Abstract
Background: Hepatitis C virus (HCV) in diffuse large B-cell lymphoma (DLBCL) is associated with a higher prevalence and distinctive clinical characteristics and outcomes. Methods: A retrospective analysis of adult DLBCL patients from 2011 to 2015 was studied. Results: A total of 206 adult DLBCL were enrolled with 22 (10.7%) HCV-positive patients. Compared to HCV-negative patients, the HCV-positive group had a poor performance status (p = 0.011), lower platelet count (p = 0.029), and higher spleen and liver involvement incidences (liver involvement, p = 0.027, spleen involvement, p = 0.026), and they received fewer cycles of chemotherapy significantly due to morbidity and mortality (p = 0.048). Overall survival was shorter in HCV-positive DLBCL (25.3 months in HCV-positive vs. not reached (NR), p = 0.049). With multivariate analysis, poor performance status (p < 0.001), advanced stage (p < 0.001), less chemotherapy cycles (p < 0.001), and the presence of liver toxicity (p = 0.001) contributed to poor OS in DLBCL. Among HCV-positive DLBCL, the severity of liver fibrosis was the main risk factor related to death. Conclusion: Inferior survival of HCV-positive DLBCL was observed and associated with poor performance status, higher numbers of complications, and intolerance of treatment, leading to fewer therapy. Therefore, anti-HCV therapy, such as direct-acting antiviral agents, might benefit these patients in the future.
Keywords: hepatitis C virus, diffuse large B-cell lymphoma, survival, fibrosis, performance status, liver toxicity
1. Introduction
Hepatitis C virus (HCV) infection is one of the major causes of chronic liver disease around the world with a prevalence rate of around 3% [1]. In addition to hepatic function, various extrahepatic manifestations, including vasculitis, glomerulonephrisis, and a wide range of lymphoproliferative disorders, have been associated with HCV infections, resulting in further morbidities and mortalities [2,3]. HCV infection are also associated with B-cell non-Hodgkin lymphoma (NHL) with distinct clinical presentation and outcome, especially in diffuse large B-cell lymphoma (DLBCL) and marginal zone lymphoma (MZL) in recent epidemiological studies [4,5,6,7,8,9]. Due to the fact that direct anti-HCV therapy induces hematologic response in indolent NHL patients with HCV infection, HCV infection has been linked to NHL, and front-line therapy of asymptomatic indolent NHLs is recommended [10,11]. DLBCL, the most common NHL, is an aggressive type of NHL with specific genetic features and clinical presentations [12]. Although HCV-positive DLBCLs display specific clinical features and outcomes, the optimal treatment and timing of anti-HCV therapy in aggressive lymphoma is still uncertain [1,13,14,15,16]. Previous reports showed that HCV-positive patients with DLBCL exhibited worse overall survival (OS), and the incidence of severe hepatic toxicity in HCV-positive patients was significantly higher than that of HCV-negative patients [13,17]. However, all these studies only included small numbers of HCV-positive patients, and not all studies regarding HCV infection and DLBCL showed consistent results.
In addition, the seroprevalence of anti-HCV antigen in Taiwan is around 1 to 5.4% in the general population [18,19,20], and a retrospective study that investigated the association of HCV and lymphoma in southern Taiwan by Chuang et al. displayed that the incidence of HCV infection among lymphoma patients was significantly higher than that in healthy controls (11.0% vs. 1.8%, p < 0.001) [21]. The differences of HCV infection in NHL patients reflect geographic differences in HCV epidemiology [1,22,23]. The purpose of this study is to analyze clinicopathological characteristics, tolerance to chemotherapy, and clinical outcomes of DLBCL patients between HCV-positive and HCV-negative groups in order to determine the impact of HCV infection in this population.
2. Materials and Methods
2.1. Study Population
In this retrospective observational study, newly diagnosed DLBCL patients who were managed at Kaohsiung Medical University Hospital between January 2011 and December 2015 were enrolled. Clinical characteristics, including patients’ demographics, baseline laboratory and biochemical data, hepatitis virus serology including hepatitis B virus (HBV) and HCV, and FIB (fibrosis)-4 index were collected and analyzed from medical records. Lymphoma was treated according to the treatment guideline in the hospital. The study was approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(I)-20210119).
2.2. Risk Groups and Outcomes
Patients were categorized as HCV-positive or HCV-negative groups based on the presence or absence of anti-HCV antibody. Since the interferon-free direct-acting antiviral agent (DAA) regimens were added to the reimbursement list in Taiwan for HCV treatment in 2017, patients were only followed until 2017 to eliminate the confounding factor of DAA treatment. All HCV-positive patients in this study did not receive interferon-free DAA prior to or during chemotherapy. All DLBCL patients with HBsAg-positive received antiviral agents for HBV prophylaxis. Performance status was evaluated by Eastern Cooperative Oncology Group (ECOG) scale [24]. The FIB-4 index was used to assess the severity of liver fibrosis as it is considered to be a useful fibrosis scoring system in HCV. A cutoff of >3.25 had a positive predictive value of 65% and a specificity of 97% to predict advanced fibrosis [25,26]. In Taiwan, liver cirrhosis was diagnosed by the image, either sonography or computed tomography or by FIB-4 index ≥ 6.5. The international prognostic index (IPI) score divided patients into four groups: low, low-intermediate, high-intermediate, and high for risk stratification [27], and the revised international prognostic index (R-IPI) classified patients into three groups: very good, good, and poor [28], which was calculated and used for analysis. The treatment response was evaluated by computed tomography (CT) or positron emission tomography–computed tomography (PET-CT) every 3–6 months according to response criteria [29]. The first time of treatment response evaluation was at the time after 3–4 courses of chemoimmunotherapy. The liver toxicity was calculated by the common terminology criteria for adverse events (CTACE), version 4. Progression-free survival (PFS) was defined as the length of time from the date of diagnosis to the date of progression or death. Overall survival (OS) was defined as the length of time from the date of diagnosis to the last date of follow-up or death. Liver function was monitored during every course of treatment during chemotherapy and around every 3 months after chemotherapy.
2.3. Statistical Analysis
Patient characteristics between HCV-positive and HCV-negative groups were analyzed using descriptive statistics and are presented as frequencies, percentages, and means with standard deviations. Student’s t-test or nonparametric statistics were utilized to test for statistically significant differences in continuous variables, whereas the chi-square or Fisher’s exact test was used for categorical variables. OS and PFS was estimated using the Kaplan–Meier method. Variables with a p-value of less than 0.05 in the univariate analysis of overall survival were subsequently subjected to multivariate analysis using a Cox regression model. A two-tailed p-value < 0.05 was considered statistically significant.
