Key Points
Question
What is the placebo effect magnitude in different treatment modalities used for management of patients with treatment-resistant depression?
Findings
In this systematic review and meta-analysis of 3228 patients with treatment-resistant depression in 50 randomized clinical trials, the placebo effect size was large and consistent across treatment modalities. Response and remission rates associated with placebo effect were comparable across modalities.
Meaning
The findings of this study suggest a placebo effect size benchmark may be used to interpret the findings of past and future clinical trials.
Abstract
Importance
The placebo effect in depression clinical trials is a substantial factor associated with failure to establish efficacy of novel and repurposed treatments. However, the magnitude of the placebo effect and whether it differs across treatment modalities in treatment-resistant depression (TRD) is unclear.
Objective
To examine the magnitude of the placebo effect in patients with TRD across different treatment modalities and its possible moderators.
Data Sources
Searches were conducted on MEDLINE, Web of Science, and PsychInfo from inception to June 21, 2021.
Study Selection
Randomized clinical trials (RCTs) were included if they recruited patients with TRD and randomized them to a placebo or sham arm and a pharmacotherapy, brain stimulation, or psychotherapy arm.
Data Extraction and Synthesis
Independent reviewers used standard forms for data extraction and quality assessment. Random-effects analyses and standard pairwise meta-analyses were performed.
Main Outcomes and Measures
The primary outcome was the Hedges g value for the reported depression scales. Secondary outcomes included moderators assessed via meta-regression and response and remission rates. Heterogeneity was assessed with the I2 test, and publication bias was evaluated using the Egger test and a funnel plot. Cochrane Risk of Bias Tool was used to estimate risks.
Results
Fifty RCTs were included involving various types of placebo or sham interventions with a total of 3228 participants (mean [SD] age, 45.8 [6.0] years; 1769 [54.8%] female). The pooled placebo effect size for all modalities was large (g = 1.05; 95% CI, 0.91-1.1); the placebo effect size in RCTs of specific treatment modalities did not significantly differ. Similarly, response and remission rates associated with placebo were comparable across modalities. Heterogeneity was large. Three variables were associated with a larger placebo effect size: open-label prospective treatment before double-blind placebo randomization (β = 0.35; 95% CI, 0.11 to 0.59; P = .004), later year of publication (β = 0.03; 95% CI, 0.003 to 0.05; P = .03), and industry-sponsored trials (β = 0.34; 95% CI, 0.09 to 0.58; P = .007). The number of failed interventions was associated with the probability a smaller placebo effect size (β = −0.12; 95% CI, −0.23 to −0.01, P = .03). The Egger test result was not significant for small studies’ effects.
Conclusions and Relevance
This analysis may provide a benchmark for past and future clinical RCTs that recruit patients with TRD standardizing an expected placebo effect size.
This systematic review and meta-analysis examines the placebo effect shown in randomized clinical trials of patients with treatment-resistant depression.
Introduction
Major depressive disorder (MDD) is a relapsing and remitting illness, and many patients do not respond to available treatments.1,2 The standard to evaluate a new intervention for MDD uses a randomized clinical trial (RCT) with placebo as the control design that can distinguish the benefit of an active treatment compared with the nonspecific benefit of the placebo response. The placebo response is defined as the therapeutic effect produced by a placebo or sham intervention that is not due to any inherent properties of the placebo. Several novel or repurposed treatments have not been able to establish efficacy in the context of large placebo effects.3 This phenomenon is a challenge for researchers; however, attention has begun to focus on trying to understand and quantify the magnitude of the placebo response.1,2
In RCTs of non–treatment-resistant depression (non-TRD) in patients with MDD,4,5,6,7,8,9,10,11,12,13,14 the placebo effect has been found to have a large magnitude and to be associated with several factors, although with some inconsistent findings. These factors have included later publication years, number of trial arms, multicenter setting, dosing schedule, increased length of the trial, sham device placement, the magnitude of active response, early score fluctuations, and inflation of baseline severity.4,5,6,7,8,9,10,11,12,13,14 A large meta-analysis evaluating the placebo effect in depression (256 RCTs; n = 26 324) found placebo-response rates of about 35% to 40%.13 However, this analysis included only RCTs of antidepressant drugs in patients without TRD.13 Treatment-resistant depression is commonly defined as the lack of response to 2 separate antidepressant trials of adequate dose and duration. Previous meta-analyses have suggested that TRD is associated with a smaller placebo effect than non-TRD (repetitive transcranial magnetic stimulation [rTMS] and escitalopram trials).15,16 Nonetheless, to our knowledge, no analysis has assessed the placebo effect in patients with TRD receiving other treatment modalities.
Patients with TRD often receive multiple treatment modalities and have lower rates of response and remission; thus, one would expect them to experience less benefit from the nonspecific effects of treatment. This lack of response highlights the importance of quantifying the placebo effect in TRD, how it may differ across treatment modalities, and what may contribute to it. An appreciation of the expected placebo effect of specific treatment modalities in TRD would provide a benchmark to inform interpretation of past and future RCTs. We therefore conducted a systematic review and meta-analysis to quantify the placebo effect across treatment modalities in TRD. We explored the methodological, demographic, and clinical variables that may contribute to placebo effect. This quantitative analysis is needed to interpret previous RCTs and emerging treatments as well as potentially identifying the beneficial aspect of the placebo effect.
Methods
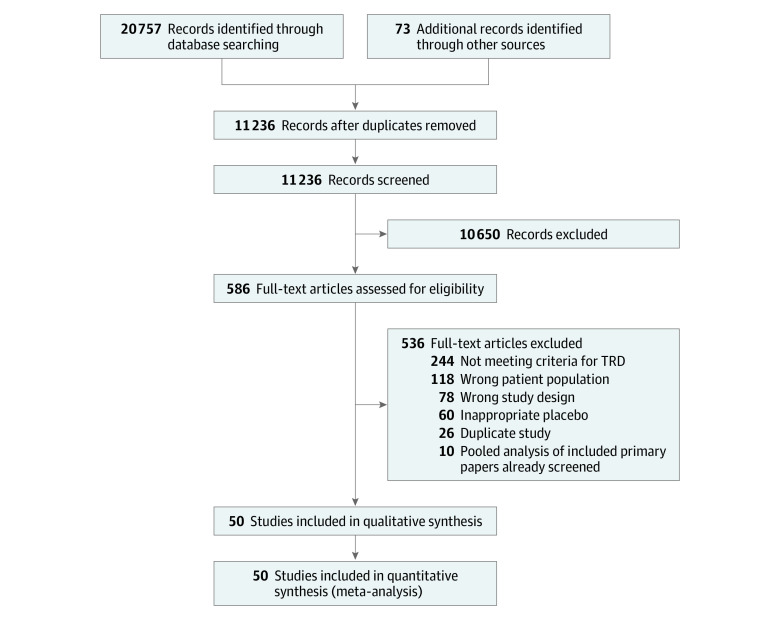
Searches were conducted on MEDLINE, Web of Science, and PsychInfo from inception to June 21, 2021 (eAppendix 2 in the Supplement). In addition, references from relevant reviews were searched.15,17,18,19,20,21 Figure 1 provides an overview of the number of studies screened and full texts reviewed. This study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline. The protocol was developed a priori and was published and registered22,23 (trial registration: PROSPERO Identifier: CRD42020190465).
Figure 1. Preferred Reporting Items for Systematic Reviews and Meta-analyses Flow Diagram for Inclusion of Studies.
Study designs were limited to parallel-arm, double-blind placebo-controlled RCTs or the first phase of randomized crossover trials that exclusively recruited patients with TRD and randomized them to at least 1 placebo arm, as specified in the protocol.22 Included active interventions were pharmacologic and somatic treatments (Maudsley Treatment Inventory) and psychotherapeutic interventions from the National Institute for Health and Care Excellence (NICE) guidelines.24,25 A placebo arm was defined as an inert pill placebo, parenteral placebo, sham device, or sham therapy that does not include any theoretical active property to induce the proposed therapeutic effect. Wait list or treatment as usual were not included as placebo interventions. Randomized clinical trials that used an open-label prospective antidepressant treatment phase to establish treatment resistance before randomization were included. Exclusion criteria were studies in which more than 15% of participants had bipolar depression, included patients with other psychiatric disorders, had sample sizes of less than 10 participants (5 per treatment arm), or used a noninert placebo arm.
Data were extracted by 3 of us (L.B.R., J.K., L.S.M.) and discrepancies were resolved by consensus. Data were confirmed by 1 of us (B.D.M.J.). Variables extracted included demographic characteristics, methodologic data, clinical/outcome data, and placebo-specific data. The demographic characteristics were age and sex. Methodological data included treatment strategy (augmentation vs monotherapy), multicenter setting, length of the trial (calculated as number of days for double-blind treatment), number of treatment arms, year of publication, trial design (ie, whether the trial used open-label prospective treatment to determine treatment resistance before double-blind randomization), and whether the trial was industry sponsored (defined as being sponsored by a pharmaceutical company or a medical device manufacturer). Randomized clinical trials funded by the US Department of Veterans Affairs; government agencies, such as the Canadian Institutes of Health Research or National Institute of Mental Health; nonprofit foundations, such as National Alliance for Research on Schizophrenia & Depression, or Brain & Behavior Research Foundation; or hospitals or universities were not considered to be industry sponsored. Clinical/outcome data comprised treatment modality, sample size, mean (SD) scores of depression rating scales at baseline and end point in both the placebo and active groups, number of responders/remitters (as defined by the individual studies), number of failed antidepressant trials, past depressive episodes, and length of the current episode. Placebo-specific data included route (eg, oral vs parenteral) of administration and number of days receiving placebo. This variable was calculated by determining how many days during the trial period participants would receive placebo or sham intervention.
