Abstract
Purpose of the Review
Idiopathic inflammatory myopathies are a heterogeneous group of autoimmune disorders. The presence of different autoantibodies allows clinicians to define distinct phenotypes. Antibodies against the melanoma differentiation-associated protein 5 gene, also called anti-MDA5 antibodies, are associated with a characteristic phenotype, the clinically amyopathic dermatomyositis with rapidly progressive interstitial lung disease. This review aims to analyze the different pharmacological options for the treatment of rapidly progressive interstitial lung disease in patients with anti-MDA5 antibodies.
Recent Findings
Evidence-based therapeutic recommendations suggest that the best initial approach to treat these patients is an early combination of immunosuppressive drugs including either glucocorticoids and calcineurin inhibitors or a triple therapy adding intravenous cyclophosphamide. Tofacitinib, a Janus kinase inhibitor, could be useful according to recent reports. High ferritin plasma levels, generalized worsening of pulmonary infiltrates, and ground-glass opacities should be considered predictive factors of a bad outcome. In this scenario, clinicians should consider rescue therapies such as therapeutic plasma exchange, polymyxin-B hemoperfusion, veno-venous extracorporeal membrane oxygenation, or even lung transplantation.
Summary
Combined immunosuppressive treatment should be considered the first-line therapy for patients with anti-MDA5 rapidly progressive interstitial lung disease. Aggressive rescue therapies may be useful in refractory patients.
Keywords: Rapidly progressive interstitial lung disease, Inflammatory myopathy, Anti-MDA5 antibody, Clinically amyopathic dermatomyositis, Immunosuppressive therapy
Introduction
Idiopathic inflammatory myopathies (IIM) are heterogeneous disorders of immunologic origin that affect multiple organs and systems, including the muscles, skin, lungs, and the joints [1••]. Around 70% of patients with IIM develop autoantibodies that can be categorized as myositis-specific antibodies (MSA) and myositis-associated antibodies (MAA). Myositis-associated antibodies can be detected in other autoimmune diseases whereas MSA are generally positive only in patients with myositis and mutually exclusive. The presence of MSA is of great support for the clinicians, allowing to differentiate between distinct myositis phenotypes [1••].
More than a third of patients with myositis have some type of interstitial lung disease (ILD) [2]. A proportion of those patients with myositis and ILD experience a rapidly progressive course. A new autoantibody, the anti-MDA5, described in 2005 by Sato et al. [3] and directed against the melanoma differentiated gene 5 protein (MDA5), also known as interferon-induced helicase C domain-containing protein 1 (IFIH1), is associated with the rapidly progressive course of the ILD.
Patients with anti-MDA5 autoantibodies show a clinical phenotype characterized by amyopathic dermatomyositis (CADM) with rapidly progressive interstitial lung disease (RP-ILD) [4–7]. Other features such as skin ulcers, polyarthritis, or overt clinical myopathy are also described in patients with anti-MDA5 autoantibodies, but RP-ILD is by far the most severe manifestation of the disease [8].
This review aims to analyze the standard pharmacological options for the treatment of the RP-ILD associated with anti-MDA5 autoantibodies. Moreover, we also discuss aggressive therapeutic alternatives reserved as rescue therapy, such as plasmapheresis, polymyxin B-hemoperfusion, extracorporeal membrane oxygenation (ECMO), or lung transplantation.
Physiopathology—role of MDA5, interferon, and innate immunity
The MDA5/IFIH1 protein participates in a well-recognized innate antiviral response, unchaining the production of type I interferons upon detection of viral double-stranded RNA at the cytosol. Otherwise, the gain of function mutations in IFIH1 has been associated with a distinct spectrum of autoimmune or autoinflammatory states [9, 10].
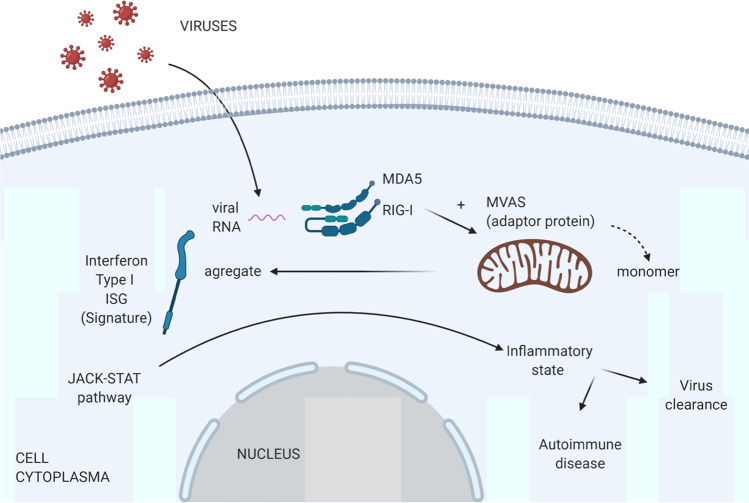
MDA5 is a member of the retinoic-acid inducible gene (RIG)-I-like helicases, which act as cytoplasmic RNA sensors. MDA5-RNA virus complexes trigger the activation of mitochondrial antiviral proteins (MAVS), which enable nuclear translocation of interferon regulatory factors (IRF) and induction of type I interferons and interferon-stimulated genes (ISGs). In turn, ISGs not only display a variety of antiviral actions inside infected cells but also boost inflammatory responses as a result of paracrine activation through the JAK-STAT signaling pathway [10]. Altogether, the upregulation of ISGs has been termed the interferon “signature” (Fig. 1).
Fig. 1.
Role of MDA5 in cells. MDA5/RIG-I molecules act as sensors of viral RNA activating the interferon pathway by means of MAVS aggregation from monomeric filaments. The next step is the activation of the JAK-STAT pathway generating an inflammatory state that helps to clear the virus but also can trigger an autoimmune disease. ISG, interferon-stimulated genes; MVAS, mitochondrial antiviral signaling; MDA5, melanoma differentiated gene 5 protein
Epidemiological data has revealed a geographical clustering and defined seasonal epidemics of anti-MDA5-positive CADM cases [11], which supports the role of environmental factors, possibly RNA virus infections, in the development of the disease.
