Abstract
Background
Assessing the pre- and post-test probability of disease in the context of routine health care is challenging. We wished to study how test performance parameters relating to clinical utility vary by clinical indication in a ‘real-world’ setting.
Methods
The diagnostic accuracy of serum total B12 and Active-B12® (holotranscobalamin) was evaluated in a primary care population, using serum methylmalonic acid as the reference standard. We used electronic requesting to establish the clinical indication for each request. Routine requests from primary care for serum total B12 were included if creatinine was also measured and estimated glomerular filtration rate was at least 60 mL/min/1.73 m2.
Results
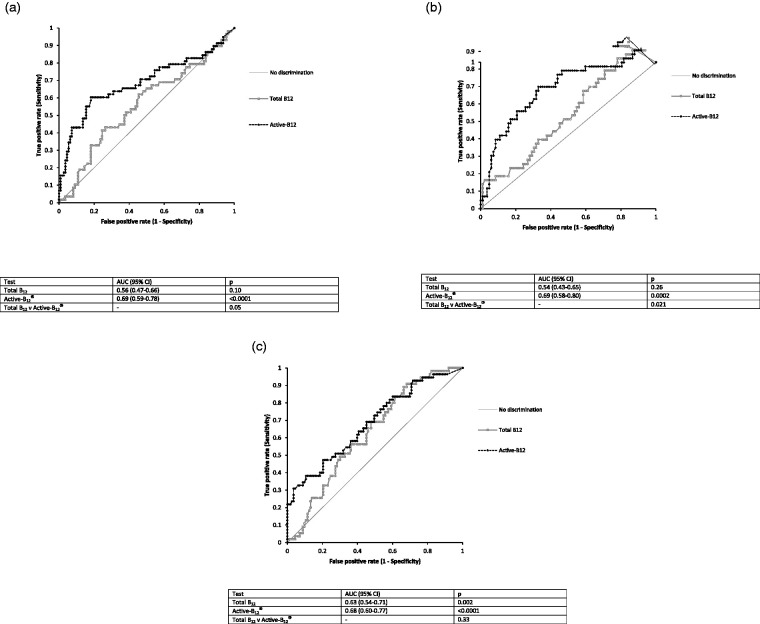
Clinical indications included peripheral neuropathy (n = 168), anaemia (n = 168), cognitive decline (n = 125), suspected dietary deficiency (n = 76), other (n = 362). For peripheral neuropathy, the area under the receiver operator curve ± 95% confidence interval (AUC ± CI) was 0.63 (0.54–0.71) (P = 0.002) for total B12 and 0.68 (0.60–0.77) (P < 0.0001) for Active-B12®. For anaemia, AUC ± CI was 0.56 (0.47–0.66) (P = 0.10) for total B12 and 0.69 (0.59–0.78) (P < 0.0001) for Active-B12®. For cognitive decline, AUC ± CI was 0.54 (0.43–0.65) (P = 0.26) for total B12 and 0.69 (0.58–0.80) (P = 0.0002) for Active-B12®. The pre–post-test change in probability of disease varied by clinical indication.
Conclusion
Combining diagnostic accuracy studies and electronic testing in a ‘real-world’ setting allows clinical utility to be assessed by clinical indication. Wider application of this would permit more personalised laboratory medicine. In this study, diagnostic performance of total B12 and Active-B12® varied across all indications. Active-B12® provided better discrimination, but this may have reflected the cut-offs used.
Keywords: Evidence-based medicine, clinical utility, Youden cut-offs, diagnostic uncertainty, electronic test requesting, ‘real-world’ setting, vitamin B12
Introduction
Diagnostic investigations are most useful at the point of maximum diagnostic uncertainty, i.e. when the potential gap between pre- and post-test probability of disease is greatest. In the context of routine health care, it is difficult to study the application of this principle to laboratory investigations. Practical challenges include: first, the existence of multiple requests and request sources, each with a potentially different pretest probability of disease; second, the need for information about the clinical indication for the request; third, the need for a validated reference standard. As a result of these difficulties, ‘real-world’ studies of the clinical utility of tests, and how this varies by clinical indication, are rare.