If the mean (SD) scores of the depression rating scales were not reported in the study, the corresponding author was contacted by email. If no reply was obtained, we extracted the data from the study graphs. Moreover, when only the SD of the change was reported, we imputed the data as recommended by the Cochrane group.26 For studies that included an open-label prospective (run-in) treatment phase, effect size was calculated from the start of the double-blind randomized phase.
The quality of the studies was assessed independently by 2 of us (B.D.M.J. and C.R.W.) and recorded using the Cochrane Risk of Bias tool.27 Thus, 5 bias domains were assessed: selection (randomization and allocation concealment), performance (blinding of participants and personnel), attrition (incomplete outcome data), detection (blinding of outcome assessment), and reporting (selective outcome reporting). For a judgment of the overall risk of bias, we followed the Cochrane recommendation: low risk of bias (low risk for all domains or some concerns of bias for 1 domain), unclear risk of bias (2 or 3 domains were rated as presenting some cause for concern), and high risk of bias (>3 domains with some bias concerns and/or ≥1domain with high risk of bias).
The analyses were performed using Stata, version 17 (StataCorp LLC) software. Considering that most studies have small samples, we used the Hedges g to estimate the effect size of the placebo response. The effect size was computed based on the baseline and end point sample sizes and means (SDs) of the primary depression score scales of the placebo group of each study. A random-effects model (restricted maximum-likelihood method instead of fixed-effect models was used considering that study heterogeneity would be high). The model provides wider 95% CIs compared with fixed-effects models, thereby providing a more conservative estimation of summary effect size.28 The model assumes that the effect sizes of the studies are different from each other and represent a random sample from a larger population of studies.29 The components of the random-effects model applied in this meta-analysis are described in eAppendix 1 in the Supplement. Heterogeneity among studies was assessed with I2 analysis and was considered high when presenting a value greater than or equal to 50% as suggested elsewhere.30 Small studies effects were assessed using the Begg modified funnel plot, the Duval and Tweedie trim-and-fill procedure, and the Egger regression intercept. The Egger test was considered significant for small studies’ effects when presenting findings significant at P < .05.
Meta-regression and subgroup analyses were applied to explore potential moderators of the placebo effect. To increase overall sample size, treatment modalities were pooled for the meta-regression. Meta-regressions were conducted using only 1 variable at a time. Subgroup analysis was conducted to compare the effect size of the placebo response among treatment modalities. Values were considered statistically significant when presenting findings at P < .05 for both meta-regression and subgroup analysis. The mean of the percentage of responders and remitters for each treatment modality was also assessed. In addition, a sensitivity analysis was conducted looking at low risk of bias.
Results
Our search yielded 11 236 studies that were screened; full texts of 605 articles were reviewed and 50 RCTs (N = 3228) were included.31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80 the mean (SD) age of the participants was 45.8 (6.0) years, and the mean (SD) proportion of women was 54.8% (20.7%) (Figure 1 and Table 1). Additional clinical characteristics are described in eTable 3 in the Supplement. Sixteen RCTs were classified to have a low risk of bias; 19, an unclear risk of bias, and 16, a high risk (eTable 1 in the Supplement). No psychotherapy RCTs met our eligibility criteria. Placebo or sham interventions were categorized as pill placebo, liquid placebo (trials in this analysis used ayahuasca, a South American psychoactive brew), parenteral placebo, sham rTMS, sham transcranial direct current stimulation (tDCS), or sham invasive brain stimulation.
Table 1. Demographic, Clinical, and Methodological Characteristics of Included Studies in the Systematic Review and Meta-analysis.
| Source | Demographic characteristics | Methodologic characteristics | Placebo characteristics | Overall risk of bias | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | Women No. (%) | Age, mean (SD), y | Depression score | Design | Augmentation | Treatment | Placebo route | Total days receiving placebo | Times placebo was measured | ||
| Bakim et al,31 2012 | 12 | 11 (91.7) | 44.4 (10.2) | HDRS-17 | SC, treatment not prospective, 3 treatment arms, 42 d | Yes | rTMS | Noninvasive brain stimulation | 30 | 6 | Unclear |
| Barbee et al,32 2011 | 48 | 33 (68.8) | 45.8 (11) | MADRS | MC, prospective treatment, 2 arms, 70 d | Yes | Lamotrigine | Oral | 70 | 6 | Low |
| Bauer et al,75 2019 | 441 | 302 (68.5) | 46.4 (12.1) | MADRS | MC, prospective treatment, 2 arms, 168 d | Yes | Brexiprazol | Oral | 168 | 9 | High |
| Bennabi et al,74 2015 | 12 | 5 (45.5) | 59.9 (15.4) | HDRS-21 | SC, treatment not prospective, 2 arms, 35 d | Yes | tDCS | Noninvasive brain stimulation | 5 | 3 | Unclear |
| Berman et al,73 2007 | 176 | 113 (64.2) | 44.2 (10.9) | MADRS | MC, prospective treatment, 2 arms, 42 d | Yes | Aripiprazole | Oral | 42 | 6 | Low |
| Berman et al,33 2009 | 172 | 117 (68.0) | 45.6 (11.3) | MADRS | MC, prospective treatment, 2 arms, 42 d | Yes | Aripiprazole | Oral | 42 | 6 | Unclear |
| Blumberger et al,35 2012 | 22 | 14 (70.0) | 45.8 (13.4) | HDRS-17 | SC, treatment not prospective, 3 arms, 42 d | Yes | rTMS | Noninvasive brain stimulation | 30 | 6 | Low |
| Blumberger et al,34 2012 | 11 | 10 (90.9) | 49.7 (9.4) | HDRS-17 | SC, treatment not prospective, 2 arms, 21 d | Yes | tDCS | Noninvasive brain stimulation | 15 | 2 | Low |
| Blumberger et al,36 2016 | 41 | 24 (59.0) | 48.1 (12.0) | HAMD-17 | SC, treatment not prospective, 3 arms, 42 d | Yes | rTMS | Noninvasive brain stimulation | 30 | 2 | Low |
| Boutros et al,37 2002 | 9 | 1 (11.0) | 52 (7) | HDRS-25 | SC, treatment not prospective, 2 arms, 14 d | Yes | rTMS | Noninvasive brain stimulation | 10 | 5 | Low |
| Chen et al,38 2013 | 10 | 4 (40.0) | 47.3 (3.5) | HAMD-17 | SC, treatment not prospective, 2 arms, 14 d | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | High |
| Coenen et al,72 2019 | 8 | 3 (37.5) | 49.9 (12) | MADRS | SC, treatment not prospective, 2 arms, 56 d | Yes | DBS | Invasive brain stimulation | 1 | 4 | High |
| Concerto et al,39 2015 | 15 | 7 (46.7) | 53 (6.7) | HDRS | SC, treatment not prospective, 2 arms, 28 d | Yes | rTMS | Noninvasive brain stimulation | 20 | 1 | High |
| Dougherty et al,40 2015 | 14 | 5 (35.7) | 48.9 (8.9) | MADRS | MC, treatment not prospective, 2 arms, 112 d | Yes | DBS | Invasive brain stimulation | 1 | 6 | Low |
