Abstract
Some COVID-19 patients suffer complications from anti-viral immune responses which can lead to both a dangerous cytokine storm and development of blood-borne factors that render severe thrombotic events more likely. The precise immune response profile is likely, therefore, to determine and predict patient outcomes and also represents a target for intervention. Anti-viral T cell exhaustion in the early stages is associated with disease progression. Dysregulation of T cell functions, which precedes cytokine storm development and neutrophil expansion in alveolar tissues heralds damaging pathology.T cell function, cytokine production and factors that attract neutrophils to the lung can be modified through targeting molecules that can modulate T cell responses. Manipulating T cell responses by targeting the PI3K/Akt/mTOR pathway could provide the means to control the immune response in COVID-19 patients. During the initial anti-viral response, T cell effector function can be enhanced by delaying anti-viral exhaustion through inhibiting PI3K and Akt. Additionally, immune dysregulation can be addressed by enhancing immune suppressor functions by targeting downstream mTOR, an important intracellular modulator of cellular metabolism. Targeting this signalling pathway also has potential to prevent formation of thrombi due to its role in platelet activation. Furthermore, this signalling pathway is essential for SARS-cov-2 virus replication in host cells and its inhibition could, therefore, reduce viral load. The ultimate goal is to identify targets that can quickly control the immune response in COVID-19 patients to improve patient outcome. Targeting different levels of the PI3K/Akt/mTOR signalling pathway could potentially achieve this during each stage of the disease.
Keywords: COVID-19, PI3K, Akt, mTOR, Immune modulation, Thrombosis
1. Introduction
The COVD-19 pandemic caused by the SARS-cov-2 virus is a global crisis with the number of confirmed cases exceeding 214 million with more than 4.4 million deaths reported by the World Health Organisation so far [1].
There are often different stages of the disease, with initial flu-like symptoms typically associated with an anti-viral immune response, followed, in some patients, by a more dangerous cytokine storm characterised by an uncontrolled immune response [2]. This second stage is associated with higher morbidity and mortality.
A third stage is characterised by an increased risk of severe thrombotic events in very ill patients with high serum levels of the d-Dimer thrombotic marker [3].
Although more than 4.9 billion vaccine doses have been administered globally [1], we are far from relying solely on a preventive approach and therapies are still needed to deal with different stages of the disease in those infected. This need is further emphasized by the emergence of new variants in different parts of the world.
Two of the main goals identified for the management of COVID-19 patients include the diagnosis and management of hyperinflammation and the detection and management of increased coagulation, both observed in severe cases. The former can be recognised by fever with elevated C-reactive protein while the latter by an increase in d-dimer and prothrombin time with reduced fibrinogen [4].
Here, we will briefly explore how targeting the PI3K/Akt/mTOR pathway can address these two goals and dissect how mitigating different components of the pathway can benefit COVID-19 patients in different phases of the disease.
2. Immune dysregulation
2.1. Suboptimal anti-viral immune response
Severe cases of COVID-19 are attributed to malfunctions in the immune response to this virus. The suboptimal function of T cells in responding to the virus in early stages is associated with severe disease. Disease progression was reported to correlate with the exhaustion phenotype of T cells, typically characterised by high PD-1 expression [5]. This exhaustion is more commonly observed in an ageing immune system, hence the correlation between the patient age and the severity of the disease.
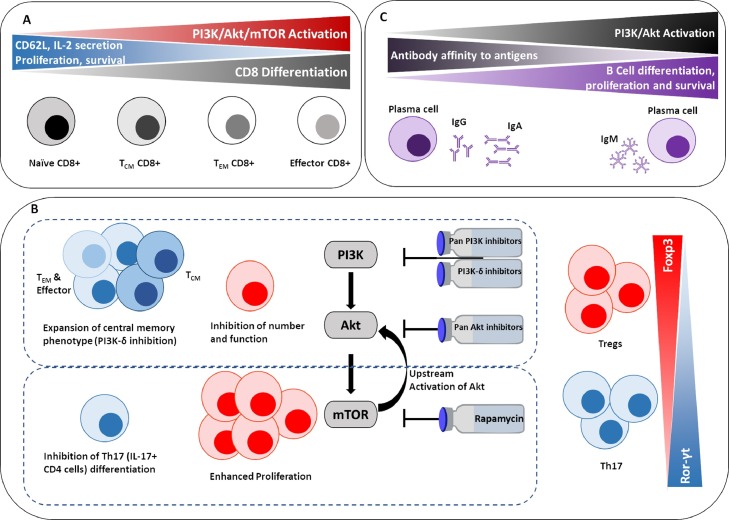
Therefore, during the early stage of the disease, enhancing the effector arm of the immune response is crucial. Many of the immune cells involved in the response to COVID-19 are modulated by the PI3K/Akt pathway (Fig. 1 ), specifically, the PI3K-δ isoform is thought to be responsible for the modulation of the function of CD8 [6], Tregs [7, 8], B cells, mast cells [9] and neutrophils [10]. It therefore comes as no surprise that mutations in this isoform are associated with an increased risk of respiratory tract infections, including a gain of function mutation that leads to increased activation of Akt resulting in activation induced death in lymphocytes [11]. It was therefore suggested that PI3K-δ is a potential target in the management of COVID-19 [12], as this was effective in allergic lung inflammation [9, 13]. Targeting PI3K with a small molecule pan inhibitor that targets all PI3K isoforms is also thought to be effective in the treatment of lung disease murine models including asthma, fibrosis [14] and allergic lung inflammation by reducing both inflammatory cytokines and the number of eosinophils, neutrophils and lymphocytes [13].
Fig. 1.
A simplified diagram illustrating the role of the PI3K/Akt/mTOR pathway in regulating different lymphocyte responses including A) CD8 T cells [6, 16, 17] B) Tregs and Th17 [7, 18, 19, 35, 36] C) B cells [66, 67]. TCM =central memory, TEM =effector memory.