3. Results
3.1. Baseline Characteristics
A total of 206 DLBCL patients were enrolled in the study. Among them, twenty-two (10.7%) were HCV-positive. The characteristics at the time of diagnosis are listed in Table 1. Compared to HCV-negative patients, HCV-positive patients seemed to have a significantly lower platelet count (186.7 ± 68.8 × 103/μL vs. 236.2 ± 102.9 × 103/μL, p = 0.029) and a trend of older age (mean age 67.23 ± 17.13 vs. 61.24 ± 15.27, p = 0.088). HCV-negative patients had better performance status compared to HCV-positive patients (p = 0.011). Higher spleen and liver involvement rates were observed in HCV-positive patients compared to HCV-negative (liver involvement: 18.2% in HCV-positive vs. 4.3% in HCV-negative, p = 0.027, spleen involvement: 31.8% in HCV-positive vs. 13.6% in HCV-negative, p = 0.026). More HCV-positive patients had elevated aminotransferase compared to HCV-negative population, however, not to a statistical significant degree (31.8% vs. 16.3%, p = 0.073). There were no significant differences on sex, B symptoms, bone marrow involvement, IPI score, R-IPI score, HBsAg status, white blood cell (WBC) count, hemoglobin, albumin, lactate dehydrogenase (LDH) levels, alanine aminotransferase (GPT) levels, Beta-2 macroglobulin levels, and the percentage of receiving treatment between two groups.
Table 1.
Baseline characteristics of HCV-negative and HCV-positive DLBCL patients.
| Variables | HCV-Negative DLBCL | HCV-Positive DLBCL | |
|---|---|---|---|
| N = 184 | N = 22 | p Value | |
| Age, (Mean ± SD) | 61.24 ± 15.27 | 67.23 ± 17.13 | 0.088 |
| Gender, N(%) | 0.618 | ||
| Male | 94(51.1%) | 10(45.5%) | |
| Female | 90(48.9%) | 12(54.5%) | |
| ECOG, N(%) | 0.011 * | ||
| 0 | 79(42.9%) | 6(27.3%) | |
| 1 | 78(42.4%) | 12(54.5%) | |
| 2 | 13(7.1%) | 1(4.5%) | |
| 3 | 11(6.0%) | 0(0%) | |
| 4 | 3(1.6%) | 3(13.6%) | |
| Ann Arbor stage, N(%) | 0.957 | ||
| I-II | 68(37.0%) | 8(36.4%) | |
| III-IV | 116(63.0%) | 14(63.6%) | |
| B symptoms, N(%) | 74(40.2%) | 13(59.1%) | 0.090 |
| Liver involvement, N(%) | 8(4.3%) | 4 4(18.2%) | 0.027 * |
| Spleen involvement, N(%) | 25(13.6%) | 7(31.8%) | 0.026 * |
| Bone marrow involvement, N(%) | 38(20.7%) | 4(18.2%) | 1.000 |
| R-IPI score, N(%) | 0.202 | ||
| Very good | 15(8.2%) | 0(0%) | |
| Good | 90(48.9%) | 9(40.9%) | |
| Poor | 79(42.9%) | 13(59.1%) | |
| IPI score, N(%) | 0.448 | ||
| Low | 57(31.0%) | 6(27.3%) | |
| Low-intermediate | 48(26.1%) | 3(13.6%) | |
| High-intermediate | 41(22.3%) | 6(27.3%) | |
| High | 38(20.7%) | 7(31.8%) | |
| HBsAg positive, N(%) | 38(20.7%) | 2(9.1%) | 0.261 |
| Abnormal liver funtion at baseline, N(%) | 30(16.3%) | 7(31.8%) | 0.073 |
| WBC (Mean ± SD) | 7554.6 ± 3474.0 | 7480.0 ± 2823.8 | 0.923 |
| Hgb (Mean ± SD) | 11.8 ± 2.3 | 11.6 ± 1.7 | 0.687 |
| Platelet (×103, Mean ± SD) | 236.2 ± 102.9 | 186.7 ± 68.8 | 0.029 * |
| Albumin (Mean ± SD) | 3.6 ± 0.6 | 3.6 ± 0.6 | 0.969 |
| LDH (Mean ± SD) | 366.9 ± 453.2 | 430.9 ± 450.3 | 0.532 |
| GOT (Mean ± SD) | 48.2 ± 134.5 | 58.1 ± 77.5 | 0.734 |
| GPT (Mean ± SD) | 43.7 ± 135.0 | 34.9 ± 28.6 | 0.763 |
| Beta-2 microglobulin (Mean ± SD) | 303.5 ± 273.2 | 332.9 ± 116.1 | 0.627 |
*: p < 0.05, ECOG: Eastern Cooperative Oncology Group, R-IPI: revised international prognostic index, IPI: international prognostic index, SD: standard deviation, WBC: white blood cell, Hgb: hemoglobin, LDH: lactate dehydrogenase, GPT: alanine aminotransferase.
3.2. Response to Therapy and Outcomes
Patients were managed by guidelines according to clinical status. Most (92.9%) of the HCV-negative patients and 86.4% of the HCV-positive patients received treatment respectively without statistical difference (p = 0.388) with most of them (74.3% in HCV-negative, 68.4% in HCV-positive) receiving an R-CHOP (rituximab, cyclophosphamide, daunorubicin, oncovin, and prednisolone) regimen. The details of the chemotherapy regimen were presented in Table 2, and there was no significant difference in treatment regimen between the HCV-positive and HCV-negative population. The HCV-positive population received fewer cycles of chemotherapy compared with HCV-negative patients (mean cycles of chemotherapy: 4.8 ± 3.5 in HCV-positive vs. 6.3 ± 3.4 in HCV-negative, p = 0.048) (Table 2). A lower evaluation of treatment response during the follow-up period was found in HCV-positive patients compared to the HCV-negative patient group (31.8% in HCV positive vs. 14.7% in HCV-negative, p = 0.041). Among these HCV-positive patients who were unable to be evaluated, four of seven patients received limited treatment with three of them receiving only one cycle of chemotherapy due to early mortality from sepsis and/or other morbidities. Three of seven patients had supportive management due to old age (n = 2, over 75 years old) and severe liver cirrhosis (n = 1).
Table 2.