Intriguingly, the pulmonary phenotype of zoonotic coronaviruses, including Middle East respiratory syndrome coronavirus (MERS-CoV), severe acute respiratory syndrome coronavirus (SARS-CoV), and SARS-CoV-2, the etiologic agent of the current COVID-19 pandemic, resemble that of patients with anti-MDA5 CADM-associated RP-ILD. Patients infected with these airborne transmitted viruses are at risk of developing severe pneumonia and adult distress respiratory syndrome (ADRS). Notably, not only the cytokine profiles but also chest computed tomography scans show striking similarities [12]. Moreover, due to its long size and 5′ capped structure, SARS-CoV-2 is predicted to primarily interact with the MDA5 cytosolic receptor, being the latter probably involved in the innate response against COVID-19 [13]. Nevertheless, none of our SARS-CoV-2-infected patients who developed ADRS tested positive for anti-MDA5 antibodies (personal communication).
The lack of MDA5/IFIH1 cytoplasmic RNA sensor in pangolins (mammalian order Pholidota), an intermediate host for different species of coronavirus and identified as a possible source of SARS-CoV-2, could prevent the innate immune response in these animals against the coronavirus and may explain why they tolerate the infection without developing a severe inflammatory state, as is the case in human beings [14, 15]. Altogether, these findings raise intriguing questions about the role of viruses and its consequences, the autoimmune/inflammatory reaction, in anti-MDA5 syndrome etiopathogenesis [16, 17].
Pharmacologic treatment and its rationale
Immunosuppressive drugs
The first line of treatment in patients with anti-MDA5 CADM patients with RP-ILD includes several types of immunosuppressive agents. A recent review focused on evidence-based therapeutic recommendations [18••] suggests that the best initial approach to manage these patients is a combined treatment using various immunosuppressive drugs, being the first options either a combination of glucocorticoids and a calcineurin inhibitor (cyclosporine or tacrolimus) or a triple therapy adding intravenous cyclophosphamide to the other two drugs.
This combined strategy is supported by several recent reports [19, 20, 21•], including a multicenter prospective study [21•] from Japan. In this study, a total of 29 adult Japanese patients with new-onset anti-MDA5-positive dermatomyositis and ILD were enrolled from 2014 to 2017 and treated with a combined immunosuppressive regimen of high-dose glucocorticoids, tacrolimus, and intravenous cyclophosphamide. The 6-month survival of the patients who received this combined therapy was significantly higher than the patients who received a step-up strategy starting with high-dose glucocorticoids followed by stepwise addition of other immunosuppressants (89% vs 33%; p < 0.0001).
These findings were replicated in a retrospective observational study [20], also from Japan, which included 11 patients with anti-MDA5-positive CADM and RP-ILD who were treated early with a combination of high-dose glucocorticoids, i.v. cyclophosphamide and tacrolimus, experiencing a significant improvement in their pulmonary forced vital capacity. Even though this study did not have a control group, no differences in mortality were observed when comparing patients with other dermatomyositis phenotypes (e.g., anti-synthetase or anti-TIF1γ). As a potential drawback of this approach, the authors alerted of frequent cytomegalovirus reactivations induced by the profound immunosuppression of the patients.
In those patients where calcineurin inhibitors are not tolerated (e.g., severe hypertension or difficult to manage diabetes), other immunosuppressants such as mycophenolate mofetil or biologic therapies such as the anti-CD20 humanized monoclonal antibody rituximab [22–24] should be considered.
Other drugs or biological therapies have also shown promising results, including basiliximab [25], an anti-CD25/sIL-2R monoclonal antibody, or tofacitinib [26, 27, 28•, 29–31] (a Janus kinase 1/3 inhibitor). Considering the proposed physiopathology of the anti-MDA5 syndrome, the JAK/STAT system is a particularly attractive therapeutic target in the anti-MDA5 syndrome, since not only type I and II interferons but also additional proinflammatory cytokines, including IL6, are involved in this pathway.
Specifically, two studies addressed the role of tofacitinib for the treatment of the RP-ILD in anti-MDA5-positive CADM patients. The first study [26] included a case series of 5 patients who received tofacitinib at a dose of 10 mg/day after failed to respond to a triple therapy with high-dose glucocorticoids, cyclosporine, and cyclophosphamide. The authors compared the outcomes of these 5 patients with 6 historical patients who had been treated with triple therapy without tofacitinib. Both groups of patients were comparable regarding major factors of poor prognosis, including high serum ferritin levels, worsening pulmonary infiltrates, and generalized ground-glass opacities. Of note, three of the tofacitinib group had a favorable response, while the rest, including all patients from the historical cohort, died. Alternatively, cytomegalovirus reactivation was constant in patients who received triple therapy and tofacitinib. The second study [28•], from China, was an open-label clinical study from a single-center evaluating the efficacy of tofacitinib at the early stages of anti-MDA5-positive CADM-associated ILD. From July 2017 to September 2018, a total of 18 consecutive patients were enrolled and received treatment mostly with glucocorticoids and tofacitinib (5 mg/12 h) and only occasionally with other immunosuppressants (2 patients received cyclosporine, and 1 mycophenolate mofetil). When compared with a historical control group of 25 patients treated with a step-wise immunosuppressive approach based on cyclosporine, mycophenolate mofetil, and/or cyclophosphamide, the survival at 6 months was significantly higher in the group treated with tofacitinib than in the historical control group. An issue to note in this study was that patients achieved a favorable outcome in the absence of severe immunosuppression, which helped to avoid infectious complications such as cytomegalovirus reactivation, or other fastidious opportunistic infections.
Genetic background in anti-MDA5 therapeutics
The recent identification of new myositis-associated genome-wide loci [32–34] may help to improve our capacity to identify potential targets for therapeutics. This could be the case of the gene product TYK2 which is targeted by tofacitinib, FAM167A-BLK targeted by nintedanib, or the truncated WDFY4 gene in Japanese patients, which enhances the MDA5-mediated nuclear-factor kappa B (NF-kB) activation in a calcineurin-dependent fashion.
How to identify the best therapeutic strategy?
