Abstract
The landscape in dentistry is changing as emerging studies continue to reveal that periodontal health impacts systemic health, and vice versa. Population studies, clinical studies, and in vitro animal studies underscore the critical importance of oral health to systemic health. These inextricable relationships come to the forefront as oral diseases, such as periodontal disease, take root. Special populations bring to bear the multimodal relationships between oral and systemic health. Specifically, periodontal disease has been associated with diabetes, metabolic syndrome, obesity, eating disorders, liver disease, cardiovascular disease, Alzheimer disease, rheumatoid arthritis, adverse pregnancy outcomes, and cancer. Although bidirectional relationships are recognized, the potential for multiple comorbidities, relationships, and connections (multimodal relationships) also exists. Proposed mechanisms that mediate this connection between oral and systemic health include predisposing and precipitating factors, such as genetic factors (gene polymorphisms), environmental factors (stress, habits—such as smoking and high‐fat diets/consumption of highly processed foods), medications, microbial dysbiosis and bacteremias/viremias/microbemias, and an altered host immune response. Thus, in a susceptible host, these predisposing and precipitating factors trigger the onset of periodontal disease and systemic disease/conditions. Further, high‐throughput sequencing technologies are shedding light on the dark matter that comprises the oral microbiome. This has resulted in better characterization of the oral microbial dysbiosis, including putative bacterial periodontopathogens and shifts in oral virome composition during disease. Multiple laboratory and clinical studies have illustrated that both eukaryotic and prokaryotic viruses within subgingival plaque and periodontal tissues affect periodontal inflammation, putative periodontopathogens, and the host immune response. Although the association between herpesviruses and periodontitis and the degree to which these viruses directly aggravate periodontal tissue damage remain unclear, the benefits to periodontal health found from prolonged administration of antivirals in immunocompromised or immunodeficient individuals demonstrates that specific populations are possibly more susceptible to viral periodontopathogens. Thus, it may be important to further examine the implications of viral pathogen involvement in periodontitis and perhaps it is time to embrace the viral dark matter within the periodontal environment to fully comprehend the pathogenesis and systemic implications of periodontitis. Emerging data from the coronavirus disease 2019 pandemic further underscores the inextricable connection between oral and systemic health, with high levels of the severe acute respiratory syndrome coronavirus 2 angiotensin‐converting enzyme 2 receptor noted on oral tissues (tongue) and an allostatic load or overload paradigm of chronic stress likely contributing to rapid breakdown of oral/dental, periodontal, and peri‐implant tissues. These associations exist within a framework of viremias/bacteremias/microbemias, systemic inflammation, and/or disturbances of the immune system in a susceptible host. A thorough review of systemic and oral diseases and conditions and their mechanistic, predisposing, and precipitating factors are paramount to better addressing the oral and systemic health and needs of our patients.
Keywords: microbemias, oral health, periodontal disease, susceptible host, systemic disease, systemic inflammation
1. OVERVIEW
Oral and periodontal health impact systemic health, and vice versa. Epidemiologic and large population studies, clinical studies, and in vitro animal studies underscore the critical importance of oral health to systemic health. These inextricable connections come to the forefront as oral diseases, such as periodontal disease, take root. Special at‐risk populations bring to bear this bidirectional relationship between oral and systemic health. The concept of a risk or susceptible host, which emerged from the classic longitudinal studies on the natural history of periodontal disease in tea laborers and in studies on identical twins, highlights a genetic basis/predisposition to periodontal disease.1, 2 Thus, in susceptible hosts, as periodontal disease affects major organ systems and conditions (kidney, liver, heart, gastrointestinal tract, respiratory tract, adipose tissue, brain, neuroendocrine system/stress, reproductive system, skeletal system/joints, aging, cancer), associations between periodontal disease and systemic diseases or conditions affecting special populations emerge. Specifically, periodontal disease has been associated with diabetes, metabolic syndrome, obesity, eating disorders, liver disease, cardiovascular disease, Alzheimer disease, rheumatoid arthritis, adverse pregnancy outcomes, and cancer (Figure 1). Although bidirectional relationships are discussed, the potential for multiple comorbidities, relationships, and connections (multimodal relationships) also exists. Proposed mechanisms that mediate this connection between oral and systemic health include predisposing and precipitating factors, such as genetic factors (gene polymorphisms), environmental factors (stress, habits—such as smoking and dietary habits that include high‐fat diets/consumption of highly processed foods), medications, microbial dysbiosis and bacteremias, and an altered host immune response (Figure 1). These associations exist within a framework of chronic bacteremias, systemic inflammation, and/or disturbances of the immune system.
