Abstract
Objective:
To evaluate broad-spectrum intravenous antibiotic use before and after the implementation of a revised febrile neutropenia management algorithm in a population of adults with hematologic malignancies.
Design:
Quasi-experimental study.
Setting and population:
Patients admitted between 2014 and 2018 to the Adult Malignant Hematology service of an acute-care hospital in the United States.
Methods:
Aggregate data for adult malignant hematology service were obtained for population-level antibiotic use: days of therapy (DOT), C. difficile infections, bacterial bloodstream infections, intensive care unit (ICU) length of stay, and in-hospital mortality. All rates are reported per 1,000 patient days before the implementation of an febrile neutropenia management algorithm (July 2014–May 2016) and after the intervention (June 2016–December 2018). These data were compared using interrupted time series analysis.
Results:
In total, 2,014 patients comprised 6,788 encounters and 89,612 patient days during the study period. Broad-spectrum intravenous (IV) antibiotic use decreased by 5.7% with immediate reductions in meropenem and vancomycin use by 22 (P = .02) and 15 (P = .001) DOT per 1,000 patient days, respectively. Bacterial bloodstream infection rates significantly increased following algorithm implementation. No differences were observed in the use of other antibiotics or safety outcomes including C. difficile infection, ICU length of stay, and in-hospital mortality.
Conclusions:
Reductions in vancomycin and meropenem were observed following the implementation of a more stringent febrile neutropenia management algorithm, without evidence of adverse outcomes. Successful implementation occurred through a collaborative effort and continues to be a core reinforcement strategy at our institution. Future studies evaluating patient-level data may identify further stewardship opportunities in this population.
Nearly 80% of patients undergoing chemotherapy for leukemia or receiving hematopoietic stem-cell transplant (HSCT) will experience at least 1 episode of febrile neutropenia.1 Fevers without microbiologically documented infection or clinically documented infection comprise >50% of febrile neutropenia episodes.1 Consensus guidelines provide mixed recommendations about whether empirical antibiotic treatment should continue until neutrophil recovery for febrile neutropenia patients without an identified source.2,3 Much of the data supporting empirical antibiotic treatment until neutrophil recovery were generated before routine antibiotic prophylaxis in high-risk neutropenia became widespread, and also at a time when antibiotic resistance was less common.4 Although it is imperative to promptly initiate and continue empirical antibiotic treatment in patients with concerns for bacterial infections, it is also essential to steward unnecessary antibiotic exposure to minimize resistance development and other collateral damage of antibiotic use. Furthermore, recent observational studies and a randomized trial demonstrated that discontinuation of empirical antibiotic treatment in clinically stable patients with fever resolution and no microbiologically documented infection or clinically documented infection may be safe despite persistent neutropenia.5–11 In June 2016, a multidisciplinary task force at University of California–San Francisco (UCSF) Medical Center convened to revise its febrile neutropenia management algorithm.
The study objective was to evaluate the association of the revised febrile neutropenia algorithm intervention with intravenous (IV) antibiotic utilization and related outcomes in adult patients on the malignant hematology service. We hypothesized that there would be reductions in IV antibiotic days of therapy (DOT) without adverse clinical outcomes following algorithm implementation.
Methods
Study design
This investigation was a quasi-experimental study using aggregated electronic medical record data of all patients on the adult malignant hematology service at the UCSF Medical Center between July 2014 and December 2018. The inclusion criteria were patients aged ≥18 years who spent at least 1 census day on the malignant hematology service during the study period. Patients who were transferred to the pediatric malignant hematology unit were excluded regardless of age because the algorithm was only implemented for the adult services.
In June 2016, our adult antimicrobial stewardship program and malignant hematology service implemented a revised febrile neutropenia management algorithm as recommended by a multidisciplinary task force of physicians, pharmacists, and nurses from the malignant hematology, hospital medicine, and infectious diseases services at UCSF. The 3 main revisions to the algorithm were (1) empirical vancomycin use should be considered in hemodynamically unstable patients and/or those with methicillin-resistant Staphylococcus aureus (MRSA) risk factors; (2) broadening from cefepime to an antipseudomonal carbapenem antibiotic (eg, meropenem) should be reserved for hemodynamically unstable patients after 24 hours; and (3) febrile neutropenia empirical antibiotic treatment (eg, cefepime) was continued for a defined duration of 10 days regardless of neutrophil recovery in patients with resolved fevers and no microbiologically documented infection or clinically documented infection. Patients with microbiologically documented infection or clinically documented infection should complete a standard treatment course for that infection. Patients resumed febrile neutropenia prophylaxis with levofloxacin following course completion. Prior to these revisions, vancomycin and broad-spectrum gram-negative antibiotic use in febrile neutropenia patients were not standardized to specific clinical criteria (eg, MRSA risk factors or hemodynamic instability). The UCSF Institutional Review Board approved this study.