| Fava et al,77 2015 | 85 | 56 (65.9) | 45.9 (10.6) | MADRS | MC, prospective treatment, 2 arms, 42 d | Yes | nAChR antagonist | Oral | 42 | 5 | High |
| Fava et al,41 2018 | 81 | 61 (75.3) | 45.2 (10.2) | MADRS | MC, prospective treatment, 3 arms, 56 d | Yes | Cariprazine | Oral | 56 | 5 | Low |
| Fitzgerald et al,42 2012 | 20 | 8 (40.0) | 44.9 (15.7) | HDRS-17 | SC, treatment not prospective, 3 arms, 21 d | Yes | rTMS | Noninvasive brain stimulation | 15 | 1 | High |
| Garcia-Toro et al,43 2001 | 18 | 8 (44.4) | 50 (11) | HDRS-21 | SC, treatment not prospective, 2 arms, 14 | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Garcia-Toro et al,76 2006 | 10 | 7 (70.0) | 47.2 (11.8) | HDRS-21 | SC, treatment not prospective, 3 arms, 14 | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Heresco-Levy et al,44 2013 | 13 | 8 (61.5) | 53 (10.2) | HDRS-21 | MC, treatment not prospective, 2 arms, 42 d | Yes | D-cycloserine | Oral | 42 | 3 | Low |
| Hobart et al,45 2018 | 206 | 144 (71.3) | 41.8 (11.7) | MADRS | MC, prospective treatment, 3 arms, 42 d | Yes | Brexiprazol | Oral | 42 | Low | |
| Hobart et al,71 2018 | 202 | 149 (72.3) | 42.7 (12.5) | MADRS | MC, prospective treatment, 2 arms, 42 | Yes | Brexiprazol | Oral | 42 | 6 | Unclear |
| Holtzheimer et al.46 2004 | 8 | 3 (42.9) | 45.4 (4.9) | HDRS-17 | SC, treatment not prospective, 2 arms 14 d | No | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Holtzheimer et al,70 2017 | 30 | 17 (57.0) | 48.7 (0.6) | MADRS | MC, treatment not prospective, 2 arms, 183 d | Yes | DBS | Invasive brain stimulation | 1 | 2 | Low |
| Husain et al,69 2017 | 20 | 11 (55.0) | 34.9 (2.5) | HDRS-17 | MC, treatment not prospective, 2 arms, 84 d | Yes | MiNocycline | Oral | 84 | 3 | Unclear |
| Ionescu et al,68 2019 | 13 | 3 (23.0) | 45.3 (11.7) | HAMD-28 | SC, treatment not prospective, 2 arms, 21 d | Yes | Ketamine | Parenteral | 6 | 6 | Low |
| Kamijima et al,66 2013 | 195 | 80 (41.0) | 38.7 (9.2) | MADRS | MC, prospective treatment, 3 arms, 42 d | Yes | Aripiprazole | Oral | 42 | 6 | Unclear |
| Kamijima et al,67 2018 | 203 | 72 (35.5) | 39.5 (11.8) | MADRS | MC, prospective treatment, 2 arms, 42 d | Yes | Aripiprazole | Oral | 42 | 6 | Low |
| Kauffmann et al,47 2004 | 5 | 4 (91.7) | 51.7 (17.2) | HDRS-21 | SC, treatment not prospective, 2 arms, 14 d | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Li et al,48 2014 | 15 | 11 (73.3) | 46.9 (NA) | HDRS-17 | SC, treatment not prospective, 4 arms, 14 d | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Marcus et al,49 2008 | 190 | 128 (67.4) | 44.4 (10.7) | MADRS | 42 | Yes | Aripiprazole | Oral | 42 | 6 | High |
| McAllister-Williams et al,50 2016 | 82 | 52 (63.0) | 45.2 (10.4) | MADRS | MC, treatment not prospective, 2 arms, 35 d | Yes | Metyrapone | Oral | 21 | 2 | Low |
| Chen et al,65 2019 | 16 | 13 (81.3) | 49.9 (8.1) | MADRS | SC, treatment not prospective, 3 arms, 3 d | Yes | Ketamine | Parenteral | 1 | 1 | High |
| Nierenberg et al,64 2003 | 17 | 7 (41.2) | 39.7 (11.9) | HSRD-17 | 42 | Yes | Lithium | Oral | 42 | 3 | Unclear |
| Palhano-Fontes et al.63 2019 | 15 | 10 (66.7) | 44.2 (12.0) | HDRS-17 | SC, treatment not prospective, 2 arms, 7 d | No | Ayahuasca | Oral | 1 | 3 | Unclear |
| Pallanti et al,51 2010 | 20 | 12 (60.0) | 47.9 (9.1) | HDRS-17 | SC, treatment not prospective, 3 arms, 21 d | Yes | rTMS | Noninvasive brain stimulation | 15 | 3 | High |
| Santos et al,52 2008 | 17 | 11 (65.0) | 29 (NA) | MADRS | SC, treatment not prospective, 2 arms, 56 d | Yes | Lamotrigine | Oral | 56 | 4 | High |
| Shelton et al,53 2001 | 10 | NA | NA | MADRS | SC, prospective treatment, 3 arms, 56 d | Yes | Olanzapine | Oral | 56 | 8 | High |
| Su et al,62 2017 | 24 | 15 (62.5) | 48.6 (8.2) | HDRS-17 | SC, treatment not prospective, 3 arms, 5 d | Yes | Ketamine | Parenteral | 1 | 8 | Unclear |
| Thase et al,54 2015 | 191 | 137 (71.6) | 45.2 (11.3) | MADRS | MC, prospective treatment, 2 arms, 42 d | Yes | Brexpiprazole | Oral | 42 | 6 | High |
| Thase et al,55 2015 | 221 | 146 (66.1) | 46.6 (11) | MADRS | MC, prospective treatment, 3 arms, 42 d | Yes | Brexpiprazole | Oral | 42 | 6 | High |
| Theleritis et al,56 2017 | 20 | 10 (50) | 38 (9.9) | HDRS-17 | SC, treatment not prospective, 4 arms, 21 d | No | rTMS | Noninvasive brain stimulation | 15 | 3 | Unclear |
| Theleritis et al,56 2017 | 24 | 10 (31.6) | 39.4 (8.9) | HDRS-17 | SC, treatment not prospective, 4 arms, 21 d | No | rTMS | Noninvasive brain stimulation | 15 | 3 | Unclear |
| Triggs et al,57 2010 (left sham) | 7 | 2 (28.6) | 41.9 (14.1) | HAMD-24 | SC, treatment not prospective, 4 arms, 14 d | Yes | rTMS | Noninvasive brain stimulation | 10 | 2 | Unclear |
| Triggs et al,57 2010 (right sham) | 7 | 4 (57.1) | 46.6 (20.2) | HAMD-24 | SC, treatment not prospective, 4 arms, 14 d | Yes | rTMS | Invasive Brain Stimulation | 10 | 2 | Unclear |
| Yesavage et al,61 2018 | 83 | 18 (21.7) | 54.8 (12.6) | HSRD-24 | MC, treatment not prospective, 2 arms, 42 d | Yes | rTMS | Noninvasive brain stimulation | 30 | 3 | Low |
| Zheng et al,60 2010 | 15 | 5 (33.3) | 26.7 (4.3) | HAMD-17 | SC, treatment not prospective, 2 arms, 28 d | Yes | rTMS | Noninvasive brain stimulation | 20 | 1 | High |
| Palm et al,59 2012 | 11 | 8 (72) | 58 (12) | HAMD-24 | SC, treatment not prospective, 2 arms, 14 d | Yes | tCDS | Noninvasive brain stimulation | 10 | 2 | Low |
| van Eijndhoven et al,58 2020 | 16 | 13 (81) | 49.7 (11) | HDRS-17 | SC, treatment not prospective, 2 arms, 20 | Yes | rTMS | Noninvasive brain stimulation | 35 | 5 | Unclear |
| Rush et al,79 2005 | 110 | 73 (66) | 45.9 (9) | HAMD-24 | MC, treatment not prospective, 2 arms, 70 d | Yes | Vagus nerve stimulation | Invasive brain stimulation | 70 | 7 | High |
| Baeken et al,80 2013 | 11 | 5 (45) | 47.3 (13.6) | HDRS | SC, treatment not prospective, 2 arms, 7 d | No | rTMS | Noninvasive brain stimulation | 4 | 1 | High |
| Padberg et al,78 1999 | 6 | 4 (66.6) | 43.3 (11.6) | HDRS | SC, treatment not prospective, 3 arms, 5 d | Yes | rTMS | Noninvasive brain stimulation | 5 | 2 | Unclear |
Abbreviations: DBS, deep brain stimulation; HDRS, Hamilton Depression Rating Scale; MADRS, Montgomery-Åsberg Depression Rating Scale; MC, multicenter; SC, single-center; rTMS, repetitive transcranial magnetic stimulation; tDCS, transcranial direct current stimulation.
Studies were pooled based on treatment modality to maintain sufficient similarity in the pairwise analysis. The effect sizes for each treatment modality were pill placebo, g = 1.14 (95% CI, 0.99 to 1.30; I2 = 80.25%); parenteral placebo, g = 1.33 (95% CI, 0.63 to 2.04; I2 = 62.28%); liquid placebo, g = 0.45 (95% CI, −0.26 to 1.15; I2 = 0%); rTMS, g = 0.89 (95% CI, 0.63 to 1.15; I2 = 62.14%); tDCS, g = 1.32 (95% CI, 0.53 to 2.11; I2 = 52.57%); and invasive brain stimulation, g = 0.86 (95% CI, 0.58 to 1.14; I2 = 15.48%). The pooled effect size for all treatment modalities was g = 1.05 (95% CI, 0.91 to 1.18) with an overall I2 = 76.19% (Figure 2 and Figure 3).
Figure 2. Main Placebo Effect Size of Included Pill and tDCS Studies.
