Abstract
Background
Inflammation underlies both the pathogenesis and prognosis in patients with acute aortic dissection (AAD). This study aimed to assess the association of ICU admission of white blood cell count (WBCc) with post-discharge mortality in these patients.
Methods
Clinical data were extracted from the MIMIC-III V1.4 database. After adjusted to covariables, Cox regression analysis and Kaplan–Meier survival curve were performed to determine the relationship between WBCc on admission and post-discharge mortality (30-day, 90-day, 1-year and 5-year) in AAD patients. Subgroup analysis and receiver operating characteristic (ROC) curve analysis were used to test the performance of WBCc in predicting mortality in AAD patients.
Results
A total of 325 eligible patients were divided into 2 groups: normal-WBCc group (≤ 11 k/uL) and high-WBCc group (> 11 K/uL). In univariate Cox regression analysis, high WBCc was significant risk predictor of 30-day, 90-day, 1-year and 5-year mortality [hazard ratio (HR), 95% CI, P 2.58 1.36–4.91 0.004; 3.16 1.76–5.70 0.000; 2.74 1.57–4.79 0.000; 2.10 1.23–3.54 0.006]. After adjusting for age and other risks, high WBCc remained a significant predictor of 30-day, 90-day and 1-year mortality in AAD patients (HR, 95% CI, P 1.994 1.058–3.76 0.033; 2.118 1.175–3.819 0.013; 2.37 1.343–4.181 0.003). The area under ROC curve of WBCc for predicting 30-day, 90-day, 1-year and 5-year mortality were 0.69, 0.70, 0.66 and 0.61, respectively. The results from subgroups analysis showed that there was no interaction in most strata and patients who were younger than 69 years of age or had history of respiratory disease with an elevated WBCc had an excess risk of 30-day mortality (HR, 95% CI, P 3.18 1.41–7.14 0.005; 3.84 1.05–14.13 0.043).
Conclusions
Higher than normal WBCc on admission may predict post-discharge mortality in patients with AAD.
Keywords: White blood cell count, Acute aortic dissection, Post-discharge mortality, MIMIC-III database
Background
Acute aortic dissection (AAD) is a devastating cardiovascular disease with urgent onset, rapid progression, and high mortality [1]. Statistics figures showed that the mortality rate of AAD was an increase of 1–2% per hour after the onset of symptoms [2], and was ranged from 36 to 72% in the intensive care unit (ICU) during first 48 h [3]. Thus, it is of great significance for risk stratification and management to evaluate the risks factors for clinical outcomes in AAD patients.
Inflammation is involved in the occurrence and development of AAD [4, 5]. In recent years, some studies showed that inflammatory reactants such as D-dimer [6], C-reactive protein (CRP) [7], platelet count (PLTc) [8] and fibrinogen [9] were associated with the clinical outcomes of AAD patients, but these results are controversial and need to be further verified in larger population and longer follow-up time. White blood cell count (WBCc) is a common clinical biochemical index to reflect acute inflammation, which has been used to detect vascular inflammation and predict cardiovascular risk [10]. Recently, elevated WBCc on admission was found to be related to increased in-hospital death in patents with AAD [11, 12]. However, the relationship between it and post-discharge mortality were poorly defined. This study aimed to evaluate and analyze the prognostic of WBCc on post-discharge mortality among AAD patients.
Methods
This was a retrospective study based on a publicly available Medical Information Mart for Intensive Care (MIMIC) III database. It is a large, single-center database containing comprehensive medical information for more than 60,000 ICU admissions at Beth Israel Deaconess Medical Center (BIDMC) in Boston, Massachusetts from 2001 to 2012 [13]. MIMIC-III data are Health Insurance Portability and Accountability Act of 1996 (HIPAA) compliant, and all investigators with data access (MEG, RG) were approved by PhysioNet. Information available in MIMIC-III includes general information (i.e., demographics, insurance, ethnicity, etc.), treatment process (i.e., charted clinical observations, laboratory tests, physiological scores, medications, surgery, etc.) and survival data.
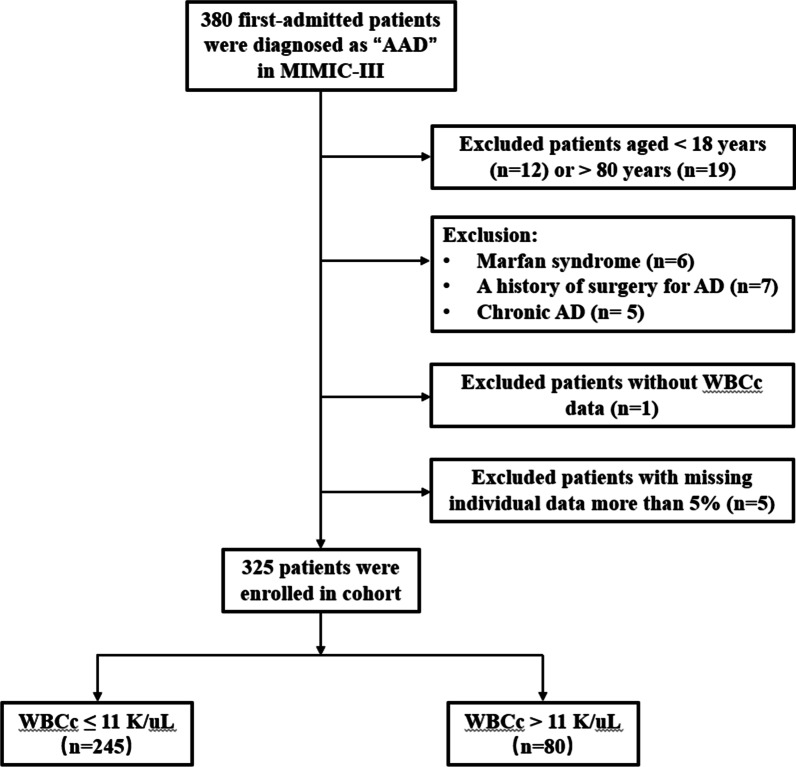
We included patients with AAD including both Stanford type A and type B based on the International Classification of Diseases 9th Edition (ICD-9) code in MIMIC-III database. Of these patients, we excluded those including: (1) For the adult study and avoiding more confounding factors, patients aged < 18 years or > 80 years were excluded; (2) patients who had a clear etiology, such as Marfan syndrome, iatrogenic aortic dissection (AD) secondary to cardiac surgery, a history of surgery for AD, or chronic AD; (3) no WBCc data; (4) missing individual data including demographics, laboratory tests, comorbidities, etc. more than 5%. Enrolled AAD patients were divided into 2 groups according to the admission WBCc > 11 K/uL and ≤ 11 K/uL as a cut-off value for normal. The complete process was shown in Fig. 1.
