Abstract
Background
Remission and low disease activity (LDA) have been proposed as the treatment goals for patients with systemic lupus erythematosus (SLE). Several definitions for each have been proposed in the literature.
Objective
To assess the impact of remission/LDA according to various definitions on relevant outcomes in patients with SLE.
Methods
This systematic literature review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses using PubMed (1946–week 2, April 2021), Cochrane library (1985–week 2, week 2, April 2021) and EMBASE (1974–week 2, April 2021). We included longitudinal and cross-sectional studies in patients with SLE reporting the impact of remission and LDA (regardless their definition) on mortality, damage accrual, flares, health-related quality of life and other outcomes (cardiovascular risk, hospitalisation and direct costs). The quality of evidence was evaluated using the Newcastle-Ottawa Scale.
Results
We identified 7497 articles; of them, 31 studies met the inclusion criteria and were evaluated. Some articles reported a positive association with survival, although this was not confirmed in all of them. Organ damage accrual was the most frequently reported outcome, and remission and LDA were reported as protective of this outcome (risk measures varying from 0.04 to 0.95 depending on the definition). Similarly, both states were associated with a lower probability of SLE flares, hospitalisations and a better health-related quality of life, in particular the physical domain.
Conclusion
Remission and LDA are associated with improvement in multiple outcomes in patients with SLE, thus reinforcing their relevance in clinical practice.
PROSPERO registration number
CRD42020162724.
Keywords: systemic lupus erythematosus, outcome assessment, health care, quality of life
Key messages.
What is already known about this subject?
Remission and low disease activity (LDA) have been reported as potential targets in the systemic lupus erythematosus (SLE) treatment.
What does this study add?
Remission and LDA (regardless of the definitions used) are associated with better outcomes.
How might this impact on clinical practice or future developments?
Remission and LDA should be considered as the target for the management of patients with SLE.
However, it is important to have a uniform definition of both.
Introduction
A treat-to-target (T2T) strategy has been proposed for several chronic diseases in order to improve the affected patients’ treatment, and thus, their outcome; in systemic lupus erythematosus (SLE), however, a uniform definition of treatment goals is lacking.
The ideal goal is remission, which was defined in 2015 and modified in 2021 by the DORIS (Definition Of Remission In SLE) group as the absence of clinical disease activity (Clinical Systemic Lupus Erythematosus Disease Activity Index (SLEDAI)=0 and Physician Global Assessment (PGA)<0.5), with no or minimal intake of glucocorticoids (prednisone daily dose not higher than 5 mg/day) and/or immunosuppressive drugs on stable maintenance dose.1 2 However, some modifications of this definition have been reported in the literature.
Nevertheless, as remission state is not achieved frequently,3–5 low disease activity (LDA) has been proposed as an alternative target. To this end, there are several definitions about LDA in the literature; for example, the Asia Pacific Lupus Consortium (APLC) has introduced the lupus low disease activity state (LLDAS): SLEDAI≤4, which allows a low level of disease activity, without activity in major organ systems or new disease activity, PGA≤1, prednisone daily dose not higher than 7.5 mg/day and/or immunosuppressive drugs on maintenance dose.6 The Toronto Lupus Cohort investigators have proposed using the term low disease activity (LDA by Toronto Lupus Cohort): SLEDAI (excluding serology)≤2, without prednisone and immunosuppressive drugs.7
All these definitions allow the use of antimalarials.
The probability of patients achieving these states seems to vary according to a number of factors including race/ethnicity, in particular African ancestry,8 9 age at diagnosis,10 previous disease activity,8 10 11 major organ involvement10 12 and treatment.8–10 Furthermore, the clinical impact of achieving such states in several clinical outcomes has been examined.13 The outcome most frequently evaluated has been organ damage accrual; in fact, in several cohorts, remission and/or LDA have been found to prevent damage, but the exact definitions used for these states have not been uniform.3 6 7 11 14–20
One of the main challenges is to validate whether all these definitions are indeed predictive of outcomes such as organ damage, death, recurrent flares, number of hospitalisations and quality of life (QoL), and which of them would be the better option. Therefore, our aim was to perform a systematic review of the current literature to assess the impact of the existing definitions of remission/LDA on relevant outcomes of patients with SLE.
Methods
Search strategies
A systematic review according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines21 was carried out. The protocol was registered with PROSPERO (CRD42020162724).
We used the electronic databases PubMed (1946-week 2, April 2021), Cochrane library (1985-week 2, week 2, April 2021) and EMBASE (1974-week 2, April 2021) were searched. We used the Medical Subject Heading (MeSH) terms and Key words in all possible combinations using Boolean operators with the following search strategy: ‘systemic lupus erythematosus’, ‘lupus’, ‘SLE’, ‘remission’, ‘low disease activity status’, ‘low lupus disease activity status’, ‘minimal disease activity’. References of all included full-text articles were hand-searched in order to find additional references from the articles that seem to be relevant for the review. Details of the full search strategy are listed in online supplemental table 1.
lupus-2021-000542supp001.pdf (22KB, pdf)
These articles were downloaded into EndNote software (V.9.3.2); duplicates were deleted. Two independent teams examined each selected article and performed data extraction independently (MFU-G and CR-S or CM-P and GP-E). In case of disagreement, a third investigator was consulted. Discrepancies were resolved by consensus. The literature review team also made every effort to identify multiple publications from a single cohort.
Criteria for the selection of studies
We included both observational studies (case–control, cross-sectional or cohort) and clinical trials on adults or children with SLE in LDA (using a validated definition) or remission (as defined by available criteria) and reporting different disease outcomes in the follow-up (mortality, damage, flare, health-related QoL (HRQoL), risk of cardiovascular disease, hospitalisations and direct healthcare cost). A minimum sample size of 100 patients was required for an article to be included. Patients needed to have similar duration of follow-up in studies that reported flare rates (using a validated definition) as percentages; alternatively, reported flares per person-years was used in cases where patients had unequal follow-up duration. Damage data, as assessed by the validated instrument (the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index (SDI)), were considered.
Studies published only as abstracts were excluded.
Articles written in English or Spanish were included. Case reports, case series, editorials, comments, letters and reviews were excluded.
Data extraction
Two reviewers independently screened all articles and applied the eligibility criteria to identify appropriate studies for inclusion; the selected articles were then abstracted, also independently, using a predetermined form. Information was collected on the study characteristics (study design, country, sample size), the number of participants, gender, age, major clinical variables (damage), definition of LDA/remission used, flare rates or flares per person-years, HRQoL scores, HRQoL instruments, hospitalisation rates, mortality rates, direct healthcare cost, definitions of cardiovascular disease and rates or risk of cardiovascular disease. If the same article reported more than one definition of the states or more than one outcome, all of them were included in the respective analyses.