Separately, accurate assessment of vitamin B12 status is problematic.1 Defining deficiency in terms of tissue stores (e.g. red cell cobalamin)2 is not always feasible, and no single analytical measurement is ideal, leading some to propose the use of combined indicators of vitamin B12 status.3 Several diagnostic reports4–8 have used methylmalonic acid (MMA) as reference standard, despite its acknowledged limitations.9 We adopted this approach in a study of the diagnostic accuracy of serum total B12 and Active-B12® (holotranscobalamin, or holoTC) in the assessment of vitamin B12 status in an otherwise unselected primary care population. We used electronic requesting to establish the clinical indication for each request. Our study is the first to report how test performance parameters relating to clinical utility vary by clinical indication in a ‘real-world’ setting.
Methods
General practitioners in 68 practices in Tayside, Scotland (population approximately 400,000) were advised of the intention to perform a study of the diagnostic accuracy of the existing measure of vitamin B12 status (i.e. total serum B12), and Active-B12® (see below for details of methods), in the setting of routine health care. Their cooperation was sought in the provision of information about the clinical indication for individual requests. Clinical information was delivered through an electronic order communications system, the Integrated Clinical Environment (ICE) (Sunquest Information Systems, Uxbridge, UK). The study was performed between 12 June 2014 and 31 July 2014.
Participants
The study population consisted of consecutive patients from whom samples were received for routine measurement of total serum vitamin B12. In order to minimise the impact of reduced glomerular filtration rate (GFR) on serum MMA,10 patients were excluded if creatinine was not concurrently requested or if estimated glomerular filtration rate (eGFR) was 60 mL/min/1.73 m2 or less. Only the results of total serum vitamin B12 were reported.
Clinical indications
Health-care professionals ordering vitamin B12 through the electronic order communications system chose from the following ‘drop-down’ list of clinical indications: unexplained anaemia; macrocytic anaemia; cognitive decline; suspected dietary deficiency; peripheral neuropathy; other.
Analytical methods
Serum total B12 was measured by chemiluminescent microparticle-based competitive immunoassay on an Advia Centaur (Siemens Healthineers, Tarrytown, NY, USA), Active-B12® by chemiluminescent microparticle-based immunoassay on an Abbott Architect i2000 (Abbott Diagnostics, Abbott Park, IL, USA), both at Ninewells Hospital in Dundee. MMA was measured using a MultiPurpose Sampler (Gerstel GmbH, Mülheim an der Ruhr, Germany) coupled to liquid chromatography tandem mass spectrometry (MPS-LC-MS/MS) at the Nutristasis Unit, Viapath, St Thomas Hospital, London.
Cut-offs used to define result categories
The lower limit of the reference interval (200 pg/mL) for serum total B12 was used, in line with the manufacturer’s recommendations. The cut-off for serum holoTC (Active-B12®) was <35 pmol/L, as used by other groups.6,11 Two separate cut-offs were used for MMA, reflecting the impact of age-related changes in glomerular function on serum MMA: >0.28 µmol/L for patients ≤65 years and >0.36 µmol/L for patients >65 years. These were defined using age-specific 97.5th percentiles in vitamin B12-replete patients in a previous study.12 Total B12, Active-B12® and MMA were measured in isolation from each other; respective assessors of each did not have access to results of the other measurements, and only laboratory staff involved in the routine measurement of total B12 had access to the clinical indication for each request.
Statistical methods
Descriptive statistics (mean, 2.5th and 97.5th centiles) were calculated for population characteristics. Test performance parameters and receiver operator curve (ROC) analysis were performed by Analyze-it (Version 2.21; Analyze-it Software Ltd, West Yorkshire, UK). Youden cut-offs were established for the three commonest indications and test performance recalculated for total B12 and Active-B12® by indication.
