Abstract
Over the years, my colleagues and I have come to realise that the likelihood of pharmaceutical drugs being able to diffuse through whatever unhindered phospholipid bilayer may exist in intact biological membranes in vivo is vanishingly low. This is because (i) most real biomembranes are mostly protein, not lipid, (ii) unlike purely lipid bilayers that can form transient aqueous channels, the high concentrations of proteins serve to stop such activity, (iii) natural evolution long ago selected against transport methods that just let any undesirable products enter a cell, (iv) transporters have now been identified for all kinds of molecules (even water) that were once thought not to require them, (v) many experiments show a massive variation in the uptake of drugs between different cells, tissues, and organisms, that cannot be explained if lipid bilayer transport is significant or if efflux were the only differentiator, and (vi) many experiments that manipulate the expression level of individual transporters as an independent variable demonstrate their role in drug and nutrient uptake (including in cytotoxicity or adverse drug reactions). This makes such transporters valuable both as a means of targeting drugs (not least anti-infectives) to selected cells or tissues and also as drug targets. The same considerations apply to the exploitation of substrate uptake and product efflux transporters in biotechnology. We are also beginning to recognise that transporters are more promiscuous, and antiporter activity is much more widespread, than had been realised, and that such processes are adaptive (i.e., were selected by natural evolution). The purpose of the present review is to summarise the above, and to rehearse and update readers on recent developments. These developments lead us to retain and indeed to strengthen our contention that for transmembrane pharmaceutical drug transport “phospholipid bilayer transport is negligible”.
Keywords: membrane transport, pharmaceuticals, drugs, energy coupling, biotechnology, ADME, DMPK, transporter engineering
1. Introduction
Over the years, two main ideas have been used to explain the mechanisms by which pharmaceutical drugs and/or substrates and products of biotechnological interest pass through the plasma (or other) membranes of the relevant organism: in a more classical analysis, it is assumed (and it really is purely an assumption [1]) that molecules can diffuse through the core of the lipid bilayer by some means. In an alternative and really entirely opposite view, which we refer to as PBIN for “phospholipid bilayer diffusion is negligible” [2], we have argued [1,2,3,4,5,6,7,8,9,10,11,12] (and these constitute background that we mainly do not rehearse again here) that this does not in fact occur in real biomembranes to any significant extent at all. Some of this background material is also available in a webinar (https://bit.ly/3yQJ1FG, accessed on 15 September 2021). The two modes are illustrated in Figure 1. One may debate what is “significant” and/or “negligible”, but anything less than 5% of a total flux really is not much of a contribution, and plenty of evidence implies that it is almost certainly less than 1%. In our view, the term “negligible” covers this more than adequately. The purpose of this review and commentary is to provide an update on some of the salient issues. Examples of the importance of transporters continue to grow apace, while—whatever may or may not happen in artificial membranes—it remains the case that the actual evidence for significant transmembrane transport solely through any bilayer portions of intact biological membranes is non-existent. We shall start by looking at why this is the case, initially by focusing on some of the major differences between real biomembranes and those artificial ones made from phospholipid bilayers.
Figure 1.
A drug D might pass through a biological membrane in one of two main ways conceptually (we do not here discuss endo- and exocytosis; the focus is only on cases where the drug is considered to cross through the membrane barrier itself). On the left is illustrated transport through phospholipid bilayers, while on the right we illustrate the use of proteinaceous solute carriers to effect entry and exit of the drug. The crux of this review is that the mode on the left does not take place at any meaningful rate in intact biological membranes (since they have a high protein content).
1.1. Biological Membrane Structure
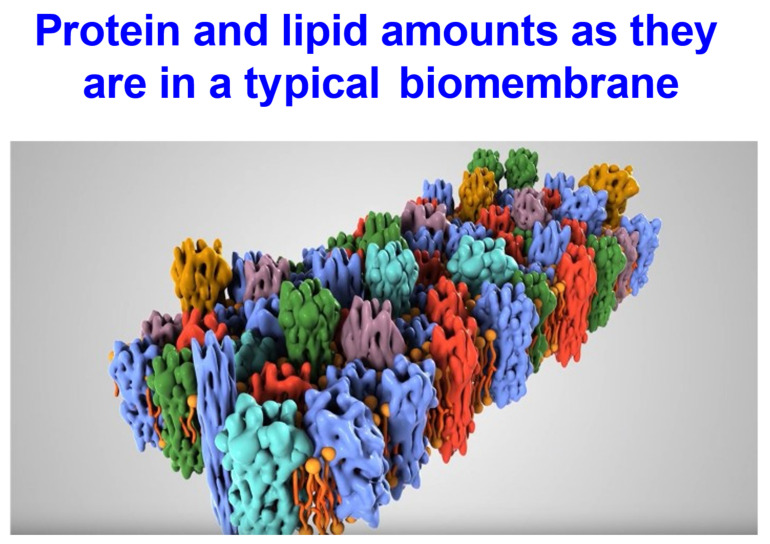
The textbook view of biological membranes is that they are to be seen as a “fluid mosaic” [13,14] of proteins embedded within a phospholipid bilayer, along with other small molecules such as sterols. The original article [13] featured a now iconic illustration (shown in Figure 3 of said article) in which a small number of proteins were embedded in and on a “sea” of phospholipids, with the proteins representing (by eye) approximately one-seventh of both the total mass and area. Unfortunately, this picture is very misleading, since biological membranes are commonly 3:1 protein:lipid by mass, and maybe 1:1 by area (not 1:7) [15,16,17,18,19,20,21,22,23,24,25,26,27,28] (see Figure 2). This widespread but erroneous mental picture, of “proteins floating in a sea of phospholipids”, has led many to suppose that from a biophysical point of view biomembranes are thus essentially just like pure lipid bilayers, in that the supposedly sparse proteins would do little or nothing to affect the kinds of properties that can be seen in purely phospholipid bilayers such as those studied in vitro [29,30,31]. However, this is emphatically not the case.
Figure 2.
Cartoon of a typical biomembrane indicating the relative paucity of phopholipid bilayer that is uninfluenced by proteins. Taken from an Open Access animation covering some of this ground and related transporter matters at https://www.youtube.com/watch?v=s23vNwLE-Jw.
Purely artificial (solely phospholipid) bilayer membranes are not terribly stable and admit the passage of small molecules (and even ions) via transient aqueous pores or channels (e.g., [32,33,34,35,36,37,38,39,40,41,42,43]). This mode of transfer clearly has to be the case if ostensibly ‘transmembrane’ transport ‘through’ them is observed, as it can easily be calculated that the free energy necessary for passing a small cation through a dielectric (such as that represented by a membrane interior of phospholipid tails) with a permittivity of ca 2 is so great that it would be unlikely to occur even over millions of years [44]. More specifically, the lateral flexing of phospholipids needed to make such transient aqueous pores simply cannot occur when the phospholipids are either bound directly to a protein or are strongly influenced thereby (as they are, e.g., [45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62]). This is likely reinforced by the well-established, substantial and dynamic lipid asymmetry between the inner and outer halves of membranes [63,64,65]. Note too that ABC transporters are responsible for moving lipids themselves around cells and within membranes [47,66,67,68,69]. In short, studying an aardvaark tells you about an aardvark, not a langoustine. In a similar way, studying sodium chloride tells you about sodium chloride, not strychnine chloride, even if they both contain chloride. Thus, in a similar way, studying pure phospholipid bilayers tells you about pure phospholipid bilayers, and not biomembranes that may happen to contain relatively small amounts phospholipids in a bilayer form. This may seem obvious when set out in this way, which is why we do it.
1.2. Transport of Drugs through Biological Membranes and the (Mis)Use of the Term “Passive”
It is now well-established that cells require proteinaceous transporters to effect the transmembrane transport of the nutrients they need to survive and to grow. Since these nutrients are mainly small molecules, it might be thought that a similar degree of acceptance would accord to the assumption that this held true for pharmaceutical drugs too, as well as for the uptake and efflux of substances of interest to the biotechnology industry. Surprisingly, as mooted above, and for some comparable reasons with bioenergetics more generally [70], this has not largely been the case. Indeed, why these and other beliefs persist despite the facts, and what to do about it, is of general philosophical and psychological interest [71,72,73,74,75,76]. In this case, it seems from our experience that it is to do with culture and education; those versed in physical organic chemistry (who are often predominant in the DMPK/ADMET sections of pharmaceutical companies but also in more traditional pharmacology schools in academia) tend not to know much about enzymology, while molecular biologists—who tend to know little of physical organic chemistry—express extreme surprise when told of the widespread belief of physical organic chemists that drugs simply pass through the bilayer portions of biomembranes. There is clearly value for individuals from both backgrounds in learning a little of each topic.
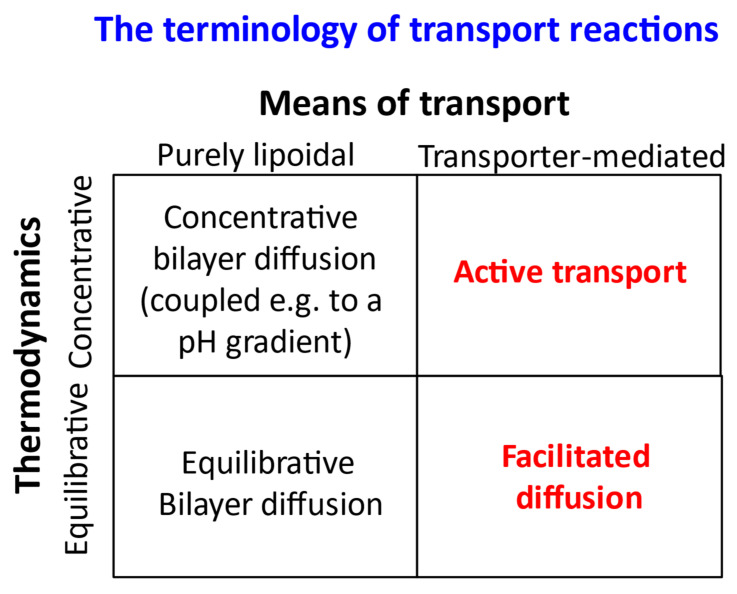
The term “passive”, as employed for instance in the phrase “passive diffusion”, is also widely misused, not least because it can be (and is) used to cover and then conflate two separate concepts. The first represents a thermodynamic meaning, where “passive” is used to mean “equilibrative”: the transporter requires no free energy and simply lets molecules pass down their concentration gradients until their concentrations (strictly, thermodynamic activities) are the same on each side of the membrane of interest (i.e., that in which the transporter is embedded). Clearly, the thermodynamic usage can (or should) have no mechanistic implications. The problem is that the same term (as in “passive diffusion” or “passive permeability”) is also used to imply a mechanism, viz that such equilibrative diffusion occurs through the phospholipid bilayer. In the worst cases, demonstration of the thermodynamic property is used to imply or even to claim the demonstration of a mechanism as transbilayer diffusion. In our view, the term “passive” has acquired so much baggage that the only solution to this (Figure 3 and reference [2]), in addition to education, is to avoid the term “passive” completely, and thus be forced to be more explicit about precisely what it is that is being claimed in any particular case.
Figure 3.
Suggested terminologies to avoid the use of the term “passive”, which is still widely misused to conflate two entirely separate concepts, one thermodynamic (hence independent of mechanism) and one mechanistic.
One feature of transport across membrane systems such as those of the popular Caco-2 cell monolayers [77,78,79,80,81,82,83,84], that (like other human tissues [85,86]; https://www.proteinatlas.org/search/slc), express hundreds of transporter proteins [87,88,89,90,91], is that the initial rate of transport of a given substrate at the same initial concentration from side A → side B can differ from that when the test is made from side B → side A, even when the transport is purely equilibrative. This has no ready explanation by or for those schooled purely in physico-chemical concepts and who believe that diffusion explains everything. By contrast, enzymologists have a perfectly straightforward explanation of this, which comes from the well-known thermodynamic Haldane relationship relating the Km and Vmax values in the forward (Km,f and Vmax,f) and reverse (Km,r and Vmax,r) directions of a reaction to its equilibrium constant Keq. While this can be found in any textbook of enzyme kinetics (e.g., [92,93,94]), or even of biochemistry, we reproduce it below:
| (Vmax,f · Km,r)/(Vmax,r · Km,f) = Keq | (1) |
Thus, even for Keq values of unity, it is easily possible to have rates that differ manyfold in the two directions for the same external concentration, just by manipulating the other values in Equation (1) while keeping their ratios consistent with it. Of course, and I stress this purposely, this is true only for enzyme- (transporter)-mediated transport, not “diffusion” through lipid bilayers [5].
1.3. Untestability of Bilayer Transport Models in Real Biomembranes
Perhaps surprisingly, the view that molecules pass through phospholipid bilayers in real biological membranes is presently untestable, because we do not have the ability to image small molecules and membranes with the atomic resolution that would be necessary to observe their transport directly. What is presently carried out most commonly is to observe molecules on one side of a membrane and later on the other side, and simply assume that they appeared there via transbilayer transport. This is a well-established and classical logical fallacy known as “affirming the precedent” or “post hoc ergo propter hoc” [1]. In a different vein, observing a molecule transferring from one side of a pure phospholipid bilayer simply does not tell you what it might do in a real membrane where there is very little bilayer that is not affected by the presence of protein. Similarly, changing the type or amount of phospholipid does not “simply” do that, because the activities of membrane proteins, including transporters [95], can vary quite substantially with changes in their adjacent lipids. Consequently, any changes in transport induced by changing lipids can perfectly well (and more accurately) be explained by their influences on the proteins embedded in the membrane.
1.4. Testability of Transporter-Mediated Models in Real Biomembranes
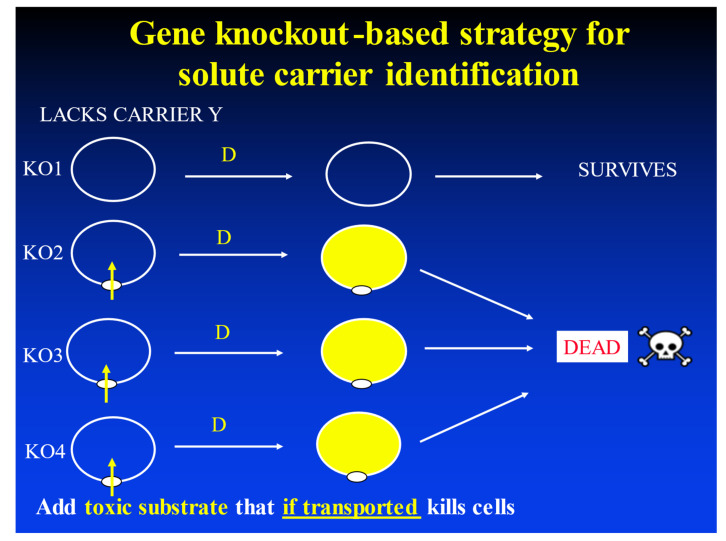
By contrast, those of us who claim that bilayer transport is negligible in real biomembranes can easily change the expression levels or activities of transporters of interest (e.g., [96,97]) and observe the concomitant effects on the transport of the small molecules of interest (as in Figure 4). (The same strategy can also be used to detect inhibitors of the transporter, e.g., [98,99].) In the paper of Winter et al. [97], the uptake and toxicity of an anticancer compound called YM155 or sepantronium bromide was decreased some 500-fold when the single transporter SLC35F2 was knocked out, and the toxicity of the compound correlated closely with that transporter’s expression level over some four orders of magnitude when explored in some 15 separate cell lines (that, as for many transporters [86,100], showed equivalently massively varying expression levels). The only reasonable (one might say plausible) explanation is that the SLC35F2 transporter normally carried some 99.5% of the flux of YM155 into the cell, and that consequently the transmembrane flux occurring by any other means, including via bilayer transport, is indeed negligible. This simple phenomenon served to explain entirely the highly variable efficacy of YM155 in a series of clinical trials.
Figure 4.
Principle of determining the substrate of a drug at toxic concentrations (e.g., [8,96,97,101,102]) by assessing the ability of cells lacking a particular transporter gene to survive its presence, while the wild-type cells, or cells knocked out for other genes not involved in the drug’s transport, are killed. Obviously, this is the extreme; there may also be degrees of resistance.
Typically, the question of the interaction between substrates and transporters can be phrased in two complementary ways: the transporter-centric question is “given a transporter of interest X, what are its substrates?”. This is the problem of de-orphanisation [103,104,105,106,107,108,109]. The complementary, substrate-centric, question is “given a substrate Y, which transporter(s) is/are responsible for its cellular uptake and/or efflux?”. The latter is the more important one for assessing the mechanisms of drug transport [8], and there are many examples (e.g., [110,111,112,113,114,115,116,117,118]) where transporter activity has been identified but where the transporters involved have not. Of course, while a known result is a partial answer to each question, the strategies for tackling them are slightly different (for a recent overview of cell-based assay methods for SLCs, see [119]). Note, in particular, that simple biophysics plus the re-use of protein motifs in evolution means that most small molecules bind to multiple targets, and most proteins can bind small molecules promiscuously (e.g., [8,9,120,121,122,123,124,125,126,127,128,129,130]).
As part of the EU-IMI ReSolute project [131] (https://re-solute.eu/), the CEMM group studied 60 cytotoxic compounds. By using an SLC-focused CRISPR-Cas9 library, they identified a series of transporters whose absence induced resistance to the drugs tested [101], much as had been achieved in smaller numbers before [96,97]. This kind of substrate-centric strategy clearly represents a potent means of identifying drug transporters, and notably it also illuminated cases of interactions. Such interactions scale exponentially with the number of candidates, but high-throughput CRISPR-Cas methods are enabling the identification of all kinds of genes involved in complex biological processes (e.g., [132,133,134,135,136,137]).
1.5. Heterogeneity of Transport and Transporters in Different Cells and Tissues
If trans-bilayer permeability were significant in real biomembranes, the free concentrations of drugs inside cells and organelles (modulo pH and potential gradients) would be more or less homogeneous. Of course, they are not, as is well-known (and as can easily be determined by chemical imaging methods, e.g., [138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159]). A particularly well-established case is that of the blood–brain barrier, where, despite the lipids not being noticeably different from those in other cells, the permeability is negligible if specific transporters are not involved [160,161,162,163,164]. What does differ greatly between different cells and tissues, of course, is the expression of particular proteins such as transporter proteins [85,86,165], with any number of large datasets now becoming available.
1.6. Role of Transporters in Biotechnology
In addition to activities mentioned in our previous reviews that were focused on transporters and biotechnology [4,12,166], a number of other authors (as reviewed, e.g., in [167,168,169,170,171,172,173,174,175,176,177,178]) have also highlighted the importance of transporters in the uptake of CO2 [179] and in the biotechnological production of substances of interest in the BioEconomy. Some recent examples include the production of amorphadiene [180], citrate [181], L-malate [182,183,184], antibiotics [185], fatty acids [186], fatty alcohols [187], olefins [188], various organic acids [176], other dicarboxylic acids [189,190], sophorolipids [173,186,191,192], in microbial fluorination (by removing a fluoride effluxer from E. coli [193]), and in the production of a variety of hydrophobic substances [191,194]. Promiscuity can be quite significant for biotechnology [182,195,196,197,198,199]. In particular, here, the promiscuity of some transporters, especially under biotechnological conditions of unphysiologically high intracellular concentrations of small molecules, means that cells can have a tendency to leak pathway intermediates with structural similarities (as judged by various means [200,201]) to products, rather than simply just excreting the desired product; this too can be manipulated via transporter engineering [171].
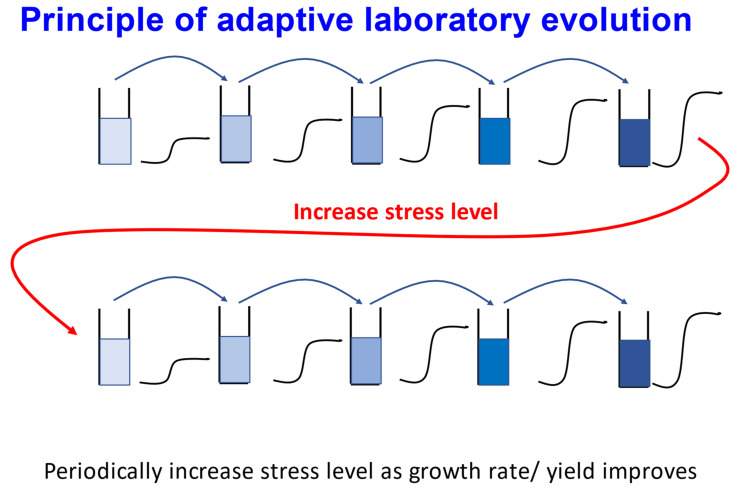
1.7. Adaptive Laboratory Evolution and Membrane Transporters
While some studies are more binary, looking for resistance to a toxic substrate via survival or death “in one go”, other strategies (such as the very elegant “variable dose analysis” [202]) are more graded. A particularly nice example is that of adaptive laboratory evolution (ALE), illustrated in Figure 5. While many very elegant examples of long-term laboratory-based bacterial evolution exist (e.g., [203,204,205,206,207,208,209,210]), a more directed focus has been where it is exploited for biotechnology (e.g., [190,211,212,213,214,215,216,217,218,219,220,221,222,223]). However, it is perfectly applicable in drug transporter studies too. As with any phenotypic selection of this type [224,225,226,227,228,229,230,231], it relies on the fact that, in a heterogeneous population, the faster-growing strains will tend to become more prevalent at the expense of the slower-growing ones. Sequencing those that take over indicates where favourable mutational events have occurred, and essentially identifies the relevant genes. It can best be run as a hypothesis-free strategy [232], given that some mutations cannot reasonably be predicted (e.g., the role of ribosomal subunits in improving methanol tolerance in E. coli [233]). In one example involving transporters [234], ALE was used to increase the tolerance to aromatic amino acids of baker’s yeast. Here, [234], the major mutation was in a transcriptional activator called Aro80, that served to increase the activity of an efflux transporter Espb6, a role (in effluxing aromatic compounds) shared with the previously known Pdr12. In another example [235], E. coli cells were made more tolerant to ionic liquids; in this case, the chief mutations in tolerant clones occurred in transport processes in the shape of mdtJI, a multidrug efflux pump, and yhdP, a largely uncharacterised transporter possibly involved [236,237] in phospholipid transport to the outer membrane.
Figure 5.
Illustration of the principle of adaptive laboratory evolution (ALE). Cells are exposed to a toxic substance that causes them to grow sub-optimally. Selection leads to strains that can revert to rates and extents of growth shown by the wild type when inoculated into fresh cultures. The stress level is increased and the process continued. Sampling and growth rate measurement can be completely automated.
As illustrated in Figure 5, the commonest means of performing ALE is in a “semi-batch” type of mode in which small inocula are used to grow cells in batch mode, while they are then sampled at the end of growth and a new inoculum introduced to a separate batch culture. What is then selected, in part, is cells that as well as having a higher growth rate also have lowered lag phases and the ability to survive better in stationary phase. Truly continuous cultures provide for a much more stringent selection, especially when carried out in a turbidostat. In a turbidostat (e.g., [227,238,239,240,241,242,243,244,245]), the growth medium is arranged such that—unlike in a chemostat—cells can grow at the maximum rate they are able to within it. The biomass in a fermentor of working volume V is controlled at a set point via a suitable probe (see, e.g., [246,247,248]). As it exceeds this level, fresh growth medium (including any inhibitors) is pumped in at an average rate over a period of v mL · min−1. Cells are washed out at the same rate. As usual [249], this rate is numerically equivalent to the growth rate = the dilution rate = v/V min−1, and may be recorded continuously. Rather surprisingly, the method has not been widely used, although McGeachy et al. [227] give a nice example that illustrated selection of mutations in the Mep3p ammonium transporter. We predict that these kinds of strategies may have much more impact in the future.
1.8. Substrate Misannotations and the Importance of Antiporter Activity in Drug Influx and Efflux
Most transporters are discovered via their effects on the uptake or efflux of a particular substrate of interest, and they are often codified accordingly. Our experience is that—just as with enzyme annotation more generally [250]—this leads to misannotation in that “any” activity discovered first is typically seen the main or even only activity. A classic example is SLC22A4, previously known as OCTN1. It had been found to catalyse the uptake of carnitine, and also that of the the non-physiological tetraethylammonium cation. However, the rates were in fact quite miserable, and it was not until Gründemann and colleagues used what was effectively an untargeted metabolomics approach [103] that it was discovered that it was in fact “really” a concentrative, sodium-coupled transporter for the nutraceutical ergothioneine [251,252] and also for the related stachydrine (proline betaine) [103,253,254]. Equivalently, this example also serves to illustrate how finding a transporter with one activity does not mean that it is the only such capability, and it is now known that at least one more transporter, viz SLC22A15 [109], can also catalyse ergothioneine uptake.
Another means of misannotation is that based on an assumption of unidirectionality (as an “influxer” or an “effluxer”) that is not warranted and follows from the fact that most kinetic assays are set up to measure either only an influx or an efflux. At one level, bidirectional transport is inevitably the case, in that most equilibrative transporters are necessarily functionally perfectly reversible; if a transporter has two uniported substrates A and B, both can pass in either direction depending on their relative concentration on either side of the membrane. More significantly, however, is the case in which the transporter is in fact an antiporter, where the transport of A in one direction is coupled to the transport of B in the other direction, whether B is measured or not. In radioisotopic assays for the uptake of A, B is usually unlabelled and hence unobserved. By contrast, so-called “untargeted” [255,256,257] mass spectrometric methods show clearly that there is a massive amount of efflux as well as influx when cells are exposed to new drug and nutrient sources [258]. In addition, the energy coupling elements of concentrative or efflux transporters are largely separate from those responsible for conducting the passage of the substrate through the membrane or can be made so [259].
“Multi-drug transporters” (MDTs) represent a particularly clear example of this; often, a transporter is labelled as an MDT involved in efflux because lowering its activity makes an organism more sensitive to a cytotoxic drug. However, in some cases (e.g., [260]), the removal of such a transporter actually makes the organism more sensitive to some other substances! Only “omics” methods in which many substances are measured simultaneously can easily disentangle this kind of behaviour.
1.9. Genome-Wide Analysis of Drug Uptake and Efflux in E. coli
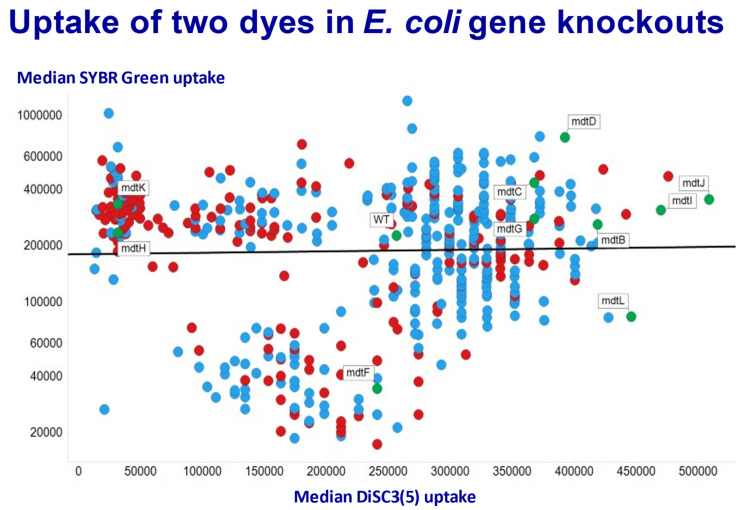
In recent work [261,262,263], we have recognised that the existence of genome-wide knockout (and overexpression) collections allows for the high-throughput assessment of the uptake of small molecule substrates. Since fluorophores are perfectly good surrogates for these purposes [264], and their uptake admits easy assessment using flow cytometry [265,266,267,268,269,270,271,272], we have been able to assess the influence of the expression of individual transporters on the uptake of various fluorophores. Figure 6 gives an example from E. coli [261], using a wild type and some 530 knockouts, of the effect of such knockouts on the steady-state uptake of two dyes, viz DiSC3(5), a carbocyanine dye responsive to membrane energisation [70], and SYBR Green, an intercalating dye whose fluorescence is massively enhanced upon binding to (especially double stranded [273]) DNA [274,275]. We here focus on the so-called multidrug transporter genes of the mdt family. Although the effect of knocking them out individually is, as expected [276], mainly (for mdt B,C,D,G,I, and J) to increase the steady-state uptake of DiSC3(5), that of mdtH and mdtK has the opposite effect, and uptake is very significantly decreased, indicating that normally these can act as influxers for this molecule. In a similar vein, the uptake of SYBR Green is lowered when mdtF and mdtL are knocked out.
Figure 6.
Dual roles of so-called multidrug transport proteins in E. coli. Data redrawn from those published with Open Access at [261]. Plotted are the median uptakes of SYBR Green and DiSC3(5) by various knockout strains relative to the Wild Type (WT). The ranges are, respectively, 70-fold and 36-fold. So-called y-genes (genes of nominally unknown function [278,279]) are encoded in red. Mdt gene knockouts are labelled (and have green symbols).
While one can never exclude the contribution of pleiotropic effects (e.g., [277]), the obvious conclusion from these kinds of genome-wide study is that many individual transporters can potentially contribute to the uptake and efflux of any stated substrate of interest. Where this is the case, knocking a single one out may not have measurable effects, since the others can “take up the slack” [276]; this does not of course then mean that the gene knocked out did not catalyse the flux of the substrate of interest—absence of evidence is not evidence of absence.
A convenient method for invoking genome-wide diversity via gene disruption is the use of transposon insertion, but this has the disadvantage that the inactivation of essential genes in haploid organisms is missed. Thus, Webber and colleagues [280] have developed a system (TraDIS-Xpress) based on a transposon linked to an inducible promoter and used this to determine transporters (and other genes) involved in resistance to triclosan [280] and to fosfomycin [281]. This kind of strategy will be vital in further uncovering transporter-mediated contributions to antimicrobial resistance (AMR).