FIGURE 1.
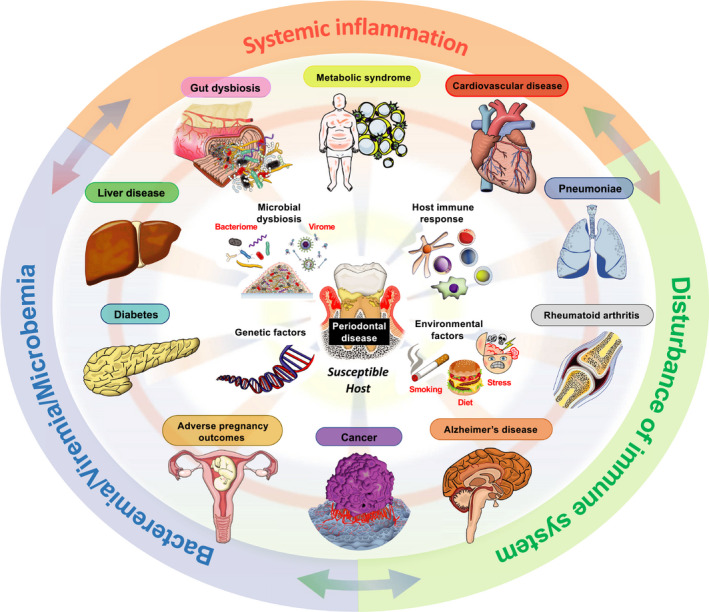
Periodontal disease associations with systemic diseases and conditions. Periodontal disease has been associated with diabetes, metabolic syndrome, obesity, eating disorders, liver disease, cardiovascular disease, Alzheimer disease, rheumatoid arthritis, adverse pregnancy outcomes, and cancer. Proposed mechanisms that mediate this connection between oral and systemic health in a susceptible host include predisposing and precipitating factors, such as genetic factors (gene polymorphisms), environmental factors (stress, habits—such as smoking and high‐fat diets/consumption of highly processed foods), medications, microbial dysbiosis and bacteremias/viremias, and an altered host immune response. These associations exist within a framework of bacteremias/viremias/microbemias, systemic inflammation, and/or disturbances of the immune system in a susceptible host. (Figure courtesy of Dr Ryutaro Kuraji, Assistant Professor, Department of Life Science Dentistry, The Nippon Dental University, Tokyo, Japan; Department of Periodontology, The Nippon Dental University School of Life Dentistry at Tokyo, Tokyo, Japan; Visiting Assistant Professor; Department of Orofacial Sciences, School of Dentistry, University of California San Francisco, San Francisco, CA, USA)
With the recent severe acute respiratory syndrome coronavirus 2 (coronavirus disease 2019) pandemic, this oral and systemic connection has once again emerged. Several chapters in this volume of Special Populations discuss this potential association. There are early and anecdotal reports of oral signs and symptoms emerging in severe acute respiratory syndrome coronavirus 2–infected individuals and/or from the stress surrounding the pandemic. This includes the well‐documented loss of taste affecting the tongue and oral cavity. It is known that the virus has a specific ability to enter the oral cavity because the tongue has the angiotensin‐converting enzyme 2 receptor, which is the primary receptor for severe acute respiratory syndrome coronavirus 2.3, 4 Other reports also indicate that severe acute respiratory syndrome coronavirus 2 may affect the hematologic system, including the bone marrow and bone healing.5, 6, 7 In addition, the cytokine storm that emerges with this disease is well known.8, 9 These altered wound healing and inflammatory processes could significantly impact the health of teeth, implants, and oral tissues, eventually causing tooth loss and implant loss. Anecdotal reports in the media and noted by clinicians, including those within our clinics at University of California San Francisco, have indicated the rapid loss and breakdown of teeth and implants during the severe acute respiratory syndrome coronavirus 2 pandemic.10 Other observations include the fracture of teeth and implants, and increased clenching and bruxism, potentially related to the stress from the pandemic.11 Thus, the rapid breakdown of oral/dental, periodontal, peri‐implant tissues being observed may be the result of a direct mechanism due to severe acute respiratory syndrome coronavirus 2 virus effects on inflammatory and wound‐healing processes in the oral cavity or as a result of chronic stress. The latter may present itself as an allostatic load or overload paradigm of chronic stress, likely contributing to the rapid breakdown/loss of tissues due to several mechanisms (neuroendocrine, altered inflammatory or wound‐healing processes). Allostatic load or overload describes the “wear and tear” on the body as a result of chronic stress and how this contributes to pathology (Figure 2).12, 13 Allostasis is the predictive regulation or stabilization of internal sensations in response to stimuli. However, with an allostatic load or overload scenario there are neuroendocrine, cardiovascular, neuroenergetic, and emotional responses that become persistently activated so that blood flow disturbances in the coronary and cerebral arteries, high blood pressure, atherogenesis, cognitive dysfunction, and depressed mood accelerate disease progression. These responses and mechanisms may impact oral health.14 As new severe acute respiratory syndrome coronavirus 2–related studies emerge, scientists and clinicians will help shed light on these observations of a potential oral‐severe acute respiratory syndrome coronavirus 2 connection.