Evaluation of the quality of the studies
The quality of the studies identified was assessed using the Newcastle-Ottawa Scale (NOS) for cohort and case–control studies a tool specifically developed to assess the quality of observational studies.15 The scoring system covers three major domains: selection of cohorts or cases and controls (maximum four points), comparability of selected groups (maximum two points) and ascertainment of either the exposure or the outcome of interest (maximum three points): the resulting score ranges from 0 to 9; a higher score represents a better methodological quality. While there is no validated cut-off value to discern between studies of good or poor quality, studies with a score of ≥7 were arbitrarily defined as being of high quality.22
Strategy for analysis synthesis
Due to the diversity of remission and LDA definitions, outcomes, heterogeneity of the results and of the different statistical tests performed in the selected articles, a meta-analysis was felt not to be feasible for most of the outcome variables; therefore, the studies selected were summarised using a narrative synthesis approach. A description and rationale were provided for grouping studies for synthesis (eg, according to outcomes type). Established metrics were used to measure the direction and magnitude effect of association between remission/LDA and outcomes (eg, OR, risk ratio (RR), HR, among others) when they were available. Summary tables and structured narrative were employed to descriptively summarise and compare each included study and to examine the heterogeneity across studies.23
Results
Study selection and characteristics of studies included
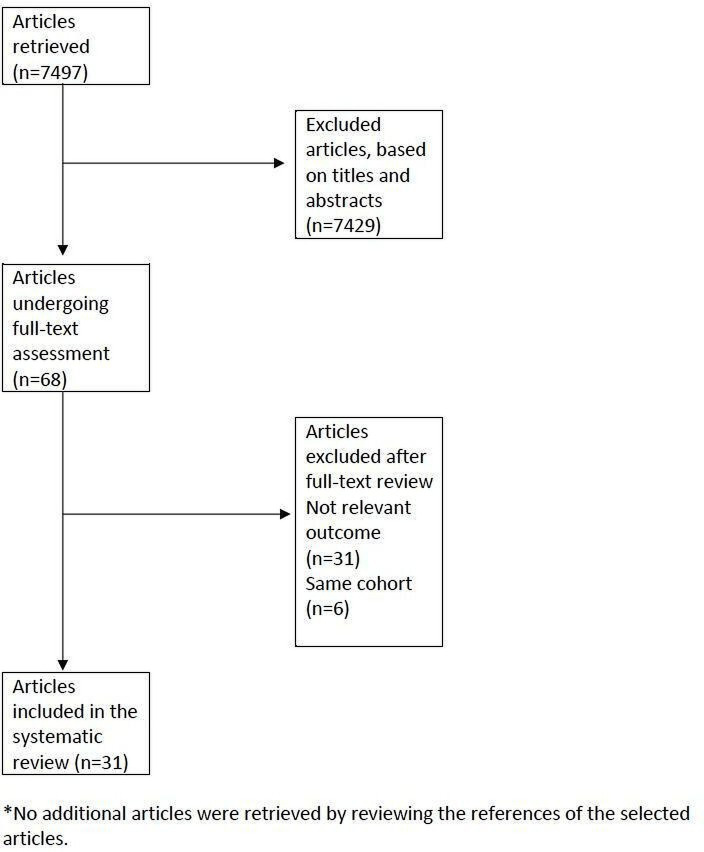
Our search identified 7497 articles, of which 31 studies met the inclusion criteria.3–5 7 11 14–18 20 24–43 The study selection process and reasons for exclusion are shown in figure 1. Four studies were cross sectional, 27 were longitudinal, 12 (38.7%) were from Europe, 10 (32.3%) from Asia and Australia, 5 (16.1%) from Latin America and 4 (12.9%) from the USA and Canada. The large majority of studies were of high quality according to NOS (table 1).
Figure 1.
PRISMA flowchart.
Table 1.
Characteristics of the articles included in this systematic review
| Remission | |||||||||||
| Authors | Country/region | Year of publication | Patients | Ethnicity, % | Cross-sectional or longitudinal | Follow-up years | NOS | ||||
| Caucasian | Asian | Mestizo/Hispanic | African descent | Others | |||||||
| Drenkard et al4 | Mexico | 1996 | 667 | NR | NR | NR | NR | NR | Longitudinal | 11.6 | 5 |
| Medina-Quiñones et al5 | UK | 2016 | 532 | 55.3 | 18.0 | NR | 10.9 | 6.8 | Longitudinal | 12.0 | 7 |
| Polachek et al7* | Canada | 2017 | 620 | NR | NR | NR | NR | NR | Longitudinal | 4.0 | 7 |
| Zen et al16 | Italy | 2017 | 293 | 100.0 | 0.0 | 0.0 | 0.0 | 0.0 | Longitudinal | 7.0 | 7 |
| Ugarte-Gil et al14* | Latin America | 2017 | 1350 | 41.8 | NR | 42.7 | 11.7 | 3.8 | Longitudinal | 2.4 | 8 |
| Tsang-A-Sjoe et al15* | The Netherlands | 2017 | 183 | 68.3 | NR | NR | NR | 31.7 | Longitudinal | 5.0 | 8 |
| Mok et al3 | Hong Kong | 2017 | 769 | 0.0 | 100.0 | 0.0 | 0.0 | 0.0 | Longitudinal | ≥5.0 | 7 |
| Petri et al18* | USA | 2018 | 1356 | 55.0 | NR | NR | 38.0 | 7.0 | Longitudinal | 3.8 | 7 |
| Tani et al11* | Italy | 2018 | 115 | NR | NR | NR | NR | NR | Longitudinal | ≥5.0 | 8 |
| Poomsalood et al24* | Thailand | 2019 | 237 | NR | NR | NR | NR | NR | Cross-sectional | NR | 7 |
| Alarcon et al20* | USA | 2019 | 558 | 28.0 | 0.0 | 35.0 | 37.0 | 0.0 | Longitudinal | NR | 8 |
| Ugarte-Gil et al25 | USA | 2019 | 483 | NR | NR | NR | NR | NR | Longitudinal | NR | 7 |
| Mathian et al26* | France | 2019 | 407 | NR | NR | NR | NR | NR | Longitudinal | 1.0 | 7 |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 10.1 | 87.7 | NR | NR | 2.2 | Longitudinal | 2.2 | 6 |
| Reátegui-Sokolova et al28* | Peru | 2019 | 315 | NR | NR | NR | NR | NR | Longitudinal | NR | 9 |
| Margiotta et al29 | Italy | 2019 | 136 | NR | NR | NR | NR | NR | Cross-sectional | NR | 7 |
| Fasano et al30 | Italy | 2019 | 294 | NR | NR | NR | NR | NR | Longitudinal | 9.0 | 7 |
| Goswami et al31 | India | 2019 | 126 | NR | NR | NR | NR | NR | Longitudinal | 0.5 | 7 |
| Tsang-A-Sjoe et al15 | The Netherlands | 2019 | 154 | 69.5 | NR | NR | NR | 30.5 | Longitudinal | 2.0 | 7 |
| Ugarte-Gil et al33* | Peru | 2020 | 208 | NR | NR | NR | NR | NR | Longitudinal | 2.2 | 9 |
| Floris et al34 * | Italy | 2020 | 116 | 100.0 | 0.0 | 0.0 | 0.0 | 0.0 | Longitudinal | 1.5 | 8 |
| Saccon et al35 | Italy | 2020 | 646 | NR | NR | NR | NR | NR | Longitudinal | 5.0 | 9 |
| Jakez-Ocampo et al36 | Mexico | 2020 | 132 | NR | NR | NR | NR | NR | Cross-sectional | NR | 6 |
| Nikfar et al37 | Iran | 2021 | 193 | NR | NR | NR | NR | NR | Longitudinal | 8.0 | 8 |
| LDA | |||||||||||
| Authors | Country/ region |
Year of publication | Patients | Ethnicity, % | Cross-sectional or longitudinal | Follow-up years | NOS | ||||
| Caucasian | Asian | Mestizo/ Hispanic |
African descent | Others | |||||||
| Polachek et al7* | Canada | 2017 | 620 | NR | NR | NR | NR | NR | Longitudinal | 4.0 | 7 |
| Ugarte-Gil et al14* | Latin America | 2017 | 1350 | 41.8 | NR | 42.7 | 11.7 | 3.8 | Longitudinal | 2.4 | 8 |
| Tsang-A-Sjoe et al15* | The Netherlands | 2017 | 183 | 68.3 | NR | NR | NR | 31.7 | Longitudinal | 5.0 | 8 |
| Golder et al39 | Asia Pacific | 2017 | 1422 | 8.0 | 90.0 | NR | NR | 2.0 | Cross-sectional | NR | 7 |
| Tani et al11* | Italy | 2018 | 115 | NR | NR | NR | NR | NR | Longitudinal | ≥5 | 8 |
| Zen et al17 | Italy | 2018 | 293 | 100.0 | 0.0 | 0.0 | 0.0 | 0.0 | Longitudinal | 7.0 | 9 |
| Petri et al18* | USA | 2018 | 1356 | 55.0 | NR | NR | 38.0 | 7.0 | Longitudinal | NR | 7 |
| Poomsalood et al24* | Thailand | 2019 | 237 | NR | NR | NR | NR | NR | Cross-sectional | NR | 7 |
| Alarcon et al20 * | USA | 2019 | 558 | 28.0 | 0.0 | 35.0 | 37.0 | 0.0 | Longitudinal | NR | 8 |
| Ugarte.Gil et al25* | USA | 2019 | 483 | NR | NR | NR | NR | NR | Longitudinal | NR | 7 |
| Mathian et al26* | France | 2019 | 407 | NR | NR | NR | NR | NR | Longitudinal | 1.0 | 7 |
| Golder et al38 | Asia Pacific | 2019 | 1707 | 10.1 | 87.7 | NR | NR | 2.2 | Longitudinal | 2.2 | 6 |
| Reategui-Sokolova et al28* | Peru | 2019 | 315 | NR | NR | NR | NR | NR | Longitudinal | NR | 9 |
| Ugarte-Gil et al33* | Peru | 2020 | 243 | NR | NR | NR | NR | NR | Longitudinal | 2.2 | 7 |
| Sharma et al40 | Norway | 2020 | 206 | 100.0 | 0.0 | 0.0 | 0.0 | 0.0 | Longitudinal | 10.0 | 6 |
| Yeo et al41 | Australia | 2020 | 200 | 52.0 | 39.5 | NR | NR | 8.5 | Longitudinal | 2.1 | 9 |
| Floris et al34* | Italy | 2020 | 116 | 100.0 | 0.0 | 0.0 | 0.0 | 0.0 | Longitudinal | 1.5 | 8 |
| Louthrenoo et al42 | Thailand | 2020 | 337 | NR | NR | NR | NR | NR | Longitudinal | 3.2 | 8 |
| Kang et al43 | Korea | 2021 | 299 | NR | NR | NR | NR | NR | Longitudinal | 4.0 | 8 |
*These articles were included for remission and LDA.
