Abstract
Introduction
Both body mass index (BMI) and waist circumference (WC) are associated with diabetes risk, and the difference between them in predictive ability for diabetes is still contentious. We conducted a population-based study to investigate and compare the association of them with diabetes by sex.
Methods
This study included a total of 4754 subjects aged 40–80 years with no diabetes at baseline between 2008 and 2017. Using multivariate Cox proportional hazards models, we calculated hazard ratios for diabetes according to tertiles of BMI or WC. Harrell's C statistics was applied to assess and compare the predictive ability of the models using BMI and WC.
Results
Both BMI and WC showed the significant positive trends with diabetes risk. In men, the extreme tertiles (BMI > 25.1 kg/m2 and WC > 88.0 cm) provided 1.58-fold or 2.04-fold higher risk compared with the first tertiles (< 22.6 kg/m2 and < 81.2 cm). In women, BMI > 24.4 kg/m2 showed 3.28-fold higher risk than the first tertile (< 21.6 kg/m2), whereas WC ≥ 78.2 cm was more than twice as likely to suffer from diabetes as WC < 78.2 cm. BMI and WC showed a comparative performance in predicting diabetes in both sexes (P value 0.447 in men, and 0.337 in women).
Conclusion
Both BMI and WC showed a positive association with diabetes and offered a comparative predictive performance for diabetes in both sexes. The cut-off points, BMI 25.1 kg/m2 and WC 88.0 cm in men and BMI 24.4 kg/m2 and WC 78.2 cm in women, might contribute to the effective prevention strategies for diabetes.
Keywords: Body mass index, Waist circumference, Diabetes, Obesity
Key Summary Points
| Why carry out this study? |
| Obesity and diabetes are significant global health burdens that are increasing at an alarming rate. Obesity is an established risk factor for type 2 diabetes, and its prevention is one of the key factors to stop diabetes pandemic. Although both body mass index and waist circumference are useful diagnostic tools for assessing obesity, differences in body fat composition cause inconsistent results which hamper right decision making and effective health intervention |
| The aim of this study was to compare the predictive ability of body mass index and waist circumference for diabetes in adults |
| What was learned from the study? |
| Body mass index and waist circumference in both sexes provided a comparable predictive ability for diabetes in a large population |
| In men and women, body mass index over 25.1 and 24.4 kg/m2 and waist circumference over 88.0 and 78.2 cm, respectively, significantly increased diabetes risk |
Introduction
Obesity has been established as a major risk factor for diabetes and as a public health challenge the world is facing. According to recent reports, diabetes is the second leading cause of high-body mass index (BMI)-related disability-adjusted life years (DAILY) worldwide [1]. In 2019, the international diabetes federation (IDF) estimated 463 million people (9.3%) with diabetes, and by 2045, the number of cases is predicted to increase up to 700 million people (10.9%) [2]. During the last few decades, rapid urbanization and the popularity of western diets have resulted in the soaring prevalence of obesity and consequent diabetes in Asian countries where more than 60% of the world’s population live [3]. In Asian upper-middle or lower-middle income countries including China, Malaysia, Thailand, and India, the intake of meat, oils, and processed foods such as packaged meals, snacks, sweetened cereals, and soft drinks have been rapidly increasing [4]. In China and Vietnam, the processed foods consumption per capita increased five-fold, while in India, the total daily intake of fat increased by 6 g per capita over the past two decades [5]. In Malaysia, the percentages of distribution of processed foods through supermarkets or hypermarkets have tripled, accelerating the nutrition transition from traditional healthy diet to the modern Westernized diet [4]. About 50% (193 million) of all diabetes cases are recorded particularly in China and India, where by 2045, the number of cases is expected to increase up to 281 million [2]. In Japan, over 40% of men and 20% of women aged over 40 years are overweight using the BMI cut-off points for Asians [6], and approximately 7 million aged 20–79 years are estimated to have diabetes [7]. A rapidly aging society coupled with sedentary lifestyles and consumption of calorie-rich foods, has contributed to the prevalence of diabetes in the country [7].
BMI and waist circumference (WC) are safe, simple, and low-cost tools to assess one’s health condition and roughly estimate the risk of obesity-related diseases including diabetes and cardiovascular diseases [8]. However, which index exhibits a better performance for diabetes risk prediction remains unclear. Two large cohort studies elucidated the positive association between BMI and diabetes risk; Kuwahara et al. [9] reported that those who developed diabetes showed a significantly higher trajectory of BMI than did those who did not develop diabetes in Japanese population, and Maskarinec et al. [10] revealed BMI was positively related to incidence of diabetes irrespective of the ethnic subgroups including Caucasian, Hawaiian, and Japanese. Another study conducted in Taiwan demonstrated that compared with WC, BMI had higher adjusted odds of having insulin resistance that is vital to the development of type 2 diabetes [11]. Meanwhile, some studies showed a comparable predictive ability of BMI and WC for diabetes to date [12–14]. A cross-sectional study from the DECODA (Diabetes Epidemiology: Collaborative Analysis of Diagnostic criteria in Asia) study [15] and the previous Chinese studies [12, 14] found that there was no significant difference between BMI and WC in area under the curve (AUC), a measure of diagnostic accuracy, for detecting diabetes, although WC showed the higher odds of diabetes. However, a growing number of reports have shown the superiority of WC as a screening tool for the prediction of diabetes [16–20]. WC is a surrogate marker of visceral fat distribution and therefore its association with the risk of diabetes in adults has been focused on, among various ethnicities including East Asian [21–24]. Jeon et al. [24] presented the high-increasing WC group showed the higher risk of diabetes over a mean follow-up of 6.2 years. The other Asian countries also supported the better predictive performance of WC for diabetes [25, 26]. The European InterAct study [27] and a Swedish population-based study [28] also showed the stronger positive association of WC with diabetes than that of BMI although both BMI and WC independently had a significant effect on diabetes risk in both sexes. Likewise, a multi-cohort study in the UK [29] also supported the findings. Thus, in the Western population, it seems to be true that WC has a greater predictive ability for diabetes than has BMI, while in Asian population there still has been conflicting evidence about it. To our best knowledge, no previous Japanese studies demonstrated a comparison of the predictive ability of BMI and WC for diabetes, although some Japanese studies compared the predictive ability of BMI and WC for cardiovascular diseases or metabolic syndrome [30, 31].
