Abstract
Introduction
We investigated bone differentiation and proliferation potencies of human bone tissue-derived mesenchymal stromal cells (hBT-MSCs) after long-term cryopreservation. We determined the presence of any morphological and characteristic changes due to freezing to identify issues that need to be solved for future clinical applications.
Subjects and methods
A total of 15 samples of hBT-MSCs that had been cryopreserved for different lengths of time, ranging from one year to 20 years (n = 3 each), were thawed and recultivated after being collected from excess iliac cancellous bone specimens of patients who underwent secondary alveolar bone grafting for cleft lip and palate in our department. We determined viability by observing calcein/EthD-stained cells under a confocal microscope, and the cell proliferation experiment was performed for one week using the Water Soluble Tetrazolium salts (WST) assay method. A confocal microscope was also used to identify any excessively accumulated senescence-associated growth factor SA-βgal. Differentiation potency was assessed in the following three groups: bone differentiation, adipocyte differentiation, and nondifferentiation induction. We examined bone/adipocyte differentiation potencies using Alizarin Red staining, Ca quantitation, and Oil Red staining after continuously culturing cells for four weeks.
Results
Viability test results indicated that the proportion of viable cells decreased as the number of years of cryopreservation increased. The cell proliferation experiment showed that cells cryopreserved for a shorter duration multiplied exponentially. In the aging test, cells cryopreserved for ≥5 years showed similar positive reactions independent of the number of years of cryopreservation. In the cell proliferation test, there was no statistically significant difference between the years of cryopreserving. We compared bone differentiation and adipocyte differentiation ability with the non-induction group, and the induction group was confirmed to have a statistical advantage. However, there was no significant difference in the induction group pertaining to different ages.
Conclusions
Samples cryopreserved for 20 years remained competent in bone and adipocyte differentiation. However, their differentiation direction tended to skew to either bone or adipocyte differentiation. Our results suggest that freezing does not accelerate aging, and samples cryopreserved for a long time are useful in future clinical applications.
Keywords: Human bone tissue-derived mesenchymal stromal cells, Multipotency, Cell proliferation potency, Cryopreservation, Transplantation
1.Introduction
Bone tissue undergoes continuous formation and resorption of bone regulated by bone cells, with old bone constantly replaced by new ones. With such an active and dynamic metabolic mechanism, bone tissue has excellent spontaneous repairability. However, spontaneous healing cannot be expected to occur in cases of complex fractures and extensive bone defects such as trauma and congenital diseases. Consequently, bone graft surgery must be performed in these cases. Bone grafting procedures include autogenous bone graft, where bone extracted from the same patient is used for grafting. Allogeneic bone grafts, where bone obtained from another person is used for grafting, and alloplastic bone grafts, where the bone used for grafting is artificial. While bone regeneration is likely with autogenous bone grafts, such grafting procedures require invasive harvesting of healthy bone, and only limited amounts are removable. In contrast, allogeneic bone grafting uses stored bone samples, and thus a sufficient quantity of bone is available. However, the heterogeneity of allogeneic bone preparations, graft immune responses, and the high infection risk assocated with this process poses significant challenges to this method. A stable supply of artificial bone is available in alloplastic bone grafts becuase it is an industrial product. However, artificial bone products are inorganic materials, which alone are usually devoid of the new bone induction activity in vivo [1]. Cleft lip and palate (CL/CP) is a disease found in approximately 1 in 500 Japanese newborns. As a result, surgery must be performed multiple times for various purposes ranging from functional to morphological improvement. The development of normal occlusion is an important therapeutic objective. For this purpose, osseous reconstruction needs to be performed in the bone defect site of the alveolar cleft to promote appropriate tooth eruption. Currently, autologous iliac crest cancellous bone is a common treatment for alveolar cleft [2,3]. This secondary bone grafting is first performed when permanent anterior teeth erupt at 5–8 years of age; however, the objective is not always achievable with a single bone graft procedure. Multiple operations are required, for example, in cases where patients are too young to harvest a sufficient amount of cancellous bone or where the bone grafts are resorbed. Accordingly, we have been researching and developing alternative bone materials by tissue engineering that could reduce the level of invasiveness of this surgery. We are conducting basic research on the clinical applications of mesenchymal cells, obtained as outgrowths in primary cultures from surplus iliac tissue, used as donors in secondary bone grafting [4,5]. This procedure is an alternative to using mesenchymal stem cells from bone marrow punctures commonly used as cell sources in studies on alternative bone materials. Thus far, we have established methods for the isolation, proliferation, and cryopreservation of bone marrow mesenchymal cells [6]. We also developed a hybrid-type bone substitute using long-term cryopreserved human bone tissue-derived mesenchymal stromal cells [7]. We have confirmed the osteogenic potency of human bone marrow-derived mesenchymal stromal cells in the dorsal subcutaneous tissue of mice [8] and during the preparation of a rat cranial bone defect model [9]. We have also reported on the bone differentiation competency and safety of samples cryopreserved for ≥10 years [10]. This study examins bone and adipocyte differentiation potencies, proliferative potencies, and freezing-induced transformation and aging of hBT-MSCs cryopreserved for ≥20 years to review potential challenges and issues associated with their long-term storage in detail.