In patients with gain-of-function mutations in PI3K-δ, it was reported that the phosphorylation of the downstream Akt is increased even in resting T cells [15]. This was associated with hyperactivation in mTOR. This genotype was found to be associated with senescent, terminally differentiated phenotype of CD8 T cells, with a significant deficiency in naïve and central memory phenotype [15]. Targeting mTOR in these patients using Rapamycin, increased the percentage of naïve T cells and rescued T cell function in vitro [15].
Lessons learnt from cancer immunotherapy suggest that small molecule inhibitors can modulate T cell responses by mitigating T cell exhaustion and restoring their effector potency. It had been previously shown that inhibiting the PI3K and Akt component of the signalling pathway, specifically the PI3K-δ and the Akt1 and Akt2 isoforms can delay the exhaustion of CD8 T cells, maintain their memory phenotype and enhance cytotoxic function [6, 16]. Targeting mTOR with Rapamycin was also found to enhance the memory phenotype of CD8 T cells [17]. Th17 CD4+ T cells treated with a PI3K-δ inhibitor were found to possess superior anti-tumour immunity, which was attributed, at least in part, to the generation of central memory like phenotype in these Th17 cells. Interestingly, this effect was not as pronounced with pan PI3K inhibition [18]. Furthermore, targeting PI3K and Akt by small molecule inhibitors can nullify the activity of the suppressive CD4+ regulatory T cells (Tregs) [7, 19]. This enhancement of the effector arm and inhibition of the suppressive arm of the immune response proved effective in enhancing the anti-tumour immune responses [6, 7, 16, 19] and have the potential of enhancing anti-viral immunity during the early stages of COVID-19.
Targeting the PI3K/Akt/mTOR pathway could be crucial in enhancing immune responses in the ageing immune system not only against the SARS-cov-2 infection, but also in response to vaccination. It was reported that mTOR inhibition using the RAD001 inhibitor improved the response to influenza vaccination in elderly patients (over the age of 65 years) as measured by serologic anti-influenza antibody response and reduced the percentage of T cells expressing PD-1 [20]. This could be vital for COVID-19 vaccination, especially with a reported correlation between disease progression in COVID-19 and T cells with high PD-1 expression [5].
2.2. Exaggerated immune response
In patients where the disease develops to a severe stage, there is an exaggerated immune response resulting in a dangerous, sometimes lethal, cytokine storm, with pro-inflammatory IL-6 being implicated as a major contributor to the catastrophic outcomes of the disease.
Understanding the immunological response to this disease can inform patient therapy. While immunosuppressive agents could be used to alleviate the cytokine storm, their use during the early phase is considered detrimental as outlined above. However, treatment of the cytokine storm with anti-inflammatory drugs including corticosteroids and anti-IL-6R antibodies has met some success. The National Institute for Health and Care Excellence in UK (NICE) has therefore added dexamethasone, (or hydrocortisone or prednisolone if not available) and Tocilizumab (an anti-IL6R antibody) to the management of COVID-19 guidelines, with Tocilizumab offered to patients who completed or are on a corticosteroid course. In the absence of Tocilizumab, another anti-IL6R antibody, Sarilumab, can be considered for hospitalised adults [21]. Interestingly, IL-6/IL-6R interaction leads to the activation of the PI3K/Akt pathway [22, 23], and therefore its inhibition using Tocilizumab could inhibit this pathway.
Steroids have different inhibitory effects dependent upon each type of proinflammatory immune cells [24]. Glucocorticoid receptor activation has a strong interaction with the PI3K/Akt pathway and dexamethasone was found to have a variable effect on the PI3K/Akt pathway in different cell types, including the inhibition of PI3K antigen induced activation in a basophilic leukaemia cell line [25]. In PBMCs from patients with severe asthma, PI3K inhibitors overcame the resistance to in vitro corticosteroid (dexamethasone) treatment and led to a significant reduction in IL-8 production [26], suggesting a potential for combining both treatments to reduce cytokine production.
Due to the exaggerated immune response, during the second stage of the SARS-cov-2 infection, supressing the effector function and enhancing the suppressor function is essential. This can be achieved by targeting the downstream molecule in the pathway; mTOR using Rapamycin. It has been shown that Rapamycin can significantly enhance the proliferation of suppressive Tregs due to a feedback loop that activates the Akt pathway [27], [28], [29], [30], [31]. This proved useful in the treatment of various autoimmune diseases. In fact, due to the effect that Akt pathway has on increasing Tregs in the lungs, it has been suggested that targeting Akt in COVID-19 patients could potentially suppress the inflammation in the patients’ lungs [32].
An important prognostic marker for severe COVID-19 disease is the neutrophil to lymphocyte (NLR) ratio in bronchial lavage [33], where higher ratios of neutrophils are associated with poorer outcomes. Th17 T cells, a subtype of T cells, are induced by IL-6 with TGF-β or IL-21/IL-1β with a primary role in recruiting neutrophils through induction of IL-8 and G-CSF [34]. Targeting mTOR by rapamycin has been shown to suppress the Th17 airway inflammation [35, 36], thus could contribute to a decrease in NLR. Rapamycin was also shown to improve the clinical outcome in patients suffering from H1N1 associated pneumonia [37], suggesting its potential use in the management of COVID-19.
3. Thrombosis
COVID-19-induced cytokine storm leads to micro- and macrovascular thrombosis development and is associated with a significant increase in d-dimer and fibrin split products. It has been shown that deaths resulting from COVID-19 are associated with significantly higher d-dimer and fibrin degradation product levels compared to survivors [38], with d-dimer levels higher than 1 µg/mL being associated with mortality in COVID-19 patients [39].
It is thought that increased inflammatory markers and ferritin concentration associated with severe COVID-19 sepsis can lead to the activation of pulmonary endothelial cells, which could lead to a dysregulation in fibrinolysis and blood vessel permeability. This will ultimately contribute to the coagulopathy observed in severe COVID-19 cases [40].
Given the clear evidence for the association between COVID-19 and thromboembolism, in addition to increased mortality associated with coagulation (71.4% of deaths versus 0.6% of survivors had disseminated intravascular coagulation during their hospital stay [38]), it is important for clinicians to be able to identify COVID-19 associated coagulopathy at an early stage and differentiate it from other conditions such as pulmonary emboli. This will aid in the identification of patients with poor prognosis and will help manage these cases with anti-coagulants as prophylactic or therapeutic measures to reduce morbidity and mortality [41, 42]. A scoring system for coagulopathy resulting from COVID-19 has therefore been proposed to help develop guidelines for anticoagulation treatment [43].