FIGURE 2.
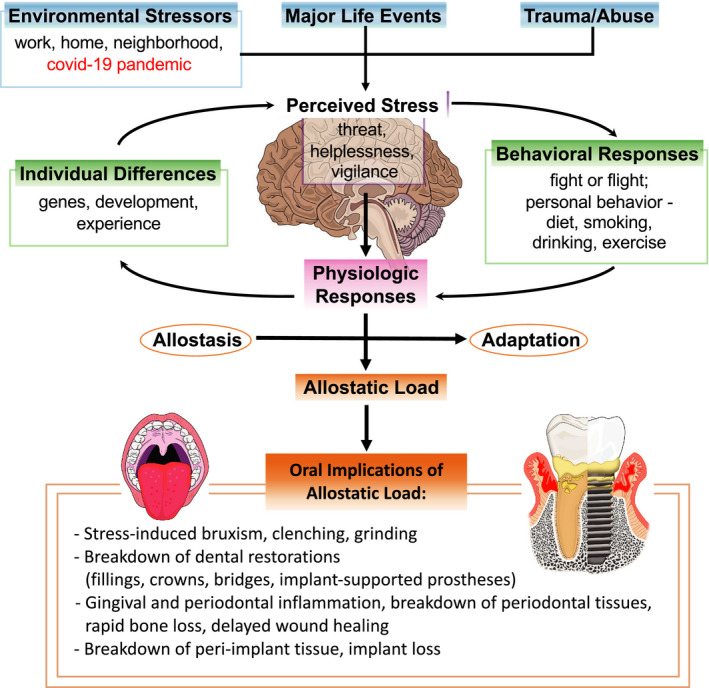
Allostatic load and implications for oral and periodontal health. Allostatic load or overload describes the “wear and tear” on the body as a result of chronic stress and how this contributes to pathology. There may be a rapid breakdown of oral, dental, periodontal, and peri‐implant tissues as a result of an allostatic load or overload. For example, from chronic stress due to the coronavirus disease 2019 (COVID‐19) pandemic, other environmental stressors, major life events, or trauma. This perceived stress is subject to individual differences and then elicits behavioral and physiologic responses. These responses result in allostasis (homeostasis), adaptation, or an allostatic load. The allostatic load or overload paradigm of chronic stress likely contributes to the rapid breakdown/loss of oral tissues due to several mechanisms (neuroendocrine, altered inflammatory or wound healing processes)
In these chapters, readers will further appreciate that the role of systemic inflammation is a recurring theme in oral‐systemic associations, including meta‐inflammation or metabolic‐associated inflammation found in obesity and other related conditions, such as metabolic syndrome and diabetes. The concept of a “two‐hit” model is presented to explain obesity‐exacerbated periodontal bone loss.15 This proposed model recognizes a previously unappreciated aspect of myeloid‐derived suppressor cell population expansion, differentiation, and activity that can directly participate in periodontal bone loss, providing new mechanistic and translational perspectives. The studies on metabolic syndrome further reveal the close and intimate relationship between systemic inflammation and the oral microbiome.16 Several authors also discuss the concept of dose‐response. For example, an increase in obesity parameters, such as body mass index, is associated with more severe periodontal disease parameters. The same has been reported for diabetes and for other diseases presented by the authors in these chapters.
