Abstract
The COVID-19 pandemic caused by SARS-CoV-2 challenges the understanding of factors affecting disease progression and severity. The identification of prognostic biomarkers and physiological processes associated with disease symptoms is relevant for the development of new diagnostic and therapeutic interventions to contribute to the control of this pandemic. To address this challenge, in this study, we used a quantitative proteomics together with multiple data analysis algorithms to characterize serum protein profiles in five cohorts from healthy to SARS-CoV-2-infected recovered (hospital discharge), nonsevere (hospitalized), and severe [at the intensive care unit (ICU)] cases with increasing systemic inflammation in comparison with healthy individuals sampled prior to the COVID-19 pandemic. The results showed significantly dysregulated proteins and associated biological processes and disorders associated to COVID-19. These results corroborated previous findings in COVID-19 studies and highlighted how the representation of dysregulated serum proteins and associated BPs increases with COVID-19 disease symptomatology from asymptomatic to severe cases. The analysis was then focused on novel disease processes and biomarkers that were correlated with disease symptomatology. To contribute to translational medicine, results corroborated the predictive value of selected immune-related biomarkers for disease recovery [Selenoprotein P (SELENOP) and Serum paraoxonase/arylesterase 1 (PON1)], severity [Carboxypeptidase B2 (CBP2)], and symptomatology [Pregnancy zone protein (PZP)] using protein-specific ELISA tests. Our results contributed to the characterization of SARS-CoV-2–host molecular interactions with potential contributions to the monitoring and control of this pandemic by using immune-related biomarkers associated with disease symptomatology.
Keywords: COVID-19, proteomic, immunology, biomarker, diagnostic, prognostic, symptomatology
Introduction
Coronavirus disease 19 (COVID-19) is a pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2, also referred as hCoV-19) with immunological dysregulation associated with disease severity (1, 2). The incidence of this pandemic is still increasing worldwide and posts a challenge for the understanding of host and virus-derived factors affecting disease severity and the identification of prognostic biomarkers and physiological processes related to COVID-19 symptomatology and relevant for the development of new diagnostic and therapeutic interventions to contribute to the control of this pandemic (3–6).
To address this challenge, proteomics constitutes a high-resolution method for the study of host response to infectious diseases, including those caused by RNA viruses (7). Quantitative proteomics has been used for the study of SARS-CoV-2 infection in various samples (e.g., serum, plasma or urine), tissues (e.g., lung), and cells (e.g., peripheral blood mononuclear or Caco-2 cells). This experimental approach has been used for the study of host anti-viral responses and the identification of biomarkers for COVID-19 disease severity, diagnostics, and treatment. Examples of these biomarkers are serum amyloid A-1 (SAA1), serum amyloid A-2 (SAA2), C-reactive protein (CRP), gelsolin (GSN), interleukins (IL-1, IL-6), serine protease inhibitors (SERPINs), progranulin (GRN), apolipoproteins (APOs), complement and pro-inflammatory factors, coagulation system, and vascular cell adhesion protein 1 (VCAM-1) (4–23). Results of proteomics analyses have shown a correlation of disease severity with inflammatory, immunological, and cancer biomarkers, metabolic suppression, neutrophil activation, hepatic and lung injury and the dysregulation of lipid transport, macrophages, platelet degranulation, and complement system pathways (4–6, 8–13, 18, 20–22, 24).
However, due to the complexity of COVID-19 symptomatology, it is important to characterize host response to SARS-CoV-2 infection in different cohorts from asymptomatic individuals to severe patients to better understand disease mechanisms and symptoms with possible medical complications at different levels, and the identification of potential diagnostic markers and drug targets (22–25). Quantitative proteomics approaches alone or in combination with other omics technologies are key to achieve this goal (24–27). To contribute in addressing this objective, herein we used a sequential window acquisition of all theoretical mass spectra (SWATH-MS) quantitative proteomics to characterize serum protein profiles in five cohorts of healthy (pre-pandemic sampling) and SARS-CoV-2-infected asymptomatic, recovered (hospital discharge), nonsevere (hospitalized), and severe [intensive care unit (ICU)] individuals. The results advanced our understanding of the molecular mechanism-driven host–SARS-CoV-2 interactions and identified immune-related prognostic biomarkers and physiological processes related to COVID-19 symptomatology.
Materials and Methods
Samples From Healthy Individuals and COVID-19 Patients
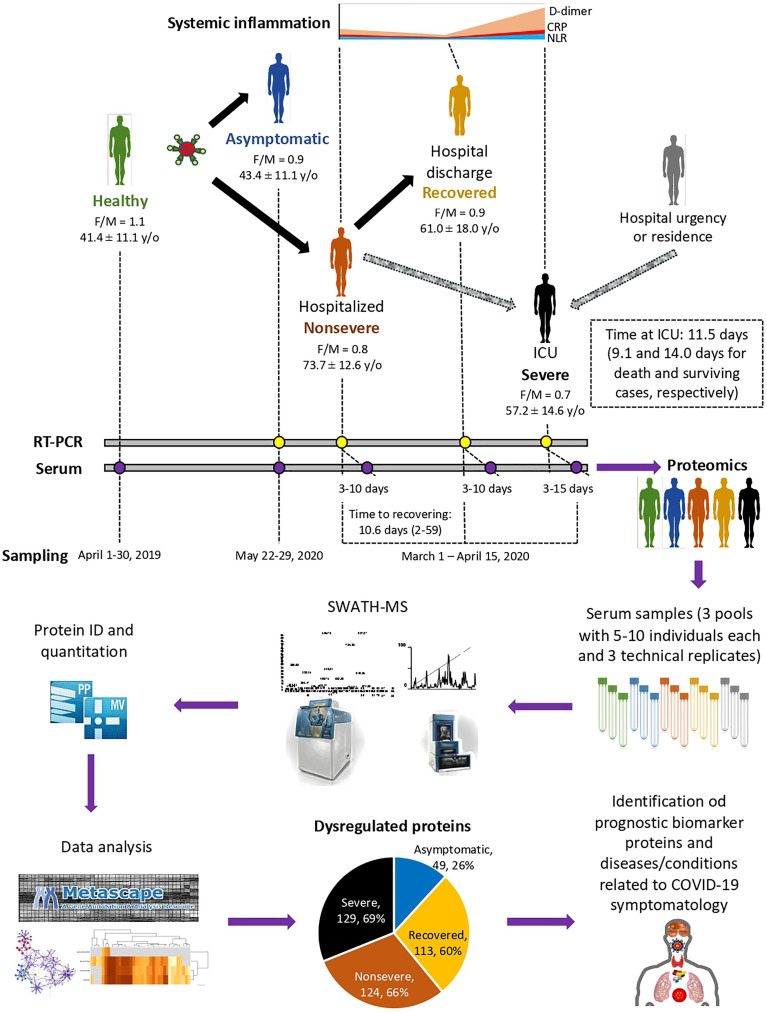
A retrospective case–control study was conducted in patients suffering from COVID-19 and healthy controls sampled at the University General Hospital of Ciudad Real (HGUCR), Spain (28, 29). Blood samples from control individuals were collected prior to the COVID-19 pandemic in April 2019. COVID-19 patients were confirmed as SARS-CoV-2-infected by IgG antibody titers or reverse transcriptase-polymerase chain reaction (RT-PCR) and sampled between March and May 2020 (28) (Figure 1). Clinical symptoms and laboratory determinations associated with COVID-19 were obtained from patient’s medical records to create cohorts of asymptomatic, nonsevere (hospitalized), recovered (hospital discharge), and severe (ICU) individuals (28). Patients were hospitalized for developing a moderate-severe clinical condition with radiologically demonstrated pneumonia and failure in blood oxygen saturation. Patients with acute respiratory failure who needed mechanical ventilation support were admitted to a hospital ICU. The patients were discharged from the hospital due to the clinical and radiological improvement of pneumonia caused by the SARS-CoV-2, along with the normalization of analytical parameters indicative of inflammation. Data can be found at Urra et al. (28) and Supplementary Table 1. Blood samples were drawn in a vacutainer tube without anticoagulant. The tube remained at rest for 15–30 min at room temperature (RT) for clotting. Subsequently, the tube was centrifuged at 1500 × g for 10 min at RT to remove the clot and obtain serum. Serum samples were heat-inactivated for 30 min at 56°C and conserved at −20°C until used for analysis. The use of samples and individual data was approved by the Ethical and Scientific Committees (University Hospital of Ciudad Real C-352 and SESCAM C-73).
Figure 1.
Individual cohorts and study design. COVID-19 patients included cohorts of asymptomatic (n = 16), recovered (hospital discharge; n = 26), nonsevere (hospitalized; n = 28), and severe (ICU; n = 25) cases with increasing systemic inflammation. Healthy individuals sampled before the COVID-19 pandemic were included in the analysis (n = 25). Female-to-male (F/M) ratio and average ± S.D. age (y/o) are shown. Additional information can be found in Urra et al. (28). A SWATH-MS proteomics approach was used for data acquisition and analysis. A retrospective case–control study was conducted in patients suffering from COVID-19 and healthy controls sampled at indicated dates using standard procedures. Serum from three pools of 5–10 individuals each with three technical replicates were used for proteomics using SWATH-MS protein identification and quantitation and data analysis using Metascape and networks of interactions between proteins and BPs using Graph Theory algorithms to identify dysregulated proteins in response to COVID-19.
Serum Proteomics
Serum samples from healthy controls (n = 25) and asymptomatic (n = 16), nonsevere (n = 28), recovered (n = 26), and severe (n = 25) COVID-19 individuals were randomly clustered in three biological pools per group (n = 5–10 samples per pool). Protein concentration in samples was determined using the BCA Protein Assay with BSA (Sigma-Aldrich) as standard. Protein serum samples (100 µg per sample) were trypsin digested using the FASP Protein Digestion Kit (Expedeon Ltd., UK) and sequencing grade trypsin (Promega, Madison, WI, USA) following the manufacturer’s recommendations. The resulting tryptic peptides were desalted onto OMIX Pipette tips C18 (Agilent Technologies, Santa Clara, CA, USA), dried down, and stored at −20°C until mass spectrometry analysis. The desalted protein digests were resuspended in 2% acetonitrile and 5% acetic acid in water and analyzed by reverse-phase liquid chromatography coupled to mass spectrometry (RP-LC-MS/MS) using an Ekspert™ nanoLC 415 system coupled online with a 6600 TripleTOF mass spectrometer (AB SCIEX; Framingham, US) through Information-Dependent Acquisition (IDA) followed by Sequential Windowed data independent Acquisition of the Total High-resolution Mass Spectra (SWATH-MS). The peptides were concentrated in a 0.1 × 20 mm C18 RP precolumn (Thermo Scientific) with a flow rate of 5 µl/min during 10 min in solvent A. Then, peptides were separated in a 0.075 × 250 mm C18 RP column (New Objective, Woburn, MA, USA) with a flow rate of 300 nl/min. Elution was done in a 120-min gradient from 5% B to 30% B followed by a 15-min gradient from 30% B to 60% B (Solvent A: 0.1% formic acid in water, solvent B: 0.1% formic acid in acetonitrile) and directly injected into the mass spectrometer for analysis.
For IDA experiments, the mass spectrometer was set to scan full spectra from 350 m/z to 1400 m/z (250 ms accumulation time) followed by up to 50 MS/MS scans (100–1500 m/z). Candidate ions with a charge state between +2 and +5 and counts per second above a minimum threshold of 100 were isolated for fragmentation. One MS/MS spectrum was collected for 100 ms, before adding those precursor ions to the exclusion list for 15 s (mass spectrometer operated by Analyst TF 1.6, ABSciex). Dynamic background subtraction was turned off. Data were acquired in high sensitivity mode with rolling collision energy on and a collision energy spread of 5. A total amount of 4 µg of total proteins was injected.
