Abstract
There is an increasing recognition of the strong links between the gut microbiome and the brain, and there is persuasive evidence that the gut microbiome plays a role in a variety of physiological processes in the central nervous system. This review summarizes findings that gut microbial composition alterations are linked to hippocampal neurogenesis, as well as the possible mechanisms of action; the existing literature suggests that microbiota influence neurogenic processes, which can result in neurological disorders. We consider this evidence from the perspectives of neuroinflammation, microbial-derived metabolites, neurotrophins, and neurotransmitters. Based on the existing research, we propose that the administration of probiotics can normalize the gut microbiome. This could therefore also represent a promising treatment strategy to counteract neurological impairment.
Key Words: gut microbiota, microbial-derived metabolites, microbiota-gut-brain axis, neurogenesis, neuroinflammation, neurological diseases, neurotrophins, probiotics
Introduction
A myriad of microorganisms reside within our body, including bacteria, fungi, parasites, and viruses. In particular, the gastrointestinal tract harbors approximately 95% of the human microbiome, and is home to more than 100 trillion bacteria (Qin et al., 2010; Bhattacharjee and Lukiw, 2013), which are collectively referred to as gut microbiota. The process of long-term symbiosis and co-evolution has resulted in a reciprocal communication between gut microbiota and the host brain; this communication occurs via the microbiota-gut-brain axis, which involves immunological, endocrine, neurological, and metabolic signaling pathways (Rhee et al., 2009; Cryan and Dinan, 2012; Janssen and Kersten, 2015; Mayer et al., 2015; Baizabal-Carvallo, 2021). In recent years, an increasing body of research has focused on the topic of the microbiota-gut-brain axis. Repeatedly, gut microbiota have been reported to participate in neurological diseases, including Parkinson’s disease (PD) (Scheperjans et al., 2015; Unger et al., 2016), Alzheimer’s disease (AD) (Vogt et al., 2017), multiple sclerosis (MS) (Kadowaki and Quintana, 2020), and autism spectrum disorder (ASD) (Liu et al., 2019). Gut microbiota have also been linked with age-related health declines that include physiological, psychological, and immunological parameters (Claesson et al., 2012) and stress-related disorders, such as depression, anxiety, and cognitive impairment (Mayer et al., 2014; Fröhlich et al., 2016). All the above-mentioned central nervous system (CNS) disorders are thought to involve pathological processes that include a decline in neurogenesis (Gage and Temple, 2013; Moreno-Jiménez et al., 2019). In this review, we review evidence of the involvement of the gut microbiome in the modulation of neurogenesis and related neurological disorders, and propose microbiota modulation as a potential therapy to rescue neurogenic decline.
Search Strategy and Selection Criteria
The studies used in this review were retrieved by replicating the search terms of Cavallucci et al. (2020). An electronic search of the PubMed database of literature on the gut microbiome and neurogenesis published from 2004 to 2020 was performed using the following search terms: gut microbiota and nervous system. The studies were further screened, and were only eligible for inclusion if the terms “neurogenesis”, “microbiota-gut-brain axis”, “neuroinflammation”, and “neurological diseases” were present in the title and/or abstract.
Neurogenesis: Implications for the Nervous System and Influencing Factors
Neurogenesis refers to the generation of new neurons by the differentiation of neural stem cells and maturation of neural progenitor cells (Gage, 2019; Cosacak et al., 2020). Albeit mainly quiescent, neurogenesis persists throughout adulthood in the mammalian brain. The subventricular zone of the lateral ventricles and the subgranular zone of the dentate gyrus in the hippocampus have been widely identified as neurogenic areas (Zhao et al., 2008).
It is generally accepted that new neurons are produced in the hippocampus of adult mammals, and that integration of these new neurons into the existing neural circuitry is critical for information processing and transmission within the nervous system. Moreover, concerning the important role of the hippocampus in sustaining normal memory and cognition (Lazarov and Hollands, 2016), it is possible that increased neurogenesis leads to a better cognitive performance, while perturbation of neurogenesis contributes to neurological impairments. Indeed, one study revealed that exercise increased the number of newborn neurons in aged mice and was associated with enhanced learning performance, which indicates that there is a strong relationship between physical activity, neurogenesis, and cognition (van Praag et al., 2005). Notably, Cavallucci et al. (2020) reported that excessive calorie intake accelerated the age-dependent decline of neurogenesis, while calorie restriction and physical exercise had the opposite effect; the authors suggested that these effects could be mediated by gut microbiota. In addition to extrinsic factors (i.e. exercise and diet), intrinsic factors, such as inflammation, hormones, and neural factors, have also been found to participate in the modulation of neurogenic processes (Zhao et al., 2008). Namely, inflammation regulates adult neurogenesis through activation of the immune system, which supports the CNS’s normal repair mechanisms and defense against injury; hormones can modulate adult neurogenesis under particular conditions, and this is partly the result of changes in the composition of blood-derived substances; and neural factors can control the proliferation of neural stem cells and play an important role in regulating synaptic plasticity, survival, and differentiation. Together, these intrinsic factors can influence both cognitive improvements and the development of neurological disorders.
Gut Microbiota: The Link with Neurogenesis and Potential Mechanisms of Action
Accumulating evidence supports the idea that gut microbiota can influence the onset and modulation of neurogenesis (Cerdó et al., 2020). Such studies have been at least partly inspired by previous findings of a pathogenic role of gut microbiota in psychiatric and neurodegenerative diseases (Fung et al., 2017). One study revealed that, compared with conventionally raised mice, germ-free (GF) mice exhibited increased adult hippocampal neurogenesis, as revealed by more proliferating cells labeled with bromo-deoxyuridine; notably, microbial colonization of GF mice at 3 weeks of age did not induce changes in adult hippocampal neurogenesis, which suggests that there is a critical 3-week window within which microbial colonization can affect adult hippocampal neurogenesis (Ogbonnaya et al., 2015). Butyric acid is produced by the fermentation of food by intestinal flora. It has been reported that young GM mice exhibit increased hippocampal neurogenesis after receiving fecal microbiota transplanted from old donor mice, concomitant with an enrichment of butyrate-producing microbiota, which was determined using metagenomic analysis (Kundu et al., 2019). In another recent study, intestinal bacteria was found to maintain the enteric nervous system in adult mice through Toll-like receptor 2-induced neurogenesis (Yarandi et al., 2020). Although the mechanisms underlying this link have not yet been elucidated, several potential mechanisms have been proposed, all of which involve an influence of the gut microbiota community on neurogenesis in the CNS.