In addition to anticoagulants, other prophylactic and therapeutic approaches are needed to manage intravascular coagulation associated with COVID-19. This is especially important given the ongoing concerns regarding the COVID-19 vaccines and the resulting hesitancy for the uptake of these life-saving vaccines.
Targeting the PI3K/Akt/mTOR pathway has the potential to prevent the formation of thrombi due to the role of this pathway in platelet activation [44]. Furthermore, in platelets, PI3K plays a crucial role for leucocyte extravasation as a response to inflammatory stimuli in different models, including the acute lung injury model [45]. Additionally, targeting the PI3K/Akt pathway using a pan PI3K inhibitor was found to inhibit the activation of platelets and reduce platelet CD40L expression and secretion in a rat sepsis model [46]. The same study reported that PI3K inhibition also impaired the sepsis associated damage to endothelial cells and suppressed the expression of biomarkers associated with the severity of sepsis [46]. Therefore, mitigating signalling in this pathway can serve the dual purpose of preventing platelet activation to prevent thrombi, while simultaneously inhibiting leucocyte extravasation into the lung, which plays a major role in the pathogenesis of severe COVID-19 disease.
In addition to thrombosis, one of the outcomes of COVID-19 is pulmonary fibrosis. Interestingly, the PI3K/Akt pathway was identified as a therapeutic target for COVID-19 associated pulmonary fibrosis and, in a bleomycin-induced lung fibrosis rat model, targeting the pathway using d-Limonene was protective against fibrosis [47].
4. Viral load
Independent of disease severity, blood levels of the inflammatory cytokines IFN-α, IFN-γ, TNF-α and TRAIL correlate with viral RNA levels in the nasal passage. During the early stages of the infection, patients who developed severe disease had increased levels of IFN-α, IL-1Ra and cytokines associated with Th1-, Th2- and Th17 T cell responses. On the other hand, patients who developed moderate disease had low levels of inflammatory markers and an increase in markers of tissue repair [48].
The immune profiling of patients with moderate or severe COVID-19 showed an increase in innate cells and a reduction in T cells in peripheral blood. Additionally, elevated plasma cytokine levels during early stages of the disease were associated with worse patient outcomes [49]. The same study also reported that at early stages of the disease, viral RNA levels in the nasopharynx did not differ between patients with moderate and severe disease, however, there was a slower decrease of viral loads in patients with severe disease who required intensive care [49]. Furthermore, there was a significant correlation between the viral load and the pro-inflammatory cytokines and there was an early increase in these cytokines in patients who died from the disease. This suggested that immune modulation at the early stages of the disease could be beneficial for patient management [49].
In a study that assessed the viral load in the saliva and in the nasopharynx of COVID-19 patients, increased salivary viral load was found to be associated with increased levels of IL-6, IL-18 and other inflammatory markers associated with severe COVID-19. More importantly, the salivary viral load was reported to correlate with disease severity and was a better predictor of COVID-19 related mortality than the nasopharyngeal viral load [50].
These findings emphasise the importance of reducing the viral load as early as possible during the disease process to improve patient outcome.
In addition to its role in modulating the immune response, the PI3K/Akt pathway has been suggested as a target for the management of COVID-19 due to its involvement in the virus entry to the cell [51].
ACE2 and CD147 expressed on host cells are the two main receptors involved in the endosomal viral entry to the cell, while TMPRSS2 and furin contribute to non-endosomal viral genome entry. Activation of CD147 and of furin leads to the induction of the PI3K/Akt pathway [52] suggesting the potential of targeting the pathway to inhibit the virus entry into the host cells.
The metabolic regulatory complex mTOR has been suggested as a target for COVID-19 therapy due to its reported role in controlling viral infection ability. While promising, this needs to be approached with caution as mTOR plays different roles in the replication and release of different viruses [53].
Interestingly, some of the approved inhibitors of the PI3K/Akt/mTOR pathway, especially rapamycin, which targets mTOR function, were shown to inhibit the replication of viruses including the Middle East respiratory syndrome coronavirus (MERS-CoV) [54] and the influenza virus [55]. This suggests their potential in inhibiting the replication of SARS-CoV-2.
The replication of SARS-CoV-2 relies, hypothetically, on changes in glucose metabolism similar to the Warburg effect in which the PI3K/Akt pathway plays an important role. Hypoxia induces the Warburg effect in pulmonary endothelial cells leading to vasoconstriction and thrombosis. The type of glycolysis could also activate pro-inflammatory immune cells and delay anti-inflammatory responses which are mostly reliant on oxidative metabolism [56]. This emphasises the importance of studying glucose metabolism, in particular the Warburg effect, as part of the management strategy for COVID-19 [57], and suggests a potential role for the PI3K/Akt/mTOR pathway as a target for mitigating viral replication.
Indeed, there is already evidence to support this. Transcriptional analysis of human pulmonary epithelial cells infected with SARS-CoV-2 identified multiple altered signalling pathways. This predicted possible therapeutic targets including two components of the PI3K/Akt/mTOR pathway; AKT and mTOR [58].
When a proteo-transcriptomics analysis was performed on a SARSCoV-2 infected cell line (Huh7, a liver cancer cell line), four pathways were found to be significantly changed during the course of infection, one of which is the mTOR signalling pathway. This was further validated, and components of the pathway, including Akt were found to be activated in a dose-dependant manner. The pan Akt inhibitor MK2206 resulted in a significant reduction in virus production as demonstrated by lower viral transcripts both in the cells and the supernatant [59]. Although inhibitors of different components of the PI3K/Akt/mTOR pathway did not show the same significant effect as MK2206 [59], simultaneously targeting PI3K and mTOR, in addition to BRD2/BRD4, identified as therapeutic targets in COVID-19, using the inhibitor SF2523 (a small molecule inhibitor that targets the PI3K isoforms α and γ, mTOR, BRD4 and DNA-PK) was effective in blocking the replication of SARS-CoV-2 in bronchial epithelial cells [60].