Fig. 1.
Study flow chart in the present study. AAD: acute aortic dissection; AD: aortic dissection; WBCc: white blood cell count
Data extraction was performed through Structured Query Language (SQL) with PostgreSQL 9.6. Baseline characteristics after ICU admission were collected, including demographics (age, gender, ethnicity, etc.), vital signs, laboratory tests, comorbidities, severity score and other data. Vital signs included systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean blood pressure (MBP). The laboratory parameters including red blood cell (RBC), red cell distribution width (RDW), hemoglobin (HB), hematocrit (HCT), platelet (PLT); activated partial thromboplastin time (APTT), prothrombin tmie (PT), international normalized ratio (INR); blood urea nitrogen (BUN), Creatinine (Cr); glucose; serum potassium, sodium, chlorine, magnesium, and total calcium were measured during the admission. Comorbidities including hypertension; diabetes; hypercholesterolemia (HC); valvular disease; stroke; coronary artery disease (CAD); congestive heart failure (CHF); atrial fibrillation (AF); liver disease; respiratory disease, acute kidney injury (AKI) and renal replacement therapy (RRT) were also collected for analysis based on the recorded ICD-9 codes in the MIMIC-III database. Severity score included sequential organ failure assessment (SOFA) score; simplified acute physiology score (SAPS II); oxford acute severity of illness score (OASIS) and systemic inflammatory response syndrome (SIRS) score.
Shapiro–Wilk tests were used to examine whether the continuous variables conform to the normal distribution. Normally distributed continuous variables were presented as the mean ± SD and non-normally distributed continuous variables were presented as the median and interquartile range (IQR). Categorical variables were presented by number and percentage. Continuous data were compared using Student t test or Mann–Whitney U test and categorical data were compared using chi-squared test as appropriate. Survival rates within normal-WBCc and high-WBCc groups were compared by the Kaplan–Meier curve and the log-rank tests. Cox regression models were conducted to evaluate the predictive value of WBCc in post-discharge including 30-day, 90-day, 1-year and 5-year mortality with hazard ratios (HRs) and 95% confidence intervals (CIs). Subgroup analyses were conducted to evaluate the WBCc and 30-day morality in different subgroups, including gender; age; hypertension; diabetes; HC; valvular disease; CHF; AF; liver disease; respiratory disease; AKI and RRT. ROC curve analyses and calculation of AUC were used to examine the performance of WBCc in predicting morality. A P value < 0.05 was considered statistically significant. All of the statistical analyses were performed by the EmpowerStats ver 2.17.8 (http://www.empowerstats.com/cn/, X&Y solutions, Inc., Boston, MA) and R software vers 3.42. The raw data showed in this study are fully available in MIMIC-III database.
Results
After reviewing the data of 380 AAD patients, a total of 325 eligible patients were enrolled in this study (detailed flow chart of patients’ selection shown in Fig. 1). The baseline characteristics of all patients are summarized in Table 1. The mean age of all patients was 68.0 (55.4–77.2) years, and 63.1% of patients (205/325) were male. According to admission WBCc, patients were divided into 2 groups including normal-WBCc group and high-WBCc group (≤ 11 K/uL; > 11 K/uL). Patients with an elevated WBCc had higher PLT, HCT, Hb, BUN and Glucose. Additionally, these patients had more CHF and higher SAPS II and SIRS scores (all P < 0.05).
Table 1.