Algorithm implementation strategy and adherence evaluation
Several key strategies were employed to implement the revised algorithm. Initial in-person meetings were held with key stakeholders on the malignant hematology service (ie, nurses, pharmacists, oncologists, hospitalists, and advanced care practitioners) to discuss the algorithm changes suggested by the task force. Upon finalization of the proposed changes, the algorithm was posted in work rooms on the malignant hematology unit as well as on the antimicrobial stewardship program website, and e-mails were sent to relevant clinician groups (ie, hospitalists, infectious disease physicians, nurse practitioners, and oncologists). Antimicrobial stewardship pharmacists and physicians performed prospective audit and feedback of patients receiving broad-spectrum intravenous (IV) antibiotics on the malignant hematology service, Monday through Friday, to support and reinforce the new algorithm. Algorithm adherence was evaluated over a 6-month period (June 28 to December 16, 2016). Adult patients with febrile neutropenia were retrospectively evaluated to determine whether the antibiotics they received were appropriate and in accordance with the new algorithm.
Study outcomes
The primary study outcome was DOT of a composite of broad-spectrum IV antibiotics commonly used for febrile neutropenia administered per 1,000 patient days.12 These antibiotics included aztreonam, cefepime, meropenem, piperacillin-tazobactam, and vancomycin. Cefepime is the choice empiric gram-negative antibiotic at our institution for febrile neutropenia and for the purposes of this analysis was combined with piperacillin-tazobactam to account for multiple drug shortages that occurred throughout our study period. Levofloxacin was removed from all analyses because it is the high-risk neutropenia prophylactic antibiotic of choice at our institution.13
Secondary study outcomes included DOT of individual broad-spectrum antibiotics and a composite of all IV antibiotics as well as Clostridioides difficile infection incidence rates, bacterial bloodstream infection incidence rates, intensive care unit (ICU) length of stay, and hospital all-cause mortality.
The Clostridioides difficile infections were defined by a positive 2-step algorithm for either the toxin protein detected via enzyme immunoassay (EIA) or the toxin gene detected by polymerase chain reaction.14 Bacterial bloodstream infections were defined by a single positive blood culture at any point during the hospital stay, and only the first culture per bacteria isolated was included in the analysis. Bacteria traditionally considered to be contaminants (eg, coagulase-negative staphylococci) were included in the analysis because these organisms can be significant in this patient population and are often treated. ICU length of stay was determined per patient. In-hospital all-cause mortality was determined by a recorded discharge disposition of the deceased patients. All secondary outcomes were standardized to 1,000 patient days.
Statistical analyses
An interrupted time series with segmented regression analysis was used to evaluate differences in monthly composite IV antibiotic DOTs and clinical outcomes associated with the implementation of a revised febrile neutropenia management algorithm.15 The study period comprised 54 months divided into 2 phases: 23 months of preintervention from July 2014 to May 2016 and 31 months of intervention from June 2016 to December 2018. This sample size provides sufficient power to detect at least a 30% change in antibiotic DOTs.16 The Durbin-Watson d statistic was used to test for potential autocorrelation of data points with d values >2 indicating no autocorrelation.