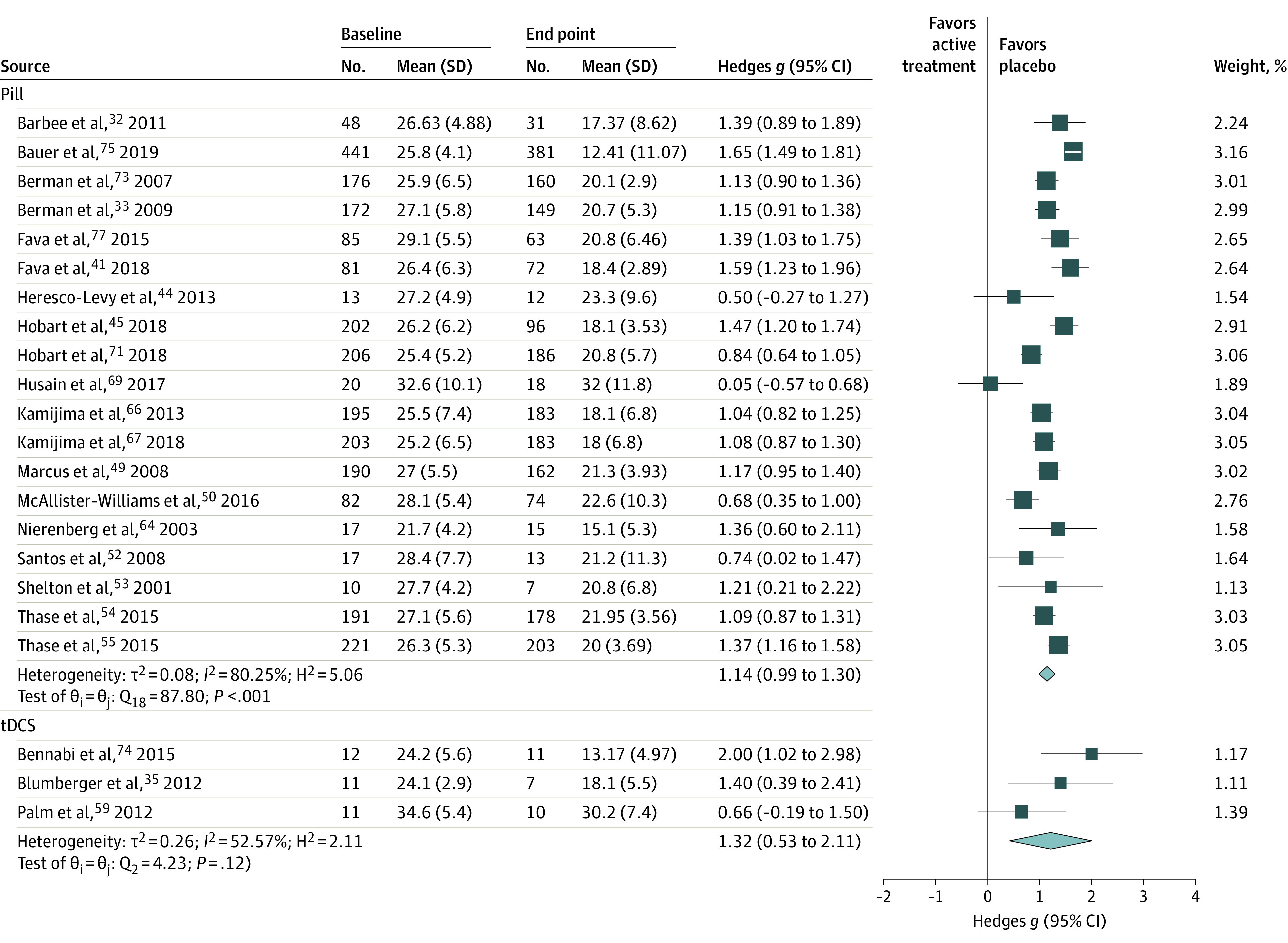
Studies were grouped by treatment modality and pooled. tDCS indicates transcranial direct current stimulation.
Figure 3. Main Placebo Effect Size of Included rTMS, Invasive Brain Stimulation, Parenteral, Ayahuasca, and Overall Studies.
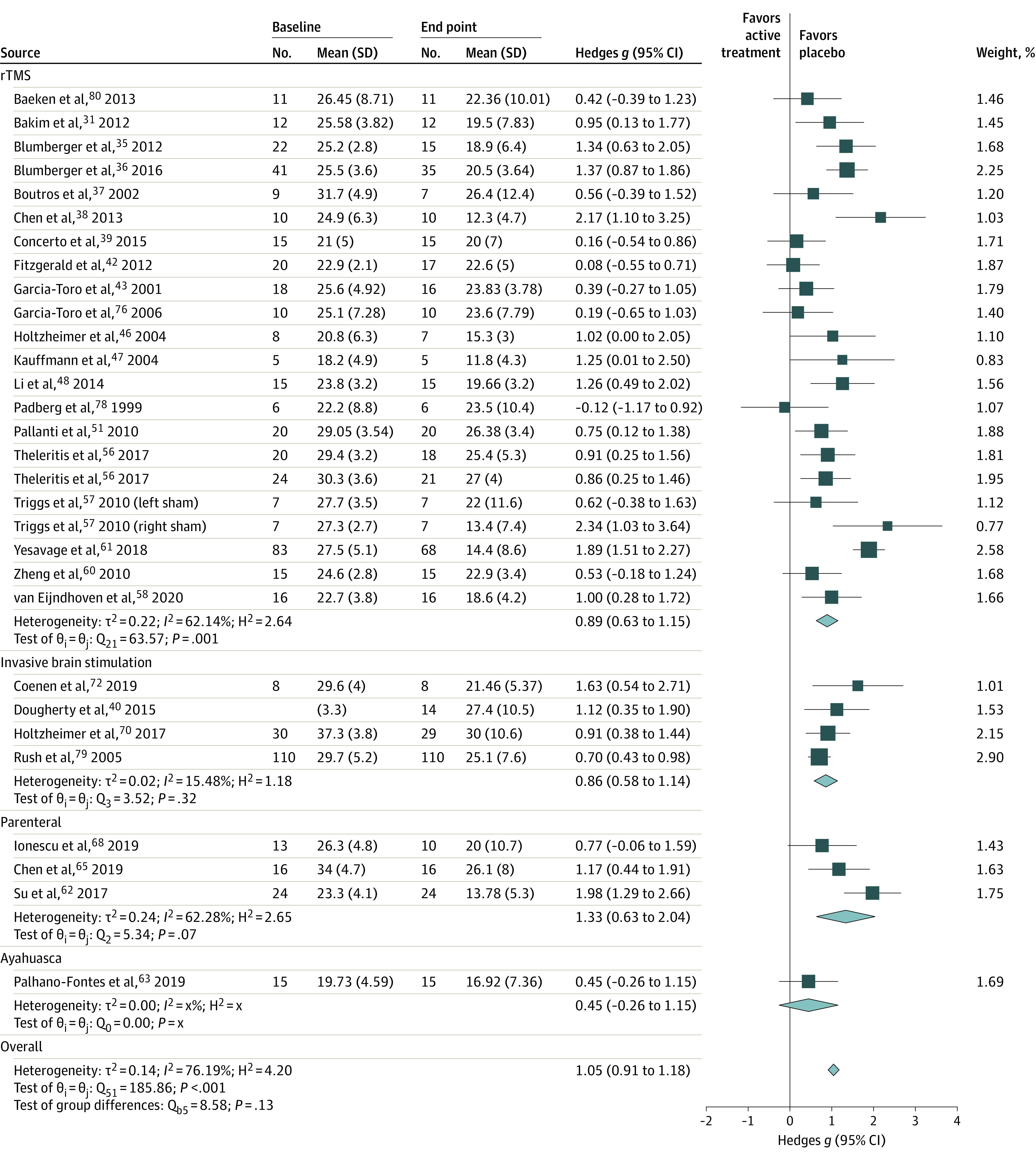
Studies were grouped by treatment modality and pooled. Ayahuasca is a South American psychoactive brew. rTMS indicates repetitive transcranial magnetic stimulation.
The funnel plot of the primary outcome showed a symmetrical distribution among the included studies (eFigure 1 in the Supplement). The Egger test corroborated this finding (z = 1.18; P = .23). We also applied the trim-and-fill method to deeply investigate any asymmetry of the funnel plot (eFigure 2 in the Supplement) and its analysis showed the presence of 5 unpublished studies. Considering these studies in the pooled analyses, the overall effect size was adjusted to g = 1.14 (95% CI, 1.00 to 1.29). The overall placebo effect size remained large when considering only studies with a low risk of bias (g = 1.18; 95% CI, 0.98 to 1.38) (eFigure 3 in the Supplement).
For a subset of RCTs that reported response rates (n = 42) and remission rates (n = 25), the pooled mean (SD) response rate was 21.2% (14.6%) and the pooled remission rate was 13.0% (9.05%). Modality-specific response and remission rates are described in Table 2.
Table 2. Modality-Specific Response and Remission Rates.
| Modality | Response | Remission | ||
|---|---|---|---|---|
| No. of studies | % Rate (SD) | No. of studies | % Rate (SD) | |
| Pill | 17 | 21.9 (8.2) | 13 | 16.6 (6.1) |
| tDCS | 2 | 18.2 (12.8) | 2 | 4.5 (6.4) |
| rTMS | 16 | 20.8 (21.4) | 7 | 11.2 (13.2) |
| Invasive brain stimulation | 4 | 19.1 (11.0) | 1 | 7.0 (0) |
| Parenteral | 2 | 22.8 (14.5) | 1 | 8 (0) |
| Liquid (Ayahuasca) | 1 | 27 (0) | 1 | 7 (0) |
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; tDCS, transcranial direct current stimulation.