Similarly, a medium-throughput drug screening library identified protein kinase inhibitors that target members of the PI3K/Akt/mTOR pathway as potential therapeutics against SARS-cov-2 infection [61].
Genome wide CRISPR screens identified host factors that are crucial for coronavirus replication (including SARS-cov-2). Autophagy related genes were found to be required for virus replication including TMEM41B and FKBP8 which interact with the PI3K complex that drives autophagy [62]. Deletions of PI3K subunits identified by CRISPR screens of two common cold coronaviruses led to a decrease in coronavirus replication including the replication of SARS-cov-2 [63].
5. Approved drugs and the potential they offer
There are few drugs that target the PI3K/Akt/mTOR pathway that have been approved by the FDA for clinical indications. Table 1 lists the main approved inhibitors and their indications in addition to a summary of their adverse reactions. As can be seen, there are two drugs that have been approved that target mTOR (rapamycin and everolimus), in addition to a number of PI3K inhibitors (pan or isoform specific). To our knowledge, currently there are no approved Akt inhibitors, although many are in clinical trials.
Table 1.
| Drug | Target | Indication approved for (FDA) | Adverse reactions |
|---|---|---|---|
| Rapamycin (Sirolimus, AY 22989 I 2190A, NSC 226080, RAPA. Rapammune…etc.) | mTOR |
|
|
| Everolimus (001, RAD, 40-O-(2-hydroxyethyl)-rapamycin, Afinitor, Certican…etc |
mTOR |
|
|
| Idelalisib (870281-82-6, CAL-101, Zydelig, GS-1101) | PI3K-δ |
|
|
| Copanlisib (1032568-63-0, BAY 80–6946, Aliqopa, BAY-80-6946) |
Pan PI3K |
|
|
| Duvelisib (COPIKTRA, 1201438-56-3, IPI-145, (S)-3-(1-((9H-Purin-6-yl)amino)ethyl)-8-chloro-2-phenylisoquinolin-1(2H)-one, INK-1197) | PI3K-δ and PI3K-γ |
|
|
| Alpelisib (1217486-61-7, BYL-719, NVP-BYL719) | PI3K-α |
|
|
| Umbralisib (UKONIQ, rp-5264, 1532533-67-7, TGR-1202) |
PI3K-δ |
|
|
While rapamycin is indicated for kidney transplant patients as a prophylaxis against organ rejection, the majority of the other drugs are indicated for cancer treatment due to their direct effects on cancer cells [64, 65].
A quick look at the adverse reactions associated with these drugs (Table 1) points to their significant impact on the immune system. While these effects could be severe, it is important to emphasise that these are associated with relatively high cancer therapeutic doses.
When used as immune modulators (as proposed in this mini review), only low doses of these drugs are needed to produce the desired effect without impacting the ability of the immune system to raise a response against infections. Therefore, the use of inhibitors in this essential pathway to modulate the immune system should be approached with caution to maximise their benefit while minimizing their adverse effects.
Given the availability of clinically approved drugs that target different components of the PI3K/Akt/mTOR pathway (Table 1), their potential in the management of COVID-19 could be explored on their own, or in combination with steroids and anti-IL6R antibodies. While the approved inhibitors that target PI3K (pan or isoform specific) can be tested for enhancing the antiviral immune response in early stages of the disease, rapamycin has a clear potential in tackling the exaggerated immune response during the later phase of the severe disease.
6. Summary
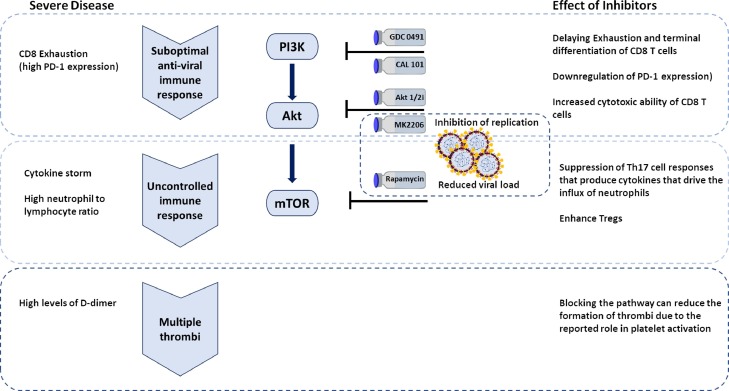
This short review provides an overview of the potential of targeting the PI3K/Akt/mTOR pathway as part of the strategy of tackling COVID-19 (Fig. 2 ). In early stages of the disease, targeting this pathway can inhibit the entry and replication of the virus and reduce the viral load thus improving patient outcome. Furthermore, during the early stages of the immune response, targeting PI3K and Akt can enhance the effector function and supress the suppressor function thus helping to eliminate the infection before it proceeds to immune dysregulation. When patients progress to uncontrolled immune responses, targeting mTOR is a potential therapeutic strategy to suppress the cytokine storm, inhibit neutrophil recruitment and enhance the suppressor regulatory T cells. Due to its involvement in clot formation, targeting this pathway has a potential protective role against thrombosis associated with severe COVID-19 cases.
Fig. 2.
The potential of targeting different components of the PI3K/Akt/mTOR pathway to tackle COVID-19.
We therefore believe that exploring the potential of this pathway is promising in tackling this global challenge.
Funding
This work was supported by Friends of ANCHOR pilot research award [grant number RS 2019 004, 2020–2021]
Declaration of Competing Interest
None.
Biographies

Rasha Abu-Eid: Dr Abu-Eid completed her PhD in Oral Pathology in 2005 from the University of Birmingham, England. After holding different positions in the University of Birmingham, University of Jordan and the University of Aberdeen, in 2011, she was successful in securing a research fellowship from the King Hussein Institute for Biotechnology and Cancer to conduct research in the field of cancer immunology in USA. In October 2016, she re-joined the University of Aberdeen as a Senior Lecturer in Oral Sciences and is currently the lead for cancer research at the Institute of Dentistry.

