Abstract
The number of patients with knee osteoarthritis, the proportion that is obese and the number undergoing unicompartmental knee arthroplasty (UKA) are all increasing. The primary aim of this systematic review was to determine the effects of obesity on outcomes in UKA. A systematic review was performed using PRISMA guidelines and the primary outcome was revision rate per 100 observed component years, with a BMI of ≥ 30 used to define obesity. The MINORS criteria and OCEBM criteria were used to assess risk of bias and level of evidence, respectively. 9 studies were included in the analysis. In total there were 4621 knees that underwent UKA. The mean age in included studies was reported to be 63 years (mean range 59.5–72 years old)) and range of follow up was 2–18 years. Four studies were OCEBM level 2b and the average MINORS score was 13. The mean revision rate in obese patients (BMI > 30) was 0.33% pa (95% CI − 3.16 to 2.5) higher than in non-obese patients, however this was not statistically significant (p = 0.82). This meta-analysis concludes that there is no significant difference in outcomes between obese and non-obese patients undergoing UKA. There is currently no evidence that obesity should be considered a definite contraindication to UKA. Further studies are needed to increase the numbers in meta-analysis to explore activity levels, surgeon’s operative data, implant design and perioperative complications and revision in more depth.
Level of evidence Level III.
Keywords: Unicompartmental knee arthroplasty, Unicondylar knee arthroplasty, Obesity, Body mass index, Reoperation
Introduction
The incidence and prevalence of obesity in the Western World is increasing [3]. According to the World Health Organisation (WHO), the prevalence of obesity has nearly doubled worldwide over the past three and half decades, particularly in developing countries [3]. Obesity is considered a major risk factor for osteoarthritis, thus the implications of this increase directly correlate to the increase in osteoarthritis (OA) and in particular knee OA [41]. Studies have shown that with every 5 kg of weight gain the risk of knee OA increases by 36% [25]. Whilst initial management of knee OA is non-operative, some patients require arthroplasty as the disease progresses and conservative methods become ineffective. Hence with the increase in knee OA, there has been research into the indications and effects of arthroplasty in this group of patients, in particular the use of unicompartmental knee arthroplasty (UKA).
Big data studies have suggested that high UKA revision is due to a caseload effect. Hence low volume surgeons and low centres have an associated higher revision rate [4, 16]. There is an assumption that UKA has a higher revision rate than total knee arthroplasty (TKA) primarily because it is easier to revise. In addition, it has other advantages, such as a more rapid recovery, fewer complications and better function. Therefore, there may be an argument that UKA may be the optimal choice for obese patients, particularly if they are young. Proponents of UKA believe that UKA can potentially be used in up to 50% of patients needing knee replacements, with the proportion being higher in younger patients [20].
However, since Kozinn and Scott highlighted in their seminal paper in 1989 that UKA is not ideal with a body weight of > 82 kg [23], most surgeons have avoided using UKA in obese patients. Despite this, some surgeons have used unicompartmental knee arthroplasty (UKA) in this situation and have challenged the view that obesity should be considered to be a contraindication [15, 35]. For example, Caivagnac showed that obesity had no adverse outcomes in UKA with a 92% 10-year survival rate [11]. Similarly, Murray et al. in a prospective study of 2438 Oxford UKA found that increasing BMI was not associated with an increasing failure rate [30]. In contrast, Kandil et al. found the overall short-term revision rate in obese patients undergoing UKA was twice as high as the revision rate in non-obese patients [18].
There is currently no published systematic review and meta-analysis evaluating the effects of obesity on UKA. Current studies investigating the effect of obesity on the outcome of UKA have been limited by short term follow-ups [24, 31], few revisions [11, 42] and small cohorts [6, 31, 38, 43]. Hence the aim of this systematic review and meta-analysis was to identify the influence of obesity, defined as a BMI as ≥ 30, on outcomes of UKA. In addition, it aims to determine whether increased BMI represents a risk factor for failure and if it does, what BMI is associated with worse outcomes and what the mechanisms of failure are. Thus, the null hypothesis is that there is no difference in revision rate for obese patients undergoing a UKA compared to non-obese patients. This study should inform surgical decision-making in UKA patient selection to improve outcomes, answer an important research question and add to the growing knowledge of the optimal management of knee OA in obese patients.
Patients and methods
Search methods
PubMed, Ovid and Web of Science were searched for clinical studies that reported the influence of BMI on outcomes following UKA for osteoarthritis between 1980 to June 2020. Obesity was defined as a BMI ≥ 30. The search terms were set with the aid of a trained librarian (Appendices). Titles and abstracts were screened for relevance using the Preferred Reporting in Systematic Reviews and Meta-analysis (PRISMA) guidelines [29].
Outcomes of interest
The primary research question was to determine whether the outcome of UKA was influenced by BMI. The primary outcome of interest was revision rate due to any cause. Initial comparison was between obese patients (BMI ≥ 30) and non-obese patients (BMI < 30). Subgroup analysis was undertaken to compare patients with a BMI ≥ 35 and BMI < 35 undergoing UKA to further evaluate the ideal BMI cut off.
Subgroup analysis was also performed by failure mechanism, specifically assessing revision for infection, aseptic loosening and unexplained pain. These three secondary outcomes were chosen, as they are main determinants of failure in UKA [28, 39]. The influence of prosthesis design, fixed vs. mobile bearing, will also be assessed. Postoperative Oxford Knee Score (OKS) and Knee Society Score (KSS) will also be evaluated comparing the two BMI groups.
Selection criteria
Comparative studies that compared revision rates for obese (BMI ≥ 30) and non-obese patients (< 30) were included. Studies that compared revision rates for patients with BMI ≥ 35 and BMI < 35 were also included.