2. Subjects and methods
2.1. Subjects
The Kitasato University Institutional Review Board approved this study (approval number: B12-101). We thawed and recultivated cryopreserved hBT-MSCs from excess iliac cancellous bone of patients who underwent secondary bone grafting at our department. A total of 15 samples from six males and nine females with a mean age of 7.7 years (range, 5–17 years) were used after cryopreservation for 1, 5, 10, 15, and 20 years (n = 3 each; Table 1). All samples were collected from iliac cancellous bone with no other underlying disease except cleft lip and palate.
Table 1.
Doner information.
2.2. Cryopreservation and recultivation of primary culture and human bone tissue-derived mesenchymal stromal cells (hBT-MSCs)
We cultured iliac bone samples collected during secondary bone grafting, but unused, in a minimum essential medium (a-MEM medium; Life Technologies Corporation, CA, USA) supplemented with 10% fetal bovine serum (FBS) (Sigma–Aldrich MO, USA), antibiotics (100 U/ml penicillin and 100 g/ml streptomycin), and 1 ng/ml bFGF in 25 cm2 flasks (Sumitomo Bakelite Co, Tokyo, Japan) at 37 °C in 5% CO2, with medium exchanges performed twice per week. At subconfluency, we subcultured the cells in 75 cm2 flasks, suspended in serum-containing CELLBANKER™ (Nippon Zenyaku Kogyo Co., Ltd., Fukushima, Japan), and cryopreserved at −80 °C.
Cryopreserved cells were thawed and recultivated in a-MEM medium at 37 °C in 5% CO2. Then, we harvested the cells at subconfluency. We used secondary passage cultures for all samples in all experiments.
2.3. Viability test
After thawing, cells were seeded at 1 × 104/50 μl in 35-mm glass dishes (IWAKI. AGC TECHNO GLASS Co., Ltd., Shizuoka, Japan), and were observed under an inverted confocal fluorescence microscope (LSM710 and LSM780 system; LSM Software ZEN 2012, Carl Zeiss Microscopy, Germany) after calcein-ethidium homodimer III staining using Live/Dead Cell Staining Kit II (TAKARA BIO INC., Tokyo, Japan).
2.4. Cell senescence test
Cells were seeded at 1 × 104/50 μl in 35-mm collagen-coated glass dishes (IWAKI. AGC TECHNO GLASS Co., Ltd., Shizuoka, Japan), and was observed under an inverted confocal fluorescence microscope (LSM710 and LSM780 system; LSM Software ZEN 2012, Carl Zeiss Microscopy, Germany) after SA-β-gal staining with Cellular Senescence Detection kit-SPIDER-bGal (DOJINDO LABORATORIES, Kumamoto, Japan).
2.5. Cell proliferation experiment
We used the following specimens for the cell proliferation test. A total of 18 samples from 11 males and seven females with a mean age of 7.4 years (range, 5–17 years) were used after cryopreservation for 0, 15, and 20 years (n = 2 each), 1 years (n = 3), 5 years (n = 4), 10 years (n = 5) (Table 1). All cases were collected from iliac cancellous bone and had no underlying disease other than cleft lip and palate.
One sample for each storage period was tested only in the cell proliferation experiment using Cell Counting Kit-8 (DOJINDO LABORATORIES, Kumamoto, Japan). Cells were seeded at 0.5 × 104 or 0.25 × 104 cells/well in 96-well plates. WST-8 (2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium) added as a coloring reagent on days 0, 1, 3, 5, and 7, and absorbance values at 450 nm were measured with a plate reader (SpectraMaxM2; Molecular Devices, CA, USA). Cell counts were converted from absorbance values according to the Lambert–Beer law [11] and were used to construct proliferation curves. For statistical evaluation, we performed the Steel–Dwass test. A p-value of < 0.05 was considered statistically significant. Data were expressed as mean ± SEM. We conducted statistical analysis using the JMP® Pro 14.2 software (SAS Institute Inc., Cary, North Carolina).
2.6. Osteogenic differentiation, adipocyte differentiation induction
Cells were seeded at 1 × 105 cells/well in 6-well plates and subjected to differentiation induction for four weeks. The medium was changed twice per week.
Osteogenic differentiation was performed in a-MEM medium (containing dexamethasone 0.1μM/ascorbic acid 0.05 mM/b-glycerophosphate 10 mM) supplemented with 10% FBS.
Adipocyte differentiation was induced in a-MEM medium (containing insulin 10 μg/ml/isobutyl methylxanthine 0.5 mM/dexamethasone 1 μM/indomethacin 0.2 mM) supplemented with 10% FBS.
The non-induction group of cells was cultured for four weeks in a-MEM medium.