Although BMI is classically used to estimate body fat mass and classify obesity, it has not been considered a sufficient measurement because it neither distinguishes fat mass from lean mass nor does it distinguish between fat types [19, 32]. These shortcomings can lead to the overestimation of body fat mass in individuals with a high lean mass, or underestimation of the risk of diabetes or cardiovascular diseases in those who have significant fat infiltration in the muscle or liver with normal BMI [21]. Sex differences significantly impact the cut-off values of BMI and WC due to the sexual dimorphisms of body fat composition [33]. Thus, the association of BMI or WC with diabetes should be assessed according to sex. Furthermore, insulin sensitivity and resistance that are closely related to body fat composition are not identical even among Asian countries [34]. Indeed, the previous studies have shown the considerable difference in optimal cut-off points of BMI or WC for diabetes depending on the country [11, 14, 25, 26, 35].
The aim of this study was to explore and compare the predictive ability of BMI and WC for diabetes in both sexes to gain further insights into the association of BMI and WC with diabetes and the optimal cut-off points of BMI and WC for diabetes in Japanese adults.
Methods
Study Design and Subjects
This study was a retrospective secondary database analysis. Our data were derived from the Iki epidemiological study of atherosclerosis and chronic kidney disease (ISSA-CKD) which is a population-based cohort study involving the residents in Iki City, Nagasaki prefecture in Japan. A total of 7895 residents, approximately 30% of the whole population in the city, underwent annual health checkups there between 2008 and 2017. Following the exclusion of 1898 residents who underwent a health checkup only once, 667 who had diabetes at baseline, and 576 who had missing data, 4754 residents were recruited for our retrospective cohort study. Our study was conducted in accordance with the Helsinki Declaration, and was approved by the Fukuoka University Clinical Research and Ethics Center (2017M010) with informed consent obtained from each participant.
Data Collection
The baseline data were collected in the year when the participant had the first health-checkup during 2008 to 2017. At health checkups, height, weight, and WC were measured with the individual wearing light clothes without shoes; information on current smoking status, alcohol consumption, and exercise was obtained using questionnaires. We categorized smoking status into 2 groups: current smoker and non-smoker. Current smoker was defined as those who had smoked 100 cigarettes or more, or smoked regularly at the baseline, and non-smoker was defined as those who did not smoke regularly at the baseline. Alcohol consumption was classified into 3 categories: daily drinker, social drinker (2 times or more per week), and non-drinker. Daily drinker was defined as subjects who drank alcohol every day irrespective of the kind or amount of it, while social drinker was defined as those who drank alcohol twice or more per week. Non-drinker was defined as those who did not drink alcohol regularly at the time of data collection. In exercise categories, subjects were categorized into two groups according to whether they regularly exercised for 30 min/day for at least twice per week or less. The BMI and WC of both sexes were divided into three categories using tertiles to summarize the association between BMI and WC and diabetes more clearly and to apply the results to clinical practice more easily (BMI: T1 < 22.6 kg/m2, T2 22.6–25.1 kg/m2, and T3 > 25.1 kg/m2, WC: T1 < 81.2 cm, T2 81.2–88.0 cm, and T3 > 88.0 cm for the men; BMI: T1 < 21.6 kg/m2, T2 21.6–24.4 kg/m2, and T3 > 24.4 kg/m2, WC: T1 < 78.2 cm, T2 78.2–86.0 cm, and T3 > 86.0 cm for the women). Blood pressure was measured twice by a trained staff and the average of the two measurements was considered as the subject’s blood pressure. The time interval of 5 min was taken between the two readings of blood pressure. Plasma glucose, triglycerides (TG), low-density lipoprotein-cholesterol (LDL-C), uric acid, alanine aminotransferase (ALT), gamma-glutamyl transpeptidase (γ-GTP), and creatinine (Cr) concentration were investigated using the enzymatic method. HbA1c was determined using a high-performance chromatographic method.
Outcome
The primary outcome of this study was the incidence of diabetes. Diabetes was defined as an increase in fasting plasma glucose ≥ 7.0 mmol/L, random plasma glucose ≥ 11.1 mmol/L, or HbA1c ≥ 6.5%, or the initiation of glucose lowering treatment, including insulin [36], which was confirmed at the last health checkup. The onset of diabetes was detected during the follow-up after the enrollment.
Statistical analysis
Continuous and categorical variables were presented as mean ± SD and percentages of each category, respectively. The Mann–Whitney U test for continuous variables and the chi-squared test for categorical variables were used to assess for the differences between both sexes. Incidence rates were calculated per 1000 person-years. Cox proportional hazards model was used to estimate the hazard ratio (HR) and 95% confidence intervals (CIs) as the effects of the variables on diabetes risk across the tertiles of the BMI and WC within each sex group. Multivariable analyses were adjusted for age, systolic blood pressure, TG, LDL-C, ALT, γ-GTP, alcohol consumption, smoking, and exercise. Finally, we calculated the area under the curve (AUC) for the Cox proportional hazards models, referred to as Harrell’s concordance Index (C-statistics) [37], to evaluate and compare the predictive powers of the models using BMI and WC. Haller’s C statistics is the probability that the survivor has the lower hazard ratio plus half the probability that the two subjects have equal hazard ratios in the data pairs [38]. The differences of the discriminatory powers were assessed using the method introduced by Newson [38]. This method estimates Harrell’s C with 95%CI of hazard models using ‘somersd’ command, and tests the null-hypothesis that there is no difference between the Harrell’s C indexes of the hazard models using ‘lincom’ command in Stata; Somers’ D is also a measure to estimate the predictive ability of models and defined as Somers’ D = Harrell’ C* 2 − 1. Two-tailed P values < 0.05 were considered statistically significant. All the analyses in this study were performed using Stata SE version 16 (StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC.).
