Abstract
Cardiac involvement is a common and serious problem in multisystem inflammatory syndrome in children (MIS-C). Echocardiographic evaluation of systolic and diastolic function by traditional, tissue Doppler and three-dimensional (3D) echocardiography was performed in consecutive 50 MIS-C patients during hospitalization and age-matched 40 healthy controls. On the day of worst left ventricular (LV) systolic function (echo-1), all left and right ventricular systolic function parameters were significantly lower (p < 0.001), E/A ratio was significantly lower, and averaged E/e′ ratio was significantly higher (median 1.5 vs. 1.8, p < 0.05; 8.9 vs. 6.3, p < 0.001 respectively) in patients compared to control. Patients were divided into 2 groups according to 3D LV ejection fraction (LVEF) on the echo-1: Group 1; LVEF < 55%, 26 patients, and group 2; LVEF ≥ 55%, 24 patients. E/e′ ratio was significantly higher in group 1 than group 2 and control at discharge (median 7.4 vs. 6.9, p = 0.005; 7.4 vs. 6.3, p < 0.001 respectively). Coronary ectasia was detected in 2 patients (z score: 2.53, 2.6 in the right coronary artery), and resolved at discharge. Compared with group 2, group 1 had significantly higher troponin-I (median 658 vs. 65 ng/L; p < 0.001), NT-pro BNP (median 14,233 vs. 1824 ng/L; p = 0.001), procalcitonin (median 10.9 vs. 2.1 µg/L; p = 0.009), ferritin (median 1234 vs. 308 µg/L; p = 0.003). The most common findings were ventricular systolic dysfunction recovering during hospitalization, and persisting LV diastolic dysfunction in the reduced LVEF group at discharge. Coronary artery involvement was rare in the acute phase of the disease. Also, in MIS-C patients, the correlation between LV systolic dysfunction and markers of inflammation and cardiac biomarkers should be considered.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00246-021-02738-3.
Keywords: MIS-C, COVID-19, Three-dimensional echocardiography, Ventricular systolic-diastolic function
Introduction
Many centers in Europe and America in 2020 have described a new syndrome associated with Covid-19 infection characterized by hyperinflammation and multiorgan involvement in children, presenting with clinical features similar to Kawasaki disease (KD) and toxic shock syndrome [1–7]. This syndrome has been named multisystem inflammatory syndrome in children (MIS‐C) associated with COVID‐19 by the Centers for Disease Control and Prevention and pediatric inflammatory multisystem syndrome temporally associated with SARS‐CoV‐2 in Europe [4, 8, 9]. MIS-C appears to be a delayed post-infectious response, 2–6 weeks after mostly asymptomatic or slightly symptomatic SARS-CoV-2 infection [10, 11].
World Health Organization reported a decrease in cases during COVID-19 pandemic in the spring–summer period in many countries, and an increasement with the autumn [12]. Similarly, a gradual increasement was seen in the number of cases from September 2020 in our country and a second peak was observed in the November–December 2020 period [13]. During this period, many children diagnosed with MIS-C were admitted to our hospital, a Covid-19 pandemic center.
In most studies, the cardiac involvement of MIS-C includes ventricular dysfunction, coronary artery dilation or aneurysms, and rarely pericarditis and valvulitis [4, 8, 9, 14].
In current practice, two-dimensional echocardiography (2DE) is the most commonly employed approach for the assessment of left ventricular (LV) function. However three-dimensional (3D) echocardiography is a more accurate and reproducible method than both M-mode and 2D Simpson’s biplane method and is as feasible as 2DE in children [15, 16]. To date, few studies performed a cardiac evaluation with conventional, tissue Doppler and 3D echocardiography in MIS-C patients.
This study aimed to evaluate systolic and diastolic function by traditional, tissue Doppler and 3D echocardiographic methods in MIS-C patients during hospitalization and to investigate the possible correlation between LV systolic function and cardiac biomarkers and other laboratory data. The secondary objectives were to assess the feasibility and reproducibility of left ventricular ejection fraction (LVEF) with 3D echocardiography in MIS-C patients.
Methods
Study Population and Protocol
A prospective single-center study was conducted at Ankara City Hospital between November 2020 and January 2021. During the study period, echocardiographic evaluation was performed in consecutive 50 MIS-C patients and age-matched 40 healthy controls. The study was approved by the Local Ethics Committee and informed written consent was obtained from the parents before enrollment.
The clinical diagnosis of MIS-C was made according to the World Health Organization (WHO) and the U.S.Centers for Disease Control and Prevention (CDC) criteria [8, 14]. MIS-C patients were classified as “KD-like”, “Kawasaki disease shock syndrome (KDSS)”, “toxic shock-like”, and “patients without either KD or shock”. Patients with Kawasaki-like presentations were defined according to the 2017 criteria of the American Heart Association, including both the classic type (fever for ≥ 5 days plus four or more clinical criteria, including bilateral bulbar non-exudative conjunctivitis, changes of the lips or oral cavity, non-suppurative laterocervical lymphadenopathy, polymorphic rash, erythema of the palms and soles, firm induration of the hands or feet, or both) and incomplete types. In incomplete types (fever for ≥ 5 days plus two or three of the aforementioned clinical criteria), the values of erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP), or both, were taken as an additional diagnostic criterion in association with the presence of anemia, thrombocytosis after 7 days of fever, hypoalbuminemia, hypertransaminasemia, leucocytosis, sterile pyuria, or an echocardiogram showing coronary aneurysms or cardiac dysfunction (i.e., left ventricular function depression, mitral valve regurgitation, or pericardial effusion) [17].
KDSS was defined as Kawasaki disease accompanied by systolic arterial hypotension, a decrease from basal systolic blood pressure of at least 20%, or the appearance of signs of peripheral hypoperfusion [18].
Toxic shock-like presentation was characterized by signs of cardiovascular collapse requiring volume resuscitation and vasopressors, and requiring intensive care. MIS-C patients who did not meet the criteria for KD-like and toxic shock-like were classified as “patients without either KD or shock”.
Exclusion criteria were underlying cardiac diseases, signs and symptoms of acute respiratory distress syndrome, receiving extracorporeal membrane oxygenation therapy, and patients with malignancy who received chemotherapy. All patients were tested for the SARS-CoV-2 virus by nasopharyngeal swab polymerase chain reaction (PCR) and immunoglobulin G antibodies.
The control group consisted of 40 healthy controls admitted to our hospital’s outpatient clinics for evaluation of heart murmurs or chest pain with no structural and functional heart defects.
Clinical and Laboratory Evaluation
Demographic, clinical, and laboratory data including C-reactive protein (CRP), procalcitonin, interleukin-6 (IL-6), ferritin, fibrinogen, D-dimer, erythrocyte sedimentation rate (ESR, the highest value before IVIG treatment was obtained), troponin-I, and N-terminal pro-brain natriuretic peptide (NT-proBNP), the highest level during hospitalization were recorded prospectively.
Serial Echocardiography Protocol
We set three-time points for analysis: echocardiogram on the day of worst left ventricular systolic function (by 3D LVEF) during the hospitalization (Echo-1); echocardiogram on the day of first improved left ventricular systolic function (Echo-2); echocardiogram on the day of discharge (Echo 3).
Echocardiographic evaluation was performed daily until the first improvement in systolic function during the acute phase of the disease in patients with impaired LV functions. Thereafter patients were evaluated on different days until discharge by echocardiography.