All types of UKA prosthesis designs (fixed bearing, mobile bearing) were included. Only studies with a minimum 1 year follow-up period were included. Any articles where the data were not extractable was excluded. The remaining inclusion and exclusion criteria are highlighted in Table 1.
Table 1.
Table of inclusion and exclusion criteria used in the systematic review
| Inclusion Criteria |
|
Comparative studies in which the outcomes were reported according to BMI Observational studies and randomised controlled trials Minimum 1 year follow up period All types of unicompartmental knee prosthesis designs Full text available Data must be extractabl |
| Exclusion Criteria |
|
Review articles, expert opinions, surgical techniques and abstracts from scientific meetings (data not available) Studies not in English language |
Data extraction and analysis
Two independent reviewers (OM and AC) performed the screening process in line with PRISMA guidelines. Following database screening, the remaining articles were analysed. Duplicates were removed. Demographic data extracted from each study included mean age, follow up period, number of knees and number of patients.
Level of evidence
The methodology of the studies was assessed using the list of criteria as recommended by the Cochrane Collaboration and an Oxford Centre for Evidence-Based Medicine (OCEBM) level of evidence (LoE) was assigned to each study [32]. A LoE of 1 is the highest level of evidence and assigned to a high-quality randomised control trial or meta-analysis.
Risk of bias
Risk of Bias was assessed using the Methodological Index for Non-Randomised Studies (MINORS) criteria [37]. The MINORS criteria are a 12-item checklist that assigns a score on various parameters to assign an overall risk of bias score out of 24 (low level risk of bias). MINORS assessment was performed to ensure there was a descriptive summary of source of bias in each study. No study was excluded based on this assessment.
Statistical analysis
The data from the included studies were pooled and analysed using REVMAN (Cochrane Collaboration, Copenhagen) review manager statistical software and R (R Core Team, Vienna) statistical software programme.
The total revision rate per 100 observed component years was calculated by multiplying the number of cases by the mean follow-up for each study. The total number of revisions was then divided by the total observed component years and multiplied by 100. The 95% confidence intervals were calculated using the Clopper Pearson exact method [10].
The weighted odds ratio and 95% confidence interval (CI) was calculated for dichotomous variables. The weighted means difference was calculated for continuous variables. The heterogeneity of the included studies was calculated using the I2 statistic to describe the percentage of variation across studies that is due to heterogeneity rather than chance [17]. A random effects model was used. A chi-square p value < 0.1 was suggestive of statistical heterogeneity.
Results
Study selection
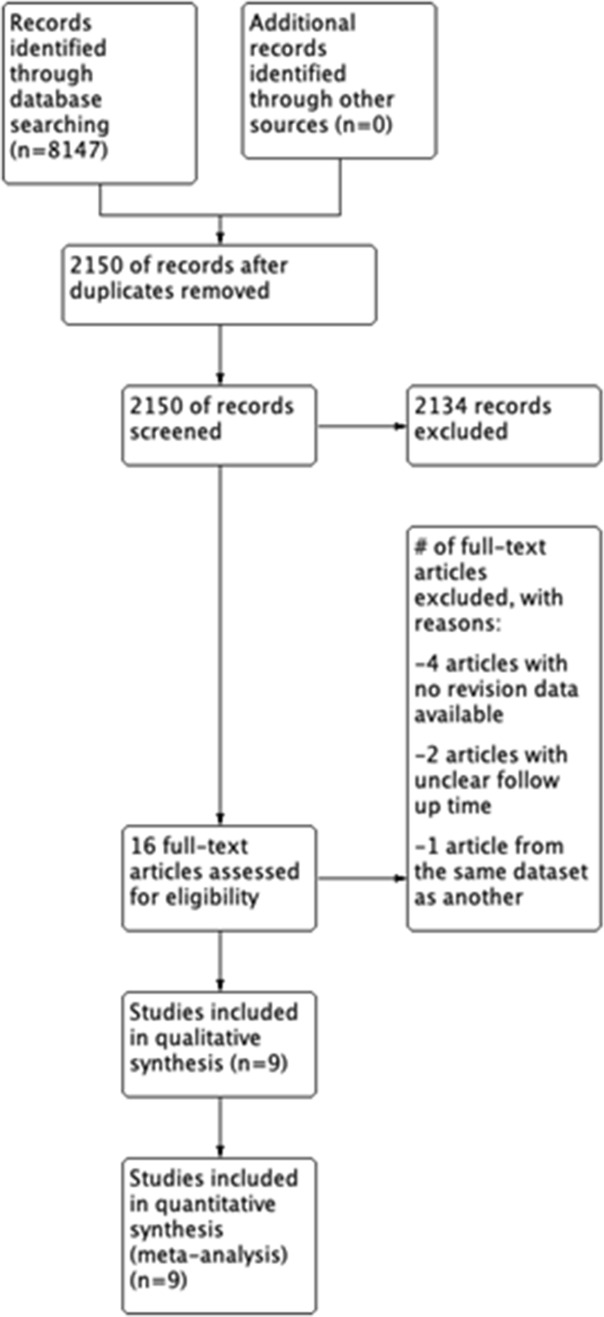
The PRISMA flow diagram showing selection of studies is illustrated in Fig. 1. Nine studies were included in the analysis having met the inclusion criteria. Four (44%) studies were prospective and five were retrospective. No studies were randomised. Eight (89%) studies could be used for a comparison of patients undergoing UKA with BMI above and below 30 and four (44%) studies could be used for comparison of patients with BMI above and below 35. In total, there were 4621 knees with a mean age of 63 years (range) that underwent UKA. Table 2 summarises the baseline characteristics and demographics of the studies.
