Highlights
-
•
Epithelial cell-mimetic (rhACE2-cytomimetic) particles bind SARS-CoV-2 spike RBD.
-
•
rhACE2-Cytomimetic particles restrict SARS-CoV-2 spike binding to cell surface.
-
•
Particle functionalized rhACE2 is stoichiometrically superior to soluble rhACE2.
-
•
Inhaled rhACE2-Cytomimetic particles limit viral spike uptake in airway epithelium.
-
•
rhACE2-Cytomimetic particles can be an effective countermeasure for COVID-19.
Keywords: COVID-19, SARS-CoV-2, Cytomimetics, Countermeasures, Angiotensin I converting enzyme (ACE2) carboxypeptidase
Abstract
The development of countermeasures that aid in the prevention and propagation of SARS-CoV-2 infections is critical to manage the continuing crisis brought about by COVID-19. Here we present a proof-of-concept study on the use of cell-mimetic microparticles (Cytomimetics) for the interference and sequestration of SARS-CoV-2 virions away from the cellular surfaces required for replication, disease manifestation, and outbreak propagation. Recombinant human ACE2 (rhACE2) functionalized onto the surface of cytomimetic particles binds the receptor binding domain (RBD) of recombinant SARS-CoV-2 spike protein with high affinity and demonstrated a stoichiometric advantage over the use of soluble rhACE2. Inhalation of rhACE2-Cytomimetic particles by mice prior to their exposure to aerosolized spike protein demonstrated the applicability of these cytomimetic particles in preventing viral protein binding to respiratory epithelial cells. Our study demonstrates the potential of an easily deliverable and highly modular technology for the control of viral infections and to complement other prophylactic countermeasures
1. Introduction
Overcoming the health, social, and economic crisis brought about by COVID-19 requires a multi-pronged approach to manage the spread of the virus. These include novel therapeutics, public education about viral transmissibility, and active prophylactic measures. While mass vaccination is the ideal approach, many obstacles still need to be overcome to reach this goal including vaccine hesitancy, and the continued emergence of new viral variants, as well as other logistical and social challenges [1,2]. Development of additional countermeasures is warranted to manage the continuing spread of COVID-19, and to deal with the emergence of variants which reduce vaccine efficacy, such as the dominant delta variant [3]. We know that the progression of COVID-19 and severity of disease are directly correlated to viral load [4,5] and gaining control over viral replication can dramatically reduce the rate of complications and shorten the course of disease. Here we propose the development and use of a novel technology, cytomimetic particles, in the form of inhalable ACE2-functionalized biomaterial particles as a prophylactic countermeasure to limit the infectivity and amplification of the viral load in people exposed to the SARS-CoV-2 virus. Rather than linking the active targeting functionality to the biomaterial particles, the cytomimetic particle approach passively leverages the tropism and affinity of the virion for specific cell surface markers to elicit the interaction in a decoy-style defensive mechanism. The SARS-CoV-2 virus has been shown to specifically target the angiotensin I converting enzyme (ACE2) carboxypeptidase [6] present in the cell surface of airway epithelial cells. Critical to this mechanism, the SARS-CoV-2 spike glycoprotein (S protein), and specifically the S1 subunit of this protein complex binds to the peptidase domain (PD) of the ACE2 protein with an equilibrium dissociation constant (KD) of approximately 15 nM [7]. We know that a significant contributing event to the current COVID-19 pandemic was the emergence of SARS-CoV-2 variants containing the D614G mutation, which resulted in an increased density of the spike protein presented on the virion surface and enhanced its infectivity [8]. Here we present a proof-of-concept study in which the use of recombinant human ACE2 (rhACE2) protein is used to functionalize HisPur particles to compete with endogenous cellular targets for the SARS-CoV-2 spike for receptor binding domain (RBD), thereby limiting its interaction with viable cell surfaces and prophylactically restricting its uptake by respiratory cells.
2. Materials and methods
2.1 Animals
All animal experimentation was approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Miami and were performed in compliance with the National Institutes of Health (Bethesda, MD, U.S.A.) policies and procedures. Two months old male mice were used for the study.
2.2 Fabrication of rhACE2-Cytomimetic particles
HisPur. NI-NTA magnetic beads (ThermoFisher Scientific, Cat# 88,832) were functionalized with an excess molar amount of recombinant human ACE2 (R&D Systems, Cat# 933-ZN-010), by overnight incubation under constant rotation in binding buffer to generate rhACE2-cytomimetic particles. Functionalized particles were magnetically separated and washed to remove unbound rhACE2 protein. Control particles were manufactured in the same manner but replacing the rhACE2 protein with bovine serum albumin (BSA) to assess non-specific interactions with the particle surface.