Echocardiographic evaluation was performed daily during the first 3 days of hospitalization in patients with preserved LV functions. Thereafter echocardiographic evaluation was performed on alternate days in the first week. In patients with preserved left ventricular systolic function by 3D LVEF; Echo-1 presents the worst LVEF value measured of during the first 3 days from hospitalization, Echo-2 presents the first better 3D LVEF value measured after Echo-1.
Echocardiographic evaluation was not done during the plasmapheresis procedure.
Echocardiographic Assessment
Transthoracic echocardiography was performed using an EPIQ 7C echocardiography system (Philips Healthcare, Andover, MA, USA). All subjects were evaluated by two experienced pediatric cardiologists. All traditional echo modes: M-mode, 2D, color, pulsed and continuous-wave Doppler (PW-CW), and tissue Doppler were used.
Standard echocardiographic measurements were made according to American Society of Echocardiography guidelines [19] including, fractional shortening (FS) calculated using M-mode, the early (E) and late (A) mitral inflow peak velocities by spectral Doppler, tricuspid annular plane systolic excursion (TAPSE) by M-mode, the systolic and early diastolic basal septal and lateral mitral annular peak velocities (e′) and systolic and diastolic lateral tricuspid annular peak velocity by tissue Doppler and z scores were calculated where appropriate [20]. Averaged E/e′ ratio was calculated as; mitral inflow peak E/mean e′ (lateral e′ + septal e′/2). The mitral annular plane systolic excursion (MAPSE) was measured by using the standard M-mode technique [21].
We divided MIS-C patients into 2 groups according to 3D LVEF at the time of echo-1: An impaired LVEF (Group 1; LVEF < 55%), a preserved LVEF (Group 2; LVEF ≥ 55%).
Three‐Dimensional Echocardiography Protocol
The 3D images were obtained immediately following the completion of the 2D protocol. The full‐volume real-time assessment was performed by 3D datasets acquired by combining 4 ECG‐gated pyramidal subvolumes. Images were acquired over four cardiac cycles using a matrix array ultrasound transducer (X5‐1 transducer, Philips Healthcare). Measurements were taken offline using a semi‐automated algorithm by QLAB (version 15.0, Philips Healthcare). From an apical full‐volume acquisition, frames for EDV and ESV measurement were identified. Endocardial contour tracing was performed with a semi‐automatic border detection algorithm and manually adjusted if needed as follows: After identifying the apex and mitral annulus on 4‐chamber and 2‐chamber slices, a preconfigured ellipse was automatically fitted to the endocardial borders of each frame and manually adjusted as required in appropriate planes. EF was derived from the EDV and ESV measurements obtained by the 3D volumes [22, 23].
Coronary Artery Assessment
The diagnosis of coronary artery abnormality was made by 2 experienced pediatric cardiologists after the echocardiographic images were examined. Echocardiographic evaluation of the coronary arteries was performed by a measurement made from inner edge to inner edge and excluding the points of branching, which may have normal focal dilation according to the 2017 American Heart Association (AHA) guideline for KD [17]. Coronary artery z scores were obtained by using the Boston z score system. Coronary artery abnormalities were classified as follows: normal < 2, dilatation ≥ 2 to < 2.5, aneurysm ≥ 2.5. Also, ectasia was defined as dilation of the coronary arteries without the presence of a segmental aneurysm [17, 24].
Reproducibility Analysis
To determine inter-observer and intra-observer variability for the measurement of 3D LVEF, 25 patients from the study and 25 controls were evaluated. For intra-observer reproducibility, the observer chose the best stored cardiac cycles for each patient. Then, the observer performed two measurements on two different days (at least 14 days between two measurements) on the same cycles. Two observers, unaware of the patients’ previous echocardiography results, performed the measurement of 3D LVEF for inter-observer reproducibility.
Statistical Analysis
Statistical Package for the Social Science (SPSS_17.0.1 for Windows; SPSS Inc) was used for statistical analysis. The two groups were compared using the independent sample t-test and Mann–Whitney U test depending on the type distribution of the data. The chi-square (χ2) test was used to compare categorical variables. Variables were tested for normality using the Shapiro–Wilk test. Repeated measures (RM) or Friedman analysis of variance (ANOVA) depending on whether parametric assumptions are met, were performed to evaluate changes of all echocardiographic parameters assessed on three different days. Wilcoxon signed rank-sum test for pairwise comparisons was used following Friedman’s analysis of variance. The independent 3 groups were assessed using the Kruskal–Wallis test followed by Tamhane’s T2 post hoc test for pairwise comparisons. The results for variables with normal distribution were reported as mean ± SD, while the nonnormally distributed parameters were reported as median (interquartile range). Categorical data are presented as numbers (n) and percentages (%). Correlation coefficients between 2 variables were calculated using Spearman’s rank-order correlation test. Intra- and inter-observer variability was analyzed according to the intraclass correlation coefficient (ICC) and the Bland–Altman method [25]. Statistical significance was defined as a two-tailed P value of < 0.05.
Results
Patient Characteristics
Fifty patients were diagnosed as MIS-C during the study period. Demographic data of the patient and control group are shown in Table 1. The patients and controls were similar in terms of age, gender, body weight, body mass index (BMI), and body surface area (BSA). Heart rate was significantly higher (p < 0.001), systolic and diastolic blood pressure were significantly lower in the MIS-C group compared to controls (p < 0.05).
Table 1.
General characteristics of the patients and controls
| MIS-C (Total) (n = 50) | Control (n = 40) | p value | |
|---|---|---|---|
| Age (years) | 10.1(7.3–14) | 10.5(7.6–13.9) | 0.66 |
| Male number, (%) | 28 (56) | 18 (45) | 0.3 |
| Body weight (kg) | 34.5(23.7–54) | 40(25–53) | 0.6 |
| BSA (m2) | 1.17(0.83–1.56) | 1.27(0.92–1.54) | 0.49 |
| BMI (kg/m2) | 18.21(16.59–20.62) | 19.03(16.82–21.8) | 0.34 |
| Heart rate (beats/min) | 120(100–138) | 85(78–95) | < 0.001* |
| Systolic BP (mm Hg) | 100(90–110) | 105(95–120) | 0.02* |
| Diastolic BP (mm Hg) | 60(51–64) | 65(60–75) | 0.02* |
BMI body mass index, BP blood pressure, BSA body surface area, MIS-C multisystem inflammatory syndrome in children
*p value < 0.05 Values are median (interquartile range) or number (%)
In this study, 5 (10%) patients were presented with KD-like, 15 (30%) patients with KDSS, 17 (34%) patients with toxic shock-like, and 13 (26%) patients without either KD or shock symptoms. Patients were treated with intravenous immunoglobulin (IVIG) (100%), corticosteroids (94%), aspirin (88%), anticoagulants (88%), inotropes/vasopressors (64%), colchicine (18%), interleukin 1 (IL-1) receptor antagonist (anakinra, 48%). Also, plasmapheresis was performed on 30% of patients. Respiratory support therapy included supplemental oxygen (26%), non-invasive mechanical ventilation (8%), and invasive mechanical ventilation (6%). The median duration of hospitalization was 15.5 days (interquartile range 12–19 days). Forty patients (80%) were admitted to the pediatric intensive care unit (PICU) and the median length of stay in the PICU was 7 days (IQR 3.25 to 9 days). The median total duration of hospitalization and duration of hospitalization in PICU was significantly longer in group 1 than group 2 [median 18 days (IQR 15 to 20) vs 13 days (IQR 10 to 16), p < 0.001; median 8 days (IQR 4 to 10) vs 2 days (IQR 0 to 5), p = 0.006, respectively]. The lowest EF was found at median 1.5 day (IQR 1–2 days) from admission to hospital in impaired LVEF group. Worst systolic cardiac function was recorded at median 1 day (IQR 1 to 2 days), and improvement of systolic cardiac function was determined at median 4 days (IQR 2 to 5 days), by 3D LVEF (Supplemental Table 1).