The best strategy to achieve a good outcome in patients with anti-MDA5-positive CADM-associated RP-ILD seems to be to start a combined immunosuppressive therapy as soon as possible. It is nonetheless challenging to identify at the very onset of the disease those patients who are at risk of suffering an RP-ILD. As mentioned above, there are several predictive factors, including the presence of high ferritin plasma levels (approx. cutoff of 1000 ng/ml, NV < 200 ng/ml) [35, 36], worsening of the pulmonary infiltrates despite treatment, and generalized ground-glass opacities, which are considered as reliable poor prognosis markers [26]. Other factors such as the serologic levels of Krebs von den Lungen-6 (KL-6) or the titer of anti-MDA5 antibodies could also be of value, albeit they are currently mostly used in research settings [37, 38].
Thus, although there are reported early diagnosis strategies and therapeutical algorithms (see Figs. 2 and 3), it is still the treating clinician who has to decide whether or not to initiate a combined immunosuppressive therapy in an individual patient.
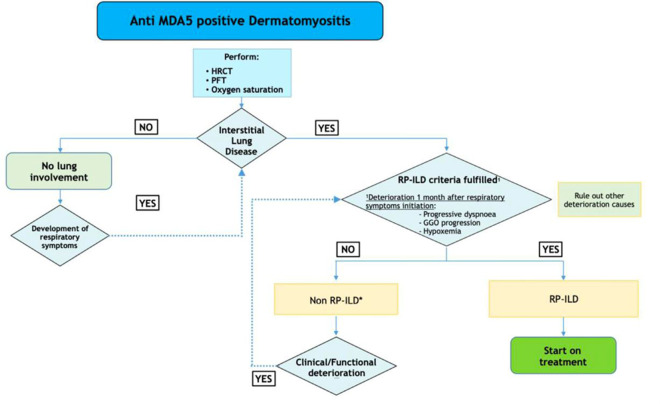
Fig. 2.
Diagnosis of RP-ILD in patients with anti-MDA5 antibodies. *Combined therapy with glucocorticoids and calcineurin antagonists is recommended especially if some risk factors are present (> 60 years old, hyperferritinemia > 500 nm/ml, C reactive protein > 1 mg/dl). HRCT, high-resolution CT scan; PFT, pulmonary function tests; RP-ILD, rapidly progressive interstitial lung disease; GGO, ground-glass opacity (see ref. [18••])
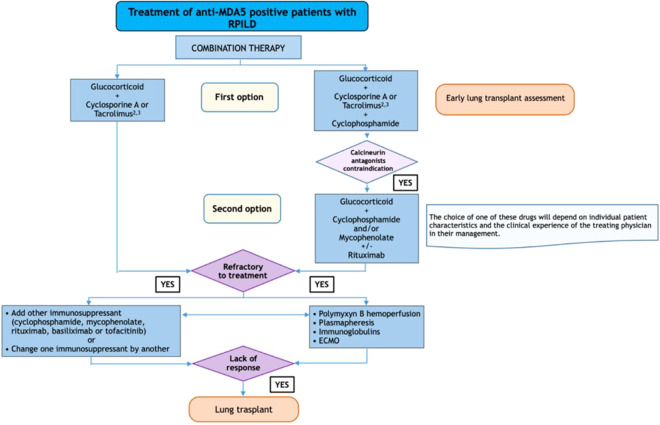
Fig. 3.
Treatment algorithm for RP-ILD patients with anti-MDA5 antibodies (see ref. [18••])
While the diagnosis of ILD relies on chest X-ray and/or high-resolution CT scans showing reticular opacities, ground-glass opacity or a honeycombing appearance, rapidly progressive ILD should be considered in cases of worsening of radiologic interstitial changes with progressive dyspnea and hypoxemia within 1 month after the onset of the respiratory symptoms [39, 40].
Pirfenidone and nintedanib, both drugs approved for the treatment of idiopathic pulmonary fibrosis [40], may be useful to some extent in the treatment of the anti-MDA5-positive CADM-associated RP-ILD patients. The first one, pirfenidone, was evaluated in a recent study from China which reported the outcome of 30 patients diagnosed with CADM-RP-ILD receiving 1800 mg/day in addition to conventional treatment with glucocorticoids and other immunosuppressive drugs. Although the drug had no impact on survival in the acute-fulminant forms, it was proven useful in the more subacute presentation. Thus, the administration of pirfenidone might play a role in improving the long-term outcome in those patients who survive [41]. Similarly, nintedanib, an intracellular inhibitor of tyrosine kinases, has demonstrated its utility not only in patients with idiopathic pulmonary fibrosis [42], but also in patients with ILD secondary to systemic sclerosis [43], and more importantly, in different diseases with ILD and a progressive course to lung fibrosis [44•]. In general, those patients who survive to the acute/fulminant form of ILD, but who may develop ILD fibrosis as a long-term manifestation, might benefit from those antifibrotic drugs. Multicenter prospective studies gathering a high number of patients may be necessary to determine the utility of these particular treatments in anti-MDA5 patients with RP-ILD.
Refractory patients
Most experts agree on defining a refractory patient as the one that does not respond to either of the two main modalities of therapy (prednisone plus calcineurin antagonists or prednisone plus cyclophosphamide plus calcineurin antagonists). Alternatively, the absence of response is defined when 1 week after the onset of the combination therapy the patient fulfills the following criteria: worsening of the respiratory symptoms (dyspnea), increasing alveolar-arterial O2 tension difference, newly emerging or expanding GGO/consolidation on chest imaging, increasing ferritin levels, and the personal impression of clinical worsening as assessed by the attending physicians. In this scenario, adding a new immunosuppressive drug (mycophenolate mofetil, rituximab, basiliximab, or tofacitinib) or switching one immunosuppressant for another are both reasonable options, emphasizing the use of the Janus kinase inhibitor tofacitinib according to the very latest studies, although the evidence is still scarce and of low quality.
Other non-pharmacologic alternative rescue therapies in these severe cases include using plasmapheresis, polymyxin B hemoperfusion, intravenous immunoglobulins, veno-venous extracorporeal membrane oxygenation (VV-ECMO), and lung transplantation.