Results
Baseline characteristics of all the participants and those classified by sex are shown in Table 1. Regarding continuous variables, age, HbA1c, LDL-C, and HDL-C were significantly higher in women than in men, and the other continuous variables were significantly higher in men. Daily drinkers (45.8%), social drinkers (28.2%), and current smokers (34.5%) were predominantly distributed in men, whereas most of the women were non-drinkers (74.9%) or non-smokers (93.1%). The number of men who work out daily (29.1%) was significantly higher than that in women (25.2%).
Table 1.
Baseline characteristics of the subjects
| Variables | Total (n = 4754) | Men (n = 2130) | Women (n = 2624) | P† |
|---|---|---|---|---|
| Age (years), mean ± SD | 59.58 ± 10.71 | 59.17 ± 10.90 | 59.92 ± 10.54 | 0.042 |
| BMI (kg/m2), mean ± SD | 23.58 ± 3.41 | 23.99 ± 3.25 | 23.24 ± 3.50 | < 0.001 |
| WC (cm), mean ± SD | 83.56 ± 9.50 | 85.10 ± 8.92 | 82.31 ± 9.76 | < 0.001 |
| SBP (mmHg), mean ± SD | 129.74 ± 19.39 | 131.14 ± 18.93 | 128.60 ± 19.69 | < 0.001 |
| DBP (mmHg), mean ± SD | 75.17 ± 11.48 | 76.89 ± 11.62 | 73.78 ± 11.18 | < 0.001 |
| FPG (mg/dL), mean ± SD | 90.92 ± 9.81 | 93.44 ± 10.33 | 88.97 ± 8.92 | < 0.001 |
| HbA1c (%), mean ± SD | 5.30 ± 0.36 | 5.28 ± 0.37 | 5.32 ± 0.35 | < 0.001 |
| TG (mg/dL), mean ± SD | 117.73 ± 80.95 | 131.27 ± 97.46 | 106.74 ± 62.41 | < 0.001 |
| LDL-C (mg/dL), mean ± SD | 122.81 ± 31.45 | 117.39 ± 30.63 | 127.22 ± 31.42 | < 0.001 |
| HDL-C (mg/dL), mean ± SD | 61.97 ± 16.19 | 57.88 ± 15.46 | 65.29 ± 16.02 | < 0.001 |
| ALT (U/L), mean ± SD | 23.07 ± 17.37 | 26.62 ± 18.54 | 20.19 ± 15.78 | < 0.001 |
| γ-GTP (U/L), mean ± SD | 41.01 ± 54.23 | 57.95 ± 70.37 | 27.25 ± 29.78 | < 0.001 |
| Uric acid (mg/dL), mean ± SD | 5.09 ± 1.36 | 5.94 ± 1.26 | 4.40 ± 1.00 | < 0.001 |
| Cr (mg/dL), mean ± SD | 0.74 ± 0.17 | 0.84 ± 0.15 | 0.65 ± 0.13 | < 0.001 |
| Alcohol, n (%) | < 0.001 | |||
| Daily drinker | 1118 (23.5) | 976 (45.8) | 142 (5.4) | |
| Social drinker | 1117 (23.5) | 600 (28.2) | 517 (19.7) | |
| Non-drinker | 2519 (53.0) | 554 (26.0) | 1965 (74.9) | |
| Smoking, n (%) | < 0.001 | |||
| Current smoker | 915 (19.3) | 734 (34.5) | 181 (6.9) | |
| Non-smoker | 3839 (80.8) | 1396 (65.5) | 2443 (93.1) | |
| Exercise, n (%) | 0.003 | |||
| Yes | 1280 (26.9) | 619 (29.1) | 661 (25.2) | |
| No | 3474 (73.1) | 1511 (70.9) | 1963 (74.8) |
BMI body mass index, WC waist circumference, SBP systolic blood pressure, FPG fasting plasma glucose, TG triglycerides, LDL-C low-density lipoprotein-cholesterol, HDL-C high-density lipoprotein-cholesterol, ALT alanine aminotransferase, γ-GTP gamma-glutamyl transpeptidase, Cr creatinine
†P value was estimated by Mann–Whitney U test for continuous variables and by chi-squared test for categorical variables
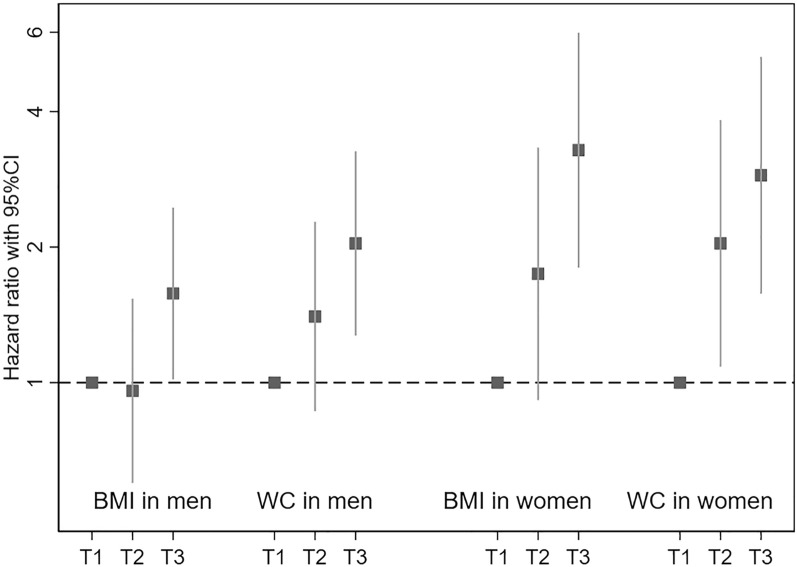
Table 2 depicts the incidence rates and crude and adjusted hazard ratios for diabetes across BMI and WC tertiles in men. During a mean follow-up of 5 years, 132 men were diagnosed with diabetes. The number of new-onset diabetes and the incidence rates consecutively increased with an increase in WC, while regarding BMI, they increased only in the third tertile compared with the first tertile. Both the crude (model 1) and the adjusted (model 2) hazard estimates obtained using Cox proportional hazards model presented a significant linear trend in the association between the two measurements and a higher diabetes risk (BMI: model 1 P 0.006, model 2 P 0.031; WC: model 1 P < 0.001, model 2 P 0.002 for trend) (Fig. 1). The adjusted hazard ratios in the highest tertile compared with the lowest one in BMI and WC were 1.58 (95% CI 1.02–2.45) and 2.04 (95% CI 1.27–3.26), respectively. The interaction between the ranks of tertiles and the type of measurements (BMI or WC) was not significant (P 0.527 for interaction).