Neuroinflammation
A well-organized inflammatory response is critical for supporting the ability of the CNS to defend against damage and maintain a normal reparative function. As resident macrophages, microglia are the most abundant innate immune cells in the brain, comprising approximately 10% of all glial cells. Microglia play an essential role in sustaining homeostasis of the CNS; they are responsible for phagocytosis of pathogens and dying cells and the modulation of inflammatory responses, and also prompt the development of functional neural circuits (Lenz and Nelson, 2018). Numerous studies have reported a close relationship between the microbiota community and the homeostatic function of microglia. Erny et al. (2015) observed more morphologically abnormal microglia in the brains of GF mice than in the brains of control mice. Furthermore, microglia in GF mice were not induced into activated phenotypes in response to the intrusion of bacteria and viruses, which highlights the critical role of microbiota in mounting an appropriate immune response in the CNS. Additionally, specific pathogen-free mice experienced antibiotic-induced microbiota depletion and microglia displayed an immature phenotype, albeit with no significant change in the number of microglia. Importantly, administration of short-chain fatty acids (SCFAs), which are a fermentation product of microbiota, normalized microglia functions in GF mice. These results suggest that the gut microbiota is related to the normal structure and function of central microglia.
Microglia have also been implicated in the development of neurodegenerative diseases. Microglia exhibit a highly activated phenotype under conditions of neurodegeneration, wherein they produce and secrete a plethora of neurotoxic pro-inflammatory mediators, including inducible nitric oxide synthase, cytokines and chemokines (e.g. interleukin (IL)-1β, IL-6, tumor necrosis factor-α, IL-8, transforming growth factor-β), and free radicals (Rubio-Perez and Morillas-Ruiz, 2012; Rodríguez-Gómez et al., 2020). Patients with ASD have been found to have more activated microglia than control patients, and the pathogenesis of ASD may be partly attributed to the dysregulation of synaptic pruning by microglia (Koyama and Ikegaya, 2015). Indeed, the involvement of microglia-mediated pruning in Alzheimer’s disease has also been reported (Rajendran and Paolicelli, 2018). Collectively, these studies indicate that microglia play a synaptotoxic role in the promotion of neurological diseases. Importantly, a recent study investigating the microbiota-gut-brain axis provided direct evidence that microbiota can influence neurogenesis by modulating microglia function (Salvo et al., 2020). The authors administered a low dose of dextran sodium sulfate to weaning mice to induce acute colonic inflammation. Weaning mice displayed behavioral deficits alongside alterations in the gut microbial composition and diminished neurogenesis in adulthood, coupled with increased gene expression for pattern recognition receptor and T-helper 17 cell-related cytokines in the hippocampus. Moreover, hippocampal microglia were activated, as revealed by increased gene expression of ionized calcium-binding adapter molecule 1.
In addition to the effects of microglial function, other aspects of the immune system may also be involved in mediating the link between gut microbiota and hippocampal neurogenesis. In one study, post-weaning mice housed alone exhibited anxiety-like behaviors and impaired learning compared with their group-housed counterparts (Dunphy-Doherty et al., 2018). Mice lacking in social interaction had an altered gut microbial composition and reduced neurogenesis compared with group-housed mice, accompanied by reduced IL-6 and IL-10 in the hippocampus. These findings indicate that adverse early-life stress, such as being housed alone, may lead to long-lasting changes in the gut microbiota, and that alterations in the gut microbial composition may affect neurogenesis via mediation of certain inflammatory cytokines, thus playing a key role in the occurrence of behavioral impairments in mice living alone. Finasteride, an inhibitor of the enzyme 5alpha-reductase, can induce depression. Diviccaro et al. (2019) found that administration of finasteride induced a short-term enhancement of neurogenesis, as demonstrated by increased proliferation in the dentate gyrus, and a long-term inhibitory effect on neurogenesis one month after finasteride treatment. Importantly, the reduction in hippocampal neurogenesis was accompanied by depressive-like behaviors, neuroinflammation and gut microbial composition alterations (Figure 1).
Figure 1.
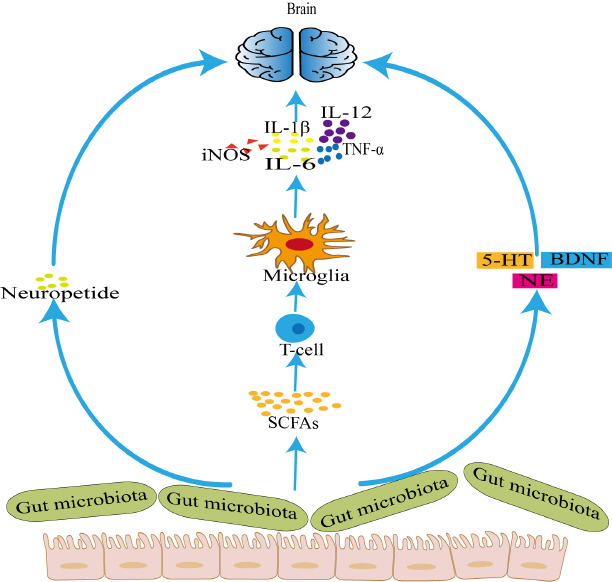
The possible relationship between gut microbiota and neuroinflammation.
The gut microbiota participates in neuroinflammation by acting on neuropeptides, producing 5-HT, NE, and BDNF. The gut microbiota can also affect the production of SCFAs, which in turn affect microglia and promote the release of inflammatory mediators, thus affecting neuroinflammation. 5-HT: 5-Hydroxytryptamine; BDNF: brain-derived neurotrophic factor; IL-12: interleukin-12; IL-1β: interleukin-1β; IL-6: interleukin-6; iNOS: inducible nitric oxide synthase; NE: noradrenaline; SCFAs: short-chain fatty acids; TNF-α: tumor necrosis factor-α.