Based on existing literature describing similar antimicrobial stewardship initiatives, we hypothesized an immediate decrease (ie, level change) as well as a sustained or continued decrease (ie, slope change) in antibiotic use because the revised algorithm became more widely adopted.17 Thus, we proposed that the most appropriate regression model capture both level and slope changes without a lag period.15 The proposed segmented regression model assumes Y t = β 0 + β 1 T t + β 2 X t + β 3 X t T t + ϵt, where Y t is the aggregated broad-spectrum IV antibiotic DOT measured monthly at time t; T t is the time since the start of the study; X t represents the intervention with preintervention months designated as 0 and intervention months designated as 1; and X t T t is the interaction term. Furthermore, β 0 is the intercept and represents the initial antibiotic DOT and β 1 is the slope in the preintervention period. Change in level (β 2) was defined as the immediate difference between the observed value at the end of the preintervention period and the beginning of the intervention periods. β 3 is the slope in the intervention period. We also calculated the difference between the change rates of the preintervention and intervention periods (β 1 + β 3). Results are reported as estimate coefficients with 95% confidence intervals. Significant P values <.05 for β 2 indicate an immediate change in antibiotic DOT following the implementation of the revised algorithm, and a significant P value for β 1 or β 3 represents a change in the slope in the preintervention or intervention period, respectively. A significant P value for the difference in the change rates between the preintervention period and intervention periods (β 1 + β 3) indicates a difference in the slopes between the preintervention period and intervention period. Newey-West standard errors were reported for coefficients estimated by ordinary least-squares regression, which was appropriate for our continuous primary outcome of broad-spectrum IV antibiotic DOT. The maximum likelihood ratio test was used to assess overall model fit. Descriptive statistics were used to compare baseline differences between the preintervention and intervention cohorts including the χ2 test or the Fisher exact test for categorical variables, and the t test or the Mann-Whitney U test for continuous variables. Time-series scatter plots were also visually inspected to identify distributions, trends, and outliers. Statistical analyses were conducted using Stata version 15.0 software (StataCorp, College Station, TX).
Results
Between July 2014 and December 2018, a total of 2,014 patients comprised 6,788 encounters and 89,612 patient days on the Adult Malignant Hematology service at UCSF Medical Center. Moreover, 892 patients comprised 2,827 encounters and 37,317 patient days in the preintervention period and 1,122 patients comprised 3,961 encounters and 52,295 patient days in the intervention period. The median age was 56 years (interquartile [IQR], 55–57) and 60% were male; these distributions were similar in both preintervention and intervention periods. The median hospital length of stay was 8.75 days (IQR, 7–10): 9 days (IQR, 7–10) in the preintervention period and 8 days (IQR, 7–10) in the intervention period (P = .78).
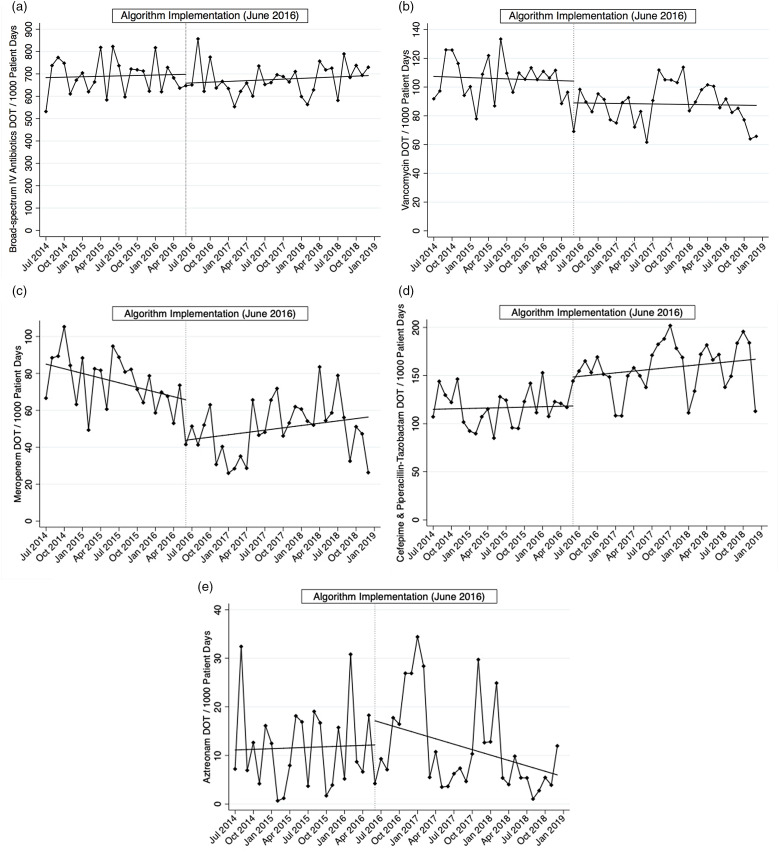
Composite IV antibiotic use decreased by 5.7% from 704 to 664 DOT per 1,000 patient days between the preintervention and intervention periods, with an absolute difference of 40 days. This difference was not significant (Table 1 and Fig. 1a). We detected were statistically significant immediate reductions in meropenem and vancomycin use by 22 DOT per 1,000 patient days (P = .02) and 15 DOT per 1,000 patient days (P = .001), respectively, whereas the use of noncarbapenem antipseudomonal antibiotics increased by 30.2 DOT per 1,000 patient days (P = .006) (Table 1 and Fig. 1b–d). These data represent ~28% and 18% reductions in meropenem and vancomycin use, respectively, from preintervention baseline rates. Use of both antibiotics continued to remain below the preintervention baseline throughout the study period. Meropenem use was decreasing in the preintervention period and despite an increasing slope in the intervention period, overall use remained below its use during the preintervention period (Fig. 1c). Similarly, vancomycin use was also decreasing in the preintervention period, and this reduction was sustained in the intervention period (Fig. 1b). Overall IV antibiotic use did not change over the intervention period (data not shown). Autocorrelation was not detected in the main analysis of the primary outcome (Durbin-Watson d statistic of 3.17) nor any of the other models.