LDA, low disease activity; NOS, Newcastle-Ottawa Scale; NR, not reported.;
Remission and LDA rates
The rates of remission and LDA varied depending on both the definition used and the population studied. Remission was more frequent in European populations being as high as 88.1% in one study, but it was as low as 3.5% when the definition excluded patients under treatment and a duration of the remission of at least 7 years. LDA was also more frequent in European populations; however, the rate depended on the definition used; as expected, the less stringent the definition, the more frequently this outcome was achieved. These data are depicted in online supplemental table 2.
lupus-2021-000542supp002.pdf (183.6KB, pdf)
Mortality
Six studies including 3933 patients evaluated mortality as an outcome, two evaluated the impact of remission and LDA on mortality, two only LDA, one only remission and one compared remission and LDA. Among the four studies reporting the impact of LDA on mortality, two of them reported a reduction on mortality (HR 0.3% and 1.4% in those in LDA and 6.9% in those active) and two did not, although the trend was similar (HR 0.30 and 0.81, p: not significant). Among the three studies evaluating the impact of remission (compared with those not on remission) on mortality; two of them reported a reduction on mortality (HR 0.08% and 5% in those in remission and 17.7% in those not in remission), whereas the other did not (HR 0.56, p value not significant). In another report, remission was not statistically different from LDA in terms of the mortality rate. These data are depicted in table 2.
Table 2.
Impact of remission and LDA on mortality*
| Authors | Patients | Follow-up years | Remission | Impact | ||||||
| Disease activity index | Immunological activity | PGA | PDN daily dose | IS use | AM use | Minimal duration | ||||
| Drenkard et al4 | 667 | 11.6 | Lack of clinical disease activity (no formal index was used) | Allowed | NR | Not allowed | Not allowed | Not allowed | 1 year | HR 0.08, p<0.001 |
| Medina-Quiñones et al5 | 532 | 12 | BILAG C, D, E | Not allowed | NR | 0 | Not allowed | Allowed | 3 years | 5% vs 17.7% (not on remission), p<0.001 |
| Polachek et al7 | 620 | 4 | SLEDAI=0 | Not allowed | NR | Not allowed | Not allowed | Allowed | 1 year | Remission vs LDA: 0.5% vs 2.5%, p=0.15 year 2 |
| Ugarte-Gil et al14 | 1350 | 2.4 | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | Mortality (reference active) HR 0.56, p=0.2623 |
| Authors | Patients | Follow-up years | LDA | Impact | |||||||
| Disease activity index | Exclusion of new activity | Major organ exclusion | PGA | Prednisone daily dose | IS drug use | Antimalarial use | Minimal duration | ||||
| Polachek et al7 | 620 | 4 | SLEDAI<=2 | Yes | No | NR | Not allowed | Not allowed | Allowed | 1 | Remission+LDA vs active 1.4% vs 6.9, p=0.02 (year 2), 3.6% vs 13.3%, p=0.004 |
| Ugarte-Gil et al14 | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission. Mortality (reference active) HR 0.81, p=0.6476 |
| Alarcon et al20,20 | 558 | NR | SLAM<=3 | No | No | NR | <=7.5 | Not allowed | Allowed | NR | Duration on LDAS RR 0.303, p=0.1360 |
| Sharma et al40 | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 50% | HR 0.31, p<0.01 |
| Sharma et al40 | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 30% | HR 0.36, p<0.05 |
| Sharma et al40 | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 70% | HR 0.25, p<0.01 |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; IS, immunosuppressive drug; LDA, low disease activity; LDAS, low disease activity status; NR, not reported; PDN, prednisone; PGA, Physician Global Assessment; RR, risk ratio; SLAM, Systemic Lupus Activity Measure; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index.
Damage accrual
Sixteen studies including 8288 patients evaluated damage accrual. In the majority of studies, both remission and LDA prevented damage accrual when compared with patients who did not attain these states (risk measures between 0.04 and 0.95 for remission and between 0.07 and 0.90 for LDA, depending on the definition). In most of the studies, LDA also included those patients who were on remission; however, depending on the definition used, there could be a difference between those in remission and those in LDA, being better to be on remission. These data are depicted in tables 3 and 4.
Table 3.