Microbial-derived metabolites
Of particular interest has been microbial-derived intermediate products, which might play a role in the gut-brain axis. SFCAs are the main products of bacterial fermentation, of which acetate, propionate, and butyrate are the most abundant. SFCAs have immuno-modulatory and anti-inflammatory properties (Sivaprakasam et al., 2016). Moreover, they can modulate the release of neuropeptides, such as serotonin and peptide YY (Filosa et al., 2018), which potentially play key roles in the gut-brain axis. SFCAs exert regulatory effects both in the peripheral nervous system and CNS, owing to the fact that they can pass through the blood–brain barrier and bind to G protein–coupled receptors (Kasubuchi et al., 2015). Although only a small amount of SCFAs reach the CNS, they may have lasting and stable effects on the brain due to the accumulation of SCFAs transported by intestinal flora to the CNS. In addition, SCFAs can affect the integrity of the blood–brain barrier, protect neurons, and indirectly regulate the secretion of 5-hydroxytryptamine by enterochromaffin cells, thus affecting emotion and memory; indeed, SCFAs have been reported to contribute to improved cognitive performance in animal models of neurodegenerative diseases (Stilling et al., 2014). For instance, Ho et al. (2018) reported that SCFAs have an inhibitory effect on the generation of beta-amyloid peptides in Alzheimer’s disease pathogenesis. In addition, Clostridium butyricum, a butyrate-producing probiotic, significantly improved neurological deficits and neuronal degeneration in a mouse model of traumatic brain injury (Li et al., 2018). Furthermore, Yang et al. (2020) found that SCFAs can prompt the growth and differentiation of human neural progenitor cells at physiologically relevant levels. Given that the expression of neurogenesis-related genes ATR, BCL2, BID, CASP8, CDK2, VEGFA, E2F1, FAS, NDN could be influenced by supplementation with SCFAs, these results suggest that SCFAs could regulate early neural system development.
Neurotrophins and neurotransmitters
Brain-derived neurotrophic factor (BDNF) is a neurotrophin that is widely expressed in the brain, and has critical roles in regulating synaptic plasticity, as well as survival and differentiation in the CNS (Ichim et al., 2012). Altered BDNF expression has been reported in several neurological disorders, including Alzheimer’s disease, PD, depression, and other psychiatric disorders (Nagahara and Tuszynski, 2011). O’Sullivan et al. (2011) found that maternal separation caused a marked increase in hippocampal BDNF in a rat model of early-life stress, while Bifidobacterium breve 6330, a probiotic, increased BDNF levels in control animals, which suggests that BDNF might be involved in the regulation of anxiety through microbiome-gut-brain axis. Furthermore, it has been reported that GF mice have altered BDNF expression levels in the hippocampus (Neufeld et al., 2011; Clarke et al., 2013). The vagus nerve is thought to serve as a communication pathway of the microbiome-gut-brain axis. One study found that adult mice subjected to subdiaphragmatic vagotomy had decreased BDNF mRNA expression in the hippocampus, as well as reduced proliferation and survival of neurons in hippocampus (O’Leary et al., 2018), which further suggests that the gut microbiota affects BDNF expression through the vagus nerve, thus mediating the bidirectional communication between the brain and intestine.
Serotonin and its precursor, tryptophan, are important signaling molecules in both the CNS and gastrointestinal tract. Serotonin exerts regulatory effects on a variety of physiological functions, including modulation of stress, anxiety, mood, and cognition (Szapacs et al., 2004; Asan et al., 2013). Furthermore, serotonin plays a critical role in sustaining hippocampal homeostasis and promoting hippocampal neurogenesis (Alenina and Klempin, 2015). Clarke et al. (2013) investigated the role of the microbiome-gut-brain axis in the hippocampal serotonergic system, and observed elevated hippocampal serotonin levels and increased plasma concentrations of tryptophan in male GF mice compared with conventionally raised controls. These results suggested that changes in the gut microbiota could affect CNS neurotransmission, and that regulation of the gut-brain axis is essential for maintaining homeostasis. Moreover, Siopi et al. (2020) found that fecal transfer from chronic mild stress mice to healthy mice impaired the antidepressant and neurogenic effects of fluoxetine, which is a standard selective serotonin reuptake inhibitor that not only promotes proliferation, differentiation, and survival of progenitor cells in the hippocampus, but also influences the plasticity of new neurons generated.
Together, these results highlight the possibility that the gut microbiome influences hippocampal neurogenesis via mediation of the serotonergic system.
Dysbiosis in Neurological Disorders
Considering that the gut microbial community could impact on hippocampal neurogenesis via multiple pathways, as described above, it is necessary to consider its role in the pathogenesis of neurological disorders, which have also been associated with abnormal neurogenesis in the CNS.
Cumulative evidence has confirmed that alterations of the gut microbiome composition are associated with the development of neurodegenerative diseases. Patients with Alzheimer’s disease have been reported to show different fecal microbial patterns to those of control individuals, with decreased abundances of Firmicutes and Actinobacteria and an increased abundance of Bacteroidetes at the phylum level (Vogt et al., 2017). In addition, while findings have not been conclusive, the abundance of Helicobacter pylori is thought to be positively associated with cognitive impairment in Alzheimer’s disease, and the pathogenic effect may be mediated by inflammatory responses (Roubaud-Baudron et al., 2012; Park et al., 2017). The imbalance of gut microbiota can aggravate degenerative changes of the CNS.
The gut microbiome has also gained increasing attention in PD, and its involvement has been strongly implicated in the modulation of this disease. Microbiota analysis has revealed increased Enterobacteria and decreased Prevotella strains in patients with PD (Scheperjans et al., 2015). Another clinical study found that, compared with healthy controls, patients with PD exhibited decreases in a range of butyrate-producing and anti-inflammatory bacterial genera, including Blautia, Coprococcus, and Roseburia, as well as increases in several potentially harmful proinflammatory Proteobacteria of the genus Ralstonia (Keshavarzian et al., 2015). Concordantly, Unger et al. (2016) found that patients with PD exhibited a reduction of the SFCA-producing bacterial family Prevotellaceae and lower SCFA concentrations relative to control patients.