Frank James Ward: Frank Ward studied Immunology at King's College London and received his PhD from the University of London in 1996. He moved to the University of Aberdeen in 2000 to pursue his main research interest, which is to investigate how the immune system maintains immunological tolerance, particularly in autoimmune disease and cancer. During the last 10 years he has worked on the less well-studied soluble isoform of CTLA-4 as a potential checkpoint inhibitor target for a range of cancers. Frank has published data in several journals based on patient focused studies around sCTLA-4 and has presented at national and international conferences.
References
- 1.World_Health_Organization, WHO Coronavirus Disease (COVID-19) Dashboard, 2021. https://covid19.who.int/. 2021).
- 2.Pedersen S.F., Ho Y.C. SARS-CoV-2: a storm is raging. J. Clin. Invest. 2020;130(5):2202–2205. doi: 10.1172/JCI137647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cui S., Chen S., Li X., Liu S., Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18(6):1421–1424. doi: 10.1111/jth.14830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stratton C.W., Tang Y.W., Lu H. Pathogenesis-Directed Therapy of 2019 Novel Coronavirus Disease. J. Med. Virol. 2020 doi: 10.1002/jmv.26610. [DOI] [PubMed] [Google Scholar]
- 5.Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., Chen L., Li M., Liu Y., Wang G., Yuan Z., Feng Z., Wu Y., Chen Y. Reduction and Functional Exhaustion of T Cells in Patients with Coronavirus Disease. (COVID-19), medRxiv preprint (2020) 2019 doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abu Eid R., Ahmad S., Lin Y., Webb M., Berrong Z., Shrimali R., Kumai T., Ananth S., Rodriguez P.C., Celis E., Janik J., Mkrtichyan M., Khleif S.N. Enhanced Therapeutic Efficacy and Memory of Tumor-Specific CD8 T Cells by Ex Vivo PI3K-delta Inhibition. Cancer Res. 2017;77(15):4135–4145. doi: 10.1158/0008-5472.CAN-16-1925. [DOI] [PubMed] [Google Scholar]
- 7.Ahmad S., Abu-Eid R., Shrimali R., Webb M., Verma V., Doroodchi A., Berrong Z., Samara R., Rodriguez P.C., Mkrtichyan M., Khleif S.N. Differential PI3Kdelta Signaling in CD4(+) T-cell Subsets Enables Selective Targeting of T Regulatory Cells to Enhance Cancer Immunotherapy. Cancer Res. 2017;77(8):1892–1904. doi: 10.1158/0008-5472.CAN-16-1839. [DOI] [PubMed] [Google Scholar]
- 8.Patton D.T., Garden O.A., Pearce W.P., Clough L.E., Monk C.R., Leung E., Rowan W.C., Sancho S., Walker L.S., Vanhaesebroeck B., Okkenhaug K. Cutting edge: the phosphoinositide 3-kinase p110 delta is critical for the function of CD4+CD25+Foxp3+ regulatory T cells. J. Immunol. 2006;177(10):6598–6602. doi: 10.4049/jimmunol.177.10.6598. [DOI] [PubMed] [Google Scholar]
- 9.Lee K.S., Lee H.K., Hayflick J.S., Lee Y.C., Puri K.D. Inhibition of phosphoinositide 3-kinase delta attenuates allergic airway inflammation and hyperresponsiveness in murine asthma model. FASEB J. 2006;20(3):455–465. doi: 10.1096/fj.05-5045com. [DOI] [PubMed] [Google Scholar]
- 10.Puri K.D., Doggett T.A., Douangpanya J., Hou Y., Tino W.T., Wilson T., Graf T., Clayton E., Turner M., Hayflick J.S., Diacovo T.G. Mechanisms and implications of phosphoinositide 3-kinase delta in promoting neutrophil trafficking into inflamed tissue. Blood. 2004;103(9):3448–3456. doi: 10.1182/blood-2003-05-1667. [DOI] [PubMed] [Google Scholar]
- 11.Angulo I., Vadas O., Garcon F., Banham-Hall E., Plagnol V., Leahy T.R., Baxendale H., Coulter T., Curtis J., Wu C., Blake-Palmer K., Perisic O., Smyth D., Maes M., Fiddler C., Juss J., Cilliers D., Markelj G., Chandra A., Farmer G., Kielkowska A., Clark J., Kracker S., Debre M., Picard C., Pellier I., Jabado N., Morris J.A., Barcenas-Morales G., Fischer A., Stephens L., Hawkins P., Barrett J.C., Abinun M., Clatworthy M., Durandy A., Doffinger R., Chilvers E.R., Cant A.J., Kumararatne D., Okkenhaug K., Williams R.L., Condliffe A., Nejentsev S. Phosphoinositide 3-kinase delta gene mutation predisposes to respiratory infection and airway damage. Science. 2013;342(6160):866–871. doi: 10.1126/science.1243292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Palma G., Pasqua T., Silvestri G., Rocca C., Gualtieri P., Barbieri A., De Bartolo A., De Lorenzo A., Angelone T., Avolio E., Botti G. PI3Kdelta Inhibition as a Potential Therapeutic Target in COVID-19. Front. Immunol. 2020;11:2094. doi: 10.3389/fimmu.2020.02094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim J.S., Jeong J.S., Kwon S.H., Kim S.R., Lee Y.C. Roles of PI3K pan-inhibitors and PI3K-delta inhibitors in allergic lung inflammation: a systematic review and meta-analysis. Sci. Rep. 2020;10(1):7608. doi: 10.1038/s41598-020-64594-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Campa C.C., Silva R.L., Margaria J.P., Pirali T., Mattos M.S., Kraemer L.R., Reis D.C., Grosa G., Copperi F., Dalmarco E.M., Lima-Junior R.C.P., Aprile S., Sala V., Dal Bello F., Prado D.S., Alves-Filho J.C., Medana C., Cassali G.D., Tron G.C., Teixeira M.M., Ciraolo E., Russo R.C., Hirsch E. Inhalation of the prodrug PI3K inhibitor CL27c improves lung function in asthma and fibrosis. Nat. Commun. 