Results
During the study period, 899 routine requests for serum total B12 were received. The breakdown of clinical indications for requests was as follows: unexplained anaemia (n = 134), macrocytic anaemia (n = 34), cognitive decline (n = 125), suspected dietary deficiency (n = 76), peripheral neuropathy (168), other (n = 362). (The anaemia categories were merged for the purposes of data analysis.) Table 1 summarises group characteristics for the three commonest indications. For the other indications, the characteristics (mean and 2.5–97.5 centiles) were as follows: (1) suspected dietary B12 deficiency: age (years) 57 (15–91), haemoglobin (g/L) 137 (109–165), haematocrit 0.42 (0.35–0.50), mean cell volume (fL) 95 (85–110), white cell count (×109/L) 6.4 (3.5–10.9), platelets (×109/L) 254 (108–406), total vitamin B12 (pg/mL) 370 (164–1004), Active-B12® (pmol/L) 65 (12–256), MMA (µmol/L) 342 (96–1869); (2) other: age (years) 61 (23–92), haemoglobin (g/L) 134 (99–165), haematocrit 0.41 (0.30–0.49), mean cell volume (fL) 94 (81–111), white cell count (×109/L) 6.8 (3.7–11.9), platelets (×109/L) 256 (130–441), total vitamin B12 (pg/mL) 388 (170–974), Active-B12® (pmol/L) 70 (17–256), MMA (µmol/L) 320 (95–1121).
Table 1.
Characteristics of study population.
| Group | Parameter | 2.5–97.5 percentile | % abnormal results (cut-off value) | |
|---|---|---|---|---|
| Total | n | |||
| Group total | 899 | |||
| Female | 560 | |||
| >65 years | 479 | |||
| Mean | ||||
| Age (years) | 64 | 23–92 | ||
| Haemoglobin (g/L) | 133 | 96–165 | 27% (<120 female, <130 male) | |
| Haematocrit | 0.41 | 0.30–0.50 | 19% (<0.37) | |
| Mean Cell Volume (fL) | 94 | 81–110 | 8% (>105) | |
| White Cell Count (×109/L) | 6.8 | 3.5–11.9 | 6% (<4 × 109) | |
| Platelets (×109/L) | 257 | 126–447 | 7% (<150 × 109) | |
| Total B12 (pg/mL) | 408 | 171–1195 | 5% (<200) | |
| Active-B12® (pmol/L) | 67 | 15–256 | 24% (<35) | |
| MMA (µmol/L) | 345 | 98–1259 | 32% (>0.28 ≤ 65yo; >0.36 > 65yo) | |
| Cognitive decline | n | |||
| Group total | 125 | |||
| Female | 80 | |||
| >65 years | 104 | |||
| Mean | ||||
| Age (y) | 77 | 44–93 | ||
| Haemoglobin (g/L) | 132 | 94–166 | 30% (<120 female, <130 male) | |
| Haematocrit | 0.41 | 0.29–0.51 | 21% (<0.37) | |
| Mean cell volume (fL) | 95 | 83–107 | 6% (>105) | |
| White cell count (×109/L) | 6.6 | 3.4–10.3 | 6% (<4 × 109) | |
| Platelets (×109/L) | 263 | 135–479 | 7% (<150 × 109) | |
| Total B12 (pg/mL) | 413 | 166–1269 | 7% (<200)yo | |
| Active-B12® (pmol/L) | 62 | 16–256 | 34% (<35)yo | |
| MMA (µmol/L) | 346 | 80–1044 | 34% (>0.28 ≤ 65yo; >0.36 > 65yo) | |
| Peripheral neuropathy | n | |||
| Group total | 168 | |||
| Female | 109 | |||
| >65 years | 58 | |||
| Mean | ||||
| Age (y) | 58 | 23–89 | ||
| Haemoglobin (g/L) | 142 | 110–168 | 7% (<120 female, <130 male) | |
| Haematocrit | 0.43 | 0.35–0.50 | 5% (<0.37) | |
| Mean cell volume (fL) | 94 | 84–106 | 2% (>105) | |
| White cell count (×109/L) | 7.2 | 3.8–13.0 | 4% (<4 × 109) | |