Laboratory Data
Immunoglobulin G test was positive and the polymerase chain reaction was negative for the SARS-CoV-2 virus in all MIS-C patients. Elevated acute phase reactants and lymphopenia were detected in all patients suggesting a hyperinflammatory state. Forty-three patients had positive serum troponin-I (> 45 ng/L) and elevated NT-pro BNP (> 500 ng/L) were detected in 48 patients. Supplemental Table 2 shows detailed laboratory results for all 50 patients.
Echocardiographic Findings
The comparison of echocardiographic evaluations performed at three different times during the hospitalization of the patients with each other and with the control group are shown in Table 2.
Table 2.
Comparison of serial conventional echocardiography findings in MIS-C with the control group
| MIS-C (n = 50) Echo (1) | MIS-C (n = 50) Echo (2) | MIS-C (n = 50) Echo (3) | Control (n = 40) | p value | |||||
|---|---|---|---|---|---|---|---|---|---|
| Echo (1) vs. Control | Echo (2) vs. Control | Echo (3) vs. Control | Echo (1) vs. Echo (2) | Echo (2) vs. Echo (3) | |||||
| MR > trivial | 25 (50) | 18 (36) | 9 (18) | 0 (0) | < 0.001* | < 0.001* | 0.005* | < 0.001* | < 0.001* |
| Pericardial effusion | 36 (72) | 32 (64) | 10 (20) | 0 (0) | < 0.001* | < 0.001* | 0.003* | 0.006* | < 0.001* |
| Pleural effusion | 26 (52) | 6 (12) | 0 (0) | 0 (0) | < 0.001* | 0.009* | – | < 0.001* | 0.009* |
| LV systolic function | |||||||||
| LVFS (%) | 30 (26–35) | 36 (31–38) | 38 (36–41) | 37 (34–41) | < 0.001* | 0.06 | 0.1 | < 0.001* | < 0.001* |
| 3D LVEF (%) | 52.8 ± 9.6 | 60.3 ± 4.8 | 65.1 ± 2.9 | 65.5 ± 4.5 | < 0.001* | < 0.001* | 0.36 | < 0.001* | < 0.001* |
| MAPSE (mm) | 10 (9–12) | 11.5 (11–13) | 14 (12.8–15.3) | 14 (13–16) | < 0.001* | < 0.001* | 0.16 | < 0.001* | < 0.001* |
| Lateral mitral s′ (cm/sn) | 8.2 ± 1.6 | 9.4 ± 1.5 | 11.2 ± 1.6 | 11.6 ± 1.8 | < 0.001* | < 0.001* | 0.09 | < 0.001* | < 0.001* |
| Septal mitral s′ (cm/sn) | 7.3 ± 1.4 | 8.3 ± 1.2 | 9 ± 1.2 | 9.2 ± 1.1 | < 0.001* | < 0.001* | 0.45 | < 0.001* | < 0.001* |
| LV diastolic function | |||||||||
| E/A ratio | 1.5 (1.19–2.13) | 1.5 (1.25–1.81) | 1.42 (1.22–1.8) | 1.8 (1.6–2.1) | 0.032* | 0.02* | < 0.001* | 0.95 | 0.35 |
| Lateral mitral e′(cm/sn) | 12.8 ± 2.2 | 13.6 ± 1.9 | 15.1 ± 2.2 | 17.4 ± 2.1 | < 0.001* | < 0.001* | < 0.001* | 0.001* | < 0.001* |
| Lateral mitral e′ z score | − 2.5 ± 0.99 | − 2.16 ± 1 | − 1.6 ± 1.09 | 0.002* | < 0.001* | ||||
| Septal mitral e′ (cm/sn) | 10.1 ± 1.6 | 10.3 ± 1.6 | 11.1 ± 1.3 | 12.9 ± 1.5 | < 0.001* | < 0.001* | < 0.001* | 0.7 | 0.006* |
| Septal mitral e′ z score | − 2.39 ± 0.83 | − 2.22 ± 0.8 | − 1.87 ± 0.68 | 0.2 | 0.009* | ||||
| Averaged E/e′ ratio | 8.6 (7.5–9.3) | 7.6 (7–8.2) | 7.1 (6.6–7.7) | 6.3 (5.8–7.1) | < 0.001* | < 0.001* | < 0.001* | < 0.001* | < 0.001* |
| Lateral/septal e′ ratio | 1.27 ± 0.14 | 1.32 ± 0.14 | 1.35 ± 0.14 | 1.35 ± 0.11 | 0.001* | 0.36 | 0.97 | 0.01 | 0.98 |
| Right ventricle | |||||||||
| TAPSE (mm) | 17 (15–19) | 18.5 (16.75–20) | 20 (18–21) | 20 (18–21) | < 0.001* | 0.16 | 0.48 | < 0.001* | < 0.001* |
| Tricuspid s′ (cm/sn) | 12.4 ± 1.8 | 13.3 ± 1.7 | 14.2 ± 1.4 | 14 ± 1.1 | < 0.001* | 0.03* | 0.36 | 0.001* | < 0.001* |
| Tricuspid e′ (cm/sn) | 15.6 ± 2.7 | 15.6 ± 2.4 | 16.3 ± 2.5 | 16.2 ± 1.4 | 0.21 | 0.15 | 0.94 | 0.98 | 0.2 |
Echo (1) presents the echo on the day of worst left ventricular function, Echo (2) presents the echo on the day of improved left ventricular function, Echo (3) presents the echo on the day of discharge. 3D LVEF three-dimensional left ventricular ejection fraction, LVFS left ventricular fractional shortening, MAPSE mitral annular plane systolic, MIS-C multisystem inflammatory syndrome in children, MR mitral regurgitation, TAPSE tricuspid annular plane systolic excursion,
*p value < 0.05 Values are presented as mean ± SD or median (interquartile range)
Echocardiography (echo-1) with the worst systolic left ventricular function in MIS-C patients, revealed 50% with mitral regurgitation (MR), 72% with pericardial effusion, and 52% with pleural effusion. None of the patients with pericardial effusion had signs of cardiac tamponade. Ten (20%) patients had small pericardial effusion and 9 (18%) patients had mild mitral regurgitation at discharge. On the Echo-1, all left and right ventricular systolic function parameters were significantly lower (p < 0.001), and as an indicator of LV diastolic dysfunction; E/A ratio, mitral lateral and septal e′ velocity, lateral/septal e′ ratio were significantly lower, and averaged E/e′ ratio was significantly higher (p < 0.05) in MIS-C patients compared to control.