1.10. Recent Approaches to the De-Orphanisation of Mammalian “Orphan” Transporters
Some 1000 genes in the human genome encode transporters [7], of which the largest class, amounting to roughly half [282,283,284,285,286,287,288], is represented by the SoLute Carrier (SLC) class (http://slc.bioparadigms.org/, accessed on 15 September 2021). As noted above, even for those that have at least one known substrate, there are doubtless many more to be found. More significantly, at this stage of knowledge, many of them are complete “orphans” in that not a single substrate is in fact known. One recent example of de-orphanisation is that of the mitochondrial transporter SLC25A51, which turns out [108,289] to be an NAD+ importer. This de-orphanisation hinged upon a successful combination of genomics, metabolomics, CRISPR-Cas9-mediated gene editing, and genetics. Other strategies include binding assays [107], the use of direct assays in Xenopus oocytes [290], and others that are covered in a recent and comprehensive review [119].
1.11. Selectivity and Drug Targeting by the Use and Exploitation of Varying Expression Profiles
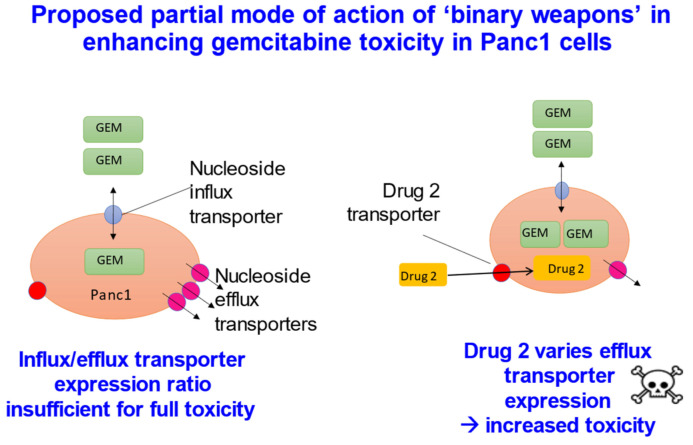
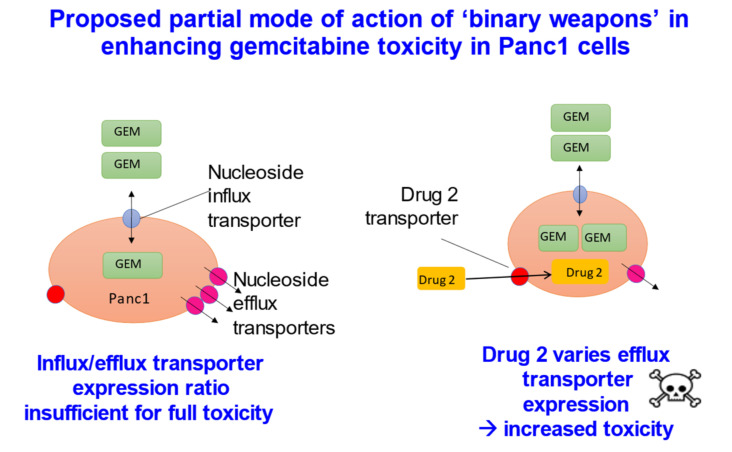
Given the need for transporters if drugs are to cross cell membranes, one can make a virtue of necessity [284] by seeking either to exploit their natural expression profiles [291,292,293] or to vary them explicitly [294], so as to target them to particular tissues [295]. The latter has obvious benefits in oncology [296,297,298,299,300], where the necessary cytotoxicity of many drugs, such as nucleoside analogues, is rather non-specific. The strategy can be used for modifying both influx and efflux transporters, though the latter is likely to prove more efficacious [276]. In one example [294] (Figure 7), the second drug in the “binary weapon” strategy decreased the expression of a relevant efflux transporter some 12-fold, in a cell-selective manner. This binary weapon strategy potentially holds much promise for targeting cytotoxic anti-cancer drugs.
Figure 7.
“Binary weapons” based on drug transporters [294]. The cytotoxic drug gemcitabine (GEM, a fluorinated cytosine nucleoside analogue) when added at a certain low concentration to Panc1 pancreatic cancer cells was barely cytotoxic (left panel). However, when a second drug was added, which was itself also non-toxic, the combination was substantially more toxic. What had occurred was that, in response to the GEM, the cells had increased the expression of the efflux transporter ABCC2 (MRP2) some 12-fold; the second drug inhibited this process, and in a cell-selective manner.
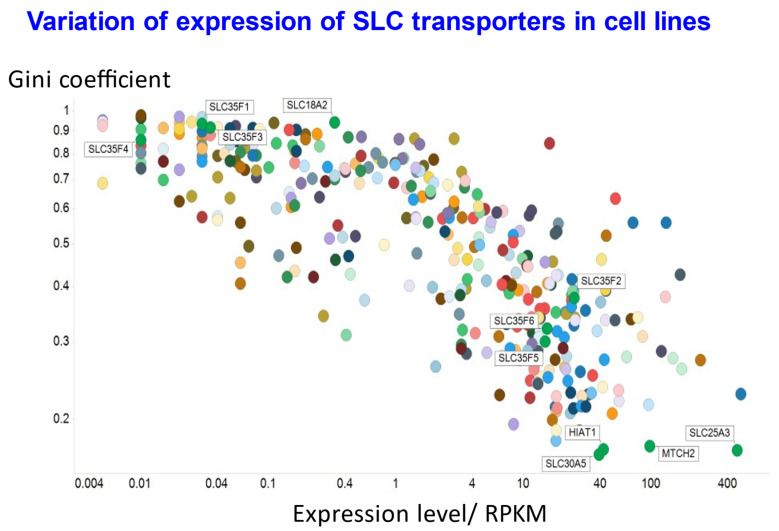
The fact that expression levels of SLCs often vary considerably was illustrated by us previously [86], using a publicly available dataset [85]. The Gini coefficient (see [86,100,301,302]) (Figure 8) describes the heterogeneity of a distribution in a simple, non-parametric manner, and takes values between zero and 1. Some of the cell line data are replotted in Figure 9, where the Gini coefficient is plotted against their median expression level as assessed by RNASeq. Specifically, we use these data to illustrate four points: (i) the very fact that there are a great many uptake transporters (at least 400), that may or may not be expressed in different cells; (ii) those transporters with the lowest Gini coefficient, indicating their relative homogeneity of expression between cell lines (which may be referred to as GiniGenes [86,100]); (iii) an example (in the form of SLC18A2, a vesicular monoamine transporter [303]) of a transporter with both a high Gini coefficient, indicating a very restricted expression, and a reasonably high expression level; and (iv) the substantial variation in Gini coefficient for the six members of the SLC35F family, with three being quite selectively expressed (SLC35F1,3,4) while the others (SLC35F2,5,6) are expressed over a very broad range of values, as shown explicitly elsewhere for SLC35F2 [97]. Overall, 61 of the 410 transporters (15%) plotted in Figure 9 have a Gini coefficient over 0.8, 105 (26%) exceed 0.7 and 173 (42%) exceed 0.5. By contrast, the Gini coefficient for income inequality in different countries (its usual domain of application [304]) https://www.indexmundi.com/facts/indicators/SI.POV.GINI/rankings shows only 14 countries/159 (9%) with a Gini coefficient exceeding 0.5.
Figure 8.
Illustration of the derivation of the non-parametric Gini coefficient for describing the inequality of a distribution (here the variation of transcript levels between cell lines). This was achieved by rank ordering the value of the different examples according to their expression levels, as indicated.
Figure 9.
Variation of Gini coefficient and median expression for 410 SLC transporters in 56 cell lines. Data were obtained from previous open access publications [85,86] and a subset replotted. A few transporters are labelled to illustrate some of the SLCs with the lowest value of the Gini coefficient (lower right), one with a high value and a reasonable expression (SLC18A2), and the six members of the SLC35F family. The transcript expression levels are expressed through a widespread normalisation (see [305], but also [306,307]) as reads per kilobase million (RPKM).
1.12. Transporters and Prodrugs
Another strategy that has been widely used to improve cellular drug uptake is the use of so-called prodrugs, in which a drug is modified by the addition of a moiety that, although without direct pharmacological activity at a target receptor, assists the passage of the drug to its target (e.g., [308,309,310,311,312,313,314,315,316,317,318]). These necessarily involve transporters, although their identity is not always known. Several recent examples showing high efficacy are based on the SLC7A5 (LAT1) transporter [319,320,321,322,323,324]. In one recent instance [321], the uptake of the anti-inflammatory salicylic acid was enhanced more than five-fold by fusing it with a phenylalanine moiety. Sometimes the prodrug is more lipophilic than its parent, and this is taken in some quarters to mean that it therefore must be passing through bilayers. As pointed out before [8], however, “in actual practice, the reformulation of a water soluble drug with lipidization modifications is difficult to execute successfully, and there is not a single example of a drug presently sold whereby medicinal chemistry was successfully used to convert a non-brain-penetrating drug into a molecule that crosses the BBB {blood–brain barrier} in pharmacologically significant amounts” [325].
1.13. Transporters and Adverse Drug Reactions
Notwithstanding all the benefits of small molecule drugs, it remains the case that they are also widely associated with various morbidities and mortalities, often referred to as “adverse drug reactions” (ADRs) (e.g., [326,327,328,329,330,331,332,333,334,335,336,337,338,339,340,341,342,343,344,345,346]). Despite the lengthy and complex regulatory hurdles that drugs must overcome before being marketed (and toxicity remains a major cause of so-called “attrition” where drug candidates are pulled before they even get to market [347,348,349,350,351,352,353]), ADRs are extremely common (and hard to anticipate, given the huge genetic and phenotypic variation in human populations [354]). Due to non-linearities in biochemical kinetics, averaging across many cells or tissues necessarily hides the true biology [267,355], and thus lumping heterogeneous cells into tissues will always miss such problems [2]. Our contention is that in many cases these ADRs are mediated via transporters, especially since when concentrative transporters can potentially cause massive accumulation in particular cells. Failure or inhibition of efflux transporters also has a major role to play in drug toxicity.
Consequently, the involvement of transporters in ADRs and drug cytotoxicity is well-established (see above and, e.g., [294,336,339,340,356,357,358,359,360,361,362,363,364,365,366,367,368,369,370,371,372,373,374,375,376,377,378,379,380,381,382,383,384,385,386,387,388,389,390,391,392,393,394]), providing further evidence for the major roles of transporters in both pharmacokinetics and pharmacodynamics.
1.14. Transporters, Antibiotics, and Antimicrobial Resistance (AMR)
Antimicrobial resistance (AMR) is a major human health problem (e.g., [272,395,396,397,398,399,400,401,402,403,404,405,406,407,408,409,410,411,412,413,414,415,416,417,418,419]). Most pertinently, efflux transporters are well-recognised as a major source of AMR (e.g., [276,397,420,421,422,423,424,425,426,427,428,429,430,431,432,433,434,435,436,437,438,439,440,441,442,443,444,445,446,447,448,449,450,451,452,453,454,455,456,457]). The outer membrane also contributes significantly to the permeability barrier in Gram-negatives (e.g., [265,434,443,444,448,458,459,460,461,462,463,464,465,466]). However, another specific area in which the role of transporters is largely unrecognised—albeit this is a specific subset of drug transport—pertains to the uptake transport of anti-infectives to their sites of action [439,467]. This can involve both the targets within the microbe and the host’s transporters when (as is common, e.g., [468,469,470,471,472,473,474,475,476,477,478,479,480,481,482,483,484,485]) the infective agents reside intracellularly. A particularly clear example is given by Mycobacterium tuberculosis, the causative agent of TB, where the very striking lack of correlation between in vivo and in vitro drug potencies is easily and necessarily explained via transporter activities [139,151,486,487,488]. Many orally prescribed antibiotics enter the host via SLC15 family members [489,490], while some of the relatively few known microbial uptake transporters for anti-infectives are listed in Table 1. What evidence there is implies that there are multiple means of uptake, which is why identifying individual transporters for successful antibiotics has proven difficult [276]. On the flipside, of course, when we recognise the relevant transporters and/or their structure–activity relationships governing cell permeability, we can exploit them [437,444,491,492,493,494,495].
Table 1.
Some examples of differential resistance to anti-microbial drugs involving uptake transporters.
| Antibiotic | Transporter | Comments | Selected Reference(s) |
|---|---|---|---|
| Aminoglycosides | [496] | ||
| Chloramphenicol | YdgR | E. coli. Proton-dependent oligopeptide transporter analogue | [497] |
| Cycloserine | [498] | ||
| 5-fluocytosine | FCY2 | Various Candida spp. | [499,500] |
| Fosfomycin | [501,502] | ||
| Pacidamycin | Opp PA14 |
Pseudomonas aeruginosa | [503] [504] |
| Pentamidine | Three adenosine-based transporters | [441,505] | |
| Quinoline antimalarials | AAT1 | [506] | |
| Reviews | [441,507,508,509] | ||
| Tetracyclines | Two unknown transporters | [510,511] |
1.15. Molecular Dynamics of Transporter Reactions
Given, as mentioned, the present impossibility of detecting the molecular pathway of drug transport directly, one alternative is to calculate it from first principles, which for these purposes means via the use of molecular dynamics (MD). MD allows the calculation “ab initio” of the molecular motions of molecules during their normal activity. Although computationally demanding (something that becomes much less of an issue over time, e.g., [512,513]), it is perfectly suited to calculating the mechanisms of substrates transport across membranes [514]. We have discussed this in more detail elsewhere [2], and so we simply include here some of the more recent developments. Thus, Jia et al. [515] could mirror precisely the experimental findings underpinning the behaviours of a xylose transporter. Other findings uncovered an electrostatic lock in emrE [516] (see also [517,518]), established the molecular basis of sodium-coupled transporters [519], and illustrated the mechanism of transporters as varied as acrB [520,521,522], the vitamin B12 importer BtuCD [523], the maltose transporter ATPase MalK2 [524], McjD (an antibacterial peptide ABC transporter from E. coli) [525], proton oligopeptide transporters [526], hexameric urea transporter UreI from Helicobacter pylori [527], and the mammalian transporters SLC1A1 (excitatory amino acid transporter EAAT3) [528], SLC2A1 (glucose transporter GLUT1) [529], SLC4A1 (“band 3” protein, bicarbonate/anion exchanger) [530], SLC6A4 [531], SLC7A10 [532]. By contrast, studies of membrane-embedded molecules such as aquaporins, when conducted with high protein concentrations resembling those in biomembranes [533,534,535,536], show that even water molecules do not pass through the bilayer (see a direct illustration at http://www3.mpibpc.mpg.de/groups/de_groot/gallery/aqp1_snapshot.jpg/). In several cases, the MD simulations are accompanied by confirmatory X-ray structures (e.g., [530,535,536]).
1.16. Uptake Transporters as Drug Targets
Although our chief interest here relates mainly to the role of solute carriers in drug disposition, we would be remiss not to mention that, largely because they have been seriously understudied [288], SLCs themselves necessarily constitute potentially valuable and novel drug targets [11,286,537,538,539,540,541,542,543,544,545,546,547,548,549,550,551,552]. It is reasonable that the technical improvements in cryo-EM will contribute to the rational design of such drugs [553], as well as the many other activities ongoing (e.g., as summarised in [131]).
1.17. What Are the “Real” Substrates of Drug Uptake Transporters?
While some drugs that are SLC substrates are, or are semi-synthetic analogues of, natural products, most modern drugs are entirely synthetic in nature, and so natural evolution had no known exposure to them. It is then at least reasonable to enquire as to what the “normal” substrates of these molecules are that happen also to allow them to transport drugs. The principle of molecular similarity (e.g., [201,554,555,556,557,558,559,560,561,562,563,564]) suggests that molecules that have similar structures should tend to have similar activities, so the question then becomes “to which molecules are marketed drugs most similar”? This is a cheminformatics question, and the answer depends in part on the nature of the structural encoding, although most encodings of “actually” similar molecules show a Tanimoto similarity exceeding 0.8 or so, a number that may be used as a kind of benchmark. Our initial assumption was that successful, marketed drug should bear structural similarities to endogenous human metabolites [565,566,567,568], which have been catalogued in metabolic reconstructions [569] and elsewhere (e.g., [570]). However, this accounts for only a small percentage (~15% [83,200,568]), and the true answer—possibly unsurprisingly, post hoc—is that most drugs actually bear similarities to natural products, whose uptake via SLCs may be assumed to be, or to have been during natural selection, of some nutritional or medical benefit to the host [200,264]. This also brings to the fore the important role of natural products in drug discovery [9,571,572,573,574,575,576,577,578,579,580,581,582,583,584,585,586]. Natural products are, of course, famous for breaking [587,588,589,590] many of the “rule of 5” [591] guidelines, and include many very clear examples of drugs that cannot possibly diffuse through lipid bilayers in intact biological cells.
1.18. Why Do SOME Solvents Increase the Rate of Drug Uptake?
Anecdotally, there is a common assumption that because solvents such as DMSO can increase the rate of drug uptake they must be doing so by solubilising the drug, or at least by assisting its solubilisation, in the phospholipid bilayer part of biomembranes. This would then be seen as some kind of evidence for the importance of bilayer transport, but in fact this does not follow at all. The issue with insoluble drugs is that transporters require their substrates to be bound, and that this normally happens via solubilisation in the aqueous phase. Rocks and crystals and amorphous solids are not direct substrates of drug transporters; molecules are. All that solvents such as DMSO are then doing is increasing the rate of solubilisation of drug solids and their presentation to the transporters as single molecules (the necessarily preferred substrates) in solution.
2. Discussion
Real biological membranes possess a high protein:lipid ratio, often as much as 3:1 by mass. Consequently, they do not remotely behave in a manner similar to pure, artificial, phospholipid bilayers. Additionally, the transport of small organic molecules (including drugs) across them commonly requires the intercession of proteinaceous transporters (e.g., [2,3,6,8,101,287,370,592,593,594,595,596,597,598,599,600,601,602,603,604,605,606,607,608,609,610,611,612,613,614,615,616,617,618,619,620]), and there is in fact no actual evidence whatsoever for any significant flux across native, undamaged biomembranes through whatever phospholipid bilayer may be present. This contrasts with a widely held set of assumptions, based in part (it is assumed) on what can be observed in pure phospholipid bilayers that admit the transport of all kinds of small molecules, albeit through transient aqueous pores.
Identifying these transporters of small molecules can be performed (e.g., [96,97,99,101,171]) by manipulating their expression, including under conditions in which their substrates are otherwise toxic. The idea is that removing (or otherwise inhibiting) a transporter that normally helps a toxic drug to enter a cell should increase the host’s resistance to it, while overexpressing the transporter would make the cells more sensitive. More generally, understanding the co-variation between the uptake of a molecule and the expression of its potential transporters gives a strong indication of which they are.
The recognition that in real biological membranes the transbilayer transport through phospholipids of small molecule drugs, nutrients, and biotechnology products is negligible also explains straightforwardly the following well-established facts:
The negligible uptake of drugs and substrates in some tissues, including via the blood–brain barrier (and equivalents in the retina, testes, and other tissues), where relevant transporters are absent;
The extreme heterogeneity of uptake of a given molecule in different organs, tissues, and organisms despite little substantive variation in their lipid physical properties;
The existence of transporters for all kinds of small molecules (even water, acetate, ammonia, glycerol, etc., as well as entirely hydrophobic molecules such as alkanes [621,622,623]) that had previously been assumed to lack them;
A variety of cases in which individual defined transporters can be shown to account for the overwhelming bulk of measured fluxes;
The need for such transporters in order to effect drug uptake, mirroring the widespread recognition that they can serve to efflux them (and thereby created resistance to their activity);
The role of transporters in drug-mediated toxicity (e.g., [366,624]);
The poor correlation between the uptake of small molecules and simple physicochemical properties such as log P or log D (many examples, such as those in [2,6,83,625,626,627]).
3. Looking to the Future
The two chief questions posed earlier (“what are the substrates for a given trasporter?” and “what are the transporters for a given substrate?”) are normally addressed experimentally, using a variety of the methods described above. As intimated above, the exponential increase in computer power will eventually allow the methods of molecular dynamics to admit these “measurements” entirely by calculations based on simple force fields, de novo. In addition, given the success of so-called deep learning [628,629,630] methods in predicting the structures of proteins [631,632,633,634,635,636,637,638,639,640,641,642,643,644,645], novel receptor–ligand interactions [646,647,648,649,650], and a variety of other protein and small molecule properties (e.g., [201,651,652,653,654,655,656,657,658,659,660,661,662,663,664,665]), it seems likely that we shall soon have available in silico methods for predicting transporter substrates directly from protein sequences, and the likeliest transporters from candidate substrate structures of interest.
4. Conclusions
On the basis of the present evidence, the transport of drugs and nutrients through phospholipid bilayers in real biomembranes is negligible. Progress in understanding drug distribution profiles is thus to be made by establishing which transporters they use, the expression profiles of those transporters, and the kinetic rate equations that they obey. Armed with these it will be possible to model, to analyse, to understand, and to exploit our principled knowledge of drug distributions both mechanistically and with confidence.
Acknowledgments
I thank many colleagues for useful discussions.
Funding
DBK thanks the BBSRC (grant BB/R014426/1) and the Novo Nordisk Foundation (Grant agreement No. NNF20CC0035580) for financial support.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kell D.B. How drugs pass through biological cell membranes—A paradigm shift in our understanding? [(accessed on 15 September 2021)];Beilstein Mag. 2016 2 doi: 10.3762/bmag.5. Available online: http://www.beilstein-institut.de/download/628/09_kell.pdf. [DOI] [Google Scholar]
- 2.Kell D.B., Oliver S.G. How drugs get into cells: Tested and testable predictions to help discriminate between transporter-mediated uptake and lipoidal bilayer diffusion. Front. Pharmacol. 2014;5:231. doi: 10.3389/fphar.2014.00231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dobson P., Lanthaler K., Oliver S.G., Kell D.B. Implications of the dominant role of cellular transporters in drug uptake. Curr. Top. Med. Chem. 2009;9:163–184. doi: 10.2174/156802609787521616. [DOI] [PubMed] [Google Scholar]
- 4.Kell D.B., Swainston N., Pir P., Oliver S.G. Membrane transporter engineering in industrial biotechnology and whole-cell biocatalysis. Trends Biotechnol. 2015;33:237–246. doi: 10.1016/j.tibtech.2015.02.001. [DOI] [PubMed] [Google Scholar]
- 5.Mendes P., Oliver S.G., Kell D.B. Fitting transporter activities to cellular drug concentrations and fluxes: Why the bumblebee can fly. Trends Pharmacol. Sci. 2015;36:710–723. doi: 10.1016/j.tips.2015.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dobson P.D., Kell D.B. Carrier-mediated cellular uptake of pharmaceutical drugs: An exception or the rule? Nat. Rev.Drug Discov. 2008;7:205–220. doi: 10.1038/nrd2438. [DOI] [PubMed] [Google Scholar]
- 7.Kell D.B., Dobson P.D., Oliver S.G. Pharmaceutical drug transport: The issues and the implications that it is essentially carrier-mediated only. Drug Discov. Today. 2011;16:704–714. doi: 10.1016/j.drudis.2011.05.010. [DOI] [PubMed] [Google Scholar]
- 8.Kell D.B., Dobson P.D., Bilsland E., Oliver S.G. The promiscuous binding of pharmaceutical drugs and their transporter-mediated uptake into cells: What we (need to) know and how we can do so. Drug Discov. Today. 2013;18:218–239. doi: 10.1016/j.drudis.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 9.Kell D.B. Finding novel pharmaceuticals in the systems biology era using multiple effective drug targets, phenotypic screening, and knowledge of transporters: Where drug discovery went wrong and how to fix it. FEBS J. 2013;280:5957–5980. doi: 10.1111/febs.12268. [DOI] [PubMed] [Google Scholar]
- 10.Kell D.B. Implications of endogenous roles of transporters for drug discovery: Hitchhiking and metabolite-likeness. Nat. Rev. Drug Discov. 2016;15:143–144. doi: 10.1038/nrd.2015.44. [DOI] [PubMed] [Google Scholar]
- 11.Kell D.B., Wright Muelas M., O’Hagan S., Day P.J. The role of drug transporters in phenotypic screening. Drug Target Rev. 2018;4:16–19. [Google Scholar]
- 12.Kell D.B. Control of metabolite efflux in microbial cell factories: Current advances and future prospects. In: El-Mansi E.M.T., Nielsen J., Mousdale D., Allman T., Carlson R., editors. Fermentation Microbiology and Biotechnology. 4th ed. CRC Press; Boca Raton, FL, USA: 2019. pp. 117–138. [Google Scholar]
- 13.Singer S.J., Nicolson G.L. The fluid mosaic model of the structure of cell membranes. Science. 1972;175:720–731. doi: 10.1126/science.175.4023.720. [DOI] [PubMed] [Google Scholar]
- 14.Nicolson G.L. The Fluid-Mosaic Model of Membrane Structure: Still relevant to understanding the structure, function and dynamics of biological membranes after more than 40 years. Biochim. Biophys. Acta. 2014;1838:1451–1466. doi: 10.1016/j.bbamem.2013.10.019. [DOI] [PubMed] [Google Scholar]
- 15.Goñi F.M. The basic structure and dynamics of cell membranes: An update of the Singer-Nicolson model. Biochim. Biophys. Acta. 2014;1838:1467–1476. doi: 10.1016/j.bbamem.2014.01.006. [DOI] [PubMed] [Google Scholar]
- 16.Guidotti G. The composition of biological membranes. Arch. Intern. Med. 1972;129:194–201. doi: 10.1001/archinte.1972.00320020038003. [DOI] [PubMed] [Google Scholar]
- 17.Engelman D.M. Membranes are more mosaic than fluid. Nature. 2005;438:578–580. doi: 10.1038/nature04394. [DOI] [PubMed] [Google Scholar]
- 18.Dupuy A.D., Engelman D.M. Protein area occupancy at the center of the red blood cell membrane. Proc. Natl. Acad. Sci. USA. 2008;105:2848–2852. doi: 10.1073/pnas.0712379105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Domański J., Marrink S.J., Schäfer L.V. Transmembrane helices can induce domain formation in crowded model membranes. Biochim. Biophys. Acta. 2012;1818:984–994. doi: 10.1016/j.bbamem.2011.08.021. [DOI] [PubMed] [Google Scholar]
- 20.Lindén M., Sens P., Phillips R. Entropic tension in crowded membranes. PLoS Comput. Biol. 2012;8:e1002431. doi: 10.1371/journal.pcbi.1002431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Goose J.E., Sansom M.S.P. Reduced Lateral Mobility of Lipids and Proteins in Crowded Membranes. PLoS Comput. Biol. 2013;9:e1003033. doi: 10.1371/journal.pcbi.1003033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Höfling F., Franosch T. Anomalous transport in the crowded world of biological cells. Rep. Prog. Phys. 2013;76:046642. doi: 10.1088/0034-4885/76/4/046602. [DOI] [PubMed] [Google Scholar]
- 23.Javanainen M., Hammaren H., Monticelli L., Jeon J.H., Miettinen M.S., Martinez-Seara H., Metzler R., Vattulainen I. Anomalous and normal diffusion of proteins and lipids in crowded lipid membranes. Faraday Discuss. 2013;161:397–417. doi: 10.1039/C2FD20085F. [DOI] [PubMed] [Google Scholar]
- 24.Guigas G., Weiss M. Effects of protein crowding on membrane systems. Biochim Biophys Acta. 2016;1858:2441–2450. doi: 10.1016/j.bbamem.2015.12.021. [DOI] [PubMed] [Google Scholar]
- 25.Jeon J.H., Javanainen M., Martinez-Seara H., Metzler R., Vattulainen I. Protein Crowding in Lipid Bilayers Gives Rise to Non-Gaussian Anomalous Lateral Diffusion of Phospholipids and Proteins. Phys. Rev. X. 2016;6:021006. doi: 10.1103/PhysRevX.6.021006. [DOI] [Google Scholar]