Fig. 1.
PRISMA flow diagram of record identification, screening and selection in meta-analysis
Table 2.
Study characteristic, demographics and LoE of studies meeting inclusion criteria
| Study | Year | Type of Study | Type of Prosthesis (Fixed bearing/mobile bearing) | No. of Patients | No. of Knees | mean Age, range (years) | BMI categories | Follow-up period (months) | Secondary outcomes of Interest | Level of Evidence (LoE) |
|---|---|---|---|---|---|---|---|---|---|---|
| Seyler [36] | 2009 | Cohort | Fixed bearing | 68 | 80 | 72(44–91) | < 30, > 30 |
60 months (24–128) |
– | 2b |
| Bonutti [8] | 2011 | Retrospective | Fixed bearing | 67 | 80 | 66.5(45–81) | < 35, > 35 |
36 months (24 to 84) |
– | 3b |
| Plate [33] | 2015 | Retrospective | Fixed bearing | 672 | 746 | 64 | < 18.5, 18.5–25, 25–30, 30–35, 35–40, 40–45, > 45 |
33.6 months (24 +) |
– | 3b |
| Zengerink [45] | 2015 | Retrospective | Fixed bearing | 130 | 141 | 60.5 | < 30, > 30 |
60 months (24–144) |
Unexplained pain | 3b |
| Venkatesh [40] | 2016 | Cohort | Fixed bearing | 148 | 175 | 61.7 | < 30, > 30 | 67.2 months (24–120) | Unexplained Pain, aseptic loosening, KSS | 2b |
| Woo [42] | 2017 | Retrospective | Fixed bearing | 673 | 673 | 62 | < 25, 25–30, 30–35, > 35 |
64 months (30–102) |
Unexplained pain, aseptic loosening | 3b |
| Murray [30] | 2013 | Cohort | Mobile bearing | – | 2438 | 64 | < 25, 25–30, 30–35, > 35, 35–40, 40–45, > 45 | 55.2 months (24–144) | Unexplained pain, infection, aseptic loosening, OKS, KSS | 2b |
| Xu [44] | 2019 | Cohort | Fixed bearing | 184 | 184 | 59.5 | > 30, < 30 | Minimum 120 months | Aseptic loosening, infection, KSS, OKS | 2b |
| Polat [34] | 2019 | Retrospective | Mobile bearing | 104 | 104 | 60.2 | > 30, < 30 |
46 months (31.4– 60.6) |
Aseptic loosening, infection | 3b |
Level of evidence
Five (56%) studies had Level 3b evidence and the remainder were Level 2b based on the OCEBM criteria (Table 2). This suggests the results of the meta-analysis may assist in forming basis of recommendations for revision rate, aseptic loosening, infection and aseptic loosening outcomes in the obese population.
Risk of bias
The mean MINORS score was 13 (range 11–20) as shown on Table 3. All the studies had clearly stated aims, adequate statistical analysis and appropriate endpoints. However, most of the studies were not prospective and did not state the baseline equivalence of groups. Furthermore, almost all the studies did not adjust for possible confounding factors in obese patients undergoing UKA; however, due to the nature of the study research question it is likely that every study will have been expected to score low on this metric. Hence, the true effect of obesity on UKA outcomes should be interpreted in context.
Table 3.
Risk of Bias assessment using the MINORS criteria for each study
| Study | Clearly stated aim | Inclusion of consecutive patients | Prospective collection of data | Endpoint appropriate to the aim of the study | Unbiased assessment of the study endpoint | Follow up period appropriate to the aim of the study | Loss of follow up less than 5% | Prospective calculation of the study size | An adequate control group | Contemporary group | Baseline equivalence of groups | Adequate statistical analysis | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Seyler [36] | 2 | 1 | 2 | 2 | 0 | 2 | 0 | 0 | 2 | 1 | 0 | 2 | 14 |
| Bonutti [8] | 2 | 0 | 0 | 2 | 0 | 2 | 0 | 2 | 2 | 1 | 0 | 2 | 13 |
| Plate [33] | 2 | 1 | 0 | 2 | 0 | 2 | 2 | 0 | 2 | 1 | 0 | 2 | 12 |
| Zengerink [45] | 2 | 0 | 0 | 2 | 0 | 2 | 2 | 0 | 2 | 1 | 0 | 2 | 13 |
| Venkatesh [40] | 2 | 0 | 0 | 2 | 0 | 2 | 1 | 0 | 2 | 1 | 0 | 2 | 12 |
| Woo [42] | 2 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 2 | 1 | 0 | 2 | 11 |
| Murray [30] | 2 | 2 | 2 | 2 | 0 | 2 | 2 | 2 | 2 | 2 | 0 | 2 | 20 |
| Xu [44] | 2 | 0 | 0 | 2 | 0 | 2 | 0 | 0 | 2 | 1 | 0 | 2 | 11 |
| Polat [34] | 2 | 1 | 0 | 2 | 0 | 2 | 0 | 0 | 2 | 1 | 0 | 2 | 12 |
Meta-analysis
Table 4 highlights the revision rate and observed component years for each study. The revision rate of UKA in studies of patients with BMI ≥ 30 was 0.33% pa higher than in patients with a BMI < 30. The difference was however not statistically significant (p = 0.82) and was small as compared to the 95% confidence interval (CI), which was from − 2.5% pa to 3.16% pa. (Fig. 2).
Table 4.