Table 2.
Incidence rates and hazard ratios for diabetes across BMI and waist circumference tertiles in men
| Men | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| BMI | WC | P†† | ||||||||
| (kg/m2) | New-onset diabetes | Incidence rate† | Hazard ratio (95% CI) | (cm) | New-onset diabetes | Incidence rate† | Hazard ratio (95% CI) | |||
| Model 1‡ | Model 2§ | Model 1‡ | Model 2§ | |||||||
| T1 (< 22.6) | 36 | 12.57 | 1.00 (Reference) | 1.00 (Reference) | T1 (< 81.2) | 28 | 10.18 | 1.00 (Reference) | 1.00 (Reference) | 0.527 |
| T2 (22.6–25.1) | 35 | 9.49 | 1.02 (0.64–1.62) | 0.96 (0.60–1.54) | T2 (81.2–88.0) | 42 | 10.79 | 1.49 (0.92–2.41) | 1.40 (0.86–2.28) | |
| T3 (> 25.1) | 61 | 15.69 | 1.74 (1.15–2.63)** | 1.58 (1.02–2.45)* | T3 (> 88.0) | 62 | 16.31 | 2.30 (1.47–3.60)*** | 2.04 (1.27–3.26)** | |
| Total | 132 | P for trend¶ | 0.006 | 0.031 | Total | 132 | P for trend¶ | < 0.001 | 0.002 | |
BMI body mass index, WC waist circumference, HR hazard ratio, CI confidence interval
†Per 1000 person-years
‡Adjusted for no covariates
§Adjusted for age, SBP, TG, LDL-C, ALT, γ-GTP, alcohol, smoking, and exercise
¶P value was estimated by Cox proportional hazard analysis
††For interaction between the rank of tertiles and the type of measurements
*P < 0.05, **P < 0.01, and ***P < 0.001
Fig. 1.
Adjusted hazard rations across BMI and WC tertiles. Adjusted hazard ratios and 95% CIs associated with diabetes risk were estimated by Cox hazard proportional models. Squares represent the hazard ratios and the vertical bars crossing the squares indicate the 95% CI. The dashed line shows hazard ratio of 1.0. BMI body mass index, WC waist circumference, CI confidence interval
Women also showed an increase in the incidence rates of diabetes with a higher BMI and WC and a significant positive trend between the tertiles of BMI and WC and diabetes risk in both crude and adjusted models (P < 0.001) (Table 3, Fig. 1). The estimated hazard ratios in the highest tertile were higher than those in men (BMI: 3.28, 95% CI 1.80–5.98; WC: 2.89, 1.58–5.29). The rank by the type of measurement interaction was also not significant in women (P 0.585 for interaction).
Table 3.
Incidence rates and hazard ratios for diabetes across BMI and waist circumference tertiles in women
| Women | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| BMI | WC | P†† | ||||||||
| (kg/m2) | New-onset diabetes | Incidence rate† | Hazard ratio (95% CI) | (cm) | New-onset diabetes | Incidence rate† | Hazard ratio (95% CI) | |||
| Model 1‡ | Model 2§ | Model 1‡ | Model 2§ | |||||||
| T1 (< 21.6) | 14 | 2.58 | 1.00 (Reference) | 1.00 (Reference) | T1 (< 78.2) | 14 | 2.51 | 1.00 (Reference) | 1.00 (Reference) | 0.585 |
| T2 (21.6–24.4) | 28 | 6.26 | 2.04 (1.07–3.87)* | 1.74 (0.91–3.33) | T2 (78.2–86.0) | 33 | 7.54 | 2.46 (1.32–4.60)** | 2.04 (1.09–3.83)* | |
| T3 (> 24.4) | 57 | 14.46 | 4.34 (2.42–7.79)*** | 3.28 (1.80–5.98)*** | T3 (> 86.0) | 52 | 13.32 | 4.18 (2.32–7.54)*** | 2.89 (1.58–5.29)** | |
| Total | 99 | P for trend¶ | < 0.001 | < 0.001 | Total | 99 | P for trend¶ | < 0.001 | < 0.001 | |
BMI body mass index, WC waist circumference, HR hazard ratio, CI confidence interval
†Per 1000 person-years
‡Adjusted for no covariates
§Adjusted for age, SBP, TG, LDL-C, ALT, γ-GTP, alcohol, smoking, and exercise
¶P value was estimated by Cox proportional hazard analysis
††For interaction between the rank of tertiles and the type of measurements
*P < 0.05, **P < 0.01, and ***P < 0.001
Table 4 illustrates a comparison of the predictive ability of Cox proportional hazards models of BMI and WC in both sexes using Harrell’s C statistics. BMI and WC showed a comparable discrimination power for diabetes in both sexes (P = 0.295 in men, P = 0.152 in women).
Table 4.
Comparison of the predictive powers of Cox proportional hazards models for diabetes between BMI and WC by sex
| Harrell’s C statistics (95% CI) | P | |
|---|---|---|
| Men | ||
| BMI | 0.6522 (0.5986–0.7058)*** | 0.295 |
| WC | 0.6624 (0.6099–0.7150)*** | |
| Women | ||
| BMI | 0.7725 (0.7285–0.8164)*** | 0.152 |
| WC | 0.7587 (0.7147–0.8027)*** |
BMI body mass index, WC waist circumference, CI confidence interval
*P < 0.05, **P < 0.01, and ***P < 0.001
Discussion
This large longitudinal study focused on the association of BMI and WC with diabetes risk in both sexes; it elucidated their significant positive effects and comparable predictive ability for diabetes. Based on our results, the optimal cut-off points of BMI and WC were 25.1 kg/m2 and 88.0 cm in men, and 24.4 kg/m2 and 78.2 cm in women, respectively. Our finding that these two body fat markers had a significant positive association with diabetes risk is consistent with the previous reports [9, 11, 12, 14, 27, 28]. A study that analyzed four UK cohorts [29] and a meta-analysis comprising a total of 20,827 subjects [15] also noted the significant impacts of a higher BMI and WC on incident diabetes. A Korean prospective cohort study of 31,118 person-years of follow-up [24] demonstrated that individuals with a mean WC of 90.1 cm had 4.5- and 8.3-fold higher risk of diabetes in men and women, respectively, compared with those with a mean WC of 69.9 cm.