2018;9(1):5232. doi: 10.1038/s41467-018-07698-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lucas C.L., Kuehn H.S., Zhao F., Niemela J.E., Deenick E.K., Palendira U., Avery D.T., Moens L., Cannons J.L., Biancalana M., Stoddard J., Ouyang W., Frucht D.M., Rao V.K., Atkinson T.P., Agharahimi A., Hussey A.A., Folio L.R., Olivier K.N., Fleisher T.A., Pittaluga S., Holland S.M., Cohen J.I., Oliveira J.B., Tangye S.G., Schwartzberg P.L., Lenardo M.J., Uzel G. Dominant-activating germline mutations in the gene encoding the PI(3)K catalytic subunit p110delta result in T cell senescence and human immunodeficiency. Nat. Immunol. 2014;15(1):88–97. doi: 10.1038/ni.2771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Abu Eid R., Friedman K.M., Mkrtichyan M., Walens A., King W., Janik J., Khleif S.N. Akt1 and -2 inhibition diminishes terminal differentiation and enhances central memory CD8(+) T-cell proliferation and survival. Oncoimmunology. 2015;4(5) doi: 10.1080/2162402X.2015.1005448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Araki K., Turner A.P., Shaffer V.O., Gangappa S., Keller S.A., Bachmann M.F., Larsen C.P., Ahmed R. mTOR regulates memory CD8 T-cell differentiation. Nature. 2009;460(7251):108–112. doi: 10.1038/nature08155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Majchrzak K., Nelson M.H., Bowers J.S., Bailey S.R., Wyatt M.M., Wrangle J.M., Rubinstein M.P., Varela J.C., Li Z., Himes R.A., Chan S.S., Paulos C.M. beta-catenin and PI3Kdelta inhibition expands precursor Th17 cells with heightened stemness and antitumor activity. JCI Insight. 2017;2(8) doi: 10.1172/jci.insight.90547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Abu-Eid R., Samara R.N., Ozbun L., Abdalla M.Y., Berzofsky J.A., Friedman K.M., Mkrtichyan M., Khleif S.N. Selective inhibition of regulatory T cells by targeting the PI3K-Akt pathway. Cancer Immunol. Res. 2014;2(11):1080–1089. doi: 10.1158/2326-6066.CIR-14-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mannick J.B., Del Giudice G., Lattanzi M., Valiante N.M., Praestgaard J., Huang B., Lonetto M.A., Maecker H.T., Kovarik J., Carson S., Glass D.J., Klickstein L.B. mTOR inhibition improves immune function in the elderly. Sci. Transl. Med. 2014;6(268) doi: 10.1126/scitranslmed.3009892. 268ra179. [DOI] [PubMed] [Google Scholar]
- 21.National_Institute_for_Health_and_Care_Excellence, COVID-19 rapid guideline: managing COVID-19, 2021. https://www.nice.org.uk/guidance/ng191/resources/fully-accessible-version-of-the-guideline-pdf-pdf-51035553326. 2021).
- 22.Zhang C., Wu Z., Li J.W., Zhao H., Wang G.Q. Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents. 2020;55(5) doi: 10.1016/j.ijantimicag.2020.105954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pelaia C., Calabrese C., Garofalo E., Bruni A., Vatrella A., Pelaia G. Therapeutic Role of Tocilizumab in SARS-CoV-2-Induced Cytokine Storm: rationale and Current Evidence. Int. J. Mol. Sci. 2021;22(6) doi: 10.3390/ijms22063059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liberman A.C., Budzinski M.L., Sokn C., Gobbini R.P., Steininger A., Arzt E. Regulatory and Mechanistic Actions of Glucocorticoids on T and Inflammatory Cells. Front Endocrinol (Lausanne) 2018;9:235. doi: 10.3389/fendo.2018.00235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Andrade M.V., Hiragun T., Beaven M.A. Dexamethasone suppresses antigen-induced activation of phosphatidylinositol 3-kinase and downstream responses in mast cells. J. Immunol. 2004;172(12):7254–7262. doi: 10.4049/jimmunol.172.12.7254. [DOI] [PubMed] [Google Scholar]
- 26.Bi J., Min Z., Yuan H., Jiang Z., Mao R., Zhu T., Liu C., Zeng Y., Song J., Du C., Chen Z. PI3K inhibitor treatment ameliorates the glucocorticoid insensitivity of PBMCs in severe asthma. Clin. Transl. Med. 2020;9(1):22. doi: 10.1186/s40169-020-0262-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Basu S., Golovina T., Mikheeva T., June C.H., Riley J.L. Cutting edge: foxp3-mediated induction of pim 2 allows human T regulatory cells to preferentially expand in rapamycin. J. Immunol. 2008;180(9):5794–5798. doi: 10.4049/jimmunol.180.9.5794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Battaglia M., Stabilini A., Roncarolo M.G. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood. 2005;105(12):4743–4748. doi: 10.1182/blood-2004-10-3932. [DOI] [PubMed] [Google Scholar]
- 29.Long S.A., Buckner J.H. Combination of rapamycin and IL-2 increases de novo induction of human CD4(+)CD25(+)FOXP3(+) T cells. J. Autoimmun. 2008;30(4):293–302. doi: 10.1016/j.jaut.2007.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Strauss L., Whiteside T.L., Knights A., Bergmann C., Knuth A., Zippelius A. Selective survival of naturally occurring human CD4+CD25+Foxp3+ regulatory T cells cultured with rapamycin. J. Immunol. 2007;178(1):320–329. doi: 10.4049/jimmunol.178.1.320. [DOI] [PubMed] [Google Scholar]
- 31.Sun S.Y., Rosenberg L.M., Wang X., Zhou Z., Yue P., Fu H., Khuri F.R. Activation of Akt and eIF4E survival pathways by rapamycin-mediated mammalian target of rapamycin inhibition. Cancer Res. 2005;65(16):7052–7058. doi: 10.1158/0008-5472.CAN-05-0917. [DOI] [PubMed] [Google Scholar]
