The worldwide battle against the coronavirus disease 2019 (COVID-19) pandemic has entered the mass vaccination phase. Israel had an early start1 and ≈90% of the adult population received the BioNTech COVID-19 (Pfizer) vaccine. Early vaccination phase involved elderly population, however, on January 26, the minimal age limit was lowered to ≥16 years. Shortly thereafter, during February to April 2021, a cluster of 8 patients acutely presented to our local emergency department with suspected myocarditis after receiving the second dose of the BioNTech COVID-19 (Pfizer) vaccine. The study was approved by our Institutional Review Board. The data that support the findings of this study are available from the corresponding author upon reasonable request.
All cases involved young (age 20–34 years) patients, all but one of male gender. Acute COVID-19 infection was excluded based on normal chest X-rays, negative swabs for COVID-19 polymerase chain reaction and negative anti-COVID-19-N antibodies but positive COVID-19-S antibodies (reflecting recent vaccination). All but one case had full viral and bacterial workup to exclude alternative causes of myocarditis.
Previous medical history was notable in 3 patients: One with celiac disease diagnosed during childhood, treated only with gluten restriction; one with asthma stable on montelukast therapy; the third patient had 2 prior perimyocarditis episodes requiring hospitalization: In 2017, the patient presented with several days of fever followed by left arm pain and elevated troponin levels. Echocardiography showed left ventricular ejection fraction (LVEF) of 45%, and he was treated with colchicine for several weeks. In 2019, he presented with fever, diarrhea, and chest discomfort. ECG showed diffuse ST-segment elevation with elevated troponin levels. Viral and bacterial workup was negative. Echocardiography showed LVEF=45%, and cardiac magnetic resonance imaging (MRI) showed LVEF=56% and positive late gadolinium enhancement.
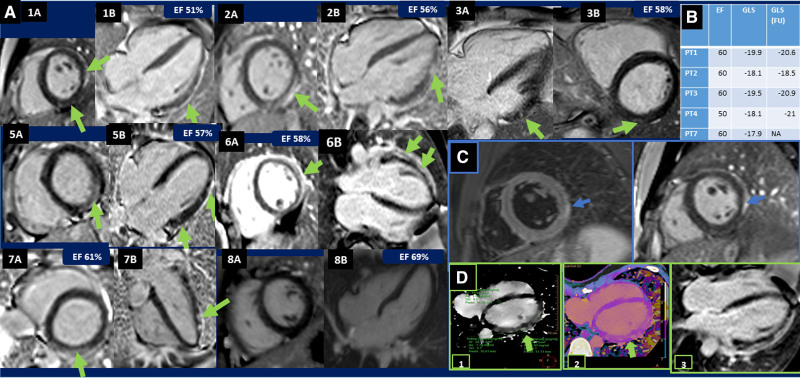
All 8 patients had an uneventful course after the first COVID-19 vaccination. However, within 3 to 7 days following the second vaccination, they developed angina-type chest pain and malaise, severe enough to prompt hospitalization. Upon admission, 5 patients had abnormal electrocardiograms (usually diffuse ST-segment changes; one patient had Q waves in II, III, AVF) and all had elevated troponin levels (median 4226 ng/L, normal <2.5 ng/L). Interestingly, troponin levels fluctuated, invariably demonstrating one peak on day 2 (median 14 153 ng/L) and a second peak (median 13 887 ng/L) on day 4. No correlation between troponin levels and severity of clinical symptoms was noted. Echocardiograms showed reduced LVEF of 45% to 50% in 3 patients and impaired speckled strain in 5 patients (median −18.1%, normal −20% to 23%, Figure). Cardiac MRI (MAGNETOM Aera 1.5T, Siemens) was available in 7/8 patients and fulfilled accepted criteria for myocardial inflammation2 in 6 patients. Patchy late gadolinium enhancement was evident in 6/7 patients, involving subepicardial and midmyocardial regions ranging from 1% to 21% (median 5.5%) myocardial involvement (Figure). In one patient, T2 mapping consistent with myocardial edema was visible along the late gadolinium enhancement. Patients with localized ST-segment elevation and abnormal LVEF, underwent invasive or noninvasive coronary imaging, demonstrating normal coronary arteries in all cases. According to our local clinical policy, 4 patients underwent spectral cardiac CT assessment (Spectral CT 7500 Philips, iodine dose 1.5 mL/kg, radiation dose 5–7 mSv) showing delayed iodine enhancement at the 7 minutes scan, with typical distribution of myocarditis (Figure). One patient, who refused to undergo an MRI was diagnosed with myocarditis according to clinical presentation, diffuse ST-segment changes, elevated troponin levels, and mild LV dysfunction on echocardiography.3 Notably, this patient was the one who had previous myocarditis episodes. Similarly, in the patient in whom MRI scan did not demonstrate typical MRI findings, the diagnosis was based on clinical, electrocardiographic, echocardiographic, and laboratory findings.