Impact of remission on damage
| Authors | Patients | Follow-up years | Remission | Impact | ||||||
| Disease activity index | Immunological activity | PGA | PDN daily dose | IS use | AM use | Minimal duration | ||||
| Polachek et al7 | 620 | 4 | SLEDAI=0 | Not allowed | NR | Not allowed | Not allowed | Allowed | 1 year | ∆SDI: 0.1 vs 0.2 (remission vs LDA only) at year 2; p=0.18 |
| Zen et al16* | 293 | 7 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 1 year | 1 year (reference <1 year) OR 0.947, p=0.946 |
| Zen et al16* | 293 | 7 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 2 years | 2 years (reference <1 year) OR 0.228, p=0.028 |
| Zen et al16* | 293 | 7 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 3 years | 3 years (reference <1 year) OR 0.116, p=0.001 |
| Zen et al16* | 293 | 7 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 4 years | 4 years (reference <1 year) OR 0.118, p=0.005 |
| Zen et al16* | 293 | 7 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 5 years | ≥5 years (reference <1 year) OR 0.044, p<0.001 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | New damage (reference active) HR 0.60, p=0.0042 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | New damage non-GC (reference active) HR 0.51, p=0.0006 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | New severe damage non-GC (reference active) HR 0.31, p=0.0101 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | New damage GC (reference active) HR 0.99, p=0.9697 |
| Tsang-A-Sjoe et al15 | 183 | 5 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 5 years | Prolonged remission (5 years) OR=0.20, p=0.001 |
| Mok et al3 | 769 | NR | C-SLEDAI=0 | Allowed | <0.5 | ≤5 | Allowed | Allowed | 5 years | No remission or remission <5 years OR damage 2.42, p<0.001 |
| Petri et al18* | 1356 | NR | C-SLEDAI=0 | Allowed | <0.5 | ≤5 | Allowed | Allowed | NR | Less than 25% RR 0.54, p<0.0001, 25%–50% on remission RR 0.47, p<0.0001, 50%–75% RR 0.43, p<0.0001, >=75%, RR 0.45, p=0.0019 (reference not remission) |
| Petri et al18* | 1356 | NR | C-SLEDAI=0 | Allowed | <0.5 | 0 | Allowed | Allowed | NR | Less than 25% RR 0.60, p<0.0001, 25%–50% on remission RR 0.66, p=0.023, 50%–75% RR 0.63, p=0.035, >=75%, RR=0.58, p=0.12 (reference not remission) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | 0 | Allowed | Allowed | NR | HR 0.64, p=0.020 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | 0 | Not allowed | Allowed | NR | HR 0.60, p=0.022 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | NR | HR 0.49, p<0.0001 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Not allowed | Allowed | NR | HR 0.58, p=0.0005 (>=50% vs <50%) |
| Golder et al27,27* | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | 0 | Allowed | Allowed | NR | HR 0.62, p=0.076 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | 0 | Not allowed | Allowed | NR | HR 0.59, p=0.083 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | <=5 | Allowed | Allowed | NR | HR 0.63, p=0.0083 (>=50% vs <50%) |
| Golder et al27* | 1707 | 2,2 | C-SLEDAI=0 | Not allowed | <0.5 | <=5 | Not allowed | Allowed | NR | HR 0.65, p=0.043 (>=50% vs <50%) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | Any | <=5 | Allowed | Allowed | 1 year | OR 0.952 (p=0.828) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | Any | <=5 | Allowed | Allowed | 2 years | OR 0.858 (p=0.471) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | Any | <=5 | Allowed | Allowed | 3 years | OR 0.912 (p=0.668) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | Any | <=5 | Allowed | Allowed | 4 years | OR 0.391 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | Any | <=5 | Allowed | Allowed | 5 years | OR 0.620 (p=0.010) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | Allowed | Allowed | Allowed | 1 year | OR 0.808 (p=0.185) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | Allowed | Allowed | Allowed | 2 years | OR 0.560 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | Allowed | Allowed | Allowed | 3 years | OR 0.427 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | Allowed | Allowed | Allowed | 4 years | OR 0.226 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | Allowed | Allowed | Allowed | 5 years | OR 0.377 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | Allowed | Allowed | Allowed | 1 year | OR 0.766 (p=0.119) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | Allowed | Allowed | Allowed | 2 years | OR 0.454 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | Allowed | Allowed | Allowed | 3 years | OR 0.512 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | Allowed | Allowed | Allowed | 4 years | OR 0.173 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | Allowed | Allowed | Allow ed |
5 years | OR 0.382 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | <=5 | Allowed | Allowed | 1 year | OR 0.764 (p=0.101) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | <=5 | Allowed | Allowed | 2 years | OR 0.495 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | <=5 | Allowed | Allowed | 3 years | OR 0.430 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | <=5 | Allowed | Allowed | 4 years | OR 0.294 (p<0.001) |
| Saccon et al35* | 646 | 5 years | Any | Allowed | <0.5 | <=5 | Allowed | Allowed | 5 years | OR 0.363 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | 1 year | OR 0.857 (p=0.326) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | 2 years | OR 0.514 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | 3 years | OR 0.459 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | 4 years | OR 0.243 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | 5 years | OR 0.397 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | <=5 | Allowed | Allowed | 1 year | OR 0.888 (p<0.471) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | <=5 | Allowed | Allowed | 2 years | OR 0.497 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | <=5 | Allowed | Allowed | 3 years | OR 0.548 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | <=5 | Allowed | Allowed | 4 years | OR 0.251 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | Any | <=5 | Allowed | Allowed | 5 years | OR 0.411 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | 1 year | OR 0.800 (p=0.167) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | 2 years | OR 0.479 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | 3 years | OR 0.438 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | 4 years | OR 0.296 (p<0.001) |
| Saccon et al35* | 646 | 5 years | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | 5 years | OR 0.384 (p<0.001) |
| Jakez-Ocampo et al36 | NR (case control) | NR | C-SLEDAI=0 | Allowed | NR | Not allowed | Not allowed | Not allowed | 10 years | Remission group: 0.68±0.67, control group 1.05±0.87 (p=0.016). No difference between those on remission with or without serological activity. |
| Floris et al34 | 116 | 1.5 | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | NR | OR=0.07, p=0.015 |
| Nikfar et al37 | 193 | 8 | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Not allowed | Allowed | 5 years | OR=0.62, p=0.047 |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; C-SLEDAI, Clinical Systemic Lupus Erythematosus Disease Activity Index; IS, immunosuppressive drug; LDAS, low disease activity status; LLDAS, lupus low disease activity state; NR, not reported; PDN, prednisone; PGA, Physician Global Assessment.;
Table 4.
Impact of LDA on damage
| Authors | Patients | Follow-up years | LDA | Impact | |||||||
| Disease activity index | Exclusion of New activity | Major Organ Exclusion | PGA | PDN daily dose | IS use | AM use | Minimal duration | ||||
| Polachek et al7 | 620 | 4 | SLEDAI<=2 | No | No | NR | Not allowed | Not allowed | Allowed | 1 year | Remission+LDA vs active: 0.15 vs 0.52, p<0.001 (year 2); 0.25 vs 0.88, p<0.001 (year 4) |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission new damage (reference active) HR 0.66, p=0.0158 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission new severe damage (reference active) HR 0.54, p=0.0614 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission new severe damage (reference active) HR 0.54, p=0.0614 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission new damage non-GC (reference active) HR 0.62, p=0.0067 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission new severe damage non-GC (reference active) HR 0.35, p=0.0206 |
| Ugarte-Gil et al14* | 1350 | 2.4 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | at least once | Excluding remission new damage GC (reference active) HR 1.34, p=0.3333 |
| Tsang-A-Sjoe et al15 | 183 | 5 | SLEDAI<=4 | No | Yes | <=2/10 | <=7.5 | Allowed | Allowed | >50% on LLDAS: OR=0.52, p=0.046 | |
| Tani et al11* | 115 | At least 5 | SLEDAI<=4 | No | Yes | <=1 | <=7.5 | Allowed | Allowed | 100% of the follow-up | ∆SDI: 0.11 vs 0.63; p<0.001 |
| Tani et al11* | 115 | at least 5 | SLEDAI<=4 | No | Yes | <=1 | <=7.5 | Allowed | Allowed | 50% | ∆SDI: 0.25 vs 0.78; p=0.004 |
| Zen et al17* | 293 | 7 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 1 year | 1 year (reference <1 year) OR 0.899, p=0.877 |
| Zen et al17,17* | 293 | 7 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 2 years | 2 years (reference <1 year) OR 0.279, p=0.036 |
| Zen et al17* | 293 | 7 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 3 years | 3 years (reference <1 year) OR 0.252, p=0.025 |
| Zen et al17* | 293 | 7 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 4 years | 4 years (reference <1 year) OR 0.122, p=0.001 |
| Zen et al17,17* | 293 | 7 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 5 years | ≥5 years (reference <1 year) OR 0.071, p<0.001 |
| Petri et al18 | 1356 | NR | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | Less than 25% RR 0.80, p=0.12, 25%–50% on remission RR 0.63, p=0.0012, 50%–75% RR 0.47, p<0.0001, >=75%, RR 0.39, p<0.0001 (reference not LLDAS) |
| Alarcon et al20 | 558 | NR | SLAM<=3 | No | No | NR | <=7.5 | Not allowed | Allowed | NR | Duration on LDAS RR: 0.1773, p<0.0001 LDAS prevented from GC-related and non-GC-related damage |
| Golder et al38* | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | HR 0.54, p<0.0001 |
| Golder et al38* | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | HR 0.59, p<0.0001 |
| Golder et al38* | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | RR 0.14 p<0.0001 |
| Sharma et al40* | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 50% | HR 0.37, p<0.01 (SDI>=3) |
| Sharma et al40* | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 30% | HR 0.57, p=0.08 (SDI>=3) |
| Sharma et al40* | 206 | 10 | SLEDAI<=4 | Yes | Yes | NR | <=7.5 | Allowed | Allowed | 70% | HR 0.38, p<0.01 (SDI>=3) |
| Floris et al34 | 116 | 1.5 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | LLDAS (not in remission) OR 0.25, p=0.049 |
| Kang et al43* | 299 | 4 | C-SLEDAI<=1 | NR | NR | NR | 5 | Allowed | Allowed | NR | B=−0.033, p=0.368 |
| Kang et al43* | 299 | 4 | C-SLEDAI<=2 | NR | NR | NR | 0 | Not allowed | allowed | NR | B=−0.093, p=0.390 |
| Kang et al43* | 299 | 4 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | B=−0.064, p=0.050 |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; C-SLEDAI, Clinical Systemic Lupus Erythematosus Disease Activity Index; IS, immunosuppressive drug; LDA, low disease activity; LDAS, low disease activity status; LLDAS, lupus low disease activity state;NR, not reported; PDN, prednisone; PGA, Physician Global Assessment; SLAM, Systemic Lupus Activity Measure.