2.7. Evaluation of differentiation potency
2.7.1. Evaluation of osteogenic differentiation
2.7.1.1. Alizarin Red staining
The cell surface was washed twice with phosphate-buffered saline (PBS) (−) and then fixed with 100% ethanol. Cells were stained for 2 min with 1.3% alizarin Red S solution (Wako Pure Chemical Corporation, Osaka, Japan), washed thrice with distilled water to remove the excess staining solution, and then allowed to dry.
2.7.1.2. Calcium quantitation
After we washed the cell surface thrice with PBS (−), we extracted calcium with 0.5 N HCl, and Espa·Ca (NIPRO, Osaka, Japan) was used to quantify the calcium with the Arsenazo III method. The Steel-Dwass test was used for statistical evaluation, and a value of P < 0.05 was considered statistically significant. Data were expressed as mean ± SEM.
2.7.2. Evaluation of adipocyte differentiation
2.7.2.1. Oil Red staining
After washing twice with PBS (−), cells were fixed with 10% formalin solution. We washed the cells successively with distilled water followed by 60% isopropanol. Then we stained the cells for 20 min with Oil Red solution (Sigma–Aldrich, MO, USA). After staining, we washed the cells successively with 60% isopropanol and then PBS. We determined the amount of generated adipocyte as count, total area, % areas, and average size using the Image J analysis software version 1.36 b (National Institutes of Health, USA http://:rsb.Info.Nih.gou/ij/).
2.8. Tile scan analysis of live/dead cell staining and senescent cell staining
For live/dead cell staining and senescent cell staining, we performed 5 × 5 tile scanning (1 sample for each age) and calculated the ratios of the mean counts. In live/dead cell staining, only live or dead cells in a single visual field are stained. In contrast, in senescent cell staining, nuclei are stained. Therefore, the number of cells was considered equal to the number of nuclei. Furthermore, for each staining, we measured the fluorescence intensity and calculated the mean value. Imaris (BITPRANE Co., Ltd.) was used for these measurements.
3. Results
3.1. Viability test
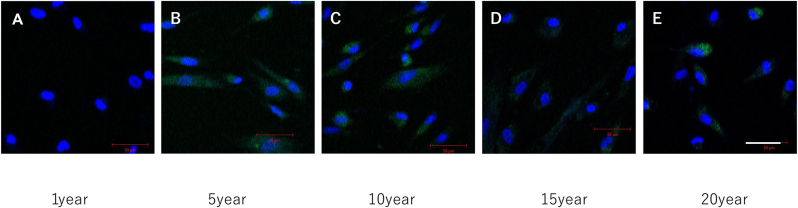
Although the proportion of dead cells increased in samples cryopreserved for ≥5 years, there was no loss of shape in viable cell staining. However, and despite the fact that the proportion of viable cells was high in samples stored for ≥10 years, some cells were not morphologically intact. In 20-year-old samples, the proportion of viable cells decreased, and we noted the marked appearance of cells that were swollen, had a disrupted membrane, or had lost their shape (Fig. 1).
Fig. 1.
Live/dead cell testing (A)1 year(B)5 year(C)10 year(D)15 year(E)20 year;bar = 50 μm. Live cell stained green fluorescence and dead cell stained red fluorescence.In samples cryopreserved for one year, cells including dead cells were found to have a regular and round shape. For samples cryopreserved for five years, live cell staining (green fluorescence) showed no loss of shape. In samples cryopreserved for ≥10 years, the proportion of viable cells was still high, but the shape of some cells had deteriorated. In samples cryopreserved for 20 years, the proportion of viable cells was decreased, and cells that were swollen, had a disrupted membrane, or had lost their shape were noticeable (E; arrows).
3.2. Cell senescence test
All cells cryopreserved for ≥5 years were positive for SA-β-gal, indicating the accumulation of a senescence-associated substance (Fig. 2).
Fig. 2.
Cell senescence (A)1 year SA-β-gal(B)5 year SA-β-gal(C)10 year SA-β-gal(D)15 year SA-β-gal(E)20 year SA-β-gal bar = 50 μm SPiDER-bGal-positive cells emit green fluorescence and their nuclei are stained dark blue. SA-β--gal-positive cells were found in all samples cryopreserved for ≥5 years.
3.3. Cell proliferation experiment
The proliferation curves revealed that there was no significant difference in the population of cells due to the length of storage period (P > 0.05) (Fig. 3).
Fig. 3.
Cell proliferation curves. Cell proliferation tests use samples that are not cryopreserved for zero years. The rate of cell proliferation tended to be faster, and the cell count on day 7 was greater for cells cryopreserved for a shorter period (such as 1 year and 0 years). For cells cryopreserved for ≥10 years, the proliferation rate did not accelerate rapidly, but the number of cells increased at a constant rate. Data represent mean ± SEM of n = 2–5 for 6 independent experiments. Steel–Dwass test of all pairs was performed, but there was no significant difference.
3.4. Confirmation of potency of differentiation into various cells
3.4.1. Bone differentiation potency
3.4.1.1. Alizarin Red staining
Overall, cells from test samples were better stained compared to the control group. We confirmed the potency of differentiation into osteoblasts for all samples (Fig. 4).