Amyotrophic lateral sclerosis (ALS) is a chronic neurodegenerative disease characterized by a progressive loss of motor neurons and muscle atrophy. An altered gut microbiome composition has been detected in a mouse model of ALS, with a reduced abundance of butyrate-producing bacteria, including Butyrivibrio fibrisolvens and Escherichia coli (Wu et al., 2015). Furthermore, a clinical study by Mazzini et al. (2018) reported variations in the gut microbial composition in patients with ALS, including higher Escherichia coli and Enterobacteria levels and lower Clostridium levels compared with control patients.
Previous work has also revealed a causal link between the gut microbial community and ASD. Children with autism have been reported to have a decreased abundance of butyrate-producing taxa, such as Ruminococcaceae and Eubacterium, accompanied by relative low levels of SCFAs (Liu et al., 2019).
MS is an autoimmune inflammatory neurodegenerative disease in which targeting SCFAs also seems to be a promising strategy. Indeed, reduced levels of SCFA-producing bacteria has been detected in patients with MS (Takewaki et al., 2020). Moreover, administration of SCFA has been associated with a better disease course in an experimental animal model of autoimmune encephalomyelitis (Melbye et al., 2019). Additionally, a high abundance of Archaea and markedly low abundances of Firmicutes and Bacteroidetes have been reported in patients with MS (Tremlett et al., 2017).
Ischemic stroke is caused by focal occlusion or arterial stenosis. Some studies have found evidence for a bidirectional communication between the intestinal axis and brain axis after stroke, and that stroke changes the composition of gut microbiota; in turn, microbiota imbalance can impact immune responses and substantially worsen the prognosis of stroke as a result (Houlden et al., 2016; Liang et al., 2020; Sadler et al., 2020). After stroke, T lymphocytes (especially γδ T-cells) migrate from the Peyer plaque or lamina propria of the small intestine to the brain and/or pia mater; intestinal microflora seem to affect the degree of neuroinflammation after stroke by regulating this transport of intestinal T cells to the brain (Benakis et al., 2016; Cryan et al., 2020). In addition to eliciting a microglia response, stroke can also induce peripheral immune cells to invade the brain parenchyma, which worsens stroke prognosis (Neumann et al., 2015; Selvaraj and Stowe, 2017). Gut microbiota and their metabolite SCFAs play a key role in stroke. SCFAs regulate the activation of microglia and the composition of immune cells, and lymphocytes play a key role in mediating the role of SCFAs in microglia activation. In addition, SCFAs may indirectly mediate post-stroke recovery and neuronal plasticity by regulating the activity of microglia (Sadler et al., 2020). Thus, dysregulated SCFA levels resulting from an imbalanced microbiota composition may impede recovery after stroke.
Possible Gut Microbiome-Based Treatment Strategies for Neurological Disorders
As mentioned above, an abnormal gut microbial composition may be causally link with neurological disorders and abnormal neurogenic processes. Thus, it is possible that interventions that normalize the microbial composition could ameliorate neurogenesis-related neurological disorders to some extent.
Probiotics are beneficial bacteria that, in sufficient quantities, can confer health benefits in the host (Gareau et al., 2010). Probiotics help to maintain immune homeostasis by regulating gastrointestinal function and inhibiting systemic immune responses (Logan and Katzman, 2005; Franco-Robles and López, 2015). Probiotics have been considered as a potential treatment for depression and anxiety. Administration of Lactobacillus helveticus combined with Bifidobacterium longum has been reported to decrease anxiety-like behaviors in both rats and humans (Messaoudi et al., 2011). In one randomized controlled trial, intake of probiotics reduced negative thoughts associated with depression (Steenbergen et al., 2015). Importantly, VSL#3, a probiotic mixture, has been shown to reduce inflammation in aged rats by decreasing IL-10 protein expression, and to contribute to the increase of BDNF mRNA and synapsin in the hippocampus (Distrutti et al., 2014). In a study investigating the effect of Lactobacillus strains on cognitive decline in aging-accelerated mice, supplementation of the subspecies K71 improved cognitive performance, which was attributed to an upregulation of hippocampal BDNF expression (Corpuz et al., 2018). In rats fed with a chronic high-fat diet, supplementation with another subspecies, HII01, has been found to increase hippocampal plasticity and attenuate brain mitochondrial dysfunction and microglial activation (Chunchai et al., 2018). Therefore, probiotics supplementation may be beneficial to the remission of mental illness and the improvement of cognitive ability. However, the pilot AMBITION study with five patients with Alzheimer’s disease (NCT03998423) was recently interrupted after SARS-CoV-2 was detected in fecal material, which serves as a reminder of the potential risk of fecal transfer.
Conclusions
Since the inception of the concept of the microbiota-gut-brain axis, much research has contributed to our knowledge about bacteria-host communication and the signaling pathways involved. The gut microbiome can influence many physiological processes, including hippocampal neurogenesis, which critically links the gut microbiome with CNS development. Albeit preliminary and mostly experimental, the existing literature indicates that abnormal gut microbial composition could serve as a pathogenic factor that contributes to neurogenic processes, thus playing a part in an array of neurological disorders. The mechanisms underlying gut microbiome-induced inhibition or promotion of neurogenesis have not yet been elucidated, but may include neural, humoral, metabolic, and immune pathways. Supplementation with probiotics could positively alter the composition and function of gut microbiome, and thus represents a promising treatment strategy for neurological diseases. However, more well-designed clinical trials are needed to evaluate the efficacy and safety of probiotics. Overall, we are still in the early stages of understanding the association between neurogenesis and gut microbiota. Future work could investigate the impact of gut microbiota in immune-mediated CNS diseases, such as MS, neuromyelitis optica, and Guillain-Barré syndrome, as well as non-immune-mediated CNS diseases, such as ASD, depression, and anxiety.
In conclusion, the study of gut microbiota holds great promise for improving the diagnosis, prognosis, and treatment of CNS diseases. SCFAs are key metabolites derived from gut microbiota that affect T cell function, thereby indirectly regulating the nerve regeneration environment. In experimental animal models of stroke, SCFAs have also been reported to promote the effectiveness of rehabilitation at the functional and morphological levels, which could aid the development of new, more effective rehabilitation strategies in patients with stroke (Sadler et al., 2020). Future work could also examine whether the number of gut microbiota can predict the severity of CNS diseases such as stroke, as well as the response to treatment. Additional studies are needed to determine the role of different microbial species in the neurogenesis and pathogenesis of neurological diseases, as well as how gut microbiota could be modulated to enhance therapeutic effects.