Table 1.
Association Between Revised Febrile Neutropenia Management Algorithm and Days of Therapy per 1,000 Patient Days of Commonly Used Intravenous Antibiotics
| Parameter | Coefficient (95% CI) | P Value |
|---|---|---|
| Primary outcome—composite broad-spectrum IV antibioticsa | ||
| Preintervention slope (β 1) | 0.64 (−3.93 to 5.21) | .78 |
| Level change immediately following intervention (β 2) | −39.6 (−109 to 29.9) | .26 |
| Slope change following intervention (β 3) | 0.49 (−4.83 to 5.80) | .85 |
| Intervention trend (β 1 + β 3) | 1.13 (−1.55 to 3.80) | .40 |
| Vancomycin | ||
| Preintervention slope (β 1) | −0.14 (−1.06 to 0.77) | .76 |
| Level change immediately following intervention (β 2) | −15.2 (−27.8 to −2.52) | .02 |
| Slope change following intervention (β 3) | 0.08 (−1.05 to 1.21) | .88 |
| Intervention trend (β 1 + β 3) | −0.06 (−0.68 to 0.57) | .86 |
| Meropenem | ||
| Preintervention slope (β 1) | −0.84 (−1.57 to −0.12) | .02 |
| Level change immediately following intervention (β 2) | −21.9 (−34.8 to −8.99) | .001 |
| Slope change following intervention (β 3) | 1.2 (0.26 to 2.26) | .01 |
| Intervention trend (β 1 + β 3) | 0.42 (−0.27 to 1.12) | .23 |
| Cefepime and piperacillin-tazobactam | ||
| Preintervention slope (β 1) | 0.15 (−0.96 to 1.26) | .79 |
| Level change immediately following intervention (β 2) | 30.2 (8.88 to 51.4) | .006 |
| Slope change following intervention (β 3) | 0.47 (−1.07 to 2.00) | .55 |
| Intervention trend (β 1 + β 3) | 0.61 (−0.45 to 1.68) | .25 |
| Aztreonam | ||
| Preintervention slope (β 1) | 0.04 (−0.46 to 0.55) | 0.86 |
| Level change immediately following intervention (β 2) | 4.99 (−6.37 to 16.3) | 0.38 |
| Slope change following intervention (β 3) | −0.42 (−1.05 to 0.21) | .19 |
| Intervention trend (β 1 + β 3) | −0.37 (−0.78 to 0.04) | .07 |
Note. CI, confidence interval.
Composite includes aztreonam, cefepime, meropenem, piperacillin-tazobactam, and vancomycin.
Fig. 1.
Segmented linear regression of IV antibiotics days of therapy (DOT) per 1,000 patient days over time. Dotted vertical line, intervention month June 2016. (a) Composite broad-spectrum IV antibiotics. (b) Vancomycin. (c) Meropenem. (d) Cefepime plus piperacillin-tazobactam. (e) Aztreonam.