Fig. 4.
AlizarinRed S staining (A)1 year(B) 5 year(C)10 year(D)15 year(E)20 year Upper, nondifferentiation induction group; lower, differentiation induction group In Alizarin Red S staining, all samples in the osteogenic differentiation group were stained regardless of the length of cryopreservation, while no samples in the nondifferentiation induction group were stained.
3.4.1.2. Calcium quantitation
No osteogenesis was observed in the non-induction group.
Samples cryopreserved for one year exhibited the lowest calcium production, as opposed to samples stored for five and 10 years, which showed comparable production levels. Calcium production levels of the 15- and 20-year-old samples were decreased but were still higher than the levels those of the 1-year-old samples (Fig. 5). There was no statistically significant difference between the ages in this regard (p > 0.05).
Fig. 5.
Ca quantitation. Values were calculated from the mean values of three samples. For the nondifferentiation induction group, the mean measurement was 0.2956, indicating virtually no production. For the induction group, significant osteogenesis was observed. The production level was the lowest for the sample cryopreserved for one year and comparable between those cryopreserved for five years and 10 years. High bone formation was observed for samples cryopreserved for up to 10 years.Unlike cells cryopreserved for one year, long-term cryopreserved cells (15–20 years) showed significantly greater levels of osteogenic potency compared to the non-induction group. Data represent mean ± SEM of n = 3 for 10 independent experiments.Steel–Dwass tests between the induction groups of all pairs were performed, but there no significant difference was noted. In comparison between the non-induced group and the induced group, in all groups, indicating a significant difference (∗p < 0.05).
3.4.2. Adipocyte differentiation
3.4.2.1. Oil Red staining
Fat droplets were confirmed in all samples that were cryopreserved for ≤15 years (Fig. 6).
Fig. 6.
Oil Red staining (A)1 year(B)5 year(C)10 year(D)15 year(E)20 year bar = 100μm Cells containing red-stained fat droplet are cells that underwent adipocyte differentiation. In this study, all samples cryopreserved for up to 15 years had a confirmed adipocyte differentiation potency. For samples cryopreserved for more than 20 years, one of three samples did not show differentiation potency, and another sample exhibited only a small cell count. For samples cryopreserved for 10 years, while the number of cells stained was small, all three samples were stained similarly, thereby verifying adipocyte differentiation potency. Using Image-j, the number of fat droplets, total area, % areas, and average size were measured and tested. Data represent mean ± SEM of n = 3 for 10 independent experiments.Steel–Dwass test between induction groups of all pairs were performed, but there was no significant difference. In comparison between the non-induced group and the induced group, in all groups, indicating a significant difference (∗p < 0.05).
In contrast, in samples cryopreserved for > 20 years, the number of confirmed cell droplets became scarce. There were no significant differences among the ages with respect to the total count, total area, % areas, and average size.
There was a significant difference between the non-induced group and the formation of lipid droplets (P < 0.05).
3.5. Tile scan analysis of live/dead cell staining and senescent cell staining
While the 5 × 5 tile scan analysis revealed that the senescence-associated factor measured tended to be higher, the osteogenic potency was still considerably high for samples stored for five years. Moreover, there were adequate levels of adipocyte differentiation for the 15- and 1-year-old samples showing low levels of overaccumulation of senescent cells (Table 2).
Table 2.
Tile scan analysis of live/dead cell staining and senescent cell staining.
| 1 year | 5 year | 10 year | 15 year | 20 year | |
|---|---|---|---|---|---|
| Live cell | 91 | 139 | 92 | 154 | 33 |
| Dead cell | 7 | 14 | 23 | 57 | 39 |
| Live/dead | 13 | 0.946 | 4 | 2.702 | 0.846 |
| SA-β-gal | 4 | 292 | 320 | 105 | 193 |
| SA-β-gal Fluorescence intensity | 0.02 | 25.7632 | 37.3993 | 24.9 | 32.4747 |
| Dapi | 87 | 37.3993 | 395 | 104 | 211 |
| Dapi Fluorescence intensity | 121.79 | 24.9 | 164.45 | 183.09 | 219.276 |
The image analysis software program Imaris was used to analyze mean cell counts and fluorescence intensities using 5 × 5 tile scan. For live/dead cell testing, the number of cells counted as viable cells was found to have decreased considerably in samples cryopreserved for ≥15 years. In senescent cell staining, neither the number of positive cells nor the intensity of fluorescence was proportional to the duration of cryopreservation.