For SWATH quantitative analysis, 45 independent samples (three technical replicates from each of the three biological replicates for each of the five experimental groups) (8 μg each) were subjected to the cyclic data independent acquisition (DIA) of mass spectra using the SWATH variable windows calculator (V 1.0, AB SCIEX) and the SWATH acquisition method editor (AB SCIEX), similar to established methods (30). A set of 50 overlapping windows was constructed (containing 1 m/z for the window overlap), covering the precursor mass range of 400–1250 m/z. For these experiments, a 50-ms survey scan (350–1400 m/z) was acquired at the beginning of each cycle, and SWATH-MS/MS spectra were collected from 100 to 1500 m/z for 70 ms at high sensitivity mode, resulting in a cycle time of 3.6 s. Collision energy for each window was determined according to the calculation for a charge +2 ion-centered upon the window with a collision energy spread of 15.
To create a spectral library of all the detectable peptides in the samples, the IDA MS raw files were combined and subjected to database searches in unison using ProteinPilot software v. 5.0.1 (AB SCIEX) with the Paragon algorithm. Spectra identification was performed by searching against the UniProt human proteome database (75,074 entries in October 2020) with the following parameters: iodoacetamide cysteine alkylation, trypsin digestion, identification focus on biological modification, and thorough ID as search effort. The detected protein threshold was set at 0.05. To assess the quality of identifications, an independent False Discovery Rate (FDR) analysis with the target-decoy approach provided by Protein Pilot was performed. Positive identifications were considered when identified proteins reached a 1% global FDR.
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE (31) partner repository with the dataset identifier PXD024549 and 10.6019/PXD024549.
Quality Control of Proteomics Data
Quality of proteomics data was controlled at multiple levels. First, a rat ileum digest was used for the evaluation of instrument performance. Buffer A samples were run as blanks every three injections to prevent carryover. Three technical replicates were injected for each sample. For validation of serum proteomics data, protein representation for previously identified biomarkers for COVID-19 and proteomics studies were used to show correlation with disease severity (Supplementary Figures 3, 4). An enrichment analysis was conducted using the Coronascape COVID database (https://metascape.org/COVID) (32) to identify proteins found in our study as differentially represented in response to COVID-19 and reported in previous COVID-19 omics datasets.
Data Analysis
For SWATH processing, up to 10 peptides with seven transitions per protein were automatically selected by the SWATH Acquisition MicroApp 2.0 in the PeakView 2.2 software with the following parameters: 15 ppm ion library tolerance, 5 min XIC extraction window, 0.01 Da XIC width, and considering only peptides with at least 99% confidence and excluding those that were shared or contained modifications. However, to ensure reliable quantitation, only proteins with three or more peptides available for quantitation were selected for XIC peak area extraction and exported for analysis in the MarkerView 1.3 software (AB SCIEX). Global normalization according to the Total Area Sums of all detected proteins in the samples was conducted (Supplementary Data 1).
The Student’s t-test (p < 0.05) was used to perform two-sample comparisons between the averaged area sums of all the transitions derived for each protein across the nine replicate runs for each group under comparison, in order to identify proteins that were significantly differentially represented between groups (Supplementary Data 1). Protein representation was also compared between groups by Welch’s unpaired t-test (p < 0.05; https://www.graphpad.com/quickcalcs/ttest1/?Format=C) and by one-way ANOVA test followed by post-hoc Bonferroni and Holm multiple comparisons (p < 0.05; https://astatsa.com/OneWay_Anova_with_TukeyHSD/) (4). Proteins with significant differences between healthy individuals and one of the COVID-19 cohorts only were selected for heatmap analysis of z-score using complete linkage and Spearman rank correlation (http://www.heatmapper.ca/expression/). Data were separately analyzed for overrepresented and underrepresented proteins using the Metascape gene annotation and analysis resource (https://metascape.org/gp/index.html#/main/step1) (Supplementary Figure 1).
To evaluate the network of interactions between proteins and BPs, a network was built using data for each protein and the BPs in which it is involved (Supplementary Data 2). This network reflects the importance of each protein on each BP according to its representation. The purpose was to obtain a general framework based on previous network developments using Graph Theory algorithms, which were revealed to be adequate for the purpose of representing these relationships (33). Networks exhibit nodes and the relationships between these components (links). Each node represents a protein or a BP. The network is directed, as each edge links each protein “to” one or multiple BPs. Several indices measure network properties from which the relationships among proteins and BPs are derived. The weighted degree (WG) is one of the most basic measures of a network, representing the number of links leaving (or arriving at) a given node after weighting by the total number of records containing this interaction. In this context, a protein always links to a BP with a “strength” derived from its representation. The WG provides an estimation of the strength of the association but does not evaluate the importance of each node in the context of the network. We used the Page Rank (PR) index to calculate the importance of each node in the complete network (34). This index calculates the number of links of each protein to one or several BPs, together with its weighted degree. The PR of each protein is calculated according to the authority (i.e., the relative importance) of each BP. The PR is an index that assigns a universal rank to nodes based on the importance of the other nodes to which it is linked and the WG. We calculated PR for each cohort (healthy, asymptomatic, recovered, nonsevere, and severe COVID-19 cases) and built separate networks for each condition. Then, we calculated how PR of both proteins and BPs changed in each group. We were looking for prominent changes in the nodes of the network, using an approach based on the distribution of values and the semantic rules of Fuzzy Logic (35). For each node of the network, we selected all the nodes that were in the first quintile (i.e., lowest values) of the PR’s distribution of groups “healthy” and “asymptomatic” and that were in the last quintile (i.e., high values) of distribution of groups “nonsevere” and “severe”. The opposite selection (highest versus lowest) was also carried out. After relating these queries by the operator “AND” according to Fuzzy Logic rules, each node was ranked between 0 (no change) and 1 (maximum change). We arbitrarily removed the nodes with values lower than 0.5. We also evaluated the weighted nestedness of each network as a measure of structuring. A network is more coherent and robust (i.e., resilient to node removal) if structuring is high. Nestedness is a measure in ecological system networks that emanates from the way elements are linked. It should be noted that the absence of nestedness does not mean the absence of a pattern. Nestedness is not a feature of the network, but a consequence of the WD sequences (36). Since most of the available algorithms evaluate the nestedness using only the pattern presence/absence (i.e., interaction/not interaction), we adhered to the approach provided by the software WINE (37) since it also accounts for the weights of the interactions in quantitative data matrices (proteins and BPs in our application) that include the number of events of each interaction and the strength of such interaction, or the representation of the proteins involved in each BP.
Determination of IL-1 and IL-4 Serum Levels
Serum levels of IL-1 and IL-4 were determined by ELISA (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s instructions. Briefly, 96-microwell plates coated in duplicate with anti-human IL-1β or IL-4 were washed twice with 400 µl/well of wash buffer and 100 µl of human IL-1β or IL-4 standard (20.00 pg/ml) at serial dilutions (1:2, 1:4, 1:8, 1:16, and 1:32), 100 µl/well of sera at 1:2 dilution, and 100 µl/well of sample diluent as negative control. Then, 50 µl/well of biotin conjugate were added to all wells. After incubation for 2 h at RT and three washes with 400 µl/well of wash buffer, 100 µl/well of streptaviding-HRP were added to all wells. After incubation for 1 h at RT and three washes with 400 µl/well of wash buffer, 100 µl/well of 3,3′,5,5′-Tetramethylbenzidine or TMB substrate solution were added to all wells. As soon as the Standard 1 well reached an O.D. of 0.9 at 620 nm, the colorimetric reaction was stopped with 100 µl/well of stop solution and the absorbance was measured in a spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) at an O.D. of 450 nm; 0.05 Human IL-1β or IL-4 concentration (pg/ml) in each sample was calculated from the obtained standard curve. The results were compared between different groups by one-way ANOVA test with post-hoc Tukey Honestly Significant Difference (HSD) (https://astatsa.com/OneWay_Anova_with_TukeyHSD/; p = 0.05).
Validation of Selected Serum Protein Biomarkers
Serum samples from cohorts included in the proteomics analysis plus additional samples of healthy controls (n = 37) and asymptomatic (n = 18), nonsevere (n = 29), recovered (n = 27), and severe (n = 25) COVID-19 individuals were used for validation analysis. Serum levels of PZP, SELENOP, CBP2, and PON1 were determined by ELISA (MyBioSource, Inc., San Diego, CA, USA, provided by bioNova Científica S.L., Madrid, Spain) following the manufacturer’s protocol available online (PZP, MBS2706073, https://www.mybiosource.com/human-elisa-kits/pregnancy-zone-protein-pzp/2706073; SELENOP, MBS163893, https://www.mybiosource.com/human-elisa-kits/selenoprotein-p-se-p/163893; CPB2, MBS703133, https://www.mybiosource.com/cpb2-human-elisa-kits/carboxypeptidase-b2-plasma/703133; PON1, MBS2883206, https://www.mybiosource.com/pon1-human-elisa-kits/serum-paraoxonase-arylesterase-1/2883206). The results were compared between different groups by one-way ANOVA test with post-hoc Tukey HSD (https://astatsa.com/OneWay_Anova_with_TukeyHSD/; p = 0.05). Proteomics and ELISA data were compared by Spearman’s Rho (rs) correlation analysis (https://www.socscistatistics.com/tests/spearman/default2.aspx; p = 0.05).
Results
Variations in Differential Serum Protein Profiles and Affected Biological Processes According to COVID-19 Disease Symptomatology
The study was conducted using a SWATH-MS quantitative proteomics to characterize serum protein profiles in COVID-19 patient cohorts from asymptomatic to recovered (hospital discharge), nonsevere (hospitalized), and severe (ICU) cases with increasing systemic inflammation in comparison with healthy individuals sampled prior to the COVID-19 pandemic (Figure 1). A total of 189 proteins were identified in serum samples from all cohorts included in the study (Supplementary Data 1). Of them, 49, 113, 124, and 129 proteins were significantly dysregulated in asymptomatic, recovered, nonsevere, and severe cases when compared to healthy controls, respectively (Figure 1; Supplementary Figure 1 and Data 1). As expected, immunoglobulins, high-density lipoproteins (HDL) and complement cascade represented 32% (60/189), 23% (44/189), and 12% (22/189) of identified serum proteins, respectively (Supplementary Data 1).
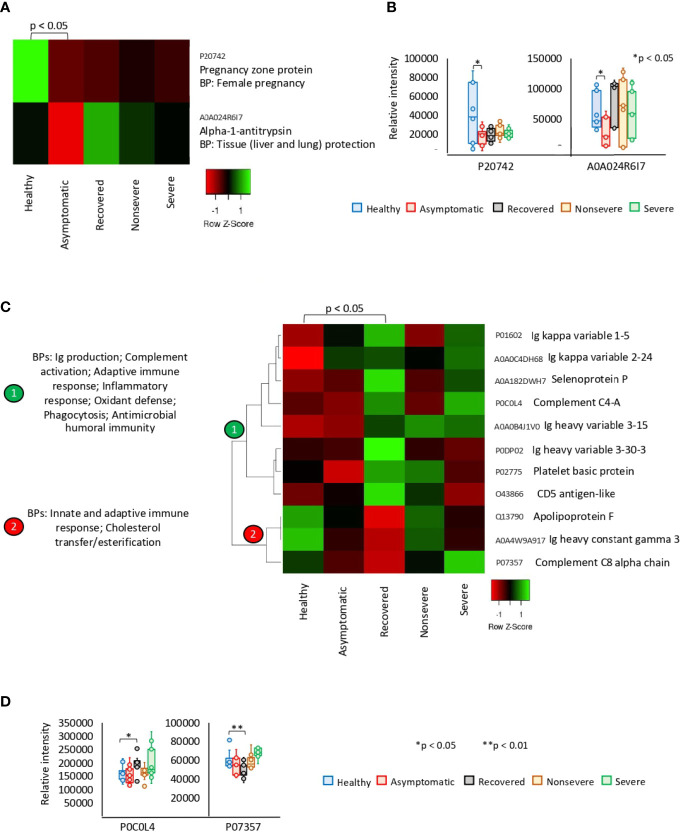
Of the significantly dysregulated proteins, Pregnancy zone protein (PZP) and Alpha-1-antitrypsin (SERPINA1) were identified as underrepresented in asymptomatic cases only (Figures 2A, B). These proteins are involved in biological processes (BPs) of female pregnancy and tissue protection. In recovered COVID-19 cases, 11 proteins were exclusively significantly dysregulated and grouped in two clades of overrepresented (n = 8) and underrepresented (n = 3) proteins (Figures 2C, D). Patient’s recovery was associated with dysregulation of immune response; increased complement activation, inflammatory response, and oxidant defense; and decrease in cholesterol transfer/esterification.