Flare
Five studies including 3033 patients evaluated longitudinally the occurrence of flares after achieving these states. Remission and LDA reduced the probability of flares in all studies included, regardless of the definition used (HR between 0.26 and 0.70 for remission and between 0.41 and 0.74 for LDA); however, the longer the duration of the state, the lower the risk. Only one study compare remission versus LDA and it did not find a statistically significant difference. These data are depicted in table 5.
Table 5.
Impact of remission and LDA on flare*
| Authors |
Country/ region |
Year of publication | Patients | Follow-up years | Remission | Impact | ||||||
| Disease activity index | Immunological activity | PGA | PDN daily dose | IS use | AM use | Minimal duration | ||||||
| Polachek et al7 | Canada | 2017 | 620 | 4 | SLEDAI=0 | Not allowed | NR | Not allowed | Not allowed | Allowed | 1 year | Remission vs LDA 5% vs 4.4%, p=0.8, year 2 |
| Mathian et al26 | France | 2019 | 407 | 1 | C-SLEDAI=0 | Allowed | NR | <=5 | Allowed | Allowed | NR | For each year remission, HR 0.7, p=0.02 |
| Golder et al27 38 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | 0 | Allowed | Allowed | NR | HR 0.39, p<0.0001 (>=50% vs<50%) |
| Golder et al27,27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | 0 | Not allowed | Allowed | NR | HR 0.36; p<0.0001 (>=50% vs<50%) |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Allowed | Allowed | NR | HR 0.54 p<0.0001 (>=50% vs<50%) |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Allowed | <0.5 | <=5 | Not allowed | Allowed | NR | HR 0.52 p<0.0001 (>=50% vs <50%) |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | 0 | Allowed | Allowed | NR | HR 0.28 p<0.0001 (>=50% vs <50%) |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | 0 | Not allowed | Allowed | NR | HR 0.26, p<0.0001(>=50% vs <50%) |
| Golder et al27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | <=5 | Allowed | Allowed | NR | HR 0.43, p<0.0001 (>=50% vs <50%) |
| Golder et al27,27 | Asia Pacific | 2019 | 1707 | 2.2 | C-SLEDAI=0 | Not allowed | <0.5 | <=5 | Not allowed | Allowed | NR | HR 0.41, p<0.0001 (>=50% vs <50%) |
| Authors |
Country/region |
Year of publication | Patients | Follow-up years | LDA | Impact |
|||||||
| Disease activity index | Exclusion of new activity | Major organ exclusion | PGA | PDN daily dose | IS use | AM use | Minimal duration | ||||||
| Polachek et al7 | Canada | 2017 | 620 | 4 | SLEDAI<=2 | Yes | No | NR | Not allowed | Not allowed | Allowed | 1 | Remission+LDA vs active: 4.8 vs 14.6, p<0.001 (year 2), 3.7 vs 14.8, p=0.007 (year 4) |
| Golder et al38 | Asia Pacific | 2019 | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | Any visit | HR 0.65 (any flare), p<0.0001 |
| Golder et al38 | Asia Pacific | 2019 | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | Any visit | HR 0.74 (mild-moderate flare), p<0.0001 |
| Golder et al38 | Asia Pacific | 2019 | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | Any visit | HR 0.59 (severe flare), p<0.0001 |
| Golder et al38 | Asia Pacific | 2019 | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | 50% of the follow-up | HR 0.41, p<0.0001 (any flare) |
| Kang et al43 | Korea | 2021 | 299 | 4 | C-SLEDAI<=1 | NR | NR | NR | 5 | Allowed | Allowed | NR | B=0.419, p=0.109 |
| Kang et al43 | Korea | 2021 | 299 | 4 | C-SLEDAI<=2 | NR | NR | NR | 0 | Not allowed | Allowed | NR | B=0.960, p=0.969 |
| Kang et al43 | Korea | 2021 | 299 | 4 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | B=0.090, p<0.001 |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; C-SLEDAI, Clinical Systemic Lupus Erythematosus Disease Activity Index; IS, immunosuppressive drug; LDA, low disease activity; NR, not reported; PDN, prednisone; PGA, Physician Global Assessment.
Health-related quality of life (HRQoL)
Ten manuscripts including 4480 patients evaluated HRQoL. Remission and LDA were associated with a better HRQoL being this impact more consistent on the physical components of HRQoL, and less so on the mental components of HRQoL. These data are depicted in tables 6 and 7.
Table 6.
Impact of remission on HRQoL*
| Authors | Country/ region |
Year of publication | Patients | Follow-up years | Remission | Domains positively associated | Domains not associated | ||||||
| Disease activity index | Immunological activity | PGA | PDN daily dose | IS use | AM use | Minimal duration | |||||||
| Mok et al3 | Hong Kong | 2017 | 769 | Cross-sectional | C-SLEDAI=0 | Allowed | <0.5 | ≤5 | Allowed | Allowed | 5 years | SF-36: >5 years vs <5 years and >5 years vs no remission role physical, vitality, social functioning, PCS >5 years vs no remission bodily pain, general health, role emotional, mental health, MCS LupusPRO: >5 years vs <5 years and >5 years vs no remission role physical, vitality, social functioning, PCS >5 years vs no remission bodily pain, general health, role emotional, mental health, MCS, remission >5 vs no remission desire/goal |
SF-36: >5 years vs <5 years and >5 years vs no remission symptoms, medications, procreation, physical health, emotional, HRQoL total. >5 years vs <5 years remission pain, image LupusPRO: >5 years vs <5 years and >5 years vs no remission cognition, social support, coping, satisfaction with medical care, non-HRQoL total. >5 years vs <5 years remission pain, image, remission >5 vs remission <years desire/goal |
| Margiotta et al29 | Italy | 2019 | 136 | Cross-sectional | C-SLEDAI=0 | Allowed | NR | <=5 | Allowed | Allowed | 5 years | SF-36: >5 years vs <5 years and no remission physical functioning, role physical, bodily pain, general health and social functioning | SF-36: >5 years vs <5 years and no remission vitality, role emotional, mental health |
| Goswami et al31 | India | 2019 | 126 | Cross-sectional | C-SLEDAI=0 | Allowed | <0.5 | Allowed | Allowed | Allowed | NR | SF-36: Complete remission was associated with a better PCS than clinical remission | |
| Poomsalood et al24 | Thailand | 2019 | 237 | Cross-sectional | C-SLEDAI=0 | Allowed | NR | <=5 | Allowed | Allowed | 1 year | SLEQOL: Remission vs not on remission activities, symptom, treatment, mood, self-image, total QoL univariable | SLEQOL: Remission vs not on remission. Physical, remission vs LDA NS all univariable |
| Tsang-A-Sjoe et al15 | The Netherlands | 2019 | 154 | 2 | C-SLEDAI=0 | Allowed | <=2/10 | <=5 | Allowed | Allowed | NR | SF-36: Remission on and off therapy and PCS | SF-36: Remission on and off therapy and MCS |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; C-SLEDAI, Clinical Systemic Lupus Erythematosus Disease Activity Index; HRQoL, health-related quality of life; IS, immunosuppressive drug; LDAS, low disease activity; LLDAS, lupus low disease activity state; MCS, Mental Component Summary; NR, not reported; PCS, Physical Component Summary; PDN, prednisone; PGA, Physician Global Assessment; SF-36, 36-Item Short Form Health Survey; SLEQOL, Systemic Lupus Erythematosus Quality of Life.
Table 7.