4. Discussion
4.1. Cryopreservation
Cryopreservation has long been attempted as a means to store cells for long periods without changing their properties. Our laboratory has been cryopreserving and storing cells using the same technique under the guidance of the same engineer for over 20 years. Since FBS is changed slightly every few years, the manufacturer will also change almost every 3–5 years. Each time, three to five types of serum samples are collected and examined to assess their ability to proliferate for approximately one week with the same cells (iliac cancellous bone). In our lab, we also perform an FBS lot check. More specifically, we select and use cells with the same growth ability as the serum used. Under these conditions, we consider the usefulness of cells in long-term cryopreservation in our department. In general, cells suffer damage from freezing in two years [12]: physical damage associated with the formation of intracellular ice crystals and damage associated with hyperosmolarity during freezing. The freezing rate is considered the most important factor affecting cell viability after being frozen-thawed. If the freezing rate is too fast [13], intracellular ice crystals are formed, which damage the organelle and cause cell lysis upon thawing to destroy the cell wall. In contrast, if the freezing rate is too slow [14], the extracellular osmotic pressure increases with the freezing of the extracellular solution, moisture moves from the supercooled cells, and intracellular dehydration proceeds. The optimal freezing rate depends on two independent factors: cell size and water permeability [15]. In hematopoietic stem cells, a cooling rate of −1 °C / min using the program freezer is optimal [16], but is acceptable up to −3 °C / min. Since this cooling rate [17] can be obtained by simply remaining in a freezer at −70°C to −80 °C since the beginning, this method is widely used in current clinical practice. When cooled at this rate, intracellular dehydration is mild. The ice crystals formed remain small enough and do not destroy the organelle or cell wall, thus preserving cell viability [18]. Concerming the cooling rate, the method used in our department, which involves immediate storage and transportation on dry ice at −79 °C and direct placement in a deep freezer at −80°C minimizes the formation of intracellular ice crystals. It is highly possible that, when frozen cells are thawed, the ice crystals formed in the cells grow larger [19], and recrystallization occurs. Cells may need rapid warming to prevent any physical damage[20]. Generally, a heating rate of 90°C–100 °C / min or more is recommended. Since we thaw under a constant temperature bath (37 °C) at the time of thawing, we do not obtain a heating rate of 90°C–100 °C / min. The cryopreservation solution is further divided into two types: intracellular permeabilized cryoprotectant and extracellular impermeable cryoprotectant. The former is dimethylsulfoxide (DMSO) or glycerol, and is the most widely used. Examples of the latter include hydoxyethylstarch (HES) and dextran. Several studies have found that these combinations are more effective [21,22]. We do not use a combination type in our lab, but instead, we use a commercial cryopreservation solution, a 10% DMSO serum-containing product. Considering our freezing, storage again, and thawing procedures, it was inferred that there was a high possibility that damage caused by recrystallization during thawing occurred. We have previously reported that hBT-MSCs cryopreserved for ≥10 years are capable of bone and adipocyte differentiation [10] and osteogenesis in vivo [9], and as a result, they are highly reputed due to their high potential. A clinical study has shown that cells flash-frozen at −196 °C, at a temperature where cell activity is stopped [23], could be administered to the original patients after storage for up to 41 years [24]. While some previous studies have shown that samples viability after 4–8 years of cryopreservation at −80 °C, which is not such an ultra-low temperature, was maintained [25,26], the present study confirming the multipotency of mesenchymal cells cryopreserved for up to 20 years are of significance in clinical applications. This is becuase these data suggest the feasibility of long-term cryopreservation with a simpler method. In general, cryopreservation fluids (including CELLBANKER, which is used in our department) guarantee ≥80% cell viability at the time of thawing after storage of 3–5 years from the date manufactured. In this study, cells remained capable of differentiation after cryopreservation for even longer periods. No major differences in differentiation and proliferation competencies were noted among samples stored for up to 15 years (Fig. 4, Fig. 5, Fig. 6). Since floating necrotized cells due to thawing after intracellular freezing [27] (e.g., cell membrane disruption) are removed during centrifugation and supernatant aspiration, all dead cells counted in live/dead cell staining have an intact membrane structure.
In this study, the viable cell staining showed no shape destruction. In contrast, the proportion of dead cells determined with live/dead cell staining increased in samples cryopreserved for five years (exceeding the shelf life of the cell cryopreservation fluid). In samples stored for 10 years, the proportion of viable cells was high, but some cells had a disordered and collapsed shape. In contrast, in samples cryopreserved for 20 years, the proportion of viable cells decreased, and cells with disordered and collapsed shape walls were noticeable (Fig. 1). The live/dead cell testing results suggest that cells that survive thawing and undergo differentiation induction after recultivation can be cultured, presumably because they are strong cells that have withstood such a stressful process. From the above, it is suggested that even long-term cryopreserved specimens (>20 years) can maintain their differentiation potential and can thus be used as cells.