2.3 In vitro validation of cytomimetic particles
Binding efficacy of the functionalized cytomimetic particles to the SARS-CoV2 Spike protein was determined through western blotting and immunofluorescence imaging. Briefly, functionalized cytomimetic particles were combined with SARS-CoV2 recombinant Spike RBD protein (R&D Systems, Cat# 10,499-CV-100) at increasing ratios, incubated overnight under constant rotation and then the amount of bound spike protein to the rhACE2-Cytomimetic particles as well as free-floating spike protein was determined through western blotting for the S1 protein subunit (AbCam, ab272420) (Fig. 1B). The efficacy of the rhACE2-Cytomimetic particles to outcompete the ACE2 on cell surfaces for spike protein binding was determined using primary ocular surface epithelial cell cultures. Cells were pre-incubated with either rhACE2-Cytomimetic particles, or an equivalent amount of non-ACE2 functionalized HisPur beads (control) for 5 min, followed by supplementation of the culture media with 5 µg of recombinant SARS-CoV-2 spike RBD protein for 2 h. Cells were then washed 3x in PBS to remove unbound spike protein, fixed and probed for immunohistochemical imaging of spike protein (AbCam, Cat # ab272420), and ACE-2 (AbCam, Cat# ab153488). To compare the inhibitory efficacy of rhACE2 functionalized onto a cytomimetic particle formulation over the use of soluble rhACE2, we transduced Human Embryonic Kidney Epithelial Cells (HEK293, ATCC, Cat#CRL-1573) with lentiviral preparations carrying a constitutive ACE2 expression cassette (Addgene, Cat #154,985-LV) to generate stable human ACE2 overexpressing cell lines. HEK293 cells were cultured in Dulbeccos modified eagle medium (DMEM, Gibco Cat# 11,965) supplemented with 10% fetal bovine serum (FBS, Thomas Scientific, Cat# C788D86) and 1% antibiotic-antimycotic solution (Gibco, Cat# 15,240) at 37 °C in 5% CO2. For experimentation, the ACE2-HEK293 cultures were pre-incubated with 2 µg of soluble rhACE2, or an equivalent quantity of rhACE2-Cytomimetic particles to account for 2 µg of rhACE2 on their surface for 5 min. Cells were then exposed to 5 µg of SARS-CoV-2 spike protein for 4 h. After 4 h of incubation, culture supernatants were collected, the cells washed 3x in PBS, and lysed using RIPA cell lysis buffer. Cell lysates and culture supernates were then analyzed using western blots for the presence of rhACE2 or SARS-CoV-2 spike protein.
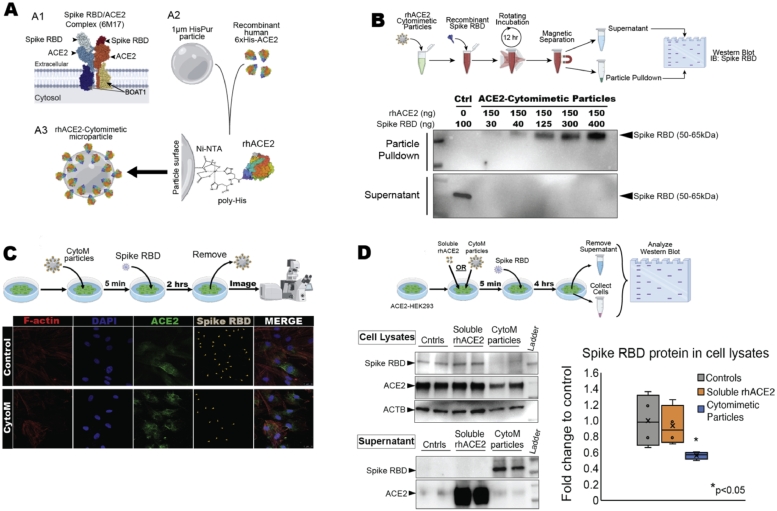
Fig. 1.