Treatment detail, response, and liver toxicity in HCV-negative and HCV-positive DLBCL patients.
| Variables | HCV-Negative DLBCL | HCV-Positive DLBCL | |
|---|---|---|---|
| N = 184 | N = 22 | p Value | |
| Receiving treatment (Yes), N(%) | 171(92.9%) | 19(86.4%) | 0.388 |
| First-line treatment regimen in receiving treatment population | 0.354 | ||
| RCHOP | 127(74.3%) | 13(68.4%) | |
| RCOP | 30(17.5%) | 6(31.6%) | |
| Other regimen (REPOCH, RMTX) | 11(6.4%) | 0(0.0%) | |
| R with prednisolone | 3(1.8%) | 0(0.0%) | |
| Chemotherapy cycles (Mean ± SD) | 6.3 ± 3.4 | 4.8 ± 3.5 | 0.048 * |
| Treatment response, N(%) | |||
| Complete response/Partial response | 105(57.1%) | 9(40.9%) | 0.150 |
| Progression or recurrent | 52(28.3%) | 6(27.3%) | 0.922 |
| Unable to evaluate response | 27(14.7%) | 7(31.8%) | 0.041 * |
| Liver toxicity | 83(45.1%) | 19(86.4%) | <0.001 * |
| ≥Grade 3 liver toxicity | 18(9.8%) | 8(36.4%) | <0.001 * |
| Treatment delay or discontinue | 23(12.5%) | 9(40.9%) | 0.001 * |
* p < 0.05, R-CHOP: rituximab, cyclophosphamide, daunorubicin, oncovin and prednisolone; R-COP: rituximab, cyclophosphamide, oncovin and prednisolone; R-EPOCH: rituximab, etoposide, cyclophosphamide, daunorubicin, oncovin and prednisolone; R: rituximab.
The progression and recurrent events were similar in HCV-positive and HCV-negative groups in evaluable patients (27.3% in HCV-positive vs. 28.3% in HCV-negative, p = 0.922). Higher incidence of liver toxicity (86.4% in HCV-positive vs. 45.1% in HCV-negative, p < 0.001) and severe liver toxicity (≥grade 3 liver toxicity, 36.4% in HCV-positive vs. 9.8% in HCV-negative, p < 0.001) were observed in HCV-positive patients than HCV-negative patients. The incidence of liver toxicity was not associated with baseline GPT levels in HCV-positive patients (20.67 IU/mL, without liver toxicity vs. 37.16 IU/mL, with liver toxicity, p = 0.366). More patients in the HCV-positive group delayed or discontinued their treatment due to toxicities than in the HCV-negative group (40.9% vs. 12.5%, p = 0.001)
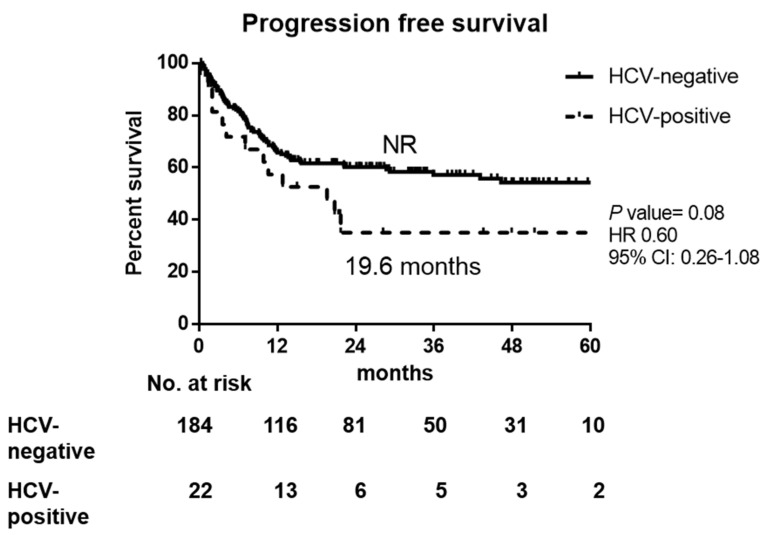
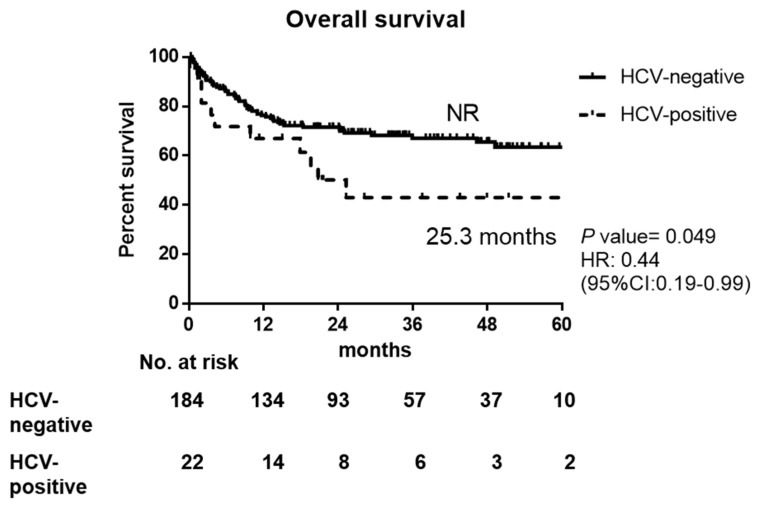
There was a trend of worse PFS in HCV-infected patients than non-infected, but no statistical significance (19.6 months in HCV-positive vs. NR in HCV-negative, p = 0.080), as shown in Figure 1. The 1-year, 2-year, and 3-year OS rates in HCV-positive and HCV-negative DLBCL patients were 67.0% vs. 76.4%, 50.2% vs. 71.6%, and 43.1% vs. 67.0%, respectively. The OS was worse in HCV-positive DLBCL patients (25.3 months in HCV-positive vs. not reached (NR) in HCV-negative, p = 0.049 by Kaplan–Meier method, Figure 2), which might result from early mortality, higher liver toxicity, and higher incidence of treatment delay/discontinuation with fewer treatment courses.
Figure 1.
Progression-free survival of HCV-positive and HCV-negative DLBCL patients. NR: not reached, HR: hazard ratio, CI: confidence interval.
Figure 2.
Overall survival of HCV-positive and HCV-negative DLBCL patients. NR: not reached, HR: hazard ration, CI: confidence interval.