Additional file: Open peer review reports 1 (90.1KB, pdf) and 2 (92.5KB, pdf) .
Footnotes
P-Reviewers: Hernández-Echeagaray E, Imbimbo BP; C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Cason N, Yu J, Song LP; T-Editor: Jia Y
Conflicts of interest:The authors declare that they have no conflict of interest.
Financial support:None.
Copyright license agreement:The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check:Checked twice by iThenticate.
Peer review:Externally peer reviewed.
Open peer reviewers:Elizabeth Hernández-Echeagaray, Universidad Nacional Autónoma de México, Mexico; Bruno P. Imbimbo, Chiesi Farmaceutici S.p.A., Italy.
References
- 1.Alenina N, Klempin F. The role of serotonin in adult hippocampal neurogenesis. Behav Brain Res. 2015;277:49–57. doi: 10.1016/j.bbr.2014.07.038. [DOI] [PubMed] [Google Scholar]
- 2.Asan E, Steinke M, Lesch KP. Serotonergic innervation of the amygdala: targets, receptors, and implications for stress and anxiety. Histochem Cell Biol. 2013;139:785–813. doi: 10.1007/s00418-013-1081-1. [DOI] [PubMed] [Google Scholar]
- 3.Baizabal-Carvallo JF. Gut microbiota: a potential therapeutic target for Parkinson’s disease. Neural Regen Res. 2021;16:287–288. doi: 10.4103/1673-5374.290896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Benakis C, Brea D, Caballero S, Faraco G, Moore J, Murphy M, Sita G, Racchumi G, Ling L, Pamer EG, Iadecola C, Anrather J. Commensal microbiota affects ischemic stroke outcome by regulating intestinal γδ T cells. Nat Med. 2016;22:516–523. doi: 10.1038/nm.4068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bhattacharjee S, Lukiw WJ. Alzheimer’s disease and the microbiome. Front Cell Neurosci. 2013;7:153. doi: 10.3389/fncel.2013.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cavallucci V, Fidaleo M, Pani G. Nutrients and neurogenesis: the emerging role of autophagy and gut microbiota. Curr Opin Pharmacol. 2020;50:46–52. doi: 10.1016/j.coph.2019.11.004. [DOI] [PubMed] [Google Scholar]
- 7.Cerdó T, Diéguez E, Campoy C. Impact of gut microbiota on neurogenesis and neurological diseases during infancy. Curr Opin Pharmacol. 2020;50:33–37. doi: 10.1016/j.coph.2019.11.006. [DOI] [PubMed] [Google Scholar]
- 8.Chunchai T, Thunapong W, Yasom S, Wanchai K, Eaimworawuthikul S, Metzler G, Lungkaphin A, Pongchaidecha A, Sirilun S, Chaiyasut C, Pratchayasakul W, Thiennimitr P, Chattipakorn N, Chattipakorn SC. Decreased microglial activation through gut-brain axis by prebiotics, probiotics, or synbiotics effectively restored cognitive function in obese-insulin resistant rats. J Neuroinflammation. 2018;15:11. doi: 10.1186/s12974-018-1055-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Claesson MJ, Jeffery IB, Conde S, Power SE, O’Connor EM, Cusack S, Harris HM, Coakley M, Lakshminarayanan B, O’Sullivan O, Fitzgerald GF, Deane J, O’Connor M, Harnedy N, O’Connor K, O’Mahony D, van Sinderen D, Wallace M, Brennan L, Stanton C, et al. Gut microbiota composition correlates with diet and health in the elderly. Nature. 2012;488:178–184. doi: 10.1038/nature11319. [DOI] [PubMed] [Google Scholar]
- 10.Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F, Dinan TG, Cryan JF. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry. 2013;18:666–673. doi: 10.1038/mp.2012.77. [DOI] [PubMed] [Google Scholar]
- 11.Corpuz HM, Ichikawa S, Arimura M, Mihara T, Kumagai T, Mitani T, Nakamura S, Katayama S. Long-term diet supplementation with Lactobacillus Paracasei K71 prevents age-related cognitive decline in senescence-accelerated mouse prone 8. Nutrients. 2018;10:762. doi: 10.3390/nu10060762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cosacak MI, Bhattarai P, Kizil C. Alzheimer’s disease, neural stem cells and neurogenesis: cellular phase at single-cell level. Neural Regen Res. 2020;15:824–827. doi: 10.4103/1673-5374.268896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13:701–712. doi: 10.1038/nrn3346. [DOI] [PubMed] [Google Scholar]
- 14.Cryan JF, O’Riordan KJ, Sandhu K, Peterson V, Dinan TG. The gut microbiome in neurological disorders. Lancet Neurol. 2020;19:179–194. doi: 10.1016/S1474-4422(19)30356-4. [DOI] [PubMed] [Google Scholar]
- 15.Distrutti E, O’Reilly JA, McDonald C, Cipriani S, Renga B, Lynch MA, Fiorucci S. Modulation of intestinal microbiota by the probiotic VSL#3 resets brain gene expression and ameliorates the age-related deficit in LTP. PLoS One. 2014;9:e106503. doi: 10.1371/journal.pone.0106503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Diviccaro S, Giatti S, Borgo F, Barcella M, Borghi E, Trejo JL, Garcia-Segura LM, Melcangi RC. Treatment of male rats with finasteride, an inhibitor of 5alpha-reductase enzyme, induces long-lasting effects on depressive-like behavior, hippocampal neurogenesis, neuroinflammation and gut microbiota composition. Psychoneuroendocrinology. 2019;99:206–215. doi: 10.1016/j.psyneuen.2018.09.021. [DOI] [PubMed] [Google Scholar]
- 17.Dunphy-Doherty F, O’Mahony SM, Peterson VL, O’Sullivan O, Crispie F, Cotter PD, Wigmore P, King MV, Cryan JF, Fone KCF. Post-weaning social isolation of rats leads to long-term disruption of the gut microbiota-immune-brain axis. Brain Behav Immun. 2018;68:261–273. doi: 10.1016/j.bbi.2017.10.024. [DOI] [PubMed] [Google Scholar]