Serial echocardiography revealed significant improvement in all left and right ventricular systolic function parameters (p < 0.001), also there was no significant difference between MIS-C patients and control on the discharge echocardiogram. Mitral lateral e′ velocity and averaged E/e′ ratio, evaluating left ventricular diastolic function, showed significant improvement in serial echocardiographic measurements (all p value < 0.05). Mitral lateral e′ velocity z scores on echo (1), echo (2), echo (3) were − 2.5 ± 0.99, − 2.16 ± 1, − 1.6 ± 1.09 respectively and mitral septal e′ velocity z scores on echo (1), echo (2), echo (3) were − 2.39 ± 0.83, − 2.22 ± 0.8, − 1.87 ± 0.68 respectively in MIS-C patients. Mitral lateral and septal e′ velocities z scores returned to the normal range at discharge in MIS-C patients. Nevertheless, compared to control, E/A ratio, mitral lateral and septal e' velocity were significantly lower, averaged E/e′ ratio was significantly higher, in MIS-C patients on all three echocardiographic evaluations (p < 0.05). There was no significant difference in tricuspid e′ velocity between MIS-C patients and control (p ≥ 0.05).
An echocardiogram based on worst 3D LVEF during the hospitalization revealed reduced LV systolic function in 26 (52%) patients (3D LVEF < 55%). In addition, three of these patients had 3D LVEF < 30% (supplemental video). However, M-mode-derived left ventricular FS (LVFS) < 30% was determined in 19 (38%) patients at the same echocardiographic evaluation. Significantly more patients had lower left ventricular systolic function with 3D LVEF measurement compared to M-mode FS (p < 0.05).
Comparison of coronary artery z scores in serial echocardiographic evaluations during hospitalization are shown in Table 3.
Table 3.
Comparison of serial echocardiographic coronary artery z scores in MIS-C patients
| Echo (1) | Echo (2) | Echo (3) | p value (echo1 vs. echo 2) | p value (echo2 vs. echo 3) | |
|---|---|---|---|---|---|
| LMCA Z score | − 0.37 ± 0.86 | − 0,6 ± 0.76 | − 0.83 ± 0.65 | 0.014* | 0.006* |
| LAD Z score | − 0.61 ± 0.92 | − 0.88 ± 0.71 | − 1.1 ± 0.63 | 0.004* | 0.002* |
| RCA Z score | − 0.04 ± 0.16 | − 0.13 ± 0.14 | − 0.48 ± 0.11 | 0.98 | 0.09* |
LAD left anterior descending, LMCA left main coronary artery, RCA right coronary artery, other abbreviations as in Table 2
*p value < 0.05 Values are presented as mean ± SD
Coronary artery abnormality was detected in only 2 of the MIS-C patients (coronary ectasia; z score: 2.53 and 2.6 in the right coronary artery on the Echo-1). Both coronary artery ectasia resolved at discharge. Except for this, no segmental aneurysms and progressive coronary lesions were detected in the MIS-C group. Serial echocardiographic evaluations of coronary arteries revealed gradually significant decreasement in left main coronary artery (LMCA) and left anterior descending (LAD) z scores (all p value < 0.05). While there was no significant change in right coronary artery (RCA) z score at the first and second echocardiographic evaluation (p ≥ 0.05), a statistically significant decrease was detected between second and third measurements (p < 0.05).
Echocardiographic findings in preserved and reduced ejection fraction groups in echo-1, on MIS-C and comparison with the control group are shown in Table 4.
Table 4.
Echocardiographic findings in preserved and reduced ejection fraction Groups in MIS-C and comparison with the control group
| MIS-C (ECHO-1) | Control | P value | ||||
|---|---|---|---|---|---|---|
| Group 1 (3D LVEF < %55) (n = 26) | Group 2 (3D LVEF ≥ %55) (n = 24) | (n = 40) | < % 55 vs. ≥ % 55 | < % 55 vs. Control | ≥ % 55 vs. Control | |
| Demographics | ||||||
| Age (years) | 10.9 (8.9–14) | 8.3 (4.5–13) | 10.5 (7.6–13.9) | 0.27 | 0.9 | 0.5 |
| Male number, (%) | 14 (54) | 14 (58) | 18 (45) | 0.9 | 0.65 | 0.44 |
| Body weight (kg) | 34.5 (30–54.3) | 31.5 (17.5–53) | 40 (25–53) | 0.39 | 0.89 | 0.52 |
| BSA (m2) | 1.17 (1.03–1.57) | 1.07 (0.66–1.54) | 1.27 (0.92–1.54) | 0.35 | 0.9 | 0.42 |
| BMI (kg/m2) | 18.2 (16.7–21.6) | 18.3 (16–20.4) | 19.1 (16.8–21.8) | 0.67 | 0.9 | 0.39 |
| Heart rate (beats/min) | 122 (104–136) | 118 (98–138) | 85 (78–95) | 0.9 | < 0.001* | < 0.001* |
| Systolic BP (mm Hg) | 99 (88–110) | 100 (90–110) | 105 (95–120) | 0.97 | 0.10 | 0.22 |
| Diastolic BP (mm Hg) | 59 (50–64) | 60 (58–68) | 65 (60–75) | 0.79 | 0.008* | 0.068 |
| MR > trivial | 22 (85) | 3 (13) | 0 (0) | < 0.001* | < 0.001* | < 0.001* |
| Pericardial effusion | 23 (88) | 13 (50) | 0 (0) | 0.007* | < 0.001* | < 0.001* |
| Pleural effusion | 18 (69) | 8 (33) | 0 (0) | 0.011* | < 0.001* | < 0.001* |
| LV systolic function | ||||||
| 3D LVEF | 47 (42–52) | 60 (57–63) | 65 (62–69) | < 0.001* | < 0.001* | < 0.001* |
| LV FS (%) | 26 (23–27) | 36 (32–38) | 37 (34–41) | < 0.001* | < 0.001* | 0.91 |
| MAPSE (mm) | 9 (8–10) | 11 (10–13) | 14 (13–16) | < 0.001* | < 0.001* | < 0.001* |
| Lateral mitral s′ (cm/sn) | 7 (6.4–8) | 9.5 (8.2–10.4) | 11.8 (10.7–12.8) | < 0.001* | < 0.001* | < 0.001* |
| Septal mitral s′ (cm/sn) | 5.9 (5.5–8.1) | 8 (7.1–8.8) | 9.3 (8.3–10.1) | 0.002* | < 0.001* | < 0.001* |
| LV diastolic function | ||||||
| E/A ratio | 2 (1.4–2.5) | 1.3 (1.1–1.5) | 1.8 (1.6–2.1) | 0.002* | 0,89 | 0,14 |
| Lateral mitral e′(cm/sn) | 12.1 (10.7–13.3) | 13.5 (12.1–14.7) | 17.2 (15.8–19.1) | 0.014* | < 0.001* | < 0.001* |
| Lateral mitral e′ z score | − 3 (− 3.38 to − 2.38) | − 2.23 (− 2.60 to − 1.37) | 0.001 | |||
| Septal mitral e′ (cm/sn) | 10 (8.9–10.5) | 10.1 (9–12.1) | 12.9 (11.5–14.1) | 0.27 | < 0.001* | < 0.001* |
| Septal mitral e′ z score | − 2.5 (− 3.04 to − 2.31) | − 2.21 (− 2.72 to − 1.42) | 0.039 | |||
| Averaged E/e′ ratio | 8.9 (8.5–9.6) | 7.9 (7.2–8.7) | 6.3 (5.8–7.1) | 0.001* | < 0.001* | < 0.001* |
| Lateral/septal e′ ratio | 1.23 (1.18–1.33) | 1.27 (1.18–1.41) | 1.33 (1.29–1.43) | 0.22 | 0.001* | 0.34 |
| Right ventricle | ||||||
| TAPSE (mm) | 16 (15–18) | 18 (16–21) | 19 (17–21) | 0.06 | < 0.001* | 0.14 |
| Tricuspid s′ (cm/sn) | 11.5 (10.8–12.9) | 13.1 (12–14) | 14.1 (13.2–14.8) | 0.05 | < 0.001* | 0.054 |
| Tricuspid e′ (cm/sn) | 14.8 (13–17.6) | 15 (13.3–17.4) | 16.3 (15.4–17.3) | 0.9 | 0.69 | 0.67 |
Reduced (group 1) and preserved (group 2) EF groups and controls were similar in terms of age, gender, body weight, BMI, and BSA. Diastolic blood pressure was significantly lower in group 1 compared to the control. Heart rates were significantly higher in group 1 and group 2 compared to controls (p < 0.05). More patients in group 1 had MR, pericardial effusion, and pleural effusion, compared to group 2 (p values; < 0.001, 0.007, 0.011 respectively).