3.3. Prognostic Factors for Overall Survival and Progression Free Survival in DLBCL Patients
Based on univariate analysis via Cox regression model, we found that older age (HR: 1.03, 95% CI: 1.01–1.04, p = 0.003), poor performance status (ECOG ≥ 2, HR: 6.22, 95% CI: 3.66–10.56, p < 0.001), advanced stage (HR 4.36, 95% CI: 2.23–8.55, p <0.001), less chemotherapy cycles (HR: 0.77, 95% CI: 0.70–0.84, p < 0.001), and the presence of liver toxicity (HR: 2.46, 95% CI: 1.47–4.12, p = 0.001) contributed to poor OS. In multivariate analysis, four key factors: advanced stage, poor performance status, liver toxicity presentation, and less chemotherapy cycles were independent poor prognostic factors for overall survival in DLBCL patients (Table 3). Regarding the progression-free survival, we found that after multivariate analysis, poor performance status (ECOG ≥ 2, HR: 2.82, 95% CI: 1.56–5.09, p < 0.001), advanced stage (HR: 3.18, 95% CI: 1.81–5.58, p < 0.001), the presence of liver toxicity (HR: 2.67, 95% CI: 1.69–4.22, p < 0.001), and less chemotherapy cycles (HR 0.90, 95% CI: 0.83–0.98, p = 0.015), contributed to poor PFS in DLBCL patients (Table 4).
Table 3.
Univariate and multivariate analysis of risk factors for overall survival.
| Univariate Analysis | Multivariate Analysis a | |||
|---|---|---|---|---|
| Variables | HR (95%CI) | p-Value | HR (95%CI) | p-Value |
| Age | 1.03(1.01–1.04) | 0.004 * | 1.00(0.98–1.02) | 0.733 |
| Gender | 0.479 | |||
| Male | 1.0 | |||
| Female | 0.84(0.52–1.36) | |||
| Hepatitis C infection | 0.054 | 0.574 | ||
| Negative | 1.0 | 1.00 | ||
| Positive | 1.89 (0.99–3.60) | 0.82(0.41–1.64) | ||
| Hepatitis B infection | 0.069 | 0.202 | ||
| Negative | 1 | 1.00 | ||
| Positive | 0.50(0.24–1.06) | 0.61(0.28–1.31) | ||
| ECOG | <0.001 * | 0.003 * | ||
| <2 | 1.00 | 1.00 | ||
| ≥2 | 6.22(3.66–10.56) | 2.64(1.39–4.99) | ||
| Stage | <0.001 * | <0.001 * | ||
| I/II | 1.0 | 1.00 | ||
| III/IV | 4.36(2.23–8.55) | 4.28(2.12–8.63) | ||
| Chemotherapy cycles | 0.77(0.70–0.84) | <0.001 * | 0.78(0.70–0.86) | <0.001 * |
| Liver toxicity | 0.001 * | <0.001 * | ||
| Yes | 2.46(1.47–4.12) | 2.73(1.62–4.60) | ||
| No | 1.00 | 1.00 | ||
*: p < 0.05, HR: hazard ratio, CI: confidence interval, ECOG: Eastern Cooperative Oncology Group. a: Multivariate analyses: variables with p < 0.1 in univariate analyses were included in multivariate analyses.
Table 4.
Univariate and multivariate analysis of risk factors for progression-free survival.
| Univariate Analysis | Multivariate Analysis a | |||
|---|---|---|---|---|
| Variables | HR (95%CI) | p-Value | HR (95%CI) | p-Value |
| Age | 1.02(1.00–1.03) | 0.033 * | 1.00(0.98–1.01) | 0.838 |
| Gender | 0.184 | |||
| Male | 1.0 | |||
| Female | 0.75(0.49–1.15) | |||
| Hepatitis C infection | 0.084 | 0.827 | ||
| Negative | 1.0 | 1.00 | ||
| Positive | 1.68 (0.93–3.03) | 0.93(0.50–1.74) | ||
| Hepatitis B infection | 0.025 * | 0.041 * | ||
| Negative | 1 | 1.00 | ||
| Positive | 0.47(0.24–0.91) | 0.49(0.25–0.97) | ||
| ECOG | <0.001 * | 0.001 * | ||
| <2 | 1.00 | 1.00 | ||
| ≥2 | 4.60(2.81–7.53) | 2.82(1.56–5.09) | ||
| Stage | <0.001 * | <0.001 * | ||
| I/II | 1.0 | 1.00 | ||
| III/IV | 3.59(2.09–6.18) | 3.18(1.81–5.58) | ||
| Chemotherapy cycles | 0.89(0.82–0.96) | 0.003 * | 0.90(0.83–0.98) | 0.015 * |
| Liver toxicity | 0.001 * | <0.001* | ||
| Yes | 2.56(1.63–4.02) | 2.67(1.69–4.22) | ||
| No | 1.00 | 1.00 | ||
*: p < 0.05, HR: hazard ratio, CI: confidence interval, ECOG: Eastern Cooperative Oncology Group a: Multivariate analyses: variables with p < 0.1 in univariate analyses were included in multivariate analyses.
3.4. Details of HCV-Positive DLBCL Patients during Follow-Up Period
The details of HCV-positive DLBCL patients are listed in Table S1. Half of HCV-positive (11 of 22) DLBCL patients died during the follow-up period. Seven died of sepsis, two died due to the progression of hepatocellular carcinoma, one died of gastrointestinal bleeding related to liver cirrhosis, and only one patient died of lymphoma progression. Based on subgroup analyses of the death events in the HCV-positive population, no difference of initial performance status, stage, IPI score, platelet count, coagulation profile (prothrombin time, PT), albumin, GPT, and LDH levels was found between the dead and alive groups. However, less chemotherapy cycles, higher percentage of severe liver toxicity, and treatment delay/discontinuation were observed in the death population, but not to a statistically significant degree. We found that only liver cirrhosis status contributed to more death events in HCV-positive patients. In HCV-positive patients, all were alive of those without liver cirrhosis (p = 0.035) or FIB-4 index less than 1.45 points (p = 0.032) (Table 5). In addition, only one of 22 HCV-positive patients started peg-interferon and ribavirin treatment for HCV at 4 months after the end of chemotherapy, and HCV viral load was 2758.2 IU/mL at that time. The HCV viral load became undetectable after peg-interferon and ribavirin status to now.
Table 5.