- 18.Erny D, Hrabě de Angelis AL, Jaitin D, Wieghofer P, Staszewski O, David E, Keren-Shaul H, Mahlakoiv T, Jakobshagen K, Buch T, Schwierzeck V, Utermöhlen O, Chun E, Garrett WS, McCoy KD, Diefenbach A, Staeheli P, Stecher B, Amit I, Prinz M. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015;18:965–977. doi: 10.1038/nn.4030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Filosa S, Di Meo F, Crispi S. Polyphenols-gut microbiota interplay and brain neuromodulation. Neural Regen Res. 2018;13:2055–2059. doi: 10.4103/1673-5374.241429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Franco-Robles E, López MG. Implication of fructans in health: immunomodulatory and antioxidant mechanisms. ScientificWorldJournal. 2015;2015:289267. doi: 10.1155/2015/289267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fröhlich EE, Farzi A, Mayerhofer R, Reichmann F, Jačan A, Wagner B, Zinser E, Bordag N, Magnes C, Fröhlich E, Kashofer K, Gorkiewicz G, Holzer P. Cognitive impairment by antibiotic-induced gut dysbiosis: Analysis of gut microbiota-brain communication. Brain Behav Immun. 2016;56:140–155. doi: 10.1016/j.bbi.2016.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fung TC, Olson CA, Hsiao EY. Interactions between the microbiota, immune and nervous systems in health and disease. Nat Neurosci. 2017;20:145–155. doi: 10.1038/nn.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gage FH. Adult neurogenesis in mammals. Science. 2019;364:827–828. doi: 10.1126/science.aav6885. [DOI] [PubMed] [Google Scholar]
- 24.Gage FH, Temple S. Neural stem cells: generating and regenerating the brain. Neuron. 2013;80:588–601. doi: 10.1016/j.neuron.2013.10.037. [DOI] [PubMed] [Google Scholar]
- 25.Gareau MG, Sherman PM, Walker WA. Probiotics and the gut microbiota in intestinal health and disease. Nat Rev Gastroenterol Hepatol. 2010;7:503–514. doi: 10.1038/nrgastro.2010.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ho L, Ono K, Tsuji M, Mazzola P, Singh R, Pasinetti GM. Protective roles of intestinal microbiota derived short chain fatty acids in Alzheimer’s disease-type beta-amyloid neuropathological mechanisms. Expert Rev Neurother. 2018;18:83–90. doi: 10.1080/14737175.2018.1400909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Houlden A, Goldrick M, Brough D, Vizi ES, Lénárt N, Martinecz B, Roberts IS, Denes A. Brain injury induces specific changes in the caecal microbiota of mice via altered autonomic activity and mucoprotein production. Brain Behav Immun. 2016;57:10–20. doi: 10.1016/j.bbi.2016.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ichim G, Tauszig-Delamasure S, Mehlen P. Neurotrophins and cell death. Exp Cell Res. 2012;318:1221–1228. doi: 10.1016/j.yexcr.2012.03.006. [DOI] [PubMed] [Google Scholar]
- 29.Janssen AW, Kersten S. The role of the gut microbiota in metabolic health. FASEB J. 2015;29:3111–3123. doi: 10.1096/fj.14-269514. [DOI] [PubMed] [Google Scholar]
- 30.Kadowaki A, Quintana FJ. The gut-CNS axis in multiple sclerosis. Trends Neurosci. 2020;43:622–634. doi: 10.1016/j.tins.2020.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kasubuchi M, Hasegawa S, Hiramatsu T, Ichimura A, Kimura I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients. 2015;7:2839–2849. doi: 10.3390/nu7042839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Keshavarzian A, Green SJ, Engen PA, Voigt RM, Naqib A, Forsyth CB, Mutlu E, Shannon KM. Colonic bacterial composition in Parkinson’s disease. Mov Disord. 2015;30:1351–1360. doi: 10.1002/mds.26307. [DOI] [PubMed] [Google Scholar]
- 33.Koyama R, Ikegaya Y. Microglia in the pathogenesis of autism spectrum disorders. Neurosci Res. 2015;100:1–5. doi: 10.1016/j.neures.2015.06.005. [DOI] [PubMed] [Google Scholar]
- 34.Kundu P, Lee HU, Garcia-Perez I, Tay EXY, Kim H, Faylon LE, Martin KA, Purbojati R, Drautz-Moses DI, Ghosh S, Nicholson JK, Schuster S, Holmes E, Pettersson S. Neurogenesis and prolongevity signaling in young germ-free mice transplanted with the gut microbiota of old mice. Sci Transl Med. 2019;11:eaau4760. doi: 10.1126/scitranslmed.aau4760. [DOI] [PubMed] [Google Scholar]
- 35.Lazarov O, Hollands C. Hippocampal neurogenesis: learning to remember. Prog Neurobiol. 2016;138-140:1–18. doi: 10.1016/j.pneurobio.2015.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lenz KM, Nelson LH. Microglia and beyond: innate immune cells as regulators of brain development and behavioral function. Front Immunol. 2018;9:698. doi: 10.3389/fimmu.2018.00698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li H, Sun J, Du J, Wang F, Fang R, Yu C, Xiong J, Chen W, Lu Z, Liu J. Clostridium butyricum exerts a neuroprotective effect in a mouse model of traumatic brain injury via the gut-brain axis. Neurogastroenterol Motil. 2018;30:e13260. doi: 10.1111/nmo.13260. [DOI] [PubMed] [Google Scholar]
- 38.Liang YF, Hao P, Duan HM, Zhao W, Gao YD, Li XG, Yang CY. Pathological and behavioral changes after ischemic stroke in adult mice. Zhongguo Zuzhi Gongcheng Yanjiu. 2020;24:5625–5631. [Google Scholar]