Another unique example of how the host‐immune response may be compromised by periodontal disease is in its association with rheumatoid arthritis. Epidemiologic evidence for an association between these two diseases shows emerging scientific information linking specific bacterial periodontal pathogens, such as Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans, in the citrullination process, leading to auto‐antibody formation and compromised immunotolerance of the susceptible patient to rheumatoid arthritis.17
In the chapter on eating disorders, the authors give an overview of the oral health impact of eating disorders with a special emphasis on the periodontium.18 Oral health professionals have a unique role to play in the early diagnosis of eating disorders because of the important impact that eating disorders have on the oral cavity. In vomiting‐associated eating disorders, the risk of erosive tooth wear is primarily localized to the palatal surfaces of the incisors. Emerging evidence also indicates a high frequency of gingivitis and gingival recessions associated with compulsive toothbrushing. A holistic approach, including oral health and functional rehabilitation, should be promoted by physicians, psychiatrists, and dentists for people with eating disorders.
The role of the oral microbiome and key members of this complex community that regulate health and oral disease, including periodontal disease, have been recognized as key mechanistic players in systemic diseases. These oral pathobionts have recently taken center stage in several diseases, including Alzheimer disease and cancers, including oral cancer. The role of the periodontal disease microflora on the initiation and progression of Alzheimer disease is presented in this volume with a focus on the potential role of the keystone pathogen, P. gingivalis, with its family of gingipain enzymes. Small‐molecule inhibitors of these bacterial gingipains and their effects on reducing biological markers of Alzheimer disease may have beneficial effects for the initiation and progression of loss of cognitive function in Alzheimer disease.19 In addition to key pathobionts, the concept of oral dysbiosis has been associated with periodontal disease and systemic diseases, including cancer. Oral dysbiosis, or an imbalance in the composition of the oral microbial flora, is discussed in this volume as it relates to the epidemiologic evidence and mechanistic underpinnings linking periodontitis to oral, gastrointestinal, lung, breast, prostate, and uterine cancers.20 The oral microbiome and its interactions with the host (oralome) is also discussed in the context of cancer. Given the associations between oral microbes and cancer, antimicrobials, antimicrobial peptides (with known anticancer properties), and probiotics may be promising tools to prevent and treat cancers, targeting both the microbes and associated carcinogenesis processes. These findings represent a novel paradigm in the pathogenesis and treatment of cancer focused on the oral microbiome and antimicrobial‐based therapies.
Shedding light on the dark matter that comprises the oral microbiome is actively being pursued. In addition to the well‐described bacteriome, the mycobiome (especially oral candidiasis) and virome are specifically discussed.21, 22 Multiple laboratory and clinical studies have illustrated that both eukaryotic and prokaryotic viruses within subgingival plaque and periodontal tissues affect periodontal inflammation, putative periodontopathogens, and the host immune response. Although the association between herpesviruses and periodontitis and the degree to which these viruses directly aggravate periodontal tissue damage remain unclear, the benefits to periodontal health found from prolonged administration of antivirals in immunocompromised or immunodeficient individuals demonstrate that specific populations are possibly more susceptible to viral periodontopathogens. Thus, it may be important to further examine the implications of viral pathogen involvement in periodontitis.