Left Ventricle Systolic Function
All left ventricular systolic function parameters were significantly lower in group 1 compared to the control and group 2 (p < 0.05). LVFS (median 36% vs. 37%; p ≥ 0.05) values were lower in group 2 compared to control, but it was not statistically significant. The other LV systolic function parameters were significantly lower in group 2 compared to control (Table 4).
Left Ventricle Diastolic Function
E/A ratio (median 2 vs. 1.3; p = 0.002) and the average E/e′ ratio (median 8.9 vs 7.9; p = 0.001) were significantly higher in group 1 compared to group 2. When both groups were compared with control, E/A ratio was not significantly different, but average E/e′ ratio was significantly higher in MIS-C patients. Lateral e′ and septal e′ velocities were significantly lower in both groups compared to control (p < 0.001). Also lateral and septal e′ z scores in group 1 were significantly lower than group 2 (p values < 0.05) and median z scores were lower than − 2 in both groups. Lateral e′ z scores in group 1 and group 2 were median − 3 (IQR: − 3.38 to − 2.38), − 2.23 (IQR − 2.60 to − 1.37), respectively, and septal e′ z scores in group 1 and group 2 were median − 2.5(IQR − 3.04 to − 2.31), 2.21(IQR − 2.72 to − 1.42), respectively (Table 4).
Right Ventricle Function
TAPSE (median 16 vs. 18 mm; p ≥ 0.05) and tricuspid s′ velocities (11.5 vs. 13.1 cm/s; p ≥ 0.05) in group 1 were lower than group 2, but it was not statistically significant. These values were significantly lower in group 1 compared to the control (p < 0.001), but there was no significant difference between group 2 and control. There was no significant difference in tricuspid e′ velocity among all groups (p ≥ 0.05).
Echocardiographic findings at discharge in group 1 and group 2 and comparison with the control group are shown in Table 5.
Table 5.
Echocardiographic findings at discharge in preserved and reduced ejection fraction groups in MIS-C and comparison with the control group
| MIS-C- ECHO (3) | Control | p value | ||||
|---|---|---|---|---|---|---|
| Group 1 (3D LVEF < %55) (n = 26) | Group 2 (3D LVEF ≥ %55) (n = 24) | (n = 40) | < %55 vs. ≥ %55 | < %55 vs. Control | ≥ %55 vs. Control | |
| LV systolic function | ||||||
| 3D LVEF | 64.5 (63–66) | 66.5(65–69) | 65( 62–69) | 0.07 | 0.71 | 0.59 |
| LV FS (%) | 38 (36–40) | 39 (37–41) | 37 (34–41) | 0.3 | 0.8 | 0.08 |
| MAPSE (mm) | 14 (12–16) | 14 (12–15) | 14 (13–16) | 0.33 | 0.07 | 0.9 |
| Lateral mitral s′ (cm/sn) | 11.2 (10.5–12.2) | 11.1 (10.2–12) | 11.8 (10.7–12.8) | 0.26 | 0.06 | 0.75 |
| Septal mitral s′ (cm/sn) | 8.9 (8.3–9.6) | 9.2 (8.5–9.8) | 9.3 (8.3–10.1) | 0.64 | 0.64 | 0.95 |
| LV diastolic function | ||||||
| E/A ratio | 1.3 (1.2–1.6) | 1.5 (1.4–1.8) | 1.8 (1.6–2.1) | 0.48 | 0.001* | 0.004* |
| Lateral mitral e′ (cm/sn) | 13.5 (12.1–16.2) | 16 (15–16.9) | 17.2 (15.8–19.1) | 0.006* | < 0.001* | 0.01* |
| Lateral mitral e′ z score | − 2.28 (− 2.73 to − 1.14) | − 1.19 (− 1.59 to − 0.44) | < 0.001* | |||
| Septal mitral e′ (cm/sn) | 11 (9.7–12) | 11.3 (10.6–12.3) | 12.9 (11.5–14.1) | 0.13 | < 0.001* | < 0.001* |
| Septal mitral e′ z score | − 2.08 (− 2.63 to − 1.63) | − 1.56 (− 2.09 to − 1.2) | 0.012* | |||
| Averaged E/e′ ratio | 7.4 (7–8) | 6.9 (6.5–7) | 6.3 (5.8–7.1) | 0.005* | < 0.001* | 0.07 |
| Lateral/septal e′ ratio | 1.33 (1.21–1.39) | 1.4 (1.32–1.47) | 1.33 (1.29–1.43) | 0.15 | 0.63 | 0.47 |
| Right ventricle | ||||||
| TAPSE (mm) | 20 (18–21) | 20 (18–22) | 19 (17–21) | 0.81 | 0.96 | 0.94 |
| Tricuspid s′ (cm/sn) | 14 (13–15) | 14.2 (13.9–15.4) | 14.1 (13.2–14.8) | 0.98 | 0.93 | 0.7 |
| Tricuspid e′ (cm/sn) | 15.6 (13.8–18) | 17 (15–18) | 16.3 (15.4–17.3) | 0.25 | 0.7 | 0.52 |
There was no difference between groups in left ventricular systolic function parameters. E/A ratio was found to be significantly lower in group 1 and group 2 compared to the control (median 1.3 vs. 1.8, p = 0.001; 1.5 vs. 1.8, p = 0.004, respectively). The averaged E/e′ ratio was still significantly higher in group 1 than group 2 and control (median 7.4 vs. 6.9, p = 0.005; 7.4 vs 6.3, p < 0.001, respectively). Mitral lateral e' velocity was significantly lower in group 1 than group 2 (median 13.5 vs 16, p = 0.006) also it was significantly lower in group 1 and group 2 compared to control (median 13.5 vs 17.2, p < 0.001; 16 vs 17.2, p = 0.01, respectively). Also lateral and septal e′ z scores in group 1 were significantly lower than group 2 (p values < 0.05) and median z scores were lower than − 2 in group 1. Lateral e′ z scores in group 1 and group 2 were median − 2.28 (IQR − 2.73 to − 1.14), − 1.19 (IQR − 1.59 to − 0.44), respectively, and septal e′ z scores in group 1 and group 2 were median − 2.08 (IQR − 2.63 to − 1.63), − 1.56 (IQR − 2.09 to − 1.2), respectively. (Table 5).
There was no difference in right ventricular systolic and diastolic parameters between all groups at discharge.