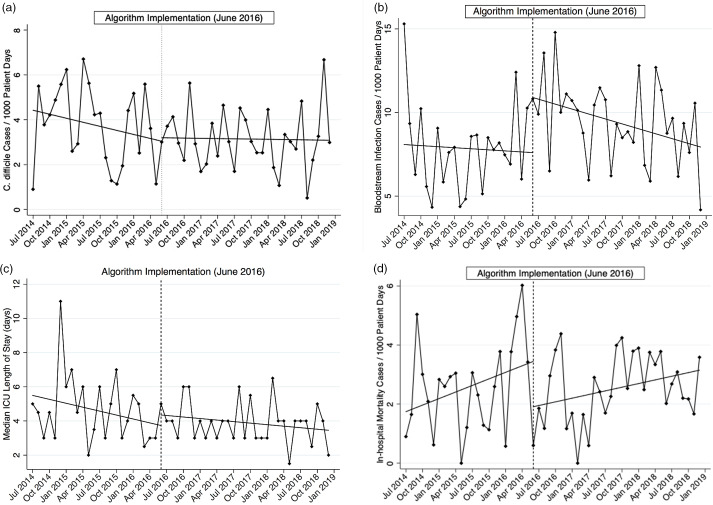
No statistically significant differences were observed in the incidence rates of C. difficile infection, ICU length of stay, or in-hospital mortality between the 2 study periods (Table 2 and Fig. 2a, c, d). However, there was an immediate and significant increase in bacterial bloodstream infection rates (Fig. 2b). The median ICU length of stay for patients admitted to the ICU was 4 days (IQR, 3–5): 4.5 days (IQR, 3–6) in the preintervention period and 4 days (IQR, 3–4) in the intervention period (P = .18).
Table 2.
Association Between Revised Febrile Neutropenia Management Algorithm and Secondary Study Outcomes Standardized to 1,000 Patient Daysa
| Parameter | Coefficient (95% CI) | P Value |
|---|---|---|
| C. difficile infection rate | ||
| Preintervention slope (β 1) | −0.06 (−0.17 to 0.05) | .28 |
| Level change immediately following intervention (β 2) | 0.15 (−1.59 to 1.90) | .86 |
| Slope change following intervention (β 3) | 0.06 (−0.07 to 0.18) | .37 |
| Intervention trend (β 1 + β 3) | −0.004 (−0.06 to 0.05) | .90 |
| Bacterial bloodstream infection rate | ||
| Preintervention slope (β 1) | −0.02 (−0.23 to 0.18) | .84 |
| Level change immediately following intervention (β 2) | 3.30 (0.97 to 5.63) | .006 |
| Slope change following intervention (β 3) | −0.08 (−0.29 to 0.14) | .47 |
| Intervention trend (β 1 + β 3) | −0.10 (−0.17 to −0.03) | .006 |
| Gram-positive bacterial bloodstream infection rate | ||
| Preintervention slope (β 1) | 0.07 (−0.13 to 0.27) | .48 |
| Level change immediately following intervention (β 2) | 2.19 (−1.15 to 5.54) | .19 |
| Slope change following intervention (β 3) | −0.19 (−0.41 to 0.04) | .10 |
| Intervention trend (β 1 + β 3) | −0.12 (−0.23 to 0.01) | .03 |
| Gram-negative bacterial bloodstream infection rate | ||
| Preintervention slope (β 1) | −0.001 (−0.07 to 0.06) | .88 |
| Level change immediately following intervention (β 2) | 1.11 (−0.29 to 2.52) | .12 |
| Slope change following intervention (β 3) | 0.01 (−0.09 to 0.11) | .85 |
| Intervention trend (β 1 + β 3) | 0.01 (−0.07 to 0.82) | .89 |
| Intensive care unit length of stay in days (median) | ||
| Preintervention slope (β 1) | −0.08 (−0.19 to 0.04) | .18 |
| Level change immediately following intervention (β 2) | 0.62 (−0.79 to 2.03) | .38 |
| Slope change following intervention (β 3) | 0.05 (−0.07 to 0.17) | .44 |
| Intervention trend (β 1 + β 3) | −0.03 (−0.07 to 0.01) | .15 |
| Hospital all-cause mortality rate | ||
| Preintervention slope (β 1) | 0.07 (−0.04 to 0.19) | .20 |
| Level change immediately following intervention (β 2) | −1.54 (−3.45 to 0.38) | .11 |
| Slope change following intervention (β 3) | −0.03 (−0.16 to 0.09) | .60 |
| Intervention trend (β 1 + β 3) | 0.04 (−0.01 to 0.09) | .11 |
Note. CI, confidence interval.
Unless otherwise specified.
Fig. 2.