Impact of remission and LDA on HRQoL*
| Authors | Country/region | Year of publication | Patients | Follow-up years | LDA | Domains positively associated | Domains not associated | |||||||
| Disease activity index | Exclusion of New activity | Major Organ Exclusion | PGA | PDN daily dose | IS use | AM use | Minimal duration | |||||||
| Golder et al39 | APLC | 2017 | 1707 | 2.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | SF-36: PCS and MCS Multivariable role physical, bodily pain, general health, social functioning, role emotional, vitality, mental health univariable | SF-36: Physical function univariable |
| Poomsalood et al24 | Thailand | 2019 | 237 | Cross-sectional | C-SLEDAI=0 | Allowed | NR | <=5 | Allowed | Allowed | 1 year | 1 year | SLEQOL: Physical, activities, symptom, treatment, mood, self-image, total QoL univariable, LLDAS vs no LDA b: 20.02, p=0.003 Multivariable |
|
| Ugarte-Gil et al25 | USA | 2019 | 472 | NR | SLAM<=3 | No | No | NR | <=7.5 | Not allowed | Allowed | NR | SF-36: PCS, MCS, physical functioning, role physical, bodily pain, general health, social functioning, role emotional, vitality, mental health | |
| Ugarte-Gil et al33 | Peru | 2020 | 243 | 2 | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | at least once | LupusQoL: Physical health, pain, planning, burden to others, emotional health, fatigue | LupusQoL: Intimate relationship, body image |
| Louthrenoo et al42 | Thailand | 2020 | 337 | 3.2 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | Global and all domains of SLEQOL, PCS and MCS SF-36 | |
| Kang et al43 | Korea | 2021 | 299 | 4 | C-SLEDAI<=1 | NR | NR | NR | 5 | Allowed | Allowed | NR | PCS and MCS SF-36 | |
| Kang et al43 | Korea | 2021 | 299 | 4 | C-SLEDAI<=2 | NR | NR | NR | 0 | Not allowed | Allowed | NR | PCS and MCS SF-36 | |
| Kang et al43 | Korea | 2021 | 299 | 4 | SLEDAI<=4 | Yes | Yes | <=1 | <=7.5 | Allowed | Allowed | NR | PCS and MCS SF-36 | |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; HRQoL, health-related quality of life; IS, immunosuppressive drug; LDAS, low disease activity; LLDAS, lupus low disease activity state;MCS, Mental Component Summary; NR, not reported; PCS, Physical Component Summary; PDN, prednisone; PGA, Physician Global Assessment; SF-36, 36-Item Short Form Health Survey; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index.
Other outcomes
Three manuscripts including 802 patients evaluated other outcomes. Being on remission and LDA was associated with a lower hospitalisation rate; LDA was associated with lower medical cost and prolonged remission with lower cardiovascular risk. These data are depicted in table 8.
Table 8.
Impact of remission and LDA on other outcomes*
| Authors | Country/region | Year of publication | Patients | Follow-up years | Remission | Impact | |||||||
| Disease activity index | Immunological activity | PGA | PDN daily dose | IS use | AM use | Minimal duration | |||||||
| Reátegui-Sokolov et al28 | Peru | 2019 | 308 | NR | SLEDAI=0 | Not allowed | NR | ≤5 | Allowed | Allowed | At least once | Outcome: Hospitalisation HR 0.45, p=0.001 |
|
| Fasano et al30 | Italy | 2019 | 294 | 9 | C-SLEDAI=0 | Allowed | NR | ≤5 | Allowed | Allowed | 5 years | Cardiovascular event: HR 0.18, p=0.023 | |
|
Authors
|
Country/region
|
Year of publication | Patients | Follow-up years | LDA |
Impact
|
|||||||
| Disease activity index | Exclusion of new activity | Major organ exclusion | PGA | Prednisone daily dose | IS drug use | Antimalarial use | Minimal duration | ||||||
| Reátegui-Sokolov et al28 | Peru | 2019 | 308 | NR | SLEDAI<=4 | No | No | NR | <=7.5 | Allowed | Allowed | At least once | Excluding remission. Outcome: Hospitalisation HR 0.50, p=0.001 |
| Yeo et al41 | Australia | 2020 | 200 | 2.1 | SLEDAI<=4 | Yes | Yes | <1 | <=7.5 | Allowed | Allowed | 50% | Outcome: Direct health cost. Ratio of geometric means 0.53, p<0.001 |
*If an article included more than one definition, a row per definition is included.
AM, antimalarials; IS, immunosuppressive drug; LDA, low disease activity; NR, not reported; PDN, prednisone; PGA, Physician Global Assessment; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index.
Discussion
Our systematic literature search showed that being in remission or LDA, regardless of the definitions used, was associated with better outcomes in patients with SLE, the most commonly reported outcomes being lower damage accrual, fewer flares and a better HRQoL. The association with a lower mortality rate was less consistently reported.
In terms of mortality, LDA was associated with lower mortality in two studies, one from the Toronto Lupus Cohort,7 which had a more stringent definition of LDA (SLEDAI ≤2 without treatment) and the other from Norway40 (which allowed a SLEDAI ≤4, excluding new activity and major organ activity, and allowing prednisone ≤7.5 mg/day and immunosuppressive drugs on maintenance dose); similarly, remission was associated with lower mortality in a study from Mexico4 and in one from the UK.5 However, in the GLADEL14 and the LUMINA20 cohorts, the association between remission and LDA and mortality was not statistically significant, although the trend was in the protective direction. This lack of association between achieving these outcomes and mortality could be due to a relatively short follow-up time in these cohorts. The Toronto Lupus Cohort compared remission and LDA and found no statistically significant difference between the two states in terms of mortality.7
Remission was associated with a lower risk of damage accrual in several cohorts from Asia, Europe, North America (USA-Canada) and Latin America3 7 14–16 18 27 34–37; however, the minimum time on remission needed to prevent damage accrual has yet to be determined. According to the Padua cohort, being in remission for less than 1 year was not protective against damage,16 whereas according to the Hopkins cohort, being in remission even less than 25% of the follow-up time prevented the accrual of damage.18,18 According to the GLADEL cohort, being in remission prevented not only the accrual of any damage but also the accrual of severe damage (an increase in the SDI of at least 3 points) and from non-glucocorticoid (GC)-related damage and severe damage.14 Additionally, the longer the duration of remission, the lower the probability of damage accrual.16 Similarly, LDA (regardless of how it was defined) has been associated with less damage accrual7 11 14 15 17 18 20 34 38 40 43; however, in the Padua cohort, being on LDA for less than 1 year did not prevent the accrual of damage.17 In the Hopkins cohort, being in LDA for less than 25% of the follow-up did not prevent the accrual of damage.18 Being in LDA prevented also severe damage accrual, non-GC and GC-related damage14; furthermore, the longer the duration of LDAS, the less the damage accrued.20 In the Toronto cohort, being on remission and LDA (SLEDAI ≤2 without treatment) did not differ in terms of the risk of damage accrual7; however, in the Padua cohort, being in remission was associated with a lower risk of damage that being on LLDAS (which allowed a SLEDAI ≤4, excluding new activity and major organ activity, and allowing prednisone ≤7.5 mg/day and immunosuppressive drugs on maintenance dose).17 Probably, the difference in the definitions used in both cohorts could explain these results. Consistent with these results, prolonged remission was associated with a lower probability of cardiovascular events.30
Being in remission or LDA reduced the risk of any flares, being those mild-moderate or severe.7 26 27 38 43 Only in the Toronto cohort remission and LDA (SLEDAI ≤2 without treatment) were compared, but no differences were found.7
Patient perspective is important in defining the optimal treatment target. In previous reports, the association between disease activity and HRQoL has been low or absent.44 Notably, remission and LDA have been found to be associated with a better HRQoL in cross-sectional and longitudinal studies regardless of whether generic or lupus-specific measures were used.3 24 25 29 31–33 39 42 43 These associations were more consistently reported in the physical than in the mental domains, probably because the mental domains are affected also by comorbid conditions such as depression, fibromyalgia and anxiety. It has been suggested that specific measures may ascertain better QoL dimensions specific to patients with SLE.44
Finally, remission and LDA could reduce hospitalisation rate; this has been reported in the Peruvian Almenara Lupus cohort28; LDA could also reduce annual medical cost as reported in a study from an Australian cohort.41 It is important to point out that this information needs to be confirmed in other populations.