- 26.Duncan A.L., Reddy T., Koldso H., Helie J., Fowler P.W., Chavent M., Sansom M.S.P. Protein crowding and lipid complexity influence the nanoscale dynamic organization of ion channels in cell membranes. Sci. Rep. 2017;7:16647. doi: 10.1038/s41598-017-16865-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Marrink S.J., Corradi V., Souza P.C.T., Ingólfsson H.I., Tieleman D.P., Sansom M.S.P. Computational Modeling of Realistic Cell Membranes. Chem. Rev. 2019;119:6184–6226. doi: 10.1021/acs.chemrev.8b00460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ando T., Uchihashi T., Scheuring S. Filming biomolecular processes by high-speed atomic force microscopy. Chem. Rev. 2014;114:3120–3188. doi: 10.1021/cr4003837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jain M.K. The Bimolecular Lipid Membrane. Van Nostrand Reinhold; New York, NY, USA: 1972. [Google Scholar]
- 30.Tien H.T. Bilayer Lipid Membranes (BLM): Theory and Practice. Marcel Dekker; New York, NY, USA: 1974. [Google Scholar]
- 31.Tien H.T., Ottova-Leitmannova A. Planar Lipid Bilayers (BLMs) and Their Applications. Elsevier; New York, NY, USA: 2003. [Google Scholar]
- 32.Marrink S.J., Jähnig F., Berendsen H.J.C. Proton transport across transient single-file water pores in a lipid membrane studied by molecular dynamics simulations. Biophys. J. 1996;71:632–647. doi: 10.1016/S0006-3495(96)79264-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Weaver J.C., Powell K.T., Mintzer R.A., Sloan S.R., Ling H. The diffusive permeability of bilayer membranes: The contribution of transient aqueous pores. Bioelectrochem. Bioenerg. 1984;12:405–412. doi: 10.1016/0302-4598(84)87019-1. [DOI] [Google Scholar]
- 34.Deamer D.W., Bramhall J. Permeability of lipid bilayers to water and ionic solutes. Chem. Phys. Lipids. 1986;40:167–188. doi: 10.1016/0009-3084(86)90069-1. [DOI] [PubMed] [Google Scholar]
- 35.Leontiadou H., Mark A.E., Marrink S.J. Molecular dynamics simulations of hydrophilic pores in lipid bilayers. Biophys. J. 2004;86:2156–2164. doi: 10.1016/S0006-3495(04)74275-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Loison C., Mareschal M., Schmid F. Pores in bilayer membranes of amphiphilic molecules: Coarse-grained molecular dynamics simulations compared with simple mesoscopic models. J. Chem. Phys. 2004;121:1890–1900. doi: 10.1063/1.1752884. [DOI] [PubMed] [Google Scholar]
- 37.Leontiadou H., Mark A.E., Marrink S.J. Ion transport across transmembrane pores. Biophys. J. 2007;92:4209–4215. doi: 10.1529/biophysj.106.101295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Marrink S.J., De Vries A.H., Tieleman D.P. Lipids on the move: Simulations of membrane pores, domains, stalks and curves. Biochim. Biophys. Acta. 2009;1788:149–168. doi: 10.1016/j.bbamem.2008.10.006. [DOI] [PubMed] [Google Scholar]
- 39.Gurtovenko A.A., Anwar J., Vattulainen I. Defect-mediated trafficking across cell membranes: Insights from in silico modeling. Chem. Rev. 2010;110:6077–6103. doi: 10.1021/cr1000783. [DOI] [PubMed] [Google Scholar]
- 40.Bennett W.F., Tieleman D.P. The importance of membrane defects-lessons from simulations. Acc. Chem. Res. 2014;47:2244–2251. doi: 10.1021/ar4002729. [DOI] [PubMed] [Google Scholar]
- 41.Bubnis G., Grubmüller H. Sequential Water and Headgroup Merger: Membrane Poration Paths and Energetics from MD Simulations. Biophys. J. 2020;119:2418–2430. doi: 10.1016/j.bpj.2020.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Akimov S.A., Volynsky P.E., Galimzyanov T.R., Kuzmin P.I., Pavlov K.V., Batishchev O.V. Pore formation in lipid membrane I: Continuous reversible trajectory from intact bilayer through hydrophobic defect to transversal pore. Sci. Rep. 2017;7:12152. doi: 10.1038/s41598-017-12127-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Akimov S.A., Volynsky P.E., Galimzyanov T.R., Kuzmin P.I., Pavlov K.V., Batishchev O.V. Pore formation in lipid membrane II: Energy landscape under external stress. Sci. Rep. 2017;7:12509. doi: 10.1038/s41598-017-12749-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Parsegian A. Energy of an ion crossing a low dielectric membrane: Solutions to four relevant electrostatic problems. Nature. 1969;221:844–846. doi: 10.1038/221844a0. [DOI] [PubMed] [Google Scholar]
- 45.Lee A.G. Lipid-protein interactions in biological membranes: A structural perspective. Biochim. Biophys. Acta. 2003;1612:1–40. doi: 10.1016/S0005-2736(03)00056-7. [DOI] [PubMed] [Google Scholar]
- 46.Tillman T.S., Cascio M. Effects of membrane lipids on ion channel structure and function. Cell Biochem. Biophys. 2003;38:161–190. doi: 10.1385/CBB:38:2:161. [DOI] [PubMed] [Google Scholar]
- 47.Van Meer G., Voelker D.R., Feigenson G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008;9:112–124. doi: 10.1038/nrm2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Niemelä P.S., Miettinen M.S., Monticelli L., Hammaren H., Bjelkmar P., Murtola T., Lindahl E., Vattulainen I. Membrane proteins diffuse as dynamic complexes with lipids. J. Am. Chem. Soc. 2010;132:7574–7575. doi: 10.1021/ja101481b. [DOI] [PubMed] [Google Scholar]
- 49.Hickey K.D., Buhr M.M. Lipid bilayer composition affects transmembrane protein orientation and function. J. Lipids. 2011;2011:208457. doi: 10.1155/2011/208457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Laganowsky A., Reading E., Allison T.M., Ulmschneider M.B., Degiacomi M.T., Baldwin A.J., Robinson C.V. Membrane proteins bind lipids selectively to modulate their structure and function. Nature. 2014;510:172–175. doi: 10.1038/nature13419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Aponte-Santamaría C., Briones R., Schenk A.D., Walz T., De Groot B.L. Molecular driving forces defining lipid positions around aquaporin-0. Proc. Natl. Acad. Sci. USA. 2012;109:9887–9892. doi: 10.1073/pnas.1121054109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Poveda J.A., Giudici A.M., Renart M.L., Molina M.L., Montoya E., Fernández-Carvajal A., Fernández-Ballester G., Encinar J.A., González-Ros J.M. Lipid modulation of ion channels through specific binding sites. Biochim. Biophys. Acta. 2014;1838:1560–1567. doi: 10.1016/j.bbamem.2013.10.023. [DOI] [PubMed] [Google Scholar]
- 53.Hedger G., Sansom M.S.P. Lipid interaction sites on channels, transporters and receptors: Recent insights from molecular dynamics simulations. Biochim. Biophys. Acta. 2016;1858:2390–2400. doi: 10.1016/j.bbamem.2016.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kalli A.C., Sansom M.S.P., Reithmeier R.A.F. Molecular dynamics simulations of the bacterial UraA H+-uracil symporter in lipid bilayers reveal a closed state and a selective interaction with cardiolipin. PLoS Comput. Biol. 2015;11:e1004123. doi: 10.1371/journal.pcbi.1004123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Newport T.D., Sansom M.S.P., Stansfeld P.J. The MemProtMD database: A resource for membrane-embedded protein structures and their lipid interactions. Nucleic Acids Res. 2019;47:D390–D397. doi: 10.1093/nar/gky1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Song C., De Groot B.L., Sansom M.S.P. Lipid Bilayer Composition Influences the Activity of the Antimicrobial Peptide Dermcidin Channel. Biophys. J. 2019;116:1658–1666. doi: 10.1016/j.bpj.2019.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gault J., Liko I., Landreh M., Shutin D., Bolla J.R., Jefferies D., Agasid M., Yen H.Y., Ladds M., Lane D.P., et al. Combining native and ‘omics’ mass spectrometry to identify endogenous ligands bound to membrane proteins. Nat. Methods. 2020;17:505–508. doi: 10.1038/s41592-020-0821-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Harayama T., Riezman H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 2018;19:281–296. doi: 10.1038/nrm.2017.138. [DOI] [PubMed] [Google Scholar]
- 59.Casares D., Escribá P.V., Rosselló C.A. Membrane Lipid Composition: Effect on Membrane and Organelle Structure, Function and Compartmentalization and Therapeutic Avenues. Int. J. Mol. Sci. 2019;20:2167. doi: 10.3390/ijms20092167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kanonenberg K., Royes J., Kedrov A., Poschmann G., Angius F., Solgadi A., Spitz O., Kleinschrodt D., Stühler K., Miroux B., et al. Shaping the lipid composition of bacterial membranes for membrane protein production. Microb. Cell Factories. 2019;18:131. doi: 10.1186/s12934-019-1182-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chorev D.S., Robinson C.V. The importance of the membrane for biophysical measurements. Nat. Chem. Biol. 2020;16:1285–1292. doi: 10.1038/s41589-020-0574-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Thompson M.J., Baenziger J.E. Ion channels as lipid sensors: From structures to mechanisms. Nat. Chem. Biol. 2020;16:1331–1342. doi: 10.1038/s41589-020-00693-3. [DOI] [PubMed] [Google Scholar]
- 63.Doktorova M., Symons J.L., Levental I. Structural and functional consequences of reversible lipid asymmetry in living membranes. Nat. Chem. Biol. 2020;16:1321–1330. doi: 10.1038/s41589-020-00688-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lorent J.H., Levental K.R., Ganesan L., Rivera-Longsworth G., Sezgin E., Doktorova M., Lyman E., Levental I. Plasma membranes are asymmetric in lipid unsaturation, packing and protein shape. Nat. Chem. Biol. 2020;16:644–652. doi: 10.1038/s41589-020-0529-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Makarova M., Owen D.M. Asymmetry across the membrane. Nat. Chem. Biol. 2020;16:605–606. doi: 10.1038/s41589-020-0545-6. [DOI] [PubMed] [Google Scholar]
- 66.Pohl A., Devaux P.F., Herrmann A. Function of prokaryotic and eukaryotic ABC proteins in lipid transport. Biochim. Biophys. Acta. 2005;1733:29–52. doi: 10.1016/j.bbalip.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 67.Quazi F., Molday R.S. Lipid transport by mammalian ABC proteins. Essays Biochem. 2011;50:265–290. doi: 10.1042/bse0500265. [DOI] [PubMed] [Google Scholar]
- 68.Borst P., Zelcer N., Van Helvoort A. ABC transporters in lipid transport. Biochim. Biophys. Acta. 2000;1486:128–144. doi: 10.1016/S1388-1981(00)00053-6. [DOI] [PubMed] [Google Scholar]
- 69.Neumann J., Rose-Sperling D., Hellmich U.A. Diverse relations between ABC transporters and lipids: An overview. Biochim. Biophys. Acta Biomembr. 2017;1859:605–618. doi: 10.1016/j.bbamem.2016.09.023. [DOI] [PubMed] [Google Scholar]
- 70.Kell D.B. A protet-based, protonic charge transfer model of energy coupling in oxidative and photosynthetic phosphorylation. Adv. Microb. Physiol. 2021;78:1–177. doi: 10.1016/bs.ampbs.2021.01.001. [DOI] [PubMed] [Google Scholar]
- 71.Kell D.B., Welch G.R. Belief: The Baggage behind Our Being. OSF Preprints; 2018. [(accessed on 15 September 2021)]. Available online: pnxcshttps://osf.io/pnxcs/ [Google Scholar]
- 72.Kahneman D. Thinking, Fast and Slow. Penguin; London, UK: 2011. [Google Scholar]
- 73.Sharot T. The Optimism Bias. Robinson; London, UK: 2012. [Google Scholar]
- 74.Sharot T. The Influential Mind. Abacus; London, UK: 2017. [Google Scholar]
- 75.Sharot T. To quell misinformation, use carrots—Not just sticks. Nature. 2021;591:347. doi: 10.1038/d41586-021-00657-0. [DOI] [PubMed] [Google Scholar]
- 76.Peterson J.C., Bourgin D.D., Agrawal M., Reichman D., Griffiths T.L. Using large-scale experiments and machine learning to discover theories of human decision-making. Science. 2021;372:1209–1214. doi: 10.1126/science.abe2629. [DOI] [PubMed] [Google Scholar]
- 77.Artursson P., Palm K., Luthman K. Caco-2 monolayers in experimental and theoretical predictions of drug transport. Adv. Drug Deliv. Rev. 1996;22:67–84. doi: 10.1016/S0169-409X(96)00415-2. [DOI] [PubMed] [Google Scholar]
- 78.Sun H., Chow E.C., Liu S., Du Y., Pang K.S. The Caco-2 cell monolayer: Usefulness and limitations. Expert Opin. Drug Metab. Toxicol. 2008;4:395–411. doi: 10.1517/17425255.4.4.395. [DOI] [PubMed] [Google Scholar]
- 79.Van Breemen R.B., Li Y. Caco-2 cell permeability assays to measure drug absorption. Expert Opin. Drug Metab. Toxicol. 2005;1:175–185. doi: 10.1517/17425255.1.2.175. [DOI] [PubMed] [Google Scholar]
- 80.Press B. Optimization of the Caco-2 permeability assay to screen drug compounds for intestinal absorption and efflux. Methods Mol. Biol. 2011;763:139–154. doi: 10.1007/978-1-61779-191-8_9. [DOI] [PubMed] [Google Scholar]
- 81.Volpe D.A. Drug-permeability and transporter assays in Caco-2 and MDCK cell lines. Future Med. Chem. 2011;3:2063–2077. doi: 10.4155/fmc.11.149. [DOI] [PubMed] [Google Scholar]
- 82.Mukhopadhya I., Murray G.I., Berry S., Thomson J., Frank B., Gwozdz G., Ekeruche-Makinde J., Shattock R., Kelly C., Iannelli F., et al. Drug transporter gene expression in human colorectal tissue and cell lines: Modulation with antiretrovirals for microbicide optimization. J. Antimicrob. Chemother. 2016;71:372–386. doi: 10.1093/jac/dkv335. [DOI] [PubMed] [Google Scholar]
- 83.O’Hagan S., Kell D.B. The apparent permeabilities of Caco-2 cells to marketed drugs: Magnitude, and independence from both biophysical properties and endogenite similarities. PeerJ. 2015;3:e1405. doi: 10.7717/peerj.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Iftikhar M., Iftikhar A., Zhang H., Gong L., Wang J. Transport, metabolism and remedial potential of functional food extracts (FFEs) in Caco-2 cells monolayer: A review. Food Res. Int. 2020;136:109240. doi: 10.1016/j.foodres.2020.109240. [DOI] [PubMed] [Google Scholar]
- 85.Thul P.J., Åkesson L., Wiking M., Mahdessian D., Geladaki A., Ait Blal H., Alm T., Asplund A., Björk L., Breckels L.M., et al. A subcellular map of the human proteome. Science. 2017;356:eaal3321. doi: 10.1126/science.aal3321. [DOI] [PubMed] [Google Scholar]
- 86.O’Hagan S., Wright Muelas M., Day P.J., Lundberg E., Kell D.B. GeneGini: Assessment via the Gini coefficient of reference ‘‘housekeeping’’ genes and diverse human transporter expression profiles. Cell Syst. 2018;6:230–244. doi: 10.1016/j.cels.2018.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sun D., Lennernäs H., Welage L.S., Barnett J.L., Landowski C.P., Foster D., Fleisher D., Lee K.D., Amidon G.L. Comparison of human duodenum and Caco-2 gene expression profiles for 12,000 gene sequences tags and correlation with permeability of 26 drugs. Pharm. Res. 2002;19:1400–1416. doi: 10.1023/A:1020483911355. [DOI] [PubMed] [Google Scholar]
- 88.Anderle P., Huang Y., Sadée W. Intestinal membrane transport of drugs and nutrients: Genomics of membrane transporters using expression microarrays. Eur. J. Pharm. Sci. 2004;21:17–24. doi: 10.1016/S0928-0987(03)00169-6. [DOI] [PubMed] [Google Scholar]
- 89.Landowski C.P., Anderle P., Sun D., Sadee W., Amidon G.L. Transporter and ion channel gene expression after Caco-2 cell differentiation using 2 different microarray technologies. AAPS J. 2004;6:e21. doi: 10.1208/aapsj060321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pshezhetsky A.V., Fedjaev M., Ashmarina L., Mazur A., Budman L., Sinnett D., Labuda D., Beaulieu J.F., Menard D., Nifant’ev I., et al. Subcellular proteomics of cell differentiation: Quantitative analysis of the plasma membrane proteome of Caco-2 cells. Proteomics. 2007;7:2201–2215. doi: 10.1002/pmic.200600956. [DOI] [PubMed] [Google Scholar]
- 91.Ahlin G., Hilgendorf C., Karlsson J., Szigyarto C.A., Uhlén M., Artursson P. Endogenous gene and protein expression of drug-transporting proteins in cell lines routinely used in drug discovery programs. Drug Metab. Dispos. 2009;37:2275–2283. doi: 10.1124/dmd.109.028654. [DOI] [PubMed] [Google Scholar]
- 92.Fersht A. Enzyme Structure and Mechanism. 2nd ed. W.H. Freeman; San Francisco, CA, USA: 1977. [Google Scholar]
- 93.Keleti T. Basic Enzyme Kinetics. Akadémiai Kiadó; Budapest, Hungary: 1986. [Google Scholar]
- 94.Cornish-Bowden A. Fundamentals of Enzyme Kinetics. 2nd ed. Portland Press; London, UK: 1995. [Google Scholar]
- 95.Denning E.J., Beckstein O. Influence of lipids on protein-mediated transmembrane transport. Chem. Phys. Lipids. 2013;169:57–71. doi: 10.1016/j.chemphyslip.2013.02.007. [DOI] [PubMed] [Google Scholar]
- 96.Lanthaler K., Bilsland E., Dobson P., Moss H.J., Pir P., Kell D.B., Oliver S.G. Genome-wide assessment of the carriers involved in the cellular uptake of drugs: A model system in yeast. BMC Biol. 2011;9:70. doi: 10.1186/1741-7007-9-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Winter G.E., Radic B., Mayor-Ruiz C., Blomen V.A., Trefzer C., Kandasamy R.K., Huber K.V.M., Gridling M., Chen D., Klampfl T., et al. The solute carrier SLC35F2 enables YM155-mediated DNA damage toxicity. Nat. Chem. Biol. 2014;10:768–773. doi: 10.1038/nchembio.1590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Birsoy K., Wang T., Possemato R., Yilmaz O.H., Koch C.E., Chen W.W., Hutchins A.W., Gultekin Y., Peterson T.R., Carette J.E., et al. MCT1-mediated transport of a toxic molecule is an effective strategy for targeting glycolytic tumors. Nat. Genet. 2013;45:104–108. doi: 10.1038/ng.2471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bailey T.L., Nieto A., McDonald P.H. A Nonradioactive High-Throughput Screening-Compatible Cell-Based Assay to Identify Inhibitors of the Monocarboxylate Transporter Protein 1. Assay Drug Dev. Technol. 2019;17:275–284. doi: 10.1089/adt.2019.936. [DOI] [PubMed] [Google Scholar]
- 100.Wright Muelas M., Mughal F., O’Hagan S., Day P.J., Kell D.B. The role and robustness of the Gini coefficient as an unbiased tool for the selection of Gini genes for normalising expression profiling data Sci. Rep. 2019;9:17960. doi: 10.1038/s41598-019-54288-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Girardi E., César-Razquin A., Lindinger S., Papakostas K., Lindinger S., Konecka J., Hemmerich J., Kickinger S., Kartnig F., Gürtl B., et al. A widespread role for SLC transmembrane transporters in resistance to cytotoxic drugs. Nat. Chem. Biol. 2020;16:469–478. doi: 10.1038/s41589-020-0483-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kell D.B. Hitchhiking into the cell. Nat. Chem. Biol. 2020;16:367–368. doi: 10.1038/s41589-020-0489-x. [DOI] [PubMed] [Google Scholar]
- 103.Gründemann D., Harlfinger S., Golz S., Geerts A., Lazar A., Berkels R., Jung N., Rubbert A., Schömig E. Discovery of the ergothioneine transporter. Proc. Natl. Acad. Sci. USA. 2005;102:5256–5261. doi: 10.1073/pnas.0408624102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Babcock J.J., Li M. Deorphanizing the human transmembrane genome: A landscape of uncharacterized membrane proteins. Acta Pharmacol. Sin. 2014;35:11–23. doi: 10.1038/aps.2013.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Stieger B., Hagenbuch B. Recent advances in understanding hepatic drug transport. F1000Research. 2016;5:2465. doi: 10.12688/f1000research.9466.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hashimoto M., Girardi E., Eichner R., Superti-Furga G. Detection of Chemical Engagement of Solute Carrier Proteins by a Cellular Thermal Shift Assay. ACS Chem. Biol. 2018;13:1480–1486. doi: 10.1021/acschembio.8b00270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Clémençon B., Lüscher B.P., Hediger M.A. Establishment of a novel microscale thermophoresis ligand-binding assay for characterization of SLC solute carriers using oligopeptide transporter PepT1 (SLC15 family) as a model system. J. Pharmacol. Toxicol. Methods. 2018;92:67–76. doi: 10.1016/j.vascn.2018.03.004. [DOI] [PubMed] [Google Scholar]
- 108.Girardi E., Agrimi G., Goldmann U., Fiume G., Lindinger S., Sedlyarov V., Srndic I., Gurtl B., Agerer B., Kartnig F., et al. Epistasis-driven identification of SLC25A51 as a regulator of human mitochondrial NAD import. Nat. Commun. 2020;11:6145. doi: 10.1038/s41467-020-19871-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yee S.W., Buitrago D., Stecula A., Ngo H.X., Chien H.C., Zou L., Koleske M.L., Giacomini K.M. Deorphaning a solute carrier 22 family member, SLC22A15, through functional genomic studies. FASEB J. 2020;34:15734–15752. doi: 10.1096/fj.202001497R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.André P., Debray M., Scherrmann J.M., Cisternino S. Clonidine transport at the mouse blood-brain barrier by a new H+ antiporter that interacts with addictive drugs. J. Cereb. Blood Flow Metab. 2009;29:1293–1304. doi: 10.1038/jcbfm.2009.54. [DOI] [PubMed] [Google Scholar]
- 111.Auvity S., Chapy H., Goutal S., Caille F., Hosten B., Smirnova M., Decleves X., Tournier N., Cisternino S. Diphenhydramine as a selective probe to study H+-antiporter function at the blood-brain barrier: Application to [11C]diphenhydramine positron emission tomography imaging. J. Cereb. Blood Flow Metab. 2017;37:2185–2195. doi: 10.1177/0271678X16662042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Chapy H., Smirnova M., Andre P., Schlatter J., Chiadmi F., Couraud P.O., Scherrmann J.M., Decleves X., Cisternino S. Carrier-mediated cocaine transport at the blood-brain barrier as a putative mechanism in addiction liability. Int. J. Neuropsychopharmacol. 2014;18:pyu001. doi: 10.1093/ijnp/pyu001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chapy H., André P., Declèves X., Scherrmann J.M., Cisternino S. A polyspecific drug/proton antiporter mediates diphenhydramine and clonidine transport at the mouse blood-retinal barrier. Br. J. Pharmacol. 2015;172:4714–4725. doi: 10.1111/bph.13246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Okura T., Hattori A., Takano Y., Sato T., Hammarlund-Udenaes M., Terasaki T., Deguchi Y. Involvement of the pyrilamine transporter, a putative organic cation transporter, in blood-brain barrier transport of oxycodone. Drug Metab. Dispos. 2008;36:2005–2013. doi: 10.1124/dmd.108.022087. [DOI] [PubMed] [Google Scholar]
- 115.Okura T., Higuchi K., Kitamura A., Deguchi Y. Proton-coupled organic cation antiporter-mediated uptake of apomorphine enantiomers in human brain capillary endothelial cell line hCMEC/D3. Biol Pharm. Bull. 2014;37:286–291. doi: 10.1248/bpb.b13-00773. [DOI] [PubMed] [Google Scholar]
- 116.Tega Y., Kubo Y., Yuzurihara C., Akanuma S., Hosoya K. Carrier-Mediated Transport of Nicotine Across the Inner Blood-Retinal Barrier: Involvement of a Novel Organic Cation Transporter Driven by an Outward H+ Gradient. J. Pharm. Sci. 2015;104:3069–3075. doi: 10.1002/jps.24453. [DOI] [PubMed] [Google Scholar]
- 117.Tega Y., Akanuma S., Kubo Y., Hosoya K. Involvement of the H+/organic cation antiporter in nicotine transport in rat liver. Drug Metab. Dispos. 2015;43:89–92. doi: 10.1124/dmd.114.061002. [DOI] [PubMed] [Google Scholar]
- 118.Dickens D., Rädisch S., Chiduza G.N., Giannoudis A., Cross M.J., Malik H., Schaeffeler E., Sison-Young R.L., Wilkinson E.L., Goldring C.E., et al. Cellular uptake of the atypical antipsychotic clozapine is a carrier-mediated process. Mol. Pharm. 2018;15:3557–3572. doi: 10.1021/acs.molpharmaceut.8b00547. [DOI] [PubMed] [Google Scholar]
- 119.Dvorak V., Wiedmer T., Ingles-Prieto A., Altermatt P., Batoulis H., Bärenz F., Bender E., Digles D., Dürrenberger F., Heitman L.H., et al. An overview of cell-based assay platforms for the solute carriers family of transporters. Front. Pharmacol. 2021;12:722889. doi: 10.3389/fphar.2021.722889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Mestres J., Gregori-Puigjané E., Valverde S., Solé R.V. The topology of drug-target interaction networks: Implicit dependence on drug properties and target families. Mol. Biosyst. 2009;5:1051–1057. doi: 10.1039/b905821b. [DOI] [PubMed] [Google Scholar]
- 121.Hu Y., Gupta-Ostermann D., Bajorath J. Exploring compound promiscuity patterns and multi-target activity spaces. Comput. Struct. Biotechnol. J. 2014;9:e201401003. doi: 10.5936/csbj.201401003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Bajorath J. Analyzing Promiscuity at the Level of Active Compounds and Targets. Mol. Inform. 2016;35:583–587. doi: 10.1002/minf.201600030. [DOI] [PubMed] [Google Scholar]
- 123.Gilberg E., Jasial S., Stumpfe D., Dimova D., Bajorath J. Highly Promiscuous Small Molecules from Biological Screening Assays Include Many Pan-Assay Interference Compounds but Also Candidates for Polypharmacology. J. Med. Chem. 2016;59:10285–10290. doi: 10.1021/acs.jmedchem.6b01314. [DOI] [PubMed] [Google Scholar]
- 124.Hu Y., Bajorath J. Entering the ‘big data’ era in medicinal chemistry: Molecular promiscuity analysis revisited. Future Sci. OA. 2017;3:FSO179. doi: 10.4155/fsoa-2017-0001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bofill A., Jalencas X., Oprea T.I., Mestres J. The human endogenous metabolome as a pharmacology baseline for drug discovery. Drug Discov. Today. 2019;24:1806–1820. doi: 10.1016/j.drudis.2019.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Cerisier N., Petitjean M., Regad L., Bayard Q., Réau M., Badel A., Camproux A.C. High Impact: The Role of Promiscuous Binding Sites in Polypharmacology. Molecules. 2019;24:2529. doi: 10.3390/molecules24142529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Feldmann C., Miljkovic F., Yonchev D., Bajorath J. Identifying Promiscuous Compounds with Activity against Different Target Classes. Molecules. 2019;24:4185. doi: 10.3390/molecules24224185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Perrin J., Werner T., Kurzawa N., Rutkowska A., Childs D.D., Kalxdorf M., Poeckel D., Stonehouse E., Strohmer K., Heller B., et al. Identifying drug targets in tissues and whole blood with thermal-shift profiling. Nat. Biotechnol. 2020;38:303–308. doi: 10.1038/s41587-019-0388-4. [DOI] [PubMed] [Google Scholar]
- 129.Yang Z.Y., He J.H., Lu A.P., Hou T.J., Cao D.S. Frequent hitters: Nuisance artifacts in high-throughput screening. Drug Discov. Today. 2020;25:657–667. doi: 10.1016/j.drudis.2020.01.014. [DOI] [PubMed] [Google Scholar]
- 130.Niphakis M.J., Lum K.M., Cognetta A.B., Correia B.E., Ichu T.A., Olucha J., Brown S.J., Kundu S., Piscitelli F., Rosen H., et al. A Global Map of Lipid-Binding Proteins and Their Ligandability in Cells. Cell. 2015;161:1668–1680. doi: 10.1016/j.cell.2015.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Superti-Furga G., Lackner D., Wiedmer T., Ingles-Prieto A., Barbosa B., Girardi E., Ulrich G., Gürtl B., Klavins K., Klimek C., et al. The RESOLUTE consortium: Unlocking SLC transporters for drug discovery. Nat. Rev. Drug Discov. 2020;19:429–430. doi: 10.1038/d41573-020-00056-6. [DOI] [PubMed] [Google Scholar]
- 132.Garst A.D., Bassalo M.C., Pines G., Lynch S.A., Halweg-Edwards A.L., Liu R., Liang L., Wang Z., Zeitoun R., Alexander W.G., et al. Genome-wide mapping of mutations at single-nucleotide resolution for protein, metabolic and genome engineering. Nat. Biotechnol. 2017;35:48–55. doi: 10.1038/nbt.3718. [DOI] [PubMed] [Google Scholar]
- 133.McGlincy N.J., Meacham Z.A., Reynaud K.K., Muller R., Baum R., Ingolia N.T. A genome-scale CRISPR interference guide library enables comprehensive phenotypic profiling in yeast. BMC Genom. 2021;22:205. doi: 10.1186/s12864-021-07518-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Stovicek V., Holkenbrink C., Borodina I. CRISPR/Cas system for yeast genome engineering: Advances and applications. FEMS Yeast Res. 2017;17:fox030. doi: 10.1093/femsyr/fox030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Anzalone A.V., Koblan L.W., Liu D.R. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat. Biotechnol. 2020;38:824–844. doi: 10.1038/s41587-020-0561-9. [DOI] [PubMed] [Google Scholar]