We also validated a growing trend in the incidence of diabetes at a relatively low BMI or WC which has been reported as an ethnic-specific trait of East Asian [15, 39]; the aforementioned meta-analysis [15] documented that Japanese participants showed a strong association between these body fat markers and diabetes risk (OR 1.91 and 1.83 in men, and OR 2.11 and 2.11 in women) albeit with a lower mean of BMI and WC (23.4 kg/m2 and 81.3 cm in men, and 23.4 kg/m2 and 74.7 cm in women) than that of the other ethnic groups.
Japanese immigrants residing in Western countries also have exhibited a higher susceptibility to body fat accumulation; a prospective study with a follow-up period of 1,119,224 person-years revealed the adjusted hazard ratios of diabetes for Japanese-Americans were higher than Caucasians and Native Hawaiians at any level of BMI including less than 22 kg/m2 [10]. In consistent with our results, the Japanese Americans showed the steadily increasing risk of diabetes with an increase of BMI before the risk soared at a BMI ≥ 25.0 kg/m2. Moreover, they revealed the almost identical incidence rates of diabetes to the mean of male incidence rates in our results (data not shown), although the incidence rates at a BMI < 25.0 kg/m2 in the whole subject were lower than our results since the Japanese Americans accounted for only 43%. According to several studies, the possible pathophysiological explanation for the predisposition in East Asians includes the onset of visceral fat accumulation, subsequent insulin resistance, and β-cell impairment, which could lead to paucity of insulin to compensate for insulin resistance, at a lower BMI or WC compared with Caucasian [9, 40]. Therefore, World Health Organization (WHO) suggests defining a BMI of 23–27.5 kg/m2 as overweight and a BMI more than 27.5 kg/m2 as obese for Asians although the standard cut-off point for obesity is 30 kg/m2 [41]. In our results, men with a BMI > 25.1 kg/m2 and women with a BMI > 24.4 kg/m2 were at a significantly higher risk for diabetes compared with the first tertiles (BMI < 22.6 kg/m2 in men and BMI < 21.6 kg/m2 in women). Almost in line with our findings, a prospective study [12] and a cross-sectional study [14] conducted in China reported the optimal cut-off points for diabetes were 24.73 kg/m2 in men and 23.18 kg/m2 in women, and 26.08 kg/m2 in men and 24.92 kg/m2 in women, respectively, while the other cross-sectional studies in Iran [35], Jordan [25], and Bangladesh [26] showed the optimal cut-off points of BMI for diabetes ranged from 21 up to 30 kg/m2. Recently, the American Diabetes Association has recommended that the BMI cut points for Asian American adults should be lowered from 25 to 23 kg/m2 based on the mounting evidence of Asian immigrants who are more susceptible to excess body fat accumulation than the other ethnic groups [10, 42]. However, lowering cut-off points can sacrifice some specificity for better sensitivity [43], and estimated cut-off points of BMI can be impacted by the mean BMI in participants, study design, and sample sizes, therefore further investigation is required.
Regarding WC, 88 cm in men and 78.2 cm in women were the thresholds to significantly raise the diabetes risk compared with those in the first tertile (WC < 81.2 cm in men and WC < 78.2 cm in women). These values are close to the cut-off points for Asians recommended by the WHO [41] and International Diabetes Federation [44] (90 cm in men and 80 cm in women). In the aforementioned studies in China [12, 14, 45] and a community-based study of Japanese-Americans in Seattle [46], their cut-off points were also close to our results, ranging from 87–90 cm in men, and from 77–84 cm in women. Although a Japanese prospective study with a median follow-up of 4.9 years [47] revealed a relatively low cut-off points of WC for men (85 cm), the lower mean WC at baseline than ours and the differences of the sample sizes and the follow-up periods might be the plausible explanation for the discrepancy between those results.
Despite the increasing evidence of better prediction ability of WC than BMI for diabetes [17, 18, 22, 26], our finding that BMI had a comparable prediction ability for diabetes as WC is supported by some published studies [12–14, 45, 48]. Recent reports have suggested that BMI, a surrogate marker of total body fat mass, increases with aging up to 75 years; aging and body fat accumulation share a similar pathophysiological pathways characterized by chronic inflammation leading to a state of β cell dysfunction and insulin resistance, resulting in diabetes [49–51].
Generally, in women, more subcutaneous fat is distributed than visceral fat, and menopause, in turn accelerates visceral fat accumulation [33]. Therefore, a stronger association between a higher WC and diabetes risk might be shown with aging. However, BMI still can remain comparably correlated with diabetes because visceral fat does not increase as drastically in men, and total body fat mass remains almost stable even after menopause [51]. Meanwhile, visceral fat mass distribution in men increases remarkably with aging, parallel to an increase in WC. This fact is incompatible with our result that BMI in men also had comparable prediction power for diabetes risk as WC. However, the discrepancy might be reasoned using a report by Jean-Pierre Després [21]; at the population level, BMI and WC strongly positively correlate although at the individual level, WC increases parallel to visceral fat distribution irrespective of BMI.
Compared to urban areas, rural areas have disadvantages in maintenance and risk assessment of obesity as an important risk factor of diabetes because of the less accessibility or availability of medical resources which assess body fat composition [52, 53]. Dual-energy X-ray absorptiometry, computed tomography, and magnetic resonance imaging can offer imaging information on subcutaneous or abdominal fat tissue compartments or adipose tissue in muscle to predict diabetes risk more accurately than anthropometric measurements [9, 51, 54], however, they are not routinely recommended due to the limited availability, exposure to radiation, and cost. As shown in our results, the acceptable predictive powers of BMI and WC for diabetes would contribute to the effective maintenance and risk assessment of obesity to prevent diabetes in regular health checkups. Also, Ma et al. [55] reported that physical exercise was less effective in preventing diabetes in Asian population than the other ethnic groups due partly to the more susceptibility to poor diet and the necessity to achieve even greater weight loss to have the same low risk of diabetes as non-overweight Caucasians. Therefore, accessible and effective prevention strategies should be more emphasized for them.