Plasmapheresis
Also known as therapeutic plasma exchange, plasmapheresis (PMF) is a procedure in which blood passes through a device allowing to separate or remove some molecules from the plasma [45]. Its rationale in anti-MDA5-positive CADM RP-ILD patients is to remove the anti-MDA5 antibodies and to absorb or eliminate cytokines or other inflammatory molecules. The reported role of several cytokines, including the IL-6, IL-10, and IL-18 in the pathophysiology of MDA5-associated RP-ILD, supports the potential utility of plasmapheresis in these patients. Moreover, plasmapheresis can be used even if there is a concurrent infectious disease, which is not unusual in severely immunosuppressed patients. Even though a recent study about therapeutic recommendations in anti-MDA5 patients plasmapheresis was categorized as a level 3 of evidence (efficacy based in case reports and case series), it was recommended as a therapeutic option for refractory patients [18••].
During the last year, several reports reinforced the role of plasmapheresis in anti-MDA5-positive CADM RP-ILD patients. In brief, Shirakashi et al. from Japan retrospectively analyzed a cohort of 13 anti-MDA5-positive dermatomyositis patients with RP-ILD, all of them refractory to combined immunosuppressive therapy with glucocorticoids, calcineurin inhibitors, and intravenous monthly cyclophosphamide pulses. Five out of eight patients who received plasmapheresis as an add-on therapy survived, in comparison with none of the 5 patients who were treated only with combined immunosuppressive therapy without plasmapheresis [46]. Similarly, another study performed also in Japan [47•] reported 10 anti-MDA5-positive CADM RP-ILD patients with combined immunosuppressive therapy refractory disease. While six of them were additionally treated with plasmapheresis and were all alive at 1 year, only 25% of the ones who did not receive plasmapheresis survived.
Some other examples are supporting the possible benefit of plasmapheresis as an additional therapy in patients with refractory disease [48–51]. Nevertheless, the variety of local protocols and fluid replacement regimes used might yield different results and bias the evaluation of the efficacy of this procedure. Furthermore, transfusion-related acute lung injury following plasma exchange sessions is an infrequent but potentially threatening complication of this treatment [52].
Globally, plasmapheresis could be considered an effective adjuvant therapy in refractory anti-MDA5-positive CADM RP-ILD patients.
Polymyxin-B hemoperfusion
The rationale for using polymyxin B-immobilized fiber column direct hemoperfusion treatment is similar to plasmapheresis, functioning as an extracorporeal blood filter that absorbs blood molecules, including toxins, proteins, or antibodies, although the underlying mechanisms remain still unclear. This technique has been recommended for the treatment of refractory septic shock with multiorgan dysfunction and endotoxinemia [53] and also an alternative therapy for patients with acute respiratory distress syndrome [54]. Several case reports and case series [55–60] support using this technique as adjuvant therapy in anti-MDA5-positive CADM-associated RP-ILD patients refractory to combined immunosuppressive therapy. An expert panel included that polymyxin B-immobilized fiber column direct hemoperfusion may be an alternative rescue treatment in patients who do not respond to combined immunosuppressive drugs, either separately or sequentially with plasmapheresis [18••].
Veno-venous extracorporeal membrane oxygenation (ECMO)
Veno-venous extracorporeal membrane oxygenation (ECMO) is a life-support technique aimed to maintain blood oxygenation in patients with severe respiratory failure. Thus, it applies to anti-MDA5-positive CADM-associated RP-ILD patients refractory to combined immunosuppressive therapy, mostly as a bridge to a most definitive solution like lung transplantation or while waiting for a response to the immunosuppressive drugs. Currently, there is scarce data on the efficacy of VV-ECMO in this type of patients [61, 62•, 63–68, 69•, 70], and was considered successful only in a few patients, either because it helped to improve the clinical status of the patient or it allowed performing a lung transplantation [67, 68, 69•, 70].
Lung transplantation
According to recent data, patients diagnosed with connective tissue diseases and interstitial lung involvement are not worse candidates for lung transplantation than those with non-connective tissue diseases [71]. Notwithstanding this, our personal experience with lung transplantation in patients with anti-MDA5-associated RP-ILD has not been encouraging. A series of three patients underwent lung transplantation in our center and all three died during the first year of follow-up. Chronic rejection, graft dysfunction, and opportunistic infections were respectively responsible for these deaths (personal communication) [72]. It is noteworthy that none of the potential specific challenges related to transplant in patients with the anti-MDA5 syndrome, including subclinical myocardial involvement, muscle weakness-induced hypoventilation, gastroesophageal reflux disease, dysphagia, malignancy, and relapse of the underlying disease, was observed in our patients. Alternatively, the fact that the three of our patients underwent transplantation as an emergency surgery might have largely contributed to the poor outcomes [73].
There is an additional experience of lung transplants reported by other authors [67, 68, 69•, 70, 74] including a total of 7 patients, all of them survived. Thus, lung transplantation is a viable therapeutic option in refractory patients with anti-MDA5-associated RP-ILD. Referring those patients to centers with experience evaluating and managing lung transplants is strongly recommended [18••].
Conclusions
The pharmacological treatment of patients with anti-MDA5-positive CADM-associated RP-ILD is a medical challenge. Currently, early combined double or triple immunosuppressive therapy with glucocorticoids plus calcineurin inhibitors with or without cyclophosphamide is the preferred therapeutic option followed by the use of mycophenolate mofetil or rituximab when calcineurin antagonists are not feasible. In refractory patients, adding one more immunosuppressive drug (mycophenolate mofetil, rituximab, basiliximab, or tofacitinib) to the current therapy or changing one immunosuppressant for another are both reasonable options. Based on its mechanism of action and recent reports, the JAK inhibitor tofacitinib may be considered a new promising agent, although further data in RP-ILD are warranted to prioritize it in the current therapeutic algorithm. Finally, although the specific pathogenesis of the RP-ILD in the anti-MDA5 syndrome is still unknown, more data on the genetic background, environmental triggers, and risk and prognostic factors may contribute to identifying patient profiles that will hopefully help to define the best patient-tailored therapy.