Study results showing observed component years and all cause annual revision rate comparing obese (BMI > 30) and non-obese (BMI < 30) patients
| Study | Year | Observed component years | Annual revision rate (%) | ||
|---|---|---|---|---|---|
| BMI < 30 | BMI > 30 | BMI < 30 | BMI > 30 | ||
| Seyler [36] | 2009 | 310 | 90 | 1.612 | 4.444 |
| Bonutti [8] | 2015 | 899 | 1190 | 1.779 | 2.269 |
| Plate [33] | 2015 | 250 | 316 | 0 | 3.164 |
| Zengerink [45] | 2016 | 655 | 325 | 0.763 | 1.538 |
| Venkatesh [40] | 2017 | 2803 | 832 | 0.178 | 0.361 |
| Woo [42] | 2013 | 5676 | 5538 | 0.599 | 0.524 |
| Murray [30] | 2019 | 1420 | 420 | 0.141 | 1.190 |
| Xu [44] | 2019 | 100 | 299 | 0 | 3.68 |
Fig. 2.
Forest plot of overall revision rate after UKA comparing BMI ≥ 30 and BMI < 30
As shown in Fig. 3, the revision rate of UKA in studies of patients with BMI ≥ 35 was 0.36% pa higher than in patients with a BMI < 30. The difference was; however, not statistically significant (p = 0.83) and was small as compared to the 95% (CI), which was from − 3.55% pa to 4.27% pa. This was associated with a low level of heterogeneity (I2 = 0%).
Fig. 3.
Forest plot of overall revision rate after UKA comparing BMI ≥ 35 and BMI < 35
Meta-analysis of subgroup analysis was performed based on the failure mechanism and implant design as shown on Table 5. From the subgroup meta-analysis (Table 5), there was no difference in the secondary outcomes except for unexplained pain and OKS outcome.
Table 5.
Meta-analysis summary of primary outcomes for revision rate comparing both BMI’s and secondary outcomes using BMI cut off of 30
| Outcome | Number of knees | Number of events | Heterogeneity, I2 (%) | p value | Odds ratio or mean difference (95% CI) |
|---|---|---|---|---|---|
| Overall revision rate (BMI > 30, BMI < 30) | 4526 | 149 | 0 | 0.82 | MD − 0.18 (95% CI − 2.36 to 2.01) |
| Overall revision rate (BMI > 35, BMI < 35) | 4028 | 104 | 0 | 0.86 | MD -0.36 (95% CI − 4.27 to 3.55) |
| Infection revision rate (BMI > 30, BMI < 30) | 757 | 10 | 0 | 0.41 | OR 1.73 (95% CI 0.47 to 6.31) |
| Aseptic loosening revision rate, BMI > 30, BMI < 30) | 975 | 6 | 0 | 0.52 | OR 1.56 (95% CI 0.41 to 5.99) |
| Unexplained pain revision rate (BMI > 30, BMI < 30) | 974 | 13 | 0 | 0.04 | OR 3.66 (95% CI 1.09 to 12.30) |
| Revision rate (mobile bearing, BMI > 30, BMI < 30) | 2542 | 49 | 0 | 0.99 | OR − 0.02(95% CI − 3.16 to 2.50) |
| Revision rate (fixed bearing, BMI > 30, BMI < 30) | 1984 | 83 | 0 | 0.73 | OR -0.80 (95% CI, − 5.29 to 3.69) |
| KSS (BMI > 30, BMI < 30) | 359 | – | 45 | 0.32 | MD 0.32 (95% CI − 2.2 to 2.84) |
| OKS (BMI > 30, BMI < 30) | 1110 | – | 0 | < 0.05 | MD − 1.81 (95% CI − 2.75 to − 0.86) |
Prosthetic design
Fixed bearing prosthesis was not associated with an increased risk of revision in obese patients (means difference 0.80, 95% CI − 5.29 to 3.69) and neither was mobile bearing prosthesis (means difference 0.02, 95% CI − 3.16 to 2.50) (Table 5).
Discussion
The study showed that there was no significant increase in the risk of revision of UKA implanted in obese patients. This was the finding whether the comparison was made between patients with a BMI above or below 30 and 35. Therefore obesity does not represent a clear risk factor for failure and should not be considered a definite contraindication for UKA. However, for patients with BMI above both 30 and 35 there was an increase in the mean revision rate (0.33% pa and 0.36% pa), which was substantially smaller than the very wide confidence intervals (− 2.5 to 3.16 and − 3.55 to 4.27). This equated to only a small difference of 3–4% in revision rate at 10 years. Furthermore, the risk of bias in the studies was appreciable (Table 3) and there was low heterogeneity in the meta-analysis. Therefore, obesity may be associated with an increased risk of revision so surgeons should continue to be cautious implanting UKA in obese patients, who should be warned that there might be an increased risk of revision.
Every patient that is treated with a UKA could have had a TKA instead. Therefore, when deciding whether to do a UKA in obese patients, it is sensible to consider not just the possible increased revision rate of UKA in the obese, but also the increased revision rate in TKA. A high-quality meta-analysis of 20 studies (15,276 patients) by Kerkhoff showed that the revision rate is higher in the obese (BMI > 30) population undergoing TKA (OR 1.79, 95% CI 1.15–2.78) [21]. Although our study used different methodology and had fewer patients, the increase in mean revision rate with obesity following UKA of about 20% was lower than the increase of about 80% that following TKA. Therefore, if there is a slight increase in revision rate following UKA with obesity this should not necessarily be considered a contraindication and might be an argument for obese patients to undergo a UKA rather than a TKR. Further analysis is warranted to compare outcomes of obese patients undergoing UKA and TKA.