Figure 2.
Exclusive differential representation of proteins in sera from COVID-19 asymptomatic and recovered cases. (A) Heatmap of proteins significantly dysregulated (Z-scored original value) in asymptomatic cases only (p < 0.05; unpaired two-sided Welch’s t-test). Biological process (BP) is shown for each protein. (B) Change in levels of two selected proteins with significant differences between asymptomatic cases and healthy controls (*p < 0.05; unpaired two-sided Welch’s t-test). (C) Heatmap of proteins significantly dysregulated (Z-scored original value) in recovered cases only (p < 0.05; unpaired two-sided Welch’s t-test). Biological processes (BPs) are shown for each cluster of proteins differentially represented in response to COVID-19 (cluster 1, overrepresented; cluster 2, underrepresented). (D) Change in levels of two selected proteins with significant differences between recovered cases and healthy controls (*p < 0.05, **p < 0.01; unpaired two-sided Welch’s t-test).
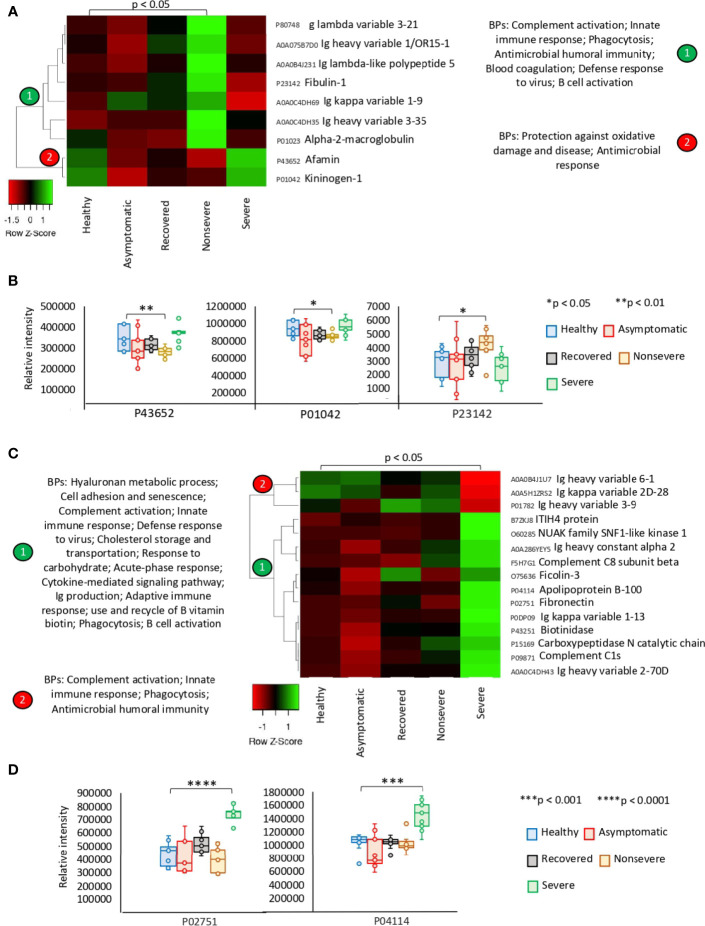
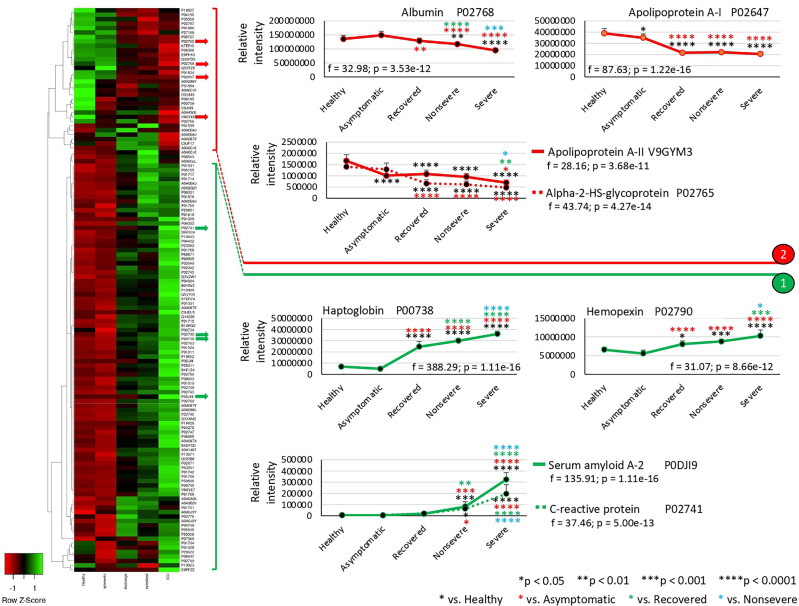
The exclusively significantly dysregulated serum proteins in nonsevere (n = 9) and severe (n = 15) patients affected multiple BPs (Figures 3A–D). In nonsevere cases, overrepresented proteins (n = 7) are involved in complement activation, immune response, and blood coagulation while underrepresented proteins (n = 2) reduce protection against oxidative damage and disease. Severe cases showed dysregulation of BPs such as immune response, metabolic processes, complement activation, and response to carbohydrate associated with overrepresented proteins (n = 12). Exclusively underrepresented proteins in severe cases (n = 3) are involved in immune response and complement activation. Proteins with multiple differential representation in sera from COVID-19 cases when compared to healthy controls (n = 128) were grouped into two clades of proteins with a tendency towards increase (n = 93) and decrease (n = 35) in representation according to disease severity (Figure 4).
Figure 3.
Exclusive differential representation of proteins in sera from COVID-19 nonsevere and severe cases. (A) Heatmap of proteins significantly dysregulated (Z-scored original value) in nonsevere cases only (p < 0.05; unpaired two-sided Welch’s t-test). Biological processes (BPs) are shown for each cluster of proteins differentially represented in response to COVID-19 (cluster 1, overrepresented; cluster 2, underrepresented). (B) Change in levels of three selected proteins with significant differences between nonsevere cases and healthy controls (*p < 0.05, **p < 0.01; unpaired two-sided Welch’s t-test). (C) Heatmap of proteins significantly dysregulated (Z-scored original value) in severe cases only (p < 0.05; unpaired two-sided Welch’s t-test). Biological processes (BPs) are shown for each cluster of proteins differentially represented in response to COVID-19 (cluster 1, overrepresented; cluster 2, underrepresented). (D) Change in levels of two selected proteins with significant differences between severe cases and healthy controls (***p < 0.001, ****p < 0.0001; unpaired two-sided Welch’s t-test).
Figure 4.
Multiple differential representation of proteins in sera from COVID-19 cases. Heatmap of proteins significantly dysregulated (Z-scored original value) in multiple COVID-19 cohorts (p < 0.05; unpaired two-sided Welch’s t-test). Clusters of proteins differentially represented in response to COVID-19 (cluster 1, overrepresented; cluster 2, underrepresented) are shown. Protein levels of four selected proteins with significant differences on each cluster were compared between groups by one-way ANOVA test followed by post-hoc Bonferroni and Holm multiple comparisons (f-values and p-values are shown) and unpaired two-sided Welch’s t-test (*p < 0.05, **p < 0.01, (***p < 0.001, ****p < 0.0001).
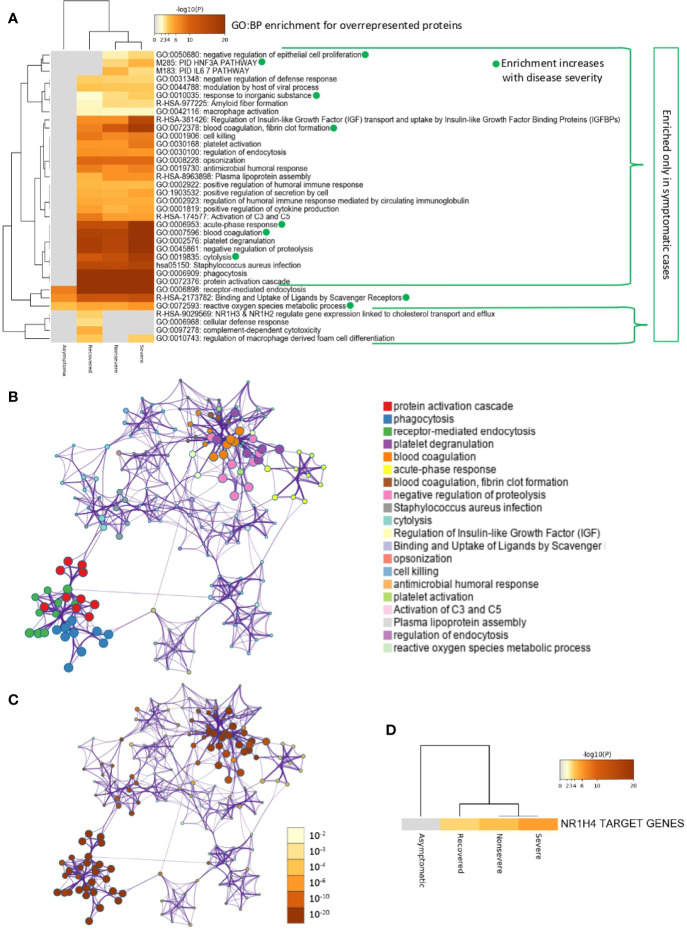
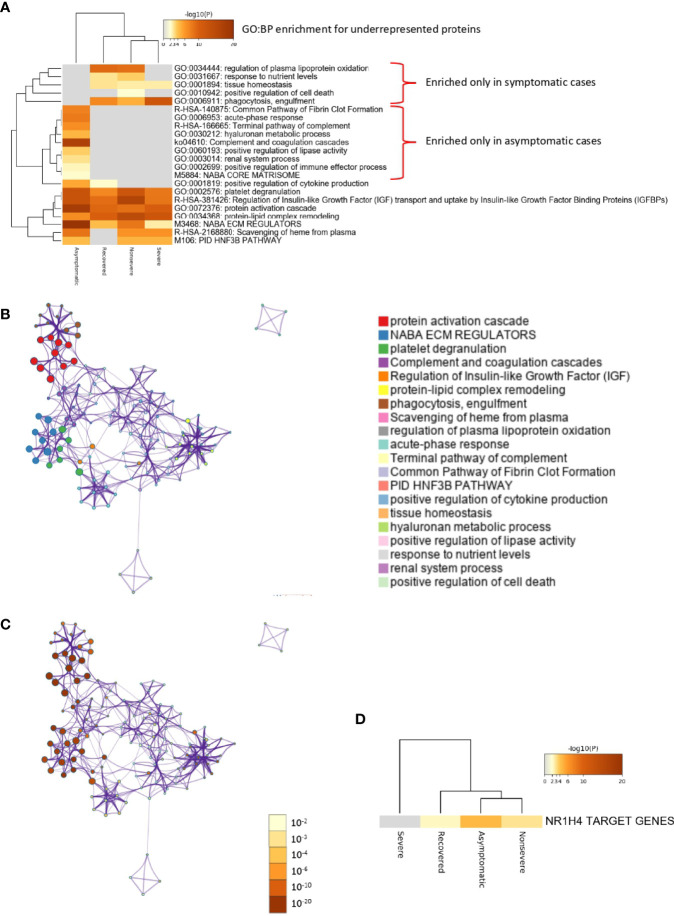
Of the multiple BPs affected by significantly dysregulated serum proteins, some were only enriched in symptomatic cases while others were enriched in asymptomatic cases (Figures 5A–D and 6A–D). For overrepresented proteins, enrichment increased with disease severity for BPs such as negative regulation of epithelial cell proliferation, FOXA1 transcription factor network (HNF3A pathway M285) coordinating function of primary airway epithelial cells, IL-6-mediated signaling events (M183), response to inorganic substance, blood coagulation, acute-phase response, cytolysis, binding and uptake of ligands by scavenger receptors, and reactive oxygen species metabolic process (Figure 5A). Underrepresented proteins were enriched only in both asymptomatic (e.g., common pathway of fibrin clot formation, acute-phase response, complement and coagulation cascade, hyaluronan metabolic process, positive regulation of lipase activity, renal system process, positive regulation of immune effector process and M5884 ensemble of genes encoding core extracellular matrix including ECM glycoproteins, collagens, and proteoglycans) and symptomatic (e.g., regulation of plasma lipoprotein oxidation, response to nutrient levels, tissue homeostasis, positive regulation of cell death, and phagocytosis) cases (Figure 6A).