Funding
This work was supported in part by the Instituto de Salud Carlos III and the European Regional Development Fund (ERDF) (grant number PI15/02100).
Declarations
Conflict of Interest
A. Selva-O’Callaghan declares that he has no conflict of interest. F. Romero-Bueno declares that he has no conflict of interest. E. Trallero-Araguás declares that he has no conflict of interest. A. Gil-Vila declares that he has no conflict of interest. J. C. Ruiz-Rodríguez declares that he has no conflict of interest. O. Sánchez-Pernaute declares that he has no conflict of interest. I. Pinal-Fernández declares that he has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Other CTD: Infammatory Myopathies and Sjogren’s
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References and Recommended Reading
- 1.Selva-O’Callaghan A, Pinal-Fernandez I, Trallero-Araguas E, Milisenda JC, Grau- Junyent JM, Mammen AL. Classification and management of adult inflammatory myopathies. Lancet Neurol. 2018;17:816–28. doi: 10.1016/S1474-4422(18)30254-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Connors GR, Christopher-Stine L, Oddis CV, Danoff SK. Interstitial lung disease associated with the idiopathic inflammatory myopathies: what progress has been made in the past 35 years? Chest. 2010;138(6):1464–1474. doi: 10.1378/chest.10-0180. [DOI] [PubMed] [Google Scholar]
- 3.Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum. 2005;52:1571–1576. doi: 10.1002/art.21023. [DOI] [PubMed] [Google Scholar]
- 4.Chen Z, Cao M, Plana MN, et al. Utility of anti-melanoma differentiation-associated gene 5 antibody measurement in identifying patients with dermatomyositis and a high risk for developing rapidly progressive interstitial lung disease: a review of the literature and a meta-analysis. Arthritis Care Res (Hoboken) 2013;65:1316–1324. doi: 10.1002/acr.21985. [DOI] [PubMed] [Google Scholar]
- 5.Labrador-Horrillo M, Martinez MA, Selva-O'Callaghan A, et al. Anti-MDA5 antibodies in a large Mediterranean population of adults with dermatomyositis. J Immunol Res. 2014;2014:290797. 10.1155/2014/290797. Epub 2014 Feb 4. PMID: 24741583; PMCID: PMC3987881. [DOI] [PMC free article] [PubMed]
- 6.Allenbach Y, Uzunhan Y, Toquet S, et al. Different phenotypes in dermatomyositis associated with anti-MDA5 antibody: study of 121 cases. Neurology. 2020;95:e70–e78. doi: 10.1212/WNL.0000000000009727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li Y, Gao X, Li Y, et al. Predictors and mortality of rapidly progressive interstitial lung disease in patients with idiopathic inflammatory myopathy: a series of 474 patients. Front Med (Lausanne) 2020;7:363. doi: 10.3389/fmed.2020.00363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hall JC, Casciola-Rosen L, Samedy LA, et al. Anti-melanoma differentiation-associated protein 5-associated dermatomyositis: expanding the clinical spectrum. Arthritis Care Res (Hoboken) 2013;65:1307–1315. doi: 10.1002/acr.21992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang DP, Zhang HY, Liao BW, et al. SRP54 Negatively regulates IFN-beta production and antiviral response by targeting RIG-I and MDA5 [published online ahead of print, 2020 Aug 7]. Virol Sin. 2020. 10.1007/s12250-020-00267-6. [DOI] [PMC free article] [PubMed]
- 10.Rice GI, Park S, Gavazzi F, et al. Genetic and phenotypic spectrum associated with IFIH1 gain-of-function. Hum Mutat. 2020;41:837–849. doi: 10.1002/humu.23975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Muro Y, Sugiura K, Hoshino K, Akiyama M, Tamakoshi K. Epidemiologic study of clinically amyopathic dermatomyositis and anti-melanoma differentiation-associated gene 5 antibodies in central Japan. Arthritis Res Ther. 2011;13:R214. doi: 10.1186/ar3547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gono T, Kaneko H, Kawaguchi Y, et al. Cytokine profiles in polymyositis and dermatomyositis complicated by rapidly progressive or chronic interstitial lung disease. Rheumatology (Oxford) 2014;53:2196–2203. doi: 10.1093/rheumatology/keu258. [DOI] [PubMed] [Google Scholar]
- 13.Gao H, Yao H, Yang S, Li L. From SARS to MERS: evidence and speculation. Front Med. 2016;10:377–382. doi: 10.1007/s11684-016-0466-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fischer H, Tschachler E, Eckhart L. Pangolins lack IFIH1/MDA5, a cytoplasmic RNA sensor that initiates innate immune defense upon coronavirus infection. Front Immunol. 2020;11:939. doi: 10.3389/fimmu.2020.00939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Giannini M, Ohana M, Nespola B, et al. Similarities between COVID-19 and anti-MDA5 syndrome: what can we learn for better care? Eur Respir J. 2020;in press. 10.1183/13993003.01618-2020. [DOI] [PMC free article] [PubMed]
- 17.Xu Q. MDA5 should be detected in severe COVID-19 patients [published online ahead of print, 2020 May 28]. Med Hypotheses. 2020;143:109890. [DOI] [PMC free article] [PubMed]
- 18.Romero-Bueno F, Diaz Del Campo P, Trallero-Araguás E, et al. Recommendations for the treatment of anti-melanoma differentiation-associated gene 5-positive dermatomyositis-associated rapidly progressive interstitial lung disease. Semin Arthritis Rheum. 2020;50:776–790. doi: 10.1016/j.semarthrit.2020.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Koga T, Kaieda S, Okamoto M, et al. Successful treatment of rapidly progressive unclassifiable idiopathic interstitial pneumonia with anti-melanoma differentiation-associated gene-5 antibody by intensive immunosuppressive therapy. Intern Med. 2018;57:1039–1043. doi: 10.2169/internalmedicine.9553-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matsuda KM, Yoshizaki A, Kuzumi A, et al. Combined immunosuppressive therapy provides favorable prognosis and increased risk of cytomegalovirus reactivation in anti-melanoma differentiation-associated gene 5 antibody-positive dermatomyositis. J Dermatol. 2020;47:483–489. doi: 10.1111/1346-8138.15274. [DOI] [PubMed] [Google Scholar]