Baseline characteristics of patients
| Characteristics | White blood cell count (K/uL) | P value | |
|---|---|---|---|
| ≤ 11 (n = 245) | > 11 (n = 80) | ||
| Demographics | |||
| Gender | 0.145 | ||
| Male | 160 (65.3%) | 45 (56.3%) | |
| Female | 85 (34.7%) | 35 (43.8%) | |
| Age, years | 69.0 (55.0–77.0) | 64.5 (55.0–77.0) | 0.378 |
| Insurance | 0.786 | ||
| Government | 16 (6.5%) | 4 (5.0%) | |
| Medicaid | 11 (4.5%) | 4 (5.0%) | |
| Medicare | 130 (53.1%) | 41 (51.3%) | |
| Private | 83 (33.9%) | 31 (38.8%) | |
| Selfpay | 5 (2.0%) | 0 (0.0%) | |
| Ethnicity | 0.846 | ||
| White | 166 (67.8%) | 56 (70.0%) | |
| Black | 32 (13.1%) | 12 (15.0%) | |
| Hispanic | 11 (4.5%) | 2 (2.5%) | |
| Others | 36 (14.7%) | 10 (12.5%) | |
| Dissection site | 0.152 | ||
| Thoracic | 143 (58.4%) | 38 (47.5%) | |
| Abdominal | 38 (15.5%) | 19 (23.8%) | |
| Thoracoabdominal | 64 (26.1%) | 23 (28.8%) | |
| Sanford type | 0.058 | ||
| A | 172 (70.2%) | 47 (58.7%) | |
| B | 73 (29.8%) | 33 (41.3%) | |
| Comorbidities | |||
| Hypertension | 142 (58.0%) | 50 (62.5%) | 0.473 |
| Diabetes | 19 (7.8%) | 8 (10.0%) | 0.528 |
| HC | 38 (15.5%) | 12 (15.0%) | 0.913 |
| Valvular disease | 22 (9.0%) | 11 (13.8%) | 0.220 |
| Stroke | 18 (7.3%) | 7 (8.8%) | 0.683 |
| CAD | 44 (18.0%) | 14 (17.5%) | 0.926 |
| CHF | 11 (4.5%) | 18 (22.5%) | 0.001 |
| Atrial fibrillation | 42 (17.1%) | 14 (17.5%) | 0.941 |
| Renal disease | 30 (12.2%) | 15 (18.8%) | 0.144 |
| Liver disease | 9 (3.7%) | 1 (1.3%) | 0.461 |
| Respiratory disease | 49 (20.0%) | 14 (17.5%) | 0.623 |
| AKI | 96 (39.2%) | 37 (46.3%) | 0.264 |
| RRT | 20 (8.2%) | 9 (11.3%) | 0.400 |
| Laboratory test | |||
| RBC, K/uL | 3.7 ± 0.6 | 3.8 ± 0.9 | 0.234 |
| PLT, K/uL | 154.0 (116.0–220.0) | 180.5 (136.5–265.5) | 0.021 |
| RDW, % | 14.4 (13.6–15.4) | 14.6 (13.7–15.3) | 0.449 |
| HCT, % | 28.0 (22.0–33.0) | 30.4 ± 6.9 | 0.006 |
| Hb, g/dL | 9.6 (7.6–11.5) | 10.3 ± 2.4 | 0.029 |
| BUN, mg/dL | 15.0 (12.0–20.0) | 20.0 (15.0–26.5) | < 0.001 |
| Creatinine, mg/dL | 1.0 (1.0–2.0) | 1.0 (1.0–2.0) | 0.064 |
| Chloride, mmol/L | 102.0 (100.0–105.0) | 102.4 ± 4.2 | 0.899 |
| Glucose, mg/dL | 100.0 (88.0–116.0) | 110.5 (91.0–126.0) | 0.013 |
| APTT, s | 28.4 (25.4–32.7) | 27.7 (24.4–33.0) | 0.571 |
| INR | 1.0 (1.0–1.0) | 1.0 (1.0–1.0) | 0.148 |
| PT, s | 13.4 (12.6–14.4) | 13.4 (12.4–14.8) | 0.769 |
| Sodium, mmol/L | 136.0 (134.0–139.0) | 136.6 ± 3.3 | 0.246 |
| Potassium, mmol/L | 3.6 (3.3–3.9) | 3.7 ± 0.6 | 0.187 |
| Total Ca2, mg/dL | 9.0 (8.0–9.0) | 8.5 (8.0–9.0) | 0.618 |
| Magnesium, mg/dL | 2.0 (1.9–2.3) | 2.0 (1.9–2.3) | 0.633 |
| SBP, mmHg | 151.0 (136.0–164.0) | 149.5 (134.5–164.5) | 0.859 |
| DBP, mmHg | 80.0 (72.0–89.0) | 82.0 (71.0–93.0) | 0.504 |
| MAP, mmHg | 102.0 (93.0–111.0) | 103.0 (92.5–114.0) | 0.590 |
| Severity score | |||
| SAPSII | 34.0 (27.0–41.0) | 40.5 ± 14.3 | 0.003 |
| OASIS | 32.0 (27.0–37.0) | 34.0 (26.0–41.5) | 0.173 |
| SOFA | 4.0 (2.0–6.0) | 4.0 (2.0–7.0) | 0.624 |
| SIRS | 2.0 (2.0–3.0) | 3.0 (3.0–4.0) | < 0.001 |
| Treatment | 0.366 | ||
| Medical | 58 (31.0%) | 22 (28.6%) | |
| Surgical | 84 (44.9%) | 30 (39.0%) | |
| Endovascular | 45 (24.1%) | 25 (32.5%) | |
Data are presented as mean ± SD, n (%), or medians (interquartile ranges). HC: hypercholesterolemia; CAD: coronary artery disease; CHF: congestive heart failure; AKI: acute kidney injury; RRT: renal replacement therapy; RBC: red blood cell; PLT: platelet; RDW: red cell distribution width; Hb: hemoglobin; HCT: hematocrit; BUN: blood urea nitrogen; APTT: activated partial thromboplastin time; PT: prothrombin time; INR: international normalized ratio; Total Ca: total calcium; SBP: systolic blood pressure; DBP: diastolic blood pressure; MBP: mean blood pressure; SAPS II: simplified acute physiology score; OASIS: oxford acute severity of illness score; SOFA: sequential organ failure assessment; SIRS: systemic inflammatory response syndrome
During the 5-year follow-up, 98 patients died. The overall 30-, 90-day and 1-, 5-year mortality rate were 14.8% (48/325), 18.8% (61/325), 22.8% (74/325) and 30.2% (98/325), respectively. In Fig. 2, the Kaplan–Meier survival curve indicated that the survival rate of high-WBCc group was significantly lower than normal-WBCc group during these periods (log-rank P < 0.01).
Fig. 2.