Taking together, being on remission or on LDA, regardless of the definitions used, is associated with better outcomes, including mortality, damage, flares, HRQoL, hospitalisation and cost. It is important, however, to point out that a uniform definition of both states is desirable in order to make these results comparable. The current definition of remission, as proposed by the DORIS group, takes into account two physician disease activity measures (clinical SLEDAI=0 and PGA<0.5) as well as treatment (prednisone daily dose not higher than 5 mg/day and/or immunosuppressive drugs on maintenance dose),1 and, even not all the studies used this definition, the large majority used 2015 or 2021 DORIS definitions1 2 or a very similar definition. LDA should be different enough from remission in order to define a group of patients with a better prognosis than those with active disease, but, not as good as the prognosis of those on remission; in this context, the definition proposed by APLC is a good option as it allows a higher level of disease activity (SLEDAI ≤4 and PGA≤1), excludes activity in major organs and new activity, and also allows a higher dose of prednisone (7.5 mg/day) and keeping the immunosuppressive drugs on maintenance dose.6 Additionally, in the KORNET cohort from Korea, LLDAS, but not LDA (SLEDAI ≤2 without treatment) or MDA (minimal disease activity) were predictive of good outcomes.43 However, more information is needed in order to determine if being on remission is better than being on LDA. About the duration of these states, it seems that achieving these states even for a short period of time is associated with better outcomes, but the longer the patient remains on these states, the better the outcomes will be.
These analyses have some limitations; first, as the studies included used different definitions for remission and LDA, a meta-analysis could not be performed. Second, the duration of follow-up in some studies reviewed was not long enough for the assessment of mortality. Third, there are only a few studies for some of the outcomes assessed; this precludes us from making stronger conclusions.
The main strength of this report is the inclusion of several different populations from across the world and several outcomes, allowing us to evaluate the real impact of remission and LDA in the prognosis of patients with SLE.
In conclusion, being in remission or LDA (regardless of the definition) is associated with improved outcomes in patients with SLE. These results reinforce the relevance of these outcomes for the management of patients with SLE.
In order to facilitate the implementation of a T2T strategy in SLE, it is important to have an uniform definition of remission1 and LDA.
Footnotes
Twitter: @mugartegil, @gponsestel
Contributors: All authors were involved in drafting or revising this article critically for important intellectual content, and all authors approved the final version to be published. MFU-G has full access to all of the data from the study and takes responsibility for their integrity and the accuracy of the analyses performed.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: MFU-G: Grant/research support from Jannsen, Pfizer, not related to this article. CM-P declared no conflict of interest. CR-S: Grant/research support from Jannsen, not related to this article. GP-E: Grant/research support from JANSSEN and GSK; consultant of JANNSEN, GSK and SANOFI; speakers bureau: Pfizer, JANNSEN and GSK, not related to this article. RFvV: Grant/research support from AbbVie, Arthrogen, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Pfizer and UCB; consultant of AbbVie, AstraZeneca, Biotest, Bristol-Myers Squibb, Celgene, GSK, Janssen, Lilly, Medac, Merck, Novartis, Pfizer, Roche and UCB, not related to this article. GB: Grant/research support from GSK, Pfizer; consultant of Novartis, SOBI, not related to this article. GA declared no conflict of interest. BAP-E: Grant/research support from GSK, Jannsen; consultant of GSK, Janssen; speakers bureau: GSK, Janssen, not related to this article.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Ethics statements
Patient consent for publication
Not required.
References
- 1.van Vollenhoven R, Bertsias G, Doria A, et al. OP0296 the 2021 Doris definition of remission in SLE – final recommendations from an International task force. Ann Rheum Dis 2021;80:181.1–2. 10.1136/annrheumdis-2021-eular.1192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.van Vollenhoven R, Voskuyl A, Bertsias G, et al. A framework for remission in SLE: consensus findings from a large international Task force on definitions of remission in SLE (DORIS). Ann Rheum Dis 2017;76:554–61. 10.1136/annrheumdis-2016-209519 [DOI] [PubMed] [Google Scholar]
- 3.Mok CC, Ho LY, Tse SM, et al. Prevalence of remission and its effect on damage and quality of life in Chinese patients with systemic lupus erythematosus. Ann Rheum Dis 2017;76:1420–5. 10.1136/annrheumdis-2016-210382 [DOI] [PubMed] [Google Scholar]
- 4.Drenkard C, Villa AR, Garcia-Padilla C, et al. Remission of systematic lupus erythematosus. Medicine 1996;75:88–98. 10.1097/00005792-199603000-00005 [DOI] [PubMed] [Google Scholar]
- 5.Medina-Quiñones CV, Ramos-Merino L, Ruiz-Sada P, et al. Analysis of complete remission in systemic lupus erythematosus patients over a 32-year period. Arthritis Care Res 2016;68:981–7. 10.1002/acr.22774 [DOI] [PubMed] [Google Scholar]
- 6.Franklyn K, Lau CS, Navarra SV, et al. Definition and initial validation of a lupus low disease activity state (LLDAS). Ann Rheum Dis 2016;75:1615–21. 10.1136/annrheumdis-2015-207726 [DOI] [PubMed] [Google Scholar]
- 7.Polachek A, Gladman DD, Su J, et al. Defining low disease activity in systemic lupus erythematosus. Arthritis Care Res 2017;69:997–1003. 10.1002/acr.23109 [DOI] [PubMed] [Google Scholar]
- 8.Wilhelm TR, Magder LS, Petri M. Remission in systemic lupus erythematosus: durable remission is rare. Ann Rheum Dis 2017;76:547–53. 10.1136/annrheumdis-2016-209489 [DOI] [PubMed] [Google Scholar]
- 9.Babaoglu H, Li J, Goldman D, et al. Predictors of predominant lupus low disease activity state (LLDAS-50). Lupus 2019;28:1648–55. 10.1177/0961203319886028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ugarte-Gil MF, Wojdyla D, Pons-Estel GJ, et al. Predictors of remission and low disease activity state in systemic lupus erythematosus: data from a multiethnic, multinational Latin American cohort. J Rheumatol 2019;46:1299–308. 10.3899/jrheum.180433 [DOI] [PubMed] [Google Scholar]
- 11.Tani C, Vagelli R, Stagnaro C, et al. Remission and low disease activity in systemic lupus erythematosus: an achievable goal even with fewer steroids? real-life data from a monocentric cohort. Lupus Sci Med 2018;5:e000234. 10.1136/lupus-2017-000234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aringer M, Costenbader K, Brinks R. Validation of new systemic lupus erythematosus classification criteria. Arthritis Rheumatol 2018;70. [Google Scholar]
- 13.Gatto M, Zen M, Iaccarino L, et al. New therapeutic strategies in systemic lupus erythematosus management. Nat Rev Rheumatol 2019;15:30–48. 10.1038/s41584-018-0133-2 [DOI] [PubMed] [Google Scholar]
- 14.Ugarte-Gil MF, Wojdyla D, Pons-Estel GJ, et al. Remission and low disease activity status (LDAS) protect lupus patients from damage occurrence: data from a multiethnic, multinational Latin American lupus cohort (GLADEL). Ann Rheum Dis 2017;76:2071–4. 10.1136/annrheumdis-2017-211814 [DOI] [PubMed] [Google Scholar]
- 15.Tsang-A-Sjoe MWP, Bultink IEM, Heslinga M, et al. Both prolonged remission and lupus low disease activity state are associated with reduced damage accrual in systemic lupus erythematosus. Rheumatology 2017;56:121–8. 10.1093/rheumatology/kew377 [DOI] [PubMed] [Google Scholar]