- 39.Liu S, Li E, Sun Z, Fu D, Duan G, Jiang M, Yu Y, Mei L, Yang P, Tang Y, Zheng P. Altered gut microbiota and short chain fatty acids in Chinese children with autism spectrum disorder. Sci Rep. 2019;9:287. doi: 10.1038/s41598-018-36430-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Logan AC, Katzman M. Major depressive disorder: probiotics may be an adjuvant therapy. Med Hypotheses. 2005;64:533–538. doi: 10.1016/j.mehy.2004.08.019. [DOI] [PubMed] [Google Scholar]
- 41.Mayer EA, Tillisch K, Gupta A. Gut/brain axis and the microbiota. J Clin Invest. 2015;125:926–938. doi: 10.1172/JCI76304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mayer EA, Knight R, Mazmanian SK, Cryan JF, Tillisch K. Gut microbes and the brain: paradigm shift in neuroscience. J Neurosci. 2014;34:15490–15496. doi: 10.1523/JNEUROSCI.3299-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mazzini L, Mogna L, De Marchi F, Amoruso A, Pane M, Aloisio I, Cionci NB, Gaggìa F, Lucenti A, Bersano E, Cantello R, Di Gioia D, Mogna G. Potential role of gut microbiota in ALS pathogenesis and possible novel therapeutic strategies. J Clin Gastroenterol. 2018;52(Suppl 1) doi: 10.1097/MCG.0000000000001042. Proceedings from the 9th Probiotics, Prebiotics and New Foods, Nutraceuticals and Botanicals for Nutrition & Human and Microbiota Health Meeting, held in Rome, Italy from September 10 to 12, 2017:S68-70. [DOI] [PubMed] [Google Scholar]
- 44.Melbye P, Olsson A, Hansen TH, Søndergaard HB, Bang Oturai A. Short-chain fatty acids and gut microbiota in multiple sclerosis. Acta Neurol Scand. 2019;139:208–219. doi: 10.1111/ane.13045. [DOI] [PubMed] [Google Scholar]
- 45.Messaoudi M, Lalonde R, Violle N, Javelot H, Desor D, Nejdi A, Bisson JF, Rougeot C, Pichelin M, Cazaubiel M, Cazaubiel JM. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr. 2011;105:755–764. doi: 10.1017/S0007114510004319. [DOI] [PubMed] [Google Scholar]
- 46.Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, Rábano A, Cafini F, Pallas-Bazarra N, Ávila J, Llorens-Martín M. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25:554–560. doi: 10.1038/s41591-019-0375-9. [DOI] [PubMed] [Google Scholar]
- 47.Nagahara AH, Tuszynski MH. Potential therapeutic uses of BDNF in neurological and psychiatric disorders. Nat Rev Drug Discov. 2011;10:209–219. doi: 10.1038/nrd3366. [DOI] [PubMed] [Google Scholar]
- 48.Neufeld KM, Kang N, Bienenstock J, Foster JA. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol Motil. 2011;23:255–264, e119. doi: 10.1111/j.1365-2982.2010.01620.x. [DOI] [PubMed] [Google Scholar]
- 49.Neumann J, Riek-Burchardt M, Herz J, Doeppner TR, König R, Hütten H, Etemire E, Männ L, Klingberg A, Fischer T, Görtler MW, Heinze HJ, Reichardt P, Schraven B, Hermann DM, Reymann KG, Gunzer M. Very-late-antigen-4 (VLA-4)-mediated brain invasion by neutrophils leads to interactions with microglia, increased ischemic injury and impaired behavior in experimental stroke. Acta Neuropathol. 2015;129:259–277. doi: 10.1007/s00401-014-1355-2. [DOI] [PubMed] [Google Scholar]
- 50.O’Leary OF, Ogbonnaya ES, Felice D, Levone BR, Conroy LC, Fitzgerald P, Bravo JA, Forsythe P, Bienenstock J, Dinan TG, Cryan JF. The vagus nerve modulates BDNF expression and neurogenesis in the hippocampus. Eur Neuropsychopharmacol. 2018;28:307–316. doi: 10.1016/j.euroneuro.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 51.O’Sullivan E, Barrett E, Grenham S, Fitzgerald P, Stanton C, Ross RP, Quigley EM, Cryan JF, Dinan TG. BDNF expression in the hippocampus of maternally separated rats: does Bifidobacterium breve 6330 alter BDNF levels. Benef Microbes. 2011;2:199–207. doi: 10.3920/BM2011.0015. [DOI] [PubMed] [Google Scholar]
- 52.Ogbonnaya ES, Clarke G, Shanahan F, Dinan TG, Cryan JF, O’Leary OF. Adult hippocampal neurogenesis is regulated by the microbiome. Biol Psychiatry. 2015;78:e7–9. doi: 10.1016/j.biopsych.2014.12.023. [DOI] [PubMed] [Google Scholar]
- 53.Park AM, Omura S, Fujita M, Sato F, Tsunoda I. Helicobacter pylori and gut microbiota in multiple sclerosis versus Alzheimer’s disease: 10 pitfalls of microbiome studies. Clin Exp Neuroimmunol. 2017;8:215–232. doi: 10.1111/cen3.12401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, Nielsen T, Pons N, Levenez F, Yamada T, Mende DR, Li J, Xu J, Li S, Li D, Cao J, Wang B, Liang H, Zheng H, Xie Y, et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464:59–65. doi: 10.1038/nature08821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rajendran L, Paolicelli RC. Microglia-mediated synapse loss in Alzheimer’s disease. J Neurosci. 2018;38:2911–2919. doi: 10.1523/JNEUROSCI.1136-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rhee SH, Pothoulakis C, Mayer EA. Principles and clinical implications of the brain-gut-enteric microbiota axis. Nat Rev Gastroenterol Hepatol. 2009;6:306–314. doi: 10.1038/nrgastro.2009.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rodríguez-Gómez JA, Kavanagh E, Engskog-Vlachos P, Engskog MKR, Herrera AJ, Espinosa-Oliva AM, Joseph B, Hajji N, Venero JL, Burguillos MA. Microglia: agents of the CNS pro-inflammatory response. Cells. 2020;9:1717. doi: 10.3390/cells9071717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Roubaud-Baudron C, Krolak-Salmon P, Quadrio I, Mégraud F, Salles N. Impact of chronic Helicobacter pylori infection on Alzheimer’s disease: preliminary results. Neurobiol Aging. 2012;33:1009.e11–9. doi: 10.1016/j.neurobiolaging.2011.10.021. [DOI] [PubMed] [Google Scholar]