| Platelets (×109/L) | 258 | 126–434 | 4% (<150 × 109) | |
| Total B12 (pg/mL) | 410 | 205–838 | 1% (<200) | |
| Active-B12® (pmol/L) | 66 | 12–256 | 27% (<35) | |
| MMA (µmol/L) | 394 | 119–2011 | 33% (>0.28 ≤ 65yo;>0.36 > 65yo) | |
| Unexplained anaemia combined with macrocytic anaemia | n | |||
| Group total | 168 | |||
| Female | 97 | |||
| >65 years | 105 | |||
| Mean | ||||
| Age (years) | 69 | 26–91 | ||
| Haemoglobin (g/L) | 121 | 87–159 | 62% (<120 female, <130 male) | |
| Haematocrit | 0.38 | 0.28–0.50 | 43% (<0.37) | |
| Mean cell volume (fL) | 95 | 79–116 | 12% (>105) | |
| White cell count (×109/L) | 6.8 | 3.0–11.9 | 10% (<4 × 109) | |
| Platelets (×109/L) | 258 | 110–448 | 11% (<150 × 109) | |
| Total B12 (pg/mL) | 391 | 144–1541 | 8% (<200) | |
| Active-B12® (pmol/L) | 67 | 15–256 | 21% (<35) | |
| MMA (µmol/L) | 347 | 84–1149 | 34% (>0.28 ≤ 65yo; >0.36 > 65yo) |
Note: the upper measuring limit of the Active-B12® assay is 256 pmol/L; all samples ≥256 pmol/L were recorded for analysis as 256 pmol/L. This does not affect % abnormal results nor the clinical performance calculations. ‘Unexplained anaemia’ and ‘macrocytic anaemia’ categories were merged. In the category ‘unexplained anaemia’, 8 patients from 134 had an elevated MCV (>105 fL). In the category ‘macrocytic anaemia’, 12 patients from 34 had an elevated MCV.
ROC curves for the three commonest specific indications are shown in Figure 1(a) to (c). For other indications, the areas under the curve ± 95% confidence intervals (AUC ± CI) were as follows for total B12: suspected dietary deficiency 0.50 (0.36–0.64); other 0.47 (0.41–0.54). For Active-B12®, the AUC ± CI were: suspected dietary deficiency 0.61 (0.47–0.75); other 0.66 (0.60–0.72). For the total population, the AUC ± CI for total B12 was 0.53 (0.49–0.57) (P = 0.075), and for Active-B12®, AUC ± CI was 0.68 (0.64–0.72) (P < 0.0001). These AUC were significantly different from each other (P < 0.0001).
Figure 1.
(a) ROC curves for total B12 and Active-B12® in the diagnosis of vitamin B12 deficiency (as defined by raised serum MMA) in anaemia. (b) ROC curves for total B12 and Active-B12® in the diagnosis of vitamin B12 deficiency (as defined by raised serum MMA) in cognitive decline. (c) ROC curves for total B12 and Active-B12® in the diagnosis of vitamin B12 deficiency (as defined by raised serum MMA) in peripheral neuropathy.
Table 2 summarises the pre- and post-test probabilities of vitamin B12 deficiency (as defined by raised serum MMA), before and after measurement of serum total B12, in each of the three commonest specific indications for requesting total B12. Data are presented using the preassigned cut-offs and using Youden cut-offs (designed to optimise differentiating ability).13 We wished to calculate the post-test probability of disease in the presence of positive and negative results, respectively; therefore, we replaced the negative predictive values (NPV) by [1–NPV]. Table 3 summarises the corresponding data for Active-B12®.
Table 2.