Segmented linear regression of secondary outcomes per 1,000 patient days over time (except ICU length of stay, which is represented as medians). Dotted vertical line, intervention month June 2016. (a) Clostridioides difficile infection. (b) Bacterial bloodstream infection. (c) Intensive care unit length of stay. (d) In-hospital mortality.
Discussion
The implementation of a revised febrile neutropenia management algorithm significantly reduced vancomycin and meropenem use at our institution. These findings are consistent with our hypothesis because the algorithm revisions directly delineated specific criteria for vancomycin and broader gram-negative antibiotics (ie, meropenem). Vancomycin and meropenem DOTs continued to remain below preintervention rates. Notably, vancomycin continued to decrease during the intervention period and represents a successful antimicrobial stewardship intervention for this high-use antibiotic.18 Although meropenem reductions were also observed, there was a compensatory increase in anti-pseudomonal β-lactam use, suggesting that although escalation of empirical antibiotic treatment to meropenem was effective, the overall duration of cefepime and piperacillin-tazobactam did not change.
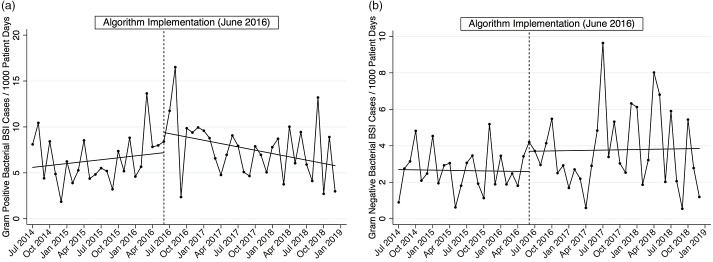
Algorithm implementation did not appear to be significantly associated with changes in C. difficile infection rates, ICU length of stay, or in-hospital all-cause mortality. We did not discern an obvious explanation (eg, changes in microbiologic culturing practices, known hospital unit outbreaks) for the observed increase in bacterial bloodstream infections following algorithm implementation. Gram-positive and gram-negative bacterial bloodstream infections reflect that of all bacterial bloodstream infections with observed increases following the intervention (Table 2 and Fig. 3). No discernible patterns were observed between the preintervention and intervention periods for concerning pathogens such as MRSA, vancomycin-resistant E. faecalis and E. faecium, extended-spectrum β-lactamase (ESBL)–producing organisms (eg, E. coli, K. pneumoniae, K. oxytoca), or P. aeruginosa (data not shown). These transiently higher rates did not correspond to an increase in in-hospital mortality. Regardless, this finding has prompted a closer evaluation using patient-level data to fully ascertain whether these were spurious increases or were related to the algorithm change.
Fig. 3.
Segmented linear regression of bacterial bloodstream infections per 1,000 patient days over time. Dotted vertical line, intervention month June 2016. (a) Gram-positive bacteria. (b) Gram-negative bacteria.
Real-world de-escalation and discontinuation strategies for management of empirical antibiotic treatment in febrile neutropenia are increasing, and results from the present study contribute and support existing evidence. A phase 4 randomized trial (ie, the “How Long” study) conducted in Spain5 enrolled 157 high-risk adult patients with febrile neutropenia initiated on empirical antibiotic treatment and assigned patients to either de-escalation when afebrile for 72 hours (experimental group) or continued on empirical antibiotic treatment until neutrophil count recovery (control group). The experimental group had more antibiotic-free days and fewer adverse events without any observed increase in harms.5 Implementing a similar de-escalation approach may help to address the compensatory increase in cefepime and piperacillin-tazobactam that we observed in our study.
La Martire et al11 also reported similar results in their interrupted time series analysis following the implementation of a de-escalation and discontinuation protocol in their high-risk neutropenic patients in France. These researchers noted an overall decrease in carbapenem use with no changes in C. difficile infections, bloodstream infections, ICU transfers, and mortality.11 Results from these 2 and several other studies collectively support recommendations put forth in the European Conference of Infections in Leukemia (ECIL) guidelines that early de-escalation is safe in hemodynamically stable patients regardless of neutrophil recovery, though the National Comprehensive Cancer Network and Infectious Diseases Society of America guidelines still list this strategy as an “alternative” approach to continuation of broad-spectrum coverage until count recovery.6,8,9,19 In a secondary analysis of their trial comparing cefepime to imipenem-cilastatin, Cherif et al9 found no association between early empirical antibiotic treatment discontinuation and rates of fever recurrences or mortality. Similarly, Slobbe et al8 found that discontinuation of empirical antibiotic treatment was safe after 72 hours in patients without an microbiologically documented infection or clinically documented infection. Snyder et al10 evaluated early de-escalation of empirical antibiotic treatment in 46 neutropenic patients and found no differences in worse outcomes compared to 74 patients who did not receive early de-escalation of empirical antibiotic treatment.10 Taken together, results from these studies support earlier empirical antibiotic treatment discontinuation in patients without microbiologically documented infection or clinically documented infection, which will be a future focus for our institution.