- 59.Rubio-Perez JM, Morillas-Ruiz JM. A review: inflammatory process in Alzheimer’s disease, role of cytokines. ScientificWorldJournal. 2012;2012:756357. doi: 10.1100/2012/756357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sadler R, Cramer JV, Heindl S, Kostidis S, Betz D, Zuurbier KR, Northoff BH, Heijink M, Goldberg MP, Plautz EJ, Roth S, Malik R, Dichgans M, Holdt LM, Benakis C, Giera M, Stowe AM, Liesz A. Short-chain fatty acids improve poststroke recovery via immunological mechanisms. J Neurosci. 2020;40:1162–1173. doi: 10.1523/JNEUROSCI.1359-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Salvo E, Stokes P, Keogh CE, Brust-Mascher I, Hennessey C, Knotts TA, Sladek JA, Rude KM, Swedek M, Rabasa G, Gareau MG. A murine model of pediatric inflammatory bowel disease causes microbiota-gut-brain axis deficits in adulthood. Am J Physiol Gastrointest Liver Physiol. 2020;319:G361–374. doi: 10.1152/ajpgi.00177.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Scheperjans F, Aho V, Pereira PA, Koskinen K, Paulin L, Pekkonen E, Haapaniemi E, Kaakkola S, Eerola-Rautio J, Pohja M, Kinnunen E, Murros K, Auvinen P. Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov Disord. 2015;30:350–358. doi: 10.1002/mds.26069. [DOI] [PubMed] [Google Scholar]
- 63.Selvaraj UM, Stowe AM. Long-term T cell responses in the brain after an ischemic stroke. Discov Med. 2017;24:323–333. [PMC free article] [PubMed] [Google Scholar]
- 64.Siopi E, Chevalier G, Katsimpardi L, Saha S, Bigot M, Moigneu C, Eberl G, Lledo PM. Changes in gut microbiota by chronic stress impair the efficacy of fluoxetine. Cell Rep. 2020;30:3682–3690e6. doi: 10.1016/j.celrep.2020.02.099. [DOI] [PubMed] [Google Scholar]
- 65.Sivaprakasam S, Prasad PD, Singh N. Benefits of short-chain fatty acids and their receptors in inflammation and carcinogenesis. Pharmacol Ther. 2016;164:144–151. doi: 10.1016/j.pharmthera.2016.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Steenbergen L, Sellaro R, van Hemert S, Bosch JA, Colzato LS. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav Immun. 2015;48:258–264. doi: 10.1016/j.bbi.2015.04.003. [DOI] [PubMed] [Google Scholar]
- 67.Stilling RM, Dinan TG, Cryan JF. Microbial genes, brain & behaviour - epigenetic regulation of the gut-brain axis. Genes Brain Behav. 2014;13:69–86. doi: 10.1111/gbb.12109. [DOI] [PubMed] [Google Scholar]
- 68.Szapacs ME, Mathews TA, Tessarollo L, Ernest Lyons W, Mamounas LA, Andrews AM. Exploring the relationship between serotonin and brain-derived neurotrophic factor: analysis of BDNF protein and extraneuronal 5-HT in mice with reduced serotonin transporter or BDNF expression. J Neurosci Methods. 2004;140:81–92. doi: 10.1016/j.jneumeth.2004.03.026. [DOI] [PubMed] [Google Scholar]
- 69.Takewaki D, Suda W, Sato W, Takayasu L, Kumar N, Kimura K, Kaga N, Mizuno T, Miyake S, Hattori M, Yamamura T. Alterations of the gut ecological and functional microenvironment in different stages of multiple sclerosis. Proc Natl Acad Sci U S A. 2020;117:22402–22412. doi: 10.1073/pnas.2011703117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tremlett H, Bauer KC, Appel-Cresswell S, Finlay BB, Waubant E. The gut microbiome in human neurological disease: A review. Ann Neurol. 2017;81:369–382. doi: 10.1002/ana.24901. [DOI] [PubMed] [Google Scholar]
- 71.Unger MM, Spiegel J, Dillmann KU, Grundmann D, Philippeit H, Bürmann J, Faßbender K, Schwiertz A, Schäfer KH. Short chain fatty acids and gut microbiota differ between patients with Parkinson’s disease and age-matched controls. Parkinsonism Relat Disord. 2016;32:66–72. doi: 10.1016/j.parkreldis.2016.08.019. [DOI] [PubMed] [Google Scholar]
- 72.van Praag H, Shubert T, Zhao C, Gage FH. Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci. 2005;25:8680–8685. doi: 10.1523/JNEUROSCI.1731-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Vogt NM, Kerby RL, Dill-McFarland KA, Harding SJ, Merluzzi AP, Johnson SC, Carlsson CM, Asthana S, Zetterberg H, Blennow K, Bendlin BB, Rey FE. Gut microbiome alterations in Alzheimer’s disease. Sci Rep. 2017;7:13537. doi: 10.1038/s41598-017-13601-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wu S, Yi J, Zhang YG, Zhou J, Sun J. Leaky intestine and impaired microbiome in an amyotrophic lateral sclerosis mouse model. Physiol Rep. 2015;3:e12356. doi: 10.14814/phy2.12356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yang LL, Millischer V, Rodin S, MacFabe DF, Villaescusa JC, Lavebratt C. Enteric short-chain fatty acids promote proliferation of human neural progenitor cells. J Neurochem. 2020;154:635–646. doi: 10.1111/jnc.14928. [DOI] [PubMed] [Google Scholar]
- 76.Yarandi SS, Kulkarni S, Saha M, Sylvia KE, Sears CL, Pasricha PJ. Intestinal bacteria maintain adult enteric nervous system and nitrergic neurons via Toll-like receptor 2-induced neurogenesis in mice. Gastroenterology. 2020;159:200–213.e8. doi: 10.1053/j.gastro.2020.03.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zhao C, Deng W, Gage FH. Mechanisms and functional implications of adult neurogenesis. Cell. 2008;132:645–660. doi: 10.1016/j.cell.2008.01.033. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