Different variables in the dead and alive groups of HCV-positive DLBCL patients.
| Variables | HCV-Positive DLBCL | ||
|---|---|---|---|
| Dead (n = 11) | Alive (n = 11) | p-Value | |
| Age, (Mean ± SD) | 69.00 ± 14.11 | 65.45 ± 20.25 | 0.639 |
| Sex | 0.198 | ||
| Male | 3(27.3%) | 7(63.6%) | |
| Female | 8(72.7%) | 4(36.4%) | |
| ECOG | 1.000 | ||
| <2 | 9(81.8%) | 9(81.8%) | |
| ≥2 | 2(18.2%) | 2(18.2%) | |
| Ann Arbor stage | 1.000 | ||
| I–II | 4(36.4%) | 4(36.4%) | |
| III–IV | 7(63.6%) | 7(63.6%) | |
| IPI score | 0.924 | ||
| Low | 3(27.3%) | 3(27.3%) | |
| Low-intermediate | 1(9.1%) | 2(18.2%) | |
| High-intermediate | 3(27.3%) | 3(27.3%) | |
| High | 4(36.4%) | 3(27.3%) | |
| GPT at diagnosis (Mean ± SD) |
42.82 ± 36.68 | 27.00 ± 15.29 | 0.209 |
| PLT at diagnosis (×103, Mean ± SD) |
184.45 ± 72.12 | 188.91 ± 68.80 | 0.884 |
| Albumin at diagnosis (Mean ± SD) |
3.53 ± 0.74 | 3.63 ± 0.58 | 0.737 |
| PT(INR) at diagnosis (Mean ± SD) |
1.05 ± 0.78 | 1.04 ± 0.53 | 0.707 |
| LDH at diagnosis (Mean ± SD) |
531.45 ± 615.90 | 330.45 ± 155.97 | 0.307 |
| Abnormal liver funtion at baseline, N(%) | 5(45.5%) | 2(18.2%) | 0.361 |
| Liver cirrhosis | 0.035 * | ||
| No | 6(54.5%) | 11(100%) | |
| Yes | 5(45.5%) | 0(0%) | |
| FIB-4 index | 0.032 * | ||
| <1.45 | 0(0%) | 4(36.4%) | |
| 1.45–3.25 | 4(36.4%) | 5(45.5%) | |
| >3.25 | 7(63.6%) | 2(18.2%) | |
| Receiving treatment | 1.000 | ||
| Yes | 9(81.8%) | 10(90.9%) | |
| No | 2(18.2%) | 1(9.1%) | |
| Chemotherapy cycles | 3.91 ± 4.04 | 5.64 ± 2.77 | 0.255 |
| ≥Grade 3 Liver toxicity | 0.183 | ||
| Yes | 6(54.5%) | 2(18.2%) | |
| No | 5(45.5%) | 9(81.8%) | |
| Treatment delay or discontinuation | 0.387 | ||
| Yes | 6(54.5%) | 3(27.3%) | |
| No | 5(45.5%) | 8(72.7%) | |
*: p < 0.05, SD: standard deviation, ECOG: Eastern Cooperative Oncology Group, IPI: international prognostic index GPT: alanine aminotransferase, PLT: platelet, PT: prothrombin time, INR: international normalized ratio, LDH: lactate dehydrogenase, FIB-4: fibrosis-4.
4. Discussion
In our study, the prevalence of HCV infection was 10.7% (22/206 patients) in DLBCL patients. It was higher than the prevalence rate of the general population in Taiwan at 4.4% (6904/157,720) of anti-HCV positive [30]. There have been some hypothetical models to describe the possible pathologic role of HCV infection in aggressive B-cell lymphoma. The direct transformation mechanism is one kind of hypothesis accounting for HCV-associated lymphomagenesis. In vitro, the infection of B-cell lines with HCV leads to somatic mutations of several oncogenes and tumor-suppressor genes such as p53, beta-catenin, and Bcl6 [31]. On the contrary, some studies support the role of HCV as an indirect transformation agent by chronically stimulating B-cell immunologic response and finally leading to lymphoma. Polyclonal or monoclonal B-cell proliferation can be detected in the blood or bone marrow of HCV patients and is associated with mixed cryoglobulinemia type II. The risk of lymphoma in patients with HCV-associated cryoglobulinemia is estimated to be 35 times higher than that in the general population [32]. All these theories are proposed to explain the high prevalence of HCV infection in lymphoma patients.
In previous retrospective studies, HCV-infected DLBCL patients shared distinctive clinical features. A higher percentage of HCV-positive DLBCL cases were associated with old age (> or =60) than HCV-negative DLBCL cases at diagnosis [14,33]. HCV-positive patients had more frequent extra-nodal involvement [13,33] and more frequent elevated lactate dehydrogenase (LDH) levels than other patients [13]. In our studies, we found that HCV-positive DLBCL patients had poorer performance status, lower platelet count, and higher probability of liver and spleen involvement than HCV-negative patients. In terms of OS and PFS, the influence of HCV infection on survival in DLBCL patients remains controversial. Studies conducted by Ennishi et al. reported a similar outcome in HCV-positive DLBCL patients compared to HCV-negative (3-year OS 75% in HCV-positive vs. 84% in HCV-negative, p = 0.07) [17]. On the contrary, two other studies presented an inferior outcome in HCV-positive patients [16,34]. Chen et al. found that three independent factors predicted a dismal OS: lower albumin level (<3 g/dL vs. ≥3 g/dL; HR = 13.21, 95% CI = 2.69–64.98, p = 0.001), presence of HCV infection (HCV-positive vs. HCV-negative; HR = 9.75, 95% CI = 1.97–48.34, p = 0.005), and poor IPI risk (high-intermediate or high vs. low-intermediate or low; HR = 5.56, 95% CI = 1.17–26.55, p = 0.031) [34]. The Fondazione Italiana Linfomi has conducted a multicenter retrospective study to explore a new prognostic system for HCV-associated DLBCL. Therefore, the HCV Prognostic Score, based on performance status, albumin level, and HCV-RNA viral load, was therefore introduced as a useful tool to predict the outcome of HCV-associated DLBCL [16].
In our study, we found that an inferior overall survival was observed in HCV-positive patients by the Kaplan–Meier method. However, after multivariate analysis, HCV infection status was not the key risk factor for overall survival. Instead, advanced stage, poor performance status, liver toxicity presentation, and less chemotherapy cycles were the main risk factors that predicted overall survival and progression-free survival. A higher percentage of HCV-positive patients’ treatment response was unable to be evaluated during the follow-up period (31.8% in HCV-positive vs. 14.7% in HCV-negative, p = 0.041). Nearly half (42.9%, three of seven) of them did not receive any treatment due to elderly age with poor performance status and severe liver cirrhosis; the rest of the patients received treatment of no more than two cycles with no possible follow-up image due to sepsis-related death. This highlighted the ability of HCV co-morbidity to hamper treatment completeness. The number of chemotherapy cycles was a major predictive factor for OS and HCV-positive patients; fewer chemotherapy cycles resulted in poor survival in the HCV-positive group. The phenomenon was also observed by Dlouhy et al., who presented higher number of complications and early deaths in HCV-positive DLBCL patients [33].