Figure 5.
Enrichment ontology clusters for differentially overrepresented proteins in sera from COVID-19 cases. (A) Statistically enriched terms (GO/KEGG biological processes; GO : BP). Accumulative hypergeometric p-values and enrichment factors were calculated and used for filtering. Remaining significant terms were then hierarchically clustered into a tree based on Kappa-statistical similarities among their protein memberships (as used in DAVID Bioinformatics Resources 6.8; https://david.ncifcrf.gov). A 0.3 Kappa score was applied as the threshold to cast the tree into term clusters. The term with the best p-value within each cluster was selected as its representative term and displayed in a dendrogram. The heatmap cells are colored by their p-values; white cells indicate the lack of enrichment for that term in the corresponding gene list. BPs in which enrichment increased with disease severity only in symptomatic cases are shown. (B) Network of enriched terms. We selected a subset of representative terms from the full cluster and convert them into a network layout. More specifically, each term is represented by a circle node, where its size is proportional to the number of input genes that fall into that term, and its color represents its cluster identity (i.e., nodes of the same color belong to the same cluster). Terms with a similarity score > 0.3 are linked by an edge (the thickness of the edge represents the similarity score). The network is visualized with Cytoscape (v3.1.2) with “force-directed” layout and with edge bundled for clarity. One term from each cluster is selected to have its term description shown as label. (C) Network of enriched terms colored by p-value. The same enrichment network has its nodes colored by p-value, as shown in the legend. The darker the color, the more statistically significant the node is (see legend for p-value ranges). (D) Quality control and association analysis. Protein lists were identified in the ontology categories Transcription_Factor_Targets. All genes in the genome were used as the enrichment background. Terms with a p-value < 0.01, a minimum count of 3, and an enrichment factor (ratio between the observed counts and the counts expected by chance) > 1.5 were collected and grouped into clusters. The algorithm used here is the same as that used in the other enrichment analyses.
Figure 6.
Enrichment ontology clusters for differentially underrepresented proteins in sera from COVID-19 cases. (A) Statistically enriched terms (GO/KEGG biological processes; GO : BP). Accumulative hypergeometric p-values and enrichment factors were calculated and used for filtering. Remaining significant terms were then hierarchically clustered into a tree based on Kappa-statistical similarities among their protein memberships (as used in DAVID Bioinformatics Resources 6.8; https://david.ncifcrf.gov). A 0.3 Kappa score was applied as the threshold to cast the tree into term clusters. The term with the best p-value within each cluster was selected as its representative term and displayed in a dendrogram. The heatmap cells are colored by their p-values; white cells indicate the lack of enrichment for that term in the corresponding gene list. BPs enriched only in symptomatic or asymptomatic cases are shown. (B) Network of enriched terms. We selected a subset of representative terms from the full cluster and convert them into a network layout. More specifically, each term is represented by a circle node, where its size is proportional to the number of input genes that fall into that term, and its color represents its cluster identity (i.e., nodes of the same color belong to the same cluster). Terms with a similarity score > 0.3 are linked by an edge (the thickness of the edge represents the similarity score). The network is visualized with Cytoscape (v3.1.2) with “force-directed” layout and with edge bundled for clarity. One term from each cluster is selected to have its term description shown as label. (C) Network of enriched terms colored by p-value. The same enrichment network has its nodes colored by p-value, as shown in the legend. The darker the color, the more statistically significant the node is (see legend for p-value ranges). (D) Quality control and association analysis. Protein lists were identified in the ontology categories Transcription_Factor_Targets. All genes in the genome were used as the enrichment background. Terms with a p-value < 0.01, a minimum count of 3, and an enrichment factor (ratio between the observed counts and the counts expected by chance) > 1.5 were collected and grouped into clusters. The algorithm used here is the same as that used in the other enrichment analyses.
The network of interactions between proteins and BPs was characterized using Graph Theory algorithms (Supplementary Figure 2 and Data 2). While visually similar, networks have deep differences in their structure. Other than obvious changes of the proteins involved (presence/absence and representation), therefore affecting the BPs, nestedness showed a decreasing magnitude according to the patient cohorts. Nestedness is maximum for healthy and asymptomatic individuals (nestedness of 12.2 and 12.1, respectively), which reflects a high structuring of the clusters (Figure 7 and Supplementary Data 2). However, networks built using proteins and BPs for nonsevere and severe patients show a clear de-structuring (nestedness of 5.1 and 3.8, respectively). The networking built with data of recovered patients shows an intermediate structure without clear differences with other cohorts in this analysis (nestedness of 10.1). These results point to a clear pattern in which some proteins (rate of change > 0.900; Supplementary Data 2) such as neutrophil defensin 3, serum amyloid A (SAA) SAA2-SAA4 readthrough, Apolipoprotein C-IV, and Fibrinogen gamma chain are associated with nonsevere and severe COVID-19 patients, therefore increasing the PR index value of the BPs. It seems that overrepresentation of selected proteins in patients with higher COVID-19 symptomatology is blocking the normal regulation of these BPs, which resulted in higher PR values in these cohorts. Networks resulting from these cohorts are de-structured, and the structure with clear clusters observed in healthy individuals is not evident. Therefore, the networks produced with proteins and the BPs in the five cohorts show critical changes. These changes include the overrepresentation of some BPs such as negative regulation by a host of viral processes, negative regulation of mononuclear cell proliferation, positive regulation of interleukins, positive regulation of chemokine production, and positive regulation of respiratory burst involved in inflammatory response that remained unaltered in healthy and recovered individuals. These results support the idea that a network construct, based on pure statistical rules, reflects the clinical status commonly observed in critical COVID-19 patients.
Figure 7.
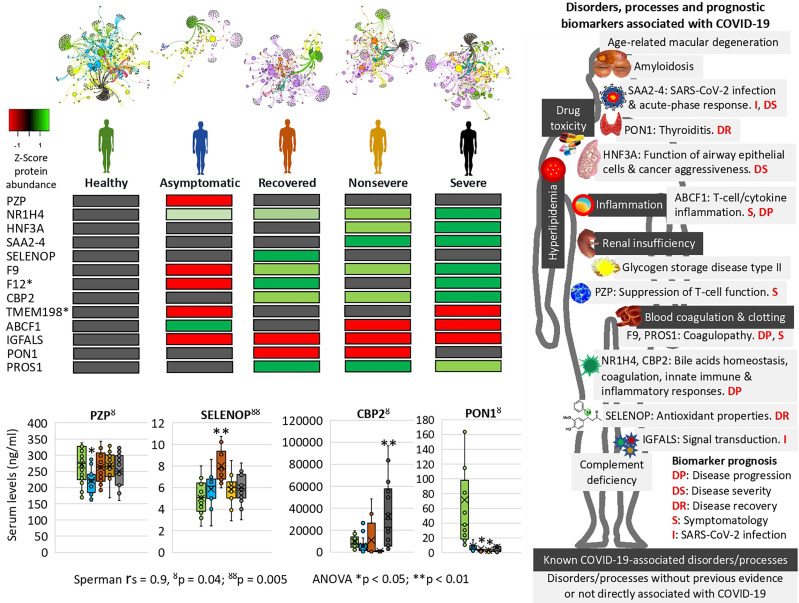
Prognostic biomarker proteins related to COVID-19 symptomatology. Network analysis of interactions between proteins and BPs reflects nestedness or structuring of the cluster’s magnitude decreasing with COVID-19 symptomatology. SWATH-MS quantitative serum proteomics identified proteins involved in physiological disorders and processes associated with COVID-19 and novel biomarker proteins with potential implications for the development of new diagnostic and therapeutic interventions to contribute to the control of this pandemic. *Unsupportive protein profile for prognostic biomarker. Selected serum biomarkers (PZP, SELENOP, CBP2, and PON1) were validated by ELISA. Change in protein serum levels with significant differences in comparison to healthy controls (*p < 0.05, **p < 0.01; one-way ANOVA test with post-hoc Tukey HSD). Proteomics and ELISA data were compared by Spearman’s Rho (rs) correlation analysis (ŏ p < 0.05, ŏŏ p < 0.01).
In correspondence with these BPs, the network of enriched terms showed that the most represented processes in proteins overrepresented in COVID-19 cohorts are protein activation cascade, phagocytosis, receptor-mediated endocytosis, platelet degranulation, blood coagulation, acute-phase response, negative regulation of proteolysis, Staphylococcus aureus infection, cytolysis, regulation of insulin-like growth factor (IGF), binding and uptake of ligands by scavenger, opsonization, cell killing, antimicrobial humoral response, platelet activation, activation of complement C3 and C5, plasma lipoprotein assembly, regulation of endocytosis, and reactive oxygen species metabolic process (Figures 5B, C). For underrepresented proteins, the most enriched processes were protein activation cascade, enzymes and their regulators involved in the remodeling of the extracellular matrix (NABA ECM regulators), platelet degranulation, complement and coagulation cascades, regulation of IGF, protein–lipid complex remodeling, phagocytosis, scavenging of heme from plasma, regulation of plasma lipoprotein oxidation, acute-phase response, terminal pathway of complement, pathway of fibrin clot formation, FOXA2 and FOXA3 transcription factor networks (HNF3B pathway M106), positive regulation of cytokine production, tissue homeostasis, hyaluronan metabolic process, positive regulation of lipase activity, response to nutrient levels, renal system process, and positive regulation of cell death (Figures 6B, C). The quality control and association analysis showed that network representation of nuclear receptor subfamily 1, group H, member 4 (NR1H4) target genes increased with disease severity (Figures 5D, 6D). Protein–protein interaction enrichment analysis resulted in complement, coagulation, and clotting cascades for overrepresented proteins and lipoprotein particle remodeling, reverse cholesterol transport, and peptide ligand-binding receptors for underrepresented proteins (Supplementary Figure 1).
Identification of Prognostic Biomarkers in Proteins Associated With COVID-19 Disease Symptomatology
For validation of serum proteomics data, an enrichment analysis was conducted using the Coronascape COVID database (https://metascape.org/COVID) to identify proteins found in our study as differentially represented in response to COVID-19 and reported in previous COVID-19 omics datasets as a correlate of disease severity (Supplementary Figures 3, 4). This analysis also identified proteins dysregulated in COVID-19 patients and potentially not previously associated with disease symptomatology (Supplementary Figure 5). Of these proteins, several were previously identified as biomarkers of severe COVID-19 in non-omics studies and were not included in further analyses (Supplementary Figure 5).
However, other proteins not previously identified in COVID-19 patients or with differences in the representation profile compared to our study were proposed as novel in relation to disease symptomatology and were used for prognostic biomarkers identification (Table 1 and Supplementary Figure 5). Of them, coagulation factor XII (F12) and transmembrane protein 198 (TMEM198) showed an unsupportive profile for biomarker prediction (Table 1 and Figure 7). TMEM198 has been associated with diabetes as observed in comorbidities of COVID-19 symptomatic cohorts included in the study (Supplementary Table 1).
Table 1.