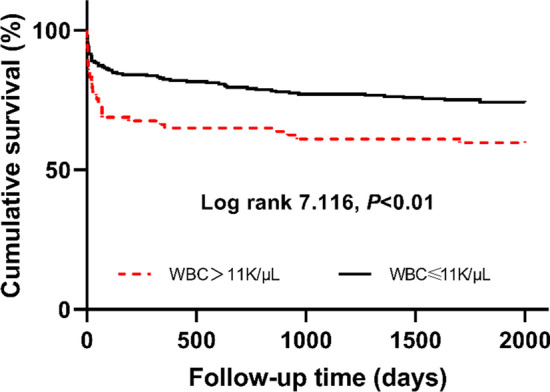
Kaplan–Meier survival curve for post-discharge survival stratified by admission white blood cell count. Survival rate were lower in high-WBCc group (log-rank P < 0.01); WBCc: white blood cell count
In order to explore the association between admission WBCc and post-discharge mortality, Cox regression analysis was performed and listed in Table 2. In the univariate Cox regression analysis, compared with the referent group (normal-WBCc: ≤ 11 K/uL), high WBCc was a significant predictor of 30-day, 90-day, 1-year and 5-year mortality in patients with AAD (HR, 95% CI, P 2.58 1.36–4.91 0.004; 3.16 1.76–5.70 0.000; 2.74 1.57–4.79 0.000; 2.10 1.23–3.54 0.006). In the multivariate Cox regression analysis, after adjusting to age, hypertension, valvular disease, stroke, CHF, atrial fibrillation, renal disease, BUN, type of AAD and treatment type, high-WBCc remained a significant predictor of 30-day, 90-day and 1-year mortality in AAD patients (HR, 95% CI, P 1.994 1.058–3.76 0.033; 2.118 1.175–3.819 0.013; 2.37 1.343–4.181 0.003), but not a predictor of 5-year mortality (HR: 1.499, 95% CI 0.932–2.411, P 0.095). Moreover, as shown in Table 3, the AUC of level in predicting 30-day, 90-day, 1-year and 5-year mortality were 0.69, 0.70, 0.66 and 0.61, respectively. Compared with other classic severity scores, WBCc showed a better performance than SIRS score.
Table 2.
Hazard Ratio (HR) (95% CIs) for mortality across groups of admission WBCc
| WBCc | Non-adjusted | Model 1 | ||
|---|---|---|---|---|
| HR (95% CIs) | P value | HR (95% CIs) | P value | |
| 30-day mortality WBCc, K/uL | ||||
| ≤ 11 K/uL | 1.0 (ref) | / | 1.0 (ref) | / |
| > 11 K/uL | 2.583 (1.361–4.905) | 0.004 | 1.994 (1.058–3.76) | 0.033 |
| 90-day mortality WBCc, K/uL | ||||
| ≤ 11 K/uL | 1.0 (ref) | / | 1.0 (ref) | / |
| > 11 K/uL | 3.161 (1.756–5.693) | < 0.001 | 2.118(1.175–3.819) | 0.013 |
| 1-year mortality WBCc, K/uL | ||||
| ≤ 11 K/uL | 1.0 (ref) | / | 1.0 (ref) | / |
| > 11 K/uL | 2.741 (1.569–4.789) | < 0.001 | 2.37 (1.343–4.181) | 0.003 |
| 5-year mortality WBCc, K/uL | ||||
| ≤ 11 K/uL | 1.0 (ref) | / | 1.0 (ref) | / |
| > 11 K/uL | 2.090 (1.234–3.541) | 0.006 | 1.499 (0.932–2.411) | 0.095 |
Non-adjusted model adjusted to: none;
Adjusted model 1 adjusted to: age, hypertension, valvular disease, stroke, CHF, atrial fibrillation, renal disease, BUN, type of AAD and treatment type
HR: Hazard Ratio; WBCc: White blood cell count; CHF: congestive heart failure; BUN: blood urea nitrogen
Table 3.
Area under receiver operating characteristic (ROC) curve of WBCc and severity scores
| Terms | AUC |
|---|---|
| 30-day mortality | |
| WBC | 0.691 |
| SAPSII | 0.796 |
| OASIS | 0.812 |
| SOFA | 0.697 |
| SIRS | 0.648 |
| 90-day mortality | |
| WBC | 0.697 |
| SAPSII | 0.779 |
| OASIS | 0.786 |
| SOFA | 0.716 |
| SIRS | 0.644 |
| 1-year mortality | |
| WBC | 0.659 |
| SAPSII | 0.745 |
| OASIS | 0.755 |
| SOFA | 0.677 |
| SIRS | 0.622 |
| 5-year mortality | |
| WBC | 0.610 |
| SAPSII | 0.701 |
| OASIS | 0.685 |
| SOFA | 0.626 |
| SIRS | 0.585 |
AUC: area under curve; P < 0.05
For further analysis, patients were divided into different subgroups (gender, age, hypertension, diabetes, HC, valvular disease, CHF, AF, liver disease, respiratory disease, AKI and RRT). As shown in Table 4, the results showed that there was no interaction in most strata (P for interaction = 0.13–1.00). Patients who were younger than 69 years of age or had a history of respiratory disease with an elevated WBCc had an excess risk of 30-day mortality (HR, 95% CI, P 3.18 1.41–7.14 0.005; 3.84 1.05–14.13 0.043).
Table 4.