Pooled meta-regression analysis revealed that industry-sponsored studies (β = 0.34; 95% CI, 0.09 to 0.59; P = .007), year of publication (β = 0.03; 95% CI, 0.003 to 0.05; P = .03), and studies that used an open-label prospective treatment phase before double-blind randomization (β = 0.35; 95% CI, 0.11 to 0.59; P = .004) had a significantly higher placebo effect. The number of failed interventions during the current episode was associated with a smaller placebo effect (β = −0.12; 95% CI, −0.23 to −0.01; P = .03) (Table 3).
Table 3. Meta-regression for Placebo Effect.
| Variable | No. | Coefficient β (95% CI) | P value |
|---|---|---|---|
| Publication year | 52 | 0.03 (0.003 to 0.05) | .03 |
| No. of treatment arms | 52 | 0.02 (−0.20 to 0.23) | .89 |
| Prospective treatment | 52 | 0.35 (0.11 to 0.59) | .004 |
| Industry sponsored | 52 | 0.34 (0.09 to 0.58) | .007 |
| Baseline depression severity | 52 | −0.006 (−0.04 to 0.03) | .74 |
| Total study length, d | 52 | 0.002 (−0.001 to 0.006) | .17 |
| Total days of placebo | 52 | 0.002 (−0.001 to 0.005) | .22 |
| Multicenter | 52 | 0.21 (−0.05 to 0.46) | .11 |
| Augmentation treatment | 52 | 0.35 (−0.13 to 0.83) | .15 |
| Age | 51 | 0.02 (−0.001 to 0.05) | .06 |
| Female % | 52 | 0.001 (−0.006 to 0.009) | .72 |
| Length of current episode | 32 | 0.0003 (−0.004 to 0.005) | .90 |
| Past episodes | 24 | −0.03 (−0.15 to 0.1) | .68 |
| Effect size of active arm | 51 | 0.08 (−0.08 to 0.25) | .31 |
| No. of failed trials | 17 | −0.12 (−0.23 to −0.01) | .03 |
Subgroup analysis found no significant differences in placebo effect among treatment modalities (eTable 2 in the Supplement). Owing to the high heterogeneity between studies, no other subgroup analysis was completed.
Discussion
In this meta-analysis, we report on the placebo effect in TRD across multiple treatment modalities. We synthesized data from 50 RCTs including 3228 participants who received either pill placebo, parenteral placebo, liquid placebo, sham rTMS, sham tDCS, or sham invasive brain stimulation. The combined placebo effect size for all interventions was large (g = 1.05) and the placebo effect sizes for each treatment modality did not significantly differ. This finding is consistent with prior analyses that have shown that placebo in MDD (non-TRD) has a large effect size, seen with RCTs of antidepressants (Cohen d = 1.69), rTMS (g = 0.8), or tDCS (g = 1.0914,15,81). Studies in other psychiatric populations have also shown a high placebo effect size, such as negative symptoms in schizophrenia (Cohen d = 2.91) and response rate (39.2%) in bipolar depression.82,83 Although all of these analyses reported a large effect size, the effect size found in RCTs involving patients with TRD is numerically smaller for pill placebo and numerically larger for sham stimulation than the effect sizes reported in these RCTs involving patients without TRD.14,15,81
In our secondary analysis, we assessed response and remission rates as reported in the individual RCTs. The pooled response rate across all treatment modalities was 23.5% and the remission rate was 15.5%. These low rates did not differ significantly across treatment modalities. A large meta-analysis in patients without TRD reported a response rate of placebo to be 35% to 40%, which is numerically higher than our rate of 21.2%.13 Another meta-analysis reported a remission rate of 22%, which is numerically higher than our finding of 13.0%.84 Earlier studies of placebo effect that have included participants with TRD have found that TRD was associated with a lower placebo response, which may explain why our response and remission rates appear to be lower than in studies involving patients without TRD.15
Our meta-regression found that RCTs that used an open-label prospective treatment phase before double-blind randomization, a more recent year of publication, and those that were industry sponsored had a larger placebo effect. A meta-analysis of placebo response in negative symptoms of schizophrenia also reported that placebo response was higher in industry-sponsored clinical trials (Cohen d = 6.72) compared with academic-funded trials (Cohen d = 1.01).82 Only pill placebo RCTs used an open-label prospective treatment phase and only 1 small rTMS RCT was industry sponsored. Previous research has also suggested that, in populations without treatment-resistant MDD, a higher placebo response was associated with more recent year of publication.85 Other studies have suggested that finding may reflect methodological changes, such as an increased number of multicenter studies.86 Placebo effect has been shown to be associated with expectancy of active treatment and increased activity in reward circuitry.6,87 Industry-sponsored RCTs are often investigating novel agents, which may lead to increased expectations of efficacy by participants, although we are unable to confirm this possibility with available data. Another potential contributing factor to the higher placebo effect in the prospective open-label treatment trials is that of delayed antidepressant response. In these studies, patients are continuing the recently started antidepressant from the prospective open-label treatment phase in combination with placebo or another active agent, which may be contributing to a delayed response. The number of failed trials was also significantly associated with a smaller placebo effect. Although only 17 of 50 studies reported on this variable, this finding is consistent with previous literature showing that a higher level of treatment resistance is associated with a smaller placebo effect.15
Our analysis did not find that the placebo effect was significantly different across treatment modalities. This finding is consistent with trials comparing rTMS with escitalopram.16 The large placebo effect in TRD does not change substantially based on modality. This lack of effect supports the notion that the nonspecific factors contributing to the placebo effect in TRD are prevalent in any clinical trial. Our findings suggest that the invasiveness of the intervention (eg, pill vs invasive brain stimulation) does not substantially affect the placebo effect in TRD. Given the consistency of the placebo effect across treatment modalities in TRD, neurobiological and common psychological factors need to be further investigated.
Our primary finding that the placebo effect in TRD is large (g = 1.05) and that it is consistent across treatment modalities may help to interpret the results of past and future RCTs. Researchers who conduct clinical trials may now compare their results with a benchmark for expected placebo effect in TRD. For instance, a placebo-controlled RCT that reports negative findings but had a placebo effect size greater than g = 1.05 could be interpreted as a false-negative; conversely, an RCT that reports positive findings with a placebo effect size less than g = 1.05 could be a false-positive. Our results also provide a context for noninferiority trials that do not use a placebo or sham control intervention. Treatments that differentiate from this benchmark (g = 1.05) by a predetermined margin would be expected to be superior to placebo.
Limitations
This study has limitations. Although we were able to include several different treatment modalities in our analysis, no psychotherapy RCTs met our eligibility criteria. Psychotherapy trials in TRD were either open-label, single arm, had an active comparator, or used treatment as usual or a wait-list condition. There were also no RCTs of electroconvulsive therapy or magnetic seizure therapy that used sham versions of these procedures in a TRD population. Another limitation is the definition of TRD. Although we used the most common definition, there is no standard TRD definition. A large proportion of studies that were excluded reported including patients with TRD even though they defined TRD based on only one failed antidepressant trial. For this reason, we were unable to include 2 large industry-sponsored trials for rTMS.88,89 Future research would benefit from an improved and consistent definition of TRD. Current definitions of TRD are homogeneous and do not consistently account for a number of important factors, such as specific treatments failed (eg, psychotherapy vs brain stimulation vs selective serotonin reuptake inhibitor vs serotonin and norepinephrine reuptake inhibitor) and psychosocial factors. Although we aimed to compare the placebo effect across treatment modalities, our results are limited by the absence of direct comparison of 2 placebo modalities (ie, sham brain stimulation vs pill placebo).
Conclusions
The present systematic review and meta-analysis compared the placebo effect in TRD across different treatment modalities. Our main finding was that the placebo effect in TRD appears to be large and consistent across treatment modalities. The effect size in the studies included in our analysis (g = 1.05) may serve as a benchmark to assess placebo effect in future TRD RCTs. Factors that increase the placebo effect appear to include using an open-label, prospective treatment phase and industry sponsorship. To better understand the placebo effect, the following improvements are needed: more consistent reporting of data, an agreement on a standard definition of TRD and its possible subgroups, and further assessment and reporting of participants’ expectations and experiences within a clinical trial.