4.2. Do cells age due to freezing?
To investigate whether the age of frozen samples depends on the duration of cryopreservation, we stained cells to detect senescence-associated b-galactosidase (SA-β-gal) as a marker of aging. The β-galactosidase activity, which functions at near-neutral pH, is measured based on the blue color intensity of the X-Gal product used as a substrate. Many previous reports have documented β-galactosidase activity at the cell or tissue level [28]. Such β-galactosidase activity has become well-known as a marker for senescence after the activity was reported to be high in cells that had undergone many rounds of passage [29]. In general, the lifespan of cells is regulated in two steps of cell senescence and crisis. The former is G1 arrest by cell cycle regulators, and the latter, which was discovered by Heyflic [30] 45 years ago, is the irreversible arrest of cell proliferation that occurs when cells reach the intrinsically allowed number of cell divisions (i.e., when telomeres shorten to a limited size). The proliferation of hBT-MSCs is finite. Unlike ES cells and cancer cells, hBT-MSCs have a very low level of telomerase activity and, theoretically, have an absolute lifespan limit due to telomere shortening. The cell division lifespan tends to be shorter as the age of the cell donor increases [31,32], and more b-galactosidase activity is induced as cells undergo more rounds of passage [29,33]. Cells in a state of cell proliferation arrest are very large and flat, making them distinct from other cells in the proliferative phase. These changes in the morphological characteristics of hBT-MSCs have been reported to be prominent immediately before proliferation. Moreover, traits such as shape,differentiation, and potency have also been reported to change. In the present study, all cell preparations were P2, and the source cells were collected from young donors (mean age, 7.9 years). Therefore, increased b-galactosidase activity was observed in cells that had undergone the freezing process. As far as staining results are concerned, no dependency on the duration of cryopreservation was identified, and the activity in samples cryopreserved for five years was as high as the activity in samples cryopreserved for 20 years (Fig. 2). In cells with high b-galactosidase activity, no correlation was found between the potency (size of potential capacity) in osteoblasts differentiation and formation of fat droplets, with high differentiation potency observed (Fig. 4, Fig. 5, Fig. 6). The results of the present study suggest that freezing is unlikely to accelerate the aging of cells over time.
4.3. Safety and future prospects of long-term cryopreserved samples
Regarding the safety of samples cryopreserved for as long as 10 years, our laboratory conducted investigations on thier chromosomal morphology, abnormalities in the p53 gene, which is a tumor suppressor gene, expression of the Myc gene, which is one of oncogenes, and morphology with G-banding, and these tests revealed no abnormalities [10]. The cultivation of hBT-MSCs using serum from safe and stable autologous blood is a useful procedure in terms of infection risk, foreign body, and immune mechanism. Currently, active basic research using allogeneic MSCs is ongoing. While MSCs are generally considered to be immunosuppressive and induce no immune responses after allogeneic transplantation [34], some studies have shown that MSCs do not induce immuno-tolerance and are immunogenic [35,36]. For children with cleft lip and palate (CL/CP), we aim to collect a part of their maxilla which will be collected at the first surgery (cheiloplasty), and use their own cryopreserved maxilla derived MSCs for secondary bone grafting in the mixed dentition stage and again, after recultivation, for secondary correction of cleft lip and nose deformities during or after adolescence. This is because with this method we will not need to perform multiple donor operations during early childhood, and thus minimize the scars caused by donor surgery. The findings obtained in the present study that used samples stored for up to 20 years provide a key to open the path toward the realization of clinical applications involving a series of treatments. Unlike treatment with allogeneic MSCs, the treatment that we are pursuing can also alleviate concerns about immune rejection because we use serum from autologous blood. Although hBT-MSCs cryopreserved for almost 20 years showed high level of b-Galactosidase activity after five years, their differentiation potency was not markedly decreased. Since the number of n was small, there were many factors that were estimated to depend on the individual potential (Fig. 7). Human-derived cells that have been in cryopreservation for more than 20 years (although not intended for long-term storage at that time) are almost nonexistent in our department. In the future, we aim to ensure universality by conducting similar reproduction experiments when the 15-year cells, which are currently stored, eventually reach their 20th year. Furthermore, issues to be addressed in future studies involve the appropriate freezing and thawing methods for bone substitute preparation, determine the cause of senescent cells emergence after freezing, and to identify the means to prevent this from occuring.
Fig. 7.
Oil Red staining、Statistical processing of Steel Dwass. Steel–Dwass test was performed, but there were many variations due to the small number of specimens including the% area, and no significant difference occurred. Total area and average size had similar results.
5. Conclusions
Long-term cryopreserved hBT-MSCs demonstrated fewer living cells when thawed for a longer freezing period. Furthermore, senescence staining data showed that all samples cryopreserved for ≥5 years were aged, but that the extent of aging was independent of the duration of cryopreservation from 5 to 20 years. Cell growth did not differ between ages, and the possibility of differentiation was confirmed in all years. Bone and fat differentiation abilities tend to be biased in either direction, but the possibility of donor origin was also considered. As future research subjects, I would like to investigate whether the above findings are due to the ability derived from donors, accumulate additional tests, determine whether this is related to cell aging, and reconfirm safety and appropriate freeze-thaw methods.
Acknowledgments
This study was supported partly by a Grant-in-Aid (C) from the Ministry of Education, Culture, Sports, Science and Technology, Japan (17K11852).