- 136.Hanna R.E., Doench J.G. Design and analysis of CRISPR-Cas experiments. Nat. Biotechnol. 2020;38:813–823. doi: 10.1038/s41587-020-0490-7. [DOI] [PubMed] [Google Scholar]
- 137.Lee H.K., Oh Y., Hong J., Lee S.H., Hur J.K. Development of CRISPR technology for precise single-base genome editing: A brief review. BMB Rep. 2021;54:98–105. doi: 10.5483/BMBRep.2021.54.2.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Prideaux B., Dartois V., Staab D., Weiner D.M., Goh A., Via L.E., Barry C.E., 3rd, Stoeckli M. High-sensitivity MALDI-MRM-MS imaging of moxifloxacin distribution in tuberculosis-infected rabbit lungs and granulomatous lesions. Anal. Chem. 2011;83:2112–2118. doi: 10.1021/ac1029049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Sarathy J.P., Dartois V., Lee E.J. The role of transport mechanisms in Mycobacterium tuberculosis drug resistance and tolerance. Pharmaceuticals. 2012;5:1210–1235. doi: 10.3390/ph5111210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Wang N., Dartois V., Carter C.L. An optimized method for the detection and spatial distribution of aminoglycoside and vancomycin antibiotics in tissue sections by mass spectrometry imaging. J. Mass Spectrom. 2021;56:e4708. doi: 10.1002/jms.4708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Römpp A., Guenther S., Takats Z., Spengler B. Mass spectrometry imaging with high resolution in mass and space (HR2 MSI) for reliable investigation of drug compound distributions on the cellular level. Anal. Bioanal. Chem. 2011;401:65–73. doi: 10.1007/s00216-011-4990-7. [DOI] [PubMed] [Google Scholar]
- 142.Lietz C.B., Gemperline E., Li L. Qualitative and quantitative mass spectrometry imaging of drugs and metabolites. Adv. Drug Deliv. Rev. 2013;65:1074–1085. doi: 10.1016/j.addr.2013.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Morosi L., Zucchetti M., D’Incalci M., Davoli E. Imaging mass spectrometry: Challenges in visualization of drug distribution in solid tumors. Curr. Opin. Pharmacol. 2013;13:807–812. doi: 10.1016/j.coph.2013.06.003. [DOI] [PubMed] [Google Scholar]
- 144.Patel K.J., Trédan O., Tannock I.F. Distribution of the anticancer drugs doxorubicin, mitoxantrone and topotecan in tumors and normal tissues. Cancer Chemother. Pharmacol. 2013;72:127–138. doi: 10.1007/s00280-013-2176-z. [DOI] [PubMed] [Google Scholar]
- 145.Nerini I.F., Morosi L., Zucchetti M., Ballerini A., Giavazzi R., D’Incalci M. lntratumor Heterogeneity and Its Impact on Drug Distribution and Sensitivity. Clin. Pharmacol. Ther. 2014;96:224–238. doi: 10.1038/clpt.2014.105. [DOI] [PubMed] [Google Scholar]
- 146.Prideaux B., Stoeckli M. Mass spectrometry imaging for drug distribution studies. J. Proteom. 2012;75:4999–5013. doi: 10.1016/j.jprot.2012.07.028. [DOI] [PubMed] [Google Scholar]
- 147.Prideaux B., ElNaggar M.S., Zimmerman M., Wiseman J.M., Li X.H., Dartois V. Mass spectrometry imaging of levofloxacin distribution in TB-infected pulmonary lesions by MALDI-MSI and continuous liquid microjunction surface sampling. Int. J. Mass Spectrom. 2015;377:699–708. doi: 10.1016/j.ijms.2014.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Swales J.G., Tucker J.W., Spreadborough M.J., Iverson S.L., Clench M.R., Webborn P.J., Goodwin R.J. Mapping drug distribution in brain tissue using liquid extraction surface analysis mass spectrometry imaging. Anal. Chem. 2015;87:10146–10152. doi: 10.1021/acs.analchem.5b02998. [DOI] [PubMed] [Google Scholar]
- 149.Mann A., Han H., Eyal S. Imaging transporters: Transforming diagnostic and therapeutic development. Clin. Pharmacol. Ther. 2016;100:479–488. doi: 10.1002/cpt.416. [DOI] [PubMed] [Google Scholar]
- 150.Esteve C., Jones E.A., Kell D.B., Boutin H., McDonnell L.A. Mass spectrometry imaging shows major derangements in neurogranin and in purine metabolism in the triple-knockout 3xTg Alzheimer-like mouse model. Biochim. Biophys. Acta. 2017;1865:747–754. doi: 10.1016/j.bbapap.2017.04.002. [DOI] [PubMed] [Google Scholar]
- 151.Prideaux B., Lenaerts A., Dartois V. Imaging and spatially resolved quantification of drug distribution in tissues by mass spectrometry. Curr. Opin. Chem. Biol. 2018;44:93–100. doi: 10.1016/j.cbpa.2018.05.007. [DOI] [PubMed] [Google Scholar]
- 152.Karlsson O., Hanrieder J. Imaging mass spectrometry in drug development and toxicology. Arch. Toxicol. 2017;91:2283–2294. doi: 10.1007/s00204-016-1905-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Sørensen I.S., Janfelt C., Nielsen M.M.B., Mortensen R.W., Knudsen N.Ø., Eriksson A.H., Pedersen A.J., Nielsen K.T. Combination of MALDI-MSI and cassette dosing for evaluation of drug distribution in human skin explant. Anal. Bioanal. Chem. 2017;409:4993–5005. doi: 10.1007/s00216-017-0443-2. [DOI] [PubMed] [Google Scholar]
- 154.Tournier N., Stieger B., Langer O. Imaging techniques to study drug transporter function in vivo. Pharmacol. Ther. 2018;189:104–122. doi: 10.1016/j.pharmthera.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 155.Son H., Jang K., Lee H., Kim S.E., Kang K.W., Lee H. Use of Molecular Imaging in Clinical Drug Development: A Systematic Review. Nucl. Med. Mol. Imaging. 2019;53:208–215. doi: 10.1007/s13139-019-00593-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Gilmore I.S., Heiles S., Pieterse C.L. Metabolic Imaging at the Single-Cell Scale: Recent Advances in Mass Spectrometry Imaging. Annu. Rev. Anal. Chem. 2019;12:201–224. doi: 10.1146/annurev-anchem-061318-115516. [DOI] [PubMed] [Google Scholar]
- 157.Mokosch A.S., Gerbig S., Grevelding C.G., Haeberlein S., Spengler B. High-resolution AP-SMALDI MSI as a tool for drug imaging in Schistosoma mansoni. Anal. Bioanal. Chem. 2021;413:2755–2766. doi: 10.1007/s00216-021-03230-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Newman C.F., Havelund R., Passarelli M.K., Marshall P.S., Francis I., West A., Alexander M.R., Gilmore I.S., Dollery C.T. Intracellular Drug Uptake-A Comparison of Single Cell Measurements Using ToF-SIMS Imaging and Quantification from Cell Populations with LC/MS/MS. Anal. Chem. 2017;89:11944–11953. doi: 10.1021/acs.analchem.7b01436. [DOI] [PubMed] [Google Scholar]
- 159.Fox B.W., Schroeder F.C. Toward spatially resolved metabolomics. Nat. Chem. Biol. 2020;16:1039–1040. doi: 10.1038/s41589-020-00650-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pardridge W.M. Blood-brain barrier endogenous transporters as therapeutic targets: A new model for small molecule CNS drug discovery. Expert Opin. Ther. Targets. 2015;19:1059–1072. doi: 10.1517/14728222.2015.1042364. [DOI] [PubMed] [Google Scholar]
- 161.Brzica H., Abdullahi W., Ibbotson K., Ronaldson P.T. Role of Transporters in Central Nervous System Drug Delivery and Blood-Brain Barrier Protection: Relevance to Treatment of Stroke. J. Cent. Nerv. Syst. Dis. 2017;9:1179573517693802. doi: 10.1177/1179573517693802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Al-Majdoub Z.M., Al Feteisi H., Achour B., Warwood S., Neuhoff S., Rostami-Hodjegan A., Barber J. Proteomic Quantification of Human Blood-Brain Barrier SLC and ABC Transporters in Healthy Individuals and Dementia Patients. Mol. Pharm. 2019;16:1220–1233. doi: 10.1021/acs.molpharmaceut.8b01189. [DOI] [PubMed] [Google Scholar]
- 163.Gomez-Zepeda D., Taghi M., Scherrmann J.M., Decleves X., Menet M.C. ABC Transporters at the Blood-Brain Interfaces, Their Study Models, and Drug Delivery Implications in Gliomas. Pharmaceutics. 2019;12:20. doi: 10.3390/pharmaceutics12010020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Williams E.I., Betterton R.D., Davis T.P., Ronaldson P.T. Transporter-Mediated Delivery of Small Molecule Drugs to the Brain: A Critical Mechanism That Can Advance Therapeutic Development for Ischemic Stroke. Pharmaceutics. 2020;12:154. doi: 10.3390/pharmaceutics12020154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Uhlén M., Fagerberg L., Hallstrom B.M., Lindskog C., Oksvold P., Mardinoglu A., Sivertsson Ǻ., Kampf C., Sjöstedt E., Asplund A., et al. Tissue-based map of the human proteome. Science. 2015;347:1260419. doi: 10.1126/science.1260419. [DOI] [PubMed] [Google Scholar]
- 166.Kell D.B. Control of metabolite efflux in microbial cell factories: Current advances and future prospects. OSF Preprints. 2018:xg9jh. doi: 10.31219/osf.io/xg9jh. [DOI] [Google Scholar]
- 167.Sauer M., Porro D., Mattanovich D., Branduardi P. Microbial production of organic acids: Expanding the markets. Trends Biotechnol. 2008;26:100–108. doi: 10.1016/j.tibtech.2007.11.006. [DOI] [PubMed] [Google Scholar]
- 168.Boyarskiy S., Tullman-Ercek D. Getting pumped: Membrane efflux transporters for enhanced biomolecule production. Curr. Opin. Chem. Biol. 2015;28:15–19. doi: 10.1016/j.cbpa.2015.05.019. [DOI] [PubMed] [Google Scholar]
- 169.Mukhopadhyay A. Tolerance engineering in bacteria for the production of advanced biofuels and chemicals. Trends Microbiol. 2015;23:498–508. doi: 10.1016/j.tim.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 170.Jones C.M., Hernández Lozada N.J., Pfleger B.F. Efflux systems in bacteria and their metabolic engineering applications. Appl. Microbiol. Biotechnol. 2015;99:9381–9393. doi: 10.1007/s00253-015-6963-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Van der Hoek S.A., Borodina I. Transporter engineering in microbial cell factories: The ins, the outs, and the in-betweens. Curr. Opin. Biotechnol. 2020;66:186–194. doi: 10.1016/j.copbio.2020.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Wang G., Møller-Hansen I., Babaei M., D’Ambrosio V., Christensen H.B., Darbani B., Jensen M.K., Borodina I. Transportome-wide engineering of Saccharomyces cerevisiae. Metab. Eng. 2021;64:52–63. doi: 10.1016/j.ymben.2021.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Jezierska S., Van Bogaert I.N.A. Crossing boundaries: The importance of cellular membranes in industrial biotechnology. J. Ind. Microbiol. Biotechnol. 2017;44:721–733. doi: 10.1007/s10295-016-1858-z. [DOI] [PubMed] [Google Scholar]
- 174.Zhu Y., Zhou C., Wang Y., Li C. Transporter Engineering for Microbial Manufacturing. Biotechnol. J. 2020;15:e1900494. doi: 10.1002/biot.201900494. [DOI] [PubMed] [Google Scholar]
- 175.Onyeabor M., Martinez R., Kurgan G., Wang X. Engineering transport systems for microbial production. Adv. Appl. Microbiol. 2020;111:33–87. doi: 10.1016/bs.aambs.2020.01.002. [DOI] [PubMed] [Google Scholar]
- 176.Soares-Silva I., Ribas D., Sousa-Silva M., Azevedo-Silva J., Rendulić T., Casal M. Membrane transporters in the bioproduction of organic acids: State of the art and future perspectives for industrial applications. FEMS Microbiol. Lett. 2020;367:fnaa118. doi: 10.1093/femsle/fnaa118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lane T.S., Rempe C.S., Davitt J., Staton M.E., Peng Y., Soltis D.E., Melkonian M., Deyholos M., Leebens-Mack J.H., Chase M., et al. Diversity of ABC transporter genes across the plant kingdom and their potential utility in biotechnology. BMC Biotechnol. 2016;16:47. doi: 10.1186/s12896-016-0277-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Nielsen J., Keasling J.D. Engineering Cellular Metabolism. Cell. 2016;164:1185–1197. doi: 10.1016/j.cell.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 179.Fang S., Huang X., Zhang X., Zhang M., Hao Y., Guo H., Liu L.N., Yu F., Zhang P. Molecular mechanism underlying transport and allosteric inhibition of bicarbonate transporter SbtA. Proc. Natl. Acad. Sci. USA. 2021;118:e2101632118. doi: 10.1073/pnas.2101632118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Zhang C., Chen X., Stephanopoulos G., Too H.P. Efflux transporter engineering markedly improves amorphadiene production in Escherichia coli. Biotechnol. Bioeng. 2016;113:1755–1763. doi: 10.1002/bit.25943. [DOI] [PubMed] [Google Scholar]
- 181.Steiger M.G., Rassinger A., Mattanovich D., Sauer M. Engineering of the citrate exporter protein enables high citric acid production in Aspergillus niger. Metab. Eng. 2019;52:224–231. doi: 10.1016/j.ymben.2018.12.004. [DOI] [PubMed] [Google Scholar]
- 182.Kurgan G., Kurgan L., Schneider A., Onyeabor M., Rodriguez-Sanchez Y., Taylor E., Martinez R., Carbonell P., Shi X., Gu H., et al. Identification of major malate export systems in an engineered malate-producing Escherichia coli aided by substrate similarity search. Appl. Microbiol. Biotechnol. 2019;103:9001–9011. doi: 10.1007/s00253-019-10164-y. [DOI] [PubMed] [Google Scholar]
- 183.Chen X., Wang Y., Dong X., Hu G., Liu L. Engineering rTCA pathway and C4-dicarboxylate transporter for L-malic acid production. Appl. Microbiol. Biotechnol. 2017;101:4041–4052. doi: 10.1007/s00253-017-8141-8. [DOI] [PubMed] [Google Scholar]
- 184.Cao W., Yan L., Li M., Liu X., Xu Y., Xie Z., Liu H. Identification and engineering a C4-dicarboxylate transporter for improvement of malic acid production in Aspergillus niger. Appl Microbiol. Biotechnol. 2020;104:9773–9783. doi: 10.1007/s00253-020-10932-1. [DOI] [PubMed] [Google Scholar]
- 185.Severi E., Thomas G.H. Antibiotic export: Transporters involved in the final step of natural product production. Microbiology. 2019;165:805–818. doi: 10.1099/mic.0.000794. [DOI] [PubMed] [Google Scholar]
- 186.Salvador López J.M., Van Bogaert I.N.A. Microbial fatty acid transport proteins and their biotechnological potential. Biotechnol. Bioeng. 2021;118:2184–2201. doi: 10.1002/bit.27735. [DOI] [PubMed] [Google Scholar]
- 187.Hu Y., Zhu Z., Nielsen J., Siewers V. Heterologous transporter expression for improved fatty alcohol secretion in yeast. Metab. Eng. 2018;45:51–58. doi: 10.1016/j.ymben.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 188.Mingardon F., Clement C., Hirano K., Nhan M., Luning E.G., Chanal A., Mukhopadhyay A. Improving olefin tolerance and production in E. coli using native and evolved AcrB. Biotechnol. Bioeng. 2015;112:879–888. doi: 10.1002/bit.25511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Darbani B., Stovicek V., Van der Hoek S.A., Borodina I. Engineering energetically efficient transport of dicarboxylic acids in yeast Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA. 2019;116:19415–19420. doi: 10.1073/pnas.1900287116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Lennen R.M., Jensen K., Mohammed E.T., Malla S., Börner R.A., Chekina K., Özdemir E., Bonde I., Koza A., Maury J., et al. Adaptive laboratory evolution reveals general and specific chemical tolerance mechanisms and enhances biochemical production. bioRxiv. 2019:634105. doi: 10.1101/634105. [DOI] [Google Scholar]
- 191.Claus S., Jenkins Sánchez L., Van Bogaert I.N.A. The role of transport proteins in the production of microbial glycolipid biosurfactants. Appl. Microbiol. Biotechnol. 2021;105:1779–1793. doi: 10.1007/s00253-021-11156-7. [DOI] [PubMed] [Google Scholar]
- 192.Jezierska S., Claus S., Van Bogaert I.N.A. Identification and importance of mitochondrial citrate carriers and ATP citrate lyase for glycolipid production in Starmerella bombicola. Appl. Microbiol. Biotechnol. 2020;104:6235–6248. doi: 10.1007/s00253-020-10702-z. [DOI] [PubMed] [Google Scholar]
- 193.Markakis K., Lowe P.T., Davison-Gates L., O’Hagan D., Rosser S.J., Elfick A. An Engineered E. coli Strain for Direct in Vivo Fluorination. ChemBioChem. 2020;21:1856–1860. doi: 10.1002/cbic.202000051. [DOI] [PubMed] [Google Scholar]
- 194.Claus S., Jezierska S., Van Bogaert I.N.A. Protein-facilitated transport of hydrophobic molecules across the yeast plasma membrane. FEBS Lett. 2019;593:1508–1527. doi: 10.1002/1873-3468.13469. [DOI] [PubMed] [Google Scholar]
- 195.Hult K., Berglund P. Enzyme promiscuity: Mechanism and applications. Trends Biotechnol. 2007;25:231–238. doi: 10.1016/j.tibtech.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 196.Babtie A.C., Bandyopadhyay S., Olguin L.F., Hollfelder F. Efficient catalytic promiscuity for chemically distinct reactions. Angew. Chem. Int. Ed. Engl. 2009;48:3692–3694. doi: 10.1002/anie.200805843. [DOI] [PubMed] [Google Scholar]
- 197.Nobeli I., Favia A.D., Thornton J.M. Protein promiscuity and its implications for biotechnology. Nat. Biotechnol. 2009;27:157–167. doi: 10.1038/nbt1519. [DOI] [PubMed] [Google Scholar]
- 198.Carbonell P., Faulon J.L. Molecular signatures-based prediction of enzyme promiscuity. Bioinformatics. 2010;26:2012–2019. doi: 10.1093/bioinformatics/btq317. [DOI] [PubMed] [Google Scholar]
- 199.Carbonell P., Lecointre G., Faulon J.L. Origins of specificity and promiscuity in metabolic networks. J. Biol. Chem. 2011;286:43994–44004. doi: 10.1074/jbc.M111.274050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.O’Hagan S., Kell D.B. Consensus rank orderings of molecular fingerprints illustrate the ‘most genuine’ similarities between marketed drugs and small endogenous human metabolites, but highlight exogenous natural products as the most important ‘natural’ drug transporter substrates. ADMET DMPK. 2017;5:85–125. [Google Scholar]
- 201.Samanta S., O’Hagan S., Swainston N., Roberts T.J., Kell D.B. VAE-Sim: A novel molecular similarity measure based on a variational autoencoder. Molecules. 2020;25:3446. doi: 10.3390/molecules25153446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Sierzputowska K., Baxter C.R., Housden B.E. Variable Dose Analysis: A Novel High-throughput RNAi Screening Method for Drosophila Cells. Bio-Protocol. 2018;8:e3112. doi: 10.21769/BioProtoc.3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Senior E., Bull A.T., Slater J.H. Enzyme evolution in a microbial community growing on the herbicide Dalapon. Nature. 1976;263:476–479. doi: 10.1038/263476a0. [DOI] [PubMed] [Google Scholar]
- 204.Barrick J.E., Yu D.S., Yoon S.H., Jeong H., Oh T.K., Schneider D., Lenski R.E., Kim J.F. Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature. 2009;461:1243–1247. doi: 10.1038/nature08480. [DOI] [PubMed] [Google Scholar]
- 205.Good B.H., McDonald M.J., Barrick J.E., Lenski R.E., Desai M.M. The dynamics of molecular evolution over 60,000 generations. Nature. 2017;551:45–50. doi: 10.1038/nature24287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Tenaillon O., Barrick J.E., Ribeck N., Deatherage D.E., Blanchard J.L., Dasgupta A., Wu G.C., Wielgoss S., Cruveiller S., Médigue C., et al. Tempo and mode of genome evolution in a 50,000-generation experiment. Nature. 2016;536:165–170. doi: 10.1038/nature18959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Khan A.I., Dinh D.M., Schneider D., Lenski R.E., Cooper T.F. Negative epistasis between beneficial mutations in an evolving bacterial population. Science. 2011;332:1193–1196. doi: 10.1126/science.1203801. [DOI] [PubMed] [Google Scholar]
- 208.Woods R.J., Barrick J.E., Cooper T.F., Shrestha U., Kauth M.R., Lenski R.E. Second-order selection for evolvability in a large Escherichia coli population. Science. 2011;331:1433–1436. doi: 10.1126/science.1198914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Lenski R.E., Wiser M.J., Ribeck N., Blount Z.D., Nahum J.R., Morris J.J., Zaman L., Turner C.B., Wade B.D., Maddamsetti R., et al. Sustained fitness gains and variability in fitness trajectories in the long-term evolution experiment with Escherichia coli. Proc. Biol. Sci. 2015;282:20152292. doi: 10.1098/rspb.2015.2292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Van den Bergh B., Swings T., Fauvart M., Michiels J. Experimental Design, Population Dynamics, and Diversity in Microbial Experimental Evolution. Microbiol. Mol. Biol. Rev. 2018;82:e00008-18. doi: 10.1128/MMBR.00008-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Dragosits M., Mattanovich D. Adaptive laboratory evolution—Principles and applications for biotechnology. Microb. Cell Factories. 2013;12:64. doi: 10.1186/1475-2859-12-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.LaCroix R.A., Palsson B.O., Feist A.M. A Model for Designing Adaptive Laboratory Evolution Experiments. Appl. Environ. Microbiol. 2017;83:e03115-16. doi: 10.1128/AEM.03115-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Mundhada H., Seoane J.M., Schneider K., Koza A., Christensen H.B., Klein T., Phaneuf P.V., Herrgard M., Feist A.M., Nielsen A.T. Increased production of L-serine in Escherichia coli through Adaptive Laboratory Evolution. Metab. Eng. 2017;39:141–150. doi: 10.1016/j.ymben.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 214.Pereira R., Wei Y., Mohamed E., Radi M., Malina C., Herrgård M.J., Feist A.M., Nielsen J., Chen Y. Adaptive laboratory evolution of tolerance to dicarboxylic acids in Saccharomyces cerevisiae. Metab. Eng. 2019;56:130–141. doi: 10.1016/j.ymben.2019.09.008. [DOI] [PubMed] [Google Scholar]
- 215.Phaneuf P.V., Gosting D., Palsson B.O., Feist A.M. ALEdb 1.0: A database of mutations from adaptive laboratory evolution experimentation. Nucleic Acids Res. 2019;47:D1164–D1171. doi: 10.1093/nar/gky983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Phaneuf P.V., Yurkovich J.T., Heckmann D., Wu M., Sandberg T.E., King Z.A., Tan J., Palsson B.O., Feist A.M. Causal mutations from adaptive laboratory evolution are outlined by multiple scales of genome annotations and condition-specificity. BMC Genom. 2020;21:514. doi: 10.1186/s12864-020-06920-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Portnoy V.A., Bezdan D., Zengler K. Adaptive laboratory evolution—Harnessing the power of biology for metabolic engineering. Curr. Opin. Biotechnol. 2011;22:590–594. doi: 10.1016/j.copbio.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 218.Reyes L.H., Kao K.C. Growth-Coupled Carotenoids Production Using Adaptive Laboratory Evolution. Methods Mol. Biol. 2018;1671:319–330. doi: 10.1007/978-1-4939-7295-1_20. [DOI] [PubMed] [Google Scholar]
- 219.Winkler J., Reyes L.H., Kao K.C. Adaptive laboratory evolution for strain engineering. Methods Mol. Biol. 2013;985:211–222. doi: 10.1007/978-1-62703-299-5_11. [DOI] [PubMed] [Google Scholar]
- 220.Godara A., Kao K.C. Adaptive laboratory evolution for growth coupled microbial production. World J. Microbiol. Biotechnol. 2020;36:175. doi: 10.1007/s11274-020-02946-8. [DOI] [PubMed] [Google Scholar]
- 221.Lee S., Kim P. Current Status and Applications of Adaptive Laboratory Evolution in Industrial Microorganisms. J. Microbiol. Biotechnol. 2020;30:793–803. doi: 10.4014/jmb.2003.03072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Sandberg T.E., Salazar M.J., Weng L.L., Palsson B.O., Feist A.M. The emergence of adaptive laboratory evolution as an efficient tool for biological discovery and industrial biotechnology. Metab. Eng. 2019;56:1–16. doi: 10.1016/j.ymben.2019.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Zhu Z., Zhang J., Ji X., Fang Z., Wu Z., Chen J., Du G. Evolutionary engineering of industrial microorganisms-strategies and applications. Appl. Microbiol. Biotechnol. 2018;102:4615–4627. doi: 10.1007/s00253-018-8937-1. [DOI] [PubMed] [Google Scholar]
- 224.Dykhuizen D.E., Hartl D.L. Selection in Chemostats. Microbiol. Rev. 1983;47:150–168. doi: 10.1128/mr.47.2.150-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Flegr J. Two distinct types of natural selection in turbidostat-like and chemostat-like ecosystems. J. Theor. Biol. 1997;188:121–126. doi: 10.1006/jtbi.1997.0458. [DOI] [Google Scholar]
- 226.Gresham D., Dunham M.J. The enduring utility of continuous culturing in experimental evolution. Genomics. 2014;104:399–405. doi: 10.1016/j.ygeno.2014.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.McGeachy A.M., Meacham Z.A., Ingolia N.T. An Accessible Continuous-Culture Turbidostat for Pooled Analysis of Complex Libraries. ACS Synth. Biol. 2019;8:844–856. doi: 10.1021/acssynbio.8b00529. [DOI] [PubMed] [Google Scholar]
- 228.Delneri D., Leong H.S., Hayes A., Davey H.M., Kell D.B., Oliver S.G. Assessing contributions to fitness of individual genes via genome-wide competition analysis. Yeast. 2003;20:S337. [Google Scholar]
- 229.Delneri D., Hoyle D.C., Gkargkas K., Cross E.J., Rash B., Zeef L., Leong H.S., Davey H.M., Hayes A., Kell D.B., et al. Identification and characterization of high-flux-control genes of yeast through competition analyses in continuous cultures. Nat. Genet. 2008;40:113–117. doi: 10.1038/ng.2007.49. [DOI] [PubMed] [Google Scholar]
- 230.Pir P., Gutteridge A., Wu J., Rash B., Kell D.B., Zhang N., Oliver S.G. The genetic control of growth rate: A systems biology study in yeast. BMC Syst. Biol. 2012;6:4. doi: 10.1186/1752-0509-6-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Wortel M.T., Bosdriesz E., Teusink B., Bruggeman F.J. Evolutionary pressures on microbial metabolic strategies in the chemostat. Sci. Rep. 2016;6:29503. doi: 10.1038/srep29503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Kell D.B., Oliver S.G. Here is the evidence, now what is the hypothesis? The complementary roles of inductive and hypothesis-driven science in the post-genomic era. Bioessays. 2004;26:99–105. doi: 10.1002/bies.10385. [DOI] [PubMed] [Google Scholar]
- 233.Bennett R.K., Gregory G.J., Gonzalez J.E., Har J.R.G., Antoniewicz M.R., Papoutsakis E.T. Improving the Methanol Tolerance of an Escherichia coli Methylotroph via Adaptive Laboratory Evolution Enhances Synthetic Methanol Utilization. Front. Microbiol. 2021;12:638426. doi: 10.3389/fmicb.2021.638426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Pereira R., Mohamed E.T., Radi M.S., Herrgård M.J., Feist A.M., Nielsen J., Chen Y. Elucidating aromatic acid tolerance at low pH in Saccharomyces cerevisiae using adaptive laboratory evolution. Proc. Natl. Acad. Sci. USA. 2020;117:27954–27961. doi: 10.1073/pnas.2013044117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Mohamed E.T., Wang S., Lennen R.M., Herrgard M.J., Simmons B.A., Singer S.W., Feist A.M. Generation of a platform strain for ionic liquid tolerance using adaptive laboratory evolution. Microb. Cell Factories. 2017;16:204. doi: 10.1186/s12934-017-0819-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Mitchell A.M., Srikumar T., Silhavy T.J. Cyclic Enterobacterial Common Antigen Maintains the Outer Membrane Permeability Barrier of Escherichia coli in a Manner Controlled by YhdP. mBio. 2018;9:e01321-18. doi: 10.1128/mBio.01321-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Grimm J., Shi H., Wang W., Mitchell A.M., Wingreen N.S., Huang K.C., Silhavy T.J. The inner membrane protein YhdP modulates the rate of anterograde phospholipid flow in Escherichia coli. Proc. Natl. Acad. Sci. USA. 2020;117:26907–26914. doi: 10.1073/pnas.2015556117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Avrahami-Moyal L., Engelberg D., Wenger J.W., Sherlock G., Braun S. Turbidostat culture of Saccharomyces cerevisiae W303-1A under selective pressure elicited by ethanol selects for mutations in SSD1 and UTH1. FEMS Yeast Res. 2012;12:521–533. doi: 10.1111/j.1567-1364.2012.00803.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Davey H.M., Davey C.L., Woodward A.M., Edmonds A.N., Lee A.W., Kell D.B. Oscillatory, stochastic and chaotic growth rate fluctuations in permittistatically-controlled yeast cultures. Biosystems. 1996;39:43–61. doi: 10.1016/0303-2647(95)01577-9. [DOI] [PubMed] [Google Scholar]