The 2 major limitations to our study are the lack of randomization and lack of patient-level data. Drawbacks of observational studies involve third-variable problems and regression to the mean; however, we chose the quasi-experimental study design with interrupted time series analysis as a more robust design to assess regression to the mean. Antibiotic prescribing patterns with seasonality trends and drug shortages (eg, cefepime and piperacillin-tazobactam) during our study period are 2 measurable time-varying confounders that might have affect our observed outcomes. A concern with seasonality is that it can lead to autocorrelation (eg, higher antibiotic use clustered in the summer months when the institution welcomes new healthcare trainees unfamiliar with institutional practices, or during the influenza season). However, autocorrelation was not detected in our models, thereby suggesting that seasonal differences in antimicrobial prescribing were not significant and/or immeasurable. Furthermore, our study period spanned nearly 4 years, with adequate observations for all seasons in the preintervention and intervention periods. Although we did not examine patient-level data, no major changes in patient case mix or other service-line factors (eg, volume of transplants) would have been anticipated to confound the primary analysis of antibiotic use during this time. This aggregate dataset allowed for population-level analysis over the 4 years and was appropriate to identify trends in IV antibiotic use and other relevant outcomes. However, patient-level data are necessary to further investigate other findings including the observed increase in bloodstream infection rates immediately following implementation. These patient level data can more specifically identify the at-risk population of patients with febrile neutropenia, can establish temporality to determine whether these increases occurred following algorithm implementation, can discern between specific pathogens, and can inform regression models to determine independent associations.
These study findings support the continued role of evidence-based management algorithms as effective intervention tools for antimicrobial stewardship programs, especially in the hematology-oncology population.20 This study also emphasizes the crucial importance of multidisciplinary contributions to stewardship efforts. We successfully engaged key stakeholders from the malignant hematology, hospital medicine and infectious diseases services to revise the algorithm. This multidisciplinary collaboration was instrumental during the implementation period of this algorithm across adult services. Results from a quality improvement project indicated that overall algorithm adherence was 57% among 166 febrile neutropenia episodes from June 28 to December 16, 2016 (data not shown). Most nonadherence (88%) was not de-escalating soon enough (eg, continuing vancomycin or escalating from cefepime to meropenem for persistent fevers in an otherwise stable patient without microbiologically documented infection or clinically documented infection). Algorithm adherence improved and ranged from 30% at the beginning to nearly 75% by the end of the 6-month evaluation, which may reflect widespread awareness and uptake of this new algorithm by our clinician stakeholders. In the setting of dwindling effective pharmacotherapy options for our patients, alternative evidence-based management strategies must be employed. Efforts to maintain these reductions in antibiotic use continue to require multifaceted approaches including routine education and also prospective audit and feedback.
Acknowledgments
Financial support
No financial support was provided relevant to this article.
Conflicts of interest
R.O. reports receiving personal fees from Amgen, Genentech, and Jazz Pharmaceuticals; she serves as site principal investigator for Astellas, Daiichi Sankyo, Genentech, and Pfizer. S.B.D. reports receiving consulting fees from Genentech and Basilea. All other authors report no conflicts of interest relevant to this article.