LVFS and 3D LVEF showed moderate negative correlation with troponin-I, NT-pro BNP, and ferritin (r value for troponin-I: − 0.63 and − 0.68, respectively; r value for NT-pro BNP: − 0.52 and − 0.57, respectively; r value for ferritin: − 0.5, and − 0.51, respectively). LVFS and 3D LVEF showed weak negative correlation with procalcitonin, CRP, and IL6 (r value for procalcitonin: − 0.44 and − 0.47, respectively; r value for CRP: − 0.34 and − 0.31, respectively; r value for IL6: − 0.29 and − 0.33, respectively) (Table 6).
Table 6.
Correlation between left ventricular systolic function and cardiac markers and acute phase reactants
| LVFS | 3D LVEF | |||
|---|---|---|---|---|
| r | p | r | p | |
| Troponin-I | − 0.63 | < 0.001** | − 0.68 | < 0,001** |
| NT-proBNP | − 0.52 | < 0.001** | − 0.57 | < 0.001** |
| CRP | − 0.34 | 0.017* | − 0.31 | 0.029* |
| Ferritin | − 0.5 | < 0.001** | − 0.51 | < 0.001** |
| IL6 | − 0.29 | 0.04* | − 0.33 | 0.02* |
| Procalcitonin | − 0.44 | 0.001** | − 0.47 | 0.001** |
r correlation coefficients, LVFS left ventricular fractional shortening, NT-proBNP N-terminal pro-brain natriuretic peptide, CRP C-reactive protein, IL6 interleukin-6, 3D
*Correlation is significant at the 0.05 level, 3D LVEF three-dimensional left ventricular ejection fraction, **Correlation is significant at the 0.01 level,
Compared with preserved LVEF group, impaired LVEF group had significantly higher troponin-I (median 658 vs. 65 ng/L; p < 0.001), NT-pro BNP (median 14,233 vs. 1824 ng/L; p = 0.001), procalcitonin (median 10.9 vs. 2.1 µg/L; p = 0.009), and ferritin levels (median 1234 vs.308 µg/L; p < 0.03) (Table 7).
Table 7.
Comparison of laboratory parameters of preserved and impaired LVEF groups
| MIS-C | p value | ||
|---|---|---|---|
| Group 1 (3D LVEF < %55) (n = 26) | Group 2 (3D LVEF ≥ %55) (n = 24) | ||
| Troponin-I (ng/L) < 45 | 658 (236–1819) | 65 (43–410) | < 0.001* |
| NT-pro BNP (ng/L) < 125 | 14,233 (43–28,859) | 1824 (1065–7621) | 0.001* |
| CRP (mg/l) < 5 | 211 (176–224) | 190 (149–217) | 0.23 |
| Procalcitonin (µg/L) | 10.9 (3.3–27.9) | 2.1 (0.6–13.8) | 0.009* |
| IL6 (pg/mL) < 2.0 | 250 (98–660) | 132 (80–256) | 0.12 |
| ESR (mm/hr) | 70 (52–106) | 69 (41–91) | 0.44 |
| Ferritin (µg/L) (22–322) | 1234 (387–1902) | 308 (185–887) | 0.003* |
| Fibrinogen (g/L) (1.7–4.2) | 6.2 (4.6–7.6) | 5.8 (4.7–6.3) | 0.3 |
| D-dimer (mg/L) < 0.55 | 4.5 (3–6.3) | 2.9 (1.7–12.7) | 0.45 |
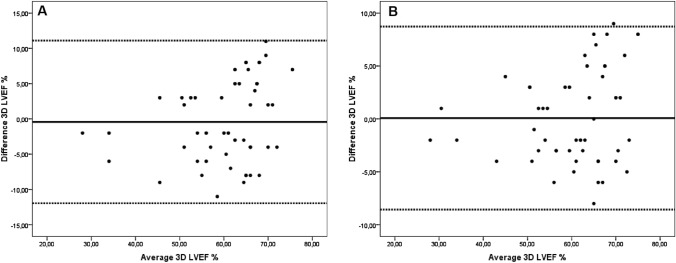
Intra-observer and inter-observer correlations were good for 3D LVEF measurements. Intraclass correlation coefficients are 0.91 for inter-observer and 0.95 for intra-observer measurements. Bland–Altman analysis for 3D LVEF showed no difference between observers or remeasurements with a single observer (Fig. 1).
Fig. 1.
A Bland–Altman plot showing inter-observer variability for measurement of three-dimensional left ventricular ejection fraction (3D LVEF %). B Bland–Altman plot showing intra-observer variability for measurement of 3D LVEF (%)
Discussion
In this study, we reported the traditional, tissue Doppler and 3D echocardiographic findings and changes during the hospitalization, in MIS-C patients. According to this study, ventricular dysfunction was the most common cardiac finding in MIS-C patients. Coronary artery involvement was found in a small number of patients in the acute phase of the disease.
In all MIS-C patients, left ventricular systolic and diastolic dysfunction and right ventricular systolic dysfunction were present in echocardiography performed when the left ventricular systolic function was the worst. In a previous study, similar findings were obtained using myocardial deformation analysis in addition to conventional echocardiographic methods in an MIS-C cohort [26]. Also, left and right ventricular systolic dysfunction was commonly reported in the MIS-C series [1, 4, 7, 27, 28].
A recent study evaluating echocardiographic findings in MIS-C patients reported no significant difference in LVEF and LVFS, in preserved LVEF group compared to the control [26]. However, in our study left ventricular systolic function parameters MAPSE, mitral lateral-septal s' velocities, and 3D LVEF were found to be significantly lower in the group with 3D LVEF ≥ 55%, than the control. In our opinion, this difference is the result of a relatively large MIS-C cohort in our study. Also, LV diastolic dysfunction was detected in both groups at the echocardiography on the day of the worst left ventricular functions. These results show that there are subtle and subclinical myocarditis findings in the group with preserved LVEF consistent with the study of Matsubara et al. [26] that showed distinct dysfunction in systolic and diastolic myocardial deformation parameters in preserved LVEF group.
All left ventricular systolic function parameters were improved at discharge in both impaired and preserved LVEF groups in our study. LV diastolic dysfunction persisted at discharge, in the impaired LVEF group. Similarly, Matsubara D. et al. [26] showed persistence of left ventricular diastolic dysfunction in early follow-up of MIS-C patients. Also, in the same study, right ventricular systolic dysfunction was detected in MIS-C patients with reduced LVEF. Consistent with these findings, in our study, right ventricular systolic function parameters decreased in the impaired LVEF group during the worst clinical period. However, there was improvement at discharge. On contrary, there was no right ventricular systolic dysfunction in preserved LVEF group.
Valverde et al. [7] reported pericardial effusion with a rate of 27.9% and MR with a rate of 42.5% in a large European cohort. Our MIS-C patients had a similar rate of MR (50%) but a higher rate of pericardial effusion (72%). In addition, MR and pericardial effusion rates were higher in the reduced LVEF group compared to preserved LVEF. Although systolic functions improved and laboratory parameters returned to normal in all patients, 20% of them had small pericardial effusion and 18% of them had mild MR at discharge. As reported previously [26], pleural effusion improved during hospitalization in all 26 patients in our study.