The strengths of our study include a large sample size and the longitudinal study design where the incidence of diabetes can be identified. Nevertheless, some limitations also should be mentioned. First, in this study, the diagnosis of diabetes was only based on HbA1c and plasma glucose levels. The lack of data on oral glucose tolerance tests might have underestimated the incidence of diabetes. Second, we did not differentiate type 1 from type 2 diabetes. However, we recruited the subjects from a population aged over 40 years, and it has been reported that type 1 diabetes accounts for 5–10% of all the cases of diabetes [56]. Therefore, the impact of this limitation might be trivial. On the other hand, the fact that all the participants were aged over 40 years might have limited the generalizability. Third, menopause status was not investigated in our study. However, epidemiological evidence has shown that the prospective positive association between BMI and WC and diabetes risk in women has been consistently observed regardless of menopausal status [21]. Forth, recruitment of those who had annual health checkups in the community might have caused selection bias and consequently underestimation of the incidence of diabetes because they are expected to be generally health conscious.
Conclusions
In conclusion, both increased BMI and WC were positively associated with diabetes risk and offered a comparative performance for the prediction of diabetes in both sexes in a Japanese large longitudinal study. Based on our results, the cut-off points, BMI 25.1 kg/m2 and WC 88.0 cm in men and BMI 24.4 kg/m2 and WC 78.2 in women, might contribute to the effective prevention strategies for diabetes.
Acknowledgements
The authors gratefully acknowledge the efforts of the many contributors to the ISSA-CKD study.
Funding
The authors received grants from the Iki City for this study, and the journal’s Rapid Service Fee was funded by the grants.
Authorship
All authors meet the International Committee of Medical Journal Editors criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
All authors contributed to this manuscript significantly. HA contributed to the conception and the design of this study. MA and HA were involved in the data analyses. MA, HF, SF, AS, MK, TM, KT, SY, TY, CY, RM, KT, KI, TY, TK, SM, KA, DK, KM, and HA contributed to the data interpretation. All authors critically revised the draft of the manuscript and approved the final report.
Disclosures
Makiko Abe, Hideyuki Fujii, Shunsuke Funakoshi, Atsushi Satoh, Miki Kawazoe, Toshiki Maeda, Kazuhiro Tada, Soichiro Yokota, Toshitaka Yamanokuchi, Chikara Yoshimura, Ryosuke Mimata, Koji Takahashi, Kenji Ito, Tetsuhiko Yasuno, Takeshi Kuga, Shigeki Mukoubara, Kozaburo Akiyoshi, Daiji Kawanami, Kosuke Masutani, and Hisatomi Arima accept accountability for all aspects of the work and have nothing to disclose.
Compliance with Ethics Guidelines
Our study was conducted in accordance with the Helsinki Declaration and was approved by the Fukuoka University Clinical Research and Ethics Center (2017M010) with informed consent obtained from each participant.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available for ethical reasons.
References
- 1.Dai H, Alsalhe TA, Chalghaf N, Riccò M, Bragazzi NL, Wu J. The global burden of disease attributable to high body mass index in 195 countries and territories, 1990–2017: an analysis of the Global Burden of Disease Study. PLoS Med. 2020;17(7):e1003198. doi: 10.1371/journal.pmed.1003198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2019;157:107843. doi: 10.1016/j.diabres.2019.107843. [DOI] [PubMed] [Google Scholar]
- 3.Nanditha A, Ma RC, Ramachandran A, et al. Diabetes in Asia and the Pacific: implications for the global epidemic. Diabetes Care. 2016;39(3):472–485. doi: 10.2337/dc15-1536. [DOI] [PubMed] [Google Scholar]
- 4.Baker P, Friel S. Food systems transformations, ultra-processed food markets and the nutrition transition in Asia. Glob Health. 2016;12(1):80. doi: 10.1186/s12992-016-0223-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Unnikrishnan R, Anjana RM, Mohan V. Diabetes in South Asians: is the phenotype different? Diabetes. 2014;63(1):53–55. doi: 10.2337/db13-1592. [DOI] [PubMed] [Google Scholar]
- 6.Hasegawa M, Akter S, Hu H, et al. Five-year cumulative incidence of overweight and obesity, and longitudinal change in body mass index in Japanese workers: the Japan Epidemiology Collaboration on Occupational Health Study. J Occup Health. 2020;62(1):e12095. doi: 10.1002/1348-9585.12095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ikeda N, Nishi N, Noda H, Noda M. Trends in prevalence and management of diabetes and related vascular risks in Japanese adults: Japan National Health and Nutrition Surveys 2003–2012. Diabetes Res Clin Pract. 2017;127:115–122. doi: 10.1016/j.diabres.2017.03.011. [DOI] [PubMed] [Google Scholar]
- 8.Huxley R, Mendis S, Zheleznyakov E, Reddy S, Chan J. Body mass index, waist circumference and waist:hip ratio as predictors of cardiovascular risk—a review of the literature. Eur J Clin Nutr. 2010;64(1):16–22. doi: 10.1038/ejcn.2009.68. [DOI] [PubMed] [Google Scholar]