Regarding the HCV-positive DLBCL population, we found that liver cirrhosis was the main factor related to death events. Unlike the whole DLBCL population, age, stage, and performance status carried no vital role in predicting survival in HCV-positive DLBCL patients. Although fewer chemotherapy cycles, higher percentage of severe liver toxicity, and treatment delay/discontinuation were observed among those patients that died, the difference was not statistically significant compared to those who remained alive. While these findings highlighted the influence of the severity of liver cirrhosis for HCV-positive DLBCL patients, the small sample size might limit the statistical findings. This result highlighted that the severity of cirrhosis was the major factor to determine survival in HCV-positive DLBCL patients and also contributed to high complications and early deaths in HCV-positive DLBCL patients.
We found higher liver toxicity and higher ≥ grade 3 liver toxicity in HCV-positive population, and the incidence of liver toxicity was not associated with initial GPT levels. One patient (4.5%, one of 22 HCV-positive) received peg-interferon and ribavirin treatment for high HCV RNA levels after chemoimmunotherapy. Since we did not have the comprehensive data of HCV viral load, it was difficult to be conclude as a HCV reactivation in our study. The increased risk of liver toxicity in the HCV-positive population was consistent with other studies. In a Japanese multicenter retrospective study of 553 DLBCL patients, HCV RNA levels significantly increased in the HCV-positive patients during immunochemotherapy, and more grade 3–4 hepatic toxicity (27%) was observed in patients with HCV-positive patients than HCV-negative patients (3%) [17]. In another Italian retrospective study of 156 HCV-positive DLBCL patients, they found that 85% of patients completed their therapeutic program without any interruption or dosage reduction and five patients (4%) had to discontinue chemotherapy due to severe hepatic function impairment (toxicity grade 3–4) [35]. These studies revealed that without initial liver dysfunction, HCV-positive patients experience a similar outcome compared to HCV-negative patients when treated with chemotherapy or immunotherapy [36]. Therefore, careful monitoring of hepatic function and viral load is suggested during the therapy, especially for those with initial liver dysfunction patients.
Some studies showed that DAA treatment could reverse the liver inflammation, fibrosis, and decrease the complications of compensated liver cirrhosis in chronic hepatitis C [37,38,39]. From the observation report of Occhipinti et al., seven DLBCL patients with HCV infection received different DAA regimens concurrently with immunochemotherapy [40]. All patients completed their scheduled treatment with no liver toxicity occurrence. Thus, the concomitant administration of DAA and immunochemotherapy for HCV-positive DLBCL seems safe and may prevent immunochemotherapy-induced liver toxicity. Further prospective studies are warranted to identify if concurrent DAA therapy with chemoimmunotherapy in HCV-positive DLBCL patients can lead to survival benefit, especially for those with advanced fibrosis.
There were some limitations in this study. First, our study was a retrospective study with limited numbers of HCV patients, which limits the scope of our conclusion. Secondly, HCV viral load was not routinely checked during 2011–2015. Therefore, we did not have the comprehensive data of HCV viral load in all HCV-positive patients. Thus, we only used the anti-HCV positivity as criteria for being HCV-positive. Thirdly, some patients had a shortened course of therapy due to mortalities, which hinders proper response evaluations, especially in HCV-positive patients. However, this study presents real-world data of clinical characteristics and outcomes of HCV-positive DLBCL patients before the introduction of DAA treatment and provided the association of HCV and DLBCL.
5. Conclusions
In our study, DLBCL patients have a higher prevalence (10.7%) of HCV infection than the general population in our area. Clinically, HCV-positive DLBCL patients had poor performance status, lower platelet count, higher probability of spleen and liver involvement, and received less chemotherapy cycles due to morbidity and mortality. With multivariate analysis, stage, performance status, liver toxicity presentation, and chemotherapy cycles were found to be the major factors for the OS and PFS in DLBCL patients. Inferior survival of HCV-positive DLBCL was contributed by poor performance status, higher numbers of complications, and intolerance of treatment, leading to fewer therapy. Severity of liver fibrosis was the main factor related to death in the HCV-positive population. As such, anti-HCV therapy such as direct-acting antiviral agents might benefit such patients in the future to improve outcomes.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jpm11090844/s1, Table S1: the details of HCV-positive DLBCL patients.
Author Contributions
Conceptualization, Y.-F.T., H.-H.H., Y.-C.L. and C.-I.Y.; methodology, Y.-F.T., C.-I.Y. and P.-F.W.; validation, C.-I.H., C.-F.H., M.-L.Y. and C.-Y.D.; resources, T.-J.Y., Y.-C.G., J.-S.D., H.-C.W., S.-F.C., Y.-F.T., H.-H.H. and Y.-C.L.; data curation, T.-M.C. and Y.-L.K.; writing—original draft preparation, Y.-F.T. and H.-H.H.; writing—review and editing, Y.-F.T., H.-H.H. and C.-M.H.; supervision, P.-F.W., C.-I.H., C.-F.H., M.-L.Y. and C.-Y.D.; project administration, Y.-F.T. and H.-H.H. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by a grant from the Kaohsiung Medical University Hospital (KMUH108-8R24 and KMUH109-9R19) and Ministry of Health and Welfare (MOHW110-TDU-B-212-124006).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(I)-20210119).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data used in the present study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Defrancesco I., Zerbi C., Rattotti S., Merli M., Bruno R., Paulli M., Arcaini L. HCV infection and non-Hodgkin lymphomas: An evolving story. Clin. Exp. Med. 2020;20:321–328. doi: 10.1007/s10238-020-00615-6. [DOI] [PubMed] [Google Scholar]