rhACE2-Cytomimetic particles and in vitro studies, (A) rhACE2-Cytomimetic particles fabrication showing the known SARS-CoV-2 interactions with ACE2 (A1, PDB: 6M17, [6]), components and functionalization chemistry utilized in this study (A2), and final conceptualized rhACE2-Cytomimetic particles (A3). (B) rhACE2-Cytomimetic particles binding capacity experiment (top) and western blot showing loading capacity of SARS-CoV-2 spike protein receptor biding domain (RBD) onto rhACE2-Cytomimetic particles (bottom). (C) rhACE2-Cytomimetic particles in vitro testing on primary human epithelial cell cultures (top), and immunohistochemistry images for F-actin, DAPI, ACE2, and Spike RBD (gray with orange arrowheads) present on human epithelial cells pre-incubated with HisPur beads (control) or rhACE2-Cytomimetic particles and exposed to SARS-CoV-2 Spike RBD protein for 2 h (bottom). (D) Comparative study between rhACE2-Cytomimetic particles and soluble rhACE2 in vitro testing on primary ACE2 overexpressing HEK293 cell cultures, human epithelial cell cultures (top). Western blot images of RBD, ACE2, and ACTB in cell lysate and supernatant fractions, and quantification of spike RBD protein in cell lysate fraction (bottom, *p < 0.05).
2.4 In vivo delivery of cytomimetic particles
Mice were anesthetized and made to inhaled rhACE2-Cytomimetic particles through a nose mask with the aid of a nebulizer for 5 min, then immediately exposed to the Spike SARS-CoV2 recombinant Spike RBD protein also through nebulization, and then maintained for 3 h under observation following recovery from anesthesia. After euthanasia the mice nasal passages and tracheas were dissected and used for either western blot or immunohistochemistry analysis.
2.5 Statistical analyses
Paired and unpaired t tests were calculated using GraphPad Prism 9, with statistical significance denoted at p < 0.05.
3. Results
Functionalized cytomimetic particles were fabricated through the conjugation of 6x His-tagged rhACE2 onto HisPur magnetic particles (Fig. 1A). In vitro validation of the binding efficacy was determined using western blot analysis, rhACE2-Cytomimetic particles bind SARS-CoV-2 Spike RBD effectively at a ratio of greater than 1:2.5 (Spike RBD: rhACE2) (Fig. 1B). The comparative affinity of the SARS-CoV-2 Spike RBD to rhACE2 on cytomimetic particles over cellular ACE2 was determined by pre-incubation of human epithelial cell cultures with rhACE2-ytomimetic particles followed by exposure to the spike RBD protein as a prophylactic use model in vitro. Immunofluorescence imaging showed significantly reduced amounts of SARS-CoV-2 Spike RBD protein bound to the cellular ACE2 receptor, indicating the rhACE2-Cytomimetic particles out compete the human cellular ACE2 as a target for the SARS-CoV-2 Spike RBD binding (Fig. 1C). We further compared the inhibition efficiency of rhACE2-Cytomimetic particles to that of soluble rhACE2, a modality that has been advanced as a therapeutic option [9,10] and demonstrate that an equivalent amount of ACE2 immobilized onto a particle surface is significantly more efficient at preventing viral spike protein uptake in ACE2 overexpressing cells (Fig. 1D).
The in vivo efficacy of rhACE2-Cytomimetic particles was determined via the inhaled delivery of rhACE2-Cytomimetic particles to mice immediately prior to their exposure to the SARS-CoV-2 Spike protein. Mice were nebulized with either saline (control) or rhACE2-Cytomimetic particles for 5 min, followed by nebulization with 2 ug total Spike RBD protein per animal. After 3 h of Spike RBD exposure, mouse nasal passages and tracheas were dissected for immunofluorescence and western blot analysis. Our results show that use of rhACE2-Cytomimetic particles limits the binding of the viral protein onto the airway epithelial lining (Fig. 2). The amount of viral Spike RBD protein bound to the endogenous mouse ACE2 (Spike/ACE2) in the nasal epithelium and turbinates was significantly lower in the rhACE2-Cytomimetic particles cohort (Fig. 2B) than in the controls. In fact, mice that had inhaled the rhACE2-Cytomimetic particles had little detectable spike RBD protein bound to the epithelial lining of the airway tree beyond the nasal cavity.
Fig. 2.
. In vivo delivery of rhACE2-Cytomimetic particles, (A) Mouse experimentation workflow and immunohistochemical images of dissected nasal passages depicting endogenous mouse ACE2 and Spike RBD co-localization (solid white arrowheads), or Spike RBD protein not co-localized to the ACE2 fluorescent signal (black arrowheads). (B) Western blot image and quantification of the spike RBD/ACE2 protein ratio isolated from mouse nasal (N) and trachea (T) tissues following 3 h of exposure to Spike RBD in the control and rhACE2-Cytomimetic particles cohort (** p < 0.01).