References
- 1.Nesher L, Rolston KV.The current spectrum of infection in cancer patients with chemotherapy related neutropenia. Infection 2014;42:5–13. [DOI] [PubMed] [Google Scholar]
- 2.Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 2011;52:e56–e93. [DOI] [PubMed] [Google Scholar]
- 3.Baden LR, Swaminathan S, Angarone M, et al. Prevention and treatment of cancer-related infections, version 2.2016, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2016;14:882–913. [DOI] [PubMed] [Google Scholar]
- 4.Taplitz RA, Kennedy EB, Bow EJ, et al. Antimicrobial prophylaxis for adult patients with cancer-related immunosuppression: ASCO and IDSA clinical practice guideline update. J Clin Oncol 2018;36:3043–3054. [DOI] [PubMed] [Google Scholar]
- 5.Aguilar-Guisado M, Espigado I, Martin-Pena A, et al. Optimisation of empirical antimicrobial therapy in patients with haematological malignancies and febrile neutropenia (How Long study): an open-label, randomised, controlled phase 4 trial. Lancet Haematol 2017;4:e573–e583. [DOI] [PubMed] [Google Scholar]
- 6.Cornelissen JJ, Rozenberg-Arska M, Dekker AW.Discontinuation of intravenous antibiotic therapy during persistent neutropenia in patients receiving prophylaxis with oral ciprofloxacin. Clin Infect Dis 1995;21:1300–1302. [DOI] [PubMed] [Google Scholar]
- 7.Horowitz HW, Holmgren D, Seiter K.Stepdown single agent antibiotic therapy for the management of the high-risk neutropenic adult with hematologic malignancies. Leuk Lymphoma 1996;23:159–163. [DOI] [PubMed] [Google Scholar]
- 8.Slobbe L, Waal L, Jongman LR, Lugtenburg PJ, Rijnders BJ.Three-Day treatment with imipenem for unexplained fever during prolonged neutropaenia in haematology patients receiving fluoroquinolone and fluconazole prophylaxis: a prospective observational safety study. Eur J Cancer 2009;45:2810–2817. [DOI] [PubMed] [Google Scholar]
- 9.Cherif H, Bjorkholm M, Engervall P, et al. A prospective, randomized study comparing cefepime and imipenem-cilastatin in the empirical treatment of febrile neutropenia in patients treated for haematological malignancies. Scand J Infect Dis 2004;36:593–600. [DOI] [PubMed] [Google Scholar]
- 10.Snyder M, Pasikhova Y, Baluch A.Early antimicrobial de-escalation and stewardship in adult hematopoietic stem cell transplantation recipients: retrospective review. Open Forum Infect Dis 2017;4:ofx226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.La Martire G, Robin C, Oubaya N, et al. De-escalation and discontinuation strategies in high-risk neutropenic patients: an interrupted time series analyses of antimicrobial consumption and impact on outcome. Eur J Clin Microbiol Infect Dis 2018;37:1931–1940. [DOI] [PubMed] [Google Scholar]
- 12.Polk RE, Fox C, Mahoney A, Letcavage J, MacDougall C.Measurement of adult antibacterial drug use in 130 US hospitals: comparison of defined daily dose and days of therapy. Clin Infect Dis 2007;44:664–670. [DOI] [PubMed] [Google Scholar]
- 13.Mikulska M, Averbuch D, Tissot F, et al. Fluoroquinolone prophylaxis in haematological cancer patients with neutropenia: ECIL critical appraisal of previous guidelines. J Infect 2018;76:20–37. [DOI] [PubMed] [Google Scholar]
- 14.McDonald LC, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis 2018;66:e1–e48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bernal JL, Cummins S, Gasparrini A.Interrupted time series regression for the evaluation of public health interventions: a tutorial. Int J Epidemiol 2017;46:348–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hawley S, Ali MS, Berencsi K, Judge A, Prieto-Alhambra D.Sample size and power considerations for ordinary least squares interrupted time series analysis: a simulation study. Clin Epidemiol 2019;11:197–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wattier RL, Levy ER, Sabnis AJ, Dvorak CC, Auerbach AD.Reducing second gram-negative antibiotic therapy on pediatric oncology and hematopoietic stem cell transplantation services. Infect Control Hosp Epidemiol 2017;38:1039–1047. [DOI] [PubMed] [Google Scholar]
- 18.Baggs J, Fridkin SK, Pollack LA, Srinivasan A, Jernigan JA.Estimating national trends in inpatient antibiotic use among US hospitals from 2006 to 2012. JAMA Intern Med 2016;176:1639–1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Averbuch D, Orasch C, Cordonnier C, et al. European guidelines for empirical antibacterial therapy for febrile neutropenic patients in the era of growing resistance: summary of the 2011 4th European Conference on Infections in Leukemia. Haematologica 2013;98:1826–1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Webb BJ, Majers J, Healy R, et al. Antimicrobial stewardship in a hematological malignancy unit: carbapenem reduction and decreased vancomycin-resistant Enterococcus infection. Clin Infect Dis 2020;71:960–967. [DOI] [PubMed] [Google Scholar]