- 240.Hoffmann S.A., Wohltat C., Muller K.M., Arndt K.M. A user-friendly, low-cost turbidostat with versatile growth rate estimation based on an extended Kalman filter. PLoS ONE. 2017;12:e0181923. doi: 10.1371/journal.pone.0181923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Markx G.H., Davey C.L., Kell D.B. The permittistat: A novel type of turbidostat. J. Gen. Microbiol. 1991;137:735–743. doi: 10.1099/00221287-137-4-735. [DOI] [Google Scholar]
- 242.Munson R.J. Turbidostats. In: Norris J.R., Ribbons D.W., editors. Methods in Microbiology. Vol. 2. Academic Press; London, UK: 1970. pp. 349–376. [Google Scholar]
- 243.Watson T.G. The Present Status and Future Prospects of the Turbidostat. J. Appl. Chem. Biotechnol. 1972;22:229–243. doi: 10.1002/jctb.5020220206. [DOI] [Google Scholar]
- 244.Guarino A., Shannon B., Marucci L., Grierson C., Savery N., Bernardo M. A low-cost,open-sourceTurbidostat design for in-vivo control experiments in Synthetic Biology. IFAC Pap. Online. 2019;52:244–248. doi: 10.1016/j.ifacol.2019.12.265. [DOI] [Google Scholar]
- 245.Takahashi C.N., Miller A.W., Ekness F., Dunham M.J., Klavins E. A low cost, customizable turbidostat for use in synthetic circuit characterization. ACS Synth. Biol. 2015;4:32–38. doi: 10.1021/sb500165g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Harris C.M., Kell D.B. The estimation of microbial biomass. Biosensors. 1985;1:17–84. doi: 10.1016/0265-928X(85)85005-7. [DOI] [PubMed] [Google Scholar]
- 247.Harris C.M., Todd R.W., Bungard S.J., Lovitt R.W., Morris J.G., Kell D.B. The dielectric permittivity of microbial suspensions at radio frequencies: A novel method for the estimation of microbial biomass. Enzym. Microb. Technol. 1987;9:181–186. doi: 10.1016/0141-0229(87)90075-5. [DOI] [Google Scholar]
- 248.Kell D.B., Markx G.H., Davey C.L., Todd R.W. Real-time monitoring of cellular biomass: Methods and applications. Trends Anal. Chem. 1990;9:190–194. doi: 10.1016/0165-9936(90)87042-K. [DOI] [Google Scholar]
- 249.Pirt S.J. Principles of Microbe and Cell Cultivation. Wiley; London, UK: 1975. [Google Scholar]
- 250.Rembeza E., Engqvist M.K. Experimental investigation of enzyme functional annotations reveals extensive annotation error. bioRxiv. 2020 doi: 10.1101/2020.12.18.423474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Borodina I., Kenny L.C., McCarthy C.M., Paramasivan K., Pretorius R., Roberts T.J., Van der Hoek S.A., Kell D.B. The biology of ergothioneine, an antioxidant nutraceutical. Nutr. Res. Rev. 2020;33:190–217. doi: 10.1017/S0954422419000301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Cheah I.K., Halliwell B. Ergothioneine, recent developments. Redox Biol. 2021;42:101868. doi: 10.1016/j.redox.2021.101868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Gründemann D. The ergothioneine transporter controls and indicates ergothioneine activity—A review. Prev. Med. 2012;54:S71–S74. doi: 10.1016/j.ypmed.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 254.Tschirka J., Kreisor M., Betz J., Gründemann D. Substrate selectivity check of the ergothioneine transporter. Drug Metab. Dispos. 2018;46:779–785. doi: 10.1124/dmd.118.080440. [DOI] [PubMed] [Google Scholar]
- 255.Broadhurst D., Goodacre R., Reinke S.N., Kuligowski J., Wilson I.D., Lewis M.R., Dunn W.B. Guidelines and considerations for the use of system suitability and quality control samples in mass spectrometry assays applied in untargeted clinical metabolomic studies. Metabolomics. 2018;14:72. doi: 10.1007/s11306-018-1367-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Dunn W.B., Erban A., Weber R.J.M., Creek D.J., Brown M., Breitling R., Hankemeier T., Goodacre R., Neumann S., Kopka J., et al. Mass Appeal: Metabolite identification in mass spectrometry-focused untargeted metabolomics. Metabolomics. 2013;9:S44–S66. doi: 10.1007/s11306-012-0434-4. [DOI] [Google Scholar]
- 257.Nash W.J., Dunn W.B. From mass to metabolite in human untargeted metabolomics: Recent advances in annotation of metabolites applying liquid chromatography-mass spectrometry data. Trends Anal. Chem. 2019;120:115324. doi: 10.1016/j.trac.2018.11.022. [DOI] [Google Scholar]
- 258.Wright Muelas M., Roberts I., Mughal F., O’Hagan S., Day P.J., Kell D.B. An untargeted metabolomics strategy to measure differences in metabolite uptake and excretion by mammalian cell lines. Metabolomics. 2020;16:107. doi: 10.1007/s11306-020-01725-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Sajid A., Lusvarghi S., Murakami M., Chufan E.E., Abel B., Gottesman M.M., Durell S.R., Ambudkar S.V. Reversing the direction of drug transport mediated by the human multidrug transporter P-glycoprotein. Proc. Natl. Acad. Sci. USA. 2020;117:29609–29617. doi: 10.1073/pnas.2016270117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Sun N., Li D., Fonzi W., Li X., Zhang L., Calderone R. Multidrug-resistant transporter Mdr1p-mediated uptake of a novel antifungal compound. Antimicrob. Agents Chemother. 2013;57:5931–5939. doi: 10.1128/AAC.01504-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Jindal S., Yang L., Day P.J., Kell D.B. Involvement of multiple influx and efflux transporters in the accumulation of cationic fluorescent dyes by Escherichia coli. BMC Microbiol. 2019;19:195. doi: 10.1186/s12866-019-1561-0. also bioRxiv 603688v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Salcedo-Sora J.E., Kell D.B. A quantitative survey of bacterial persistence in the presence of antibiotics: Towards antipersister antimicrobial discovery. Antibiotics. 2020;9:508. doi: 10.3390/antibiotics9080508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Salcedo-Sora J.E., Jindal S., O’Hagan S., Kell D.B. A palette of fluorophores that are differentially accumulated by wild-type and mutant strains of Escherichia coli: Surrogate ligands for bacterial membrane transporters. Microbiology. 2021;167:001016. doi: 10.1099/mic.0.001016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.O’Hagan S., Kell D.B. Structural similarities between some common fluorophores used in biology, marketed drugs, endogenous metabolites, and natural products. Mar. Drugs. 2020;18:582. doi: 10.3390/md18110582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Kaprelyants A.S., Kell D.B. Rapid assessment of bacterial viability and vitality using rhodamine 123 and flow cytometry. J. Appl. Bacteriol. 1992;72:410–422. doi: 10.1111/j.1365-2672.1992.tb01854.x. [DOI] [Google Scholar]
- 266.Kaprelyants A.S., Kell D.B. Dormancy in stationary-phase cultures of Micrococcus luteus: Flow cytometric analysis of starvation and resuscitation. Appl. Env. Microbiol. 1993;59:3187–3196. doi: 10.1128/aem.59.10.3187-3196.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Davey H.M., Kell D.B. Flow cytometry and cell sorting of heterogeneous microbial populations: The importance of single-cell analysis. Microbiol. Rev. 1996;60:641–696. doi: 10.1128/mr.60.4.641-696.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Buranda T., Gineste C., Wu Y., Bondu V., Perez D., Lake K.R., Edwards B.S., Sklar L.A. A High-Throughput Flow Cytometry Screen Identifies Molecules That Inhibit Hantavirus Cell Entry. SLAS Discov. 2018;23:634–645. doi: 10.1177/2472555218766623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Edwards B.S., Sklar L.A. Flow Cytometry: Impact on Early Drug Discovery. J. Biomol. Screen. 2015;20:689–707. doi: 10.1177/1087057115578273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Strouse J.J., Ivnitski-Steele I., Waller A., Young S.M., Perez D., Evangelisti A.M., Ursu O., Bologa C.G., Carter M.B., Salas V.M., et al. Fluorescent substrates for flow cytometric evaluation of efflux inhibition in ABCB1, ABCC1, and ABCG2 transporters. Anal. Biochem. 2013;437:77–87. doi: 10.1016/j.ab.2013.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Tegos G.P., Evangelisti A.M., Strouse J.J., Ursu O., Bologa C., Sklar L.A. A high throughput flow cytometric assay platform targeting transporter inhibition. Drug Disc. Today Technol. 2014;12:e95–e103. doi: 10.1016/j.ddtec.2014.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Jindal S., Thampy H., Day P.J., Kell D.B. Very rapid flow cytometric assessment of antimicrobial susceptibility during the apparent lag phase of bacterial (re)growth. Microbiology. 2019;165:439–454. doi: 10.1099/mic.0.000777. [DOI] [PubMed] [Google Scholar]
- 273.Dragan A.I., Pavlovic R., McGivney J.B., Casas-Finet J.R., Bishop E.S., Strouse R.J., Schenerman M.A., Geddes C.D. SYBR Green I: Fluorescence properties and interaction with DNA. J. Fluoresc. 2012;22:1189–1199. doi: 10.1007/s10895-012-1059-8. [DOI] [PubMed] [Google Scholar]
- 274.Berney M., Vital M., Hülshoff I., Weilenmann H.U., Egli T., Hammes F. Rapid, cultivation-independent assessment of microbial viability in drinking water. Water Res. 2008;42:4010–4018. doi: 10.1016/j.watres.2008.07.017. [DOI] [PubMed] [Google Scholar]
- 275.Hammes F., Berney M., Wang Y., Vital M., Köster O., Egli T. Flow-cytometric total bacterial cell counts as a descriptive microbiological parameter for drinking water treatment processes. Water Res. 2008;42:269–277. doi: 10.1016/j.watres.2007.07.009. [DOI] [PubMed] [Google Scholar]
- 276.Mendes P., Girardi E., Superti-Furga G., Kell D.B. Why most transporter mutations that cause antibiotic resistance are to efflux pumps rather than to import transporters. bioRxiv. 2020 doi: 10.1101/2020.01.16.909507. [DOI] [Google Scholar]
- 277.Featherstone D.E., Broadie K. Wrestling with pleiotropy: Genomic and topological analysis of the yeast gene expression network. Bioessays. 2002;24:267–274. doi: 10.1002/bies.10054. [DOI] [PubMed] [Google Scholar]
- 278.Rudd K.E. Linkage map of Escherichia coli K-12, edition 10: The physical map. Microbiol. Mol. Biol. Rev. 1998;62:985–1019. doi: 10.1128/MMBR.62.3.985-1019.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Ghatak S., King Z.A., Sastry A., Palsson B.O. The y-ome defines the 35% of Escherichia coli genes that lack experimental evidence of function. Nucleic Acids Res. 2019;47:2446–2454. doi: 10.1093/nar/gkz030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Yasir M., Turner A.K., Bastkowski S., Baker D., Page A.J., Telatin A., Phan M.-D., Monahan L., Savva G.M., Darling A., et al. TraDIS-Xpress: A high-resolution whole-genome assay identifies novel mechanisms of triclosan action and resistance. Genome Res. 2020;30:239–249. doi: 10.1101/gr.254391.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Turner A.K., Yasir M., Bastkowski S., Telatin A., Page A.J., Charles I.G., Webber M.A. A genome-wide analysis of Escherichia coli responses to fosfomycin using TraDIS-Xpress reveals novel roles for phosphonate degradation and phosphate transport systems. J. Antimicrob. Chemother. 2020;75:3144–3151. doi: 10.1093/jac/dkaa296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Höglund P.J., Nordström K.J.V., Schiöth H.B., Fredriksson R. The solute carrier families have a remarkably long evolutionary history with the majority of the human families present before divergence of Bilaterian species. Mol. Biol. Evol. 2011;28:1531–1541. doi: 10.1093/molbev/msq350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Hediger M.A., Clemencon B., Burrier R.E., Bruford E.A. The ABCs of membrane transporters in health and disease (SLC series): Introduction. Mol. Aspects Med. 2013;34:95–107. doi: 10.1016/j.mam.2012.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Rives M.L., Javitch J.A., Wickenden A.D. Potentiating SLC transporter activity: Emerging drug discovery opportunities. Biochem Pharmacol. 2017;135:1–11. doi: 10.1016/j.bcp.2017.02.010. [DOI] [PubMed] [Google Scholar]
- 285.Liu X. SLC Family Transporters. Adv. Exp. Med. Biol. 2019;1141:101–202. doi: 10.1007/978-981-13-7647-4_3. [DOI] [PubMed] [Google Scholar]
- 286.Colas C., Ung P.M.U., Schlessinger A. SLC transporters: Structure, function, and drug discovery. Medchemcomm. 2016;7:1069–1081. doi: 10.1039/C6MD00005C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Pizzagalli M.D., Bensimon A., Superti-Furga G. A guide to plasma membrane solute carrier proteins. FEBS J. 2021;288:2784–2835. doi: 10.1111/febs.15531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.César-Razquin A., Snijder B., Frappier-Brinton T., Isserlin R., Gyimesi G., Bai X., Reithmeier R.A., Hepworth D., Hediger M.A., Edwards A.M., et al. A call for systematic research on solute carriers. Cell. 2015;162:478–487. doi: 10.1016/j.cell.2015.07.022. [DOI] [PubMed] [Google Scholar]
- 289.Kory N., Uit de Bos J., Van der Rijt S., Jankovic N., Gura M., Arp N., Pena I.A., Prakash G., Chan S.H., Kunchok T., et al. MCART1/SLC25A51 is required for mitochondrial NAD transport. Sci. Adv. 2020;6:eabe5310. doi: 10.1126/sciadv.abe5310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Nour-Eldin H.H., Nørholm M.H.H., Halkier B.A. Screening for plant transporter function by expressing a normalized Arabidopsis full-length cDNA library in Xenopus oocytes. Plant. Methods. 2006;2:17. doi: 10.1186/1746-4811-2-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Pfefferkorn J.A., Litchfield J., Hutchings R., Cheng X.M., Larsen S.D., Auerbach B., Bush M.R., Lee C., Erasga N., Bowles D.M., et al. Discovery of novel hepatoselective HMG-CoA reductase inhibitors for treating hypercholesterolemia: A bench-to-bedside case study on tissue selective drug distribution. Bioorg. Med. Chem. Lett. 2011;21:2725–2731. doi: 10.1016/j.bmcl.2010.11.103. [DOI] [PubMed] [Google Scholar]
- 292.Pfefferkorn J.A., Guzman-Perez A., Litchfield J., Aiello R., Treadway J.L., Pettersen J., Minich M.L., Filipski K.J., Jones C.S., Tu M., et al. Discovery of (S)-6-(3-cyclopentyl-2-(4-(trifluoromethyl)-1H-imidazol-1-yl)propanamido)nicotini c acid as a hepatoselective glucokinase activator clinical candidate for treating type 2 diabetes mellitus. J. Med. Chem. 2012;55:1318–1333. doi: 10.1021/jm2014887. [DOI] [PubMed] [Google Scholar]
- 293.Meyer zu Schwabedissen H.E., Robert B., Hussner J., Juhnke B.O., Gliesche D., Boettcher K., Sternberg K., Schmitz K.P., Kroemer H.K. Cell specific expression of uptake transporters—A potential approach for cardiovascular drug delivery devices. Mol. Pharm. 2014;11:665–672. doi: 10.1021/mp400245g. [DOI] [PubMed] [Google Scholar]
- 294.Grixti J., O’Hagan S., Day P.J., Kell D.B. Enhancing drug efficacy and therapeutic index through cheminformatics-based selection of small molecule binary weapons that improve transporter-mediated targeting: A cytotoxicity system based on gemcitabine. Front. Pharmacol. 2017;8:155. doi: 10.3389/fphar.2017.00155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Orozco C.C., Atkinson K., Ryu S., Chang G., Keefer C., Lin J., Riccardi K., Mongillo R.K., Tess D., Filipski K.J., et al. Structural attributes influencing unbound tissue distribution. Eur. J. Med. Chem. 2020;185:111813. doi: 10.1016/j.ejmech.2019.111813. [DOI] [PubMed] [Google Scholar]
- 296.Nyquist M.D., Corella A., Burns J., Coleman I., Gao S., Tharakan R., Riggan L., Cai C., Corey E., Nelson P.S., et al. Exploiting AR-Regulated Drug Transport to Induce Sensitivity to the Survivin Inhibitor YM155. Mol. Cancer Res. 2017;15:521–531. doi: 10.1158/1541-7786.MCR-16-0315-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Nyquist M.D., Prasad B., Mostaghel E.A. Harnessing Solute Carrier Transporters for Precision Oncology. Molecules. 2017;22:539. doi: 10.3390/molecules22040539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Al-Abdulla R., Perez-Silva L., Abete L., Romero M.R., Briz O., Marin J.J.G. Unraveling ‘The Cancer Genome Atlas’ information on the role of SLC transporters in anticancer drug uptake. Expert Rev. Clin. Pharmacol. 2019;12:329–341. doi: 10.1080/17512433.2019.1581605. [DOI] [PubMed] [Google Scholar]
- 299.Huang K.M., Uddin M.E., DiGiacomo D., Lustberg M.B., Hu S., Sparreboom A. Role of SLC transporters in toxicity induced by anticancer drugs. Expert Opin. Drug Metab. Toxicol. 2020;16:493–506. doi: 10.1080/17425255.2020.1755253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Wu Z., Xu J., Liang C., Meng Q., Hua J., Wang W., Zhang B., Liu J., Yu X., Shi S. Emerging roles of the solute carrier family in pancreatic cancer. Clin. Transl. Med. 2021;11:e356. doi: 10.1002/ctm2.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Ceriani L., Verme P. The origins of the Gini index: Extracts from Variabilità e Mutabilità (1912) by Corrado Gini. J. Econ. Inequal. 2012;10:421–443. doi: 10.1007/s10888-011-9188-x. [DOI] [Google Scholar]
- 302.Lv X., Zhang G., Ren G. Gini index estimation for lifetime data. Lifetime Data Anal. 2017;23:275–304. doi: 10.1007/s10985-016-9357-0. [DOI] [PubMed] [Google Scholar]
- 303.Lawal H.O., Krantz D.E. SLC18: Vesicular neurotransmitter transporters for monoamines and acetylcholine. Mol. Aspects Med. 2013;34:360–372. doi: 10.1016/j.mam.2012.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Wilkinson R., Pickett K. The Spirit Level: Why Equality Is Better for Everyone. Penguin Books; London, UK: 2009. [Google Scholar]
- 305.Mortazavi A., Williams B.A., McCue K., Schaeffer L., Wold B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods. 2008;5:621–658. doi: 10.1038/nmeth.1226. [DOI] [PubMed] [Google Scholar]
- 306.Wagner G.P., Kin K., Lynch V.J. Measurement of mRNA abundance using RNA-seq data: RPKM measure is inconsistent among samples. Theory Biosci. 2012;131:281–285. doi: 10.1007/s12064-012-0162-3. [DOI] [PubMed] [Google Scholar]
- 307.Zhao S., Ye Z., Stanton R. Misuse of RPKM or TPM normalization when comparing across samples and sequencing protocols. RNA. 2020;26:903–909. doi: 10.1261/rna.074922.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Anand B.S., Dey S., Mitra A.K. Current prodrug strategies via membrane transporters/receptors. Expert Opin. Biol. Ther. 2002;2:607–620. doi: 10.1517/14712598.2.6.607. [DOI] [PubMed] [Google Scholar]
- 309.Clas S.D., Sanchez R.I., Nofsinger R. Chemistry-enabled drug delivery (prodrugs): Recent progress and challenges. Drug Discov. Today. 2014;19:79–87. doi: 10.1016/j.drudis.2013.08.014. [DOI] [PubMed] [Google Scholar]
- 310.Huttunen K.M., Raunio H., Rautio J. Prodrugs—From Serendipity to Rational Design. Pharmacol. Rev. 2011;63:750–771. doi: 10.1124/pr.110.003459. [DOI] [PubMed] [Google Scholar]
- 311.Majumdar S., Duvvuri S., Mitra A.K. Membrane transporter/receptor-targeted prodrug design: Strategies for human and veterinary drug development. Adv. Drug Deliv. Rev. 2004;56:1437–1452. doi: 10.1016/j.addr.2004.02.006. [DOI] [PubMed] [Google Scholar]
- 312.Mazzaferro S., Bouchemal K., Ponchel G. Oral delivery of anticancer drugs II: The prodrug strategy. Drug Discov. Today. 2013;18:93–98. doi: 10.1016/j.drudis.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 313.Sievaen E. Exploitation of bile acid transport systems in prodrug design. Molecules. 2007;12:1859–1889. doi: 10.3390/12081859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Sinokrot H., Smerat T., Najjar A., Karaman R. Advanced Prodrug Strategies in Nucleoside and Non-Nucleoside Antiviral Agents: A Review of the Recent Five Years. Molecules. 2017;22:1736. doi: 10.3390/molecules22101736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Yang C., Tirucherai G.S., Mitra A.K. Prodrug based optimal drug delivery via membrane transporter/receptor. Expert Opin. Biol. Ther. 2001;1:159–175. doi: 10.1517/14712598.1.2.159. [DOI] [PubMed] [Google Scholar]
- 316.Zhang Y., Sun J., Sun Y., Wang Y., He Z. Prodrug Design Targeting Intestinal PepT1 for Improved Oral Absorption: Design and Performance. Curr. Drug Metab. 2013;14:675–687. doi: 10.2174/1389200211314060004. [DOI] [PubMed] [Google Scholar]
- 317.Minhas G.S., Newstead S. Structural basis for prodrug recognition by the SLC15 family of proton-coupled peptide transporters. Proc. Natl. Acad. Sci. USA. 2019;116:804–809. doi: 10.1073/pnas.1813715116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Minhas G.S., Newstead S. Recent advances in understanding prodrug transport through the SLC15 family of proton-coupled transporters. Biochem. Soc. Trans. 2020;48:337–346. doi: 10.1042/BST20180302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Huttunen J., Peltokangas S., Gynther M., Natunen T., Hiltunen M., Auriola S., Ruponen M., Vellonen K.S., Huttunen K.M. L-Type Amino Acid Transporter 1 (LAT1/Lat1)-Utilizing Prodrugs Can Improve the Delivery of Drugs into Neurons, Astrocytes and Microglia. Sci. Rep. 2019;9:12860. doi: 10.1038/s41598-019-49009-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Huttunen J., Gynther M., Vellonen K.S., Huttunen K.M. L-Type amino acid transporter 1 (LAT1)-utilizing prodrugs are carrier-selective despite having low affinity for organic anion transporting polypeptides (OATPs) Int. J. Pharm. 2019;571:118714. doi: 10.1016/j.ijpharm.2019.118714. [DOI] [PubMed] [Google Scholar]
- 321.Montaser A.B., Jarvinen J., Löffler S., Huttunen J., Auriola S., Lehtonen M., Jalkanen A., Huttunen K.M. L-Type Amino Acid Transporter 1 Enables the Efficient Brain Delivery of Small-Sized Prodrug across the Blood-Brain Barrier and into Human and Mouse Brain Parenchymal Cells. ACS Chem. Neurosci. 2020;11:4301–4315. doi: 10.1021/acschemneuro.0c00564. [DOI] [PubMed] [Google Scholar]
- 322.Montaser A., Lehtonen M., Gynther M., Huttunen K.M. L-Type Amino Acid Transporter 1-Utilizing Prodrugs of Ketoprofen Can Efficiently Reduce Brain Prostaglandin Levels. Pharmaceutics. 2020;12:344. doi: 10.3390/pharmaceutics12040344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Peura L., Malmioja K., Huttunen K., Leppanen J., Hämäläinen M., Forsberg M.M., Gynther M., Rautio J., Laine K. Design, synthesis and brain uptake of LAT1-targeted amino acid prodrugs of dopamine. Pharm. Res. 2013;30:2523–2537. doi: 10.1007/s11095-012-0966-3. [DOI] [PubMed] [Google Scholar]
- 324.Puris E., Gynther M., Huttunen J., Petsalo A., Huttunen K.M. L-type amino acid transporter 1 utilizing prodrugs: How to achieve effective brain delivery and low systemic exposure of drugs. J. Control. Release. 2017;261:93–104. doi: 10.1016/j.jconrel.2017.06.023. [DOI] [PubMed] [Google Scholar]
- 325.Pardridge W.M. Blood-brain barrier delivery. Drug Discov. Today. 2007;12:54–61. doi: 10.1016/j.drudis.2006.10.013. [DOI] [PubMed] [Google Scholar]
- 326.Agbabiaka T.B., Savovic J., Ernst E. Methods for causality assessment of adverse drug reactions: A systematic review. Drug Saf. 2008;31:21–37. doi: 10.2165/00002018-200831010-00003. [DOI] [PubMed] [Google Scholar]
- 327.Davies E.C., Green C.F., Mottram D.R., Pirmohamed M. Adverse drug reactions in hospitals: A narrative review. Curr. Drug Saf. 2007;2:79–87. doi: 10.2174/157488607779315507. [DOI] [PubMed] [Google Scholar]
- 328.Hazell L., Shakir S.A. Under-reporting of adverse drug reactions: A systematic review. Drug Saf. 2006;29:385–396. doi: 10.2165/00002018-200629050-00003. [DOI] [PubMed] [Google Scholar]
- 329.Ji Z.L., Han L.Y., Yap C.W., Sun L.Z., Chen X., Chen Y.Z. Drug Adverse Reaction Target Database (DART): Proteins related to adverse drug reactions. Drug Saf. 2003;26:685–690. doi: 10.2165/00002018-200326100-00002. [DOI] [PubMed] [Google Scholar]
- 330.King C., McKenna A., Farzan N., Vijverberg S.J., Van der Schee M.P., Maitland-van der Zee A.H., Arianto L., Bisgaard H., BØnnelykke K., Berce V., et al. Pharmacogenomic associations of adverse drug reactions in asthma: Systematic review and research prioritisation. Pharm. J. 2020;20:621–628. doi: 10.1038/s41397-019-0140-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Miguel A., Azevedo L.F., Araújo M., Pereira A.C. Frequency of adverse drug reactions in hospitalized patients: A systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2012;21:1139–1154. doi: 10.1002/pds.3309. [DOI] [PubMed] [Google Scholar]
- 332.Osanlou O., Pirmohamed M., Daly A.K. Pharmacogenetics of Adverse Drug Reactions. Adv. Pharmacol. 2018;83:155–190. doi: 10.1016/bs.apha.2018.03.002. [DOI] [PubMed] [Google Scholar]
- 333.Pirmohamed M. Pharmacogenetics of idiosyncratic adverse drug reactions. Handb. Exp. Pharmacol. 2010;196:477–491. doi: 10.1007/978-3-642-00663-0_17. [DOI] [PubMed] [Google Scholar]
- 334.Pirmohamed M. Personalized Pharmacogenomics: Predicting Efficacy and Adverse Drug Reactions. Annu. Rev. Genom. Hum. Genet. 2014;15:349–370. doi: 10.1146/annurev-genom-090413-025419. [DOI] [PubMed] [Google Scholar]
- 335.Sakiris M.A., Sawan M., Hilmer S.N., Awadalla R., Gnjidic D. Prevalence of adverse drug events and adverse drug reactions in hospital among older patients with dementia: A systematic review. Br. J. Clin. Pharmacol. 2021;87:375–385. doi: 10.1111/bcp.14417. [DOI] [PubMed] [Google Scholar]
- 336.Su S.C., Chung W.H., Hung S.I. Digging up the Human Genome: Current Progress in Deciphering Adverse Drug Reactions. BioMed Res. Int. 2014;2014:824343. doi: 10.1155/2014/824343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Wei C.Y., Lee M.T., Chen Y.T. Pharmacogenomics of adverse drug reactions: Implementing personalized medicine. Hum. Mol. Genet. 2012;21:R58–R65. doi: 10.1093/hmg/dds341. [DOI] [PubMed] [Google Scholar]
- 338.Wilke R.A., Lin D.W., Roden D.M., Watkins P.B., Flockhart D., Zineh I., Giacomini K.M., Krauss R.M. Identifying genetic risk factors for serious adverse drug reactions: Current progress and challenges. Nat. Rev. Drug Discov. 2007;6:904–916. doi: 10.1038/nrd2423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Zhang L.L., Yang S., Wei W., Zhang X.J. Genetic polymorphisms affect efficacy and adverse drug reactions of DMARDs in rheumatoid arthritis. Pharm. Genom. 2014;24:531–538. doi: 10.1097/FPC.0000000000000085. [DOI] [PubMed] [Google Scholar]
- 340.Zolk O., Fromm M.F. Transporter-mediated drug uptake and efflux: Important determinants of adverse drug reactions. Clin. Pharmacol. Ther. 2011;89:798–805. doi: 10.1038/clpt.2010.354. [DOI] [PubMed] [Google Scholar]
- 341.Hakkarainen K.M., Andersson Sundell K., Petzold M., Hagg S. Prevalence and perceived preventability of self-reported adverse drug events--a population-based survey of 7099 adults. PLoS ONE. 2013;8:e73166. doi: 10.1371/journal.pone.0073166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Benfenati E. In Silico Methods for Predicting Drug Toxicity. Springer; Berlin/Heidelber, Germany: 2016. [Google Scholar]
- 343.Gyllensten H., Jönsson A.K., Hakkarainen K.M., Svensson S., Hägg S., Rehnberg C. Comparing Methods for Estimating Direct Costs of Adverse Drug Events. Value Health. 2017;20:1299–1310. doi: 10.1016/j.jval.2017.06.007. [DOI] [PubMed] [Google Scholar]
- 344.Giblin K.A., Basili D., Afzal A.M., Rosenbrier-Ribeiro L., Greene N., Barrett I., Hughes S.J., Bender A. New Associations between Drug-Induced Adverse Events in Animal Models and Humans Reveal Novel Candidate Safety Targets. Chem. Res. Toxicol. 2021;34:438–451. doi: 10.1021/acs.chemrestox.0c00311. [DOI] [PubMed] [Google Scholar]