Candidate prognostic biomarker proteins related to COVID-19 disease symptomatology.
| Proteins | Results of our study | Previous findings | Biomarker predictor | Refs |
|---|---|---|---|---|
| Selenoprotein P (SELENOP) | Overrepresented in recovered cases | Lower levels in COVID-19 patients | Disease recovery Validated by ELISA |
(38) |
| Coagulation factor IX (F9) | Overrepresented in all COVID-19 patients. Correlation with symptomatology | Decrease in protein levels from nonsevere to severe patients | Disease progression | (22, 39, 40) |
| Coagulation factor XII (F12) | Overrepresented in all but nonsevere COVID-19 patients | Not identified | Unsupportive profile | (40) |
| Carboxypeptidase B2 (CPB2) | Overrepresented in all but asymptomatic COVID-19 patients | Not identified | Disease severity Validated by ELISA |
(41) |
| Transmembrane protein 198 (TMEM198) | Underrepresented in asymptomatic and severe COVID-19 patients | Not identified | Unsupportive profile | (42, 43) |
| ATP-binding cassette sub-family F member 1 (ABCF1) | Overrepresented in asymptomatic and underrepresented in nonsevere and severe COVID-19 patients | Not identified | Symptomatology and disease progression | (44) |
| Insulin-like growth factor-binding protein complex acid labile subunit (IGFALS) | Underrepresented in all COVID-19 patients | Increase in protein levels from nonsevere to severe patients | SARS-CoV-2 infection | (22, 44) (45) (46) (47) |
| Serum paraoxonase/ arylesterase 1 (PON1) |
Underrepresented in nonsevere and recovered COVID-19 patients | Increase in protein levels from nonsevere to severe patients | Disease recovery Reduction in thyroiditis Validated by ELISA |
(22, 48, 49) |
| Pregnancy zone protein (PZP) | Underrepresented only in asymptomatic cases | Not identified | Symptomatology Validated by ELISA |
(50) |
| Vitamin K-dependent protein S (PROS1) | Overrepresented in recovered, nonsevere, and severe COVID-19 patients but with lower levels in severe cases | Associated with COVID-19 coagulopathy | Disease progression Symptomatology | (51) |
Proteins related to COVID-19 symptomatology and used for the identification of candidate prognosis biomarkers. Protein representation refers to significant differences when compared to healthy controls. Full data are disclosed in Supplementary Data 1 and Figures 5, 7.
Selected identified candidate prognostic immune-related biomarker proteins, PZP, Selenoprotein P (SELENOP), Carboxypeptidase B2 (CPB2), and Serum paraoxonase/arylesterase 1 (PON1) (Table 1), were validated by ELISA using sera from individuals of all cohorts included in the study (Figure 7). The results corroborated the predictive value of these biomarkers for disease recovery (SELENOP and PON1), severity (CBP2), and symptomatology (PZP).
Characterization of Differentially Represented Proteins in Response to COVID-19 and Associated to Other Human Diseases and Conditions to Monitor Risk Factors for Disease Symptomatology
An enrichment analysis was conducted using the DisGeNET discovery platform (https://www.disgenet.org) provided by Metascape (https://metascape.org) to identify proteins differentially represented in response to COVID-19 and associated to other human diseases and conditions with major affected physiological processes resulting in macrophage activation and coagulopathy (Figure 7 and Supplementary Figure 6). The results showed two main types of pathologies enriched with disease severity, renal insufficiency (acute kidney injury, acute kidney insufficiency, proteinuria, and nephrotic syndrome) and blood coagulation alterations (factor V Leiden mutation, activated protein C resistance, and lupus anticoagulant disorder). Alterations in blood coagulation are a consequence of the SARS-CoV-2 infection and the associated pro-inflammatory processes (52, 53). Three of the identified pathologies (factor V Leiden mutation, activated protein C resistance, and lupus anticoagulant disorder) are related to pro-coagulant alterations and have been clinically associated with COVID-19 coagulopathy (54). Renal insufficiency has been associated with poor COVID-19 prognosis (55), and the results correlated with renal disease comorbidity in COVID-19 symptomatic cohorts included in the study (Supplementary Table 1). Drug toxicity and adverse reaction to drug are likely associated with the patient’s response to drugs, which were supplied to all symptomatic patients (Supplementary Table 1). Hyperlipidemia but not complement deficiency disease correlated with clinical conditions in COVID-19 cohorts (Supplementary Table 1). Inflammation is a common condition in COVID-19 patients with increasing symptomatology with disease severity (Figure 1). Several of these disorders and COVID-19 disease severity are associated with positive regulation of interleukins (e.g., IL-6) (56) (Supplementary Figure 5A, Supplementary Data 2). However, in this study, we did not identify interleukins in the serum proteomics dataset, likely due to low protein levels in healthy and asymptomatic cases and interventions to control the so-called “cytokine storm” in symptomatic COVID-19 patients (Supplementary Figure 7). Other identified diseases such as amyloidosis, complement deficiency disease, age-related macular degeneration, and glycogen storage disease type II have not been previously directly associated with COVID-19 at least as evidenced in this study. These diseases and conditions may be used to monitor risk factors for COVID-19 disease symptomatology.
Discussion
In this study, SWATH-MS quantitative serum proteomics together with multiple data analysis algorithms was used to characterize host response to SARS-CoV-2 infection in different cohorts from asymptomatic individuals to severe patients. Due to the complexity of COVID-19 symptomatology, this approach contributed to a better understanding of disease mechanisms and symptoms with possible medical complications at different levels and the identification of potential diagnostic/prognostic biomarkers and drug targets (22–25). The results corroborated previous findings in COVID-19 studies and highlighted how the representation of dysregulated serum proteins and associated BPs increases with COVID-19 disease symptomatology from asymptomatic to severe cases (4–6, 8–13, 18, 22–25). However, the analysis was focused on results that provided new insights into COVID-19 disease symptomatology and potential biomarker proteins for diagnostic and therapeutic interventions (Figure 7).
Of the significantly dysregulated proteins, selected immune-related proteins PZP, SELENOP, PON1, and CBP2 were validated as candidate prognostic biomarkers for COVID-19 symptomatology (Table 1 and Figure 7). Of them, PZP was underrepresented in asymptomatic cases only. This protein is a broad-spectrum immunosuppressive protein that suppresses T-cell function during pregnancy to prevent fetal rejection, and its overrepresentation correlates with airway infection and bronchiectasis disease severity (50). Consequently, serum PZP protein levels may be used as a biomarker for COVID-19 disease symptomatology and prognosis of asymptomatic carriers. Selenoprotein levels related to selenium (Se) status affect immune defense and tissue homeostasis through its effect on the trafficking of tissue macrophages (57, 58), and thus SELENOP may be used as a biomarker for disease recovery. PONs have the capacity to protect cells from oxidative stress and are implicated in the pathogenesis of inflammatory diseases (59, 60). Findings suggest a role for PON1 against atherosclerosis and obesity and protective capacity against bacterial, parasitic, and viral infectious diseases (59). Regarding COVID-19, PON1 has been shown to increase in protein levels from nonsevere to severe patients (22) and we found the protein underrepresented in nonsevere and recovered patients, thus suggesting a biomarker for disease recovery. CPB2 appears to have a role in innate immunity through inactivation of complement component C5a, which can induce inflammatory pathways via C5aR receptor (41, 61). In our study, CPB2 was overrepresented in all but asymptomatic COVID-19 patients, thus providing a candidate biomarker for disease severity. As expected, the serum levels of these biomarkers correlated with the anti-SARS-CoV-2 Spike IgG levels previously shown to significantly increase from asymptomatic to severe cohorts included in this study (28).
Enrichment analyses were used to identify prognostic biomarker proteins and association to other human diseases and conditions (Figure 7 and Supplementary Figure 6). The BP enrichment and association analyses showed that network representation of nuclear receptor subfamily 1, group H, member 4 (NR1H4) target genes increased with COVID-19 disease severity (Figure 7). The farnesoid X receptor (FXR, NR1H4) encodes a ligand-activated transcription factor, which shares structural features in common with nuclear hormone receptor family that functions as a receptor for bile acids (BA) and regulation of the expression of genes involved in bile acid synthesis and transport, lipid and glucose homeostasis, and innate immune and inflammatory responses (62). NR1H4 is essential for BA homeostasis while FXR and its hepatic and intestinal target genes transcriptionally regulate BA synthesis, detoxification, secretion, and absorption in the enterohepatic circulation. Furthermore, FXR agonists as well as a fibroblast growth factor 19 (FGF19) analogue are currently tested in clinical trials for different cholestatic liver diseases (57). The FOXA1 transcription factor network (HNF3A pathway M285) BP with overrepresented proteins in response to COVID-19 increased in enrichment with disease severity (Figure 7). This pathway (https://www.gsea-msigdb.org/gsea/msigdb/cards/PID_HNF3A_PATHWAY) coordinates function of primary airway epithelial cells (63) and has been associated with more aggressive breast (64) and prostate cancer (65). Accordingly, considering disorders and processes associated with COVID-19, these proteins may be proposed as candidate prognosis biomarkers for disease progression and severity (Figure 7).
The network of interactions between proteins and BPs characterized using Graph Theory algorithms reflected patterns in correlation with COVID-19 disease severity (Figure 7 and Supplementary Figure 2). A distinctive finding using this approach was the acute-phase response SAA2–SAA4 (SAA2–4) readthrough proteins, whose overrepresentation was associated with nonsevere and severe COVID-19 patients (Figure 7). The SAA2 has been used to monitor the severity of COVID-19 and as a biomarker for SARS-CoV-2 infection (4). However, the increase in the expression of SAA2–4 coding acute-phase reactant genes or serum protein levels has not been directly associated with COVID-19 patients but with clear cell renal carcinoma (66) and lung cells (67). Therefore, these proteins constitute biomarkers for SARS-CoV-2 infection and prognosis of disease severity (Figure 7).
Other novel prognostic biomarker proteins related to COVID-19 disease symptomatology were identified (Table 1 and Figure 7) (22, 68). These biomarkers included potential prognostic tools for SARS-CoV-2 infection, disease symptomatology, progression and recovery, and reduction in thyroiditis. To contribute to the application of these findings in the clinic, some of these prognostic biomarkers were validated using protein-specific ELISA tests (Figure 7) and could be incorporated into the daily routine for disease diagnosis/prognosis. Recently, the glycoprotein Galectin-9 (Gal-9) involved in innate immunity and associated with cytokine release syndrome was identified as a surrogate diagnostic biomarker in SARS-CoV-2 infection (69). In our proteomics study, Gal-9 was not identified, but in accordance with these results, the Galectin-3-binding protein (Gal-3BP) with a role in innate immune response to viruses (70) was significantly overrepresented in all symptomatic COVID-19 cohorts (Supplementary Data 1).