- 21.Tsuji H, Nakashima R, Hosono Y, et al. Multicenter prospective study of the efficacy and safety of combined immunosuppressive therapy with high-dose glucocorticoid, tacrolimus, and cyclophosphamide in interstitial lung diseases accompanied by anti-melanoma differentiation-associated gene 5-positive dermatomyositis. Arthritis Rheumatol. 2020;72:488–498. doi: 10.1002/art.41105. [DOI] [PubMed] [Google Scholar]
- 22.Koichi Y, Aya Y, Megumi U, et al. A case of anti-MDA5-positive rapidly progressive interstitial lung disease in a patient with clinically amyopathic dermatomyositis ameliorated by rituximab, in addition to standard immunosuppressive treatment. Mod Rheumatol. 2017;27:536–540. doi: 10.3109/14397595.2015.1014140. [DOI] [PubMed] [Google Scholar]
- 23.Ogawa Y, Kishida D, Shimojima Y, Hayashi K, Sekijima Y. Effective Administration of Rituximab in Anti-MDA5 Antibody-Positive Dermatomyositis with Rapidly Progressive Interstitial Lung Disease and Refractory Cutaneous Involvement: A Case Report and Literature Review. Case Rep Rheumatol. 2017;2017:5386797. 10.1155/2017/5386797. Epub 2017 Oct 31. PMID: 29225988; PMCID: PMC5684540. [DOI] [PMC free article] [PubMed]
- 24.Tokunaga K, Hagino N. Dermatomyositis with rapidly progressive interstitial lung disease treated with rituximab: a report of 3 cases in Japan. Intern Med. 2017;56:1399–1403. doi: 10.2169/internalmedicine.56.7956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zou J, Li T, Huang X, Chen S, Guo Q, Bao C. Basiliximab may improve the survival rate of rapidly progressive interstitial pneumonia in patients with clinically amyopathic dermatomyositis with anti-MDA5 antibody. Ann RheumDis. 2014;73:1591–1593. doi: 10.1136/annrheumdis-2014-205278. [DOI] [PubMed] [Google Scholar]
- 26.Kurasawa K, Arai S, Namiki Y, et al. Tofacitinib for refractory interstitial lung diseases in anti-melanoma differentiation-associated 5 gene antibody-positive dermatomyositis. Rheumatology (Oxford) 2018;57:2114–2119. doi: 10.1093/rheumatology/key188. [DOI] [PubMed] [Google Scholar]
- 27.Kato M, Ikeda K, Kageyama T,et al. Successful Treatment for Refractory Interstitial Lung Disease and Pneumomediastinum With Multidisciplinary Therapy Including Tofacitinib in a Patient With Anti-MDA5 Antibody-Positive Dermatomyositis. J Clin Rheumatol. 2019 Jan 4. 10.1097/RHU.0000000000000984. Epub ahead of print. PMID: 30614890. [DOI] [PubMed]
- 28.Chen Z, Wang X, Ye S. Tofacitinib in amyopathic dermatomyositis-associated interstitial lung disease. N Engl J Med. 2019;381:291–3. doi: 10.1056/NEJMc1900045. [DOI] [PubMed] [Google Scholar]
- 29.Takatani A, Koga T, Fujita Y, et al. Efficacy of tofacitinib for slowly progressive interstitial lung disease in a patient with anti-MDA5 antibody-positive dermatomyositis. Clin Immunol. 2020;215:108451. doi: 10.1016/j.clim.2020.108451. [DOI] [PubMed] [Google Scholar]
- 30.Ohmura SI, Yamabe T, Naniwa T. Successful dose escalation of tofacitinib for refractory dermatomyositis and interstitial lung disease with anti-melanoma differentiation-associated gene 5 antibodies. Mod Rheumatol Case Rep. 2021;5:76–81. [DOI] [PubMed]
- 31.Kato M, Ikeda K, Kageyama T, et al. Successful treatment for refractory interstitial lung disease and pneumomediastinum with multidisciplinary therapy including tofacitinib in a patient with anti-MDA5 antibody-positive dermatomyositis. J Clin Rheumatol. 2019 doi: 10.1097/RHU.0000000000000984. [DOI] [PubMed] [Google Scholar]
- 32.Kochi Y, Kamatani Y, Kondo Y, et al. Splicing variant of WDFY4 augments MDA5 signalling and the risk of clinically amyopathic dermatomyositis. Ann Rheum Dis. 2018;77:602–611. doi: 10.1136/annrheumdis-2017-212149. [DOI] [PubMed] [Google Scholar]
- 33.Acosta-Herrera M, Kerick M, González-Serna D, et al. Genome-wide meta-analysis reveals shared new loci in systemic seropositive rheumatic diseases. Ann Rheum Dis. 2019;78:311–319. doi: 10.1136/annrheumdis-2018-214127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rothwell S, Cooper RG, Lundberg IE, et al. Dense genotyping of immune-related loci in idiopathic inflammatory myopathies confirms HLA alleles as the strongest genetic risk factor and suggests different genetic background for major clinical subgroups. Ann Rheum Dis. 2016;75:1558–1566. doi: 10.1136/annrheumdis-2015-208119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gono T, Kawaguchi Y, Ozeki E, et al. Serum ferritin correlates with activity of anti-MDA5 antibody-associated acute interstitial lung disease as a complication of dermatomyositis. Mod Rheumatol. 2011;21:223–227. doi: 10.1007/s10165-010-0371-x. [DOI] [PubMed] [Google Scholar]
- 36.Nishioka A, Tsunoda S, Abe T, et al. Serum neopterin as well as ferritin, soluble interleukin-2 receptor, KL-6 and anti-MDA5 antibody titer provide markers of the response to therapy in patients with interstitial lung disease complicating anti-MDA5 antibody-positive dermatomyositis. Mod Rheumatol. 2019;29:814–820. doi: 10.1080/14397595.2018.1548918. [DOI] [PubMed] [Google Scholar]