- 32.Somanath P.R. Is targeting Akt a viable option to treat advanced-stage COVID-19 patients? Am. J. Physiol. Lung Cell. Mol. Physiol. 2020;319(1) doi: 10.1152/ajplung.00124.2020. L45-L47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lagunas-Rangel F.A. Neutrophil-to-lymphocyte ratio and lymphocyte-to-C-reactive protein ratio in patients with severe coronavirus disease 2019 (COVID-19): a meta-analysis. J. Med. Virol. 2020 doi: 10.1002/jmv.25819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Deng J., Yu X.Q., Wang P.H. Inflammasome activation and Th17 responses. Mol. Immunol. 2019;107:142–164. doi: 10.1016/j.molimm.2018.12.024. [DOI] [PubMed] [Google Scholar]
- 35.Joean O., Hueber A., Feller F., Jirmo A.C., Lochner M., Dittrich A.M., Albrecht M. Suppression of Th17-polarized airway inflammation by rapamycin. Sci. Rep. 2017;7(1):15336. doi: 10.1038/s41598-017-15750-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang J., Jin H., Xu Y., Shan J. Rapamycin Modulate Treg/Th17 Balance via Regulating Metabolic Pathways: a Study in Mice. Transplant. Proc. 2019;51(6):2136–2140. doi: 10.1016/j.transproceed.2019.04.067. [DOI] [PubMed] [Google Scholar]
- 37.Wang C.H., Chung F.T., Lin S.M., Huang S.Y., Chou C.L., Lee K.Y., Lin T.Y., Kuo H.P. Adjuvant treatment with a mammalian target of rapamycin inhibitor, sirolimus, and steroids improves outcomes in patients with severe H1N1 pneumonia and acute respiratory failure. Crit. Care Med. 2014;42(2):313–321. doi: 10.1097/CCM.0b013e3182a2727d. [DOI] [PubMed] [Google Scholar]
- 38.Tang N., Li D.J., Wang X., Sun Z.Y. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thrombosis Haemostasis. 2020;18(4):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou F., Yu T., Du R. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study (vol 395, pg 1054, 2020) Lancet. 2020;395(10229) doi: 10.1016/S0140-6736(20)30566-3. 1038-1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McGonagle D., O'Donnell J.S., Sharif K., Emery P., Bridgewood C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2020;2(7):e437–e445. doi: 10.1016/S2665-9913(20)30121-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.A. Gasecka, J.A. Borovac, R.A. Guerreiro, M. Giustozzi, W. Parker, D. Caldeira, G. Chiva-Blanch, Thrombotic Complications in Patients with COVID-19: pathophysiological Mechanisms, Diagnosis, and Treatment, Cardiovasc Drugs Ther (2020). [DOI] [PMC free article] [PubMed]
- 42.Avila J., Long B., Holladay D., Gottlieb M. Thrombotic complications of COVID-19. Am. J. Emerg. Med. 2020 doi: 10.1016/j.ajem.2020.09.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hadid T., Kafri Z., Al-Katib A. Coagulation and anticoagulation in COVID-19. Blood Rev. 2020 doi: 10.1016/j.blre.2020.100761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen Z., Li T., Kareem K., Tran D., Griffith B.P., Wu Z.J. The role of PI3K/Akt signaling pathway in non-physiological shear stress-induced platelet activation. Artif. Organs. 2019;43(9):897–908. doi: 10.1111/aor.13465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kral-Pointner J.B., Schrottmaier W.C., Salzmann M., Mussbacher M., Schmidt G.J., Moser B., Heber S., Birnecker B., Paar H., Zellner M., Knapp S., Assinger A., Schabbauer G. Platelet PI3K Modulates Innate Leukocyte Extravasation during Acid-Induced Acute Lung Inflammation. Thromb. Haemost. 2019;19(10):1642–1654. doi: 10.1055/s-0039-1693693. [DOI] [PubMed] [Google Scholar]
- 46.Wan P., Tan X., Xiang Y., Tong H., Yu M. PI3K/AKT and CD40L Signaling Regulate Platelet Activation and Endothelial Cell Damage in Sepsis. Inflammation. 2018;41(5):1815–1824. doi: 10.1007/s10753-018-0824-5. [DOI] [PubMed] [Google Scholar]
- 47.Yang F., Chen R., Li W.Y., Zhu H.Y., Chen X.X., Hou Z.F., Cao R.S., Zang G., Li Y.X., Zhang W. d-Limonene Is a Potential Monoterpene to Inhibit PI3K/Akt/IKK-alpha/NF-kappaB p65 Signaling Pathway in Coronavirus Disease 2019 Pulmonary Fibrosis. Front Med (Lausanne) 2021;8 doi: 10.3389/fmed.2021.591830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Perlman S. COVID-19 poses a riddle for the immune system. Nature. 2020;584(7821):345–346. doi: 10.1038/d41586-020-02379-1. [DOI] [PubMed] [Google Scholar]
- 49.Lucas C., Wong P., Klein J., Castro T.B.R., Silva J., Sundaram M., Ellingson M.K., Mao T., Oh J.E., Israelow B., Takahashi T., Tokuyama M., Lu P., Venkataraman A., Park A., Mohanty S., Wang H., Wyllie A.L., Vogels C.B.F., Earnest R., Lapidus S., Ott I.M., Moore A.J., Muenker M.C., Fournier J.B., Campbell M., Odio C.D., Casanovas-Massana A., Yale I.T., Herbst R., Shaw A.C., Medzhitov R., Schulz W.L., Grubaugh N.D., Dela Cruz C., Farhadian S., Ko A.I., Omer S.B., Iwasaki A. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584(7821):463–469. doi: 10.1038/s41586-020-2588-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Silva J., Lucas C., Sundaram M., Israelow B., Wong P., Klein J., Tokuyama M., Lu P., Venkataraman A., Liu F., Mao T., Oh J.E., Park A., Casanovas-Massana A., Vogels C.B.F., Muenker C.M., Zell J., Fournier J.B., Campbell M., Chiorazzi M., Ruiz Fuentes E., Petrone M., Kalinich C.C., Ott I.M., Watkins A., Moore A.J., Nakahata M.I., Grubaugh N.D., Farhadian S., Dela Cruz C., Ko A., Schulz W.L., Ring A.M., Ma S., Omer S., Wyllie A.L., Iwasaki A. Saliva viral load is a dynamic unifying correlate of COVID-19 severity and mortality. medRxiv. 2021 [Google Scholar]