4. Discussion
With the current worldwide pandemic caused by the novel coronavirus, the evolution of the virus towards more infectious and pathogenic strains, and the mortality rate observed from respiratory complications in COVID-19 illness, novel approaches to the management of SARS-CoV-2 exposures need to be entertained. Our cell-mimetic approach utilizes a powerful and evolutionarily perfected mechanism, that of a decoy deployment strategy, which is one of the most ubiquitous defense mechanisms found in nature [11]. Moreover, mimetism is not a novel concept in medicine and is routinely used in biomedical applications to enhance the biocompatibility of medical devices [12], and for the delivery of DNA vaccines [13], chemotherapeutic payloads [14], and immunomodulatory molecules [15]. We show that a cytomimetic particle approach can successfully sequester the SARS-CoV-2 spike glycoprotein off its biological targets in the respiratory tract, potentially limiting the pathological effects of viral infections without the need for additional specialized protective gear [16], or intricate fabrication processes as described in similar studies [17], [18], [19]. We further demonstrate that surface-functionalized rhACE2 presents a stoichiometric advantage over soluble rhACE2 in its inhibition of SARS-CoV-2 spike glycoprotein binding to cellular ACE2, with the latter already having demonstrated encouraging outcomes in clinical trials [10].
Countermeasures traditionally employed during a viral outbreak, such as increased hygienic practices and social distancing, are all focused on restricting the propagation of viral transmission. Gaining control over the infectivity of an outbreak is probably the single-most important factor in overcoming an outbreak. The technology presented in this paper, while conceptually novel is relatively easy to adapt to other pathogens for which known cellular targets exist, and which are known to occur in regional, endemic, or seasonal fashions. If employed correctly, cytomimetic technology would add a molecular-level countermeasure to limit the propagation of an infection within a community as well as within the tissues of already-infected patients. The latter would result in a reduction of the rate of complications from uncontrolled viral loads in patients, thus alleviating the strain on the healthcare system, which constitutes an invaluable benefit to the management of any outbreak.
Funding
This work was supported in part by a generous philanthropic gift from Dr. Nasser Ibrahim Al-Rashid. Bascom Palmer Eye Institute is supported by NIH Center Core Grant P30EY014801 and a Research to Prevent Blindness Unrestricted Grant (New York, NY, USA).
Declaration of Competing Interest
The authors have no actual or perceived competing interests to declare.
Acknowledgments
Figures were created in part with Biorender.com. We would like to thank Dr. Galina Dvoriantchikova for her assistance in coordinating animal experimentations.
References
- 1.Mills M.C., Salisbury D. The challenges of distributing COVID-19 vaccinations. Eclinicalmedicine. 2020;31 doi: 10.1016/j.eclinm.2020.100674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Loomba S., de Figueiredo A., Piatek S.J., de Graaf K., Larson H.J. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat. Hum. Behav. 2021;5:337–348. doi: 10.1038/s41562-021-01056-1. [DOI] [PubMed] [Google Scholar]
- 3.Planas D., Veyer D., Baidaliuk A., Staropoli I., Guivel-Benhassine F., Rajah M.M., Planchais C., Porrot F., Robillard N., Puech J., Prot M., Gallais F., Gantner P., Velay A., Guen J.L., Kassis-Chikhani N., Edriss D., Belec L., Seve A., Courtellemont L., Péré H., Hocqueloux L., Fafi-Kremer S., Prazuck T., Mouquet H., Bruel T., Simon-Lorière E., Rey F.A., Schwartz O. Reduced sensitivity of SARS-CoV-2 variant delta to antibody neutralization. Nature. 2021:1–7. doi: 10.1038/s41586-021-03777-9. [DOI] [PubMed] [Google Scholar]