eTable 1. Risk of Bias
eTable 2. Subgroup Analysis of Treatment Modalities
eTable 3. Clinical Characteristics
eAppendix 1. Components of the Randomized Effect Model Used for Our Analyses
eAppendix 2. Search Strategy
eFigure 1. Funnel Plot of Included Studies
eFigure 2. Trim and Fill Funnel Plot
eFigure 3. Forest Plot of Main Placebo Effect Size Grouped by Risk of Bias
References
- 1.Rush AJ, Warden D, Wisniewski SR, et al. STAR*D: revising conventional wisdom. CNS Drugs. 2009;23(8):627-647. [DOI] [PubMed] [Google Scholar]
- 2.Johnston KM, Powell LC, Anderson IM, Szabo S, Cline S. The burden of treatment-resistant depression: a systematic review of the economic and quality of life literature. J Affect Disord. 2019;242:195-210. doi: 10.1016/j.jad.2018.06.045 [DOI] [PubMed] [Google Scholar]
- 3.Fountoulakis KN, McIntyre RS, Carvalho AF. From randomized controlled trials of antidepressant drugs to the meta-analytic synthesis of evidence: methodological aspects lead to discrepant findings. Curr Neuropharmacol. 2015;13(5):605-615. doi: 10.2174/1570159X13666150630174343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Walsh BT, Seidman SN, Sysko R, Gould M. Placebo response in studies of major depression: variable, substantial, and growing. JAMA. 2002;287(14):1840-1847. doi: 10.1001/jama.287.14.1840 [DOI] [PubMed] [Google Scholar]
- 5.Khan A, Bhat A, Kolts R, Thase ME, Brown W. Why has the antidepressant-placebo difference in antidepressant clinical trials diminished over the past three decades? CNS Neurosci Ther. 2010;16(4):217-226. doi: 10.1111/j.1755-5949.2010.00151.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sinyor M, Levitt AJ, Cheung AH, et al. Does inclusion of a placebo arm influence response to active antidepressant treatment in randomized controlled trials? Results from pooled and meta-analyses. J Clin Psychiatry. 2010;71(3):270-279. doi: 10.4088/JCP.08r04516blu [DOI] [PubMed] [Google Scholar]
- 7.Iovieno N, Papakostas GI. Does the presence of an open-label antidepressant treatment period influence study outcome in clinical trials examining augmentation/combination strategies in treatment partial responders/nonresponders with major depressive disorder? J Clin Psychiatry. 2012;73(5):676-683. doi: 10.4088/JCP.11r06978 [DOI] [PubMed] [Google Scholar]
- 8.Khan A, Khan SR, Walens G, Kolts R, Giller EL. Frequency of positive studies among fixed and flexible dose antidepressant clinical trials: an analysis of the food and drug administration summary basis of approval reports. Neuropsychopharmacology. 2003;28(3):552-557. doi: 10.1038/sj.npp.1300059 [DOI] [PubMed] [Google Scholar]
- 9.Landin R, DeBrota DJ, DeVries TA, Potter WZ, Demitrack MA. The impact of restrictive entry criterion during the placebo lead-in period. Biometrics. 2000;56(1):271-278. doi: 10.1111/j.0006-341X.2000.00271.x [DOI] [PubMed] [Google Scholar]
- 10.Rutherford BR, Cooper TM, Persaud A, Brown PJ, Sneed JR, Roose SP. Less is more in antidepressant clinical trials: a meta-analysis of the effect of visit frequency on treatment response and dropout. J Clin Psychiatry. 2013;74(7):703-715. doi: 10.4088/JCP.12r08267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Whitlock ME, Woodward PW, Alexander RC. Is high placebo response really a problem in depression trials? a critical re-analysis of depression studies. Innov Clin Neurosci. 2019;16(7-08):12-17. [PMC free article] [PubMed] [Google Scholar]
- 12.Targum SD, Cameron BR, Ferreira L, MacDonald ID. Early score fluctuation and placebo response in a study of major depressive disorder. J Psychiatr Res. 2020;121:118-125. doi: 10.1016/j.jpsychires.2019.11.014 [DOI] [PubMed] [Google Scholar]
- 13.Furukawa TA, Cipriani A, Atkinson LZ, et al. Placebo response rates in antidepressant trials: a systematic review of published and unpublished double-blind randomised controlled studies. Lancet Psychiatry. 2016;3(11):1059-1066. doi: 10.1016/S2215-0366(16)30307-8 [DOI] [PubMed] [Google Scholar]
- 14.De Smet S, Nikolin S, Moffa A, et al. Determinants of sham response in tDCS depression trials: a systematic review and meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry. 2021;109:110261. doi: 10.1016/j.pnpbp.2021.110261 [DOI] [PubMed] [Google Scholar]
- 15.Razza LB, Moffa AH, Moreno ML, et al. A systematic review and meta-analysis on placebo response to repetitive transcranial magnetic stimulation for depression trials. Prog Neuropsychopharmacol Biol Psychiatry. 2018;81:105-113. doi: 10.1016/j.pnpbp.2017.10.016 [DOI] [PubMed] [Google Scholar]
- 16.Brunoni AR, Lopes M, Kaptchuk TJ, Fregni F. Placebo response of non-pharmacological and pharmacological trials in major depression: a systematic review and meta-analysis. PLoS One. 2009;4(3):e4824. doi: 10.1371/journal.pone.0004824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li JM, Zhang Y, Su WJ, et al. Cognitive behavioral therapy for treatment-resistant depression: a systematic review and meta-analysis. Psychiatry Res. 2018;268:243-250. doi: 10.1016/j.psychres.2018.07.020 [DOI] [PubMed] [Google Scholar]
- 18.Marcantoni WS, Akoumba BS, Wassef M, et al. A systematic review and meta-analysis of the efficacy of intravenous ketamine infusion for treatment resistant depression: January 2009-January 2019. J Affect Disord. 2020;277:831-841. doi: 10.1016/j.jad.2020.09.007 [DOI] [PubMed] [Google Scholar]
- 19.Li H, Cui L, Li J, Liu Y, Chen Y. Comparative efficacy and acceptability of neuromodulation procedures in the treatment of treatment-resistant depression: a network meta-analysis of randomized controlled trials. J Affect Disord. 2021;287:115-124. doi: 10.1016/j.jad.2021.03.019 [DOI] [PubMed] [Google Scholar]
- 20.McIntyre RS, Carvalho IP, Lui LMW, et al. The effect of intravenous, intranasal, and oral ketamine in mood disorders: a meta-analysis. J Affect Disord. 2020;276:576-584. doi: 10.1016/j.jad.2020.06.050 [DOI] [PubMed] [Google Scholar]
- 21.Carter B, Strawbridge R, Husain MI, et al. Relative effectiveness of augmentation treatments for treatment-resistant depression: a systematic review and network meta-analysis. Int Rev Psychiatry. 2020;32(5-6):477-490. doi: 10.1080/09540261.2020.1765748 [DOI] [PubMed] [Google Scholar]
- 22.Jones BDM, Weissman CR, Razza LB, Husain MI, Brunoni AR, Daskalakis ZJ. Protocol for a systematic review and meta-analysis of the placebo response in treatment-resistant depression: comparison of multiple treatment modalities. BMJ Open. 2021;11(2):e041349. doi: 10.1136/bmjopen-2020-041349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shamseer L, Moher D, Clarke M, et al. ; PRISMA-P Group . Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;350:g7647. doi: 10.1136/bmj.g7647 [DOI] [PubMed] [Google Scholar]
- 24.Taylor D, Paton C, Kapur S. The Maudsley Prescribing Guidelines in Psychiatry. 12th ed. Informa Healthcare; 2015. [Google Scholar]
- 25.National Institute for Health and Care Excellence . Depression in adults: recognition and management. Published 2009. Accessed July 2, 2021. https://www.nice.org.uk/guidance/cg90 [PubMed]
- 26.16.1.3.2 Imputing standard deviations for changes from baseline. Accessed April 5, 2021. https://handbook-5-1.cochrane.org/chapter_16/16_1_3_2_imputing_standard_deviations_for_changes_from_baseline.htm
- 27.Higgins JPT, Altman DG, Gøtzsche PC, et al. ; Cochrane Bias Methods Group; Cochrane Statistical Methods Group . The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343(7829):d5928. doi: 10.1136/bmj.d5928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mutz J, Edgcumbe DR, Brunoni AR, Fu CHY. Efficacy and acceptability of non-invasive brain stimulation for the treatment of adult unipolar and bipolar depression: a systematic review and meta-analysis of randomised sham-controlled trials. Neurosci Biobehav Rev. 2018;92:291-303. doi: 10.1016/j.neubiorev.2018.05.015 [DOI] [PubMed] [Google Scholar]
- 29.StataCorp. Stata Statistical Software: Release 17. 2021. Accessed July 2, 2021. https://www.stata.com/products/
- 30.Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560. doi: 10.1136/bmj.327.7414.557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bakim B, Uzun UE, Karamustafalioglu O, et al. The combination of antidepressant drug therapy and high-frequency repetitive transcranial magnetic stimulation in medication-resistant depression. Klinik Psikofarmakoloji Bülteni-Bulletin of Clinical Psychopharmacology. 2012;22(3):244-253. doi: 10.5455/bcp.20120807092434 [DOI] [Google Scholar]