Currently there is no data from registry studies that has looked at obesity outcomes in UKA. This may be due to several factors including caseload effect, threshold of revision and incomplete data. In particular the UKA registry data seem to have significant problems with missing BMI data and in a study by Liddle et al. using the National Joint Registry (NJR) data reporting 52.9% of patients out of 41,986 did not have a recorded BMI [26]. In contrast to UKA, there seems to be clear agreement between meta-analysis in the literature and registry data on TKA and obesity outcomes with greater evidence available regarding the higher revision rate in TKA [9, 12]. Erdem et al. from the Danish knee registry found that survival was affected in patients over the age of 70 years weighing < 60 kg and > 80 kg but weight was not found to affect risk of revision in patients aged 55–70 years [14].
Subgroup analysis
When analysing all the secondary outcomes, the meta-analysis suggests that unexplained pain appears to be the most strongly associated adverse event, with the revision rate for unexplained pain increasing significantly in the obese population (Table 5). Unexplained pain also seems to account for the earliest revision rates in patients undergoing UKA with an average follow up time of 18 months until revision [1]. This may be due to the higher body mass causing bone overload. The revision of a UKA to TKA in the obese population appears to have been performed mostly in the first few years (average 2–2.2 years) in the studies included. This is in keeping with the literature where revision after UKA is seen in two peaks: early failure due to unexplained pain and late failure because of aseptic loosening [6, 13]. However, it must be stressed that due to the sparse number of patients and studies included in the subgroup analysis the results must be interpreted in context. Interestingly there appeared to be a statistically significant difference in the postoperative OKS (Table 5) between the two groups. However, this subgroup analysis only involved two studies and the mean difference may not be clinically important.
There were 757 patients included in the meta-analysis of infection. Ten patients had a superficial infection. Obesity was not associated with a significant increased risk of infection, odds ratio 1.73 (95% CI 0.47–6.31). This is unlike the outcomes of TKA where a meta-analysis of 20 studies has shown a significant increased risk of infection both in the short term and in the long term (OR 1.90, 95% CI 1.46–2.47) [21]. The risk of infection appears to be greater in TKA when compared with the infection rate in this meta-analysis of UKA (OR 1.73, 95% CI 0.47–6.31). Lum et al. looked at outcomes for 650 UKA and 1300 TKA in severely obese patients (BMI > 35) and found that there were lower deep infections rates and lower revision rates in those that underwent UKA [27]. In the recent two arm randomised controlled TOPKAT trial which compared UKA and TKA, there was no subgroup analysis comparing obese patients in either the TKA or the UKA group, however the revision rate in the UKA was lower than TKA [5].
It is generally accepted that obesity is associated with more intraoperative and postoperative complications [7, 19]. This may in part be due to the higher comorbidities in the obese population. Hence, it is interesting to note that only 4 studies documented postoperative complications and only one study documented intraoperative complications. No statistical model of the included studies adjusted for co-morbidities. It was difficult to assess the studies for baseline equivalence and confounding (Table 3) hence there is a clear need for future studies to adjust for the comorbidities using an appropriate comorbidity index tool in the statistical model.
Prosthetic design
Prosthetic design plays an important influence on UKA performance and outcomes [2]. The study included a mixture of mobile bearing and fixed bearing UKA designs. There is evidence to suggest that fixed bearing UKA results in greater contact stress on the polyethylene inset which may eventually lead to a failure associated with wear or tibial component loosening [22]. Tibial component loosening is more frequent in the all-polyethylene designs than metal-backed prosthesis [22]. Although the increase in revision rate with obesity with the fixed bearing UKR (0.8% pa) was higher than with the mobile (0.2% pa) the difference was not statistically significant.
Limitations
Owing to limited data and information available we were only able to explore the BMI thresholds of 30 and 35. The overall low number of studies heavily limited the resulting meta-analysis however by collating this available information, this study can be used as a reference point to inform knee surgeons when managing obese patients. Ideally each BMI category (e.g. 30–35, 35–40, 40–45 etc.) should be explored individually as some but not others might be contraindicated. Interestingly in the two studies that provided data about BMI > 45 there were no revisions. Setting a minimum follow-up period of one year, as part of the inclusion criteria would capture most of the early implant failures however one can argue that having no minimum follow up period would capture all the studies from a pragmatic analysis perspective. Another limitation of the study is to not include the change in patient reported outcome measures (PROMs) or postoperative PROMs as part of the meta-analysis however this was due to inconsistent reporting, missing data and heterogeneity.
The other main limitation of the meta-analysis is the decision to include all comparative studies, but this was made as it represented the best available evidence. Another challenge found was differentiating between studies that have been reported as prospective but are actually retrospective by design with prospective outcome data collection. However, the methodology scoring criteria applied showed that the studies were comparable and that pooling of the data was acceptable.
A key finding is the missing information presented in the studies. In particular, the lack of information on the performance and caseload of the surgeon performing the UKA. A study by Liddle et al. showed that surgeons performing fewer than 10 UKA’s per year had an 8-year survival of 87.9% compared with 92.4% for surgeons who performed 30 UKAs per year [26].
Conclusions
Overall, this meta-analysis shows that a higher BMI does not lead to significantly worse outcomes of patients treated with a UKA. Therefore obese patients should not be excluded from undergoing a UKA based on BMI alone. However the revision rate for unexplained pain was the most strongly associated cause of revision in the obese population. There was a trend to increased revision rates with BMI > 30 and > 35 and with fixed bearing devices. Further studies are needed to increase the numbers in meta-analysis to explore activity levels, surgeon’s operative data, implant design and perioperative complications and revision in more depth. However, based on the available evidence obesity should not be considered a contraindication to UKA.