- 37.Matsushita T, Mizumaki K, Kano M, et al. Antimelanoma differentiation-associated protein 5 antibody level is a novel tool for monitoring disease activity in rapidly progressive interstitial lung disease with dermatomyositis. Br J Dermatol. 2017;176:395–402. doi: 10.1111/bjd.14882. [DOI] [PubMed] [Google Scholar]
- 38.Sakamoto S, Okamoto M, Kaieda S, et al. Low positive titer of anti-melanoma differentiation-associated gene 5 antibody is not associated with a poor long-term outcome of interstitial lung disease in patients with dermatomyositis. Respir Investig. 2018;56:464–472. doi: 10.1016/j.resinv.2018.07.007. [DOI] [PubMed] [Google Scholar]
- 39.Raghu G, Remy-Jardin M, Myers JL, et al. Diagnosis of idiopathic pulmonary fibrosis. An official ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2018;198:e44–e68. doi: 10.1164/rccm.201807-1255ST. [DOI] [PubMed] [Google Scholar]
- 40.Raghu G, Collard HR, Egan JJ, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183:788–824. doi: 10.1164/rccm.2009-040GL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li T, Guo L, Chen Z, et al. Pirfenidone in patients with rapidly progressive interstitial lung disease associated with clinically amyopathic dermatomyositis. Sci Rep. 2016;6:33226. doi: 10.1038/srep33226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Richeldi L, du Bois RM, Raghu G, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis [published correction appears in N Engl J Med. 2015 Aug 20;373(8):782] N Engl J Med. 2014;370(22):2071–2082. doi: 10.1056/NEJMoa1402584. [DOI] [PubMed] [Google Scholar]
- 43.Distler O, Highland KB, Gahlemann M, et al. Nintedanib for systemic sclerosis-associated interstitial lung disease. N Engl J Med. 2019;380:2518–2528. doi: 10.1056/NEJMoa1903076. [DOI] [PubMed] [Google Scholar]
- 44.Flaherty KR, Wells AU, Cottin V, et al. Nintedanib in progressive fibrosing interstitial lung diseases. N Engl J Med. 2019;381:1718–27. doi: 10.1056/NEJMoa1908681. [DOI] [PubMed] [Google Scholar]
- 45.Reeves HM, Winters JL. The mechanisms of action of plasma exchange. Br J Haematol. 2014;164:342–351. doi: 10.1111/bjh.12629. [DOI] [PubMed] [Google Scholar]
- 46.Shirakashi M, Nakashima R, Tsuji H, et al. Efficacy of plasma exchange in anti-MDA5-positive dermatomyositis with interstitial lung disease under combined immunosuppressive treatment [published online ahead of print, 2020 Apr 10]. Rheumatology (Oxford). 2020;keaa123. 10.1093/rheumatology/keaa123. [DOI] [PubMed]
- 47.Abe Y, Kusaoi M, Tada K, Yamaji K, Tamura N. Successful treatment of anti-MDA5 antibody-positive refractory interstitial lung disease with plasma exchange therapy. Rheumatology (Oxford) 2020;59:767–771. doi: 10.1093/rheumatology/kez357. [DOI] [PubMed] [Google Scholar]
- 48.Ning Y, Yang G, Sun Y, Chen S, Liu Y, Shi G. Efficiency of therapeutic plasma-exchange in acute interstitial lung disease, associated with polymyositis/dermatomyositis resistant to glucocorticoids and immunosuppressive drugs: a retrospective study. Front Med (Lausanne) 2019;6:239. doi: 10.3389/fmed.2019.00239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Endo Y, Koga T, Suzuki T, et al. Successful treatment of plasma exchange for rapidly progressive interstitial lung disease with anti-MDA5 antibody-positive dermatomyositis: a case report. Medicine (Baltimore) 2018;97:e0436. doi: 10.1097/MD.0000000000010436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Saito T, Mizobuchi M, Miwa Y, et al. Anti-MDA-5 antibody-positive clinically amyopathic dermatomyositis with rapidly progressive interstitial lung disease treated with therapeutic plasma exchange: a case series [published online ahead of print, 2020 Aug 21]. J Clin Apher. 2020;10.1002/jca.21833. 10.1002/jca.21833. [DOI] [PubMed]
- 51.Yamagata A, Arita M, Tanaka A, et al. Therapeutic plasma exchange for clinically amyopathic dermatomyositis (CADM) associated with rapidly progressive interstitial pneumonia [published online ahead of print, 2020 Aug 18]. J Clin Apher. 2020;10.1002/jca.21824. 10.1002/jca.21824. [DOI] [PubMed]
- 52.Kagawa H, Tsujino K, Yamamoto Y, et al. Acute lung injury after plasma exchange in a patient with anti-MDA5 antibody-positive, rapidly progressive, interstitial lung disease. A case report. Respir Med Case Rep. 2020;29:101016. doi: 10.1016/j.rmcr.2020.101016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Klein DJ, Foster D, Walker PM, Bagshaw M, Mekonnen H, Antonelli M. Polymyxin B hemoperfusion in endotoxemic septic shock patients without extreme endotoxemia: a post hoc analysis of the EUPHRATES trial. Intensive Care Med. 2018;44:2205–2212. doi: 10.1007/s00134-018-5463-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tsushima K, Kubo K, Koizumi T, et al. Direct hemoperfusion using a polymyxin B immobilized column improves acute respiratory distress syndrome. J Clin Apher. 2002;17:97–102. doi: 10.1002/jca.10019. [DOI] [PubMed] [Google Scholar]
- 55.Okabayashi H, Ichiyasu H, Hirooka S, Akaike K, Kojima K, Jodai T, et al. Clinical effects of direct hemoperfusion using a polymyxin B-immobilized fiber column in clinically amyopathic dermatomyositis-associated rapidly progressive interstitial pneumonias. BMC Pulm Med. 2017;17:134. doi: 10.1186/s12890-017-0479-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sasaki O, Dohi M, Harada H, et al. A case of polymyxin B-immobilized fiber column treatment for rapidly progressive interstitial pneumonia associated with clinically amyopathic dermatomyositis. Case Rep Med. 2013;2013:750275. doi: 10.1155/2013/750275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Teruya A, Kawamura K, Ichikado K, Sato S, Yasuda Y, Yoshioka M. Successful polymyxin B hemoperfusion treatment associated with serial reduction of serum anti-CADM-140/MDA5 antibody levels in rapidly progressive interstitial lung disease with amyopathic dermatomyositis. Chest. 2013;144:1934–1936. doi: 10.1378/chest.13-0186. [DOI] [PubMed] [Google Scholar]