- 32.Barbee JG, Thompson TR, Jamhour NJ, et al. A double-blind placebo-controlled trial of lamotrigine as an antidepressant augmentation agent in treatment-refractory unipolar depression. J Clin Psychiatry. 2011;72(10):1405-1412. doi: 10.4088/JCP.09m05355gre [DOI] [PubMed] [Google Scholar]
- 33.Berman RM, Fava M, Thase ME, et al. Aripiprazole augmentation in major depressive disorder: a double-blind, placebo-controlled study in patients with inadequate response to antidepressants. CNS Spectr. 2009;14(4):197-206. doi: 10.1017/S1092852900020216 [DOI] [PubMed] [Google Scholar]
- 34.Blumberger DM, Tran LC, Fitzgerald PB, Hoy KE, Daskalakis ZJ. A randomized double-blind sham-controlled study of transcranial direct current stimulation for treatment-resistant major depression. Front Psychiatry. 2012;3:74. doi: 10.3389/fpsyt.2012.00074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Blumberger DM, Mulsant BH, Fitzgerald PB, et al. A randomized double-blind sham-controlled comparison of unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant major depression. World J Biol Psychiatry. 2012;13(6):423-435. doi: 10.3109/15622975.2011.579163 [DOI] [PubMed] [Google Scholar]
- 36.Blumberger DM, Maller JJ, Thomson L, et al. Unilateral and bilateral MRI-targeted repetitive transcranial magnetic stimulation for treatment-resistant depression: a randomized controlled study. J Psychiatry Neurosci. 2016;41(4):E58-E66. doi: 10.1503/jpn.150265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Boutros NN, Gueorguieva R, Hoffman RE, Oren DA, Feingold A, Berman RM. Lack of a therapeutic effect of a 2-week sub-threshold transcranial magnetic stimulation course for treatment-resistant depression. Psychiatry Res. 2002;113(3):245-254. doi: 10.1016/S0165-1781(02)00267-6 [DOI] [PubMed] [Google Scholar]
- 38.Chen SJ, Chang CH, Tsai HC, Chen ST, Lin CCh. Superior antidepressant effect occurring 1 month after rTMS: add-on rTMS for subjects with medication-resistant depression. Neuropsychiatr Dis Treat. 2013;9:397-401. doi: 10.2147/NDT.S40466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Concerto C, Lanza G, Cantone M, et al. Repetitive transcranial magnetic stimulation in patients with drug-resistant major depression: a six-month clinical follow-up study. Int J Psychiatry Clin Pract. 2015;19(4):252-258. doi: 10.3109/13651501.2015.1084329 [DOI] [PubMed] [Google Scholar]
- 40.Dougherty DD, Rezai AR, Carpenter LL, et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol Psychiatry. 2015;78(4):240-248. doi: 10.1016/j.biopsych.2014.11.023 [DOI] [PubMed] [Google Scholar]
- 41.Fava M, Durgam S, Earley W, et al. Efficacy of adjunctive low-dose cariprazine in major depressive disorder: a randomized, double-blind, placebo-controlled trial. Int Clin Psychopharmacol. 2018;33(6):312-321. doi: 10.1097/YIC.0000000000000235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fitzgerald PB, Hoy KE, Herring SE, et al. A double blind randomized trial of unilateral left and bilateral prefrontal cortex transcranial magnetic stimulation in treatment resistant major depression. J Affect Disord. 2012;139(2):193-198. doi: 10.1016/j.jad.2012.02.017 [DOI] [PubMed] [Google Scholar]
- 43.Garcia-Toro M, Mayol A, Arnillas H, et al. Modest adjunctive benefit with transcranial magnetic stimulation in medication-resistant depression. J Affect Disord. 2001;64(2-3):271-275. doi: 10.1016/S0165-0327(00)00223-8 [DOI] [PubMed] [Google Scholar]
- 44.Heresco-Levy U, Gelfin G, Bloch B, et al. A randomized add-on trial of high-dose D-cycloserine for treatment-resistant depression. Int J Neuropsychopharmacol. 2013;16(3):501-506. doi: 10.1017/S1461145712000910 [DOI] [PubMed] [Google Scholar]
- 45.Hobart M, Skuban A, Zhang P, et al. Efficacy and safety of flexibly dosed brexpiprazole for the adjunctive treatment of major depressive disorder: a randomized, active-referenced, placebo-controlled study. Curr Med Res Opin. 2018;34(4):633-642. doi: 10.1080/03007995.2018.1430220 [DOI] [PubMed] [Google Scholar]
- 46.Holtzheimer PE III, Russo J, Claypoole KH, Roy-Byrne P, Avery DH. Shorter duration of depressive episode may predict response to repetitive transcranial magnetic stimulation. Depress Anxiety. 2004;19(1):24-30. doi: 10.1002/da.10147 [DOI] [PubMed] [Google Scholar]
- 47.Kauffmann CD, Cheema MA, Miller BE. Slow right prefrontal transcranial magnetic stimulation as a treatment for medication-resistant depression: a double-blind, placebo-controlled study. Depress Anxiety. 2004;19(1):59-62. doi: 10.1002/da.10144 [DOI] [PubMed] [Google Scholar]
- 48.Li C-T, Chen M-H, Juan C-H, et al. Efficacy of prefrontal theta-burst stimulation in refractory depression: a randomized sham-controlled study. Brain. 2014;137(Pt 7):2088-2098. doi: 10.1093/brain/awu109 [DOI] [PubMed] [Google Scholar]
- 49.Marcus RN, McQuade RD, Carson WH, et al. The efficacy and safety of aripiprazole as adjunctive therapy in major depressive disorder: a second multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychopharmacol. 2008;28(2):156-165. doi: 10.1097/JCP.0b013e31816774f9 [DOI] [PubMed] [Google Scholar]
- 50.McAllister-Williams RH, Anderson IM, Finkelmeyer A, et al. ; ADD Study Team . Antidepressant augmentation with metyrapone for treatment-resistant depression (the ADD Study): a double-blind, randomised, placebo-controlled trial. Lancet Psychiatry. 2016;3(2):117-127. doi: 10.1016/S2215-0366(15)00436-8 [DOI] [PubMed] [Google Scholar]
- 51.Pallanti S, Bernardi S, Di Rollo A, Antonini S, Quercioli L. Unilateral low frequency versus sequential bilateral repetitive transcranial magnetic stimulation: is simpler better for treatment of resistant depression? Neuroscience. 2010;167(2):323-328. doi: 10.1016/j.neuroscience.2010.01.063 [DOI] [PubMed] [Google Scholar]
- 52.Santos MA, Rocha FL, Hara C. Efficacy and safety of antidepressant augmentation with lamotrigine in patients with treatment-resistant depression: a randomized, placebo-controlled, double-blind study. Prim Care Companion J Clin Psychiatry. 2008;10(3):187-190. doi: 10.4088/PCC.v10n0302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Shelton RC, Tollefson GD, Tohen M, et al. A novel augmentation strategy for treating resistant major depression. Am J Psychiatry. 2001;158(1):131-134. doi: 10.1176/appi.ajp.158.1.131 [DOI] [PubMed] [Google Scholar]
- 54.Thase ME, Youakim JM, Skuban A, et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebo-controlled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 2015;76(9):1224-1231. doi: 10.4088/JCP.14m09688 [DOI] [PubMed] [Google Scholar]
- 55.Thase ME, Youakim JM, Skuban A, et al. Adjunctive brexpiprazole 1 and 3 mg for patients with major depressive disorder following inadequate response to antidepressants: a phase 3, randomized, double-blind study. J Clin Psychiatry. 2015;76(9):1232-1240. doi: 10.4088/JCP.14m09689 [DOI] [PubMed] [Google Scholar]
- 56.Theleritis C, Sakkas P, Paparrigopoulos T, et al. Two versus one high-frequency repetitive transcranial magnetic stimulation session per day for treatment-resistant depression: a randomized sham-controlled trial. J ECT. 2017;33(3):190-197. doi: 10.1097/YCT.0000000000000387 [DOI] [PubMed] [Google Scholar]
- 57.Triggs WJ, Ricciuti N, Ward HE, et al. Right and left dorsolateral pre-frontal rTMS treatment of refractory depression: a randomized, sham-controlled trial. Psychiatry Res. 2010;178(3):467-474. doi: 10.1016/j.psychres.2010.05.009 [DOI] [PubMed] [Google Scholar]
- 58.van Eijndhoven PFP, Bartholomeus J, Möbius M, et al. A randomized controlled trial of a standard 4-week protocol of repetitive transcranial magnetic stimulation in severe treatment resistant depression. J Affect Disord. 2020;274:444-449. doi: 10.1016/j.jad.2020.05.055 [DOI] [PubMed] [Google Scholar]
- 59.Palm U, Schiller C, Fintescu Z, et al. Transcranial direct current stimulation in treatment resistant depression: a randomized double-blind, placebo-controlled study. Brain Stimul. 2012;5(3):242-251. doi: 10.1016/j.brs.2011.08.005 [DOI] [PubMed] [Google Scholar]
- 60.Zheng H, Zhang L, Li L, et al. High-frequency rTMS treatment increases left prefrontal myo-inositol in young patients with treatment-resistant depression. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(7):1189-1195. doi: 10.1016/j.pnpbp.2010.06.009 [DOI] [PubMed] [Google Scholar]