Acknowledgements
The authors would like to thank Cairns Library staff for all their assistance in accessing databases.
Abbreviations
- BMI
Body Mass Index
- CI
Confidence interval
- KSS
Knee society score
- LoE
Level of evidence
- MD
Mean difference
- MINORS
Methodological Index for non-randomised studies
- NJR
National joint registry
- OA
Osteoarthritis
- OCEBM
Oxford Centre for Evidence-Based Medicine
- OKS
Oxford Knee Score
- OR
Odds ratio
- PRISMA
Preferred reporting in systematic reviews and meta-analyses
- PROMs
Patient reported outcome measures
- TKA
Total knee arthroplasty
- UKA
Unicompartmental knee arthroplasty
- UKR
Unicompartmental knee replacement
- WHO
World Health Organisation
Appendix
Search terms for online databases
Medline (Ovid) (n = 2224)
| Term | Studies | |
|---|---|---|
| 1 | Obesity/ | 169,746 |
| 2 | Body Mass Index/ | 118,030 |
| 3 | Body weight/ | 184,387 |
| 4 | Overweight/ | 22,474 |
| 5 | Obes*.ti, ab | 274,098 |
| 6 | (Body mass index or body-mass index). ti, ab | 166,193 |
| 7 | bmi.ti, ab | 129,016 |
| 8 | Overweight.ti, ab | 62,903 |
| 9 | 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 | 629,825 |
| 10 | Arthroplasty, replacement, knee/ | 21,619 |
| 11 | (Unicompartmental or uni-compartmental or UKA or UKR).ti, ab | 2210 |
| 12 | (knee adj3(arthroplast* or replace* or prosthes*)).ti,ab | 30,537 |
| 13 | Knee prosthesis/ | 11,096 |
| 14 | 10 or 12 or 13 | 35,781 |
| 15 | 11 and 14 | 1952 |
Web of Science (n = 2510)
Obesity OR overweight OR BMI OR “Body Mass Index” OR “Body-mass index” OR “Obes*” And “Knee arthroplast*” OR “Knee Prosth*” OR “Knee Replace*” OR “Unicompartmental Knee” OR UKA OR UKR OR “Uni-compartmental”.
PubMed Search (n = 3414).
(((((((((obesity) OR Body mass index) OR body weight) OR overweight[Title/Abstract]) OR obes*[Title/Abstract]) OR ((Body mass index[Title/Abstract] OR Body-mass index)[Title/Abstract]))))) AND ((((((((((prosthesis) OR arthroplasty) OR replacement) OR arthroplasty[tw]) OR replacement[tw]) AND knee)))).
Author contributions
OM was involved with study design, data collection, statistical analysis, drafting, reviewing and editing of the manuscript. TWH was involved with data collection, statistical analysis and drafting of the manuscript. AJC was involved with data collection, drafting and editing of the manuscript. SJM, BK and DWM were involved with study design, reviewing and editing of the manuscript. All contributors met the ICMJE and journal criteria for authorship and approved the final manuscript.
Funding
Nothing to declare.
Compliance with ethical standards
Conflict of interest
OM, AJC, SJM and BK have no conflicts of interest. This is an adapted manuscript of a postgraduate university project submitted by OM. TWH owns the copyright for the Decision Aid for Medial UKR licensed through Oxford University Technology Transfer Office. DWM has an industrial grant from Zimmer Biomet paid to the University of Oxford. DWM receives consultancy payments and royalties from Zimmer Biomet relating to knee replacement.
Ethical approval
None required.
Informed consent
None required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Argenson JN, Chevrol-Benkeddache Y, Aubaniac JM. Modern unicompartmental knee arthroplasty with cement: a three to ten-year follow-up study. J Bone Joint Surg Am. 2002;84:2235–2239. doi: 10.2106/00004623-200212000-00017. [DOI] [PubMed] [Google Scholar]
- 2.Argenson JN, Parratte S. The unicompartmental knee: design and technical considerations in minimizing wear. Clin Orthop Relat Res. 2006;452:137–142. doi: 10.1097/01.blo.0000229358.19867.60. [DOI] [PubMed] [Google Scholar]
- 3.Arroyo-Johnson C, Mincey KD. Obesity Epidemiology Worldwide. Gastroenterol Clin North Am. 2016;45:571–579. doi: 10.1016/j.gtc.2016.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baker P, Jameson S, Critchley R, Reed M, Gregg P, Deehan D. Center and surgeon volume influence the revision rate following unicondylar knee replacement: an analysis of 23,400 medial cemented unicondylar knee replacements. J Bone Joint Surg Am. 2013;95:702–709. doi: 10.2106/JBJS.L.00520. [DOI] [PubMed] [Google Scholar]
- 5.Beard DJ, Davies LJ, Cook JA, MacLennan G, Price A, Kent S, et al. The clinical and cost-effectiveness of total versus partial knee replacement in patients with medial compartment osteoarthritis (TOPKAT): 5-year outcomes of a randomised controlled trial. Lancet (Lond, Engl) 2019;394:746–756. doi: 10.1016/S0140-6736(19)31281-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Berend KR, Lombardi AV, Jr, Mallory TH, Adams JB, Groseth KL. Early failure of minimally invasive unicompartmental knee arthroplasty is associated with obesity. Clin Orthop Relat Res. 2005;440:60–66. doi: 10.1097/01.blo.0000187062.65691.e3. [DOI] [PubMed] [Google Scholar]