- 58.Furusawa H, Sugiura M, Mitaka C, Inase N. Direct hemoperfusion with polymyxin B-immobilized fibre treatment for acute exacerbation of interstitial pneumonia. Respirology. 2017;22:1357–1362. doi: 10.1111/resp.13054. [DOI] [PubMed] [Google Scholar]
- 59.Ichiyasu H, Horio Y, Tsumura S, Hirosako S, Sakamoto Y, Sakata S, et al. Favorable outcome with hemoperfusion of polymyxin B-immobilized fiber column for rapidly progressive interstitial pneumonia associated with clinically amyopathic dermatomyositis: report of three cases. Mod Rheumatol. 2014;24:361–365. doi: 10.3109/14397595.2013.852847. [DOI] [PubMed] [Google Scholar]
- 60.Ichiyasu H, Horio Y, Masunaga A, Migiyama Y, Sakamoto Y, Jodai T, et al. Efficacy of direct hemoperfusion using polymyxin B-immobilized fiber column (PMXDHP) in rapidly progressive interstitial pneumonias: results of a historical control study and a review of previous studies. Ther Adv Respir Dis. 2017;11:261–275. doi: 10.1177/1753465817708950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Broome M, Palmer K, Schersten H, Frenckner B, Nilsson F. Prolonged extracorporeal membrane oxygenation and circulatory support as bridge to lung transplant. Ann Thorac Surg. 2008;86:1357–1360. doi: 10.1016/j.athoracsur.2008.03.053. [DOI] [PubMed] [Google Scholar]
- 62.Vuillard C, Pineton de Chambrun M, de Prost N, Guerin C, Schmidt M, Dargent A, et al. Clinical features and outcome of patients with acute respiratory failure revealing anti-synthetase or anti-MDA-5 dermato-pulmonary syndrome: a French multicenter retrospective study. Ann Intensive Care. 2018;8:87. doi: 10.1186/s13613-018-0433-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Alqatari S, Riddell P, Harney S, Henry M, Murphy G. MDA-5 associated rapidly progressive interstitial lung disease with recurrent pneumothoraxes: a case report. BMC Pulm Med. 2018;18:59. doi: 10.1186/s12890-018-0622-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gorka J, Szczeklik W, Wludarczyk A, Loboda P, Chmura L, Musial J. Rapidly progressive interstitial lung fibrosis in a patient with amyopathic dermatomyositis and antiMDA5 antibodies. Pol Arch Med Wewn. 2015;125:685–686. doi: 10.20452/pamw.3055. [DOI] [PubMed] [Google Scholar]
- 65.Aoyama J, Hayashi H, Yajima C, et al. Anti-MDA5 antibody-positive rapidly progressive interstitial pneumonia without cutaneous manifestations. Respir Med Case Rep. 2019;26:193–196. doi: 10.1016/j.rmcr.2019.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li ZY, Gill E, Mo F, Reyes C. Double anti-PL-7 and anti-MDA-5 positive amyopathic dermatomyositis with rapidly progressive interstitial lung disease in a Hispanic patient. BMC Pulm Med. 2020;20:220. doi: 10.1186/s12890-020-01256-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Leclair V, Labirua-Iturburu A, Lundberg IE. Successful lung transplantation in a case of rapidly progressive interstitial lung disease associated with antimelanoma differentiation-associated gene 5 antibodies. J Rheumatol. 2018;45(4):581–583. doi: 10.3899/jrheum.171047. [DOI] [PubMed] [Google Scholar]
- 68.Deitchman AR, Kalchiem-Dekel O, Todd N, Reed RM. Rapidly progressive interstitial lung disease due to anti-melanoma differentiation associated protein-5 requiring a bilateral lung transplant, and complicated by kennel cough. Respir Med Case Rep. 2019;28:100886. doi: 10.1016/j.rmcr.2019.100886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Huang K, Vinik O, Shojania K, et al. Clinical spectrum and therapeutics in Canadian patients with anti-melanoma differentiation associated gene 5 (MDA5)-positive dermatomyositis: a case-based review. Rheumatol Int. 2019;39:1971–81. doi: 10.1007/s00296-019-04398-2. [DOI] [PubMed] [Google Scholar]
- 70.Pacot L, Pouchot J, De Prost N, et al. Interstitial lung disease-complicated anti-MDA5 antibody in clinically amyopathic dermatomyositis patients: report of two cases with distinct clinical features. Front Med (Lausanne) 2020;7:77. doi: 10.3389/fmed.2020.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Prieto-Peña D, Martínez-Meñaca A, Calderón-Goercke M, et al. Long-term survival of lung transplantation for interstitial lung disease associated with connective tissue diseases: a study of 26 cases from a referral centre. Clin Exp Rheumatol. 2020;38:615–620. [PubMed] [Google Scholar]
- 72.Selva-O’Callaghan A, Labrador-Horrillo M, Munoz-Gall X, et al. Polymyositis/dermatomyositis-associated lung disease: analysis of a series of 81 patients. Lupus. 2005;14:534–542. doi: 10.1191/0961203305lu2158oa. [DOI] [PubMed] [Google Scholar]
- 73.Dotan Y, Vaidy A, Shapiro WB, Zhao H, Dass C, Toyoda Y, et al. Effect of acute exacerbation of idiopathic pulmonary fibrosis on lung transplantation outcome. Chest. 2018;154:818–26. [DOI] [PubMed]
- 74.Shoji T, Bando T, Fujinaga T, et al. Living-donor lobar lung transplantation for rapidly progressive interstitial pneumonia associated with clinically amyopathic dermatomyositis: report of a case. Gen Thorac Cardiovasc Surg. 2013;61:32–4. [DOI] [PubMed]