- 345.Insani W.N., Whittlesea C., Alwafi H., Man K.K.C., Chapman S., Wei L. Prevalence of adverse drug reactions in the primary care setting: A systematic review and meta-analysis. PLoS ONE. 2021;16:e0252161. doi: 10.1371/journal.pone.0252161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Tong H., Phan N.V.T., Nguyen T.T., Nguyen D.V., Vo N.S., Le L. Review on Databases and Bioinformatic Approaches on Pharmacogenomics of Adverse Drug Reactions. Pharmgenom. Pers. Med. 2021;14:61–75. doi: 10.2147/PGPM.S290781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Cook D., Brown D., Alexander R., March R., Morgan P., Satterthwaite G., Pangalos M.N. Lessons learned from the fate of AstraZeneca’s drug pipeline: A five-dimensional framework. Nat. Rev. Drug Discov. 2014;13:419–431. doi: 10.1038/nrd4309. [DOI] [PubMed] [Google Scholar]
- 348.Morgan P., Brown D.G., Lennard S., Anderton M.J., Barrett J.C., Eriksson U., Fidock M., Hamren B., Johnson A., March R.E., et al. Impact of a five-dimensional framework on R&D productivity at AstraZeneca. Nat. Rev. Drug Discov. 2018;17:167–181. doi: 10.1038/nrd.2017.244. [DOI] [PubMed] [Google Scholar]
- 349.Kola I., Landis J. Can the pharmaceutical industry reduce attrition rates? Nat. Rev. Drug Discov. 2004;3:711–715. doi: 10.1038/nrd1470. [DOI] [PubMed] [Google Scholar]
- 350.Leeson P.D., Empfield J.R. Reducing the risk of drug attrition associated with physicochemical properties. Annu. Rep. Med. Chem. 2010;45:393–407. [Google Scholar]
- 351.Muthas D., Boyer S., Hasselgren C. A critical assessment of modeling safety-related drug attrition. MedChemComm. 2013;4:1058–1065. doi: 10.1039/c3md00072a. [DOI] [Google Scholar]
- 352.Waring M.J., Arrowsmith J., Leach A.R., Leeson P.D., Mandrell S., Owen R.M., Pairaudeau G., Pennie W.D., Pickett S.D., Wang J., et al. An analysis of the attrition of drug candidates from four major pharmaceutical companies. Nat. Rev. Drug Discov. 2015;14:475–486. doi: 10.1038/nrd4609. [DOI] [PubMed] [Google Scholar]
- 353.Liu Z., Delavan B., Roberts R., Tong W. Lessons Learned from Two Decades of Anticancer Drugs. Trends Pharmacol. Sci. 2017;38:852–872. doi: 10.1016/j.tips.2017.06.005. [DOI] [PubMed] [Google Scholar]
- 354.Williams R.J. Biochemical Individuality. John Wiley; New York, NY, USA: 1956. [Google Scholar]
- 355.Kell D.B., Ryder H.M., Kaprelyants A.S., Westerhoff H.V. Quantifying heterogeneity: Flow cytometry of bacterial cultures. Antonie van Leeuwenhoek. 1991;60:145–158. doi: 10.1007/BF00430362. [DOI] [PubMed] [Google Scholar]
- 356.Amur S., Zineh I., Abernethy D.R., Huang S.M., Lesko L.J. Pharmacogenomics and adverse drug reactions. Pers. Med. 2010;7:633–642. doi: 10.2217/pme.10.63. [DOI] [PubMed] [Google Scholar]
- 357.Clarke J.D., Cherrington N.J. Genetics or environment in drug transport: The case of organic anion transporting polypeptides and adverse drug reactions. Expert Opin. Drug Metab. Toxicol. 2012;8:349–360. doi: 10.1517/17425255.2012.656087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Daly A.K. Using Genome-Wide Association Studies to Identify Genes Important in Serious Adverse Drug Reactions. Annu. Rev. Pharmacol. Toxicol. 2012;52:21–35. doi: 10.1146/annurev-pharmtox-010611-134743. [DOI] [PubMed] [Google Scholar]
- 359.Giorgi M.A., Caroli C., Arazi H.C., Di Girolamo G. Pharmacogenomics and adverse drug reactions: The case of statins. Expert Opin. Pharmacother. 2011;12:1499–1509. doi: 10.1517/14656566.2011.563734. [DOI] [PubMed] [Google Scholar]
- 360.Kato M., Fukuda T., Wakeno M., Fukuda K., Okugawa G., Ikenaga Y., Yamashita M., Takekita Y., Nobuhara K., Azuma J., et al. Effects of the serotonin type 2A, 3A and 3B receptor and the serotonin transporter genes on paroxetine and fluvoxamine efficacy and adverse drug reactions in depressed Japanese patients. Neuropsychobiology. 2006;53:186–195. doi: 10.1159/000094727. [DOI] [PubMed] [Google Scholar]
- 361.Mealey K.L. Adverse drug reactions in veterinary patients associated with drug transporters. Vet. Clin. N. Am. Small Anim. Pract. 2013;43:1067–1078. doi: 10.1016/j.cvsm.2013.04.004. [DOI] [PubMed] [Google Scholar]
- 362.Meyer U.A. Pharmacogenetics and adverse drug reactions. Lancet. 2000;356:1667–1671. doi: 10.1016/S0140-6736(00)03167-6. [DOI] [PubMed] [Google Scholar]
- 363.Rollason V., Lloret-Linares C., Lorenzini K.I., Daali Y., Gex-Fabry M., Piguet V., Besson M., Samer C., Desmeules J. Evaluation of Phenotypic and Genotypic Variations of Drug Metabolising Enzymes and Transporters in Chronic Pain Patients Facing Adverse Drug Reactions or Non-Response to Analgesics: A Retrospective Study. J. Pers. Med. 2020;10:198. doi: 10.3390/jpm10040198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Zhou Y., Zhang G.Q., Wei Y.H., Zhang J.P., Zhang G.R., Ren J.X., Duan H.G., Rao Z., Wu X.A. The impact of drug transporters on adverse drug reaction. Eur. J. Drug Metab. Pharm. 2013;38:77–85. doi: 10.1007/s13318-013-0117-1. [DOI] [PubMed] [Google Scholar]
- 365.Bodo A., Bakos E., Szeri F., Varadi A., Sarkadi B. The role of multidrug transporters in drug availability, metabolism and toxicity. Toxicol. Lett. 2003;140–141:133–143. doi: 10.1016/S0378-4274(02)00497-6. [DOI] [PubMed] [Google Scholar]
- 366.Ciarimboli G. Role of organic cation transporters in drug-induced toxicity. Expert Opin. Drug Metab. Toxicol. 2011;7:159–174. doi: 10.1517/17425255.2011.547474. [DOI] [PubMed] [Google Scholar]
- 367.Ciarimboli G., Holle S.K., Vollenbrocker B., Hagos Y., Reuter S., Burckhardt G., Bierer S., Herrmann E., Pavenstadt H., Rossi R., et al. New Clues for Nephrotoxicity Induced by Ifosfamide: Preferential Renal Uptake via the Human Organic Cation Transporter 2. Mol. Pharm. 2011;8:270–279. doi: 10.1021/mp100329u. [DOI] [PubMed] [Google Scholar]
- 368.Damaraju V.L., Mowles D., Yao S., Ng A., Young J.D., Cass C.E., Tong Z. Role of human nucleoside transporters in the uptake and cytotoxicity of azacitidine and decitabine. Nucleosides Nucleotides Nucleic Acids. 2012;31:236–255. doi: 10.1080/15257770.2011.652330. [DOI] [PubMed] [Google Scholar]
- 369.Damaraju V.L., Scriver T., Mowles D., Kuzma M., Ryan A.J., Cass C.E., Sawyer M.B. Erlotinib, Gefitinib, and Vandetanib Inhibit Human Nucleoside Transporters and Protect Cancer Cells from Gemcitabine Cytotoxicity. Clin. Cancer Res. 2014;20:176–186. doi: 10.1158/1078-0432.CCR-13-2293. [DOI] [PubMed] [Google Scholar]
- 370.DeGorter M.K., Xia C.Q., Yang J.J., Kim R.B. Drug Transporters in Drug Efficacy and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2012;52:249–273. doi: 10.1146/annurev-pharmtox-010611-134529. [DOI] [PubMed] [Google Scholar]
- 371.Elwi A.N., Damaraju V.L., Kuzma M.L., Baldwin S.A., Young J.D., Sawyer M.B., Cass C.E. Human concentrative nucleoside transporter 3 is a determinant of fludarabine transportability and cytotoxicity in human renal proximal tubule cell cultures. Cancer Chemother. Pharmacol. 2009;63:289–301. doi: 10.1007/s00280-008-0739-1. [DOI] [PubMed] [Google Scholar]
- 372.Feng B., El-Kattan A.F., Radi Z.A. Renal transporters in drug disposition, drug-drug interactions, and nephrotoxicity. Curr. Protoc. Toxicol. 2012;53:23.3.1–23.3.15. doi: 10.1002/0471140856.tx2303s53. [DOI] [PubMed] [Google Scholar]
- 373.Fischer A., Hoeger S.J., Stemmer K., Feurstein D.J., Knobeloch D., Nussler A., Dietrich D.R. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010;245:9–20. doi: 10.1016/j.taap.2010.02.006. [DOI] [PubMed] [Google Scholar]
- 374.Fisel P., Renner O., Nies A.T., Schwab M., Schaeffeler E. Solute carrier transporter and drug-related nephrotoxicity: The impact of proximal tubule cell models for preclinical research. Expert Opin. Drug Metab. Toxicol. 2014;10:395–408. doi: 10.1517/17425255.2014.876990. [DOI] [PubMed] [Google Scholar]
- 375.Huang K.M., Hu S., Sparreboom A. Drug transporters and anthracycline-induced cardiotoxicity. Pharmacogenomics. 2018;19:883–888. doi: 10.2217/pgs-2018-0056. [DOI] [PubMed] [Google Scholar]
- 376.Jabir R.S., Naidu R., Annuar M.A., Ho G.F., Munisamy M., Stanslas J. Pharmacogenetics of taxanes: Impact of gene polymorphisms of drug transporters on pharmacokinetics and toxicity. Pharmacogenomics. 2012;13:1979–1988. doi: 10.2217/pgs.12.165. [DOI] [PubMed] [Google Scholar]
- 377.Kamal M.A., Keep R.F., Smith D.E. Role and relevance of PEPT2 in drug disposition, dynamics, and toxicity. Drug Metab. Pharm. 2008;23:236–242. doi: 10.2133/dmpk.23.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Krajcsi P., Vereczkey L. Transporter-drug interactions and transporter-mediated toxicity in the liver/hepatocyte. Preface. Drug Metab. Rev. 2010;42:379. doi: 10.3109/03602530903492129. [DOI] [PubMed] [Google Scholar]
- 379.Li S., Chen Y., Zhang S., More S.S., Huang X., Giacomini K.M. Role of organic cation transporter 1, OCT1 in the pharmacokinetics and toxicity of cis-diammine(pyridine)chloroplatinum(II) and oxaliplatin in mice. Pharm. Res. 2011;28:610–625. doi: 10.1007/s11095-010-0312-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Mor A.L., Kaminski T.W., Karbowska M., Pawlak D. New Insight into Organic Anion Transporters from the Perspective of Potentially Important Interactions and Drugs Toxicity. J. Physiol. Pharmacol. 2018;69:307–324. doi: 10.26402/jpp.2018.3.01. [DOI] [PubMed] [Google Scholar]
- 381.More S.S., Li S., Yee S.W., Chen L., Xu Z., Jablons D.M., Giacomini K.M. Organic cation transporters modulate the uptake and cytotoxicity of picoplatin, a third-generation platinum analogue. Mol. Cancer Ther. 2010;9:1058–1069. doi: 10.1158/1535-7163.MCT-09-1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Nakamura T., Yonezawa A., Hashimoto S., Katsura T., Inui K. Disruption of multidrug and toxin extrusion MATE1 potentiates cisplatin-induced nephrotoxicity. Biochem. Pharmacol. 2010;80:1762–1767. doi: 10.1016/j.bcp.2010.08.019. [DOI] [PubMed] [Google Scholar]
- 383.Niemi M. Transporter pharmacogenetics and statin toxicity. Clin. Pharmacol. Ther. 2010;87:130–133. doi: 10.1038/clpt.2009.197. [DOI] [PubMed] [Google Scholar]
- 384.Parmar S., Seeringer A., Denich D., Gärtner F., Pitterle K., Syrovets T., Ohmle B., Stingl J.C. Variability in transport and biotransformation of cytarabine is associated with its toxicity in peripheral blood mononuclear cells. Pharmacogenomics. 2011;12:503–514. doi: 10.2217/pgs.10.200. [DOI] [PubMed] [Google Scholar]
- 385.Schuetz J.D., Swaan P.W., Tweedie D.J. The role of transporters in toxicity and disease. Drug Metab. Dispos. 2014;42:541–545. doi: 10.1124/dmd.114.057539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Sprowl J.A., Ciarimboli G., Lancaster C.S., Giovinazzo H., Gibson A.A., Du G.Q., Janke L.J., Cavaletti G., Shields A.F., Sparreboom A. Oxaliplatin-induced neurotoxicity is dependent on the organic cation transporter OCT2. Proc. Natl. Acad. Sci. USA. 2013;110:11199–11204. doi: 10.1073/pnas.1305321110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Szakács G., Váradi A., Özvegy-Laczka C., Sarkadi B. The role of ABC transporters in drug absorption, distribution, metabolism, excretion and toxicity (ADME-Tox) Drug Discov. Today. 2008;13:379–393. doi: 10.1016/j.drudis.2007.12.010. [DOI] [PubMed] [Google Scholar]
- 388.Visscher H., Rassekh S.R., Sandor G.S., Caron H.N., Van Dalen E.C., Kremer L.C., Van der Pal H.J., Rogers P.C., Rieder M.J., Carleton B.C., et al. Genetic variants in SLC22A17 and SLC22A7 are associated with anthracycline-induced cardiotoxicity in children. Pharmacogenomics. 2015;16:1065–1076. doi: 10.2217/pgs.15.61. [DOI] [PubMed] [Google Scholar]
- 389.Wang L., Sweet D.H. Renal organic anion transporters (SLC22 family): Expression, regulation, roles in toxicity, and impact on injury and disease. AAPS J. 2013;15:53–69. doi: 10.1208/s12248-012-9413-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Zhang S., Lovejoy K.S., Shima J.E., Lagpacan L.L., Shu Y., Lapuk A., Chen Y., Komori T., Gray J.W., Chen X., et al. Organic cation transporters are determinants of oxaliplatin cytotoxicity. Cancer Res. 2006;66:8847–8857. doi: 10.1158/0008-5472.CAN-06-0769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Evers R., Piquette-Miller M., Polli J.W., Russel F.G.M., Sprowl J.A., Tohyama K., Ware J.A., De Wildt S.N., Xie W., Brouwer K.L.R., et al. Disease-Associated Changes in Drug Transporters May Impact the Pharmacokinetics and/or Toxicity of Drugs: A White Paper From the International Transporter Consortium. Clin. Pharmacol. Ther. 2018;104:900–915. doi: 10.1002/cpt.1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Hu S., Sprowl J.A. Strategies to Reduce Solute Carrier-Mediated Toxicity. Clin. Pharmacol. Ther. 2018;104:799–802. doi: 10.1002/cpt.1185. [DOI] [PubMed] [Google Scholar]
- 393.Chu X., Liao M., Shen H., Yoshida K., Zur A.A., Arya V., Galetin A., Giacomini K.M., Hanna I., Kusuhara H., et al. International Transporter Consortium, Clinical Probes and Endogenous Biomarkers as Substrates for Transporter Drug-Drug Interaction Evaluation: Perspectives From the International Transporter Consortium. Clin. Pharmacol. Ther. 2018;104:836–864. doi: 10.1002/cpt.1216. [DOI] [PubMed] [Google Scholar]
- 394.Anderson J.T., Huang K.M., Lustberg M.B., Sparreboom A., Hu S. Reviews of Physiology Biochemistry and Pharmacology. Springer; Berlin/Heidelberg, Germany: 2020. Solute carrier transportome in chemotherapy-induced adverse drug reactions. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.O’Neill J. Vaccines and Alternative Approaches: Reducing Our Dependence on Antimicrobials. The Review on Antimicrobial Resistance. The Wellcome Trust and HM Government; London, UK: 2016. [Google Scholar]
- 396.O’Neill J. Tackling Drug-Resistant Infections Globally: An. Overview of Our Work. The Review on Antimicrobial Resistance. The Wellcome Trust and HM Government; London, UK: 2016. [Google Scholar]
- 397.Piddock L., Garneau-Tsodikova S., Garner C. Ask the experts: How to curb antibiotic resistance and plug the antibiotics gap? Future Med. Chem. 2016;8:1027–1032. doi: 10.4155/fmc-2014-0032. [DOI] [PubMed] [Google Scholar]
- 398.Crofts T.S., Gasparrini A.J., Dantas G. Next-generation approaches to understand and combat the antibiotic resistome. Nat. Rev. Microbiol. 2017;15:422–434. doi: 10.1038/nrmicro.2017.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Wells V., Piddock L.J.V. Addressing antimicrobial resistance in the UK and Europe. Lancet Infect. Dis. 2017;17:1230–1231. doi: 10.1016/S1473-3099(17)30633-3. [DOI] [PubMed] [Google Scholar]
- 400.Baker S., Thomson N., Weill F.X., Holt K.E. Genomic insights into the emergence and spread of antimicrobial-resistant bacterial pathogens. Science. 2018;360:733–738. doi: 10.1126/science.aar3777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Annunziato G. Strategies to Overcome Antimicrobial Resistance (AMR) Making Use of Non-Essential Target Inhibitors: A Review. Int. J. Mol. Sci. 2019;20:5844. doi: 10.3390/ijms20235844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Dougan G., Dowson C., Overington J., Next Generation Antibiotic Discovery Symposium P. Meeting the discovery challenge of drug-resistant infections: Progress and focusing resources. Drug Discov. Today. 2019;24:452–461. doi: 10.1016/j.drudis.2018.11.015. [DOI] [PubMed] [Google Scholar]
- 403.Roope L.S.J., Smith R.D., Pouwels K.B., Buchanan J., Abel L., Eibich P., Butler C.C., Tan P.S., Walker A.S., Robotham J.V., et al. The challenge of antimicrobial resistance: What economics can contribute. Science. 2019;364:eaau4679. doi: 10.1126/science.aau4679. [DOI] [PubMed] [Google Scholar]
- 404.Turner N.A., Sharma-Kuinkel B.K., Maskarinec S.A., Eichenberger E.M., Shah P.P., Carugati M., Holland T.L., Fowler V.G. Methicillin-resistant Staphylococcus aureus: An overview of basic and clinical research. Nat. Rev. Microbiol. 2019;17:203–218. doi: 10.1038/s41579-018-0147-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Antwi A.N., Stewart A., Crosbie M. Fighting antibiotic resistance: A narrative review of public knowledge, attitudes, and perceptions of antibiotics use. Perspect. Public Health. 2020;140:338–350. doi: 10.1177/1757913920921209. [DOI] [PubMed] [Google Scholar]
- 406.Diallo O.O., Baron S.A., Abat C., Colson P., Chaudet H., Rolain J.M. Antibiotic resistance surveillance systems: A review. J. Glob. Antimicrob. Resist. 2020;23:430–438. doi: 10.1016/j.jgar.2020.10.009. [DOI] [PubMed] [Google Scholar]
- 407.Kavvas E.S., Yang L., Monk J.M., Heckmann D., Palsson B.O. A biochemically-interpretable machine learning classifier for microbial GWAS. Nat. Commun. 2020;11:2580. doi: 10.1038/s41467-020-16310-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Jit M., Ng D.H.L., Luangasanatip N., Sandmann F., Atkins K.E., Robotham J.V., Pouwels K.B. Quantifying the economic cost of antibiotic resistance and the impact of related interventions: Rapid methodological review, conceptual framework and recommendations for future studies. BMC Med. 2020;18:38. doi: 10.1186/s12916-020-1507-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Khan J., Tarar S.M., Gul I., Nawaz U., Arshad M. Challenges of antibiotic resistance biofilms and potential combating strategies: A review. 3 Biotech. 2021;11:169. doi: 10.1007/s13205-021-02707-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.De Kraker M.E.A., Stewardson A.J., Harbarth S. Will 10 Million People Die a Year due to Antimicrobial Resistance by 2050? PLoS Med. 2016;13:e1002184. doi: 10.1371/journal.pmed.1002184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Hoffman P.S. Antibacterial Discovery: 21st Century Challenges. Antibiotics. 2020;9:213. doi: 10.3390/antibiotics9050213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Lu J., Sheldenkar A., Lwin M.O. A decade of antimicrobial resistance research in social science fields: A scientometric review. Antimicrob. Resist. Infect. Control. 2020;9:178. doi: 10.1186/s13756-020-00834-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.De Kraker M.E.A., Lipsitch M. Burden of Antimicrobial Resistance: Compared to What? Epidemiol. Rev. 2021:mxab001. doi: 10.1093/epirev/mxab001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Schuts E.C., Boyd A., Muller A.E., Mouton J.W., Prins J.M. The Effect of Antibiotic Restriction Programs on Prevalence of Antimicrobial Resistance: A Systematic Review and Meta-Analysis. Open Forum Infect. Dis. 2021;8:ofab070. doi: 10.1093/ofid/ofab070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Ali J., Rafiq Q.A., Ratcliffe E. Antimicrobial resistance mechanisms and potential synthetic treatments. Future Sci. OA. 2018;4:FSO290. doi: 10.4155/fsoa-2017-0109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Blair J.M.A., Webber M.A., Baylay A.J., Ogbolu D.O., Piddock L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015;13:42–51. doi: 10.1038/nrmicro3380. [DOI] [PubMed] [Google Scholar]
- 417.Abouelhassan Y., Garrison A.T., Yang H., Chavez-Riveros A., Burch G.M., Huigens R.W., 3rd Recent Progress in Natural-Product-Inspired Programs Aimed To Address Antibiotic Resistance and Tolerance. J. Med. Chem. 2019;62:7618–7642. doi: 10.1021/acs.jmedchem.9b00370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Silver L.L. Challenges of antibacterial discovery. Clin. Microbiol. Rev. 2011;24:71–109. doi: 10.1128/CMR.00030-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.White T.A., Kell D.B. Comparative genomic assessment of novel broad-spectrum targets for antibacterial drugs. Comp. Funct. Genomics. 2004;5:304–327. doi: 10.1002/cfg.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Payne D.J., Gwynn M.N., Holmes D.J., Pompliano D.L. Drugs for bad bugs: Confronting the challenges of antibacterial discovery. Nat. Rev. Drug Discov. 2007;6:29–40. doi: 10.1038/nrd2201. [DOI] [PubMed] [Google Scholar]
- 421.Yılmaz Ç., Özcengiz G. Antibiotics: Pharmacokinetics, toxicity, resistance and multidrug efflux pumps. Biochem. Pharmacol. 2017;133:43–62. doi: 10.1016/j.bcp.2016.10.005. [DOI] [PubMed] [Google Scholar]
- 422.Ahmad I., Nawaz N., Dermani F.K., Kohlan A.K., Saidijam M., Patching S.G. Bacterial Multidrug Efflux Proteins: A Major Mechanism of Antimicrobial Resistance. Curr. Drug Targets. 2018;19:1–13. doi: 10.2174/1389450119666180426103300. [DOI] [Google Scholar]
- 423.Chopra I., Roberts M. Tetracycline antibiotics: Mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol. Biol Rev. 2001;65:232–260. doi: 10.1128/MMBR.65.2.232-260.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Phillips-Jones M.K., Harding S.E. Antimicrobial resistance (AMR) nanomachines-mechanisms for fluoroquinolone and glycopeptide recognition, efflux and/or deactivation. Biophys. Rev. 2018;10:347–362. doi: 10.1007/s12551-018-0404-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Putman M., Van Veen H.W., Konings W.N. Molecular properties of bacterial multidrug transporters. Microbiol. Mol. Biol. Rev. 2000;64:672–693. doi: 10.1128/MMBR.64.4.672-693.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Piddock L.J. Multidrug-resistance efflux pumps—Not just for resistance. Nat. Rev. Microbiol. 2006;4:629–636. doi: 10.1038/nrmicro1464. [DOI] [PubMed] [Google Scholar]
- 427.Piddock L.J.V. Clinically relevant chromosomally encoded multidrug resistance efflux pumps in bacteria. Clin. Microbiol. Rev. 2006;19:382–402. doi: 10.1128/CMR.19.2.382-402.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 428.Piddock L.J.V. The 2019 Garrod Lecture: MDR efflux in Gram-negative bacteria-how understanding resistance led to a new tool for drug discovery. J. Antimicrob. Chemother. 2019;74:3128–3134. doi: 10.1093/jac/dkz370. [DOI] [PubMed] [Google Scholar]
- 429.Nikaido H. Multidrug resistance in bacteria. Annu. Rev. Biochem. 2009;78:119–146. doi: 10.1146/annurev.biochem.78.082907.145923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Li X.Z., Plésiat P., Nikaido H. The challenge of efflux-mediated antibiotic resistance in Gram-negative bacteria. Clin. Microbiol. Rev. 2015;28:337–418. doi: 10.1128/CMR.00117-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Brown E.D., Wright G.D. Antibacterial drug discovery in the resistance era. Nature. 2016;529:336–343. doi: 10.1038/nature17042. [DOI] [PubMed] [Google Scholar]
- 432.Du D., Wang-Kan X., Neuberger A., Van Veen H.W., Pos K.M., Piddock L.J.V., Luisi B.F. Multidrug efflux pumps: Structure, function and regulation. Nat. Rev. Microbiol. 2018;16:523–539. doi: 10.1038/s41579-018-0048-6. [DOI] [PubMed] [Google Scholar]
- 433.Zwama M., Yamaguchi A. Molecular mechanisms of AcrB-mediated multidrug export. Res. Microbiol. 2018;169:372–383. doi: 10.1016/j.resmic.2018.05.005. [DOI] [PubMed] [Google Scholar]
- 434.Zgurskaya H.I., Rybenkov V.V., Krishnamoorthy G., Leus I.V. Trans-envelope multidrug efflux pumps of Gram-negative bacteria and their synergism with the outer membrane barrier. Res. Microbiol. 2018;169:351–356. doi: 10.1016/j.resmic.2018.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Zgurskaya H.I., Walker J.K., Parks J.M., Rybenkov V.V. Multidrug Efflux Pumps and the Two-Faced Janus of Substrates and Inhibitors. Acc. Chem. Res. 2021;54:930–939. doi: 10.1021/acs.accounts.0c00843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.De Oliveira D.M.P., Forde B.M., Kidd T.J., Harris P.N.A., Schembri M.A., Beatson S.A., Paterson D.L., Walker M.J. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 2020;33:e00181-19. doi: 10.1128/CMR.00181-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Zhao S., Adamiak J.W., Bonifay V., Mehla J., Zgurskaya H.I., Tan D.S. Defining new chemical space for drug penetration into Gram-negative bacteria. Nat. Chem. Biol. 2020;16:1293–1302. doi: 10.1038/s41589-020-00674-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 438.Rahman T., Yarnall B., Doyle D.A. Efflux drug transporters at the forefront of antimicrobial resistance. Eur. Biophys. J. 2017;46:647–653. doi: 10.1007/s00249-017-1238-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Perlin M.H., Andrews J., Toh S.S. Essential Letters in the Fungal Alphabet: ABC and MFS Transporters and Their Roles in Survival and Pathogenicity. Adv. Genet. 2014;85:201–253. doi: 10.1016/B978-0-12-800271-1.00004-4. [DOI] [PubMed] [Google Scholar]
- 440.Capela R., Moreira R., Lopes F. An Overview of Drug Resistance in Protozoal Diseases. Int. J. Mol. Sci. 2019;20:5748. doi: 10.3390/ijms20225748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.De Koning H.P. The Drugs of Sleeping Sickness: Their Mechanisms of Action and Resistance, and a Brief History. Trop Med. Infect. Dis. 2020;5:14. doi: 10.3390/tropicalmed5010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 442.Spengler G., Kincses A., Gajdács M., Amaral L. New roads leading to old destinations: Efflux pumps as targets to reverse multidrug resistance in bacteria. Molecules. 2017;22:468. doi: 10.3390/molecules22030468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Krishnamoorthy G., Leus I.V., Weeks J.W., Wolloscheck D., Rybenkov V.V., Zgurskaya H.I. Synergy between Active Efflux and Outer Membrane Diffusion Defines Rules of Antibiotic Permeation into Gram-Negative Bacteria. mBio. 2017;8:e01172-17. doi: 10.1128/mBio.01172-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 444.Rybenkov V.V., Zgurskaya H.I., Ganguly C., Leus I.V., Zhang Z., Moniruzzaman M. The Whole Is Bigger than the Sum of Its Parts: Drug Transport in the Context of Two Membranes with Active Efflux. Chem. Rev. 2021;121:5597–5631. doi: 10.1021/acs.chemrev.0c01137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Nikaido H., Pagès J.M. Broad-specificity efflux pumps and their role in multidrug resistance of Gram-negative bacteria. FEMS Microbiol. Rev. 2012;36:340–363. doi: 10.1111/j.1574-6976.2011.00290.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 446.Bolla J.M., Alibert-Franco S., Handzlik J., Chevalier J., Mahamoud A., Boyer G., Kieć-Kononowicz K., Pagès J.M. Strategies for bypassing the membrane barrier in multidrug resistant Gram-negative bacteria. FEBS Lett. 2011;585:1682–1690. doi: 10.1016/j.febslet.2011.04.054. [DOI] [PubMed] [Google Scholar]
- 447.Otrębska-Machaj E., Chevalier J., Handzlik J., Szymańska E., Schabikowski J., Boyer G., Bolla J.M., Kieć-Kononowicz K., Pagès J.M., Alibert S. Efflux Pump Blockers in Gram-Negative Bacteria: The New Generation of Hydantoin Based-Modulators to Improve Antibiotic Activity. Front. Microbiol. 2016;7:622. doi: 10.3389/fmicb.2016.00622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Vila J., Marti S., Sánchez-Céspedes J. Porins, efflux pumps and multidrug resistance in Acinetobacter baumannii. J. Antimicrob. Chemother. 2007;59:1210–1215. doi: 10.1093/jac/dkl509. [DOI] [PubMed] [Google Scholar]
- 449.Schweizer H.P. Understanding efflux in Gram-negative bacteria: Opportunities for drug discovery. Expert Opin. Drug Discov. 2012;7:633–642. doi: 10.1517/17460441.2012.688949. [DOI] [PubMed] [Google Scholar]
- 450.Kourtesi C., Ball A.R., Huang Y.Y., Jachak S.M., Vera D.M., Khondkar P., Gibbons S., Hamblin M.R., Tegos G.P. Microbial efflux systems and inhibitors: Approaches to drug discovery and the challenge of clinical implementation. Open Microbiol. J. 2013;7:34–52. doi: 10.2174/1874285801307010034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.Venter H., Mowla R., Ohene-Agyei T., Ma S.T. RND-type drug efflux pumps from Gram-negative bacteria: Molecular mechanism and inhibition. Front. Microbiol. 2015;6:377. doi: 10.3389/fmicb.2015.00377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 452.Alibert S., N’gompaza Diarra J., Hernandez J., Stutzmann A., Fouad M., Boyer G., Pagès J.M. Multidrug efflux pumps and their role in antibiotic and antiseptic resistance: A pharmacodynamic perspective. Expert Opin. Drug Met. Toxicol. 2017;13:301–309. doi: 10.1080/17425255.2017.1251581. [DOI] [PubMed] [Google Scholar]