Figure.
Echocardiography, magnetic resonance imaging (MRI), and cardiac computed tomography (CT) findings. A, Late gadolinium enhancement (LGE) short-axis and 4-chamber images that demonstrate both linear (1A, 2A, 5A, 6A, 6B, and 7A) and patchy (1A, 1B, 2B, 3A, 3B, 5B, 6B, and 7B) subepicardial and midmyocardial LGE patterns. B, Table showing patients with reduced global longitudinal strain (GLS), their ejection fraction (EF) and GLS measured at a follow-up (FU) visit a median of 18 wk after their hospitalization. C, Patient no. 1, cardiac MRI, oblique short-axis views. Left: T2W dark blood (turbo inversion recovery magnitude) image showing high signal intensity corresponding to edema in the lateral wall. Right: subepicardial LGE in the lateral wall. D, Showing both MRI and cardiac CT, 4-chamber view of patient no. 6 (1+2)-delayed enhancement 7 min postinjection using prospective electrocardiography-triggered axial scans set at middiastolic phase (78%) using detector-based spectral CT (Spectral CT 7500, Philips Healthcare, Best, the Netherlands). 1: Iodine density image. 2: conventional overlay Z-effective, showing patchy midepicardial and subepicardial delayed iodine enhancement of the lateral wall. 3: Cardiac MR showing patchy midepicardial and subepicardial LGE of the lateral wall.
Resolution of symptoms, consistent decrease of troponin levels, without significant complications, invariably occurred in all patients within 7 days. One patient developed a short run of asymptomatic monomorphic ventricular tachycardia which resolved spontaneously. At discharge, 2 patients had slightly depressed LVEF on echocardiography, whereas the other 6 patients had normal LV function. No patient was discharged with home ECG monitoring.
Speckled strain echocardiography was done in 6/8 patients, 18 weeks (median) following the index episode and showed mildly reduced global longitudinal strain in only one patient.
Influenza vaccine-associated myocarditis is well described. The mechanism is unknown but appears to involve inappropriate immune responses which may also be triggered by mRNA vaccines along with other potential mechanisms4 and is emphasized by the presence of autoimmune-related diseases in 3 of our patients. The male predominance noted here is intriguing.
Previous reports suggested a possible association between the COVID-19 vaccine and myocarditis.5
We present a cluster with nearly identical clinical, laboratory and imaging findings along with a clear timeline link to the date of the second BioNTech vaccination. Notwithstanding, there is no precedent to this mass vaccination effort in history and a coincidental cluster of unrelated myocarditis cannot be excluded. Finally, our findings suggest that despite this potential vaccine-associated complication, most patients will have a benign course of disease and high rates of recovery.
Sources of Funding
None.
Disclosures
None.
Nonstandard Abbreviations and Acronyms
- COVID-19
- coronavirus disease 2019
- LVEF
- left ventricular ejection fraction
- MRI
- magnetic resonance imaging
For Sources of Funding and Disclosures, see page 910.
Contributor Information
Dana Viskin, Email: danaviskin@gmail.com.
Yan Topilsky, Email: topilskyyan@gmail.com.
Galit Aviram, Email: aviramgalit@hotmail.com.
Tomer Mann, Email: manntomer@gmail.com.
Sapir Sadon, Email: Sapirsadon8@gmail.com.
Yitzhac Hadad, Email: yitzhach@tlvmc.gov.il.
Nir Flint, Email: flintn@gmail.com.
Haim Shmilovich, Email: haim.shmilovich@gmail.com.
Shmuel Banai, Email: banais@netvision.net.il.
References
- 1.Dagan N, Barda N, Kepten E, Miron O, Perchik S, Katz MA, Hernán MA, Lipsitch M, Reis B, Balicer RD. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N Engl J Med. 2021; 384:1412–1423. doi: 10.1056/NEJMoa2101765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U, Kindermann I, Gutberlet M, Cooper LT, Liu P, et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018; 72:3158–3176. doi: 10.1016/j.jacc.2018.09.072 [DOI] [PubMed] [Google Scholar]
- 3.Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Heliö T, Heymans S, Jahns R, et al. ; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013; 34:2636–48, 2648a. doi: 10.1093/eurheartj/eht210 [DOI] [PubMed] [Google Scholar]
- 4.Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines - a new era in vaccinology. Nat Rev Drug Discov. 2018; 17:261–279. doi: 10.1038/nrd.2017.243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Prevention CfDCa. Centers for Diseases Control and Prevention-Vaccines and Related Biological Products Advisory Committee June 10, 2021, Meeting Presentation. Accessed on June 22, 2021. https://www.fda.gov/media/150054/download Posted on June 10, 2021