Subgroup analysis of the association with admission WBCc and 30-day mortality
| Number of patients | WBCc | P for interaction | |||
|---|---|---|---|---|---|
| ≤ 11 K/uL | P value | > 11 K/uL | |||
| Gender | 0.676 | ||||
| Male | 205 | ref | 0.001 | 3.799 (1.695–8.513) | |
| Female | 120 | ref | 0.550 | 1.392 (0.471–4.113) | |
| Age | 0.013 | ||||
| < 69 | 167 | ref | 0.005 | 3.175 (1.412–7.141) | |
| ≥ 69 | 158 | ref | 0.405 | 1.609 (0.525–4.934) | |
| Hypertension | 0.260 | ||||
| No | 133 | ref | 0.017 | 3.179 (1.235–8.181) | |
| Yes | 192 | ref | 0.067 | 2.286 (0.943–5.543) | |
| Diabetes | 0.085 | ||||
| No | 298 | ref | 0.013 | 2.378 (1.204–4.694) | |
| Yes | 27 | ref | 0.119 | 5.100 (0.658–39.548) | |
| HC | 0.529 | ||||
| No | 275 | ref | 0.001 | 3.261 (1.636–6.500) | |
| Yes | 50 | ref | 0.524 | 0.485 (0.052–4.487) | |
| Valvular disease | 0.246 | ||||
| No | 292 | ref | 0.003 | 2.843 (1.416–5.709) | |
| Yes | 33 | ref | 0.774 | 1.275 (0.242–6.704) | |
| CHF | 0.119 | ||||
| No | 296 | ref | 0.017 | 2.348 (1.163–4.741) | |
| Yes | 29 | ref | 0.999 | / | |
| AF | 0.148 | ||||
| No | 269 | ref | 0.011 | 2.504 (1.230–5.098) | |
| Yes | 56 | ref | 0.153 | 2.960 (0.668–13.118) | |
| Liver disease | 1.000 | ||||
| No | 315 | ref | 0.002 | 2.738 (1.429–5.248) | |
| Yes | 10 | ref | 1.000 | 0.000 | |
| Respiratory disease | 0.005 | ||||
| No | 262 | ref | 0.024 | 2.369 (1.119–5.015) | |
| Yes | 63 | ref | 0.043 | 3.844 (1.046–14.127) | |
| AKI | 0.216 | ||||
| No | 192 | ref | 0.004 | 3.458 (1.473–8.121) | |
| Yes | 133 | ref | 0.256 | 1.761 (0.663–4.679) | |
| RRT | 0.129 | ||||
| No | 296 | ref | 0.008 | 2.519 (1.269–4.999) | |
| Yes | 29 | ref | 0.270 | 2.833 (0.445–18.042) | |
WBCc: White blood cell count; HC: hypercholesterolemia; CHF: congestive heart failure; AF: atrial fibrillation; AKI: acute kidney injury; RRT: renal replacement therapy
Discussion
This observational retrospective study based on a large sample cohort analyzed the relationship between admission WBCc in AAD patients and their clinical outcomes. Our results indicated that a high-WBCc on admission in patients with AAD was associated with poor clinical outcomes. After adjustment using a multivariate Cox analysis, the WBCc is an independent predictor to 30-day, 90-day and 1-year mortality. AUC analysis indicated that the WBCc had a better performance than SIRS score in predicting mortality in patients with AAD. Moreover, a subgroup analysis showed that high-WBCc on admission carried an excess risk of 30-days mortality in patients who were younger than 69 years of age or had a history of respiratory disease.
AAD is an acutely presenting, severe disease and its simple and effective biomarker for evaluating the prognosis is still lacking [14]. Inflammation promotes medial degradation of aortic artery and increases artery wall weakness and rupture [15, 16]. In recent decades, studies showed that several indicators of the inflammatory reaction including CRP level [17], D-dimer level [18] and PLTc [19] were associated with clinical outcomes in acute aortic syndrome (AAS). The WBCc is a sensitive and non-specific inflammation biomarker and its elevation also has been observed in AAD patients [11, 12, 20, 21]. However, the results of further studies on the association between the WBCc and prognosis of patients with AAD were inconsistent. A French study [20] with a Western cohort showed that the WBCc was not associated with in-hospital death in patients with AD (OR = 2.80, 95% CI 0.80–12.58, P = 0.12), but its sample size was relatively small (n = 94). Recently, two studies from China [11, 12] respectively found that, the WBCc could predict in-hospital, but failed to post-discharge mortality. The differences in underlying diseases, type of AD and sample quantity may partially explain the inconsistency.
In the present study, approximately a quarter of AAD patients showed an elevated WBCc on admission. These patients had higher PLT, HCT, Hb, BUN and Glucose, and had more CHF, higher SAPS II and SIRS scores. Moreover, a novel finding in our study was that the admission WBCc could predict post-discharge including 30-day, 90-day and 1-year mortality of AAD patients, which has rarely been reported before. The results from subgroup analysis and AUC analysis proved an excellent performance of the WBCc in predicting mortality in different periods in AAD patients. Compared with other classic severity scores, the WBCc showed a better performance than SIRS score. The progression of AAD significantly increases the level of vascular inflammation and the White blood cell (WBC) is an inflammatory reactant in the early stage of AAD. It has been proved that WBC can activate endothelial and microvascular damage, resulting in release of pro-inflammatory cytokines that contribute to a profound degradation of collagen and the extracellular matrix (ECM) related to smooth muscle cell (SMC) depletion, elastic fiber fragmentation and atherosclerosis underling aortic wall irreversible remodeling and weakness, which promote the progression of AAD and its postoperative recurrence [15]. In addition, clinical studies showed that an increased WBCc on admission was related to some serious postoperative complications, such as sepsis, hemorrhage, delirium, stroke and myocardial infarction, which might be one of the reasons for the poor prognosis and death [22–25]. In this study, a definite trend is evident that the incidence of malperfusion (including myocardial infarction, ischemic stroke and acute renal injury) in the high-WBCc group were higher than those in the normal-WBCc group, although there was no statistical difference (P = 0.094; P = 0.683; P = 0.264, respectively). Besides, the deaths in both groups were due to proximal or distal extension of dissection, aortic rupture, hemorrhage, pericardial tamponade, myocardial infarction, arch vessel occlusion causing stroke, visceral ischemia. These results are consistent with previous studies and suggests that the elevated WBC may affect clinical outcomes by promoting the progression of AAD and increasing complications. What’s more, our study showed that the mortality rates at 30-day, 90-day, 1-year and 5-year in high-WBCc group were significantly higher than those in the normal-WBCc group (P = 0.003; P < 0.001; P < 0.001; P = 0.006, respectively), but Kaplan Meyer survival curve indicated that the main drop in high-WBCc group was concentrated at the beginning of the curve. This is most likely due to the severity of the disease including malperfusion or comorbidities (e.g. CHF) in these patients as mentioned above. After initial drop, the survival curves in two groups didn’t separate further. This result suggested that admission WBCc were more associated with 30-day, 90-day and 1-year outcomes, which was also shown in the Cox regression analysis.