The oral cavity exists as a highly dynamic microbial environment that harbors many distinct substrata and microenvironments housing diverse microbial communities. The oral cavity also is presented with near‐constant environmental challenges, including host diet, salivary flow, masticatory forces, and introduction of exogenous microbes. The composition of the oral microbiome is shaped throughout life by factors that include host genetics and maternal transmission, as well as by environmental factors, such as dietary habits, oral hygiene practice, medications, and systemic factors. This dynamic ecosystem presents opportunities for oral microbial dysbiosis and the development of dental and periodontal diseases.23 The application of both in vitro and culture‐independent approaches has broadened the mechanistic understandings of complex polymicrobial communities within the oral cavity, as well as the environmental, local, and systemic underpinnings that influence the dynamics of the oral microbiome. In the chapter on the oral microbiome, the authors discuss the current understanding of microbial communities within the oral cavity and the influences and challenges upon this system that encourage homeostasis or provoke microbiome perturbation, and which thus contribute to states of oral health or disease. To counter the disease‐associated dysbiosis of the oral cavity, strategies have been proposed to reestablish a “healthy” microbiome via the use of probiotics. The “Probiotics for periodontal health–current molecular findings” chapter reviews the literature on the use of probiotics for modifying the oral microbial composition toward a beneficial state that might alleviate disease progression.24
Bidirectional relationships are another common theme present for several diseases associated with periodontal disease, including diabetes, metabolic syndrome, obesity, Alzheimer disease, and other diseases discussed in this volume. With regard to periodontitis and metabolic syndrome, the vast majority of studies point to an association between these two conditions and also demonstrate that periodontitis can contribute to the development of or can worsen metabolic syndrome.16 Also, it is well established in the cognitively impaired patient that a lack of adherence to dental hygiene routines and professional care leads to increases in the prevalence and severity of dental conditions, leading to increased loss of teeth. More recent evidence has indicated a possible role of the microbiota of dental plaque associated with periodontal diseases in the development and progression of Alzheimer disease, thereby supporting a two‐way interaction of these two diseases.19 Although bidirectional relationships are frequently acknowledged in this framework, the potential for multiple comorbidities/associations, relationships, and connections also exists. For example, biological mechanisms potentially linking obesity and periodontal disease are adiposity‐associated hyperinflammation, microbial dysbiosis, altered immune response, specific genetic polymorphisms, and increased stress.25
Several functional axes also emerge when discussing mechanistic associations between periodontal disease and systemic diseases, including, among others, the oral‐liver, oral‐kidney, and oral‐brain axes. The global burden of periodontal disease combined with the recent nonalcoholic fatty liver disease epidemic has important clinical and public health implications. A subset of nonalcoholic fatty liver disease can develop into more severe and progressive forms, namely nonalcoholic steatohepatitis. Emerging evidence suggests an association between periodontal disease and liver diseases, and thus the authors of the chapter on liver disease propose the term periodontal disease–related nonalcoholic fatty liver disease or periodontal disease–related nonalcoholic steatohepatitis.26 These associations indicate the existence of an oral‐liver axis. The chapter on Alzheimer disease suggests that new therapies are needed to address the potential upstream events that may precede overt signs of Alzheimer disease.19 One of these approaches would be to target the various bacterial, viral, and other microbial pathogens associated with periodontal disease that can translocate into the bloodstream and then to distal sites, such as the brain. Such microbial translocation would lead to local inflammation and buildup of the hallmark signs of Alzheimer disease, including beta‐amyloid deposits, tau fragmentation and tangles, breakdown of host protective molecules (such as the apolipoproteins), and neuron toxicity. This process highlights the importance of the oral‐brain axis.
Aging is associated with the development of disease, including periodontal disease. Further, the elderly population is predicted to double in 30 years, which will have a significant impact that the dental profession needs to plan for. Periodontal disease is one of the many diseases and conditions that increase in prevalence with age. In addition to the traditional focus on individual age‐related conditions, there is now a greater recognition that multisystem conditions, such as frailty, play an important role in the health of older populations.27 Frailty is a clinical condition in older adults that increases the risk of adverse health outcomes. There is likely a bidirectional relationship between periodontal disease and frailty. Comorbid systemic diseases, poor physical functioning, and limited ability to self‐care in frail older people have been implicated to be underlying the association between frailty and periodontal disease. In addition, both frailty and periodontal disease also have strong associations with inflammatory dysregulation and other age‐related pathophysiologic changes that may similarly underlie their development and progression.