- 2.Negro F., Forton D., Craxi A., Sulkowski M.S., Feld J.J., Manns M.P. Extrahepatic morbidity and mortality of chronic hepatitis C. Gastroenterology. 2015;149:1345–1360. doi: 10.1053/j.gastro.2015.08.035. [DOI] [PubMed] [Google Scholar]
- 3.Dammacco F., Lauletta G., Russi S., Leone P., Tucci M., Manno C., Monaco S., Ferrari S., Vacca A., Racanelli V. Clinical practice: Hepatitis C virus infection, cryoglobulinemia and cryoglobulinemic vasculitis. Clin. Exp. Med. 2019;19:1–21. doi: 10.1007/s10238-018-0536-z. [DOI] [PubMed] [Google Scholar]
- 4.Spinelli J.J., Lai A.S., Krajden M., Andonov A., Gascoyne R.D., Connors J.M., Brooks-Wilson A.R., Gallagher R.P. Hepatitis C virus and risk of non-Hodgkin lymphoma in British Columbia, Canada. Int. J. Cancer. 2008;122:630–633. doi: 10.1002/ijc.23105. [DOI] [PubMed] [Google Scholar]
- 5.de Sanjose S., Benavente Y., Vajdic C.M., Engels E.A., Morton L.M., Bracci P.M., Spinelli J.J., Zheng T., Zhang Y., Franceschi S., et al. Hepatitis C and non-Hodgkin lymphoma among 4784 cases and 6269 controls from the International Lymphoma Epidemiology Consortium. Clin. Gastroenterol. Hepatol. 2008;6:451–458. doi: 10.1016/j.cgh.2008.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Viswanatha D.S., Dogan A. Hepatitis C virus and lymphoma. J. Clin. Pathol. 2007;60:1378–1383. doi: 10.1136/jcp.2007.051870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mihaila R.G. Hepatitis C virus—associated B cell non-Hodgkin’s lymphoma. World J. Gastroenterol. 2016;22:6214–6223. doi: 10.3748/wjg.v22.i27.6214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Suarez F., Lortholary O., Hermine O., Lecuit M. Infection-associated lymphomas derived from marginal zone B cells: A model of antigen-driven lymphoproliferation. Blood. 2006;107:3034–3044. doi: 10.1182/blood-2005-09-3679. [DOI] [PubMed] [Google Scholar]
- 9.Provencio M., Sanchez A., Bonilla F., Espana P. Association between hepatitis C virus and non-Hodgkin’s lymphomas. J. Clin. Oncol. 2006;24:3513. doi: 10.1200/JCO.2006.06.4329. [DOI] [PubMed] [Google Scholar]
- 10.Zignego A.L., Ramos-Casals M., Ferri C., Saadoun D., Arcaini L., Roccatello D., Antonelli A., Desbois A.C., Comarmond C., Gragnani L., et al. International therapeutic guidelines for patients with HCV-related extrahepatic disorders. A multidisciplinary expert statement. Autoimmun. Rev. 2017;16:523–541. doi: 10.1016/j.autrev.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 11.Arcaini L., Bruno R. Hepatitis C virus infection and antiviral treatment in marginal zone lymphomas. Curr. Clin. Pharm. 2010;5:74–81. doi: 10.2174/157488410791110751. [DOI] [PubMed] [Google Scholar]
- 12.Sehn L.H., Salles G. Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2021;384:842–858. doi: 10.1056/NEJMra2027612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Besson C., Canioni D., Lepage E., Pol S., Morel P., Lederlin P., Van Hoof A., Tilly H., Gaulard P., Coiffier B., et al. Characteristics and outcome of diffuse large B-cell lymphoma in hepatitis C virus-positive patients in LNH 93 and LNH 98 Groupe d’Etude des Lymphomes de l’Adulte programs. J. Clin. Oncol. 2006;24:953–960. doi: 10.1200/JCO.2005.01.5016. [DOI] [PubMed] [Google Scholar]
- 14.Park B.B., Kim J.S., Lee Y.Y., Kang H.J., Ryoo B.Y., Kang J.H., Kim H.Y., Kim B.S., Oh S.Y., Kwon H.C., et al. Clinical characteristics and outcome for hepatitis C virus-positive diffuse large B-cell lymphoma. Leuk. Lymphoma. 2008;49:88–94. doi: 10.1080/10428190701732861. [DOI] [PubMed] [Google Scholar]
- 15.Visco C., Finotto S. Hepatitis C virus and diffuse large B-cell lymphoma: Pathogenesis, behavior and treatment. World J. Gastroenterol. 2014;20:11054–11061. doi: 10.3748/wjg.v20.i32.11054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Merli M., Visco C., Spina M., Luminari S., Ferretti V.V., Gotti M., Rattotti S., Fiaccadori V., Rusconi C., Targhetta C., et al. Outcome prediction of diffuse large B-cell lymphomas associated with hepatitis C virus infection: A study on behalf of the Fondazione Italiana Linfomi. Haematologica. 2014;99:489–496. doi: 10.3324/haematol.2013.094318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ennishi D., Maeda Y., Niitsu N., Kojima M., Izutsu K., Takizawa J., Kusumoto S., Okamoto M., Yokoyama M., Takamatsu Y., et al. Hepatic toxicity and prognosis in hepatitis C virus-infected patients with diffuse large B-cell lymphoma treated with rituximab-containing chemotherapy regimens: A Japanese multicenter analysis. Blood. 2010;116:5119–5125. doi: 10.1182/blood-2010-06-289231. [DOI] [PubMed] [Google Scholar]
- 18.Sun C.A., Chen H.C., Lu C.F., You S.L., Mau Y.C., Ho M.S., Lin S.H., Chen C.J. Transmission of hepatitis C virus in Taiwan: Prevalence and risk factors based on a nationwide survey. J. Med. Virol. 1999;59:290–296. doi: 10.1002/(SICI)1096-9071(199911)59:3<290::AID-JMV5>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 19.Chen Y.Y., Chen C.L., Chen J.W., Hsu N.T., Wei S.T., Hou S.M., Lu S.N., Chen P.J. Secular Trends and Geographic Maps of Hepatitis C Virus Infection among 4 Million Blood Donors in Taiwan from 1999 to 2017. Hepatol. Commun. 2020;4:1193–1205. doi: 10.1002/hep4.1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sun C.A., Chen H.C., Lu S.N., Chen C.J., Lu C.F., You S.L., Lin S.H. Persistent hyperendemicity of hepatitis C virus infection in Taiwan: The important role of iatrogenic risk factors. J. Med. Virol. 2001;65:30–34. doi: 10.1002/jmv.1097. [DOI] [PubMed] [Google Scholar]
- 21.Chuang S.-S., Liao Y.-L., Chang S.-T., Hsieh Y.-C., Kuo S.-Y., Lu C.-L., Hwang W.-S., Lin I.-H., Tsao C.-J., Huang W.-T. Hepatitis C virus infection is significantly associated with malignant lymphoma in Taiwan, particularly with nodal and splenic marginal zone lymphomas. J. Clin. Pathol. 2010;63:595–598. doi: 10.1136/jcp.2010.076810. [DOI] [PubMed] [Google Scholar]