- 453.Blair J.M.A., Piddock L.J.V. How to measure export via bacterial multidrug resistance efflux pumps. MBio. 2016;7:e00840-16. doi: 10.1128/mBio.00840-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Colclough A.L., Alav I., Whittle E.E., Pugh H.L., Darby E.M., Legood S.W., McNeil H.E., Blair J.M. RND efflux pumps in Gram-negative bacteria; regulation, structure and role in antibiotic resistance. Future Microbiol. 2020;15:143–157. doi: 10.2217/fmb-2019-0235. [DOI] [PubMed] [Google Scholar]
- 455.Seukep A.J., Kuete V., Nahar L., Sarker S.D., Guo M. Plant-derived secondary metabolites as the main source of efflux pump inhibitors and methods for identification. J. Pharm. Anal. 2020;10:277–290. doi: 10.1016/j.jpha.2019.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Slipski C.J., Zhanel G.G., Bay D.C. Biocide Selective TolC-Independent Efflux Pumps in Enterobacteriaceae. J. Membr. Biol. 2018;251:15–33. doi: 10.1007/s00232-017-9992-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Altinöz E., Altuner E.M. Antibiotic Resistance and Efflux Pumps. Int. J. Innov. Res. Rev. 2019;3:1–9. [Google Scholar]
- 458.Zhou Y., Joubran C., Miller-Vedam L., Isabella V., Nayar A., Tentarelli S., Miller A. Thinking outside the “bug”: A unique assay to measure intracellular drug penetration in gram-negative bacteria. Anal. Chem. 2015;87:3579–3584. doi: 10.1021/ac504880r. [DOI] [PubMed] [Google Scholar]
- 459.Krishnamoorthy G., Wolloscheck D., Weeks J.W., Croft C., Rybenkov V.V., Zgurskaya H.I. Breaking the Permeability Barrier of Escherichia coli by Controlled Hyperporination of the Outer Membrane. Antimicrob. Agents Chemother. 2016;60:7372–7381. doi: 10.1128/AAC.01882-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Coldham N.G., Webber M., Woodward M.J., Piddock L.J.V. A 96-well plate fluorescence assay for assessment of cellular permeability and active efflux in Salmonella enterica serovar Typhimurium and Escherichia coli. J. Antimicrob. Chemother. 2010;65:1655–1663. doi: 10.1093/jac/dkq169. [DOI] [PubMed] [Google Scholar]
- 461.Six D.A., Krucker T., Leeds J.A. Advances and challenges in bacterial compound accumulation assays for drug discovery. Curr. Opin. Chem. Biol. 2018;44:9–15. doi: 10.1016/j.cbpa.2018.05.005. [DOI] [PubMed] [Google Scholar]
- 462.Widya M., Pasutti W.D., Sachdeva M., Simmons R.L., Tamrakar P., Krucker T., Six D.A. Development and Optimization of a Higher-Throughput Bacterial Compound Accumulation Assay. ACS Infect. Dis. 2019;5:394–405. doi: 10.1021/acsinfecdis.8b00299. [DOI] [PubMed] [Google Scholar]
- 463.Alav I., Kobylka J., Kuth M.S., Pos K.M., Picard M., Blair J.M.A., Bavro V.N. Structure, Assembly, and Function of Tripartite Efflux and Type 1 Secretion Systems in Gram-Negative Bacteria. Chem. Rev. 2021;121:5479–5596. doi: 10.1021/acs.chemrev.1c00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Vergalli J., Bodrenko I.V., Masi M., Moynie L., Acosta-Gutiérrez S., Naismith J.H., Davin-Regli A., Ceccarelli M., Van den Berg B., Winterhalter M., et al. Porins and small-molecule translocation across the outer membrane of Gram-negative bacteria. Nat. Rev. Microbiol. 2020;18:164–176. doi: 10.1038/s41579-019-0294-2. [DOI] [PubMed] [Google Scholar]
- 465.Masi M., Winterhalter M., Pages J.M. Outer Membrane Porins. Subcell. Biochem. 2019;92:79–123. doi: 10.1007/978-3-030-18768-2_4. [DOI] [PubMed] [Google Scholar]
- 466.Masi M., Réfrégiers M., Pos K.M., Pagès J.M. Mechanisms of envelope permeability and antibiotic influx and efflux in Gram-negative bacteria. Nat. Microbiol. 2017;2:17001. doi: 10.1038/nmicrobiol.2017.1. [DOI] [PubMed] [Google Scholar]
- 467.Galocha M., Costa I.V., Teixeira M.C. Carrier-Mediated Drug Uptake in Fungal Pathogens. Genes. 2020;11:1324. doi: 10.3390/genes11111324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 468.Jantsch J., Chikkaballi D., Hensel M. Cellular aspects of immunity to intracellular Salmonella enterica. Immunol. Rev. 2011;240:185–195. doi: 10.1111/j.1600-065X.2010.00981.x. [DOI] [PubMed] [Google Scholar]
- 469.Price C.T.D., Al-Quadan T., Santic M., Rosenshine I., Abu Kwaik Y. Host proteasomal degradation generates amino acids essential for intracellular bacterial growth. Science. 2011;334:1553–1557. doi: 10.1126/science.1212868. [DOI] [PubMed] [Google Scholar]
- 470.Bravo-Santano N., Ellis J.K., Mateos L.M., Calle Y., Keun H.C., Behrends V., Letek M. Intracellular Staphylococcus aureus Modulates Host Central Carbon Metabolism To Activate Autophagy. mSphere. 2018;3:e00374-18. doi: 10.1128/mSphere.00374-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Thwaites G.E., Gant V. Are bloodstream leukocytes Trojan Horses for the metastasis of Staphylococcus aureus? Nat. Rev. Microbiol. 2011;9:215–222. doi: 10.1038/nrmicro2508. [DOI] [PubMed] [Google Scholar]
- 472.Kell D.B., Potgieter M., Pretorius E. Individuality, phenotypic differentiation, dormancy and ‘persistence’ in culturable bacterial systems: Commonalities shared by environmental, laboratory, and clinical microbiology. F1000Research. 2015;4:179. doi: 10.12688/f1000research.6709.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Kell D.B., Pretorius E. On the translocation of bacteria and their lipopolysaccharides between blood and peripheral locations in chronic, inflammatory diseases: The central roles of LPS and LPS-induced cell death. Integr. Biol. 2015;7:1339–1377. doi: 10.1039/c5ib00158g. [DOI] [PubMed] [Google Scholar]
- 474.Casadevall A. Evolution of intracellular pathogens. Annu. Rev. Microbiol. 2008;62:19–33. doi: 10.1146/annurev.micro.61.080706.093305. [DOI] [PubMed] [Google Scholar]
- 475.Silva M.T., Pestana N.T. The in vivo extracellular life of facultative intracellular bacterial parasites: Role in pathogenesis. Immunobiology. 2013;218:325–337. doi: 10.1016/j.imbio.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 476.Leon-Sicairos N., Reyes-Cortes R., Guadrón-Llanos A.M., Madueña-Molina J., Leon-Sicairos C., Canizalez-Román A. Strategies of Intracellular Pathogens for Obtaining Iron from the Environment. Biomed. Res. Int. 2015;2015:476534. doi: 10.1155/2015/476534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 477.Von Bargen K., Gorvel J.P., Salcedo S.P. Internal affairs: Investigating the Brucella intracellular lifestyle. FEMS Microbiol. Rev. 2012;36:533–562. doi: 10.1111/j.1574-6976.2012.00334.x. [DOI] [PubMed] [Google Scholar]
- 478.McClure E.E., Chavez A.S.O., Shaw D.K., Carlyon J.A., Ganta R.R., Noh S.M., Wood D.O., Bavoil P.M., Brayton K.A., Martinez J.J., et al. Engineering of obligate intracellular bacteria: Progress, challenges and paradigms. Nat. Rev. Microbiol. 2017;15:544–558. doi: 10.1038/nrmicro.2017.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Garzoni C., Kelley W.L. Staphylococcus aureus: New evidence for intracellular persistence. Trends Microbiol. 2009;17:59–65. doi: 10.1016/j.tim.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 480.Garzoni C., Kelley W.L. Return of the Trojan horse: Intracellular phenotype switching and immune evasion by Staphylococcus aureus. EMBO Mol. Med. 2011;3:115–117. doi: 10.1002/emmm.201100123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Takeuchi H., Furuta N., Morisaki I., Amano A. Exit of intracellular Porphyromonas gingivalis from gingival epithelial cells is mediated by endocytic recycling pathway. Cell Microbiol. 2011;13:677–691. doi: 10.1111/j.1462-5822.2010.01564.x. [DOI] [PubMed] [Google Scholar]
- 482.Proal A.D., Marshall T.G. Re-framing the theory of autoimmunity in the era of the microbiome: Persistent pathogens, autoantibodies, and molecular mimicry. Discov. Med. 2018;140:299–308. [PubMed] [Google Scholar]
- 483.Proal A.D., VanElzakker M.B. Pathogens Hijack Host Cell Metabolism: Intracellular Infection as a Driver of the Warburg Effect in Cancer and Other Chronic Inflammatory Conditions. Immunometabolism. 2020;3:e210003. [Google Scholar]
- 484.Hunstad D.A., Justice S.S. Intracellular lifestyles and immune evasion strategies of uropathogenic Escherichia coli. Annu. Rev. Microbiol. 2010;64:203–221. doi: 10.1146/annurev.micro.112408.134258. [DOI] [PubMed] [Google Scholar]
- 485.Potgieter M., Bester J., Kell D.B., Pretorius E. The dormant blood microbiome in chronic, inflammatory diseases. FEMS Microbiol. Rev. 2015;39:567–591. doi: 10.1093/femsre/fuv013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Fullam E., Young R.J. Physicochemical properties and Mycobacterium tuberculosis transporters: Keys to efficacious antitubercular drugs? RSC Med. Chem. 2021;12:43. doi: 10.1039/D0MD00265H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 487.Prideaux B., Via L.E., Zimmerman M.D., Eum S., Sarathy J., O’Brien P., Chen C., Kaya F., Weiner D.M., Chen P.Y., et al. The association between sterilizing activity and drug distribution into tuberculosis lesions. Nat. Med. 2015;21:1223–1227. doi: 10.1038/nm.3937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Santucci P., Greenwood D.J., Fearns A., Chen K., Jiang H., Gutierrez M.G. Intracellular localisation of Mycobacterium tuberculosis affects efficacy of the antibiotic pyrazinamide. Nat. Commun. 2021;12:3816. doi: 10.1038/s41467-021-24127-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Smith D.E., Clémençon B., Hediger M.A. Proton-coupled oligopeptide transporter family SLC15: Physiological, pharmacological and pathological implications. Mol. Aspects Med. 2013;34:323–336. doi: 10.1016/j.mam.2012.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Samsudin F., Parker J.L., Sansom M.S.P., Newstead S., Fowler P.W. Accurate Prediction of Ligand Affinities for a Proton-Dependent Oligopeptide Transporter. Cell Chem. Biol. 2016;23:299–309. doi: 10.1016/j.chembiol.2015.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 491.Richter M.F., Hergenrother P.J. The challenge of converting Gram-positive-only compounds into broad-spectrum antibiotics. Ann. N. Y. Acad. Sci. 2019;1435:18–38. doi: 10.1111/nyas.13598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 492.Parker E.N., Drown B.S., Geddes E.J., Lee H.Y., Ismail N., Lau G.W., Hergenrother P.J. Implementation of permeation rules leads to a FabI inhibitor with activity against Gram-negative pathogens. Nat. Microbiol. 2020;5:67–75. doi: 10.1038/s41564-019-0604-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 493.Perlmutter S.J., Geddes E.J., Drown B.S., Motika S.E., Lee M.R., Hergenrother P.J. Compound Uptake into E. coli Can Be Facilitated by N-Alkyl Guanidiniums and Pyridiniums. ACS Infect. Dis. 2021;7:162–173. doi: 10.1021/acsinfecdis.0c00715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 494.Muñoz K.A., Hergenrother P.J. Facilitating Compound Entry as a Means to Discover Antibiotics for Gram-Negative Bacteria. Acc. Chem. Res. 2021;54:1322–1333. doi: 10.1021/acs.accounts.0c00895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 495.Davis T.D., Gerry C.J., Tan D.S. General platform for systematic quantitative evaluation of small-molecule permeability in bacteria. ACS Chem. Biol. 2014;9:2535–2544. doi: 10.1021/cb5003015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 496.Aires J.R., Nikaido H. Aminoglycosides are captured from both periplasm and cytoplasm by the AcrD multidrug efflux transporter of Escherichia coli. J. Bacteriol. 2005;187:1923–1929. doi: 10.1128/JB.187.6.1923-1929.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.Prabhala B.K., Aduri N.G., Sharma N., Shaheen A., Sharma A., Iqbal M., Hansen P.R., Brasen C., Gajhede M., Rahman M., et al. The prototypical proton-coupled oligopeptide transporter YdgR from Escherichia coli facilitates chloramphenicol uptake into bacterial cells. J. Biol. Chem. 2018;293:1007–1017. doi: 10.1074/jbc.M117.805960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 498.Chen J.M., Uplekar S., Gordon S.V., Cole S.T. A point mutation in cycA partially contributes to the D-cycloserine resistance trait of Mycobacterium bovis BCG vaccine strains. PLoS ONE. 2012;7:e43467. doi: 10.1371/journal.pone.0043467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Chapeland-Leclerc F., Bouchoux J., Goumar A., Chastin C., Villard J., Noël T. Inactivation of the FCY2 gene encoding purine-cytosine permease promotes cross-resistance to flucytosine and fluconazole in Candida lusitaniae. Antimicrob. Agents Chemother. 2005;49:3101–3108. doi: 10.1128/AAC.49.8.3101-3108.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 500.Chen Y.N., Lo H.J., Wu C.C., Ko H.C., Chang T.P., Yang Y.L. Loss of heterozygosity of FCY2 leading to the development of flucytosine resistance in Candida tropicalis. Antimicrob. Agents Chemother. 2011;55:2506–2514. doi: 10.1128/AAC.01777-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 501.Takahata S., Ida T., Hiraishi T., Sakakibara S., Maebashi K., Terada S., Muratani T., Matsumoto T., Nakahama C., Tomono K. Molecular mechanisms of fosfomycin resistance in clinical isolates of Escherichia coli. Int. J. Antimicrob. Agents. 2010;35:333–337. doi: 10.1016/j.ijantimicag.2009.11.011. [DOI] [PubMed] [Google Scholar]
- 502.Ballestero-Téllez M., Docobo-Pérez F., Portillo-Calderón I., Rodríguez-Martínez J.M., Racero L., Ramos-Guelfo M.S., Blázquez J., Rodríguez-Baño J., Pascual A. Molecular insights into fosfomycin resistance in Escherichia coli. J. Antimicrob. Chemother. 2017;72:1303–1309. doi: 10.1093/jac/dkw573. [DOI] [PubMed] [Google Scholar]
- 503.Mistry A., Warren M.S., Cusick J.K., Karkhoff-Schweizer R.R., Lomovskaya O., Schweizer H.P. High-level pacidamycin resistance in Pseudomonas aeruginosa is mediated by an opp oligopeptide permease encoded by the opp-fabI operon. Antimicrob. Agents Chemother. 2013;57:5565–5571. doi: 10.1128/AAC.01198-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 504.Pletzer D., Braun Y., Dubiley S., Lafon C., Kohler T., Page M.G., Mourez M., Severinov K., Weingart H. The Pseudomonas aeruginosa PA14 ABC Transporter NppA1A2BCD Is Required for Uptake of Peptidyl Nucleoside Antibiotics. J. Bacteriol. 2015;197:2217–2228. doi: 10.1128/JB.00234-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 505.De Koning H.P. Uptake of pentamidine in Trypanosoma brucei brucei is mediated by three distinct transporters: Implications for cross-resistance with arsenicals. Mol. Pharmacol. 2001;59:586–592. doi: 10.1124/mol.59.3.586. [DOI] [PubMed] [Google Scholar]
- 506.Tindall S.M., Vallières C., Lakhani D.H., Islahudin F., Ting K.N., Avery S.V. Heterologous Expression of a Novel Drug Transporter from the Malaria Parasite Alters Resistance to Quinoline Antimalarials. Sci. Rep. 2018;8:2464. doi: 10.1038/s41598-018-20816-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 507.Chopra I. Molecular mechanisms involved in the transport of antibiotics into bacteria. Parasitology. 1988;96:S25–S44. doi: 10.1017/S0031182000085966. [DOI] [PubMed] [Google Scholar]
- 508.Chopra I. Penetration of antibiotics to their target sites. J. Antimicrob. Chemother. 1990;26:607–609. doi: 10.1093/jac/26.5.607-a. [DOI] [PubMed] [Google Scholar]
- 509.Delespaux V., De Koning H.P. Transporters in antiparasitic drug development and resistance. In: Flohe L., Koch O., Jäger T., editors. Antiparasitic and Antibacterial Drug Discovery: Trypanosomatidae. Wiley-Blackwell; London, UK: 2013. pp. 335–349. [Google Scholar]
- 510.McMurry L., Levy S.B. Two transport systems for tetracycline in sensitive Escherichia coli: Critical role for an initial rapid uptake system insensitive to energy inhibitors. Antimicrob. Agents Chemother. 1978;14:201–209. doi: 10.1128/AAC.14.2.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Smith M.C., Chopra I. Energetics of tetracycline transport into Escherichia coli. Antimicrob. Agents Chemother. 1984;25:446–449. doi: 10.1128/AAC.25.4.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 512.Hutson M. The language machines. Nature. 2021;591:22–25. doi: 10.1038/d41586-021-00530-0. [DOI] [PubMed] [Google Scholar]
- 513.Shrivastava A.D., Swainston N., Samanta S., Roberts I., Wright Muelas M., Kell D.B. MassGenie: A transformer-based deep learning method for identifying small molecules from their mass spectra. bioRxiv. 2021 doi: 10.1101/2021.06.25.449969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 514.Vermaas J.V., Trebesch N., Mayne C.G., Thangapandian S., Shekhar M., Mahinthichaichan P., Baylon J.L., Jiang T., Wang Y., Muller M.P., et al. Microscopic Characterization of Membrane Transporter Function by In Silico Modeling and Simulation. Methods Enzymol. 2016;578:373–428. doi: 10.1016/bs.mie.2016.05.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Jia R., Martens C., Shekhar M., Pant S., Pellowe G.A., Lau A.M., Findlay H.E., Harris N.J., Tajkhorshid E., Booth P.J., et al. Hydrogen-deuterium exchange mass spectrometry captures distinct dynamics upon substrate and inhibitor binding to a transporter. Nat. Commun. 2020;11:6162. doi: 10.1038/s41467-020-20032-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 516.Vermaas J.V., Rempe S.B., Tajkhorshid E. Electrostatic lock in the transport cycle of the multidrug resistance transporter EmrE. Proc. Natl. Acad. Sci. USA. 2018;115:E7502–E7511. doi: 10.1073/pnas.1722399115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 517.Padariya M., Kalathiya U., Baginski M. Structural and dynamic changes adopted by EmrE, multidrug transporter protein--Studies by molecular dynamics simulation. Biochim. Biophys. Acta. 2015;1848:2065–2074. doi: 10.1016/j.bbamem.2015.05.014. [DOI] [PubMed] [Google Scholar]
- 518.Padariya M., Kalathiya U., Baginski M. Structural and dynamic insights on the EmrE protein with TPP+ and related substrates through molecular dynamics simulations. Chem. Phys. Lipids. 2018;212:1–11. doi: 10.1016/j.chemphyslip.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 519.Li J., Zhao Z., Tajkhorshid E. Locking Two Rigid-body Bundles in an Outward-Facing Conformation: The Ion-coupling Mechanism in a LeuT-fold Transporter. Sci. Rep. 2019;9:19479. doi: 10.1038/s41598-019-55722-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 520.Zuo Z., Weng J., Wang W. Insights into the Inhibitory Mechanism of D13-9001 to the Multidrug Transporter AcrB through Molecular Dynamics Simulations. J. Phys. Chem. B. 2016;120:2145–2154. doi: 10.1021/acs.jpcb.5b11942. [DOI] [PubMed] [Google Scholar]
- 521.Jamshidi S., Sutton J.M., Rahman K.M. Mapping the Dynamic Functions and Structural Features of AcrB Efflux Pump Transporter Using Accelerated Molecular Dynamics Simulations. Sci. Rep. 2018;8:10470. doi: 10.1038/s41598-018-28531-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 522.Johnson R.M., Fais C., Parmar M., Cheruvara H., Marshall R.L., Hesketh S.J., Feasey M.C., Ruggerone P., Vargiu A.V., Postis V.L.G., et al. Cryo-EM Structure and Molecular Dynamics Analysis of the Fluoroquinolone Resistant Mutant of the AcrB Transporter from Salmonella. Microorganisms. 2020;8:943. doi: 10.3390/microorganisms8060943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Pan C., Weng J., Wang W. Conformational Dynamics and Protein-Substrate Interaction of ABC Transporter BtuCD at the Occluded State Revealed by Molecular Dynamics Simulations. Biochemistry. 2016;55:6897–6907. doi: 10.1021/acs.biochem.6b00386. [DOI] [PubMed] [Google Scholar]
- 524.Hsu W.L., Furuta T., Sakurai M. Analysis of the Free Energy Landscapes for the Opening-Closing Dynamics of the Maltose Transporter ATPase MalK2 Using Enhanced-Sampling Molecular Dynamics Simulation. J. Phys. Chem. B. 2015;119:9717–9725. doi: 10.1021/acs.jpcb.5b05432. [DOI] [PubMed] [Google Scholar]
- 525.Gu R.X., Corradi V., Singh G., Choudhury H.G., Beis K., Tieleman D.P. Conformational Changes of the Antibacterial Peptide ATP Binding Cassette Transporter McjD Revealed by Molecular Dynamics Simulations. Biochemistry. 2015;54:5989–5998. doi: 10.1021/acs.biochem.5b00753. [DOI] [PubMed] [Google Scholar]
- 526.Immadisetty K., Hettige J., Moradi M. What Can and Cannot Be Learned from Molecular Dynamics Simulations of Bacterial Proton-Coupled Oligopeptide Transporter GkPOT? J. Phys. Chem. B. 2017;121:3644–3656. doi: 10.1021/acs.jpcb.6b09733. [DOI] [PubMed] [Google Scholar]
- 527.Cáceres-Delpiano J., Teneb J., Mansilla R., Garcia A., Salas-Burgos A. Variations in periplasmic loop interactions determine the pH-dependent activity of the hexameric urea transporter UreI from Helicobacter pylori: A molecular dynamics study. BMC Struct. Biol. 2015;15:11. doi: 10.1186/s12900-015-0038-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Heinzelmann G., Kuyucak S. Molecular dynamics simulations elucidate the mechanism of proton transport in the glutamate transporter EAAT3. Biophys. J. 2014;106:2675–2683. doi: 10.1016/j.bpj.2014.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 529.Park M.S. Molecular Dynamics Simulations of the Human Glucose Transporter GLUT1. PLoS ONE. 2015;10:e0125361. doi: 10.1371/journal.pone.0125361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 530.Reithmeier R.A.F., Casey J.R., Kalli A.C., Sansom M.S.P., Alguel Y., Iwata S. Band 3, the human red cell chloride/bicarbonate anion exchanger (AE1, SLC4A1), in a structural context. Biochim. Biophys. Acta. 2016;1858:1507–1532. doi: 10.1016/j.bbamem.2016.03.030. [DOI] [PubMed] [Google Scholar]
- 531.Zhang Y., Zheng G., Fu T., Hong J., Li F., Yao X., Xue W., Zhu F. The binding mode of vilazodone in the human serotonin transporter elucidated by ligand docking and molecular dynamics simulations. Phys. Chem. Chem. Phys. 2020;22:5132–5144. doi: 10.1039/C9CP05764A. [DOI] [PubMed] [Google Scholar]
- 532.Mikou A., Cabaye A., Goupil A., Bertrand H.O., Mothet J.P., Acher F.C. Asc-1 Transporter (SLC7A10): Homology Models And Molecular Dynamics Insights Into The First Steps Of The Transport Mechanism. Sci. Rep. 2020;10:3731. doi: 10.1038/s41598-020-60617-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Briones R., Aponte-Santamaria C., De Groot B.L. Localization and Ordering of Lipids Around Aquaporin-0: Protein and Lipid Mobility Effects. Front. Physiol. 2017;8:124. doi: 10.3389/fphys.2017.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 534.Saboe P.O., Rapisarda C., Kaptan S., Hsiao Y.S., Summers S.R., De Zorzi R., Dukovski D., Yu J., De Groot B.L., Kumar M., et al. Role of Pore-Lining Residues in Defining the Rate of Water Conduction by Aquaporin-0. Biophys. J. 2017;112:953–965. doi: 10.1016/j.bpj.2017.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 535.De Maré S.W., Venskutonyte R., Eltschkner S., De Groot B.L., Lindkvist-Petersson K. Structural Basis for Glycerol Efflux and Selectivity of Human Aquaporin 7. Structure. 2020;28:215–222.e3. doi: 10.1016/j.str.2019.11.011. [DOI] [PubMed] [Google Scholar]
- 536.Moss F.J., Mahinthichaichan P., Lodowski D.T., Kowatz T., Tajkhorshid E., Engel A., Boron W.F., Vahedi-Faridi A. Aquaporin-7: A Dynamic Aquaglyceroporin With Greater Water and Glycerol Permeability Than Its Bacterial Homolog GlpF. Front. Physiol. 2020;11:728. doi: 10.3389/fphys.2020.00728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 537.Casiraghi A., Bensimon A., Superti-Furga G. Recent developments in ligands and chemical probes targeting solute carrier transporters. Curr. Opin. Chem. Biol. 2021;62:53–63. doi: 10.1016/j.cbpa.2021.01.012. [DOI] [PubMed] [Google Scholar]
- 538.Betters J.L., Yu L. Transporters as drug targets: Discovery and development of NPC1L1 inhibitors. Clin. Pharmacol. Ther. 2010;87:117–121. doi: 10.1038/clpt.2009.209. [DOI] [PubMed] [Google Scholar]
- 539.Ecker G., Chiba P. Transporters as Drug Carriers: Structure, Function, Substrates. Wiley/VCH; Weinheim, Germany: 2009. [Google Scholar]
- 540.Ecker G.F., Clausen R.P., Sitte H.H. Transporters as Drug Targets. Wiley; New York, NY, USA: 2017. [Google Scholar]
- 541.Lin L., Yee S.W., Kim R.B., Giacomini K.M. SLC transporters as therapeutic targets: Emerging opportunities. Nat. Rev. Drug Discov. 2015;14:543–560. doi: 10.1038/nrd4626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 542.Qosa H., Mohamed L.A., Alqahtani S., Abuasal B.S., Hill R.A., Kaddoumi A. Transporters as Drug Targets in Neurological Diseases. Clin. Pharmacol. Ther. 2016;100:441–453. doi: 10.1002/cpt.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 543.César-Razquin A., Girardi E., Yang M., Brehme M., Saez-Rodriguez J., Superti-Furga G. In silico prioritization of transporter-drug relationships from drug sensitivity screens. Front. Pharmacol. 2018;9:1011. doi: 10.3389/fphar.2018.01011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 544.Garibsingh R.A., Schlessinger A. Advances and Challenges in Rational Drug Design for SLCs. Trends Pharmacol. Sci. 2019;40:790–800. doi: 10.1016/j.tips.2019.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 545.Scalise M., Console L., Galluccio M., Pochini L., Indiveri C. Chemical Targeting of Membrane Transporters: Insights into Structure/Function Relationships. ACS Omega. 2020;5:2069–2080. doi: 10.1021/acsomega.9b04078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 546.Nakanishi T., Tamai I. Solute carrier transporters as targets for drug delivery and pharmacological intervention for chemotherapy. J. Pharm. Sci. 2011;100:3731–3750. doi: 10.1002/jps.22576. [DOI] [PubMed] [Google Scholar]
- 547.Schumann T., Konig J., Henke C., Willmes D.M., Bornstein S.R., Jordan J., Fromm M.F., Birkenfeld A.L. Solute Carrier Transporters as Potential Targets for the Treatment of Metabolic Disease. Pharmacol. Rev. 2020;72:343–379. doi: 10.1124/pr.118.015735. [DOI] [PubMed] [Google Scholar]
- 548.Zhang Y., Zhang Y., Sun K., Meng Z., Chen L. The SLC transporter in nutrient and metabolic sensing, regulation, and drug development. J. Mol. Cell Biol. 2019;11:1–13. doi: 10.1093/jmcb/mjy052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 549.Li M., Zhang S., Yang B. Urea Transporters Identified as Novel Diuretic Drug Targets. Curr. Drug Targets. 2020;21:279–287. doi: 10.2174/1389450120666191129101915. [DOI] [PubMed] [Google Scholar]
- 550.Zeden M.S., Burke Ó., Vallely M., Fingleton C., O’Gara J.P. Exploring amino acid and peptide transporters as therapeutic targets to attenuate virulence and antibiotic resistance in Staphylococcus aureus. PLoS Pathog. 2021;17:e1009093. doi: 10.1371/journal.ppat.1009093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 551.Rochette L., Meloux A., Zeller M., Malka G., Cottin Y., Vergely C. Mitochondrial SLC25 Carriers: Novel Targets for Cancer Therapy. Molecules. 2020;25:2417. doi: 10.3390/molecules25102417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 552.Wang W.W., Gallo L., Jadhav A., Hawkins R., Parker C.G. The Druggability of Solute Carriers. J. Med. Chem. 2020;63:3834–3867. doi: 10.1021/acs.jmedchem.9b01237. [DOI] [PubMed] [Google Scholar]