Preventative and noninvasive treatment, supportive periodontal therapy, and patient‐specific maintenance plans are imperative to maintaining oral health in the older population. Multiple coexisting changes, including xerostomia, altered wound healing, altered bone physiology, altered microbiome, and diminished plaque control, can add complexity to periodontal management. Considerations of the patient's general health, the selected periodontal treatment plan, and the selected completed restorative procedures need to be considered. In the chapter on treatment considerations in the older adult with periodontal disease, the authors review how age, comorbidities, oral hygiene, and restorative dental procedures collectively influence the treatment and management of the periodontium in the elderly.28
In addition, as the USA has an aging population, the number of individuals with cognitive impairment and periodontitis is increasing, since both conditions/diseases increase with age. Dental informed‐consent best practices for dental/periodontal treatment of individuals with cognitive impairment have not been explored, yet they warrant consideration, since complex dental treatments to address periodontal needs/edentulism raise challenges for informed consent in elderly with cognitive impairment. In the chapter on dental informed consent challenges and considerations for the cognitively impaired patients, the authors review information to help practitioners better understand this topic and develop best practices in dentistry for informed consent of patients with cognitive impairment that need extensive dental treatment, including surgical and implant therapy.29
Women are particularly susceptible to developing gingival problems during pregnancy. In addition, periodontal disease in pregnant women may lead to adverse outcomes for both mother and infant, which has serious clinical and public health implications.30 Both scenarios have been extensively examined and call attention to pregnant women as an important and vulnerable population in relation to periodontal health. Recent evidence further suggests that there is an association between the presence of periodontal disease and a pregnancy‐associated oral dysbiosis. Although this oral dysbiosis and pathogenic periodontal bacteria are considered to be associated with adverse pregnancy outcomes, it is still not clear how an oral dysbiosis during pregnancy can modulate oral diseases and birth outcomes. In the chapter on oral microbiome shifts during pregnancy and adverse pregnancy outcomes, the authors summarize the current literature on normal changes in the oral microbiome that occur during pregnancy; the pathogenic changes in the oral microbiome thought to occur in association with adverse pregnancy outcomes; and the association between the placental microbiome and the oral microbiome.31
Tobacco use contributes to more mortality and morbidity globally than any other behavioral risk factor, and it adversely effects the oral cavity and the periodontium. New or emerging tobacco and/or nicotine products have been brought to market, namely electronic cigarettes, including heated tobacco and other noncombustible nicotine products. Cannabis (marijuana) use is also increasing, and it, too, has risks for oral health. In the chapter on tobacco and nicotine products, the authors review selected tobacco and nicotine products with an emphasis on their implications for periodontal disease risk and clinical management.32 The authors also provide strategies for tobacco‐use counselling and cessation support that dental professionals can implement in practice.
A lesson learned from the chapters in this volume on special populations is that health providers would be remiss not to engage in a more thorough medical and dental history review of their patients prior to developing treatment plans, including previously unappreciated oral and medical risks. This emerging landscape warrants new considerations of the oral‐systemic connections to maximize oral and systemic health.
In summary, these reviews convey that special populations reflect the oral‐systemic connections that manifest around periodontal disease. Common mechanisms and factors—namely, inflammation, the oral microbiome and its dysbiosis, stress, diet, genetics, habits, and the environment—play into these associations. A thorough review of systemic and oral diseases and conditions and other mechanistic, predisposing, and precipitating factors is paramount to better addressing the oral and systemic health and needs of our patients.
Kapila YL. Oral health’s inextricable connection to systemic health: Special populations bring to bear multimodal relationships and factors connecting periodontal disease to systemic diseases and conditions. Periodontol 2000. 2021;87:11–16. 10.1111/prd.12398
Funding information:
This work was supported by funding from the AAP Sunstar Innovation Grant, NIH R01 DE025225 grant, and Larry Berkelhammer funds to Yvonne L. Kapila.
REFERENCES
- 1.Löe H, Anerud A, Boysen H, Morrison E. Natural history of periodontal disease in man. Rapid, moderate and no loss of attachment in Sri Lankan laborers 14 to 46 years of age. J Clin Periodontol. 1986;13:431‐445. [DOI] [PubMed] [Google Scholar]
- 2.Michalowicz BS. Genetic and heritable risk factors in periodontal disease. J Periodontol. 1994;65:479‐488. [DOI] [PubMed] [Google Scholar]
- 3.Kuraji R, Kapila YL. High expression of ACE2 receptor of 2019‐nCoV on the epithelial cells of oral mucosa. PracticeUpdate. https://www.practiceupdate. 2020. [DOI] [PMC free article] [PubMed]
- 4.Xu H, Zhong L, Deng J, et al. High expression of ACE2 receptor of 2019‐nCoV on the epithelial cells of oral mucosa. Intl J Oral Sci. 2020;12:8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.de Oliveira Toledo SL, Sousa Nogueira L, das Graças Carvalho M, Alves Rios DR, de Barros Pinheiro M. COVID‐19: review and hematologic impact. Clin Chim Acta. 2020;19:170‐176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Disser NP, De Micheli AJ, Schonk MM, et al. Musculoskeletal consequences of COVID‐19. J Bone Joint Surg. 2020;102:1197‐1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tao H, Ge G, Li W, et al. Dysimmunity and inflammatory storm: watch out for bone lesions in COVID‐19 infection. Med Hypotheses. 2020;145:110332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tang Y, Liu J, Zhang D, Xu Z, Ji J, Wen C. Cytokine storm in COVID‐19: the current evidence and treatment strategies. Front Immunol. 2020;11:1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Candia P, Prattichizzo F, Garavelli S, Matarese G. T cells: warriors of SARS‐CoV‐2 infection. Trends Immunol. 2021;42:18‐30.S1471–4906(20)30260‐X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yan W. Their teeth fell out. Was it another Covid‐19 consequence? The New York Times https://www.nytimes.com/2020/11/26/health/covid‐teeth‐falling‐out.html. Accessed November 28, 2020.