- 9.Kuwahara K, Honda T, Nakagawa T, Yamamoto S, Hayashi T, Mizoue T. Body mass index trajectory patterns and changes in visceral fat and glucose metabolism before the onset of type 2 diabetes. Sci Rep. 2017;7:43521. doi: 10.1038/srep43521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Maskarinec G, Erber E, Grandinetti A, et al. Diabetes incidence based on linkages with health plans: the multiethnic cohort. Diabetes. 2009;58(8):1732–1738. doi: 10.2337/db08-1685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cheng YH, Tsao YC, Tzeng IS, et al. Body mass index and waist circumference are better predictors of insulin resistance than total body fat percentage in middle-aged and elderly Taiwanese. Medicine (Baltim) 2017;96(39):e8126. doi: 10.1097/MD.0000000000008126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu J, Gong L, Li Q, et al. A novel visceral adiposity index for prediction of type 2 diabetes and pre-diabetes in Chinese adults: a 5-year prospective study. Sci Rep. 2017;7(1):13784. doi: 10.1038/s41598-017-14251-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fujita M, Sato Y, Nagashima K, Takahashi S, Hata A. Predictive power of a body shape index for development of diabetes, hypertension, and dyslipidemia in Japanese adults: a retrospective cohort study. PLoS One. 2015;10(6):e0128972. doi: 10.1371/journal.pone.0128972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wei J, Liu X, Xue H, Wang Y, Shi Z. Comparisons of visceral adiposity index, body shape index, body mass index and waist circumference and their associations with diabetes mellitus in adults. Nutrients. 2019;11(7):1580. doi: 10.3390/nu11071580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nyamdorj R, Qiao Q, Lam TH, et al. BMI compared with central obesity indicators in relation to diabetes and hypertension in Asians. Obesity (Silver Spring) 2008;16(7):1622–1635. doi: 10.1038/oby.2008.73. [DOI] [PubMed] [Google Scholar]
- 16.Mirzaei M, Khajeh M. Comparison of anthropometric indices (body mass index, waist circumference, waist to hip ratio and waist to height ratio) in predicting risk of type II diabetes in the population of Yazd, Iran. Diabetes Metab Syndr. 2018;12(5):677–682. doi: 10.1016/j.dsx.2018.04.026. [DOI] [PubMed] [Google Scholar]
- 17.Hou X, Chen S, Hu G, et al. Stronger associations of waist circumference and waist-to-height ratio with diabetes than BMI in Chinese adults. Diabetes Res Clin Pract. 2019;147:9–18. doi: 10.1016/j.diabres.2018.07.029. [DOI] [PubMed] [Google Scholar]
- 18.Hartwig S, Kluttig A, Tiller D, et al. Anthropometric markers and their association with incident type 2 diabetes mellitus: which marker is best for prediction? Pooled analysis of four German population-based cohort studies and comparison with a nationwide cohort study. BMJ Open. 2016;6(1):e009266. doi: 10.1136/bmjopen-2015-009266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cicero AF, D’Addato S, Reggi A, Marchesini G, Borghi C. Gender difference in hepatic steatosis index and lipid accumulation product ability to predict incident metabolic syndrome in the historical cohort of the Brisighella Heart Study. Metab Syndr Relat Disord. 2013;11(6):412–416. doi: 10.1089/met.2012.0147. [DOI] [PubMed] [Google Scholar]
- 20.Ross R, Neeland IJ, Yamashita S, et al. Waist circumference as a vital sign in clinical practice: a consensus statement from the IAS and ICCR Working Group on visceral obesity. Nat Rev Endocrinol. 2020;16(3):177–189. doi: 10.1038/s41574-019-0310-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Després JP. Body fat distribution and risk of cardiovascular disease: an update. Circulation. 2012;126(10):1301–1313. doi: 10.1161/CIRCULATIONAHA.111.067264. [DOI] [PubMed] [Google Scholar]
- 22.Fan Y, Wang R, Ding L, et al. Waist circumference and its changes are more strongly associated with the risk of type 2 diabetes than body mass index and changes in body weight in Chinese adults. J Nutr. 2020;150(5):1259–1265. doi: 10.1093/jn/nxaa014. [DOI] [PubMed] [Google Scholar]
- 23.Haghighatdoost F, Amini M, Feizi A, Iraj B. Are body mass index and waist circumference significant predictors of diabetes and prediabetes risk: results from a population based cohort study. World J Diabetes. 2017;8(7):365–373. doi: 10.4239/wjd.v8.i7.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jeon J, Jung KJ, Jee SH. Waist circumference trajectories and risk of type 2 diabetes mellitus in Korean population: the Korean genome and epidemiology study (KoGES) BMC Public Health. 2019;19(1):741. doi: 10.1186/s12889-019-7077-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Khader Y, Batieha A, Jaddou H, El-Khateeb M, Ajlouni K. The performance of anthropometric measures to predict diabetes mellitus and hypertension among adults in Jordan. BMC Public Health. 2019;19(1):1416. doi: 10.1186/s12889-019-7801-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bhowmik B, Munir SB, Diep LM, et al. Anthropometric indicators of obesity for identifying cardiometabolic risk factors in a rural Bangladeshi population. J Diabetes Investig. 2013;4(4):361–368. doi: 10.1111/jdi.12053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Langenberg C, Sharp SJ, Schulze MB, et al. Long-term risk of incident type 2 diabetes and measures of overall and regional obesity: the EPIC-InterAct case-cohort study. PLoS Med. 2012;9(6):e1001230. doi: 10.1371/journal.pmed.1001230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nordström A, Hadrévi J, Olsson T, Franks PW, Nordström P. Higher prevalence of type 2 diabetes in men than in women is associated with differences in visceral fat mass. J Clin Endocrinol Metab. 2016;101(10):3740–3746. doi: 10.1210/jc.2016-1915. [DOI] [PubMed] [Google Scholar]
- 29.Taylor AE, Ebrahim S, Ben-Shlomo Y, et al. Comparison of the associations of body mass index and measures of central adiposity and fat mass with coronary heart disease, diabetes, and all-cause mortality: a study using data from 4 UK cohorts. Am J Clin Nutr. 2010;91(3):547–556. doi: 10.3945/ajcn.2009.28757. [DOI] [PubMed] [Google Scholar]
- 30.Suka M, Miwa Y, Ono Y, Yanagisawa H. BMI, waist circumference, and clustering of cardiovascular risk factors in Japanese adults. Environ Health Prev Med. 2011;16(2):90–96. doi: 10.1007/s12199-010-0169-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Oda E, Kawai R. Comparison among body mass index (BMI), waist circumference (WC), and percent body fat (%BF) as anthropometric markers for the clustering of metabolic risk factors in Japanese. Intern Med. 2010;49(15):1477–1482. doi: 10.2169/internalmedicine.49.3363. [DOI] [PubMed] [Google Scholar]
- 32.Okorodudu DO, Jumean MF, Montori VM, et al. Diagnostic performance of body mass index to identify obesity as defined by body adiposity: a systematic review and meta-analysis. Int J Obes. 2010;34(5):791–799. doi: 10.1038/ijo.2010.5. [DOI] [PubMed] [Google Scholar]