Clinical utility of Total B12 by clinical indication.
|
Using pre-assigned cut-off (200 pg/mL) |
Using Youden cut-offsa |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication | Abnormal results (n) | PPV (%) | Δ in probability (%) with low result | 1 – NPV (%) | Δ in probability (%) with normal result | Youden cut-off (pg/mL) | Abnormal results (n) | PPV (%) | Δ in probability (%) with low result | 1–NPV (%) | Δ in probability (%) with normal result | Prevalence (%) |
| Anaemian = 168 | 11 | 18.2 | –18.4 | 39.0 | +2.4 | 287 | 46 | 50.0 | +13.4 | 30.7 | –5.9 | 36.6 |
| Cognitive declinen = 125 | 9 | 77.8 | +43.4 | 31.0 | –3.4 | 200 | 9 | 77.8 | +43.4 | 31.0 | –3.4 | 34.4 |
| Peripheral neuropathyn = 168 | 2 | 50.0 | +17.3 | 32.5 | –0.2 | 481 | 127 | 39.4 | +6.7 | 12.2 | –20.5 | 32.7 |
Δ = change; PPV (positive predictive value) = post-test probability of vitamin B12 deficiency in presence of a low total B12 result; (1–NPV [negative predictive value]) = post-test probability of vitamin B12 deficiency in presence of a normal total B12 result.
Note: Probability of vitamin B12 deficiency (as defined by raised serum MMA) before and after measurement of total B12, for three clinical indications. Pretest probability (prevalence) is shown, along with the post-test probability associated with low and normal results. For ease of reference, the changes in probability are also shown. Data are presented using preassigned cut-offs, and separately using Youden cut-offs.
aThe cut-offs that optimise differentiating ability when equal weight is given to sensitivity and specificity.
Table 3.
Clinical utility of Active-B12® by clinical indication.
|
Using pre-assigned cut-off (35 pmol/L) |
Using Youden cut-offsa |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication | Abnormal results (n) | PPV (%) | Δ in probability (%) with low result | 1 – NPV (%) | Δ in probability (%) with normal result | Youden cut-off (pg/mL) | Abnormal results (n) | PPV (%) | Δ in probability (%) with low result | 1 – NPV (%) | Δ in probability (%) with normal result | Prevalence (%) |
| Anaemian = 168 | 29 | 75.9 | +39.3 | 26.7 | –9.9 | 43 | 44 | 68.2 | +31.6 | 22.2 | –14.4 | 36.6 |
| Cognitive declinen = 125 | 43 | 55.8 | +21.4 | 23.2 | –11.2 | 44 | 54 | 52.6 | +18.2 | 19.1 | –15.3 | 34.4 |
| Peripheral neuropathyn = 168 | 46 | 50.0 | +17.3 | 26.2 | –6.5 | 29 | 30 | 63.6 | +30.9 | 25.2 | –7.5 | 32.7 |
Δ = change; PPV (positive predictive value) = post-test probability of vitamin B12 deficiency in presence of a low Active-B12®result; (1–NPV [negative predictive value]) = post-test probability of vitamin B12 deficiency in presence of a normal Active-B12®result.
Note: Probability of vitamin B12 deficiency (as defined by raised serum MMA) before and after measurement of Active-B12®, for three clinical indications. Pretest probability (prevalence) is shown, along with the post-test probability associated with low and normal results. For ease of reference, the changes in probability are also shown. Data are presented using pre-assigned cut-offs, and separately using Youden cut-offs.aThe cut-offs that optimise differentiating ability when equal weight is given to sensitivity and specificity.
Discussion
Optimal targeting of diagnostic tests in the setting of normal care is challenging. Evaluations of diagnostic accuracy are usually performed in well-defined and pre-selected populations, whereas tests in routine use are usually applied to multiple populations that vary in terms of relevant parameters (e.g. prevalence). In the current study, we have addressed this issue by performing a diagnostic evaluation in the setting of normal care.
We identified several distinct populations, based on the clinical indication for the vitamin B12 request. Patients with cognitive decline were, as anticipated, older than other patient groups, and had higher creatinine, reflecting reduced glomerular function. Interestingly, prevalence of vitamin B12 deficiency, as defined by raised serum MMA, was broadly similar in the patient groups defined by the three commonest specific indications, ranging from 32.7% in patients with peripheral neuropathy to 36.6% in patients with anaemia. This is important, since any differences in PPV and [1–NPV] across clinical indications are therefore not attributable to large differences in prevalence.