- 11.Zinko C. Bay Area dentists spot 2020 stress in clenched jaws and cracked teeth. https://www.sfchronicle.com/culture/article/Bay‐Area‐dentists‐spot‐2020‐stress‐in‐clenched‐15764993.php. December 2, 2020. San Francisco Chronicle.
- 12.McEwen BS. Protection and damage from acute and chronic stress: allostasis and allostatic overload and relevance to the pathophysiology of psychiatric disorders. Ann N Y Acad Sci. 2004;1032:1–7. [DOI] [PubMed] [Google Scholar]
- 13.McEwen BS. Neurobiological and systemic effects of chronic stress. Chronic Stress. 2017;1:2470547017692328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Decker AM, Kapila Y, Wang H‐L. The psychobiological links between chronic stress–related diseases, periodontal/peri‐implant diseases, and wound healing. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kwack KH, Maglaras V, Thiyagarajan R, Zhang L, Kirkwood KL. Myeloid‐derived suppressor cells in obesity‐associated periodontal disease: a conceptual model. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pirih FQ, Monajemzadeh S, Singh N, et al. Association between metabolic syndrome and periodontitis—the role of lipids, inflammatory cytokines, altered host response, and the microbiome. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.González‐Febles J, Sanz M. Periodontitis and rheumatoid arthritis: what have we learned about their connection and their treatment. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 18.Rangé H, Colon P, Godart N, Kapila Y, Bouchard P. Eating disorders through the periodontal lens. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ryder MI, Xenoudi P. Alzheimer disease and the periodontal patient: new insights, connections, and therapies. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 20.Radaic A, Ganther S, Kamarajan P, Yom SS, Grandis JR, Kapila Y. Paradigm shift in the pathogenesis and treatment of oral cancer focused on the oralome and antimicrobial‐based therapeutics. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Villar CC, Dongari‐Bagtzoglou A. Fungal diseases: oral dysbiosis in susceptible hosts. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 22.Martinez A, Kuraji R, Kapila Y. The human oral virome—shedding light on the dark matter. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sedghi L, DiMassa V, Harrington A, Lynch S, Kapila Y. The oral microbiome: the role of key organisms and complex networks in oral health and disease. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nguyen T, Brody H, Radaic A, Kapila Y. Probiotics for periodontal health—current molecular findings. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ganesan S, Vazana S, Stuhr S. Waistline to the gumline: relationship of obesity and periodontal disease—biological and management considerations. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 26.Kuraji R, Sekino S, Kapila Y, Numabe Y. Periodontal disease–related nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: an emerging concept of oral‐liver axis. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Clark D, Kotronia E, Ramsay SE. Aging, and periodontal disease: basic biological considerations. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Curtis DA, Lin G‐H, Rajendran Y, Tsegazeab G, Suryadevara J, Kapila Y. Treatment planning considerations in the older adult with periodontal disease. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 29.Alsaleh A, Kapila Y, Shahriar I, Kapila Y. Dental informed consent challenges and considerations for cognitively impaired patients. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Raju K, Berens L. Periodontology and pregnancy: an overview of biomedical and epidemiological evidence. Periodontol 2000. 2021. [DOI] [PubMed] [Google Scholar]
- 31.Ye C, Kapila Y. Oral microbiome changes during pregnancy and adverse pregnancy outcomes. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chaffee BW, Couch ET, Vora MV, Holliday RS. Oral and periodontal implications of tobacco and nicotine products. Periodontol 2000. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]