- 7.Bergin PF, Russell GV. The effects of obesity in orthopaedic care. Instr Course Lect. 2015;64:11–24. [PubMed] [Google Scholar]
- 8.Bonutti PM, Goddard MS, Zywiel MG, Khanuja HS, Johnson AJ, Mont MA. Outcomes of unicompartmental knee arthroplasty stratified by body mass index. J Arthroplasty. 2011;26:1149–1153. doi: 10.1016/j.arth.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 9.Boyce L, Prasad A, Barrett M, Dawson-Bowling S, Millington S, Hanna SA, et al. The outcomes of total knee arthroplasty in morbidly obese patients: a systematic review of the literature. Arch Orthop Trauma Surg. 2019;139:553–560. doi: 10.1007/s00402-019-03127-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Campi S, Pandit HG, Dodd CAF, Murray DW. Cementless fixation in medial unicompartmental knee arthroplasty: a systematic review. Knee Surg Sports Traumatol Arthrosc. 2017;25:736–745. doi: 10.1007/s00167-016-4244-5. [DOI] [PubMed] [Google Scholar]
- 11.Cavaignac E, Lafontan V, Reina N, Pailhe R, Wargny M, Laffosse JM, et al. Obesity has no adverse effect on the outcome of unicompartmental knee replacement at a minimum follow-up of seven years. Bone Joint J. 2013;95-b:1064–1068. doi: 10.1302/0301-620X.95B8.31370. [DOI] [PubMed] [Google Scholar]
- 12.Chaudhry H, Ponnusamy K, Somerville L, McCalden RW, Marsh J, Vasarhelyi EM. Revision rates and functional outcomes among severely, morbidly, and super-obese patients following primary total knee arthroplasty: a systematic review and meta-analysis. JBJS Rev. 2019;7:e9–e9. doi: 10.2106/JBJS.RVW.18.00184. [DOI] [PubMed] [Google Scholar]
- 13.Epinette JA, Brunschweiler B, Mertl P, Mole D, Cazenave A. Unicompartmental knee arthroplasty modes of failure: wear is not the main reason for failure: a multicentre study of 418 failed knees. Orthop Traumatol Surg Res. 2012;98:S124–130. doi: 10.1016/j.otsr.2012.07.002. [DOI] [PubMed] [Google Scholar]
- 14.Gøttsche D, Gromov K, Viborg PH, Bräuner EV, Pedersen AB, Troelsen A. Weight affects survival of primary total knee arthroplasty: study based on the Danish Knee Arthroplasty Register with 67,810 patients and a median follow-up time of 5 years. Acta Orthop. 2019;90:60–66. doi: 10.1080/17453674.2018.1540091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hamilton TW, Pandit HG, Jenkins C, Mellon SJ, Dodd CAF, Murray DW. Evidence-based indications for mobile-bearing unicompartmental knee arthroplasty in a consecutive cohort of thousand knees. J Arthroplasty. 2017;32:1779–1785. doi: 10.1016/j.arth.2016.12.036. [DOI] [PubMed] [Google Scholar]
- 16.Hamilton TW, Rizkalla JM, Kontochristos L, Marks BE, Mellon SJ, Dodd CAF, et al. The interaction of caseload and usage in determining outcomes of unicompartmental knee arthroplasty: a meta-analysis. J Arthroplasty. 2017;32:3228–3237.e3222. doi: 10.1016/j.arth.2017.04.063. [DOI] [PubMed] [Google Scholar]
- 17.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kandil A, Werner BC, Gwathmey WF, Browne JA. Obesity, morbid obesity and their related medical comorbidities are associated with increased complications and revision rates after unicompartmental knee arthroplasty. J Arthroplasty. 2015;30:456–460. doi: 10.1016/j.arth.2014.10.016. [DOI] [PubMed] [Google Scholar]
- 19.Kempegowda H, Richard R, Borade A, Tawari A, Graham J, Suk M, et al. Obesity is associated with high perioperative complications among surgically treated intertrochanteric fracture of the femur. J Orthop Trauma. 2017;31:352–357. doi: 10.1097/BOT.0000000000000825. [DOI] [PubMed] [Google Scholar]
- 20.Kennedy JA, Matharu GS, Hamilton TW, Mellon SJ, Murray DW. Age and outcomes of medial meniscal-bearing unicompartmental knee arthroplasty. J Arthroplasty. 2018;33:3153–3159. doi: 10.1016/j.arth.2018.06.014. [DOI] [PubMed] [Google Scholar]
- 21.Kerkhoffs GM, Servien E, Dunn W, Dahm D, Bramer JA, Haverkamp D. The influence of obesity on the complication rate and outcome of total knee arthroplasty: a meta-analysis and systematic literature review. J Bone Joint Surg Am. 2012;94:1839–1844. doi: 10.2106/JBJS.K.00820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim KT, Lee S, Lee JI, Kim JW. Analysis and treatment of complications after unicompartmental knee arthroplasty. Knee Surg Relat Res. 2016;28:46–54. doi: 10.5792/ksrr.2016.28.1.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kozinn SC, Scott R. Unicondylar knee arthroplasty. J Bone Joint Surg Am. 1989;71:145–150. doi: 10.2106/00004623-198971010-00023. [DOI] [PubMed] [Google Scholar]
- 24.Kuipers BM, Kollen BJ, Bots PC, Burger BJ, van Raay JJ, Tulp NJ, et al. Factors associated with reduced early survival in the Oxford phase III medial unicompartment knee replacement. Knee. 2010;17:48–52. doi: 10.1016/j.knee.2009.07.005. [DOI] [PubMed] [Google Scholar]
- 25.Lementowski PW, Zelicof SB. Obesity and osteoarthritis. Am J Orthop (Belle Mead NJ) 2008;37:148–151. [PubMed] [Google Scholar]