For all categories of indication, measurement of Active-B12® reliably differentiated vitamin B12 deficiency from non-deficiency, and for all the AUC for Active-B12® was greater than for total B12, although in the case of peripheral neuropathy, the difference was not statistically significant. However, these findings must be interpreted with caution in the context of the wider literature. The effect on the clinical utility of holoTC of applying different MMA cut-offs,8 and more widely, the need to base cut-offs (for all vitamin B12 biomarkers) on adverse outcomes,14 have previously been highlighted. The age-specific MMA cut-offs applied here, particularly in patients >65 years, may have affected sensitivity and specificity; other limitations include those of using MMA as a reference standard,15 and the exclusion of patients where creatinine was not requested, or where eGFR was 60 mL/min/1.73 m2 or less. On the wider issue, the ability of holoTC to predict, for example, neurological outcomes like cognitive decline in well-designed studies16–19 has been variable, and its role as a biomarker of clinically meaningful vitamin B12 deficiency remains inconclusive. Recent assessments15,20 acknowledge the limitations of holoTC and other markers and endorse the adoption of algorithm-based approaches that combine more than one measure of vitamin B12 status.3 This seems reasonable, and our findings do not provide a basis for challenging this position.
With these caveats, low Active-B12® results were more useful than normal ones; the increase in probability of vitamin B12 deficiency seen with a low result was higher, for all three of the commonest specific indications, than the decrease in probability associated with a normal result (see Table 3). This was true both when the preassigned cut-offs and when Youden cut-offs were used. As anticipated, the PPV ranking for these indications corresponded to the prevalence ranking (when the preassigned cut-off was used), with the highest PPV associated with anaemia and the lowest with peripheral neuropathy.
We have shown in the current study that it is possible to apply the principles of evidence-based medicine to routine requesting. Electronic requesting is now commonplace in health care, and, with the co-operation of clinical colleagues, can readily be harnessed to studies of diagnostic accuracy. In the presence of a validated reference standard, it is possible to establish prevalence by clinical indication and requesting source. The cost of diagnostic studies involving validated reference standards should be offset against the potential savings enabled by greater precision in laboratory requesting, for example through the application of local algorithms.
Acknowledgements
Reagents were provided by Axis-Shield Diagnostics.
Footnotes
Declaration of conflicting interests: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Craig was an employee of Axis-Shield Diagnostics at the time of this research.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval: Not applicable.
Guarantor: MJM.
Contributorship: MJM had the idea for the study and wrote the first draft of the paper. DC performed data analysis. FB, ME and MH assisted with the laboratory processing of samples, and commented on drafts of the paper. ED and WAB commented on drafts of the paper.
ORCID iD
Michael J Murphy https://orcid.org/0000-0001-6735-125X
References
- 1.Hughes CF, McNulty H.Assessing biomarker status of vitamin B12 in the laboratory: no simple solution. Ann Clin Biochem 2018; 55: 188–189. [DOI] [PubMed] [Google Scholar]
- 2.Valente E, Scott JM, Ueland P-M, et al. Diagnostic accuracy of holotranscobalamin, methylmalonic acid, serum cobalamin, and other indicators of tissue vitamin B12 status in the elderly. Clin Chem 2011; 57: 856–863. [DOI] [PubMed] [Google Scholar]
