARS‐CoV‐2 lineage B.1.1.7 in England: insights from linking epidemiological and genetic data. medRxiv. 2021;414:2020.12.30.20249034. [Google Scholar]
- 43.Faria NR, Claro I, Candido DS, Franco LAM, Andrade PS et al., Genomic characterisation 434 of an emergent SARS‐CoV‐2 lineage in Manaus: preliminary findings. Virological.org.