- 61.Yesavage JA, Fairchild JK, Mi Z, et al. ; VA Cooperative Studies Program Study Team . Effect of repetitive transcranial magnetic stimulation on treatment-resistant major depression in US veterans: a randomized clinical trial. JAMA Psychiatry. 2018;75(9):884-893. doi: 10.1001/jamapsychiatry.2018.1483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Su TP, Chen MH, Li CT, et al. Dose-related effects of adjunctive ketamine in Taiwanese patients with treatment-resistant depression. Neuropsychopharmacology. 2017;42(13):2482-2492. doi: 10.1038/npp.2017.94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Palhano-Fontes F, Barreto D, Onias H, et al. Rapid antidepressant effects of the psychedelic ayahuasca in treatment-resistant depression: a randomized placebo-controlled trial. Psychol Med. 2019;49(4):655-663. doi: 10.1017/S0033291718001356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nierenberg AA, Papakostas GI, Petersen T, et al. Lithium augmentation of nortriptyline for subjects resistant to multiple antidepressants. J Clin Psychopharmacol. 2003;23(1):92-95. doi: 10.1097/00004714-200302000-00013 [DOI] [PubMed] [Google Scholar]
- 65.Chen MH, Lin WC, Tu PC, et al. Antidepressant and antisuicidal effects of ketamine on the functional connectivity of prefrontal cortex-related circuits in treatment-resistant depression: a double-blind, placebo-controlled, randomized, longitudinal resting fMRI study. J Affect Disord. 2019;259:15-20. doi: 10.1016/j.jad.2019.08.022 [DOI] [PubMed] [Google Scholar]
- 66.Kamijima K, Higuchi T, Ishigooka J, et al. ; ADMIRE Study Group . Aripiprazole augmentation to antidepressant therapy in Japanese patients with major depressive disorder: a randomized, double-blind, placebo-controlled study (ADMIRE study). J Affect Disord. 2013;151(3):899-905. doi: 10.1016/j.jad.2013.07.035 [DOI] [PubMed] [Google Scholar]
- 67.Kamijima K, Kimura M, Kuwahara K, Kitayama Y, Tadori Y. Randomized, double-blind comparison of aripiprazole/sertraline combination and placebo/sertraline combination in patients with major depressive disorder. Psychiatry Clin Neurosci. 2018;72(8):591-601. doi: 10.1111/pcn.12663 [DOI] [PubMed] [Google Scholar]
- 68.Ionescu DF, Bentley KH, Eikermann M, et al. Repeat-dose ketamine augmentation for treatment-resistant depression with chronic suicidal ideation: a randomized, double blind, placebo controlled trial. J Affect Disord. 2019;243:516-524. doi: 10.1016/j.jad.2018.09.037 [DOI] [PubMed] [Google Scholar]
- 69.Husain MI, Chaudhry IB, Husain N, et al. Minocycline as an adjunct for treatment-resistant depressive symptoms: a pilot randomised placebo-controlled trial. J Psychopharmacol. 2017;31(9):1166-1175. doi: 10.1177/0269881117724352 [DOI] [PubMed] [Google Scholar]
- 70.Holtzheimer PE, Husain MM, Lisanby SH, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry. 2017;4(11):839-849. doi: 10.1016/S2215-0366(17)30371-1 [DOI] [PubMed] [Google Scholar]
- 71.Hobart M, Skuban A, Zhang P, et al. A randomized, placebo-controlled study of the efficacy and safety of fixed-dose brexpiprazole 2 mg/d as adjunctive treatment of adults with major depressive disorder. J Clin Psychiatry. 2018;79(4):17m12058. doi: 10.4088/JCP.17m12058 [DOI] [PubMed] [Google Scholar]
- 72.Coenen VA, Bewernick BH, Kayser S, et al. Superolateral medial forebrain bundle deep brain stimulation in major depression: a gateway trial. Neuropsychopharmacology. 2019;44(7):1224-1232. doi: 10.1038/s41386-019-0369-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Berman RM, Marcus RN, Swanink R, et al. The efficacy and safety of aripiprazole as adjunctive therapy in major depressive disorder: a multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2007;68(6):843-853. doi: 10.4088/JCP.v68n0604 [DOI] [PubMed] [Google Scholar]
- 74.Bennabi D, Nicolier M, Monnin J, et al. Pilot study of feasibility of the effect of treatment with tDCS in patients suffering from treatment-resistant depression treated with escitalopram. Clin Neurophysiol. 2015;126(6):1185-1189. doi: 10.1016/j.clinph.2014.09.026 [DOI] [PubMed] [Google Scholar]
- 75.Bauer M, Hefting N, Lindsten A, Josiassen MK, Hobart M. A randomised, placebo-controlled 24-week study evaluating adjunctive brexpiprazole in patients with major depressive disorder. Acta Neuropsychiatr. 2019;31(1):27-35. doi: 10.1017/neu.2018.23 [DOI] [PubMed] [Google Scholar]
- 76.Garcia-Toro M, Salva J, Daumal J, et al. High (20-Hz) and low (1-Hz) frequency transcranial magnetic stimulation as adjuvant treatment in medication-resistant depression. Psychiatry Res. 2006;146(1):53-57. doi: 10.1016/j.pscychresns.2004.08.005 [DOI] [PubMed] [Google Scholar]
- 77.Fava M, Ramey T, Pickering E, Kinrys G, Boyer S, Altstiel L. A randomized, double-blind, placebo-controlled phase 2 study of the augmentation of a nicotinic acetylcholine receptor partial agonist in depression: is there a relationship to leptin levels? J Clin Psychopharmacol. 2015;35(1):51-56. doi: 10.1097/JCP.0000000000000245 [DOI] [PubMed] [Google Scholar]
- 78.Padberg F, Zwanzger P, Thoma H, et al. Repetitive transcranial magnetic stimulation (rTMS) in pharmacotherapy-refractory major depression: comparative study of fast, slow and sham rTMS. Psychiatry Res. 1999;88(3):163-171. doi: 10.1016/S0165-1781(99)00092-X [DOI] [PubMed] [Google Scholar]
- 79.Rush AJ, Marangell LB, Sackeim HA, et al. Vagus nerve stimulation for treatment-resistant depression: a randomized, controlled acute phase trial. Biol Psychiatry. 2005;58(5):347-354. doi: 10.1016/j.biopsych.2005.05.025 [DOI] [PubMed] [Google Scholar]
- 80.Baeken C, Vanderhasselt MA, Remue J, et al. Intensive HF-rTMS treatment in refractory medication-resistant unipolar depressed patients. J Affect Disord. 2013;151(2):625-631. doi: 10.1016/j.jad.2013.07.008 [DOI] [PubMed] [Google Scholar]
- 81.Rief W, Nestoriuc Y, Weiss S, Welzel E, Barsky AJ, Hofmann SG. Meta-analysis of the placebo response in antidepressant trials. J Affect Disord. 2009;118(1-3):1-8. doi: 10.1016/j.jad.2009.01.029 [DOI] [PubMed] [Google Scholar]
- 82.Fraguas D, Díaz-Caneja CM, Pina-Camacho L, Umbricht D, Arango C. Predictors of placebo response in pharmacological clinical trials of negative symptoms in schizophrenia: a meta-regression analysis. Schizophr Bull. 2019;45(1):57-68. doi: 10.1093/schbul/sbx192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nierenberg AA, Østergaard SD, Iovieno N, Walker RSW, Fava M, Papakostas GI. Predictors of placebo response in bipolar depression. Int Clin Psychopharmacol. 2015;30(2):59-66. doi: 10.1097/YIC.0000000000000058 [DOI] [PubMed] [Google Scholar]
- 84.Meister R, Jansen A, Härter M, Nestoriuc Y, Kriston L. Placebo and nocebo reactions in randomized trials of pharmacological treatments for persistent depressive disorder: a meta-regression analysis. J Affect Disord. 2017;215:288-298. doi: 10.1016/j.jad.2017.03.024 [DOI] [PubMed] [Google Scholar]
- 85.Khan A, Fahl Mar K, Faucett J, Khan Schilling S, Brown WA. Has the rising placebo response impacted antidepressant clinical trial outcome? data from the US Food and Drug Administration, 1987-2013. World Psychiatry. 2017;16(2):181-192. doi: 10.1002/wps.20421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Furukawa TA, Cipriani A, Leucht S, et al. Is placebo response in antidepressant trials rising or not? a reanalysis of datasets to conclude this long-lasting controversy. Evid Based Ment Health. 2018;21(1):1-3. doi: 10.1136/eb-2017-102827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Jilch S, Sel R, Shariat SF. Medical practice and placebo response: an inseparable bond? Wien Klin Wochenschr. 2020;132(9-10):228-231. doi: 10.1007/s00508-020-01626-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.George MS, Lisanby SH, Avery D, et al. Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder: a sham-controlled randomized trial. Arch Gen Psychiatry. 2010;67(5):507-516. doi: 10.1001/archgenpsychiatry.2010.46 [DOI] [PubMed] [Google Scholar]
- 89.O’Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216. doi: 10.1016/j.biopsych.2007.01.018 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eTable 1. Risk of Bias
eTable 2. Subgroup Analysis of Treatment Modalities
eTable 3. Clinical Characteristics
eAppendix 1. Components of the Randomized Effect Model Used for Our Analyses
eAppendix 2. Search Strategy
eFigure 1. Funnel Plot of Included Studies
eFigure 2. Trim and Fill Funnel Plot
eFigure 3. Forest Plot of Main Placebo Effect Size Grouped by Risk of Bias