In the subgroup analysis, there was no interaction in most strata, which proved the reliability of the WBCc on admission predicting mortality in AAD patients. We also found that AAD patients who were younger than 69 years of age or had a history of respiratory disease with an elevated WBCc had an excess risk of 30-days mortality. On the one hand, we analyzed the data of major organ perfusion in young patients with the high-WBCc and elderly patients with high-WBCc. A definite trend is evident that the incidence of acute renal injury in the young patients were higher than elderly patients (50.0% vs. 41.2%, P = 0.434). It has long been known that malperfusion such as acute kidney injury was an independent predictor of mortality in AAD [26, 27]. Although there is no statistical difference in this result, it suggested that the higher risk of 30-day mortality in younger patients with high-WBCc may be associated with this complication, which need to be confirmed in a larger population. On the other hand, postoperative hypoxemia is quite common in patients with AAD and is associated with poor clinical outcomes [28]. Previous studies have shown that AAD patients complicated with respiratory diseases such as chronic obstructive pulmonary disease (COPD) are more likely to have hypoxemia or develop into acute respiratory failure after surgery, which has been proved to be an independent predictor of mortality in AAD [29, 30]. Besides, patients with respiratory diseases such as COPD, asthma and tuberculosis accompanied with an increased WBCc have a higher mortality rate [31, 32]. Perhaps, it could explain the reason why AAD patients who had a history of respiratory disease with an elevated WBCc had an excess risk of 30-days mortality. Our results indicated that more severe measures need to be taken in both of the above situations.
It is first time to reveal the potential value of the WBCc as a prognostic biomarker of post-discharge mortality in AAD patients. Combined with previous studies, our results provide further evidence of the utility of this stable and convenient indicator predicting prognosis in AAD patients. In the future, additional researches are needed to further understand the role of different types of WBC or some of their components in the prognosis of AAD patients, which provide the possibility for the application of targeted intervention in the treatment of AAD.
There are several limitations need to be mentioned in the study. Firstly, this study is a single-center observation research, which may not be universally representative. Secondly, we only focus on the admission WBCc. Observation of changes of the WBCc at different periods may provide more information in assessing its prognostic value. Thirdly, there are some missing data in this study, such as the timing of admission from onset, the severity of dissection and the surgical factors, which could also affect the WBCc. More on the above indicators evaluation and in-depth mechanism exploration should be conducted in the future.
Conclusions
In summary, the present study indicated that higher than normal WBCc on admission is an independent predictor for the post-discharge mortality in patients with AAD.
Acknowledgements
We would like to thank LT for statistical assistance.
Abbreviations
- AAD
Acute aortic dissection
- AD
Aortic dissection
- WBCc
White blood cell count
- ROC
Receiver operating characteristic
- ICU
Intensive care unit
- CRP
C-reactive protein
- PLTc
Platelet count
- MIMIC
Medical Information Mart for Intensive Care
- BIDMC
Beth Israel Deaconess Medical Center
- HIPAA
Health Insurance Portability and Accountability Act of 1996
- ICD-9
International Classification of Diseases 9th Edition
- SQL
Structured Query Language
- SBP
Systolic blood pressure
- DBP
Diastolic blood pressure
- MBP
Mean blood pressure
- RBC
Red blood cell
- RDW
Red cell distribution width
- HB
Hemoglobin
- HCT
Hematocrit
- PLT
Platelet
- APTT
Activated partial thromboplastin time
- PT
Prothrombin time
- INR
International normalized ratio
- BUN
Blood urea nitrogen
- Cr
Creatinine
- HC
Hypercholesterolemia
- CAD
Coronary artery disease
- CHF
Congestive heart failure
- AF
Atrial fibrillation
- AKI
Acute kidney injury
- RRT
Renal replacement therapy
- SOFA
Sequential organ failure assessment
- SAPS II
Simplified acute physiology score
- OASIS
Oxford acute severity of illness score
- SIRS
Systemic inflammatory response syndrome
- IQR
Interquartile range
- HRs
Hazard ratios
- CIs
Confidence intervals
- AAS
Acute aortic syndrome
- WBC
White blood cell
- ECM
Extracellular matrix
- SMC
Smooth muscle cell
- COPD
Chronic obstructive pulmonary disease
Authors' contributions
CYZ and ZLF contributed equally to this work. QX and XLC conceptualized this research aim, planned the analyses and guided the literature review. HB and XLC extracted the data from the MIMIC-III database. CYZ and ZLF participated in processing and analyzing the data. CYZ wrote the first draft of the paper. GQL and RZS revised and commented on the draft and overall responsibility. All authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
The datasets generated and/or analysed during the current study are available in the Physionet repository, https://physionet.org/physiobank /database/mimic 3cdb.