At the level of other human diseases and conditions, findings revealed potential disorders associated with COVID-19 (Figure 7 and Supplementary Figure 6). Hyperlipidemia and other forms of dyslipidemia have been associated with COVID-19 severity (71) and may be related to FXR and NR1H4 BP enrichment. Amyloidosis in its different forms is caused by deposition of immunoglobulin light chains and have not been previously associated with COVID-19 except for the management of patients with this condition (72). Accordingly, immunoglobulin lambda and kappa variable light chains were overrepresented in nonsevere and severe patients when compared to healthy individuals (Figures 3A, C). Another interesting finding was the complement deficiency disease (Figure 7). The complement cascade that is directly associated with blood coagulation alterations (73, 74) has been implicated in COVID-19 pathology (Supplementary Figure 4) (75). However, in our study, complement, coagulation, and clotting cascades were clearly directly associated with COVID-19 severity, which may explain the association with complement deficiency disease and thrombosis disorders. One of the pathologies identified in our analysis was the age-related macular degeneration (Figure 7). This pathology is directly associated with dysregulation of complement regulators such as factor H, which is treated with these factors as therapeutic interventions (76) and has not been associated with COVID-19 but with the primary systemic amyloidosis identified here as enriched with disease severity (Figure 7) (77). Another pathology identified as a correlate of disease severity was glycogen storage disease type II, a lysosomal disease not previously related to COVID-19. The immunity to glycan Galα1-3Galβ1-(3)4GlcNAc-R (α-Gal), which was recently related to tick bites and allergic reactions to mammalian meat consumption (alpha-gal syndrome) (78, 79), has been implicated in the protective response to COVID-19 (28, 80). Complement component C3 and hemoglobin subunit beta (HBB) were associated with the immune response to α-Gal in the zebrafish animal model (81) and were both significantly overrepresented in COVID-19 patients when compared to healthy individuals (Supplementary Data 1). In humans, the endogenous source of α-Gal is gut bacteria (78), and glycan metabolism has a key role in shaping microbiota composition (82). Therefore, the dysregulation in C3 and HBB serum protein levels observed in COVID-19 cohorts and previously reported in response to α-Gal51 may be due to gut microbiota dysbiosis associated to SARS-CoV-2 infection and COVID-19 severity (83, 84) (Supplementary Figure 8). Apolipoprotein A (APOA) isoforms A-I, A-II, and A-IV were significantly dysregulated in COVID-19 patients and serum protein levels decreased with disease symptomatology (Figure 4 and Supplementary Data 1). Lipoprotein(a)-containing APOAs are endogenous triggers of innate immunity and can induce trained immunity (TRIM) (85), thus suggesting that TRIM associated with bacillus Calmette-Guérin (BCG) vaccination may be affected in COVID-19 patients (86, 87) (Supplementary Figure 8). Altogether, these disorders and physiological processes should be considered to improve monitoring of COVID-19 symptomatology and as potential targets for therapeutic interventions to reduce the risk for severe symptoms and mortality (23, 24).
A better understanding of COVID-19 on human molecular pathophysiology is required for the identification of new biomarkers and diagnostic and therapeutic targets. By August 9, 2021, 56 publications appear in PubMed with search keywords “covid AND serum AND proteomic” (https://pubmed.ncbi.nlm.nih.gov/?term=covid+serum+proteomic&sort=date). These publications confirmed previous results in studies with different cohorts, populations, and settings and/or provided new serum biomarkers related to disease progression and symptomatology. For example, among the latest publications on this list, Pavel et al. (88) confirmed the association between Th2/Th1 cytokine imbalance and COVID-19 risk mortality; Singh et al. (89) confirmed the increase in serum inflammatory markers in COVID-19 patients; Mitamura et al. (90) confirmed cytokine storm in severe COVID-19 patients; Lazari et al. (91) confirmed and validated SAA1 and SAA2 proteins as biomarkers in low- and high-risk COVID-19 patients; Völlmy et al. (92) proposed various serum proteins as biomarkers to predict mortality in COVID-19 patients; Geyer et al. (93) showed a functional association between serum proteins, biological processes, and clinical parameters between COVID-19 patients and symptomatic but PCR-negative individuals; Laudanski et al. (94) identified serum proteins with potential role in COVID-19 pathology; and Gutmann et al. (95) found mannose binding lectin 2 and pentraxin-3 (PTX3) of the innate immune system as positively associated with COVID-19 mortality.
Our study is the first to provide serum proteomic profiles of cohorts of SARS-CoV-2-infected recovered (hospital discharge), nonsevere (hospitalized), and severe (ICU) cases with increasing systemic inflammation in comparison with healthy individuals sampled prior to the COVID-19 pandemic. The results not only confirmed previous results but provided new serum biomarkers, BPs, and physiological disorders related to disease progression and symptomatology (Figure 7). The confirmation of previous results in studies conducted with different cohorts and populations as shown here for the first time in Spain is important to validate diagnostic and therapeutic interventions at a global scale affecting this pandemic. The new prognostic biomarkers associated with COVID-19 reported here not only serve in conjunction with diagnostic RNA, antigen, and antibody detection tests to complement other previously identified biomarkers such as IL-6, but also provide the possibility of using highly abundant serum proteins for prognosis of disease severity (e.g., CBP2, up to 0.1 mg/ml), asymptomatic carriers (e.g., PZP, up to 350 ng/ml), or disease recovery (e.g., PON1, up to 160 ng/ml). The disorders and processes associated with the new biomarkers identified in this study provide clinical tools for the evaluation and treatment of SARS-CoV-2 infection and disease symptomatology and progression (Figure 7). For example, detection of high HNF3A levels in nonsevere or severe patients suggests their diagnosis and treatment to reduce airway dilatation with production of large cysts associated with function of airway epithelial cells (96).
The main limitations of this study include the following (a) possible effect on serum protein representation of immunosuppressive treatments to control the cytokine storm in symptomatic COVID-19 patients (Supplementary Table 1); (b) impact of comorbidities associated or not to COVID-19 (Supplementary Table 1); (c) serum samples were collected when the main circulating SARS-CoV-2 variant was WIV04/2019 and thus possible differences with other variants in the serum protein response to infection and caused pathologies should be considered; (d) although serum proteomics analysis was conducted with samples on each cohort including three pools of 5–10 individuals each (Figure 1), studies with a larger number of samples and/or on individual cases may provide case-by-case differences in serum protein representation; and (e) as samples were collected from a retrospective study (28), the effect of some factors such as age (oldest in nonsevere cases; Figure 1) but not sex ratio (similar in all groups; Figure 1) may affect protein representation. However, because age did not show significant differences between severe and asymptomatic or heathy cohorts, possible differences in age-related serum protein representation should not affect the main results of the study.
In conclusion and despite these limitations, the SWATH-MS quantitative serum proteomics used in our study together with multiple data analysis algorithms contributed to the characterization of SARS-CoV-2–host molecular interactions and advanced translational medicine by identifying prognostic biomarker proteins and physiological disorders with potential implications for disease diagnosis/prognosis contributing to the control of the COVID-19 pandemic. The identified biomarkers for disease recovery (SELENOP and PON1), severity (CBP2), and symptomatology (PZP) could be used for disease prognosis. For example, in some cases, hospitalized nonsevere patients could progress to disease recovery (hospital discharge) or severity (ICU). In our study, the results showed that some of these biomarkers may be used to evaluate the risk of hospitalized patients to develop severe symptoms.
Data Availability Statement
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD024549 and 10.6019/PXD024549. The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Ethics Statement
The studies involving human participants were reviewed and approved by Ethical and Scientific Committees (University Hospital of Ciudad Real C-352 and SESCAM C-73). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Author Contributions
JF, JU, MV, and CG designed the study. MV, SA-J, EF-C, MC, and IGF performed the experiments. MV, JF, JU, FR-d-R, NJ-C, and AE-P collected and analyzed data. JF, CG, JU, IGF, and MV supervised the project. All authors wrote the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This research was partially funded by Junta de Comunidades de Castilla-La Mancha (JCCM), Spain and EU-FEDER (grants MYCOTRAINING SBPLY/19/180501/000174 and GALINFEC SBPLY/17/180501/000185). EF-C has a predoctoral grant from UCLM. MC was funded by the Ministerio de Ciencia, Innovación y Universidades, Spain (grant FJC-2018-038277-I). IGF was supported by UCLM. MV was supported by UCLM and EU-FEDER.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We acknowledge the University of Castilla La Mancha (UCLM, Spain) for support to Group SaBio.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2021.730710/full#supplementary-material
Analysis of differentially represented proteins in response to COVID-19.
High-resolution network analysis of interactions between proteins and BPs characterized using Graph Theory algorithms.
Serum proteomics data for previously identified biomarkers for COVID-19 disease severity.
Metascape analysis of differentially represented proteins and previously reported in COVID-19 omics datasets.
Differential representation of proteins not previously reported in COVID-19 omics datasets.
Association of differentially represented proteins in response to COVID-19 to other human diseases and conditions.
Cytokine (IL-1 and IL-4) response in COVID-19 symptomatic patients and healthy controls.
Mechanisms of potential disorders associated with COVID-19.
Clinical parameters and comorbidities in COVID-19 asymptomatic and symptomatic cohorts.
Serum proteomics analysis.
Network analysis of interactions between proteins and BPs characterized using Graph Theory algorithms. Please see the Supplementary Material section of the Author guidelines for details on the different file types accepted.
References