Coronary artery dilation and aneurysms have been described in MIS-C patients, however, the pathologic mechanism has not been elucidated [1, 27–29]. Although the incidence of coronary artery abnormalities in MIS-C varied significantly between reports, most of the large series reported coronary changes in 8–24% of patients [7, 27, 28]. Coronary artery ectasia was detected in only 2 patients (4%) in our cohort (z score: 2.53 and 2.6 in the right coronary artery), and they recovered until discharge. All patients were given IVIG in this study. However, we cannot speculate whether the use of IVIG or other treatments had a protective effect on coronary artery involvement like Kawasaki disease. There are no data about the mid- and long-term impact of MIS-C on coronary arteries. Therefore, MIS-C patients need long-term follow-up for coronary artery evolution, whether or not there is coronary artery involvement in the acute period. As emphasized in a previous study [26], we avoided using the term “prominent” coronary arteries, taking into account the phenomenon of transient coronary dilatation associated with underlying febrile illness. Even though pericoronary brightness is seen in echocardiography in most of the MIS-C patients, we did not include this terminology in our evaluation because it was removed in the 2017 American Heart Association (AHA) guideline for KD [17].
Despite the underlying mechanism of myocardial dysfunction in MIS‐C patients has not been completely clarified, the potential mechanism for myocardial injury is explained by dysregulated inflammatory response related to cytokine storm, similar to acute COVID‐19 infection. Cytokine storm is characterized by elevated inflammatory markers and elevated pro-inflammatory cytokines such as IL6. [28, 29]. Frequently elevated troponin (68–95%) and BNP or NT-Pro BNP levels (78–100%) and their association with the presentation of shock and LV dysfunction were reported in a meta-analysis evaluating cardiac involvement in MIS-C patients [30]. When all MIS-C patients were evaluated in this study, a weak-moderate correlation was found between cardiac markers, acute phase reactants, and 3D LVEF, LVFS. Moreover, when subgroup analysis was performed according to 3D LVEF, cardiac markers (troponin-I and NT-proBNP) and some acute phase reactants (procalcitonin and ferritin) were significantly higher in the impaired LVEF group. These findings show that as the inflammation increases, the picture of myocarditis worsens, and both ventricles' function deteriorates.
Evaluation of the left ventricle with 3D echocardiography was reported as an accurate and reproducible method in children as well as many validation studies have been conducted on this subject. [15, 16, 31–33]. Measurement of left ventricular volumes and EF by 3DE was stated as a highly repeatable method in expert consensus document of European Association of Cardiovascular Imaging and the American Society of Echocardiography [23]. These findings have also been validated in neonates and infants with small LV volumes and high heart rates [34]. Our study showed that measurement of volume LVEF with 3D echocardiography is a valuable, feasible, and reproducible method for the evaluation of LV systolic function in MIS-C patients. In the cardiac evaluation of MIS-C patients, Theocharis P et al. [35] reported LV systolic dysfunction with 3D LVEF in more patients compared to M-mode FS although it was not statistically significant. In our study, which was conducted with a larger patient group, LV systolic dysfunction with 3D LVEF was detected in statistically significantly more patients than with Mode FS. We think that especially in patients with borderline left ventricular systolic dysfunction with LVFS, the volume LVEF measured by 3D echocardiography is more beneficial in demonstrating global low systolic dysfunction due to myocarditis associated wall motion abnormality in MIS-C patients.
Limitations
This was a single-center, prospective study with enough sample size. However, we are aware that MIS-C is a novel disease and our study has some limitations. One of the limitations of the study was the acquisition of three-dimensional echocardiographic images, especially in children < 5 years old. In addition, imaging was performed by pediatric cardiology specialists under strict infection-control regulations. Another controversial issue is the impact of high heart rate in MIS-C groups on the accuracy of the diastolic parameter assessment. Therefore we calculated z score for appropriate diastolic function parameters according to references. We did not adjust parameters for heart rate as reference data are presented without heart rate correction. As hemodynamic changes might occur during plasmapheresis treatment, echocardiographic images were not taken during this procedure. We demonstrated acute and subacute echo findings in MIS-C patients, but further studies are needed to evaluate the long-term cardiac involvement of these patients.
Conclusion
Our results showed that the most common echocardiographic findings in MIS-C patients are ventricular dysfunction associated with the myocarditis-like condition, pericardial effusion, MR, and rare coronary artery involvement in the acute phase of the disease. We think that a correlation between left ventricular systolic dysfunction and the severity of inflammation and cardiac biomarkers should be kept in mind in MIS-C patients. In this study, ventricular systolic function recovered during hospitalization, but LV diastolic dysfunction persisted in the reduced LVEF group. However, mid- and long-term follow-up studies are needed for both coronary artery involvement and left ventricular diastolic dysfunction in MIS-C patients.
Supplementary Information
Below is the link to the electronic supplementary material.
Author Contributions
1st author: Design and writing of the article with literature review. 2nd, 3rd, and 4th author: Critical revision of article. 5th, 6th, and 7th author: Data analysis. 8th and 9th author: Literature review and revision of manuscript. 10th and 11th author: Revision of the manuscript.
Funding
The authors and contributors received no funding during the production of this manuscript.
Declarations
Conflict of interest
Authors report no conflicts of interest.
Ethical Approval
The protocol was approved by the Local Ethics Committee of Ankara City Hospital. Informed written consent was obtained from the parents before enrollment.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Whittaker E, Bamford A, Kenny J, et al. Clinical characteristics of 58 children with a pediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2. JAMA. 2020;324(3):259–269. doi: 10.1001/jama.2020.10369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Riphagen S, Gomez X, Gonzalez-Martinez C, Wilkinson N, Theocharis P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet (Lond, Engl) 2020;395(10237):1607–1608. doi: 10.1016/S0140-6736(20)31094-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Verdoni L, Mazza A, Gervasoni A, et al. An outbreak of severe Kawasaki-like disease at the Italian epicentre of the SARS-CoV-2 epidemic: an observational cohort study. Lancet (Lond, Engl) 2020;395(10239):1771–1778. doi: 10.1016/S0140-6736(20)31103-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Belhadjer Z, Méot M, Bajolle F, et al. Acute heart failure in multisystem inflammatory syndrome in children in the context of global SARS-CoV-2 pandemic. Circulation. 2020;142(5):429–436. doi: 10.1161/CIRCULATIONAHA.120.048360. [DOI] [PubMed] [Google Scholar]
- 5.Ramcharan T, Nolan O, Lai CY, et al. Paediatric inflammatory multisystem syndrome: temporally associated with SARS-CoV-2 (PIMS-TS): cardiac features, management and short-term outcomes at a UK Tertiary paediatric hospital. Pediatr Cardiol. 2020;41(7):1391–1401. doi: 10.1007/s00246-020-02391-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheung EW, Zachariah P, Gorelik M, et al. Multisystem inflammatory syndrome related to COVID-19 in previously healthy children and adolescents in New York City. JAMA. 2020;324(3):294–296. doi: 10.1001/jama.2020.10374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Valverde I, Singh Y, Sanchez-de-Toledo J, et al. Acute cardiovascular manifestations in 286 children with multisystem inflammatory syndrome associated with COVID-19 infection in Europe. Circulation. 2021;143(1):21–32. doi: 10.1161/CIRCULATIONAHA.120.050065. [DOI] [PubMed] [Google Scholar]
- 8.CDC Health Alert Network. Multisystem inflammatory syndrome in children (MIS-C) associated with coronavirus disease 2019 (COVID-19). Available at: https://emergency.cdc.gov/han/2020/han00432.asp Accessed 21 June 2020
- 9.Royal College of Pediatrics and Child Health. Guidance: paediatric multisystem inflammatory syndrome temporally associated with COVID-19. Available at: https://www.rcpch.ac.uk/sites/default/files/2020-05/COVID-19-Paediatric-multisystem-%20inflammatory%20syndrome-20200501.pdf Accessed 16 Aug 2020.