We would like to extend our appreciation to PhD. Masahiro Fukaya, PhD.Yoshinobu Hara (Department of Anatomy, Kitasato University School of Medicine) and Ms.Yukiko Matsuo (Department of Thoracic surgery, Kitasato University School of Medicine) for their great contribution to this study. We are also deeply grateful to the members of many departments of the Kitasato University Hospital for their valuable help with this study.
Footnotes
Peer review under responsibility of the Japanese Society for Regenerative Medicine.
References
- 1.Katsube Y., Yuba S., Ohgushi H. Regenerative medicine library 6; the Japanese society for regenerative medicine. Asakura Publishing Co., Ltd.; 2012. Bone regeneration using mesenchymal stem cells. Skeletal system; pp. 92–110. [Google Scholar]
- 2.Orth D., Howard R.D. An improved appliance for the pre-surgical orthodontic preparation of the cleft case for the secondary bone grafting. Br J Plast Surg. 1969;22:90–93. [PubMed] [Google Scholar]
- 3.Boyne P.J., Sands N.R. Secondary bone grafting of residual alveolar and palatal clefts. J Oral Surg. 1972;30:87–92. [PubMed] [Google Scholar]
- 4.Wongchuensoontorn C., Liebehenschel N., Schwarz U., Schmelzeisen R., Gutwald R. Application of a new chair-side method for the harvest of mesenchymal stem cells in a patient with nonunion of a fracture of the atrophic mandible-A case report. J Cranio-Maxillo-Fac Surg. 2009;37:155–161. doi: 10.1016/j.jcms.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 5.Behnia H., Khojasteh A., Soleimani M., Tehranchi A., Atashi A. Repair of alveolar cleft defect with mesenchymal stem cells and platelet derived growth factors. J Cranio-Maxillo-Fac Surg. 2012;40:2–7. doi: 10.1016/j.jcms.2011.02.003. [DOI] [PubMed] [Google Scholar]
- 6.Shimakura Y., Yamazaki Y., Uchinuma E. Experimental study on bone formation potential of cryopreserved human bone marrow mesenchymal cell/hydroxyapatite complex in the presence of recombinant human bone morphogenetic protein-2. J Craniofac Surg. 2003;14:108–116. doi: 10.1097/00001665-200301000-00021. [DOI] [PubMed] [Google Scholar]
- 7.Matsuo A., Yamazaki Y., Takase C., Aoyagi K., Uchinuma E. Osteogenic potential of cryopreserved human bone marrow-derived mesenchymal stem cells cultured with autologous serum. J Craniofac Surg. 2008;19:693–700. doi: 10.1097/SCS.0b013e318163f2cc. [DOI] [PubMed] [Google Scholar]
- 8.Takeda A., Yamazaki Y., Baba K., Ishiguro M., Aoyagi K., Ikemoto S. Osteogenic potential of human bone marrow-derived mesenchymal stromal cells cultured in autologus serum: a preliminary study. J Oral Maxillofac Surg. 2012;70:469–476. doi: 10.1016/j.joms.2012.03.021. [DOI] [PubMed] [Google Scholar]
- 9.Sugimogo T., Yamazaki Y., Kumazawa K., Sone Y., Takeda A., Uchinuma E. The significance of performing osteogenic differentiation in human bone tissue-derived mesenchymal stromal cells. J Oral Tissue Eng. 2013;11:103–112. [Google Scholar]
- 10.Kumazawa K., Sugimogo T., Yamazaki Y., Sone Y., Takeda A., Uchinuma E. Osteogenic potential, multipotency, and cytogenetic safety of human bone tissue-derived mesenchymal stromal cells (hBT-MSCs) after long-term cryopreservation. Kitasato Med J. 2014;44:95–103. [Google Scholar]
- 11.Koike H. Principle of spectrophotometry and measurement of action spectra and quantum yield. Low Temp Sci. 2009;67:431–447. [Google Scholar]
- 12.Mazur P. Cryobiology;The freezing of biological systems. Science. 1970;168:939–949. doi: 10.1126/science.168.3934.939. [DOI] [PubMed] [Google Scholar]
- 13.Mazur P. The role of intracellular freezing in the death of cells cooled at super optimal rates. Cryobiology. 1977;14:251–272. doi: 10.1016/0011-2240(77)90175-4. [DOI] [PubMed] [Google Scholar]
- 14.Bank H., Mazur P. Relation between ultrastructure and viability of frozen-thawed Chinese hamster tissue-culture cells. Exp Cell Res. 1972;71:441–454. doi: 10.1016/0014-4827(72)90314-x. [DOI] [PubMed] [Google Scholar]
- 15.Mazur P. The role of cell membranes in the freezing of yeast and other single cells. Ann NY Acad Sci. 1965;124:658–676. doi: 10.1111/j.1749-6632.1965.tb45420.x. [DOI] [PubMed] [Google Scholar]
- 16.Leibo S.P., Mazur P. The role of cooling rates in low temperature preservation. Cryobiology. 1971;2:447–452. doi: 10.1016/0011-2240(71)90035-6. [DOI] [PubMed] [Google Scholar]