- 51.Khezri M.R. PI3K/AKT signaling pathway: a possible target for adjuvant therapy in COVID-19. Hum. Cell. 2021 doi: 10.1007/s13577-021-00484-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lokhande A.S., Devarajan P.V. A review on possible mechanistic insights of Nitazoxanide for repurposing in COVID-19. Eur. J. Pharmacol. 2021;891 doi: 10.1016/j.ejphar.2020.173748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Maiese K. The Mechanistic Target of Rapamycin (mTOR): novel Considerations as an Antiviral Treatment. Curr Neurovasc Res. 2020;17(3):332–337. doi: 10.2174/1567202617666200425205122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kindrachuk J., Ork B., Hart B.J., Mazur S., Holbrook M.R., Frieman M.B., Traynor D., Johnson R.F., Dyall J., Kuhn J.H., Olinger G.G., Hensley L.E., Jahrling P.B. Antiviral potential of ERK/MAPK and PI3K/AKT/mTOR signaling modulation for Middle East respiratory syndrome coronavirus infection as identified by temporal kinome analysis. Antimicrob. Agents Chemother. 2015;59(2):1088–1099. doi: 10.1128/AAC.03659-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kuss-Duerkop S.K., Wang J., Mena I., White K., Metreveli G., Sakthivel R., Mata M.A., Munoz-Moreno R., Chen X., Krammer F., Diamond M.S., Chen Z.J., Garcia-Sastre A., Fontoura B.M.A. Influenza virus differentially activates mTORC1 and mTORC2 signaling to maximize late stage replication. PLoS Pathog. 2017;13(9) doi: 10.1371/journal.ppat.1006635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pålsson-McDermott E.M., O'Neill L.A.J. Targeting immunometabolism as an anti-inflammatory strategy. Cell Res. 2020;30(4):300–314. doi: 10.1038/s41422-020-0291-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Icard P., Lincet H., Wu Z., Coquerel A., Forgez P., Alifano M., Fournel L. The key role of Warburg effect in SARS-CoV-2 replication and associated inflammatory response. Biochimie. 2021;180:169–177. doi: 10.1016/j.biochi.2020.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fagone P., Ciurleo R., Lombardo S.D., Iacobello C., Palermo C.I., Shoenfeld Y., Bendtzen K., Bramanti P., Nicoletti F. Transcriptional landscape of SARS-CoV-2 infection dismantles pathogenic pathways activated by the virus, proposes unique sex-specific differences and predicts tailored therapeutic strategies. Autoimmun. Rev. 2020;19(7) doi: 10.1016/j.autrev.2020.102571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Appelberg S., Gupta S., Svensson Akusjarvi S., Ambikan A.T., Mikaeloff F., Saccon E., Vegvari A., Benfeitas R., Sperk M., Stahlberg M., Krishnan S., Singh K., Penninger J.M., Mirazimi A., Neogi U. Dysregulation in Akt/mTOR/HIF-1 signaling identified by proteo-transcriptomics of SARS-CoV-2 infected cells. Emerg Microbes Infect. 2020;9(1):1748–1760. doi: 10.1080/22221751.2020.1799723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Acharya A., Pandey K., Thurman M., Challagundala K.B., Vann K.R., Kutateladze T.G., Morales G.A., Durden D.L., Byrareddy S.N. Blockade of SARS-CoV-2 infection in vitro by highly potent PI3K-alpha/mTOR/BRD4 inhibitor. bioRxiv. 2021 [Google Scholar]
- 61.Garcia G., Jr., Sharma A., Ramaiah A., Sen C., Purkayastha A., Kohn D.B., Parcells M.S., Beck S., Kim H., Bakowski M.A., Kirkpatrick M.G., Riva L., Wolff K.C., Han B., Yuen C., Ulmert D., Purbey P.K., Scumpia P., Beutler N., Rogers T.F., Chatterjee A.K., Gabriel G., Bartenschlager R., Gomperts B., Svendsen C.N., Betz U.A.K., Damoiseaux R.D., Arumugaswami V. Antiviral drug screen identifies DNA-damage response inhibitor as potent blocker of SARS-CoV-2 replication. Cell Rep. 2021;35(1) doi: 10.1016/j.celrep.2021.108940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kratzel A., Kelly J.N., Brueggemann Y., Portmann J., V'kovski P., Todt D., Ebert N., Steinmann E., Dijkman R., Zimmer G., Pfaender S., Thiel V. A genome-wide CRISPR screen identifies interactors of the autophagy pathway as conserved coronavirus targets. bioRxiv. 2021 doi: 10.1371/journal.pbio.3001490. 2021.02.24.432634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang R., Simoneau C.R., Kulsuptrakul J., Bouhaddou M., Travisano K., Hayashi J.M., Carlson-Stevermer J., Oki J., Holden K., Krogan N.J., Ott M., Puschnik A.S. Functional genomic screens identify human host factors for SARS-CoV-2 and common cold coronaviruses. bioRxiv. 2020 doi: 10.1016/j.cell.2020.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.FDA, FDA-Approved Drugs, 2021. https://www.accessdata.fda.gov/scripts/cder/daf/.
- 65.National_Center_for_Biotechnology_Information, PubChem Compound Summary, 2021. https://pubchem.ncbi.nlm.nih.gov. (Accessed August 30, 2021 2021).
- 66.Limon J.J., Fruman D.A. Akt and mTOR in B Cell Activation and Differentiation. Front. Immunol. 2012;3:228. doi: 10.3389/fimmu.2012.00228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Abdelrasoul H., Werner M., Setz C.S., Okkenhaug K., Jumaa H. PI3K induces B-cell development and regulates B cell identity. Sci. Rep. 2018;8(1):1327. doi: 10.1038/s41598-018-19460-5. [DOI] [PMC free article] [PubMed] [Google Scholar]