- 10.Belot A, Antona D, Renolleau S, et al. SARSCoV- 2-related paediatric inflammatory multisystem syndrome, an epidemiological study, France, 1 March to 17 May 2020. Euro Surveill. 2020;25(22):2001010. doi: 10.2807/1560-7917.ES.2020.25.22.2001010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Godfred-Cato S, Bryant B, Leung J, et al. COVID-19-associated multisystem inflammatory syndrome in children United States, March–July 2020. MMWR Morb Mortal Wkly Rep. 2020;69(32):1074–1080. doi: 10.15585/mmwr.mm6932e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.World Health Organization. WHO (2021) WHO Coronavirus Disease (COVID-19) Dashboard. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports.
- 13.Worldometer COVID-19 coronavirus pandemic. Available at: https://www.worldometers.info/coronavirus/country/turkey/ Accessed 21 June 2021
- 14.World Health Organization Scientific Brief (2020). Multisystem inflammatory syndrome in children and adolescents with COVID-19 Available at: https://www.who.int/publications/i/item/multisystem-inflammatory-syndrome-in-childrenand-adolescents-with-covid-19. Accessed 21 June 2020.
- 15.Lu X, Xie M, Tomberlin D, et al. How accurately, reproducibly, and efficiently can we measure left ventricular indices using M-mode, 2-dimensional, and 3-dimensional echocardiography in children? Am Heart J. 2008;155(5):946–953. doi: 10.1016/j.ahj.2007.11.034. [DOI] [PubMed] [Google Scholar]
- 16.Balluz R, Liu L, Zhou X, Ge S. Real time three-dimensional echocardiography for quantification of ventricular volumes, mass, and function in children with congenital and acquired heart diseases. Echocardiography (Mount Kisco, N. Y.) 2013;30(4):472–482. doi: 10.1111/echo.12132. [DOI] [PubMed] [Google Scholar]
- 17.McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135(17):e927–e999. doi: 10.1161/CIR.0000000000000484. [DOI] [PubMed] [Google Scholar]
- 18.Kanegaye JT, Wilder MS, Molkara D, et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783–e789. doi: 10.1542/peds.2008-1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lopez L, Colan SD, Frommelt PC, et al. Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the Pediatric Measurements Writing Group of the American Society of Echocardiography Pediatric and Congenital Heart Disease Council. J Am Soc Echocardiogr. 2010;23(5):465–577. doi: 10.1016/j.echo.2010.03.019. [DOI] [PubMed] [Google Scholar]
- 20.Dallaire F, Slorach C, Hui W, et al. Reference values for pulse wave Doppler and tissue Doppler imaging in pediatric echocardiography. Circ Cardiovasc Imaging. 2015;8(2):e002167. doi: 10.1161/CIRCIMAGING.114.002167. [DOI] [PubMed] [Google Scholar]
- 21.Willenheimer R, Cline C, Erhardt L, Israelsson B. Left ventricular atrioventricular plane displacement: an echocardiographic technique for rapid assessment of prognosis in heart failure. Heart (British Cardiac Society) 1997;78(3):230–236. doi: 10.1136/hrt.78.3.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vaidya GN, Salgado BC, Badar F, John A, Stoddard MF. Two-dimensional strain echocardiography-derived left ventricular ejection fraction, volumes, and global systolic dyssynchrony index: Comparison with three-dimensional echocardiography. Echocardiography (Mount Kisco, N.Y.) 2019;36(6):1054–1065. doi: 10.1111/echo.14362. [DOI] [PubMed] [Google Scholar]
- 23.Simpson J, Lopez L, Acar P, et al. Three-dimensional echocardiography in congenital heart disease: an expert consensus document from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. Eur Heart J Cardiovasc Imaging. 2016;17(10):1071–1097. doi: 10.1093/ehjci/jew172. [DOI] [PubMed] [Google Scholar]
- 24.McCrindle BW, Li JS, Minich LL, et al. Coronary artery involvement in children with Kawasaki disease: risk factors from analysis of serial normalized measurements. Circulation. 2007;116(2):174–179. doi: 10.1161/CIRCULATIONAHA.107.690875. [DOI] [PubMed] [Google Scholar]
- 25.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet (Lond, Engl) 1986;1(8476):307–310. doi: 10.1016/S0140-6736(86)90837-8. [DOI] [PubMed] [Google Scholar]
- 26.Matsubara D, Kauffman HL, Wang Y, et al. Echocardiographic findings in pediatric multisystem inflammatory syndrome associated with COVID-19 in the United States. J Am Coll Cardiol. 2020;76(17):1947–1961. doi: 10.1016/j.jacc.2020.08.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Feldstein LR, Rose EB, Horwitz SM, et al. Multisystem inflammatory syndrome in U.S. children and adolescents. New Engl J Med. 2020;383(4):334–346. doi: 10.1056/NEJMoa2021680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dufort EM, Koumans EH, Chow EJ, et al. Multisystem inflammatory syndrome in children in New York State. N Engl J Med. 2020;383(4):347–358. doi: 10.1056/NEJMoa2021756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Alsaied T, Tremoulet AH, Burns JC, et al. Review of cardiac involvement in multisystem inflammatory syndrome in children. Circulation. 2021;143(1):78–88. doi: 10.1161/CIRCULATIONAHA.120.049836. [DOI] [PubMed] [Google Scholar]
- 30.Yasuhara J, Watanabe K, Takagi H, Sumitomo N, Kuno T. COVID-19 and multisystem inflammatory syndrome in children: a systematic review and meta-analysis. Pediatr Pulmonol. 2021;56(5):837–848. doi: 10.1002/ppul.2524528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ylänen K, Eerola A, Vettenranta K, Poutanen T. Three-dimensional echocardiography and cardiac magnetic resonance imaging in the screening of long-term survivors of childhood cancer after cardiotoxic therapy. Am J Cardiol. 2014;113(11):1886–1892. doi: 10.1016/j.amjcard.2014.03.019. [DOI] [PubMed] [Google Scholar]
- 32.Poutanen T, Ikonen A, Jokinen E, Vainio P, Tikanoja T. Transthoracic three-dimensional echocardiography is as good as magnetic resonance imaging in measuring dynamic changes in left ventricular volume during the heart cycle in children. Euro J Echocardiogr. 2001;2(1):31–39. doi: 10.1053/euje.2000.0054. [DOI] [PubMed] [Google Scholar]
- 33.Krell K, Laser KT, Dalla-Pozza R, et al. Real-time three-dimensional echocardiography of the left ventricle-pediatric percentiles and head-to-head comparison of different contour-finding algorithms: a multicenter study. J Am Soc Echocardiogr. 2018;31(6):702–711.e13. doi: 10.1016/j.echo.2018.01.018. [DOI] [PubMed] [Google Scholar]
- 34.Friedberg MK, Su X, Tworetzky W, Soriano BD, Powell AJ, Marx GR. Validation of 3D echocardiographic assessment of left ventricular volumes, mass, and ejection fraction in neonates and infants with congenital heart disease: a comparison study with cardiac MRI. Circ Cardiovasc Imaging. 2010;3(6):735–742. doi: 10.1161/CIRCIMAGING.109.928663. [DOI] [PubMed] [Google Scholar]
- 35.Theocharis P, Wong J, Pushparajah K, et al. Multimodality cardiac evaluation in children and young adults with multisystem inflammation associated with COVID-19. Eur Heart J Cardiovasc Imaging. 2021;22(8):896–903. doi: 10.1093/ehjci/jeaa212. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.