- 33.Tramunt B, Smati S, Grandgeorge N, et al. Sex differences in metabolic regulation and diabetes susceptibility. Diabetologia. 2020;63(3):453–461. doi: 10.1007/s00125-019-05040-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tan VM, Lee YS, Venkataraman K, et al. Ethnic differences in insulin sensitivity and beta-cell function among Asian men. Nutr Diabetes. 2015;5(7):e173. doi: 10.1038/nutd.2015.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shabnam AA, Homa K, Reza MT, Bagher L, Hossein FM, Hamidreza A. Cut-off points of waist circumference and body mass index for detecting diabetes, hypercholesterolemia and hypertension according to National Non-Communicable Disease Risk Factors Surveillance in Iran. Arch Med Sci. 2012;8(4):614–621. doi: 10.5114/aoms.2012.30284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Haneda M, Noda M, Origasa H, et al. Japanese clinical practice guideline for diabetes 2016. J Diabetes Investig. 2018;9(3):657–697. doi: 10.1111/jdi.12810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Harrell FE, Jr, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15(4):361–387. doi: 10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 38.Newson RB. Comparing the predictive powers of survival models using Harrell’s C or Somers’ D. Stand Genom Sci. 2010;10(3):339–358. [Google Scholar]
- 39.Karter AJ, Schillinger D, Adams AS, et al. Elevated rates of diabetes in Pacific Islanders and Asian subgroups: the diabetes study of Northern California (DISTANCE) Diabetes Care. 2013;36(3):574–579. doi: 10.2337/dc12-0722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ma RC, Chan JC. Type 2 diabetes in East Asians: similarities and differences with populations in Europe and the United States. Ann N Y Acad Sci. 2013;1281(1):64–91. doi: 10.1111/nyas.12098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.World Health Organization Expert Consultation Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363(9403):157–163. doi: 10.1016/S0140-6736(03)15268-3. [DOI] [PubMed] [Google Scholar]
- 42.American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(Suppl 1):S14–31. [DOI] [PubMed]
- 43.Hsia DS, Larrivee S, Cefalu WT, Johnson WD. Impact of lowering BMI cut points as recommended in the revised American Diabetes Association’s Standards of Medical Care in diabetes-2015 on diabetes screening in Asian Americans. Diabetes Care. 2015;38(11):2166–2168. doi: 10.2337/dc15-0299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alberti KGMM, Zimmet P, Shaw J. Metabolic syndrome—a new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet Med. 2006;23(5):469–480. doi: 10.1111/j.1464-5491.2006.01858.x. [DOI] [PubMed] [Google Scholar]
- 45.Wei J, Liu X, Xue H, Wang Y, Shi Z. Comparisons of visceral adiposity index, body shape index, body mass index and waist circumference and their associations with diabetes mellitus in adults. Nutrients. 2019;11(7):1580. doi: 10.3390/nu11071580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fujimoto WY, Boyko EJ, Hayashi T, et al. Risk factors for type 2 diabetes: lessons learned from Japanese Americans in Seattle. J Diabetes Investig. 2012;3(3):212–224. doi: 10.1111/j.2040-1124.2012.00195.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hu H, Kurotani K, Sasaki N, et al. Optimal waist circumference cut-off points and ability of different metabolic syndrome criteria for predicting diabetes in Japanese men and women: Japan Epidemiology Collaboration on Occupational Health Study. BMC Public Health. 2016;16:220. doi: 10.1186/s12889-016-2856-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vazquez G, Duval S, Jacobs DR, Jr, Silventoinen K. Comparison of body mass index, waist circumference, and waist/hip ratio in predicting incident diabetes: a meta-analysis. Epidemiol Rev. 2007;29:115–128. doi: 10.1093/epirev/mxm008. [DOI] [PubMed] [Google Scholar]
- 49.Johnson AR, Milner JJ, Makowski L. The inflammation highway: metabolism accelerates inflammatory traffic in obesity. Immunol Rev. 2012;249(1):218–238. doi: 10.1111/j.1600-065X.2012.01151.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cerf ME. Beta cell physiological dynamics and dysfunctional transitions in response to islet inflammation in obesity and diabetes. Metabolites. 2020;10(11):452. doi: 10.3390/metabo10110452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ponti F, Santoro A, Mercatelli D, et al. Aging and imaging assessment of body composition: from fat to facts. Front Endocrinol (Lausanne) 2020;10:861–861. doi: 10.3389/fendo.2019.00861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Qin VM, McPake B, Raban MZ, et al. Rural and urban differences in health system performance among older Chinese adults: cross-sectional analysis of a national sample. BMC Health Serv Res. 2020;20(1):372. doi: 10.1186/s12913-020-05194-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dugani SB, Mielke MM, Vella A. Burden and management of type 2 diabetes in rural United States. Diabetes Metab Res Rev. 2021;37(5):e3410. doi: 10.1002/dmrr.3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ponti F, Santoro A, Mercatelli D, et al. Aging and imaging assessment of body composition: from fat to facts. Front Endocrinol (Lausanne) 2019;10:861. doi: 10.3389/fendo.2019.00861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ma Y, Hébert JR, Manson JE, et al. Determinants of racial/ethnic disparities in incidence of diabetes in postmenopausal women in the U.S.: The Women’s Health Initiative 1993–2009. Diabetes Care. 2012;35(11):2226–2234. doi: 10.2337/dc12-0412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chia CW, Egan JM, Ferrucci L. Age-related changes in glucose metabolism, hyperglycemia, and cardiovascular risk. Circ Res. 2018;123(7):886–904. doi: 10.1161/CIRCRESAHA.118.312806. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available for ethical reasons.