Declarations
Ethics approval and consent to participate
The establishment of this database was approved by the Massachusetts Institute of Technology (Cambridge, MA) and Beth Israel Deaconess Medical Center (Boston, MA), and consent was obtained for the original data collection. The data is publicly available (in the MIMIC-III database) hence ethical approval statement and the informed consent is not required for the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Chiyuan Zhang and Zuli Fu have contributed equally to this work.
Contributor Information
Xuliang Chen, Email: chenxuliang04@163.com.
Qian Xu, Email: elsiexu403@163.com.
References
- 1.Nienaber CA, Clough RE. Management of acute aortic dissection. Lancet (London, England) 2015;385(9970):800–811. doi: 10.1016/S0140-6736(14)61005-9. [DOI] [PubMed] [Google Scholar]
- 2.Nienaber CA, Eagle KA. Aortic dissection: new frontiers in diagnosis and management: part I: from etiology to diagnostic strategies. Circulation. 2003;108(5):628–635. doi: 10.1161/01.CIR.0000087009.16755.E4. [DOI] [PubMed] [Google Scholar]
- 3.Evangelista A, Isselbacher EM, Bossone E, Gleason TG, Eusanio MD, Sechtem U, et al. Insights from the international registry of acute aortic dissection: a 20-year experience of collaborative clinical research. Circulation. 2018;137(17):1846–1860. doi: 10.1161/CIRCULATIONAHA.117.031264. [DOI] [PubMed] [Google Scholar]
- 4.Nienaber CA, Clough RE, Sakalihasan N, Suzuki T, Gibbs R, Mussa F, et al. Aortic dissection. Nat Rev Dis Primers. 2016;2:16053. doi: 10.1038/nrdp.2016.53. [DOI] [PubMed] [Google Scholar]
- 5.Liu Z, Luo H, Zhang L, Huang Y, Liu B, Ma K, et al. Hyperhomocysteinemia exaggerates adventitial inflammation and angiotensin II-induced abdominal aortic aneurysm in mice. Circ Res. 2012;111(10):1261–1273. doi: 10.1161/CIRCRESAHA.112.270520. [DOI] [PubMed] [Google Scholar]
- 6.Mori K, Tamune H, Tanaka H, Nakamura M. Admission values of D-dimer and C-reactive protein (CRP) predict the long-term outcomes in acute aortic dissection. Internal Med (Tokyo, Japan) 2016;55(14):1837–1843. doi: 10.2169/internalmedicine.55.6404. [DOI] [PubMed] [Google Scholar]
- 7.Wen D, Du X, Dong JZ, Zhou XL, Ma CS. Value of D-dimer and C reactive protein in predicting inhospital death in acute aortic dissection. Heart (British Cardiac Society) 2013;99(16):1192–1197. doi: 10.1136/heartjnl-2013-304158. [DOI] [PubMed] [Google Scholar]
- 8.Li DZ, Chen QJ, Sun HP, Zeng R, Zeng Z, Gao XM, et al. Mean platelet volume to platelet count ratio predicts in-hospital complications and long-term mortality in type A acute aortic dissection. Blood Coagul Fibrinolysis Int J Haemost Thromb. 2016;27(6):653–659. doi: 10.1097/MBC.0000000000000449. [DOI] [PubMed] [Google Scholar]
- 9.Guan X, Gong M, Wang X, Zhu J, Liu Y, Sun L, et al. Low preoperative fibrinogen level is risk factor for neurological complications in acute aortic dissection. Medicine. 2018;97(21):e10830. doi: 10.1097/MD.0000000000010830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shah B, Baber U, Pocock SJ, Krucoff MW, Ariti C, Gibson CM, et al. White blood cell count and major adverse cardiovascular events after percutaneous coronary intervention in the contemporary era: insights from the PARIS Study (Patterns of Non-Adherence to Anti-Platelet Regimens in Stented Patients Registry) Circ Cardiovasc Interv. 2017 doi: 10.1161/CIRCINTERVENTIONS.117.004981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fan X, Huang B, Lu H, Zhao Z, Lu Z, Yang Y, et al. Impact of admission white blood cell count on short- and long-term mortality in patients with type a acute aortic dissection: an observational study. Medicine. 2015;94(42):e1761. doi: 10.1097/MD.0000000000001761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen ZR, Huang B, Lu HS, Zhao ZH, Hui RT, Yang YM, et al. Admission white blood cell count predicts short-term clinical outcomes in patients with uncomplicated Stanford type B acute aortic dissection. J Geriatr Cardiol JGC. 2017;14(1):49–56. doi: 10.11909/j.issn.1671-5411.2017.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Johnson AE, Pollard TJ, Shen L, Lehman LW, Feng M, Ghassemi M, et al. MIMIC-III, a freely accessible critical care database. Sci Data. 2016;3:160035. doi: 10.1038/sdata.2016.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mussa FF, Horton JD, Moridzadeh R, Nicholson J, Trimarchi S, Eagle KA. Acute aortic dissection and intramural hematoma: a systematic review. JAMA. 2016;316(7):754–763. doi: 10.1001/jama.2016.10026. [DOI] [PubMed] [Google Scholar]
- 15.Cifani N, Proietta M, Tritapepe L, Di Gioia C, Ferri L, Taurino M, et al. Stanford-A acute aortic dissection, inflammation, and metalloproteinases: a review. Ann Med. 2015;47(6):441–446. doi: 10.3109/07853890.2015.1073346. [DOI] [PubMed] [Google Scholar]