- 16.Zen M, Iaccarino L, Gatto M, et al. The effect of different durations of remission on damage accrual: results from a prospective monocentric cohort of Caucasian patients. Ann Rheum Dis 2017;76:562–5. 10.1136/annrheumdis-2016-210154 [DOI] [PubMed] [Google Scholar]
- 17.Zen M, Iaccarino L, Gatto M, et al. Lupus low disease activity state is associated with a decrease in damage progression in Caucasian patients with SLE, but overlaps with remission. Ann Rheum Dis 2018;77:104–10. 10.1136/annrheumdis-2017-211613 [DOI] [PubMed] [Google Scholar]
- 18.Petri M, Magder LS. Comparison of remission and lupus low disease activity state in damage prevention in a United States systemic lupus erythematosus cohort. Arthritis Rheumatol 2018;70:1790–5. 10.1002/art.40571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Piga M, Floris A, Cappellazzo G, et al. Failure to achieve lupus low disease activity state (LLDAS) six months after diagnosis is associated with early damage accrual in Caucasian patients with systemic lupus erythematosus. Arthritis Res Ther 2017;19:247. 10.1186/s13075-017-1451-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alarcón GS, Ugarte-Gil MF, Pons-Estel G, et al. Remission and low disease activity state (LDAS) are protective of intermediate and long-term outcomes in SLE patients. results from LUMINA (LXXVIII), a multiethnic, multicenter US cohort. Lupus 2019;28:423–6. 10.1177/0961203319826693 [DOI] [PubMed] [Google Scholar]
- 21.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009;339:b2535. 10.1136/bmj.b2535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wells G, Shea B, O'connell D. The Newcastle–Ottawa scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp
- 23.Campbell M, McKenzie JE, Sowden A, et al. Synthesis without meta-analysis (swim) in systematic reviews: reporting guideline. BMJ 2020;368:l6890. 10.1136/bmj.l6890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Poomsalood N, Narongroeknawin P, Chaiamnuay S, et al. Prolonged clinical remission and low disease activity statuses are associated with better quality of life in systemic lupus erythematosus. Lupus 2019;28:1189–96. 10.1177/0961203319862614 [DOI] [PubMed] [Google Scholar]
- 25.Ugarte-Gil MF, Pons-Estel GJ, Vila LM, et al. Time in remission and low disease activity state (LDAS) are associated with a better quality of life in patients with systemic lupus erythematosus: results from LUMINA (LXXIX), a multiethnic, multicentre US cohort. RMD Open 2019;5:e000955. 10.1136/rmdopen-2019-000955 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mathian A, Mouries-Martin S, Dorgham K, et al. Ultrasensitive serum interferon-α quantification during SLE remission identifies patients at risk for relapse. Ann Rheum Dis 2019;78:1669–76. 10.1136/annrheumdis-2019-215571 [DOI] [PubMed] [Google Scholar]
- 27.Golder V, Kandane-Rathnayake R, Huq M, et al. Evaluation of remission definitions for systemic lupus erythematosus: a prospective cohort study. Lancet Rheumatol 2019;1:e103–10. 10.1016/S2665-9913(19)30048-7 [DOI] [PubMed] [Google Scholar]
- 28.Reátegui-Sokolova C, Rodríguez-Bellido Z, Gamboa-Cárdenas RV, et al. Remission and low disease activity state prevent hospitalizations in systemic lupus erythematosus patients. Lupus 2019;28:1344–9. 10.1177/0961203319876998 [DOI] [PubMed] [Google Scholar]
- 29.Margiotta DPE, Fasano S, Basta F, et al. The association between duration of remission, fatigue, depression and health-related quality of life in Italian patients with systemic lupus erythematosus. Lupus 2019;28:1705–11. 10.1177/0961203319884651 [DOI] [PubMed] [Google Scholar]
- 30.Fasano S, Margiotta DPE, Pierro L, et al. Prolonged remission is associated with a reduced risk of cardiovascular disease in patients with systemic lupus erythematosus: a GIRRCS (Gruppo Italiano di Ricerca in Reumatologia clinica E Sperimentale) study. Clin Rheumatol 2019;38:457–63. 10.1007/s10067-018-4286-9 [DOI] [PubMed] [Google Scholar]
- 31.Goswami RP, Chatterjee R, Ghosh P, et al. Quality of life among female patients with systemic lupus erythematosus in remission. Rheumatol Int 2019;39:1351–8. 10.1007/s00296-019-04329-1 [DOI] [PubMed] [Google Scholar]
- 32.Tsang-A-Sjoe MWP, Bultink IEM, Heslinga M, et al. The relationship between remission and health-related quality of life in a cohort of SLE patients. Rheumatology 2019;58:628–35. 10.1093/rheumatology/key349 [DOI] [PubMed] [Google Scholar]
- 33.Ugarte-Gil MF, Gamboa-Cárdenas RV, Reátegui-Sokolova C, et al. Better health-related quality of life in systemic lupus erythematosus predicted by low disease activity State/Remission: data from the Peruvian Almenara lupus cohort. Arthritis Care Res 2020;72:1159–62. 10.1002/acr.24009 [DOI] [PubMed] [Google Scholar]
- 34.Floris A, Piga M, Perra D, et al. Treatment target in newly diagnosed systemic lupus erythematosus: the association of lupus low disease activity state and remission with lower Accrual of early damage. Arthritis Care Res 2020;72:1794–9. 10.1002/acr.24086 [DOI] [PubMed] [Google Scholar]
- 35.Saccon F, Zen M, Gatto M, et al. Remission in systemic lupus erythematosus: testing different definitions in a large multicentre cohort. Ann Rheum Dis 2020;79:943–50. 10.1136/annrheumdis-2020-217070 [DOI] [PubMed] [Google Scholar]
- 36.Jakez-Ocampo J, Rodriguez-Armida M, Fragoso-Loyo H, et al. Clinical characteristics of systemic lupus erythematosus patients in long-term remission without treatment. Clin Rheumatol 2020;39:3365–71. 10.1007/s10067-020-05379-8 [DOI] [PubMed] [Google Scholar]
- 37.Nikfar M, Malek Mahdavi A, Khabbazi A, et al. Long-term remission in patients with systemic lupus erythematosus. Int J Clin Pract 2021;75:e13909. 10.1111/ijcp.13909 [DOI] [PubMed] [Google Scholar]
- 38.Golder V, Kandane-Rathnayake R, Huq M, et al. Lupus low disease activity state as a treatment endpoint for systemic lupus erythematosus: a prospective validation study. The Lancet Rheumatology 2019;1:e95–102. 10.1016/S2665-9913(19)30037-2 [DOI] [PubMed] [Google Scholar]
- 39.Golder V, Kandane-Rathnayake R, Hoi AY-B, et al. Association of the lupus low disease activity state (LLDAS) with health-related quality of life in a multinational prospective study. Arthritis Res Ther 2017;19:62. 10.1186/s13075-017-1256-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sharma C, Raymond W, Eilertsen G, et al. Association of achieving lupus low disease activity state fifty percent of the time with both reduced damage Accrual and mortality in patients with systemic lupus erythematosus. Arthritis Care Res 2020;72:447–51. 10.1002/acr.23867 [DOI] [PubMed] [Google Scholar]
- 41.Yeo AL, Koelmeyer R, Kandane-Rathnayake R, et al. Lupus low disease activity state and reduced direct health care costs in patients with systemic lupus erythematosus. Arthritis Care Res 2020;72:1289–95. 10.1002/acr.24023 [DOI] [PubMed] [Google Scholar]
- 42.Louthrenoo W, Kasitanon N, Morand E, et al. Comparison of performance of specific (SLEQOL) and generic (SF36) health-related quality of life questionnaires and their associations with disease status of systemic lupus erythematosus: a longitudinal study. Arthritis Res Ther 2020;22:8. 10.1186/s13075-020-2095-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kang J-H, Shin M-H, Choi S-E, et al. Comparison of three different definitions of low disease activity in patients with systemic lupus erythematosus and their prognostic utilities. Rheumatology 2021;60:762–6. 10.1093/rheumatology/keaa407 [DOI] [PubMed] [Google Scholar]
- 44.Elera-Fitzcarrald C, Fuentes A, González LA, et al. Factors affecting quality of life in patients with systemic lupus erythematosus: important considerations and potential interventions. Expert Rev Clin Immunol 2018;14:915–31. 10.1080/1744666X.2018.1529566 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
lupus-2021-000542supp001.pdf (22KB, pdf)
lupus-2021-000542supp002.pdf (183.6KB, pdf)
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.