- 1.Alm E, Broberg EK, Connor T, Hodcroft EB, Komissarov AB, Maurer-Stroh S, et al. WHO European Region Sequencing Laboratories and GISAID EpiCoV Group; WHO European Region Sequencing Laboratories and GISAID EpiCoV Group. Geographical and Temporal Distribution of SARS-CoV-2 Clades in the WHO European Region, January to June 2020. Euro Surveill (2020) 25(32):2001410. 10.2807/1560-7917.ES.2020.25.32.2001410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mortaz E, Tabarsi P, Varahram M, Folkerts G, Adcock IM. The Immune Response and Immunopathology of COVID-19. Front Immunol (2020) 11:2037. 10.3389/fimmu.2020.02037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gortázar C, Del-Río FJR, Domínguez L, de la Fuente J. Host or Pathogen-Related Factors in COVID-19 Severity? Lancet (2020) 396(10260):1396–7. 10.1016/S0140-6736(20)32215-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shen B, Yi X, Sun Y, Bi X, Du J, Zhang C, et al. Proteomic and Metabolomic Characterization of COVID-19 Patient Sera. Cell (2020) 182(1):59–72.e15. 10.1016/j.cell.2020.05.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shu T, Ning W, Wu D, Xu J, Han Q, Huang M, et al. Plasma Proteomics Identify Biomarkers and Pathogenesis of COVID-19. Immunity (2020) 53(5):1108–22.e5. 10.1016/j.immuni.2020.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Messner CB, Demichev V, Wendisch D, Michalick L, White M, Freiwald A, et al. Ultra-High-Throughput Clinical Proteomics Reveals Classifiers of COVID-19 Infection. Cell Syst (2020) 11(1):11–24.e4. 10.1016/j.cels.2020.05.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sperk M, van Domselaar R, Rodriguez JE, Mikaeloff F, Sá Vinhas B, Saccon E, et al. Utility of Proteomics in Emerging and Re-Emerging Infectious Diseases Caused by RNA Viruses. J Proteome Res (2020) 19(11):4259–74. 10.1021/acs.jproteome.0c00380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li J, Guo M, Tian X, Wang X, Yang X, Wu P, et al. Virus-Host Interactome and Proteomic Survey Reveal Potential Virulence Factors Influencing SARS-CoV-2 Pathogenesis. Med (NY) (2021) 2(1):99–112.e7. 10.1016/j.medj.2020.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yang Y, Shen C, Li J, Yuan J, Wei J, Huang F, et al. Plasma IP-10 and MCP-3 Levels Are Highly Associated With Disease Severity and Predict the Progression of COVID-19. J Allergy Clin Immunol (2020) 146(1):119–27.e4. 10.1016/j.jaci.2020.04.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wei X, Su J, Yang K, Wei J, Wan H, Cao X, et al. Elevations of Serum Cancer Biomarkers Correlate With Severity of COVID-19. J Med Virol (2020) 92(10):2036–41. 10.1002/jmv.25957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Overmyer KA, Shishkova E, Miller IJ, Balnis J, Bernstein MN, Peters-Clarke TM, et al. Large-Scale Multi-Omic Analysis of COVID-19 Severity. Cell Syst (2021) 12(1):23–40.e7. 10.1016/j.cels.2020.10.003 S2405-4712(20)30371-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.D’Alessandro A, Thomas T, Dzieciatkowska M, Hill RC, Francis RO, Hudson KE, et al. Serum Proteomics in COVID-19 Patients: Altered Coagulation and Complement Status as a Function of IL-6 Level. J Proteome Res (2020) 19(11):4417–27. 10.1021/acs.jproteome.0c00365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Su Y, Chen D, Yuan D, Lausted C, Choi J, Dai CL, et al. Multi-Omics Resolves a Sharp Disease-State Shift Between Mild and Moderate COVID-19. Cell (2020) 183(6):1479–95.e20. 10.1016/j.cell.2020.10.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu M, Chen Y, Xia H, Wang C, Tan CY, Cai X, et al. Transcriptional and Proteomic Insights Into the Host Response in Fatal COVID-19 Cases. Proc Natl Acad Sci USA (2020) 117(45):28336–43. 10.1073/pnas.2018030117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bojkova D, Klann K, Koch B, Widera M, Krause D, Ciesek S, et al. Proteomics of SARS-CoV-2-Infected Host Cells Reveals Therapy Targets. Nature (2020) 583(7816):469–72. 10.1038/s41586-020-2332-7 [DOI] [PubMed] [Google Scholar]
- 16.Rieder M, Wirth L, Pollmeier L, Jeserich M, Goller I, Baldus N, et al. Serum Protein Profiling Reveals a Specific Upregulation of the Immunomodulatory Protein Progranulin in COVID-19. J Infect Dis (2021) 223(5):775–84. 10.1093/infdis/jiaa741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tian W, Zhang N, Jin R, Feng Y, Wang S, Gao S, et al. Immune Suppression in the Early Stage of COVID-19 Disease. Nat Commun (2020) 11(1):5859. 10.1038/s41467-020-19706-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Begue F, Tanaka S, Mouktadi Z, Rondeau P, Veeren B, Diotel N, et al. Altered High-Density Lipoprotein Composition and Functions During Severe COVID-19. Sci Rep (2021) 11(1):2291. 10.1038/s41598-021-81638-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yan L, Yi J, Huang C, Zhang J, Fu S, Li Z, et al. Rapid Detection of COVID-19 Using MALDI-TOF-Based Serum Peptidome Profiling. Anal Chem (2021) 93(11):4782–7. 10.1021/acs.analchem.0c04590 [DOI] [PubMed] [Google Scholar]
- 20.Liu X, Cao Y, Fu H, Wei J, Chen J, Hu J, et al. Proteomics Analysis of Serum From COVID-19 Patients. ACS Omega (2021) 6(11):7951–8. 10.1021/acsomega.1c00616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Barberis E, Vanella VV, Falasca M, Caneapero V, Cappellano G, Raineri D, et al. Circulating Exosomes Are Strongly Involved in SARS-CoV-2 Infection. Front Mol Biosci (2021) 8:632290. 10.3389/fmolb.2021.632290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee JS, Han D, Kim SY, Hong KH, Jang MJ, Kim MJ, et al. Longitudinal Proteomic Profiling Provides Insights Into Host Response and Proteome Dynamics in COVID-19 Progression. Proteomics (2021) 4:e2000278. 10.1002/pmic.202000278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen Y, Yao H, Zhang N, Wu J, Gao S, Guo J, et al. Proteomic Analysis Identifies Prolonged Disturbances in Pathways Related to Cholesterol Metabolism and Myocardium Function in the COVID-19 Recovery Stage. J Proteome Res (2021) 20(7):3463–74. 10.1021/acs.jproteome.1c00054 [DOI] [PubMed] [Google Scholar]
- 24.Praissman J, Wells L. Proteomics-Based Insights Into the SARS-CoV-2 Mediated COVID-19 Pandemic: A Review of the First Year of Research. Mol Cell Proteomics (2021) 20:100103. 10.1016/j.mcpro.2021.100103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Doykov I, Hällqvist J, Gilmour KC, Grandjean L, Mills K, Heywood WE. ‘The Long Tail of Covid-19’ - The Detection of a Prolonged Inflammatory Response After a SARS-CoV-2 Infection in Asymptomatic and Mildly Affected Patients. F1000Res (2020) 9:1349. 10.12688/f1000research.27287.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jaiswal S, Kumar M, Mandeep, Sunita, Singh Y, Shukla P. Systems Biology Approaches for Therapeutics Development Against COVID-19. Front Cell Infect Microbiol (2020) 10:560240. 10.3389/fcimb.2020.560240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rana R, Rathi V, Ganguly NK. A Comprehensive Overview of Proteomics Approach for COVID 19: New Perspectives in Target Therapy Strategies. J Proteins Proteom (2020) 2:1–10. 10.1007/s42485-020-00052-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Urra JM, Ferreras-Colino E, Contreras M, Cabrera CM, Fernández de Mera IG, Villar M, et al. The Antibody Response to the Glycan α-Gal Correlates With COVID-19 Disease Symptoms. J Med Virol (2020) 3:10.1002/jmv.26575. 10.1002/jmv.26575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.de la Fuente J, Urra JM, Contreras M, Pacheco I, Ferreras-Colino E, Doncel-Pérez E, et al. A Dataset for the Analysis of Antibody Response to Glycan Alpha-Gal in Individuals With Immune-Mediated Disorders. F1000Res (2020) 9:1366. 10.12688/f1000research.27495.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gillet LC, Navarro P, Tate S, Röst H, Selevsek N, Reiter L, et al. Targeted Data Extraction of the MS/MS Spectra Generated by Data-Independent Acquisition: A New Concept for Consistent and Accurate Proteome Analysis. Mol Cell Proteomics (2012) 11(6):O111.016717. 10.1074/mcp.O111.016717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Perez-Riverol Y, Csordas A, Bai J, Bernal-Llinares M, Hewapathirana S, Kundu DJ, et al. The PRIDE Database and Related Tools and Resources in 2019: Improving Support for Quantification Data. Nucleic Acids Res (2019) 47(D1):D442–50. 10.1093/nar/gky1106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou Y, Zhou B, Pache L, Chang M, Khodabakhshi AH, Tanaseichuk O, et al. Metascape Provides a Biologist-Oriented Resource for the Analysis of Systems-Level Datasets. Nat Commun (2019) 10(1):1523. 10.1038/s41467-019-09234-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Estrada-Peña A, Villar M, Artigas-Jerónimo S, López V, Alberdi P, Cabezas-Cruz A, et al. Use of Graph Theory to Characterize Human and Arthropod Vector Cell Protein Response to Infection With Anaplasma Phagocytophilum. Front Cell Infect Microbiol (2018) 8:265. 10.3389/fcimb.2018.00265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Halu A, Mondragón RJ, Panzarasa P, Bianconi G. Multiplex PageRank. PloS One (2013) 8(10):e78293. 10.1371/journal.pone.0078293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dubois D, Prade H. What Are Fuzzy Rules and How to Use Them. Fuzzy sets Syst (1996) 84(2):169–85. 10.1016/0165-0114(96)00066-8 [DOI] [Google Scholar]
- 36.Payrató-Borràs C, Hernández L, Moreno Y. Breaking the Spell of Nestedness: The Entropic Origin of Nestedness in Mutualistic Systems. Phys Rev X (2019) 9:031024. 10.1103/PhysRevX.9.031024 [DOI] [Google Scholar]
- 37.Galeano J, Pastor JM, Iriondo JM. Weighted-Interaction Nestedness Estimator (WINE): A New Estimator to Calculate Over Frequency Matrices. Environ Modelling Software (2009) 24:1342–6. 10.1016/j.envsoft.2009.05.014 [DOI] [Google Scholar]
- 38.Heller RA, Sun Q, Hackler J, Seelig J, Seibert L, Cherkezov A, et al. Prediction of Survival Odds in COVID-19 by Zinc, Age and Selenoprotein P as Composite Biomarker. Redox Biol (2021) 38:101764. 10.1016/j.redox.2020.101764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bowles L, Platton S, Yartey N, Dave M, Lee K, Hart DP, et al. Lupus Anticoagulant and Abnormal Coagulation Tests in Patients With Covid-19. N Engl J Med (2020) 383(3):288–90. 10.1056/NEJMc2013656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Martín-Rojas RM, Pérez-Rus G, Delgado-Pinos VE, Domingo-González A, Regalado-Artamendi I, Alba-Urdiales N, et al. COVID-19 Coagulopathy: An in-Depth Analysis of the Coagulation System. Eur J Haematol (2020) 105(6):741–50. 10.1111/ejh.13501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Leung LLK, Morser J. Carboxypeptidase B2 and Carboxypeptidase N in the Crosstalk Between Coagulation, Thrombosis, Inflammation, and Innate Immunity. J Thromb Haemost (2018). 10.1111/jth.14199 [DOI] [PubMed] [Google Scholar]
- 42.Liang J, Fu Y, Cruciat CM, Jia S, Wang Y, Tong Z, et al. Transmembrane Protein 198 Promotes LRP6 Phosphorylation and Wnt Signaling Activation. Mol Cell Biol (2011) 31(13):2577–90. 10.1128/MCB.05103-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Moparthi L, Koch S. Wnt Signaling in Intestinal Inflammation. Differentiation (2019) 108:24–32. 10.1016/j.diff.2019.01.002 [DOI] [PubMed] [Google Scholar]
- 44.Powell TR, Tansey KE, Breen G, Farmer AE, Craig IW, Uher R, et al. ATP-Binding Cassette Sub-Family F Member 1 (ABCF1) Is Identified as a Putative Therapeutic Target of Escitalopram in the Inflammatory Cytokine Pathway. J Psychopharmacol (2013) 27(7):609–15. 10.1177/0269881113490329 [DOI] [PubMed] [Google Scholar]
- 45.Zhang L, Song QF, Jin JJ, Huang P, Wang ZP, Xie XF, et al. Differential Protein Analysis of Serum Exosomes Post-Intravenous Immunoglobulin Therapy in Patients With Kawasaki Disease. Cardiol Young (2017) 27(9):1786–96. 10.1017/S1047951117001433 [DOI] [PubMed] [Google Scholar]