- 16.Xu H, Du S, Fang B, Li C, Jia X, Zheng S, et al. VSMC-specific EP4 deletion exacerbates angiotensin II-induced aortic dissection by increasing vascular inflammation and blood pressure. Proc Natl Acad Sci USA. 2019;116(17):8457–8462. doi: 10.1073/pnas.1902119116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vrsalović M, Vrsalović PA. Admission C-reactive protein and outcomes in acute aortic dissection: a systematic review. Croat Med J. 2019;60(4):309–315. doi: 10.3325/cmj.2019.60.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gorla R, Erbel R, Kahlert P, Tsagakis K, Jakob H, Mahabadi AA, et al. Diagnostic role and prognostic implications of D-dimer in different classes of acute aortic syndromes. Eur Heart J Acute Cardiovasc Care. 2017;6(5):379–388. doi: 10.1177/2048872615594500. [DOI] [PubMed] [Google Scholar]
- 19.Huang B, Tian L, Fan X, Zhu J, Liang Y, Yang Y. Low admission platelet counts predicts increased risk of in-hospital mortality in patients with type A acute aortic dissection. Int J Cardiol. 2014;172(3):e484–e486. doi: 10.1016/j.ijcard.2014.01.001. [DOI] [PubMed] [Google Scholar]
- 20.Shimada S, Nakamura H, Kurooka A, Nishioka N, Sugimura K, Ino H, et al. Fever associated with acute aortic dissection. Circ J. 2007;71(5):766–771. doi: 10.1253/circj.71.766. [DOI] [PubMed] [Google Scholar]
- 21.Wen D, Wu HY, Jiang XJ, Zhang HM, Zhou XL, Li JJ, et al. Role of plasma C-reactive protein and white blood cell count in predicting in-hospital clinical events of acute type A aortic dissection. Chin Med J. 2011;124(17):2678–2682. [PubMed] [Google Scholar]
- 22.Arnaoutoglou E, Kouvelos G, Tzimas P, Laou E, Bouris V, Papadopoulos G, et al. Relationship between normal preoperative white blood cell count and major adverse events after endovascular repair for abdominal aortic aneurysm: results of a pilot study. J Clin Anesth. 2017;36:201–205. doi: 10.1016/j.jclinane.2016.10.032. [DOI] [PubMed] [Google Scholar]
- 23.Garbens A, Wallis CJD, Bjarnason G, Kulkarni GS, Nathens AB, Nam RK, et al. Platelet to white blood cell ratio predicts 30-day postoperative infectious complications in patients undergoing radical nephrectomy for renal malignancy. Canadian Urological Association = journal Journal de l'Association des urologues du Canada. 2017;11(11):E414-e20. doi: 10.5489/cuaj.4478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Heredia-Rodríguez M, Bustamante-Munguira J, Lorenzo M, Gómez-Sánchez E, Álvarez FJ, Fierro I, et al. Procalcitonin and white blood cells, combined predictors of infection in cardiac surgery patients. J Surg Res. 2017;212:187–194. doi: 10.1016/j.jss.2017.01.021. [DOI] [PubMed] [Google Scholar]
- 25.Kotfis K, Ślozowska J, Safranow K, Szylińska A, Listewnik M. The practical use of white cell inflammatory biomarkers in prediction of postoperative delirium after cardiac surgery. Brain Sci. 2019;9(11):308. doi: 10.3390/brainsci9110308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Czerny M, Schoenhoff F, Etz C, Englberger L, Khaladj N, Zierer A, et al. The impact of pre-operative malperfusion on outcome in acute type A aortic dissection: results from the GERAADA registry. J Am Coll Cardiol. 2015;65(24):2628–2635. doi: 10.1016/j.jacc.2015.04.030. [DOI] [PubMed] [Google Scholar]
- 27.Tsai HS, Tsai FC, Chen YC, Wu LS, Chen SW, Chu JJ, et al. Impact of acute kidney injury on one-year survival after surgery for aortic dissection. Ann Thorac Surg. 2012;94(5):1407–1412. doi: 10.1016/j.athoracsur.2012.05.104. [DOI] [PubMed] [Google Scholar]
- 28.Ge H, Jiang Y, Jin Q, Wan L, Qian X, Zhang Z. Nomogram for the prediction of postoperative hypoxemia in patients with acute aortic dissection. BMC Anesthesiol. 2018;18(1):146. doi: 10.1186/s12871-018-0612-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Baldawi M, Baldawi M, Krafcik B, Al-Jubouri M, Markowiak S, Osman M, et al. Factors associated with increased mortality after isolated abdominal aortic dissection repair. Ann Vasc Surg. 2019;60:171–177. doi: 10.1016/j.avsg.2019.03.038. [DOI] [PubMed] [Google Scholar]
- 30.Khoynezhad A, Donayre CE, Smith J, Kopchok GE, Walot I, White RA. Risk factors for early and late mortality after thoracic endovascular aortic repair. J Thorac Cardiovasc Surg. 2008;135(5):1103–1109. doi: 10.1016/j.jtcvs.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 31.Li XC, Cao XP, Guo MZ, Xie M, Liu XS. Trends and risk factors of mortality and disability adjusted life years for chronic respiratory diseases from 1990 to 2017: systematic analysis for the Global Burden of Disease Study 2017. BMJ (Clin Res ed). 2020;368:m234. doi: 10.1136/bmj.m234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ruggiero C, Metter EJ, Cherubini A, Maggio M, Sen R, Najjar SS, et al. White blood cell count and mortality in the Baltimore Longitudinal Study of Aging. J Am Coll Cardiol. 2007;49(18):1841–1850. doi: 10.1016/j.jacc.2007.01.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analysed during the current study are available in the Physionet repository, https://physionet.org/physiobank /database/mimic 3cdb.