- 553.Ceska T., Chung C.W., Cooke R., Phillips C., Williams P.A. Cryo-EM in drug discovery. Biochem. Soc. Trans. 2019;47:281–293. doi: 10.1042/BST20180267. [DOI] [PubMed] [Google Scholar]
- 554.Bajorath J. Molecular Similarity Concepts for Informatics Applications. Methods Mol. Biol. 2017;1526:231–245. doi: 10.1007/978-1-4939-6613-4_13. [DOI] [PubMed] [Google Scholar]
- 555.Bender A., Glen R.C. Molecular similarity: A key technique in molecular informatics. Org. Biomol. Chem. 2004;2:3204–3218. doi: 10.1039/b409813g. [DOI] [PubMed] [Google Scholar]
- 556.Bender A., Jenkins J.L., Li Q.L., Adams S.E., Cannon E.O., Glen R.C. Molecular Similarity: Advances in Methods, Applications and Validations in Virtual Screening and QSAR. Annu. Rep. Comput. Chem. 2006;2:141–168. doi: 10.1016/S1574-1400(06)02009-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 557.Eckert H., Bajorath J. Molecular similarity analysis in virtual screening: Foundations, limitations and novel approaches. Drug Discov. Today. 2007;12:225–233. doi: 10.1016/j.drudis.2007.01.011. [DOI] [PubMed] [Google Scholar]
- 558.Floris M., Olla S. Molecular Similarity in Computational Toxicology. Methods Mol. Biol. 2018;1800:171–179. doi: 10.1007/978-1-4939-7899-1_7. [DOI] [PubMed] [Google Scholar]
- 559.Ginn C.M.R., Willett P., Bradshaw J. Virtual Screening: An Alternative or Complement to High Throughput Screening? Volume 20. Springer; Dordrecht, The Netherlands: 2000. Combination of molecular similarity measures using data fusion; pp. 1–16. [Google Scholar]
- 560.Johnson M.A., Maggiora G.M. Concepts and Applications of Molecular Similarity. Wiley; New York, NY, USA: 1990. [Google Scholar]
- 561.Maggiora G.M., Shanmugasundaram V. Molecular Similarity Measures. Methods Mol. Biol. 2011;672:39–100. doi: 10.1007/978-1-60761-839-3_2. [DOI] [PubMed] [Google Scholar]
- 562.Medina-Franco J.L., Maggiora G.M. Molecular similarity analysis. In: Bajorath J., editor. Chemoinformatics for Drug Discovery. Wiley; Hoboken, NJ, USA: 2014. pp. 343–399. [Google Scholar]
- 563.Zahoránszky-Kőhalmi G., Bologa C.G., Oprea T.I. Impact of similarity threshold on the topology of molecular similarity networks and clustering outcomes. J. Cheminfor. 2016;8:16. doi: 10.1186/s13321-016-0127-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 564.Gasteiger J. Handbook of Chemoinformatics: From Data to Knowledge. Wiley/VCH; Weinheim, Germany: 2003. [Google Scholar]
- 565.O’Hagan S., Swainston N., Handl J., Kell D.B. A ‘rule of 0.5′ for the metabolite-likeness of approved pharmaceutical drugs. Metabolomics. 2015;11:323–339. doi: 10.1007/s11306-014-0733-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.O’Hagan S., Kell D.B. Understanding the foundations of the structural similarities between marketed drugs and endogenous human metabolites. Front. Pharmacol. 2015;6:105. doi: 10.3389/fphar.2015.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 567.O’Hagan S., Kell D.B. MetMaxStruct: A Tversky-similarity-based strategy for analysing the (sub)structural similarities of drugs and endogenous metabolites. Front. Pharmacol. 2016;7:266. doi: 10.3389/fphar.2016.00266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 568.O’Hagan S., Kell D.B. Analysis of drug-endogenous human metabolite similarities in terms of their maximum common substructures. J. Cheminformatics. 2017;9:18. doi: 10.1186/s13321-017-0198-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 569.Thiele I., Swainston N., Fleming R.M.T., Hoppe A., Sahoo S., Aurich M.K., Haraldsdottír H., Mo M.L., Rolfsson O., Stobbe M.D., et al. A community-driven global reconstruction of human metabolism. Nat. Biotechnol. 2013;31:419–425. doi: 10.1038/nbt.2488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 570.Wishart D.S., Feunang Y.D., Marcu A., Guo A.C., Liang K., Vazquez-Fresno R., Sajed T., Johnson D., Li C., Karu N., et al. HMDB 4.0: The human metabolome database for 2018. Nucleic Acids Res. 2018;46:D608–D617. doi: 10.1093/nar/gkx1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 571.Rosen J., Gottfries J., Muresan S., Backlund A., Oprea T.I. Novel chemical space exploration via natural products. J. Med. Chem. 2009;52:1953–1962. doi: 10.1021/jm801514w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 572.Butler M.S., Robertson A.A.B., Cooper M.A. Natural product and natural product derived drugs in clinical trials. Nat. Prod. Rep. 2014;31:1612–1661. doi: 10.1039/C4NP00064A. [DOI] [PubMed] [Google Scholar]
- 573.Doak B.C., Over B., Giordanetto F., Kihlberg J. Oral druggable space beyond the rule of 5: Insights from drugs and clinical candidates. Chem. Biol. 2014;21:1115–1142. doi: 10.1016/j.chembiol.2014.08.013. [DOI] [PubMed] [Google Scholar]
- 574.Doak B.C., Kihlberg J. Drug discovery beyond the rule of 5—Opportunities and challenges. Expert Opin. Drug Discov. 2017;12:115–119. doi: 10.1080/17460441.2017.1264385. [DOI] [PubMed] [Google Scholar]
- 575.Harvey A.L. Natural products in drug discovery. Drug Discov. Today. 2008;13:894–901. doi: 10.1016/j.drudis.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 576.Ganesan A. The impact of natural products upon modern drug discovery. Curr. Opin. Chem. Biol. 2008;12:306–317. doi: 10.1016/j.cbpa.2008.03.016. [DOI] [PubMed] [Google Scholar]
- 577.Cragg G.M., Newman D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta. 2013;1830:3670–3695. doi: 10.1016/j.bbagen.2013.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 578.Newman D.J., Cragg G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020;83:770–803. doi: 10.1021/acs.jnatprod.9b01285. [DOI] [PubMed] [Google Scholar]
- 579.Dias D.A., Urban S., Roessner U. A historical overview of natural products in drug discovery. Metabolites. 2012;2:303–336. doi: 10.3390/metabo2020303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 580.Ji H.F., Li X.J., Zhang H.Y. Natural products and drug discovery Can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia? EMBO Rep. 2009;10:194–200. doi: 10.1038/embor.2009.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 581.Lahlou M. The Success of Natural Products in Drug Discovery. Pharmacol. Pharm. 2013;4:17–31. doi: 10.4236/pp.2013.43A003. [DOI] [Google Scholar]
- 582.Luo F., Gu J., Chen L., Xu X. Systems pharmacology strategies for anticancer drug discovery based on natural products. Mol. Biosyst. 2014;10:1912–1917. doi: 10.1039/c4mb00105b. [DOI] [PubMed] [Google Scholar]
- 583.Silva T., Reis J., Teixeira J., Borges F. Alzheimer’s disease, enzyme targets and drug discovery struggles: From natural products to drug prototypes. Ageing Res. Rev. 2014;15:116–145. doi: 10.1016/j.arr.2014.03.008. [DOI] [PubMed] [Google Scholar]
- 584.Wright G.D. Something old, something new: Revisiting natural products in antibiotic drug discovery. Can. J. Microbiol. 2014;60:147–154. doi: 10.1139/cjm-2014-0063. [DOI] [PubMed] [Google Scholar]
- 585.Wright G.D. Unlocking the potential of natural products in drug discovery. Microb. Biotechnol. 2019;12:55–57. doi: 10.1111/1751-7915.13351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 586.Zheng C.L., Wang J.A., Liu J.L., Pei M.J., Huang C., Wang Y.H. System-level multi-target drug discovery from natural products with applications to cardiovascular diseases. Mol. Divers. 2014;18:621–635. doi: 10.1007/s11030-014-9521-y. [DOI] [PubMed] [Google Scholar]
- 587.Camp D., Garavelas A., Campitelli M. Analysis of Physicochemical Properties for Drugs of Natural Origin. J. Nat. Prod. 2015;78:1370–1382. doi: 10.1021/acs.jnatprod.5b00255. [DOI] [PubMed] [Google Scholar]
- 588.Lipinski C.A. Rule of five in 2015 and beyond: Target and ligand structural limitations, ligand chemistry structure and drug discovery project decisions. Adv. Drug Deliv. Rev. 2016;101:34–41. doi: 10.1016/j.addr.2016.04.029. [DOI] [PubMed] [Google Scholar]
- 589.Walters W.P. Going further than Lipinski’s rule in drug design. Exp. Opin. Drug Discov. 2012;7:99–107. doi: 10.1517/17460441.2012.648612. [DOI] [PubMed] [Google Scholar]
- 590.Zhang M.Q., Wilkinson B. Drug discovery beyond the ‘rule-of-five’. Curr. Opin. Biotechnol. 2007;18:478–488. doi: 10.1016/j.copbio.2007.10.005. [DOI] [PubMed] [Google Scholar]
- 591.Lipinski C.A., Lombardo F., Dominy B.W., Feeney P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997;23:3–25. doi: 10.1016/S0169-409X(96)00423-1. [DOI] [PubMed] [Google Scholar]
- 592.Giacomini K.M., Huang S.M., Tweedie D.J., Benet L.Z., Brouwer K.L., Chu X., Dahlin A., Evers R., Fischer V., Hillgren K.M., et al. Membrane transporters in drug development. Nat. Rev. Drug Discov. 2010;9:215–236. doi: 10.1038/nrd3028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 593.Giacomini K.M., Galetin A., Huang S.M. The International Transporter Consortium: Summarizing Advances in the Role of Transporters in Drug Development. Clin. Pharmacol. Ther. 2018;104:766–771. doi: 10.1002/cpt.1224. [DOI] [PubMed] [Google Scholar]
- 594.Müller J., Keiser M., Drozdzik M., Oswald S. Expression, regulation and function of intestinal drug transporters: An update. Biol. Chem. 2017;398:175–192. doi: 10.1515/hsz-2016-0259. [DOI] [PubMed] [Google Scholar]
- 595.Sai Y., Tsuji A. Transporter-mediated drug delivery: Recent progress and experimental approaches. Drug Discov. Today. 2004;9:712–720. doi: 10.1016/S1359-6446(04)03198-8. [DOI] [PubMed] [Google Scholar]
- 596.Liu X. Transporter-Mediated Drug-Drug Interactions and Their Significance. Adv. Exp. Med. Biol. 2019;1141:241–291. doi: 10.1007/978-981-13-7647-4_5. [DOI] [PubMed] [Google Scholar]
- 597.Saunders N.R., Dziegielewska K.M., Møllgård K., Habgood M.D. Recent Developments in Understanding Barrier Mechanisms in the Developing Brain: Drugs and Drug Transporters in Pregnancy, Susceptibility or Protection in the Fetal Brain? Annu. Rev. Pharmacol. Toxicol. 2019;59:487–505. doi: 10.1146/annurev-pharmtox-010818-021430. [DOI] [PubMed] [Google Scholar]
- 598.Darbani B., Kell D.B., Borodina I. Energetic evolution of cellular transportomes. BMC Genom. 2018;19:418. doi: 10.1186/s12864-018-4816-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 599.König J., Müller F., Fromm M.F. Transporters and drug-drug interactions: Important determinants of drug disposition and effects. Pharmacol. Rev. 2013;65:944–966. doi: 10.1124/pr.113.007518. [DOI] [PubMed] [Google Scholar]
- 600.Franke R.M., Gardner E.R., Sparreboom A. Pharmacogenetics of drug transporters. Curr. Pharm. Des. 2010;16:220–230. doi: 10.2174/138161210790112683. [DOI] [PubMed] [Google Scholar]
- 601.Harwood M.D., Neuhoff S., Carlson G.L., Warhurst G., Rostami-Hodjegan A. Absolute abundance and function of intestinal drug transporters: A prerequisite for fully mechanistic in vitro-in vivo extrapolation of oral drug absorption. Biopharm. Drug Dispos. 2013;34:2–28. doi: 10.1002/bdd.1810. [DOI] [PubMed] [Google Scholar]
- 602.Ishikawa T., Tsuji A., Inui K., Sai Y., Anzai N., Wada M., Endou H., Sumino Y. The genetic polymorphism of drug transporters: Functional analysis approaches. Pharmacogenomics. 2004;5:67–99. doi: 10.1517/phgs.5.1.67.25683. [DOI] [PubMed] [Google Scholar]
- 603.Ivanyuk A., Livio F., Biollaz J., Buclin T. Renal Drug Transporters and Drug Interactions. Clin. Pharmacokinet. 2017;56:825–892. doi: 10.1007/s40262-017-0506-8. [DOI] [PubMed] [Google Scholar]
- 604.Lai Y., Sampson K.E., Stevens J.C. Evaluation of drug transporter interactions in drug discovery and development. Comb. Chem. High Throughput Screen. 2010;13:112–134. doi: 10.2174/138620710790596772. [DOI] [PubMed] [Google Scholar]
- 605.Lai Y., Hsiao P. Beyond the ITC White Paper: Emerging sciences in drug transporters and opportunities for drug development. Curr. Pharm. Des. 2014;20:1577–1594. doi: 10.2174/13816128113199990467. [DOI] [PubMed] [Google Scholar]
- 606.Lee W., Ha J.M., Sugiyama Y. Post-translational regulation of the major drug transporters in the families of organic anion transporters and organic anion-transporting polypeptides. J. Biol. Chem. 2020;295:17349–17364. doi: 10.1074/jbc.REV120.009132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 607.Neul C., Schaeffeler E., Sparreboom A., Laufer S., Schwab M., Nies A.T. Impact of Membrane Drug Transporters on Resistance to Small-Molecule Tyrosine Kinase Inhibitors. Trends Pharmacol. Sci. 2016;37:904–932. doi: 10.1016/j.tips.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 608.Nigam S.K. What do drug transporters really do? Nat. Rev. Drug Discov. 2015;14:29–44. doi: 10.1038/nrd4461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 609.Petzinger E., Geyer J. Drug transporters in pharmacokinetics. Naunyn Schmiedebergs Arch. Pharmacol. 2006;372:465–475. doi: 10.1007/s00210-006-0042-9. [DOI] [PubMed] [Google Scholar]
- 610.Rodrigues A.D., Taskar K.S., Kusuhara H., Sugiyama Y. Endogenous Probes for Drug Transporters: Balancing Vision With Reality. Clin. Pharmacol. Ther. 2018;103:434–448. doi: 10.1002/cpt.749. [DOI] [PubMed] [Google Scholar]
- 611.Thwaites D.T., Anderson C.M. H+-coupled nutrient, micronutrient and drug transporters in the mammalian small intestine. Exp. Physiol. 2007;92:603–619. doi: 10.1113/expphysiol.2005.029959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 612.Unadkat J.D., Dahlin A., Vijay S. Placental drug transporters. Curr. Drug Metab. 2004;5:125–131. doi: 10.2174/1389200043489171. [DOI] [PubMed] [Google Scholar]
- 613.Zhang L., Strong J.M., Qiu W., Lesko L.J., Huang S.M. Scientific perspectives on drug transporters and their role in drug interactionst. Mol. Pharm. 2006;3:62–69. doi: 10.1021/mp050095h. [DOI] [PubMed] [Google Scholar]
- 614.Vora B., Green E.A.E., Khuri N., Ballgren F., Sirota M., Giacomini K.M. Drug-nutrient interactions: Discovering prescription drug inhibitors of the thiamine transporter ThTR-2 (SLC19A3) Am. J. Clin. Nutr. 2020;111:110–121. doi: 10.1093/ajcn/nqz255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 615.Terada T., Inui K. Gene expression and regulation of drug transporters in the intestine and kidney. Biochem. Pharmacol. 2007;73:440–449. doi: 10.1016/j.bcp.2006.10.010. [DOI] [PubMed] [Google Scholar]
- 616.Sugiyama Y., Steffansen B. Transporters in Drug Development: Discovery, Optimization, Clinical Study and Regulation. AAPS/Springer; New York, NY, USA: 2013. [Google Scholar]
- 617.Koepsell H. Organic Cation Transporters in Health and Disease. Pharmacol. Rev. 2020;72:253–319. doi: 10.1124/pr.118.015578. [DOI] [PubMed] [Google Scholar]
- 618.Schlessinger A., Welch M.A., Van Vlijmen H., Korzekwa K., Swaan P.W., Matsson P. Molecular Modeling of Drug-Transporter Interactions-An International Transporter Consortium Perspective. Clin. Pharmacol. Ther. 2018;104:818–835. doi: 10.1002/cpt.1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 619.Zamek-Gliszczynski M.J., Giacomini K.M., Zhang L. Emerging Clinical Importance of Hepatic Organic Cation Transporter 1 (OCT1) in Drug Pharmacokinetics, Dynamics, Pharmacogenetic Variability, and Drug Interactions. Clin. Pharmacol. Ther. 2018;103:758–760. doi: 10.1002/cpt.941. [DOI] [PubMed] [Google Scholar]
- 620.Zamek-Gliszczynski M.J., Taub M.E., Chothe P.P., Chu X., Giacomini K.M., Kim R.B., Ray A.S., Stocker S.L., Unadkat J.D., Wittwer M.B., et al. International Transporter, C., Transporters in Drug Development: 2018 ITC Recommendations for Transporters of Emerging Clinical Importance. Clin. Pharmacol. Ther. 2018;104:890–899. doi: 10.1002/cpt.1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 621.Julsing M.K., Schrewe M., Cornelissen S., Hermann I., Schmid A., Bühler B. Outer membrane protein AlkL boosts biocatalytic oxyfunctionalization of hydrophobic substrates in Escherichia coli. Appl. Environ. Microbiol. 2012;78:5724–5733. doi: 10.1128/AEM.00949-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 622.Call T.P., Akhtar M.K., Baganz F., Grant C. Modulating the import of medium-chain alkanes in E. coli through tuned expression of FadL. J. Biol. Eng. 2016;10:5. doi: 10.1186/s13036-016-0026-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 623.Cornelissen S., Julsing M.K., Volmer J., Riechert O., Schmid A., Bühler B. Whole-cell-based CYP153A6-catalyzed (S)-limonene hydroxylation efficiency depends on host background and profits from monoterpene uptake via AlkL. Biotechnol. Bioeng. 2013;110:1282–1292. doi: 10.1002/bit.24801. [DOI] [PubMed] [Google Scholar]
- 624.Ciarimboli G., Gautron S., Schlatter E. Organic Cation Transporters: Integration of Physiology, Pathology and Pharmacology. Springer; Berilin/Heidelberg, Germany: 2016. [Google Scholar]
- 625.Beck J.G., Chatterjee J., Laufer B., Kiran M.U., Frank A.O., Neubauer S., Ovadia O., Greenberg S., Gilon C., Hoffman A., et al. Intestinal Permeability of Cyclic Peptides: Common Key Backbone Motifs Identified. J. Am. Chem. Soc. 2012;134:12125–12133. doi: 10.1021/ja303200d. [DOI] [PubMed] [Google Scholar]
- 626.Corti G., Maestrelli F., Cirri M., Zerrouk N., Mura P. Development and evaluation of an in vitro method for prediction of human drug absorption—II. Demonstration of the method suitability. Eur. J. Pharm. Sci. 2006;27:354–362. doi: 10.1016/j.ejps.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 627.Skolnik S., Lin X., Wang J., Chen X.H., He T., Zhang B. Towards prediction of in vivo intestinal absorption using a 96-well Caco-2 assay. J. Pharm. Sci. 2010;99:3246–3265. doi: 10.1002/jps.22080. [DOI] [PubMed] [Google Scholar]
- 628.Kell D.B., Samanta S., Swainston N. Deep learning and generative methods in cheminformatics and chemical biology: Navigating small molecule space intelligently Biochem. J. 2020;477:4559–4580. doi: 10.1042/BCJ20200781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 629.LeCun Y., Bengio Y., Hinton G. Deep learning. Nature. 2015;521:436–444. doi: 10.1038/nature14539. [DOI] [PubMed] [Google Scholar]
- 630.Schmidhuber J. Deep learning in neural networks: An overview. Neural Netw. 2015;61:85–117. doi: 10.1016/j.neunet.2014.09.003. [DOI] [PubMed] [Google Scholar]
- 631.Paliwal K., Lyons J., Heffernan R. A Short Review of Deep Learning Neural Networks in Protein Structure Prediction Problems. Adv. Tech. Biol. Med. 2015;3:3. doi: 10.4172/2379-1764.1000139. [DOI] [Google Scholar]
- 632.Torrisi M., Pollastri G., Le Q. Deep learning methods in protein structure prediction. Comput. Struct. Biotechnol. J. 2020;18:1301–1310. doi: 10.1016/j.csbj.2019.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 633.Torrisi M., Pollastri G. Brewery: Deep learning and deeper profiles for the prediction of 1D protein structure annotations. Bioinformatics. 2020;36:3897–3898. doi: 10.1093/bioinformatics/btaa204. [DOI] [PubMed] [Google Scholar]
- 634.Wang J., Cao H., Zhang J.Z.H., Qi Y. Computational Protein Design with Deep Learning Neural Networks. Sci. Rep. 2018;8:6349. doi: 10.1038/s41598-018-24760-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 635.Xu J. Distance-based protein folding powered by deep learning. Proc. Natl. Acad. Sci. USA. 2019;116:16856–16865. doi: 10.1073/pnas.1821309116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 636.Drori I., Thaker D., Srivatsa A., Jeong D., Wang Y., Nan L., Wu F., Leggas D., Lei J., Lu W., et al. Accurate Protein Structure Prediction by Embeddings and Deep Learning Representations. [(accessed on 15 September 2021)];arXiv. 2019 Available online: https://arxiv.org/abs/1911.05531v1.1911.05531v1 [Google Scholar]
- 637.Senior A.W., Evans R., Jumper J., Kirkpatrick J., Sifre L., Green T., Qin C., Zidek A., Nelson A.W.R., Bridgland A., et al. Protein structure prediction using multiple deep neural networks in the 13th Critical Assessment of Protein Structure Prediction (CASP13) Proteins. 2019;87:1141–1148. doi: 10.1002/prot.25834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 638.Senior A.W., Evans R., Jumper J., Kirkpatrick J., Sifre L., Green T., Qin C., Zidek A., Nelson A.W.R., Bridgland A., et al. Improved protein structure prediction using potentials from deep learning. Nature. 2020;577:706–710. doi: 10.1038/s41586-019-1923-7. [DOI] [PubMed] [Google Scholar]
- 639.Jumper J., Evans R., Pritzel A., Green T., Figurnov M., Ronneberger O., Tunyasuvunakool K., Bates R., Zidek A., Potapenko A., et al. Highly accurate protein structure prediction with AlphaFold. Nature. 2021;596:583–589. doi: 10.1038/s41586-021-03819-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 640.Tunyasuvunakool K., Adler J., Wu Z., Green T., Zielinski M., Zidek A., Bridgland A., Cowie A., Meyer C., Laydon A., et al. Highly accurate protein structure prediction for the human proteome. Nature. 2021;596:590–596. doi: 10.1038/s41586-021-03828-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 641.Pereira J., Simpkin A.J., Hartmann M.D., Rigden D.J., Keegan R.M., Lupas A.N. High-accuracy protein structure prediction in CASP14. Proteins. 2021 doi: 10.1002/prot.26171. [DOI] [PubMed] [Google Scholar]
- 642.Simpkin A.J., Sánchez Rodríguez F., Mesdaghi S., Kryshtafovych A., Rigden D.J. Evaluation of model refinement in CASP14. Proteins. 2021 doi: 10.1002/prot.26185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 643.Baek M., DiMaio F., Anishchenko I., Dauparas J., Ovchinnikov S., Lee G.R., Wang J., Cong Q., Kinch L.N., Schaeffer R.D., et al. Accurate prediction of protein structures and interactions using a 3-track network. bioRxiv. 2021 doi: 10.1101/2021.06.14.448402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 644.Bouatta N., Sorger P., AlQuraishi M. Protein structure prediction by AlphaFold2: Are attention and symmetries all you need? Acta Crystallogr. D Struct. Biol. 2021;77:982–991. doi: 10.1107/S2059798321007531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 645.Callaway E. DeepMind’s AI predicts structures for a vast trove of proteins. Nature. 2021;595:635. doi: 10.1038/d41586-021-02025-4. [DOI] [Google Scholar]
- 646.Stepniewska-Dziubinska M.M., Zielenkiewicz P., Siedlecki P. Development and evaluation of a deep learning model for protein-ligand binding affinity prediction. Bioinformatics. 2018;34:3666–3674. doi: 10.1093/bioinformatics/bty374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 647.Tian K., Shao M., Wang Y., Guan J., Zhou S. Boosting compound-protein interaction prediction by deep learning. Methods. 2016;110:64–72. doi: 10.1016/j.ymeth.2016.06.024. [DOI] [PubMed] [Google Scholar]
- 648.Verma N., Qu X., Trozzi F., Elsaied M., Karki N., Tao Y., Zoltowski B., Larson E.C., Kraka E. SSnet: A Deep Learning Approach for Protein-Ligand Interaction Prediction. Int J. Mol. Sci. 2021;22:1392. doi: 10.3390/ijms22031392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 649.Jiménez-Luna J., Grisoni F., Weskamp N., Schneider G. Artificial intelligence in drug discovery: Recent advances and future perspectives. Expert Opin. Drug Discov. 2021;16:949–959. doi: 10.1080/17460441.2021.1909567. [DOI] [PubMed] [Google Scholar]
- 650.Laine E., Eismann S., Elofsson A., Grudinin S. Protein sequence-to-structure learning: Is this the end(-to-end revolution)? [(accessed on 15 September 2021)];arXiv. 2021 doi: 10.1002/prot.26235. Available online: https://arxiv.org/abs/2105.07407.2105.07407 [DOI] [PubMed] [Google Scholar]
- 651.Arús-Pous J., Probst D., Reymond J.L. Deep Learning Invades Drug Design and Synthesis. Chimia. 2018;72:70–71. doi: 10.2533/chimia.2018.70. [DOI] [PubMed] [Google Scholar]
- 652.Arús-Pous J., Blaschke T., Ulander S., Reymond J.L., Chen H., Engkvist O. Exploring the GDB-13 chemical space using deep generative models. J. Cheminfor. 2019;11:20. doi: 10.1186/s13321-019-0341-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Jurtz V.I., Johansen A.R., Nielsen M., Almagro Armenteros J.J., Nielsen H., Sønderby C.K., Winther O., Sønderby S.K. An introduction to deep learning on biological sequence data: Examples and solutions. Bioinformatics. 2017;33:3685–3690. doi: 10.1093/bioinformatics/btx531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 654.Gómez-Bombarelli R., Wei J.N., Duvenaud D., Hernández-Lobato J.M., Sánchez-Lengeling B., Sheberla D., Aguilera-Iparraguirre J., Hirzel T.D., Adams R.P., Aspuru-Guzik A. Automatic Chemical Design Using a Data-Driven Continuous Representation of Molecules. ACS Cent. Sci. 2018;4:268–276. doi: 10.1021/acscentsci.7b00572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 655.Khemchandani Y., O’Hagan S., Samanta S., Swainston N., Roberts T.J., Bollegala D., Kell D.B. DeepGraphMolGen, a multiobjective, computational strategy for generating molecules with desirable properties: A graph convolution and reinforcement learning approach. J. Cheminfor. 2020;12:53. doi: 10.1186/s13321-020-00454-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 656.Shrivastava A.D., Kell D.B. FragNet, a contrastive learning-based transformer model for clustering, interpreting, visualising and navigating chemical space. Molecules. 2021;26:2065. doi: 10.3390/molecules26072065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 657.Karimi M., Wu D., Wang Z., Shen Y. DeepAffinity: Interpretable deep learning of compound-protein affinity through unified recurrent and convolutional neural networks. Bioinformatics. 2019;35:3329–3338. doi: 10.1093/bioinformatics/btz111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 658.Biswas S., Khimulya G., Alley E.C., Esvelt K.M., Church G.M. Low-N protein engineering with data-efficient deep learning. Nat. Methods. 2021;18:389–396. doi: 10.1038/s41592-021-01100-y. [DOI] [PubMed] [Google Scholar]
- 659.Wu Z., Yang K.K., Liszka M., Lee A., Batzilla A., Wernick D., Weiner D.P., Arnold F.H. Signal peptides generated by attention-based neural networks. ACS Synth. Biol. 2020;9:2154–2161. doi: 10.1021/acssynbio.0c00219. [DOI] [PubMed] [Google Scholar]
- 660.Elnaggar A., Heinzinger M., Dallago C., Rihawi G., Wang Y., Jones L., Gibbs T., Feher T., Angerer C., Steinegger M., et al. ProtTrans: Towards Cracking the Language of Life’s Code Through Self-Supervised Deep Learning and High Performance Computing. [(accessed on 15 September 2021)];arXiv. 2020 Available online: https://arxiv.org/abs/2007.06225.2007.06225 [Google Scholar]
- 661.Riesselman A.J., Ingraham J.B., Marks D.S. Deep generative models of genetic variation capture the effects of mutations. Nat. Methods. 2018;15:816–822. doi: 10.1038/s41592-018-0138-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 662.Volk M.J., Lourentzou I., Mishra S., Vo L.T., Zhai C., Zhao H. Biosystems Design by Machine Learning. ACS Synth. Biol. 2020;9:1514–1533. doi: 10.1021/acssynbio.0c00129. [DOI] [PubMed] [Google Scholar]
- 663.Mulligan V.K. Current directions in combining simulation-based macromolecular modeling approaches with deep learning. Expert Opin. Drug Discov. 2021;16:1025–1044. doi: 10.1080/17460441.2021.1918097. [DOI] [PubMed] [Google Scholar]
- 664.Meinen B.A., Bahl C.D. Breakthroughs in computational design methods open up new frontiers for de novo protein engineering. Protein Eng. Des. Sel. 2021;34:gzab007. doi: 10.1093/protein/gzab007. [DOI] [PubMed] [Google Scholar]
- 665.Kreutter D., Schwaller P., Reymond J.-L. Predicting enzymatic reactions with a molecular transformer. Chem. Sci. 2021;12:8648–8659. doi: 10.1039/D1SC02362D. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.