- 46.Minghui Z, Kunhua H, Yunwen B, Hongmei L, Jing L, Shaowen W, et al. Analysis of Differentially Expressed Proteins Involved in Autoimmune Cirrhosis and Normal Serum by iTRAQ Proteomics. Proteomics Clin Appl (2019) 13(3):e1700153. 10.1002/prca.201700153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Winn BJ. Is There a Role for Insulin-Like Growth Factor Inhibition in the Treatment of COVID-19-Related Adult Respiratory Distress Syndrome? Med Hypotheses (2020) 144:110167. 10.1016/j.mehy.2020.110167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Aviram M. Introduction to Paraoxonases. J Lipids (2012) 2012:687273. 10.1155/2012/687273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Marasinghe SSK, Sivakanesan R. Increased Paraoxonase and Arylesterase Activity in Thyroiditis Patients Compared to Healthy Individuals: PS137. Porto BioMed J (2017) 2(5):178. 10.1016/j.pbj.2017.07.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Finch S, Shoemark A, Dicker AJ, Keir HR, Smith A, Ong S, et al. Pregnancy Zone Protein Is Associated With Airway Infection, Neutrophil Extracellular Trap Formation, and Disease Severity in Bronchiectasis. Am J Respir Crit Care Med (2019) 200(8):992–1001. 10.1164/rccm.201812-2351OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chatterjee S, Sengupta T, Majumder S, Majumder R. COVID-19: A Probable Role of the Anticoagulant Protein S in Managing COVID-19-Associated Coagulopathy. Aging (Albany NY) (2020) 12(16):15954–61. 10.18632/aging.103869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Iba T, Levy JH, Levi M, Thachil J. Coagulopathy in COVID-19. J Thromb Haemost (2020) 18(9):2103–9. 10.1111/jth.14975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Iba T, Connors JM, Levy JH. The Coagulopathy, Endotheliopathy, and Vasculitis of COVID-19. Inflammation Res (2020) 69(12):1181–9. 10.1007/s00011-020-01401-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Iba T, Levy JH, Connors JM, Warkentin TE, Thachil J, Levi M. The Unique Characteristics of COVID-19 Coagulopathy. Crit Care (2020) 24(1):360. 10.1186/s13054-020-03077-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, et al. Kidney Disease Is Associated With in-Hospital Death of Patients With COVID-19. Kidney Int (2020) 97(5):829–38. 10.1016/j.kint.2020.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Broman N, Rantasärkkä K, Feuth T, Valtonen M, Waris M, Hohenthal U, et al. IL-6 and Other Biomarkers as Predictors of Severity in COVID-19. Ann Med (2020) 11:1–5. 10.1080/07853890.2020.1840621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Carlson BA, Yoo MH, Sano Y, Sengupta A, Kim JY, Irons R, et al. Selenoproteins Regulate Macrophage Invasiveness and Extracellular Matrix-Related Gene Expression. BMC Immunol (2009) 10:57. 10.1186/1471-2172-10-57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Huang Z, Rose AH, Hoffmann PR. The Role of Selenium in Inflammation and Immunity: From Molecular Mechanisms to Therapeutic Opportunities. Antioxid Redox Signal (2012) 16(7):705–43. 10.1089/ars.2011.4145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Farid AS, Horii Y. Modulation of Paraoxonases During Infectious Diseases and Its Potential Impact on Atherosclerosis. Lipids Health Dis (2012) 11:92. 10.1186/1476-511X-11-92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Teiber JF, Horke S, Haines DC, Chowdhary PK, Xiao J, Kramer GL, et al. Dominant Role of Paraoxonases in Inactivation of the Pseudomonas Aeruginosa Quorum-Sensing Signal N-(3-Oxododecanoyl)-L-Homoserine Lactone. Infect Immun (2008) 76(6):2512–9. 10.1128/IAI.01606-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Merle NS, Noe R, Halbwachs-Mecarelli L, Fremeaux-Bacchi V, Roumenina LT. Complement System Part II: Role in Immunity. Front Immunol (2015) 6:257. 10.3389/fimmu.2015.00257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Keitel V, Dröge C, Häussinger D. Targeting FXR in Cholestasis. Handb Exp Pharmacol (2019) 256:299–324. 10.1007/164_2019_231 [DOI] [PubMed] [Google Scholar]
- 63.Paranjapye A, Mutolo MJ, Ebron JS, Leir SH, Harris A. The FOXA1 Transcriptional Network Coordinates Key Functions of Primary Human Airway Epithelial Cells. Am J Physiol Lung Cell Mol Physiol (2020) 319(1):L126–36. 10.1152/ajplung.00023.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kumar U, Ardasheva A, Mahmud Z, Coombes RC, Yagüe E. FOXA1 Is a Determinant of Drug Resistance in Breast Cancer Cells. Breast Cancer Res Treat (2021) 186(2):317–26. 10.1007/s10549-020-06068-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Teng M, Zhou S, Cai C, Lupien M, He HH. Pioneer of Prostate Cancer: Past, Present and the Future of FOXA1. Protein Cell (2021) 12(1):29–38. 10.1007/s13238-020-00786-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zamora-Fuentes JM, Hernández-Lemus E, Espinal-Enríquez J. Gene Expression and Co-Expression Networks Are Strongly Altered Through Stages in Clear Cell Renal Carcinoma. Front Genet (2020) 11:578679. 10.3389/fgene.2020.578679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.McCall MN, Illei PB, Halushka MK. Complex Sources of Variation in Tissue Expression Data: Analysis of the GTEx Lung Transcriptome. Am J Hum Genet (2016) 99(3):624–35. 10.1016/j.ajhg.2016.07.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Vabret N, Britton GJ, Gruber C, Hegde S, Kim J, Kuksin M, et al. Sinai Immunology Review Project. Immunology of COVID-19: Current State of the Science. Immunity (2020) 52(6):910–41. 10.1016/j.immuni.2020.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bozorgmehr N, Mashhouri S, Perez Rosero E, Xu L, Shahbaz S, Sligl W, et al. Galectin-9, a Player in Cytokine Release Syndrome and a Surrogate Diagnostic Biomarker in SARS-CoV-2 Infection. mBio (2021) 12(3):e00384–21. 10.1128/mBio.00384-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Loimaranta V, Hepojoki J, Laaksoaho O, Pulliainen AT. Galectin-3-Binding Protein: A Multitask Glycoprotein With Innate Immunity Functions in Viral and Bacterial Infections. J Leukoc Biol (2018) 104(4):777–86. 10.1002/JLB.3VMR0118-036R [DOI] [PubMed] [Google Scholar]
- 71.Choi GJ, Kim HM, Kang H. The Potential Role of Dyslipidemia in COVID-19 Severity: An Umbrella Review of Systematic Reviews. J Lipid Atheroscler (2020) 9(3):435–48. 10.12997/jla.2020.9.3.435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kastritis E, Wechalekar A, Schönland S, Sanchorawala V, Merlini G, Palladini G, et al. Challenges in the Management of Patients With Systemic Light Chain (AL) Amyloidosis During the COVID-19 Pandemic. Br J Haematol (2020) 190(3):346–57. 10.1111/bjh.16898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Conway EM, Pryzdial ELG. Is the COVID-19 Thrombotic Catastrophe Complement-Connected? J Thromb Haemost (2020) 18(11):2812–22. 10.1111/jth.15050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Perico L, Benigni A, Casiraghi F, Ng LFP, Renia L, Remuzzi G. Immunity, Endothelial Injury and Complement-Induced Coagulopathy in COVID-19. Nat Rev Nephrol (2021) 17(1):46–64. 10.1038/s41581-020-00357-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Java A, Apicelli AJ, Liszewski MK, Coler-Reilly A, Atkinson JP, Kim AH, et al. The Complement System in COVID-19: Friend and Foe? JCI Insight (2020) 5(15):e140711. 10.1172/jci.insight.140711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Maugeri A, Barchitta M, Mazzone MG, Giuliano F, Agodi A. Complement System and Age-Related Macular Degeneration: Implications of Gene-Environment Interaction for Preventive and Personalized Medicine. BioMed Res Int (2018) 2018:7532507. 10.1155/2018/7532507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Mullins RF, Russell SR, Anderson DH, Hageman GS. Drusen Associated With Aging and Age-Related Macular Degeneration Contain Proteins Common to Extracellular Deposits Associated With Atherosclerosis, Elastosis, Amyloidosis, and Dense Deposit Disease. FASEB J (2000) 14(7):835–46. 10.1096/fasebj.14.7.835 [DOI] [PubMed] [Google Scholar]
- 78.Cabezas-Cruz A, Hodžić A, Román-Carrasco P, Mateos-Hernández L, Duscher GG, Sinha DK, et al. Environmental and Molecular Drivers of the α-Gal Syndrome. Front Immunol (2019) 10:1210. 10.3389/fimmu.2019.01210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Young I, Prematunge C, Pussegoda K, Corrin T, Waddell L. Tick Exposures and Alpha-Gal Syndrome: A Systematic Review of the Evidence. Ticks Tick Borne Dis (2021) 12(3):101674. 10.1016/j.ttbdis.2021.101674 [DOI] [PubMed] [Google Scholar]
- 80.de la Fuente J, Gortázar C, Cabezas-Cruz A. Immunity to Glycan α-Gal and Possibilities for the Control of COVID-19. Immunotherapy (2021) 13(3):185–8. 10.2217/imt-2020-0247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pacheco I, Contreras M, Villar M, Risalde MA, Alberdi P, Cabezas-Cruz A, et al. Vaccination With Alpha-Gal Protects Against Mycobacterial Infection in the Zebrafish Model of Tuberculosis. Vaccines (Basel) (2020) 8(2):195. 10.3390/vaccines8020195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Koropatkin NM, Cameron EA, Martens EC. How Glycan Metabolism Shapes the Human Gut Microbiota. Nat Rev Microbiol (2012) 10(5):323–35. 10.1038/nrmicro2746 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ferreira C, Viana SD, Reis F. Is Gut Microbiota Dysbiosis a Predictor of Increased Susceptibility to Poor Outcome of COVID-19 Patients? An Update. Microorganisms (2020) 9(1):53. 10.3390/microorganisms9010053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Luo J J, Liang S, Jin F. Gut Microbiota in Antiviral Strategy From Bats to Humans: A Missing Link in COVID-19. Sci China Life Sci (2021) 27:1–15. 10.1007/s11427-020-1847-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Netea MG, Domínguez-Andrés J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E, et al. Defining Trained Immunity and Its Role in Health and Disease. Nat Rev Immunol (2020) 20(6):375–88. 10.1038/s41577-020-0285-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hamiel U, Kozer E, Youngster I. SARS-CoV-2 Rates in BCG-Vaccinated and Unvaccinated Young Adults. JAMA (2020) 323(22):2340–1. 10.1001/jama.2020.8189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.de la Fuente J, Armas O, Sánchez-Rodríguez L, Gortázar C, Lukashev AN. COVID-BCG Collaborative Working Group, Citizen Science Initiative Points at Childhood BCG Vaccination as a Risk Factor for COVID-19. Transbound Emerg Dis (2021). 10.1111/tbed.14097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pavel AB, Glickman JW, Michels JR, Kim-Schulze S, Miller RL, Guttman-Yassky E. Th2/Th1 Cytokine Imbalance Is Associated With Higher COVID-19 Risk Mortality. Front Genet (2021) 12:706902. 10.3389/fgene.2021.706902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Singh R, Hemati H, Bajpai M, Yadav P, Maheshwari A, Kumar S, et al. Sustained Expression of Inflammatory Monocytes and Activated T Cells in COVID-19 Patients and Recovered Convalescent Plasma Donors. Immun Inflamm Dis (2021). 10.1002/iid3.476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mitamura Y, Schulz D, Oro S, Li N, Kolm I, Lang C, et al. Cutaneous and Systemic Hyperinflammation Drives Maculopapular Drug Exanthema in Severely Ill COVID-19 Patients. Allergy (2021). 10.1111/all.14983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lazari LC, Ghilardi FR, Rosa-Fernandes L, Assis DM, Nicolau JC, Santiago VF, et al. Prognostic Accuracy of MALDI-TOF Mass Spectrometric Analysis of Plasma in COVID-19. Life Sci Alliance (2021) 4(8):e202000946. 10.26508/lsa.202000946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Völlmy F, van den Toorn H, Zenezini Chiozzi R, Zucchetti O, Papi A, Volta CA, et al. A Serum Proteome Signature to Predict Mortality in Severe COVID-19 Patients. Life Sci Alliance (2021) 4(9):e202101099. 10.26508/lsa.202101099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Geyer PE, Arend FM, Doll S, Louiset ML, Virreira Winter S, Müller-Reif JB, et al. High-Resolution Serum Proteome Trajectories in COVID-19 Reveal Patient-Specific Seroconversion. EMBO Mol Med (2021) 7:e14167. 10.15252/emmm.202114167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Laudanski K, Jihane H, Antalosky B, Ghani D, Phan U, Hernandez R, et al. Unbiased Analysis of Temporal Changes in Immune Serum Markers in Acute COVID-19 Infection With Emphasis on Organ Failure, Anti-Viral Treatment, and Demographic Characteristics. Front Immunol (2021) 12:650465. 10.3389/fimmu.2021.650465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Gutmann C, Takov K, Burnap SA, Singh B, Ali H, Theofilatos K, et al. SARS-CoV-2 RNAemia and Proteomic Trajectories Inform Prognostication in COVID-19 Patients Admitted to Intensive Care. Nat Commun (2021) 12(1):3406. 10.1038/s41467-021-23494-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Park S, Lee EJ. Diagnosis and Treatment of Cystic Lung Disease. Korean J Intern Med (2017) 32(2):229–38. 10.3904/kjim.2016.242 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Analysis of differentially represented proteins in response to COVID-19.
High-resolution network analysis of interactions between proteins and BPs characterized using Graph Theory algorithms.
Serum proteomics data for previously identified biomarkers for COVID-19 disease severity.
Metascape analysis of differentially represented proteins and previously reported in COVID-19 omics datasets.
Differential representation of proteins not previously reported in COVID-19 omics datasets.
Association of differentially represented proteins in response to COVID-19 to other human diseases and conditions.
Cytokine (IL-1 and IL-4) response in COVID-19 symptomatic patients and healthy controls.
Mechanisms of potential disorders associated with COVID-19.
Clinical parameters and comorbidities in COVID-19 asymptomatic and symptomatic cohorts.
Serum proteomics analysis.
Network analysis of interactions between proteins and BPs characterized using Graph Theory algorithms. Please see the Supplementary Material section of the Author guidelines for details on the different file types accepted.
Data Availability Statement
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD024549 and 10.6019/PXD024549. The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.