- 3.Fedosov SN, Brito A, Miller JW, et al. Combined indicator of vitamin B12 status: modification for missing biomarkers and folate status and recommendations for revised cut-points. Clin Chem Lab Med 2015; 53: 1215–1225. [DOI] [PubMed] [Google Scholar]
- 4.Risch M, Meier DW, Sakem B, et al. Vitamin B12 and folate levels in healthy Swiss senior citizens: a prospective study evaluating reference intervals and decision limits. BMC Geriatr 2015; 15: 82–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clarke R, Sherliker P, Hin H, et al. Detection of vitamin B12 deficiency in older people by measuring vitamin B12 or the active fraction of vitamin B12, holotranscobalamin. Clin Chem 2007; 53: 963–970. [DOI] [PubMed] [Google Scholar]
- 6.Obeid R, Herrmann W.Holotranscobalamin in laboratory diagnosis of cobalamin deficiency compared to total cobalamin and methylmalonic acid. Clin Chem Lab Med 2007; 45: 1746–1750. [DOI] [PubMed] [Google Scholar]
- 7.Schrempf W, Eulitz M, Neumeister V, et al. Utility of measuring vitamin B12 and its active fraction, holotranscobalamin, in neurological vitamin B12 deficiency syndromes. J Neurol 2011; 258: 393–401. [DOI] [PubMed] [Google Scholar]
- 8.Heil SG, de Jonge R, de Rotte CFJ, et al. Screening for metabolic vitamin B12 deficiency by holotranscobalamin in patients suspected of vitamin B12 deficiency: a multicentre study. Ann Clin Biochem 2012; 49: 184–189. [DOI] [PubMed] [Google Scholar]
- 9.Green R, Allen LH, Bjørke-Monsen AL, et al. Vitamin B12 deficiency. Nat Rev Dis Primers 2017; 3: 20. [DOI] [PubMed] [Google Scholar]
- 10.Lewerin C, Ljungman S, Nilsson-Ehle H.Glomerular filtration rate as measured by serum cystatin C is an important determinant of plasma homocysteine and serum methylmalonic acid in the elderly. J Intern Med 2007; 261: 65–73. [DOI] [PubMed] [Google Scholar]
- 11.Miller JW, Garrod MG, Rockwood AL, et al. Measurement of total vitamin B12 and holotranscobalamin, singly and in combination, in screening for metabolic vitamin B12 deficiency. Clin Chem 2006; 52: 278–285. [DOI] [PubMed] [Google Scholar]
- 12.Vogiatzoglou A, Oulhaj A, Smith AD, et al. Determinants of plasma methylmalonic acid in a large population: implications for assessment of vitamin B12 status. Clin Chem 2009; 55: 2198–2206. [DOI] [PubMed] [Google Scholar]
- 13.Youden WJ.Index for rating diagnostic tests. Cancer 1950; 3: 32–35. [DOI] [PubMed] [Google Scholar]
- 14.Yetley EA, Pfeiffer CM, Phinney KW, et al. Biomarkers of vitamin B12 status in NHANES: a roundtable summary. Am J Clin Nutr 2011; 94: 313S–321S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wolffenbuttel BHR, Wouters HJCM, Heiner-Fokkema MR, et al. The many faces of cobalamin (vitamin B12) deficiency. Mayo Clin Proc Innov Qual Outcomes 2019; 3: 200–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clarke R, Birks J, Nexo E, et al. Low vitamin B-12 status and risk of cognitive decline in older adults. Am J Clin Nutr 2007; 86: 1384–1391. [DOI] [PubMed] [Google Scholar]
- 17.Kivipelto M, Annerbo S, Hultdin J, et al. Homocysteine and holo-transcobalamin and the risk of dementia and Alzheimer's disease: a prospective study. Eur J Neurol 2009; 16: 808–813. [DOI] [PubMed] [Google Scholar]
- 18.Hooshmand B, Solomon A, Kareholt I, et al. Homocysteine and holotranscobalamin and the risk of Alzheimer disease: a longitudinal study. Neurology 2010; 75: 1408–1414. [DOI] [PubMed] [Google Scholar]
- 19.Miles LM, Allen E, Mills K, et al. Vitamin B12 status and neurologic function in older people: a cross-sectional analysis of baseline trial data from the Older People and Enhanced Neurological Function (OPEN) study. Am J Clin Nutr 2016; 104: 790–796. [DOI] [PubMed] [Google Scholar]
- 20.Harrington DJ.Laboratory assessment of vitamin B12 status. J Clin Pathol 2017; 70: 168–173. [DOI] [PubMed] [Google Scholar]