- 26.Liddle AD, Pandit H, Judge A, Murray DW. Effect of surgical caseload on revision rate following total and unicompartmental knee replacement. J Bone Joint Surg Am. 2016;98:1–8. doi: 10.2106/JBJS.N.00487. [DOI] [PubMed] [Google Scholar]
- 27.Lum ZC, Crawford DA, Lombardi AV, Jr, Hurst JM, Morris MJ, Adams JB, et al. Early comparative outcomes of unicompartmental and total knee arthroplasty in severely obese patients. Knee. 2018;25:161–166. doi: 10.1016/j.knee.2017.10.006. [DOI] [PubMed] [Google Scholar]
- 28.Mohammad HR, Strickland L, Hamilton TW, Murray DW. Long-term outcomes of over 8,000 medial Oxford Phase 3 Unicompartmental Knees-a systematic review. Acta Orthop. 2018;89:101–107. doi: 10.1080/17453674.2017.1367577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097–e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Murray DW, Pandit H, Weston-Simons JS, Jenkins C, Gill HS, Lombardi AV, et al. Does body mass index affect the outcome of unicompartmental knee replacement? Knee. 2013;20:461–465. doi: 10.1016/j.knee.2012.09.017. [DOI] [PubMed] [Google Scholar]
- 31.Naal FD, Neuerburg C, Salzmann GM, Kriner M, von Knoch F, Preiss S, et al. Association of body mass index and clinical outcome 2 years after unicompartmental knee arthroplasty. Arch Orthop Trauma Surg. 2009;129:463–468. doi: 10.1007/s00402-008-0633-7. [DOI] [PubMed] [Google Scholar]
- 32.OCEBM Levels of Evidence Working Group. The Oxford Levels of Evidence 2. 2011; https://www.cebm.net/index.aspx?o=5653. Accessed 20.03.2020.
- 33.Plate JF, Augart MA, Seyler TM, Bracey DN, Hoggard A, Akbar M, et al. Obesity has no effect on outcomes following unicompartmental knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2017;25:645–651. doi: 10.1007/s00167-015-3597-5. [DOI] [PubMed] [Google Scholar]
- 34.Polat AE, Polat B, Gürpınar T, Çarkçı E, Güler O. The effect of morbid obesity (BMI ≥ 35 kg/m(2)) on functional outcome and complication rate following unicompartmental knee arthroplasty: a case–control study. J Orthop Surg Res. 2019;14:266–266. doi: 10.1186/s13018-019-1316-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rodríguez-Merchán EC, Gómez-Cardero P. Unicompartmental knee arthroplasty: current indications, technical issues and results. EFORT Open Rev. 2018;3:363–373. doi: 10.1302/2058-5241.3.170048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Seyler TM, Mont MA, Lai LP, Xie J, Marker DR, Zywiel MG, et al. Mid-term results and factors affecting outcome of a metal-backed unicompartmental knee design: a case series. J Orthop Surg Res. 2009;4:39. doi: 10.1186/1749-799X-4-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for non-randomized studies (minors): development and validation of a new instrument. ANZ J Surg. 2003;73:712–716. doi: 10.1046/j.1445-2197.2003.02748.x. [DOI] [PubMed] [Google Scholar]
- 38.Tabor OB, Jr, Tabor OB, Bernard M, Wan JY. Unicompartmental knee arthroplasty: long-term success in middle-age and obese patients. J Surg Orthop Adv. 2005;14:59–63. [PubMed] [Google Scholar]
- 39.Vasso M, Corona K, D'Apolito R, Mazzitelli G, Panni AS. Unicompartmental knee arthroplasty: modes of failure and conversion to total knee arthroplasty. Joints. 2017;5:44–50. doi: 10.1055/s-0037-1601414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Venkatesh HK, Maheswaran SS. Mid-term results of Miller–Galante unicompartmental knee replacement for medial compartment knee osteoarthritis. J Orthop Traumatol. 2016;17:199–206. doi: 10.1007/s10195-015-0385-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wills AK, Black S, Cooper R, Coppack RJ, Hardy R, Martin KR, et al. Life course body mass index and risk of knee osteoarthritis at the age of 53 years: evidence from the 1946 British birth cohort study. Ann Rheum Dis. 2012;71:655–660. doi: 10.1136/ard.2011.154021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Woo YL, Chen YQ, Lai MC, Tay KJ, Chia SL, Lo NN, et al. Does obesity influence early outcome of fixed-bearing unicompartmental knee arthroplasty? J Orthop Surg (Hong Kong) 2017;25:2309499016684297. doi: 10.1177/2309499016684297. [DOI] [PubMed] [Google Scholar]
- 43.Xing Z, Katz J, Jiranek W. Unicompartmental knee arthroplasty: factors influencing the outcome. J Knee Surg. 2012;25:369–373. doi: 10.1055/s-0031-1299666. [DOI] [PubMed] [Google Scholar]
- 44.Xu S, Lim WJ, Chen JY, Lo NN, Chia SL, Tay DKJ, et al. The influence of obesity on clinical outcomes of fixed-bearing unicompartmental knee arthroplasty: a ten-syear follow-up study. Bone Joint J. 2019;101-b:213–220. doi: 10.1302/0301-620X.101B2.BJJ-2018-0969.R2. [DOI] [PubMed] [Google Scholar]
- 45.Zengerink I, Duivenvoorden T, Niesten D, Verburg H, Bloem R, Mathijssen N. Obesity does not influence the outcome after unicompartmental knee arthroplasty. Acta Orthop Belg. 2015;81:776–783. [PubMed] [Google Scholar]