- 17.Mazur P. Theoretical and experimental effects of cooling and warming velocity on the survival of frozen and thawed cells. Cryobiology. 1966;2:181–192. doi: 10.1016/s0011-2240(66)80165-7. [DOI] [PubMed] [Google Scholar]
- 18.Forman Sj K.G., Thomas E.D. .Black Well Scientific Publications; Boston: 1994. Bone marrow transplantation; pp. 299–308. [Google Scholar]
- 19.Luyet B.L., Rapatz G. Pattern of ice formation in some aqueous solutions. Biodynamica. 1958;8:1–6. [PubMed] [Google Scholar]
- 20.Leibo S.P., Farrant J., Mazur P., Hanna M.G., Smith L.H. Effect of freezing on marrow stem cell suspensions;interactions of cooling and warming rates in the presence of PVP,sucrose of glycerol. Cryobiology. 1970;6:315–321. doi: 10.1016/s0011-2240(70)80086-4. [DOI] [PubMed] [Google Scholar]
- 21.Makino S., Harada M., Akashi K., Taniguchi S., Shibuya T., Inaba S., Niho Y. A simplified method for cryopreservation of peripheral blood stem cells at -80 degrees C without rate-controlled freezing. Bone Marrow Transplant. 1991;8:239–244. [PubMed] [Google Scholar]
- 22.Nadamanee A., Schmidt G.M., Sniecinsky I. Storage of unfractionated bone marrow(BM)without rate-controlled freezing is equivalent to standard technique for short time storage. Blood. 1991;78:251A. [Google Scholar]
- 23.Asano T., Kenmochi T., Isono K. Organ preservation. J Jpn Surgical Society. 1996;97:958–963. [PubMed] [Google Scholar]
- 24.Sumida Y., Kitamura T., Motomura N., Saito A., Yamamoto S. Study on long-term cryopreservation method for human bone marrow stem cells (-196°C, for up to 41 years) – extension of storage life by Repetitive freeze-thaw-in vivo or-in vitro subculture (RFTIVIVTSC) in the presence of various growth factors and results of cultivation of peripheral vein and subcutaneous adipose tissue as mesenchymal cell sources. J J Transfus Cell Ther. 2016;60:94–95. [Google Scholar]
- 25.Ance B., Uodis B., Sergey N., Dace S., Agnese E. Characterization of human adipose-derived stem cells cultured in autologous serum after subsequent pssaging and long term cryopreservation. J Stem Cell. 2014;9:135–148. [PubMed] [Google Scholar]
- 26.MiyamotoY Ikeuchi M., Noguchi H., Hayashi S. Long-tern cryopereservation of human and other mammalian cells at -80°C for 8 years. Cell Med. 2018;10:7. doi: 10.1177/2155179017733148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leibo S.P. In: Charles M.S., Ivan L., editors. vol. 2. Karger; Basel: 1981. Cryobiology of immune competent cells; pp. 66–76. (The immune system). [Google Scholar]
- 28.Gary R.K., Kindell S.M. Quantitative assay of senescence-associated beta-galactosidase activity in mammalian cell extracts. Anal Biochem. 2005;343:329–334. doi: 10.1016/j.ab.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 29.Yumura W., Imasawa T., Suganuma S. Accelerated tubular cell senescence in SMP30 knockout mice. Histol Histopathol. 2006;21:1151–1156. doi: 10.14670/HH-21.1151. [DOI] [PubMed] [Google Scholar]
- 30.Heyflick L., Moorhead P.S. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585–621. doi: 10.1016/0014-4827(61)90192-6. [DOI] [PubMed] [Google Scholar]
- 31.Umezawa A. Replicative cellular aging of mesenchymal stem cells. Regen Med. 2008;7:49–52. [Google Scholar]
- 32.Zhang K., Ikeda Y., Kasugai S., Ikeda M. Extended culture conditions for multipotent bone marrow-derived mesenchymal stem cells. Kokubyo Gakkai Zasshi. 2016;83:13–24. [PubMed] [Google Scholar]
- 33.Goberdhan P.D., Xinhua L., George B., Meileen A., Glynis S. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Cell Biol. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lalu M.M., McIntyre L., Pugliese C. Safety of cell therapy with mesenchymal stromal cells(SafeCell).a systematic review and meta-analysis of clinical trials. PLoS. 2012;7 doi: 10.1371/journal.pone.0047559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kotobuki N., Katsube Y., Katou Y., Tadokoro M., Hirose M., Ohgushi H. In vivo survival and osteogenic differentiation of allogeneic rat bone marrow mesenchymal stem cells(MSCs) Cell Transplant. 2008;17:705–712. doi: 10.3727/096368908786092793. [DOI] [PubMed] [Google Scholar]
- 36.Eliopoulos N., Stagg J., Leieune L., Pommey S., Galipeau J. Allogeneic marrow stromal cells are immune rejected by MHC class Ⅰ-and class class Ⅱ-mismatched recipient mice. Blood. 2005;106:4057–4065. doi: 10.1182/blood-2005-03-1004. [DOI] [PubMed] [Google Scholar]