- 22.Mele A., Pulsoni A., Bianco E., Musto P., Szklo A., Sanpaolo M.G., Iannitto E., De Renzo A., Martino B., Liso V., et al. Hepatitis C virus and B-cell non-Hodgkin lymphomas: An Italian multicenter case-control study. Blood. 2003;102:996–999. doi: 10.1182/blood-2002-10-3230. [DOI] [PubMed] [Google Scholar]
- 23.Giannoulis E., Economopoulos T., Mandraveli K., Giannoulis K., Nikolaides C., Zervou E., Papageorgiou E., Zoulas D., Tourkantonis A., Giannopoulos G., et al. The prevalence of hepatitis C and hepatitis G virus infection in patients with B cell non-Hodgkin lymphomas in Greece: A Hellenic Cooperative Oncology Group Study. Acta Haematol. 2004;112:189–193. doi: 10.1159/000081270. [DOI] [PubMed] [Google Scholar]
- 24.Oken M.M., Creech R.H., Tormey D.C., Horton J., Davis T.E., McFadden E.T., Carbone P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982;5:649–655. doi: 10.1097/00000421-198212000-00014. [DOI] [PubMed] [Google Scholar]
- 25.Vallet-Pichard A., Mallet V., Nalpas B., Verkarre V., Nalpas A., Dhalluin-Venier V., Fontaine H., Pol S. FIB-4: An inexpensive and accurate marker of fibrosis in HCV infection. comparison with liver biopsy and fibrotest. Hepatology. 2007;46:32–36. doi: 10.1002/hep.21669. [DOI] [PubMed] [Google Scholar]
- 26.Sterling R.K., Lissen E., Clumeck N., Sola R., Correa M.C., Montaner J., Sulkowski M.S., Torriani F.J., Dieterich D.T., Thomas D.L., et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317–1325. doi: 10.1002/hep.21178. [DOI] [PubMed] [Google Scholar]
- 27.The International Non-Hodgkin’s Lymphoma Prognostic Factors Project A Predictive Model for Aggressive Non-Hodgkin’s Lymphoma. N. Engl. J. Med. 1993;329:987–994. doi: 10.1056/NEJM199309303291402. [DOI] [PubMed] [Google Scholar]
- 28.Sehn L.H., Berry B., Chhanabhai M., Fitzgerald C., Gill K., Hoskins P., Klasa R., Savage K.J., Shenkier T., Sutherland J., et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood. 2007;109:1857–1861. doi: 10.1182/blood-2006-08-038257. [DOI] [PubMed] [Google Scholar]
- 29.Cheson B.D., Fisher R.I., Barrington S.F., Cavalli F., Schwartz L.H., Zucca E., Lister T.A. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014;32:3059–3068. doi: 10.1200/JCO.2013.54.8800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen C.-H., Yang P.-M., Huang G.-T., Lee H.-S., Sung J.-L., Sheu J.-C. Estimation of Seroprevalence of Hepatitis B Virus and Hepatitis C Virus in Taiwan from a Large-scale Survey of Free Hepatitis Screening Participants. J. Formos. Med. Assoc. 2007;106:148–155. doi: 10.1016/S0929-6646(09)60231-X. [DOI] [PubMed] [Google Scholar]
- 31.Machida K., Cheng K.T., Sung V.M., Shimodaira S., Lindsay K.L., Levine A.M., Lai M.Y., Lai M.M. Hepatitis C virus induces a mutator phenotype: Enhanced mutations of immunoglobulin and protooncogenes. Proc. Natl. Acad. Sci. USA. 2004;101:4262–4267. doi: 10.1073/pnas.0303971101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Saadoun D., Landau D.A., Calabrese L.H., Cacoub P.P. Hepatitis C-associated mixed cryoglobulinaemia: A crossroad between autoimmunity and lymphoproliferation. Rheumatology. 2007;46:1234–1242. doi: 10.1093/rheumatology/kem132. [DOI] [PubMed] [Google Scholar]
- 33.Dlouhy I., Torrente M., Lens S., Rovira J., Magnano L., Giné E., Delgado J., Balagué O., Martínez A., Campo E., et al. Clinico-biological characteristics and outcome of hepatitis C virus-positive patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Ann. Hematol. 2017;96:405–410. doi: 10.1007/s00277-016-2903-8. [DOI] [PubMed] [Google Scholar]
- 34.Chen Y.Y., Huang C.E., Liang F.W., Lu C.H., Chen P.T., Lee K.D., Chen C.C. Prognostic impact of hepatitis C virus infection in patients with diffuse large B-cell lymphoma treated with immunochemotherapy in the context of a novel prognostic index. Cancer Epidemiol. 2015;39:382–387. doi: 10.1016/j.canep.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 35.Visco C., Arcaini L., Brusamolino E., Burcheri S., Ambrosetti A., Merli M., Bonoldi E., Chilosi M., Viglio A., Lazzarino M., et al. Distinctive natural history in hepatitis C virus positive diffuse large B-cell lymphoma: Analysis of 156 patients from northern Italy. Ann. Oncol. 2006;17:1434–1440. doi: 10.1093/annonc/mdl131. [DOI] [PubMed] [Google Scholar]
- 36.Nooka A., Shenoy P.J., Sinha R., Lonial S., Flowers C.R. Hepatitis C reactivation in patients who have diffuse large B-cell lymphoma treated with rituximab: A case report and review of literature. Clin. Lymphoma Myeloma Leuk. 2011;11:379–384. doi: 10.1016/j.clml.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 37.Krassenburg L.A.P., Maan R., Ramji A., Manns M.P., Cornberg M., Wedemeyer H., de Knegt R.J., Hansen B.E., Janssen H.L.A., de Man R.A., et al. Clinical outcomes following DAA therapy in patients with HCV-related cirrhosis depend on disease severity. J. Hepatol. 2021;74:1053–1063. doi: 10.1016/j.jhep.2020.11.021. [DOI] [PubMed] [Google Scholar]
- 38.Hsu W.F., Lai H.C., Su W.P., Lin C.H., Chuang P.H., Chen S.H., Chen H.Y., Wang H.W., Huang G.T., Peng C.Y. Rapid decline of noninvasive fibrosis index values in patients with hepatitis C receiving treatment with direct-acting antiviral agents. BMC Gastroenterol. 2019;19:63. doi: 10.1186/s12876-019-0973-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cheng C.H., Chu C.Y., Chen H.L., Lin I.T., Wu C.H., Lee Y.K., Hu P.J., Bair M.J. Direct-acting antiviral therapy of chronic hepatitis C improves liver fibrosis, assessed by histological examination and laboratory markers. J. Med. Assoc. 2021;120:1259–1268. doi: 10.1016/j.jfma.2020.11.018. [DOI] [PubMed] [Google Scholar]
- 40.Occhipinti V., Farina L., Vigano M., Capecchi M., Labanca S., Fanetti I., Corradini P., Rumi M. Concomitant therapy with direct-acting antivirals and chemoimmunotherapy in HCV-associated diffuse large B-cell lymphoma. Dig. Liver Dis. 2019;51:719–723. doi: 10.1016/j.dld.2018.10.019. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data used in the present study are available from the corresponding author upon reasonable request.