- 4.Hung I.F.N., Cheng V.C.C., Wu A.K.L., Tang B.S.F., Chan K.H., Chu C.M., Wong M.M.L., Hui W.T., Poon L.L.M., Tse D.M.W., Chan K.S., Woo P.C.Y., Lau S.K.P., Peiris J.S.M., Yuen K.Y. Viral loads in clinical specimens and SARS manifestations. Emerg. Infect. Dis. 2004;10:1550–1557. doi: 10.3201/eid1009.040058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/s0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yan R., Zhang Y., Li Y., Xia L., Guo Y., Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444–1448. doi: 10.1126/science.abb2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.-.L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang L., Jackson C.B., Mou H., Ojha A., Peng H., Quinlan B.D., Rangarajan E.S., Pan A., Vanderheiden A., Suthar M.S., Li W., Izard T., Rader C., Farzan M., Choe H. SARS-CoV-2 spike-protein D614G mutation increases virion spike density and infectivity. Nat. Commun. 2020;11:6013. doi: 10.1038/s41467-020-19808-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Monteil V., Kwon H., Prado P., Hagelkrüys A., Wimmer R.A., Stahl M., Leopoldi A., Garreta E., del Pozo C.H., Prosper F., Romero J.P., Wirnsberger G., Zhang H., Slutsky A.S., Conder R., Montserrat N., Mirazimi A., Penninger J.M. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181:905–913. doi: 10.1016/j.cell.2020.04.004. e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zoufaly A., Poglitsch M., Aberle J.H., Hoepler W., Seitz T., Traugott M., Grieb A., Pawelka E., Laferl H., Wenisch C., Neuhold S., Haider D., Stiasny K., Bergthaler A., Puchhammer-Stoeckl E., Mirazimi A., Montserrat N., Zhang H., Slutsky A.S., Penninger J.M. Human recombinant soluble ACE2 in severe COVID-19. Lancet Respir. Med. 2020;8:1154–1158. doi: 10.1016/s2213-2600(20)30418-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Starrett A. Adaptive resemblance: a unifying concept for mimicry and crypsis. Biol. J. Linn. Soc. 1993;48:299–317. doi: 10.1111/j.1095-8312.1993.tb02093.x. [DOI] [Google Scholar]
- 12.Zhou L., Tan G.-.X., Ning C.-.Y. Modification of biomaterials surface by mimetic cell membrane to improve biocompatibility. Front. Mater. Sci. 2014;8:325–331. doi: 10.1007/s11706-014-0265-5. [DOI] [Google Scholar]
- 13.Boyoglu S., Vig K., Pillai S., Rangari V., Dennis V.A., Khazi F., Singh S.R. Enhanced delivery and expression of a nanoencapsulated DNA vaccine vector for respiratory syncytial virus. Nanomed. Nanotechnol. Biol. Med. 2009;5:463–472. doi: 10.1016/j.nano.2009.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Zhang J., Tang H., Liu Z., Chen B. Effects of major parameters of nanoparticles on their physical and chemical properties and recent application of nanodrug delivery system in targeted chemotherapy. Int. J. Nanomed. 2017;12:8483–8493. doi: 10.2147/ijn.s148359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ben-Akiva E., Witte S.E., Meyer R.A., Rhodes K.R., Green J.J. Polymeric micro- and nanoparticles for immune modulation. Biomater. Sci-Uk. 2018;7:14–30. doi: 10.1039/c8bm01285g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Aydemir D., Ulusu N.N. Correspondence: angiotensin-converting enzyme 2 coated nanoparticles containing respiratory masks, chewing gums and nasal filters may be used for protection against COVID-19 infection. Travel Med. Infect. Dis. 2020;37 doi: 10.1016/j.tmaid.2020.101697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garrido P.F., Calvelo M., Blanco-González A., Veleiro U., Suárez F., Conde D., Cabezón A., Piñeiro Á., Garcia-Fandino R. The lord of the nanorings: cyclodextrins and the battle against SARS-CoV-2. Int. J. Pharmaceut. 2020;588 doi: 10.1016/j.ijpharm.2020.119689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rao L., Xia S., Xu W., Tian R., Yu G., Gu C., Pan P., Meng Q.-.F., Cai X., Qu D., Lu L., Xie Y., Jiang S., Chen X. Decoy nanoparticles protect against COVID-19 by concurrently adsorbing viruses and inflammatory cytokines. Proc. Natl. Acad. Sci. 2020;117:27141–27147. doi: 10.1073/pnas.2014352117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang C., Wang S., Chen Y., Zhao J., Han S., Zhao G., Kang J., Liu Y., Wang L., Wang X., Xu Y., Wang S., Huang Y., Wang J., Zhao J. Membrane nanoparticles derived from ACE2-rich cells block SARS-CoV-2 infection. ACS Nano. 2021;15:6340–6351. doi: 10.1021/acsnano.0c06836. [DOI] [PubMed] [Google Scholar]